Abstract
The gastrointestinal (GI) tract is the most commonly affected internal organ system in systemic sclerosis (SSc). SSc may lead to impaired function in any region of the GI tract, from the esophagus to the anorectum, causing significant morbidity, as well as mortality in patient subsets. Given the low prevalence of SSc in the community, many rheumatologists may not have a systematic framework for diagnosing or treating the GI complaints in this disease. These practice recommendations aim to summarize and consolidate the current guidelines from the fields of gastroenterology and rheumatology, and establish a symptom-based framework for diagnosis and management based on available evidence in the literature. Subject areas in need of additional research are also identified.
Keywords: systemic sclerosis, scleroderma, gastrointestinal, management
1.0. INTRODUCTION
Systemic sclerosis (SSc) is a rare immune-mediated rheumatic disease that is associated with high morbidity and mortality.[1] Gastrointestinal (GI) disease is a frequent manifestation of SSc and affects up to 90% of cases, making it the most commonly affected internal organ system.[2] Mortality is high among patients with severe GI complications (i.e. pseudo-obstruction, malabsorption, requiring total parenteral nutrition), and falls just behind that of pulmonary arterial hypertension and pulmonary fibrosis. GI symptoms are common and attributed to a variety of factors such as delayed GI transit, dysbiosis, and dietary or medication intolerances.[3–6] The heterogeneous clinical presentation, limited understanding of disease pathogenesis, absent biomarkers of GI disease activity, and reduced familiarity with managing SSc-GI complications can lead to significant challenges in clinical practices and ultimately suboptimal patient care.[1]
Data to guide the diagnosis and management of SSc-GI complications is limited; however, several groups have developed expert consensus-based recommendations to help address this challenge. The European League Against Rheumatism (EULAR) Scleroderma Trials and Research group and United Kingdom scleroderma study group (UKSSG) each published clinical recommendations[7,8] while acknowledging limited data and a lack of controlled trials in SSc patients. However, the Canadian Scleroderma Research Group (CSRG) found that the aforementioned guidelines did not significantly change rheumatologists’ behavior when treating SSc-GI patients, citing symptom-guided care as a possible explanation.[9]
With these limitations in mind, the goals of this review are to provide a symptom-based practical systematic approach, integrating data from the rheumatologic and GI literature, to guide the diagnosis and treatment of GI complications in patients with SSc. Symptoms were classified into six groupings that are clinically relevant to the SSc population:
Oropharyngeal dysphagia, globus sensation
Heartburn, reflux, esophageal dysphagia
Nausea, vomiting, bloating, abdominal distention
Diarrhea
Constipation
Fecal incontinence
A focused differential of common considerations for each of these groups (in the context of SSc) can be found in Table 1. With guidance from an expert clinical gastroenterologist (TA), diagnostic schema for these symptom groups (see Figures 1 and 2) were developed using current gastroenterology and rheumatology guidelines, and incorporating relevant clinical studies of the SSc population when available. We also aimed to provide a quick resource representing all available clinical trial data related to the management of SSc-related GI manifestations (see Table 2).
Table 1.
Differential Diagnosis and Diagnostic Considerations for Gastrointestinal Symptoms in Systemic Sclerosis
| Symptom | Differential Diagnosis and Potential Complications | Considerations for Diagnostic Evaluation |
|---|---|---|
| Oropharyngeal | Proximal Esophageal Dysmotility | Modified Barium Swallow |
| Dysphagia | Pharyngeal Myositis | High-Resolution Esophageal Manometry |
| Globus Sensation | Sicca Syndrome | Laboratory Studies for Myopathy: CK, aldolase |
| Myasthenia Gravis | Antibodies for Myasthenia Gravis | |
| Heartburn | Gastroesophageal Reflux Disease | Esophagogastroduodenoscopy (EGD) |
| Reflux | Esophageal Body Dysmotility | High-Resolution Esophageal Manometry |
| Esophageal Dysphagia | Esophageal Strictures | pH Monitoring |
| Lower Esophageal Sphincter Dysfunction | ||
| Hiatal Hernia | ||
| Eosinophilic Esophagitis | ||
| Esophageal Candidiasis and/or Other Infection | ||
| Barrett’s Esophagus | ||
| Esophageal Adenocarcinoma | ||
| Nausea | Gastroparesis | EGD |
| Vomiting | Abnormal Gastric Accommodation | 4 Hour Scintigraphy-Based Gastric Emptying |
| Bloating | Peptic Ulcer Disease | Study |
| Distention | H. pylori Infection | Urea Breath Testing or H. pylori Stool Antigen |
| Small and/or Large Bowel Dysmotility | Testing | |
| Pneumatosis Cystoides Intestinalis | Abdominal Film | |
| Assessment of Small or Large Bowel Transit | ||
| Diarrhea | Spontaneous Bacterial Overgrowth Syndrome | Hydrogen/Methane Breath Test |
| Small Bowel Dysmotility | Celiac Disease Biomarker Testing +/− EGD | |
| Malabsorption | C. difficile Stool Antigen Testing | |
| Overflow Diarrhea | Infectious Diarrhea Testing | |
| Infectious Diarrhea | Labs to Rule Out Malabsorption | |
| Inflammatory Bowel Disease | Colonoscopy | |
| Constipation | Colonic Dysmotility | Abdominal Film |
| Fecal Impaction | Assessment of Large Bowel Transit | |
| Colon Cancer | Colonoscopy | |
| Anorectal Dysfunction | ||
| Fecal Incontinence | Internal Anal Sphincter Dysfunction | Digital Rectal Examination |
| Diarrhea | Anorectal Manometry | |
| Control/Treat Diarrhea |
Figure 1.
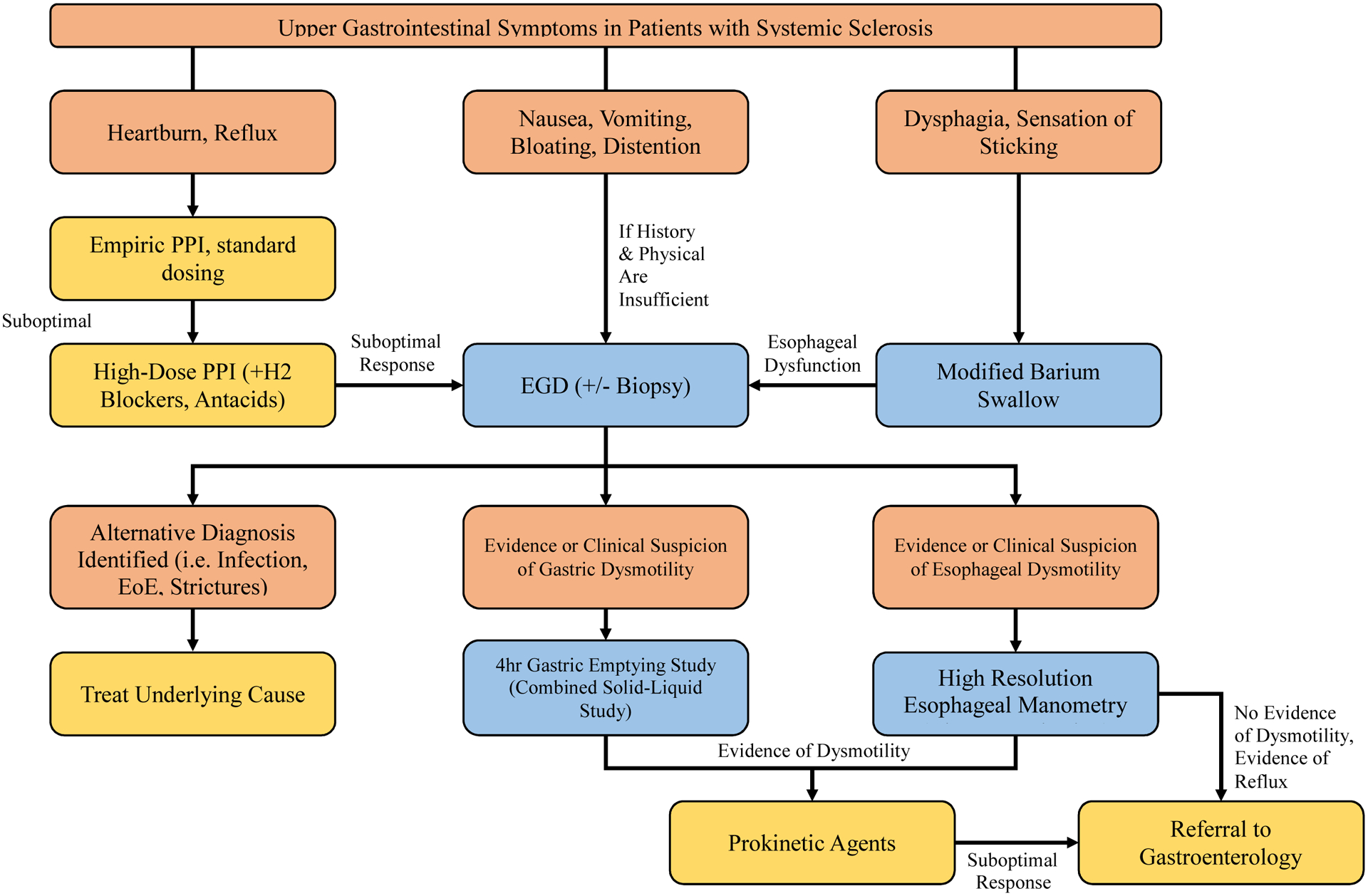
A Proposed Diagnostic Protocol for Upper Gastrointestinal Symptoms in Systemic Sclerosis
Figure 2.

