Abstract
Neoadjuvant treatment has become the standard of care for locally advanced rectal cancer for many years. Several neoadjuvant therapeutic options are currently used, the most common being conventionally fractionated radiotherapy (or long-course radiotherapy) administered concomitantly with chemotherapy and hypofractionated radiotherapy (or short-course radiotherapy). This meta-analysis will give a better overview of the results of several studies that compare long-course radio-chemotherapy with short-course radiotherapy, emphasizing on the severe acute and late toxicities and the postoperative results of the analyzed studies.
After identification, analysis and verification of eligibility criteria, eight studies were included in the meta-analysis. The methodological quality of the selected studies was assessed using the classic Oxford quality grading system (Jadad scale).
The results obtained in this meta-analysis shows us that we can safely use both short-course radiotherapy and long-course radio-chemotherapy as neoadjuvant treatment for locally advanced rectal cancer, without significant differences regarding to severe acute or late toxicities, positive resection margins R+ or the number of local pelvic relapses at three years.
Keywords:short-course radiotherapy, long-course radiotherapy, neoadjuvant treatment, rectal cancer, locally advanced rectal cancer, radio-chemotherapy.
INTRODUCTION
Neoadjuvant treatment has become the standard of care for locally advanced rectal cancer for many years (1). Several neoadjuvant therapeutic options are currently used, the most common being conventionally fractionated radiotherapy administered concomitantly with chemotherapy (or long-course radiotherapy, LCRT + CHT) and hypofractionated radiotherapy (or short-course radiotherapy, SCRT). The first option is widely used in the United States of America and in many treatment centers in Europe while the latter is mostly used in Northern Europe (2). Some studies that compare the two treatment regimens show similar outcomes for overall survival (3-7), disease free survival (3, 5-7), local relapse (3-7) or distant metastasis rate (3-5, 7). However, the choice of treatment scheme remains at the discretion of the radiation oncologist and there is no consensus on the optimal therapeutic option corresponding to the particularities of each patient.
Definition, epidemiology
Colorectal cancer is the third most common cancer after breast and lung cancer and the second leading cause of death worldwide after lung cancer. Although its incidence has remained relatively unchanged in the last two years, global mortality caused by colorectal cancer has increased by 0.2% (8).
Epidemiological data from Romania show a higher incidence of colorectal cancer compared to global epidemiological data. Thus, colorectal cancer was the most diagnosed neoplasm in 2020 in our country, about 13% of new cases of cancer (8).
Long-course radiotherapy represents the conventionally fractionated radiotherapy, with doses of 180 to 200 centi-Gray per fraction, administered in 25 to 28 daily fractions (five days per week) up to a total dose of 4 500 to 5 040 centi-Gray. Chemotherapy is administered concomitantly, the most frequent cytostatic agents being capecitabine with a dose of 825 mg/m2 twice daily or 5-fluorouracil with a dose of 1 200 mg/m² daily. Surgery is scheduled 4 to 12 weeks after radiotherapy ends, usually in the sixth or seventh week. The administration of adjuvant chemotherapy remains at the decision of the attending oncologist (9).
Short-course radiotherapy represents the hypofractionated radiotherapy, with doses of 500 centi-Gray per fraction, administered in five daily fractions over the course of a week up to a total dose of 2 500 centi-Gray. Usually, no chemotherapy is administered concomitantly. Surgery is scheduled in the first 10 days after radiotherapy, most frequently in days 5 to 7, or it can be delayed similar to long-course radiotherapy. Some studies have demonstrated that there is no difference in efficacy with immediate versus delayed surgery after short-course radiotherapy (10-12).
Purpose
Can this meta-analysis give a better overview of the results of several studies that compare longcourse radio-chemotherapy with short-course radiotherapy, by emphasizing the severe acute and late toxicities and the postoperative results of the analyzed studies?
MATERIALS AND METHODS
Inclusion and exclusion criteria
The meta-analysis includes prospective, double- blinded, randomized and retrospective studies that compare the long-course and short-course radiotherapy regimens in patients with locally advanced rectal cancer and that contain information on at least one of the following: severe acute toxicity, severe late toxicity, positive postoperative circumferential margins, local pelvic relapse rate at three years without taking into account distant metastases.
