SUMMARY
Background:
MET signaling is a key driver of papillary renal cell carcinoma (PRCC). Given that there is no optimal therapy for metastatic PRCC, we sought to compare an existing standard (sunitinib) to MET kinase inhibitors.
Methods:
We conducted a randomized, open-label, phase II trial involving patients with metastatic PRCC who had received up to one prior therapy (excluding vascular endothelial growth factor-directed agents). Patients were assigned to receive sunitinib, cabozantinib, crizotinib or savolitinib, with stratification by receipt of prior therapy and PRCC subtype. Progression-free survival (PFS) was the primary endpoint. With 41 eligible patients per arm, there was 85% power to detect a 75% improvement in median PFS in each experimental arm compared to sunitinib, employing a one-sided alpha of 0.10 for each test.
Findings:
Overall, 152 patients were enrolled. Enrollment to savolitinib (N=29) and crizotinib (N=28) arms was halted after a pre-specified futility analysis; planned accrual was completed for both sunitinib (N=46) and cabozantinib (n=44) arms. PFS was longer with cabozantinib versus sunitinib (9.0 months vs. 5.6 months; hazard ratio for progression or death, 0.60; 95% CI 0.37–0.97, P=0.019 [1-sided]). Response rate for cabozantinib was 23% versus 4% for sunitinib (2-sided P=0.010). Savolitinib and crizotinib did not improve PFS relative to sunitinib. Grade 3 or 4 adverse events occurred in 69%, 74%, 37% and 39% of patients receiving sunitinib, cabozantinib, crizotinib and savolitinib, respectively; one grade 5 thromboembolic event was seen with cabozantinib.
Interpretation:
Cabozantinib resulted in significantly longer PFS when compared to sunitinib in patients with metastatic PRCC.
Keywords: sunitinib, savolitinib, crizotinib, cabozantinib, papillary, MET, renal cell carcinoma
Introduction
Papillary renal cell carcinoma (PRCC) is a rare malignancy seen in approximately 15% of patients with kidney cancer.1 It can be further histologically sub-classified as either type 1, characterized by small cells in a single layer with scant basophilic cytoplasm and oval nuclei, or type 2, characterized by large cells with eosinophilic cytoplasm and spherical nuclei with prominent nucleoli and pseudostratification.2,3 Genomic and molecular characterization of PRCC has identified the presence of mutations and copy number alterations in the MET protooncogene. Although more frequently found in type 1, recent studies suggest that molecular alterations in the MET oncogene and/or activated MET pathway signaling also occur in type 2 disease.2,4,5
Since the introduction of vascular endothelial growth factor (VEGF)-directed therapies for metastatic clear cell RCC, the treatment paradigm for metastatic PRCC has evolved in parallel. Randomized comparisons suggest modestly improved clinical outcomes with sunitinib, a VEGF-directed multikinase inhibitor, when compared to everolimus, a mammalian target of rapamycin (mTOR) inhibitor, but these studies are limited by substantial heterogeneity given inclusion of a wide array of non-clear cell histologies beyond PRCC.6,7 Studies limited to PRCC suggest progression-free survival (PFS) with sunitinib in the range of 1.6–6.0 months, with response rates ranging from 0–13% in the same studies.8,9 Nevertheless, sunitinib has become the reference standard of care for patients with advanced or metastatic PRCC.10
The finding that MET biology is important in PRCC has led to the investigation of small molecule MET inhibitors in this disease.2,4,5 Agents such as crizotinib, savolitinib, and cabozantinib have shown potent MET inhibitory properties in preclinical models, with inhibitory concentration-50 (IC50) of 11, 4.0 and 1.3 nM, respectively.11–13 The CREATE trial evaluated crizotinib in patients with type 1 disease, demonstrating that a small proportion of patients could achieve durable responses.14 A phase II study of savolitinib showed encouraging responses in mPRCC patients with MET-driven disease.15 Most recently, in a study of non-clear cell RCC patients treated with cabozantinib, those with papillary disease appeared to derive a benefit when compared to historical standards.16 These results are limited by the lack of a relevant contemporary comparator group. We therefore designed SWOG 1500, a randomized phase II trial comparing sunitinib to cabozantinib, crizotinib and savolitinib, with the aim of determining if MET-directed therapy could improve clinical outcomes relative to conventional VEGF-directed agents.
Methods
Study Design and Participants
Eligible patients were 18 years or older and had pathologically verified PRCC. Available tissue submission for retrospective central adjudication of PRCC subtype was mandated. Patients were allowed to receive up to one prior systemic therapy, excluding VEGF- or MET-directed agents. Patients with known brain metastases who had received adequate treatment were eligible. Eligible patients had a Zubrod performance status of 0–1 and adequate organ and marrow function. Patients with significant intercurrent illness or contraindications to study medications were excluded. Patients were randomized uniformly to the control arm of sunitinib or one of three investigational arms: cabozantinib, crizotinib, and savolitinib. Randomization was dynamically balanced on two stratification factors: the number of prior therapies (0 versus 1) and PRCC subtype (type 1 versus type 2 versus NOS) as designated by local pathologic review.17
Procedures
All study medications were supplied by the National Cancer Institute. Sunitinib was administered orally at a dose of 50 mg 4 weeks on, 2 weeks off, with dose reductions to 37.5 mg and 25 mg permitted. Cabozantinib was administered at 60 mg oral daily with dose reductions to 40 mg and 20 mg permitted. Crizotinib was administered at 250 mg twice daily with dose reductions to 200 mg twice daily and 250 mg once daily permitted. Savolitinib was dosed at 600 mg oral daily with dose reductions to 400 mg and 200 mg oral daily permitted. Patients received study therapy until radiographic progression or clinical deterioration/unacceptable toxicity, as determined by the investigator.
