Abstract
COVID-19 is an infectious disease that affects the respiratory system and is caused by the novel coronavirus SARS-CoV-2. It was first reported in Wuhan, China, on December 31, 2019, and has affected the entire world. This pandemic has caused serious health, economic and social problems. In this situation, the only solution to combat COVID-19 is to accelerate the development of antiviral drugs and vaccines to mitigate the virus and develop better antiviral methods and excellent diagnostic and prevention techniques. With the development of nanotechnology, nanoparticles are being introduced to control COVID-19. Graphene oxide (GO), an oxidized derivative of graphene, is currently used in the medical field to treat certain diseases such as cancer. It is characterized by very important antiviral properties that allow its use in treating certain infectious diseases. The GO antiviral mechanism is discussed by the virus inactivation and/or the host cell receptor or by the physicochemical destruction of viral species. Moreover, the very high surface/volume ratio of GO allows the fixation of biomolecules by simple absorption. This paper summarizes the different studies performed on GO's antiviral activities and discusses GO-based biosensors for virus detection and approaches for prevention.
Keywords: COVID-19, Coronavirus SARS-CoV-2, Pandemic, Graphene oxide, Antiviral activity, Biosensors, Detection, Prevention
1. Introduction
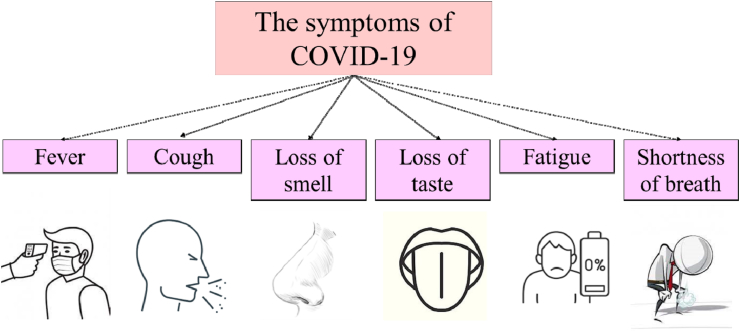
Coronavirus 2019 or COVID-19 is an infectious disease caused by the new coronavirus. This virus is part of the SARS-CoV-2 strain of coronavirus. Transmission of the virus occurs through direct contact with respiratory droplets generated by an infected person and direct contact with surfaces contaminated by the virus. The most common symptoms include fever, cough, fatigue, shortness of breath, loss of smell and taste (Maladie à coronavirus (2020) (Fig. 1). COVID-19 has become a public health problem worldwide. The World Health Organization (WHO) called COVID-19 a pandemic on March 11, 2020, stressing that a pandemic is the first time a pandemic is caused by a coronavirus (« Allocution liminaire du, 2020). The penetration of SARS-CoV-2 into human cells is based on its interaction with the angiotensin-2 converting enzyme (ACE2) present in the lungs. The lungs are therefore highly affected by SARS-CoV-2 (Hanff et al., 2020). The COVID-19 virus comprises spherical envelope particles containing positive single-stranded RNA associated with a nucleoprotein in a capsid, including protein (Mousavizadehet and Ghasemi, 2020; Seah et al., 2020). Several researchers are carrying out studies to develop a vaccine against this pandemic which has caused a serious problem for societies at the economic, social and health levels. Given the increasing number of infected patients and the possibility of SARS-CoV-2, which may transform into a more dangerous form due to mutations, scientists have been forced to adopt new strategies that focus primarily on new medications with antimicrobial antiviral properties (Möritz et al., 2001). The formulation of an effective vaccine against the new coronavirus started a few months after the pandemic. Some studies have suggested using previous vaccines or strategies for SARS-CoV-2 vaccine development (Weingartlet al., 2004). Other studies suggest that some vaccines under development may be effective. The development of neutralizing antibodies has already been studied to treat SARS-CoV-2 (Bisht et al., 2005; Kamet al., 2007). With the development of nanotechnology, nanoparticles are used in many applications, including biomedical applications. Graphene oxide (GO) is the oxidized form of graphene. It is a monoatomic layer nanomaterial formed by the oxidation of graphite. It is usually prepared by the Hummers' method (Hummers et and Offeman, 1958). Due to its functional groups such as epoxide, carboxyl and hydroxyl groups, GO is able to disperse in many solvents (Paredes et al., 2008). Graphene and GO play an important role in the medical and electronic fields due to their antibacterial properties and detection capabilities (Yeet al., 2015). In the last several decades, two-dimensional materials have been the most widely used materials for the manufacture of biosensors due to their high affinity, cost-effectiveness and ease of use (Nguyen et al., 2020). Studies have shown that GO and graphene have antibacterial and antiviral properties (Akhavan et al., 2012). It has been proven that the antibacterial properties of graphene derivatives are mainly due to their electron movement towards bacteria. This migration causes cytoplasmic efflux, decreases metabolism, affects the lipid membrane, induces oxidative stress, produces reactive oxygen species (ROS), loss of glutathione, and ultimately causes the death of the bacteria (DasariShareena et al., 2018). Seifi et al. showed that different factors affect the antibacterial performance of graphene materials, including the shape, size, functional group and electrical conductivity of graphene flakes, as well as the concentration, contact time and pH value of the graphene suspensions used in the corresponding microbial tests (Seifiet and Kamali, 2021). Besides, the GO has a higher negative charge, which makes it possible to have a high affinity for positively charged viruses (Yeet al., 2015). On the other hand, it has been shown that conjugation of GO with antibodies allows rapid detection of targeted viruses (Afroj et al., 2020). The current literature has suggested that antiviral mechanisms of graphene materials may be related to processes such as virus and/or host cell receptor inactivation, electrostatic trapping and physicochemical destruction of viral species. These effects can be enhanced by functionalizing and/or decorating carbons with species that improve graphene-virus interactions (Seifiet and Reza Kamali, 2021). Furthermore, functionalized graphene has demonstrated good viral capture capacity which, combined with heat or light inactivation, could be used as a disinfectant. Graphene sensor arrays can be implemented on standard utility textiles and for drug efficacy screening (Palmieriet and Papi, 2020). The company Zen Graphene Solutions (TSXV: ZEN) developed a GO-based composite ink functionalized by silver nanoparticles that fights the first strains of coronavirus (« Graphene ink may be use, 2020; Kumar et al., 2020). Therefore, it is necessary to emphasize the importance of using GO-based nanoparticles to develop antiviral agents and combat infectious diseases, including COVID-19, which has posed a threat to human health and the global economy. In this paper, we report the mechanism of viruses entry into host cells. Then we examine the most significant results of the antiviral activity of GO on selected viruses and the GO antiviral activity through photocatalysis, as well as we study the antiviral effects of the GO-silver complex to fight viruses. In addition, we discuss the use of GO and graphene as biosensors for virus detection. Finally, we present some applications of GO for protection against viruses.
Fig. 1.
Symptoms of COVID-19.
