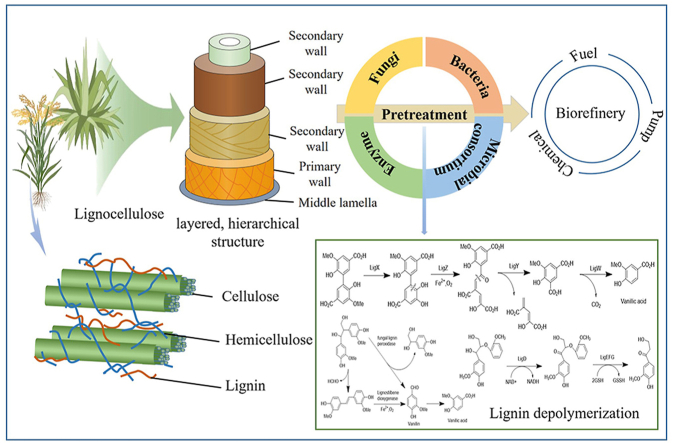
Abstract
Lignocellulose utilization has been gaining great attention worldwide due to its abundance, accessibility, renewability and recyclability. Destruction and dissociation of the cross-linked, hierarchical structure within cellulose hemicellulose and lignin is the key procedure during chemical utilization of lignocellulose. Of the pretreatments, biological treatment, which can effectively target the complex structures, is attractive due to its mild reaction conditions and environmentally friendly characteristics. Herein, we report a comprehensive review of the current biological pretreatments for lignocellulose dissociation and their corresponding degradation mechanisms. Firstly, we analyze the layered, hierarchical structure of cell wall, and the cross-linked network between cellulose, hemicellulose and lignin, then highlight that the cracking of β-aryl ether is considered the key to lignin degradation because of its dominant position. Secondly, we explore the effect of biological pretreatments, such as fungi, bacteria, microbial consortium, and enzymes, on substrate structure and degradation efficiency. Additionally, combining biological pretreatment with other methods (chemical methods and catalytic materials) may reduce the time necessary for the whole process, which also help to strengthen the lignocellulose dissociation efficiency. Thirdly, we summarize the related applications of lignocellulose, such as fuel production, chemicals platform, and bio-pulping, which could effectively alleviate the energy pressure through bioconversion into high value-added products. Based on reviewing of current progress of lignocellulose pretreatment, the challenges and future prospects are emphasized. Genetic engineering and other technologies to modify strains or enzymes for improved biotransformation efficiency will be the focus of future research.
Keywords: Lignocellulose, Lignin barrier, Cross-linked structure, Biological pretreatment, Biochemical platform
Graphical abstract
1. Introduction
According to reports, fossil fuels and other nonrenewable resources are in short supply. In this context, the utilization of renewable energy and its derivatives as substitutes can effectively reduce the dependence on nonrenewable energy, thereby achieving sustainable development and economic benefits [1]. Lignocellulose is an abundant renewable biomass resource on earth, with an annual output of more than 20 billion tons [2], including wood, plants, agricultural waste, forest residues and municipal solid waste, which can be converted into eco-friendly high value-added products [3]. It was reported that if all straw waste in China could be converted into hydrogen, the energy generated would be equivalent to a total of 170 million tons of standard coal [4]. Moreover, the production of biofuels from lignocellulose could alleviate the pressure caused by rapid energy consumption. Biofuel conversion technologies, such as those producing bioethanol, biohydrogen and biomethane, are mature. Moreover, high-value chemicals produced from lignocellulose increase the profitability.
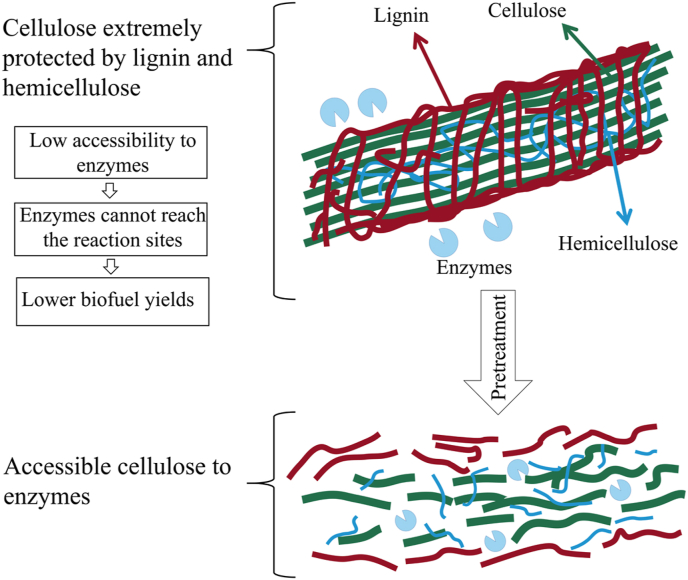
However, the large-scale utilization of lignocellulose is still challenging. Enzymatic hydrolysis of cellulose and hemicellulose is a key step in the transformation process, but the complex cross-linked structure of lignocellulose hinders the process by inhibiting enzymatic interactions (Fig. 1) [5], and the unproductive binding between enzymes and lignin also inhibits the enzymatic hydrolysis process [6]. In recent years, pretreatment has emerged as an effective approach to overcome these bottlenecks, thereby eliminating the compact and complex structure of biomass components and improving the accessibility of enzymes to cellulose. Pretreatment can accelerate the dissociation of fibers, promote biomass conversion, and hydrolyze polysaccharides into monosaccharides. Pretreatment also decreases the lignin contents, cellulose crystallinity and energy consumption [7].
Fig. 1.
Function of pretreatment [5].
In the lignocellulose process, effective pretreatment should meet the following requirements: (1) conducive to subsequent hydrolysis; (2) avoid producing by-products that inhibit enzyme saccharification; (3) be economically feasible; and (4) reduce pollution and waste of resources. At present, physical, chemical and biological treatments have been widely used for lignocellulose pretreatment. Physical treatments include mechanical crushing, ultrasonic treatment, microwave treatment, but these consume a lot of energy and are usually used as an auxiliary method. Chemical treatments have the advantage of high efficiency. For instance, rice straw treated with KOH and urea could obtain 92.38% sugar yield after 32.47% of lignin was removed under the optimal conditions [8]. However, the traditional chemical reagents have some disadvantages, such as the incomplete recovery of polysaccharides, possible generation of inhibitors and the risk of environmental pollution, thereby affecting the production efficiency [9].
Recently, ionic liquids have been used in pretreatment due to their non-flammable, non-volatile and recyclable characteristics [10]. When rice straw was treated with 1-H-3-methylmorpholinium chloride ionic liquid with water as co-solvent for 5 h, the ethanol production yield increased from 21.9% to 64% [11]. However, the high cost of ionic liquids limits large-scale implementation. Therefore, other types of green solvents have been developed as good substitutes for traditional reagents. It was reported that a high lignin removal rate of 73.17% was obtained after pretreatment with 10% ethylenediamine (EDA) and 200 °C. Compared with traditional alkali treatment, the residual EDA in wastewater can be separated by simple evaporation [12]. In addition, deep eutectic solvent (DES) has been shown to be a highly cost-effective and promising solvent [13]. Despite the fact that the lignin removal rate of DES might not be as high as that of alkali reagents, lignin pretreated with DES was substantially depolymerized and no condensation structure was observed [14]. The lignin from the corn stover was effectively removed after pretreatment with DES consisting of quaternary ammonium salts and hydrogen donors. After hydrolysis, no unfavorable inhibitors to the subsequent fermentation process were produced, indicating that DES possesses good biocompatibility [15]. In addition, it was found that the energy required for DES pretreatment was 28% and 72% lower than that of NaOH pretreatment and the steam explosion process, respectively [16]. These results all reflect the potential value of DES. Nevertheless, there are still challenges in the application of DES, whose inherent properties such as high viscosity and hygroscopicity may limit its application in biomass pretreatment. At the same time, further research is needed to identify ways in which to efficiently recover DES [10].
In short, traditional physical and chemical pretreatments require special equipment, and inhibitors can be produced in the process, which is inconducive to subsequent fermentation. Due to the great influence of pretreatment on downstream technologies, from the overall economic perspective, the eco-friendly characteristics make biological pretreatment stand out. Relatively speaking, biological methods consume less energy and do not produce inhibitors during the treatment process because it is carried out under mild conditions. There are a large number of microorganisms in nature that can be applied in biological pretreatment. The degradation capability of microorganisms mainly originates from their unique enzymatic systems. Lignolytic enzymes such as lignin peroxidase, manganese peroxidase and laccase have been applied to open the recalcitrant structure of lignocellulose. Numerous studies have shown that white rot fungi are highly selective for lignin, which can improve the hydrolysis efficiency and reduce energy consumption. At present, several studies are seeking to understand the degradation mechanism of different strains. However, fungal pretreatment still has some challenges, such as improving the stability and tolerance of strains, selecting appropriate strains and improving degradation efficiency.
In biological pretreatment bacteria with strong viability, which could adapt well to variation in the microenvironment, have also been isolated and applied. Effective biodegradation can be accomplished with microbial consortium comprising different strains. Synergism of consortium is the key to degradation. Due to higher substrate utilization rates, microbial consortium has gradually replaced the single strain and has become the first choice for this process. Furthermore, combining biological pretreatment with other methods has been a research hotspot. Various technologies have also been developed to improve the biological pretreatment process, with a focus on gene editing technology.
In addition to being widely accepted in new fields, lignocellulose has been utilized in traditional pulping and papermaking for thousands of years. With technological advancements, lignocellulosic derivatives have been used in agriculture, industry and other fields. In line with the requirements of green technology, the introduction of microorganisms or enzymes for biological modification is conducive to saving energy consumption. Relevant studies have indicated that cellulases could improve the performance of pulp dissolution [17]. Xylanase is mainly used to assist in pulp bleaching to reduce the formation of organic halogens and consumption of chemicals [18]. The reduction in total production costs ensures sustainable development of the pulp and paper industry.
This review specifically summarizes and discusses the research progress of biological pretreatments to date, providing a theoretical basis for the selection of strains. Additionally, the insightful degradation mechanisms are further clarified, and the methods for improving the process are discussed. Finally, the application of lignocellulose in fuel, chemical and pulping industries is introduced, and the influence of pretreatment on resource utilization is highlighted.
2. Microstructure characteristics of biomass fiber cell wall
2.1. Layered structure of fibrous cell wall
Plant cell walls are divided into three layers: primary walls surrounding the growing cells or cells with growth ability; secondary walls containing lignin-thickened structure and surrounding special cells such as fiber cells; and the intercellular layer [19]. The primary wall is composed of cellulose, hemicellulose and pectin, containing multi-layer nanostructures. In each layer, the cellulose microfibrils form a network structure, hemicelluloses combine with it non-covalently, and pectin acts as the matrix carrier network [20]. The secondary wall is a complex of cellulose, hemicellulose and lignin, where lignin is closely connected with cellulose through xylan. The supramolecular network formed by this complex compound provides mechanical strength, enabling plants to stand upright [21].
During the process of enzymatic cell wall deconstruction, the enzymatic dissolution mechanisms of bacteria and fungi are different. Bacterial cellulase attached to the surface of the cell wall, cell horn and plasmodesmata, and separated the cell wall from the composite interlayer (CML). In contrast, fungal cellulase can adhere to the surface and penetrate the secondary wall, which can dissolve the cell wall evenly without affecting the CML [22].
2.2. Structural characteristics of lignocellulose
Lignocellulose has a complex structure, which is mainly composed of cellulose, hemicellulose, and lignin (Fig. 2) [23]. Cellulose is a macromolecular polysaccharide with d-glucose as the structural unit and linearly connected by β-1,4 glycosidic bonds. Its content is higher than lignin and hemicellulose. Each glucose molecule constituting cellulose contains five hydroxyl groups, which can form intermolecular and intramolecular hydrogen bonds. A large number of hydrogen bonds interact with each other between the cellulose chains to form a network. Meanwhile, the existence of crystalline and amorphous regions of cellulose makes cellulose very difficult to degrade [24,25].
Fig. 2.
Schematic diagram of lignocellulose: a) the structure of cell wall; b) three components of lignocellulose [23].
Hemicellulose is a heterogeneous polymer composed of pentose and hexose [26]. Unlike cellulose, hemicellulose has only the amorphous region and can easily interact with cellulose due to its own structure and properties [27]. The hemicellulose polysaccharides are closely bonded with the cellulose microfibrils through hydrogen bonds [19].
Lignin is an aromatic polymer composed of p-coumarin, coniferol and sinapyl alcohol [28,29]. Its stable aromatic structure is the major obstacle to hydrolysis. Ether bonds, single bonds and ester bonds in lignin could be depolymerized into aromatic monomers or compounds through cracking, hydrolysis, dehydroxylation, reduction and oxidation [30]. Among the various linkages, the cracking of β-aryl ether is considered the key to lignin degradation due to its dominant position [31].
In lignocellulose, cellulose and hemicellulose are coated with lignin. Specifically, lignin is combined with hemicellulose through covalent and non-covalent bonds. The α-ether bond is the primary lignin-carbohydrate bond, which exists between lignin and the C-6 position of mannose [32]. The physical barrier of lignin provides protection and mechanical stability to the plant cell wall [33]. However, this structure also hinders the separation and recovery of these three components, resulting in the difficult natural degradation of biomass. Cellulose crystallinity, lignin barrier, accessible surface area and biomass heterogeneity are the limiting factors of enzymatic hydrolysis that lead to a low saccharification rate and high production cost [34]. Therefore, disrupting its structure through pretreatment is a very key step in the production process.
3. Biological pretreatment
3.1. Features of different pretreatment processes
Pretreatment could change the composition and structure of lignocellulose biomass to achieve improved accessibility of enzymes and obtain higher sugar yields. In the past few decades, through innovative research, pretreatment processes have continuously broken through technical limitations. The ultimate goal of all these pretreatment processes is to meet industrial requirements, so as to achieve large-scale economic production. Biological pretreatment has been reviewed by different authors, as shown in Table 1. The biological pretreatment process can be roughly divided into fungi, bacteria, microbial consortium, and enzyme treatments. It has the advantages of high cost-effectiveness. The current biological system comprises strains and enzymes with various functions. An insightful understanding of the reaction mechanism and degradation effect is conducive to designing a reasonable operation scheme. Table 2 summarizes the advantages and disadvantages of different biological pretreatment processes. Generally speaking, fungi have outstanding degradation ability and are the best choice for production. Bacteria, which have mostly been isolated from the environment, have strong viability. However, the degradation ability of a single strain is limited. For the purpose of achieving high-efficiency production, microbial consortium combining multiple strains are applied. Alternatively, the direct application of enzymes is another treatment method. The corresponding enzymes can be selected in terms of the substrate structure for targeted treatment. Different pretreatments are discussed as follows.
Table 1.
List of review papers focusing on biological pretreatment methods for lignocellulosic materials.
| NO. | Review highlights | Review gaps | Year | Reference |
|---|---|---|---|---|
| 1 |
|
|
2012 | [35] |
| 2 |
|
|
2012 | [36] |
| 3 |
|
|
2016 | [4] |
| 4 |
|
|
2017 | [37] |
| 5 |
|
|
2018 | [38] |
| 6 |
|
|
2018 | [1] |
| 7 |
|
|
2019 | [3] |
| 8 |
|
|
2021 | [39] |
Table 2.