Case presentations, case series or review studies were not included in the meta-analysis. Studies that included patients diagnosed with distant metastases at the time of enrollment were also excluded. In order to be recorded for analysis, we selected studies that had a total dose of at least 20 Gray on the short-course regimen and a minimum of 45 Gray on the long-course regimen.
Data selection
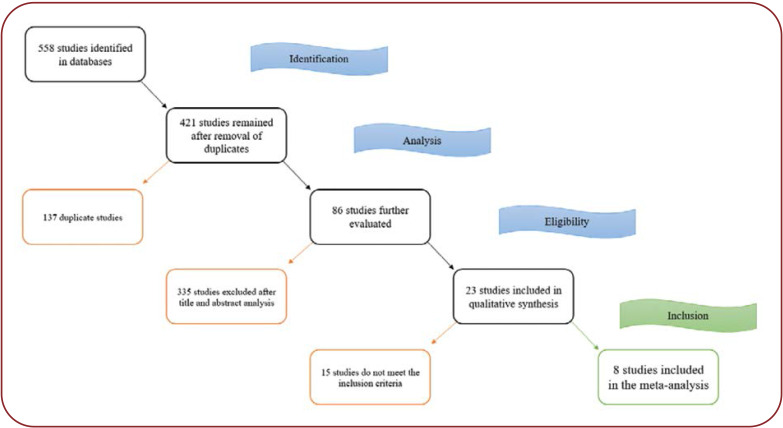
Relevant studies have been identified in the PubMed, Cochrane and Embase databases since 2000, searching for keywords (radiotherapy, neoadjuvant treatment, rectal cancer, shortcourse radiotherapy, long-course radiotherapy) in the search engines of these databases. Of the 558 identified studies, 137 were duplicates, 335 studies did not present relevant information for this meta-analysis and another 15 studies did not meet the inclusion criteria. Finally, eight studies were included in the meta-analysis.
Data analysis and quality assurance
The methodological quality of the selected studies was assessed using the classic Oxford quality grading system (Jadad scale), with a quality score of the studies ranging from 0 to 5, depending on the presence or absence of randomization, double- blinding and completeness of data (13).
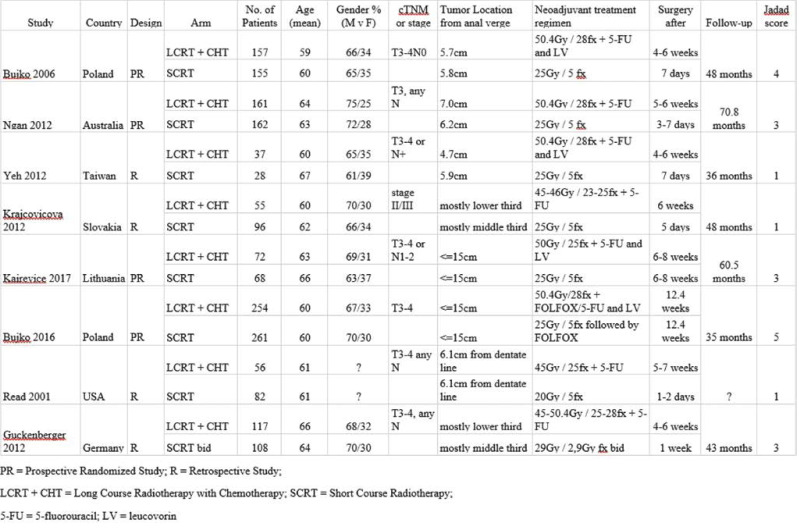
The following data was extracted from the included studies: general data about the population (country of the study, year of the study, study design, number of patients included on each arm, age, gender), TNM stage, tumor location, treatment regimens, interval to surgery, follow- up period and the variables to be analyzed: quantitative variables (scales of acute and late toxicities, grade of positive oncological margins) or nominal dichotomous variables (local relapse rate at three years).
The meta-analysis was performed using Review Manager Software 5.4 (RevMan 5.4, Cochrane Collaboration) and included dichotomous data. The results were obtained by the fixed effects analysis model and included the p score for statistical significance (p <0.05 being considered statistically significant). For all the data, risk ratio and the 95% confidence interval were calculated using the Mantel-Haenszel statistical method. Heterogeneity between studies was quantified using the Chi² and I² inconsistency test. The graphical representation of the data was performed for each comparison using the “forest plot” model.
RESULTS
The following variables were analyzed in this meta-analysis: frequency of severe acute toxicity during or immediately after neoadjuvant treatment (grade 3-4 on the Common Terminology Criteria for Adverse Events CTCAE or Radiation Therapy Oncology Group RTOG scale); frequency of severe late toxicity after neoadjuvant treatment (grade 3-4 on the Common Terminology Criteria for Adverse Events CTCAE or Radiation Therapy Oncology Group RTOG scale); presence of positive postoperative circumferential margins (R>0); and local relapse rate at three years, without taking into account distant metastases.
Severe toxicities observed
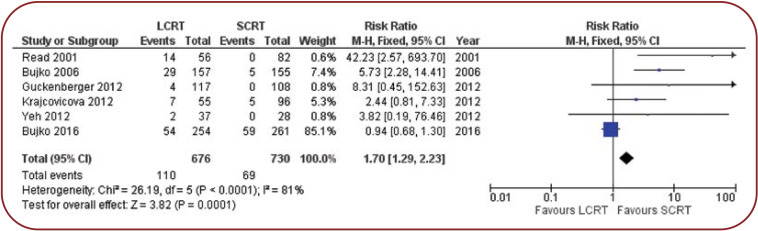
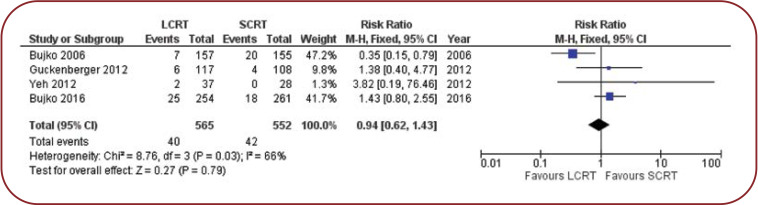
Consistent with the findings of other studies (3, 5, 14, 15), this meta-analysis shows that short-course hypofractionated radiotherapy has a favorable short-term safety profile when comparing it to conventional long-course fractional radiotherapy. In the case of LCRT + CHT we observe a higher number of events (of severe acute toxicities), even though the total number of patients in the study who received the LCRT+CHT scheme is lower. The most common acute toxicities observed in the studies were gastrointestinal toxicities.
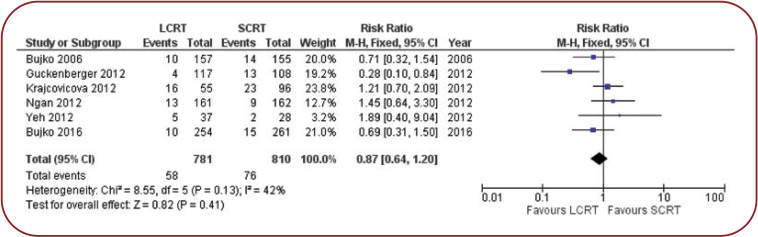
The results observed in the analysis of severe late reactions slightly favors the long-course treatment scheme. The number of events (severe late toxicities) is lower for LCRT+CHT than for SCRT and the heterogeneity of the studies is satisfactory with an I2 inconsistency test <50%.
Postoperative results and local relapse rate
By analyzing the rate of positive resection margins (microscopic R1 or macroscopic R2) in the included studies, we observe that both treatment schemes obtain similar results. Although there are several comparative studies between LCRT+CHT vs. SCRT, only Bujko et al in 2006 (3) and 2016 (7) and Guckenberger et al in 2012 (15) provide comprehensive information on resection margins.
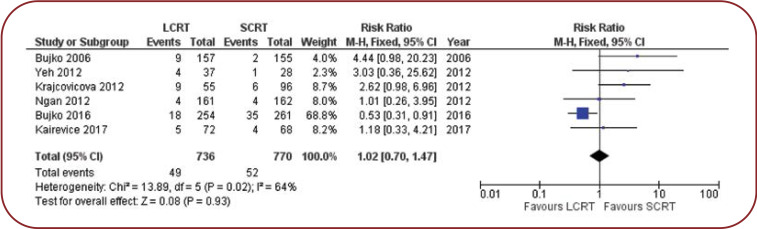
We analyzed studies that reported local relapse events, noting that we strictly selected local pelvic recurrences and did not include distant metastases. The results in this case are approximately equal for both regimens, which confirms the safety of both treatments in terms of local control.
DISCUSSION
Many authors have obtained a favorable safety profile for SCRT when talking about severe acute toxicities, with results being statistically significant (p <0.05). In 2016, Bujko et al (7) suggests the opposite, but by a very small margin (n = 59 for SCRT vs. n = 54 for LCRT+CHT). These findings may support the idea that for vulnerable people such as elderly or poor performance status (PS) patients with significant comorbidities, shorter care and milder acute side effects will increase the likelihood of administering the entire treatment without interruption. If the long-term treatment option is chosen for this category of patients, there is a higher risk of discontinuing treatment or even cessation, which has detrimental consequences on the final oncological outcome.
A confusing factor to consider is that most of the studies used the CTCAE scale as a reference for adverse reactions, while Read et al (14) used the RTOG toxicity scale. Also, Krajcovicova et al did not give details about the severity scale used by them (8).
In terms of severe late reactions, this metaanalysis justifies the choice of long-course radiotherapy in younger patients with fewer comorbidities and a longer life expectancy, despite the risk of developing more serious side effects during or immediately after treatment. Studies conducted by Bujko et al in 2006 (3) and 2016 (7) showed a rate between 5-10% of severe late reactions, but below 1% mortality caused by these complications. Both LCRT+CHT and SCRT have an adequate safety profile.
Postoperative oncological margins are a wellknown risk factor for all oncological conditions treated with surgery. Surgery at 6-8 weeks after radiotherapy is more difficult to perform due to the development of fibrosis within the pelvic tissues, but by allowing radio-chemotherapy to act for a longer time, a reduction in tumor size is obtained in many patients – downstaging (5, 6, 14) or even complete disappearance of the tumor – complete pathological response (3, 7). In the case of immediate surgery, the pelvic tissues do not suffer significantly from radiotherapy and the anatomical planes are easier to be identified by the surgeon. When removing the tumor immediately after radiotherapy, both the chances of having a significant reduction in tumor size and the chances of completely sterilizing the tumor and obtaining a complete pathological response are lower (3, 7, 14). Another essential factor in order to obtain negative resection margins is the quality of radiological investigations and their careful and complete interpretation, thus guiding both the radiation oncologist and the surgeon towards the optimal treatment method by correctly assessing the TNM stage (16).
Similarly to the resection margins, another variable of oncological importance is the local relapse rate observed in the first three years (17). Although the studies included in the analysis of this variable have a moderate heterogeneity (I2=64%), the number of events (local relapses) is almost equal n=49 vs. n=52. Surprisingly, the long-course regimen, even though using concomitant chemotherapy, does not achieve superior results of local control compared to radiotherapy alone in the neoadjuvant setting.
In the case of local relapse at three years, which was observed in the study conducted by Krajcovicova et al in 2012 (8), it should be taken into account that out of the 55 patients who underwent conventional long-course radiotherapy, only 33 subjects received concomitant chemotherapy, so the results have an additional confounding factor.
A new treatment option under investigation, total neoadjuvant treatment (TNT), which involves the administration of short course radiotherapy or long course radio-chemotherapy followed by neoadjuvant chemotherapy and surgery, might have better results regarding the rate of distant metastasis, probably through early destruction of micrometastases unidentifiable by current investigations (18). Exposure to ionizing radiation increases the risk of oncogenesis (19), which is why some researchers are further investigating the possibility of completely excluding radiotherapy from neoadjuvant treatment (20, 21).
CONCLUSION
The results obtained in this meta-analysis show that we can use both the short-course radiotherapy and the long-course radiotherapy schemes in the neoadjuvant treatment of locally advanced rectal cancer without having significant differences in terms of severe acute or late toxicities.
From the postoperative result point of view, we can expect similar rates of positive margins R+ after both treatment options, which can vary from 0% (5) to 12.9% (3). These results are satisfactory for the available treatment options and are superior to treatment regimens used before 2000, such as surgery alone or with adjuvant radiotherapy (14).
The local pelvic relapse rate at three years, excluding distant metastases, is almost identical in the comparison made in this meta-analysis, despite the fact that one of the schemes involves radiotherapy alone and the other one associated chemotherapy.
To guide the radiation oncologist in choosing the optimal treatment regimen, a treatment prediction model based on clinical and/or paraclinical data of each patient should be proposed and validated. This model should balance the risks of severe acute side effects, which can lead to treatment discontinuation, major complications or even death, with the risk of severe late side effects (22) and the desired oncological outcome.
Conflict of interests: none declared
Financial support: none declared.
Acknowledgements: Special thanks to Assoc. Prof. VERGA I. Nicolae, MD, PhD, PhD Coordinator, Head of Department of Radiotherapy, “Coltea” Clinical Hospital, Bucharest, Romania.
FIGURE 1.
Study selection process scheme
TABLE 1.
Laboratory values on admission
FIGURE 2.
Meta-analysis of severe acute toxicities in LCRT+CHT vs. SCRT
FIGURE 3.
Meta-analysis of severe late toxicities in LCRT+CHT vs. SCRT
FIGURE 4.
Meta-analysis of positive circumferential margins in LCRT+CHT vs. SCRT
FIGURE 5.
Meta-analysis of the local relapse rate at three years in LCRT+CHT vs. SCRT
Contributor Information
Horia-Dan LISCU, “Carol Davila” University of Medicine and Pharmacy, Discipline of Radiation Oncology and Medical Imaging, Bucharest, Romania; Department of Radiotherapy, “Coltea” Clinical Hospital, Bucharest, Romania.
Andreea-Iuliana MIRON, “Carol Davila” University of Medicine and Pharmacy, Discipline of Radiation Oncology and Medical Imaging, Bucharest, Romania; Department of Oncology, “Coltea” Clinical Hospital, Bucharest, Romania.
Andra-Raluca RUSEA, Clinical Laboratory of Medical Analysis, Emergency University Hospital of Bucharest, Bucharest, Romania.
Ana-Maria Nicoleta OPREA, Department of Radiotherapy, “Coltea” Clinical Hospital, Bucharest, Romania.
Ruxandra MITRE, Department of Oncology, “Coltea” Clinical Hospital, Bucharest, Romania.
Alexandru HERDEA, Department of Pediatric Orthopaedics, “Grigore Alexandrescu” Children’s Emergency Hospital, Bucharest, Romania.
Razvan NEGREANU, Department of Oncology, “Coltea” Clinical Hospital, Bucharest, Romania.
References
- 1.Halperin EC, et al. Perez & Brady's principles and practice of radiation oncology. Radiation therapy for gastrointestinal cancers. 2019.
- 2.Hong T, Das P, eds. Radiation therapy for gastrointestinal cancers. Springer International Publishing. 2017.
- 3.Bujko K, Nowacki MP, Nasierowska-Guttmejer A, et al. Long-term results of a randomized trial comparing preoperative short-course radiotherapy with preoperative conventionally fractionated chemoradiation for rectal cancer. British Journal of Surgery. 2006;93:1215–1223. doi: 10.1002/bjs.5506. [DOI] [PubMed] [Google Scholar]
- 4.Ngan SY, Burmeister B, Fisher RJ, et al. Randomized trial of short-course radiotherapy versus long-course chemoradiation comparing rates of local recurrence in patients with T3 rectal cancer: Trans-Tasman Radiation Oncology Group trial 01.04. J Clin Oncol. 2012;30:3827–3833. doi: 10.1200/JCO.2012.42.9597. [DOI] [PubMed] [Google Scholar]
- 5.Yeh CH, Chen MF, Lai CH, et al. Comparison of treatment results between surgery alone, preoperative short-course radiotherapy, or long-course concurrent chemoradiotherapy in locally advanced rectal cancer”. Int J Clin Oncol. 2012;17:482–490. doi: 10.1007/s10147-011-0317-0. [DOI] [PubMed] [Google Scholar]
- 6.Krajcovicova I, Boljesikova E, Sandorova M, et al. Preoperative radiotherapy of locally advanced rectal cancer: clinical outcome of short-course and long-course treatment with or without concomitant chemotherapy. Klin Onkol. 2012;25:364–369. [PubMed] [Google Scholar]
- 7.Bujko K, Wyrwicz L, Rutkowski A, et al. Long-course oxaliplatin-based preoperative chemoradiation versus 5× 5 Gy and consolidation chemotherapy for cT4 or fixed cT3 rectal cancer: results of a randomized phase III study. Annals of Oncology. 2016;27:834–842. doi: 10.1093/annonc/mdw062. [DOI] [PubMed] [Google Scholar]
- 9.Gamaleldin M, Church JM, Stocchi L, et al. Is routine use of adjuvant chemotherapy for rectal cancer with complete pathological response justified? The American Journal of Surgery. 2017;213:478–483. doi: 10.1016/j.amjsurg.2016.11.028. [DOI] [PubMed] [Google Scholar]
- 10.Liu S-X, et al. Short-course Versus Long-course Preoperative Radiotherapy plus Delayed Surgery in the Treatment of Rectal Cancer: a Meta-analysis. Asian Pacific Journal of Cancer Prevention 2015. Asian Pacific Organization for Cancer Prevention. 2015;16:5755. doi: 10.7314/apjcp.2015.16.14.5755. [DOI] [PubMed] [Google Scholar]
- 11.Radu C, Berglund A, Påhlman L, Glimelius B. Short-course preoperative radiotherapy with delayed surgery in rectal cancer–a retrospective study. Radiotherapy and Oncology. 2008;87:343–349. doi: 10.1016/j.radonc.2007.11.025. [DOI] [PubMed] [Google Scholar]
- 12.Zhou ZR, Liu SX, Zhang TS, et al. Short-course preoperative radiotherapy with immediate surgery versus long-course chemoradiation with delayed surgery in the treatment of rectal cancer: a systematic review and meta-analysis. Surgical Oncology. 2014;23:211–221. doi: 10.1016/j.suronc.2014.10.003. [DOI] [PubMed] [Google Scholar]
- 13.Jadad AR, Moore RA, Carroll D. Assessing the quality of reports of randomized clinical trials: is blinding necessary? Controlled Clinical Trials. 1996;17:1–12. doi: 10.1016/0197-2456(95)00134-4. [DOI] [PubMed] [Google Scholar]
- 14.Read TE, McNevin MS, Gross EK, et al. Neoadjuvant therapy for adenocarcinoma of the rectum: tumor response and acute toxicity. Diseases of the Colon & Rectum. 2001;44:513–52. doi: 10.1007/BF02234323. [DOI] [PubMed] [Google Scholar]
- 15.Guckenberger M, Saur G, Wehner D, et al. Comparison of preoperative short-course radiotherapy and long-course radiochemotherapy for locally advanced rectal cancer. Strahlentherapie und Onkologie. 2012;188:551–557. doi: 10.1007/s00066-012-0131-2. [DOI] [PubMed] [Google Scholar]
- 16.Burduja M, Ionescu RT, Verga N. Accurate and efficient intracranial hemorrhage detection and subtype classification in 3D CT scans with convolutional and long short-term memory neural networks. Sensors. 2020;20:5611. doi: 10.3390/s20195611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wasserberg N, Gutman H. Resection margins in modern rectal cancer surgery. J Surg Oncol. 2008;98:611–615. doi: 10.1002/jso.21036. [DOI] [PubMed] [Google Scholar]
- 18.Liu S, Jiang T, Xiao L, et al. Total neoadjuvant therapy (TNT) versus standard neoadjuvant chemoradiotherapy for locally advanced rectal cancer: a systematic review and meta‐analysis. The Oncologist. 2021;26:e1555–e1566. doi: 10.1002/onco.13824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Scarlat F, Scarisoreanu A, Verga N. Absorbed dose distributions using the isodensitometric method for exposures with filter employed for mammographies. Romanian Reports in Physics. 2013;65:168–177. [Google Scholar]
- 20.Ishii Y, Hasegawa H, Endo T, et al. Medium-term results of neoadjuvant systemic chemotherapy using irinotecan, 5-fluorouracil, and leucovorin in patients with locally advanced rectal cancer. Eur J Surg Oncol. 2010;36:1061–1065. doi: 10.1016/j.ejso.2010.05.017. [DOI] [PubMed] [Google Scholar]
- 21.Uehara K, Hiramatsu K, Maeda A, et al. Neoadjuvant oxaliplatin and capecitabine and bevacizumab without radiotherapy for poor-risk rectal cancer: N-SOG 03 Phase II trial. Jpn J Clin Oncol. 2013;43:964–971. doi: 10.1093/jjco/hyt115. [DOI] [PubMed] [Google Scholar]
- 22.Glynne-Jones R, Harrison, M, Hughes R. Challenges in the neoadjuvant treatment of rectal cancer: balancing the risk of recurrence and quality of life. Cancer/Radiothérapie. 2013;17:675–685. doi: 10.1016/j.canrad.2013.06.043. [DOI] [PubMed] [Google Scholar]