Abstract
Anastomotic leak (AL) represents one of the most detrimental complications after colorectal surgery. The patient-related factors and surgery-related factors leading to AL have been identified in previous studies. Through early identification and timely adjustment of risk factors, preventive measures can be taken to reduce potential AL. However, there are still many problems associated with AL. The debate about preventive measures such as preoperative mechanical bowel preparation (MBP), intraoperative drainage, and surgical scope also continues. Recently, the gut microbiota has received more attention due to its important role in various diseases. Although the underlying mechanisms of gut microbiota on AL have not been validated completely, new strategies that manipulate intrinsic mechanisms are expected to prevent and treat AL. Moreover, laboratory examinations for AL prediction and methods for blood perfusion assessment are likely to be promoted in clinical practice. This review outlines possible risk factors for AL and suggests some preventive measures in terms of patient, surgery, and gut microbiota.
Keywords: anastomotic leakage, risk factor, gut microbiota, colorectal cancer, prevention
Introduction
Colorectal cancer (CRC) is a common intestinal malignancy. In 2020, about 1.9 million additional CRC cases and 935,000 deaths from CRC were reported globally, making it the third cause of cancer incidence and the second cause of cancer death. 1 This disease seriously affects human health and consumes social and medical resources, placing a heavy burden on the country. At present, the treatments for CRC mainly include surgical techniques, radiotherapy, chemotherapy, and targeted therapy. Surgery is reckoned as the main strategy of CRC treatment. With the improvement of surgical resection and anastomosis techniques and the application of new instruments, surgeons have made significant progress in the radical treatment of CRC. However, surgical complications are inevitable, and anastomotic leak (AL) is one of the most feared complications after CRC surgery with serious impacts on morbidity and mortality. AL may increase the risk of low anterior resection syndrome and the mortality in elderly patients.2,3 Besides, AL also prolongs the hospital stay, increases the reoperation rate, and is associated with tumor recurrence and reduced long-term survival rate. 4 Therefore, early identification of risk factors for AL and timely related prevention are of great significance for surgical safety.
The diagnostic criteria for AL have not been completely established. The International Study Group of Rectal Cancer (ISREC) put forward relevant opinions for rectal AL and defined it as the intercommunication of internal and external intestinal space caused by anastomotic defects. 5 Rectal AL can be classified into 3 grades according to its severity: Grade A, no obvious clinical symptoms and signs, requiring no special intervention; Grade B, atypical clinical symptoms and signs, requiring close monitoring and conservative treatment; Grade C, peritoneal irritation and related abdominal infection, requiring reoperation. 5 However, rectal AL and colonic AL should be considered separately due to their differences in anatomy and anastomosis techniques. An expert consensus from a Delphi procedure agreed that the general definition of colorectal AL can follow the ISREC definition. 6 Besides, it is recommended that AL can be graded by ISREC and Clavien-Dindo classifications simultaneously. The clinical diagnosis of AL is based on the comprehensive judgment from many aspects, such as clinical manifestations, laboratory results, imaging examination, and surgical exploration. The details are as follows: (1) Recurrent fever and signs of persistent abdominal pain or peritonitis with increased inflammatory indicators after surgery; (2) Observation of fecal matter or pus from the abdominal (pelvic) drainage tube or surgical incision; (3) Indication of radiography or CT examination; and (4) Detection of colonoscopy or surgical exploration or digital rectal examination.
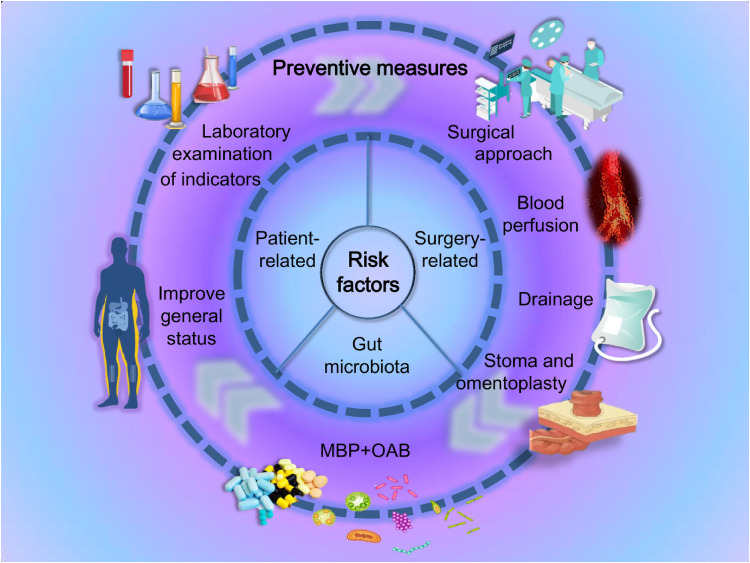
Several studies have identified risk factors of AL, mainly including general condition and laboratory examination of patients and surgery-related factors. Poor general conditions can reduce patients’ tolerance to surgery and delay their recovery. In addition, abnormal laboratory values may be an indication of postoperative complications. In terms of surgery-related factors, surgical technique, blood perfusion, and anastomotic tension are key factors affecting anastomotic healing outcomes. Recently, the relationship between gut microbiota and AL has attracted extensive attention, prompting a rethinking of traditional risk factors for AL. Since the gut microbiota plays an important role in the development of AL, the research on this topic is a new direction in AL prevention. Through early identification of risk factors and prediction of AL occurrence, timely attention can be paid to high-risk groups for targeted prevention, thus minimizing AL. In this review, we summarize patient-related (including laboratory examination) and surgery-related risk factors for AL, focusing on the potential value of gut microbiota. Moreover, some possible prophylaxis measures for AL are introduced and recommended based on risk factors (Figure 1).
Figure 1.
Preventive measures for anastomotic leak (AL) in terms of patient, surgery, and gut microbiota.
Abbreviations: MBP, mechanical bowel preparation; OAB, oral antibiotics.
General Condition and Laboratory Examination of Patients
General Condition of Patients
Male, obesity, malnutrition, hypoproteinemia, diabetes, preoperative neoadjuvant chemoradiotherapy, and other risk factors may exert an unfavorable effect on AL. Male patients usually have a narrower pelvis than female patients, leading to inadequate exposure to the surgical field and limited operation in rectal surgery. 7 A retrospective study reported that pelvic dimensions may be useful predictors for AL. 8 Another possible reason why male patients have a higher AL rate is that more visceral fat in males may negatively affect the metabolism and surgery than that in females. 9 Surgery for obese patients is more challenging and obesity is a significant risk factor for AL. 10 Malnutrition may cause prolonged anastomotic healing time, and it is considered to be a risk factor of AL. 11 In addition, lower preoperative and early postoperative serum albumin levels could signify AL,12,13 in which nutritional support should be provided. Diabetic patients have metabolic disorders and weak anti-infection ability, resulting in delayed healing of anastomosis and AL. A meta-analysis showed that diabetes was significantly and independently associated with an increased risk of AL during colorectal surgery. 14 However, a prospective cohort study suggested that AL was associated with intraoperative hyperglycemia rather than diabetes. 15 Neoadjuvant chemoradiotherapy can reduce tumor load and tumor stage and is recommended for some advanced patients. A study assessed the extent of anastomotic margin damage in patients undergoing anterior resection with neoadjuvant chemoradiotherapy. The results indicated that radiotherapy affected microvessel density and caused radiation injury of anastomosis. 16 Moreover, a randomized controlled trial (RCT) analysis of patients after rectal cancer resection suggested that preoperative radiotherapy was also associated with anastomotic stenosis. 17 Contrary to these findings, a meta-analysis reported that neoadjuvant therapy did not increase the incidence of AL. 18 The correlation between neoadjuvant therapy and AL needs further studies to confirm. Other risk factors, such as the use of corticosteroids and nonsteroidal anti-inflammatory drugs (NSAIDs), alcohol overdose, smoking, and anemia, appear to be associated with AL,19–22 which also require more warranted evidence.
Based on the above studies, the patients themselves can implement some modifiable measures, such as smoking and alcohol cessation and weight control. A retrospective study showed that the duration of hospitalization can be reduced by preoperative smoking cessation. 23 Meanwhile, the surgeons need to prioritize the control of patients’ basic diseases and the improvement of their general status before surgery. For example, by regulating blood glucose and blood pressure, correcting water and electrolyte disorders, and providing proper nutritional supplements, patients can have better operation tolerance and organ protection. These measures aiming to improve patients’ general condition may be helpful for AL prevention. Besides, a prospective RCT reported that early enteral nutrition after rectal surgery could stimulate intestinal function, reducing postoperative AL and ileus. 24 Improving the nutritional status of patients in the early postoperative period is also important. Another study revealed that AL incidence was reduced by changing clinical measures, including the avoidance of NSAIDs, maintenance of intraoperative fluid balance, and the exclusion of emergency resection with primary anastomosis. 25 These measures seem to be associated with the improvement of patients’ general condition. However, the association between the use of NSAIDs and AL is not confirmed. For instance, an observational study on anterior resections for rectal cancer suggested that NSAIDs treatment did not increase the risk of AL. 26
Laboratory Examination
Some inflammatory indicators are valuable for the early identification of AL because the inflammation of the anastomotic site occurs earlier than AL. Detection of inflammatory indicators in blood or drainage fluid, such as white blood cell (WBC), C-reactive protein (CRP), procalcitonin (PCT), and related chemokines, is not only convenient and timely but also cheap and less harmful. These inflammatory indicators are usually detected preoperatively and postoperatively.
