Abstract
INTRODUCTION:
Pancreatitis is a complex syndrome that results from many etiologies. Large well-characterized cohorts are needed to further understand disease risk and prognosis.
METHODS:
A pancreatitis cohort of more than 4,200 patients and 24,000 controls were identified in the UK BioBank (UKBB) consortium. A descriptive analysis was completed, comparing patients with acute (AP) and chronic pancreatitis (CP). The Toxic-metabolic, Idiopathic, Genetic, Autoimmune, Recurrent, and severe pancreatitis and Obstructive checklist Version 2 classification was applied to patients with AP and CP and compared with the control population.
RESULTS:
CP prevalence in the UKBB is 163 per 100,000. AP incidence increased from 21.4/100,000 per year from 2001 to 2005 to 48.2/100,000 per year between 2016 and 2020. Gallstones and smoking were confirmed as key risk factors for AP and CP, respectively. Both populations carry multiple risk factors and a high burden of comorbidities, including benign and malignant neoplastic disorders.
DISCUSSION:
The UKBB serves as a rich cohort to evaluate pancreatitis. Disease burden of AP and CP was high in this population. The association of common risk factors identified in other cohort studies was confirmed in this study. Further analysis is needed to link genomic risks and biomarkers with disease features in this population.
INTRODUCTION
Pancreatitis is a progressive, complex, destructive, inflammatory syndrome of the pancreas with a high risk of morbidity and mortality. Multiple etiologies follow what can be an unpredictable course that may include acute (AP), recurrent acute (RAP), and chronic (CP) pancreatitis. This sequence typically begins with a sentinel AP event (1–4). Approximately 20% of first-attack patients with AP develop recurrent attacks. Thirty-five percent of patients with RAP continue to end-stage CP. There is great variability in the rate of progression and severity of disease, which may in part be related to the underlying etiology, environmental exposures, and genetic factors.
Frameworks to assess pancreatitis risk factors and etiology exist, including the Toxic-metabolic, Idiopathic, Genetic, Autoimmune, Recurrent, and severe pancreatitis and Obstructive (TIGAR-O) checklist (5). This framework assesses environmental and genetic factors to determine the etiology of an individual's pancreatitis. Recently, the updated TIGAR-O Version 2 (TIGAR-O V2) was published to provide additional specificity to identifying and describing pancreatitis mechanism and etiology (6).
Previous studies have described the demographics, risk factors, and outcomes in AP and CP for North America (6–8), Europe (9–11) China (12), Japan (13,14), and the world (15). The North American Pancreatitis Study 2 (NAPS2) used the TIGAR-O V1 to a pancreatitis cohort to evaluate the multiple factors contributing to RAP and CP (8). NAPS2 has since generated a pipeline of insights into the mechanisms and outcomes of pancreatitis, including environmental and genomic factors (16–22). In addition, the Dutch Chronic Pancreatitis Registry (CARE) provides a robust chronic pancreatitis dataset (23).
The goal of this study is to supplement the current understanding of pancreatitis by describing the AP and CP population in the population-based UK BioBank (UKBB) (24). The UKBB was established to allow for prospective epidemiological studies on chronic diseases (25–28). Detailed records are kept on all participants to assure complete information. The UKBB offers a unique opportunity to investigate pancreatitis in a large well-documented population with well-described healthy and disease comparator populations. We describe a population of more than 4,200 individuals diagnosed with pancreatitis along with their comorbidities, risk factors, and potential etiologies, adding new insights into this complex syndrome.
METHODS
Study subjects
All study subjects were taken from the UKBB cohort that contains genetic and phenotypic data collected on more than 502,000 individuals from the United Kingdom general population aged 40 to 69, recruited from 2006 to 2010 (25–28). Pancreatitis phenotypes were determined using a 2-step process (Figure 1). First, all possible pancreatitis subjects were collected using a combination of data fields (DFs): self-reported pancreatitis (DF:20002), International Classification of Disease 9th Revision (ICD-9) codes (DF:41271), International Classification of Disease 10th Revision (ICD-10) codes (DF:41270), death registry for primary and secondary cause of death (DF:40001, DF:40002), and “Date (ICD-10 code) first reported” (K85 = DF:131682 and K86 = DF:131684). As the “Date K86 first reported” field included non-CP-related diagnostic codes, and self-reported pancreatitis did not differentiate between AP and CP, our confirmed pancreatitis groups consisted of only ICD-9, ICD-10, and death certificate data. No ICD-10 code exists for RAP, but subjects with multiple AP codes across ICD-9 and ICD-10 were considered as RAP. A random sample of 24,000 UKBB subjects was chosen to act as a control group in a roughly 6:1 ratio of controls to the total number of all cases providing 80% power to detect a small effect size (Cohen d = 0.2) for the smaller case group (CP) at a genome wide significance level of P < 5e-8. The only exclusion criterion for the controls was absence from the “all possible pancreatitis” group described above. These controls were neither age nor sex matched because age is a TIGAR-O V2 risk factor. The list of all ICD-9, ICD-10, and medication codes used for curation of data can be found in Supplemental Table S1 (see Supplementary Digital Content 1, http://links.lww.com/CTG/A747).
Figure 1.
Study design and subject selection criteria. Step 1 (a) identifying all acute and chronic pancreatitis subjects from 6 separate data fields: 1.1 ICD-9 hospitalization data, 1.2 ICD-10 hospitalization data, 1.3 death records (primary and secondary), 1.4 self-reported, 1.5 date of first diagnosis K85, and 1.6 date of first diagnosis K86. All possible pancreatitis subjects are combined in 1.7. Step 2 (b) combining the list of analysis subjects using 2.1 all confirmed subjects with CP from the results of 1.1, 1.2, and 1.3 in step 1; 2.2 all confirmed subjects with AP from 1.1, 1.2, and 1.3 but excluding all subjects with CP from 2.1; and a random sample of controls excluding all possible pancreatitis identified in 1.7. AP, acute pancreatitis; CP, chronic pancreatitis; ICD-9 and ICD-10, International Classification of Disease 9th Revision and 10th Revision.
TIGAR-O classification
Each of the risk factors listed in TIGAR-O V2 were tabulated for cases and controls to the closest approximation allowed by the structure of the genetic and phenotypic data of the UKBB. The TIGAR-O system was designed for etiology-based classification of chronic pancreatitis, for both clinical and research purposes (8). Some risk factors, such as alcohol abuse, were richly characterized in the UKBB, whereas others, such as AP severity, were not captured at all. In addition, a curated list of common comorbidities is embedded within the TIGAR-O V2. These included celiac disease, cystic fibrosis (CF), gallstones, hypercalcemia, hyperlipidemia, hypertriglyceridemia, inflammatory bowel disease (IBD)—separated as Crohn's disease and ulcerative colitis—and pancreatic cancer.
Comorbidity analysis data selection
ICD-10 comorbidities for the AP-only, CP and control populations were tabulated and compared across all available diagnostic codes. Comorbidity data were collected from the UKBB data field 41270 (Diagnoses ICD-10), which comprises up to 213 codes per person. The complete list of ICD-10 codes can be found on the UKBB data showcase (https://bit.ly/3qgT4lu).
Statistical analysis
Descriptive analyses were presented as counts and proportions for categorical data and mean ± SD or median and interquartile range for continuous data. Univariate comparisons were performed using the Fisher exact test, or logistic regression for categorical data and the Mann-Whitney-U or Kruskal-Wallis test for continuous data. To adjust for multiple comparisons, the Bonferroni procedure was adopted for calculating adjusted P-values. Two-tailed P-values < 0.05 after correction for multiple comparisons were considered statistically significant.
TIGAR-O V2 risk factors and pancreatitis comorbidities were tested for statistical significance in pancreatitis (AP-only or CP) cases vs controls using the Fisher exact test. Odds ratios (OR) were computed using unconditional maximum likelihood estimation, with confidence intervals using normal approximation with the R epitools package and built-in Fisher test function. All data analysis and visualization were performed using the R Project software (Version 4.0.3).
RESULTS
Participant demographic in the UKBB
Of the 6,625 possible pancreatitis subjects identified, 4,256 had a confirmed pancreatitis (AP or CP) diagnosis. A total of 3,758 subjects had at least one AP attack; 447 of these had more than a single AP diagnosis and 529 also had a CP diagnosis. After excluding subjects with both AP and CP codes, 3,229 AP-only, 1,027 CP, and 24,000 control subjects were used for this analysis.
