Abstract
Smoking poses a threat to global public health. This study analyzed data from the 2016–2018 National Health and Nutrition Examination Survey to investigate smoking’s impact on periodontal health and identify potential risk factors associated with poor periodontal health in Korean adults. The final study population was 9178 patients, with 4161 men and 5017 women. The dependent variable was the Community Periodontal Index (CPI), to investigate periodontal disease risks. Smoking was the independent variable and was divided into three groups. The chi-squared test and multivariable logistic regression analyses were used in this study. Current smokers had a higher risk of periodontal disease than non-smokers (males OR: 1.78, 95% CIs = 1.43–2.23, females OR: 1.44, 95% CIs = 1.04–1.99). Age, educational level, and dental checkups affected periodontal disease. Men with a higher number of pack years had a higher risk of periodontal disease than non-smokers (OR: 1.84, 95% CIs = 1.38–2.47). Men who quit smoking for less than five years had a higher risk of periodontal disease than non-smokers but lower than current smokers (current OR: 1.78, 95% CIs = 1.43–2.23, ex OR: 1.42, 95% CIs = 1.04–1.96). Those who had quit smoking for less than five years had a higher risk of periodontal disease than non-smokers but lower than current smokers (males OR: 1.42, 95% CIs = 1.04–1.96, females OR: 1.11, 95% CIs = 1.71–1.74). It is necessary to motivate smokers by educating them on the importance of early smoking cessation.
Keywords: periodontal health, periodontal disease, smoking, Community Periodontal Index (CPI)
1. Introduction
Smoking is one of the biggest threats to public health [1]. According to the World Health Organization, more than 8 million people have been killed, including approximately 1.2 million deaths from exposure to secondhand smoke [2]. Moreover, since tobacco has more than 7000 toxic chemicals [3], smoking is associated with numerous preventable chronic diseases [4]. In Korea, the smoking rate has been decreasing; however, as of 2018, the prevalence of daily smoking among men in Korea reached 30.5%, the third-highest rate among the Organization for Economic Co-operation and Development (OECD) members [5]. The authorities have made intensive efforts to eliminate tobacco use by implementing strong and effective tobacco control policies and measures, such as cigarette tax hikes and media campaigns [4,6].
The association between smoking and various diseases, including major causes of death, has been well-established. A cohort study in the US reported that smokers had a higher risk of developing bladder cancer and pancreatic cancer than non-smokers [7]. Another study found that smokers were more likely to have elevated levels of blood insulin and triglycerides compared to non-smokers [8,9].
Smoking can negatively impact the oral cavity, particularly in non-inflammatory oral diseases [10]. Harmful substances in tobacco products, such as nicotine, can harm the gingival tissue, decrease blood flow to the gums, and compromise the immune system [11]. Tobacco use can increase susceptibility to oral infections, stain teeth, cause dryness in the mouth, and delay the healing of oral wounds [12].
Periodontal diseases are considered to be chronic destructive inflammatory diseases [13]. They are characterized by the destruction of the periodontal tissue, loss of adhesion to connective tissues, loss of alveolar bone, and the formation of pathological sacs around the teeth [14,15,16]. In addition, poor periodontal health is associated with systemic diseases, such as cancer, heart disease, and diabetes; therefore, management is important [17,18,19]. Previous studies have shown that smoking is associated with poor periodontal health, even among young adults [20]. Another study in Korea revealed that quitting smoking within a decade could potentially improve periodontal health for former smokers [21]. A study in the US, which used large-scale data, concluded that smoking is a significant risk factor for periodontitis and may account for more than 50% of periodontitis in adults [22].
While previous studies have examined the association between smoking and periodontal diseases, additional evidence is needed to encourage healthy habits that promote smoking cessation. This study aimed to investigate the relationship between smoking and the risk of periodontal diseases in Korean adults, using a nationwide cross-sectional survey with a large sample size. Furthermore, this study aimed to provide more robust evidence for the importance of early smoking cessation by analyzing the relationship between smoking cessation in five-year intervals, which was more detailed than in previous studies.
2. Materials and Methods
2.1. Data
The data for this study were obtained from the 2016–2018 Korea National Health and Nutrition Examination Survey (KNHANES) and used a separate raw dataset (HNYN_OE). The KNHANES has been conducted by the Korea Disease Control and Prevention Agency (KDCA) since 1998 to investigate national statistics through a survey of the health level, health-related behavior, and nutritional status of 10,000 Koreans annually. The KDCA Research Ethics Review Board approved the data collection protocols for the KNHANES. The data are available for download from the KDCA website (https://knhanes.kdca.go.kr/knhanes/sub03/sub03_02_05.do, accessed on 1 January 2023). Thus, this study did not need extra approval from the ethics review board. The KNHANES is a self-reported survey using a stratified, two-stage, clustered sampling design conducted annually for South Koreans of all ages, divided into three age groups: (children: 1–11 years old, adolescents: 12–18 years old, and adults: 19 years or older).
2.2. Study Population
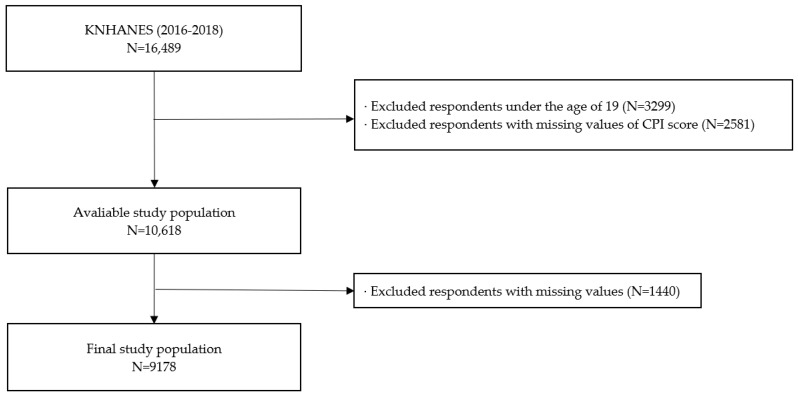
The total number of participants who completed the health examination survey for KNHANES 2016–2018 was 16,489 (7485 males and 9004 females). The exclusion criteria consisted of three categories: (a) under 19 years of age (N = 3299), (b) unable to perform oral examination due to tooth loss (N = 2581), and (c) missing values in health assessment or survey (N = 1440). The final study population was 9178, with 4161 men and 5017 women (Figure 1).