Outcomes
The primary endpoint was PFS, defined as the time from randomization to the time of radiographic or clinical progression, symptomatic deterioration or death from any cause, whichever occurred first. Secondary endpoints included toxicity, objective response rate and overall survival, defined as the time from randomization to death from any cause. Patients were assessed with computerized tomography or magnetic resonance imaging at baseline and every 12 weeks after randomization. Assessments continued until radiographic progression or discontinuation of study treatment, with further limited follow-up requested up to 3 years following randomization. Safety was evaluated by participating investigators using National Cancer Institute Common Terminology Criteria for Adverse Events, version 4.0, and response and progression was assessed using Response Evaluation Criteria in Solid Tumors (RECIST), version 1.1.
Pathologic Assessment
As part of the trial, eligible subjects submitted archival formalin-fixed, paraffin embedded tumor material. Participating sites were required to submit the pathology report and a representative hematoxylin/eosin stained slide to be scanned at 40X using Aperio Digital Pathology Slide Scanners. Three urologic pathologists with expertise in PRCC independently reviewed all cases and when needed, the prior pathology reports for review of previously performed local immunostains. Cases were considered to have papillary type 1, type 2 or not otherwise specified (NOS)/unclassified papillary histology by each pathologist. Designation of papillary histology was based on previously described features.3,18 Cases with mixed features or not falling into either category was considered NOS/unclassified papillary. When there was disagreement, the case was classified according to the majority or in the case of no agreement, re-reviewed. If needed after re-review, a group discussion resolved classification or consideration of NOS/unclassified.
Trial Oversight
The full protocol is available in Supplementary Materials 1. The initial version and all subsequent amendments were approved by the National Cancer Institute Central Institutional Review Board. The study was conducted in accordance with the International Conference on Harmonization of Good Clinical Practice guidelines, and the principles of the Declaration of Helsinki. Written consent was obtained from all patients.
Statistical Analyses
The primary endpoint was PFS, as defined above. We estimated that with 164 eligible patients (41 patients per arm), we would have 85% power to detect a 75% improvement in median PFS in any one of the three experimental arms relative to sunitinib (1-sided alpha of 0.10). This was based on the assumption of a median PFS in the sunitinib arm of 6.0 months.8 A proportional hazards (PH) model was used to evaluate each pairwise treatment comparison of PFS, adjusting for the two stratification factors as covariates in the model. Although one-sided p-values are reported (as specified in the protocol), we also report 95% confidence intervals for descriptive purposes to allow for evaluation of a two-sided 0.05 significance level.
A futility analysis was pre-specified to be conducted after fifteen PFS events were recorded on each experimental arm. If the PFS hazard ratio for an experimental arm versus sunitinib was greater than 1, corresponding to inferior PFS on the experimental arm, that arm would be recommended for closure. In prespecified, supportive analyses, the prognostic significance of histologic subtype (local designation and central review) was evaluated along with an assessment of their possible treatment modification by fitting an interaction term in the PH model for PFS.
Role of the funding source
The funder of the study had no role in study design, data collection, data analysis, data interpretation, or writing of the report. All authors had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Results
Patients
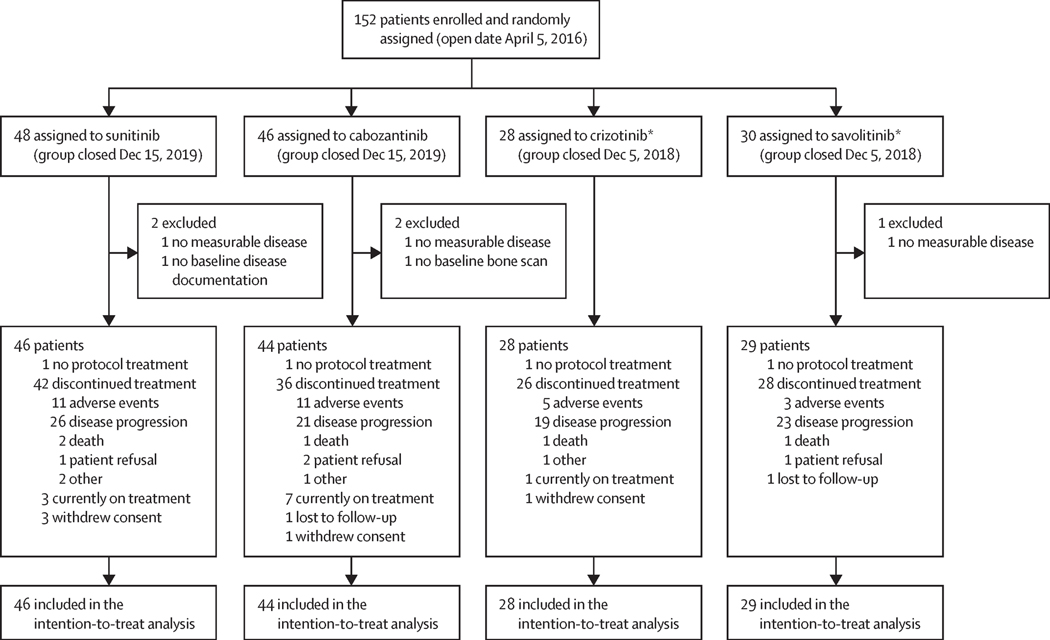
From April 2016 to December 2019, 152 patients were enrolled at 65 centers throughout the US and Canada. At the time of data cutoff on October 16, 2020, 5 patients were deemed ineligible due to inadequate baseline disease assessment or no evidence of metastatic disease (see CONSORT diagram; Figure 1). 147 eligible patients were randomized to receive sunitinib (46 patients), cabozantinib (n=44), savolitinib (n=29) and crizotinib (n=28). All eligible patients were used in the intention-to-treat population for analysis of efficacy. One patient in each arm received no protocol therapy, and they were excluded from the safety assessment.
Figure 1.
CONSORT diagram.
