Abstract
Background:
Inferior outcomes are reported in adolescents with Hodgkin lymphoma (HL). Whether these differences persist within clinical trials is unknown. We examined survival by age in patients receiving response-adapted therapy for classical HL (cHL) on phase 3 Children’s Oncology Group (COG) trials.
Methods:
This analysis included 1,907 patients (1 – 21 years [y]) with newly diagnosed HL enrolled on AHOD0431 (low-risk; NCT00302003), AHOD0031 (intermediate risk; NCT00025259), or AHOD0831 (high risk; NCT01026220) (2002 – 2012). Event-free (EFS) and overall survival (OS) were evaluated between patients <15y and ≥15y. Cox proportional hazards models estimated survival adjusting for disease and treatment factors both overall and among patients with mixed cellularity (MC) or non-MC (nodular sclerosing and not-otherwise-specified) disease.
Findings:
At median follow-up of 7.4 years (IQR 4.3–10.2), patients ≥15y (vs. younger) had worse EFS (p= 0.0038; 5-year: 80% [95% CI: 78 – 83] vs. 86% [95% CI: 83 – 88]), and worse OS (p=0.0012; 5-year 96% [95% CI: 95 – 97] vs. 99% [95% CI: 98 – 99]). Among patients with MC, those ≥15y (vs. younger) had significantly worse EFS in unadjusted (p=0.0039; 5-year 77% [95% CI: 65 – 86] vs. 94% [95% CI: 88 – 97]) and multivariable models (HR: 3.72, 95% CI: 1.56–8 .91, p=0.0032). Among the non-MC group, age ≥15y conferred significantly increased risk of event (HR: 1.3; 95% CI: 1.03 – 1.68, p= 0.027). Overall, patients ≥15y (vs. younger) were three-times more likely to die (HR: 3 08, 95% CI: 1·49, 6·39, p=0.003).
Interpretation:
Adolescents treated on COG HL trials had worse EFS and increased risk of death compared to children. Findings highlight the need for prospective studies to examine tumor and host biology, and to test novel therapies across the age spectrum.
Funding:
National Institutes of Health; St. Baldrick’s Foundation; Lymphoma Research Foundation
Introduction
Hodgkin lymphoma (HL) accounts for 18% of annual cancer diagnoses in adolescents/young adults (AYA) (defined by the National Cancer Institute [NCI] as 15 – 39 years).1 Today, five-year event-free survival (EFS) and overall survival (OS) in HL exceed 85% and 95%, respectively.(1) This success is attributed largely to (1) expanded use of risk-based, multimodal therapy (chemotherapy + radiotherapy [RT]), and (2) sequential therapeutic trials to continuously refine the standard-of-care. Despite overall progress, population-based (1, 2) studies report worse outcomes in adolescents compared to other age groups. (3, 4) Proposed hypotheses for these disparities include age-related differences in healthcare access,4 clinical trial enrollment,(5) survivorship care,(6–8) as well as the unique psychosocial needs of the adolescent-aged population. Another potential explanation for age-related survival differences in HL is the possibility that host or tumor biology, and by extension, therapeutic efficacy may differ across the age spectrum. To determine the independent effect of age on HL survival we examined outcomes among patients receiving risk-based, dose-dense, response-adapted therapy for newly diagnosed HL on Children’s Oncology Group (COG) trials.
Methods
Study design and participants:
This retrospective analysis included patients 1 – 21 years, diagnosed with classical HL (cHL) and enrolled on one of three phase 3 COG trials in the United States (U.S.) and Canada between 2002 and 2012. Trials were defined by risk group according to Ann Arbor stage and B-symptoms, and all used a response-adapted approach. In brief, AHOD0031(9) (National Clinical Trials (NCT) identifier: NCT00025259) enrolled intermediate risk patients from September 23, 2002 to October 9, 2009; AHOD0431(10) (NCT00302003) enrolled low-risk patients from February 13, 2006 to April 3, 2009; AHOD0831(11) (NCT01026220) enrolled high-risk patients from December 7, 2009 to January 19, 2012 (appendix p 2, 5). Patients eligible for enrollment at participating centers were 1 – 21 years with histologically confirmed cHL, and no prior therapy. Eligible histologic subgroups included: Mixed cellularity (MC), nodular sclerosing (NS), lymphocyte predominant (LP) and cHL, not-otherwise-specified (NOS). Each trial was approved by central and local institutional review boards, and written informed consent was obtained from participants or guardians at the time of enrollment.
Procedures:
Age groups were defined according to NCI definitions (4) with 15 years serving as the threshold to delineate younger (1 – 14 years) vs. older groups (15 – 21 years). Patients with NS and cHL, NOS histology were combined into one group (hereafter referred to as non-MC) after sensitivity analyses demonstrated no significant difference in baseline demographics or outcomes between the groups.
Outcomes:
The primary outcomes of this analysis were EFS and OS by age (<15 years vs. ≥15 years). Events included relapse, subsequent malignant neoplasm (SMN), and death. Exploratory outcomes included post-relapse survival by age, as well as EFS and OS by age using a threshold of 12 years.
Statistical analysis:
Patient characteristics were compared across age groups using the x2 test for categorical variables, the Cochran-Armitage Trend Test for ordered categorical variables, and the t-test for continuous variables. Event-free survival was measured from the date of enrollment to the date of first relapse, SMN, or death from any cause. Only first events were counted. Cumulative incidence of relapse was estimated with SMN and death as competing events and was compared across groups by the k-sample test.(12) Patients with no events were censored at last contact. Overall survival was measured from the date of study enrollment to the date of death; EFS, OS, and post-relapse survival were estimated with the Kaplan-Meier method and were compared between age groups with the log-rank test.
Cox proportional hazards regression models identified variables independently associated with survival. Pre-selected variables known or hypothesized to be associated with HL outcome (race/ethnicity, histology, B-symptoms, bulky disease, Ann Arbor stage, RT, protocol) were included in Cox models (2, 13). A significant interaction was confirmed between age and histology and thus outcomes were evaluated within each histologic subgroup (MC vs. non-MC) with interaction terms included in final multivariable models. Due to a high event-rate among patients enrolled on AHOD0431, sensitivity analyses were conducted to evaluate EFS by age among patients enrolled on AHOD0031 and AHOD0831 only (appendix p 2 – 3). Hazard ratios (HR) with 95% confidence intervals (CI) were estimated by final models and p-values < 0.05 were statistically significant.