Pancreatitis cases in the UKBB skew slightly older than those sampled for the control population with a median age of 73 vs 70 years (Table 1). There is a significantly higher proportion of men in the CP population (66.4%) vs the control population (45.3%), but not for the AP-only group (45.9%). Most UKBB participants have European ancestry (White, British, English, any other White background; 94.8% cases, 94.2% controls), with the next-most common ancestry being Asian (Indian, Pakistani, Bangladeshi, any other Asian background; 2.1% cases, 1.9% controls), followed by Black (Caribbean, African, any other Black background; 1.2% cases, 1.5% controls). There were no significant differences in ancestry between groups.
Table 1.
Demographic information of controls, pancreatitis cases, and cases with AP, CP in particular, as determined by ICD-10 code
| Variable | Level | Control (N = 24000) | AP only (N = 3,229) | CP (N = 1,027) | P-value |
| Age (yr) | Median {IQR} | 70 {62, 75} | 73 {66, 77} | 72 {65, 77} | <1e-04 |
| Sex | Male (%) | 10,876 (45.3) | 1,483 (45.9) | 682 (66.4) | <1e-04 |
| Ethnicity | White | 22,611 (94.2) | 3,068 (95.0) | 967 (94.2) | |
| Other | 354 (1.5) | 42 (1.3) | 15 (1.5) | ||
| Asian | 461 (1.9) | 69 (2.1) | 21 (2.0) | ||
| Black | 366 (1.5) | 31 (1.0) | 18 (1.8) | ||
| Chinese | 64 (0.3) | 4 (0.1) | 3 (0.3) | ||
| Mixed | 144 (0.6) | 15 (0.5) | 3 (0.3) | 0.22 |
P-values in bold meet or exceed the pre-determined Bonferonni cutoff value.
AP, acute pancreatitis; CP, chronic pancreatitis; ICD-10, International Classification of Disease 10th Revision; IQR, interquartile range.
Ethnic grouping is shown as asked by the initial Assessment Centre visit questionnaire. White includes British and Irish; Black includes Caribbean and African; Asian includes Indian, Pakistani, and Bangladeshi. Age compared using Kruskal-Wallis test, sex, mortality status, and ethnicity using χ2 test.
Of the 3,229 AP-only cases, 0.8% have the ICD-10 code for idiopathic AP (n = 25), 22.7% have biliary AP (n = 732), 2.1% have alcoholic AP (n = 69), 0.7% have drug-induced AP (n = 22), 2.7% have other AP (n = 88), and 34.6% have unspecified AP (n = 1,116). The remaining AP cases (n = 1,177, 36.5%) only had the parent K85 code and no subcodes to denote etiology. More than 99% of AP cases were late onset (≥35 years of age at AP onset). Of the AP cases, 447 (13.8%) had ICD-10 codes showing more than one AP attack. Of these, 163 (36.4%) also had an ICD-10 code for CP, leaving 283 (63.6%) remaining in the AP-only group.
The 1,027 CP cases were further separated by ICD-10 code as alcohol-induced CP (n = 233) or other CP (n = 794), making up 23% and 77% of CP cases, respectively. Many subjects with CP had comorbid AP diagnoses (529 [51.5%] CP + AP vs 498 [48.5%] CP alone) and 98.6% of CP cases were late onset (>35 years of age at CP onset).
Observing other pancreatic disorders in the UKBB, 8.01% of UKBB participants have ICD-10 codes for diabetes mellitus (E08-E13, n = 40,252), 7.03% have T2D codes with no T1D code (n = 35,311), and 1% have T1D codes (n = 4,792). More than 3 million individuals in the United Kingdom are diagnosed with diabetes, resulting in a prevalence in excess of 6% with most cases being T2D (29). As of January 2021, there have been 1,829 (0.36%) subjects diagnosed with pancreatic cancer. CF is rare in this adult population (>40 years of age), with only 139 participants having CF codes (see Supplemental Table S2, see Supplementary Digital Content 2, http://links.lww.com/CTG/A748).
Pancreatitis prevalence and incidence in the UKBB
Of the initial 502,485 UKBB participants, 469,092 were living at the time of analysis. AP incidence from 2000 to 2020 increased from 21.4/yr per 100,000 in 2001–2005 to 48.2/yr per 100,000 for 2016–2020. Confirmed CP in UKBB has a prevalence of 163 per 100,000 (0.163%; n = 764), and the incidence increased from 5.9/yr per 100,000 to 12.8/yr per 100,000 in the same time intervals as seen in Table 2.
Table 2.
Mean incidence of AP and CP over 5-yr intervals for the previous 2 decades
| Date range | Mean AP incidence/100,000 | Mean age of first AP DX | Mean CP incidence/100,000 | Mean age of first CP DX |
| 2001–2005 | 21.4/yr | 53.6 | 5.9/yr | 50.9 |
| 2006–2010 | 28.7/yr | 58.4 | 8.7/yr | 57.0 |
| 2011–2015 | 40.4/yr | 63.2 | 9.5/yr | 63.3 |
| 2016–2020 | 48.2/yr | 67.9 | 12.8/yr | 68.8 |
P-values in bold meet or exceed the pre-determined Bonferonni cutoff value.
AP, acute pancreatitis; CP, chronic pancreatitis; DX, diagnosis.
TIGAR-O risk factors
In Tables 3 and 5, there are 24 statistical tests against TIGAR-O V2 risk factors (12 AP-only and 12 CP) along with 20 predefined comorbidities in Tables 4 and 6 (10 AP-only and 10 CP), giving a Bonferroni critical value for significance of 0.05/50 = 0.001.
Table 3.
TIGAR-O V2 risk and etiology factors in the UK Biobank between acute pancreatitis cases and controls compared using univariate logistic regression
| Variable | Control (N = 24000) | AP only (N = 3,229) | OR (95% CI) | P (Wald test) | P (LR- test) |
| Average daily drink REF. = 0–1 drinks/d |
14,095 (58.7) | 2,226 (68.9) | <0.001 | ||
| 1–2 drinks/d | 6,568 (27.4) | 653 (20.2) | 0.63 (0.57,0.69) | <0.001 | |
| 3–4 drinks/d | 2,587 (10.8) | 245 (7.6) | 0.6 (0.52,0.69) | <0.001 | |
| 5+ drinks/d | 750 (3.1) | 105 (3.3) | 0.89 (0.72,1.09) | 0.259 | |
| Smoking status REF. = never |
13,090 (54.9) | 1,592 (49.8) | <0.001 | ||
| Former | 8,236 (34.5) | 1,226 (38.4) | 1.22 (1.13,1.33) | <0.001 | |
| Current | 2,516 (10.6) | 377 (11.8) | 1.23 (1.09,1.39) | <0.001 | |
| Calcium >3 | 2 (0.0) | 3 (0.1) | 11.17 (1.87,66.87) | 0.008 | 0.01 |
| Triglycerides mg/dL REF. = (0, 199) |
17,297 (77.3) | 2,088 (69.7) | <0.001 | ||
| (199, 500) | 4,919 (22.0) | 864 (28.9) | 1.46 (1.34,1.58) | <0.001 | |
| (500, 1E+03) | 168 (0.8) | 42 (1.4) | 2.07 (1.47.2.91) | <0.001 | |
| At risk medication | 90 (0.4) | 34 (1.1) | 2.83 (1.9.4.2) | <0.001 | <0.001 |
| Chronic kidney disease | 841 (3.5) | 357 (11.1) | 3.42 (3.01.3.9) | <0.001 | <0.001 |
| On dialysis | 51 (0.2) | 26 (0.8) | 3.81 (2.37.6.12) | <0.001 | <0.001 |
| Renal transplant | 19 (0.1) | 6 (0.2) | 2.35 (0.94.5.89) | 0.068 | 0.092 |
| Chemotherapy | 1800 (7.5) | 424 (13.1) | 1.86 (1.67.2.09) | <0.001 | <0.001 |
| Any diabetes mellitus | 2073 (8.6) | 652 (20.2) | 2.68 (2.43.2.95) | <0.001 | <0.001 |
| BMI: REF. = (17, 9.25) | 7,848 (32.9) | 611 (19.1) | <0.001 | ||
| (10, 17.9) | 59 (0.2) | 10 (0.3) | 2.18 (1.11.4.28) | 0.024 | |
| (25, 30) | 10144 (42.5) | 1,284 (40.1) | 1.63 (1.47.1.8) | <0.001 | |
| (30, 80) | 5,792 (24.3) | 1,299 (40.5) | 2.88 (2.6.3.19) | <0.001 | |
| Congenital malformation of the pancreas | 0 (0.0) | 1 (0.0) | NS | 0.916 | 0.039 |
P-values in bold meet or exceed the pre-determined Bonferonni cutoff value.