A Proposed Diagnostic Protocol for Lower Gastrointestinal Symptoms in Systemic Sclerosis
Table 2.
Clinical tiuls of therapeutics lor gastrointestinal disease in systemic sclerosis.
| Investigators | Study | Type of Study | Number of SSc Patients | Key Findings | |
|---|---|---|---|---|---|
| Prokinetic Drugs and Therapies (Upper GI Tract) | Ramirez-Mata, M., et al. [41] | Metoclopramide (1 dose of 20mg PO); effect on esophageal aperistalsis and LES pressure | Randomized cross-over trial | 14 | Metoclopramide was associated with increased frequency of esophageal contractions and increased LES pressure in some patients |
| Johnson, D.A., et al. [58] | Metoclopramide (10mg PO before meals and at bedtime); effect on esophageal motility and gastric emptying | Clinical trial | 12 | Metoclopramide was associated with increased LES pressures and reduced gastric emptying delay in most patients | |
| Horowitz, M., et al. [95] | Cisapride (10mg PO QID); effect on gastric/esophageal emptying and upper GI symptoms | Clinical trial | 8 | Cisapride was associated with significant improvement in gastric emptying and improved upper GI symptoms; there was no significant improvement in esophageal emptying | |
| Kahan, A., et al. [40] | Cisapride (1 dose of 10mg IV); effect on LES pressure and gastric contractions | Randomized cross-over trial | 20 | Cisapride was associated with increased LES pressure and increased number of fundic gastric contractions | |
| Fiorucci, S., et al. [96] | Erythromycin (2mg/kg/hr IV) in SSc patients vs no disease control; effect on gastric and gallbladder emptying | Non-randomized control trial | 12 | Erythromycin was associated with accelerated gastric and gallbladder motility in all patients | |
| Wang, S.J., et al [89] | Cisapride (10mg PO TID); effect on oesophageal transit | Randomized cross-over trial | 12 | Cisapride did not have an impact on oesophageal transit times | |
| Sallam, H., et al. [62] | Transcutaneous electrical nerve stimulation (acupoints ST36 and PC6); effect on GI symptom scores | Clinical trial | 17 | Prolonged transcutaneous electrical nerve stimulation therapy was associated with improved GI symptom scores | |
| McNearney, T.A., et al. [63] | Transcutaneous electrical nerve stimulation (acupoints ST36 and PC6); effect of gastric myoelectrical activity and frequency of awakening due to abdominal pain/bloating | Clinical trial | 17 | Prolonged transcutaneous electrical nerve stimulation therapy was associated with significantly decreased nighttime awakenings due to abdominal pain | |
| Ariyasu, H., et al. [97] | Grehlin (5.0mcg/kg IV) vs placebo; effect on gastric emptying | Randomized cross-over trial | 10 | Grehlin was associated with accelerated gastric emptying times | |
| Anti-Reflux Drugs | Horikoshi, T., etal. [98] | Cimetidine (200mg IV) vs Famotidine (20mg IV) vs positive control (sodium bicarbonate) and negative control; effect on gastric motility and LES pressure | Non-randomized control trial | 41 | Cimetidine and Famotidine were associated with significant increases in LES pressure, however when corrected for changes in gastric phasic motor activity this significance was limited to Cimetidine and now Sodium Bicarbonate |
| Pakozdi, A., et al. [27] | Lansoprazole (30mg PO daily) vs placebo; evaluation for long-term benefit in symptom control and effect on the progression of oesophageal dysmotility | Randomized control trial | 24 | Lansoprazole was not associated with long-term benefit for symptoms, and had no effect on prevention of progression of esophageal dysmotility | |
| Karamanolis, G.P., et al. [45] | Buspirone (20mg PO daily); effect as add-on therapy to PPI for LES pressures and reflux/regurgitation scores | Clinical trial | 30 | Buspirone was associated with increased LES pressure and improved reflux and regurgitation scores | |
| Foocharoen, C., et al. [48] | Domperidone (10mg PO TID) vs Algycon (1 tablet TID); effect on GERD symptoms as add-on therapy with omeprazole (20mg PO BID) | Randomized control trial | 148 | Domperidone and algycon were equally effective as add-on therapies to omeprazole, showing significant improvement in GERD symptoms and quality of life scores | |
| Prokinetic Drugs (Lower GI Tract) | Soudah, H.C., et al. [77] | Octreotide (10mcg SC) in SSc patients vs no disease control; effect on motility and bacterial overgrowth | Clinical trial | 5 | Octreotide was associated with inducing migrating complexes, as well as reducing hydrogen breath excretion with reduced symptoms |
| Wang, S.J., et al. [88] | Cisapride (10mg PO TID); effect on colonic transit | Clinical trial | 16 | Cisapride was associated with increased colonic transit | |
| Nikou, G.C., et al. [78] | Octreotide (0.1mg BID SC or 20mg monthly IM); effect on small bowel disease unresponsive to other prokinetic agents | Clinical trial | 7 | Octreotide was associated with significant reduction in symptom severity | |
| Vigone, B., et al. [44] | Prucalopride (20mg PO daily) vs placebo; evaluation of intestinal dysmotility and symptom scoring | Randomized cross-over trial | 40 | Prucalopride was associated with increased intestinal evacuations, improved reflux and bloating symptoms, and improved constipation scores | |
| Probiotic Drugs and Therapies | Frech, T.M., et al. [80] | Probiotics (Bifidobacterium infantis 1 billion units daily or lactobacillus rhamnosusGG 1 billion units daily); effect on GI symptom scoring | Clinical trial | 10 | Probiotics were associated with improvement in reflux scores at 2 months |
| Low, A.H.L., et al [99] | Probiotics (Vivomixx® 1800 billion units daily) vs placebo; effect on GI symptom scoring | Randomized control trial | 40 | Probiotics were associated with improvement in reflux scores at 120 days | |
| Marighela, T.F., et al. [100] | Probiotics (Invictus Lacto Pró 1 capsule daily) vs placebo; effect on GI symptom scoring | Randomized control trial | 73 | No significant difference in GI symptom scoring | |
| Garcia-Collinot, G., et al. [81] | Probiotic (Saccharomyces boulardii 200mg BID for 7 days) vs Metronidazole (500mg BID for 7 days) vs Combination Therapy (Metronidazole 500mg BID for 7 days and Saccharomyces boulardii 200mg for 14 days); treatment of SIBO | Randomized clinical trial | 40 | Combination therapy was more effective than probiotic monotherapy, which was more effective than metronidazole monotherapy in treating SIBO | |
| Fretheim, H., et al. [82] | Fecal microbiota transplant (single-donor, commercially-available anaerobic cultivated human intestinal microbiota) vs placebo; effect on GI symptom scoring | Randomized control trial | 10 | Fecal microbiota transplant was associated with mild transient adverse events and improved symptom scoring |
SSc: Systemic Sclerosis: mg: milligrams: PO: per os (by mouth): LES: lower esophageal sphincter: QID: quater in die (four times daily): Cl: gastrointestinal: IV: intravenous: kg: kilograms: hr: hour: TID: ter in die (three times daily): GERD: gastroesophageal reflux disease: BID: bis in die (twice daily): SC: subcutaneous: IM: intramuscularly: SIBO: spontaneous bacterial overgrowth syndrome.
2.0. OROPHARYNGEAL DYSPHAGIA OR GLOBUS SENSATION
Subjective dysphagia is a common complaint among patients with SSc. Dysphagia can involve the oral cavity, pharynx, and/or esophagus.[10] Esophageal sources of dysphagia have different diagnostic considerations and are thus discussed separately in this review (see Section 3.0). In the oropharynx, dry mouth (often related to sicca symptoms) may contribute to oropharyngeal dysfunction, as the reduction in salivary flow may impede the ease of normal swallowing.[11] This may be complicated by fibrosis of the base of the tongue, which may further impair normal swallowing.[12] Patients with SSc may also experience pharyngeal inflammation related to uncontrolled gastroesophageal reflux disease (GERD), often manifesting as hoarseness or dry cough. Additionally overlap autoimmune syndromes, such as cases of co-existing inflammatory myositis[13] or myasthenia gravis,[14] may contribute to dysphagia by impairing normal pharyngeal muscle function.
2.1. Use of a Modified Barium Swallow to Determine Anatomic Origin of Dysphagia
When evaluating for pure dysphagia in patients without reflux symptoms, the UKSSG and the American College of Radiology both recommend a modified Barium swallow study (also known as a video fluoroscopy swallow study) as a reasonable first diagnostic test based on expert consensus.[8,15] Clinical trials[16] support its use in SSc, highlighting its ability to identify the anatomic source of dysphagia as originating from the oral cavity, pharynx, and/or esophagus. Patients with concurrent reflux symptoms or evidence of esophageal dysmotility by the Barium swallow study should be further assessed by esophagogastroduodenoscopy (EGD), as outlined in Section 3.2.