Toxicities on COG trials are captured through investigator submission of adverse events using NCT Common Terminology Criteria for Adverse Events (CTCAE)(14). We explored the association between grade 3 or higher hematologic toxicity (myelosuppression) and EFS by age. In this analysis, HR represents the hazards of event by age when myelosuppression (yes/no), modeled as a time-dependent variable, is included (appendix p 6) (15).
Post-relapse survival by age was measured from date of first relapse to date of death. Backward selection was used to determine the most robust models by removing variables with p-values >0.2. Final models included: Age at diagnosis, time-to-relapse, race/ethnicity, and initial protocol. For descriptive purposes causes of death after relapse were categorized as HL-related, toxicity or treatment related, and other/unknown. We additionally explored whether age ≥12 years (vs. younger) defined a group with inferior EFS and OS.(16) All analyses used SAS software (SAS/STAT User’s Guide, Version 9.3; SAS Institute, Cary, NC).
Role of the funding source: Funders had no role in study design, conduct, analysis, interpretation, or reporting. All authors had full access to the data, and all had final responsibility regarding the decision to submit for publication.
Results
Between 2002 and 2012, 2,155 patients enrolled on three trials, 1,907 (88%) of whom were included in this analysis. Among 248 (12%) of 2,155 patients excluded, 91 (37%) of 248 had LP histology, 73 (29%) of 248 withdrew consent after one cycle, and 84 (34%) of 248 were not treated in the U.S. or Canada. These patients were excluded to allow for evaluation of Census-based measures of area deprivation and insurance status (Figure 1). Median age of the cohort was 15.3 years (IQR 13.0–16.9), and 1,036 (54%) of 1,907 were ≥15 years; 968 (51%) of 1,907 were male. 1,227 (64%) of 1,907 were non-Hispanic White, 211 (11%) of 1,907 were non-Hispanic Black, and 312 (16%) of 1,907 were Hispanic race/ethnicity (Table 1). In total, 196 (10%) of 1,907 patients had MC histology and 1,711 (90%) of 1,907 patients had non-MC. A significantly higher proportion of patients <15 years vs. older had MC disease (130 [15%] of 871 vs. 66 [6%] of 1,036) p<0.0001. Finally, 748 (72%) of 1,036 patients ≥15 years vs. 553 (63%) of 871 patients <15 years received RT based on protocol-directed interim response criteria (p<0.0001) (Table 1).
Figure 1:
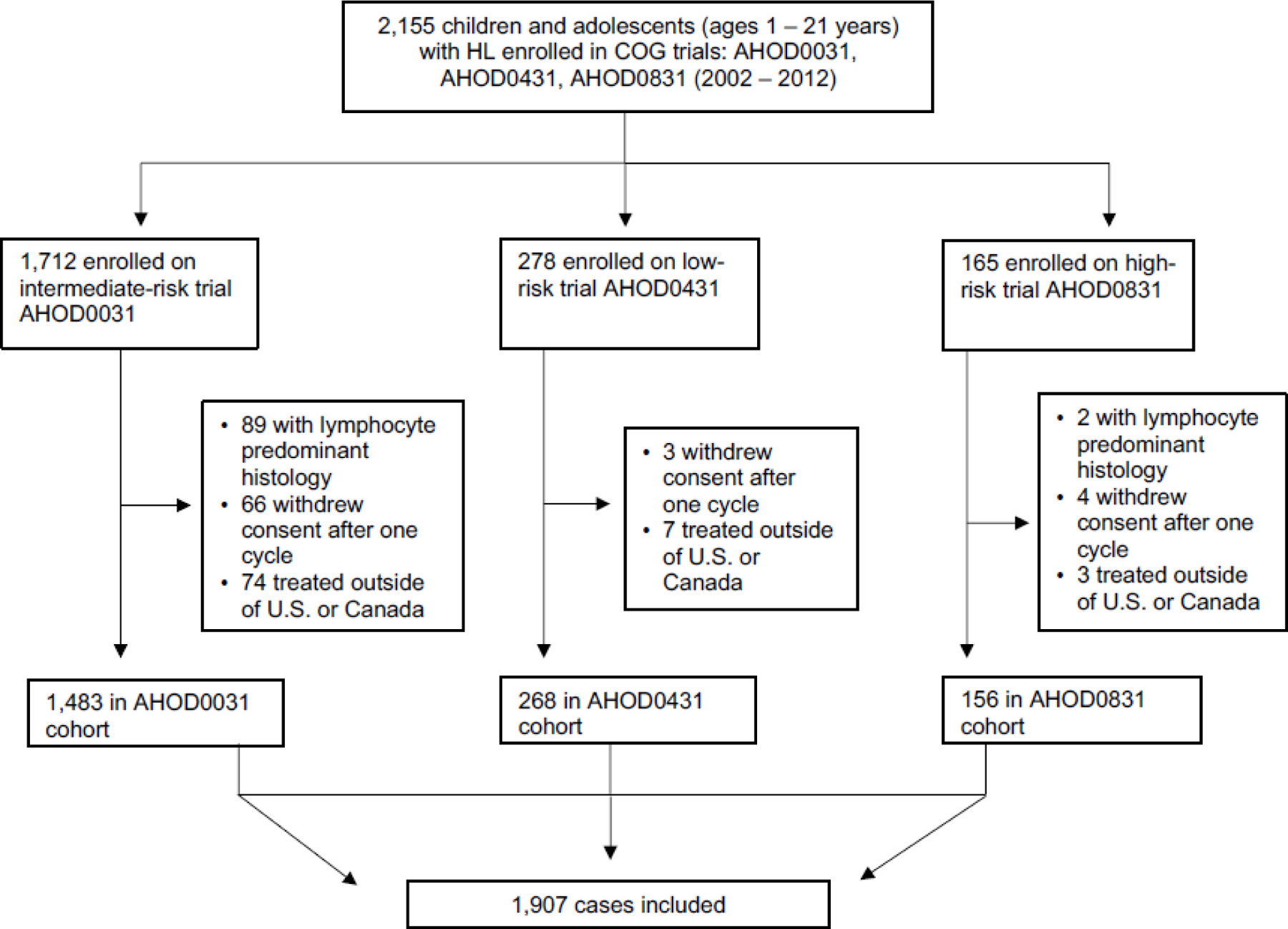
Cohort selection
Table 1.