Advantages and disadvantages of different biological pretreatment.
| Pretreatment | Advantages | Disadvantages |
|---|---|---|
| Fungi |
|
|
| Bacteria |
|
|
| Microbial consortium |
|
|
| Enzyme |
|
|
3.2. Fungi
Fungi, such as white rot fungi and brown rot fungi, are widely used in the microbial treatment of lignocellulose. White rot fungi are preferred for fungal pretreatment due to their high selectivity in lignin degradation [36]. Accumulating studies have shown that white-rot fungi degrade lignin by secreting extracellular enzymes such as laccase and peroxidase. Ma et al. [40] reported a newly isolated white-rot basidiomycete Peniophora incarnate T-7 that could be employed for poplar wood pretreatment. Significant groups of lignin-degrading enzymes, including laccase, multicopper oxidase, peroxidase and other auxiliary enzymes, were identified in proteins secreted by the strain. After 7-day pretreatment, the cellulose loss rate was only 16%, while the lignin removal rate was as high as 70%, indicating that Peniophora incarnate T-7 could selectively degrade lignin.
It was reported that the lignin-degrading enzyme system of Cerrena unicolor SP02 included lignin oxidase and lignin-degrading auxiliary enzymes, of which lignin peroxidase, laccase, manganese peroxidase and cellobiose dehydrogenase were members. Of the 5595 genes annotated in the KOG categories of the strain, 276 and 325 functional genes that played an indispensable role in enzyme secretion and lignin degradation were classified as “Intracellular trafficking, secretion, and vesicular transport” and “Carbohydrate transport and metabolism”, respectively [41]. In a previous study, the mechanism of lignin degradation by white rot fungus was studied, and the results indicated that Ceriporiopsis subvermispora could effectively cleave the β-O-4 aryl ether bonds between the lignin units to degrade lignin. Lignin trimer, the smallest β-O-4 aryl ether oligomer, was considered to describe the in situ cleavage pathway of β-O-4 aryl ethers. The oxidation pathway of the middle aromatic ring B (Fig. 3) included Cα-oxidation, Cα-Cβ cleavage, β-|O-4 cleavage, and β-O-|4 cleavage, of which Cα-Cβ cleavage was a major reaction underlying ligninolysis [42].
Fig. 3.
In situ cleavage routes of β-O-4 aryl ethers initiated [42].
Among the white-rot fungi, Phanerochaete Chrysosporium is one of the most representative strains. It degrades lignin by secreting extracellular enzymes such as LiP and MnP, but does not secrete laccase. Additionally, P. Chrysosporium is considered to be a non-selective fungus that can simultaneously degrade lignin, cellulose and hemicellulose [43]. In a previous study, the activities of carboxymethyl cellulase and MnP enzyme were detected during the degradation of ramie stalks. Glycoside hydrolases, glucosyl transferases and carbohydrate-binding modules were strongly expressed in P. Chrysosporium secretion. These results suggest that cellulose, hemicellulose and pectin could be effectively degraded [44]. Liu et al. [45] studied the structural changes in wheat straw during the degradation of P. Chrysosporium using visualization technology. Fungal treatment changed the surface of the straw from hydrophobic to hydrophilic, revealing holes. After 5 days of culture, hyphae were successfully colonized, and the hyphae were observed in the epidermal cells, vascular bundles, parenchyma cells and lignified sclerenchyma cells of the straw. Since lignin was distributed in vascular bundles, it was speculated that hyphae could directly penetrate the primary and secondary walls into the lumen of the straw to degrade lignin. This study provides further insight into degradation mechanism of microorganisms.
Unlike white rot fungi, brown rot fungi mainly act on lignocellulose carbohydrates without affecting lignin [46]. Previous results indicate that the degradation of brown rot fungi occurs in the S2 layer of cell wall, and its degradation mechanisms include oxidation and hydrolysis, which are staggered by differential gene expression [47]. In the early stage of degradation, brown rot fungi produce hydroxyl radicals (·OH) through non-enzymatic extracellular Fenton reaction (H2O2 + Fe2+ → OH− + Fe3+ + ·OH), which randomly attack the cell wall of lignocellulose, resulting in the depolymerization of carbohydrates (Fig. 4) [[48], [49], [50]]. In this process, lignin is modified, and some derivatives continue to participate in the reaction as electron sources. The second stage involves carbohydrate-degrading enzymes mediated saccharification, which provides digestible sugars for fungal metabolism [51]. For instance, Zhu et al. [52] inoculated the brown-rot fungus Gloeophyllum trabeum on rice straw to accelerate the degradation of cellulose and hemicellulose and provide carbon sources for microorganisms. After inoculation, the FPase activity was improved, and the cellulose degradation rate was as high as 40.2%. Meanwhile, G. trabeum could produce a high level of xylanase to degrade hemicellulose. Additionally, the inoculation of G. trabeum could affect succession of the microbial community structure by affecting the major physical and chemical parameters (germination index, cellulose and hemicellulose). The same strain was used in the brown-rot decay of Masson pine to further understand the deconstruction mode of wood cell walls. The amorphous polysaccharide was preferentially degraded in the early stage, while the reactive oxygen species produced by non-enzymatic chelator-mediated Fenton (CMF) system promoted the depolymerization of crystalline cellulose. It was found that the released sugars were mainly mannose and glucose, and a large amount of mannose was released before glucose, indicating that the non-enzymatic CMF system was attacked by hemicellulose first and then cellulose [50].
Fig. 4.
Model for non-enzymatic deconstruction of lignocellulose cell walls in incipient brown-rot decay. a) Deconstruction of the cell wall structure; b) Chelator-mediated Fenton reaction in wood cell walls; c) Depolymerization of polysaccharide components; d) Deconstruction of the elementary fibril structure [50]. (For interpretation of the references to colour in this figure legend, the reader is referred to the Web version of this article.)
3.3. Bacteria
In addition to fungi, bacteria are also widely used in the biomass pretreatment process. For instance, the related strains, such as Bacillus, harbour abundant genes encoding glycoside hydrolases and glycosyl transferases that are used for cellulose degradation [53]. As a dominant strain, Bacillus inoculated on rice straw could improve the microenvironment and accelerate the assembly of bacterial communities to degrade lignocellulose [54]. Muñoz et al. [55] studied marine bacteria with cellulose degradation ability and screened nine strains with endoglucanase activity from Mytilus chinensis and Mesodesma donacium. These strains were identified as Raoultella, Chryseobacterium, Aeromonas and Pseudomonas. The degradation experiment of filter paper indicated that all strains had degradation ability. In contrast, Raoultella ornithinolytica strains MA5 and MC3 and Aeromonas bivalvium strain MC23 had better effects, degrading 5.26%, 4.23% and 3.51% respectively. The microalgae pretreatment using strains MA5 and MC3 found that the cell wall of microalgae was significantly damaged at 30 °C, which was conducive to subsequent biotransformation and had certain advantages in reducing the energy cost.
Under the action of bacterial enzymes, lignin is degraded through a variety of metabolic pathways (Table 3). The β-ketoadipate pathway is considered to be a common pathway of lignin degradation. Granja travez et al. [58] analyzed 13 lignin-degrading bacteria and found a total of 10 bacteria containing the β-ketoadipate pathway. Additionally, the genes related to the biphenyl degradation pathway, propionic acid degradation pathway, gentisate acid degradation pathway and phthalic acid degradation pathway were also found in different strains. Bacillus ligniniphilus L1 has been shown to significantly degrade lignin. A total of 3 types of lignin degradation pathways were observed in the strain L1, namely the gentisate pathway, the benzoic acid pathway, and the β-ketoadipate pathway, of which the β-ketoadipate pathway included the catechuate and the protocatechuate branches. The lignin provided energy to support the growth of strain L1 at 50 °C, and 38.9% of the lignin was degraded by strain L1 during the 7 day incubation. Proteomic studies showed that the key proteins related to lignin degradation, such as 2-cys peroxiredoxins, short chain dehydrogenase, carbon-monoxide dehydrogenase, acetyl-CoA acetyltransferase, enoyl-CoA hydratase, ferredoxin, and formate dehydrogenase were up-regulated [59]. In the study of Tsegaye et al. [60], Ochrobactrum oryzae BMP03, isolated from alkali lignin medium, grew at the optimum temperature of 30 °C and pH of 6. Under the optimum conditions, the strain secreting laccase and peroxidase to degrade lignin reached the logarithmic phase within the sixth and ninth days. Klebsiella pneumoniae NX-1, Pseudomonas putida NX-1, and Ochrobactrum tritici NX-1 could also grow and metabolize using kraft lignin as the only carbon source, but the enzyme secretion ability of these strains varied to a great extent (Lac, LiP and MnP) [61]. Overall, these results show that the degradation modes of different strains are variable. There are many studies on this aspect, and understanding this process is helpful for strain selection.
Table 3.
Pathways for degradation of lignin.
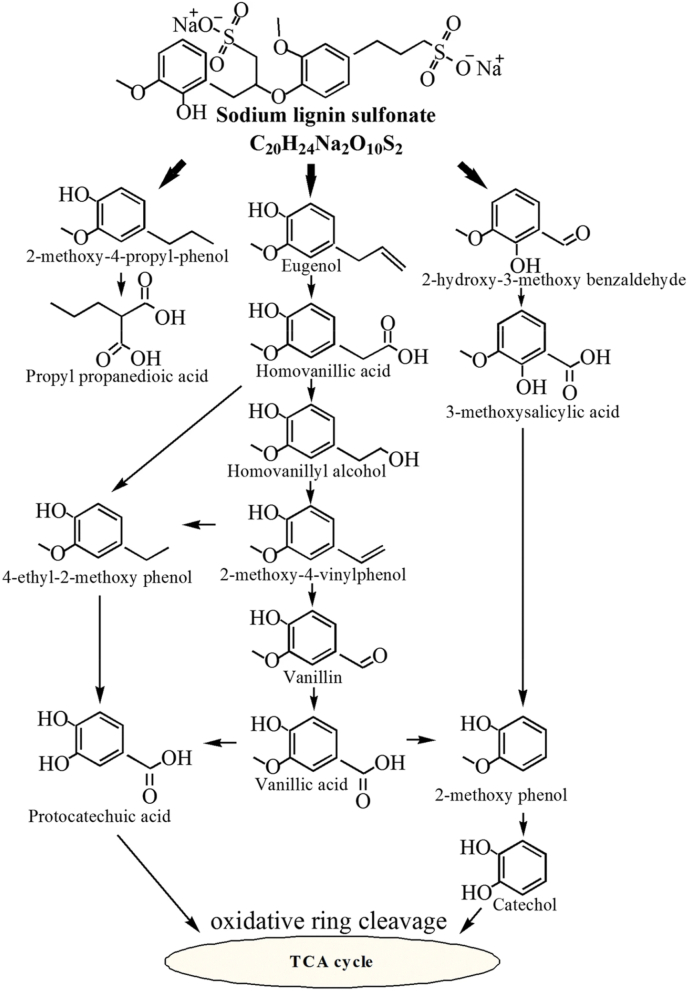
Bacteria can better adapt to extreme environments than fungi, which helps to expand the scope of the application of the process. Cortes-Tolalpa et al. [62] used wheat straw as a carbon and energy source to screen salt marsh soil microorganisms, and obtained a salt tolerant lignocellulose-degradating microbial consortium. It was found that under high salt conditions, bacteria (including Joostella marina, Flavobacterium beibuense, Algoriphagus ratkowskyi, Pseudomonas putida and Halomonas Meridiana) replaced fungi to occupy the main position, playing a major role in degradation. These strains had the ability to secrete salt tolerant enzymes and were powerful candidates to improve the degradation efficiency and reduce production costs. Jiang et al. [63] isolated a low temperature tolerant bacterium Arthrobacter sp. C2 that could secrete Lip and MnP to degrade lignin. The strain reached the maximum degradation rate (39.2%) at 15 °C using sodium lignosulfonate as a substrate and showed activity in the pH range of 3–10. The possible degradation processes included cleavage of carbon oxygen bond, demethylation of the methoxy group, oxidation of the hydroxyl group and decarboxylation of the carboxyl group. The relevant degradation pathways speculated based on the analysis results (Fig. 5) were different from the degradation processes of mesophilic bacteria. Therefore, the reasonable application of Arthrobacter sp. C2 in pretreatment was inferred to improve the operability of the process. Other examples included Cedecea lapagei, which was tolerant to a wide pH range and could survive in black liquor. The laccase activity was detected in the lignin medium at pH 6–8 inoculated by C. lapagei, while superoxide dismutase, glutathione S-transferase, alkyl hydroperoxide reductase and glutathione reductase were found on the cellulose or hemicellulose containing medium. Additionally, the study found other enzymes related to lignin and carbohydrate degradation, which could play a vital role in industrial processes [64].
Fig. 5.
Proposed degradation pathways of sodium lignin sulfonate by psychrotrophic Arthrobacter sp. C2 [63].
3.4. Microbial consortium
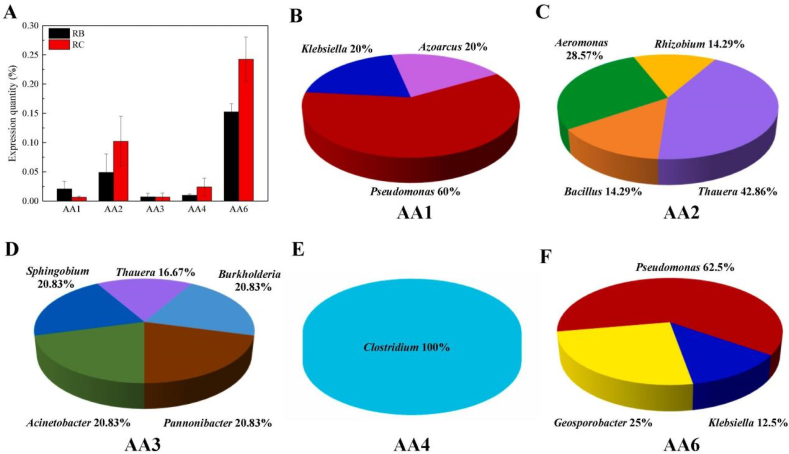
Microbial consortium, which can complete tasks too complex for monomers through division of labor and cooperation, contains various microorganisms [65]. Table 4 lists examples of various microbial consortium for pretreating biomass. For example, the strains in the microbial consortium LTF-27 mainly included Alcaligenes, Parabacteroides, Lysinibacillus, Clostridium and Sphingobacterium. They could secrete various enzymes like catalase, endoglucanase and xylanase, which played a significant role in lignocellulose degradation. The weight loss rates of hemicellulose and cellulose reached 71.7% and 65.6% after treatment at 15 °C for 20 days [66]. In another study, the microbial complex LDC showed high lignin-degrading enzyme activity (MnP, LiP and Lac). Additionally, several family genes related to lignin degradation, AA1, AA2, AA3, AA4 and AA6, were found in LDC. After LDC inoculation, the expression levels of AA2, AA4 and AA6 increased by 112%, 165% and 67% over time, respectively. Among them, most AA1 (60%) and AA6 (62.5%) were secreted by Pseudomonas (Fig. 6), accounting for a large proportion in LDC. Klebsiella, Azoarcus, Thaurea and Aeromonas were also the source microorganisms of these genes, which completed degradation through the division of labor and cooperation [67].
Table 4.
Degradation effects of different microbial consortium.
| Microbial consortium | Biomass | Operating Conditions | Treatment effect | References |
|---|---|---|---|---|
| LTF-27 | Rice straw | Incubate at 15 °C for 20 days | The weight loss of cellulose, hemicelluloses and lignin was 71.7%, 65.6% and 12.5% | [66] |
| LDC | Rice straw | The inoculation amount was 10% (v/v), and static incubate at 30 °C for 7 days | The degradation rates of cellulose, hemicellulose and lignin were 5.70%, 63.08% and 31.18% | [67] |
| CB | Corn stalk | Under conditions of temperature (32 °C), pH (3.5), solid-liquid ratio (10%), culture time (20 d) | The degradation rates of lignin, cellulose and hemicellulose were 43.36%, 31.29%, 48.36% | [69] |
| LCDC | Sawdust | Incubate at 30 ± 1 °C for 10 days | Reduction in cellulose, hemicelluloses, and lignin contents by 37.5%, 39.6%, and 56.7% | [70] |
| SST-4 | Birchwood sawdust | Incubate at 35 °C for 20 days, with an agitation level of 130 rpm | Degradation of cellulose, hemicellulose, and lignin estimated as 61.5%, 55.9% and 19.46% | [71] |
Fig. 6.