AP, acute pancreatitis; CI, confidence interval; CP, chronic pancreatitis; LR, likelihood ratio; OR, odds ratio.
TIGAR-O V2 risk and etiology factors in the UK Biobank between AP only cases and controls (random sampling of non-AP and non-CP UK Biobank participants) compared using univariate logistic regression. Likelihood ratio test P-value presented for the significance test of the variable as a whole. Wald test P-value presented for significance test from baseline. The Bonferroni corrected cutoff value for significance is P <0.001.
Table 5.
TIGAR-O V2 risk and etiology factors in the UK Biobank between CP cases and controls compared using univariate logistic regression
| Variable | Control (N = 24000) | CP (N = 1,027) | OR (95% CI) | P (Wald test) | P (LR-test) |
| Average daily drink: REF. = 0–1 drinks/d |
14095 (58.7) | 642 (62.5) | <0.001 | ||
| 1-2 drinks/d | 6,568 (27.4) | 179 (17.4) | 0.6 (0.51, 0.71) | <0.001 | |
| 3-4 drinks/d | 2,587 (10.8) | 120 (11.7) | 1.02 (0.83, 1.24) | 0.858 | |
| 5 + drinks/d | 750 (3.1) | 86 (8.4) | 2.52 (1.99, 3.19) | <0.001 | |
| Smoking status REF. = Never |
13,090 (54.9) | 348 (34.1) | <0.001 | ||
| Former | 8,236 (34.5) | 366 (35.8) | 1.67 (1.44, 1.94) | <0.001 | |
| Current | 2,516 (10.6) | 308 (30.1) | 4.6 (3.93, 5.4) | <0.001 | |
| Calcium >3 | 2 (0.0) | 0 (0.0) | NS | 0.952 | 0.687 |
| Triglycerides mg/dL: REF. =(0, 199) | 17,297 (77.3) | 619 (67.5) | <0.001 | ||
| (199, 500) | 4,919 (22.0) | 278 (30.3) | 1.58 (1.37, 1.83) | <0.001 | |
| (500, 1E+03) | 168 (0.8) | 20 (2.2) | 3.33 (2.08, 5.33) | <0.001 | |
| At risk medication | 90 (0.4) | 16 (1.6) | 4.2 (2.46, 7.18) | <0.001 | <0.001 |
| Chronic kidney disease | 841 (3.5) | 147 (14.3) | 4.6 (3.81, 5.55) | <0.001 | <0.001 |
| On dialysis | 51 (0.2) | 23 (2.2) | 10.76 (6.55, 17.67) | <0.001 | <0.001 |
| Renal transplant | 19 (0.1) | 5 (0.5) | 6.17 (2.3, 16.57) | <0.001 | 0.003 |
| On chemotherapy | 1800 (7.5) | 155 (15.1) | 2.19 (1.84, 2.62) | <0.001 | <0.001 |
| Any diabetes mellitus | 2073 (8.6) | 426 (41.5) | 7.5 (6.57, 8.56) | <0.001 | <0.001 |
| BMI: REF. = (17, 9.25) | 7,848 (32.9) | 321 (31.8) | <0.001 | ||
| (10, 17.9) | 59 (0.2) | 16 (1.6) | 6.63 (3.77, 11.65) | <0.001 | |
| (25, 30) | 10144 (42.5) | 375 (37.2) | 0.9 (0.78, 1.05) | 0.192 | |
| (30, 80) | 5,792 (24.3) | 297 (29.4) | 1.25 (1.07, 1.47) | 0.006 | |
| Congenital malformation of the pancreas | 0 (0.0) | 6 (0.6) | a | 0.906 | <0.001 |
P-values in bold meet or exceed the pre-determined Bonferonni cutoff value.
Non-computable value due to division by zero.
AP, acute pancreatitis; CI, confidence interval; CP, chronic pancreatitis; LR, likelihood ratio; OR, odds ratio.
TIGAR-O V2 risk and etiology factors in the UK Biobank between CP and controls (random sampling of non-AP and non-CP UK Biobank participants) compared using univariate logistic regression. Likelihood ratio (LR) tests P-value presented for the significance test of the variable as a whole. The Wald test P-value presented for significance test from baseline. The Bonferroni corrected cutoff value for significance is P < 0.001.
Table 4.
Curated list of common comorbidities in the UK Biobank between acute pancreatitis-only cases and controls compared using univariate logistic regression
| Variable | Control (N = 24000) | AP only (N = 3,229) | OR (95% CI) | P (Wald test) | P (LR-test) |
| Gall stones | 1,061 (4.4) | 1917 (59.4) | 31.85 (29.01.34.97) | <0.001 | <0.001 |
| Hyperlipidemia | 3,380 (14.1) | 909 (28.2) | 2.39 (2.2.2.6) | <0.001 | <0.001 |
| Hypertrygliceridemia | 6 (0.0) | 2 (0.1) | 2.48 (0.5.12.28) | 0.266 | 0.306 |
| Hypercalcemia | 160 (0.7) | 84 (2.6) | 3.98 (3.05.5.2) | <0.001 | <0.001 |
| Pancreatic cancer | 49 (0.2) | 69 (2.1) | 10.67 (7.39.15.42) | <0.001 | <0.001 |
| CF | 8 (0.0) | 2 (0.1) | 1.86 (0.39.8.76) | 0.433 | 0.462 |
| Celiac | 256 (1.1) | 59 (1.8) | 1.73 (1.3.2.3) | <0.001 | <0.001 |
| Crohn’s disease | 167 (0.7) | 58 (1.8) | 2.61 (1.93.3.53) | <0.001 | <0.001 |
| Ulcerative colitis | 312 (1.3) | 68 (2.1) | 1.63 (1.25.2.13) | <0.001 | <0.001 |
| Alcohol drinker status: REF. = never | 965 (4.0) | 214 (6.7) | <0.001 | ||
| Former | 869 (3.6) | 231 (7.2) | 1.2 (0.97.1.47) | 0.087 | |
| Current | 22,082 (92.3) | 2,765 (86.1) | 0.56 (0.48.0.66) | <0.001 |
P-values in bold meet or exceed the pre-determined Bonferonni cutoff value.
AP, acute pancreatitis; CI, confidence interval; CP, chronic pancreatitis; LR, likelihood ratio; OR, odds ratio.
Curated list of common comorbidities in the UK Biobank between AP-only cases and controls (random sampling of non-AP and non-CP UK Biobank participants) compared using univariate logistic regression. Likelihood ratio test P-value presented for the significance test of the variable as a whole. The Wald test P-value presented for significance test from baseline. The Bonferroni corrected cutoff value for significance is P <0.001.
Table 6.
Curated list of common comorbidities in the UK Biobank between CP cases and controls compared using univariate logistic regression
| Variable | Control (N = 24,000) | CP (N = 1,027) | OR (95% CI) | P (Wald test) | P (LR-test) |
| Gallstones | 1,061 (4.4) | 333 (32.4) | 10.45 (9.05, 12.07) | <0.001 | <0.001 |
| Hyperlipidemia | 3,380 (14.1) | 356 (34.7) | 3.24 (2.83, 3.7) | <0.001 | <0.001 |
| Hypertriglyceridemia | 6 (0.0) | 6 (0.6) | 23.5 (7.57, 72.99) | <0.001 | <0.001 |
| Hypercalcemia | 160 (0.7) | 43 (4.2) | 6.51 (4.62, 9.17) | <0.001 | <0.001 |
| Pancreatic cancer | 49 (0.2) | 48 (4.7) | 23.97 (16.01, 35.86) | <0.001 | <0.001 |
| CF | 8 (0.0) | 1 (0.1) | 2.92 (0.37, 23.39) | 0.312 | 0.378 |
| Celiac | 256 (1.1) | 48 (4.7) | 4.55 (3.32, 6.23) | <0.001 | <0.001 |
| Crohn's disease | 167 (0.7) | 12 (1.2) | 1.69 (0.94, 3.04) | 0.082 | 0.106 |
| Ulcerative colitis | 312 (1.3) | 32 (3.1) | 2.44 (1.69, 3.53) | <0.001 | <0.001 |
| Alcohol drinker status: REF. = never | 965 (4.0) | 52 (5.1) | <0.001 | ||
| Former | 869 (3.6) | 176 (17.4) | 3.76 (2.72, 5.19) | <0.001 | |
| Current | 22,082 (92.3) | 785 (77.5) | 0.66 (0.49, 0.88) | 0.005 |
P-values in bold meet or exceed the pre-determined Bonferonni cutoff value.
AP, acute pancreatitis; CI, confidence interval; CP, chronic pancreatitis; LR, likelihood ratio; OR, odds ratio.