1.1. Mechanism of virus entry into cells
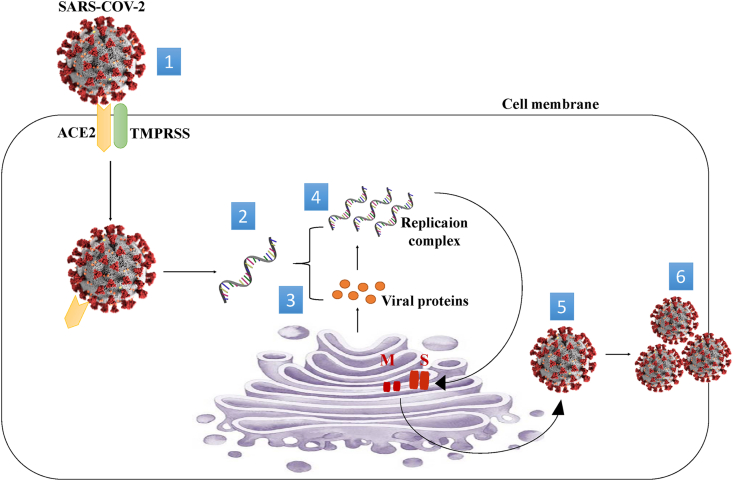
CoVs belong to the order Nidovirales, the family Coronaviridae and the subfamily Coronavirinae. The latter can be classified into four genera: alphacoronavirus, betacoronavirus, deltacoronavirus and gammacoronavirus (Cui et al., 2019; Adams et and Carstens, 2012). The first two are mammalian pathogens, the third is thought to affect avian and mammalian species, and the last infects birds (Li, 2016; Wooet al., 2012). Bats are the source of the alpha- and betacoronavirus genes, while birds originate the gamma- and delta coronavirus genes (Lauet al., 2018; Wooet al., 2012). To fight viruses effectively is necessary to know in detail how viruses can enter host cells. In general, viruses can enter host cells and multiply to form new viruses. The process of virus multiplication involves several steps, including attachment, penetration, uncoating, replication, assembly and release. Attachment is the first step in the infection process. It consists of bringing the viral surface into contact with the cell membrane. It is done by glycoproteins incorporated into the viral envelope for enveloped viruses or by capsid proteins for naked viruses. These proteins or glycoproteins bind to receptors located on the cytoplasmic membrane of the host cell. Coronavirus's entrance to the cell, first bind to a cell surface receptor for viral binding, then enter the endosomes and eventually fuse the viral and lysosomal membranes (Li, 2016). The SARS-CoV-2 S protein uses the cellular ACE2 receptor, a metalloprotease whose primary function is to degrade angiotensin II to angiotensin 1-7- to enter the host cell (Zhouet al., 2020). In SARS-CoV-1, studies have shown that binding the S1 subunit to ACE2 leads to a conformational change in the S protein, exposing S2 and allowing endocytosis and subsequent membrane fusion (de Wit et al., 2016). Besides, this membrane fusion involves cleavage by the cell surface protease TMPRSS2 at the S1/S2 junction and another S2 site (Hoffmannet al., 2020). In addition, studies have shown that the glycoprotein of SARS-CoV-2 hosts a furin cleavage site at the boundary between the S1/S2 subunits, which is treated during viral biosynthesis (Walls et al., 2020). After the fusion step with the host cell membrane and the subsequent release of viral genetic material into the cytoplasm, the cellular machinery translates the replicate gene into two polyproteins (pp1a and pp1ab) cleaved into many proteins essential to the viral cycle. It includes two viral proteases and an RNA-dependent RNA polymerase that assemble to form a large transcription and replication complex (de Wilde et al., 2017; de Wit et al., 2016). Then the synthesized RNA strands are combined with proteins to form the nucleocapsid and the assembly with envelope glycoproteins allows the budding of new viral particles (Fig. 2).
Fig. 2.
Mechanism of SARS-CoV-2 entry into the cell. 1 Spike protein on the virion binds to ACE2. TMPRSS2, an enzyme, helps the virion enter. 2 The virion releases its RNA. 3 Some RNA is translated into proteins by cell machinery. 4 Some of these proteins form a replication complex to make more RNA. 5 Proteins and RNA are assembled into a new virion in the Golgi and 6 Released.
1.2. Antiviral activity of GO
GO is the oxidized form of graphene with hydroxyl, epoxide, ketone and carboxyl functional groups located on its surface. The presence of oxygen on the edges and basal planes of GO increases its hydrophilicity, water dispersibility and bonding capacity compared to graphene (Aliyev et al., 2019; Ege et al., 2017). Due to its physicochemical properties, high surface-to-volume ratio, and extremely high mechanical strength, GO is widely used in the medical field as an antibacterial and anti-cancer agent (Rhazouaniet al., 2021). However, studies have shown that due to its two-dimensional structure, sharp edges, and negatively charged surface, this nanomaterial can interact with viruses and disrupt their plasma membrane or generate reactive oxygen species (« Synergistic Antiviral A, 2020). The antiviral activity of GO and reduced GO (rGO) was evaluated against PRV (a DNA virus) and PEDV (an RNA virus). PRV is a porcine herpes virus responsible for Aujesky's disease. At the same time, PEDV is a coronavirus that infects pigs and causes diarrhea and dehydration, leading to the animal's death. This study found that GO significantly inhibits infection of PRV and PEDV for a 2-log reduction in virus titers at non-cytotoxic concentrations. The strong antiviral activity of GO and rGO can be attributed to the unique monolayer structure and the negative charge. GO was shown to have strong antiviral activity when conjugated with PVP, a non-ionic polymer, but not with PDDA, a cationic polymer. Besides, GO inhibited the entry of viruses into host cells by causing structural destruction (Yeet al., 2015).
Ziem and his colleagues thermally produced polysulfated dendritic polyglycerol functionalized rGO sheets. This functionalized rGO material showed antiviral performance against various viruses, including orthopoxviruses, herpes simplex virus type 1 (HSV-1) and equine herpesvirus type 1 (EHV-1). These viruses cause major health problems worldwide, such as HSV-1 and orthopoxviruses that infect humans and EHV-1 that infects horses. The binding of polar polymers to the carbon material has been shown to result in improved water solubility. This also results in a bio-inert multifunctional surface, easily accessible for further post-modification and biological interactions. Subsequent sulphation of the material promotes its interaction with viruses. Here, graphene scaffolds with their large surface area offer the possibility to increase the valence of dendritic polyglycerol sulfate groups (Z. Bet al., 2016; Ziem et al., 2017).