2.2. Additional Workup for Oropharyngeal Dysphagia
If a Barium swallow study suggests dysphagia originating at the level of the oropharynx, overlap syndromes should be considered. The presence of sicca symptoms can be confirmed by a thorough history and physical exam, taking particular care to note any reported xerostomia (dry mouth), oral sores, or glossodynia that may contribute to dysphagia.[17] Salivary gland biopsies and Sjogren’s Syndrome antibodies do not correlate with sicca symptom severity in SSc patients, and thus are likely unnecessary as they would not affect management.[18] A comprehensive review of the diagnosis and treatment options of Sjogren’s Syndrome and/or sicca was recently published here.[17] Elevations in serum creatinine kinase (CK), aldolase, or antibodies associated with autoimmune myositis or myasthenia gravis (e.g. anti-acetylcholine receptor or muscle specific tyrosine kinase [MUSK] antibodies) would provide support for an underlying myopathy as a potential contributor to pharyngeal muscle weakness.[14,19] Although the identification of myositis or myasthenia gravis is important in determining optimal treatment strategies for oropharyngeal dysfunction, the nuances of their treatment are outside the scope of this review but may be found here.[20,21] Practice points for oropharyngeal dysphagia and/or globus sensation can be found in Table 3.
Table 3.
Practice Points and Research Agenda for Oropharyngeal Dysphagia or Globus Sensation in Systemic Sclerosis
Practice Points:
|
Research Agenda:
|
SSc: Systemic Sclerosis; CK: creatine kinase; MUSK: muscle-specific tyrosine kinase
3.0. HEARTBURN, REFLUX, OR ESOPHAGEAL DYSPHAGIA
Estimates suggest that approximately 90% of patients with SSc report symptoms suggestive of esophageal involvement, most commonly heartburn or reflux. Dysphagia, regurgitation, chronic cough, and hoarseness are other frequently reported symptoms. These are attributed to chronic, inadequately controlled baseline GERD, reduced lower esophageal sphincter (LES) pressures, and hypomotility of the distal two-thirds of the esophagus in SSc leading to reduced clearance.[22,23]
The Montreal definition for GERD, adopted by the American Gastroenterological Association (AGA), defines GERD as “a condition which develops when the reflux of stomach content causes troublesome symptoms and/or complications,”[24] further clarifying the signifier “troublesome” to mean a symptom or complication with an adverse effect on the well-being of the individual. The reported incidence of GERD symptoms is much higher in SSc than in the general population, and thus patients with SSc are at higher risk for complications such as strictures, esophageal erosions/ulcerations, Barrett’s esophagus, and adenocarcinoma. Clinicians should also have an awareness of overlap disorders such as eosinophilic esophagitis (EoE), hiatal hernia, or gastroparesis, which may mimic or exacerbate GERD symptoms.[23,25]
3.1. Empiric Acid Suppression Therapy
The AGA guidelines for the management of GERD[24] outline that daily standard-dose proton pump inhibitors (PPIs) (see Table 4) should be considered first-line therapy based on strong support from clinical trial data. PPIs are beneficial both for symptom management and for treating complications such as esophageal ulcers and strictures.[7] This recommendation is supported by a Cochrane review[26] that concluded that PPIs are superior to histamine-2 receptor antagonists (H2RAs), which in turn are superior to placebo in the treatment of symptomatic reflux and healing of esophagitis. No significant difference was found in the efficacy among different PPIs (esomeprazole, lansoprazole, omeprazole, pantoprazole, rabeprazole) by meta-analysis. In patients that initially responded to therapy, this symptomatic benefit extended to the 6- and 12-month follow up periods. While the only small randomized control trial[27] for PPIs in SSc did not see benefit at 12-month follow up, this study only investigated lansoprazole and did not assess for outcomes related to esophageal healing. Acknowledging the limitations of this small study, chronic PPI therapy in SSc is guided by abundant data from the GI literature, with both the EULAR and the UKSSG guidelines in agreement with the AGA’s recommendation.[7,8]
Table 4.
Standard Dosing for Proton Pump Inhibitors (PPIs) and Histamine-2 Receptor Antagonists (H2RAs)
| Standard Dosing | |
|---|---|
| PPIs | Esomeprazole 40mg daily |
| Lansoprazole 30mg daily | |
| Pantoprazole 40mg daily | |
| Omeprazole 20mg daily | |
| Rabeprazole 20mg daily | |
| H2RAs | Cimetidine 400mg twice daily |
| Famotidine 20mg twice daily | |
| Nizatidine 150mg twice daily | |
| Ranitidine 150mgtwice daily |
While limited data exists for the efficacy of twice-daily dosing of PPIs, the AGA guidelines[24] state that gastroenterologist expert opinion supports this increased dosing for patients with breakthrough symptoms. The guidelines propose H2RAs as an alternative acid suppressant when patients experience significant adverse effects from PPIs.[24] Expert consensus across gastroenterologists and rheumatologists (when dealing with reflux in SSc) is that all medications should be titrated to optimize symptom control, while utilizing the lowest possible therapeutic medication dose.[8,24] Any patients that continue to experience symptoms despite maximal acid-suppression therapy should be considered “treatment failures” and should undergo additional diagnostic evaluation.
3.2. Workup for Treatment-Refractory Reflux
The AGA and UKSSG both recommend that EGD should be utilized in treatment-refractory GERD to rule out other complications and diagnoses, particularly if “red flag” or concerning symptoms, such as dysphagia, are present.[8,24] Appropriate treatment for esophageal strictures, EoE, esophageal candidiasis or other infections, esophageal adenocarcinoma, etc., should be pursued if present.[28–30] While there is evidence[31] that suggests esophageal mucosal abnormalities may predate symptoms in SSc, further research is needed to determine the utility of screening asymptomatic patients with EGD and/or the role of prophylactic acid suppression in this patient population. Currently, the EULAR recommendations state that prescribing empiric PPI therapy in asymptomatic patients should be done with caution, given that long-term use may be associated with nutritional deficiencies and increased infection risk.[7]
3.3. Esophageal Dysmotility Evaluation and Consideration of Prokinetic Therapies
If EGD does not reveal an alternative cause for reflux symptoms in SSc, then an esophageal dysmotility evaluation is warranted.[23] Symptoms suggestive of esophageal dysmotility include dysphagia with both solids and liquids and chronic reflux (note: dysphagia primarily with solids would be more suggestive of a structural issue).[32] Gastric dysmotility must also be considered, as the two may co-exist and the diagnostics and management may differ from the presence of esophageal dysmotility alone; please see Section 4.0 for further discussion of this subject.
Multiple gastroenterology expert societies[33–35] recommend high-resolution esophageal manometry (HREM) as an appropriate follow-up test to EGD for confirming a diagnosis of esophageal dysmotility or lower esophageal sphincter (LES) dysfunction in the general population. Multiple small studies[36,37] provide support for the use of HREM in SSc for this indication. The utility of screening and treatment for dysmotility in asymptomatic SSc patients is an area that needs additional research, as growing evidence supports an association between chronic regurgitation, micro-aspiration, and the presence of interstitial lung disease. Therefore, identifying and treating asymptomatic, uncontrolled dysphagia may positively impact long-term outcomes.[38,39]
Both the EULAR and UKSSG guidelines recommend the use of prokinetic agents for SSc patients with GI dysmotility.[7,8] Notably there are very few clinical trials examining the use of prokinetic agents in SSc, particularly in the treatment of esophageal dysmotility. Two drugs, cisapride and metoclopramide, were specifically studied in SSc as treatments for esophageal dysmotility, each demonstrating benefit in small randomized cross-over trials.[40,41] However, cisapride has since been restricted in the United States due to its known association with QT prolongation leading to an increased risk of developing torsades de pointes, and the AGA guidelines for GERD recommend against the use of metoclopramide citing lack of high-quality data and a negative toxicity profile.[8,24] Prucalopride, which has a similar mechanism to cisapride but also a significantly improved cardiac profile, has demonstrated an enhancing effect on esophageal motility in general population studies,[42,43] but has not been studied in SSc-esophageal disease.[44] Another drug, buspirone, has shown benefit as add-on therapy to PPIs in SSc in one clinical trial.[45] Bethanechol is an additional promising agent seen to provide benefit in a small study of esophageal dysmotility.[46] Rheumatologist expert consensus recommends the use of prokinetic agents in the treatment of dysphagia on a case-by-case basis, as there is a great need for research on the long-term efficacy of these agents in the SSc population.[7]
3.4. Combination Therapy in Refractory Reflux
Studies examining combinations of acid suppressant pharmacotherapies demonstrated no significant benefit in the general population, though studies in the SSc population are limited.[24] A new area of interest is combining acid suppression with promotility agents and other classes of medications to improve symptoms in patients with treatment-refractory disease.
For example, buspirone, domperidone, and algycon were each found to improve reflux symptoms when used in combination with PPIs in small placebo-control trials within the SSc population.[47,48] However, given the lack of long-term outcome data in combination therapy and the potential for adverse events with select agents, such decisions should generally be made in consultation with an experienced gastroenterologist.
3.5. Anti-Reflux Surgery in SSc
While the management of pharmacotherapy-refractive reflux symptoms in the general population typically involves surgery, this approach should be avoided when possible in patients with SSc.[8,24] Multiple studies[49–52] have found that while surgical interventions (such as Nissen fundoplication, Belsey fundoplication, Collis gastroplasty, and Roux-en-Y gastric bypass) are effective in immediately reducing reflux symptoms in SSc, a significant number of cases ultimately develop postoperative dysphagia as a complication. Interestingly, one retrospective review[51] compared Roux-en-Y gastric bypass to several other surgical interventions for GERD, and found that Roux-en-Y was associated with fewer symptoms of dysphagia at 21-month follow-up. However, this procedure is also associated with increased incidence of small intestinal bacterial overgrowth (SIBO). Given the already increased prevalence of small bowel dysmotility and SIBO in the SSc patient population, this is a procedure that should still be pursued with caution.[29] Practice points for heartburn, reflux, and/or esophageal dysphagia can be found in Table 5.
Table 5.