Preoperative detection of inflammatory indicators is useful for the recognition of high-risk anastomoses, by which surgeons could provide early surveillance for patients. A retrospective study on patients undergoing left-sided CRC resection suggested that preoperative WBC count was an effective predictor of AL. 27 The AL group had a considerably higher WBC number than that in the non-AL group before surgery. In CRC patients with AL, a matched case–control study indicated that the preoperative serum levels of C-X-C motif chemokine 6 (CXCL6) and C-C motif chemokine 11 (CCL11) were elevated for rectal cancer, and the preoperative level of high-sensitivity CRP was increased for colonic cancer, respectively. 28 These preoperative indicators may be potentially associated with AL.
Apart from preoperative indicators, postoperative indicators are also helpful for earlier diagnosis of AL. Through timely detection and comprehensive analysis of postoperative indicators, possible intervention and appropriate treatment can be performed. The elevation of PCT on the first postoperative day was found to be an effective indicator for the early prediction of AL. 29 A multicenter prospective study showed that the detection of CRP on the fourth postoperative day of CRC could improve the accuracy of AL diagnosis compared to PCT or neutrophil count, and a high negative predictive value of CRP could guide the safe discharge time of patients. 30 In addition, combined detection of CRP trajectory and cut-off points may be also valuable in accurate diagnosis of AL. 31 Apart from detecting blood inflammation indicators, a recent study suggested that high bacteriological concentration in drainage fluid was a reliable indicator for the prediction and diagnosis of AL. 32 When AL occurred, gut bacteria moved into the abdominal cavity, contributing to increased bacterial concentrations in the drainage fluid. Moreover, a multicenter cohort study examined relevant blood and drainage fluid indicators. The results suggested that the combined detection of serum CRP and matrix metalloproteinase-9 (MMP-9) in drainage fluid may help predict AL earlier. 33 This combined measurement may be beneficial to improving the accuracy of AL diagnosis.
However, detection of inflammatory indicator alone is insufficient for AL diagnosis and needs to be combined with other aspects. The sensitivity and specificity of indicators and the optimal time for detection also require more assessment.
Surgery-Related Factors
Surgical Approach and Anastomosis Type
Emergency surgery is usually performed when a patient develops acute peritonitis or ileus without adequate preoperative preparation, which complicates the operation and increases the risk of postoperative complications. A retrospective study reported that emergency surgery was an independent risk factor for AL or death after AL. 34 For elective surgery, laparoscopic surgery is gaining more attention than open surgery. Several studies have shown no significant difference in surgical safety, postoperative recurrence rate, and disease-free survival rate between open and laparoscopic surgery.35–37 Although laparoscopic surgery may suffer from a lack of touch or inadequate cutting angles, it offers advantages in intraoperative bleeding reduction, precise nerve protection, and pain relief for patients. A retrospective study reported that laparoscopic and robotic surgery significantly reduced the risk of postoperative complications (including AL) and length of hospital stay compared with open surgery after colectomy. 38 Both laparoscopic and robotic surgery are labeled as minimally invasive, with the possibility of better clinical outcomes from their continued use. Another study found that in certain rectal cancer patients at high risks (eg, males, high BMI, and T4 stage), the surgical method was an independent predictor of AL, and that robotic surgery showed possible advantages in AL prevention compared to laparoscopic surgery. 39 Those selective patients need higher surgical precision, making robotic surgery more appropriate for them. Moreover, robotic surgery was also reported to have reduced operating time, decreased conversion rates, and less blood loss than laparoscopic surgery in patients with high-risk rectal cancer. 40 However, a multicenter RCT (ROLARR study) showed that the rate of conversion to open laparotomy was not significantly reduced in patients with robotic rectal cancer resection than in those with laparoscopic rectal cancer resection. 41 There was also no statistical difference in complications and 30-day mortality between robotic and laparoscopic rectal cancer surgeries. 41 The advantages of robotic surgery need more rigorous evaluation prior to widespread application.
The anastomosis of different intestinal tubes has varying impacts on AL due to different physiological characteristics. In intestinal reconstruction in CRC, ileum-colon anastomosis, colon-colon anastomosis, and colon-rectal (anal) anastomosis are generally performed. Previous studies have shown that low rectal anastomosis is an independent risk factor for AL. 42 A low colon-rectal (anal) anastomosis in the limited pelvic space may complicate the operation, resulting in high tension and poor blood flow of the anastomosis. These disadvantages are associated with the high rate of AL in rectal cancer surgery. Ileum-colon anastomosis and colon-colon anastomosis are usually applied in colon cancer surgery. Compared with rectal anastomoses, colon anastomoses have a significantly lower risk of AL. 43
According to different anastomosis techniques, the anastomosis can also be divided into stapled anastomosis, hand-sewn anastomosis, end-to-end anastomosis, end-to-side anastomosis, side-to-side anastomosis, intracorporeal anastomosis, and extracorporeal anastomosis. A systematic review including six RCTs suggested a lower incidence of AL in stapled ileum-colon anastomosis compared with hand-sewn anastomosis. 44 Similarly, a retrospective study showed that stapled ileum-colon anastomosis was associated with less leakage. 45 The stapled anastomosis was considered to have reduced tissue manipulation and less intraoperative contamination. However, another study reported a higher AL rate for stapled ileum-colon anastomosis than for hand-sewn anastomosis. 46 Besides, a retrospective study investigated whether the type of anastomosis affects the severity and treatment of AL. The results indicated that patients with hand-sewn anastomosis had fewer clinical complications and received more conservative treatments than those with stapled anastomosis. 47 One reason is that stapled anastomosis with a single-layer technique may not be as safe as hand-sewn anastomosis. These results appear to be contradictory in determining the preferred anastomosis technique.
A comparison of side-to-side, end-to-end, and end-to-side anastomosis is shown in Table 1. Side-to-side anastomosis is a safe and reliable technique with a wider diameter of anastomosis and better blood supply. The ileocolonic side-to-side anastomosis was reported to have a shorter operation time and fewer anastomotic errors (anastomotic bleeding and incomplete incision margin) than end-to-side anastomosis, but the incidence of AL is similar. 48 However, some studies suggested that the incidence of AL is higher in side-to-side anastomosis than in end-to-side anastomosis due to the number of cutting times and the length of the stitching line. 45 A recent meta-analysis of six RCTs described that end-to-side anastomosis reduced the risk of AL compared to end-to-end anastomosis after low anterior resection. 49 Another retrospective study was consistent with this conclusion, demonstrating that end-to-side anastomosis outperformed end-to-end anastomosis in vascular perfusion. 50 Although the AL rate varies between different types of anastomosis, they are all designed to ensure tension-free and adequate anastomotic blood perfusion. Careful inspection and reinforcement of the anastomosis are necessary prior to the completion of the anastomosis. A meta-analysis enrolling 1957 patients showed no difference between intracorporeal and extracorporeal anastomosis in postoperative complications and AL. However, intracorporeal anastomosis had the advantages of less bleeding, smaller incisions, and faster postoperative recovery. 51
Table 1.
Comparison of Different Anastomosis Methods.