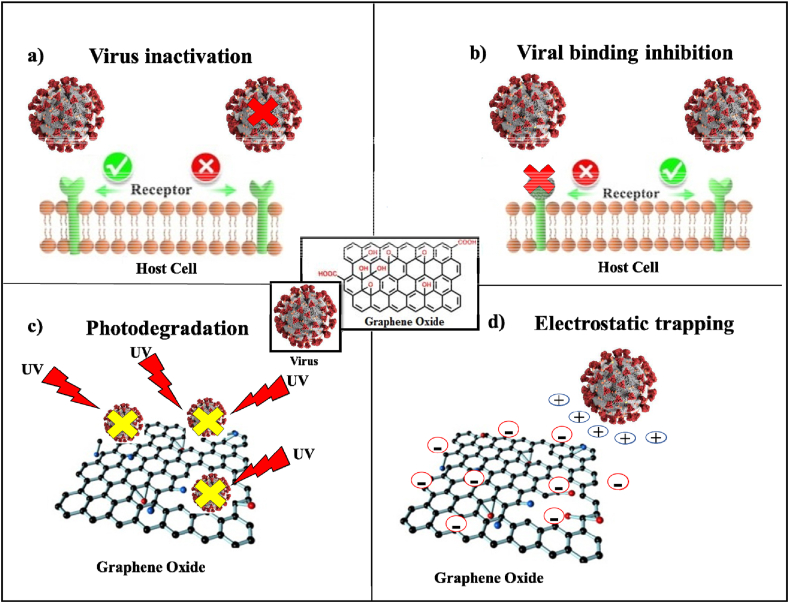
In vivo and in vitro studies have suggested that the hypericin-GO complex (GO/HY) has antiviral activity against New Duck Virus Disease (NDRV), a serious infectious disease of poultry. Hypericin has broad-spectrum antiviral activity against various viruses such as chronic hepatitis C virus and Sendai virus. The antiviral activity of the GO/HY complex was evaluated in DF-1 cells and ducklings contaminated with NDRV TH11. The GO/HY complex showed dose-dependent inhibition of NDRV replication, which can be attributed to virus inactivation or inhibition of virus binding (Fig. 3). Indirect immunofluorescence assay showed significantly suppressed protein expression in NDRV-infected DF-1 cells treated with GO/HY. In addition, GO/HY prolonged the survival time of the ducklings by reducing the pathological lesions caused by the infection and inhibiting viral replication in the liver and lungs (D. Xet al., 2019).
Fig. 3.
Description of the main mechanisms of GO's antiviral activity, a) including virus inactivation, b) viral binding inhibition, c) photodegradation and d) electrostatic trapping.
Another study showed that GO could effectively capture environmental viruses such as EV71 (a virus responsible for hand, foot and mouth disease) and H9N2 (endemic gastrointestinal avian influenza A virus). It may break down their surface proteins and extract viral RNA in an aqueous environment through GO surface bioreduction. The GO has been proven to be an excellent nanomaterial for high throughput virus detection and disinfection, demonstrating its great potential to prevent environmental infections (Songet al., 2015).
On the other hand, curcumin functionalized GO effectively inhibits respiratory syncytial virus (RSV) infection. This single-stranded RNA virus is considered the major viral pathogen of the lower respiratory tract in infants. This study showed that the composite could prevent RSV from infecting host cells by directly inactivating the virus and inhibiting viral binding. In addition, the curcumin functionalized GO showed high biocompatibility with host cells. These results indicated that the composite could provide new insights into antiviral therapy for RSV infection (Yang et al., 2017) (Table 1).
Table 1.
The antiviral activity of GO.
| Virus | Family | Species | Nucleic acid | Viral envelope | GO | Ref |
|---|---|---|---|---|---|---|
| RSV | Pneumoviridae | Human orthopneumovirus | RNA | enveloped | GO | Yang et al. (2017) |
| VSV | Rhabdoviridae | Indiana vesicolorum | RNA | enveloped | GO | Gholamiet al. (2017) |
| H9N2 | Orthomyxoviride | Influenza A | RNA | enveloped | GO | Songet al. (2015) |
| IBDV | Birnaviridae | Infectious Bursal disease virus | RNA | Non-enveloped | GO-AgNPs | Chen et al. (2016) |
| NDRV | Reoviridae | – | RNA | Non-enveloped | GO/HY | D. Xet al. (2019) |
| FCoV | Coronaviridae | Alphacoronavirus 1 | RNA | enveloped | GO-AgNPs | Chen et al. (2016) |
| PEDV | Coronaviridae | Porcine epidemic diarrhea virus | RNA | enveloped | GO | Yeet al. (2015) |
Researchers have shown that nanocomposites consisting of GO and partially reduced sulfonated GO inhibited herpes simplex virus type 1 (HSV-1) infections by preventing HSV-1 from attaching to host cells (Sametband et al., 2014). As already mentioned, the physicochemical properties of the surface of GO, rGO and structures based on these nanocarbons play a key role in their antiviral activities by affecting cell viability (Daset al., 2013). In this sense, it has been observed that the nano-topology of graphene layers has a significant implication on the physical/mechanical properties of graphene-enhanced alumina nanofibre scaffolds, which in turn play an important role in their antiviral performance against human pathogenic viruses (HPeV1 and IAV) by binding to viruses. Furthermore, since the performance of graphene materials relies on surface-dependent reactions, their concentration and exposure period should also be important factors influencing their overall antiviral activity. A good example is the work of Yang et al. (2017), which have investigated the concentration-dependent antiviral performance of curcumin-loaded, β-cyclodextrin-functionalized sulfonated graphene composite (GSCC) against the respiratory syncytial virus (RSV). RSV is an enveloped, negative-sense, single-stranded RNA virus belonging to the family Paramyxoviridae. This virus causes respiratory disease in infants and young children (Krishnan et al., 2004).
1.3. Antiviral activity of GO by photocatalysis
Recently, research on photocatalysis has been heavily explored, especially by using solar irradiation to trigger many photochemical reactions, such as generation of photocurrents, water and air purification, wastewater treatment, disinfection, and self-cleaning. Titanium dioxide TiO2 is considered an effective photocatalyst for water purification. Moreover, it can be used as an antibacterial agent due to its high oxidation capacity and super-hydrophilic character (Zaleska, 2008). Although TiO2 has interesting physicochemical properties, including high activity, stability, abundance, and low toxicity (Hoffmann et al., 1995; Wanget al., 2019), its application is limited due to charge carrier recombination and the inability to absorb visible light. In this regard, TiO2 has been modified by various elements and species, including doping, surface modification and coupling (Zaleska, 2008). As a result, graphene, GO and rGO have also been proposed for the surface modification of TiO2 and other wide bandgap semiconductors due to their large specific surface area, efficient adsorption of reagents, high conductivity, flexible structure and high stability (Lambertet al., 2009).
The GO has photocatalytic characteristics. It has been proven that GO has an excellent photocatalytic activity which could also be exploited to inhibit virus activity (Krishnamoorthy et al., 2011). For constant photodegradation, the virus must remain close to the surface of the GO under UV irradiation (Fig. 3). Therefore, the photocatalytic activities of the photocatalyst were studied under a wide spectrum of visible light. The aptamer-enhanced GO caused the inactivation of viruses and damaged the viruses' capsid proteins and nucleic acids during irradiation (Hu et al., 2012).