Demographics and clinical characteristics of study cohort overall, and by age.
| All N (%) | <15 years N (%) | 15 − 21 years N (%) | p-value | |
|---|---|---|---|---|
| 1.907 100) | 871 (46) | 1,036 (54) | ||
| Race/ethnicity | ||||
| Non-Hispanic White | 1227 (64) | 511 (59) | 716 (69) | <0.0001 |
| Non-Hispanic Black | 211 (11) | 107 (12) | 104 (10) | |
| Hispanic | 312 (16) | 180 (21) | 132 (13) | |
| Asian/Pacific + Other/unknown | 157 (8) | 73 (8) | 84 (8) | |
| Sex | ||||
| Male | 968 (51) | 487 (56) | 481 (46) | <0.0001 |
| Female | 968 (51) | 487 (56) | 481 (46) | |
| COG Study | ||||
| AHOD0031 | 1483 (78) | 684 (79) | 799 (77) | 0.22 |
| AHOD0431 | 268 (14) | 126 (14) | 142 (14) | |
| AHOD0831 | 156 (8) | 61 (7) | 95 (9) | |
| Histology | ||||
| Nodular sclerosing | 1547 (81) | 665 (76) | 882 (85) | <0.0001 |
| Mixed cellularity | 196 (10) | 130 (15) | 66 (6) | |
| Classical HL, NOS | 108 (6) | 48 (6) | 60 (6) | |
| Unknown | 56 (3) | 28 (3) | 28 (3) | |
| Ann Arbor Stage | ||||
| I | 145 (8) | 90 (10) | 55 (5) | 0.093 |
| II | 1097 (58) | 449 (52) | 648 (63) | |
| III | 352 (18) | 169 (19) | 183 (18) | |
| IV | 313 (16) | 163 (19) | 150 (14) | |
| B-symptoms | ||||
| No | 1415 (74) | 683 (78) | 732 (71) | 0.0006 |
| Yes | 490 (26) | 187 (21) | 303 (30) | |
| Unknown | 2 (<1) | 1 (<1) | 1 (<1) | |
| Bulky disease | ||||
| No | 639 (34) | 300 (34) | 339 (33) | 0.48 |
| Yes | 1254 (66) | 563 (65) | 691 (67) | |
| Unknown | 14 (<1) | 8 (<1) | 6 (<1) | |
| Radiation therapy | ||||
| No | 606 (32) | 318 (37) | 288 (28) | <0.0001 |
| Yes | 1301 (68) | 553 (63) | 748 (72) | |
| Insurance | ||||
| Private | 1136 (60) | 488 (56) | 648 (63) | 0.0003 |
| Government | 624 (33) | 319 (37) | 305 (29) | |
| Self-pay or No Insurance | 50 (3) | 14 (2) | 36 (3) | |
| Other + Unknown | 97 (5) | 50 (6) | 47 (5) | |
Median follow-up was 7.4 years (IQR 4.3–10.2) and differed between younger and older patients. Among patients <15 years, median follow-up was 8.2 years (IQR 5.0 – 10.5) vs. 6.6 years (IQR 3.8 – 9.9) in patients ≥15 years (p< 0.0001) (appendix p 2). Pooled 5-year EFS and OS were 83% (95% CI: 81 –85) and 97% (95% CI: 96 –98), respectively. Of 340 on-study events, 303 (89%) were disease relapse, 26 (8%) were SMN, and 11 (3%) were on-study deaths. Overall, 26 (1.4%) of 1,907 patients experienced SMN, and this did not differ by age. In unadjusted analyses, age ≥15 years (vs. younger) conferred significantly worse EFS (p=0.0038; 5-year: 80% [95% CI: 78–83] vs. 86% [95% CI: 83–88]) and significantly worse OS (p=0.0012; 5-year: 96% [95% CI: 95–97] vs. 99% [95% CI: 98–99]). In multivariable models these associations remained significant: Compared with younger patients, those ≥15 years had 40% increased risk of event (HR: 1.4, 95% CI: 1.1, 1.8, p=0.004) and 3-times higher risk of death overall (HR: 3.08, 95% CI: 1.49, 6.39, p=0.004) (Table 3).
Table 3.
Multivariable model of overall survival (OS) among 1,907 patients treated for HL on Children’s Oncology Group (COG) trials (2002 – 2012).*
| Variables | OS | |||
|---|---|---|---|---|
| HR | 95% CI | p-value | ||
| Age (R: <15) | ||||
| ≥15 | 3.08 | 1.49, 6.39 | 0.003 | |
| Sex (R: Female) | ||||
| Male | 1.27 | 0.69, 2.34 | 0.44 | |
| Race/ethnicity (R: Non-Hispanic White) | ||||
| Non-Hispanic Black | 2.37 | 1.05, 5.39 | 0.11 | |
| Hispanic | 2.32 | 1.06, 5.06 | ||
| Asian/Pacific Island | 0.80 | 0.11, 5.97 | ||
| Other | 0.74 | 0.10, 5.55 | ||
| Study (R: AHOD0031) | ||||
| AHOD0431 | 0.30 | 0.03, 2.73 | 0.45 | |
| AHOD0831 | 0.74 | 0.19, 2.86 | ||
| Histology (R: Mixed Cellularity [MC]) | ||||
| Non-MC | 1.30 | 0.39, 4.30 | 0.67 | |
| Stage (R: I and II) | ||||
| III | 0.86 | 0.31, 2.37 | 0.72 | |
| IV | 1.31 | 0.47, 3.71 | ||
| B symptoms (R: No) | ||||
| Yes | 2.06 | 0.94, 4.54 | 0.07 | |
| Bulky Disease (R: No) | ||||
| Yes | 1.69 | 0.70, 4.05 | 0.24 | |
| Radiation (R: Yes) | ||||
| No | 0.98 | 0.47, 2.04 | 0.96 | |
| Payment (R: Private) | ||||
| Government | 0.93 | 0.48, 1.82 | 0.91 | |
| Other + Unknown | 0.68 | 0.16, 2.97 | ||
| Self-Pay or No Insurance | 0.55 | 0.07, 4.15 | ||
Abbreviations: HR: Hazard ratio; CI: confidence interval; R: Reference; non-MC Histology (includes Nodular Sclerosing and cHL, NOS: Classical Hodgkin lymphoma, not-otherwise-specified).