| Applications | Instruments | Advantages | Disadvantages | |
|---|---|---|---|---|
| Side-to-side (functional end-to-end anastomosis) | Ileocolonic and colon-colon anastomosis | Linear cutter stapler |
|
|
| End-to-side | All types of anastomosis | Circular stapler |
|
Possible more anastomotic bleeding and inadequate donuts compared to side-to-side anastomosis. |
| End-to-end | Mainly colorectal (anal) anastomosis | Double-stapled technique (circular stapler and linear cutter stapler) | Compliance with physiological intestinal motility requirements. |
|
Laparoscopic and robotic surgeries are recommended as the preferred surgical approaches for suitable patients due to their safe and minimally invasive concept. Although the incidence of AL in stapled and hand-sewn anastomosis is controversial, the use of stapled anastomosis in laparoscopic surgery may take less time. 52 Besides, selecting the appropriate anastomotic device and performing adequate compression before anastomosis are necessary. The type of anastomosis is determined by the surgical approach, intestinal tension, and the experience of the surgeon. After the completion of anastomosis, the anastomosis needs to be carefully examined to minimize AL. A retrospective study inspected the anastomotic integrity of patients by air leak tests, showing a significant decrease in the incidence of AL in the testing group. 53 A positive air leak test needs to be seriously conducted with the additional anastomotic repair. Intraoperative colonoscopy can also be performed to assess anastomotic integrity, offering the advantage of direct visualization of the anastomosis. Patients undergoing anterior resection using intraoperative colonoscopy had lower AL rates than in those who did not use colonoscopy. 54 In addition, the treatment of reinforcing sutures as needed seems to be feasible, which can reduce AL rates in patients with rectal cancer surgery. 55
Assessment of Blood Supply
Favorable blood perfusion is one of the key factors in anastomotic healing. The location of vessel ligation can affect the blood supply to the anastomosis and the incidence of postoperative AL. There is still no consensus on the level of ligation of the inferior mesenteric artery (IMA) in CRC surgery. Two ligation methods are usually involved: one is the direct ligation at the root of IMA, allowing for complete removal of surrounding lymph nodes and ensuring the radical resection of the tumor; the other is to preserve the left colic artery (LCA) with ligation at the distal end of the IMA. The second method can improve anastomotic blood perfusion and decrease the rate of AL due to the vascular supply from the LCA. 56 Nevertheless, preservation of the LCA may result in lymph node remnants, making a high degree of lymph node dissection necessary for the IMA root and surrounding LCA. A recent meta-analysis compared the outcomes of 2 IMA ligation methods. The results showed that the group of low ligation (LCA preservation) combined with high lymph node clearance had a lower AL rate, longer operative time, less hospital stay, and fewer total lymph nodes than the group of high ligation (no LCA preservation), but the lymph nodes around IMA were comparable. 57 Although lymph node dissection may require more operative time, it also reduces the possibility of lymphatic tumor metastasis. In addition, the equivalent amount of IMA root lymph nodes in both groups also indicated the effectiveness and safety of this ligation method. Therefore, intraoperative low ligation with high lymph node dissection appears to be feasible, which may improve anastomotic perfusion and ensure radical tumor resection. However, a population-based study 58 and a meta-analysis including 5 RCTs 59 suggested that there was no association between high ligation and AL. Moreover, a nationwide cohort study on patients with rectal cancer reported that different levels of IMA ligation had no influence on any oncological outcome. 60
In recent years, indocyanine green fluorescence imaging (ICG⁃FI) has been gradually applied to assess the blood perfusion in the anastomosis, thus providing a reference for surgeons during CRC surgery. After ICG-FA shows poor blood perfusion, the transect line can be shifted to a site with better perfusion, enhancing anastomosis healing and reducing the AL rate. An RCT including 380 patients undergoing colorectal anastomosis showed that ICG⁃FI was associated with decreased AL rates in low anastomosis (4-8 cm from the anal verge) but not in high anastomosis (9-15 cm). 61 A prospective, multicenter clinical trial (PILLAR II) reported the use of ICG⁃FI in 147 patients undergoing left-sided colectomy and anterior resection. Among these patients, 11 changed their surgical plan due to inadequate anastomotic blood perfusion, resulting in no subsequent AL. 62 Furthermore, a recent meta-analysis involving 11,047 CRC patients indicated that ICG⁃FI was a safe, simple, and convenient technique for the effective assessment of blood perfusion, and its intraoperative application reduced the incidence of AL. 63 Previously, the blood flow of anastomosis was mostly confirmed by observing the intestine color and palpating the vascular pulsation, which depended on the subjective experience of the surgeons. The application of ICG can facilitate the localization of the disconnected intestinal canal, assess the blood perfusion of the anastomosis, and avoid distal ischemia, demonstrating its potential in AL prevention. However, a multicenter RCT (PILLAR III study) reported that the use of ICG⁃FI did not decrease the incidence of clinical AL and added no evident advantage in patients undergoing low anterior resection. 64 Furthermore, an ongoing RCT (IntAct study), aiming to recruit 880 patients, may provide more valuable references for the efficacy of ICG⁃FI. 65 Therefore, more rigorous confirmation is required before the wide application of ICG⁃FI.
Besides ICG⁃FI, laser Doppler flowmetry is also a useful method to evaluate mucosal blood perfusion. A prospective study aimed to compare intestinal blood perfusion measured by laser Doppler flowmetry before bowel manipulation and after vascular ligation. 66 The authors found that there was a linear correlation between reduction in blood perfusion at the rectal stump and the incidence of AL. In another study, the blood flow measurements with laser Doppler flowmetry were made preoperatively and postoperatively, aiming to compare blood flow changes around the anastomosis in relation to total mesorectal excision (TME) and partial mesorectal excision (PME). 67 The results showed that TME group had a decreased blood flow of the rectum postoperatively than PME group, which may indicate the different AL rates in TME and PME.
Intraoperative Drainage
Whether pelvic (or abdominal) drainage could reduce the incidence of AL is controversial. A multicenter RCT involving 494 patients with rectal surgery reported no correlation between drainage placement and postoperative complications, reoperation rate, or length of stay, suggesting that the pelvic drainage has no validated benefits and routine efficacy. 68 More seriously, a meta-analysis including 4 RCTs demonstrated that pelvic drainage might even increase the risk of postoperative ileus. 69 Different views consider pelvic drainage as a safe and effective way to prevent postoperative AL and reoperation. 70 In patients who underwent anterior rectal resection, the placement of pelvic drainage showed a lower rate of AL and reintervention compared with those without placement. Drainage around the anastomosis can reduce the accumulation of fluid and abscess, thereby promoting the healing of anastomosis and preventing the occurrence of AL. Moreover, the severity of AL can be moderately alleviated by drainage and flushing of the tube, which may decrease the necessity for additional surgery. For example, a retrospective study reported that the placement of double-lumen irrigation-suction tube was associated with decreased rate of grade C AL in patients undergoing sigmoid colon cancer radical resection. 71
Transanal drainage tube placement has also received increasing attention. The pressure in the anal canal is elevated after surgery, and the tension in the anastomotic can be reduced by placing a transanal drainage tube. The tube placement also allows the drainage of bowel contents and reduces contamination at the anastomosis, decreasing the incidence of AL. A recent meta-analysis including 2 RCTs and 13 observational studies showed that the incidence of AL was reduced in patients undergoing anterior resection with the placement of a transanal tube than those who did not, indicating that the use of a transanal tube can be considered for preventing AL. 72 Besides, postoperative fecal volume from the tube was a possible predictor of AL. 73 AL rates were higher in patients with a daily fecal volume of more than 100 mL /day for 2 or more days than in those of less than 2 days. 73 Moreover, the placement of a transanal tube may help cure confined peritonitis caused by fecal leakage into the abdominal cavity, thus reducing the reoperation rates of AL. 74 However, 2 recent multicenter RCTs suggested no significant benefit of the transanal tube for preventing AL after laparoscopic rectal cancer surgery.75,76 Furthermore, the material of the drainage tube, the depth of placement, and the duration of placement are generally determined by surgeon preference rather than a uniform consensus. A study evaluating the different outcomes of 3 types of transanal tubes showed that patients undergoing rectal cancer surgery with Fr32 silicone tubes had the lowest incidence of AL and the best drainage outcomes. 77
Although the debates continue, pelvic drainage tubes can be routinely placed during surgery because of their ability to limit the development of infection and alleviate the deterioration of AL. Once the anastomosis breaks down, the presence of drainage offers the possibility of conservative treatment. Besides, as displayed in chapter 2.2, the laboratory examination of drainage fluid can help predict the occurrence of AL. As for transanal tube placement, it is time-saving, cost-effective, and less harmful, which may be a viable option for patients with high-risk factors. However, given the data from the 2 RCTs,75,76 the recommendation of drainage tubes should be cautious.
Prophylactic Stoma and Omentoplasty
There has been no consensus on whether a prophylactic stoma will reduce the incidence of AL. A retrospective study suggested that prophylactic stoma was not associated with AL but reduced its severity and the necessity for secondary surgery. 78 Another meta-analysis of 5 RCTs found that patients without prophylactic stoma after rectal cancer surgery had a higher incidence of AL than those with a stoma, while the incidence of other postoperative complications was lower. 79 It is recognized that prophylactic stoma can reduce the extent of serious complications such as AL-associated peritonitis by diverting intestinal contents, thereby reducing the possibility of urgent abdominal reoperation. It is beneficial for patients with a high risk of developing AL. In addition, the quality of life, stoma complications, and economic burden of patients who undergo stoma need to be focused on. A recent meta-analysis compared the postoperative complications of ileostomy and colostomy in patients undergoing anterior resection. The results showed that although ileostomy may be associated with ileus and hydroelectrolyte disturbances, the incidence of stoma prolapse and retraction, incisional hernia, infection, and parastomal hernia was lower, making the ileostomy more recommendable. 80 Moreover, prophylactic stoma may increase the risk of postoperative permanent stoma in patients with anterior resection. 81 Therefore, a routine prophylactic stoma is not advisable. The decision to perform a prophylactic stoma requires an assessment of the patient's risk factors, a judgment of the procedure's progress, and the experience of the surgeons.
In addition to the prophylactic stoma, omentoplasty has become a new technique to prevent AL by wrapping the anastomosis with an omentum, providing additional support to strengthen anastomosis and improve neovascularization. 82 A prospective RCT showed a significant reduction in the incidence of AL, postoperative infection, and peritonitis in patients after intestinal resection and omentoplasty. 83 However, several studies suggested that this technique was ineffective in reducing the incidence of AL.84,85 Additionally, a meta-analysis reported that omentoplasty significantly reduced the incidence of AL in the esophageal anastomosis but not in colorectal anastomoses. 86 There are few studies on this new technique, and more evidence is needed to demonstrate its effectiveness in reducing the AL rate.