Curated list of common comorbidities in the UK Biobank between CP cases and controls (random sampling of non-AP and non-CP UK Biobank participants) compared using univariate logistic regression. Likelihood ratio tests P-value presented for the significance test of the variable as a whole. The Wald test P-value presented for significance test from baseline. The Bonferroni corrected cutoff value for significance is P < 0.001.
Compared with controls (Table 3), patients in the AP-only group were more likely to be a current smoker; have higher nonfasting triglycerides; be on at-risk medications, chemotherapy, or dialysis; and have body mass index designated obesity or diabetes mellitus. Alcohol consumption does not seem to be associated with AP in this cohort because the number of self-reported light drinkers is significantly higher in patients compared with controls, although drinking categories at the time of the AP event is not known. Likewise, there is no significant difference between AP-only patients and controls in calcium levels >3 mmol/L, renal transplant status, nor congenital malformations of the pancreas.
Compared with controls (Table 5), subjects with CP show a similar pattern to AP but with some important differences. Similar to the AP-only group, subjects with CP are more likely to be current smokers; have higher nonfasting triglycerides; be on at-risk factor medications, chemotherapy, or dialysis; and have a diagnosis for diabetes mellitus. In each of these cases, the OR is much higher in CP vs controls, compared with the AP-only group with the most striking being that patients with CP are much more likely to be current smokers (OR = 4.6 [3.93, 5.4] in CP, OR = 1.23 [1.09, 1.39] in AP-only) and more likely to have been diagnosed with diabetes mellitus than patients with AP (OR = 7.5 [6.57, 8.5]) in CP, OR = 2.68 [2.43, 2.95] in AP-only). This is consistent with the known risk of developing DM after AP (30,31). Unlike in the AP-only group, subjects with CP are more likely to consume 5 or more drinks per day and are significantly more likely to be former drinkers. Although significant in CP, the OR for congenital malformations of the pancreas could not be calculated because of the rarity of studies necessary to make the diagnosis in controls. Obesity is not significantly associated in the CP population after Bonferroni correction, whereas this association is much stronger in the AP population (compare Tables 3 and 5). As is the case in AP-only, there is no significant difference between patients with CP and controls in calcium levels >3 mmol/L or renal transplant status.
From a curated list of 9 common comorbidities taken from the TIGAR-O V2 checklist, CF and hypertriglyceridemia are rarely diagnosed in the UKBB cohort as seen in Tables 4 and 6. Hypertriglyceridemia was significantly associated with CP but not the AP only group. Gallstones were a significant comorbidity in both AP only and CP, but with a stronger association in AP (AP only OR = 31.85 [29.01, 34.97], CP OR = 10.45 [9.05, 12.07]). Hyperlipidemia, hypercalcemia, celiac disease, ulcerative colitis, and pancreatic cancer all showed a significant increase in prevalence from controls to AP only and an additional increase in CP. Crohn's disease was the only comorbidity more common in AP only than in CP.
Pancreatitis comorbidities
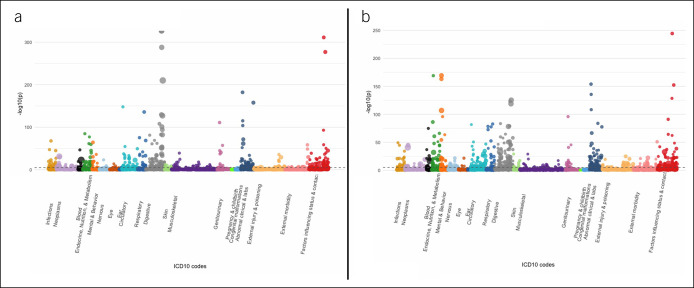
Many significant comorbidities were observed in pancreatitis cases with high OR compared with the control population. The scope of ICD-10 codes is broad, with some codes specifying disease and others specifying a myriad of signs, symptoms, tests, and external causes of injury or disease. With 5,124 ICD-10 codes available in the data set, rounding up to 5,500 comparisons gives a Bonferroni critical value for significance of 0.05/5,500 = 9.09e-6. For simplicity, using a cutoff value of 9e-6, 447 significant associations with AP and 547 significant associations with CP were identified. Of these, 75 are unique to the AP only group, 175 are unique to the CP group, and 372 are held in common. See Tables 7 and 8 for the top 25 most significant comorbidities by ICD-10 code for AP only and CP; see Supplemental Tables S3 and S4 (Supplementary Digital Contents 3 and 4, http://links.lww.com/CTG/A749 and http://links.lww.com/CTG/A750) for the full listings of all significant comorbidities. A visual representation of the comorbidities is shown in Figure 2.
Table 7.
Top 25 AP comorbidities by significance, excluding all other K85 and K86 ICD-10 diagnoses
| Code | Name | ICD-10 block | ICD-10 chapter | AP+ | AP− | Control+ | Control− | P-value | OR | Lower | Upper |
| K801 | K80.1 calculus of gallbladder with other cholecystitis | K80-K87 disorders of gallbladder, biliary tract and pancreas | Diseases of the digestive system | 774 | 2,455 | 379 | 23,621 | 0 | 19.65 | 17.26 | 22.37 |
| K802 | K80.2 calculus of gallbladder without cholecystitis | K80-K87 disorders of gallbladder, biliary tract and pancreas | Diseases of the digestive system | 1,331 | 1,898 | 630 | 23,370 | 0 | 26.01 | 23.40 | 28.91 |
| K805 | K80.5 calculus of bile duct without cholangitis or cholecystitis | K80-K87 disorders of gallbladder, biliary tract and pancreas | Diseases of the digestive system | 475 | 2,754 | 198 | 23,802 | 1.94E-288 | 20.73 | 17.48 | 24.59 |
| Z904 | Z90.4 acquired absence of other parts of digestive tract | Z80-Z99 persons with potential health hazards related to family and personal history and certain conditions influencing health status | Factors influencing health status and contact with health services | 721 | 2,508 | 818 | 23,182 | 1.39E-277 | 8.15 | 7.31 | 9.08 |
| K838 | K83.8 other specified diseases of biliary tract | K80-K87 disorders of gallbladder, biliary tract and pancreas | Diseases of the digestive system | 299 | 2,930 | 69 | 23,931 | 1.18E-210 | 35.39 | 27.17 | 46.11 |
| R101 | R10.1 pain localised to upper abdomen | R10-R19 symptoms and signs involving the digestive system and abdomen | Symptoms, signs and abnormal clinical and laboratory findings, not elsewhere classified | 584 | 2,645 | 852 | 23,148 | 9.33E-183 | 6.00 | 5.36 | 6.71 |
| R945 | R94.5 abnormal results of liver function studies | R90-R94 abnormal findings on diagnostic imaging and in function studies, without diagnosis | Symptoms, signs and abnormal clinical and laboratory findings, not elsewhere classified | 394 | 2,835 | 395 | 23,605 | 1.84E-158 | 8.31 | 7.18 | 9.60 |
| I10 | I10 essential (primary) hypertension | I10-I15 hypertensive diseases | Diseases of the circulatory system | 1,672 | 1,557 | 6,824 | 17,176 | 1.30E-148 | 2.70 | 2.51 | 2.91 |
| J90 | J90 pleural effusion, not elsewhere classified | J90-J94 other diseases of pleura | Diseases of the respiratory system | 452 | 2,777 | 675 | 23,325 | 1.80E-136 | 5.62 | 4.96 | 6.38 |
| K800 | K80.0 calculus of gallbladder with acute cholecystitis | K80-K87 disorders of gallbladder, biliary tract and pancreas | Diseases of the digestive system | 228 | 3,001 | 108 | 23,892 | 3.29E-130 | 16.81 | 13.33 | 21.20 |
| K831 | K83.1 obstruction of bile duct | K80-K87 disorders of gallbladder, biliary tract and pancreas | Diseases of the digestive system | 196 | 3,033 | 61 | 23,939 | 9.68E-128 | 25.36 | 18.98 | 33.89 |
| R104 | R10.4 other and unspecified abdominal pain | R10-R19 symptoms and signs involving the digestive system and abdomen | Symptoms, signs and abnormal clinical and laboratory findings, not elsewhere classified | 588 | 2,641 | 1,344 | 22,656 | 9.13E-116 | 3.75 | 3.38 | 4.17 |
| N179 | N17.9 acute renal failure, unspecified | N17-N19 renal failure | Diseases of the genitourinary system | 483 | 2,746 | 952 | 23,048 | 1.03E-111 | 4.26 | 3.79 | 4.78 |
| K660 | K66.0 peritoneal adhesions | K65-K67 diseases of peritoneum | Diseases of the digestive system | 306 | 2,923 | 365 | 23,635 | 9.78E-109 | 6.78 | 5.80 | 7.93 |
| R11 | R11 nausea and vomiting | R10-R19 symptoms and signs involving the digestive system and abdomen | Symptoms, signs and abnormal clinical and laboratory findings, not elsewhere classified | 510 | 2,719 | 1,118 | 22,882 | 1.28E-104 | 3.84 | 3.43 | 4.29 |
| K811 | K81.1 chronic cholecystitis | K80-K87 disorders of gallbladder, biliary tract and pancreas | Diseases of the digestive system | 177 | 3,052 | 82 | 23,918 | 8.36E-102 | 16.92 | 12.98 | 22.04 |
