Abstract
After decades of research and development concerning cancer treatment, cancer is still at large and very much a threat to the global human population. Cancer remedies have been sought from all possible directions, including chemicals, irradiation, nanomaterials, natural compounds, and the like. In this current review, we surveyed the milestones achieved by green tea catechins and what has been accomplished in cancer therapy. Specifically, we have assessed the synergistic anticarcinogenic effects when green tea catechins (GTCs) are combined with other antioxidant-rich natural compounds. Living in an age of inadequacies, combinatorial approaches are gaining momentum, and GTCs have progressed much, yet there are insufficiencies that can be improvised when combined with natural antioxidant compounds. This review highlights that there are not many reports in this specific area and encourages and recommends research attention in this direction. The antioxidant/prooxidant mechanisms of GTCs have also been highlighted. The current scenario and the future of such combinatorial approaches have been addressed, and the lacunae in this aspect have been discussed.
Keywords: green tea catechins, antioxidant, natural compounds, anticancer, mechanisms, apoptosis
1. Introduction
Cancer is a multifactorial disorder that typically arises through the influence of diverse genetic and environmental factors [1]. Cancer claimed nearly 10 million lives in 2020, the highest number of deaths through a specific disease [2]. One out of six mortalities has been statistically proven to be due to cancer. Furthermore, it is predicted that the number of global morbidities due to cancer by 2025 will number around 20 million [3,4]. Even with the advent of cutting-edge technologies for generating cancer models, genetic networks, and molecular and cellular interactions in cancer models revealing novel targets for cancer therapies [5], the disease is still at large. Despite the fact that enormous progress has been made in identifying drug targets and therapeutic molecules for cancer cell treatment, the drug resistance of cancer cells against the currently available anticancer drugs is a major bottleneck in cancer therapy [6]. In addition to that, cancer stem cells [7], insufficient bioavailability [8], and side effects of anticancer drugs [9] are also major setbacks in the progress of cancer treatments. These factors instigate the urge to explore alternative novel treatment strategies to overcome the hurdles of the present cancer therapies.
Molecules from a wide range of natural bioresources such as bacteria, fungi, plants, protozoa, and animals possess cancer therapeutic potential [10]. Among the natural products, Camptothecin and Taxol, which were initially isolated from the bark of Camptotheca acuminata and also from many fungal species, have been well-proven to be effective against many cancers and are under human clinical trials [11,12,13,14,15]. In addition to that, various other plant-derived compounds also exhibit anticancer activity, for instance, hematoxylin from the heartwood of Haematoxylon campechianum [16], Eucalyptin A from the fruits of Eucalyptus globulus [17], Pseudolaric acid B from the root bark of Pseudolarix kaempferi [18,19], Parthenolide, a sesquiterpene lactone, from the shoots of Tanacetum parthenium [20], exhibited anticancer activity by acting on different cancer targets [21].
Similarly, numerous natural compounds that exhibit anticancer potential have also been discovered in marine resources. Among these, the very first marine organism that gave a glimpse of an anticancer compound (arabino nucleosides and cytosine arabinose) was the Caribbean sponge, Cryptotethya crypta [20]. Despite natural compounds being constantly explored for anticancer potential from all sources and some being already approved as drug candidates for chemotherapy, the WHO specifically suggested that chemoprevention is the most effective therapy for various cancers, which include breast malignancies [22].
Therefore, screening of natural compounds with chemopreventive properties has been widely carried out. For example, ethanolic extracts of Arenosclera brasiliensis [23] and Haliclona koremella [24], marine sponges, were identified with chemoprevention properties. Further, the antioxidants, namely verongiaquinol and manzamine A, of the Red Sea sponge Aplysina sp. and Acanthostrongylophora sp., respectively, also exhibited antimutagenic potential [25]. Carotenoids, a group of natural compounds found across most forms of life, such as bacteria, algae, fungi, plants, and animals, have proven to have tremendous antioxidant potential and are also used as chemopreventive agents [26].
Tea is yet another important source of innumerable natural compounds with chemopreventive capacity. Among the various variants of teas, green tea is the best-studied system for its cancer chemopreventive and chemotherapeutic effects [27,28]. It has been both approved and proven that green tea reduces the risk of breast, prostate, thyroid, colorectal, stomach, esophageal, and prostate cancers [29,30,31,32,33,34]. The chemopreventive activity of green tea is mediated through catechins (flavan-3-ols), and the major bioactive component of the green tea extracts are: (−)-epicatechin (EC), (−)-epigallocatechin (EGC), (−)-epicatechin-3-gallate (ECG), and (−)-epigallocatechin-3-gallate (EGCG) [35] among which EGCG is the most abundant and biologically active [36].
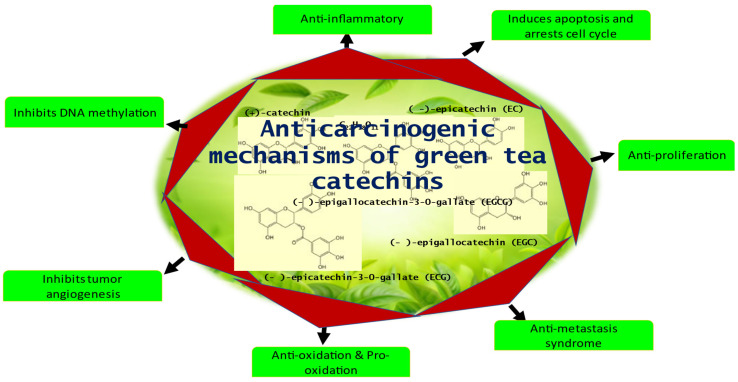
Among the other catechins, EGCG is well known for its inhibitory activity at all stages of cancer initiation, promotion, and progression. Other catechins, such as ECG and EGC, are relatively lower. EGCG inhibits protein kinases affecting cell growth and activating protein kinases linked to cell apoptosis and suppresses proteinases such as matrix metalloproteinase (MMPs) via inhibition of cancer cell migration, invasion, and metastasis. In addition, EGCG possesses antioxidant, anti-inflammatory, antiproliferative, antiangiogenic, and anti-metastatic effects by modulating signaling pathways, enzymatic activity, and protein kinases [37,38]. Figure 1 enlists the various anticarcinogenic mechanisms of GTCs. However, in humans, the plasma bioavailability of GTCs is very low, which has been in part attributed to their oxidation, metabolism, and efflux [39,40,41,42].
Figure 1.
The Anticarcinogenic mechanisms of GTCs.
In the present review, we have broadly reviewed the biological benefits of green tea catechins, and the specific anticancer applications and the milestones achieved through green tea catechins have been consolidated and presented. Finally, the main objective of this review is to highlight the combinatorial effect of catechins combined with various natural compounds. The future of such a combinatorial approach has been presented, and the lapses in the current knowledge have been addressed.
2. Biological Activity of Green Tea Catechins
Along with amino acids, proteins, lipids, and catechins, also known as flavan-3-ols, are a class of naturally occurring phenol/polyphenolic chemicals that are found in Camellia sinesis [43,44]. Until now, 8 different catechins, including C ((−)-catechin, EC ((−)-epicatechin), ECG ((−)-epicatechingallate), EGC ((−)-epigallocatechin), EGCG ((−)-epigallocatechin gallate), GC ((−)-gallocatechin), CG ((−)-catechingallate), and GCG ((−)-gallocatechin gallate), have been extracted from green tea, of which EC, EGC, ECG and EGCG are the dominant ones [45,46,47]. The biological properties of catechins include: antioxidants, anti-tumor, anti-inflammatory, anti-microbial, anti-viral, anti-diabetic, anti-obesity, and hypotensive effects [48,49,50,51].