On the other hand, GO has been shown to have a higher bactericidal activity than graphene (NoorunnisaKhanamet and Hasan, 2019). GO concentration significantly influences bacterial viability; GO forms floating scaffolds at low concentrations, promoting bacterial growth, while at high levels, GO forms scaffolds capable of inhibiting bacterial growth (Palmieriet al., 2017). Furthermore, it has been suggested that the morphology of the GO membrane (roughness, sharpness and curvature) is important for the bactericidal effect; since the rougher or sharper membrane was shown a higher bactericidal activity (AbC. Set and Is, 2019). Sun et al. (Sunet al., 2017), have studied the antibacterial activity of the GO/graphite carbon nitride nanocomposite (GO/g-C3N4), as a free metal photocatalyst, against Escherichia coli (E. coli). The composite 100 μg/mL GO/g-C3N4 was found to kill 97.9% of E. coli after 120 min of visible light irradiation. This study facilitates a thorough understanding of the mechanism of visible light disinfection and provides an ideal candidate as a sterilizing agent (Sunet al., 2017).
1.4. Antiviral activity of GO–Ag
Studies have shown that silver nanoparticles (AgNPs) exhibit antiviral activity by interacting with viruses (Elechiguerraet al., 2005). The mechanism of inactivation of HIV-1 by AgNPs depends on physical binding via the glycoprotein gp120. Due to this interaction, AgNPs prevent the virus from binding to host cells. Interactions of HIV-1 with AgNPs have only been found in the nanoparticle range of 1–10 nm. Therefore, these interactions are dependent on particle size. The combination of AgNPs that exhibit antiviral activity with GO characterized by antimicrobial potential has shown antiviral potential against both enveloped and non-enveloped viruses. The study of the antiviral activity of GO with AgNPs was performed with enveloped and non-enveloped viruses, feline coronavirus (FCoV) with envelopes and infectious bursal disease virus (IBDV) without envelopes. Viral inhibition tests used to identify the antiviral activity of the GO-AgNPs complex showed that this complex inhibited 25% of FCoV infection and 23% of IBDV infection, while GO alone inhibited only 16% of FCoV infection but showed no antiviral activity against IBDV infection. According to these results, the combination of GO with AgNPs showed better antiviral activity compared to GO or silver alone. So, an application of this nanocomposite can protect against viruses (Chen et al., 2016). In addition to alloys and coatings, nanoparticles' antiviral performance has also been interesting in powder form. For example, silver nanoparticles of 2–15 nm show antiviral activity against COVID-19 (Jeremiah et al., 2020). However, as metal nanoparticles are highly reactive in powder form, the application of metal oxide powders is important.
Another similar study was done by Du et al. (Duet al., 2018) to prevent viral entry into cells. In this study, nanocomposites of AgNPs-modified GO were self-assembled by interfacial electrostatic forces. They used Porcine Reproductive and Respiratory Syndrome Virus (PRRSV), an RNA virus with a high impact on pigs, as a model. The results of this study reported that exposure to GO-AgNPs nanocomposites suppressed PRRSV infection. As a result, GO-AgNPs nanocomposites were found to have a very strong inhibitory effect compared to GO and AgNPs alone. Furthermore, studies on the mechanism of inhibition of antiviral activity showed that GO-AgNPs nanocomposites could prevent PRRSV from entering host cells, with an inhibition efficiency of 59.2%. In the same sense, the use of GO-AgNPs nanocomposites has improved the production of interferon-α (IFN-α) and IFN-stimulating genes (ISGs), which cause direct inhibition of virus proliferation (Duet al., 2018).
1.5. Graphene and GO sensors to detect viruses
Currently, countries around the world are working to combat COVID-19. For this reason, researchers and research laboratories have been trying to develop and produce test kits that are essential to detect a low viral load at an early stage. In general, reliable, rapid and specific detection of infectious pathogens is the best way to monitor epidemics and control their spread. Two main methods have been used to detect SARS-CoV-2 infection. The first method is based on the test of the CRISPR (clustered regularly interspaced short palindromic repeats) (Broughtonet al., 2020). It uses the genetic sequence of the virus and includes genomic sequencing. At the same time, the second method is based on the detection of antibodies specific to viral antigens (Riccòet al., 2020). Recent studies have suggested that nanosensors are considered the most promising approach to detect the novel SARS-CoV-2 (Laghribet al., 2020). The evolution of different carbon-based nanomaterials such as carbon nanotubes, graphene, and GO has enabled researchers to produce a new biosensor class. Especially, graphene is considered an interesting element for the manufacture of biosensors thanks to its many specific properties. Several studies are working on the development of a graphene-based sensor for the detection and control of COVID-19. Extensive research has successfully developed a transistor-based biosensor that detects SARS-CoV-2. The biosensing device comprises graphene sheets coated with a field-effect transistor (FET) with an antibody specific to the SARS-CoV-2 spike protein. This device is a highly sensitive immunodiagnostic method for COVID-19 that requires no pre-treatment or labeling sample (Seoet al., 2020). Nguyen and his collaborators have developed a chemo-phonon graphene-based biosensing system capable of inducing the SARS-CoV-2 spike protein at the 3.75 and 1 fg/mL limit in artificial saliva with phosphate-buffered saline. This system also showed selectivity for saliva proteins and MERS-CoV spike proteins (Nguyen et al., 2021).
Another study proposed a new technique based on electrochemical impedance spectroscopy for the early detection and classification of dengue virus (DENV). It was shown that the charge transfer resistance of a gold electrode coated with a GO-reinforced polymer was influenced by the type of virus and the amount exposed on the surface. The surfaces of the GO-polymers were functionalized by the DENV component using a self-assembly process that makes the polymer surface more selective and susceptible to the virus (Navakul et al., 2017). Other scientists have successfully developed an optical sensor for the DENV virus protein E, based on cadmium sulfide quantum dots composed of an amine-functionalized thin film of GO. A specific monoclonal antibody was covalently attached to the sensor to detect the targeted E proteins. This sensor exhibited an excellent detection limit with high potential sensitivity and stronger binding to DENV protein E (Omar et al., 2019). Afsahi et al. have developed a portable and inexpensive graphene biosensor to detect the Zika virus with a highly specific immobilized monoclonal antibody. The field-effect biosensor with monoclonal antibodies covalently bound to the graphene can detect Zika virus antigens at an early stage of the disease (Afsahiet al., 2018).
Joshi et al. constructed an electrochemical immunosensor from rGO thermal flakes derived from shellac to detect the H1N1 influenza virus. These rGO-based thermal sensors are used to fabricate various immunosensors and exhibit high stability and reproducibility (Joshi et al., 2020) (Kumar et al., 2020). On the other hand, it has been shown that rGO nanoparticles decorated with gold nanoparticles can be used as an antigen-functionalized surface to detect the existence of the base antigen of the hepatitis B virus (Mfet al., 2018). A recent study has established a biosensing platform by 3D nanoimprinting of three-dimensional electrodes, coating the electrodes with rGO nanoflakes and immobilizing specific viral antigens on the rGO nanoflakes. The detection of S1 and RBD antibodies is specific and does not cross-react with other antibodies such as RBD, S1 and nucleocapsid antibodies or with proteins such as interleukin-6. In addition, this platform can be used to detect biomarkers of other viruses such as HIV and Zika (Aliet al., 1002). Another study has successfully created an rGO-based watermarking sensor for proximity detection using the electrostatic gate principle. The sensor showed an exceptional response to electrostatic potential with high sensitivity, which is fundamental for determining the proximity of objects. Object detection was possible up to 20 cm. The sensor is ultra-thin, highly skin-compliant and has a transfer process that allows it to be tattooed onto rough substrates, unlike other graphene-based proximity sensors. The sensor acts as a warning system against unwarranted contact with objects in public places and could be useful in preventing the spread of the virus (Kedambaimooleet al., 2021).