Expression levels of AA1, AA2, AA3, AA4, and AA6 in different incubation periods (A). Microorganisms from which the AA1 (B), AA2 (C), AA3 (D), AA4 (E), and AA6 (F) come during the peak stage of lignin degradation [67].
There are many combinations in microbial consortium, so it is essential to build a consortium with a positive promotion effect. In this process, there can be no vicious competition among the strains, otherwise it will lead to a low degradation efficiency. Zhang et al. [68] combined the isolated Bacillus sp. strain A7 with Clostridium sp. strain AA3 and Bacteroides sp. strain AA4 in different ways and found that there was a negative relationship between the strains A7 and AA4, which might not have a positive impact on extracellular enzymes secretion. Therefore, not all strains can be combined and applied, and careful consideration should be given to the selection of strains. Limited by this factor, Chu et al. [69] selected Phanerochaete chrysosporium, Trametes versicolor and Pleurotus ostreatus to construct compound bacteria (CB). During the culture process, it was observed that the mycelium of the three strains fused well, indicating that there was no antagonism between the strains. The mycelium of CB could diffuse through the intercellular space and degrade by utilizing the intracellular substances to secrete lignin-degrading enzymes. Its degradation ability was significantly higher than a single strain.
3.5. Enzymes
Biological pretreatment can also be carried out by directly adding enzymes, thereby eliminating the requirement of microbial growth. Enzymes can be obtained from plants, animals and microorganisms. Commercial enzymes are obtained from microorganisms (bacteria and fungi) due to their ease of large scale cultivation. According to Bhushan et al. [72], pretreatment with a crude enzyme produced by Aspergillus fischeri could effectively disintegrate the cell wall of Chlorella pyrenoidosa and release about 76.3% sugar under the best treatment conditions.
Among the enzymes related to pretreatment, laccase, a polyphenol oxidase containing copper, plays an indispensable role in degradation. Zhu et al. [73] constructed a laccase-inactive mutant and compared its lignin degradation effect with Bacillus ligniniphilus L1 to investigate the importance of laccase. The laccase encoding gene of strain L1 was expressed and purified in Escherichia coli. The obtained laccase exhibited outstanding thermal tolerance and thermal stability. Lignin could be degraded by laccase through cleavage of lignin side chain, oxidation of Cα and demethylation reaction. As a result, this enzyme could be a promising candidate for industrial production. Compared to laccase, Lip has higher redox potential and can oxidize non-phenolic structures in the absence of medium. During the specific catalytic process, Lip generates the compound I intermediate under the reduction of H2O2. Compound I is reduced to compound II through two consecutive single electron reductions, and then Lip is returned to the iron oxidation state [74]. In the degradation process catalyzed by MnP, H2O2 is used as an oxidant to convert Mn2+ into smaller chelating agent medium Mn3+, so as to better penetrate into the dense structure [75,76]. In addition to these commonly used enzymes, many other enzymes have demonstrated the ability to remove lignin. For example, the oxidase found in ascomycete Parascedosporium putredinis NO1 had β-ether enzyme activity, which could degrade without the cofactors. Besides, the oxidase could release the tricin subunit in lignin (Fig. 7) [77]. Manganese superoxide dismutase [78], glutathione lyase, Cα-dehydrogenase and glutathione transferase [79] also play an indispensable role in lignin degradation.
Fig. 7.
Changes in lignin aromatic and side chain regional components from (A) wheat control and (B) enzyme-treated wheat [77].
Enzyme catalysis has the characteristic of high efficiency, but the degradation ability of a single enzyme is limited due to the complex and diverse structure of lignin. Therefore, current research mostly focuses on the degradation of composite enzymes. For example, enzyme ultraflo® L (including endo-1,3 (4)-β-glucanase, cellulase, xylanase and feruloyl esterase) could significantly reduce the lignin content in corn cob and vine trimming shoots (VTS), where the lignin percentage in the VTS was reduced by more than 5 times [80]. Cakmak et al. [81] studied the effect of treating Porphyridium cruentum with mixed enzymes (cellulase, protease and viscozyme). The results showed that the effect of mixed enzyme treatment was better than the single enzyme treatment, demonstrating a synergistic effect. Protease was responsible for degrading cell proteins. Viscozyme was a carbohydrase mix that could degrade xylan in the cell wall. In another study, cellulase, glucose hydrolase and an enzyme mixture (cellulase, glucose hydrolase and xylanase) were applied to a microalgae dissolution at dosages of 0.5% and 1% and the results showed that the dissolution rate of 1% cellulase and the enzyme mixture was faster, and the final soluble volatile solid (680 mg vs soluble/L) was higher than the 0.5% dosage. Among them, the mixed enzyme had a synergistic effect. Cellulase hydrolyzes cellulose, while glucose hydrolase and xylanase hydrolyze hemicellulose. The mixed enzyme showed better performance than the single enzyme [82]. Additionally, in the enzyme treatment process, the type and dosage of enzyme can be adjusted according to different substrates. Ometto et al. [83] selected appropriate enzymes for pretreatment according to the composition of the algal cell wall. These enzymes showed different effects under the same treatment conditions due to structural differences of algae. It was found that the soluble chemical oxygen demand released by the mixed enzyme treatment was more than twice that of the single enzyme. Compared with thermal treatment, thermal hydrolysis and ultrasonic treatment, enzymatic hydrolysis preferentially acted on the cell wall components (Fig. 8). After enzymatic treatment, the concentration of the soluble carbohydrates was increased.
Fig. 8.
Microstructure of S. obliquus and C. sorokiniana cells, untreated (1) and after thermal hydrolysis (2), ultrasound (3) and enzymatic (4) pre-treatment [83].
Overall, the effects of various parameters (pH, temperature, the enzyme load, substrate load, etc.) should be considered during enzymatic hydrolysis. With the in-depth study of enzyme degradation characteristics and mechanisms, the challenges for improving the stability, catalytic activity and degradation efficiency have been further explored. Future research is expected to be combined with genetic engineering to obtain the enzymes needed for industrial production.
4. Methods for improving process
4.1. Two-step fungal treatment
Researchers have proposed an effective combined biological treatment method, namely a two-step fungal treatment using white-rot fungi and brown-rot fungi, to improve the degradation efficiency. White-rot fungi and brown-rot fungi have different effects, so their combination can provide excellent results. Wang et al. [84] pretreated corn cobs with three species of white-rot fungi, namely Trichaptum biforme (Dui16067) and Trametes orientalis (Cui6300 and Cui6319). Then the brown-rot fungus Fomitopsis pinicola (Cui12330) was added for further degradation over a short time. White-rot fungi liberated ligninolytic enzymes to degrade lignin, while the brown-rot fungus selectively removed hemicellulose and had no significant impact on the cellulose content. Meanwhile, the depolymerization of the lignin S type occurred during brown rot fungus treatment. After the two-step pretreatment, the corn cob structure was destructed more seriously and the hydrolysis efficiency was significantly improved. This method showed the advantages of less processing time and was conducive to industrial production. Hermosilla et al. [85] also observed a synergistic effect in wheat straw samples successively inoculated with white rot fungi and brown rot fungi, and the glucose recovery rate was higher than the single strain culture.
Optimizing the co-culture mode and conditions of fungi is an effective approach to improve the utilization rate of lignocellulose. Researchers found that Trichoderma viride was not suitable for simultaneous culture with Phanerochaete Chrysosporium, but successive inoculation of the two fungi could improve the degradation efficiency. The cellulase activity detected during this culture mode was higher than in the co-culture mode. The experimental results showed that under the optimum culture conditions, such as an inoculation concentration of 14% (w/v), 1:1.4 solid-liquid ratio and culture temperature of 30 °C, the experimental group inoculated with P. chrysosporium for 4 days and then inoculated with T. viride for 4 days showed the highest lignin and cellulose degradation rates of 26.38% and 33.29% respectively [86].
4.2. Combination of biological pretreatment and other methods
4.2.1. Combination of biological pretreatment and chemical pretreatment
Since a single pretreatment scheme is not applicable to all processes, the combination of different pretreatment methods might produce synergistic effects and optimize the defects of separate pretreatment processes, thereby improving biomass degradation efficiency. Biological pretreatments could be combined with chemical processes. It was reported that combining biological and chemical treatments could shorten the process time and reduce the production of inhibitors [7]. Previous studies have shown that the combined treatment with phosphoric acid and white rot fungi increased the theoretical ethanol yield from 27.9% to 62.8% [87]. In another study, Liang et al. [88] first pretreated straw with 1% (W/V) NaOH solution for 24 h to provide more growth sites for subsequently inoculated microorganisms. The OEM2 microbial consortium screened out in the experiment was mainly composed of 6 bacteria phyla, which had stable and efficient lignocellulose degradation ability. After 10 generations of transfer culture, the lignocellulose degradation rate was still above 35%. During OEM2 inoculation, the concentration of xylanase was higher than CMCase and laccase, and the decomposition rate of hemicellulose (85.38%) was higher than cellulose (75.95%) and lignin (18.90%).
Fenton reagent was also commonly used in this process because it could effectively destroy the structure of rice straw and increase the binding sites of enzymes. At the same time, the activities of CMCase, FPase, xylanase, Lip and MnP were improved [89]. In the subsequent study, researchers added functional bacterial agents on the basis of Fenton-pretreated rice straw. During the Fenton treatment, the free radicals were produced to promote the oxidation and degradation of lignocellulose. After inoculation, the diversity and richness of the microbial community were improved, and the endoglucanase and polyphenol oxidase activities showed a fluctuating increasing trend, accelerating lignocellulose degradation. This combined pretreatment method improved the abundance of key genes, as well as the correlation between the key genes and core colonies [90].
In addition to the above reagents, ionic liquids and eutectic solvents are also used in the pretreatment process. Therefore, the selection of reagents and the relationship between reagents and microorganisms have been extensively investigated in subsequent studies.
4.2.2. Combination of biological pretreatment and catalysis system
In addition to selecting appropriate pretreatment methods and finding the best treatment conditions, adding auxiliary materials is also an effective means to improve the degradation efficiency. The combination of metal nanoparticles and enzymes has been shown to improve the catalytic activity and expand the scope of reactions. Compared with free enzymes, the stability of enzymes immobilized on metal nanoparticles, in terms of temperature and pH, was significantly improved [91]. Additionally, the metal nanoparticles could increase enzyme activity by increasing the electron transfer rate. The optimum concentration leads to a significant surface effect, while a higher optimum concentration leads to increased oxidative stress, resulting in cell death [92]. Huang et al. [93] explored the degradation performance of Phanerochaete Chrysosporium under different proportions of Fe3O4 NMS (0%, 2.5%, 5%, 10%). In the degradation process, Fe3O4 NMS served as an iron source for supplying Fe2+ and Fe3+ to strengthen the Fenton reaction and an energy source for strain growth. The addition of Fe3O4 NMS increased the Lip enzyme activity of the strain, and the highest enzyme activity of 2.5% was 169.78 U g−1. Meanwhile, the production of CMCase and xylanase was also promoted to a certain extent. In another study, sugarcane bagasse (SB) pretreated with protease activated by magnesium oxide nanoparticles (MgN) showed an 18-fold higher lignin removal rate compared with untreated samples. Similarly, in the presence of MgN, the yield of reducing sugar and glucose in xylanase-treated SB samples was significantly increased, demonstrating that this treatment has potential significance for ethanol production [94].
The existence of metal ions will also affect the enzyme activity. For instance, Mn2+ and Cu2+ could significantly affect MNP activity. Under static liquid culture conditions, the addition of 1 mM Mn2+ increased the enzyme activity of Trametes trogii S0301 to the maximum level (about 16.5 U/mL) on the 6th day. The addition of Cu2+ (0.2 and 2 mM) also improved the MNP activity. Mn2+ and Cu2+ were used as inducers of the MNP gene to induce the Group I MnP subfamily (T_trogii_09901, 09904, 09903 and 09906) and Group II MnP subfamily (T_trogii_11,984, 11,971, 11,985 and 11,983) genes, respectively [95]. The role of Mn2+ was also reflected in the degradation of wheat straw by Irpex lacteus. The addition of Mn2+ increased the enzyme yield and induced MNP secretion. The mediated oxidation reaction of MNP led to the degradation of a large amount of hemicellulose. This metal ion had a positive impact on the glucose yield [96]. In another study, Li et al. [97] compared the degradation effect of Pleurotus ostreatus haucc 162 with and without Mn2+, and the results showed that the presence of Mn2+ enhanced the depolymerization of lignin and significantly damaged the crosslinking structure. After the addition of Mn2+, the contents of polysaccharide monooxygenase and 1,4-benzoquinone reductase were increased, which played a significant role in the driving of Fenton reaction. Other examples include Fe2+, Ag2+, Mg2+, Mn+ and Co+, which could significantly enhance the activity of xylanase extracted and purified from Aspergillus oryzae LC1 at a concentration of 2 mM. The enzyme could effectively hydrolyze untreated sugarcane bagasse, wheat straw and wheat bran [98].
In the pretreatment process, biochar could also be added to improve the treatment effect. As a common catalyst, biochar is prepared from biomass through pyrolysis. Its microporous structure could provide a suitable growth environment for microorganisms [99]. The addition of biochar could increase the abundance of microbial sequences related to carbohydrate metabolism and stimulate microbial activity [100]. In order to obtain higher efficiency, improving the performance of the catalyst is conducive to the reaction. For instance, the acidity of biochar (Ni/AC, Co/AC and Ni–Co/AC) modified by the metal catalyst was improved and the depolymerization effect of lignin was strengthened. Of them, the high acidity of the Ni–Co/AC catalyst could promote the cracking of lignin C–C and C–O–C bonds [101].
The above materials showed good catalytic effects on the oxidative degradation of lignin under different conditions. The current research aims to improve the conversion efficiency and stability of the catalysts. Considering the need for environmental protection, the harmful by-products should be avoided in the catalytic process. The preparation of green reusable catalysts is an effective means to reduce the cost.
5. Biotransformation of lignocellulose
5.1. Fuel production
5.1.1. Bioethanol
The production of bioethanol is usually divided into three steps: (1) pretreatment of raw materials, (2) enzymatic hydrolysis of polysaccharides into monosaccharides, and (3) microbial fermentation to convert monosaccharides into ethanol [102]. Biomass is derived from a wide range of sources and is one of the raw materials for ethanol production, such as energy crops. As members of lignocellulose, energy crops show good ethanol production capacity, the characteristics of fast growth and adaptability to adverse environment. Muthuvelu et al. [103] evaluated the bioethanol production potential of four lignocellulosic materials (Typha angustifolia, Arundo donax, Saccharum arundinaceum, and Ipomoea carnea). In this process, various enzymes, such as laccase, cellulase, and β-glucosidase were co-immobilized for catalysis due to the low treatment efficiency and long treatment time of a single free enzyme. The co-immobilized enzyme system facilitated hydrolysis to release sugar monomers while degrading the biomass. The produced hydrolysate was fermented by Saccharomyces cerevisiae. The ethanol concentrations of the four lignocellulose were 3.58 ± 1.14, 3.98 ± 0.87, 4.18 ± 1.14 and 4.43 ± 0.98 g/L, respectively. The results showed that Saccharum arundinaceum and Ipomoea carnea were better and could be used as raw materials for ethanol production.