These properties of green tea are found helpful for the treatment of obesity, diabetes, cardiovascular disease (CVD), nervous problems, and oral hygiene. For instance, catechins have been shown to regulate cell growth and nourishment and cause programmed cell death of tumor cells [50,52,53,54]. Numerous ailments and diseases, such as aging, arthritis, cancer, cardiovascular disease, diabetes, and obesity, include inflammation as the common component. The ability to reduce protein denaturation and boost the generation of anti-inflammatory cytokines are two of green tea’s overall anti-inflammatory capabilities [55]. Oxidative stress resulting from ROS has serious health implications, as it can damage DNA, affect protein folding, and reduce the ability of the mitochondria to produce ATP. As a result, the brain’s cognitive abilities may decline and may lead to conditions such as Alzheimer’s and Parkinson’s. Green tea’s anti-inflammatory and antioxidant qualities help protect neurons, and studies have demonstrated that its metabolites can pass the blood-brain barrier [56,57,58,59,60]. By binding to ROS, green tea’s antioxidant nature help neutralize free radicals inside the body and upregulates the activity of antioxidant enzymes [61,62]. Likewise, green tea’s anti-inflammatory and antioxidant properties have an impact on treating CVD. Additionally, regular intake of green tea has become instrumental in preventing atherosclerosis, lower total cholesterol levels, and enhancing the low-density lipoprotein (LDL) to high-density lipoprotein (HDL) ratio [63,64]. Specific symptoms, such as increased waist circumference, higher plasma triglycerides, increased LDL/HDL ratio, raised fasting blood glucose, and elevated blood pressure, are closely linked to metabolic syndromes such as diabetes and obesity [65,66]. Insulin resistance and perhaps reduced insulin production are additional features of type 2 diabetes. Green tea has been demonstrated to boost glucose-induced insulin production and improve receptor sensitivity for insulin [67,68]. Green tea has been demonstrated to decrease blood pressure and control obesity by improving HDL and reducing LDL, triglycerides, and body waist circumference by inhibiting digestive enzymes and fat absorption [69,70,71].
According to published research, green tea is proven to be antibacterial against a majority of oral microorganisms. Additionally, it has been demonstrated to enhance oral health by raising oral peroxidase activity, delaying the onset and progression of periodontitis, lowering dentin erosion and tooth loss, and thus contributing to the reduction of bad breath [72,73,74,75,76]. By interacting with the Pneumolysin and Sortase A of Streptococcus pneumoniae, epigallocatechin gallate (EGCG) was discovered to regulate antibiotic resistance [77]. Green tea was discovered to have a far higher level of antibacterial activity packed in the nano-sized particles that diffuse out from teabags compared to micro- and macro-sized particles. The bactericidal action was due to the abundance and higher active surface area of the catechins [78]. In a 2015 study, Deepak Kumar et al. examined the antimicrobial properties of 12 synthetic derivatives of catechins. Three of these derivatives of catechins exhibited antibacterial activity, and one of these compounds also demonstrated high antifungal activity. The docking investigation evidenced the catechins’ affinity for the ATP-binding region of DNA gyrase as the driving force behind the antibacterial or antifungal effect. There has been extensive research on the antibacterial properties of GTCs on microbes. It has been demonstrated that green tea can fight these germs both directly and indirectly and that it can also work synergistically along with routine antibiotics. The anti-inflammatory and antioxidant properties of green tea, together with other well-known health advantages, may also aid in the antibacterial effects. Studies on Escherichia coli revealed that exposure to green tea polyphenols (GTPs) caused significant changes in the expression of 17 genes, with nine genes being upregulated and eight genes being downregulated [79,80,81].
The presence of many structural -OH groups invest strong antioxidant property of the GTCs. More than ten families of chemicals make up the chemical formulation of green tea. Phenolic acids, polyphenolic compounds, amino acids, and lipids make up the primary constituents of green tea [43,44,82,83,84,85]. Due to the beneficial aspects of GTCs mentioned above, it has been recommended as a dietary product on a daily basis [86].
3. Anticancer Activity of Green Tea Catechins
Most of green tea’s anti-tumor properties are catechin-regulated, with EGCG having the strongest impact. EGCG has the highest inhibitory activity, followed by ECG > EGC > EC. Moreover, combinations of catechins have shown enhanced anti-tumor activity than isolated EGCG due to their combined effect. According to Fujiki et al. [87], GTCs have a variety of anti-mutagenic and anticarcinogenic effects on human malignancies, including those of the breast, esophagus, colon, prostate, small intestine, stomach, lung, and liver. Several of these effects will be discussed in this section.
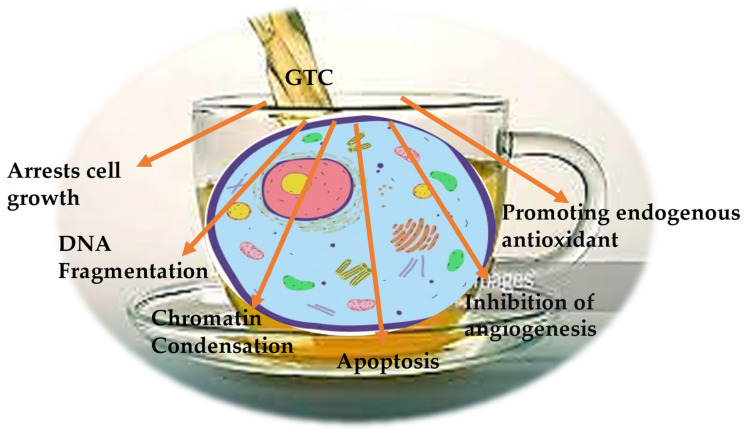
It is believed that green tea compounds, GTC mixtures, or pure EGCG are capable of influencing the carcinogenesis cycle, tumor origin, proliferation, and growth. This has been realized based on investigations using animal models and cancer cell lines [86]. For in-vivo investigations, xenograft tumor models of injecting human tumor cells subcutaneously into naked mice were conducted. Tumors typically develop over time depending on the cell concentrations injected. Mice were administered EGCG via intraperitoneal injection, dietary water or feed, and oral gavage, with varying concentrations of catechins consumed for the entire treatment in accordance with various experimental designs. Though the dosage of GTCs is varied with respect to the type of cancer cells studied, relatively fixed concentrations of EGCG (5–200 µM) were used predominantly. GTCs have been proven to reduce telomerase in cell lines, induce cell death, arrest cell cycles, and beneficially target cell receptors through binding with receptor tyrosine kinases (RTKs) [88]. GTCs have the capacity to both neutralize and generate ROS [89]. The antioxidant ability of GTCs has been ranked in the order ECG > EGCG > EGC > EC [90], and the prooxidant activity of GTCs has been reported to be crucial for programmed cell death and suppression of cancer cell development. GTCs are good demethylating agents and can be used as epigenetic modifiers to change the histone and the transcription of miRNAs in order to epigenetically control cellular processes and inhibit oncogene expression [91,92]. It has been established that GTCs, such as EGCG, can inhibit cancer growth in numerous mechanisms involving many molecular cues. Initially, EGCG binds with one or more target proteins, in this case usually a transmembrane receptor such as kinases, and modifies the regulation of signaling and metabolic processes that are crucial for the growth of cancerous cells. Figure 2 gives an overview of the various cancer-related processes that GTCs affect and interplay with.
Figure 2.
The anticancer mechanisms of GTCs.