Gut Microbiota
The Relationship Between Microbiota and AL
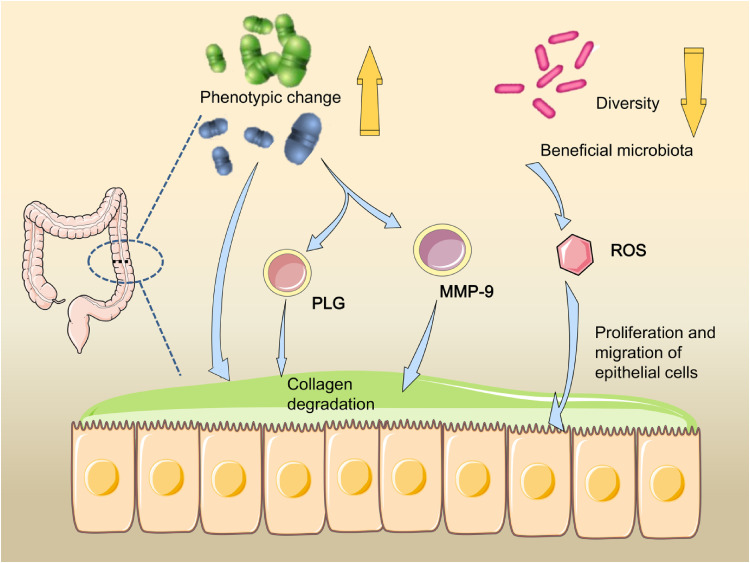
The underlying causes and molecular mechanisms of AL have not been fully elucidated. Even a patient without any risk factors may develop AL after surgery without technical errors. Such errors cannot be attributed to the individual patient's problems or the surgeon's technique factors. In recent years, the incidence of AL has not decreased significantly with medical advances. Hence, the relationship between gut microbiota and AL gradually receives more concern, and the dysbiosis of gut microbiota may significantly affect AL. Intestinal repair and microbiota adaptation are generally in dynamic homeostasis. When the homeostasis is not maintained, the number and type of microbiota change accordingly, and some possible adverse events (eg, AL) occur, further deepening the imbalance. The specific mechanism of gut microbial dysbiosis causing AL is unclear, and the following mechanisms are currently possible (Figure 2).
Figure 2.
The possible mechanisms of gut microbiota on anastomotic leak (AL). The altered phenotype makes the strain aggressive and virulent, activating MMP-9 and PLG, causing collagen degradation and AL. Besides, low microbiota diversity and decreased beneficial microbiota may reduce the proliferation and migration of ROS-mediated epithelial cells, thereby delaying anastomotic healing.
Abbreviations: MMP-9, matrix metalloproteinase-9; PLG, plasminogen; ROS, reactive oxygen species.
First, the diversity of gut microbiota has a correlation with AL. Reduced diversity may cause an increase in undesirable microbiota and a decrease in beneficial microbiota, resulting in compromised intestinal resistance and an additional risk of adverse effects. Van Praagh et al 87 found that the microbial diversity in the anastomotic tissue of patients who developed AL was low, with a high abundance of Lachnospiraceae and Bacteroidaceae. These 2 strains are associated with the degradation of mucin, which is a major component of the intestinal mucus layer. Functioning mucus layer is necessary for intestinal protection and its disruption leads to impaired anastomotic healing. 88 In addition, some species of microbiota are proven to promote anastomotic healing. For example, Lactobacillus could promote the secretion of reactive oxygen species (ROS) by NADPH oxidase 1 (Nox1), 89 enhancing the proliferation and migration of epithelial cells and maintaining the intestinal barrier. 90 Besides, some beneficial microbiota, such as Lactobacillus, are identified to directly inhibit pathogens. 91 A study showed that the abundance of Lactobacillus was greatly reduced after intestinal anastomotic surgery in rats. 92 Once these beneficial microbiotas are reduced, the anastomosis healing may also be affected.
Another mechanism is the phenotypic change in the gut microbiota. After some external or internal stimulus, the microbiota transforms into an aggressive, virulent strain. In addition, a 500-fold increase in the Enterococcus genus was found in rat intestinal resection anastomosis tissue. 92 Among its strains, Enterococcus faecalis was demonstrated to promote collagen degradation and activate MMP9 in host intestinal tissues, leading to the development of AL. 93 E faecalis can also bind and hyperactivate human plasminogen (PLG), producing supraphysiologic collagenolysis. 94 The high collagen degrading activity of strains was associated with their transformed phenotype, characterized by stronger virulence. In a rat model receiving preoperative radiotherapy for colectomy, Olivas et al 95 found the incidence of AL was higher in the group treated with radiotherapy and colonization of Pseudomonas aeruginosa than in the group with only radiotherapy. The reason may be that the radiotherapy-induced phenotypic changes in the strain, exhibiting enhanced collagenase activity, high invasiveness, and destruction of intestinal epithelial cells. 95 Moreover, morphine can also induce the transformation of P aeruginosa to a more virulent phenotype, leading to the destruction of the intestinal mucus layer. 96 By analyzing the strain composition in tissue specimens from CRC patients, Mima et al 97 reported a correlation between the high abundance of Bifidobacterium genus and increased risk of postoperative AL. The authors speculated that Bifidobacterium might contribute to the development of AL by affecting the blood flow and oxygen supply to the anastomosis or other intestinal microbiota, causing poor perfusion and tissue hypoxia. 97 This indirect speculation needs to be supported by more direct evidence.
By detecting the composition and alteration of the intestinal microbiota in patients, the relevant microbiota can be used as a marker for predicting AL. More microbiota associated with AL need to be clarified to explore their interactions and their role in the development of AL, thus providing new opportunities for the treatment of AL by targeting their underlying mechanisms.
Gut Microbiota in AL Prevention
The dysbiosis of gut microbiota causing AL may be affected by multiple factors in the perioperative period, and the reasonable control or exploitation of these factors can help prevent AL.
Preoperative mechanical bowel preparation (MBP) generally involves oral laxatives and warm saline enemas. It allows the cleanses of the intestines, reducing intestinal contents and facilitating surgical operations. However, studies showed that MBP affected the composition of intestinal microbiota and reduced their diversity, 98 which may impact the development of AL. Whether MBP can be routinely performed preoperatively is controversial. A meta-analysis concluded that MBP had no advantage in reducing the risk of AL, 99 and another study suggested no effect of MBP on the clinical severity of AL. 100 Therefore, it may be feasible to remove preoperative MBP, thus eliminating the effects of MBP on the patient's intestinal microbiota. Moreover, the use of antibiotics also affects intestinal microbiota, including their composition, diversity, and resistance. The effects have a long duration, which reduces AL by acting on pathogens detrimental to intestinal recovery and anastomotic healing. 101 In a rat model, Shogan et al 93 reported that topical application of antibiotics to the anastomosis effectively eliminated E faecalis and limited its role in tissue degradation, thus preventing AL. They further demonstrated that the application of the intravenous antibiotic had no such effect on rats and failed to achieve complete clearance of E faecalis from the anastomotic tissues in patients with colon surgery. These results suggested that the effectiveness of antibiotics may be affected by different ways of application. A recent meta-analysis including 6 RCTs indicated that oral antibiotics (OABs) with intravenous antibiotics significantly reduced the incidence of surgical site infections and AL in patients with laparoscopic CRC surgery compared with intravenous antibiotics alone. 102 Both oral and intravenous antibiotics are necessary for AL prevention. Additionally, selective antibiotics may be more effective in reducing AL than broad-spectrum antibiotics. 103 The reason may be that broad-spectrum antibiotics could lead to a broader clearance of microbiota and the dysbiosis of a patient's intestinal microbiota, while selective antibiotics could be more targeted to specific pathogens including aerobic Gram-negative bacteria, Staphylococcus aureus, and fungi. 104 Oral selective antibiotics, such as tobramycin, colistin, and amphotericin B, can be administered to decontaminate the digestive tract. 104 Thus, the benefits of the preoperative prophylactic application of selective antibiotics may be greater than its adverse effects on intestinal microbiota. To decrease the incidence of surgical site infection and AL, patients are advised to receive preoperative selective oral and intravenous antibiotic prophylaxis, and routine administration of MBP remains controversial and needs to be supported by more evidence.
In addition to MBP and antibiotics, some new adjuvant methods acting on the gut microbiota have been explored and applied for AL prevention. Tranexamic acid (TXA) enema could inhibit pathogen-mediated PLG activation and reduce collagen degradation at the anastomosis, thereby preventing AL in mice. 105 Depletion of intestinal phosphate concentrations was demonstrated to convert P aeruginosa to a more virulent phenotype. 106 Oral polyphosphate was an alternative method of AL prevention, which attenuates virulence and decreases collagenase activity of intestinal pathogens in mice, promoting healing of pathogen-induced AL. 107 Probiotics are dietary supplements of live microorganisms that maintain the balance of intestinal microbiota for beneficial effects, and prebiotics can promote the growth and function of probiotics. Synbiotics are a combination of probiotics and prebiotics. Sapidis et al 108 combined synbiotics with glutamine to evaluate anastomosis healing in rats, which showed reduced bacterial translocation, improved anastomotic mechanical strength, and decreased risk of AL. An RCT revealed that perioperative administration of probiotics significantly reduced the incidence of surgical site infection and AL after colorectal surgery, 109 possibly due to partial restoration of microbial diversity and effects on immune function. 110 The underlying mechanism of probiotics on AL needs to be further explored.
Although the above results need to be validated in further studies, the gut microbiota certainly has unique advantages and potential in predicting and treating AL. The occurrence of AL is not solely due to patient-related risk factors and the surgeon's surgical technique but is influenced by various factors such as gut microbiota. In-depth exploration of the mechanisms underlying the role of gut microbiota in AL is valuable for overcoming the challenge of AL.
Discussion
As a detrimental complication after colorectal surgery, AL has been a key concern and hardship for surgeons. Raising the awareness of risk factors for AL can be worthwhile in preventing AL. With increasing risk factors for AL, surgeons need to be more alert to the occurrence of AL and prevent it in all aspects. Preoperatively, the basic physical condition of the patient needs to be improved. Intraoperatively, careful attention should be paid to the scope of bowel resection, preservation of blood vessels, selection of suitable surgical instruments and anastomotic methods, and inspection of the anastomosis. Additionally, routine pelvic drainage is recommended due to its advantage for the prediction and treatment of AL, while prophylactic stoma and the placement of transanal drains are dependent on the situation. Postoperatively, the relevant indicators have to be closely detected, and different treatments could be given according to the severity of AL.