Rabee et al. [104] explored the feasibility of using rumen microorganisms from camels and sheep to hydrolyze barley straw to produce ethanol. After anaerobic cultivation for 3 days at 39 °C and pH 6.8, the hydrolysate was fermented to obtain a total ethanol yield of 0.92 and 0.88 mg/mL respectively. These microbiota contained a large number of cellulose-decomposing bacteria and had great potential in fuel production. Additionally, bamboo could be used as a raw material for ethanol production. Bacillus velezensis LC1 had good cellulose and hemicellulose degradation abilities. After degradation of bamboo shoots for 6 days, 55.30 ± 1.40 mg/L glucose and 488.81 ± 45.06 mg/L xylose were obtained. The hydrolysate was then fermented with S. cerevisiae and Escherichia coli KO11. Of them, S. cerevisiae fermented glucose and E. coli KO11 fermented xylose. The ethanol yield of 7.21 ± 0.24 g/L was obtained after 96 h. This result revealed the excellent value of bamboo shoots and the strong biotransformation ability of Bacillus velezensis LC1 [105]. Other relevant studies include grass [106], Arthrospira (Spirulina) platensis [107], etc.
In a follow-up study, researchers found various cellulose-degrading bacteria in insects, which could be used in the biotransformation process. Dar et al. [108] isolated 15 strains of bacteria from the gut system of the red flour beetle Tribolium castaneum, of which strain RSP75 showed the highest cellulolytic activity. Strain RSP75 was identified as Bacillus altitudinis, which showed great adaptability to the acidic conditions of pH 5.0. During co-culturing with S. cerevisiae, B. altitudinis RSP75 attacked the cellulose β-1,4 glycosidic bonds and released cellobiose, which was further hydrolyzed to glucose by cellobiase. Hemicellulose was cut into short xylan by endoxylanase, and then hydrolyzed to pentacarbon sugar by exoxylanase (Fig. 9). These sugars were metabolized through the Embden-Meyerhof-Parnas pathway and then converted to ethanol by yeast anaerobic fermentation.
Fig. 9.
Predicted mechanism/pathways used by B. altitudinis RSP75 for ethanol production [108].
However, it is noteworthy that S. cerevisiae cannot ferment xylose. Therefore, selecting the strains that ferment pentose is an effective way to improve biomass utilization. As a xylose fermentation yeast, Scheffersomyces stipitis can convert hexose and pentose into ethanol, which is suitable for industrial production. However, S. stipitis has a low tolerance to inhibitory chemicals [109]. To overcome this problem, Pereira et al. [110] improved the tolerance of Pichia pastoris to inhibitors such as acetic acid and lignosulfonate in hardwood sulfite wastewater by evolutionary engineering. Compared with the parent strain, the ethanol conversion rate increased by 6%. In another study, an anaerobic thermophilic bacterium Thermoanaerobacter italicus Pentocrobe 411 was reported for ethanol conversion. The strain showed good tolerance to sodium acetate and furfural and could ferment hexose and pentose. When corn cob, oil palm empty fruit string and oil palm leaf were fermented, 98–99% sugar conversion (glucose, xylose and arabinose) was obtained, and the maximum ethanol concentration was 94–106 g/L [111]. The discovery of these strains is crucial to improving the yield of ethanol. However, the efficient use of strains and the design of a reasonable fermentation process still need further investigation.
5.1.2. Biohydrogen
Biohydrogen is produced by photo fermentation (PF), dark fermentation (DF) and bio photolysis [112]. DF has become a common method in the lignocellulose-based hydrogen production process due to its low energy consumption [113]. The DF of rice straw after enzymatic hydrolysis with Clostridium pasteurianum obtained the maximum hydrogen yield of 23.96 mL/L/h in 96 h [114].
The combined application of DF and PF is also one of the most effective approaches to biohydrogen production due to its easy operation and high efficiency. Lu et al. [115] successfully established a co-culture model for the cellulose-degrading bacteria Clostridium cellulovorans 743B and photosynthetic bacteria Rhodopseudomonas palustris CGA009. The degree of cellulose degradation and hydrogen production in this mode was significantly higher than those of the monocultures. Clostridium cellulovorans used cellulose as a carbon source to produce hydrogen and volatile fatty acids (VFAs), while R. Palustris used VFAs produced by DF to stabilize the pH and produce hydrogen by PF. Co-cultured bacteria can be combined in various ways. Laurinavichene et al. [116] selected Clostridium butyricum and Rhodobacter sphaeroides for co-culture. The results demonstrated that R. Sphaeroides could inhibit C. butyricum. The hydrogen production capacity of the co-culture was lower than the single culture. Therefore, the relationship between the co-cultured microorganisms should be taken into consideration when selecting the co-cultured strains to avoid the occurrence of inhibition phenomenon leading to a poor final effect.
Finding the strains with high degradation and transformation has become a research hotspot in the field of biohydrogen production. Yin et al. [117] evaluated the hydrogen production capacity of the facultative anaerobic bacterium Enterococcus faecium INET2. Temperature, initial pH, substrate concentration and inoculation ratio were the parameters affecting hydrogen production. The comparative experimental results demonstrated that the optimal conditions were culture temperature of 35 °C, initial pH of 7, substrate concentration of 15 g/L glucose and inoculation ratio of 10%. When the cumulative hydrogen production peaked (130 mL H2/100 mL), the substrate degradation rate was as high as 93.3%. The thermophilic Thermoanaerobacterium sp. strain F6 exhibited the capacity to directly convert lignocellulose into hydrogen. Hydrogen production was enhanced with the increase in the xylan concentration, and the hydrogen production from 60 g/L xylan was 19 times higher than that of 5 g/L xylan. The strain grew well on unpretreated sugarcane bagasse (composed of 44% of cellulose and 34% of hemicellulose), indicating that the strain was capable of producing cellulose and hemicellulose-degrading enzymes. However, the presence of lignin prevents hydrogen production, so it is necessary to remove lignin during the process of biohydrogen production [118]. Zhao et al. [119] used Phanerochaete Chrysosporium to degrade corn straw. After treatment, the lignin content and crystallinity decreased, and the lignin removal rate reached 34.3% in 15 days. After the samples treated for 15 days were co-cultured with the crude cellulase extracted from Trichoderma viride for 96 h, the saccharification rate of the enzyme reached 47.3%, which was significantly higher than the untreated straw. Subsequently, the enzymatic hydrolysate was converted into hydrogen by Thermoanaerobacterium thermosaccharolyticum W16. After 36 h of fermentation, the cumulative hydrogen production of straw reached the highest value of 2812 mL/L. The above results revealed the importance of pretreatment, suggesting it to be an economic method to combine with enzyme saccharification.
5.1.3. Biomethane
Anaerobic digestion (AD) of lignocellulose is a complex process that can be divided into four phases: hydrolysis, fermentation, acidogenesis and methanogenesis [120]. Pretreatment before AD is a key step in the methane production process. Previous reports have demonstrated that although energy crops, agricultural residues and other biomass are rich in sources, pretreatment is necessary to maximize the utilization of raw materials and improve gas production. After pretreatment, the stubbornness of lignocellulose biomass was reduced and the accessibility of enzymes was improved [121].
Table 5 lists the methane yields from biomass after different biological pretreatments. The daily methane production of sawdust treated with lignocellulose-degrading microbial consortium (composed of bacteria and yeast) reached a maximum of 9.8 L kg−1 VS d−1 after 18 days of AD, while the daily methane production of untreated sawdust showed a maximum of 7.4 L kg−1 VS d−1 after 20 days of AD. It can be seen that pretreatment could improve methane production and accelerate the methane production rate [70]. Peng et al. [122] cultivated Clostridium cellustolyticum for AD using cellulose-disaccharide and wheat straw as carbon sources, respectively, to enhance the hydrolysis of substrates. Both biofortification methods could improve the biochemical methane potential. This result demonstrated the potential of biofortification to improve the efficiency of AD. However, it is worth ensuring that the strains used are able to adapt to the complex systems.
Table 5.
Effect of lignocellulose pretreatment on methane yield.
| Pretreatment method | Biomass | Operating Conditions | Methane yield | References |
|---|---|---|---|---|
| R. ornithinolytica strain MC3 | N.gaditana | The mixture was incubated for 48 h at 30 °C | 262.84 mL g VSS−1 | [55] |
| R. ornithinolytica strain MA5 | 282.92 mL g VSS−1 | |||
| Bacterial consortium SST-4 | Birchwood sawdust mixed with creosote | Treatment at 35 °C | 162.8 L/kg VS | [71] |
| Mixed enzyme | Scenedesmus obliquus | Treatment at 105 °C and 120 °C | 1050 ± 201 mL gVS−1 | [83] |
| Chlorella sorokiniana | 775 ± 253 mL gVS−1 | |||
| Arthrospira maxima | 1197 ± 254 mL gVS−1 | |||
| Chaetomium globosporum | Wheat | 36 °C temperature, 81% moisture content and 31 days residence time | 358 mL/gVS | [123] |
| Pearl millet straw | ||||
| Trichoderma reesei TISTR 3080 | Empty fruit bunches | A moisture content of 80% (w/w)with an incubation time of 15 days | 311.7 mL CH4 g−1 VS | [124] |
| Pleurotus ostreatus DSM 11191 | A moisture content of 85% (w/w)with an incubation time of 10 days | 267.1 mL CH4 g−1 VS |
Contaminated materials used in construction, industry and other fields, such as wood treated with creosote, can also be used in this process. Furthermore, researchers have screened a bacterial consortium SST-4 (including Acinetobacter calcoaceticus, Shewanella putrefaciens, Bacillus cereus and Novosphingobium taihuense) for degradation, detoxification and later transformation to reduce pollution. SST-4 showed cellular activity and a creosote degradation ability in the pH range of 5–8. Its treated biomass showed significant advantages in the process of AD, with an improved methane conversion rate. This study revealed the ability of SST-4 in bioremediation, degradation and transformation, and highlighted the role of pretreatment [71].
In the process of methane production, optimizing pretreatment conditions could improve the output and economic benefit, thereby improving the overall process. Yadav et al. [123] analyzed the effects of temperature, residence time and moisture content when Chaetomium globosporum was used to treat wheat and pearl millet straw to overcome this bottleneck. It was found that temperature and moisture content had a significant effect on the release of reducing sugar i.e., the increase of temperature and moisture content increased the release of reducing sugar. Additionally, the interaction effect between the two affected the treatment results (Fig. 10). The results showed that under the optimum conditions (36 °C temperature, 81% moisture content and 31 days residence time), lignin and cellulose were effectively degraded and the release of reducing sugar was significantly increased. Compared to the methane production potential of untreated and treated samples, the methane production of untreated wheat and pearl millet stalks were 263 mL/gVS and 226 mL/gVS respectively. After treatment, the methane production was increased to 358 mL/gVS. Suksong et al. [124] found that high moisture content conditions could promote microbial growth and metabolism, and corresponded to the high degradation of the material microstructure. Under the pretreatment conditions of 80–85% (w/w) water content and 10–15 days of culture time, Trichoderma reesei and Pleurotus ostreatus showed excellent degradation effects. The author summarized the kinetic parameters of methane production from oil palm empty fruit bunch pretreated with fungi through solid-state AD, and found that pretreatment could improve biomethane production and energy recovery, and shorten the lag phase time of the AD process. Therefore, biological pretreatment of raw materials and optimization of the process conditions are important means to adapt to commercial production.
Fig. 10.
Surface plots of interaction between (a) temperature and moisture content; (b) temperature and residence time; (c) residence time and moisture content [123].
5.2. Preparation of platform chemicals
Lignocellulose can be prepared into environment-friendly materials. For instance, composite materials with plant fiber as reinforcement have biodegradable properties. Research shows that the biocomposite flowerpot (BFP) made from straw fiber with a hydrolyzed soybean protein isolate/urea/formaldehyde copolymer-based adhesive had good biodegradation under the joint action of water and microorganisms. Introduction of the hydrolyzed soybean protein isolate improved the degradation rate of BFP. Of them, the combination with a higher C/N ratio was more conducive to microbial growth. Meanwhile, nitrate ions formed by nitrobacteria could promote plant growth [125]. In addition to being used for fuel production, lignocellulose components and derivatives can also be used for the production of fine chemicals. Cellulose is widely used in various fields. The non-toxicity of cellulose to cells makes it possible to develop biocompatible materials for 3D printing (biological ink) based on nano cellulose [126]. Additionally, the charged nature of the nano cellulose interface is helpful for improving the performance of hydrogels, promoting the dispersion of fillers or adjusting the physical properties [127]. Hemicellulose can be used to produce xylitol, which is obtained from hydrolysate by yeast. Bacteria and fungi can also be used to produce xylitol. Therefore, selective gene manipulation to improve the titer and productivity will become a production goal. For instance, approaches to improve xylitol production include optimizing the selective xylose transport system; overexpressing xylose reductase (XR); deleting xylitol dehydrogenase gene (XDH); and avoiding xylitol conversion into xylose [128]. For example, Jeon et al. [129] introduced the XR gene into the xylitol dehydrogenase gene disrupted Candida tropicalis to achieve high-level expression of XR. When cultured in a mixture of glucose and xylose, the production rate of the recombinant C. tropicalis was 73% higher than the parent strain, and the xylitol yield was 96%.
Effective utilization of lignin is very important for today's society. The various characteristics of lignin also help to improve the properties of materials. For instance, lignin could act as an ultraviolet absorber in polymer film. Film with 5% (w/w) lignin derivative could provide 91% UVA protection and 100% UVB protection. The mechanical properties and thermal stability of this film were also improved. This material is expected to be used in food or drug packaging [130]. The ferulic acid-based epoxy resin prepared using lignin-derived ferulic acid showed higher crosslinking density and tensile strength, and had good processability. The molten resin could maintain a liquid state for 3 h at room temperature, which could replace the traditional bisphenol A epoxy resin [131]. In addition, other products such as vanillin, 4-vinyl guaiacol and polyhydroxyalkanoates, can play an important role in the fields of spice, food and packaging material industries, as outlined in Table 6.
Table 6.
Conversion of lignin-related materials to chemicals by microorganism.
| Microorganism | Substrate | Chemical | Yield | References |
|---|---|---|---|---|
| Rhodococcus jostii RHA1 | Wheat straw | Vanillin | a maximum yield of 96 mg/L after 144 h | [132] |
| Pseudomonas putida KT2440 | Ferulic acid | Vanillin | the vanillin molar yield up to 86% within 3 h | [133] |
| Engineered P.putida KT2440 | Wheat straw | Ferulic acid | a high titer of 8.3 ± 3.2 mg L−1 | [134] |
| Oceanimonas doudorofii | Lignin and its derivatives (sinapinic acid and syringic acid) | Polyhydroxyalkanoate | The yields of PHA ranged from 0.2 to 2.7 wt % | [135] |
| Pseudomonas putida A514 | Vanillic acid | Polyhydroxyalkanoate | PHA titer increased to 246 mg/Liter via transcription level optimization | [136] |
| Rhodococcus opacus PD630 | Lignin | Lipid | a yield of 0.95 ± 0.01 g L−1 after a 4 d fermentation | [137] |
| Engineered P.putida KT2440 | Lignin-derived substrates | Pyruvate and l-lactate | an 82.5 ± 3.5% (wt/wt) yield of pyruvatel and l-lactate combined | [138] |
| Pseudomonas putida | Catechol | cis,cis-muconic acid | an MA titer of 64.2 g L−1 | [139] |
| Engineered Burkholderia glumae strain BGR1 | p-Coumaric acid | p-hydroxybenzoic acid | 10 mM pCA resulted in a high conversion yield of pHBA (89.5%) | [140] |
| Pseudomonas putida KT2440 | Softwood lignin | cis,cis-muconic acid | ∼20% yield (mol/mol) | [141] |
| Sphingobium sp. SYK-6 | Hardwood lignin | ∼45% yield (mol/mol) |
5.3. Bio-pulping
Using biotechnology to remove lignin during the biological pulping process is a potential alternative to chemical treatment as it can reduce the generation of toxic substances in wastewater. It has been reported that the depolymerization of rice straw with commercial microorganisms (mainly composed of Streptomyces rochei, Bacillus licheniformis, Bacillus subtilis and Aspergillus oryzae) could destroy the ester bond between lignin and hemicellulose, improve the crystallinity of fiber and improve the mechanical properties under an appropriate fermentation time. In the subsequent preparation of a composite membrane, pretreatment significantly shortened the required beating time and reduced the energy consumption by 43.75%. Meanwhile, the tensile index of the material was increased from 11.26 N/mg to 15.68 N/mg [142]. In another study, cotton pulp pretreated with 0.1% cellulase saved up to 26% of the energy. The tensile index, burst index and tear index of the paper were increased by 11.2%, 9.0% and 8.2%, respectively. In fact, better fibrosis was obtained by enzyme treatment, which improved the combination between fibers [143].