Breast cancers are the most prevalent cancers among women around the world and appear due to many factors [93]. Epidemiological research in China has demonstrated that drinking green tea has positive effects on breast cancer prevention and recurrence, particularly for women who drink more than 4 cups of tea each day [94,95,96]. Numerous researchers have examined the mechanism through which catechins inhibit malignancy. Tea catechins, such as EGCG, ECG, and EC, have a potent antiangiogenic impact that inhibits cell proliferation and triggers apoptosis of breast cancer cells by neutralizing reactive oxygen species (ROS)-induced oxidative stress [97,98]. This was supported by reports validating the effect of EGCG on breast cancer cell inhibition and migration by down-regulating the phosphoinositide 3-kinases (PI3K)/Akt (Protein kinase B) and tumor protein p53/B-cell lymphoma (Bcl)-2 signaling pathways as well as modification of telomerase [99,100]. Additionally, prior research has demonstrated that tea catechins affect cell membrane receptors and restrict the spread of breast cancer cells by lowering levels of neo-nourishing factors such as vascular endothelial growth factor (VEGF) and epidermal growth factor (EGFR) and blocking the chemical signaling and activation of protein transcription-3 (STAT-3) and nuclear factor kappa B (NF-κB) [101,102]. Moreover, EGCG can inhibit breast cancer progression by hindering the focal adhesion kinase (FAK) signaling pathway, binding target proteins such as estrogen receptors (ERs) [101,103]; their antiproliferative activity by blocking ERβ-specific inhibitor is also reported [104]. Additionally, it has been suggested that EGCG modulates signal peptide-CUB-EGF domain-containing protein 2 (SCUBE2) gene expression in breast cancer cells and DNA methyltransferases [105]. Deb and colleagues typically found that 20 μM EGCG reduced the expression of the epigenetically suppressed TIMP-3 gene [106]. Additionally, ER + progesterone receptors (PR) + cancer cells were used to evaluate the anticancer effects of EGCG by epigenetic downregulation of ER- through p38 mitogen-activated protein kinase(p38MAPK)/casein kinase 2 (CK2) activation [107]. Animal models have demonstrated that administering EGCG inhibits the growth of cancerous breast tissue. The observations in a xenograft model revealed that EGCG inhibited pro-tumor macrophage invasion and macrophage 2 (M2) polarization after intraperitoneal administration of 10 mg/kg EGCG [108]. GTCs are crucial for the prevention and treatment of human breast cancer owing to their potential to target the molecular cues responsible for breast cancer.
One of the worst malignancies in the world is hepatocellular carcinoma (HCC), an invasive liver cancer [109]. Numerous scientific and clinical investigations have looked closely at the anti-hepatocellular carcinoma properties of green tea catechins. Now it is known that EGCG can suppress the development and spread of hepatocellular tumors by inducing apoptosis, controlling autophagy, and acting as an anti-angiogenic agent [110]. EGCG has been shown to inhibit the growth of human HCC cells in in vitro studies using hepatic cancer cell lines by blocking the phosphorylation of the tyrosine-kinase receptor insulin-like growth factor 1 receptor (IGF-1R), initiating cell death by activating Caspase-9 and -3, suppressing Bcl-2, cyclooxygenase-2 (COX-2) and lipogenic enzymes, regulating the levels of VEGF and its receptor (VEGFR-2), NF-κB and ERK1/2, actuate AMPK (adenosine monophosphate-activated protein) and reactive oxygen species (ROS)-mediated membrane permeability [111]. The beneficial effects of GTCs intake were tested in various animal models, and its ability to suppress HCC growth, proliferation, and trigger apoptosis has been confirmed. It was hypothesized that GTCs prohibit hepatocyte stem cell growth, excitation of adenosine monophosphate-activated protein kinase (AMPK) protein inside the liver, and regulation of epigenetic expression [110,112]. Additionally, EGCG reduced the activity of the VEGF/VEGFR or IGF/IGF1R signaling axis, which in turn reduced the development of HCC xenografts [113,114]. Therefore, it is thought that GTCs, particularly EGCG, may be useful in preventing hepatic cancer.
Prostate cancer is one of the most common diseases affecting men and is currently a serious public health issue [115]. It is observed that the regular intake of green tea catechins (for 1 year) not only reduces the serum IGF-1, VEGF, and the target antigen for the prostate but also prevents the advancement of mature intraepithelial neoplasia to prostate cancer [116,117]. Recently, green tea has been utilized as a radiosensitizer for the effective treatment of prostate cancer via radiotherapy [118]. Numerous studies have suggested that GTCs hindered prostate cancer progression at various stages of the disease by reducing the expression of the prostate-specific antigen (PSA), the transcriptional activity of the androgen receptor (AR), the activation of chromatin proteins, and DNA methylation of genes [119,120]. Additionally, Gupta et al. (2000) showed that treatment of prostate cancer with various concentrations of EGCG (20, 40, and 80 µM) could pronounce notable effects on G0/G1 phase arrest and cause cellular death by upregulation of p21 waf1 expression [121]. Furthermore, the same group identified the cdk inhibitor-cyclin-cdk machinery as the relevant bio-molecular pathway for EGCG-instigated cell cycle overactivity and apoptosis. EGCG (5 µM) has also been noted to constrain cell migration and incursion via impacting lipid rafts to disable stimulation of the tyrosine protein kinase Met receptor [122]. The involvement of upregulated p53, p21, Bcl-2-associated X protein(bax), and cysteine protease-9 (CASP-9a) expression and DNA-binding protein inhibitor (ID2) downregulation was other also linked to prostate cell death [123,124,125]. The therapeutic efficacy of EGCG against prostate tumorigenesis was reported in transgenic adenocarcinoma of mouse prostate (TRAMP). The study results on mice illustrated that a 0.06% uptake of EGCG in drinking water for 28 weeks reduced prostate cancer proliferation and induced their death by down-regulating molecular mediators that support cancerous phenotype [126,127]. Based on the aforementioned research, EGCG can be used as an attractive chemo-agent for prostate cancer prevention.
The prevalence of gastrointestinal (GI) cancers poses a grave threat due to their high risks and mortality in humans [128]. According to an epidemiological study, drinking green tea can lower the incidence of GI cancer, especially those pertaining to the esophagus and colorectum [129]. In fact, green tea’s role in the prevention of esophageal cancer was validated by a meta-analysis of clinical studies done in China [130]. Several works, both in vitro and in vivo, have demonstrated GTCs’ influence on gastric and colorectal cancer. Some of the effects on tumor vascularization, DNA methylation, reduced tumor cell growth and proliferation, and programmed cell death were all postulated. It is postulated that EGCG hinders cancerous cell formation and invasion by disrupting the molecular signals of cancer cells. Moreover, EGCG was proposed to prompt G1 cell phase arrest, Glycogen synthase kinase 3 (GSK-3) and protein phosphatase 2A(PP2A) phosphorylation and decrease epidermal growth factor receptor (EGFR), human epidermal growth factor receptor 2 (HER2) activation and wingless/Integrated (Wnt)/catenin signal pathways [131,132]. According to other studies [133,134]. EGCG can also end up arresting the cell cycle by impairing the transcription factor and signaling pathways such as (activator protein (AP-1), Akt, ERK1/2, p38, MAPK, NF-B and also cause a reduction in the level of essential anti-apoptotic proteins. Similarly, Umeda et al. [135] found that, at a physiological dose of 1 M EGCG, EGCG stimulated G2/M cell cycle arrest and activated laminin (receptor) mediated myosin phosphatase. Adachi and colleagues conducted investigations with the goal of treating diseases related to obesity. These studies revealed that EGCG reduced the development of colorectal cancer cells by decreasing the expression of a variety of Receptor Tyrosine Kinas (RTK)-mediated cell signaling pathways [136,137]. Activating Bax and Caspase-3, lowering the levels of proliferation protein cyclin D1 and regulator protein Bcl-2 in the NF-B channels, and inhibiting the G0-G1 phase have all been observed in esophageal cell lines [138]. The production of VEGF and VEGFA, as well as the activation of the VEGF/VEGFR axis, have also been reported to be inhibited by EGCG [139] in colorectal cancer cells. EGCG has been shown to modulate the epigenome to regulate the methylation degree of the transcriptional regulator Retinod X Receptor (RXR) promoter [140]. Animal, model-based studies validated the anticancer effects of EGCG on N-nitro methylbenzylamine (NMBA)-induced rat esophageal carcinogenesis via targeting cyclin D1 and affecting COX-2 expression [141]. Additionally, EGCG oral dosages of 5, 10, and 20 mg/kg led to a reduction in colorectal cancer cell volume. This resulted from EGCG’s antagonistic effect on the expression of Notch signaling. Additionally, researchers examined the effects of intraperitoneal injection of 1.5 mg/d of EGCG on angiogenesis and tumor formation in naked mice bearing stomach cancer xenografts. Possible causes included reduced VEGF and CD31 expression [139,140,142]. In a colon carcinogenesis model, EGCG suppressed the activation of the IGF/IGF1R axis and the VEGF/VEGFR axis, according to [113,143]. Overall, the studies mentioned above show that GTCs hold a prominent position in gastrointestinal tract carcinogenesis prevention and cure.