Practice Points and Research Agenda for Heartburn, Reflux, or Esophageal Dysphagia in Systemic Sclerosis
Practice Points:
|
Research Agenda:
|
PPI: proton pump inhibitor; SSc: Systemic Sclerosis; H2RA: histamine-2 receptor antagonist; GI: gastrointestinal; EGD: esophagogastroduodenoscopy; GERD: gastroesophageal reflux disease; HREM: high resolution esophageal manometry
4.0. NAUSEA, VOMITING, BLOATING, OR ABDOMINAL DISTENTION
The differential diagnosis for nausea, vomiting, bloating, and/or abdominal distention symptoms is broad and should receive a thorough standard evaluation prior to pursuit of SSc-specific disease processes. The AGA has published an excellent technical review[53] encompassing the evaluation of both acute and chronic nausea/vomiting symptoms, but particularly important considerations for the SSc population should include GI dysmotility, dysbiosis (including SIBO), and malabsorption. Assessment of the adverse effects of medications, particularly immunosuppressives used in the management of SSc (e.g. mycophenolate mofetil, nintedanib, methotrexate) and dose-adjustments when necessary should be considered prior to pursuing further GI testing.
Though symptoms do not consistently associate with specific areas of GI dysmotility, if nausea, vomiting, bloating, and/or abdominal distention are coupled with diarrhea and/or constipation, small or large bowel involvement should receive additional consideration as discussed in Sections 5.0 and 6.0.[23] In contrast, overlying symptoms of refractory reflux, epigastric pain, early satiety, and/or postprandial fullness may suggest an underlying gastric dysmotility (such as gastroparesis or abnormal gastric accommodation), which affects up to 50% of SSc patients.[23,54]
4.1. Evaluation of Gastric Motility
Gastroparesis is defined as “a syndrome of objectively delayed gastric emptying in the absence of mechanical obstruction and cardinal symptoms including early satiety, postprandial fullness, nausea, vomiting, bloating, and upper abdominal pain.”[55] AGA guidelines[53] recommend EGD to assess for peptic ulcers, Helicobacter pylori infection, and gastric outlet obstruction. The UKSSG guidelines are in agreement and recommend follow-up testing with gastric emptying studies or ultrasonography if symptoms are suggestive of gastric dysmotility but there is no evidence of outlet obstruction.[8] The 4-hour gastric emptying scintigraphy study is the diagnostic standard for gastroparesis, and a combined solid-liquid phase study shows increased sensitivity.[55,56]
4.2. Management of Gastric Dysmotility
In patients with confirmed gastric dysmotility, the UKSSG guidelines recommend dietary modification with or without adjunct prokinetic therapies as the mainstay of treatment.[8] While the studies[57] available on dietary modification in SSc are small and limited in scope, dietary counseling may recommend such interventions as a low-fat and low-fiber diet, small meal sizes, and high-calorie liquids to optimize nutrition.[55] There is very limited data specific to SSc in the use of prokinetic agents for the treatment of gastric dysmotility, and thus existing guidelines recommend that these agents be used with caution.
Presently metoclopramide is the only medication that is formally approved in the United States for the long-term treatment of gastroparesis.[55] One small trial[58] has shown the benefit of improved gastric emptying times extends to SSc patients as well. Unfortunately, significant extrapyramidal side effects such as tardive dyskinesia warrant caution in its use. For this reason, the lowest possible therapeutic dose is recommended (starting with 5mg before meals and at bedtime, with a maximal total daily dose of 40mg). Drug holidays should be initiated whenever possible, and physicians should maintain a low threshold to discontinue therapy, particularly in the setting of any new neurologic symptoms.[55]
Domperidone, a dopamine-2 antagonist similar to metoclopramide with fewer side effects, can be obtained only through special government programs in the United States. Intravenous erythromycin is another consideration, although this medication is limited by its multiple times a day dosing and chronic propensity towards tachyphylaxis.[55] Prucalopride has also been identified in general population studies[59] as a new effective agent for the treatment of gastroparesis. Patients with persistent or progressive symptoms despite dietary modification and/or adjunct prokinetics may be treated together with an experienced gastroenterologist for consideration of alternative or combination therapies (e.g. mirtazapine, buspirone, pyridostigmine, etc.).[60,61] Recent studies[62,63] suggest that transcutaneous electrical nerve stimulation may also improve gastric transit in SSc patients, but larger studies are needed to prove clinical efficacy.
An evaluation by a gastroenterologist and nutritionist is particularly important when managing patients with progressive weight loss and/or malnutrition attributed to GI dysfunction. Patients may need an assessment for supplemental enteral or parenteral access for nutrition if they are unable to maintain a normal BMI and balanced diet on oral intake alone.[55,64] Practice points for nausea, vomiting, bloating, and/or distention can be found in Table 6.
Table 6.
Practice Points and Research Agenda for Nausea, Vomiting, Bloating, or Distention in Systemic Sclerosis
Practice Points:
|
Research Agenda:
|
SSc: Systemic Sclerosis; EGD: esophagogastroduodenoscopy
5.0. DIARRHEA
Diarrhea is another common presenting GI complaint in SSc, estimated to affect approximately 50% of patients.[8] Causes of diarrhea in this patient population are frequently attributed to SIBO, malabsorption, and GI tract dysmotility; however, an assessment for other common causes of diarrhea such as infection and inflammatory bowel disease should also be pursued.[64] Clostridium difficile infection is of particular concern given high exposure to the medical environment and frequent use of antibiotics (in the setting of immunosuppression, SIBO, digital ulcers, etc.). Excellent reviews for the workup of acute and chronic diarrhea in the general population have been published by the American Academy of Family Physicians and New England Journal of Medicine, respectively, and are provided here for reference.[65,66]
5.1. Clinical Presentation, Diagnosis, and Treatment of SIBO in SSc
Patients with SSc are at increased risk for SIBO due to the presence of small and/or large bowel dysmotility (leading to luminal stasis of food) and chronic exposure to high-dose acid suppression therapy, which reduces the physiologic gastric acid protection against excessive bacterial colonization of the GI tract.[67] SIBO is defined by the American Journal of Gastroenterology as “a condition in which the small bowel is colonized by excessive numbers of aerobic and anaerobic microbes that are normally found in the large intestine.”[68] This may present as a number of symptoms, such as episodic loose stools, halitosis, abdominal discomfort, distention, or bloating.[8,69] Symptoms may also present more insidiously as malnutrition, and thus rheumatologists should maintain a high index of suspicion for this diagnosis.[67] Identifying the true prevalence of SIBO in the SSc population remains challenging due to poor diagnostic testing options and therefore a high incidence of empiric antibiotic therapy trials. The gold standard test for diagnosis, jejunal culture, is often prohibited by the need for general anesthesia, which is frequently contraindicated in SSc due to the cardiopulmonary complications of the disease.[70] With these significant limitations, the incidence of SIBO in SSc is estimated to be somewhere between 30–62%.[69]
Currently, the utility of diagnostic testing versus empiric therapy in clinical cases with high suspicion for SIBO in SSc is unknown. The only validated diagnostic test for SIBO in SSc, given the relative contraindication for the gold standard jejunal culture, is the hydrogen or methane breath test following a glucose or lactulose bolus.[64] The EULAR guidelines recommend empiric courses of intermittent or rotating broad-spectrum antibiotics for the treatment of SIBO in SSc, based on expert consensus, acknowledging a lack of randomized control trial data available in SSc. No recommendation is made regarding the utility of diagnostic testing.[7] The UKSSG recommendations do not provide definitive guidance regarding diagnostic testing versus empiric treatment, stating that hydrogen breath testing may be used for diagnosis and empiric antibiotics may be used for treatment.[8] Finally, the AGA’s recent clinical practice update for SIBO, not specific to SSc, acknowledges that empiric antibiotics are the mainstay of treatment based on poor-quality data, and that hydrogen/methane breath testing may be useful in identifying the subset of patients that will respond to therapy.[71] Additional research is needed in the general population, as well as in SSc, to further define the risk-benefit ratio of empiric therapy versus initial breath testing. The AGA guidelines do state, however, that repeated diagnostic testing is not necessary if patients respond to antibiotic therapy.[71]
As a whole, antibiotic treatment for SIBO remains empiric with limited data to support one antibiotic regimen versus another. Rifaximin has the most robust data to support its use, with successful clinical trials in both the general population[72] and SSc population.[73] There is no consensus on the appropriate dosing of rifaximin for SIBO, and thus the current AGA guidelines[71] suggest a regimen of dividing a total daily dose of 800–1200mg throughout the day. There is also no consensus on length of antibiotic therapy, with studies ranging 1–4 weeks of therapy. Rate of recurrence is known to be high, cited as up to 44% of cases by 9 months, and thus the AGA, EULAR, and UKSSG guidelines suggest that some patients may need repeated or rotating antibiotic courses.[7,8,71] Other antibiotics that have been studied in SSc for the treatment of SIBO, with moderate success, include ciprofloxacin, norfloxacin, amoxicillin, and metronidazole.[69] A full list of proposed antibiotic regimens for SIBO from the AGA is listed in Table 7.[71]
Table 7.