| K828 | K82.8 other specified diseases of gallbladder | K80-K87 disorders of gallbladder, biliary tract and pancreas | Diseases of the digestive system | 174 | 3,055 | 93 | 23,907 | 8.07E-95 | 14.64 | 11.35 | 18.89 |
| Z864 | Z86.4 personal history of psychoactive substance abuse | Z80-Z99 persons with potential health hazards related to family and personal history and certain conditions influencing health status | Factors influencing health status and contact with health services | 910 | 2,319 | 3,195 | 20,805 | 1.13E-93 | 2.56 | 2.35 | 2.78 |
| E119 | E11.9 without complications | E10-E14 diabetes mellitus | Endocrine, nutritional and metabolic diseases | 610 | 2,619 | 1,764 | 22,236 | 1.66E-85 | 2.94 | 2.66 | 3.25 |
| K760 | K76.0 fatty (change of) liver, not elsewhere classified | K70-K77 diseases of liver | Diseases of the digestive system | 223 | 3,006 | 246 | 23,754 | 2.50E-83 | 7.16 | 5.95 | 8.62 |
| K830 | K83.0 cholangitis | K80-K87 disorders of gallbladder, biliary tract and pancreas | Diseases of the digestive system | 140 | 3,089 | 60 | 23,940 | 8.06E-83 | 18.08 | 13.33 | 24.53 |
| K219 | K21.9 gastro-oesophageal reflux disease without oesophagitis | K20-K31 diseases of oesophagus, stomach and duodenum | Diseases of the digestive system | 620 | 2,609 | 1,855 | 22,145 | 5.40E-82 | 2.84 | 2.57 | 3.13 |
| E780 | E78.0 pure hypercholesterolaemia | E70-E90 metabolic disorders | Endocrine, nutritional and metabolic diseases | 849 | 2,380 | 3,119 | 20,881 | 6.87E-78 | 2.39 | 2.19 | 2.61 |
| J181 | J18.1 lobar pneumonia, unspecified | J09-J18 Influenza and pneumonia | Diseases of the respiratory system | 349 | 2,880 | 715 | 23,285 | 3.72E-76 | 3.95 | 3.45 | 4.51 |
| R18 | R18 ascites | R10-R19 symptoms and signs involving the digestive system and abdomen | Symptoms, signs and abnormal clinical and laboratory findings, not elsewhere classified | 177 | 3,052 | 170 | 23,830 | 2.93E-72 | 8.13 | 6.56 | 10.07 |
P-values in bold meet or exceed the pre-determined Bonferonni cutoff value.
AP, acute pancreatitis; ICD-10, International Classification of Disease 10th Revision; OR, odds ratio.
Significance and OR calculated using the Fisher exact test.
Table 8.
Top 25 CP comorbidities by significance, excluding all other K85 and K86 ICD-10 diagnoses
| Code | Name | ICD-10 block | ICD-10 chapter | CP+ | CP− | Control+ | Control− | P-value | OR | Lower | Upper |
| Z871 | Z87.1 personal history of diseases of the digestive system | Z80-Z99 persons with potential health hazards related to family and personal history and certain conditions influencing health status | Factors influencing health status and contact with health services | 473 | 554 | 1,610 | 22,390 | 4.69E-245 | 11.87 | 10.40 | 13.56 |
| F102 | F10.2 Dependence syndrome (alcohol) | F10-F19 Mental and behavioural disorders due to psychoactive substance use | Mental and behavioural disorders | 189 | 838 | 159 | 23,841 | 4.37E-170 | 33.82 | 27.09 | 42.22 |
| E119 | E11.9 without complications | E10-E14 diabetes mellitus | Endocrine, nutritional and metabolic diseases | 405 | 622 | 1,764 | 22,236 | 9.77E-170 | 8.21 | 7.18 | 9.39 |
| F101 | F10.1 harmful use (alcohol) | F10-F19 Mental and behavioural disorders due to psychoactive substance use | Mental and behavioural disorders | 206 | 821 | 252 | 23,748 | 7.00E-164 | 23.65 | 19.42 | 28.79 |
| R101 | R10.1 pain localised to upper abdomen | R10-R19 symptoms and signs involving the digestive system and abdomen | Symptoms, signs and abnormal clinical and laboratory findings, not elsewhere classified | 291 | 736 | 852 | 23,148 | 1.17E-154 | 10.74 | 9.23 | 12.51 |
| Z904 | Z90.4 acquired absence of other parts of digestive tract | Z80-Z99 persons with potential health hazards related to family and personal history and certain conditions influencing health status | Factors influencing health status and contact with health services | 285 | 742 | 818 | 23,182 | 4.72E-153 | 10.89 | 9.34 | 12.69 |
| R104 | R10.4 other and unspecified abdominal pain | R10-R19 symptoms and signs involving the digestive system and abdomen | Symptoms, signs and abnormal clinical and laboratory findings, not elsewhere classified | 324 | 703 | 1,344 | 22,656 | 2.67E-136 | 7.77 | 6.74 | 8.96 |
| Z864 | Z86.4 personal history of psychoactive substance abuse | Z80-Z99 persons with potential health hazards related to family and personal history and certain conditions influencing health status | Factors influencing health status and contact with health services | 465 | 562 | 3,195 | 20,805 | 3.01E-129 | 5.39 | 4.74 | 6.13 |
| K838 | K83.8 other specified diseases of biliary tract | K80-K87 disorders of gallbladder, biliary tract and pancreas | Diseases of the digestive system | 126 | 901 | 69 | 23,931 | 4.25E-126 | 48.50 | 35.90 | 65.53 |
| K802 | K80.2 calculus of gallbladder without cholecystitis | K80-K87 disorders of gallbladder, biliary tract and pancreas | Diseases of the digestive system | 229 | 798 | 630 | 23,370 | 4.81E-124 | 10.65 | 9.01 | 12.58 |
| K831 | K83.1 obstruction of bile duct | K80-K87 disorders of gallbladder, biliary tract and pancreas | Diseases of the digestive system | 118 | 909 | 61 | 23,939 | 1.35E-119 | 50.94 | 37.14 | 69.88 |
| R11 | R11 nausea and vomiting | R10-R19 symptoms and signs involving the digestive system and abdomen | Symptoms, signs and abnormal clinical and laboratory findings, not elsewhere classified | 267 | 760 | 1,118 | 22,882 | 4.92E-109 | 7.19 | 6.18 | 8.37 |
| F103 | F10.3 Withdrawal state (alcohol) | F10-F19 Mental and behavioural disorders due to psychoactive substance use | Mental and behavioural disorders | 107 | 920 | 57 | 23,943 | 1.31E-107 | 48.85 | 35.19 | 67.82 |
| N179 | N17.9 acute renal failure, unspecified | N17-N19 renal failure | Diseases of the genitourinary system | 234 | 793 | 952 | 23,048 | 1.16E-96 | 7.14 | 6.09 | 8.38 |
| F171 | F17.1 harmful use | F10-F19 Mental and behavioural disorders due to psychoactive substance use | Mental and behavioural disorders | 249 | 778 | 1,101 | 22,899 | 1.42E-96 | 6.66 | 5.70 | 7.77 |
| Z720 | Z72.0 tobacco use | Z70-Z76 persons encountering health services in other circumstances | Factors influencing health status and contact with health services | 198 | 829 | 681 | 23,319 | 8.77E-92 | 8.18 | 6.88 | 9.72 |
| E109 | E10.9 without complications | E10-E14 diabetes mellitus | Endocrine, nutritional and metabolic diseases | 130 | 897 | 232 | 23,768 | 5.67E-87 | 14.85 | 11.86 | 18.59 |
| R634 | R63.4 abnormal weight loss | R50-R69 general symptoms and signs | Symptoms, signs and abnormal clinical and laboratory findings, not elsewhere classified | 182 | 845 | 620 | 23,380 | 1.39E-84 | 8.12 | 6.79 | 9.71 |
| K297 | K29.7 gastritis, unspecified | K20-K31 diseases of oesophagus, stomach and duodenum | Diseases of the digestive system | 248 | 779 | 1,273 | 22,727 | 3.93E-84 | 5.68 | 4.87 | 6.63 |
| J90 | J90 pleural effusion, not elsewhere classified | J90-J94 other diseases of pleura | Diseases of the respiratory system | 187 | 840 | 675 | 23,325 | 1.83E-83 | 7.69 | 6.45 | 9.17 |
| I10 | I10 essential (primary) hypertension | I10-I15 hypertensive diseases | Diseases of the circulatory system | 595 | 432 | 6,824 | 17,176 | 2.38E-82 | 3.47 | 3.05 | 3.94 |
| K760 | K76.0 fatty (change of) liver, not elsewhere classified | K70-K77 diseases of liver | Diseases of the digestive system | 125 | 902 | 246 | 23,754 | 1.07E-79 | 13.38 | 10.68 | 16.76 |
| J181 | J18.1 lobar pneumonia, unspecified | J09-J18 Influenza and pneumonia | Diseases of the respiratory system | 186 | 841 | 715 | 23,285 | 3.67E-79 | 7.20 | 6.04 | 8.58 |
| K709 | K70.9 alcoholic liver disease, unspecified | K70-K77 diseases of liver | Diseases of the digestive system | 79 | 948 | 43 | 23,957 | 5.05E-79 | 46.43 | 31.84 | 67.69 |
| R945 | R94.5 abnormal results of liver function studies | R90-R94 abnormal findings on diagnostic imaging and in function studies, without diagnosis | Symptoms, signs and abnormal clinical and laboratory findings, not elsewhere classified | 146 | 881 | 395 | 23,605 | 2.56E-78 | 9.90 | 8.10 | 12.11 |
AP, acute pancreatitis; ICD-10, International Classification of Disease 10th Revision; OR, odds ratio.