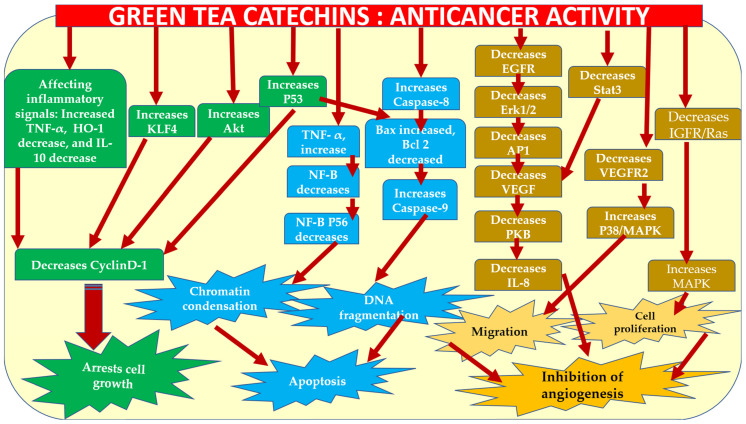
About 25% of cancer-related deaths are caused by lung cancer. Lung cancer claims more lives each year than colon, breast, and prostate cancers combined [144] (https://www.cancer.org/cancer/lung-cancer/about/key-statistics.html, accessed on 12 January 2023) Various results on the effect of GTCs consumption in the suppression of chemically induced lung tumors in transgenetic rodents are available. Similar to the aforementioned cancers, GTCs have been found to have both curative and preventive effects on lung cancer through obstruction of key protein kinases and regulation of molecular signals and gene expression of cyclin D1, Bcl-2, p21, p53, VEGF, Bax COX-2, Caspase-3, -7, and -9 [145,146,147]. Lu and colleagues [148] corroborated the hindering ability of GTCs to be more effective than caffeine against lung carcinogenesis in experiments with 4-(methylnitrosamino)-1-(3-Pyridyl)-1-Butanone (NNK)-induced A/J mice, administering 0.5% Polyphenon E (65% EGCG) or 0.044% caffeine as dietary fluid for 52 weeks. It was also claimed that EGCG could significantly inhibit phosphorylation [148]. Similarly, H1299 and Lu99, types of non-small cell lung cancer (NSCLC), when treated with 50 and 100 µM EGCG. were shown to suppress metastasis of lung carcinoma. For the probable mechanism of inhibition of epithelial-mesenchymal transition (EMT), phosphorylation of matrix metalloproteinases-9 (MMP-9) with tyrosine and focal Adhesion Kinase (FAK) activities were suggested [149,150,151]. The effect of EGCG (0–20 µM) on H1299 cells has shown the affinity of EGCG attachment with rat sarcoma (Ras)-GTPase activating protein src Homology-3 (SH3) domain-binding protein 1 (G3BP1), production of ROS and cancer cell apoptosis [152,153]. Treatments with 10–100 µM EGCG have shown remarkable effects on lung carcinogenesis and its spread by blocking multiple protein receptors, nicotine-driven migration, and differentiation of Non-small cell lung cancer (NSCLC) cells [154,155]. Likewise, treatment of NSCLC with EGCG (0–120 µM) has been shown to disrupt the enzyme expression of telomerase along with Caspase-3 and -9 activities [156]. As a result, catechins can function as targeted agents to stop or slow the development of lung tumors. As summarized in this section, GTCs’ anticancer effects are indeed diverse and well investigated, and authoritatively confirmed. Figure 3 summarizes the various signaling pathways and molecular targets of GTCs behind the anticancer activity of GTCs.
Figure 3.
The molecular targets and signaling pathways behind the anticancer activity of GTCs. TNF—tumor necrosis factor; HO-1—Heme oxygenase-1; IL—interleukin; KLF4—Kruppel-like factor 4.
4. Anticancer Activity of Catechins Combined with Natural Compounds
With the proven anticarcinogenic activity of GTCs gaining eminence, various combinatorial approaches, where GTCs were combined with other anticancer drugs/natural compounds, have also been tested. Generally, natural compounds hold a clear advantage over drugs for reasons such as their natural availability, abundance, and bio-safety. The combined effect of tea catechins (EGCG) with other natural compounds have shown a complementary effect in exerting anticancerous property due to improved bioavailability of pro-apoptotic metabolites. The various mechanisms of catechins in enhancing anticancer effects when combined with other natural substances are compiled in Table 1. For example, the combination of curcumin and catechins has shown better anticancer effects on a wide range of cancer cells [157,158,159,160,161,162]. Other bioactive substances, such as 6-gingerol, pan-axadiol, sulforaphane, and pterostilbene, have been able to exhibit mutual interactions with GTCs like that of curcumin [163,164,165,166]. Natural components such as Piperine and genistein are others that have been observed to enhance EGCG bioavailability by hindering glucuronidation and MRP-mediated efflux, thereby controlling the mobility of EGCG within the gastrointestinal region [167,168]. Furthermore, it has been reported that combining GTCs with ascorbic acid, sucrose, quercetin, and carbohydrate has enhanced the bioaccessibility and intestinal absorption of catechins [169,170]. It has been demonstrated that luteolin, in combination with EGCG, has enhanced anti-lung cancer properties via the increased activation of cancer-suppressor p53 protein [171]. Similarly, Papi and colleagues concluded that the mixtures of vitexin-2-oxyloside (40 g/mL), raphasatin (5 g/mL), with EGCG (10 g/mL) can inhibit colon cancer cell proliferation and trigger its death via the mitochondrial-mediated pathway [172]. More recent work on integrating a silibinin mixture with EGCG has shown a more promising way to target cancer endothelial cells [173].
Table 1.