Few OS events limited assessment of OS by histology in patients with MC disease.
The effect of age on EFS differed by histologic subgroup. In patients with non-MC histology, age ≥15 years vs. younger was associated with significantly inferior EFS (p=0.047; 5-year: 80% [95% CI: 78–83]) vs. 84% [95% CI: 81–87]), (Figure 2). Among patients with MC histology, age ≥15 years vs. younger was also associated with inferior EFS (p=0.0039; 5-year: 77% [95% CI: 65 – 86] vs. 94% (95% CI: 88 – 97]) (Figure 2). These differences in EFS remained significant in multivariable models when interaction terms between age and histology were included. Specifically, patients ≥15 years (vs. younger) with non-MC histology had 1.3-times higher hazard of event (HR: 1.3 [95% CI: 1.03–1.68], p=0.027) and those ≥15 years (vs. younger) with MC histology had 3.7-times higher hazard of event (HR: 3.72, 95% CI: 1.56– 8.91, p=0.0032) (Table 2). Due to a high event-rate among patients enrolled on AHOD0431, a sensitivity analysis was conducted to examine adjusted EFS among patients enrolled on AHOD0031 and AHOD0831 only. In these models, age ≥15 years and non-MC disease remained significantly associated with worse EFS (HR: 1.40; 95% CI: 1.08, 1.82. p=0.013), as did age ≥15 years and MC disease (HR: 3.08; 95% CI: 1.26, 7.54, p=0.014) (appendix p 2 – 3). Severe myelosuppression occurred in 1,278 (67%) of 1,907 patients; this did not differ between older and younger patients (p= 0.42). When this toxicity was included in a model with age, it attenuated the effect of older age on EFS (appendix p 6).
Figure 2.
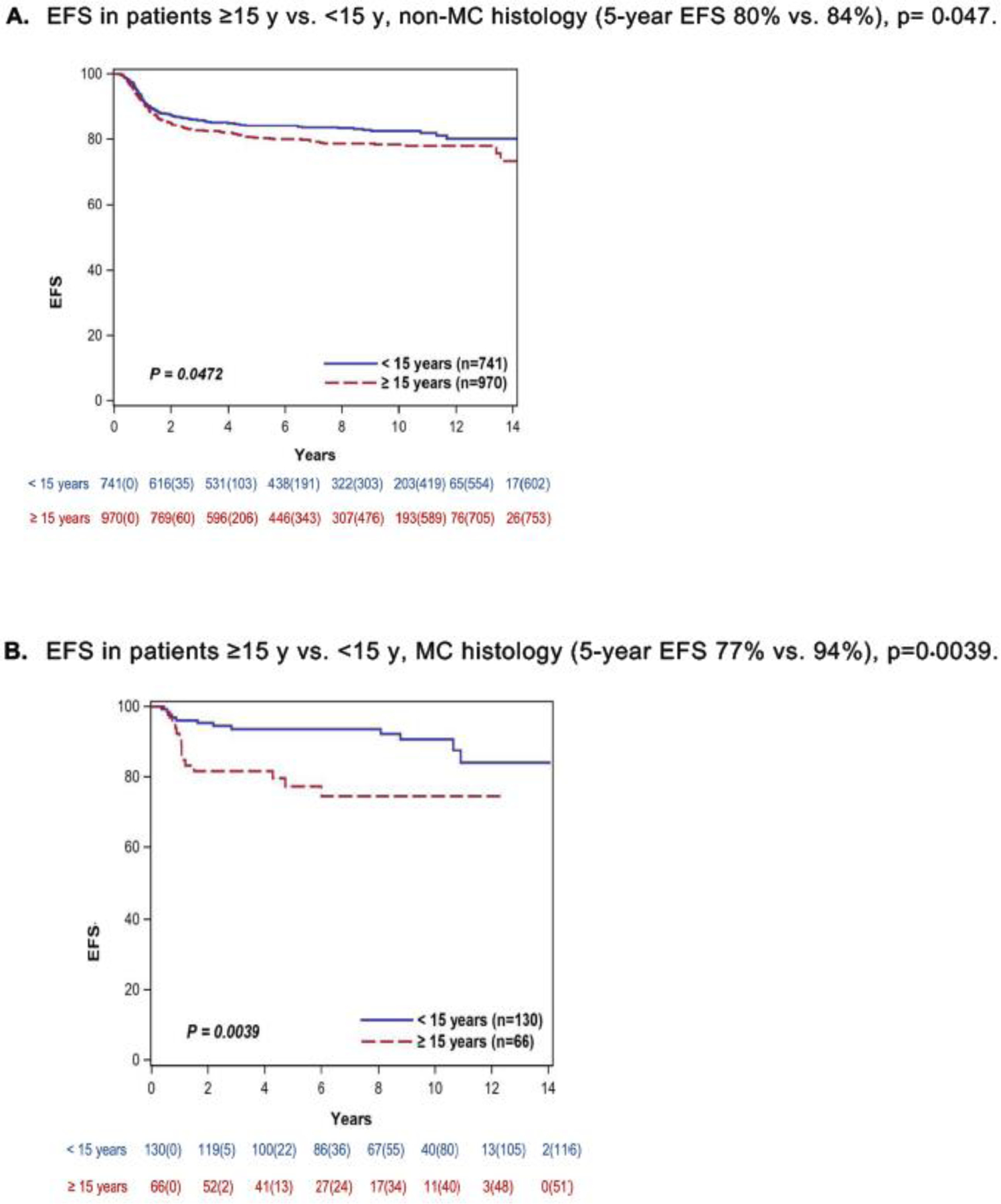
(A and B): EFS by age ≥15 years vs. <15 years and histologic subgroups in 1,907 patients (<1 – 21 years) enrolled on phase 3 Children’s Oncology Group trials for newly diagnosed Hodgkin lymphoma (HL) (2002 – 2012). The mixed cellularity (MC) group included 196 (10%) of the cohort of 1,907 patients. The non-MC group included 1,711 (90%) of 1,907 patients and was comprised of 1,547 (90%) of 1,711 patients with nodular sclerosing (NS) histology, 108 (6.5%) of 1,711 with classical HL, not-otherwise-specified, and 56 (3.5%) of 1,711 with histology unknown.