Significance and OR calculated using Fisher exact test.
Figure 2.
Manhattan plot of AP (a) and CP (b) comorbidities with other ICD-10 diagnoses by significance value. Each circle is scaled by the side of the odds ratio for that comorbidity. ICD-10 codes colored and labeled by ICD-10 chapter titles. Dashed line at Bonferroni corrected P-value of 9 × 10−6. AP, acute pancreatitis; CP, chronic pancreatitis; ICD-10, International Classification of Disease 10th Revision.
Of the comorbidities significantly associated with both AP and CP, many are also associated with the pancreas, including “other specified diseases of the pancreas,” abdominal pain, and pancreatic cancer. Some notable comorbidities include primary hypertension (I10), abnormal results of liver function studies (R94.5), type 2 diabetes mellitus (E11), alcohol and tobacco use (F10, Z72.1, F17, and Z72.0), calculus of the gallbladder (K80), gastritis (K29), removal of gallbladder (Z904), and mood disorders including depression (F32.9) and anxiety (F41.9).
Significantly associated comorbidities unique to AP-only are often risk factors for developing AP, such as disease of the gallbladder (K80 and K82), cancer of the biliary tract (C24), history of cancer (Z85), Crohn's disease (K50.9) (possibly linked to medications causing AP such as azathioprine or 6-mercaptopurine (32)), and obesity (E66.8). Significantly associated comorbidities unique to CP are often the results of CP diagnosis or chronic systemic inflammation including benign neoplasms of the colon (D12), insulin-dependent diabetes (E10), chronic kidney disease, chronic pain, and opioid abuse. CP also has a host of diagnoses related to alcohol dependency and abuse (F10).
DISCUSSION
The UKBB is an extremely large and well-curated data set with more than 502,000 adult participants (25–28). Using data from 4,256 patients with pancreatitis in the UKBB, we explored risk factors for pancreatitis and its subtypes AP and CP, presenting our findings within the TIGAR-O V2 framework (6). Our results reaffirm many existing reports on pancreatitis risk factors showing increased risk of pancreatitis, especially in patients who smoke cigarettes and consume more than 5 drinks of alcohol per day. However, it is the first comprehensive population-based study to systematically compare each of the independent risk factors in the TIGAR-O V2 list and evaluate their relative contribution to overall risk in both AP and CP.
AP, an acute event where most patients recover, is best measured in incidence, whereas CP, a continuous condition, may be measured in both incidence and prevalence. The National Institute for Health and Care Excellence (NICE) (2020) guideline for pancreatitis reports 56 AP cases per 100,000 people per year in the UK and 5 new CP cases per 100,000 people per year in Western Europe (33). Other studies show that the incidence of AP throughout Europe ranges from 4.6 up to 100 per 100,000 per year (34–38). Several of these studies have shown an increase in incidence with age similar to what we see in the UKBB. Our findings fall well into this range and show a very distinct increase in incidence from 21.4 to 48.2 per 100,000 per year coinciding with the overall aging of our cohort. This increase in incidence is likely a combination of detection bias and the cumulative environmental effects in an aging cohort. The difficulty of calculating incidence on a closed, older population is that the denominator of the incidence calculation shrinks with the mortality of the population, artificially inflating the raw incidence.
Prevalence of CP in Europe is shown to be between 16 and 154 per 100,000 (11, 39, 40). A recent study showed the prevalence of CP in the United States to be 73 per 100,000 with a peak of 135 per 100,000 for the 46–55-year age group (41). The UKBB data show a higher prevalence of 163 per 100,000 and a doubling in incidence from 5.9 to 12.8 per 100,000 per year similar to what we see in AP. The current CP prevalence and incidence in the UKBB presented here more closely matches the estimates from the recent Danish study with a prevalence of 154 and an incidence of 12.6 per 100,000 per year (11).
ICD-9 and ICD-10 codes in the UKBB are a poor surrogate of etiology in both AP and CP with 71.1% of AP listed as either “unspecified” or with no code at all and 77% of CP cases classified as “other.” The TIGAR-O V2 system was created to assess the heterogeneous risk factors contributing to the etiology of RAP and CP. Our TIGAR-O V2 analysis shows that nearly 62.4% of all subjects with AP in the UKBB could be classified as biliary AP, although only 22.7% have the corresponding ICD-10 code. In addition, 22% of subjects classified with the biliary AP ICD-10 code have no other gallstone-related codes in their medical history. Gallstones are considered a rare etiology for CP, yet we found 32.4% of subjects with CP to have a history of gallstones. Alcoholic AP, often the second most common etiology across many countries, may be highly underreported in the UKBB. Alcoholic CP is considered the most common etiology of CP but once again is underrepresented in this population with only 27% receiving an alcoholic CP ICD-10 code.
A diagnosis of hypertriglyceridemia, considered both an AP and CP etiology as well as a risk factor for severe AP, is extremely rare in the UKBB, although the number of subjects with nonfasting triglycerides >500 mg/dL is considerably higher in all categories. Autoimmune diseases such as IBD and celiac disease are enriched in both AP and CP groups and may make up as much as 6% of AP and 9% of CP etiologic risk. We also see higher than normal rates of DM, hyperlipidemia, chronic kidney disease, and pancreatic cancer in both AP and CP cohorts. Finally, it is only in CP where we see an increase in the number of current smokers.
The very high fraction of patients on chemotherapy in the CP group (15%) was unexpected, as was the prevalence of chemotherapy in the control group (7.5%). However, the median age of subjects with CP was 72 years and 70 years in control subjects. Cancer is an age-related disorder with the incidence of most cancers increasing rapidly beginning in middle age (42,43). The prevalence of all cancers in the general UK population is approximately 3.6% (44), which should be lower than the prevalence in the older population represented in the UKBB. Chemotherapy is a nonspecific identifier because ‘chemotherapeutic agents’ are used in a variety of inflammatory disorders, such as rheumatologic disease and IBD. There is also a higher prevalence of benign and malignant neoplastic conditions in the CP population compared with controls that may explain this finding. The etiology of this enrichment may be a result of underlying host factors, environmental exposures such as tobacco use, or chronic systemic inflammation. This finding and potential association require further investigation.
The extensive ICD-10 data set linked to the large, real-world cohort in the UKBB allows for an unbiased detection of conditions associated with AP and CP. Several conditions strongly associated with AP and CP are well-known risk factors, such as gallstones and alcohol use. An unexpected but strongly linked condition associated with both AP and CP was essential hypertension (I10). The mechanistic link between hypertension both AP (OR = 2.70 [2.51,2.91]) and CP (OR = 3.47 [3.05,3.94]) is not clear. Hypertension is a syndrome of multiple etiologies, and it is plausible that multiple factors contribute to both hypertension and pancreatitis, such as abnormal cholesterol and bile metabolism, smoking, and diet factors.