Median age of the cohort was 66 years (range 29–89) and 76% were male. As of the data cutoff, 3, 7, 1, and 0 patients were still receiving protocol therapy with sunitinib, cabozantinib, crizotinib, and savolitinib, respectively. Baseline characteristics by arm and pooled across arms are shown in Table 1. Prior therapy was received by 10 patients (7%), and the most common prior therapy was nivolumab with ipilimumab, which was received by 4 patients.
Table 1.
Patient characteristics.
| All Patients (n=147) | Sunitinib (n=46) | Cabozantinib (n=44) | Crizotinib (n=28) | Savolitinib (n=29) | |
|---|---|---|---|---|---|
| Median Age (Range), years (IQR) | 66 (58,75) | 65 (58,73) | 65 (58,75) | 68 (61,75) | 67 (58,72) |
| Males [n (%)] | 112 (76%) | 35 (76%) | 36 (82%) | 22 (79%) | 19 (66%) |
| Race [n (%)] | |||||
| White | 114 (78%) | 39 (85%) | 32 (73%) | 22 (79%) | 21 (72%) |
| Black | 21 (14%) | 5 (11%) | 9 (20%) | 4 (14%) | 3 (10%) |
| Other | 6 (4%) | 2 (4%) | 3 (7%) | 2 (8%) | 5 (16%) |
| Unknown | 6 (4%) | 0 (0%) | 2 (5%) | 1 (4%) | 3 (10%) |
| Prior Systemic Therapy [n (%)] | 10 (7%) | 3 (7%) | 2 (5%) | 2 (7%) | 3 (10%) |
| Histological Subtype [n (%)] (Local assessment) | |||||
| Type I | 27 (18%) | 8 (17%) | 9 (20%) | 5 (18%) | 5 (17%) |
| Type II | 79 (54%) | 24 (52%) | 25 (57%) | 15 (54%) | 15 (52%) |
| Not otherwise specified | 41 (28%) | 14 (30%) | 10 (23%) | 8 (29%) | 9 (31%) |
| Histologic Subtype [n (%)] (Central assessment) | |||||
| Type I | 41 (28%) | 12 (26%) | 14 (32%) | 9 (32%) | 6 (21%) |
| Type II | 63 (43%) | 21 (46%) | 16 (36%) | 13 (46%) | 13 (45%) |
| Mixed/Other | 35 (24%) | 11 (24%) | 12 (27%) | 5 (18%) | 7 (24%) |
| Missing | 8 (5%) | 2 (4%) | 2 (5%) | 1 (4%) | 3 (10%) |
| IMDC Risk Group [n (%)] | |||||
| Favorable | 38 (26%) | 14 (30%) | 10 (23%) | 8 (29%) | 6 (30%) |
| Intermediate | 89 (61%) | 26 (57%) | 28 (64%) | 16 (57%) | 19 (66%) |
| High | 20 (14%) | 6 (13%) | 6 (14%) | 4 (14%) | 4 (14%) |
| Zubrod PS [n (%)] | |||||
| 0 | 91 (62%) | 29 (63%) | 29 (66%) | 18 (64%) | 15 (52%) |
| 1 | 56 (38%) | 17 (37%) | 15 (34%) | 10 (36%) | 14 (48%) |
| Prior Nephrectomy [n (%)] | 113 (77%) | 34 (74%) | 32 (73%) | 26 (72%) | 21 (72%) |
| Metastatic Sites of Interest [n (%)] | |||||
| Bone | 26 (18%) | 7 (15%) | 6 (14%) | 5 (18%) | 8 (28%) |
| Central nervous system | 1 (< 1%) | 0 (0%) | 0 (0%) | 1 (< 1%) | 0 (0%) |
Efficacy
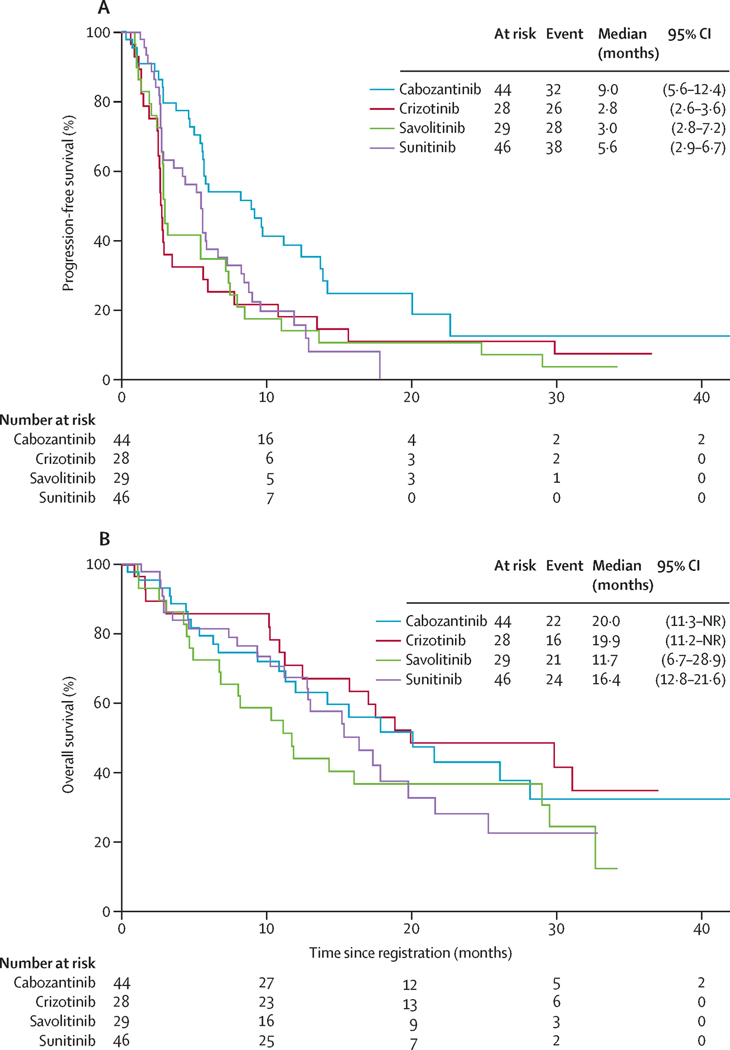
At the time of pre-specified interim analysis, savolitinib and crizotinib demonstrated a hazard ratio for PFS greater than 1 relative to sunitinib (see Supplementary Materials 2 for further details). The DSMC subsequently recommended that accrual be stopped to the savolitinib and crizotinib arms, and this recommendation was accepted by the NCI. The trial was then temporarily closed on December 5, 2018 so the protocol and consent could be amended to reflect the updated design. The trial was reopened to accrual to the remaining arms (sunitinib and cabozantinib) on January 22, 2019. A subsequent futility analysis was conducted in which the DSMC recommended continued accrual to the cabozantinib and sunitinib arms to the planned full accrual of 45 patients per arm. At final analysis, median PFS was 5.6 months for sunitinib and 9.0 months, 2.8 months, and 3.0 months for cabozantinib, crizotinib and savolitinib, respectively (Figure 2a). The covariate-adjusted hazard ratio for PFS in the comparison of cabozantinib to sunitinib was 0.60 (95%CI 0.37–0.97 [1-sided P-value=0.019]).