Development of the biological bleaching technology can reduce the consumption of chlorine-based chemicals. Shintani et al. [144] used Geotrichum candidum Dec 1 to decolorize oxygen delignified kraft pulp (OKP). When the pulp concentration was 25% for 6 days, the kappa value of the OKP decreased from 12.4 points to 10.4 points. Bacteria may be more conducive to the bleaching process than fungi. In a previous study, Streptomyces rutgersensis UTMC 2445 showed xylan decomposition activity. Its 5-day fermentation broth was applied to the bleaching of birch and oriental water gangqing mixed pulp. Under the optimum conditions (30 °C, treatment for 6 h and pH of 5), the whiteness increased by 7%, and the strain reduced the amount of chemical bleach in an environment-friendly manner [145].
Therefore, the use of biotechnologies for delignification of biomass materials and pulp wastewater can bring a series of benefits. Relevant studies have confirmed that biotechnology could reduce energy consumption. Therefore, it is essential to achieve more efficient degradation and improve the pulp performance for industrial production.
6. Economic evaluation
As previously mentioned, the utilization of lignocellulosic biomass to produce fuel and chemicals is an option to help alleviate the energy crisis. During the biorefinery process, a variety of conversion technologies have been applied. However, there are still challenges for achieving commercial development. Pretreatment and enzymatic hydrolysis are defined as the biorefinery core steps, accounting for more than half of the total cost. Therefore, it is vital to choose a suitable method. Physical and chemical treatments have the characteristics of rapidity and a high saccharification rate, but the chemical process tends to corrode the equipment and increase the maintenance costs. Acid pretreatment may produce phenolics that inhibit cellulase, which thus requires detoxification. Moreover, in order to facilitate subsequent enzymatic fermentation, a washing process is commonly required after alkali pretreatment, which increases the water consumption and generates a large amount of wastewater. Although wastewater can be recycled, the existing technologies cannot achieve complete recovery. In comparison, biological pretreatment does not require chemical reagents or other management operations, and the input cost of the preliminary process is lower than other treatments.
In the downstream process, acid hydrolysis can also be used to release sugar. The acid catalysts can function without pretreatment, and the hydrolysis rate is faster than that of enzymes. However, the difficulty in separating the acid and sugar has become the major obstacle to commercialization [146]. Therefore, biological treatment has become the preferred process. Bioconversion generally involves three steps, namely enzyme production, enzyme hydrolysis and fermentation. The economic benefits associated with different combinations are variable. To reduce the expenditure as much as possible, existing research integrates enzymatic hydrolysis and fermentation, which simplifies the operation steps. Compared to separate hydrolysis and fermentation (SHF) processes, simultaneous saccharification and fermentation (SSF) has a low contamination risk and increases the hydrogen yield [147]. Conversely, the consolidated bioprocessing (CPB) process encompasses the whole biotransformation process, further reducing the investment cost. It is estimated that CPB is able to reduce the total cost of biological processing by 77.8% [148]. For example, Myceliophthora thermophila has a large number of genes encoding lignocellulolytic enzymes that can rapidly degrade polysaccharides. During the CPB process, lignocellulose could be directly converted into four carbon 1,4-diacids using engineered Myceliophthora thermophila without adding hydrolytic enzymes [149].
Moreover, the development of new enzymes is another strategy to achieve sustainable development. For instance, lytic polysaccharide monooxygenase can cleave glycosidic bonds, while the synergy between LPMO and cellulase can promote cellulose conversion [150].
7. Future directions and challenges
The complex structure of lignocellulose hinders the contact of enzymes. Appropriate depolymerization is needed to improve the biotransformation efficiency. Although various microorganisms have been shown to be suitable for lignocellulose processes, the ability of a single strain is limited. Therefore, it is essential to find strains with high efficiency and high stability to improve industrial production. At present, the known microorganisms are only a small part of the natural system, and most microorganisms have not been recognized and developed. Microorganisms are widely distributed in soil, sludge, salt marshes and animal intestines. The required microorganisms can be screened by cultivation under specific conditions. Therefore, using genetic engineering and other technologies to modify strains or enzymes could be a potential strategy.
So far, many studies have reported the construction of engineered microorganisms. For instance, Coconi-Linares et al. [151] genetically modified Phanerochaete Chrysosporium for heterologous expression of the vpl2 (multifunctional peroxidase) optimized gene as a host. The transformation step of the strain during the transformation process was mediated by shock wave, showing that the recombinant strain could express MnP1, LiPH8 and VPL2 simultaneously, and the enzyme activity was increased significantly. This study provides a feasible operation scheme for increasing the yield of lignin-degrading enzymes. Chiang et al. [152] constructed an E. coli BL-G lacking ptsG (glucose specific transporter) gene, which could ferment xylose and glucose simultaneously, but the glucose utilization rate was low. Therefore, the glucose facilitator gene of Zymomonas mobilis was introduced to improve the glucose utilization ability of the strain. Additionally, a series of operations were carried out on the strain, such as increasing the activity of pentose phosphate pathway and deleting organic acid synthesis genes to improve the utilization rate of xylose. The final strain BL-A4 showed high utilization of xylose and glucose, and the ethanol yield was up to 29 g/L, accounting for 97% of the theoretical value.
Additionally, as an effective method to improve the degradation process, the catalyst should not only improve the degradation efficiency, but also play a role in biotransformation to improve the output. This is especially true for carbon based catalysts, which have a wide range of sources and low cost, resulting in a greater utilization value. The development of a high-efficiency catalyst in an economically appropriate range will be the key goal of the future research. Subsequent studies should also focus on how to shorten the process time, eliminate the lag period, expand the substrate scope and comprehensively improve the value utilization of lignocellulose.
The application of nanotechnology also provides support for the development of biotechnologies. The large surface area, high catalytic efficiency and strong specificity of nanoparticles are key factors [153]. At the same time, the application of nanoparticles can form more active sites and accelerate the reaction rate. In addition, the combined application of nanotechnology and hydrolases in biorefinery has piqued widespread interest. The recyclability of magnetic nanoparticles is crucial for achieving sustainable development. Nanoparticles, nanotubes, nanosheets and other nanoscale materials can be used as supporting materials for enzymes [154]. Furthermore, compared with traditional pretreatment methods, nanotechnology has fewer restrictions. The catalytic performance of hydrolases immobilized on nanomaterials is enhanced, making this technique suitable for more refined production processes. Previous research has illustrated that immobilization could improve the thermostability and reusability of the enzymes [155]. In one such study, cellulase immobilized on functionalized magnetic nanospheres by electrostatic adsorption exhibited 87% activity recovery and broader pH stability [156]. Similarly, although the activity of Aspergillus niger cellulase immobilized onto β-cyclodextrin-conjugated magnetic particles decreased, it could be reused 16 times, and a higher amount of glucose could be obtained relative to that of free cellulase [157]. In conclusion, immobilized enzymes have great economic value in high-value production and can be effectively applied in various lignocellulose processes.
8. Conclusion
This review mainly focuses on lignocellulose degradation technologies related to biological methods. In this regard, enzyme treatment has gained increasing attention due to the advantages of mild and simple biological treatment conditions. The current research directions mainly focus on the following aspects: (1) searching for new efficient functional strains or enzymes; (2) deeply understanding the degradation mechanisms; (3) determining the best pretreatment conditions; (4) improving the function of strains through genetic engineering; and (5) adding auxiliary materials to strengthen the process.
Additionally, this article summarizes the potential value of lignocellulose in high-value utilization. As a renewable resource, lignocellulose could be a potential alternative to fossil fuels. In the process of biotransformation, high-yield bacteria screening, designing reasonable transformation process, improving strain stability, and avoiding feedback inhibition could meet the needs of industrial mass production. However, finding an efficient and universal solution is highly necessary for achieving sustainable development and improving economic benefits.
Overall, the biological refining of lignocellulose is still a complex process. When strengthening a single step, we should also pay attention to the relationship between different processes, and improve the process performance on the basis of biotechnology combined with other methods, so as to realize the efficient utilization of lignocellulose.
Credit author statement
Zengyou Wu: Writing – original draft, Writing – review & editing, Kun Peng: Software, Data curation, Software, Visualization, Yin Zhang: Writing – review & editing, Mei Wang: Review & editing, Validation, Cheng Yong: Validation, Formal analysis, Investigation, Ling Chen: Validation, Investigation, Ping Qu: Methodology, Project administration, Funding acquisition, Validation, Hongying Huang: Methodology, Project administration, Funding acquisition, Validation, Resources, Enhui Sun: Conceptualization, Co-Supervision, Methodology, Project administration, Funding acquisition, Validation, Resources, Writing review & editing, Mingzhu Pan: Conceptualization, Supervision, Methodology, Validation, Formal analysis, Review & editing.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgments
This research was supported by the Exploring and Overturning Innovation Program of Jiangsu Academy of Agricultural Sciences (ZX(21)1221), Key Research & Development Program of Jiangsu Province (BE2020335), Jiangsu Province Agricultural Independent Innovation Fund (CX(19)2003), and National Natural Science Foundation of China (41807132).
Contributor Information
Enhui Sun, Email: enhsun@126.com.
Mingzhu Pan, Email: mzpan@njfu.edu.cn.
Data availability
The data that has been used is confidential.
References
- 1.Baruah J., Nath B.K., Sharma R., Kumar S., Deka R.C., Baruah D.C., Kalita E. Recent trends in the pretreatment of lignocellulosic biomass for value-added products. Front. Energy Res. 2018;6:141. doi: 10.3389/fenrg.2018.00141. [DOI] [Google Scholar]
- 2.Liu Y.J., Li B., Feng Y.G., Cui Q. Consolidated bio-saccharification: leading lignocellulose bioconversion into the real world. Biotechnol. Adv. 2020;40 doi: 10.1016/j.biotechadv.2020.107535. [DOI] [PubMed] [Google Scholar]
- 3.López-Mondéjar R., Algora C., Baldrian P. Lignocellulolytic systems of soil bacteria: a vast and diverse toolbox for biotechnological conversion processes. Biotechnol. Adv. 2019;37 doi: 10.1016/j.biotechadv.2019.03.013. [DOI] [PubMed] [Google Scholar]
- 4.Ren N.Q., Zhao L., Chen C., Guo W.Q., Cao G.L. A review on bioconversion of lignocellulosic biomass to H2: key challenges and new insights. Bioresour. Technol. 2016;215:92–99. doi: 10.1016/j.biortech.2016.03.124. [DOI] [PubMed] [Google Scholar]
- 5.Shirkavand E., Baroutian S., Gapes D.J., Young B.R. Combination of fungal and physicochemical processes for lignocellulosic biomass pretreatment – a review. Renew. Sustain. Energy Rev. 2016;54:217–234. doi: 10.1016/j.rser.2015.10.003. [DOI] [Google Scholar]
- 6.Liu H., Sun J.L., Leu S.Y., Chen S.C. Toward a fundamental understanding of cellulase-lignin interactions in the whole slurry enzymatic saccharification process. Biofuels, Bioprod. Biorefin. 2016;10:648–663. doi: 10.1002/bbb.1670. [DOI] [Google Scholar]
- 7.Yoo C.G., Meng X.Z., Pu Y.Q., Ragauskas A.J. The critical role of lignin in lignocellulosic biomass conversion and recent pretreatment strategies: a comprehensive review. Bioresour. Technol. 2020;301 doi: 10.1016/j.biortech.2020.122784. [DOI] [PubMed] [Google Scholar]
- 8.Zahoor W. Wang, Tan X.S., Imtiaz M., Wang Q.F., Miao C.L., Yuan Z.H., Zhuang X.S. Rice straw pretreatment with KOH/urea for enhancing sugar yield and ethanol production at low temperature. Ind. Crop. Prod. 2021;170 doi: 10.1016/j.indcrop.2021.113776. [DOI] [Google Scholar]
- 9.Tarrsini M., Ng Q.H., Teoh Y.P., Shuit S.H., Ooi Z.X., Kunasundari B. Biomass Convers. Biorefin.; 2021. Structural and Composition Modification of Harum Manis Mango (Mangifera Indica) Leaves via Chemical Pretreatment for Bioethanol Production. [DOI] [Google Scholar]