Proposed Antibiotic Regimens for Spontaneous Bacterial Overgrowth Syndrome (SIBO)
| Antibiotic | Recommended Dosing for SIBO Treatment |
|---|---|
| Amoxicillin-clavulanic acid | 500–125mg three times daily |
| Ciprofloxacin | 250mg twice daily |
| Doxycycline | 100mg twice daily |
| Metronidazole | 250mg three times daily |
| Neomycin | 500mg twice daily |
| Norfloxacin | 800mg daily |
| Rifaximin | 800–1200mg daily total in divided doses |
| Tetracycline | 250mg four times daily |
| Trimethoprim-sulfamethoxazole | 1 double strength tablet twice daily |
5.2. Evaluation and Treatment of Small Intestinal Dysmotility
Dysmotility of the small bowel is estimated to affect anywhere between 40–88% of patients with SSc, although more precise figures are challenging to define as studies have shown that motility abnormalities likely precede symptoms. Complications of small bowel dysmotility may include SIBO, chronic intestinal pseudo-obstruction (CIPO), or pneumatosis cystoides intestinalis (a rare complication of SSc where air-filled cysts develop within the submucosa of the intestines, which have a propensity to spontaneously rupture and cause pneumoperitoneum).[74] One screening test that may be particularly beneficial is a plain abdominal radiograph, which can distinguish overflow diarrhea secondary to constipation or chronic intestinal pseudo-obstruction from other causes.[75] Appropriate bowel regimen therapies and additional workup for constipation are discussed in Section 6.0. While manometry may be diagnostic for small bowel dysmotility, this method is time-intensive and the need for routine anesthesia limits its use to patients without significant cardiopulmonary complications.[64] Scintigraphy is recommended as an alternative diagnostic tool to assess small bowel transit times.[76] Given the relative safety and non-invasive approach of scintigraphy, it may be a reasonable to obtain prior to the initiation of anti-diarrheal agents or other motility agents.
Data supporting motility agents in the management of small intestinal SSc remains limited, although both octreotide and prucalopride have demonstrated symptomatic benefit in studies.[44,77,78] Pyridostigmine is an additional agent that shows promise in the treatment of CIPO, as demonstrated in pediatric case series.[79] For the management of diarrhea symptoms, the UKSSG guidelines recommend lifestyle modification through diet to increase stool consistency and/or a trial of loperamide.[8,57] Importantly, anti-diarrheal agents should not be utilized until a thorough evaluation of other treatable causes of diarrhea is complete.
5.3. Gut Microbiome Modification
A new area of study for GI symptom therapy involves modulation of the GI luminal microbiome. The UKSSG guidelines state that probiotics “may be useful in some patients” with SSc, but do not provide additional recommendations regarding strains, treatment strategies, or duration of use.[8] The probiotics Bifidobacterium infantis and Lactobacillus rhamnosusGG subjectively improve bloating, distention, and reflux in patients with SSc.[80] Data suggests that combination therapy of Saccharomyces boulardii with metronidazole may be more effective treatment for SIBO than either agent alone in SSc.[81] Fecal transplant has also demonstrated symptomatic benefit for SSc in a small randomized control trial.[82] Ultimately the data for all microbiome modulation agents is still limited and this is an area that requires further study in this population. Practice points for diarrhea can be found in Table 8.
Table 8.
Practice Points and Research Agenda for Diarrhea in Systemic Sclerosis
Practice Points:
|
Research Agenda:
|
SSc: Systemic Sclerosis; SIBO: spontaneous bacterial overgrowth syndrome
6.0. CONSTIPATION
Patients with SSc are prone to experience constipation due to colonic dysmotility leading to reduced transit times.[64] The AGA defines constipation as a subjective patient symptom, which may include the traditional “definition” of infrequent bowel movements (generally less than 3 weekly), but may also be broadened to include symptoms such as “hard stools, a feeling of incomplete evacuation, abdominal discomfort, bloating, and distention.”[83] Given the range of clinical presentations, a careful history and physical is essential to determine the nature of symptoms and to rule out contributory causes, such as endocrine or metabolic sources;[8] the AGA’s clinical position statement is provided here for reference.[83] Notably, colonoscopy is an appropriate first step for those patients who have not completed age-appropriate colon cancer screenings, or are exhibiting additional “alarm symptoms” such as sudden-onset constipation, blood in stools, anemia on lab work, or unintentional weight loss.[83]
6.1. Therapeutic Laxative Trial for Constipation
In patients who do not warrant initial colonoscopy screening or have no endoscopic findings to explain constipation symptoms, the AGA recommends an empiric trial of stool-bulking agents and/or laxatives to help improve symptoms.[83] The UKSSG guidelines suggest breaking down cases into three distinct symptom groups: normal urge with difficulty emptying, infrequent urge with difficulty emptying, and normal urge with normal emptying. Stool-bulking agents such as fiber may have symptomatic benefit in those patients with difficulty emptying but a normal urge to void. However, caution must be used in patients with SSc who have significantly delayed colonic transit, as stool bulking may not adequately stimulate bowel movements as would be expected in patients with a normal colon, and therefore may exacerbate bloating or distention.[84] Osmotic laxatives (such as polyethylene glycol or milk of magnesia) may benefit patients with an infrequent urge to void and difficulty evacuating the colon, however they can have the drawback of worsening bloating symptoms. Stimulant laxatives (such as bisacodyl or glycerol suppositories) may improve symptoms in those with a normal urge to void and normal sensation of emptying.[8,83] Newer agents that rheumatologists may utilize include colonic secretory agents such as lubiprostone, linaclotide, and plecanatide; studies[85–87] in the general population have demonstrated their symptomatic benefit in the treatment of chronic idiopathic constipation. Notably, these agents have not yet been specifically studied in the SSc population.
6.2. Assessment of Colonic Motility and Prokinetic Therapies
Patients who fail a therapeutic laxative trial for constipation are candidates for additional colonic transit studies, according to the AGA. These can include the use of radio-opaque markers or scintigraphy, following a rule out of defecatory disorders (please see Section 7.0 for additional discussion of fecal incontinence).[83] These diagnostic tests have been used in the SSc population with success.[88] Following a confirmation of slow colonic transit, patients may be considered for prokinetic therapy. Cisparide and prucalopride demonstrate symptomatic benefit in SSc patients experiencing constipation.[44,89] Prucalopride was also validated in general population studies, identifying this is an effective alternative medication in the treatment of constipation.[90] Practice points for constipation can be found in Table 9.
Table 9.
Practice Points and Research Agenda for Constipation in Systemic Sclerosis
Practice Points:
|
Research Agenda:
|
SSc: Systemic Sclerosis
7.0. FECAL INCONTINENCE
Symptoms of anorectal dysfunction are reported in up to 70% of patients with SSc, making this the second most involved portion of the GI tract following the esophagus.[74] The most commonly reported symptom is that of fecal incontinence (FI), defined by the American College of Gastroenterology (ACG) as the “involuntary loss of solid or liquid feces.”[91] Patients with SSc have been found in clinical studies[92] to have an impaired recto-anal inhibitory response (RAIR) that contributes to internal anal sphincter dysfunction, and consequently, FI. However, prior to RAIR assessment, the ACG guidelines recommend identifying whether patients have any other conditions that may predispose to FI symptoms, such as diarrhea, pelvic floor trauma (particularly obstetric injury), and neuropathy.[91] An evaluation of diarrhea in SSc is further discussed in Section 5.0.
7.1. Digital Rectal Examination and Conservative Management of FI
The ACG currently provides a strong recommendation[91] for performing a digital rectal examination (DRE) to assess for anal sphincter tone and presence of rectal masses if history and physical exam do not provide an alternative predisposing source; there are no rheumatology society guidelines presently on the subject of FI. Reduced internal anal sphincter tone would support an impaired RAIR that may be seen in SSc.[92] Patients with loose stools, diarrhea, or constipation that are contributing to FI should be managed with stool-modifying medications, discussed further in Sections 5.0 and 6.0, respectively. The ACG recommends that these medications are trialed, if clinically appropriate, prior to further diagnostic testing.[91]
7.2. Anorectal Manometry, Pelvic Physical Therapy, and Invasive Management of FI
In patients who do not respond to conservative management, the ACG provides a strong recommendation for anorectal manometry, balloon expulsion testing, and rectal sensation evaluation for follow-up diagnostic testing.[91] Presently only anorectal manometry has been studied[37] in the SSc population, and thus the clinical utility of the latter two diagnostics for SSc is currently unknown. Anorectal manometry may be used to confirm an impaired RAIR, and normal testing should prompt the reconsideration of alternative diagnoses.[37] For patients with confirmed anorectal dysfunction that have not responded to conservative management, the ACG provides a strong recommendation for pelvic floor rehabilitation therapy.[91] Studies[93] have demonstrated the benefits of this therapy extends to the SSc population as well.
The ACG guidelines outline a few alternative invasive options for the management of FI in patients that do not respond to symptom-modifying therapies or pelvis exercises. In the general population, minimally invasive sphincter bulking agents are given a weak recommendation and sacral nerve stimulation is given a strong recommendation. Additional surgical options include sphincteroplasty, artificial anal sphincters, and colostomy.[91] Sacral nerve stimulation has been studied in SSc with favorable outcomes,[94] but additional research is needed to clarify the benefit of other invasive management options. Pursual of these options in FI should not be conducted without the assistance of a specialized gastroenterologist or anorectal surgeon. Practice points for fecal incontinence can be found in Table 10.
Table 10.
Practice Points and Research Agenda for Fecal Incontinence in Systemic Sclerosis
Practice Points:
|
Research Agenda:
|
SSc: Systemic Sclerosis; FI: fecal incontinence
8.0. SUMMARY
In the evaluation of SSc-GI disease, a symptoms-based approach is practical and may help to guide the initial diagnostic assessment. While GI disease is common in SSc, there remains a great need for additional research understanding disease mechanism and assessing the efficacy of both known and novel therapies for these patients. Risk stratification of asymptomatic patients is also an area that needs further study, as the utility of empiric therapies to prevent or slow progression of symptoms is not known. Clinicians should aim to treat symptoms to optimize quality of life and potentially reduce the risk of known GI complications in SSc.