Anticarcinogenic effects of GTCs when combined with antioxidant-rich anticancerous natural compounds.
| GTC Component | Natural Compounds | Cancer Type | Anticarcinogenic Activity | Molecular Mechanism | References |
|---|---|---|---|---|---|
| EGCG | Curcumin | MCF-7 breast cancer | Inhibition of cancer cell growth and induction of apoptosis | Activation of caspase-dependent apoptosis, inhibition of P-gP pump function | [160] |
| EGCG | Curcumin | MDA-MB-231 breast cancer | Inhibition of cancer cell growth | G(2)/M-phase arrest. Decreased VEGFR-1 protein expression in tumours |
[159] |
| Epicatechin | Curcumin | HL-60 myeloid leukaemia | Inhibition of cancer cell growth | Cell cycle arrest at S phase | [174] |
| EGCG | Curcumin | Normal, premalignant and malignant oral cells | Inhibition of cancer cell growth | Cell cycle arrest at G1 phase and S/G2M | [158] |
| EGCG | Curcumin | A549 and NCI-H460 lung cancer | Inhibition of cancer cell and tumour growth | Cell cycle arrest at G1 and S/G2 phases via cyclin D1 and cyclin B1 inhibition | [162] |
| EGCG | Curcumin | PC3—human prostate cancer | Inhibition of cancer cell and tumour growth | Arrests S and G2/M phases by upregulated expression of p-21 | [157] |
| EGCG | Curcumin and lovastatin | SKGT-4 and TE-8 esophageal cancer | Inhibition of cancer cell and tumour growth | Suppression of mitotic signal transduction pathway through Phosphorylation/dephosporylation of /Erk1/2, c-Jun and COX-2 | [161] |
| EGCG | Silibinin | Non-small-cell lung cancer cells | Inhibition of angiogenesis and cell migration of endothelial and lung tumour cells | Antiangiogenic activity via VEGF, VEGFR2, and miR-17–92 cluster and miR-19b cluster | [173] |
| EGCG | Luteolin | esophageal cancer cell lines—TE-8 and SKGT-4 | Inhibition of growth and induction of apoptosis of cancer cells and tumour | induction of p53-dependent apoptotic pathways | [171] |
| EGCG | 6-Gingerol | 1321 N1 and LN18 glioma cells | Inhibition of cancer cell growth and induce apoptosis | Apoptosis induction by activated Caspase-3 and Annexin-V FITC/PI | [166] |
| EGCG | Panaxadiol (PD) | HCT-116 and SW-480 human colorectal cancers | Inhibits cancer cell growth and induces apoptosis | Cell cycle arrest at G1 and G2/M | [163] |
| EGCG | Sulforaphane (SFN) |
PC3 prostate cancer | Arrests cancer cells | Inhibition of genes in AP-1 pathway inhibits cell proliferation, differentiation, apoptosis, angiogenesis and tumour invasion | [165] |
| EGCG | Pterostilbene | MIA PaCa-2 and PANC-1 Pancreatic cancer cells | Inhibits growth of cancer cells and induces apoptosis | Induction of cell cycle arrest and cell apoptosis | [164] |
| EGCG | Paclitaxel (Taxol) | Breast cancer cells (4T1, MCF-7, and MDA-MB231) | Inhibits growth of cancer cells and induces apoptosis | JNK phosphorylation and cell death | [175] |
| EGCG | taxane (i.e., paclitaxel and docetaxel) | Human Prostrate cancer cells PC-3ML cells | Inhibition of growth and induction of apoptosis | Induction of cell cycle arrest and cell apoptosis | [176] |
| EC epicatechin |
Panaxadio | HCT-116 human colorectal cancer cells |
Inhibition of cancer cell growth | Mechanism not reported | [177] |
| EGCG | Cisplatin/ tamoxifen |
1321N1 cells and U87-MG cells/ biliary tract cancer cells | Cytotoxicity on Cancer cells | Inhibition of telomerase and induction of cell cycle arrest | [178,179] |
| EGCG | doxorubicin | Drug-resistant KB-A1 cells | Cytotoxicity on Cancer cells | Modulating P-glycoprotein efflux pump | [180] |
| EGCG | doxorubicin (DOX) | human oral epidermoid carcinoma (KB-A-1) | In vivo reversal of doxorubicin resistanceby (-)-epigallocatechin gallate in a solid human carcinoma | Induction of cell apoptosis | [181] |
| EGCG | Vitexin-2-o- xyloside, Raphasatin | Colon cancer cells—LoVo and CaCo-2; Breast cancer cells—MDA-MB-231 and MCF-7 |
Inhibition of cancer cell growth and induction of apoptosis | Arrests Cell cycle at G0/G1 phases and activating ROS- mediated mitochondrial apoptotic pathways | [172] |
| EGCG and ECG | DOX | Chemoresistant hepatocellular carcinoma (HCC) cell line BEL-7404 | Inhibition of cancer cell and tumour growth. | Inhibition of P-glycoprotein efflux pump | [182] |
Abbreviations: EC—(−)-epicatechin; EGC—(−)-epigallocatechin; ECG—(−)-epicatechin-3-gallate; EGCG—(−)-epigallocatechin-3-gallate; ROS—reactive oxygen species; JNK—Jun N-terminal kinase; AP1—activating protein; VEGF—vascular endothelial growth factor; COX—Cyclooxygenase; Erk—Extracellular signal-regulated kinase; FITC/PI—fluorescein isothiocyanate; PI, propidium iodide; miR—microRNAs.
5. Anticarcinogenic Antioxidant/Prooxidant Effects of Catechins
The specific chemical structure of polyphenols found in green tea (the presence of a minimum of five hydroxyl groups) has a substantial impact on antioxidant capacity [183,184,185,186,187,188]. This is because green tea contains at least five hydroxyl groups. The di/tri-hydroxy structure of the B and D rings [185], as well as the meta-5,7-dihydroxy group at the A ring [189,190], help in the chelation of transition metal ions [183,184]. For synthesizing any novel anticancer molecule(s) based on the structure of catechins, (i) the molecule should be of epicatechin type; (ii) it should have as many galloyl moieties because this will increase the number of hydroxyls that can bind to DNA and Cu(II) and reducing it to Cu(I) [183]. Nevertheless, in certain cases, they might also exhibit prooxidative effects [183,184,191]. The actual anticancer mechanism is through the regulation of catechins within an intracellular pool of nitro-oxidative stress [192]. This is the reason why polyphenolic chemicals, especially catechins at high dosages, can also result in adverse effects [193] because catechins are a type of antioxidant. Hence, at high dosages, induction of prooxidative stress as well as oxygen damage to the constituents of the cell result. Close connectivity between inflammation and prooxidative action also exists [193]. Catechins have qualities that are both antioxidant and prooxidant, and both properties are dependent on the same variables (OH groups) inside the molecule [183]. Oxidation of polyphenols results in reactive oxygen species and electrophilic quinones that can damage cells [194,195]. Thus, this prooxidative nature of catechins should also be seriously taken into account since it plays a vital role in the etiopathogenesis of degenerative disorders leading to cancer [193]. Figure 4 summarizes the anticancer activity of catechin exercised via its antioxidant activity.
Figure 4.
The molecular targets and signaling pathways are affected via the antioxidant activity of GTCs resulting in the anticancer effect.
6. Challenges, Future Recommendations, and Conclusions
One of the major challenges facing the biomedical application of GTCs is their low bioavailability. Certain progress has been made in this related aspect; however, clear resolutions are far from being achieved. One of the key strategies towards enhancing the antitumor potential of GTCs for improving their bioavailability was through combining catechins with other phytochemicals and anticancer compounds through nanostructure-based delivery systems and molecular modifications. Standalone GTCs, in spite of their high reputation, have their own limitations, this is where combinatorial approaches take lead positions. Combination therapies with catechins can exert synergistic effects through interaction of catechins with bioactives or drugs, leading to reduction of the side effect of conventional chemotherapy and enhanced anticancer potential. Chemically modifying EGCG or other catechins, using nanotechnological as well as drug delivery approaches has a wider perspective in improving its anticancer effect [40]. There is a lot to expect from GTCs in the field of anticancer research. This review prompts more research attention in these areas. There are abundant in vitro investigations; more in vivo investigations are required to understand and improvise the available resources from GTCs for human benefit.