- 10.Wang W., Lee D.J. Lignocellulosic biomass pretreatment by deep eutectic solvents on lignin extraction and saccharification enhancement: a review. Bioresour. Technol. 2021;339 doi: 10.1016/j.biortech.2021.125587. [DOI] [PubMed] [Google Scholar]
- 11.Mohammadi M., Shafiei M., Abdolmaleki A., Karimi K., Mikkola J.P., Larsson C. A morpholinium ionic liquid for rice straw pretreatment to enhance ethanol production. Ind. Crop. Prod. 2019;139 doi: 10.1016/j.indcrop.2019.111494. [DOI] [Google Scholar]
- 12.Chen H., Huo W.Z., Wang B.X., Wang Y., Wen H., Cai D., Zhang C.W., Wu Y.L., Qin P.Y. L-lactic acid production by simultaneous saccharification and fermentation of dilute ethylediamine pre-treated rice straw. Ind. Crop. Prod. 2019;141 doi: 10.1016/j.indcrop.2019.111749. [DOI] [Google Scholar]
- 13.Melro E., Alves L., Antunes F.E., Medronho B. A brief overview on lignin dissolution. J. Mol. Liq. 2018;265:578–584. doi: 10.1016/j.molliq.2018.06.021. [DOI] [Google Scholar]
- 14.Satlewal A., Agrawal R., Das P., Bhagia S., Pu Y.Q., Puri S.K., Ramakumar S.S.V., Ragauskas A.J. Assessing the facile pretreatments of bagasse for efficient enzymatic conversion and their impacts on structural and chemical properties. ACS Sustain. Chem. Eng. 2019;7:1095–1104. doi: 10.1021/acssuschemeng.8b04773. [DOI] [Google Scholar]
- 15.Xu G.C., Ding J.C., Han R.Z., Dong J.J., Ni Y. Enhancing cellulose accessibility of corn stover by deep eutectic solvent pretreatment for butanol fermentation. Bioresour. Technol. 2016;203:364–369. doi: 10.1016/j.biortech.2015.11.002. [DOI] [PubMed] [Google Scholar]
- 16.Procentese A., Raganati F., Olivieri G., Russo M.E., Rehmann L., Marzocchella A. Low-energy biomass pretreatment with deep eutectic solvents for bio-butanol production. Bioresour. Technol. 2017;243:464–473. doi: 10.1016/j.biortech.2017.06.143. [DOI] [PubMed] [Google Scholar]
- 17.Duan C., Wang X.Q., Zhang Y.L., Xu Y.J., Ni Y.H. Fractionation and cellulase treatment for enhancing the properties of kraft-based dissolving pulp. Bioresour. Technol. 2016;224:439–444. doi: 10.1016/j.biortech.2016.10.077. [DOI] [PubMed] [Google Scholar]
- 18.Nie S., Wang S., Qin C., Yao S., Ebonka J.F., Song X., Li K. Removal of hexenuronic acid by xylanase to reduce adsorbable organic halides formation in chlorine dioxide bleaching of bagasse pulp. Bioresour. Technol. 2015;196:413–417. doi: 10.1016/j.biortech.2015.07.115. [DOI] [PubMed] [Google Scholar]
- 19.Keegstra K. Plant cell walls. Plant Physiol. 2010;154:483–486. doi: 10.1104/pp.110.161240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhang Y., Yu J.Y., Wang X., Durachko D.M., Zhang S.L., Cosgrove D.J. Molecular insights into the complex mechanics of plant epidermal cell walls. Science. 2021;372:706–711. doi: 10.1126/science.abf2824. [DOI] [PubMed] [Google Scholar]
- 21.Kang X., Kirui A., Widanage M.C.D., Mentink-Vigier F., Cosgrove D.J., Wang T. Lignin-polysaccharide interactions in plant secondary cell walls revealed by solid-state NMR. Nat. Commun. 2019;10:347. doi: 10.1038/s41467-018-08252-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ding S.Y., Liu Y.S., Zeng Y.N., Himmel M.E., Baker J.O., Bayer E.A. How does plant cell wall nanoscale Architecture correlate with enzymatic digestibility? Science. 2012;338:1055–1060. doi: 10.1126/science.1227491. [DOI] [PubMed] [Google Scholar]
- 23.Menon V., Rao M. Trends in bioconversion of lignocellulose: biofuels, platform chemicals & biorefinery concept. Prog. Energy Combust. Sci. 2012;38:522–550. doi: 10.1016/j.pecs.2012.02.002. [DOI] [Google Scholar]
- 24.Shill K., Miller K., Clark D.S., Blanch H.W. A model for optimizing the enzymatic hydrolysis of ionic liquid-pretreated lignocellulose. Bioresour. Technol. 2012;126:290–297. doi: 10.1016/j.biortech.2012.08.062. [DOI] [PubMed] [Google Scholar]
- 25.Kassaye S., Pant K.K., Jain S. Synergistic effect of ionic liquid and dilute sulphuric acid in the hydrolysis of microcrystalline cellulose. Fuel Process. Technol. 2016;148:289–294. doi: 10.1016/j.fuproc.2015.12.032. [DOI] [Google Scholar]
- 26.Kumar G., Dharmaraja J., Arvindnarayan S., Shoban S., Bakonyi P., Saratale G.D., Nemestothy N., Belafi-Bako K., Yoon J.J., Kim S.H. A comprehensive review on thermochemical, biological, biochemical and hybrid conversion methods of bio-derived lignocellulosic molecules into renewable fuels. Fuel. 2019;251:352–367. doi: 10.1016/j.fuel.2019.04.049. [DOI] [Google Scholar]
- 27.Woiciechowski A.L., Neto C.J.D., Vandenberghe L.P.D., Neto D.P.D., Sydney A.C.N., Letti L.A., Karp S.G., Torres L.A.Z., Soccol C.R. Lignocellulosic biomass: acid and alkaline pretreatments and their effects on biomass recalcitrance – conventional processing and recent advances. Bioresour. Technol. 2020;304 doi: 10.1016/j.biortech.2020.122848. [DOI] [PubMed] [Google Scholar]
- 28.Ma J.P., Shi S., Jia X.Q., Xia F., Ma H., Gao J., Xu J. Advances in catalytic conversion of lignocellulose to chemicals and liquid fuels. J. Energy Chem. 2019;36:74–86. doi: 10.1016/j.jechem.2019.04.026. [DOI] [Google Scholar]
- 29.Wang F.Q., Ouyang D.H., Zhou Z.Y., Page S.J., Liu D.H., Zhao X.B. Lignocellulosic biomass as sustainable feedstock and materials for power generation and energy storage. J. Energy Chem. 2020;57:247–280. doi: 10.1016/j.jechem.2020.08.060. [DOI] [Google Scholar]
- 30.Ponnusamy V.K., Nguyen D.D., Dharmaraja J., Shobana S., Banu J.R., Saratale R.G., Chang S.W., Kumar G. A review on lignin structure, pretreatments, fermentation reactions and biorefinery potential. Bioresour. Technol. 2019;271:462–472. doi: 10.1016/j.biortech.2018.09.070. [DOI] [PubMed] [Google Scholar]
- 31.Chen Z., Wan C.X. Biological valorization strategies for converting lignin into fuels and chemicals. Renew. Sustain. Energy Rev. 2017;73:610–621. doi: 10.1016/j.rser.2017.01.166. [DOI] [Google Scholar]
- 32.Nishimura H., Kamiya A., Nagata T., Katahira M., Watanabe T. Direct evidence for α ether linkage between lignin and carbohydrates in wood cell walls. Sci. Rep. 2018;8:6538. doi: 10.1038/s41598-018-24328-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Liu Q.Q., Luo L., Lignins L.Q. Zheng. Biosynthesis and biological functions in plants. Int. J. Mol. Sci. 2018;19:335–351. doi: 10.3390/ijms19020335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.dos Santos A.C., Ximenes E., Kim Y., Ladisch M.R. Lignin–enzyme interactions in the hydrolysis of lignocellulosic biomass. Trends Biotechnol. 2019;37:518–531. doi: 10.1016/j.tibtech.2018.10.010. [DOI] [PubMed] [Google Scholar]
- 35.Parawira W. Enzyme research and applications in biotechnological intensification of biogas production. Crit. Rev. Biotechnol. 2012;32:172–186. doi: 10.3109/07388551.2011.595384. [DOI] [PubMed] [Google Scholar]
- 36.Wan C.X., Li Y.B. Fungal pretreatment of lignocellulosic biomass. Biotechnol. Adv. 2012;30:1447–1457. doi: 10.1016/j.biotechadv.2012.03.003. [DOI] [PubMed] [Google Scholar]
- 37.Asina F.N.U., Brzonova I., Kozliak E., Kubatova A., Ji Y. Microbial treatment of industrial lignin: successes, problems and challenges. Renew. Sustain. Energy Rev. 2017;77:1179–1205. doi: 10.1016/j.rser.2017.03.098. [DOI] [Google Scholar]
- 38.Guo H.L., Wang X.D., Lee D.J. Proteomic researches for lignocellulose-degrading enzymes: a mini-review. Bioresour. Technol. 2018;265:532–541. doi: 10.1016/j.biortech.2018.05.101. [DOI] [PubMed] [Google Scholar]
- 39.Saini S., Sharma K.K. Fungal lignocellulolytic enzymes and lignocellulose: a critical review on their contribution to multiproduct biorefinery and global biofuel research. Int. J. Biol. Macromol. 2021;193:2304–2319. doi: 10.1016/j.ijbiomac.2021.11.063. [DOI] [PubMed] [Google Scholar]
- 40.Ma J.S., Yue H.M., Li H.Q., Zhang J., Zhang Y.H., Wang X.L., Gong S., Liu G.Q. Selective delignification of poplar wood with a newly isolated white-rot basidiomycete Peniophora incarnata T-7 by submerged fermentation to enhance saccharification. Biotechnol. Biofuels. 2021;14:135. doi: 10.1186/s13068-021-01986-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Zhang Z.C., Shah A.M., Mohamed H., Zhang Y., Tsiklauri N., Song Y.D. Genomic studies of white-rot fungus Cerrena unicolor SP02 provide insights into food safety value-added utilization of non-food lignocellulosic biomass. J. Fungi. 2021;7:835. doi: 10.3390/jof7100835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.van Erven G., Hilgers R., de Waard P., Gladbeek E.J., van Berkel W.J.H., Kabel M.A. Elucidation of in situ ligninolysis mechanisms of the selective white-rot fungus Ceriporiopsis subvermispora. ACS Sustain. Chem. Eng. 2019;7:16757–16764. doi: 10.1021/acssuschemeng.9b04235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Zheng M.H., Zuo S.S., Niu D.Z., Jiang D., Tao Y., Xu C.C. Effect of four species of white rot fungi on the chemical composition and in vitro rumen degradability of naked oat straw. Waste Biomass Valori. 2021;12:435–443. doi: 10.1007/s12649-020-00991-w. [DOI] [Google Scholar]
- 44.Xie C.L., Gong W.B., Zhu Z.H., Zhou Y.J., Xu C., Yan L., Hu Z.X., Ai L.Z., Peng Y.D. Comparative secretome of white-rot fungi reveals co-regulated carbohydrate-active enzymes associated with selective ligninolysis of ramie stalks. Microb. Biotechnol. 2021;14:911–922. doi: 10.1111/1751-7915.13647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Liu L., Qian C., Jiang L., Yu H.Q. Direct three-dimensional characterization and multiscale visualization of wheat straw deconstruction by white rot fungus. Environ. Sci. Technol. 2014;48:9819–9825. doi: 10.1021/es5020983. [DOI] [PubMed] [Google Scholar]
- 46.Zhang J.W., Silverstein K.A.T., Castao J.D., Figueroa M., Schilling J.S. Gene regulation shifts shed light on fungal adaption in plant biomass decomposers. mBio. 2019;10 doi: 10.1128/mBio.02176-19. e02176-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Presley G.N., Schilling J.S. Distinct growth and secretome strategies for two taxonomically divergent Brown rot fungi. Appl. Environ. Microbiol. 2017;83 doi: 10.1128/AEM.02987-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ray M.J., Leak D.J., Spanu P.D., Murphy R.J. Brown rot fungal early stage decay mechanism as a biological pretreatment for softwood biomass in biofuel production. Biomass Bioenergy. 2010;34:1257–1262. doi: 10.1016/j.biombioe.2010.03.015. [DOI] [Google Scholar]
- 49.Zhang J.W., Presley G.N., Hammel K.E., Ryu J.S., Menke J.R., Figueroa M., Hu D.H., Orr G., Schilling J.S. Localizing gene regulation reveals a staggered wood decay mechanism for the brown rot fungus Postia placenta. Proc. Natl. Acad. Sci. U. S. A. 2016;113:10968–10973. doi: 10.1073/pnas.1608454113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zhu Y., Li W., Meng D.S., Li X.J., Goodell B. Non-enzymatic modification of the crystalline structure and chemistry of Masson pine in brown-rot decay. Carbohydr. Polym. 2022;286 doi: 10.1016/j.carbpol.2022.119242. [DOI] [PubMed] [Google Scholar]
- 51.Arantes V., Jellison J., Goodell B. Peculiarities of brown-rot fungi and biochemical Fenton reaction with regard to their potential as a model for bioprocessing biomass. Appl. Microbiol. Biotechnol. 2012;94:323–338. doi: 10.1007/s00253-012-3954-y. [DOI] [PubMed] [Google Scholar]
- 52.Zhu N., Zhu Y.Y., Li B.Q., Jin H.M., Dong Y.W. Increased enzyme activities and fungal degraders by Gloeophyllum trabeum inoculation improve lignocellulose degradation efficiency during manure-straw composting. Bioresour. Technol. 2021;337 doi: 10.1016/j.biortech.2021.125427. [DOI] [PubMed] [Google Scholar]
- 53.Cai L., Chen T.B., Zheng S.W., Liu H.T., Zheng G.D. Decomposition of lignocellulose and readily degradable carbohydrates during sewage sludge biodrying, insights of the potential role of microorganisms from a metagenomic analysis. Chemosphere. 2018;201:127–136. doi: 10.1016/j.chemosphere.2018.02.177. [DOI] [PubMed] [Google Scholar]
- 54.Zhang S.B., Xia T.Y., Wang J.L., Zhao Y., Xie X.Y., Wei Z.M., Zhang X., Song C.H., Song X.Y. Role of Bacillus inoculation in rice straw composting and bacterial community stability after inoculation: unite resistance or individual collapse. Bioresour. Technol. 2021;337 doi: 10.1016/j.biortech.2021.125464. [DOI] [PubMed] [Google Scholar]
- 55.Muñoz C., Hidalgo C., Zapata M., Jeison D., Riquelme C., Rivas M. Use of cellulolytic marine bacteria for enzymatic pretreatment in microalgal biogas production. Appl. Environ. Microbiol. 2014;80:4199–4206. doi: 10.1128/AEM.00827-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Bugg T.D.H., Ahmad M., Hardiman E.M., Rahmanpour R. Pathways for degradation of lignin in bacteria and fungi. Nat. Prod. Rep. 2011;28(12):1883–1896. doi: 10.1039/c1np00042j. [DOI] [PubMed] [Google Scholar]
- 57.Becker J., Kuhl M., Kohlstedt M., Starck S., Wittmann C. Metabolic engineering of Corynebacterium glutamicum for the production of cis, cis-muconic acid from lignin. Microb. Cell Factories. 2018;17:115. doi: 10.1186/s12934-018-0963-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Granja-Travez R.S., Persinoti G.F., Squina F.M., Bugg T.D.H. Functional genomic analysis of bacterial lignin degraders: diversity in mechanisms of lignin oxidation and metabolism. Appl. Microbiol. Biotechnol. 2020;104:3305–3320. doi: 10.1007/s00253-019-10318-y. [DOI] [PubMed] [Google Scholar]
- 59.Zhu D.C., Zhang P.P., Xie C.X., Zhang W.M., Sun J.Z., Qian W.J., Yang B. Biodegradation of alkaline lignin by Bacillus ligniniphilus L1. Biotechnol. Biofuels. 2017;10:44. doi: 10.1186/s13068-017-0735-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Tsegaye B., Balomajumder C., Roy P. Isolation and characterization of novel lignolytic, cellulolytic, and hemicellulolytic bacteria from wood-feeding termite cryptotermes brevis. Int. Microbiol. 2018;22:29–39. doi: 10.1007/s10123-018-0024-z. [DOI] [PubMed] [Google Scholar]
- 61.Xu Z.X., Qin L., Cai M.F., Hua W.B., Jin M.J. Biodegradation of kraft lignin by newly isolated Klebsiella pneumoniae, Pseudomonas putida, and Ochrobactrum tritici strains. Environ. Sci. Pollut. Res. 2018;25:14171–14181. doi: 10.1007/s11356-018-1633-y. [DOI] [PubMed] [Google Scholar]