Table 2.
Multivariable model of event-free survival (EFS) in 1,907 patients (1 – 22 years) treated for HL on Children’s Oncology Group (COG) trials (2002 – 2012)
| Variables | EFS | |||
|---|---|---|---|---|
| HR | 95% CI | p-value | ||
| Sex (R: Female) | ||||
| Male | 1.09 | 0.87, 1.34 | 0.46 | |
| Race (R: Non-Hispanic White) | ||||
| Non-Hispanic Black | 0.90 | 0.61, 1.32 | 0.76 | |
| Hispanic | 1.16 | 0.85, 1.60 | ||
| Asian/Pacific Island | 1.22 | 0.66, 2.26 | ||
| Other | 0.95 | 0.55, 1.65 | ||
| Study (R: AHOD0031) | ||||
| AHOD0431 | 3.46 | 2.11, 5.66 | <0.0001 | |
| AHOD0831 | 0.49 | 0.28, 0.84 | ||
| Age * Histology interaction | ||||
| MC * Age (R: <15) | ≥ 15 | 3.72 | (1.56, 8.91) | 0 020 |
| Non-MC * Age (R: <15) | ≥ 15 | 1.32 | (1.03, 1.68) | |
| Stage (R: I and II) | ||||
| III | 1.47 | (1.00, 2.16) | <0.0001 | |
| IV | 2.49 | (1.72, 3.62) | ||
| B symptoms (R: No) | ||||
| Yes | 2.11 | (1.50, 2.96) | <0.0001 | |
| Bulky Disease (R: No) | ||||
| Yes | 2.27 | (1.60, 3.23) | <0.0001 | |
| Radiation (R: Yes) | ||||
| No | 1.56 | (1.22, 2.00) | 0.0001 | |
| Payment (R: Private) | ||||
| Government | 0.87 | (0.68, 1.13) | 0.19 | |
| Other + Unknown | 1.18 | (0.74, 1.89) | ||
| Self-Pay or None | 0.45 | (0.19, 1.12) | ||
Abbreviations: HR: Hazard ratio; CI: confidence interval
Median time to relapse was 10 year (IQR 0.7–1.6) and did not differ by age. In total, 303 (16%) of 1,907 patients had disease recurrence. At median post-relapse follow-up of 5.6 years (IQR 2.1–8.5), survival was significantly worse in those ≥15 years vs. younger (p=0.0042; 5-year post-relapse OS: 80% [95% CI: 72 – 85] vs. 93% [95% CI: 85 – 97) (appendix p 2, 8). This remained significant in multivariable models: age ≥15 years conferred significantly increased risk of post-relapse mortality (HR: 2.7; 95% CI: 1.2, 6.1, p= 0.014) (Table 4). In total, 41 (14%) of 303 patients died post-relapse; HL progression was the reported cause in (13) 32% of 41.
When evaluating the age threshold of 12 years, unadjusted EFS was worse in those ≥12 years (vs. younger) (p=0.024; 5-year: 82% [95% CI: 80 – 84] vs. 88% [95% CI: 84 – 91]). Overall survival did not differ by age when using a threshold of 12 years (p= 0.20). In multivariable models, age ≥12 years (vs. younger) with non-MC histology remained significantly associated with inferior EFS (HR: 1.48; 95% CI: 1.03, 2.13, p= 0.030) (appendix p 7). Due to a small sample size with few deaths in the MC group, adjusted OS was not evaluated using an age threshold of 12.
Discussion
In patients receiving risk-based, response-adapted therapy for HL on COG trials, those age ≥15 years had worse EFS and OS than younger patients. In a novel finding, we identified that the magnitude of the effect of age on EFS differed by histology. Among patients with MC disease, age ≥15 years (vs. younger) conferred almost 4-times higher risk of event. Consistent with both population-based and prior consortium studies, we found that age ≥15 years was associated with worse survival within those enrolled on a clinical trial; this included post-relapse outcomes, which may reflect differences in salvage therapy among patients transitioning from pediatric to adult care. Finally, we found that inferior EFS in adolescents extended below the NCI-defined lower age bound for AYA (15 years), and that patients as young as 12 years were at increased risk for treatment failure on the included trials. Studies of age-related differences in HL outcomes at the population-level have highlighted under-enrollment of AYAs on clinical trials as a potential driver. While clinical trial enrollment is certainly a contributor, findings from this work support the additional hypotheses that HL in adolescents may be biologically distinct from similar histopathologic entities in children, and/or that differences in drug exposure, chemotherapy sensitivity, or salvage treatment options may also be important. For these patients, prospective studies, powered to measure lymphoma and host biology, treatment tolerability, and tumor response across the age spectrum are needed. Furthermore, that age ≥15 years conferred increased risk of death despite outstanding OS in the pooled cohort speaks to the possibility of differences in post-treatment follow-up or post-relapse care in patients aging out of pediatric oncology.