Thus far, binary combinations have been worked out with GTCs, trinary or ternary composites, combining two or three more versatile and dynamic properties of natural compounds (for example, EGCG/cur/lovastatin), need be attempted since the success rates in overcoming the limitations of catechins though such multiple combinations can be relatively higher. Moreover, through the course of the review, we could find only a handful of research articles that reported GTCs/natural compound combinations, with most studies localized around the use of curcumin. Nature has more to offer than just curcumin, and there are diverse natural antioxidant compounds reputed for their strong anticancer activity (for example, resveratrol, indole-3-carbinol, vitamin D, chrysin, celastrol, and betulinic acid); this review urges more expansion through attempting many other available natural options. Additionally, EGCG is, for the most part, worked on GTC; the other green tea catechins also possess unique properties, and combinations of two or more catechins with natural compounds could exhibit more versatile anticarcinogenic properties.
GTCs’ systemic action mechanism in the human body is far from understood; it is necessary that we obtain a more comprehensive understanding of EGCG-mediated anticancer mechanisms by exploring whether the relative molecules are direct targets for EGCG and then substantiate the in vivo relevance of the reported mechanisms. Targeted delivery by attaching specific ligands to nanoparticles is another area that has not been looked into in detail. The clinical translation of GTCs, their pharmacokinetics, and drug delivery aspects need more investigation [40]. The results from clinical trials with EGCG are not all positive. A study performed on 49 patients with various tumors reported no major antitumor responses when using GTE. The catechin concentrations and administration methods, as well as other parameters, need to be optimized to obtain a clear picture. More importantly, the safety aspects and the side effects of GTCs should be investigated in clinical trials, and the related mechanisms should be theoretically/practically demonstrated.
The various anticarcinogenic achievements of GTCs were reviewed, and the progress made in anticancer therapies was summarized. The combinatorial enhancement of the ant-cancer attributes of GTCs, through integration with renowned natural compounds invested with anticancer properties, has been specifically highlighted, and the future directions proposed.
Author Contributions
J.G., M.M. and S.S.C.P., preparation of the original draft, and revisions; J.-W.O., participated in the review and revisions, funding. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
This research received no external funding.
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Wu S., Zhu W., Thompson P., Hannun Y.A. Evaluating Intrinsic and Non-Intrinsic Cancer Risk Factors. Nat. Commun. 2018;9:3490. doi: 10.1038/s41467-018-05467-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.WHO Cancer Key Facts. [(accessed on 3 January 2023)]; Available online: https://www.who.int/news-room/fact-sheets/detail/cancer.
- 3.Ferlay J., Soerjomataram I., Dikshit R., Eser S., Mathers C., Rebelo M., Parkin D.M., Forman D., Bray F. Cancer Incidence and Mortality Worldwide: Sources, Methods and Major Patterns in GLOBOCAN 2012. Int. J. Cancer. 2015;136:E359–E386. doi: 10.1002/ijc.29210. [DOI] [PubMed] [Google Scholar]
- 4.Ferlay J., Colombet M., Soerjomataram I., Mathers C., Parkin D.M., Piñeros M., Znaor A., Bray F. Estimating the Global Cancer Incidence and Mortality in 2018: GLOBOCAN Sources and Methods. Int. J. Cancer. 2019;144:1941–1953. doi: 10.1002/ijc.31937. [DOI] [PubMed] [Google Scholar]
- 5.Praetorius N.P., Mandal T.K. Engineered Nanoparticles in Cancer Therapy. Recent Pat. Drug Deliv. Formul. 2007;1:37–51. doi: 10.2174/187221107779814104. [DOI] [PubMed] [Google Scholar]
- 6.Donnenberg V.S., Donnenberg A.D. Multiple Drug Resistance in Cancer Revisited: The Cancer Stem Cell Hypothesis. J. Clin. Pharmacol. 2005;45:872–877. doi: 10.1177/0091270005276905. [DOI] [PubMed] [Google Scholar]
- 7.Velasco-Velázquez M.A., Homsi N., De La Fuente M., Pestell R.G. Breast Cancer Stem Cells. Int. J. Biochem. Cell Biol. 2012;44:573–577. doi: 10.1016/j.biocel.2011.12.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.LaVan D.A., McGuire T., Langer R. Small-Scale Systems for in Vivo Drug Delivery. Nat. Biotechnol. 2003;21:1184–1191. doi: 10.1038/nbt876. [DOI] [PubMed] [Google Scholar]
- 9.Illouz F., Braun D., Briet C., Schweizer U., Rodien P. Endocrine Side-Effects of Anti-Cancer Drugs: Thyroid Effects of Tyrosine Kinase Inhibitors. Eur. J. Endocrinol. 2014;171:R91–R99. doi: 10.1530/EJE-14-0198. [DOI] [PubMed] [Google Scholar]
- 10.Jiang T., Yang T., Chen Y., Miao Y., Xu Y., Jiang H., Yang M., Mao C. Emulating Interactions between Microorganisms and Tumor Microenvironment to Develop Cancer Theranostics. Theranostics. 2022;12:2833–2859. doi: 10.7150/thno.70719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Stork G., Schultz A.G. The Total Synthesis of Dl-Camptothecin. J. Am. Chem. Soc. 1971;93:4074–4075. doi: 10.1021/ja00745a056. [DOI] [PubMed] [Google Scholar]
- 12.Volkmann R., Danishefsky S., Eggler J., Solomon D.M. Total Synthesis of (+-)-Camptothecine. J. Am. Chem. Soc. 1971;93:5576–5577. doi: 10.1021/ja00750a045. [DOI] [Google Scholar]
- 13.Ejima A., Terasawa H., Sugimori M., Tagawa H. Asymmetric Synthesis of (S)-Camptothecin. Tetrahedron Lett. 1989;30:2639–2640. doi: 10.1016/S0040-4039(00)99086-5. [DOI] [Google Scholar]
- 14.Bennasar M.-L., Juan C., Bosch J. A Short Synthesis of Camptothecin via A2-Fluoro-1,4-Dihydropyridine. Chem. Commun. 2000:2459–2460. doi: 10.1039/b007814j. [DOI] [Google Scholar]
- 15.Bennasar M.-L., Zulaica E., Juan C., Alonso Y., Bosch J. Addition of Ester Enolates to N-Alkyl-2-Fluoropyridinium Salts: Total Synthesis of (±)-20-Deoxycamptothecin and (+)-Camptothecin. J. Org. Chem. 2002;67:7465–7474. doi: 10.1021/jo026173j. [DOI] [PubMed] [Google Scholar]
- 16.Lin L.-G., Xie H., Li H.-L., Tong L.-J., Tang C.-P., Ke C.-Q., Liu Q.-F., Lin L.-P., Geng M.-Y., Jiang H., et al. Naturally Occurring Homoisoflavonoids Function as Potent Protein Tyrosine Kinase Inhibitors by C-Src-Based High-Throughput Screening. J. Med. Chem. 2008;51:4419–4429. doi: 10.1021/jm701501x. [DOI] [PubMed] [Google Scholar]