Graphene-based sensors have been used in the medical field to detect human diseases, but they have also been used in food, agriculture and aquaculture to combat certain viruses. Natarajan et al. have developed a sensitive and selective diagnostic method for the white spot syndrome virus, which is deadly in aquaculture. This method manufactures a simple electrochemical immunosensor consisting of a vitreous carbon electrode modified with immobilized GO and stained with methylene blue. This biosensor allowed a very sensitive detection within 35±5min (« An Elegant Analysis of, 2017).
1.6. GO textile for protection against virus
As already mentioned, GO-based nanomaterials are currently used in many fields, including the medical domain. With their extraordinary physicochemical and biological properties such as antimicrobial, antiviral and conductivity properties, GO nanosheets can be used to manufacture protective equipment against infectious diseases. As a result, these properties may offer new advantages for controlling SARS-CoV-2 and reducing its spread. Researchers have successfully manufactured a multifunctional cotton fabric with high electrical conductivity and ultra-strong UV protective properties by applying a dispersion of nanosheets of GO to the fabric surface using the vacuum filtration deposition method. Besides, the resulting fabric was assembled with polyaniline by an in situ chemical polymerization process. The results showed that the ultra-strong protection against UV radiation and the electrical conductivity is higher than the control cotton. Moreover, even a 10-fold repeated washing with water showed no effect on the electrical conductivity and the effectiveness of UV protection (Kumar et al., 2020; Tang, 2020).
A recent study by De Maio et al. (De Maioet al., 2021) showed that pre-incubation of the virus with soluble GO inhibits the infection of VERO cells by SARS-CoV-2. Furthermore, when graphene/GO-functionalized polyurethane or cotton was in contact with SARS-CoV-2, the infectivity of the tissue was almost completely inhibited. The results of this study provide a new nanomaterial-based strategy to increase the effectiveness of protective equipment against SARS-CoV-2 significantly.
Another study used ion implantation to modify the surface property and structure of the GO. Nanosheets of GO were introduced into a cotton fabric by microwave radiation-induced cross-linking and bombarded with different Fe3+ ions. The results of this study proved that GO could be successfully combined with cotton fabrics under microwave irradiation. Also, the scanning electron microscopy analysis results revealed that the nanosheets of GO presented protrusions and nanopores after being treated by ion implantation. Besides, Raman and atomic force microscopy analyses demonstrated the appearance of a new structure such as vortices. As a result, ion-implanted GO cotton has shown a higher antibacterial activity compared to control. Moreover, the prepared nanomaterials have excellent mechanical, antibacterial properties and wash durability even after being washed 60 times (Hu et al., 2019).
Pan et al. applied the GO-modified N-halamine polymer precursor to cotton fabrics by a conventional dip-drying method. Subsequently, the functionalized cotton fabrics were reduced in situ by treatment with L-ascorbic acid. The coated cotton fabrics were then treated with household bleach for increased antibacterial activity. In addition, the coated fabrics were also given a high hydrophobicity, indicating a high self-cleaning ability (Pan et al., 2018).
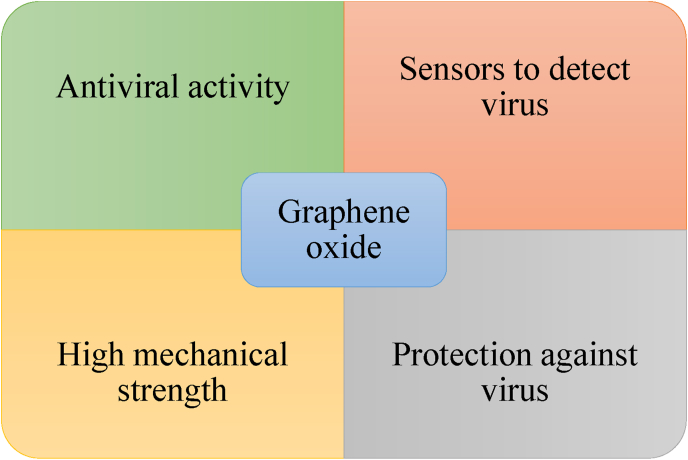
Fig. 4 show the potential multifunction of GO that may be used against the virus at various level. Thus, it can be said that GO can be used as a biosensor for the detection of viruses and also to generate protective equipment against infectious diseases due to its mechanical properties. Moreover, it can be used as an antiviral agent due to its important properties (Fig. 4).
Fig. 4.
Representative uses of GO to combat viruses.
2. Conclusion and perspectives
GO-based nanomaterials are interesting candidates to be used in various fields, especially for medical and electronic applications, due to their promising physicochemical properties, including the high surface/volume ratio, surface charge, size and shape, as well as the optical, electronic and biological properties. Nanotechnology-based approaches are biocompatible and feasible to deal with various types of viral infections, including COVID-19. Also, GO has a higher negative charge which allows its interaction with positively charged viruses. This characteristic explains the fact that it has an important antiviral activity against certain viruses. In parallel, GO and graphene have the ability to inactivate various viruses by different mechanisms, including virus inactivation, viral binding inhibition, photodegradation and electrostatic trapping. Besides, it has been shown that GO and graphene are very useful for high throughput diagnostics, using a transistor-based biosensor that detects SARS-CoV-2. They can also be used to make protective equipment against infectious diseases.
In response to the global COVID-19 pandemic, we summarized the antiviral activity of GO in general. Furthermore, we presented the role of GO-based biosensors for virus diagnosis and the application of this nanomaterial for infection protection. In addition, there is a need for further research to develop new GO-based antivirals to fight SARS-CoV-2. Finally, several issues remain to be resolved, such as cytotoxicity, which must be fully assessed to ensure a biocompatible therapeutic product without undesirable side effects. Studies to date indicate that the toxicity of GO may depend on its size, methods of synthesis, route of administration and exposure time. Therefore, there is a need to better understand the toxicological mechanisms of GO in order to benefit from the advantages of nanotechnologies in the medical field and also to minimize the health risks.
CRediT authorship contribution statement
Asmaa Rhazouani: Writing – original draft. Khalid Aziz: Figures Conception and Editing. Halima Gamrani: Reviewing and Editing. Lhoucine Gebrati: Reviewing and Editing. Md Sahab Uddin: Reviewing and Editing. Aziz Faissal: Reviewing and Editing, Supervision.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have influenced the work reported in this paper.