- 62.Cortes-Tolalpa L., Norder J., van Elsas J.D., Salles J.F. Halotolerant microbial consortia able to degrade highly recalcitrant plant biomass substrate. Appl. Microbiol. Biotechnol. 2018;102:2913–2927. doi: 10.1007/s00253-017-8714-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Jiang C., Cheng Y., Zang H.L., Chen X., Wang Y., Zhang Y.T., Wang J.M., Shen X.H., Li C.Y. Biodegradation of lignin and the associated degradation pathway by psychrotrophic Arthrobacter sp. C2 from the cold region of China. Cellulose. 2020;27:1423–1440. doi: 10.1007/s10570-019-02858-3. [DOI] [Google Scholar]
- 64.Mathews S.L., Epps M.J., Blackburn R.K., Goshe M.B., Grunden A.M., Dunn R.R. Public questions spur the discovery of new bacterial species associated with lignin bioconversion of industrial waste. R. Soc. Open Sci. 2019;6 doi: 10.1098/rsos.180748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Walls L.E., Rios-Solis L. Sustainable production of microbial isoprenoid derived advanced biojet fuels using different generation feedstocks: a review. Front. Bioeng. Biotechnol. 2020;8 doi: 10.3389/fbioe.2020.599560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Zheng G.X., Yin T., Lu Z.X., Boboua S.Y.B., Li J.C., Zhou W.L. Degradation of rice straw at low temperature using a novel microbial consortium LTF-27 with efficient ability. Bioresour. Technol. 2020;304 doi: 10.1016/j.biortech.2020.123064. [DOI] [PubMed] [Google Scholar]
- 67.Xu C.F., Su X., Wang J.H., Zhang F.Z., Shen G.N., Yuan Y., Yan L., Tang H.Z., Song F.Q., Wang W.D. Characteristics and functional bacteria in a microbial consortium for rice straw lignin-degrading. Bioresour. Technol. 2021;331 doi: 10.1016/j.biortech.2021.125066. [DOI] [PubMed] [Google Scholar]
- 68.Zhang D.D., Wang Y., Zheng D., Guo P., Cheng W., Cui Z.J. New combination of xylanolytic bacteria isolated from the lignocellulose degradation microbial consortium XDC-2 with enhanced xylanase activity. Bioresour. Technol. 2016;221:686–690. doi: 10.1016/j.biortech.2016.09.087. [DOI] [PubMed] [Google Scholar]
- 69.Chu X.D., Awasthi M.K., Liu Y.Y.N., Cheng Q.S., Qu J.B., Sun Y. Studies on the degradation of corn straw by combined bacterial cultures. Bioresour. Technol. 2021;320 doi: 10.1016/j.actatropica.2020.124174. [DOI] [PubMed] [Google Scholar]
- 70.Ali S.S., Abomohra A., Sun J.Z. Effective bio-pretreatment of sawdust waste with a novel microbial consortium for enhanced biomethanation. Bioresour. Technol. 2017;238:425–432. doi: 10.1016/j.biortech.2017.03.187. [DOI] [PubMed] [Google Scholar]
- 71.Ali S.S., Jiao H.X., Mustafa A.M., Koutra E., El-Sapagh S., Kornaros M., Elsamahy T., Khalil M., Bulgariu L., Sun J.Z. Construction of a novel microbial consortium valued for the effective degradation and detoxification of creosote-treated sawdust along with enhanced methane production. J. Hazard Mater. 2021;418 doi: 10.1016/j.jhazmat.2021.126091. [DOI] [PubMed] [Google Scholar]
- 72.Bhushan S., Rana M.S., Bhandari M., Sharma A.K., Simsek H., Prajapati S.K. Enzymatic pretreatment of algal biomass has different optimal conditions for biogas and bioethanol routes. Chemosphere. 2021;284 doi: 10.1016/j.chemosphere.2021.131264. [DOI] [PubMed] [Google Scholar]
- 73.Zhu D.C., Liang N., Zhang R.X., Ahmad F., Zhang W.M., Yang B., Wu J., Geng A., Gabriel M., Sun J.Z. Insight into depolymerization mechanism of bacterial laccase for lignin. ACS Sustain. Chem. Eng. 2020;8:12920–12933. doi: 10.1021/acssuschemeng.0c03457. [DOI] [Google Scholar]
- 74.Pollegioni L., Tonin F., Rosini E. Lignin-degrading enzymes. FEBS J. 2015;282:1190–1213. doi: 10.1111/febs.13224. [DOI] [PubMed] [Google Scholar]
- 75.Wang X.L., Yao B., Su X.Y. Linking enzymatic oxidative degradation of lignin to organics detoxification. Int. J. Mol. Sci. 2018;19:3373. doi: 10.3390/ijms19113373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Macdonald J., Goacher R.E., Abou-Zaid M., Master E.R. Comparative analysis of lignin peroxidase and manganese peroxidase activity on coniferous and deciduous wood using ToF-SIMS. Appl. Microbiol. Biotechnol. 2016;100:8013–8020. doi: 10.1007/s00253-016-7560-2. [DOI] [PubMed] [Google Scholar]
- 77.Oates N.C., Abood A., Schirmacher A.M., Alessi A.M., Bird S.M., Bennett J.P., Leadbeater D.R., Li Y., Dowle A.A., Liu S. A multi-omics approach to lignocellulolytic enzyme discovery reveals a new ligninase activity from Parascedosporium putredinis NO1. Proc. Natl. Acad. Sci. U. S. A. 2021;118 doi: 10.1073/pnas.2008888118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Rashid G.M.M., Taylor C.R., Liu Y.Q.X., Zhang X.Y., Rea D., Fulop V., Bugg T.D.H. Identification of manganese superoxide dismutase from sphingobacterium sp. T2 as a novel bacterial enzyme for lignin oxidation. ACS Chem. Biol. 2015;10:2286–2294. doi: 10.1021/acschembio.5b00298. [DOI] [PubMed] [Google Scholar]
- 79.Mnich E., Vanholme R., Oyarce P., Liu S., Lu F.C., Goeminne G., Jorgensen B., Motawie M.S., Boerjan W., Ralph J., Ulvskov P., Moller B.L., Bjarnholt N., Harholt J. Degradation of lignin β-aryl ether units in Arabidopsis thaliana expressing LigD, LigF and LigG from Sphingomonas paucimobilis SYK-6. Plant Biotechnol. J. 2017;15:581–593. doi: 10.1111/pbi.12655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Pérez-Rodríguez N., Garcia-Bernet D., Dominguez J.M. Effects of enzymatic hydrolysis and ultrasounds pretreatments on corn cob and vine trimming shoots for biogas production. Bioresour. Technol. 2016;221:130–138. doi: 10.1016/j.biortech.2016.09.013. [DOI] [PubMed] [Google Scholar]
- 81.Cakmak E.K., Ugurlu A. Enhanced biogas production of red microalgae via enzymatic pretreatment and preliminary economic assessment. Algal Res. 2020;50 doi: 10.1016/j.algal.2020.101979. [DOI] [Google Scholar]
- 82.Passos F., Hom-Diaz A., Blanquez P., Vicent T., Ferrer I. Improving biogas production from microalgae by enzymatic pretreatment. Bioresour. Technol. 2016;199:347–351. doi: 10.1016/j.biortech.2015.08.084. [DOI] [PubMed] [Google Scholar]
- 83.Ometto F., Quiroga G., Psenicka P., Whitton R., Jefferson B., Villa R. Impacts of microalgae pre-treatments for improved anaerobic digestion: thermal treatment, thermal hydrolysis, ultrasound and enzymatic hydrolysis. Water Res. 2014;65:350–361. doi: 10.1016/j.watres.2014.07.040. [DOI] [PubMed] [Google Scholar]
- 84.Wang R.Z., You T.T., Yang G.H., Xu F. Efficient short time white rot Brown rot fungal pretreatments for the enhancement of enzymatic saccharification of corn cobs. ACS Sustain. Chem. Eng. 2017;5:10849–10857. doi: 10.1021/acssuschemeng.7b02786. [DOI] [Google Scholar]
- 85.Hermosilla E., Rubilar O., Schalchli H., da Silva A.S., Ferreira-Leitao V., Diez M.C. Sequential white-rot and brown-rot fungal pretreatment of wheat straw as a promising alternative for complementary mild treatments. Waste Manage. (Tucson, Ariz.) 2018;79:240–250. doi: 10.1016/j.wasman.2018.07.044. [DOI] [PubMed] [Google Scholar]
- 86.Chen K.J., Tang J.C., Xu B.H., Lan S.L., Cao Y.K. Degradation enhancement of rice straw by co-culture of Phanerochaete chrysosporium and Trichoderma viride. Sci. Rep. 2019;9 doi: 10.1038/s41598-019-56123-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Ishola M.M., Isroi, Taherzadeh M.J. Effect of fungal and phosphoric acid pretreatment on ethanol production from oil palm empty fruit bunches (OPEFB) Bioresour. Technol. 2014;165:9–12. doi: 10.1016/j.biortech.2014.02.053. [DOI] [PubMed] [Google Scholar]
- 88.Liang J.J., Fang X.X., Lin Y.Q., Wang D.H. A new screened microbial consortium OEM2 for lignocellulosic biomass deconstruction and chlorophenols detoxification. J. Hazard Mater. 2018;347:341–348. doi: 10.1016/j.jhazmat.2018.01.023. [DOI] [PubMed] [Google Scholar]
- 89.Wu D., Wei Z.M., Zhao Y., Zhao X.Y., Mohamed T.A., Zhu L.J., Wu J.Q., Meng Q.Q., Yao C.H., Zhao R. Improved lignocellulose degradation efficiency based on Fenton pretreatment during rice straw composting. Bioresour. Technol. 2019;294 doi: 10.1016/j.biortech.2019.122132. [DOI] [PubMed] [Google Scholar]
- 90.Wu D., Xia T.Y., Zhang Y.X., Wei Z.M., Qu F.T., Zheng G.R., Song C.H., Zhao Y., Kang K.J., Yang H.Y. Identifying driving factors of humic acid formation during rice straw composting based on Fenton pretreatment with bacterial inoculation. Bioresour. Technol. 2021;337 doi: 10.1016/j.biortech.2021.125403. [DOI] [PubMed] [Google Scholar]
- 91.Gao S.Q., Liu Y.T., Wang L.H., Wang Z.H., Liu P.B., Gao J., Jiang Y.J. Incorporation of metals and enzymes with porous imine molecule cages for highly efficient semiheterogeneous chemoenzymatic catalysis. ACS Catal. 2021;11:5544–5553. doi: 10.1021/acscatal.1c00587. [DOI] [Google Scholar]
- 92.Gadhe A., Sonawane S.S., Varma M.N. Influence of nickel and hematite nanoparticle powder on the production of biohydrogen from complex distillery wastewater in batch fermentation. Int. J. Hydrogen Energy. 2015;40:10734–10743. doi: 10.1016/j.ijhydene.2015.05.198. [DOI] [Google Scholar]
- 93.Huang D.L., Li T., Xe P., Zeng G.M., Chen M., Lai C., Cheng M., Guo X.Y., Chen S., Li Z.H. Deciphering the Fenton-reaction-aid lignocellulose degradation pattern by Phanerochaete chrysosporium with ferroferric oxide nanomaterials: enzyme secretion, straw humification and structural alteration. Bioresour. Technol. 2019;276:335–342. doi: 10.1016/j.biortech.2019.01.013. [DOI] [PubMed] [Google Scholar]
- 94.Dutta N., Saha M.K. Nanoparticle-induced enzyme pretreatment method for increased glucose production from lignocellulosic biomass under cold conditions. J. Sci. Food Agric. 2019;99:767–780. doi: 10.1002/jsfa.9245. [DOI] [PubMed] [Google Scholar]
- 95.Zhang Y., Dong Z.Q., Luo Y., Yang E., Xu H.N., Chagan I., Yan J.P. The manganese peroxidase gene family of Trametes trogii: gene identification and expression patterns using various metal ions under different culture conditions. Microorganisms. 2021;9:2595. doi: 10.3390/microorganisms9122595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Salvachúa D., Martínez A.T., Tien M., Lopez-Lucendo M.F., Garcia F., de los Rios V., Martinez M.J., Prieto A. Differential proteomic analysis of the secretome of Irpex lacteus and other white-rot fungi during wheat straw pretreatment. Biotechnol. Biofuels. 2013;6:115. doi: 10.1186/1754-6834-6-115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Li F., Zhang J.L., Ma F.Y., Chen Q., Xiao Q.Y., Zhang X.Y., Xie S.X., Yu H.B. Lytic polysaccharide monooxygenases promote oxidative cleavage of lignin and lignin–carbohydrate complexes during fungal degradation of lignocellulose. Environ. Microbiol. 2021;23:4547–4560. doi: 10.1111/1462-2920.15648. [DOI] [PubMed] [Google Scholar]
- 98.Bhardwaj N., Kumar B., Agarwal K., Chaturvedi V., Verma P. Purification and characterization of a thermo-acid/alkali stable xylanases from Aspergillus oryzae LC1 and its application in Xylo-oligosaccharides production from lignocellulosic agricultural wastes. Int. J. Biol. Macromol. 2018;122:1191–1202. doi: 10.1016/j.ijbiomac.2018.09.070. [DOI] [PubMed] [Google Scholar]
- 99.Liu Y.H., He P.J., Shao L.M., Zhang H., Lu F. Significant enhancement by biochar of caproate production via chain elongation. Water Res. 2017;119:150–159. doi: 10.1016/j.watres.2017.04.050. [DOI] [PubMed] [Google Scholar]
- 100.Zhou G.X., Xu X.F., Qiu X.W., Zhang J.B. Biochar influences the succession of microbial communities and the metabolic functions during rice straw composting with pig manure. Bioresour. Technol. 2019;272:10–18. doi: 10.1016/j.biortech.2018.09.135. [DOI] [PubMed] [Google Scholar]
- 101.Biswas B., Kumar A., Kaur R., Krishna B.B., Bhaskar T. Catalytic hydrothermal liquefaction of alkali lignin over activated bio-char supported bimetallic catalyst. Bioresour. Technol. 2021;337 doi: 10.1016/j.biortech.2021.125439. [DOI] [PubMed] [Google Scholar]
- 102.Rosales-Calderon O., Arantes V. A review on commercial-scale high-value products that can be produced alongside cellulosic ethanol. Biotechnol. Biofuels. 2019;12:240. doi: 10.1186/s13068-019-1529-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Muthuvelu K.S., Rajarathinam R., Manickam N.K., Uthandi S. Development of co-immobilized tri-enzyme biocatalytic system for one-pot pretreatment of four different perennial lignocellulosic biomass and evaluation of their bioethanol production potential. Bioresour. Technol. 2018;269:227–236. doi: 10.1016/j.biortech.2018.08.091. [DOI] [PubMed] [Google Scholar]
- 104.Rabee A.E., Sayed Alahl A.A., Mebarek L., Ishaq S.L. Fibrolytic rumen bacteria of camel and sheep and their applications in the bioconversion of barley straw to soluble sugars for biofuel production. PLoS One. 2022;17 doi: 10.1371/journal.pone.0262304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Li Y.Q., Lei L., Zheng L., Xiao X.M., Tang H., Luo C.B. Genome sequencing of gut symbiotic Bacillus velezensis LC1 for bioethanol production from bamboo shoots. Biotechnol. Biofuels. 2020;13:34. doi: 10.1186/s13068-020-1671-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Mohapatra S., Mishra C., Behera S.S., Thatoi H. Application of pretreatment, fermentation and molecular techniques for enhancing bioethanol production from grass biomass – a review. Renew. Sustain. Energy Rev. 2017;78:1007–1032. doi: 10.1016/j.rser.2017.05.026. [DOI] [Google Scholar]
- 107.Aikawa S., Joseph A., Yamada R., Izumi Y., Yamagishi T., Matsuda F., Kawai H., Chang J.S., Hasunuma T., Kondo A. Direct conversion of Spirulina to ethanol without pretreatment or enzymatic hydrolysis processes. Energy Environ. Sci. 2013;6:1844–1849. doi: 10.1039/c3ee40305j. [DOI] [Google Scholar]
- 108.Dar M.A., Dhole N.P., Xie R.R., Pawar K.D., Ullah K., Rahi P., Pandit R.S., Sun J.Z. Valorization potential of a novel bacterial strain, Bacillus altitudinis RSP75, towards lignocellulose bioconversion: an assessment of symbiotic bacteria from the stored grain pest, Tribolium castaneum. Microorganisms. 2021;9:1952. doi: 10.3390/microorganisms9091952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Duque A., Alvarez C., Domenech P., Manzanares P., Moreno A.D. Advanced bioethanol production: from novel raw materials to integrated biorefineries. Processes. 2021;9:206. doi: 10.3390/pr9020206. [DOI] [Google Scholar]