- 17.Yang S.-P., Zhang X.-W., Ai J., Gan L.-S., Xu J.-B., Wang Y., Su Z.-S., Wang L., Ding J., Geng M.-Y., et al. Potent HGF/c-Met Axis Inhibitors from Eucalyptus Globulus: The Coupling of Phloroglucinol and Sesquiterpenoid Is Essential for the Activity. J. Med. Chem. 2012;55:8183–8187. doi: 10.1021/jm3007454. [DOI] [PubMed] [Google Scholar]
- 18.Li M.-H., Miao Z.-H., Tan W.-F., Yue J.-M., Zhang C., Lin L.-P., Zhang X.-W., Ding J. Pseudolaric Acid B Inhibits Angiogenesis and Reduces Hypoxia-Inducible Factor 1α by Promoting Proteasome-Mediated Degradation. Clin. Cancer Res. 2004;10:8266–8274. doi: 10.1158/1078-0432.CCR-04-0951. [DOI] [PubMed] [Google Scholar]
- 19.Yu B., Li M.-H., Wang W., Wang Y.-Q., Jiang Y., Yang S.-P., Yue J.-M., Ding J., Miao Z.-H. Pseudolaric Acid B-Driven Phosphorylation of c-Jun Impairs Its Role in Stabilizing HIF-1alpha: A Novel Function-Converter Model. J. Mol. Med. 2012;90:971–981. doi: 10.1007/s00109-012-0865-4. [DOI] [PubMed] [Google Scholar]
- 20.Zhu X., Yuan C., Tian C., Li C., Nie F., Song X., Zeng R., Wu D., Hao X., Li L. The Plant Sesquiterpene Lactone Parthenolide Inhibits Wnt/β-Catenin Signaling by Blocking Synthesis of the Transcriptional Regulators TCF4/LEF1. J. Biol. Chem. 2018;293:5335–5344. doi: 10.1074/jbc.M117.819300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Huang M., Lu J.-J., Ding J. Natural Products in Cancer Therapy: Past, Present and Future. Nat. Prod. Bioprospect. 2021;11:5–13. doi: 10.1007/s13659-020-00293-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gu K.J., Li G. An Overview of Cancer Prevention: Chemoprevention and Immunoprevention. J. Cancer Prev. 2020;25:127–135. doi: 10.15430/JCP.2020.25.3.127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Torres Y.R., Berlinck R.G.S., Nascimento G.G.F., Fortier S.C., Pessoa C., de Moraes M.O. Antibacterial Activity against Resistant Bacteria and Cytotoxicity of Four Alkaloid Toxins Isolated from the Marine Sponge Arenosclera Brasiliensis. Toxicon. 2002;40:885–891. doi: 10.1016/S0041-0101(01)00286-0. [DOI] [PubMed] [Google Scholar]
- 24.Dobiáš L., Černá M., Rössner P., Šrám R. Genotoxicity and Carcinogenicity of Metronidazole. Mutat. Res./Rev. Genet. Toxicol. 1994;317:177–194. doi: 10.1016/0165-1110(94)90001-9. [DOI] [PubMed] [Google Scholar]
- 25.Calcabrini C., Catanzaro E., Bishayee A., Turrini E., Fimognari C. Marine Sponge Natural Products with Anticancer Potential: An Updated Review. Mar. Drugs. 2017;15:310. doi: 10.3390/md15100310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Shanmugham V., Subban R. Capsanthin-Loaded Micelles: Preparation, Characterization and in Vitro Evaluation of Cytotoxicity Using MDA-MB-231 Breast Cancer Cell Line. Food Technol. Biotechnol. 2022;60:350–360. doi: 10.17113/ftb.60.03.22.7405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chen X., Han Y., Meng H., Li W., Li Q., Luo Y., Wang C., Xie J., Wu L., Zhang X., et al. Characteristics of the Emulsion Stabilized by Polysaccharide Conjugates Alkali-Extracted from Green Tea Residue and Its Protective Effect on Catechins. Ind. Crops Prod. 2019;140:111611. doi: 10.1016/j.indcrop.2019.111611. [DOI] [Google Scholar]
- 28.Han Y., Cheng Z., Zhang Y., Zhang N., Zhu X., Chen X., Shao Y., Cheng Y., Wang C., Luo Y., et al. Effect of Metal Ions and PH on the Emulsifying Properties of Polysaccharide Conjugates Prepared from Low-Grade Green Tea. Food Hydrocoll. 2020;102:105624. doi: 10.1016/j.foodhyd.2019.105624. [DOI] [Google Scholar]
- 29.Guo Y., Zhi F., Chen P., Zhao K., Xiang H., Mao Q., Wang X., Zhang X. Green Tea and the Risk of Prostate Cancer: A Systematic Review and Meta-Analysis. Medicine. 2017;96:e6426. doi: 10.1097/MD.0000000000006426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ma S., Wang C., Bai J., Wang X., Li C. Association of Tea Consumption and the Risk of Thyroid Cancer: A Meta-Analysis. Int. J. Clin. Exp. Med. 2015;8:14345–14351. [PMC free article] [PubMed] [Google Scholar]
- 31.Najaf Najafi M., Salehi M., Ghazanfarpour M., Hoseini Z.S., Khadem-Rezaiyan M. The Association between Green Tea Consumption and Breast Cancer Risk: A Systematic Review and Meta-Analysis. Phytother. Res. 2018;32:1855–1864. doi: 10.1002/ptr.6124. [DOI] [PubMed] [Google Scholar]
- 32.Khan N., Mukhtar H. Modulation of Signaling Pathways in Prostate Cancer by Green Tea Polyphenols. Biochem. Pharmacol. 2013;85:667–672. doi: 10.1016/j.bcp.2012.09.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Sawada N. Risk and Preventive Factors for Prostate Cancer in Japan: The Japan Public Health Center-Based Prospective (JPHC) Study. J. Epidemiol. 2017;27:2–7. doi: 10.1016/j.je.2016.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zhang L., Ho C.-T., Zhou J., Santos J.S., Armstrong L., Granato D. Chemistry and Biological Activities of Processed Camellia sinensis Teas: A Comprehensive Review. Compr. Rev. Food Sci. Food Saf. 2019;18:1474–1495. doi: 10.1111/1541-4337.12479. [DOI] [PubMed] [Google Scholar]
- 35.Chen X., Du Y., Wu L., Xie J., Chen X., Hu B., Wu Z., Yao Q., Li Q. Effects of Tea-Polysaccharide Conjugates and Metal Ions on Precipitate Formation by Epigallocatechin Gallate and Caffeine, the Key Components of Green Tea Infusion. J. Agric. Food Chem. 2019;67:3744–3751. doi: 10.1021/acs.jafc.8b06681. [DOI] [PubMed] [Google Scholar]
- 36.Shirakami Y., Shimizu M. Possible Mechanisms of Green Tea and Its Constituents against Cancer. Molecules. 2018;23:2284. doi: 10.3390/molecules23092284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Oz H.S., Chen T., de Villiers W.J.S. Green Tea Polyphenols and Sulfasalazine Have Parallel Anti-Inflammatory Properties in Colitis Models. Front. Immunol. 2013;4:132. doi: 10.3389/fimmu.2013.00132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wang J., Man G.C.W., Chan T.H., Kwong J., Wang C.C. A Prodrug of Green Tea Polyphenol (−)-Epigallocatechin-3-Gallate (Pro-EGCG) Serves as a Novel Angiogenesis Inhibitor in Endometrial Cancer. Cancer Lett. 2018;412:10–20. doi: 10.1016/j.canlet.2017.09.054. [DOI] [PubMed] [Google Scholar]
- 39.Kale A., Gawande S., Kotwal S., Netke S., Roomi W., Ivanov V., Niedzwiecki A., Rath M. Studies on the Effects of Oral Administration of Nutrient Mixture, Quercetin and Red Onions on the Bioavailability of Epigallocatechin Gallate from Green Tea Extract. Phytother. Res. 2010;24((Suppl. S1)):S48–S55. doi: 10.1002/ptr.2899. [DOI] [PubMed] [Google Scholar]