References
- Alayande A.B., Chae S., et al. Kim I.S. « Surface morphology-dependent spontaneous bacterial behaviors on graphene oxide membranes ». Separ. Purif. Technol. 2019;226:68–74. [Google Scholar]
- Adams et M.J., Carstens E.B. « ratification vote on taxonomic proposals to the international committee on taxonomy of viruses (2012) ». Arch. Virol. 2012;157(7):1411–1422. doi: 10.1007/s00705-012-1299-6. juill. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Afroj S., Tan S., Abdelkader A., Novoselov K., et al. Karim N. « highly conductive, scalable, and machine washable graphene-based E-textiles for multifunctional wearable electronic applications ». Adv. Funct. Mater. 2020;30:2000293. doi: 10.1002/adfm.202000293. avr. [DOI] [Google Scholar]
- Afsahi S., et al. « Novel graphene-based biosensor for early detection of Zika virus infection ». Biosens. Bioelectron. 2018;100:85–88. doi: 10.1016/j.bios.2017.08.051. févr. [DOI] [PubMed] [Google Scholar]
- Akhavan O., Choobtashani M., Ghaderi et E. « protein degradation and RNA efflux of viruses photocatalyzed by graphene–tungsten oxide composite under visible light irradiation ». J. Phys. Chem. C. 2012;116(17):9653–9659. doi: 10.1021/jp301707m. mai. [DOI] [Google Scholar]
- M. A. Ali et al., « Sensing of COVID-19 Antibodies in Seconds via Aerosol Jet Nanoprinted Reduced-Graphene-Oxide-Coated 3D Electrodes », Advanced Materials (Deerfield Beach, Fla.), doi: 10.1002/adma.202006647. [DOI] [PMC free article] [PubMed]
- Aliyev E., Filiz V., Khan M.M., Lee Y.J., Abetz C., et al. Abetz V. « structural characterization of graphene oxide: surface functional groups and fractionated oxidative debris ». Nanomaterials (Basel) 2019;9(8):E1180. doi: 10.3390/nano9081180. août. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bisht H., Roberts A., Vogel L., Subbarao K., et al. Moss B. « Neutralizing antibody and protective immunity to SARS coronavirus infection of mice induced by a soluble recombinant polypeptide containing an N-terminal segment of the spike glycoprotein ». Virology. avr. 2005;334(2):160–165. doi: 10.1016/j.virol.2005.01.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Broughton J.P., et al. « CRISPR-Cas12-based detection of SARS-CoV-2 ». Nat. Biotechnol. 2020;38(7):870–874. doi: 10.1038/s41587-020-0513-4. juill. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Y.-N., Hsueh Y.-H., Hsieh C.-T., Tzou D.-Y., et al. Chang P.-L. « antiviral activity of graphene-silver nanocomposites against non-enveloped and enveloped viruses ». Int. J. Environ. Res. Publ. Health. 2016;13(4):430. doi: 10.3390/ijerph13040430. avr. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cui J., Li F., et al. Shi Z.-L. « Origin and evolution of pathogenic coronaviruses ». Nat. Rev. Microbiol. 2019;17(3):181–192. doi: 10.1038/s41579-018-0118-9. mars. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Du X., et al. « Hypericin-loaded graphene oxide protects ducks against a novel duck reovirus. », Mater Sci Eng C Mater Biol Appl. 2019;105 doi: 10.1016/j.msec.2019.110052. 110052-110052, août. [DOI] [PubMed] [Google Scholar]
- Dasari Shareena T.P., McShan D., Dasmahapatra A.K., et al. Tchounwou P.B. « A review on graphene-based nanomaterials in biomedical applications and risks in environment and health ». Nano-Micro Lett. 2018;10(3):53. doi: 10.1007/s40820-018-0206-4. mai. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Das S., et al. « Oxygenated functional group density on graphene oxide: its effect on cell toxicity ». Part. Part. Syst. Char. 2013;30(2):148–157. doi: 10.1002/ppsc.201200066. févr. [DOI] [Google Scholar]
- De Maio F., et al. « Graphene nanoplatelet and graphene oxide functionalization of face mask materials inhibits infectivity of trapped SARS-CoV-2 ». iScience. 2021;24(7) doi: 10.1016/j.isci.2021.102788. juill. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Wilde A.H., Snijder E.J., Kikkert M., et al. van Hemert M.J. « host factors in coronavirus replication ». Roles of Host Gene and Non-coding RNA Expression in Virus Infection. 2017;419:1–42. doi: 10.1007/82_2017_25. juin. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Wit E., van Doremalen N., Falzarano D., et al. Munster V.J. « SARS and MERS: recent insights into emerging coronaviruses ». Nat. Rev. Microbiol. 2016;14(8):523–534. doi: 10.1038/nrmicro.2016.81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Du T., et al. « antiviral activity of graphene oxide–silver nanocomposites by preventing viral entry and activation of the antiviral innate immune response ». ACS Appl. Bio Mater. 2018;1(5):1286–1293. doi: 10.1021/acsabm.8b00154. nov. [DOI] [PubMed] [Google Scholar]
- Ege D., Kamali A.R., et al. Boccaccini A.R. « graphene oxide/polymer-based biomaterials ». Adv. Eng. Mater. 2017;19(12):1700627. doi: 10.1002/adem.201700627. [DOI] [Google Scholar]
- Elechiguerra J.L., et al. « Interaction of silver nanoparticles with HIV-1 ». J. Nanobiotechnol. 2005;3(1):6. doi: 10.1186/1477-3155-3-6. juin. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gholami M.F., et al. « functionalized graphene as extracellular matrix mimics: toward well-defined 2D nanomaterials for multivalent virus interactions ». Adv. Funct. Mater. 2017;27(15):1606477. doi: 10.1002/adfm.201606477. [DOI] [Google Scholar]
- Hanff T.C., Harhay M.O., Brown T.S., Cohen J.B., et al. Mohareb A.M. « is there an association between COVID-19 mortality and the renin-angiotensin system? A call for epidemiologic investigations ». Clin. Infect. Dis. 2020;71(15):870–874. doi: 10.1093/cid/ciaa329. juill. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoffmann M.R., Martin S.T., Choi W., et al. Bahnemann D.W. « environmental applications of semiconductor photocatalysis ». Chem. Rev. 1995;95(1):69–96. doi: 10.1021/cr00033a004. janv. [DOI] [Google Scholar]
- Hoffmann M., et al. « SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor ». Cell. 2020;181(2):271–280. doi: 10.1016/j.cell.2020.02.052. e8, avr. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu X., Mu L., Wen J., et al. Zhou Q. « Covalently synthesized graphene oxide-aptamer nanosheets for efficient visible-light photocatalysis of nucleic acids and proteins of viruses ». Carbon. 2012;8(50):2772–2781. doi: 10.1016/j.carbon.2012.02.038. [DOI] [Google Scholar]