Timely predictive indicators, convenient and fast operating instruments, effective methods for blood perfusion assessment, and utilization of microbiota in AL are prospects worthy of further efforts. Predictive indicators can be obtained from plasma and drainage fluid, and multiple combined detections may provide a more accurate reference. The application of different staplers has simplified surgical procedures and improved surgical techniques, promoting gastrointestinal reconstruction. Designing new instruments suitable for surgery with minimal tissue damage can reduce postoperative complications. Since adequate anastomotic blood perfusion is a prerequisite for healing, the real-time and objective intraoperative assessment of anastomotic perfusion is particularly critical. New techniques for evaluating perfusion need to be extensively developed. Microbiota is associated with AL and functions as a key factor in various diseases. Regulating microbial diversity and manipulating intrinsic mechanisms can achieve more beneficial effects on human health. More promisingly, patients can personally target their treatment according to the individual layout of microbiota.
Although considerable progress has been made in the prevention and treatment of AL, some of the following limitations remain. First, the diagnostic criteria for AL are not consistent among different centers. Some insidious asymptomatic AL may be unrecognized, resulting in different reported incidences of AL and biased results of related studies. In addition, most of the indicators for predicting AL have no uniform optimal critical values. Only statistically significant correlation cannot be widely used in clinical applications. The changes in indicators are also affected by surgery, blood transfusions, and drugs, which need to be combined with the specific situation of patients in clinical practice to provide guiding value for diagnosing AL. Moreover, multiple risk factors for the eventual occurrence of AL may interact with each other. Therefore, more basic studies are required to clarify the underlying causes of AL, especially the relationship between gut microbiota and AL. Finally, the current explanation of AL risk factors is incomplete because most studies are limited to single-center, small-sample, and retrospective. Designing and implementing more multicenter, large-sample, prospective trials are promising in the future.
Conclusion
Early diagnosis and prevention of AL based on the identification of risk factors is reliable. Apart from the current risk factors for AL, understanding the role of gut microbiota in AL and the molecular mechanism of AL development is crucial for the prevention and treatment of AL. With the development of new technologies and a deeper understanding of AL at the molecular level, the challenges of AL will eventually be overcome.
Abbreviations
- AL
anastomotic leak
- CRC
colorectal cancer
- CRP
C-reactive protein
- CXCL6
C-X-C motif chemokine 6
- CCL11
C-C motif chemokine 11
- IL
interleukin
- IMA
inferior mesenteric artery
- ICG⁃FI
indocyanine green fluorescence imaging
- ISREC
International Study Group of Rectal Cancer
- LCA
left colic artery
- MBP
mechanical bowel preparation
- MMP-9
matrix metalloproteinase-9
- Nox1
NADPH oxidase 1
- NSAIDs
nonsteroidal anti-inflammatory drugs
- OABs
oral antibiotics
- PCT
procalcitonin
- PLG
plasminogen
- PME
partial mesorectal excision
- RCT
randomized controlled trial
- ROS
reactive oxygen species
- TME
total mesorectal excision
- TXA
tranexamic acid
- WBC
white blood cell
Footnotes
Declaration of Conflicting Interests: The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Education Project of Jilin University, the Financial Department of Jilin Province, Science and Technology Department of Jilin Province, and National Natural Science Foundation of China, (grant numbers 419070600046 and 45121031D024, No. 2019SCZT045, No. YDZJ202201ZYTS004, and No. 32000953).
ORCID iD: Jiannan Li https://orcid.org/0000-0001-9744-7666
References
- 1.Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209-249. doi: 10.3322/caac.21660. [DOI] [PubMed] [Google Scholar]
- 2.Boström P, Haapamäki MM, Rutegård J, Matthiessen P, Rutegård M. Population-based cohort study of the impact on postoperative mortality of anastomotic leakage after anterior resection for rectal cancer. BJS Open. 2019;3(1):106-111. doi: 10.1002/bjs5.50106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Jutesten H, Buchwald P, Angenete E, Rutegard M, Lydrup ML. High risk of low anterior resection syndrome in long-term follow-up after anastomotic leakage in anterior resection for rectal cancer. Dis Colon Rectum. 2021;65(10):1264-1273. doi: 10.1097/dcr.0000000000002334 [DOI] [PubMed] [Google Scholar]
- 4.Ha GW, Kim JH, Lee MR. Oncologic impact of anastomotic leakage following colorectal cancer surgery: a systematic review and meta-analysis. Ann Surg Oncol. 2017;24(11):3289-3299. doi: 10.1245/s10434-017-5881-8. [DOI] [PubMed] [Google Scholar]
- 5.Rahbari NN, Weitz J, Hohenberger W, et al. Definition and grading of anastomotic leakage following anterior resection of the rectum: a proposal by the international study group of rectal cancer. Surgery. 2010;147(3):339-351. doi: 10.1016/j.surg.2009.10.012. [DOI] [PubMed] [Google Scholar]
- 6.van Helsdingen CP, Jongen AC, de Jonge WJ, Bouvy ND, Derikx JP. Consensus on the definition of colorectal anastomotic leakage: a modified Delphi study. World J Gastroenterol. 2020;26(23):3293-3303. doi: 10.3748/wjg.v26.i23.3293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zhou C, Wu X-r, Liu X-h, et al. Male gender is associated with an increased risk of anastomotic leak in rectal cancer patients after total mesorectal excision. Gastroenterol Rep (Oxf). 2018;6(2):137-143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Yu ZL, Liu XH, Liu HS, et al. Impact of pelvic dimensions on anastomotic leak after anterior resection for patients with rectal cancer. Surg Endosc. 2021;35(5):2134-2143. doi: 10.1007/s00464-020-07617-1. [DOI] [PubMed] [Google Scholar]
- 9.Verduin WM, Warps AK, van den Helder R, Doodeman HJ, Houdijk APJ. Visceral fat and anastomotic leakage after colon cancer resection. Dis Colon Rectum. 2021;64(2):163-170. doi: 10.1097/dcr.0000000000001779. [DOI] [PubMed] [Google Scholar]
- 10.Nugent TS, Kelly ME, Donlon NE, et al. Obesity and anastomotic leak rates in colorectal cancer: a meta-analysis. Int J Colorectal Dis. 2021;36(9):1819-1829. doi: 10.1007/s00384-021-03909-7. [DOI] [PubMed] [Google Scholar]
- 11.Xu H, Kong F. Malnutrition-Related factors increased the risk of anastomotic leak for rectal cancer patients undergoing surgery. Biomed Res Int. 2020;2020:5059670. doi: 10.1155/2020/5059670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lai CC, You JF, Yeh CY, et al. Low preoperative serum albumin in colon cancer: a risk factor for poor outcome. Int J Colorectal Dis. 2011;26(4):473-481. doi: 10.1007/s00384-010-1113-4. [DOI] [PubMed] [Google Scholar]
- 13.Shimura T, Toiyama Y, Hiro J, et al. Monitoring perioperative serum albumin can identify anastomotic leakage in colorectal cancer patients with curative intent. Asian J Surg. 2018;41(1):30-38. doi: 10.1016/j.asjsur.2016.07.009. [DOI] [PubMed] [Google Scholar]
- 14.Lin X, Li J, Chen W, et al. Diabetes and risk of anastomotic leakage after gastrointestinal surgery. J Surg Res. 2015;196(2):294-301. doi: 10.1016/j.jss.2015.03.017. [DOI] [PubMed] [Google Scholar]
- 15.Reudink M, Huisman DE, van Rooijen SJ, et al. Association between intraoperative blood glucose and anastomotic leakage in colorectal surgery. J Gastrointest Surg. 2021;25(10):2619-2627. doi: 10.1007/s11605-021-04933-2. [DOI] [PubMed] [Google Scholar]
- 16.Qin Q, Zhu Y, Wu P, et al. Radiation-induced injury on surgical margins: a clue to anastomotic leakage after rectal-cancer resection with neoadjuvant chemoradiotherapy? Gastroenterol Rep (Oxf). 2019;7(2):98-106. doi: 10.1093/gastro/goy042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Qin Q, Ma T, Deng Y, et al. Impact of preoperative radiotherapy on anastomotic leakage and stenosis after rectal cancer resection: post Hoc analysis of a randomized controlled trial. Dis Colon Rectum. 2016;59(10):934-942. doi: 10.1097/dcr.0000000000000665. [DOI] [PubMed] [Google Scholar]
- 18.Hu MH, Huang RK, Zhao RS, Yang KL, Wang H. Does neoadjuvant therapy increase the incidence of anastomotic leakage after anterior resection for mid and low rectal cancer? A systematic review and meta-analysis. Colorectal Dis. 2017;19(1):16-26. doi: 10.1111/codi.13424. [DOI] [PubMed] [Google Scholar]