Funding Statement:
NIH/NIAMS K23 AR071473 to ZM; Scleroderma Research Foundation to ZM; Jerome L. Greene Foundation to ZM; Scleroderma Research Foundation
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Conflict of interest statement: None of the authors received any financial support or other benefits from commercial sources for the work reported in this manuscript, nor do any of the authors have any financial interests, which could create a potential conflict of interest or appearance thereof.
References
- [1].Denton CP, Khanna D. Systemic sclerosis. Lancet 2017October7;390(10103):1685–99. [DOI] [PubMed] [Google Scholar]
- [2].Hong BY, Giang R, Mbuagbaw L, Larche M, Thabane L. Factors associated with development of gastrointestinal problems in patients with scleroderma: a systematic review. Syst.Rev 2015December30;4:188,015-0176-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].McMahan ZH, Tucker AE, Perin J, Volkmann ER, Kulkarni S, Ziessman HA, et al. The relationship between gastrointestinal transit, Medsger GI severity, and UCLA GIT 2.0 symptoms in patients with systemic sclerosis. Arthritis Care.Res.(Hoboken) 2020October16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [4].Marie I, Leroi AM, Gourcerol G, Levesque H, Ménard JF, Ducrotte P. Fructose Malabsorption in Systemic Sclerosis. Medicine (Baltimore) 2015September;94(39):e1601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Volkmann ER, Hoffmann-Vold AM, Chang YL, Jacobs JP, Tillisch K, Mayer EA, et al. Systemic sclerosis is associated with specific alterations in gastrointestinal microbiota in two independent cohorts. BMJ Open Gastroenterol. 2017April1;4(1):e000134,2017–000134. eCollection 2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Kröner PT, Tolaymat OA, Bowman AW, Abril A, Lacy BE. Gastrointestinal Manifestations of Rheumatological Diseases. Am.J.Gastroenterol 2019September;114(9):1441–54. [DOI] [PubMed] [Google Scholar]
- [7].Kowal-Bielecka O, Fransen J, Avouac J, Becker M, Kulak A, Allanore Y, et al. Update of EULAR recommendations for the treatment of systemic sclerosis. Ann.Rheum.Dis 2017August;76(8):1327–39. [DOI] [PubMed] [Google Scholar]
- [8].Hansi N, Thoua N, Carulli M, Chakravarty K, Lal S, Smyth A, et al. Consensus best practice pathway of the UK scleroderma study group: gastrointestinal manifestations of systemic sclerosis. Clin.Exp.Rheumatol 2014Nov-Dec;32(6 Suppl 86):S,214–21. [PubMed] [Google Scholar]
- [9].Pope J, Harding S, Khimdas S, Bonner A, Canadian Scleroderma Research Group, Baron M. Agreement with guidelines from a large database for management of systemic sclerosis: results from the Canadian Scleroderma Research Group. J.Rheumatol 2012March;39(3):524–31. [DOI] [PubMed] [Google Scholar]
- [10].Eyigor S, Sezgin B, Kuntman BD, Karabulut G, Zihni Yargucu F, Ozturk K, et al. Oropharyngeal swallowing functions are impaired in patients with scleroderma. Clin.Exp.Rheumatol 2020May-Jun;38 Suppl125(3):169–70. [PubMed] [Google Scholar]
- [11].Shah AA, Wigley FM. Often forgotten manifestations of systemic sclerosis. Rheum.Dis.Clin.North Am 2008February;34(1):221,38; ix. [DOI] [PubMed] [Google Scholar]
- [12].Bali V, Dabra S, Behl AB, Bali R. A rare case of hidebound disease with dental implications. Dent.Res.J.(Isfahan) 2013July;10(4):556–61. [PMC free article] [PubMed] [Google Scholar]
- [13].Paik JJ. Muscle disease in scleroderma. Curr.Opin.Rheumatol 2018November;30(6):576–80. [DOI] [PubMed] [Google Scholar]
- [14].Zivković SA, Medsger TA,Jr. Myasthenia gravis and scleroderma: two cases and a review of the literature. Clin.Neurol.Neurosurg 2007May;109(4):388–91. [DOI] [PubMed] [Google Scholar]
- [15].Expert Panel on Gastrointestinal Imaging:, Levy AD, Carucci LR, Bartel TB, Cash BD, Chang KJ, et al. ACR Appropriateness Criteria(®) Dysphagia. J.Am.Coll.Radiol 2019May;16(5S):S104–15. [DOI] [PubMed] [Google Scholar]
- [16].Fraticelli P, Pisani AM, Benfaremo D, De Marino L, Campioni D, Carboni N, et al. Videofluorography swallow study in patients with systemic sclerosis: correlation with clinical and radiological features. Clin.Exp.Rheumatol 2019Jul-Aug;37 Suppl119(4):108–14. [PubMed] [Google Scholar]
- [17].Vivino FB, Bunya VY, Massaro-Giordano G, Johr CR, Giattino SL, Schorpion A, et al. Sjogren’s syndrome: An update on disease pathogenesis, clinical manifestations and treatment. Clin.Immunol 2019June;203:81–121. [DOI] [PubMed] [Google Scholar]
- [18].Salliot C, Mouthon L, Ardizzone M, Sibilia J, Guillevin L, Gottenberg JE, et al. Sjogren’s syndrome is associated with and not secondary to systemic sclerosis. Rheumatology (Oxford) 2007February;46(2):321–6. [DOI] [PubMed] [Google Scholar]
- [19].Bohlmeyer TJ, Wu AH, Perryman MB. Evaluation of laboratory tests as a guide to diagnosis and therapy of myositis. Rheum.Dis.Clin.North Am 1994November;20(4):845–56. [PubMed] [Google Scholar]
- [20].Oddis CV, Aggarwal R. Treatment in myositis. Nat.Rev.Rheumatol 2018May;14(5):279–89. [DOI] [PubMed] [Google Scholar]
- [21].Narayanaswami P, Sanders DB, Wolfe G, Benatar M, Cea G, Evoli A, et al. International consensus guidance for management of myasthenia gravis: 2020 update. Neurology 2020November3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].Rose S, Young MA, Reynolds JC. Gastrointestinal manifestations of scleroderma. Gastroenterol.Clin.North Am 1998September;27(3):563–94. [DOI] [PubMed] [Google Scholar]
- [23].McFarlane IM, Bhamra MS, Kreps A, Iqbal S, Al-Ani F, Saladini-Aponte C, et al. Gastrointestinal Manifestations of Systemic Sclerosis. Rheumatology (Sunnyvale) 2018;8(1):235. doi: 10.4172/2161,1149.1000235.Epub 2018March30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [24].Kahrilas PJ, Shaheen NJ, Vaezi MF, American Gastroenterological Association Institute, Clinical Practice and Quality Management Committee. American Gastroenterological Association Institute technical review on the management of gastroesophageal reflux disease. Gastroenterology 2008October;135(4):1392,1413, 1413.e1–5. [DOI] [PubMed] [Google Scholar]
- [25].Richter JE, Rubenstein JH. Presentation and Epidemiology of Gastroesophageal Reflux Disease. Gastroenterology 2018January;154(2):267–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [26].Khan M, Santana J, Donnellan C, Preston C, Moayyedi P. Medical treatments in the short term management of reflux oesophagitis. Cochrane Database Syst.Rev 2007April18;(2):CD003244. doi(2):CD003244. [DOI] [PubMed] [Google Scholar]
- [27].Pakozdi A, Wilson H, Black CM, Denton CP. Does long term therapy with lansoprazole slow progression of oesophageal involvement in systemic sclerosis? Clin.Exp.Rheumatol 2009May-Jun;27(3 Suppl 54):5–8. [PubMed] [Google Scholar]
- [28].Iwakiri K, Kinoshita Y, Habu Y, Oshima T, Manabe N, Fujiwara Y, et al. Evidence-based clinical practice guidelines for gastroesophageal reflux disease 2015. J.Gastroenterol 2016August;51(8):751–67. [DOI] [PubMed] [Google Scholar]
- [29].Carlson DA, Hinchcliff M, Pandolfino JE. Advances in the evaluation and management of esophageal disease of systemic sclerosis. Curr.Rheumatol.Rep 2015January;17(1):475,014–0475-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [30].Abonia JP, Wen T, Stucke EM, Grotjan T, Griffith MS, Kemme KA, et al. High prevalence of eosinophilic esophagitis in patients with inherited connective tissue disorders. J.Allergy Clin.Immunol 2013August;132(2):378–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [31].Thonhofer R, Siegel C, Trummer M, Graninger W. Early endoscopy in systemic sclerosis without gastrointestinal symptoms. Rheumatol.Int 2012January;32(1):165–8. [DOI] [PubMed] [Google Scholar]
- [32].Johnston BT. Oesophageal dysphagia: a stepwise approach to diagnosis and management. Lancet Gastroenterol.Hepatol 2017August;2(8):604–9. [DOI] [PubMed] [Google Scholar]
- [33].Gyawali CP, Carlson DA, Chen JW, Patel A, Wong RJ, Yadlapati RH. ACG Clinical Guidelines: Clinical Use of Esophageal Physiologic Testing. Am.J.Gastroenterol 2020September;115(9):1412–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [34].Liu LWC, Andrews CN, Armstrong D, Diamant N, Jaffer N, Lazarescu A, et al. Clinical Practice Guidelines for the Assessment of Uninvestigated Esophageal Dysphagia. J.Can.Assoc.Gastroenterol 2018February9;1(1):5–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [35].Trudgill NJ, Sifrim D, Sweis R, Fullard M, Basu K, McCord M, et al. British Society of Gastroenterology guidelines for oesophageal manometry and oesophageal reflux monitoring. Gut 2019October;68(10):1731–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [36].Aggarwal N, Lopez R, Gabbard S, Wadhwa N, Devaki P, Thota PN. Spectrum of esophageal dysmotility in systemic sclerosis on high-resolution esophageal manometry as defined by Chicago classification. Dis.Esophagus 2017December1;30(12):1–6. [DOI] [PubMed] [Google Scholar]
- [37].Luciano L, Granel B, Bernit E, Harle JR, Baumstarck K, Grimaud JC, et al. Esophageal and anorectal involvement in systemic sclerosis: a systematic assessment with high resolution manometry. Clin.Exp.Rheumatol 2016Sep-Oct;34 Suppl100(5):63–9. [PubMed] [Google Scholar]
- [38].Tétreault MP, Kahrilas P. GI Manifestations With a Focus on the Esophagus: Recent Progress in Understanding Pathogenesis. Curr.Rheumatol.Rep 2019July3;21(8):42,019–0841-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [39].Denaxas K, Ladas SD, Karamanolis GP. Evaluation and management of esophageal manifestations in systemic sclerosis. Ann.Gastroenterol 2018Mar-Apr;31(2):165–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [40].Kahan A, Chaussade S, Gaudric M, Freitag B, Amor B, Menkes CJ, et al. The effect of cisapride on gastro-oesophageal dysfunction in systemic sclerosis: a controlled manometric study. Br.J.Clin.Pharmacol 1991June;31(6):683–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [41].Ramirez-Mata M, Ibañez G, Alarcon-Segovia D. Stimulatory effect of metoclopramide on the esophagus and lower esophageal sphincter of patients of patients with PSS. Arthritis Rheum. 1977Jan-Feb;20(1):30–4. [DOI] [PubMed] [Google Scholar]
- [42].Lei WY, Hung JS, Liu TT, Yi CH, Chen CL. Influence of prucalopride on esophageal secondary peristalsis in reflux patients with ineffective motility. J.Gastroenterol.Hepatol 2018March;33(3):650–5. [DOI] [PubMed] [Google Scholar]
- [43].Yi CH, Lei WY, Hung JS, Liu TT, Chen CL. Effects of prucalopride on esophageal secondary peristalsis in humans. Clin.Transl.Gastroenterol 2016November10;7(11):e202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [44].Vigone B, Caronni M, Severino A, Bellocchi C, Baldassarri AR, Fraquelli M, et al. Preliminary safety and efficacy profile of prucalopride in the treatment of systemic sclerosis (SSc)-related intestinal involvement: results from the open label cross-over PROGASS study. Arthritis Res.Ther 2017June20;19(1):145,017–1340-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [45].Karamanolis GP, Panopoulos S, Denaxas K, Karlaftis A, Zorbala A, Kamberoglou D, et al. The 5-HT1A receptor agonist buspirone improves esophageal motor function and symptoms in systemic sclerosis: a 4-week, open-label trial. Arthritis Res.Ther 2016September1;18(1):195,016–1094-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [46].Agrawal A, Hila A, Tutuian R, Mainie I, Castell DO. Bethanechol improves smooth muscle function in patients with severe ineffective esophageal motility. J.Clin.Gastroenterol 2007April;41(4):366–70. [DOI] [PubMed] [Google Scholar]
- [47].Karamanolis GP, Denaxas K, Panopoulos S, Bournia KV, Zorbala A, Kamberoglou D, et al. Severe oesophageal disease and its associations with systemic sclerosis. Clin.Exp.Rheumatol 2017Sep-Oct;35 Suppl106(4):82–5. [PubMed] [Google Scholar]
- [48].Foocharoen C, Chunlertrith K, Mairiang P, Mahakkanukrauh A, Suwannaroj S, Namvijit S, et al. Effectiveness of add-on therapy with domperidone vs alginic acid in proton pump inhibitor partial response gastro-oesophageal reflux disease in systemic sclerosis: randomized placebo-controlled trial. Rheumatology (Oxford) 2017February;56(2):214–22. [DOI] [PubMed] [Google Scholar]
- [49].Orringer MB, Orringer JS, Dabich L, Zarafonetis CJ. Combined Collis gastroplasty--fundoplication operations for scleroderma reflux esophagitis. Surgery 1981October;90(4):624–30. [PubMed] [Google Scholar]
- [50].Poirier NC, Taillefer R, Topart P, Duranceau A. Antireflux operations in patients with scleroderma. Ann.Thorac.Surg 1994July;58(1):66,72; discussion 72–3. [DOI] [PubMed] [Google Scholar]
- [51].Kent MS, Luketich JD, Irshad K, Awais O, Alvelo-Rivera M, Churilla P, et al. Comparison of surgical approaches to recalcitrant gastroesophageal reflux disease in the patient with scleroderma. Ann.Thorac.Surg 2007November;84(5):1710,5; discussion 1715–6. [DOI] [PubMed] [Google Scholar]
- [52].Mansour KA, Malone CE. Surgery for scleroderma of the esophagus: a 12-year experience. Ann.Thorac.Surg 1988November;46(5):513–4. [DOI] [PubMed] [Google Scholar]
- [53].Quigley EM, Hasler WL, Parkman HP. AGA technical review on nausea and vomiting. Gastroenterology 2001January;120(1):263–86. [DOI] [PubMed] [Google Scholar]
- [54].Gyger G, Baron M. Systemic Sclerosis: Gastrointestinal Disease and Its Management. Rheum.Dis.Clin.North Am 2015August;41(3):459–73. [DOI] [PubMed] [Google Scholar]
- [55].Camilleri M, Parkman HP, Shafi MA, Abell TL, Gerson L, American College of Gastroenterology. Clinical guideline: management of gastroparesis. Am.J.Gastroenterol 2013January;108(1):18,37; quiz 38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [56].Miller JB, Gandhi N, Clarke J, McMahan Z. Gastrointestinal Involvement in Systemic Sclerosis: An Update. J.Clin.Rheumatol 2018September;24(6):328–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [57].Smith E, Pauling JD. The efficacy of dietary intervention on gastrointestinal involvement in systemic sclerosis: A systematic literature review. Semin.Arthritis Rheum 2019August;49(1):112–8. [DOI] [PubMed] [Google Scholar]
- [58].Johnson DA, Drane WE, Curran J, Benjamin SB, Chobanian SJ, Karvelis K, et al. Metoclopramide response in patients with progressive systemic sclerosis. Effect on esophageal and gastric motility abnormalities. Arch.Intern.Med 1987September;147(9):1597–601. [PubMed] [Google Scholar]
- [59].Carbone F, Van den Houte K, Clevers E, Andrews CN, Papathanasopoulos A, Holvoet L, et al. Prucalopride in Gastroparesis: A Randomized Placebo-Controlled Crossover Study. Am.J.Gastroenterol 2019August;114(8):1265–74. [DOI] [PubMed] [Google Scholar]
- [60].Malamood M, Roberts A, Kataria R, Parkman HP, Schey R. Mirtazapine for symptom control in refractory gastroparesis. Drug Des.Devel.Ther 2017March30;11:1035–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [61].Ahuja NK, Mische L, Clarke JO, Wigley FM, McMahan ZH. Pyridostigmine for the treatment of gastrointestinal symptoms in systemic sclerosis. Semin.Arthritis Rheum 2018August;48(1):111–6. [DOI] [PubMed] [Google Scholar]
- [62].Sallam H, McNearney TA, Doshi D, Chen JD. Transcutaneous electrical nerve stimulation (TENS) improves upper GI symptoms and balances the sympathovagal activity in scleroderma patients. Dig.Dis.Sci 2007May;52(5):1329–37. [DOI] [PubMed] [Google Scholar]
- [63].McNearney TA, Sallam HS, Hunnicutt SE, Doshi D, Chen JD. Prolonged treatment with transcutaneous electrical nerve stimulation (TENS) modulates neuro-gastric motility and plasma levels of vasoactive intestinal peptide (VIP), motilin and interleukin-6 (IL-6) in systemic sclerosis. Clin.Exp.Rheumatol 2013Mar-Apr;31(2 Suppl 76):140–50. [PubMed] [Google Scholar]
- [64].Sakkas LI, Simopoulou T, Daoussis D, Liossis SN, Potamianos S. Intestinal Involvement in Systemic Sclerosis: A Clinical Review. Dig.Dis.Sci 2018April;63(4):834–44. [DOI] [PubMed] [Google Scholar]
- [65].Barr W, Smith A. Acute diarrhea. Am.Fam.Physician 2014February1;89(3):180–9. [PubMed] [Google Scholar]
- [66].Donowitz M, Kokke FT, Saidi R. Evaluation of patients with chronic diarrhea. N.Engl.J.Med 1995March16;332(11):725–9. [DOI] [PubMed] [Google Scholar]