Most other conditions strongly associated with AP and CP are either symptoms (R10.X), treatments (Y83.6), complications (N17.9), or sequela (E10, E11) of the conditions. Recent studies from the NAPS2 (45,46) and multicenter CP pain study (47) cohorts demonstrated a strong association with depression and anxiety in patients with pancreatitis, especially those with pain. A striking but not surprising finding was the relative high risk of intentional self-harm (ICD-10 X60-X65 family; e.g. suicide attempts) in patients with CP (ORs 8–28) (see Table S4, Supplementary Digital Content 4, http://links.lww.com/CTG/A750). This is consistent with the findings of the very low quality of life in patients with CP (17,20) and desperation of patients with little hope of improvement.
Our study has a number of limitations. Despite the high-quality curation on behalf of the UKBB, we cannot rule out that there may be errors in the assignment of ICD codes to our cases. A meta-analysis of ICD codes for pancreatitis found a positive predictive value of 0.71 for all pancreatitis (48). Unfortunately, less than 30% of AP and CP cases in the UKBB have an etiology coded into their medical record, making it difficult to differentiate risk factors. The lack of a coded etiology could be an artifact of how ICD-10 codes are used in the UK NHS. In our comorbidity analysis, timing of the ICD-10 comorbidities has not been taken into consideration; therefore, we cannot assess causation.
We were not able to assess all aspects of the TIGAR-O V2 checklist because some items are either not collected or are not clearly differentiated. Specifically, genetic risks were difficult to compare because of differences in the genotyping array in the UKBB. Alcohol consumption surveys in the UKBB were taken at 2 time points and may not adequately reflect the influence of alcohol on the risks of AP or CP at the time of diagnosis. Mean patient age was higher in the UKBB (60–73 years old) than in the NAPS2 group (46–59 years old) (8) likely because of differing inclusion criteria as the UKBB enrolled patients between 40 and 69 years of age at recruitment (49), whereas the NAPS2 cohort was recruited from adult referral centers. The effect of the later age of enrollment and the prospective follow-up time may influence the low number of idiopathic pancreatitis and subjects with CF. Likewise, it may increase the number of significant age-related comorbidities and also be more heavily weighting AP toward biliary etiology.
The TIGAR-O risk checklist, including genetic risk variants, was originally created using the NAPS2 cohort. Similar to the UKBB, the NAPS2 cohort was primarily patients of White, European ancestry, although NAPS2 had a much higher percentage of self-identified African ancestry subjects (5–11% NAPS2 vs 1.1–1.7% UKBB). These factors limit the generalizability of the data to more diverse populations as others have shown that genetic variants associated with pancreatitis in Europeans are rare in those with African ancestry (50).
Future directions include an intensive genomic analysis to further characterize this population. Such an analysis may aid in understanding and identifying risk factors associated with disease onset, progression, and disease severity not seen in the comorbidity analysis. Additional studies may include more detailed examination of the various comorbidities and their interactions. The analysis presented here serves as the foundation for future investigations into the complex syndrome of pancreatitis using this well-described cohort.
CONFLICTS OF INTEREST
Guarantor of the article: David C. Whitcomb MD, PhD.
Specific author contributions: P.G., B.B., and M.H. were involved in planning and conducting the study. D.S. and P.G. collected the data. D.S., P.G., and M.E. completed the data analysis. All authors contributed to data interpretation. D.S., P.G., S.M., C.B., M.H., and M.E. created the initial draft of the manuscript. All authors participated in manuscript revision and editing, and reviewed and approved the final draft of the manuscript submitted for review.
Financial support: No external funding sources were used for this analysis and manuscript preparation.
Potential competing interests: P.G. and C.S.O. are employees of Ariel Precision Medicine. M.E. and C.B. are consultants to Ariel Precision Medicine. D.C.W. is a cofounder of Ariel Precision Medicine and a chief scientific officer. B.B. is an employee of DNAnexus. D.S. and M.H. are former employees of Ariel Precision Medicine.
Study Highlights.
WHAT IS KNOWN
✓ Pancreatitis is a complex syndrome with multiple etiology and comorbidities.
✓ Pancreatitis epidemiologic data vary greatly globally.
✓ Accurate prediction and prognostication of disease progression remains enigmatic.
WHAT IS NEW HERE
✓ Initial description of the 4,200 + patients with pancreatitis identified in the UK BioBank.
✓ The estimated prevalence of chronic pancreatitis in this population is higher than similar cohorts.
✓ Unexpected finding of increased prevalence of hypertension, neoplastic disorders, and chemotherapy use in patients with CP was identified and warrants further evaluation.
Supplementary Material
ACKNOWLEDGEMENTS
This research was conducted using the data obtained as of March 2021 from UKBB Application 48065 “Pancreatic Disease Haplotype Association Studies” (PI: Mark Haupt MD).
Footnotes
SUPPLEMENTARY MATERIAL accompanies this paper at http://links.lww.com/CTG/A747, http://links.lww.com/CTG/A748, http://links.lww.com/CTG/A749, http://links.lww.com/CTG/A750
Contributor Information
Daniel M. Spagnolo, Email: daniel.m.spagnolo@gmail.com.
Phil J. Greer, Email: pgreer@arielmedicine.com.
Celeste Shelton Ohlsen, Email: cshelton@arielmedicine.com.
Shannon Mance, Email: shm98@pitt.edu.
Mitchell Ellison, Email: mellison@arielmedicine.com.
Cameron Breze, Email: cbreze@arielmedicine.com.
Ben Busby, Email: bbusby@dnanexus.com.
David C. Whitcomb, Email: whitcomb@pancreas.org.
REFERENCES
- 1.Machicado JD, Yadav D. Epidemiology of recurrent acute and chronic pancreatitis: Similarities and differences. Dig Dis Sci 2017;62(7):1683–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Whitcomb DC. Hereditary pancreatitis: New insights into acute and chronic pancreatitis. Gut 1999;45(3):317–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sankaran SJ, Xiao AY, Wu LM, et al. Frequency of progression from acute to chronic pancreatitis and risk factors: A meta-analysis. Gastroenterology 2015;149(6):1490–500.e1. [DOI] [PubMed] [Google Scholar]
- 4.Ahmed Ali U, Issa Y, Hagenaars JC, et al. Risk of recurrent pancreatitis and progression to chronic pancreatitis after a first episode of acute pancreatitis. Clin Gastroenterol Hepatol 2016;14(5):738–46. [DOI] [PubMed] [Google Scholar]
- 5.Etemad B, Whitcomb DC. Chronic pancreatitis: Diagnosis, classification, and new genetic developments. Gastroenterology 2001;120(3):682–707. [DOI] [PubMed] [Google Scholar]
- 6.Whitcomb DC. North American Pancreatitis Study Group. Pancreatitis: TIGAR-O version 2 risk/etiology checklist with topic reviews, updates, and use primers. Clin Transl Gastroenterol 2019;10(6):e00027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Nawaz H, Mounzer R, Yadav D, et al. Revised atlanta and determinant-based classification: Application in a prospective cohort of acute pancreatitis patients. Am J Gastroenterol 2013;108(12):1911–7. [DOI] [PubMed] [Google Scholar]
- 8.Whitcomb DC, Yadav D, Adam S, et al. Multicenter approach to recurrent acute and chronic pancreatitis in the United States: The North American pancreatitis study 2 (NAPS2). Pancreatology 2008;8(4-5):520–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Moran RA, García-Rayado G, de la Iglesia-García D, et al. Influence of age, body mass index and comorbidity on major outcomes in acute pancreatitis, a prospective nation-wide multicentre study. United Eur Gastroenterol J 2018;6(10):1508–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Párniczky A, Kui B, Szentesi A, et al. Prospective, multicentre, nationwide clinical data from 600 cases of acute pancreatitis. PLoS One 2016;11(10):e0165309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Olesen SS, Mortensen LH, Zinck E, et al. Time trends in incidence and prevalence of chronic pancreatitis: A 25‐year population‐based nationwide study. United Eur Gastroenterol J 2021;9(1):82–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zhu Y, Pan X, Zeng H, et al. A study on the etiology, severity, and mortality of 3260 patients with acute pancreatitis according to the revised Atlanta classification in Jiangxi, China over an 8-year period. Pancreas 2017;46(4):504–9. [DOI] [PubMed] [Google Scholar]