- Hu J., Liu J., Gan L., et al. Long M. « surface-modified graphene oxide-based cotton fabric by ion implantation for enhancing antibacterial activity ». ACS Sustain. Chem. Eng. 2019;7(8):7686–7692. doi: 10.1021/acssuschemeng.8b06361. avr. [DOI] [Google Scholar]
- Hummers et W.S., Offeman R.E. « preparation of graphitic oxide ». J. Am. Chem. Soc. 1958 doi: 10.1021/ja01539a017. mars. [DOI] [Google Scholar]
- Jeremiah S.S., Miyakawa K., Morita T., Yamaoka Y., et al. Ryo A. « Potent antiviral effect of silver nanoparticles on SARS-CoV-2 ». Biochem. Biophys. Res. Commun. 2020;533(1):195–200. doi: 10.1016/j.bbrc.2020.09.018. nov. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joshi S.R., Sharma A., Kim G.-H., et al. Jang J. « Low cost synthesis of reduced graphene oxide using biopolymer for influenza virus sensor ». Mater Sci Eng C Mater Biol Appl. 2020;108:110465. doi: 10.1016/j.msec.2019.110465. mars. [DOI] [PubMed] [Google Scholar]
- Kam Y.W., et al. « Antibodies against trimeric S glycoprotein protect hamsters against SARS-CoV challenge despite their capacity to mediate FcγRII-dependent entry into B cells in vitro ». Vaccine. 2007;25(4):729–740. doi: 10.1016/j.vaccine.2006.08.011. janv. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kedambaimoole V., et al. « reduced graphene oxide tattoo as wearable proximity sensor ». Advanced Electronic Materials. 2021;7(4):2001214. doi: 10.1002/aelm.202001214. [DOI] [Google Scholar]
- Krishnamoorthy K., Mohan R., et al. Kim S.-J. 2011. « Graphene Oxide as a Photocatalytic Material ». juin. [DOI] [Google Scholar]
- Krishnan S., Halonen M., et al. Welliver R.C. « Innate immune responses in respiratory syncytial virus infections ». Viral Immunol. 2004;17(2):220–233. doi: 10.1089/0882824041310612. [DOI] [PubMed] [Google Scholar]
- Kumar A., Sharma K., et al. Dixit A.R. « Role of graphene in biosensor and protective textile against viruses ». Med. Hypotheses. 2020;144:110253. doi: 10.1016/j.mehy.2020.110253. nov. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar Raghav et P., Mohanty S. « Are graphene and graphene-derived products capable of preventing COVID-19 infection? ». Med. Hypotheses. 2020;144:110031. doi: 10.1016/j.mehy.2020.110031. nov. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laghrib F., et al. « Current progress on COVID-19 related to biosensing technologies: new opportunity for detection and monitoring of viruses ». Microchem. J. 2020 doi: 10.1016/j.microc.2020.105606. oct. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lambert T.N., et al. « synthesis and characterization of Titania−Graphene nanocomposites ». J. Phys. Chem. C. 2009;113(46):19812–19823. doi: 10.1021/jp905456f. nov. [DOI] [Google Scholar]
- Lau S.K.P., et al. « discovery and sequence analysis of four deltacoronaviruses from birds in the Middle East reveal interspecies jumping with recombination as a potential mechanism for avian-to-avian and avian-to-mammalian transmission ». J. Virol. 2018;92(15) doi: 10.1128/JVI.00265-18. e00265-18, août. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li F. « structure, function, and evolution of coronavirus spike proteins ». Annual Review of Virology. 2016;3(1):237–261. doi: 10.1146/annurev-virology-110615-042301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mf A.M., et al. « Gold nanoparticle-decorated reduced-graphene oxide targeting anti hepatitis B virus core antigen. Bioelectrochemistry. 2018;122:199–205. doi: 10.1016/j.bioelechem.2018.04.004. avr. [DOI] [PubMed] [Google Scholar]
- Möritz M., Peters H., Nipko B., et al. Rüden H. « Capability of air filters to retain airborne bacteria and molds in heating, ventilating and air-conditioning (HVAC) systems ». Int. J. Hyg Environ. Health. 2001;203(5):401–409. doi: 10.1078/1438-4639-00054. janv. [DOI] [PubMed] [Google Scholar]
- Mousavizadeh L., et al. Ghasemi S. « Genotype and phenotype of COVID-19: their roles in pathogenesis ». J. Microbiol. Immunol. Infect. 2020 doi: 10.1016/j.jmii.2020.03.022. mars. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Navakul K., Warakulwit C., Yenchitsomanus P.-T., Panya A., Lieberzeit P.A., et al. Sangma C. « A novel method for dengue virus detection and antibody screening using a graphene-polymer based electrochemical biosensor ». Nanomedicine. 2017;13(2):549–557. doi: 10.1016/j.nano.2016.08.009. [DOI] [PubMed] [Google Scholar]
- Nguyen E.P., Silva C. de C.C., et al. Merkoçi A. « Recent advancement in biomedical applications on the surface of two-dimensional materials: from biosensing to tissue engineering ». Nanoscale. oct. 2020;12(37):19043–19067. doi: 10.1039/D0NR05287F. [DOI] [PubMed] [Google Scholar]
- Nguyen N.H.L., Kim S., Lindemann G., et al. Berry V. « COVID-19 spike protein induced phononic modification in antibody-coupled graphene for viral detection application ». ACS Nano. 2021 doi: 10.1021/acsnano.1c02549. juin. [DOI] [PubMed] [Google Scholar]
- Noorunnisa Khanam P., et al. Hasan A. « Biosynthesis and characterization of graphene by using non-toxic reducing agent from Allium Cepa extract: antibacterial properties ». Int. J. Biol. Macromol. 2019;126:151–158. doi: 10.1016/j.ijbiomac.2018.12.213. avr. [DOI] [PubMed] [Google Scholar]
- Omar N.A.S., Fen Y.W., Abdullah J., Zaid M.H.M., Daniyal W.M.E.M.M., et al. Mahdi M.A. « Sensitive surface plasmon resonance performance of cadmium sulfide quantum dots-amine functionalized graphene oxide based thin film towards dengue virus E-protein ». Opt Laser. Technol. 2019;114:204–208. doi: 10.1016/j.optlastec.2019.01.038. juin. [DOI] [Google Scholar]
- Palmieri V., et al. « bacteria meet graphene: modulation of graphene oxide nanosheet interaction with human pathogens for effective antimicrobial therapy ». ACS Biomater. Sci. Eng. 2017;3(4):619–627. doi: 10.1021/acsbiomaterials.6b00812. avr. [DOI] [PubMed] [Google Scholar]
- Palmieri V., et al. Papi M. « Can graphene take part in the fight against COVID-19? ». Nano Today. 2020;33:100883. doi: 10.1016/j.nantod.2020.100883. août. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pan N., Liu Y., Ren X., et al. Huang T.-S. 2018. « Fabrication of Cotton Fabrics through In-Situ Reduction of Polymeric N-Halamine Modified Graphene Oxide with Enhanced Ultraviolet-Blocking, Self-Cleaning, and Highly Efficient, and Monitorable Antibacterial Properties », Colloids And Surfaces.https://agris.fao.org/agris-search/search.do?recordID=US201800443362 Consulté le: oct. 26, 2020. [En ligne]. Disponible sur: [Google Scholar]
- Paredes J.I., Villar-Rodil S., Martínez-Alonso A., et al. Tascón J.M.D. « Graphene oxide dispersions in organic solvents ». Langmuir. oct. 2008;24(19):10560–10564. doi: 10.1021/la801744a. [DOI] [PubMed] [Google Scholar]
- Rhazouani A., et al. « synthesis and toxicity of graphene oxide nanoparticles: a literature review of in vitro and in vivo studies ». BioMed Res. Int. 2021;2021:5518999. doi: 10.1155/2021/5518999. juin. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riccò M., et al. « point-of-care diagnostic tests for detecting SARS-CoV-2 antibodies: a systematic review and meta-analysis of real-world data ». J. Clin. Med. 2020;9(5) doi: 10.3390/jcm9051515. Art. no 5. mai. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sametband M., Kalt I., Gedanken A., et al. Sarid R. « herpes simplex virus type-1 attachment inhibition by functionalized graphene oxide ». ACS Appl. Mater. Interfaces. 2014;6(2):1228–1235. doi: 10.1021/am405040z. janv. [DOI] [PubMed] [Google Scholar]
- Seah I., Su X., et al. Lingam G. « Revisiting the dangers of the coronavirus in the ophthalmology practice ». Eye. 2020;34(7) doi: 10.1038/s41433-020-0790-7. Art. no 7, juill. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seifi T., et al. Kamali A.R. « Anti-pathogenic activity of graphene nanomaterials: a review ». Colloids Surf. B Biointerfaces. 2021;199:111509. doi: 10.1016/j.colsurfb.2020.111509. mars. [DOI] [PubMed] [Google Scholar]
- Seifi T., et al. Reza Kamali A. « Antiviral performance of graphene-based materials with emphasis on COVID-19: a review ». Medicine in Drug Discovery. 2021;11:100099. doi: 10.1016/j.medidd.2021.100099. sept. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seo G., et al. « rapid detection of COVID-19 causative virus (SARS-CoV-2) in human nasopharyngeal swab specimens using field-effect transistor-based biosensor ». ACS Nano. 2020;14(4):5135–5142. doi: 10.1021/acsnano.0c02823. avr. [DOI] [PubMed] [Google Scholar]
- Song Z., et al. « Virus capture and destruction by label-free graphene oxide for detection and disinfection applications ». Small. 2015;11(9–10):1171–1176. doi: 10.1002/smll.201401706. mars. [DOI] [PubMed] [Google Scholar]
- Sun L., et al. « antibacterial activity of graphene oxide/g-C3N4 composite through photocatalytic disinfection under visible light ». ACS Sustain. Chem. Eng. 2017;5(10):8693–8701. doi: 10.1021/acssuschemeng.7b01431. oct. [DOI] [Google Scholar]
- Tang X. « Functionalization of cotton fabric with graphene oxide nanosheet and polyaniline for conductive and UV blocking properties ». 2020. https://www.academia.edu/10856034/Functionalization_of_cotton_fabric_with_graphene_oxide_nanosheet_and_polyaniline_for_conductive_and_UV_blocking_properties Consulté le: oct. 26 [En ligne]. Disponible sur:
- Walls A.C., Park Y.-J., Tortorici M.A., Wall A., McGuire A.T., et al. Veesler D. « structure, function, and antigenicity of the SARS-CoV-2 spike glycoprotein ». Cell. 2020;181(2):281–292. doi: 10.1016/j.cell.2020.02.058. e6, avr. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang K., et al. « morphology- and crystalline composition-governed activity of titania-based photocatalysts: overview and perspective ». Catalysts. 2019;9(12) doi: 10.3390/catal9121054. Art. no 12, déc. [DOI] [Google Scholar]
- Weingartl H., et al. « Immunization with modified vaccinia virus Ankara-based recombinant vaccine against severe acute respiratory syndrome is associated with enhanced hepatitis in ferrets ». J. Virol. nov. 2004;78(22):12672–12676. doi: 10.1128/JVI.78.22.12672-12676.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woo P.C.Y., et al. « Discovery of seven novel Mammalian and avian coronaviruses in the genus deltacoronavirus supports bat coronaviruses as the gene source of alphacoronavirus and betacoronavirus and avian coronaviruses as the gene source of gammacoronavirus and deltacoronavirus ». J. Virol. 2012;86(7):3995–4008. doi: 10.1128/JVI.06540-11. avr. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang X.X., Li C.M., Li Y.F., Wang J., et al. Huang C.Z. « Synergistic antiviral effect of curcumin functionalized graphene oxide against respiratory syncytial virus infection ». Nanoscale. 2017;9(41):16086–16092. doi: 10.1039/C7NR06520E. oct. [DOI] [PubMed] [Google Scholar]
- Ye S., et al. « antiviral activity of graphene oxide: how sharp edged structure and charge matter ». ACS Appl. Mater. Interfaces. sept. 2015;7(38):21571–21579. doi: 10.1021/acsami.5b06876. [DOI] [PubMed] [Google Scholar]
- Ziem B., et al. « highly efficient multivalent 2D nanosystems for inhibition of orthopoxvirus particles. », Adv Healthc Mater. 2016;5(22):2922–2930. doi: 10.1002/adhm.201600812. sept. [DOI] [PubMed] [Google Scholar]
- Zaleska A. « doped-TiO2: a review ». Recent Pat. Eng. 2008;2(3):157–164. doi: 10.2174/187221208786306289. nov. [DOI] [Google Scholar]
- Zhou P., et al. « A pneumonia outbreak associated with a new coronavirus of probable bat origin ». Nature. 2020;579(7798):270–273. doi: 10.1038/s41586-020-2012-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ziem B., Azab W., Gholami M.F., Rabe J.P., Osterrieder N., et al. Haag R. « Size-dependent inhibition of herpesvirus cellular entry by polyvalent nanoarchitectures ». Nanoscale. 2017;9(11):3774–3783. doi: 10.1039/c7nr00611j. mars. [DOI] [PubMed] [Google Scholar]
- « Allocution liminaire du Directeur général de l’OMS lors du point presse sur la COVID-19 - 11 mars 2020 ». éhttps://www.who.int/fr/dg/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---11-march-2020 (consulté le oct. 27, 2020).
- « an elegant analysis of white spot syndrome virus using a graphene oxide/methylene blue based electrochemical immunosensor platform ». Sci. Rep. 2017;7(1) doi: 10.1038/srep46169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- « Graphene Ink May Be Used to Fight Coronavirus ». MINING.COM; 2020. https://www.mining.com/graphene-ink-may-be-used-to-fight-novel-coronavirus/ mai 01. (consulté le nov. 11, 2020) [Google Scholar]
- « Maladie à coronavirus (COVID-19) ». https://www.unicef.org/morocco/recits/maladie-%C3%A0-coronavirus-covid-19 (consulté le oct. 27, 2020)
- « synergistic antiviral activity of graphene oxide and common antiviral agents | STATNANO ». https://statnano.com/news/67505/Synergistic-Antiviral-Activity-of-Graphene-Oxide-and-Common-Antiviral-Agents (consulté le oct. 10, 2020)