Figure 2.
Kaplan-Meier analysis of progression-free survival (a) and overall survival (b).
The confirmed overall response rate was 4%, 23%, 0% and 3% with sunitinib, cabozantinib, crizotinib and savolitinib, respectively (Supplemental Table 1). Two patients (5%) achieved a complete response with cabozantinib – no complete responses were observed on the other treatment arms. The response rate associated with cabozantinib was significantly higher than that with sunitinib (2-sided P-value=0.010). Median overall survival was 16.4 months for sunitinib, and was 20.0, 19.9 and 11.7 months for cabozantinib, crizotinib and savolitinib, respectively (Figure 2b). The hazard ratio for the survival comparison between cabozantinib and sunitinib was 0.84 (95%CI 0.47–1.51). No significant differences in overall survival were observed between treatment arms. In Supplemental Table 2 we report the cross-classification of locally assessed histologic subtype (a stratification factor) and centrally assessed histologic subtype. The estimated treatment effect within each histologic subset was consistently in the favorable direction for cabozantinib (Supplemental Table 3).
Safety
The rate of treatment discontinuation due to adverse events (attributed to study medication) was highest with sunitinib (11 patients, 24%), followed by cabozantinib (10 patients, 23%), crizotinib (4 patients, 16%), and savolitinib (3 patients, 10%). Sixteen patients remained on protocol treatment at the time of last follow-up. Adverse events occurred in 93%, 98%, 93% and 93% of patients receiving sunitinib, cabozantinib, crizotinib and savolitinib, respectively (Table 2). Grade 3 or 4 adverse events occurred in 69%, 74%, 37% and 39% of patients treated with sunitinib, cabozantinib, crizotinib, and savolitinib, respectively. The most common grade 3 or 4 adverse events with sunitinib were hypertension (17%), anemia (13%) and decrease in white blood cell count (11%). The most common grade 3 or 4 adverse events with cabozantinib were hypertension (32%), hand-foot syndrome (20%), and fatigue (13%). The most common grade 3 or 4 adverse events with crizotinib were fatigue and increase in ALT levels (7% for both). The most common grade 3 or 4 adverse events with savolitinib were hyponatremia (10%), thrombocytopenia (7%), and limb swelling (7%). One grade 4 adverse event (i.e., death) within 30 days of the last dose of study medication was reported in one patient receiving cabozantinib, secondary to thromboembolic event.
Table 2.
Adverse events occurring in 10% or more of any treatment arm*
| Sunitinib | Cabozantinib | Crizotinib | Savolitinib | |||||
|---|---|---|---|---|---|---|---|---|
| Toxicity | Any Grade [n (%)] | Grade 3 or 4 [n (%)] | Any Grade [n (%)] | Grade 3 or 4 [n (%)] | Any Grade [n (%)] | Grade 3 or 4 [n (%)] | Any Grade [n (%)] | Grade 3 or 4 [n (%)] |
| Overall | 42 (93) | 31 (68) | 42 (97) | 32 (74) | 25 (92) | 10 (37) | 26 (92) | 11 (39) |
| Fatigue | 26 (57) | 3 (6) | 30 (69) | 6 (13) | 13 (48) | 2 (7) | 14 (50) | 0 (0) |
| Diarrhea | 22 (48) | 3 (6) | 24 (55) | 2 (4) | 12 (44) | 0 (0) | 3 (10) | 0 (0) |
| Nausea | 19 (42) | 4 (8) | 15 (34) | 0 (0) | 7 (25) | 0 (0) | 12 (42) | 0 (0) |
| Hypertension | 16 (35) | 8 (17) | 28 (65) | 14 (32) | 3 (11) | 0 (0) | 6 (21) | 1 (3) |
| Anorexia | 14 (31) | 0 (0) | 19 (44) | 1 (2) | 5 (18) | 0 (0) | 6 (21) | 0 (0) |
| Mucositis oral | 13 (28) | 0 (0) | 16 (37) | 1 (2) | 1 (3) | 0 (0) | 0 (0) | 0 (0) |
| Hand-Foot syndrome | 11 (24) | 0 (0) | 21 (48) | 9 (20) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Vomiting | 11 (24) | 1 (2) | 6 (13) | 0 (0) | 2 (7) | 0 (0) | 2 (7) | 0 (0) |
| Dehydration | 5 (11) | 1 (2) | 1 (2) | 0 (0) | 0 (0) | 0 (0) | 1 (3) | 1 (3) |
| Abdominal pain | 3 (6) | 1 (2) | 6 (13) | 3 (6) | 1 (3) | 0 (0) | 1 (3) | 0 (0) |
| Dyspnea | 3 (6) | 0 (0) | 5 (11) | 0 (0) | 4 (14) | 0 (0) | 2 (7) | 1 (3) |
| Edema limbs | 3 (6) | 0 (0) | 3 (6) | 0 (0) | 6 (22) | 1 (3) | 8 (28) | 2 (7) |
| GI disorders-Other, specify | 3 (6) | 0 (0) | 7 (16) | 1 (2) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Pain in extremity | 3 (6) | 0 (0) | 6 (13) | 1 (2) | 2 (7) | 1 (3) | 1 (3) | 0 (0) |
| Rash maculo-papular | 3 (6) | 0 (0) | 8 (18) | 0 (0) | 2 (7) | 1 (3) | 2 (7) | 1 (3) |
| Thromboembolic event | 1 (2) | 0 (0) | 8 (18) | 5 (11) | 0 (0) | 0 (0) | 3 (10) | 2 (7) |
| Platelet count decreased | 18 (40) | 2 (4) | 8 (18) | 0 (0) | 0 (0) | 0 (0) | 1 (3) | 0 (0) |
| Anemia | 15 (33) | 6 (13) | 10 (23) | 0 (0) | 6 (22) | 1 (3) | 2 (7) | 0 (0) |
| White blood cell decreased | 13 (28) | 5 (11) | 9 (20) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Neutrophil count decreased | 11 (24) | 4 (8) | 7 (16) | 0 (0) | 1 (3) | 0 (0) | 0 (0) | 0 (0) |
| Lymphocyte count decreased | 10 (22) | 2 (4) | 6 (13) | 0 (0) | 2 (7) | 0 (0) | 3 (10) | 0 (0) |
| AST increased | 8 (17) | 1 (2) | 14 (32) | 0 (0) | 7 (25) | 1 (3) | 2 (7) | 0 (0) |
| Proteinuria | 7 (15) | 1 (2) | 7 (16) | 1 (2) | 0 (0) | 0 (0) | 1 (3) | 0 (0) |
| ALT increased | 6 (13) | 1 (2) | 13 (30) | 1 (2) | 8 (29) | 2 (7) | 3 (10) | 0 (0) |
| Hypoalbuminemia | 6 (13) | 1 (2) | 6 (13) | 0 (0) | 4 (14) | 1 (3) | 5 (17) | 0 (0) |
| Hyponatremia | 4 (8) | 2 (4) | 3 (6) | 3 (6) | 0 (0) | 0 (0) | 7 (25) | 3 (10) |
| Hypophosphatemia | 3 (6) | 0 (0) | 11 (25) | 4 (9) | 1 (3) | 0 (0) | 0 (0) | 0 (0) |
| Hypocalcemia | 1 (2) | 0 (0) | 10 (23) | 1 (2) | 3 (11) | 0 (0) | 3 (10) | 0 (0) |
| Hypomagnesemia | 0 (0) | 0 (0) | 9 (20) | 2 (4) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Hypomagnesemia | 0 (0) | 0 (0) | 9 (20) | 2 (4) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
One grade 5 event was observed with cabozantinib secondary to thromboembolic event.
Discussion
This is the first randomized trial specific to metastatic PRCC to show a clinically and statistically significant benefit with a targeted therapy over an existing standard. We observed a significant improvement in both PFS and response rate with the dual VEGF/MET inhibitor cabozantinib relative to sunitinib. The more selective MET inhibitors crizotinib and savolitinib did not appear to have superior clinical activity versus sunitinib; in fact, those investigational arms were halted early in the study based on a pre-planned futility analysis. Our findings imply that PRCC may be driven by both VEGF- and MET-signaling pathways and thus may be more amenable to targeting with a dual VEGF/MET inhibitor such as cabozantinib.
MET alterations are abundant in patients with PRCC – the frequency is higher in patients with type 1 versus type 2 disease.4 Although alterations in VHL and resultant increases in VEGF have classically been associated with clear cell disease, some series point to similar, if not higher, expression of VEGF in PRCC.19 Activity with the dual VEGF and MET inhibitor foretinib has previously been observed in PRCC. A phase II study exploring foretinib in this setting produced a PFS of 9.3 months, similar to what we observed in the current study.20 Both foretinib and cabozantinib also have affinity for AXL, a receptor tyrosine kinase that belongs to the TAM family and is overexpressed in both clear cell and papillary RCC histologies, and may also serve as an important therapeutic target.21 As small molecules inhibit diverse targets, the key driver of cabozantinib response in PRCC remains to be determined.
Of interest will be the MET status of the PRCC tumors in the current study. A recent randomized study comparing sunitinib to savolitinib in metastatic PRCC was designed solely for those with presumably MET-driven disease, defined as gains in chromosome 7, MET amplification, MET kinase domain variations, or HGF alterations.22 That study was terminated after accrual of 60 patients; subsequent analysis did show a numerically higher response rate with savolitinib as compared to sunitinib (27% versus 7%). Savolitinib does appear to have more pronounced activity in the context of MET alteration. A single-arm, phase II study of savolitinib including 109 unselected patients with PRCC showed a response rate of 18% in MET-driven disease versus 0% in patients with MET independent disease.15 Crizotinib may similarly fare better in PRCC patients bearing MET alteration. In the European Organization for the Research and Treatment of Cancer (EORTC) CREATE trial enrolling patients with type I PRCC, 2 of 4 patients (50%) with MET mutation garnered responses with crizotinib. In contrast, only 1/16 patients (6.3%) lacking MET mutation responded.14 One might speculate that the savolitinib and crizotinib arms in the current study may have fared better had we employed a biomarker-based design. In our current trial, biospecimens were prospectively collected for pre-planned molecular characterization of MET pathway activation, which was hypothesized to be a predictive biomarker in the pooled MET inhibitor groups vs sunitinib.
In the 98 cases in which type 1 or type 2 designation was rendered both locally and with central path review, a concordant diagnosis was offered in 62 of the 98 cases (63%). In 41 of the 147 eligible, randomized patients (28%), a diagnosis of “papillary histology NOS” was rendered. These data highlight the challenges in assigning an accurate histologic subtype. Ultimately, since the estimated clinical benefit with cabozantinib was observed across both type 1 and 2 disease, it is unclear whether this designation will have clinical relevance. As noted, the presence of MET alterations has been identified in both subtypes. The ongoing molecular studies in this trial may provide clarity as to which specific molecular phenotypes are associated with response or resistance to the systemic therapies given as part of the study.
Limitations of the current study include the heterogeneity among the subtypes of metastatic PRCC assessed. At one point during its development, it was suggested the study be limited to PRCC type 1 patients exclusively; however, it was felt that the relative infrequency of this subtype and need for real-time central pathology review would represent a barrier to accrual. Another initial consideration was to restrict eligibility to patients with documented alterations in the MET protooncogene or MET pathway activation. This was thought to be infeasible, as delays for tissue procurement and screening might be prohibitive in a potentially rapidly growing disease. Our study was also not designed to assess differences in overall survival. Crossover to other potentially active treatments following the study could confound the assessment of overall survival. Emerging prospective studies utilizing checkpoint inhibitors such as pembrolizumab recently suggest activity in non-clear cell subtypes.23 Use of these therapies would need to be accounted for whilst interpreting overall survival.
One must consider that the open-label nature of our study could also bias the primary endpoint of investigator-assessed PFS. Although funding was not able to facilitate a central review of imaging in the current study, there is precedent for this in the context of other cooperative group studies. The randomized, phase II CABOSUN clinical trial, comparing cabozantinib and sunitinib in advanced clear cell RCC, did perform a post hoc central review of imaging.24 Finally, our study was a randomized phase II effort – ideally, a phase III study would be performed to establish a standard of care for this disease. The feasibility of such an endeavor is questionable. Our study, despite being an effort across North America, accrued at a pace of only roughly 3.5 patients per month. As noted previously, other attempted phase III efforts such as the SAVOIR study have closed prematurely.
These limitations notwithstanding, our study provides compelling evidence that cabozantinib could be considered a new benchmark for patients with metastatic PRCC. Single-arm trials have shown compelling evidence for novel combinations of targeted therapy with immunotherapy – for instance, a trial combining savolitinib and durvalumab yielded a response rate of 27% in patients with advanced papillary RCC.25 Given the emerging activity with cabozantinib-based combinations with checkpoint inhibitors in both clear cell and non-clear cell RCC, a study comparing cabozantinib with or without checkpoint inhibitor in papillary RCC would be of interest.26,27
Supplementary Material
Research in Context.
Evidence before this study
A search of Pubmed along with major urology and oncology congresses was conducted. Articles and research abstracts in English that were related to metastatic papillary renal cell carcinoma were searched between the dates of June 1, 2010 and June 1, 2020. MeSH terms “papillary renal cell carcinoma”, “papillary kidney cancer”, “targeted therapy”, “VEGF-TKI” and “MET inhibitor” were used. The results of the search imply that there is no obvious standard of care for the management of metastatic papillary renal cell carcinoma, with no randomized trials conducted exclusively in this population. Agents that have been developed for clear cell renal cell carcinoma, such as sunitinib, have been applied in this disease, but results have been modest at best, with progression-free survival estimates ranging from 1.5 to 6 months. Biological evidence has emerged in recent years supporting the role of the MET protooncogene as a driver of papillary kidney cancer biology. With that in mind, several single-arm, prospective trials have evaluated MET inhibitors (e.g., foretinib and savolitinib) with encouraging results. However, in the only attempt to compare MET inhibition to VEGF inhibition in papillary renal cell carcinoma to date (the prospective, randomized phase III SAVOIR trial), the study was terminated prematurely with no conclusive results.
Added value of this study
SWOG 1500 study is the first completed randomized trial assessing targeted therapies specifically in patients with metastatic papillary kidney cancer. The study compared sunitinib to three putative MET inhibitors (cabozantinib, crizotinib, and savolitinib). Of these MET inhibitors, cabozantinib has the strongest affinity for MET and additionally blocks the VEGF receptor. The study met its primary endpoint by demonstrating a significant benefit in progression-free survival with cabozantinib versus sunitinib; notably, response rate was also considerably higher with cabozantinib as compared to sunitinib. Equally important, the study showed that the MET inhibitors crizotinib and savolitinib did not compare favorably to sunitinib – the respective study arms were closed at the time of an interim futility analysis. The safety profiles of all drugs were consistent with previous studies.
Implications of all the available evidence
SWOG 1500 is the first study comparing systemic therapies for metastatic papillary kidney cancer to meet its primary endpoint. The response rate and progression-free survival associated with cabozantinib in this study establish a benchmark for future trials in this disease state. Existing US Food and Drug Administration and European Medicine Agency labels for cabozantinib and sunitinib for the front-line treatment of advanced renal cell carcinoma are agnostic to histology – therefore, based on the results of SWOG 1500, cabozantinib could immediately supplant sunitinib as a clinical standard of care for patients with metastatic papillary kidney cancer.
Acknowledgements
SWOG 1500 was supported by NIH/National Cancer Institute grants CA180888, CA180819, CA180820, CA180821, CA180863, and CA180868.
Funding: Funded by NIH/National Cancer Institute grants CA180888, CA180819, CA180820, CA180821, CA180863, and CA180868; SWOG 1500 ClinicalTrials.gov number, NCT02761057.)
Contributors
SKP, CT, IMT, NBH, DJG, DYH, BS, MS, MT, PH, AAd, VN GAB, UV, AAl TZ, SC, MP, and PNL contributed to the conceptualization, methodology, and investigation of the trial and manuscript. SKP, CT, MP, and JW acquired funding for the trial. SKP, CT, MP, and PNL contributed to the formal analysis of the data and accessed and verified the data. SKP, CT, NBH, DJG, DYH, JW and PNL were responsible for the supervision of the trial and manuscript development. All authors contributed equally to the writing, review, and editing of the manuscript and approve of the final version.
Declaration of Interests
SKP reports personal fees from Genentech, personal fees from Aveo, personal fees from Eisai, personal fees from Roche, personal fees from Pfizer, personal fees from Novartis, personal fees from Exelixis, personal fees from Ipsen, personal fees from BMS, personal fees from Astellas, outside the submitted work.
IMT reports grants from SWOG - NIH/NCI grant, during the conduct of the study.
NBH reports personal fees from Exelixis, outside the submitted work.
DJG reports personal fees from American Assoc for Cancer Research, grants and personal fees from Astellas, grants and personal fees from Astrazeneca, personal fees from Axess Oncology, personal fees and other from Bayer H/C, personal fees from BMS, grants from Calithera, personal fees from Capio Biosciences, personal fees from Constellation Pharmaceuticals, personal fees from EMD Serono, grants, personal fees and other from Exelixis, personal fees from Flatrion, personal fees from Ipsen, grants and personal fees from Janssen Pharmaceuticals, personal fees from Merck, personal fees from Michael J Hennessey Assoc, personal fees from Millennium Medical Publishing, personal fees from Modra Pharmaceitucals, personal fees from Myovant Sciences, personal fees from NCI Genitourinary SC member, personal fees from Nektar Therapeutics, grants from Novartis, personal fees from Physician Education Resource, grants and personal fees from Pfizer, personal fees from Propella TX, grants, personal fees and other from Sanofi, personal fees from UroGPO, personal fees and other from UroToday, personal fees from Vizuri Health Sciences, outside the submitted work
DYH reports grants from Canadian Clinical Trials Group during the conduct of the study; personal fees from Pfizer, personal fees from Ipsen, personal fees from Astra Zeneca, personal fees from BMS, personal fees from Merck, outside the submitted work.
MS reports grants from Janssen Oncology, grants from Advaxis, grants from Harpoon, grants from Bristol-Myers Squibb, grants from Genocea Biosciences, grants from Lilly, grants from Nektar, grants and personal fees from Xencor, grants from Tmunity Therapeutics, grants from Exelixis, personal fees from Merck Sharp & Dohme, outside the submitted work.
VN reports grants and personal fees from Pfizer, grants and personal fees from Janssen, grants from Merck, grants from Bristol-Myers Squibb, personal fees from Myovant Sciences, personal fees from Regeneron, outside the submitted work.
GAB reports personal fees from Pfizer, personal fees from BMS, personal fees from Ipsen, personal fees from Merck, outside the submitted work.
UV reports grants and personal fees from BMS, grants and personal fees from Exelixis, grants and personal fees from Alkermes, grants and personal fees from Merck, personal fees from Bayer, personal fees from Sanofi, personal fees from Pfizer, outside the submitted work.
AAl reports grants and personal fees from AstraZeneca, personal fees from Pfizer, outside the submitted work
TZ reports grants from Acerta, grants from Novartis, grants from Merrimack, grants from Abbvie/Stemcentrx, grants and personal fees from Merck, grants from Regeneron, grants from Mirati Therapeutics, grants and personal fees from Janssen, grants and personal fees from Astra Zeneca, grants and personal fees from Pfizer, grants from Omniseq, grants from PGDx, grants and personal fees from SeaGen, personal fees from Genentech Roche, personal fees from Exelixis, personal fees from Amgen, personal fees from BMS, personal fees from Sanofi-Aventis, personal fees from Pharmacyclics, personal fees from Foundation Medicine, personal fees from MJH Life Sciences, personal fees from Calithera Biosciences, personal fees from Dendreon, other from Capio Biosciences, other from Archimmune Therapeutics, personal fees from Nanorobotics, outside the submitted work.
CT, BS, MT, PH, AAd, SC, MP, JW, and PNL report no conflicts of interest.
Footnotes
Data Sharing
Access to de-identified participant data and additional related documents such as study protocol, statistical analysis plan, and informed consent form will be granted according to the merit of a written application assessed by the SWOG GU Committee. SWOG, as part of the NCI’s National Clinical Trials Network, adheres to their policy of making clinical trial data available per request. Requests will be considered from 3 months of publication of this manuscript. Requests must be sent to spal@coh.org. Regarding use of the data, a data usage agreement will be executed between the requesting investigator and SWOG.
References
- 1.Zhang T, Gong J, Maia MC, Pal SK. Systemic Therapy for Non-Clear Cell Renal Cell Carcinoma. Am Soc Clin Oncol Educ Book 2017; 37: 337–42. [DOI] [PubMed] [Google Scholar]
- 2.Cancer Genome Atlas Research Network, Linehan WM, Spellman PT, et al. Comprehensive Molecular Characterization of Papillary Renal-Cell Carcinoma. N Engl J Med 2016; 374: 135–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Moch H, Cubilla AL, Humphrey PA, Reuter VE, Ulbright TM. The 2016 WHO Classification of Tumours of the Urinary System and Male Genital Organs—Part A: Renal, Penile, and Testicular Tumours. European Urology 2016; 70: 93–105. [DOI] [PubMed] [Google Scholar]
- 4.Pal SK, Ali SM, Yakirevich E, et al. Characterization of Clinical Cases of Advanced Papillary Renal Cell Carcinoma via Comprehensive Genomic Profiling. Eur Urol 2018; 73: 71–8. [DOI] [PubMed] [Google Scholar]
- 5.Albiges L, Guegan J, Le Formal A, et al. MET is a potential target across all papillary renal cell carcinomas: result from a large molecular study of pRCC with CGH array and matching gene expression array. Clin Cancer Res 2014; 20: 3411–21. [DOI] [PubMed] [Google Scholar]
- 6.Armstrong AJ, Halabi S, Eisen T, et al. Everolimus versus sunitinib for patients with metastatic non-clear cell renal cell carcinoma (ASPEN): a multicentre, open-label, randomised phase 2 trial. Lancet Oncol 2016; 17: 378–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Tannir NM, Jonasch E, Albiges L, et al. Everolimus Versus Sunitinib Prospective Evaluation in Metastatic Non-Clear Cell Renal Cell Carcinoma (ESPN): A Randomized Multicenter Phase 2 Trial. Eur Urol 2016; 69: 866–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ravaud A, Oudard S, De Fromont M, et al. First-line treatment with sunitinib for type 1 and type 2 locally advanced or metastatic papillary renal cell carcinoma: a phase II study (SUPAP) by the French Genitourinary Group (GETUG)†. Ann Oncol 2015; 26: 1123–8. [DOI] [PubMed] [Google Scholar]
- 9.Tannir NM, Plimack E, Ng C, et al. A phase 2 trial of sunitinib in patients with advanced non-clear cell renal cell carcinoma. Eur Urol 2012; 62: 1013–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.National Comprehensive Cancer Network (NCCN) Guidelines for Kidney Cancer, Version 1.2021 (Available at http://www.nccn.org; last accessed November 25, 2020.). . [DOI] [PMC free article] [PubMed]
- 11.Yakes FM, Chen J, Tan J, et al. Cabozantinib (XL184), a novel MET and VEGFR2 inhibitor, simultaneously suppresses metastasis, angiogenesis, and tumor growth. Mol Cancer Ther 2011; 10: 2298–308. [DOI] [PubMed] [Google Scholar]
- 12.Zou HY, Li Q, Lee JH, et al. An orally available small-molecule inhibitor of c-Met, PF-2341066, exhibits cytoreductive antitumor efficacy through antiproliferative and antiangiogenic mechanisms. Cancer Res 2007; 67: 4408–17. [DOI] [PubMed] [Google Scholar]
- 13.Gavine PR, Ren Y, Han L, et al. Volitinib, a potent and highly selective c-Met inhibitor, effectively blocks c-Met signaling and growth in c-MET amplified gastric cancer patient-derived tumor xenograft models. Mol Oncol 2015; 9: 323–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Schöffski P, Wozniak A, Escudier B, et al. Crizotinib achieves long-lasting disease control in advanced papillary renal-cell carcinoma type 1 patients with MET mutations or amplification. EORTC 90101 CREATE trial. Eur J Cancer 2017; 87: 147–63. [DOI] [PubMed] [Google Scholar]
- 15.Choueiri TK, Plimack E, Arkenau H-T, et al. Biomarker-Based Phase II Trial of Savolitinib in Patients With Advanced Papillary Renal Cell Cancer. J Clin Oncol 2017; 35: 2993–3001. [DOI] [PubMed] [Google Scholar]
- 16.Martínez Chanzá N, Xie W, Asim Bilen M, et al. Cabozantinib in advanced non-clear-cell renal cell carcinoma: a multicentre, retrospective, cohort study. Lancet Oncol 2019; 20: 581–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Pocock SJ, Simon R. Sequential treatment assignment with balancing for prognostic factors in the controlled clinical trial. Biometrics 1975; 31: 103–15. [PubMed] [Google Scholar]
- 18.Srigley JR, Delahunt B, Eble JN, et al. The International Society of Urological Pathology (ISUP) Vancouver Classification of Renal Neoplasia. Am J Surg Pathol 2013; 37: 1469–89. [DOI] [PubMed] [Google Scholar]
- 19.Song SH, Jeong IG, You D, et al. VEGF/VEGFR2 and PDGF-B/PDGFR-β expression in non-metastatic renal cell carcinoma: a retrospective study in 1,091 consecutive patients. Int J Clin Exp Pathol 2014; 7: 7681–9. [PMC free article] [PubMed] [Google Scholar]
- 20.Choueiri TK, Vaishampayan U, Rosenberg JE, et al. Phase II and biomarker study of the dual MET/VEGFR2 inhibitor foretinib in patients with papillary renal cell carcinoma. Journal of Clinical Oncology: Official Journal of the American Society of Clinical Oncology 2013; 31: 181–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Yu H, Liu R, Ma B, et al. Axl receptor tyrosine kinase is a potential therapeutic target in renal cell carcinoma. Br J Cancer 2015; 113: 616–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Choueiri TK, Heng DYC, Lee JL, et al. Efficacy of Savolitinib vs Sunitinib in Patients With MET-Driven Papillary Renal Cell Carcinoma: The SAVOIR Phase 3 Randomized Clinical Trial. JAMA Oncol 2020; published online May 29. DOI: 10.1001/jamaoncol.2020.2218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.McDermott DF, Lee J-L, Ziobro M, et al. First-line pembrolizumab (pembro) monotherapy for advanced non-clear cell renal cell carcinoma (nccRCC): Results from KEYNOTE-427 cohort B. JCO 2019; 37: 546–546. [Google Scholar]
- 24.Choueiri TK, Hessel C, Halabi S, et al. Cabozantinib versus sunitinib as initial therapy for metastatic renal cell carcinoma of intermediate or poor risk (Alliance A031203 CABOSUN randomised trial): Progression-free survival by independent review and overall survival update. Eur J Cancer 2018; 94: 115–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Suarez Rodriguez C, Larkin JMG, Patel P, et al. Overall survival results for durvalumab and savolitinib in metastatic papillary renal cancer. JCO 2020; 38: 619–619. [Google Scholar]
- 26.Choueiri TK, Powles T, Burotto M, et al. 696O_PR Nivolumab + cabozantinib vs sunitinib in first-line treatment for advanced renal cell carcinoma: First results from the randomized phase III CheckMate 9ER trial. Annals of Oncology 2020; 31: S1159. [Google Scholar]
- 27.McGregor BA, Agarwal N, Suarez C, et al. 709P Cabozantinib (C) in combination with atezolizumab (A) in non-clear cell renal cell carcinoma (nccRCC): Results from cohort 10 of the COSMIC-021 study. Annals of Oncology 2020; 31: S558. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.