- 13.Masamune A, Kikuta K, Hamada S, et al. Clinical practice of acute pancreatitis in Japan: An analysis of nationwide epidemiological survey in 2016. Pancreatology 2020;20(4):629–36. [DOI] [PubMed] [Google Scholar]
- 14.Masamune A, Kikuta K, Kume K, et al. Nationwide epidemiological survey of chronic pancreatitis in Japan: Introduction and validation of the new Japanese diagnostic criteria 2019. J Gastroenterol 2020;55(11):1062–71. [DOI] [PubMed] [Google Scholar]
- 15.Matta B, Gougol A, Gao X, et al. Worldwide variations in demographics, management, and outcomes of acute pancreatitis. Clin Gastroenterol Hepatol 2020;18(7):1567–75.e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Burton F, Alkaade S, Collins D, et al. Use and perceived effectiveness of non-analgesic medical therapies for chronic pancreatitis in the United States. Aliment Pharmacol Ther 2011;33(1):149–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mullady DK, Yadav D, Amann ST, et al. Type of pain, pain-associated complications, quality of life, disability and resource utilisation in chronic pancreatitis: A prospective cohort study. Gut 2011;60(1):77–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Greer JB, LaRusch J, Brand RE, et al. ABO blood group and chronic pancreatitis risk in the NAPS2 cohort. Pancreas 2011;40(8):1188–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wilcox CM, Yadav D, Ye T, et al. Chronic pancreatitis pain pattern and severity are independent of abdominal imaging findings. Clin Gastroenterol Hepatol 2015;13(3):552–60; quiz e28-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Machicado JD, Amann ST, Anderson MA, et al. Quality of life in chronic pancreatitis is determined by constant pain, disability/unemployment, current smoking, and associated Co-morbidities. Am J Gastroenterol 2017;112(4):633–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Conwell DL, Banks PA, Sandhu BS, et al. Validation of demographics, etiology, and risk factors for chronic pancreatitis in the USA: A report of the North American pancreas study (NAPS) group. Dig Dis Sci 2017;62(8):2133–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Schwarzenberg SJ, Uc A, Zimmerman B, et al. Chronic pancreatitis: Pediatric and adult cohorts show similarities in disease progress despite different risk factors. J Pediatr Gastroenterol Nutr 2019;68(4):566–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ahmed Ali U, Issa Y, van Goor H, et al. Dutch chronic pancreatitis registry (CARE): Design and rationale of a nationwide prospective evaluation and follow-up. Pancreatology 2015;15(1):46–52. [DOI] [PubMed] [Google Scholar]
- 24.UK Biobank Consortium. UK Biobank Website (https://www.ukbiobank.ac.uk). Accessed February 1, 2021. [Google Scholar]
- 25.Allen N, Sudlow C, Downey P, et al. UK Biobank: Current status and what it means for epidemiology. Heal Pol Technol 2012;1(3):123–6. [Google Scholar]
- 26.Allen NE, Sudlow C, Peakman T, et al. UK biobank data: Come and get it. Sci Transl Med 2014;6(224):224ed4. [DOI] [PubMed] [Google Scholar]
- 27.Collins R. What makes UK Biobank special?. Lancet 2012;379(9822):1173–4. [DOI] [PubMed] [Google Scholar]
- 28.Sudlow C, Gallacher J, Allen N, et al. UK biobank: An open access resource for identifying the causes of a wide range of complex diseases of middle and old age. Plos Med 2015;12(3):e1001779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Centre H and SCI. Quality and Outcomes Framework Achievement, Prevalence and Exceptions Data, 2011/12. Technical Annex, 2012. (http://www.hscic.gov.uk/catalogue/PUB08135/qof-11-12-rep.pdf%5Cnpapers3://publication/uuid/444B68C6-CA5B-4CCF-9E44-F738131EDA7E). Accessed March 1, 2021. [Google Scholar]
- 30.Umapathy C, Raina A, Saligram S, et al. Natural history after acute necrotizing pancreatitis: A large US tertiary care experience. J Gastrointest Surg 2016;20(11):1844–53. [DOI] [PubMed] [Google Scholar]
- 31.Vipperla K, Papachristou GI, Slivka A, et al. Risk of new-onset diabetes is determined by severity of acute pancreatitis. Pancreas 2016;45(1):e14–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Gjuladin-Hellon T, Iheozor-Ejiofor Z, Gordon M, et al. Azathioprine and 6-mercaptopurine for maintenance of surgically-induced remission in Crohn's disease. Cochrane Database Syst Rev 2019;8:CD010233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Excellence National Institute for Health and Care. Pancreatitis. NICE Guideline 104, 2020. (www.nice.org.uk/guidance/ng104. Accessed July 15, 2021. [Google Scholar]
- 34.Roberts SE, Morrison-Rees S, John A, et al. The incidence and aetiology of acute pancreatitis across Europe. Pancreatology 2017;17(2):155–65. [DOI] [PubMed] [Google Scholar]
- 35.PanWessex Study Group; Wessex Surgical Trainee Research Collaborative; Mirnezami A, Knight B, Moran B, et al. Population-based observational study of acute pancreatitis in southern England. Ann R Coll Surg Engl 2019;101(7):487–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Xiao AY, Tan MLY, Wu LM, et al. Global incidence and mortality of pancreatic diseases: A systematic review, meta-analysis, and meta-regression of population-based cohort studies. Lancet Gastroenterol Hepatol 2016;1(1):45–55. [DOI] [PubMed] [Google Scholar]
- 37.Knudsen JS, Heide-Jørgensen U, Mortensen FV, et al. Acute pancreatitis: 31-Year trends in incidence and mortality - a Danish population-based cohort study. Pancreatology 2020;20(7):1332–9. [DOI] [PubMed] [Google Scholar]
- 38.Floyd A, Pedersen L, Nielsen GL, et al. Secular trends in incidence and 30-day case fatality of acute pancreatitis in North jutland county, Denmark: A register-based study from 1981-2000. Scand J Gastroenterol 2002;37(12):1461–5. [DOI] [PubMed] [Google Scholar]
- 39.Lévy P, Barthet M, Mollard BR, et al. Estimation of the prevalence and incidence of chronic pancreatitis and its complications. Gastroentérologie Clin Biol 2006;30(6-7):838–44. [DOI] [PubMed] [Google Scholar]
- 40.Capurso G, Archibugi L, Pasquali P, et al. Prevalence of chronic pancreatitis: Results of a primary care physician-based population study. Dig Liver Dis 2017;49(5):535–9. [DOI] [PubMed] [Google Scholar]
- 41.Machicado JD, Dudekula A, Tang G, et al. Period prevalence of chronic pancreatitis diagnosis from 2001–2013 in the commercially insured population of the United States. Pancreatology 2019;19(6):813–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.White MC, Holman DM, Boehm JE, et al. Age and cancer risk: A potentially modifiable relationship. Am J Prev Med 2014;46(3 Suppl 1):S7–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Group USCSW. US Cancer Statistics: 1999-2009 Incidence and Mortality Web-Based Report. Atlanta GA USDHHS, CDC, Natl Cancer Inst. Published online 2013. [Google Scholar]
- 44.Pallin ND, Beeken RJ, Pritchard-Jones K, et al. Therapeutic radiographers' delivery of health behaviour change advice to those living with and beyond cancer: A qualitative study. BMJ Open 2020;10(8):e039909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Dunbar EK, Greer PJ, Amann ST, et al. Pain experience in pancreatitis:strong association of genetic risk loci for anxiety and PTSD in patients with severe, constant, and constant-severe pain. Am J Gastroenterol 2021;116(10):2128–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Dunbar E, Greer PJ, Melhem N, et al. Constant-severe pain in chronic pancreatitis is associated with genetic loci for major depression in the NAPS2 cohort. J Gastroenterol 2020;55(10):1000–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Phillips AE, Faghih M, Drewes AM, et al. Psychiatric comorbidity in patients with chronic pancreatitis associates with pain and reduced quality of life. Am J Gastroenterol 2020;115(12):2077–85. [DOI] [PubMed] [Google Scholar]
- 48.Xiao AY, Tan ML, Plana MN, et al. The use of international classification of diseases codes to identify patients with pancreatitis: A systematic review and meta-analysis of diagnostic accuracy studies. Clin Transl Gastroenterol 2018;9(10):191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Bycroft C, Freeman C, Petkova D, et al. The UK Biobank resource with deep phenotyping and genomic data. Nature 2018;562(7726):203–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Phillips AE, LaRusch J, Greer P, et al. Known genetic susceptibility factors for chronic pancreatitis in patients of European ancestry are rare in patients of African ancestry. Pancreatology 2018;18(5):528–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.