- 40.Bimonte S., Albino V., Piccirillo M., Nasto A., Molino C., Palaia R., Cascella M. Epigallocatechin-3-Gallate in the Prevention and Treatment of Hepatocellular Carcinoma: Experimental Findings and Translational Perspectives. Drug Des. Dev. Ther. 2019;13:611–621. doi: 10.2147/DDDT.S180079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Stevens J.F., Maier C.S. The Chemistry of Gut Microbial Metabolism of Polyphenols. Phytochem. Rev. 2016;15:425–444. doi: 10.1007/s11101-016-9459-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Cheng Z., Zhang Z., Han Y., Wang J., Wang Y., Chen X., Shao Y., Cheng Y., Zhou W., Lu X., et al. A Review on Anti-Cancer Effect of Green Tea Catechins. J. Funct. Foods. 2020;74:104172. doi: 10.1016/j.jff.2020.104172. [DOI] [Google Scholar]
- 43.Ahmad N., Mukhtar H. Green Tea Polyphenols and Cancer: Biologic Mechanisms and Practical Implications. Nutr. Rev. 1999;57:78–83. doi: 10.1111/j.1753-4887.1999.tb06927.x. [DOI] [PubMed] [Google Scholar]
- 44.Tadano N., Du C.-K., Yumoto F., Morimoto S., Ohta M., Xie M.-F., Nagata K., Zhan D.-Y., Lu Q.-W., Miwa Y., et al. Biological Actions of Green Tea Catechins on Cardiac Troponin C. Br. J. Pharmacol. 2010;161:1034–1043. doi: 10.1111/j.1476-5381.2010.00942.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Jin Y., Jin C.H., Row K.H. Separation of Catechin Compounds from Different Teas. Biotechnol. J. 2006;1:209–213. doi: 10.1002/biot.200500019. [DOI] [PubMed] [Google Scholar]
- 46.Fung S.-T., Ho C.K., Choi S.-W., Chung W.-Y., Benzie I.F.F. Comparison of Catechin Profiles in Human Plasma and Urine after Single Dosing and Regular Intake of Green Tea (Camellia sinensis) Br. J. Nutr. 2013;109:2199–2207. doi: 10.1017/S0007114512004370. [DOI] [PubMed] [Google Scholar]
- 47.Bae J., Kim N., Shin Y., Kim S.-Y., Kim Y.-J. Activity of Catechins and Their Applications. Biomed. Dermatol. 2020;4:8. doi: 10.1186/s41702-020-0057-8. [DOI] [Google Scholar]
- 48.Hamilton-Miller J.M.T. Anti-Cariogenic Properties of Tea (Camellia sinensis) J. Med. Microbiol. 2001;50:299–302. doi: 10.1099/0022-1317-50-4-299. [DOI] [PubMed] [Google Scholar]
- 49.Maity R., Chatterjee M., Banerjee A., Das A., Mishra R., Mazumder S., Chanda N. Gold Nanoparticle-Assisted Enhancement in the Anti-Cancer Properties of Theaflavin against Human Ovarian Cancer Cells. Mater. Sci. Eng. C Mater. Biol. Appl. 2019;104:109909. doi: 10.1016/j.msec.2019.109909. [DOI] [PubMed] [Google Scholar]
- 50.Subramani C., Natesh R.K. Molecular Mechanisms and Biological Implications of Green Tea Polyphenol, (−)-Epigallocatechin-3-Gallate. J. Pharma Biosci. Technol. 2013;1:54–63. [Google Scholar]
- 51.Chacko S.M., Thambi P.T., Kuttan R., Nishigaki I. Beneficial Effects of Green Tea: A Literature Review. Chin. Med. 2010;5:13. doi: 10.1186/1749-8546-5-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Crew K.D., Ho K.A., Brown P., Greenlee H., Bevers T.B., Arun B., Sneige N., Hudis C., McArthur H.L., Chang J., et al. Effects of a Green Tea Extract, Polyphenon E, on Systemic Biomarkers of Growth Factor Signalling in Women with Hormone Receptor-Negative Breast Cancer. J. Hum. Nutr. Diet. 2015;28:272–282. doi: 10.1111/jhn.12229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Li M.-J., Yin Y.-C., Wang J., Jiang Y.-F. Green Tea Compounds in Breast Cancer Prevention and Treatment. World J. Clin. Oncol. 2014;5:520–528. doi: 10.5306/wjco.v5.i3.520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Shirakami Y., Sakai H., Kochi T., Seishima M., Shimizu M. Catechins and Its Role in Chronic Diseases. Adv. Exp. Med. Biol. 2016;929:67–90. doi: 10.1007/978-3-319-41342-6_4. [DOI] [PubMed] [Google Scholar]
- 55.Chatterjee P., Chandra S., Dey P., Bhattacharya S. Evaluation of Anti-Inflammatory Effects of Green Tea and Black Tea: A Comparative in Vitro Study. J. Adv. Pharm. Technol. Res. 2012;3:136–138. doi: 10.4103/2231-4040.97298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Faria A., Pestana D., Teixeira D., Azevedo J., De F.V., Mateus N., Calhau C. Flavonoid transport across RBE4 cells: A blood-brain barrier model. Cell. Mol. Biol. Lett. 2010;15:234–241. doi: 10.2478/s11658-010-0006-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Figueira I., Garcia G., Pimpão R.C., Terrasso A.P., Costa I., Almeida A.F., Tavares L., Pais T.F., Pinto P., Ventura M.R., et al. Polyphenols Journey through Blood-Brain Barrier towards Neuronal Protection. Sci. Rep. 2017;7:11456. doi: 10.1038/s41598-017-11512-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Mancini E., Beglinger C., Drewe J., Zanchi D., Lang U.E., Borgwardt S. Green Tea Effects on Cognition, Mood and Human Brain Function: A Systematic Review. Phytomedicine. 2017;34:26–37. doi: 10.1016/j.phymed.2017.07.008. [DOI] [PubMed] [Google Scholar]
- 59.Scholey A., Downey L.A., Ciorciari J., Pipingas A., Nolidin K., Finn M., Wines M., Catchlove S., Terrens A., Barlow E., et al. Acute Neurocognitive Effects of Epigallocatechin Gallate (EGCG) Appetite. 2012;58:767–770. doi: 10.1016/j.appet.2011.11.016. [DOI] [PubMed] [Google Scholar]
- 60.Vauzour D. Dietary Polyphenols as Modulators of Brain Functions: Biological Actions and Molecular Mechanisms Underpinning Their Beneficial Effects. Oxid. Med. Cell. Longev. 2012;2012:914273. doi: 10.1155/2012/914273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Newsome B.J., Petriello M.C., Han S.G., Murphy M.O., Eske K.E., Sunkara M., Morris A.J., Hennig B. Green Tea Diet Decreases PCB 126-Induced Oxidative Stress in Mice by up-Regulating Antioxidant Enzymes. J. Nutr. Biochem. 2014;25:126–135. doi: 10.1016/j.jnutbio.2013.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Tsai C.-F., Hsu Y.-W., Ting H.-C., Huang C.-F., Yen C.-C. The in Vivo Antioxidant and Antifibrotic Properties of Green Tea (Camellia sinensis, Theaceae) Food Chem. 2013;136:1337–1344. doi: 10.1016/j.foodchem.2012.09.063. [DOI] [PubMed] [Google Scholar]
- 63.Islam M.A. Cardiovascular Effects of Green Tea Catechins: Progress and Promise. Recent Pat. Cardiovasc. Drug Discov. 2012;7:88–99. doi: 10.2174/157489012801227292. [DOI] [PubMed] [Google Scholar]
- 64.Bhardwaj P., Khanna D. Green Tea Catechins: Defensive Role in Cardiovascular Disorders. Chin. J. Nat. Med. 2013;11:345–353. doi: 10.1016/S1875-5364(13)60051-5. [DOI] [PubMed] [Google Scholar]
- 65.Grandl G., Wolfrum C. Hemostasis, Endothelial Stress, Inflammation, and the Metabolic Syndrome. Semin. Immunopathol. 2018;40:215–224. doi: 10.1007/s00281-017-0666-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Iqbal J., Al Qarni A., Hawwari A., Alghanem A.F., Ahmed G. Metabolic Syndrome, Dyslipidemia and Regulation of Lipoprotein Metabolism. