Abstract
The authors report a case of a 57-year-old woman who was successfully treated with a percutaneous embolization procedure for a renal arteriovenous fistula that developed as a complication of a kidney biopsy. An acute kidney injury that failed to resolve with medical management prompted further investigation with a renal biopsy. Five hours after the kidney biopsy, the patient became hemodynamically unstable with a blood pressure of 77 of 52 mm Hg. A stat abdominal computed tomography scan without contrast discovered a large left-sided perinephric hematoma that measured up to 11.5 cm with a moderate amount of perinephric blood. An angiogram subsequently demonstrated the presence of an arteriovenous fistula at the inferior pole of the left kidney. Several 2 × 3 mm and 3 × 3 mm coils were deployed into two separate segmental branches of the inferior pole, and the post embolization angiogram confirmed resolution of the previously visualized arteriovenous fistula.
Keywords: Interventional radiology, Kidney biopsy, Arteriovenous fistula, Embolization, Nephrology
Introduction
A percutaneous kidney biopsy is a minimally invasive procedure that can be performed for numerous reasons, including establishing an accurate diagnosis, distinguishing acute versus chronic pathology, and directing optimal medical therapy for renal disease. Arteriovenous fistula (AVF) formation may occur in up to 18% of patients after percutaneous renal biopsy [1]. An AVF is an abnormal connection or passageway between an artery and a vein. If a vein is inadvertently punctured during an attempted biopsy, inadequate hemostasis may allow persistent bleeding from an arterial puncture site to track into the venous puncture site, creating an AVF.
AVFs may remain asymptomatic and spontaneously resolve or cause complications such as hematuria, hypertension, and/or renal insufficiency [1]. Large or multiple AVFs can produce clinically significant effects by shunting blood from the arterial to the venous circulation, forcing the heart to pump additional volume which may lead to high-output cardiac failure. A percutaneous embolization technique is a proven, effective treatment option for patients who have acquired an AVF after a kidney biopsy [2]. Alternative treatment approaches include observation for spontaneous resolution, and surgery as a last resort for refractory cases.
Case presentation
A 57-year-old female patient was instructed by her oncologist to go to the emergency department for complaints of increasing weakness, diarrhea, and vomiting for 2 weeks, along with a rising creatinine. She has a past medical history of metastatic pancreatic cancer currently being treated with chemotherapy. Her vitals upon admission included a blood pressure of 131 of 68 mm Hg, heart rate of 97 bpm, respiratory rate of 17 br/min, and an O2 saturation of 100% on room air. Initial labs revealed a low hemoglobin of 8.6 g/dL, low sodium of 130 mmol/L, low carbon dioxide of 18 mmol/L, elevated blood urea nitrogen of 56 mg/dL, elevated creatinine of 4.4 mg/dL, along with potassium, chloride, calcium, and glucose levels measured within normal limits. Her baseline creatinine was 0.8 mg/dL. A urinalysis was performed and demonstrated cloudy urine with 100 mg/dL of protein and 29 RBCs per HPF. The emergency room physician admitted the patient for treatment of an acute kidney injury (AKI) and temporarily withheld chemotherapy.
An abdominal ultrasound was performed and displayed increased echogenicity of both kidneys compatible with medical renal disease along with mild right hydronephrosis. Interventional radiology was consulted to perform a kidney biopsy after 5 days of medical AKI treatment failed to restore her baseline kidney function. Preprocedural CT imaging was employed to target the lower pole of the left kidney. Preprocedural coagulation studies were within normal limits. Using intermittent CT guidance, a 17-gauge coaxial introducer needle was advanced into the lower pole of the left kidney, where 3 core samples of renal tissue were obtained using an 18-gauge Bard biopsy needle (Fig. 1). Estimated blood loss was minimal and there were no intraoperative complications.
Fig. 1.
CT scan displaying the biopsy needle in the lower pole of the left kidney.
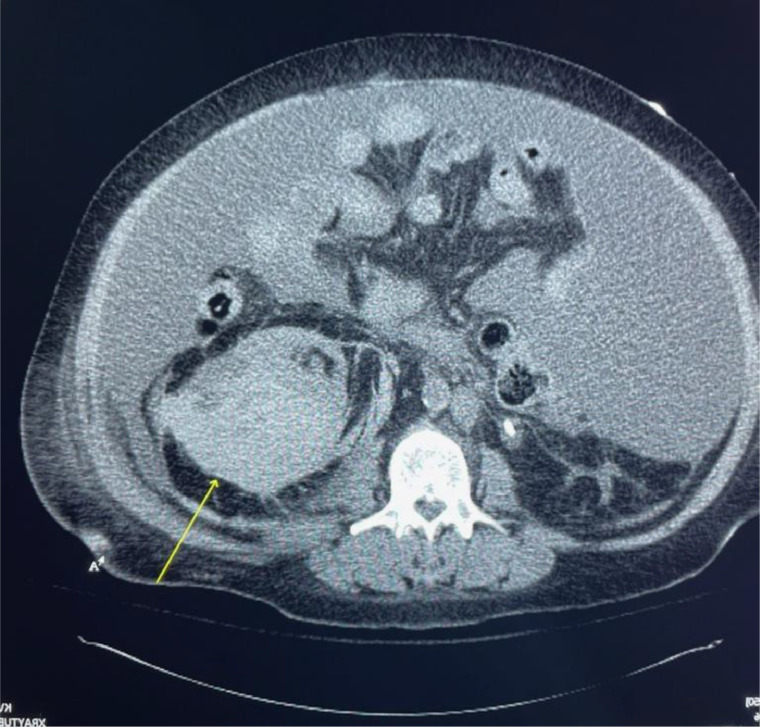
Roughly 5 hours after the procedure, a rapid response team was called as the patient's blood pressure dropped to 77 of 52 mmHg with a mean arterial pressure of 50 mm Hg, a heart rate of 79 bpm, and an O2 saturation of 94% on room air. An initial 250 mL bolus of normal saline elevated her blood pressure to 83 of 54 mm Hg, and a second 250 mL bolus led to a blood pressure of 83 of 53 mm Hg. A third and final 500 mL bolus was administered and raised her blood pressure up to 106 of 66 mm Hg. A stat complete blood count and complete metabolic panel revealed a hemoglobin level of 6.4 g/dL and the patient received 1 unit of packed red blood cells. A stat CT scan of the abdomen and chest without contrast revealed a large left-sided perinephric hematoma measuring up to 11.5 cm with a moderate amount of perinephric blood (Fig. 2). The patient was referred back to interventional radiology for a renal angiogram with possible embolization.
Fig. 2.
Abdominal CT scan displaying a large left-sided perinephric hematoma.
Access into the right common femoral artery was obtained under ultrasound guidance using a micropuncture needle after anesthetizing the skin. A microwire was passed and a 6 French introducer sheath was placed. Continuous infusion of saline was initiated through the sheath. A Bentson wire was advanced to the mid-abdominal aorta. A catheter was used to access the left renal artery and an angiogram was performed. The angiogram demonstrated no evidence for active extravasastion of blood. Prompt visualization of a draining vein from the inferior pole of the left kidney was consistent with an arteriovenous fistula (Fig. 3). A microcatheter was used to subselect an inferior segmental branch and an additional angiogram was performed. The angiogram confirmed the presence of an arteriovenous fistula at the inferior pole of the left kidney (Fig. 4). Several 2 × 3 mm and 3 × 3 mm coils were deployed into 2 separate segmental branches of the inferior pole. Post embolization angiogram demonstrated resolution of the previously visualized arteriovenous fistula (Fig. 5). The catheters and vascular sheath were removed and hemostasis was achieved with a Starclose closure device followed by manual compression. The patient tolerated the procedure well without any immediate complications. She was discharged home three days after the embolization procedure with diagnoses of AKI with acute tubular necrosis, thrombotic microangiopathy, and a retroperitoneal bleed postbiopsy.
Fig. 3.
Renal angiogram showing abnormal enhancement at the inferior pole of the left kidney, raising suspicion for an arteriovenous fistula.
Fig. 4.
Second renal angiogram confirmed rapid visualization of draining inferior renal vein (inferior arrow) and inferior vena cava (superior arrow) consistent with an arteriovenous fistula.
Fig. 5.
Post embolization angiogram demonstrates resolution of the previously visualized arteriovenous fistula.
Discussion
An AVF, as noted in the above case, is a rare but serious complication that can occur following a renal biopsy. Multiple acute hemodynamic changes can be observed from a large AVF, all of which, if not corrected, could lead to patient demise. Immediately following the formation of a large AVF, there is a drastic decrease in systemic vascular resistance, which subsequently causes an increase in stroke volume, heart rate, and cardiac output [3,4]. The systemic vascular resistance decreases, in part, because the fistula allows blood to bypass the high resistance of the systemic arterioles and diverts blood directly into the low resistance venous system. This increased blood flow and sheer stress on the venous system stimulates the vascular endothelium to release nitrous oxide and other endothelial relaxing factors, which in turn dilates the arteries, further reducing systemic vascular resistance [3,4].