- 19.McDermott FD, Heeney A, Kelly ME, Steele RJ, Carlson GL, Winter DC. Systematic review of preoperative, intraoperative and postoperative risk factors for colorectal anastomotic leaks. Br J Surg. 2015;102(5):462-479. doi: 10.1002/bjs.9697. [DOI] [PubMed] [Google Scholar]
- 20.Huang Y, Tang SR, Young CJ. Nonsteroidal anti-inflammatory drugs and anastomotic dehiscence after colorectal surgery: a meta-analysis. ANZ J Surg. 2018;88(10):959-965. doi: 10.1111/ans.14322. [DOI] [PubMed] [Google Scholar]
- 21.Eriksen TF, Lassen CB, Gögenur I. Treatment with corticosteroids and the risk of anastomotic leakage following lower gastrointestinal surgery: a literature survey. Colorectal Dis. 2014;16(5):O154-O160. doi: 10.1111/codi.12490. [DOI] [PubMed] [Google Scholar]
- 22.Choudhuri AH, Uppal R, Kumar M. Influence of non-surgical risk factors on anastomotic leakage after major gastrointestinal surgery: audit from a tertiary care teaching institute. Int J Crit Illn Inj Sci. 2013;3(4):246-249. doi: 10.4103/2229-5151.124117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Inoue Y, Katoh T, Masuda S, et al. Perioperative complications of abdominal surgery in smokers. J Anesth. 2020;34(5):712-718. doi: 10.1007/s00540-020-02815-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Boelens PG, Heesakkers FF, Luyer MD, et al. Reduction of postoperative ileus by early enteral nutrition in patients undergoing major rectal surgery: prospective, randomized, controlled trial. Ann Surg. 2014;259(4):649-655. doi: 10.1097/sla.0000000000000288. [DOI] [PubMed] [Google Scholar]
- 25.Iversen H, Ahlberg M, Lindqvist M, Buchli C. Changes in clinical practice reduce the rate of anastomotic leakage after colorectal resections. World J Surg. 2018;42(7):2234-2241. doi: 10.1007/s00268-017-4423-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hultberg D K, Angenete E, Lydrup ML, Rutegård J, Matthiessen P, Rutegård M. Nonsteroidal anti-inflammatory drugs and the risk of anastomotic leakage after anterior resection for rectal cancer. Eur J Surg Oncol. 2017;43(10):1908-1914. doi: 10.1016/j.ejso.2017.06.010. [DOI] [PubMed] [Google Scholar]
- 27.Morimoto M, Taniguchi K, Yamamoto O, Naka T, Sugitani A, Fujiwara Y. Preoperative white blood cell count predicts anastomotic leakage in patients with left-sided colorectal cancer. PLoS One. 2021;16(10):e0258713. doi: 10.1371/journal.pone.0258713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Holmgren K, Jonsson P, Lundin C, et al. Preoperative biomarkers related to inflammation may identify high-risk anastomoses in colorectal cancer surgery: explorative study. BJS Open. 2022;6(3):zrac072. doi: 10.1093/bjsopen/zrac072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zielińska-Borkowska U, Dib N, Tarnowski W, Skirecki T. Monitoring of procalcitonin but not interleukin-6 is useful for the early prediction of anastomotic leakage after colorectal surgery. Clin Chem Lab Med. 2017;55(7):1053-1059. doi: 10.1515/cclm-2016-0736. [DOI] [PubMed] [Google Scholar]
- 30.Hernandez A S, Frasson M, García-Granero A, et al. Diagnostic accuracy of C-reactive protein, procalcitonin and neutrophils for the early detection of anastomotic leakage after colorectal resection: a multicentric, prospective study. Colorectal Dis. 2021;23(10):2723-2730. doi: 10.1111/codi.15845. [DOI] [PubMed] [Google Scholar]
- 31.Stephensen BD, Reid F, Shaikh S, Carroll R, Smith SR, Pockney P. C-reactive protein trajectory to predict colorectal anastomotic leak: PREDICT study. Br J Surg. 2020;107(13):1832-1837. doi: 10.1002/bjs.11812. [DOI] [PubMed] [Google Scholar]
- 32.Ge W, Gong HY, Xia YQ, Shao LH, Shen H, Chen G. Bacteriological concentration of peritoneal drainage fluid could make an early diagnosis of anastomotic leakage following rectal resection. Sci Rep. 2021;11(1):23156. doi: 10.1038/s41598-021-02649-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Sparreboom CL, Komen N, Rizopoulos D, et al. A multicentre cohort study of serum and peritoneal biomarkers to predict anastomotic leakage after rectal cancer resection. Colorectal Dis. 2020;22(1):36-45. doi: 10.1111/codi.14789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bakker IS, Grossmann I, Henneman D, Havenga K, Wiggers T. Risk factors for anastomotic leakage and leak-related mortality after colonic cancer surgery in a nationwide audit. Br J Surg. 2014;101(4):424-432. discussion 432. doi:10.1002/bjs.9395. [DOI] [PubMed] [Google Scholar]
- 35.Martínez-Pérez A, Carra MC, Brunetti F, de'Angelis N. Short-term clinical outcomes of laparoscopic vs open rectal excision for rectal cancer: a systematic review and meta-analysis. World J Gastroenterol. 2017;23(44):7906-7916. doi: 10.3748/wjg.v23.i44.7906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Stevenson ARL, Solomon MJ, Brown CSB, et al. Disease-free survival and local recurrence after laparoscopic-assisted resection or open resection for rectal cancer: the Australasian laparoscopic cancer of the Rectum randomized clinical trial. Ann Surg. 2019;269(4):596-602. doi: 10.1097/sla.0000000000003021. [DOI] [PubMed] [Google Scholar]
- 37.Bonjer HJ, Deijen CL, Abis GA, et al. A randomized trial of laparoscopic versus open surgery for rectal cancer. N Engl J Med. 2015;372(14):1324-1332. doi: 10.1056/NEJMoa1414882. [DOI] [PubMed] [Google Scholar]
- 38.Wei D, Johnston S, Goldstein L, Nagle D. Minimally invasive colectomy is associated with reduced risk of anastomotic leak and other major perioperative complications and reduced hospital resource utilization as compared with open surgery: a retrospective population-based study of comparative effectiveness and trends of surgical approach. Surg Endosc. 2020;34(2):610-621. doi: 10.1007/s00464-019-06805-y. [DOI] [PubMed] [Google Scholar]
- 39.Numata M, Kazama K, Onodera A, et al. Short-term outcomes following robotic-assisted laparoscopic surgery for technically demanding rectal cancer. Anticancer Res. 2020;40(4):2337-2342. doi: 10.21873/anticanres.14201. [DOI] [PubMed] [Google Scholar]
- 40.Ahmed J, Cao H, Panteleimonitis S, Khan J, Parvaiz A. Robotic vs laparoscopic rectal surgery in high-risk patients. Colorectal Dis. 2017;19(12):1092-1099. doi: 10.1111/codi.13783. [DOI] [PubMed] [Google Scholar]
- 41.Jayne D, Pigazzi A, Marshall H, et al. Effect of robotic-assisted vs conventional laparoscopic surgery on risk of conversion to open laparotomy among patients undergoing resection for rectal cancer: the ROLARR randomized clinical trial. Jama. 2017;318(16):1569-1580. doi: 10.1001/jama.2017.7219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Pommergaard HC, Gessler B, Burcharth J, Angenete E, Haglind E, Rosenberg J. Preoperative risk factors for anastomotic leakage after resection for colorectal cancer: a systematic review and meta-analysis. Colorectal Dis. 2014;16(9):662-671. doi: 10.1111/codi.12618. [DOI] [PubMed] [Google Scholar]
- 43.Jongen ACHM. Novel insights in the pathophysiology of colorectal anastomotic leakage. Maastricht University. 2019. 10.26481/dis.20190926aj [DOI]
- 44.Luglio G, Corcione F. Stapled versus handsewn methods for ileocolic anastomoses. Tech Coloproctol. 2019;23(11):1093-1095. doi: 10.1007/s10151-019-02105-8. [DOI] [PubMed] [Google Scholar]
- 45.Puleo S, Sofia M, Trovato MA, et al. Ileocolonic anastomosis: preferred techniques in 999 patients. A multicentric study. Surg Today. 2013;43(10):1145-1149. doi: 10.1007/s00595-012-0381-8. [DOI] [PubMed] [Google Scholar]
- 46.Nordholm-Carstensen A, Schnack Rasmussen M, Krarup PM. Increased leak rates following stapled versus handsewn ileocolic anastomosis in patients with right-sided colon cancer: a nationwide cohort study. Dis Colon Rectum. 2019;62(5):542-548. doi: 10.1097/dcr.0000000000001289. [DOI] [PubMed] [Google Scholar]
- 47.Espin E, Vallribera F, Kreisler E, Biondo S. Clinical impact of leakage in patients with handsewn vs stapled anastomosis after right hemicolectomy: a retrospective study. Colorectal Dis. 2020;22(10):1286-1292. doi: 10.1111/codi.15098. [DOI] [PubMed] [Google Scholar]
- 48.Liu Z, Wang G, Yang M, et al. Ileocolonic anastomosis after right hemicolectomy for colon cancer: functional end-to-end or end-to-side? World J Surg Oncol. 2014;12:306. doi: 10.1186/1477-7819-12-306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.McKechnie T, Sharma S, Daniel R, Eskicioglu C. End-to-end versus end-to-side anastomosis for low anterior resection: a systematic review and meta-analysis of randomized controlled trials. Surgery. 2021;170(2):397-404. doi: 10.1016/j.surg.2020.12.030. [DOI] [PubMed] [Google Scholar]