Disease-related factors associated with inferior survival in HL include (but are not limited to) advanced stage, B-symptoms, and bulky adenopathy, two of which were increased in older adolescents at presentation in our cohort. In population-based studies, not receiving RT is also predictive of poor outcomes, particularly in AYAs, which was similarly observed in our cohort.(17) The observation of inferior EFS among adolescents in this cohort is consistent with reports from both the German Society of Pediatric Oncology and Hematology-Hodgkin’s Disease (GPOH-HD)-2002 study and the Pediatric Oncology Group (POG) 8725 Protocol which both reported inferior EFS in patients ≥13 years vs. younger.(18, 19) In contrast, Fernandez et al. reported no difference in EFS between younger and older (>15 years) patients treated on POG studies P9425 and P9426.(20) On these trials, patients received response-adapted chemotherapy, however all received RT, which may have led to improved survival among the older group.(21) A recent study by Burnelli et al. reported similar survival in children and adolescents treated on Italian Association of Pediatric Hematology and Oncology (AIEOP) protocols between 1996 and 2017; all intermediate and high-risk patients on these protocols received RT and LP patients were included in their analysis.(22)
Given the association between RT and HL outcomes, it is notable that the response-adapted approach in COG trials resulted in omission of RT in >30% of patients. This is relevant because multivariable models identified that not receiving RT was independently associated with higher risk of event. It is additionally notable that older adolescents were more likely than younger patients in our cohort to require RT, suggesting slow or incomplete chemotherapy response by interim disease evaluation among the adolescents given the COG response-adapted approach. Despite this, older patients were more likely to have disease recurrence, even after adjusting for RT in multivariable models. This finding speaks to the possibility of differences in chemotherapy sensitivity across age groups, or of differences in actual amount of drug delivered.
That adolescents in our cohort had worse outcomes than younger children treated on the same trials underscores the need to better understand how not only current imaging-based response criteria, but also drug delivery and treatment tolerability differ by age. Factors impacting drug exposure broadly include dosing adjustments, treatment tolerability and adherence. Though we were unable to measure adherence, it is possible that older patients were more likely to skip doses of steroids, as observed in adolescents taking oral chemotherapy for acute lymphoblastic leukemia.(23) Dosing guidelines in the protocols could have also impacted drug delivery as they suggested adjusting chemotherapy doses for ideal body weight. This could have resulted in under-treatment of overweight or obese patients, however data to evaluate this hypothesis were unavailable. Exploratory models revealed that the presence of hematologic toxicity attenuated the EFS differences by age. Though additional work is needed to further understand these findings, they support the possibility that differences in drug exposure between younger and older patients, either due to dosing variations, pharmacokinetics, or adjustments at the discretion of treating providers may partly explain the differences in EFS between younger and older patients in our cohort. Prospective studies are needed to explore this further.
Patients ≥15 years with MC histology had significantly higher hazard of event compared to younger patients with MC disease. Treatment for children and adolescents with cHL has been largely agnostic of histology in North American and European studies, until the recent separation of LP histology. Bazzeh and colleagues examined the impact of histology on HL outcomes of children and adults using population-level data from 1998 – 2005.(24) Findings from this analysis revealed that patients in the “non-NS” subgroup, which included both MC and LP, had worse outcomes in adults but not children.(24) Our cohort did not include patients with LP disease, however the pattern of findings was similar: Older patients with MC histology had inferior EFS. These observations suggest first, that MC histology deserves more focus across all age groups, and second, that for older patients, MC histology may define a group in whom response-adaptation may be suboptimal.
While the effect of age on EFS in our cohort differed by histology, the extent to which HL in adolescents is truly biologically distinct from the same histopathological entity in other age groups remains unknown.(25) Histology may be a proxy for other molecular or biologic factors that change with age, including variations in the immune microenvironment, initial viral exposure, or in body morphometrics and hormonal changes associated with puberty and growth. (26) An infectious etiology for HL has long been suggested by its epidemiologic characteristics, and much of the data point specifically to Epstein-Barr virus (EBV). Furthermore, MC disease is more frequently associated with younger age at presentation, as well as with EBV, again supporting biologic or molecular phenomena that may change across the age spectrum. Further rationale for a molecular influence is supported by the fact that the Scott Predictor, a study of gene expression profiling in adults identified a 23-gene signature that was predictive of survival in adults(27) with advanced stage HL, but that was not predictive in children.(28) As the field moves forward, correlative studies from upcoming trials can use banked specimens to examine whether the distribution of prognostic molecular biomarkers varies by age and histology.
Post-relapse survival was significantly worse in patients ≥15 years. Dependence on salvage therapy to maintain OS has allowed for reduction of RT in those demonstrating chemotherapy responsiveness up-front. Fortunately, salvage therapies are highly successful, however access to, and receipt of these therapies (e.g. stem cell transplantation [SCT]) may vary. The median age at relapse in our cohort was 18 years. Due to life transitions (29) these patients may not have received second-line therapy at their original treating institutions. Thus, part of the difference in post-relapse survival may result from transitions between pediatric and adult settings, and/or from a difference in type of salvage. The present COG data have limited information about post-relapse therapy, however additional analyses using larger linked datasets are underway.
The clinical trial setting of cancer cooperative groups presents several advantages to examining survival outcomes by age, and is one of the strengths of this analysis. Standardized enrollment requirements ensure uniform assessment of stage, histology, and HL characteristics. Identical treatment strategies ensure that factors impacting cancer care during up-front therapy are largely controlled for, and standard capture of events allow for reliable assessments of outcome. Limitations are also related to the structure of these trials, namely the capacity for detailed dosing data and longer follow up of disease outcomes, particularly in an AYA population (appendix p 2). During up-front therapy, legacy studies did not uniformly proscribe or capture dose modifications or treatment delays, nor did they collect complete data on body mass index. Due to small numbers we were not powered to determine whether high rates of early relapse in adolescents with MC disease contributed to their inferior OS outcomes. Given that MC histology is less prevalent in North American and European populations, analyses to understand this subtype will require larger cohorts facilitated by data-sharing and harmonization across consortia. Finally, details about salvage therapy were largely unavailable, which leaves important questions about post-relapse mortality unanswered.