As the systemic vascular resistance declines, there is an acute fall in both central and peripheral blood pressure. With central filling pressures decreasing, the sympathetic nervous system attempts to compensate by increasing the contractility of the heart as well as increasing the heart rate [6,7]. The increase in cardiac output subsequently increases the venous return to the heart, which can lead to ventricular dilation and high-output heart failure [5].
If left untreated, an AVF may progressively enlarge and lead to local edema and pain, as well as ischemia due to redirection of arterial blood flow. Chronic changes associated with an AVF include worsening ventricular dilation and function, left ventricular hypertrophy, and pulmonary hypertension [5]. Eventually, deleterious myocardial remodeling can occur, furthering myocardial dysfunction [8,9].
Conclusion
Formation of an AVF is a rare but serious adverse event that may occur following a percutaneous renal biopsy in up to 18% of patients, as documented in this case. It is important to document this complication and highlight the successful treatment strategies that can be employed to improve patient outcomes.
Patient consent
Patient written informed consent was not obtained for the publication of this report and accompanying images as no patient identifiers were utilized.
Footnotes
Acknowledgments: The authors would like to acknowledge Douglass Sherard, MD, for providing the case information and images.
Competing Interests: The authors have declared that no competing interests exist.
References
- 1.Harrison KL, Nghiem HV, Coldwell DM, Davis CL. Renal dysfunction due to an arteriovenous fistula in a transplant recipient. J Am Soc Nephrol. 1994;5(6):1300–1306. doi: 10.1681/ASN.V561300. DecPMID: 7893994. [DOI] [PubMed] [Google Scholar]
- 2.Kopecna L, Mach V, Prochazka J. Arteriovenous fistula as a complication of renal biopsy. Bratisl Lek Listy. 2005;106(6-7):218–220. PMID: 16201740. [PubMed] [Google Scholar]
- 3.Korsheed Shvan, Eldehni Mohamed.T., John Stephen G., Fluck Richard J., McIntyre Christopher W. Effects of arteriovenous fistula formation on arterial stiffness and cardiovascular performance and function. Nephrology Dialysis Transplantation. 2011;26(10):3296–3302. doi: 10.1093/ndt/gfq851. IssueOctoberPages. [DOI] [PubMed] [Google Scholar]
- 4.Mitchell G.F., Parise H., Vita J.A., Larson M.G., Warner E., Keaney J.F. Local shear stress and brachial artery flow-mediated dilation. Hypertension. 2004;44(2):134–139. doi: 10.1161/01.hyp.0000137305.77635.68. [DOI] [PubMed] [Google Scholar]
- 5.Reddy Y.N., Obokata M., Dean P.G., Melenovsky V., Nath K.A., Borlaug B.A. Long-term cardiovascular changes following creation of arteriovenous fistula in patients with end stage renal disease. Eur Heart J. 2017;38(24):1913–1923. doi: 10.1093/eurheartj/ehx045. [DOI] [PubMed] [Google Scholar]
- 6.Korsheed S., Eldehni M.T., John S.G., Fluck R.J., McIntyre C.W. Effects of arteriovenous fistula formation on arterial stiffness and cardiovascular performance and function. Nephrology Dialysis Transplantation. 2011;26(10):3296–3302. doi: 10.1093/ndt/gfq851. [DOI] [PubMed] [Google Scholar]
- 7.Basile C, Lomonte C, Vernaglione L, Casucci C, Antonelli M, Losurdo N. The relationship between the flow of arteriovenous fistula and cardiac output in haemodialysis patients. Nephrol Dial Transplant. 2008;23:282. doi: 10.1093/ndt/gfm549. [DOI] [PubMed] [Google Scholar]
- 8.London GM. Left ventricular alterations and end-stage renal disease. Nephrol Dial Transplant. 2002;17(Suppl 1):29. doi: 10.1093/ndt/17.suppl_1.29. [DOI] [PubMed] [Google Scholar]
- 9.Ori Y, Korzets A, Katz M, Erman A, Weinstein T, Malachi T. The contribution of an arteriovenous access for hemodialysis to left ventricular hypertrophy. Am J Kidney Dis. 2002;40:745. doi: 10.1053/ajkd.2002.35685. [DOI] [PubMed] [Google Scholar]