Figure 1.
Flowchart of the study participants displaying the inclusion and exclusion criteria.; CPI, Community Periodontal Index; KNHANES, Korea National Health and Nutrition Examination Survey.
2.3. Variables
The dependent variable in this study was the Community Periodontal Index (CPI), used to measure the risk of periodontal disease. The oral health examinations were conducted by public health dentists and local public health dentists at the city and provincial levels under the supervision of the Korea Disease Control and Prevention Agency (KDCA). The risk to periodontal health was assessed by dividing the upper and lower jaws into three sections and recording the highest CPI score for each section. The CPI score was based on periodontal pocket depth, calculus attachment, and gingival bleeding measurements. The scores ranged from 0 to 4, with 0 indicating healthy, 1 indicating bleeding, 2 indicating dental calculus, 3 indicating a superficial periodontal pocket of 4–5 mm, and 4 indicating a deep periodontal pocket of 6 mm or more. Using the sum of the CPI scores, we assessed the risk of periodontal disease as the outcome variable.
The independent variable was the smoking status, classified into three groups: non-smokers, ex-smokers, and current smokers. Smoking status was based on the question, “Do you currently smoke cigarettes?”. We also used pack years and smoking cessation status as variables in the subgroup analysis. Pack years indicate the number of cigarettes a person has smoked in their lifetime, calculated by multiplying the total number of cigarettes smoked per day by the total number of years a person smoked.
The covariate variables were controlled for, as potential confounding factors. These included socioeconomic factors, such as sex, age, household income, and region, and factors related to health behaviors, such as current drinking status and physical activity. Oral health habits were also included as covariates. Teeth brushing frequency was investigated, based on the number of times teeth were brushed during the previous day, while dental checkup status was surveyed based on the question, “Did you have a dental checkup in the past 12 months?”.
2.4. Statistical Analysis
A chi-squared test was conducted to explore the general characteristics of the study population. The general characteristics of the final study population were represented as frequency and percentage. To assess the relationship between smoking and periodontal disease using the sum of the CPI scores in adults, we used multivariable logistic regression analysis with covariate adjustment. Subgroup analyses were performed to evaluate the relationship between pack years, smoking cessation status, and periodontal disease. All the results were presented as odds ratios (ORs) and 95% confidence intervals (CIs). The analyses were performed using stratified sampling variables. All the estimates were estimated using weighted variables to generalize the data. SAS version 9.4 software (SAS Institute, Cary, NC, USA) was used for all the statistical analyses. Statistical significance was determined as a two-sided p-value of <0.05.
3. Results
Table 1 summarizes the characteristics of the study population, classified according to sex. Of the 9178 participants, 4161 were male (45.3%), and 5017 were female (54.7%). A total of 3042 (73.1%) males and 3143 (62.6%) females had periodontal disease risks, as expressed by the CPI. Among the males, 1484 (35.7%) were current smokers, 1623 (39.0%) were ex-smokers, and 1054 (25.3%) were non-smokers. Among the females, 282 (5.6%) were current smokers, 344 (6.9%) were ex-smokers, and 4391 (87.5%) were non-smokers.
Table 1.
General characteristics of the study population.
| Variables | Community Periodontal Index (CPI) | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Male | Female | ||||||||||||||
| Total | No | Yes | p-Value | Total | No | Yes | p-Value | ||||||||
| N | % | N | % | N | % | N | % | N | % | N | % | ||||
| Total (N = 9178) | 4161 | 45.3 | 1119 | 26.9 | 3042 | 73.1 | 5017 | 54.7 | 1874 | 37.4 | 3143 | 62.6 | |||
| Smoking Behavior | <0.0001 | 0.0130 | |||||||||||||
| Non-smoker | 1054 | 25.3 | 376 | 35.7 | 678 | 64.3 | 4391 | 87.5 | 1668 | 38.0 | 2723 | 62.0 | |||
| Ex-smoker | 1623 | 39.0 | 419 | 25.8 | 1204 | 74.2 | 344 | 6.9 | 123 | 35.8 | 221 | 64.2 | |||
| Current smoker | 1484 | 35.7 | 324 | 21.8 | 1160 | 78.2 | 282 | 5.6 | 83 | 29.4 | 199 | 70.6 | |||
| Age | <0.0001 | <0.0001 | |||||||||||||
| 20–29 | 645 | 15.5 | 276 | 42.8 | 369 | 57.2 | 688 | 13.7 | 361 | 52.5 | 327 | 47.5 | |||
| 30–39 | 800 | 19.2 | 250 | 31.3 | 550 | 68.8 | 1007 | 20.1 | 453 | 45.0 | 554 | 55.0 | |||
| 40–49 | 936 | 22.5 | 238 | 25.4 | 698 | 74.6 | 1215 | 24.2 | 447 | 36.8 | 768 | 63.2 | |||