- 50.Kato H, Ishida T, Nitori N, et al. Efficacy of side-to-end anastomosis to prevent anastomotic leakage after anterior resection for rectal cancer. Mol Clin Oncol. 2022;16(2):44. doi: 10.3892/mco.2021.2477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Wu Q, Jin C, Hu T, Wei M, Wang Z. Intracorporeal versus extracorporeal anastomosis in laparoscopic right colectomy: a systematic review and meta-analysis. J Laparoendosc Adv Surg Tech A. 2017;27(4):348-357. doi: 10.1089/lap.2016.0485. [DOI] [PubMed] [Google Scholar]
- 52.Schineis C, Fenzl T, Aschenbrenner K, et al. Stapled intestinal anastomoses are more cost effective than hand-sewn anastomoses in a diagnosis related group system. Surgeon. 2021;19(6):321-328. doi: 10.1016/j.surge.2020.09.002. [DOI] [PubMed] [Google Scholar]
- 53.Allaix ME, Lena A, Degiuli M, et al. Intraoperative air leak test reduces the rate of postoperative anastomotic leak: analysis of 777 laparoscopic left-sided colon resections. Surg Endosc. 2019;33(5):1592-1599. doi: 10.1007/s00464-018-6421-8. [DOI] [PubMed] [Google Scholar]
- 54.Yang SY, Han J, Han YD, et al. Intraoperative colonoscopy for the assessment and prevention of anastomotic leakage in low anterior resection for rectal cancer. Int J Colorectal Dis. 2017;32(5):709-714. doi: 10.1007/s00384-017-2767-y. [DOI] [PubMed] [Google Scholar]
- 55.Maeda K, Nagahara H, Shibutani M, et al. Efficacy of intracorporeal reinforcing sutures for anastomotic leakage after laparoscopic surgery for rectal cancer. Surg Endosc. 2015;29(12):3535-3542. doi: 10.1007/s00464-015-4104-2. [DOI] [PubMed] [Google Scholar]
- 56.Tang X, Zhang M, Wang C, He Q, Sun G, Qu H. Preservation of the left colic artery and superior rectal artery in laparoscopic surgery can reduce anastomotic leakage in sigmoid colon cancer. J Minim Access Surg. 2021;17(2):208-212. doi: 10.4103/jmas.JMAS_15_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Yin TC, Chen YC, Su WC, et al. Low ligation Plus high dissection versus high ligation of the Inferior mesenteric artery in sigmoid colon and rectal cancer surgery: a meta-analysis. Front Oncol. 2021;11:774782. doi: 10.3389/fonc.2021.774782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Rutegård M, Hemmingsson O, Matthiessen P, Rutegård J. High tie in anterior resection for rectal cancer confers no increased risk of anastomotic leakage. Br J Surg. 2012;99(1):127-132. doi: 10.1002/bjs.7712. [DOI] [PubMed] [Google Scholar]
- 59.Kong M, Chen H, Xin Y, Jiang Y, Han Y, Sheng H. High ligation of the inferior mesenteric artery and anastomotic leakage in anterior resection for rectal cancer: a systematic review and meta-analysis of randomized controlled trial studies. Colorectal Dis. 2021;23(3):614-624. doi: 10.1111/codi.15419. [DOI] [PubMed] [Google Scholar]
- 60.Boström P, Hultberg DK, Häggström J, et al. Oncological impact of high vascular tie after surgery for rectal cancer: a nationwide cohort study. Ann Surg. 2021;274(3):e236-e244. doi: 10.1097/sla.0000000000003663. [DOI] [PubMed] [Google Scholar]
- 61.Alekseev M, Rybakov E, Shelygin Y, Chernyshov S, Zarodnyuk I. A study investigating the perfusion of colorectal anastomoses using fluorescence angiography: results of the FLAG randomized trial. Colorectal Dis. 2020;22(9):1147-1153. doi: 10.1111/codi.15037. [DOI] [PubMed] [Google Scholar]
- 62.Jafari MD, Wexner SD, Martz JE, et al. Perfusion assessment in laparoscopic left-sided/anterior resection (PILLAR II): a multi-institutional study. J Am Coll Surg. 2015;220(1):82-92.e1. doi: 10.1016/j.jamcollsurg.2014.09.015. [DOI] [PubMed] [Google Scholar]
- 63.Safiejko K, Tarkowski R, Kozlowski TP, et al. Safety and efficacy of indocyanine green in colorectal cancer surgery: a systematic review and meta-analysis of 11,047 patients. Cancers (Basel). 2022;14(4):1036. doi: 10.3390/cancers14041036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Jafari MD, Pigazzi A, McLemore EC, et al. Perfusion assessment in left-sided/low anterior resection (PILLAR III): a randomized, controlled, parallel, multicenter study assessing perfusion outcomes with PINPOINT near-infrared fluorescence imaging in low anterior resection. Dis Colon Rectum. 2021;64(8):995-1002. doi: 10.1097/dcr.0000000000002007. [DOI] [PubMed] [Google Scholar]
- 65.Armstrong G, Croft J, Corrigan N, et al. Intact: intra-operative fluorescence angiography to prevent anastomotic leak in rectal cancer surgery: a randomized controlled trial. Colorectal Dis. 2018;20(8):O226-o234. doi: 10.1111/codi.14257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Vignali A, Gianotti L, Braga M, Radaelli G, Malvezzi L, Di Carlo V. Altered microperfusion at the rectal stump is predictive for rectal anastomotic leak. Dis Colon Rectum. 2000;43(1):76-82. doi: 10.1007/bf02237248. [DOI] [PubMed] [Google Scholar]
- 67.Back E, Brännström F, Svensson J, et al. Mucosal blood flow in the remaining rectal stump is more affected by total than partial mesorectal excision in patients undergoing anterior resection: a key to understanding differing rates of anastomotic leakage? Langenbecks Arch Surg. 2021;406(6):1971-1977. doi: 10.1007/s00423-021-02182-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Denost Q, Rouanet P, Faucheron JL, et al. To drain or not to drain infraperitoneal anastomosis after rectal excision for cancer: the GRECCAR 5 randomized trial. Ann Surg. 2017;265(3):474-480. doi: 10.1097/sla.0000000000001991. [DOI] [PubMed] [Google Scholar]
- 69.Guerra F, Giuliani G, Coletta D, et al. A meta-analysis of randomized controlled trials on the use of suction drains following rectal surgery. Dig Surg. 2018;35(6):482-490. doi: 10.1159/000485139. [DOI] [PubMed] [Google Scholar]
- 70.Rondelli F, Bugiantella W, Vedovati MC, et al. To drain or not to drain extraperitoneal colorectal anastomosis? A systematic review and meta-analysis. Colorectal Dis. 2014;16(2):O35-O42. doi: 10.1111/codi.12491. [DOI] [PubMed] [Google Scholar]
- 71.Yang F, Liu D, Xu X, et al. A double-lumen irrigation-suction tube placed during operation could reduce the risk of grade C anastomotic leakage resulting from selective sigmoid colon cancer radical resection. Langenbecks Arch Surg. 2020;405(7):1007-1016. doi: 10.1007/s00423-020-01959-z. [DOI] [PubMed] [Google Scholar]
- 72.Choy KT, Yang TWW, Heriot A, Warrier SK, Kong JC. Does rectal tube/transanal stent placement after an anterior resection for rectal cancer reduce anastomotic leak? A systematic review and meta-analysis. Int J Colorectal Dis. 2021;36(6):1123-1132. doi: 10.1007/s00384-021-03851-8. [DOI] [PubMed] [Google Scholar]
- 73.Kawada K, Takahashi R, Hida K, Sakai Y. Impact of transanal drainage tube on anastomotic leakage after laparoscopic low anterior resection. Int J Colorectal Dis. 2018;33(3):337-340. doi: 10.1007/s00384-017-2952-z. [DOI] [PubMed] [Google Scholar]
- 74.Wang S, Zhang Z, Liu M, Li S, Jiang C. Efficacy of transanal tube placement after anterior resection for rectal cancer: a systematic review and meta-analysis. World J Surg Oncol. 2016;14:92. doi: 10.1186/s12957-016-0854-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Zhao S, Zhang L, Gao F, et al. Transanal drainage tube use for preventing anastomotic leakage after laparoscopic low anterior resection in patients with rectal cancer: a randomized clinical trial. JAMA Surg. 2021;156(12):1151-1158. doi: 10.1001/jamasurg.2021.4568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Tamura K, Matsuda K, Horiuchi T, et al. Laparoscopic anterior resection with or without transanal tube for rectal cancer patients - A multicenter randomized controlled trial. Am J Surg. 2021;222(3):606-612. doi: 10.1016/j.amjsurg.2020.12.054. [DOI] [PubMed] [Google Scholar]
- 77.Luo Y, Zhu CK, Wu DQ, Zhou LB, Wang CS. Effect comparison of three different types of transanal drainage tubes after anterior resection for rectal cancer. BMC Surg. 2020;20(1):166. doi: 10.1186/s12893-020-00811-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Shiomi A, Ito M, Saito N, et al. Diverting stoma in rectal cancer surgery. A retrospective study of 329 patients from Japanese cancer centers. Int J Colorectal Dis. 2011;26(1):79-87. doi: 10.1007/s00384-010-1036-0. [DOI] [PubMed] [Google Scholar]
- 79.Ahmad NZ, Abbas MH, Khan SU, Parvaiz A. A meta-analysis of the role of diverting ileostomy after rectal cancer surgery. Int J Colorectal Dis. 2021;36(3):445-455. doi: 10.1007/s00384-020-03771-z. [DOI] [PubMed] [Google Scholar]
- 80.Du R, Zhou J, Tong G, et al. Postoperative morbidity and mortality after anterior resection with preventive diverting loop ileostomy versus loop colostomy for rectal cancer: a updated systematic review and meta-analysis. Eur J Surg Oncol. 2021;47(7):1514-1525. doi: 10.1016/j.ejso.2021.01.030. [DOI] [PubMed] [Google Scholar]
- 81.Holmgren K, Häggström J, Haapamäki MM, Matthiessen P, Rutegård J, Rutegård M. Defunctioning stomas may reduce chances of a stoma-free outcome after anterior resection for rectal cancer. Colorectal Dis. 2021;23(11):2859-2869. doi: 10.1111/codi.15836. [DOI] [PubMed] [Google Scholar]
- 82.Hayari L, Hershko DD, Shoshani H, Maor R, Mordecovich D, Shoshani G. Omentopexy improves vascularization and decreases stricture formation of esophageal anastomoses in a dog model. J Pediatr Surg. 2004;39(4):540-544. doi: 10.1016/j.jpedsurg.2003.12.013. [DOI] [PubMed] [Google Scholar]
- 83.Nasiri S, Mirminachi B, Taherimehr R, Shadbakhsh R, Hojat M. The effect of omentoplasty on the rate of anastomotic leakage after intestinal resection: a randomized controlled trial. Am Surg. 2017;83(2):157-161. [PubMed] [Google Scholar]
- 84.Ozben V, Aytac E, Liu X, Ozuner G. Does omental pedicle flap reduce anastomotic leak and septic complications after rectal cancer surgery? Int J Surg. 2016;27:53-57. doi: 10.1016/j.ijsu.2015.12.070. [DOI] [PubMed] [Google Scholar]
- 85.Ozben V, Ozuner G, Isik O, Gorgun E, Stocchi L, Liu X. Impact of omentoplasty on anastomotic leak and septic complications after low pelvic anastomosis: a study from the NSQIP database. Int J Colorectal Dis. 2018;33(12):1733-1739. doi: 10.1007/s00384-018-3151-2. [DOI] [PubMed] [Google Scholar]
- 86.Wiggins T, Markar SR, Arya S, Hanna GB. Anastomotic reinforcement with omentoplasty following gastrointestinal anastomosis: a systematic review and meta-analysis. Surg Oncol. 2015;24(3):181-186. doi: 10.1016/j.suronc.2015.06.011. [DOI] [PubMed] [Google Scholar]
- 87.van Praagh JB, de Goffau MC, Bakker IS, et al. Mucus microbiome of anastomotic tissue during surgery has predictive value for colorectal anastomotic leakage. Ann Surg. 2019;269(5):911-916. doi: 10.1097/sla.0000000000002651. [DOI] [PubMed] [Google Scholar]
- 88.Bosmans JW, Jongen AC, Birchenough GM, et al. Functional mucous layer and healing of proximal colonic anastomoses in an experimental model. Br J Surg. 2017;104(5):619-630. doi: 10.1002/bjs.10456. [DOI] [PubMed] [Google Scholar]
- 89.Jones RM, Luo L, Ardita CS, et al. Symbiotic lactobacilli stimulate gut epithelial proliferation via nox-mediated generation of reactive oxygen species. Embo J. 2013;32(23):3017-3028. doi: 10.1038/emboj.2013.224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Jones RM, Mercante JW, Neish AS. Reactive oxygen production induced by the gut microbiota: pharmacotherapeutic implications. Curr Med Chem. 2012;19(10):1519-1529. doi: 10.2174/092986712799828283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Lukic J, Chen V, Strahinic I, et al. Probiotics or pro-healers: the role of beneficial bacteria in tissue repair. Wound Repair Regen. 2017;25(6):912-922. doi: 10.1111/wrr.12607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Shogan BD, Smith DP, Christley S, Gilbert JA, Zaborina O, Alverdy JC. Intestinal anastomotic injury alters spatially defined microbiome composition and function. Microbiome. 2014;2:35. doi: 10.1186/2049-2618-2-35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Shogan BD, Belogortseva N, Luong PM, et al. Collagen degradation and MMP9 activation by Enterococcus faecalis contribute to intestinal anastomotic leak. Sci Transl Med. 2015;7(286):286ra68. doi: 10.1126/scitranslmed.3010658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Jacobson RA, Wienholts K, Williamson AJ, et al. Enterococcus faecalis exploits the human fibrinolytic system to drive excess collagenolysis: implications in gut healing and identification of druggable targets. Am J Physiol Gastrointest Liver Physiol. 2020;318(1):G1-g9. doi: 10.1152/ajpgi.00236.2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Olivas AD, Shogan BD, Valuckaite V, et al. Intestinal tissues induce an SNP mutation in Pseudomonas aeruginosa that enhances its virulence: possible role in anastomotic leak. PLoS One. 2012;7(8):e44326. doi: 10.1371/journal.pone.0044326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Babrowski T, Holbrook C, Moss J, et al. Pseudomonas aeruginosa virulence expression is directly activated by morphine and is capable of causing lethal gut-derived sepsis in mice during chronic morphine administration. Ann Surg. 2012;255(2):386-393. doi: 10.1097/SLA.0b013e3182331870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Mima K, Sakamoto Y, Kosumi K, et al. Mucosal cancer-associated microbes and anastomotic leakage after resection of colorectal carcinoma. Surg Oncol. 2020;32:63-68. doi: 10.1016/j.suronc.2019.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Harrell L, Wang Y, Antonopoulos D, et al. Standard colonic lavage alters the natural state of mucosal-associated microbiota in the human colon. PLoS One. 2012;7(2):e32545. doi: 10.1371/journal.pone.0032545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Leenen JPL, Hentzen J, Ockhuijsen HDL. Effectiveness of mechanical bowel preparation versus no preparation on anastomotic leakage in colorectal surgery: a systematic review and meta-analysis. Updates Surg. 2019;71(2):227-236. doi: 10.1007/s13304-018-0526-4. [DOI] [PubMed] [Google Scholar]
- 100.Ji WB, Hahn KY, Kwak JM, et al. Mechanical bowel preparation does not affect clinical severity of anastomotic leakage in rectal cancer surgery. World J Surg. 2017;41(5):1366-1374. doi: 10.1007/s00268-016-3839-9. [DOI] [PubMed] [Google Scholar]
- 101.Jernberg C, Löfmark S, Edlund C, Jansson JK. Long-term ecological impacts of antibiotic administration on the human intestinal microbiota. Isme J. 2007;1(1):56-66. doi: 10.1038/ismej.2007.3. [DOI] [PubMed] [Google Scholar]
- 102.Sangiorgio G, Vacante M, Basile F, Biondi A. Oral and parenteral vs. Parenteral antibiotic prophylaxis for patients undergoing laparoscopic colorectal resection: an intervention review with meta-analysis. Antibiotics (Basel). 2021;11(1):21. doi: 10.3390/antibiotics11010021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Grewal S, Reuvers JRD, Abis GSA, et al. Oral antibiotic prophylaxis reduces surgical site infection and anastomotic leakage in patients undergoing colorectal cancer surgery. Biomedicines. 2021;9(9):1184. doi: 10.3390/biomedicines9091184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Abis GSA, Stockmann H, Bonjer HJ, et al. Randomized clinical trial of selective decontamination of the digestive tract in elective colorectal cancer surgery (SELECT trial). Br J Surg. 2019;106(4):355-363. doi: 10.1002/bjs.11117. [DOI] [PubMed] [Google Scholar]
- 105.Jacobson RA, Williamson AJ, Wienholts K, et al. Prevention of anastomotic leak via local application of tranexamic acid to target bacterial-mediated plasminogen activation: a practical solution to a Complex problem. Ann Surg. 2021;274(6):e1038-e1046. doi: 10.1097/sla.0000000000003733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Long J, Zaborina O, Holbrook C, Zaborin A, Alverdy J. Depletion of intestinal phosphate after operative injury activates the virulence of P aeruginosa causing lethal gut-derived sepsis. Surgery. 2008;144(2):189-197. doi: 10.1016/j.surg.2008.03.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Hyoju SK, Klabbers RE, Aaron M, et al. Oral polyphosphate suppresses bacterial collagenase production and prevents anastomotic leak due to Serratia marcescens and Pseudomonas aeruginosa. Ann Surg. 2018;267(6):1112-1118. doi: 10.1097/sla.0000000000002167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Sapidis N, Tziouvaras C, Ioannidis O, Kalaitsidou I, Botsios D. The effect of glutamine and synbiotics on the healing of colonic anastomosis. Rev Esp Enferm Dig. 2014;106(4):255-262. [PubMed] [Google Scholar]
- 109.Kotzampassi K, Stavrou G, Damoraki G, et al. A four-probiotics regimen reduces postoperative complications after colorectal surgery: a randomized, double-blind, placebo-controlled study. World J Surg. 2015;39(11):2776-2783. doi: 10.1007/s00268-015-3071-z. [DOI] [PubMed] [Google Scholar]
- 110.Stavrou G, Kotzampassi K. Gut microbiome, surgical complications and probiotics. Ann Gastroenterol. 2017;30(1):45-53. doi: 10.20524/aog.2016.0086. [DOI] [PMC free article] [PubMed] [Google Scholar]