- 110.Pereira S.R., Nogue V.S.I., Frazao C.J.R., Serafim L.S., Gorwa-Grauslund M.F., Xavier A.M.R.B. Adaptation of Scheffersomyces stipitis to hardwood spent sulfite liquor by evolutionary engineering. Biotechnol. Biofuels. 2015;8:50. doi: 10.1186/s13068-015-0234-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Andersen R.L., Jensen K.M., Mikkelsen M.J. Continuous ethanol fermentation of pretreated lignocellulosic biomasses, waste biomasses, molasses and syrup using the anaerobic, thermophilic bacterium thermoanaerobacter italicus Pentocrobe 411. PLoS One. 2015;10 doi: 10.1371/journal.pone.0136060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Yadav M., Paritosh K., Vivekanand V. Lignocellulose to bio-hydrogen: an overview on recent developments. Int. J. Hydrogen Energy. 2020;45:18195–18210. doi: 10.1016/j.ijhydene.2019.10.027. [DOI] [Google Scholar]
- 113.Soowski G., Konkol I., Cenian A. Production of hydrogen and methane from lignocellulose waste by fermentation. A review of chemical pretreatment for enhancing the efficiency of the digestion process. J. Clean. Prod. 2020;267 doi: 10.1016/j.jclepro.2020.121721. [DOI] [Google Scholar]
- 114.Srivastava N., Srivastava M., Kushwaha D., Gupta V.K., Manikanta A., Ramteke P.W., Mishra P.K. Efficient dark fermentative hydrogen production from enzyme hydrolyzed rice straw by Clostridium pasteurianum (MTCC116) Bioresour. Technol. 2017;238:552–558. doi: 10.1016/j.biortech.2017.04.077. [DOI] [PubMed] [Google Scholar]
- 115.Lu H.Y., Lee P.K.H. Effects of cellulose concentrations on the syntrophic interactions between Clostridium cellulovorans 743B and Rhodopseudomonas palustris CGA009 in coculture fermentation for biohydrogen production. Int. J. Hydrogen Energy. 2015;40:11800–11808. doi: 10.1016/j.ijhydene.2015.05.135. [DOI] [Google Scholar]
- 116.Laurinavichene T., Tsygankov A. Hydrogen photoproduction by co-culture Clostridium butyricum and Rhodobacter sphaeroides. Int. J. Hydrogen Energy. 2015;40:14116–14123. doi: 10.1016/j.ijhydene.2015.08.086. [DOI] [Google Scholar]
- 117.Yin Y.N., Wang J.L. Characterization and hydrogen production performance of a novel strain Enterococcus faecium INET2 isolated from gamma irradiated sludge. Int. J. Hydrogen Energy. 2016;41:22793–22801. doi: 10.1016/j.ijhydene.2016.10.059. [DOI] [Google Scholar]
- 118.Jiang Y.J., Lu J.S., Lu Y., Wu R.F., Dong W.L., Zhou J., Jiang M., Xin F.X. Efficient hydrogen production from lignocellulosic feedstocks by a newly isolated thermophlic Thermoanaerobacterium sp. strain F6. Int. J. Hydrogen Energy. 2019;44:14380–14386. doi: 10.1016/j.ijhydene.2019.01.226. [DOI] [Google Scholar]
- 119.Zhao L., Cao G.L., Wang A.J., Ren H.Y., Dong D., Liu Z.N., Guan X.Y., Xu C.J., Ren N.Q. Fungal pretreatment of cornstalk with Phanerochaete chrysosporium for enhancing enzymatic saccharification and hydrogen production. Bioresour. Technol. 2012;114:365–369. doi: 10.1016/j.biortech.2012.03.076. [DOI] [PubMed] [Google Scholar]
- 120.Hu Y.S., Hao X.D., Wang J.M., Cao Y.L. Enhancing anaerobic digestion of lignocellulosic materials in excess sludge by bioaugmentation and pre-treatment. Waste Manage. (Tucson, Ariz.) 2016;49:55–63. doi: 10.1016/j.wasman.2015.12.006. [DOI] [PubMed] [Google Scholar]
- 121.Vasco-Correa J., Khanal S., Manandhar A., Shah A. Anaerobic digestion for bioenergy production: global status, environmental and techno-economic implications, and government policies. Bioresour. Technol. 2018;247:1015–1026. doi: 10.1016/j.biortech.2017.09.004. [DOI] [PubMed] [Google Scholar]
- 122.Peng X.W., Börner R.A., Nges I.A., Liu J. Impact of bioaugmentation on biochemical methane potential for wheat straw with addition of Clostridium cellulolyticum. Bioresour. Technol. 2014;152:567–571. doi: 10.1016/j.biortech.2013.11.067. [DOI] [PubMed] [Google Scholar]
- 123.Yadav M., Singh A., Balan V., Pareek N., Vivekanand V. Biological treatment of lignocellulosic biomass by Chaetomium globosporum: process derivation and improved biogas production. Int. J. Biol. Macromol. 2019;128:176–183. doi: 10.1016/j.ijbiomac.2019.01.118. [DOI] [PubMed] [Google Scholar]
- 124.Suksong W., Wongfaed N., Sangsri B., Kongjan P., Prasertsan P., Podmirseg S.M., Insam H., O-Thong S. Enhanced solid-state biomethanisation of oil palm empty fruit bunches following fungal pretreatment. Ind. Crop. Prod. 2020;145 doi: 10.1016/j.indcrop.2020.112099. [DOI] [Google Scholar]
- 125.Sun E.H., Liao G.F., Zhan Q., Q P., Wu G.F., Huang H.Y. Biodegradable copolymer-based composites made from straw fiber for biocomposite flowerpots application. Compos. B Eng. 2019;165:193–198. doi: 10.1016/j.compositesb.2018.11.121. [DOI] [Google Scholar]
- 126.Dai L., Cheng T., Duan C., Zhao W., Zhang W.P., Zou X.J., Aspler J., Ni Y.H. 3D printing using plant-derived cellulose and its derivatives: a review. Carbohydr. Polym. 2019;203:71–86. doi: 10.1016/j.carbpol.2018.09.027. [DOI] [PubMed] [Google Scholar]
- 127.De France K.J., Hoare T., Cranston E.D. Review of hydrogels and aerogels containing nanocellulose. Chem. Mater. 2017;29:4609–4631. doi: 10.1021/acs.chemmater.7b00531. [DOI] [Google Scholar]
- 128.Dasgupta D., Bandhu S., Adhikari D.K., Ghosh D. Challenges and prospects of xylitol production with whole cell bio-catalysis: a review. Microbiol. Res. 2017;197:9–21. doi: 10.1016/j.micres.2016.12.012. [DOI] [PubMed] [Google Scholar]
- 129.Jeon W.Y., Yoon B.H., Ko B.S., Shim W.Y., Kim J.H. Xylitol production is increased by expression of codon-optimized Neurospora crassa xylose reductase gene in Candida tropicalis. Bioproc. Biosyst. Eng. 2012;35:191–198. doi: 10.1007/s00449-011-0618-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Shikinaka K., Nakamura M., Otsuka Y. Strong UV absorption by nanoparticulated lignin in polymer films with reinforcement of mechanical properties. Polymer. 2020;190 doi: 10.1016/j.polymer.2020.122254. [DOI] [Google Scholar]
- 131.Ye J.L., Ma S.Q., Wang B.B., Chen Q.M., Huang K., Xu X.W., Li Q., Wang S., Lu N., Zhu J. High-performance bio-based epoxies from ferulic acid and furfuryl alcohol: synthesis and properties. Green Chem. 2021;23:1772–1781. doi: 10.1039/d0gc03946b. [DOI] [Google Scholar]
- 132.Sainsbury P.D., Hardiman E.M., Ahmad M., Otani H., Seghezzi N., Eltis L.D., Bugg T.D.H. Breaking down lignin to high-value chemicals: the conversion of lignocellulose to vanillin in a gene deletion mutant of Rhodococcus jostii RHA1. ACS Chem. Biol. 2013;8:2151–2156. doi: 10.1021/cb400505a. [DOI] [PubMed] [Google Scholar]
- 133.Graf N., Altenbuchner J. Genetic engineering of Pseudomonas putida KT2440 for rapid and high-yield production of vanillin from ferulic acid. Appl. Microbiol. Biotechnol. 2014;98:137–149. doi: 10.1007/s00253-013-5303-1. [DOI] [PubMed] [Google Scholar]
- 134.Williamson J.J., Bahrin N., Hardiman E.M., Bugg T.D.H. Production of substituted styrene bioproducts from lignin and lignocellulose using engineered Pseudomonas putida KT2440. Biotechnol. J. 2020;15 doi: 10.1002/biot.201900571. [DOI] [PubMed] [Google Scholar]
- 135.Numata K., Morisaki K. Screening of marine bacteria to synthesize polyhydroxyalkanoate from lignin: contribution of lignin derivatives to biosynthesis by Oceanimonas doudoroffii. ACS Sustain. Chem. Eng. 2015;3:569–573. doi: 10.1021/acssuschemeng.5b00031. [DOI] [Google Scholar]
- 136.Wang X.P., Lin L., Dong J.D., Ling J., Wang W.P., Wang H.L., Zhang Z.C., Yu X.W. Simultaneous improvements of Pseudomonas cell growth and polyhydroxyalkanoate production from a lignin derivative for lignin-consolidated bioprocessing. Appl. Environ. Microbiol. 2018;84 doi: 10.1128/AEM.01469-18. e01469-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Xie S.X., Sun S., Lin F.R., Li M.Z., Pu Y.Q., Cheng Y.B., Xu B., Liu Z.H., Sousa L.D., Dale B.E., Ragauskas A.J., Dai S.Y., Yuan J.S. Mechanism-guided design of highly efficient protein secretion and lipid conversion for biomanufacturing and biorefining. Adv. Sci. 2019;6 doi: 10.1002/advs.201801980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Johnson C.W., Beckham G.T. Aromatic catabolic pathway selection for optimal production of pyruvate and lactate from lignin. Metab. Eng. 2015;28:240–247. doi: 10.1016/j.ymben.2015.01.005. [DOI] [PubMed] [Google Scholar]
- 139.Kohlstedt M., Starck S., Barton N., Stolzenberger J., Selzer M., Mehlmann K., Schneider R., Pleissner D., Rinkel J., Dickschat J.S., Venus J., van Duuren J.B.J.H., Wittmann C. From lignin to nylon: cascaded chemical and biochemical conversion using metabolically engineered Pseudomonas putida. Metab. Eng. 2018;47:279–293. doi: 10.1016/j.ymben.2018.03.003. [DOI] [PubMed] [Google Scholar]
- 140.Jung D.H., Kim E.J., Jung E., Kazlauskas R.J., Choi K.Y., Kim B.G. Production of p- hydroxybenzoic acid from p- coumaric acid by Burkholderia glumae BGR1. Biotechnol. Bioeng. 2016;113:1493–1503. doi: 10.1002/bit.25908. [DOI] [PubMed] [Google Scholar]
- 141.Sonoki T., Takahashi K., Sugita H., Hatamura M., Azuma Y., Sato T., Suzuki S., Kamimura N., Masai E. Glucose-free cis, cis -muconic acid production via new metabolic designs corresponding to the heterogeneity of lignin. ACS Sustain. Chem. Eng. 2017;6:1256–1264. doi: 10.1021/acssuschemeng.7b03597. [DOI] [Google Scholar]
- 142.Sun E.H., Zhang Y., Yong C., Qu P., Huang H.Y., Xu Y.D. Biological fermentation pretreatment accelerated the depolymerization of straw fiber and its mechanical properties as raw material for mulch film. J. Clean. Prod. 2020;284 doi: 10.1016/j.jclepro.2020.124688. [DOI] [Google Scholar]
- 143.Gupta C., Jain P., Kumar D., Dixit A.K., Jain R.K. Production of cellulase enzyme from isolated fungus and its application as efficient refining aid for production of security paper. Int. J. Appl. Microbiol. Biotechnol. Res. 2015;3:11–19. [Google Scholar]
- 144.Shintani N., Shoda M. Decolorization of oxygen-delignified bleaching effluent and biobleaching of oxygen-delignified kraft pulp by non-white-rot fungus Geotrichum candidum Dec 1. J. Environ. Sci. 2013;25:S164–S168. doi: 10.1016/S1001-0742(14)60649-5. [DOI] [PubMed] [Google Scholar]
- 145.Hamedi J., Fakhri A.V., Mahdavi S. Biobleaching of mechanical paper pulp using Streptomyces rutgersensis UTMC 2445, isolated from a lignocellulose-rich soil. J. Appl. Microbiol. 2019;128:161–170. doi: 10.1111/jam.14489. [DOI] [PubMed] [Google Scholar]
- 146.Zhou Z.Y., Liu D.H., Zhao X.B. Conversion of lignocellulose to biofuels and chemicals via sugar platform: an updated review on chemistry and mechanisms of acid hydrolysis of lignocellulose. Renew. Sustain. Energy Rev. 2021;146 doi: 10.1016/j.rser.2021.111169. [DOI] [Google Scholar]
- 147.Rorke D.C.S., Lekha P., Kana G.E.B., Sithole B.B. Surfactant-assisted green liquor dregs pretreatment to enhance the digestibility of paper mill sludge. Int. J. Hydrogen Energy. 2021;46:21359–21371. doi: 10.1016/j.ijhydene.2021.04.018. [DOI] [Google Scholar]
- 148.Lynd L.R., van Zyl W.H., McBride J.E., Laser M. Consolidated bioprocessing of cellulosic biomass: an update. Curr. Opin. Biotechnol. 2005;16:577–593. doi: 10.1016/j.copbio.2005.08.009. [DOI] [PubMed] [Google Scholar]
- 149.Li J.G., Lin L.C., Sun T., Xu J., Ji J.X., Liu Q., Tian C.G. Direct production of commodity chemicals from lignocellulose using Myceliophthora thermophile. Metab. Eng. 2020;61:416–426. doi: 10.1016/j.ymben.2019.05.007. [DOI] [PubMed] [Google Scholar]
- 150.Eibinger M., Ganner T., Bubner P., Rosker S., Kracher D., Haltrich D., Ludwig R., Plank H., Nidetzky B. Cellulose surface degradation by a lytic polysaccharide monooxygenase and its effect on cellulase hydrolytic efficiency. J. Biol. Chem. 2014;289:35929–35938. doi: 10.1074/jbc.M114.602227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Coconi-Linares N., Magana-Ortiz D., Guzman-Ortiz D.A., Fernandez F., Loske A.M., Gomez-Lim M.A. High-yield production of manganese peroxidase, lignin peroxidase, and versatile peroxidase in Phanerochaete chrysosporium. Appl. Microbiol. Biotechnol. 2014;98:9283–9294. doi: 10.1007/s00253-014-6105-9. [DOI] [PubMed] [Google Scholar]
- 152.Chiang C.J., Lee H.M., Guo H.J., Wang Z.W., Lin L.J., Chao Y.P. Systematic approach to engineer Escherichia coli pathways for Co-utilization of a glucose–xylose mixture. J. Agric. Food Chem. 2013;61:7583–7590. doi: 10.1021/jf401230r. [DOI] [PubMed] [Google Scholar]
- 153.Dhanya B.S., Mishra A., Chandel A.K., Verma M.L. Development of sustainable approaches for converting the organic waste to bioenergy. Sci. Total Environ. 2020;723 doi: 10.1016/j.scitotenv.2020.138109. [DOI] [PubMed] [Google Scholar]
- 154.Verma M.L., Puri M., Barrow C.J. Recent trends in nanomaterials immobilised enzymes for biofuel production. Crit. Rev. Biotechnol. 2014;36:108–119. doi: 10.3109/07388551.2014.928811. [DOI] [PubMed] [Google Scholar]
- 155.Abraham R.E., Verma M.L., Barrow C.J., Puri M. Suitability of magnetic nanoparticle immobilised cellulases in enhancing enzymatic saccharification of pretreated hemp biomass. Biotechnol. Biofuels. 2014;7 doi: 10.1186/1754-6834-7-90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Zhang W.J., Qiu J.H., Feng H.X., Zang L.M., Sakai E. Increase in stability of cellulase immobilized on functionalized magnetic nanospheres. J. Magn. Magn Mater. 2015;375:117–123. doi: 10.1016/j.jmmm.2014.09.067. [DOI] [Google Scholar]
- 157.Huang P.J., Chang K.L., Hsieh J.F., Chen S.T. Catalysis of rice straw hydrolysis by the combination of immobilized cellulase from Aspergillus niger on β-cyclodextrin-Fe3O4 nanoparticles and ionic liquid. BioMed Res. Int. 2015;2015 doi: 10.1155/2015/409103. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data that has been used is confidential.