- [67].Polkowska-Pruszyńska B, Gerkowicz A, Szczepanik-Kułak P, Krasowska D. Small intestinal bacterial overgrowth in systemic sclerosis: a review of the literature. Arch.Dermatol.Res 2019January;311(1):1–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [68].Rezaie A, Buresi M, Lembo A, Lin H, McCallum R, Rao S, et al. Hydrogen and Methane-Based Breath Testing in Gastrointestinal Disorders: The North American Consensus. Am.J.Gastroenterol 2017May;112(5):775–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [69].Pittman N, Rawn SM, Wang M, Masetto A, Beattie KA, Larché M. Treatment of small intestinal bacterial overgrowth in systemic sclerosis: a systematic review. Rheumatology (Oxford) 2018October1;57(10):1802–11. [DOI] [PubMed] [Google Scholar]
- [70].Sawadpanich K, Soison P, Chunlertrith K, Mairiang P, Sukeepaisarnjaroen W, Sangchan A, et al. Prevalence and associated factors of small intestinal bacterial overgrowth among systemic sclerosis patients. Int.J.Rheum.Dis 2019April;22(4):695–9. [DOI] [PubMed] [Google Scholar]
- [71].Quigley EMM, Murray JA, Pimentel M. AGA Clinical Practice Update on Small Intestinal Bacterial Overgrowth: Expert Review. Gastroenterology 2020October;159(4):1526–32. [DOI] [PubMed] [Google Scholar]
- [72].Gatta L, Scarpignato C. Systematic review with meta-analysis: rifaximin is effective and safe for the treatment of small intestine bacterial overgrowth. Aliment.Pharmacol.Ther 2017March;45(5):604–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [73].Parodi A, Sessarego M, Greco A, Bazzica M, Filaci G, Setti M, et al. Small intestinal bacterial overgrowth in patients suffering from scleroderma: clinical effectiveness of its eradication. Am.J.Gastroenterol 2008May;103(5):1257–62. [DOI] [PubMed] [Google Scholar]
- [74].Sallam H, McNearney TA, Chen JD. Systematic review: pathophysiology and management of gastrointestinal dysmotility in systemic sclerosis (scleroderma). Aliment.Pharmacol.Ther 2006March15;23(6):691–712. [DOI] [PubMed] [Google Scholar]
- [75].Sattar B, Chokshi RV. Colonic and Anorectal Manifestations of Systemic Sclerosis. Curr.Gastroenterol.Rep 2019July8;21(7):33,019-0699-0. [DOI] [PubMed] [Google Scholar]
- [76].von der Ohe MR, Camilleri M. Measurement of small bowel and colonic transit: indications and methods. Mayo Clin.Proc 1992December;67(12):1169–79. [DOI] [PubMed] [Google Scholar]
- [77].Soudah HC, Hasler WL, Owyang C. Effect of octreotide on intestinal motility and bacterial overgrowth in scleroderma. N.Engl.J.Med 1991November21;325(21):1461–7. [DOI] [PubMed] [Google Scholar]
- [78].Nikou GC, Toumpanakis C, Katsiari C, Charalambopoulos D, Sfikakis PP. Treatment of small intestinal disease in systemic sclerosis with octreotide: a prospective study in seven patients. J.Clin.Rheumatol 2007June;13(3):119–23. [DOI] [PubMed] [Google Scholar]
- [79].Manini ML, Camilleri M, Grothe R, Di Lorenzo C. Application of Pyridostigmine in Pediatric Gastrointestinal Motility Disorders: A Case Series. Paediatr.Drugs 2018April;20(2):173–80. [DOI] [PubMed] [Google Scholar]
- [80].Frech TM, Khanna D, Maranian P, Frech EJ, Sawitzke AD, Murtaugh MA. Probiotics for the treatment of systemic sclerosis-associated gastrointestinal bloating/ distention. Clin.Exp.Rheumatol 2011Mar-Apr;29(2 Suppl 65):S22–5. [PubMed] [Google Scholar]
- [81].García-Collinot G, Madrigal-Santillán EO, Martínez-Bencomo MA, Carranza-Muleiro RA, Jara LJ, Vera-Lastra O, et al. Effectiveness of Saccharomyces boulardii and Metronidazole for Small Intestinal Bacterial Overgrowth in Systemic Sclerosis. Dig.Dis.Sci 2020April;65(4):1134–43. [DOI] [PubMed] [Google Scholar]
- [82].Fretheim H, Chung BK, Didriksen H, Bækkevold ES, Midtvedt Ø, Brunborg C, et al. Fecal microbiota transplantation in systemic sclerosis: A double-blind, placebo-controlled randomized pilot trial. PLoS One 2020May21;15(5):e0232739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [83].Locke GR 3rd, Pemberton JH, Phillips SF. American Gastroenterological Association Medical Position Statement: guidelines on constipation. Gastroenterology 2000December;119(6):1761–6. [DOI] [PubMed] [Google Scholar]
- [84].McMahan ZH, Hummers LK. Systemic sclerosis--challenges for clinical practice. Nat.Rev.Rheumatol 2013February;9(2):90–100. [DOI] [PubMed] [Google Scholar]
- [85].Johanson JF, Morton D, Geenen J, Ueno R. Multicenter, 4-week, double-blind, randomized, placebo-controlled trial of lubiprostone, a locally-acting type-2 chloride channel activator, in patients with chronic constipation. Am.J.Gastroenterol 2008January;103(1):170–7. [DOI] [PubMed] [Google Scholar]
- [86].Miner PB Jr, Koltun WD, Wiener GJ, De La Portilla M, Prieto B, Shailubhai K, et al. A Randomized Phase III Clinical Trial of Plecanatide, a Uroguanylin Analog, in Patients With Chronic Idiopathic Constipation. Am.J.Gastroenterol 2017April;112(4):613–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [87].Lembo AJ, Kurtz CB, Macdougall JE, Lavins BJ, Currie MG, Fitch DA, et al. Efficacy of linaclotide for patients with chronic constipation. Gastroenterology 2010March;138(3):886,95.e1. [DOI] [PubMed] [Google Scholar]
- [88].Wang SJ, Lan JL, Lan JL, Chen DY, Chen YH, Hsieh TY, et al. Effects of cisapride on colonic transit in patients with progressive systemic sclerosis. Clin.Rheumatol 2002August;21(4):271–4. [DOI] [PubMed] [Google Scholar]
- [89].Wang SJ, La JL, Chen DY, Chen YH, Hsieh TY, Lin WY. Effects of cisapride on oesophageal transit of solids in patients with progressive systemic sclerosis. Clin.Rheumatol 2002February;21(1):43–5. [DOI] [PubMed] [Google Scholar]
- [90].Bassotti G, Usai Satta P, Bellini M. Prucalopride for the treatment of constipation: a view from 2015 and beyond. Expert Rev.Gastroenterol.Hepatol 2019March;13(3):257–62. [DOI] [PubMed] [Google Scholar]
- [91].Wald A, Bharucha AE, Cosman BC, Whitehead WE. ACG clinical guideline: management of benign anorectal disorders. Am.J.Gastroenterol 2014August;109(8):1141,57; (Quiz) 1058. [DOI] [PubMed] [Google Scholar]
- [92].Heyt GJ, Oh MK, Alemzadeh N, Rivera S, Jimenez SA, Rattan S, et al. Impaired rectoanal inhibitory response in scleroderma (systemic sclerosis): an association with fecal incontinence. Dig.Dis.Sci 2004June;49(6):1040–5. [DOI] [PubMed] [Google Scholar]
- [93].Collins J, Mazor Y, Jones M, Kellow J, Malcolm A. Efficacy of anorectal biofeedback in scleroderma patients with fecal incontinence: a case-control study. Scand.J.Gastroenterol 2016December;51(12):1433–8. [DOI] [PubMed] [Google Scholar]
- [94].Kenefick NJ, Vaizey CJ, Nicholls RJ, Cohen R, Kamm MA. Sacral nerve stimulation for faecal incontinence due to systemic sclerosis. Gut 2002December;51(6):881–3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [95].Horowitz M, Maddern GJ, Maddox A, Wishart J, Chatterton BE, Shearman DJ. Effects of cisapride on gastric and esophageal emptying in progressive systemic sclerosis. Gastroenterology 1987August;93(2):311–5. [DOI] [PubMed] [Google Scholar]
- [96].Fiorucci S, Distrutti E, Bassotti G, Gerli R, Chiucchiù S, Betti C, et al. Effect of erythromycin administration on upper gastrointestinal motility in scleroderma patients. Scand.J.Gastroenterol 1994September;29(9):807–13. [DOI] [PubMed] [Google Scholar]
- [97].Ariyasu H, Iwakura H, Yukawa N, Murayama T, Yokode M, Tada H, et al. Clinical effects of ghrelin on gastrointestinal involvement in patients with systemic sclerosis. Endocr.J 2014;61(7):735–42. [DOI] [PubMed] [Google Scholar]
- [98].Horikoshi T, Matsuzaki T, Sekiguchi T. Effect of H2-receptor antagonists cimetidine and famotidine on interdigestive gastric motor activity and lower esophageal sphincter pressure in progressive systemic sclerosis. Intern.Med 1994July;33(7):407–12. [DOI] [PubMed] [Google Scholar]
- [99].Low AHL, Teng GG, Pettersson S, de Sessions PF, Ho EXP, Fan Q, et al. A double-blind randomized placebo-controlled trial of probiotics in systemic sclerosis associated gastrointestinal disease. Semin.Arthritis Rheum 2019December;49(3):411–9. [DOI] [PubMed] [Google Scholar]
- [100].Marighela TF, Arismendi MI, Marvulle V, Brunialti MKC, Salomão R, Kayser C. Effect of probiotics on gastrointestinal symptoms and immune parameters in systemic sclerosis: a randomized placebo-controlled trial. Rheumatology (Oxford) 2019November1;58(11):1985–90. [DOI] [PubMed] [Google Scholar]