Conclusion
Although pediatric HL has been a success story over the last four decades, the present analysis highlights an unresolved survival gap in adolescents. The observation that older patients in this cohort had disproportionately high event rates and increased risk of early mortality in the controlled treatment setting supports the need for novel biology-based approaches in adolescents with both newly diagnosed and recurrent disease. Our finding of higher relapse rates in patients ≥12 years supports the eligibility criteria of the current COG collaboration in the National Clinical Trials Network (NCTN) protocol for advanced stage HL (SWOG S1826, ClinicalTrials.gov identifier: NCT03907488). In 2020, the National Comprehensive Cancer Network convened a working group of leaders representing pediatric and adult consortia to coordinate development of AYA-focused clinical trials in HL. (30) The opening of these trials is proof of principle that joint-efforts across pediatric and adult consortia are changing the framework of how we manage and study HL, which in turn, will provide novel insights into how best to optimize care and improve outcomes for the adolescent HL population.
Supplementary Material
RESEARCH IN CONTEXT.
Evidence before this study:
While Hodgkin lymphoma is widely considered a highly curable malignancy, numerous studies have reported inferior outcomes in adolescent and young adult (AYA) patients compared to children and older adults. Whether these age-related differences are observed in patients enrolled on phase 3 clinical trials is not known. We searched PubMed through September, 2021, for published analyses assessing the effect of age on survival outcomes of pediatric and adolescent patients treated for early or advanced stage Hodgkin lymphoma with response-adapted therapy on cooperative group clinical trials. The search terms (“adolescent” or “AYA” or “pediatric” or “childhood”) AND “Hodgkin” AND (“clinical trial” or “phase 3”) AND “response-adapted” were used. We identified no published reports examining the effects of age on survival, across all stages, in patients receiving response-adapted therapy for newly diagnosed Hodgkin lymphoma on contemporary clinical trials.
Added value of this study:
This is the largest analysis to date examining the independent effect of age on survival of children and adolescents receiving response-based therapy for early or advanced stage Hodgkin lymphoma on Children’s Oncology Group clinical trials. The analytic cohort included 1,907 children and adolescents (1 – 21 years old; 968 [51%] male and 939 [49%] female), with classical Hodgkin lymphoma enrolled on one of three phase 3 trials in North America between 2002 and 2012. The analysis revealed that among patients enrolled on clinical trials, older (versus younger) age was associated with significantly worse event-free and overall survival. The analysis also revealed that the magnitude of the effect of age on event-free survival varied across classical Hodgkin lymphoma histologic subgroups. Specifically, among patients with mixed cellularity disease, age 15 years and older was associated with almost 4-times higher hazard of event compared with those less than 15 years. Across all patients, age 15 years or older conferred 300% increased risk of all-cause mortality.
Implications of all the available evidence:
Adolescent patients receiving response-adapted therapy for Hodgkin lymphoma on Children’s Oncology Group trials had worse EFS and increased risk of early mortality when compared with younger children. These findings support the hypothesis that classical Hodgkin lymphoma in adolescents may be biologically distinct from similar histopathologic entities in children. Findings additionally highlight the need for prospective studies to both examine tumor and host biology, and to test novel therapeutic approaches across the age spectrum.
Acknowledgments:
This work was supported in part by the National Institutes of Health and National Clinical Trials Network (NCTN) Operations Center Grant No. U10CA180886, NCTN Statistics & Data Center Grant U10CA180899; St. Baldrick’s Foundation; COG Chair Grant No. U10CA98543, COG Statistics and Data Center Grant No. U10CA098413; Lymphoma Research Foundation; KL2 Career Development Award Grant No. KL2-TR001874, Irving Institute for Clinical & Translational Research, Columbia University Irving Medical Center.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Declaration of interests: We declare no competing interests.
Data Sharing: Consistent with the policies of the National Institutes of Health, The Children’s Oncology Group Hodgkin Lymphoma Committee will consider data sharing requests from researchers investigating questions regarding childhood and adolescent Hodgkin lymphoma. Investigators who wish to use individual patient data from one or more COG studies must make a formal request to the Group. Requests for access to COG protocol research data may be sent to: datarequest@childrensoncologygroup.org.
References
- 1.Kahn JM, Kelly KM. Adolescent and young adult Hodgkin lymphoma: Raising the bar through collaborative science and multidisciplinary care. Pediatr Blood Cancer 2018;65(7):e27033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kahn JM, Keegan TH, Tao L, Abrahao R, Bleyer A, Viny AD. Racial disparities in the survival of American children, adolescents, and young adults with acute lymphoblastic leukemia, acute myelogenous leukemia, and Hodgkin lymphoma. Cancer 2016;122(17):2723–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Henderson TO, Parsons SK, Wroblewski KE, Chen L, Hong F, Smith SM, et al. Outcomes in adolescents and young adults with Hodgkin lymphoma treated on US cooperative group protocols: An adult intergroup (E2496) and Children’s Oncology Group (COG AHOD0031) comparative analysis. Cancer 2018;124(1):136–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Crombie JL, LaCasce AS. Current considerations in AYA Hodgkin lymphoma. Br J Haematol 2019;184(1):72–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bleyer A, Tai E, Siegel S. Role of clinical trials in survival progress of American adolescents and young adults with cancer-and lack thereof. Pediatr Blood Cancer 2018;65(8):e27074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Keegan THM, Parsons HM. Adolescent angst: enrollment on clinical trials. Hematology Am Soc Hematol Educ Program 2018;2018(1):154–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Weiss AR, Nichols CR, Freyer DR. Enhancing Adolescent and Young Adult Oncology Research Within the National Clinical Trials Network: Rationale, Progress, and Emerging Strategies. Semin Oncol 2015;42(5):740–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wolfson J, Sun CL, Wyatt L, Stock W, Bhatia S. Impact of treatment site on disparities in outcome among adolescent and young adults with Hodgkin lymphoma. Leukemia 2017;31(6):1450–3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Friedman DL, Chen L, Wolden S, Buxton A, McCarten K, FitzGerald TJ, et al. Dose-intensive response-based chemotherapy and radiation therapy for children and adolescents with newly diagnosed intermediate-risk hodgkin lymphoma: a report from the Children’s Oncology Group Study AHOD0031. J Clin Oncol 2014;32(32):3651–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Keller FG, Castellino SM, Chen L, et al. Results of the AHOD0431 trial of response adapted therapy and a salvage strategy for limited stage, classical Hodgkin lymphoma: A report from the Children’s Oncology Group. Cancer 2018;124(15):3210–3219. doi: 10.1002/cncr.31519 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kelly KM, Cole PD, Pei Q, Bush R, Roberts KB, Hodgson DC, et al. Response-adapted therapy for the treatment of children with newly diagnosed high risk Hodgkin lymphoma (AHOD0831): a report from the Children’s Oncology Group. Br J Haematol 2019;187(1):39–48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gray RJ. A Class of K-Sample Tests for Comparing the Cumulative Incidence of a Competing Risk. The Annals of Statistics 16(3), 1141–1154. [Google Scholar]
- 13.Kahn JM, Kelly KM, Pei Q, Bush R, Friedman DL, Keller FG, et al. Survival by Race and Ethnicity in Pediatric and Adolescent Patients With Hodgkin Lymphoma: A Children’s Oncology Group Study. J Clin Oncol 2019;37(32):3009–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Trotti A, Colevas AD, Setser A, Rusch V, Jaques D, Budach V, et al. CTCAE v3.0: development of a comprehensive grading system for the adverse effects of cancer treatment. Semin Radiat Oncol 2003;13(3):176–81. [DOI] [PubMed] [Google Scholar]
- 15.Valeri L , and VanderWeele TJ Mediation analysis allowing for exposure–mediator interactions and causal interpretation: theoretical assumptions and implementation with SAS and SPSS macros. Psychol. Methods 2013; 18:137–150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gore L, Ivy SP, Balis FM, Rubin E, Thornton K, Donoghue M, et al. Modernizing Clinical Trial Eligibility: Recommendations of the American Society of Clinical Oncology-Friends of Cancer Research Minimum Age Working Group. J Clin Oncol 2017;35(33):3781–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kahn JM, Maguire FB, Li Q, Abrahao R, Flerlage JE, Alvarez E, et al. Initial cancer treatment and survival in children, adolescents, and young adults with Hodgkin lymphoma: A population-based study. Cancer 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Weiner MA, Leventhal B, Brecher ML, Marcus RB, Cantor A, Gieser PW, et al. Randomized study of intensive MOPP-ABVD with or without low-dose total-nodal radiation therapy in the treatment of stages IIB, IIIA2, IIIB, and IV Hodgkin’s disease in pediatric patients: a Pediatric Oncology Group study. J Clin Oncol 1997;15(8):2769–79. [DOI] [PubMed] [Google Scholar]
- 19.Mauz-Korholz C, Hasenclever D, Dorffel W, Ruschke K, Pelz T, Voigt A, et al. Procarbazine-free OEPA-COPDAC chemotherapy in boys and standard OPPA-COPP in girls have comparable effectiveness in pediatric Hodgkin’s lymphoma: the GPOH-HD-2002 study. J Clin Oncol 2010;28(23):3680–6. [DOI] [PubMed] [Google Scholar]
- 20.Fernandez KS, Schwartz CL, Chen L, Constine LS, Chauvenet A, de Alarcon PA. Outcome of adolescents and young adults compared to children with Hodgkin lymphoma treated with response-based chemotherapy on pediatric protocols: A Children’s Oncology Group report. Pediatr Blood Cancer 2017;64(12). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Castellino SM, Parsons SK, Kelly KM. Closing the survivorship gap in children and adolescents with Hodgkin lymphoma. Br J Haematol 2019;187(5):573–87. [DOI] [PubMed] [Google Scholar]
- 22.Burnelli R, Fiumana G, Rondelli R, Pillon M, Sala A, Garaventa A, et al. Comparison of Hodgkin’s Lymphoma in Children and Adolescents. A Twenty Year Experience with MH’96 and LH2004 AIEOP (Italian Association of Pediatric Hematology and Oncology) Protocols. Cancers (Basel) 2020;12(6). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bhatia S, Hageman L, Chen Y, Wong FL, McQuaid EL, Duncan C, et al. Effect of a Daily Text Messaging and Directly Supervised Therapy Intervention on Oral Mercaptopurine Adherence in Children With Acute Lymphoblastic Leukemia: A Randomized Clinical Trial. JAMA Netw Open 2020;3(8):e2014205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Bazzeh F, Rihani R, Howard S, Sultan I. Comparing adult and pediatric Hodgkin lymphoma in the Surveillance, Epidemiology and End Results Program, 1988–2005: an analysis of 21 734 cases. Leuk Lymphoma 2010;51(12):2198–207. [DOI] [PubMed] [Google Scholar]
- 25.Mottok AJR, Chun Chan F, et al. . Prediction of primary treat- ment outcome using gene expression profiling of pre-treatment biopsies obtained from childhood and adolescent Hodgkin lymphoma patients. Blood 2015;126:175. [Google Scholar]
- 26.Kelly KM. Hodgkin lymphoma in children and adolescents: improving the therapeutic index. Hematology Am Soc Hematol Educ Program 2015;2015:514–21. [DOI] [PubMed] [Google Scholar]
- 27.Scott DW, Chan FC, Hong F, Rogic S, Tan KL, Meissner B, et al. Gene expression-based model using formalin-fixed paraffin-embedded biopsies predicts overall survival in advanced-stage classical Hodgkin lymphoma. J Clin Oncol 2013;31(6):692–700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Mottok A, Steidl C. Biology of classical Hodgkin lymphoma: implications for prognosis and novel therapies. Blood 2018;131(15):1654–65. [DOI] [PubMed] [Google Scholar]
- 29.Ferrari A, Stark D, Peccatori FA, Fern L, Laurence V, Gaspar N, et al. Adolescents and young adults (AYA) with cancer: a position paper from the AYA Working Group of the European Society for Medical Oncology (ESMO) and the European Society for Paediatric Oncology (SIOPE). ESMO Open 2021;6(2):100096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Coccia PF, Pappo AS, Beaupin L, Borges VF, Borinstein SC, Chugh R, et al. Adolescent and Young Adult Oncology, Version 2.2018, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw 2018;16(1):66–97. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


