Abstract
Tubulointerstitial fibrosis (TIF), a common end result of almost all progressive chronic kidney diseases (CKD), is also the best predictor of kidney survival. Almost all cells in the kidney are involved in the progression of TIF. Myofibroblasts, the primary producers of extracellular matrix, have previously received a great deal of attention; however, a large body of emerging evidence reveals that proximal tubule (PT) plays a central role in TIF progression. In response to injury, renal tubular epithelial cells (TECs) transform into inflammatory and fibroblastic cells, producing various bioactive molecules that drive interstitial inflammation and fibrosis. Here we reviewed the increasing evidence for the key role of the PT in promoting TIF in tubulointerstitial and glomerular injury and discussed the therapeutic targets and carrier systems involving the PT that holds particular promise for treating patients with fibrotic nephropathy.
Keywords: Proximal tubule, Tubulointerstitial fibrosis, Extracellular matrix, Chronic kidney disease
1. Introduction
It is estimated that 9.1% of the global population suffers from chronic kidney disease (CKD) to some degree, and a significant proportion of patients progress to end-stage renal failure, necessitating lifelong dialysis or kidney transplantation [1]. Studies have shown that CKD is an important trigger for cardiovascular diseases [1]. As global CKD mortality has risen sharply, the economic burden is increasing by the day [1]. Diabetes mellitus, hypertension, glomerular inflammation, genetic disorders and drugs are common causative factors of CKD [2]. Despite its multiple etiologies, CKD shares a common progressive process in which normal renal parenchyma is replaced by matrix proteins such as collagen I, III, IV and fibronectin [3,4]. Renal fibrosis is defined as the accumulation and deposition of scar inside the renal parenchyma, which is manifested by TIF, glomerulosclerosis and arteriosclerosis [5]. Since tubules make up the majority of the renal parenchyma, renal fibrosis is generally referred to as TIF [6,7]. Despite the fact that not all renal injuries target the renal tubules, the severity of TIF in renal biopsies is the best prognostic indicator of CKD [8]. Therefore, understanding the pathogenesis of TIF progression is thus critical for the development of new therapies for CKD.
The development and progression of TIF involves alterations in many cell types, such as the activation and expansion of interstitial myofibroblasts, infiltration of inflammatory cells (e.g., macrophages), renal tubular atrophy and perivascular fibrosis and other cellular processes [9]. Because of the complexities of this process, determining the relative position of each cellular compartment in the progression of TIF is difficult. Myofibroblasts and their precursors have been shown in studies to be the primary producers of extracellular matrix (ECM), and thus have been the focus of attention in TIF progression [10]. However, a growing number of studies have recently shed light on the role of the proximal tubule (PT) in TIF progression. As a special tubular segment adjacent to the glomerulus, the PT is not only a target of injury but also an important mediator of TIF progression. Drugs that target proximal tubular cells (PTCs) may provide new treatment options for TIF by reducing drug toxicity and/or by increasing the efficacy of antifibrotic drugs. The aim of this review is to provide a reference for revealing the importance of PT in TIF as well as an overview of therapies targeting PT.
2. The focus in TIF is the PT rather than the distal tubule (DT)
The renal unit consists of filtration units (e.g., glomeruli) and a tubular system (consisting of PT, Henle's collaterals, DT, and collecting ducts) along which substances can be exchanged between blood and urine. The tubular system is in charge of the reabsorption of endogenous compounds, such as water, salt and glucose, as well as drug molecules that cross the tubular cells by passive or active transport.
PT, rather than DT, contributes most to TIF for three reasons. Firstly, the amount and reabsorption capacity of PT are greater than those of DT. PT represent the largest part of the kidney and play the most important role in the recovery of substances from urine, accounting for the reabsorption of approximately two-thirds of the filtrate volume [11]. In contrast, DT reabsorbs only 5–10% of the filtered sodium and chloride under normal conditions [12].
Second, PT epithelial cells are less resistant to injury than DT epithelial cells. A multitude of factors, including ischemia/reperfusion (I/R) and associated oxidative stress, nephrotoxins, inflammation, and immune system abnormalities, predispose kidneys to acute kidney injury (AKI), which can proceed to CKD and fibrosis [13]. Although AKI can cause immediate direct damage to the DT and PT epithelium, PT is more vulnerable to injury due to their high rate of oxygen consumption and relative lack of endogenous antioxidant defense and anti-apoptotic proteins [13,14]. In addition, PT is particularly susceptible to mitochondrial toxicity and reactive oxygen species (ROS) are more frequently produced in PT than in DT [15]. In contrast, the epithelium of DT is not as sensitive as the epithelium of PT, DT epithelium shows a stronger resistance to hypoxic stress compared to PT [13]. Moreover, DT epithelium is less sensitive to cell death. A typical example is that after ischemic injury, DT epithelial cells can protect sensitive PT from injury by adaptively increasing the expression of survival factors and increasing the synthesis and secretion of reparative growth factors [13].
Third, the PT is also primarily responsible for compound secretion into the urine. It is inevitable that it will come into contact with harmful substances and be damaged. Therefore, the impairment of PT's function is more pivotal in renal injury and fibrosis than DT.
PT damage has been demonstrated in a number of renal fibrotic diseases, including unilateral ureteral obstruction (UUO), polycystic kidney disease, nephrotic cystinosis, congenital nephrotic syndrome, and diabetic nephropathy (DN) (the latter two are considered “glomerular” diseases) [14]. Among them, the attention of fibrogenesis started to focus on PT due to the first identification of atubular glomeruli in the UUO model [14]. Tubular atrophy, one of the pathological manifestations of renal fibrosis, is the main determinant of renal parenchymal loss. It has been experimentally proven that the large loss of renal parenchyma induced by UUO is due to a 65% reduction in the mass of PT [16]. In addition to causing tubular atrophy, glomerulotubular disruption, atubular glomeruli formation in glomerular, tubular, and toxic nephropathies are PT's responses to injury [14]. DN is an incidental disorder of diabetes mellitus and is one of the major causes of CKD, although it is considered to be a disease with glomerular lesion. Studies have shown that the earliest site of response in the renal unit is the PT rather than the glomerulus [14]. In a high glucose environment, PT cells become hypertrophic due to increased intracellular glucose; feedback from the glomerulus triggers excessive PT reabsorption and ultrafiltration [17]. Even if only the PT is injured, it can lead to a cascade of peritubular inflammation, TIF, and glomerulosclerotic events [14]. Taken together, the most prominent features of AKI and CKD are characterized by direct or secondary injury to PT cells [14], which further highlights the key role of PT in fibrotic events.
3. Major cellular events of PT in TIF
According to the sequence of cellular events following renal injury, the pathogenesis of TIF can be divided into four overlapping phases: initiation, activation, execution and progression [18]. However, TIF is actually a dynamic process in which many events occur simultaneously with the participation of PT.
3.1. Role of PT injury on inflammatory cells infiltration
Following tubular injury, inflammation serves as the initial event that triggers the onset of fibrosis. This process can be summarized as the activation and infiltration of inflammatory cells, which results in the production of molecules such as ROS that damage the tissue and induce the production of fibrotic cytokines and growth factors (Fig. 1) [19]. The sequence of those events is the driving force that prompts fibroblasts and renal TECs to undergo phenotypic activation or transformation and to produce a large number of ECM components [18]. Inflammation that does not resolve after a chronic injury can create a vicious cycle of inflammation, tissue damage, and fibrosis.
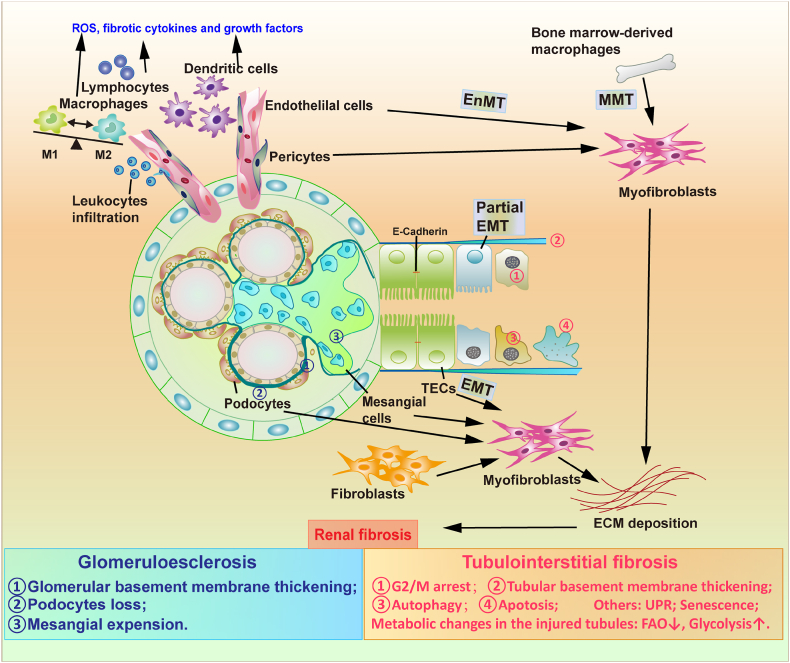
Fig. 1.
Major events in renal fibrosis. Inflammation is activated as the initiating event when the kidney is subjected to injury. Inflammatory cells, including T-lymphocytes, macrophages, and dendritic cells, infiltrate the peritubular region resulting in the production of molecules such as ROS that damage the tissue and induce the production of fibrotic cytokines and growth factors. The inflammatory microenvironment that develops after renal injury activates fibroblasts, TECs, endothelial cells, podocytes, pericytes, mesangial cells and macrophages converting into myofibroblasts to produce matrix. Depending on the severity and duration of injury, renal tubular cells exhibit a wide range of responses, such as proliferation, activated unfolded protein response (UPR), autophagy, senescence, G2/M arrest, apoptosis and EMT. There is a lot of controversy on the occurrence and subsequent effect of EMT. There are currently two views of EMT: complete EMT (absence of epithelial markers, expression of mesenchymal markers, and crossing tubular basement membrane into myofibroblasts) (abbreviated as EMT) and partial EMT (TECs acquire mesenchymal characteristics and are capable of producing a variety of pro-fibrotic factors and cytokines, but remain attached to the basement membrane). Metabolic changes that damage the renal tubules such as decreased FAO and upregulation of glycolysis can promote the process of TIF. The damage also impairs the glomerular structure, as glomerulosclerosis develops as a result of the loss of podocytes, thickening ofGBM, and expansion of the mesangial cells. Glomerulosclerosis and TIF together make up renal fibrosis (pathological changes in the vasculature are not shown in this figure).
PT injury can promote the occurrence of inflammation, but accumulating evidence also reveals the inhibitory effect of PT on inflammation, suggesting that PT may play a dual role in the occurrence and development of inflammation. An example is that although uric acid has been shown to directly activate PT cells, accelerate macrophage recruitment and promote differentiation of the M1 phenotype, PT can also antagonize TIFs, reducing inflammation by reducing the expression of pro-inflammatory factors and increasing the activation of anti-inflammatory factors [20]. In addition to macrophages, the recruitment of other inflammatory cells is also regulated by PT. For example, proximal tubular epithelial cells (PTECs) suppress pro-inflammatory immune responses in the tubular compartment by regulating the function of autologous lymphocytes, macrophages, and dendritic cells (DCs) [21,22]. In addition to upregulating a range of inhibitory molecules in response to inflammatory stimuli, human PTEC is able to and regulate antibody production in autologous B cells [23]. Colony-stimulating factor-1 (CSF-1), produced primarily by PT, can stimulate resident renal macrophages and dendritic cells, participating in recovery from AKI in response to either I/R or a model of selective PT injury induced by diphtheria-toxin-induced apoptosis in transgenic mice expressing the human diphtheria toxin receptor on PT cells [24]. Preservation of PT peroxisome proliferator-activated receptor (PPARα) expression significantly reduces the expression of pro-inflammatory cytokines such as interleukin-1 beta (IL-1β), interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-α), and significantly increases the expression of anti-inflammatory cytokines IL-10 and arginase-1, thereby reducing TIF and inflammation [25]. In conclusion, although studies have indicated that PT has both pro-inflammatory and anti-inflammatory effects, it is needed to determine which effect predominates in the TIF process, and it is also necessary to investigate whether the pro-inflammatory and anti-inflammatory mechanisms of PT interact or not.
3.2. The contribution of renal TECs to the development of TIF
The inflammatory microenvironment that develops after renal injury activates myofibroblasts to produce matrix, which is a central event in TIF. Clarifying the origin of myofibroblasts is particularly crucial because they are key producers in ECM production. Myofibroblast formation, proliferation, and ECM production are regulated by various growth factors and hormones [26], such as TGF-β [27], connective tissue growth factor (CTGF) [28,29], fibroblast growth factor (FGF) [30], platelet-derived growth factor (PDGF) [31], interleukin-1 (IL-1) [32], TNF-α [33], Angiotensin II (Ang II) [34], and aldosterone [35]. Although it has been shown that fibroblasts, TECs, endothelial cells, podocytes, pericytes, mesangial cells and macrophages are capable of converting into myofibroblasts and producing ECM (Fig. 1) [[36], [37], [38]], it has been commonly assumed that fibroblasts are the primary source of myofibroblasts [18,38]. Since renal tubules make up the largest proportion of the renal parenchyma, the process of epithelial-mesenchymal transition (EMT) cannot be ignored.
EMT, a cellular phenotypic transformation process that occurs during embryonic development, tumor metastasis, and organ fibrosis, has received a surge of attention in the development of TIF in recent years [39,40]. Early research suggested that a large proportion of myofibroblasts are transformed from PTECs by EMT [41]. However, recent data are disappointing. Recent research indicating that epithelial cells contribute little to the myofibroblast population after injury [[42], [43], [44], [45], [46]]. One possible reason for this biphasic outcome is that it appears to be more difficult to demonstrate EMT in vivo compared to the relative ease of demonstrating EMT in vitro. Therefore, the conclusions drawn by in vitro experiments are not necessarily exactly equivalent to the actual situation in vivo.
The question of whether EMT is real has long been debated, and both sides of the argument have data to back it up, making the answer even more elusive. As previously stated, it is difficult to track complete EMT process (without epithelial markers, mesenchymal marker expression, and crossover of TBM into myofibroblasts). Recent studies from different teams have shown that after injury, renal TECs exhibit mesenchymal characteristics, produce multiple profibrotic factors and cytokines, but remain attached to the basement membrane [42,[47], [48], [49]]. On this basis, a new concept of “partial epithelial-mesenchymal conversion (pEMT)" was proposed [47,49,50]. This theory may provide new answers to explain the contribution of renal TEC to the development of renal fibrosis. Moreover, injury-mediated Snail1 reactivation induces pEMT in TEC that can signal to the mesenchyme to promote myofibroblast differentiation and fibrosis and maintain inflammation without direct conversion to myofibroblasts [50]. These findings reconcile the conflicting findings regarding the role of EMT in renal fibrosis and provide avenues for the design of new anti-fibrotic therapies. Although the importance of fibroblast activation for TIF is unquestionable, the above findings suggest that EMT may be one of the major determinants of TIF development and progression.
3.3. Response of PT to persistent injury
The PT has long been considered a target of injury, but new evidence suggests that it also plays an important role in the development and progression of TIF. The renal tubules constitute to the majority of the renal parenchyma and are major targets of various metabolic, immune, ischemic, and toxic injuries. Depending on the severity and duration of injury, renal tubular cells exhibit a wide range of responses, such as proliferation, de-differentiation, activated unfolded protein response (UPR), autophagy, senescence, growth arrest, apoptosis and EMT. The following is an overview of the role of PT in these events.
3.3.1. PT injury is sufficient to cause TIF
Recent studies have shown that PT injury precedes other cellular compartment involvement, and PT injury alone is sufficient to cause TIF. Two independent research teams established PT-specific mouse injury models by crossing mice containing Cre-inducible diphtheria toxin receptor with Cre mice driven by the Six2 promoter or Ndrg1-Cre (ERT2) [51,52]. The results showed that different doses and frequencies of injection of diphtheria toxin had different degrees of PT damage. The subsequent events also changed. Injection of high-dose diphtheria toxin induces sublethal epithelial injury only in PT [51,52]. A single injection of low-dose diphtheria toxin induces only mild injury in PT. While this mild PT injury triggers reversible fibrosis, repeated mild injury is capable of causing persistent TIF, inflammation, capillary thinning, and the formation of underlying glomerulosclerosis [51,52]. Those data suggest that the severity and frequency of PT epithelial-specific injury have a significant impact on the progression of CKD.
3.3.2. PT de-differentiation is a double-edged sword in the development of TIF
Sustained renal injury is accompanied by a sustained process of de-differentiation, and the reduced response of de-differentiated cells to decreased ATP and energy supply allows the cells to survive under hypoxic conditions [53]. Highly differentiated cells expressing multiple channels and transporters. When PT is damaged, the PT epithelium de-differentiates and loses expression of typical epithelial proteins such as E-Cadherin and ZO-1. It has been reported that inhibition of EMT program preserved TEC integrity, restored de-differentiation, and alleviated fibrosis and immune infiltration in three renal injury models, namely, UUO, NTN, and folic acid (FA) nephropathy [49]. However, de-differentiation cannot be simply defined as a beneficial process, as it has also been shown in UUO and FA nephropathy models that TIF is aggravated when genetically targeting Snail1 expression, a key transcription factor in TECs de-differentiation [50]. In addition, members of other transcription factors associated with de-differentiation (SNAI, ZEB, TWIST) families can also attenuate TIF by regulating the inflammatory process [54]. However, the causal relationship between inflammation and de-differentiation is difficult to define.
The mechanism of sustained epithelial cell de-differentiation may, on the one hand, be through the production of paracrine factors, such as TGF-β, to stimulate the production of ECM by adjacent pericytes/myofibroblasts to induce TIF progression. On the other hand, it may promote TIF by exacerbating ECM deposition through cell-autonomous production of collagen proteins such as COL1A2 protein [55,56].
3.3.3. The complex relationship between autophagy and apoptosis for TIF
Autophagy regulates intracellular homeostasis through phagocytosis of cytoplasmic components. There is still no consensus on whether autophagy plays a beneficial [[57], [58], [59]] or detrimental [[60], [61], [62], [63]] role in AKI and CKD, as autophagy is often detected at some point in the disease and the database of detection throughout the process needs to be expanded. It is generally accepted that autophagy may play a protective role in the early stages of injury, but if the injury is too severe, it may promote the development of apoptosis or cell degeneration.
Signaling activated during autophagy may interfere with or compromise cell apoptosis pathways [[64], [65], [66]]. Autophagy can cooperate with apoptotic mechanisms by acting upstream of apoptosis, converging with apoptotic pathways or mediating steps downstream of apoptosis [67]. Some severely damaged TECs that should undergo apoptosis may also survive by the aid of autophagy to undergo maladaptive repair and phenotypic changes, becoming a source of pro-inflammatory and pro-fibrotic cytokines that exacerbate renal fibrosis [68]. The studies available so far suggest that autophagy appears preceding apoptosis [60]. Experiments in a 14-day UUO mice model showed that autophagy in PT was detectable in the first 7 days of the obstructed kidney and tended to increase progressively, with a decreasing trend in the second 7 days. In contrast, the onset of apoptosis begins on day 3 and increases in a time-dependent manner thereafter [69]. In primary PTCs cultured in vitro, the up-regulation of autophagy increased ECM deposition and apoptosis after TGF-β1 administration [70]. Increased ROS induced TECs damage and activated autophagy, which contributes to its proapoptotic effect [71,72]. However, the contribution of the mechanism of interaction between autophagy and apoptosis to TIF needs to be further studied.
3.3.4. The activated unfolded protein response (UPR) promotes TIF
UPR is a defense mechanism adopted by cells in response to endoplasmic reticulum (ER) stress [73]. In the process of renal injury, UPR is considered as a protection mechanism. ER stress occurs when kidney cells are stimulated by TGFβ1 or PDGF, followed by activation of the protective UPR pathway [74]. UPR restores protein homeostasis in vivo by inhibiting protein translation, introducing ER-associated molecular chaperones to encourage the refolding of unfolded proteins, and activating the ER-associated protein degradation (ERAD) system to remove unfolded proteins and maintain cell survival [75]. Sustained ER stress exacerbates activation of the major UPR branches (IRE1α/XBP1, PERK/ATF4, ATF6), inducing expression of numerous genes involved in inflammation, cell death, autophagy, and oxidative stress [76]. Activation of UPR in response to sustained ER stress during TIF may activate inflammatory pathways, and the inflammatory response further promotes the production of fibers, leading to the occurrence of TIF. For example, in cultured human renal cortical tubular cells, glucose deprivation activates UPR, which in turn activates NF-kB and promotes the transcription of proinflammatory cytokines and chemokines like IL-6, IL-8, TNF-α, RANTES, and MCP-1 [77]. Furthermore, the UPR may collaborate with autophagy to reduce protein misfolding and its consequences in kidney disease [78].
3.3.5. Senescence could be a potential target for kidney protection
Aging, ROS, and other stresses can cause senescence (e.g., DNA damage) [10]. It is believed that senescence of renal tubular cells may be one of the protective mechanisms of the kidney. Although acute senescence is beneficial, abnormal accumulation of senescent cells and persistent signaling can impair renal function and advance renal disease [79]. Although epithelial cell senescence is an irreversible growth arrest, senescent epithelial cells are metabolically active and contain a senescence-associated secretory phenotype (SASP) that can recruit inflammatory cells. Administration of the highly selective adrenergic receptor α2-ar agonist Dexmedetomidine (Dex) reduced the number of senescent renal tubular cells and decreased the expression of inflammatory markers [79]. This implies that senescence may be able to increase inflammatory paracrine secretion, and in this respect, senescence may play a role in exacerbating TIF.
3.3.6. Cell cycle phase arrest in PTECs
In damaged kidneys, some cells are arrested in the G1 or G2 phase in order to be capable of repairing DNA damage and to prevent mutagenesis of damaged cells. However, persistent arrest of renal TECs in the G2 phase leads to increased production of the pro-fibrotic cytokines TGF-β and CTGF in the JNK-dependent pathway [80]. Studies have shown that inhibition of cell cycle arrest can attenuate TIF. Data showed that administration of the histone deacetylase inhibitor methyl 4-(phenylthio) butyrate to mice reduced the number of G2/M-phase cells while attenuating the extent of tubular atrophy and TIF [81]. In addition, blockade of G2/M arrest can also be mediated by Atg5-mediated autophagy of PTECs [82]. The occurrence of EMT promotes the occurrence of cell cycle arrest, too. In the experimentally induced mouse model of renal fibrosis, conditional deletion of Twist1 or Snai1 in PTEC prevented the progression of EMT, restored the renal parenchyma repair and regeneration process related to cell proliferation and dedifferentiation, and weakened TIF [49]. Moreover, the percentage of renal TECs arrested in the G2/M phase after AKI is positively correlated with the odds of CKD [80,81,83]. The specific role of PT cell G1 phase arrest in CKD is unknown.
3.3.7. 2.3.7. increased anaerobic glycolysis in PT leads to increased TIF
Although total renal energy expenditure decreases in CKD, the metabolism of surviving nephrons increases to support compensatory changes in reabsorption [84]. This is due to the fact that when a nephron is lost, the remaining nephron must use more energy in the near term to keep the kidneys functioning. However, this process cannot be sustained indefinitely, therefore tubular loss and fibrosis eventually occur.
When injured, PT undergoes anaerobic glycolysis rather than utilizing the preferred energy substrate fatty acid oxidation (FAO) [85]. When mitochondrial function is impaired, glycolysis is increased probably to help renal tubule survive [86]. However, the downsides of increased glycolysis in renal TEC are also evident. This is because the reprogramming of energy metabolism affects the amount and function of podocytes and exacerbates the extent of TIF [87]. Sustained glycolysis leading to PT atrophy is another cause of exacerbation of TIF. However, it is unclear whether this sustained glycolysis persists in chronic injury and how it contributes to TIF progression [10]. Both in vitro- and in vivo-experiments in CKD showed a decrease in FAO [88,89]. The fatty acid transport protein Slc27a2 deficiency prevented lipid accumulation and TIF [88]. Therefore, this suggests that we can mitigate TIF in terms of enhancing FAO and inhibiting glycolysis.
3.4. PT and glomeruli - a destiny community
The PT is the primary sensor and effector of CKD and AKI progression. In renal injury, PT and glomeruli appear to be a destiny community. Due to their mitochondria-rich and oxidative phosphorylation-dependent nature, PT are particularly susceptible to various forms of injury (obstructive, ischemic, hypoxic, oxidative and metabolic), leading to cell death and eventual formation of tubular glomeruli. In turn, damage to glomeruli also causes tubular damage and leads to TIF [14]. As an example, treating human PTECs with particles isolated from differentiated untreated human podocyte medium upregulated the phosphorylation levels of p38 and Smad3 and increased the expression of fibronectin and type IV collagen, both of which are important components of the ECM [90]. This suggests that glomeruli can interact with PTCs to promote the progression of fibrosis.
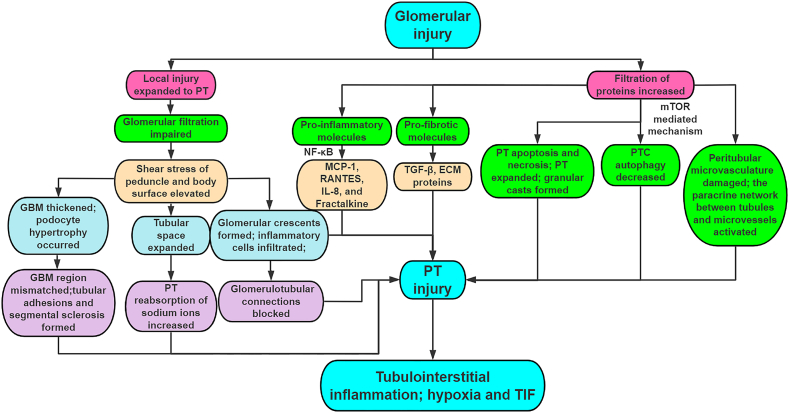
Currently, there are two views on the mechanism by which glomerular injury mediates the involvement of PT in TIF (Fig. 2). One side of the argument suggests that glomerular injury leads to tubular atrophy by triggering tubular mesenchymal injury or local injury, and that this effect later extends to PT, leading to PT apoptosis/necrosis [10]. The main role of the glomerulus is to filter blood flowing through the capillary wall, and the glomerular capillary pressure increases when the kidney is damaged, with a consequent increase in the tensile forces acting on the capillary wall structures, thus causing further tubular damage due to impaired glomerular filtration [91]. Glomerular hyperfiltration increases the flow of ultrafiltrate into the Bowman's capsule space, which leads to elevated shear stresses on the peduncle and body surface [91]. Glomerular basement membrane (GBM) growth and podocyte hypertrophy occur as subsequent events to the increased shear stress. In addition, the growth capacity of podocytes is limited due to hypertrophy. In addition, the ability of podocytes to grow is limited due to hypertrophy. A mismatch between the GBM region and the GBM region covered by peduncle results in detachment of live podocytes, adhesion of capillaries to the parietal epithelium, formation of tubular adhesions, and segmental sclerosis. In addition to causing hypertrophy of glomerular cells, mechanical stress leads to expansion of the tubular space to allow increased reabsorption of sodium in the PT and activation leading to tubulointerstitial inflammation, hypoxia and TIF [91]. Glomerular injury, on the one hand, affects the PT via local expansion of crescentic or inflammatory cells. On the other hand, glomerular injury may lead to impaired PT by blocking glomerulotubular connections through local expansion of crescent bodies or inflammatory cells [10]. Even non-glomerular injuries like the UUO and diabetes have been linked to these atubular glomeruli [92,93].
Fig. 2.
Mechanisms of glomerular injury mediating PT TIF. Local PT extension and increased protein reabsorption are two mechanisms by which glomerular injury cause TIF that are not mutually exclusive, and their arguments can be explained by model differences.
An alternative view is that glomerular injury, through the release of autocrine and paracrine signals, leads to the reabsorption of albumin and associated protein/lipid load by the PT, which in turn promotes TIF [10]. The progression of proteinuria disease is related to the phenotypic transformation of PTC pro-inflammation and pro-fibrosis. Early in vitro results have shown that albumin activates the NF-κB pathway [94], causing the synthesis of pro-inflammatory molecules in PT including monocyte chemotactic protein (MCP) −1 [95], RANTES [96], interleukin-8 (IL-8) [97] and fractalkine [98]. In addition, albumin exposure promotes the production of fibrosis-promoting factor TGF-β and its type II receptors [99,100], as well as the accumulation of the in vitro ECM components collagen IV, laminin, fibronectin [101] and collagen I [102]. In addition to TIF, elevated albumin concentration also induces PT apoptosis [103], expansion and the appearance of granular casts and necrosis [104] in vitro. All these indicate that PT damage plays a significant role in the pathophysiology of proteinuric hypertension [104]. Pathological albumin exposure may exacerbate TIF by inhibiting PT autophagy. In human PTECs line HK-2, excessive albumin absorption and degradation inhibits PT autophagy through an mTOR mediated mechanism, whereas the use of the mTOR inhibitor rapamycin enhances autophagy and attenuates renal tubular damage induced by proteinuria [62,105]. Studies have shown that proteinuria may also aggravate TIF by damaging the microvessels around the renal tubules and activating the paracrine network between the renal tubules and microvessels [106]. Although various experimental results support the second side view, three areas of question remain: Is the quantity and quality of extracorporeal proteinuria consistent with the disease status? Does albumin alone cause PT damage? What is the causal relationship between the acquired phenomena and proteinuria?
In summary, the two mechanisms of TIF induced by glomerular injury, local PT expansion and increased protein reabsorption, are not mutually exclusive from the current data, although different arguments are based on different disease models.
4. PT-targeted drugs for the treatment of CKD
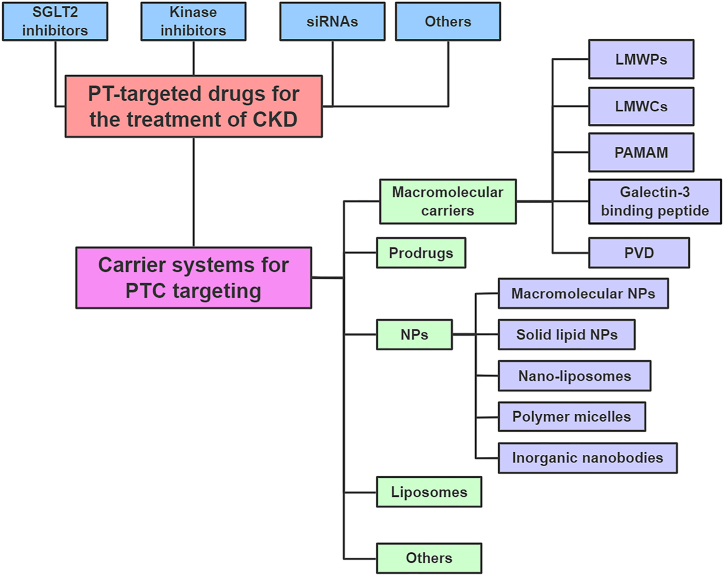
CKD is usually asymptomatic in the early stage, although having different basic pathological foundations; as a result, CKD is frequently diagnosed in the late stage [107]. Finding targets that play a key role in TIF events is particularly critical for developing effective treatments for CKD [108]. Although drugs that inhibit the function of angiotensin II (AT II), such as ACE inhibitors and AT1 receptor antagonists, have been shown to delay the progression of CKD, monotherapy with AT II inhibitors does not change the eventual progression of CKD to end-stage renal disease (ESRD) [109]. Given the critical role of PTECs in the pathogenesis of renal TIF, targeting disease-activating pathways in TECs appears to be a promising strategy to halt or reverse the progression of renal disease. In fact, increasing efforts have been made to develop therapies for renal fibrosis based on the TIF mechanism mediated by PT damage, and considerable results have been achieved (Fig. 3) [110].
Fig. 3.
PT-targeted drugs and delivery systems for the treatment of CKD.
4.1. Sodium-glucose cotransport protein-2 (SGLT2) inhibitors for PT targeting
PTtargeting drugs may provide new ideas for TIF treatment by improving the renal efficacy of anti-fibrosis agents and/or reducing drug toxicity. In diabetic conditions, the amount of SGLT2 glucose reabsorption in PTC is greatly increased due to increased glomerular glucose filtration excess. Increased glucose levels accumulated in PT may inhibit levels of sirtuin3 (Sirt3), thereby inducing the accumulation of hypoxia-inducing factor (HIF) -1α, the formation of pyruvate kinase muscle isoform M2 (PKM2) dimer, and the phosphorylation of signal activated transcription 3 (STAT3), leading to abnormal glycolysis, involved in the transformation and/or programming processes of fibrosis [111]. The glucose transporter 2 (GLUT2) transports accumulated glucose to the vascular side in a gradient-dependent manner [112]. The PT-specific drug, SGLT2 inhibitor empagliflozin, activates the renal tubule-glomerular feedback by blocking the reabsorption of sodium and glucose in PT, so as to increase sodium transport from distal renal tubules to macula, thereby improving filtration burden and reducing blood glucose level, effectively improving renal function and reducing proteinuria in human DN [113]. SGLT2 inhibitors reach the PT brush border membrane through glomerular filtration [114]. It has been shown that SGLT2 inhibitors primarily act on the S1/S2 segment of PT to enhance glucose transport to the downstream urinary tract. In the UUO model, when defects of urogenic glucose transport occur in the basal layer of PTCs, the use of SGLT2 inhibitors can regulate glycolysis to alleviate renal fibrosis [115]. SGLT2 inhibitors can ameliorate kidney damage induced by Ang II [116]. In addition, SGLT2 inhibitors may have the potential to lower blood pressure and mitigate the toxicity of PT glucose reabsorption through diuretic effects. As a result, it is anticipated that PT-specific therapy may open up new treatment options for CKD caused by DN, but until then, solid evidence of the underlying mechanisms based on an analysis of earlier clinical trials is still required.
4.2. Kinase inhibitors for PT targeting
Different in vitro and in vivo studies have shown that kinase inhibitors have promising effects in the treatment of renal fibrosis. TGF-β1 receptor kinase inhibitor targeting PT can inhibit TGF-β1-induced procollagen-1α1 gene expression in HK-2 cells, inhibit the activation of PT cells and fibroblasts, and reduce renal inflammation in UUO rats [117]. Multiple signaling pathways are involved in tubular cell activation in TIF. Therefore, targeting PT with a multitargeted kinase inhibitor such as Sunitinib analog, a multitargeted tyrosine kinase inhibitor, to simultaneously inhibit these cascades appears to be an attractive strategy [118]. Research has shown that a specific member of the serine kinase family, the Fyn kinase, which are involved in the activation of endoplasmic reticulum stress in the diabetic environment leading to PT damage, are feasible targets for the development of therapeutic drugs for DN [119]. Targeted inhibition of Rho kinase pathway in PT cells significantly reduces inflammation and lymphangiogenesis in acute renal allograft rejection [120]. In addition, kinase inhibitors such as p38 mitogen-activated protein kinase (p38 MAPK) inhibitors [121,122]and PDGF receptor kinase inhibitors have also been shown to be effective in the treatment of CKD [123].
4.3. Small interference RNAs (siRNAs) for PT targeting
Targeting the activated pathway in the PT by siRNA has been demonstrated to inhibit TIF and other CKD in various models. The accumulation of siRNA in the kidney is dependent on the ability of siRNA to be freely filtered by the glomeruli, the rate of glomerular filtration, and the uptake of siRNA by TECs. Studies have shown that AQP1 siRNA targeting mouse PTECs was able to knock down water channel aquaporin 1 (AQP1) by up to 50% [124]. Treatment of mice with siRNA targeting apoptosis-related proteins (p53, PKCδ and γGT) protected PT from cisplatin [125]. [126,127] Core fucosylation, which is associated with several biological and pathological functions (e.g., cell signaling, cell migration and inflammatory response), is a unique pattern of protein glycosylation, and silencing its related gene α-1, 6-fucosyltransferase (FUT8) with siRNA can effectively attenuate both endocytic and non-endocytic damage induced by albumin [128]. [[129], [130], [131]] Specific delivery of siRNA targeting meprin-1β, a metalloproteinase of polarized epithelial brush-border membranes, and p53, a key protein in cell-cycle arrest or apoptosis, to PTCs attenuated renal injury after nephrotoxic injury, with subsequent reductions in fibrosis and immune-cell infiltration [132]. In addition, siRNA targeting growth factors, such as CTGF [126,127] and TGF-β1 [129,133], and other profibrotic molecules, such as high glucose-1 [130] and heat shock proteins [131], and molecules associated with apoptosis and inflammation, such as caspase7 [134], have been successfully used in animal trials for the treatment of TIF.
To date, numerous kinase inhibitors and siRNA targeting the activated pathway in the PT have shown great potential for the treatment of CKD in preclinical experiments. However, there are still many difficulties in clinical implementation. For example, the treatment of renal fibrosis with a multitargeted kinase inhibitor may be more effective, but the risk of side effects may also be increased [135]. Although RNA-based strategies offer great therapeutic potential for the treatment of various human diseases, including kidney diseases, the challenges of clinical application, in vivo delivery and stability, and off-target effects need to be overcome.
5. Carrier systems for PTC targeting
In order to reduce the effect of drugs on the affected tissues and control the release of drugs, targeted delivery techniques, including the delivery of kinase inhibitors and siRNA as mentioned above, have been developed and applied. To date, multiple drug delivery systems have been used for the treatment of CKD, most of which are targeted at PTC (Fig. 3) [[136], [137], [138]]. At present, the commonly used delivery systems for targeting PT include macromolecular carriers, prodrugs, nanoparticles (NPs), and liposomes [136]. The successful clinical application of anti-tumor NPs formulations makes NPs the most promising candidates for clinical application in CKD, so the following review will focus on the research findings of NPs.
5.1. Macromolecular carriers
Macromolecular carriers are usually bioactive proteins such as enzymes, immune proteins and peptide hormones in the circulation with molecular weight less than 30,000Da [136]. These macromolecules mainly include: low molecular weight proteins (LMWPs) (lysozyme is the most studied) [139], low molecular weight chitosans (LMWCs) [140], l-serine (Ser)-modified polyamidoamine dendrimer (PAMAM) [141], galectin-3 binding peptide [142], and poly (vinylpyrrolidone-co-dimethyl maleic acid) (PVD) [141]. Although macromolecular carriers have been proved to be promising at the level of animal model experiments, the drawbacks of macromolecular carriers need to be overcome, such as uncertain immunogenicity and compatibility, high batch variability, wide molecular weight range, poor solubility, insufficient drug loading, and possible adverse effects with GBM [143], before they can be widely used in clinical treatment.
5.2. NPs
To deliver the targeted drug to the kidney, the drug is either synthesized into NPs or supplied through a nanodelivery device. A wide range of NPs delivery systems, including macromolecular NPs, solid lipid NPs, nanoliposomes, polymer micelles, and inorganic nanobodies, could theoretically be used for targeted delivery to various cells in the kidney [[144], [145], [146]]. Specific NPs can target ligands to specific cells or tissues, so more and more NPs delivery devices are being used to treat kidney disease [147]. An increasing number of NPs delivery devices are being used for the treatment of renal diseases. The mechanism of renal targeted drug delivery is as follows: drugs carrying NPs enter the kidney through the renal artery flowing through the afferent small arteries, where they either remain in the blood or are filtered from the glomerular capillaries. NPs have the ability to act on specific renal components such as the glycocalyx, endothelial cells, and GBM. Following filtration, NPs can interact with the podocytes in the Bowman's lumen. NPs are reabsorbed and interact with PTEC as they flow through PT with the filtrates [148].
The use of NPs as a vehicle for drug delivery has been successful in the clinical treatment of PT-related nephropathy [149]. Mesoscale nanoparticles (MNPs) with a diameter of 400 nm of poly lactic-co-glycolic acid conjugated to polyethylene glycol (PLGA-PEG) created by Williams et al. can avoid mononuclear phagocytic system and selectively target PT [149,150]. The mechanism of this process may be that the increased uptake pressure in the peritubular capillaries of the renal tubules leads to the release of MNPs into the tubular interstitium between the capillaries and the tubules, where they are subsequently endocytosed by the endothelial cells of the tubules. The uptake efficiency of MNPs in kidney is 26–94 times higher than that in other organs [149], which may be one of the factors for the successful targeting of PT drug delivery by MNPs. Targeting of NPs to the PT is achieved by glomerular filtration of carbon nanotubes translocated to these nuclei [151], an approach that has been applied to treat AKI with oligonucleotides [132]. MNPs are preferred over other organs (>30-fold selectivity) localized in the basolateral region of PTECs. Recent studies have shown that mice treated with NF-κB essential modulator binding peptide (NBP MNP) have a significant protective effect against ischemic AKI, with reduced tubular inflammation, necrosis and apoptosis [152]. In addition, PEG is believed to be necessary to achieve renal targeting and avoid binding to complements [149,153].
The most intriguing advantage of the renal-targeted MNP-mediated selective drug delivery approach is that it allows for the delivery of lower drug doses while improving therapeutic specificity and potentially reducing systemic toxicity [152]. The good news is that although the levels of these NP substances in the kidney are not yet known, observations of renal serum and urine samples and histological levels suggest that selective targeting of the kidney, long-term renal localization, or drug release do not have a negative effect on renal function [150], suggesting promising prospects for the development of drugs that target the renal delivery of NPs.
6. Complications of CKD
Although the most common pathological manifestation of CKD is some forms of kidney fibrosis, it usually has a variety of complications, including: metabolic acidosis, anemia, arterial hypertension, hyperuricemia and increased effective circulating fluid volume, mineral bone disorders, dyslipidemia, endocrine abnormalities, and growth disorders [154]. Among these complications, cardiovascular diseases related to dyslipidemia, hyperuricemia and hypertension are the leading causes of death in patients with CKD [155]. CKD complications can be improved by alternative treatments such as hemodialysis, peritoneal dialysis and kidney transplantation [154]. However, due to the high cost of treatment and the fact that dialysis does not prolong lifespan and poses a major challenge to the quality of life of patients, there is still a long way to go to find new treatment methods. Given the substantial barriers to kidney transplantation, exploring regeneration of injured nephron and adaptation to poor repair may be the way forward for future work.
7. Discussion
Renal fibrogenesis is a very complex and dynamic process in which almost all types of cells in the kidney are involved. Over the past decade, our understanding of TIF, particularly with regard to how inflammation, fibroblast activation, and damage to renal tubules and microvasculature lead to fibrosis, has been significantly enhanced. We are now able to better decipher the diverse origins and phenotypic heterogeneity of ECM-producing myofibroblasts, but still know very little about the contribution of each fibrotic factor, individually or collectively, to TIF. In the past, we have focused on developing therapies for CKD from myofibroblasts, the primary producers of ECM, with unfortunately limited success.
Therefore, we systematically review the central role of PT rather than DT in the occurrence and development of TIF, the main cellular events involved in the process of TIF, the interaction between PT and glomeruli in TIF, the research status of PT-targeted drugs for the treatment of CKD and the current popular PT-targeted drug delivery systems. We tried to provide a reference for better understanding of researches on the causes of TIF and CKD, as well as the development of drugs for the treatment of various kidney diseases. Unfortunately, due to words limit, we do not further discuss the mechanisms underlying the uptake, absorption and clearance of drugs targeting PTs and their carriers.
As the role of PT in mediating TIF continues to be revealed, and various SGLT2 inhibitors, kinase inhibitors and siRNA targeting PT have achieved great success in various preclinical experiments, it is suggested that PT may be a key entry point for inhibiting TIF and treating CKD. The research and development of various PT-targeted delivery systems and the successful clinical application of anti-tumor NP preparations suggest that the development of PT-targeted NP delivery systems may be a key measure for the treatment of CKD in the future [156]. In addition, efforts to find specific markers of myofibroblasts will help us to understand more deeply the process of ECM generation and degradation.
And finally, as an increasing number of studies have shown that hypertension, diabetes and obesity are major contributing factors to CKD [157,158], we should not forget that non-pharmacological approaches, i.e. lifestyle changes, are still key to effectively prevent the underlying diseases as well as renal fibrosis and CKD.
Author contribution statement
All authors listed have significantly contributed to the development and the writing of this article.
Funding statement
Dr Xiangping Qu was supported by Natural Science Foundation of Hunan Province [2020JJ4688], National Natural Science Foundation of China [81570026].
Xiaoqun Qin was supported by National Natural Science Foundation of China [81970033].
Yang Xiang was supported by Natural Science Foundation of Hunan Province [2020JJ4776].
Chi Liu was supported by National Natural Science Foundation of China [82070034], Natural Science Foundation of Hunan Province [2021JJ30898], Open Foundation of Hunan College Innovation Program [20K142].
Lang Pan was supported by National Natural Science Foundation of China [31900424], Natural Science Foundation of Hunan Province [2019JJ50760].
Data availability statement
No data was used for the research described in the article.
Declaration of interest’s statement
The authors declare no competing interests.
References
- 1.Bikbov B., Purcell C.A., Levey A.S., Smith M., Abdoli A., Abebe M., Adebayo O.M., Afarideh M., Agarwal S.K., Agudelo-Botero M., et al. Global, regional, and national burden of chronic kidney disease, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2020;395(10225):709–733. doi: 10.1016/S0140-6736(20)30045-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hill N.R., Fatoba S.T., Oke J.L., Hirst J.A., O'Callaghan C.A., Lasserson D.S., Hobbs F.D. Global prevalence of chronic kidney disease - a systematic review and meta-analysis. PLoS One. 2016;11(7) doi: 10.1371/journal.pone.0158765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Federica Genovese1 A.A.M., Diana Julie Leeming1. Morten Asser Karsdal1. Peter Boor. The extracellular matrix in the kidney: a source of novel non-invasive biomarkers of kidney fibrosis? Fibrogenesis Tissue Repair. 2014;7(1):4. doi: 10.1186/1755-1536-7-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zeisberg M., Neilson E.G. Mechanisms of tubulointerstitial fibrosis. J. Am. Soc. Nephrol. 2010;21(11):1819–1834. doi: 10.1681/ASN.2010080793. [DOI] [PubMed] [Google Scholar]
- 5.Djudjaj S., Boor P. Cellular and molecular mechanisms of kidney fibrosis. Mol. Aspect. Med. 2019;65:16–36. doi: 10.1016/j.mam.2018.06.002. [DOI] [PubMed] [Google Scholar]
- 6.Kaissling B., Lehir M., Kriz W. Renal epithelial injury and fibrosis. Biochim. Biophys. Acta. 2013;1832(7):931–939. doi: 10.1016/j.bbadis.2013.02.010. [DOI] [PubMed] [Google Scholar]
- 7.Nogueira A., Pires M.J., Oliveira P.A. Pathophysiological mechanisms of renal fibrosis: a review of animal models and therapeutic strategies. In Vivo. 2017;31(1):1–22. doi: 10.21873/invivo.11019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Nath K.A. Tubulointerstitial changes as a major determinant. Am. J. Kidney Dis. 1992;20(21):21. doi: 10.1016/s0272-6386(12)80312-x. 17. [DOI] [PubMed] [Google Scholar]
- 9.Bulow R.D., Boor P. Extracellular matrix in kidney fibrosis: more than just a scaffold. J. Histochem. Cytochem. 2019;67(9):643–661. doi: 10.1369/0022155419849388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gewin L.S. Renal fibrosis: primacy of the proximal tubule. Matrix Biol. 2018;68–69:248–262. doi: 10.1016/j.matbio.2018.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Stormark T.A., Strommen K., Iversen B.M., Matre K. Three-dimensional ultrasonography can detect the modulation of kidney volume in two-kidney, one-clip hypertensive rats. Ultrasound Med. Biol. 2007;33(12):1882–1888. doi: 10.1016/j.ultrasmedbio.2007.06.010. [DOI] [PubMed] [Google Scholar]
- 12.Reilly R.F., Ellison D.H. Mammalian distal tubule: physiology, pathophysiology, and molecular anatomy. Physiol. Rev. 2000 Jan;80(81):277–313. doi: 10.1152/physrev.2000.80.1.277. 80. [DOI] [PubMed] [Google Scholar]
- 13.Gobe G.C., Johnson D.W. Distal tubular epithelial cells of the kidney: potential support for proximal tubular cell survival after renal injury. Int. J. Biochem. Cell Biol. 2007;39(9):1551–1561. doi: 10.1016/j.biocel.2007.04.025. [DOI] [PubMed] [Google Scholar]
- 14.Chevalier R.L. The proximal tubule is the primary target of injury and progression of kidney disease: role of the glomerulotubular junction. Am. J. Physiol. Ren. Physiol. 2016;311(1):F145–F161. doi: 10.1152/ajprenal.00164.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hall A.M., Unwin R.J., Parker N., Duchen M.R. Multiphoton imaging reveals differences in mitochondrial function between nephron segments. J. Am. Soc. Nephrol. 2009;20(6):1293–1302. doi: 10.1681/ASN.2008070759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Forbes M.S., Thornhill B.A., Minor J.J., Gordon K.A., Galarreta C.I., Chevalier R.L. Fight-or-flight: murine unilateral ureteral obstruction causes extensive proximal tubular degeneration, collecting duct dilatation, and minimal fibrosis. Am. J. Physiol. Ren. Physiol. 2012;303(1):F120–F129. doi: 10.1152/ajprenal.00110.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Vallon V. The proximal tubule in the pathophysiology of the diabetic kidney. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2011;300(5):R1009–R1022. doi: 10.1152/ajpregu.00809.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Liu Y. Cellular and molecular mechanisms of renal fibrosis. Nat. Rev. Nephrol. 2011;7(12):684–696. doi: 10.1038/nrneph.2011.149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Vernon M.A., Mylonas K.J., Hughes J. Macrophages and renal fibrosis. Semin. Nephrol. 2010;30(3):302–317. doi: 10.1016/j.semnephrol.2010.03.004. [DOI] [PubMed] [Google Scholar]
- 20.Kim S.M., Lee S.H., Kim Y.G., Kim S.Y., Seo J.W., Choi Y.W., Kim D.J., Jeong K.H., Lee T.W., Ihm C.G., et al. Hyperuricemia-induced NLRP3 activation of macrophages contributes to the progression of diabetic nephropathy. Am. J. Physiol. Ren. Physiol. 2015;308(9):F993–F1003. doi: 10.1152/ajprenal.00637.2014. [DOI] [PubMed] [Google Scholar]
- 21.Kassianos A.J., Sampangi S., Wang X., Roper K.E., Beagley K., Healy H., Wilkinson R. Human proximal tubule epithelial cells modulate autologous dendritic cell function. Nephrol. Dial. Transplant. 2013;28(2):303–312. doi: 10.1093/ndt/gfs136. [DOI] [PubMed] [Google Scholar]
- 22.Wilkinson R., Wang X., Roper K.E., Healy H. Activated human renal tubular cells inhibit autologous immune responses. Nephrol. Dial. Transplant. 2011;26(5):1483–1492. doi: 10.1093/ndt/gfq677. [DOI] [PubMed] [Google Scholar]
- 23.Sampangi S., Wang X., Beagley K.W., Klein T., Afrin S., Healy H., Wilkinson R., Kassianos A.J. Human proximal tubule epithelial cells modulate autologous B-cell function. Nephrol. Dial. Transplant. 2015;30(10):1674–1683. doi: 10.1093/ndt/gfv242. [DOI] [PubMed] [Google Scholar]
- 24.Wang Y., Chang J., Yao B., Niu A., Kelly E., Breeggemann M.C., Abboud Werner S.L., Harris R.C., Zhang M.Z. Proximal tubule-derived colony stimulating factor-1 mediates polarization of renal macrophages and dendritic cells, and recovery in acute kidney injury. Kidney Int. 2015;88(6):1274–1282. doi: 10.1038/ki.2015.295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Li S., Mariappan N., Megyesi J., Shank B., Kannan K., Theus S., Price P.M., Duffield J.S., Portilla D. Proximal tubule PPARalpha attenuates renal fibrosis and inflammation caused by unilateral ureteral obstruction. Am. J. Physiol. Ren. Physiol. 2013;305(5):F618–F627. doi: 10.1152/ajprenal.00309.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Meran S., Steadman R. Fibroblasts and myofibroblasts in renal fibrosis. Int. J. Exp. Pathol. 2011;92(3):158–167. doi: 10.1111/j.1365-2613.2011.00764.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Fan J.M., Ng Y.Y., Hill P.A., Nikolic-Paterson D.J., Mu W., Atkins R.C., Lan H.Y. Transforming growth factor-beta regulates tubular epithelial-myofibroblast transdifferentiation in vitro. Kidney Int. 1999;56(4):1455–1467. doi: 10.1046/j.1523-1755.1999.00656.x. [DOI] [PubMed] [Google Scholar]
- 28.Rayego-Mateos S., Morgado-Pascual J.L., Rodrigues-Diez R.R., Rodrigues-Diez R., Falke L.L., Mezzano S., Ortiz A., Egido J., Goldschmeding R., Ruiz-Ortega M. Connective tissue growth factor induces renal fibrosis via epidermal growth factor receptor activation. J. Pathol. 2018;244(2):227–241. doi: 10.1002/path.5007. [DOI] [PubMed] [Google Scholar]
- 29.Yokoi H., Mukoyama M., Nagae T., Mori K., Suganami T., Sawai K., Yoshioka T., Koshikawa M., Nishida T., Takigawa M., et al. Reduction in connective tissue growth factor by antisense treatment ameliorates renal tubulointerstitial fibrosis. J. Am. Soc. Nephrol. 2004;15(6):1430–1440. doi: 10.1097/01.asn.0000130565.69170.85. [DOI] [PubMed] [Google Scholar]
- 30.Shaker A.M., Rakha N.K., El-Shehaby A.M.R., Ramzy T., Hamza W.M., Elkhatib M.M. The significance of fibroblast growth factor-2 and kidney injury molecule-1 as biomarker of interstitial renal fibrosis in glomerulonephritis. Egyp. J. Inter. Med. 2022;34(1) [Google Scholar]
- 31.Chen Y.T., Chang F.C., Wu C.F., Chou Y.H., Hsu H.L., Chiang W.C., Shen J., Chen Y.M., Wu K.D., Tsai T.J., et al. Platelet-derived growth factor receptor signaling activates pericyte-myofibroblast transition in obstructive and post-ischemic kidney fibrosis. Kidney Int. 2011;80(11):1170–1181. doi: 10.1038/ki.2011.208. [DOI] [PubMed] [Google Scholar]
- 32.Jiang K., Zhang Y., He F., Zhang M., Li T., Tu Z., Xu D., Zhang M., Han L., Guo L., et al. A negative feedback loop involving NF-kappaB/TIR8 regulates IL-1beta-induced epithelial- myofibroblast transdifferentiation in human tubular cells. J. Cell Commun. Signal. 2021;15(3):393–403. doi: 10.1007/s12079-021-00620-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Zhou J., Cheng H., Wang Z., Chen H., Suo C., Zhang H., Zhang J., Yang Y., Geng L., Gu M., et al. Bortezomib attenuates renal interstitial fibrosis in kidney transplantation via regulating the EMT induced by TNF-alpha-Smurf1-Akt-mTOR-P70S6K pathway. J. Cell Mol. Med. 2019;23(8):5390–5402. doi: 10.1111/jcmm.14420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Balakumar P., Sambathkumar R., Mahadevan N., Muhsinah A.B., Alsayari A., Venkateswaramurthy N., Jagadeesh G. A potential role of the renin-angiotensin-aldosterone system in epithelial-to-mesenchymal transition-induced renal abnormalities: mechanisms and therapeutic implications. Pharmacol. Res. 2019;146 doi: 10.1016/j.phrs.2019.104314. [DOI] [PubMed] [Google Scholar]
- 35.Li Y., Wang X., Zhang L., Yuan X., Hao J., Ni J., Hao L. Upregulation of allograft inflammatory factor1 expression and secretion by macrophages stimulated with aldosterone promotes renal fibroblasts to a profibrotic phenotype. Int. J. Mol. Med. 2018;42(2):861–872. doi: 10.3892/ijmm.2018.3667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Yan H., Xu J., Xu Z., Yang B., Luo P., He Q. Defining therapeutic targets for renal fibrosis: exploiting the biology of pathogenesis. Biomed. Pharmacother. 2021:143. doi: 10.1016/j.biopha.2021.112115. [DOI] [PubMed] [Google Scholar]
- 37.Liu B.C., Tang T.T., Lv L.L. How tubular epithelial cell injury contributes to renal fibrosis. Adv. Exp. Med. Biol. 2019;1165:233–252. doi: 10.1007/978-981-13-8871-2_11. [DOI] [PubMed] [Google Scholar]
- 38.Sun Y.B., Qu X., Caruana G., Li J. The origin of renal fibroblasts/myofibroblasts and the signals that trigger fibrosis. Differentiation. 2016;92(3):102–107. doi: 10.1016/j.diff.2016.05.008. [DOI] [PubMed] [Google Scholar]
- 39.Nieto M.A., Huang R.Y., Jackson R.A. Thiery JP: emt: 2016. Cell. 2016;166(1):21–45. doi: 10.1016/j.cell.2016.06.028. [DOI] [PubMed] [Google Scholar]
- 40.Sleeman J.P., Thiery J.P. SnapShot: the epithelial-mesenchymal transition. Cell. 2011;145(1):162. doi: 10.1016/j.cell.2011.03.029. e161. [DOI] [PubMed] [Google Scholar]
- 41.Iwano M., Plieth D., Danoff T.M., Xue C., Okada H., Neilson E.G. Evidence that fibroblasts derive from epithelium during tissue fibrosis. J. Clin. Invest. 2002;110(3):341–350. doi: 10.1172/JCI15518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.LeBleu V.S., Taduri G., O'Connell J., Teng Y., Cooke V.G., Woda C., Sugimoto H., Kalluri R. Origin and function of myofibroblasts in kidney fibrosis. Nat. Med. 2013;19(8):1047–1053. doi: 10.1038/nm.3218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Humphreys B.D., Lin S.L., Kobayashi A., Hudson T.E., Nowlin B.T., Bonventre J.V., Valerius M.T., McMahon A.P., Duffield J.S. Fate tracing reveals the pericyte and not epithelial origin of myofibroblasts in kidney fibrosis. Am. J. Pathol. 2010;176(1):85–97. doi: 10.2353/ajpath.2010.090517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Koesters R., Kaissling B., Lehir M., Picard N., Theilig F., Gebhardt R., Glick A.B., Hahnel B., Hosser H., Grone H.J., et al. Tubular overexpression of transforming growth factor-beta1 induces autophagy and fibrosis but not mesenchymal transition of renal epithelial cells. Am. J. Pathol. 2010;177(2):632–643. doi: 10.2353/ajpath.2010.091012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Kriz W., Kaissling B., Le Hir M. Epithelial-mesenchymal transition (EMT) in kidney fibrosis: fact or fantasy? J. Clin. Invest. 2011;121(2):468–474. doi: 10.1172/JCI44595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Li L., Zepeda-Orozco D., Black R., Lin F. Autophagy is a component of epithelial cell fate in obstructive uropathy. Am. J. Pathol. 2010;176(4):1767–1778. doi: 10.2353/ajpath.2010.090345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Lovisa S., Zeisberg M., Kalluri R. Partial epithelial-to-mesenchymal transition and other new mechanisms of kidney fibrosis. Trends Endocrinol. Metabol. 2016;27(10):681–695. doi: 10.1016/j.tem.2016.06.004. [DOI] [PubMed] [Google Scholar]
- 48.Sheng L., Zhuang S. New insights into the role and mechanism of partial epithelial-mesenchymal transition in kidney fibrosis. Front. Physiol. 2020;11 doi: 10.3389/fphys.2020.569322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Lovisa S., LeBleu V.S., Tampe B., Sugimoto H., Vadnagara K., Carstens J.L., Wu C.C., Hagos Y., Burckhardt B.C., Pentcheva-Hoang T., et al. Epithelial-to-mesenchymal transition induces cell cycle arrest and parenchymal damage in renal fibrosis. Nat. Med. 2015;21(9):998–1009. doi: 10.1038/nm.3902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Grande M.T., Sanchez-Laorden B., Lopez-Blau C., De Frutos C.A., Boutet A., Arevalo M., Rowe R.G., Weiss S.J., Lopez-Novoa J.M., Nieto M.A. Snail1-induced partial epithelial-to-mesenchymal transition drives renal fibrosis in mice and can be targeted to reverse established disease. Nat. Med. 2015;21(9):989–997. doi: 10.1038/nm.3901. [DOI] [PubMed] [Google Scholar]
- 51.Grgic I., Campanholle G., Bijol V., Wang C., Sabbisetti V.S., Ichimura T., Humphreys B.D., Bonventre J.V. Targeted proximal tubule injury triggers interstitial fibrosis and glomerulosclerosis. Kidney Int. 2012;82(2):172–183. doi: 10.1038/ki.2012.20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Takaori K., Nakamura J., Yamamoto S., Nakata H., Sato Y., Takase M., Nameta M., Yamamoto T., Economides A.N., Kohno K., et al. Severity and frequency of proximal tubule injury determines renal prognosis. J. Am. Soc. Nephrol. 2016;27(8):2393–2406. doi: 10.1681/ASN.2015060647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Kang H.M., Ahn S.H., Choi P., Ko Y.A., Han S.H., Chinga F., Park A.S., Tao J., Sharma K., Pullman J., et al. Defective fatty acid oxidation in renal tubular epithelial cells has a key role in kidney fibrosis development. Nat. Med. 2015;21(1):37–46. doi: 10.1038/nm.3762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Goossens S., Vandamme N., Van Vlierberghe P., Berx G. EMT transcription factors in cancer development re-evaluated: beyond EMT and MET. Biochim. Biophys. Acta Rev. Canc. 2017;1868(2):584–591. doi: 10.1016/j.bbcan.2017.06.006. [DOI] [PubMed] [Google Scholar]
- 55.Fragiadaki M., Witherden A.S., Kaneko T., Sonnylal S., Pusey C.D., Bou-Gharios G., Mason R.M. Interstitial fibrosis is associated with increased COL1A2 transcription in AA-injured renal tubular epithelial cells in vivo. Matrix Biol. 2011;30(7–8):396–403. doi: 10.1016/j.matbio.2011.07.004. [DOI] [PubMed] [Google Scholar]
- 56.Okada H., Kikuta T., Kobayashi T., Inoue T., Kanno Y., Takigawa M., Sugaya T., Kopp J.B., Suzuki H. Connective tissue growth factor expressed in tubular epithelium plays a pivotal role in renal fibrogenesis. J. Am. Soc. Nephrol. 2005;16(1):133–143. doi: 10.1681/ASN.2004040339. [DOI] [PubMed] [Google Scholar]
- 57.Fu Y., Xiang Y., Wu W., Cai J., Tang C., Dong Z. Persistent activation of autophagy after cisplatin nephrotoxicity promotes renal fibrosis and chronic kidney disease. Front. Pharmacol. 2022;13 doi: 10.3389/fphar.2022.918732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Zhou J., Fan Y., Zhong J., Huang Z., Huang T., Lin S., Chen H. TAK1 mediates excessive autophagy via p38 and ERK in cisplatin-induced acute kidney injury. J. Cell Mol. Med. 2018;22(5):2908–2921. doi: 10.1111/jcmm.13585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Liu B., Sun T., Li H., Qiu S., Li Y., Zhang D. Proximal tubular RAGE mediated the renal fibrosis in UUO model mice via upregulation of autophagy. Cell Death Dis. 2022;13(4):399. doi: 10.1038/s41419-022-04856-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Guan X., Qian Y., Shen Y., Zhang L., Du Y., Dai H., Qian J., Yan Y. Autophagy protects renal tubular cells against ischemia/reperfusion injury in a time-dependent manner. Cell. Physiol. Biochem. 2015;36(1):285–298. doi: 10.1159/000374071. [DOI] [PubMed] [Google Scholar]
- 61.Ma Z., Li L., Livingston M.J., Zhang D., Mi Q., Zhang M., Ding H.F., Huo Y., Mei C., Dong Z. p53/microRNA-214/ULK1 axis impairs renal tubular autophagy in diabetic kidney disease. J. Clin. Invest. 2020;130(9):5011–5026. doi: 10.1172/JCI135536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Vizza D., Perri A., Toteda G., Lupinacci S., Perrotta I., Lofaro D., Leone F., Gigliotti P., La Russa A., Bonofiglio R. Rapamycin-induced autophagy protects proximal tubular renal cells against proteinuric damage through the transcriptional activation of the nerve growth factor receptor NGFR. Autophagy. 2018;14(6):1028–1042. doi: 10.1080/15548627.2018.1448740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Nam S.A., Kim W.-Y., Kim J.W., Park S.H., Kim H.L., Lee M.-S., Komatsu M., Ha H., Lim J.H., Park C.W., et al. Autophagy attenuates tubulointerstital fibrosis through regulating transforming growth factor-β and NLRP3 inflammasome signaling pathway. Cell Death Dis. 2019;10(2) doi: 10.1038/s41419-019-1356-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Levine B., Yuan J. Autophagy in cell death: an innocent convict? J. Clin. Invest. 2005;115(10):2679–2688. doi: 10.1172/JCI26390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Maiuri M.C., Zalckvar E., Kimchi A., Kroemer G. Self-eating and self-killing: crosstalk between autophagy and apoptosis. Nat. Rev. Mol. Cell Biol. 2007;8(9):741–752. doi: 10.1038/nrm2239. [DOI] [PubMed] [Google Scholar]
- 66.Thorburn A. Apoptosis and autophagy: regulatory connections between two supposedly different processes. Apoptosis. 2008;13(1):1–9. doi: 10.1007/s10495-007-0154-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Scarlatti F., Granata R., Meijer A.J., Codogno P. Does autophagy have a license to kill mammalian cells? Cell Death Differ. 2009;16(1):12–20. doi: 10.1038/cdd.2008.101. [DOI] [PubMed] [Google Scholar]
- 68.Baisantry A., Bhayana S., Rong S., Ermeling E., Wrede C., Hegermann J., Pennekamp P., Sorensen-Zender I., Haller H., Melk A., et al. Autophagy induces prosenescent changes in proximal tubular S3 segments. J. Am. Soc. Nephrol. 2016;27(6):1609–1616. doi: 10.1681/ASN.2014111059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Xu Y., Ruan S., Wu X., Chen H., Zheng K., Fu B. Autophagy and apoptosis in tubular cells following unilateral ureteral obstruction are associated with mitochondrial oxidative stress. Int. J. Mol. Med. 2013;31(3):628–636. doi: 10.3892/ijmm.2013.1232. [DOI] [PubMed] [Google Scholar]
- 70.Livingston M.J., Ding H.F., Huang S., Hill J.A., Yin X.M., Dong Z. Persistent activation of autophagy in kidney tubular cells promotes renal interstitial fibrosis during unilateral ureteral obstruction. Autophagy. 2016;12(6):976–998. doi: 10.1080/15548627.2016.1166317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Xu Y., Yang S., Huang J., Ruan S., Zheng Z., Lin J. Tgf-beta1 induces autophagy and promotes apoptosis in renal tubular epithelial cells. Int. J. Mol. Med. 2012;29(5):781–790. doi: 10.3892/ijmm.2012.911. [DOI] [PubMed] [Google Scholar]
- 72.Deng J.K., Zhang X., Wu H.L., Gan Y., Ye L., Zheng H., Zhu Z., Liu W.J., Liu H.F. ROS-ERK pathway as dual mediators of cellular injury and autophagy-associated adaptive response in urinary protein-irritated renal tubular epithelial cells. J. Diabetes Res. 2021;2021 doi: 10.1155/2021/6614848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Hetz C., Martinon F., Rodriguez D., Glimcher L.H. The unfolded protein response: integrating stress signals through the stress sensor IRE1alpha. Physiol. Rev. 2011;91(4):1219–1243. doi: 10.1152/physrev.00001.2011. [DOI] [PubMed] [Google Scholar]
- 74.Dihazi H., Dihazi G.H., Bibi A., Eltoweissy M., Mueller C.A., Asif A.R., Rubel D., Vasko R., Mueller G.A. Secretion of ERP57 is important for extracellular matrix accumulation and progression of renal fibrosis, and is an early sign of disease onset. J. Cell Sci. 2013;126(Pt 16):3649–3663. doi: 10.1242/jcs.125088. [DOI] [PubMed] [Google Scholar]
- 75.Lenna S., Trojanowska M. The role of endoplasmic reticulum stress and the unfolded protein response in fibrosis. Curr. Opin. Rheumatol. 2012;24(6):663–668. doi: 10.1097/BOR.0b013e3283588dbb. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Diaz-Bulnes P., Saiz M.L., Corte-Iglesias V., Rodrigues-Diez R.R., Bernardo Florez A., Ruiz Bernet C., Martin Martin C., Ruiz-Ortega M., Suarez-Alvarez B., Lopez-Larrea C. Demethylation of H3K9 and H3K27 contributes to the tubular renal damage triggered by endoplasmic reticulum stress. Antioxidants. 2022;11(7) doi: 10.3390/antiox11071355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Fougeray S., Bouvier N., Beaune P., Legendre C., Anglicheau D., Thervet E., Pallet N. Metabolic stress promotes renal tubular inflammation by triggering the unfolded protein response. Cell Death Dis. 2011;2:e143. doi: 10.1038/cddis.2011.26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Cybulsky A.V. Endoplasmic reticulum stress, the unfolded protein response and autophagy in kidney diseases. Nat. Rev. Nephrol. 2017;13(11):681–696. doi: 10.1038/nrneph.2017.129. [DOI] [PubMed] [Google Scholar]
- 79.Li Q., Chen C., Chen X., Han M., Li J. Dexmedetomidine attenuates renal fibrosis via alpha2-adrenergic receptor-dependent inhibition of cellular senescence after renal ischemia/reperfusion. Life Sci. 2018;207:1–8. doi: 10.1016/j.lfs.2018.05.003. [DOI] [PubMed] [Google Scholar]
- 80.Yang L., Besschetnova T.Y., Brooks C.R., Shah J.V., Bonventre J.V. Epithelial cell cycle arrest in G2/M mediates kidney fibrosis after injury. Nat. Med. 2010;16(5):535–543. doi: 10.1038/nm.2144. 531pp. following 143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Cianciolo Cosentino C., Skrypnyk N.I., Brilli L.L., Chiba T., Novitskaya T., Woods C., West J., Korotchenko V.N., McDermott L., Day B.W., et al. Histone deacetylase inhibitor enhances recovery after AKI. J. Am. Soc. Nephrol. 2013;24(6):943–953. doi: 10.1681/ASN.2012111055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Li H., Peng X., Wang Y., Cao S., Xiong L., Fan J., Wang Y., Zhuang S., Yu X., Mao H. Atg5-mediated autophagy deficiency in proximal tubules promotes cell cycle G2/M arrest and renal fibrosis. Autophagy. 2016;12(9):1472–1486. doi: 10.1080/15548627.2016.1190071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Tang J., Liu N., Tolbert E., Ponnusamy M., Ma L., Gong R., Bayliss G., Yan H., Zhuang S. Sustained activation of EGFR triggers renal fibrogenesis after acute kidney injury. Am. J. Pathol. 2013;183(1):160–172. doi: 10.1016/j.ajpath.2013.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Harris D.C., Chan L., Schrier R.W. Remnant kidney hypermetabolism and progression of chronic renal failure. Am. J. Physiol. Lung Cell Mol. Physiol. 1988;254(252 Pt 252):F267–F276. doi: 10.1152/ajprenal.1988.254.2.F267. [DOI] [PubMed] [Google Scholar]
- 85.Chihanga T., Ma Q., Nicholson J.D., Ruby H.N., Edelmann R.E., Devarajan P., Kennedy M.A. NMR spectroscopy and electron microscopy identification of metabolic and ultrastructural changes to the kidney following ischemia-reperfusion injury. Am. J. Physiol. Ren. Physiol. 2018;314(2):F154–F166. doi: 10.1152/ajprenal.00363.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Lan R., Geng H., Singha P.K., Saikumar P., Bottinger E.P., Weinberg J.M., Venkatachalam M.A. Mitochondrial pathology and glycolytic shift during proximal tubule atrophy after ischemic AKI. J. Am. Soc. Nephrol. 2016;27(11):3356–3367. doi: 10.1681/ASN.2015020177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Li M., Jia F., Zhou H., J D., Yang M. Elevated aerobic glycolysis in renal tubular epithelial cells influences the proliferation and differentiation of podocytes and promotes renal interstitial fibrosis. Eur. Rev. Med. Pharmacol. Sci. 2018;22(16):5082–5090. doi: 10.26355/eurrev_201808_15701. [DOI] [PubMed] [Google Scholar]
- 88.Cheng L., Ge M., Lan Z., Ma Z., Chi W., Kuang W., Sun K., Zhao X., Liu Y., Feng Y., et al. Zoledronate dysregulates fatty acid metabolism in renal tubular epithelial cells to induce nephrotoxicity. Arch. Toxicol. 2018;92(1):469–485. doi: 10.1007/s00204-017-2048-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Li J., Yang Y., Li Q., Wei S., Zhou Y., Yu W., Xue L., Zhou L., Shen L., Lu G., et al. STAT6 contributes to renal fibrosis by modulating PPARalpha-mediated tubular fatty acid oxidation. Cell Death Dis. 2022;13(1):66. doi: 10.1038/s41419-022-04515-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Munkonda M.N., Akbari S., Landry C., Sun S., Xiao F., Turner M., Holterman C.E., Nasrallah R., Hebert R.L., Kennedy C.R.J., et al. Podocyte-derived microparticles promote proximal tubule fibrotic signaling via p38 MAPK and CD36. J. Extracell. Vesicles. 2018;7(1) doi: 10.1080/20013078.2018.1432206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Chagnac A., Zingerman B., Rozen-Zvi B., Herman-Edelstein M. Consequences of glomerular hyperfiltration: the role of physical forces in the pathogenesis of chronic kidney disease in diabetes and obesity. Nephron. 2019;143(1):38–42. doi: 10.1159/000499486. [DOI] [PubMed] [Google Scholar]
- 92.Najafian B., Kim Y., Crosson J.T., Mauer M. Atubular glomeruli and glomerulotubular junction abnormalities in diabetic nephropathy. J. Am. Soc. Nephrol. 2003;14(4):908–917. doi: 10.1097/01.asn.0000057854.32413.81. [DOI] [PubMed] [Google Scholar]
- 93.Forbes M.S., Thornhill B.A., Chevalier R.L. Proximal tubular injury and rapid formation of atubular glomeruli in mice with unilateral ureteral obstruction: a new look at an old model. Am. J. Physiol. Ren. Physiol. 2011;301(1):F110–F117. doi: 10.1152/ajprenal.00022.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Zoja C., Garcia P.B., Remuzzi G. The role of chemokines in progressive renal disease. Front. Biosci. 2009;14(15):1815–1822. doi: 10.2741/3343. [DOI] [PubMed] [Google Scholar]
- 95.Wang Y., Chen J., Chen L., Tay Y.C., Rangan G.K., Harris D.C. Protein overload stimulates RANTES production by proximal tubular cells depending on NF-kappa B activation. J. Am. Soc. Nephrol. 1997;8(10):1537–1545. [Google Scholar]
- 96.Zoja C., Donadelli R., Colleoni S., Figliuzzi M., Bonazzola S., Morigi M., Remuzzi G. Protein overload stimulates RANTES production by proximal tubular cells depending on NF-kappa B activation. Kidney Int. 1998;53(56):1608–1615. doi: 10.1046/j.1523-1755.1998.00905.x. [DOI] [PubMed] [Google Scholar]
- 97.Tang S., Leung J.C.K., Abe K., Chan K.W., Chan L.Y.Y., Chan T.M., Lai K.N. Albumin stimulates interleukin-8 expression in proximal tubular epithelial cells in vitro and in vivo. J. Clin. Invest. 2003;111(4):515–527. doi: 10.1172/JCI16079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Donadelli R., Zanchi C., Morigi M., Buelli S., Batani C., Tomasoni S., Corna D., Rottoli D., Benigni A., Abbate M., et al. Protein overload induces fractalkine upregulation in proximal tubular cells through nuclear factor kappaB- and p38 mitogen-activated protein kinase-dependent pathways. J. Am. Soc. Nephrol. 2003;14(10):2436–2446. doi: 10.1097/01.asn.0000089564.55411.7f. [DOI] [PubMed] [Google Scholar]
- 99.Yard B.A., Chorianopoulos E., Herr D., Fjvd Woude. Regulation of endothelin-1 and transforming growth factor-beta1 production in cultured proximal tubular cells by albumin and heparan sulphate glycosaminoglycans. Nephrol. Dial. Transplant. 2001;16(19):1769–1775. doi: 10.1093/ndt/16.9.1769. [DOI] [PubMed] [Google Scholar]
- 100.Wolf G., Schroeder R., Ziyadeh F.N., Stahl R.A.K. Albumin up-regulates the type II transforming growth factor-beta receptor in cultured proximal tubular cells. Kidney Int. 2004;66(65):1849–1858. doi: 10.1111/j.1523-1755.2004.00958.x. [DOI] [PubMed] [Google Scholar]
- 101.Stephan J.P., Mao W., Filvaroff E., Cai L., Rabkin R., Pan G. Albumin stimulates the accumulation of extracellular matrix in renal tubular epithelial cells. Am. J. Nephrol. 2004;24(1):14–19. doi: 10.1159/000075347. [DOI] [PubMed] [Google Scholar]
- 102.Hu J., Wang W., Zhang F., Li P.L., Boini K.M., Yi F., Li N. Hypoxia inducible factor-1alpha mediates the profibrotic effect of albumin in renal tubular cells. Sci. Rep. 2017;7(1) doi: 10.1038/s41598-017-15972-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Burlaka I., Nilsson L.M., Scott L., Holtback U., Eklof A.C., Fogo A.B., Brismar H., Aperia A. Prevention of apoptosis averts glomerular tubular disconnection and podocyte loss in proteinuric kidney disease. Kidney Int. 2016;90(1):135–148. doi: 10.1016/j.kint.2016.03.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Endres B.T., Sandoval R.M., Rhodes G.J., Campos-Bilderback S.B., Kamocka M.M., McDermott-Roe C., Staruschenko A., Molitoris B.A., Geurts A.M., Palygin O. Intravital imaging of the kidney in a rat model of salt-sensitive hypertension. Am. J. Physiol. Ren. Physiol. 2017;313(2):F163–F173. doi: 10.1152/ajprenal.00466.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Nolin A.C., Mulhern R.M., Panchenko M.V., Pisarek-Horowitz A., Wang Z., Shirihai O., Borkan S.C., Havasi A. Proteinuria causes dysfunctional autophagy in the proximal tubule. Am. J. Physiol. Ren. Physiol. 2016;311(6):F1271–F1279. doi: 10.1152/ajprenal.00125.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Liu A., Wang X., Hu X., Deng Y., Wen X., Lin B., Zhou M., Wang W., Luo Y., Deng J., et al. Core fucosylation involvement in the paracrine regulation of proteinuria-induced renal interstitial fibrosis evaluated with the use of a microfluidic chip. Acta Biomater. 2022;142:99–112. doi: 10.1016/j.actbio.2022.02.020. [DOI] [PubMed] [Google Scholar]
- 107.Krol E., Rutkowski B., Czarniak P., Kraszewska E., Lizakowski S., Szubert R., Czekalski S., Sulowicz W., Wiecek A. Early detection of chronic kidney disease: results of the PolNef study. Am. J. Nephrol. 2009;29(3):264–273. doi: 10.1159/000158526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Francois H., Chatziantoniou C. Renal fibrosis: recent translational aspects. Matrix Biol. 2018;68–69:318–332. doi: 10.1016/j.matbio.2017.12.013. [DOI] [PubMed] [Google Scholar]
- 109.Ripley E. Complementary effects of angiotensin-converting enzyme inhibitors and angiotensin receptor blockers in slowing the progression of chronic kidney disease. Am. Heart J. 2009;157(6 Suppl):S7–S16. doi: 10.1016/j.ahj.2009.04.008. [DOI] [PubMed] [Google Scholar]
- 110.Christensen E.I., Birn H., Storm T., Weyer K., Nielsen R. Endocytic receptors in the renal proximal tubule. Physiology. 2012;27(4):223–236. doi: 10.1152/physiol.00022.2012. [DOI] [PubMed] [Google Scholar]
- 111.Vallon V. Glucose transporters in the kidney in health and disease. Pflügers Archiv. 2020;472(9):1345–1370. doi: 10.1007/s00424-020-02361-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Morita M., Kanasaki K. Sodium-glucose cotransporter-2 inhibitors for diabetic kidney disease: targeting Warburg effects in proximal tubular cells. Diabetes Metab. 2020;46(5):353–361. doi: 10.1016/j.diabet.2020.06.005. [DOI] [PubMed] [Google Scholar]
- 113.Wanner C., Inzucchi S.E., Lachin J.M., Fitchett D., von Eynatten M., Mattheus M., Johansen O.E., Woerle H.J., Broedl U.C., Zinman B., et al. Empagliflozin and progression of kidney disease in type 2 diabetes. N. Engl. J. Med. 2016;375(4):323–334. doi: 10.1056/NEJMoa1515920. [DOI] [PubMed] [Google Scholar]
- 114.Gallo L.A., Wright E.M., Vallon V. Probing SGLT2 as a therapeutic target for diabetes: basic physiology and consequences. Diabetes Vasc. Dis. Res. 2015;12(2):78–89. doi: 10.1177/1479164114561992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Ding H., Jiang L., Xu J., Bai F., Zhou Y., Yuan Q., Luo J., Zen K., Yang J. Inhibiting aerobic glycolysis suppresses renal interstitial fibroblast activation and renal fibrosis. Am. J. Physiol. Ren. Physiol. 2017;313(3):F561–F575. doi: 10.1152/ajprenal.00036.2017. [DOI] [PubMed] [Google Scholar]
- 116.Miyata K.N., Lo C.S., Zhao S., Liao M.C., Pang Y., Chang S.Y., Peng J., Kretzler M., Filep J.G., Ingelfinger J.R., et al. Angiotensin II up-regulates sodium-glucose co-transporter 2 expression and SGLT2 inhibitor attenuates Ang II-induced hypertensive renal injury in mice. Clin. Sci. (Lond.) 2021;135(7):943–961. doi: 10.1042/CS20210094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Prakash J., de Borst M.H., van Loenen-Weemaes A.M., Lacombe M., Opdam F., van Goor H., Meijer D.K., Moolenaar F., Poelstra K., Kok R.J. Cell-specific delivery of a transforming growth factor-beta type I receptor kinase inhibitor to proximal tubular cells for the treatment of renal fibrosis. Pharm. Res. (N. Y.) 2008;25(10):2427–2439. doi: 10.1007/s11095-007-9515-x. [DOI] [PubMed] [Google Scholar]
- 118.Kok R., Dolman Harmsen, Pieters Sparidans, Lacombe Szokol, Orfi Keri, Storm, et al. Targeting of a platinum-bound sunitinib analog to renal proximal tubular cells. Int. J. Nanomed. 2012;7:417–433. doi: 10.2147/IJN.S26485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Dorotea Debra, Jiang Songling, Eun Seon Pak. Son J.B., Choi H.G., Ahn S.-M., Ha H. Pan-Src kinase inhibitor treatment attenuates diabetic kidney injury via inhibition of Fyn kinase-mediated endoplasmic reticulum stress. Exp. Mol. Med. 2022;54(58):1086–1097. doi: 10.1038/s12276-022-00810-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Poosti F., Yazdani S., Dolman M.E., Kok R.J., Chen C., Ding G., Lacombe M., Prakash J., van den Born J., Hillebrands J.L., et al. Targeted inhibition of renal Rho kinase reduces macrophage infiltration and lymphangiogenesis in acute renal allograft rejection. Eur. J. Pharmacol. 2012;694(1–3):111–119. doi: 10.1016/j.ejphar.2012.08.010. [DOI] [PubMed] [Google Scholar]
- 121.Prakash J., Sandovici M., Saluja V., Lacombe M., Schaapveld R.Q., de Borst M.H., van Goor H., Henning R.H., Proost J.H., Moolenaar F., et al. Intracellular delivery of the p38 mitogen-activated protein kinase inhibitor SB202190 [4-(4-fluorophenyl)-2-(4-hydroxyphenyl)-5-(4-pyridyl)1H-imidazole] in renal tubular cells: a novel strategy to treat renal fibrosis. J. Pharmacol. Exp. Therapeut. 2006;319(1):8–19. doi: 10.1124/jpet.106.106054. [DOI] [PubMed] [Google Scholar]
- 122.Nishida M., Okumura Y., Sato H., Hamaoka K. Delayed inhibition of p38 mitogen-activated protein kinase ameliorates renal fibrosis in obstructive nephropathy. Nephrol. Dial. Transplant. 2008;23(8):2520–2524. doi: 10.1093/ndt/gfn309. [DOI] [PubMed] [Google Scholar]
- 123.Ludewig D., Kosmehl H., Sommer M., Böhmer F.-D., Stein G. PDGF receptor kinase blocker AG1295 attenuates interstitial fibrosis in rat kidney after unilateral obstruction. Cell Tissue Res. 2000;299(1):97–103. doi: 10.1007/s004419900118. [DOI] [PubMed] [Google Scholar]
- 124.Gao S., Hein S., Dagnaes-Hansen F., Weyer K., Yang C., Nielsen R., Christensen E.I., Fenton R.A., Kjems J. Megalin-mediated specific uptake of chitosan/siRNA nanoparticles in mouse kidney proximal tubule epithelial cells enables AQP1 gene silencing. Theranostics. 2014;4(10):1039–1051. doi: 10.7150/thno.7866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Aydin E., Cebeci A., Lekesizcan A. Prevention of cisplatin-induced nephrotoxicity by kidney-targeted siRNA delivery. Int. J. Pharm. 2022;628 doi: 10.1016/j.ijpharm.2022.122268. [DOI] [PubMed] [Google Scholar]
- 126.Liu Y., Li W., Liu H., Peng Y., Yang Q., Xiao L., Liu Y., Liu F. Inhibition effect of small interfering RNA of connective tissue growth factor on the expression of extracellular matrix molecules in cultured human renal proximal tubular cells. Ren. Fail. 2014;36(2):278–284. doi: 10.3109/0886022X.2013.846866. [DOI] [PubMed] [Google Scholar]
- 127.Khaja F., Jayawardena D., Kuzmis A., Onyuksel H. Targeted sterically stabilized phospholipid siRNA nanomedicine for hepatic and renal fibrosis. Nanomaterials. 2016;6(1) doi: 10.3390/nano6010008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Wang D., Fang M., Shen N., Li L., Wang W., Wang L., Lin H. Inhibiting post-translational core fucosylation protects against albumin-induced proximal tubular epithelial cell injury. Am. J. Transl. Res. 2017;9(10):4415–4427. [PMC free article] [PubMed] [Google Scholar]
- 129.Hwang M., Kim H.J., Noh H.J., Chang Y.C., Chae Y.M., Kim K.H., Jeon J.P., Lee T.S., Oh H.K., Lee Y.S., et al. TGF-beta1 siRNA suppresses the tubulointerstitial fibrosis in the kidney of ureteral obstruction. Exp. Mol. Pathol. 2006;81(1):48–54. doi: 10.1016/j.yexmp.2005.11.005. [DOI] [PubMed] [Google Scholar]
- 130.Murphy M., Docherty N.G., Griffin B., Howlin J., McArdle E., McMahon R., Schmid H., Kretzler M., Droguett A., Mezzano S., et al. IHG-1 amplifies TGF-beta1 signaling and is increased in renal fibrosis. J. Am. Soc. Nephrol. 2008;19(9):1672–1680. doi: 10.1681/ASN.2007101080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Xia Z., Abe K., Furusu A., Miyazaki M., Obata Y., Tabata Y., Koji T., Kohno S. Suppression of renal tubulointerstitial fibrosis by small interfering RNA targeting heat shock protein 47. Am. J. Nephrol. 2008;28(1):34–46. doi: 10.1159/000108759. [DOI] [PubMed] [Google Scholar]
- 132.Alidori S., Akhavein N., Thorek D.L., Behling K., Romin Y., Queen D., Beattie B.J., Manova-Todorova K., Bergkvist M., Scheinberg D.A., et al. Targeted fibrillar nanocarbon RNAi treatment of acute kidney injury. Sci. Transl. Med. 2016;8(331):331ra339. doi: 10.1126/scitranslmed.aac9647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Wang M., Chen D.Q., Wang M.C., Chen H., Chen L., Liu D., Zhao H., Zhao Y.Y. Poricoic acid ZA, a novel RAS inhibitor, attenuates tubulo-interstitial fibrosis and podocyte injury by inhibiting TGF-beta/Smad signaling pathway. Phytomedicine. 2017;36:243–253. doi: 10.1016/j.phymed.2017.10.008. [DOI] [PubMed] [Google Scholar]
- 134.Ke Y., Tang H., Ye C., Lei C.T., Wang Y.M., Su H., Zhang C., Jiang H.J. Role and association of inflammatory and apoptotic caspases in renal tubulointerstitial fibrosis. Kidney Blood Press. Res. 2016;41(5):643–653. doi: 10.1159/000447933. [DOI] [PubMed] [Google Scholar]
- 135.Force T., Krause D.S., Van Etten R.A. Molecular mechanisms of cardiotoxicity of tyrosine kinase inhibition. Nat. Rev. Cancer. 2007;7(5):332–344. doi: 10.1038/nrc2106. [DOI] [PubMed] [Google Scholar]
- 136.Zhou P., Sun X., Zhang Z. Kidney-targeted drug delivery systems. Acta Pharm. Sin. B. 2014;4(1):37–42. doi: 10.1016/j.apsb.2013.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Dolman M.E., Harmsen S., Storm G., Hennink W.E., Kok R.J. Drug targeting to the kidney: advances in the active targeting of therapeutics to proximal tubular cells. Adv. Drug Deliv. Rev. 2010;62(14):1344–1357. doi: 10.1016/j.addr.2010.07.011. [DOI] [PubMed] [Google Scholar]
- 138.Nastase M.V., Zeng-Brouwers J., Wygrecka M., Schaefer L. Targeting renal fibrosis: mechanisms and drug delivery systems. Adv. Drug Deliv. Rev. 2018;129:295–307. doi: 10.1016/j.addr.2017.12.019. [DOI] [PubMed] [Google Scholar]
- 139.Zhang Z., Zheng Q., Han J., Gao G., Liu J., Gong T., Gu Z., Huang Y., Sun X., He Q. The targeting of 14-succinate triptolide-lysozyme conjugate to proximal renal tubular epithelial cells. Biomaterials. 2009;30(7):1372–1381. doi: 10.1016/j.biomaterials.2008.11.035. [DOI] [PubMed] [Google Scholar]
- 140.Yuan Z.X., Sun X., Gong T., Ding H., Fu Y., Zhang Z.R. Randomly 50% N-acetylated low molecular weight chitosan as a novel renal targeting carrier. J. Drug Target. 2007;15(4):269–278. doi: 10.1080/10611860701289875. [DOI] [PubMed] [Google Scholar]
- 141.Matsuura S., Katsumi H., Suzuki H., Hirai N., Takashima R., Morishita M., Sakane T., Yamamoto A. L-cysteine and L-serine modified dendrimer with multiple reduced thiols as a kidney-targeting reactive oxygen species scavenger to prevent RenalIschemia/reperfusion injury. Pharmaceutics. 2018;10(14):251. doi: 10.3390/pharmaceutics10040251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Yamamoto Y., Tsutsumi Y., Yoshioka Y., Kamada H., Sato-Kamada K., Okamoto T., Mukai Y., Shibata H., Nakagawa S., Mayumi T. Poly(vinylpyrrolidone-co-dimethyl maleic acid) as a novel renal targeting carrier. J. Contr. Release. 2004;95(2):229–237. doi: 10.1016/j.jconrel.2003.11.017. [DOI] [PubMed] [Google Scholar]
- 143.Yuan Z.-X., Shang Z., Gu J., He L. Renal targeting delivery systems. Future Med. Chem. 2019;11(17):2237–2240. doi: 10.4155/fmc-2019-0152. [DOI] [PubMed] [Google Scholar]
- 144.Yuan Z.X., Mo J., Zhao G., Shu G., Fu H.L., Zhao W. Targeting strategies for renal cell carcinoma: from renal cancer cells to renal cancer stem cells. Front. Pharmacol. 2016;7:423. doi: 10.3389/fphar.2016.00423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Shamsi A., Ahmed A., Bano B. Structural transition of kidney cystatin induced by silicon dioxide nanoparticles: an implication for renal diseases. Int. J. Biol. Macromol. 2017;94(Pt B):754–761. doi: 10.1016/j.ijbiomac.2016.10.019. [DOI] [PubMed] [Google Scholar]
- 146.Reshi M.S., Shrivastava S., Jaswal A., Sinha N., Uthra C., Shukla S. Gold nanoparticles ameliorate acetaminophen induced hepato-renal injury in rats. Exp. Toxicol. Pathol. 2017;69(4):231–240. doi: 10.1016/j.etp.2017.01.009. [DOI] [PubMed] [Google Scholar]
- 147.Ma Y., Cai F., Li Y., Chen J., Han F., Lin W. A review of the application of nanoparticles in the diagnosis and treatment of chronic kidney disease. Bioact. Mater. 2020;5(3):732–743. doi: 10.1016/j.bioactmat.2020.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Merlin J.P.J., Li X. Role of nanotechnology and their perspectives in the treatment of kidney diseases. Front. Genet. 2021;12 doi: 10.3389/fgene.2021.817974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Williams R.M., Shah J., Ng B.D., Minton D.R., Gudas L.J., Park C.Y., Heller D.A. Mesoscale nanoparticles selectively target the renal proximal tubule epithelium. Nano Lett. 2015;15(4):2358–2364. doi: 10.1021/nl504610d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Williams R.M., Shah J., Tian H.S., Chen X., Geissmann F., Jaimes E.A., Heller D.A. Selective nanoparticle targeting of the renal tubules. Hypertension. 2018;71(1):87–94. doi: 10.1161/HYPERTENSIONAHA.117.09843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Ruggiero A., Villa C.H., Bander E., Rey D.A., Bergkvist M., Batt C.A., Manova-Todorova K., Deen W.M., Scheinberg D.A., McDevitt M.R. Paradoxical glomerular filtration of carbon nanotubes. Proc. Natl. Acad. Sci. U. S. A. 2010;107(27):12369–12374. doi: 10.1073/pnas.0913667107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Han S.J., Williams R.M., Kim M., Heller D.A., D'Agati V., Schmidt-Supprian M., Lee H.T. Renal proximal tubular NEMO plays a critical role in ischemic acute kidney injury. JCI Insight. 2020;5(19) doi: 10.1172/jci.insight.139246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Gref R., Minamitake Y., Peracchia M.T., Trubetskoy V., Torchilin V., Langer R. Biodegradable long-circulating polymeric nanospheres. Science. 1994;263(5153):1600–5153. doi: 10.1126/science.8128245. [DOI] [PubMed] [Google Scholar]
- 154.Romagnani P., Remuzzi G., Glassock R., Levin A., Jager K.J., Tonelli M., Massy Z., Wanner C., Anders H.J. Chronic kidney disease. Nat. Rev. Dis. Prim. 2017;3 doi: 10.1038/nrdp.2017.88. [DOI] [PubMed] [Google Scholar]
- 155.Thomas B., Matsushita K., Abate K.H., Al-Aly Z., Arnlov J., Asayama K., Atkins R., Badawi A., Ballew S.H., Banerjee A., et al. Global cardiovascular and renal outcomes of reduced GFR. J. Am. Soc. Nephrol. 2017;28(7):2167–2179. doi: 10.1681/ASN.2016050562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Li M., Jia F., Zhou H., Di J., Yang M. Elevated aerobic glycolysis in renal tubular epithelial cells influences the proliferation and differentiation of podocytes and promotes renal interstitial fibrosis. Eur. Rev. Med. Pharmacol. Sci. 2018 doi: 10.26355/eurrev_201808_15701. (Eur Rev Med Pharmacol Sci.) [DOI] [PubMed] [Google Scholar]
- 157.Shabaka A., Cases-Corona C., Fernandez-Juarez G. Therapeutic insights in chronic kidney disease progression. Front. Med. 2021;8(23):645187. doi: 10.3389/fmed.2021.645187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Pugh D., Gallacher P.J., Dhaun N. Management of hypertension in chronic kidney disease. Drugs. 2019;79(4):365–379. doi: 10.1007/s40265-019-1064-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No data was used for the research described in the article.