| 50–59 | 810 | 19.5 | 175 | 21.6 | 635 | 78.4 | 1044 | 20.8 | 319 | 30.6 | 725 | 69.4 | |||
| 60–69 | 585 | 14.1 | 114 | 19.5 | 471 | 80.5 | 718 | 14.3 | 202 | 28.1 | 516 | 71.9 | |||
| ≥70 | 385 | 9.3 | 66 | 17.1 | 319 | 82.9 | 345 | 6.9 | 92 | 26.7 | 253 | 73.3 | |||
| Marital status | <0.0001 | <0.0001 | |||||||||||||
| Married | 2916 | 70.1 | 683 | 23.4 | 2233 | 76.6 | 3507 | 69.9 | 1234 | 35.2 | 2273 | 64.8 | |||
| Divorced | 146 | 3.5 | 30 | 20.5 | 116 | 79.5 | 302 | 6.0 | 80 | 26.5 | 222 | 73.5 | |||
| Single | 1099 | 26.4 | 406 | 36.9 | 693 | 63.1 | 1208 | 24.1 | 560 | 46.4 | 648 | 53.6 | |||
| Educational level | <0.0001 | <0.0001 | |||||||||||||
| Middle school | 673 | 16.2 | 85 | 12.6 | 588 | 87.4 | 1057 | 21.1 | 250 | 23.7 | 807 | 76.3 | |||
| High school | 1498 | 36.0 | 416 | 27.8 | 1082 | 72.2 | 1767 | 35.2 | 652 | 36.9 | 1115 | 63.1 | |||
| College | 1990 | 47.8 | 618 | 31.1 | 1372 | 68.9 | 2193 | 43.7 | 972 | 44.3 | 1221 | 55.7 | |||
| Household income | <0.0001 | <0.0001 | |||||||||||||
| Low | 477 | 11.5 | 93 | 19.5 | 384 | 80.5 | 632 | 12.6 | 179 | 28.3 | 453 | 71.7 | |||
| Mid-low | 939 | 22.6 | 230 | 24.5 | 709 | 75.5 | 1239 | 24.7 | 449 | 36.2 | 790 | 63.8 | |||
| Mid-high | 1280 | 30.8 | 345 | 27.0 | 935 | 73.0 | 1526 | 30.4 | 592 | 38.8 | 934 | 61.2 | |||
| High | 1465 | 35.2 | 451 | 30.8 | 1014 | 69.2 | 1620 | 32.3 | 654 | 40.4 | 966 | 59.6 | |||
| Region | <0.0001 | <0.0001 | |||||||||||||
| Metropolitan | 1973 | 47.4 | 533 | 27.0 | 1440 | 73.0 | 2437 | 48.6 | 927 | 38.0 | 1510 | 62.0 | |||
| Urban | 1556 | 37.4 | 479 | 30.8 | 1077 | 69.2 | 1891 | 37.7 | 798 | 42.2 | 1093 | 57.8 | |||
| Rural | 632 | 15.2 | 107 | 16.9 | 525 | 83.1 | 689 | 13.7 | 149 | 21.6 | 540 | 78.4 | |||
| Perceived stress level | 0.2388 | 0.1054 | |||||||||||||
| Low | 636 | 15.3 | 150 | 23.6 | 486 | 76.4 | 663 | 13.2 | 234 | 35.3 | 429 | 64.7 | |||
| Mid-low | 2433 | 58.5 | 667 | 27.4 | 1766 | 72.6 | 2858 | 57.0 | 1059 | 37.1 | 1799 | 62.9 | |||
| Mid-high | 934 | 22.4 | 259 | 27.7 | 675 | 72.3 | 1214 | 24.2 | 485 | 40.0 | 729 | 60.0 | |||
| High | 158 | 3.8 | 43 | 27.2 | 115 | 72.8 | 282 | 5.6 | 96 | 34.0 | 186 | 66.0 | |||
| Occupational categories | <0.0001 | <0.0001 | |||||||||||||
| White | 1433 | 34.4 | 463 | 32.3 | 970 | 67.7 | 1419 | 28.3 | 633 | 44.6 | 786 | 55.4 | |||
| Pink | 495 | 11.9 | 128 | 25.9 | 367 | 74.1 | 846 | 16.9 | 284 | 33.6 | 562 | 66.4 | |||
| Blue | 1309 | 31.5 | 229 | 17.5 | 1080 | 82.5 | 657 | 13.1 | 174 | 26.5 | 483 | 73.5 | |||
| Inoccupation | 924 | 22.2 | 299 | 32.4 | 625 | 67.6 | 2095 | 41.8 | 783 | 37.4 | 1312 | 62.6 | |||
| Current drinking status | <0.0001 | 0.7788 | |||||||||||||
| Never | 1019 | 24.5 | 274 | 26.9 | 745 | 73.1 | 2457 | 49.0 | 914 | 37.2 | 1543 | 62.8 | |||
| Monthly | 1640 | 39.4 | 515 | 31.4 | 1125 | 68.6 | 1847 | 36.8 | 700 | 37.9 | 1147 | 62.1 | |||
| Weekly | 1502 | 36.1 | 330 | 22.0 | 1172 | 78.0 | 713 | 14.2 | 260 | 36.5 | 453 | 63.5 | |||
| Physical activity | <0.0001 | 0.0002 | |||||||||||||
| Adequate | 2077 | 49.9 | 614 | 29.6 | 1463 | 70.4 | 2210 | 44.1 | 888 | 40.2 | 1322 | 59.8 | |||
| Inadequate | 2084 | 50.1 | 505 | 24.2 | 1579 | 75.8 | 2807 | 55.9 | 986 | 35.1 | 1821 | 64.9 | |||
| Teeth Brushing Frequency | <0.0001 | <0.0001 | |||||||||||||
| ≤1 | 464 | 11.2 | 102 | 22.0 | 362 | 78.0 | 164 | 3.3 | 43 | 26.2 | 121 | 73.8 | |||
| 2 | 1694 | 40.7 | 399 | 23.6 | 1295 | 76.4 | 1779 | 35.5 | 619 | 34.8 | 1160 | 65.2 | |||
| ≥3 | 2003 | 48.1 | 618 | 30.9 | 1385 | 69.1 | 3074 | 61.3 | 1212 | 39.4 | 1862 | 60.6 | |||
| Dental checkup status | <0.0001 | <0.0001 | |||||||||||||
| No | 2616 | 62.9 | 609 | 23.3 | 2007 | 76.7 | 3016 | 60.1 | 1004 | 33.3 | 2012 | 77.7 | |||
| Yes | 1545 | 37.1 | 510 | 33.0 | 1035 | 67.0 | 2001 | 39.9 | 870 | 24.0 | 1131 | 76.0 | |||
Table 2 presents the multivariate logistic regression analysis results that explore the association between smoking and periodontal disease while adjusting for covariates. The smokers had a higher risk of periodontal disease than the non-smokers. While the ex-smokers were statistically insignificant, the current smokers were significant for males (OR: 1.78, 95% CIs = 1.43–2.23) and females (OR: 1.44, 95% CIs = 1.04–1.99). As age increased, the participants showed an elevated risk of periodontal disease. The participants with a middle school education or lower had a higher risk of periodontal disease than those with a college education (males OR: 1.63, 95% CIs = 1.15–2.23, females OR: 1.59, 95% CIs = 1.18–2.14). The individuals who did not receive dental checkups were likelier to have periodontal diseases (males OR: 1.62, 95% CIs = 1.36–1.93, females OR: 1.60, 95% CIs = 1.39–1.84).
Table 2.
Results of factors associated with smoking and community periodontal index.
| Variables | Community Periodontal Index (CPI) | ||||
|---|---|---|---|---|---|
| Male | Female | ||||
| OR | 95% CIs | OR | 95% CIs | ||
| Smoking Behavior | |||||
| Non-smoker | 1.00 | 1.00 | |||
| Ex-smoker | 1.20 | (0.95–1.50) | 1.19 | (0.89–1.60) | |
| Current smoker | 1.78 * | (1.43–2.23) | 1.44 * | (1.04–1.99) | |
| Age | |||||
| 20–29 | 1.00 | 1.00 | |||
| 30–39 | 1.41 | (0.99–2.00) | 1.18 | (0.91–1.54) | |
| 40–49 | 1.77 * | (1.24–2.51) | 1.63 * | (1.24–2.15) | |
| 50–59 | 1.83 * | (1.24–2.69) | 1.93 * | (1.44–2.57) | |
| 60–69 | 2.36 * | (1.53–3.64) | 1.79 * | (1.27–2.52) | |
| ≥70 | 2.87 * | (1.70–4.83) | 1.88 * | (1.20–2.96) | |
| Marital status | |||||
| Married | 1.00 | 1.00 | |||
| Divorced | 0.77 | (0.51–1.17) | 1.36 | (0.97–1.92) | |
| Single | 0.86 | (0.64–1.14) | 0.79 * | (0.63–0.98) | |
| Educational level | |||||
| Middle school | 1.63 * | (1.15–2.23) | 1.59 * | (1.18–2.14) | |
| High school | 1.10 | (0.89–1.35) | 1.10 | (0.92–1.31) | |
| College | 1.00 | 1.00 | |||
| Household income | |||||
| Low | 1.47 * | (1.01–2.15) | 1.03 | (0.78–1.37) | |
| Mid-low | 1.15 | (0.90–1.46) | 0.99 | (0.82–1.21) | |
| Mid-high | 1.05 | (0.86–1.28) | 0.93 | (0.78–1.12) | |
| High | 1.00 | 1.00 | |||
| Region | |||||
| Metropolitan | 1.00 | 1.00 | |||
| Urban | 0.77 | (0.59–1.02) | 0.73 | (0.57–0.92) | |
| Rural | 1.39 | (0.91–2.10) | 1.78 * | (1.19–2.67) | |
| Perceived stress level | |||||
| Low | 1.00 | 1.00 | |||
| Mid-low | 0.89 | (0.69–1.14) | 1.02 | (0.83–1.26) | |
| Mid-high | 0.93 | (0.68–1.27) | 0.98 | (0.77–1.25) | |
| High | 0.94 | (0.59–1.50) | 1.25 | (0.881.77) | |
| Occupational categories | |||||
| White | 1.00 | 1.00 | |||
| Pink | 1.13 | (0.85–1.52) | 1.17 | (0.92–1.48) | |
| Blue | 1.48 * | (1.17–1.89) | 1.06 | (0.80–1.41) | |
| Inoccupation | 0.72 * | (0.57–0.91) | 1.00 | (0.83–1.20) | |
| Current drinking status | |||||
| Never | 1.00 | 1.00 | |||
| Monthly | 0.92 | (0.74–1.14) | 1.24 * | (1.07–1.43) | |
| Weekly | 1.21 | (0.95–1.55) | 1.17 | (0.95–1.45) | |
| Physical activity | |||||
| Adequate | 1.00 | 1.00 | |||
| Inadequate | 1.03 | (0.87–1.22) | 1.04 | (0.90–1.21) | |
| Teeth Brushing Frequency | |||||
| ≤1 | 1.17 | (0.87–1.57) | 1.57 * | (1.03–2.39) | |
| 2 | 1.21 * | (1.02–1.43) | 1.02 | (0.88–1.17) | |
| ≥3 | 1.00 | 1.00 | |||
| Dental checkup status | |||||
| No | 1.62 * | (1.36–1.93) | 1.60 * | (1.39–1.84) | |
| Yes | 1.00 | 1.00 | |||
* p-value < 0.05.
Table 3 presents the results of the subgroup analysis for the independent variables stratified by smoking behavior. Most of the ex-smokers did not have significant results. The observed results were more significant in the males than in the females. The risk of periodontal disease generally increased with age in men who are current smokers but was not statistically significant in their 50s. The current smokers had a higher risk of periodontal disease in all education levels, and the risk was highest for those with middle school education or lower (OR: 3.15; 95% CIs = 1.37–7.21). The current smokers had a risk of periodontal disease, regardless of their physical activity status (male, adequate: OR = 1.83, 95% CIs = 1.35–2.50; inadequate: OR = 1.77, 95% CIs = 1.30–2.42). Similarly, regardless of whether they received regular dental checkups, current smokers had a higher risk of periodontal disease (male, checkups: OR = 1.90, 95% CIs = 1.40–2.59; no checkups: OR = 1.73, 95% CIs = 1.29–2.33).
Table 3.
Results of subgroup analysis stratified by independent variables.
| Variables | Community Periodontal Index (CPI) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Male | Female | |||||||||
| Non | Ex-Smoker | Current Smoker | Non | Ex-Smoker | Current Smoker | |||||
| OR | OR | 95% CIs | OR | 95% CIs | OR | OR | 95% CIs | OR | 95% CIs | |
| Age | ||||||||||
| 20–29 | 1.00 | 1.10 | (0.63–1.92) | 1.82 * | (1.16–2.86) | 1.00 | 0.83 | (0.45–1.55) | 0.87 | (0.41–1.83) |
| 30–39 | 1.00 | 1.40 | (0.85–2.31) | 1.78 * | (1.16–2.73) | 1.00 | 1.23 | (0.76–2.00) | 2.73 * | (1.45–5.17) |
| 40–49 | 1.00 | 1.31 | (0.84–2.04) | 2.21 * | (1.37–3.29) | 1.00 | 1.34 | (0.74–2.42) | 1.37 | (0.72–2.61) |
| 50–59 | 1.00 | 0.88 | (0.49–1.57) | 1.34 | (0.72–2.48) | 1.00 | 1.88 | (0.72–4.92) | 1.62 | (0.64–4.10) |
| 60–69 | 1.00 | 1.38 | (0.75–2.54) | 2.57 * | (1.10–6.03) | 1.00 | 0.98 | (0.32–1.71) | 1.76 | (0.36–8.70) |
| ≥70 | 1.00 | 1.59 | (0.68–3.71) | 2.71 | (0.68–10.71) | 1.00 | 3.56 | (0.45–28.27) | - | - |
| Marital status | ||||||||||
| Married | 1.00 | 1.26 | (0.96–1.65) | 1.91 * | (1.44–2.53) | 1.00 | 1.18 | (0.81–1.72) | 1.51 | (0.81–1.72) |
| Divorced | 1.00 | 0.07 * | (0.01–0.89) | 0.06 | (0.00–1.32) | 1.00 | 0.90 | (0.34–2.35) | 4.44 * | (1.49–13.27) |
| Single | 1.00 | 1.06 | (0.65–1.72) | 1.70 * | (1.18–2.43) | 1.00 | 1.14 | (0.70–1.86) | 1.07 | (0.61–1.87) |
| Educational level | ||||||||||
| Middle school | 1.00 | 1.70 | (0.87–3.30) | 3.15 * | (1.37–7.21) | 1.00 | 1.12 | (0.43–2.90) | 11.40 * | (2.93–44.45) |
| High school | 1.00 | 1.21 | (0.83–1.77) | 1.67 * | (1.14–2.44) | 1.00 | 1.14 | (0.68–1.93) | 1.25 | (0.77–2.03) |
| College | 1.00 | 1.14 | (0.85–1.52) | 1.76 * | (1.34–2.32) | 1.00 | 1.19 | (0.80–1.77) | 1.13 | (0.66–1.93) |
| Household income | ||||||||||
| Low | 1.00 | 0.78 | (0.37–1.67) | 1.95 | (0.84–4.55) | 1.00 | 1.28 | (0.44–3.72) | 2.73 | (0.95–7.87) |
| Mid-low | 1.00 | 1.38 * | (1.47–4.45) | 2.56 * | (1.47–4.47) | 1.00 | 1.45 | (0.83–2.54) | 1.61 | (0.85–3.05) |
| Mid-high | 1.00 | 1.17 | (0.78–1.74) | 1.82 * | (1.22–2.71) | 1.00 | 1.16 | (0.72–1.88) | 1.27 | (0.72–2.27) |
| High | 1.00 | 1.19 | (0.85–1.68) | 1.51 * | (1.09–2.11) | 1.00 | 1.04 | (0.61–1.77) | 1.14 | (0.60–2.17) |
| Region | ||||||||||
| Metropolitan | 1.00 | 1.39 * | (1.00–1.95) | 2.16 * | (1.57–2.95) | 1.00 | 1.19 | (0.80–1.77) | 0.95 | (0.59–1.55) |
| Urban | 1.00 | 1.22 | (0.88–1.70) | 1.75 * | (1.24–2.47) | 1.00 | 1.18 | (0.76–1.86) | 2.24 * | (1.32–3.80) |
| Rural | 1.00 | 0.43 | (0.19–1.00) | 0.74 | (0.35–1.59) | 1.00 | 1.19 | (0.32–4.40) | 1.48 | (0.67–3.29) |
| Perceived stress level | ||||||||||
| Low | 1.00 | 1.41 | (0.82–2.42) | 4.02 * | (1.96–8.27) | 1.00 | 1.52 | (0.59–3.91) | 1.96 | (0.63–6.12) |
| Mid-low | 1.00 | 1.16 | (0.87–1.54) | 1.72 * | (1.29–2.31) | 1.00 | 1.17 | (0.78–1.76) | 1.48 | (0.93–2.36) |
| Mid-high | 1.00 | 1.28 | (0.79–2.06) | 1.87 * | (1.18–2.96) | 1.00 | 1.18 | (0.64–2.19) | 1.25 | (0.70–2.23) |
| High | 1.00 | 0.55 | (0.13–2.37) | 0.75 | (0.23–2.43) | 1.00 | 0.93 | (0.40–2.16) | 1.29 | (0.51–3.27) |
| Occupational categories | ||||||||||
| White | 1.00 | 1.19 | (0.86–1.63) | 1.68 * | (1.21–2.33) | 1.00 | 1.21 | (0.76–1.93) | 1.23 | (0.64–2.38) |
| Pink | 1.00 | 0.72 | (0.33–1.54) | 1.29 | (0.71–2.36) | 1.00 | 0.95 | (0.46–1.97) | 1.57 | (0.79–3.12) |
| Blue | 1.00 | 1.08 | (0.71–1.65) | 1.80 * | (1.16–2.80) | 1.00 | 0.65 | (0.23–1.82) | 2.92 * | (1.07–1.82) |
| Inoccupation | 1.00 | 1.57 | (0.99–2.48) | 2.10 * | (1.28–3.43) | 1.00 | 1.44 | (0.91–2.28) | 1.26 | (0.75–2.28) |
| Current drinking status | ||||||||||
| Never | 1.00 | 1.48 | (0.98–2.23) | 1.90 * | (1.23–2.94) | 1.00 | 1.22 | (0.74–2.01) | 1.84 * | (1.01–3.38) |
| Monthly | 1.00 | 1.27 | (0.92–1.76) | 1.88 * | (1.37–2.57) | 1.00 | 1.32 | (0.87–1.99) | 1.12 | (0.67–1.89) |
| Weekly | 1.00 | 0.87 | (0.56–1.36) | 1.45 | (0.95–2.21) | 1.00 | 1.09 | (0.59–2.11) | 1.59 | (0.95–2.66) |
| Physical activity | ||||||||||
| Adequate | 1.00 | 1.12 | (0.83–1.50) | 1.83 * | (1.35–2.50) | 1.00 | 0.97 | (0.66–1.43) | 1.11 | (0.68–1.81) |
| Inadequate | 1.00 | 1.32 | (0.96–1.82) | 1.77 * | (1.30–2.42) | 1.00 | 1.43 | (0.96–2.21) | 1.88 * | (1.26–2.79) |
| Teeth Brushing Frequency | ||||||||||
| ≤1 | 1.00 | 1.12 | (0.52–2.39) | 1.32 | (0.62–2.84) | 1.00 | 4.59 | (0.64–32.98) | 3.18 | (0.59–17.18) |
| 2 | 1.00 | 1.52 * | (1.04–2.22) | 1.41 * | (1.48–3.05) | 1.00 | 1.26 | (0.83–1.92) | 1.21 | (0.71–2.05) |
| ≥3 | 1.00 | 1.05 | (0.77–1.42) | 1.70 * | (1.26–2.29) | 1.00 | 1.16 | (0.81–1.65) | 1.65 * | (1.08–2.51) |
| Dental checkup status | ||||||||||
| No | 1.00 | 1.06 | (0.79–1.43) | 1.73 * | (1.29–2.33) | 1.00 | 1.05 | (0.74–1.48) | 1.95 * | (1.29–2.95) |
| Yes | 1.00 | 1.39 * | (1.01–1.91) | 1.90 * | (1.40–2.59) | 1.00 | 1.57 * | (1.02–2.42) | 0.82 | (0.48–1.38) |
* p-value < 0.05.
The results of the subgroup analysis, which were stratified by pack years and smoking cessation, are presented in Table 4. The males showed a statistically significant positive association. Those with a higher number of pack years had a higher risk of periodontal diseases than the non-smokers (over 20 pack years OR: 1.84, 95% CIs = 1.38–2.47). Those who had quit smoking for less than five years had a higher risk of periodontal disease than the non-smokers but lower than the current smokers (males OR: 1.42, 95% CIs = 1.04–1.96; females OR: 1.11, 95% CIs = 1.71–1.74).
Table 4.
Results of subgroup analysis stratified by pack years and smoking cessation.
| Variables | Community Periodontal Index (CPI) | ||||
|---|---|---|---|---|---|
| Male | Female | ||||
| OR | 95% CIs | OR | 95% CIs | ||
| Pack-Years | |||||
| Non-smoker | 1.00 | 1.00 | |||
| Pack Years < 5 | 1.11 | (0.87–1.42) | 1.16 | (0.90–1.50) | |
| 5 ≤ Pack Years < 10 | 1.75 * | (1.34–2.27) | 1.71 | (0.94–3.13) | |
| 10 ≤ Pack Years < 15 | 1.47 * | (1.10–2.27) | 1.47 | (0.72–3.00) | |
| 15 ≤ Pack Years < 20 | 1.80 * | (1.27–2.56) | 1.96 | (0.62–6.18) | |
| 20 ≤ Pack Years | 1.84 * | (1.38–2.47) | 2.69 | (0.90–8.05) | |
| Smoking Cessation Status | |||||
| Non-smoker | 1.00 | 1.00 | |||
| Ex-smoker (5 yr. > Cessation) | 1.42 * | (1.04–1.96) | 1.11 * | (1.71–1.74) | |
| Ex-smoker (5 yr. ≤ Cessation < 10 yr.) | 1.69 * | (1.16–2.47) | 1.07 | (0.61–1.87) | |
| Ex-smoker (10 yr. ≤ Cessation) | 0.93 | (0.71–1.21) | 1.32 | (0.81–2.14) | |
* p-value < 0.05.
4. Discussion
Despite the reduction in smoking prevalence over the past 30 years, the total number of smokers has increased from 0.99 billion in 1990 to 1.14 billion in 2019 worldwide, due to population growth [23]. The American Academy of Periodontology has pointed out that smoking negatively impacts the healing and treatment of periodontitis [24]. The purpose of the study was two main issues. First, we used a nationwide survey with a large sample size to investigate the association between smoking and periodontal disease. Second, we attempted to support the importance of early smoking cessation by analyzing the relationship between smoking cessation in five-year intervals compared to the previous studies using ten-year intervals.
The mechanisms underlying the association between smoking and periodontal disease were the following. Smoking stimulates the establishment of pathogenic microflora, diminishes the immune host response, and elevates the release of inflammatory mediators [14,15,16,25]. As smokers are more likely to absorb pathogenic microorganisms than non-smokers, previous studies have reported an increase in particular pathogens in smokers, such as Actinobacillus actinomycetemcomitans and Bacteroides forsythus, although the pathogen levels may have varied, based on the methods used in the studies [14,26,27]. Smoking can affect host inflammatory and immune responses, such as the immunosuppressive effects of macrophages on cell-mediated immune responses, inhibition of human periodontal ligament fibroblast migration, and repression of alkaline phosphatase production by nicotine [28,29]. As with this mechanism, Table 2 shows current smokers had a higher risk of periodontal diseases than non-smokers. It supports previous studies’ results that smoking is a risk factor for oral health, even among young smokers [20]. Additionally, the results in Table 2 are in the same vein as previous studies, showing that smoking significantly influences periodontitis, using a large sample in the US [22].
Notable points in Table 2 were the results of age, education level, and dental checkup status variables. In the case of age, it was consistent with the results of previous studies that the prevalence of periodontal disease tends to increase as the age of participants increases. Previous studies in Brazil and India have reported that age increases affect the severity and prevalence of periodontal disease, regardless of gender [30,31]. The education level affected periodontal health. Middle school or lower education participants had a higher risk of periodontal disease than those with a college or higher education. As some studies have reported similar findings [22,32], education progressively decreases the risk of periodontal diseases. This finding implies that education regarding periodontal health is important. There was a higher risk of periodontal disease in people who did not undergo oral examinations, which supports previous studies that those who regularly underwent oral examinations had a lower risk of periodontal disease than those who did not [33,34].
In Table 3, male smokers in their 50s were not statistically significant. This counterintuitive finding can be explained by aging, which affects tooth loss [4,35]. Even if good physical activity habits and dental checkups were regularly undertaken, the current smokers had a higher risk of periodontal disease than the non-smokers. This result could explain that current smokers cannot avoid the risk of periodontal disease, even if they have good health habits.
As shown in Table 4, the men with high pack years had a higher risk of periodontal disease. However, the women showed statistically insignificant results. The results can be explained in the WHO Framework Convention on Tobacco Control (WHO FCTC) context. The WHO FCTC emphasized the need to consider gender when developing tobacco control strategies, as perceptions of smoking habits related to gender continue to differ depending on social contexts and cultural norms. Specifically, in Confucian Asian countries, there is still a tendency for views on female smoking to be more conservative than those on male smoking [36]. The smoking rate of women is also increasing in Korea. However, considering the social context, the data on the female smoking rate collected by voluntary reporting may not be accurate, due to the opposing views of some female smokers.
We found that ex-smokers with relatively short smoking cessation periods had a lower risk of periodontal disease than current smokers. This result can be compared to a previous study that reported the possibility of reversing the risk of periodontal disease if an individual quits smoking for ten years [15]. The sentence that smoking is harmful was too clear and simple, but the results of this study tried to support the importance of early smoking cessation. It could motivate smokers to quit smoking by revealing that people who quit smoking for a relatively short period, fewer than five years, have a lower risk of periodontal disease.
This study had several limitations. First, clarifying an inverse causal association was difficult since it was a cross-sectional study. Second, the KNHANES data were collected through a self-reported survey. The data on smoking behavior, health habits, and socioeconomic variables may not be accurately estimated. There was a possibility of recall bias. Third, it was impossible to identify the type of smoking, such as whether the participants used conventional cigarettes, e-cigarettes, or both. In addition, we could not use biological indicators, such as urine cotinine, in the subjects. Therefore, further studies are needed, considering these limitations.
Despite these limitations, our study has several strengths. The main strength of this study is the use of nationally representative, large, and high-quality data. KNHANES was conducted using a random cluster design, which can generalize the study’s results to the general population. Second, oral health examination datasets collected by public health doctors may effectively estimate periodontal disease risks. It was possible to estimate the risk of periodontal disease more precisely by using the CPI score through a doctor’s examination than by using the participant’s subjective oral symptom self-reported survey. Third, our study supported the importance of early smoking cessation. Compared to previous studies, we provided more proactive evidence for the importance of early smoking cessation.
5. Conclusions
This study demonstrated a strong association between smoking and periodontal disease in South Korean adults. Long-term smoking was closely related to poor periodontal health. The findings that even a relatively short period of smoking cessation, less than five years, had a positive impact on periodontal disease could be a powerful motivator for smokers. There is a need for effective tobacco control measures to reduce the prevalence of periodontitis. Tailored smoking cessation policies and educational interventions, highlighting the benefits of short-term smoking abstinence in reducing periodontal disease risk, could encourage current smokers to quit and ultimately improve public oral health.
Acknowledgments
The authors all agree with the manuscript and express appreciation to the professor and our colleagues from the Department of Public Health, Graduate School of Yonsei University, for their advice on our manuscript. The authors are responsible for the contents, including the data analysis. The statements in the study do not necessarily represent the official views of the Korea Disease Control and Prevention Agency.
Author Contributions
K.-Y.S. designed this study, performed the statistical analysis, and drafted and completed the manuscript. Y.S.J. (Yun Seo Jang), Y.S.J. (Ye Seul Jang) and N.N. contributed to the concept and design of the study and revised the manuscript. E.-C.P. conceived, designed, and directed this study. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
The KNHANES survey collection protocols were approved by the KDCA Research Ethics Review Board. This survey did not require extra approval from the ethics review board because the data were accessible for downloading on the website.
Informed Consent Statement
Patient consent was waived due to the use of open-source data.
Data Availability Statement
The dataset used in this study is publicly accessible (https://knhanes.kdca.go.kr/knhanes/sub03/sub03_02_05.do (accessed on 1 January 2023)).
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
The authors received no specific funding for this study.
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.WHO World Health Organization Fact Sheets of Tobacco. [(accessed on 11 November 2021)]. Available online: https://www.who.int/news-room/fact-sheets/detail/tobacco.
- 2.IHME Global Burden of Disease Results-GBD Estimate of Oral Health. [(accessed on 11 November 2022)]. Available online: https://vizhub.healthdata.org/gbd-results/
- 3.Varghese C., Troisi G., Schotte K., Prasad V.M., Claire S.M.S. World No Tobacco Day 2019 puts the spotlight on lung health. J. Thorac. Dis. 2019;11:2639–2642. doi: 10.21037/jtd.2019.06.38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Jeong W., Choi D.W., Kim Y.K., Lee H.J., Lee S.A., Park E.C., Jang S.I. Associations of electronic and conventional cigarette use with periodontal disease in South Korean adults. J. Periodontol. 2020;91:55–64. doi: 10.1002/JPER.19-0060. [DOI] [PubMed] [Google Scholar]
- 5.OECD Organisation for Economic Co-operation and Development-Health Statistics 2021. [(accessed on 16 January 2023)]. Available online: https://data.oecd.org/searchresults/?q=
- 6.Lee S., Kimm H., Yun J.E., Jee S.H. Public Health Challenges of Electronic Cigarettes in South Korea. J. Prev. Med. Public Health. 2011;44:235–241. doi: 10.3961/jpmph.2011.44.6.235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Inoue-Choi M., Hartge P., Liao L.M., Caporaso N., Freedman N.D. Association between long-term low-intensity cigarette smoking and incidence of smoking-related cancer in the national institutes of health-AARP cohort. Int. J. Cancer. 2017;142:271–280. doi: 10.1002/ijc.31059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Huh Y., Han K., Choi M.-J., Kim J.H., Kim S.M., Nam G.E. Association of Smoking Status With the Risk of Type 2 Diabetes Among Young Adults: A Nationwide Cohort Study in South Korea. Nicotine Tob. Res. 2022;24:1234–1240. doi: 10.1093/ntr/ntac044. [DOI] [PubMed] [Google Scholar]
- 9.Will J.C., Galuska D.A., Ford E.S., Mokdad A., Calle E.E. Cigarette smoking and diabetes mellitus: Evidence of a positive association from a large prospective cohort study. Int. J. Epidemiol. 2001;30:540–546. doi: 10.1093/ije/30.3.540. [DOI] [PubMed] [Google Scholar]
- 10.Johnson G.K., Guthmiller J.M. The impact of cigarette smoking on periodontal disease and treatment. Periodontology 2000. 2007;44:178–194. doi: 10.1111/j.1600-0757.2007.00212.x. [DOI] [PubMed] [Google Scholar]
- 11.Gajendra S., McIntosh S., Ghosh S. Effects of tobacco product use on oral health and the role of oral healthcare providers in cessation: A narrative review. Tob. Induc. Dis. 2023;21:12. doi: 10.18332/tid/157203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Agnihotri R., Gaur S. Implications of tobacco smoking on the oral health of older adults. Geriatr. Gerontol. Int. 2014;14:526–540. doi: 10.1111/ggi.12285. [DOI] [PubMed] [Google Scholar]
- 13.Hussein B.J., Khalil A.M., Atallah H.N., Ali N.A. Assessment of salivary interleukin-17 and vitamin d levels in smokers with severe periodontitis patients and healthy individuals. J. Pharm. Negat. Results. 2023;2:7147–7155. [Google Scholar]
- 14.Kinane D., Chestnutt I. Smoking and periodontal disease. Crit. Rev. Oral Biol. Med. 2000;11:356–365. doi: 10.1177/10454411000110030501. [DOI] [PubMed] [Google Scholar]
- 15.Fiorini T., Musskopf M.L., Oppermann R.V., Susin C. Is There a Positive Effect of Smoking Cessation on Periodontal Health? A Systematic Review. J. Periodontol. 2014;85:83–91. doi: 10.1902/jop.2013.130047. [DOI] [PubMed] [Google Scholar]
- 16.Leite F.R., Nascimento G.G., Scheutz F., López R. Effect of Smoking on Periodontitis: A Systematic Review and Meta-regression. Am. J. Prev. Med. 2018;54:831–841. doi: 10.1016/j.amepre.2018.02.014. [DOI] [PubMed] [Google Scholar]
- 17.Han D.-H., Lim S., Kim J.-B. The Association of Smoking and Diabetes With Periodontitis in a Korean Population. J. Periodontol. 2012;83:1397–1406. doi: 10.1902/jop.2012.110686. [DOI] [PubMed] [Google Scholar]
- 18.Hong M.-H. TRelationship between Oral Health Behavior, Chronic Disease and Periodontal Disease in Middle and Older Adults According to Gender. J. Korea Acad.-Ind. Coop. Soc. 2018;19:403–410. [Google Scholar]
- 19.Petrenya N., Hopstock L.A., Holde G.E., Oscarson N., Jönsson B. Relationship between periodontitis and risk of cardiovascular disease: Insights from the Tromsø Study. J. Periodontol. 2022;93:1353–1365. doi: 10.1002/JPER.22-0004. [DOI] [PubMed] [Google Scholar]
- 20.Machuca G., Rosales I., LaCalle J.R., Machuca C., Bullon P. Effect of Cigarette Smoking on Periodontal Status of Healthy Young Adults. J. Periodontol. 2000;71:73–78. doi: 10.1902/jop.2000.71.1.73. [DOI] [PubMed] [Google Scholar]
- 21.Jang A.-Y., Lee J.-K., Shin J., Lee H.-Y. Association between Smoking and Periodontal Disease in Korean Adults: The Fifth Korea National Health and Nutrition Examination Survey (2010 and 2012) Korean J. Fam. Med. 2016;37:117–122. doi: 10.4082/kjfm.2016.37.2.117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Tomar S.L., Asma S. Smoking-Attributable Periodontitis in the United States: Findings From NHANES III. J. Periodontol. 2000;71:743–751. doi: 10.1902/jop.2000.71.5.743. [DOI] [PubMed] [Google Scholar]
- 23.Reitsma M.B., Flor L.S., Mullany E.C., Gupta V., Hay S.I., Gakidou E. Spatial, temporal, and demographic patterns in prevalence of smoking tobacco use and initiation among young people in 204 countries and territories, 1990–2019. Lancet Public Health. 2021;6:e472–e481. doi: 10.1016/S2468-2667(21)00102-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Research S. Position paper: Tobacco use and the periodontal patient. J. Periodontol. 1999;70:1419–1427. doi: 10.1902/jop.1999.70.11.1419. [DOI] [PubMed] [Google Scholar]
- 25.Apatzidou D.A. The role of cigarette smoking in periodontal disease and treatment outcomes of dental implant therapy. Periodontology 2000. 2022;90:45–61. doi: 10.1111/prd.12449. [DOI] [PubMed] [Google Scholar]
- 26.Haffajee A., Cugini M., Dibart S., Smith C., Kent Jr R., Socransky S. The effect of SRP on the clinical and microbiological parameters of periodontal diseases. J. Clin. Periodontol. 1997;24:324–334. doi: 10.1111/j.1600-051X.1997.tb00765.x. [DOI] [PubMed] [Google Scholar]
- 27.Zambon J., Grossi S., Machtei E., Ho A., Dunford R., Genco R. Cigarette Smoking Increases the Risk for Subgingival Infection With Periodontal Pathogens. J. Periodontol. 1996;67:1050–1054. doi: 10.1902/jop.1996.67.10s.1050. [DOI] [PubMed] [Google Scholar]
- 28.Giannopoulou C., Geinoz A., Cimasoni G. Effects of nicotine on periodontal ligament fibroblasts in vitro. J. Clin. Periodontol. 1999;26:49–55. doi: 10.1034/j.1600-051X.1999.260109.x. [DOI] [PubMed] [Google Scholar]
- 29.Arcavi L., Benowitz N.L. Cigarette smoking and infection. Arch. Intern. Med. 2004;164:2206–2216. doi: 10.1001/archinte.164.20.2206. [DOI] [PubMed] [Google Scholar]
- 30.Selvaraj S., Naing N.N., Wan-Arfah N., Djearamane S., Wong L.S., Subramaniyan V., Fuloria N.K., Sekar M., Fuloria S., de Abreu M.H.N.G. Epidemiological Factors of Periodontal Disease Among South Indian Adults. J. Multidiscip. Healthc. 2022;15:1547–1557. doi: 10.2147/JMDH.S374480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Figueiredo A., Soares S., Lopes H., dos Santos J.N., Ramalho L.M.P., Cangussu M.C., Cury P.R. Destructive periodontal disease in adult Indians from Northeast Brazil: Cross-sectional study of prevalence and risk indicators. J. Clin. Periodontol. 2013;40:1001–1006. doi: 10.1111/jcpe.12147. [DOI] [PubMed] [Google Scholar]
- 32.Zini A., Sgan-Cohen H.D., Marcenes W. Socio-economic position, smoking, and plaque: A pathway to severe chronic periodontitis. J. Clin. Periodontol. 2010;38:229–235. doi: 10.1111/j.1600-051X.2010.01689.x. [DOI] [PubMed] [Google Scholar]
- 33.Lang W.P., Farghaly M.M., Ronis D.L. The relation of preventive dental behaviors to periodontal health status. J. Clin. Periodontol. 1994;21:194–198. doi: 10.1111/j.1600-051X.1994.tb00303.x. [DOI] [PubMed] [Google Scholar]
- 34.Takami Y., Nakagaki H., Morita I., Tsuboi S., Takami S., Suzuki N., Niwa H., Ogura Y. Blood Test Values and Community Periodontal Index Scores in Medical Checkup Recipients. J. Periodontol. 2003;74:1778–1784. doi: 10.1902/jop.2003.74.12.1778. [DOI] [PubMed] [Google Scholar]
- 35.Ji M.G. Relationship between adults’ smoking realities and periodontal disease-2009 Korea National Health and Nutrition Examination Survey data. J. Korea Inst. Electron. Commun. Sci. 2012;7:917–924. [Google Scholar]
- 36.FCTC WHO Framework Convention on Tobacco Control. [(accessed on 16 January 2023)]. Available online: https://fctc.who.int/publications/i/item/9241591013.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The dataset used in this study is publicly accessible (https://knhanes.kdca.go.kr/knhanes/sub03/sub03_02_05.do (accessed on 1 January 2023)).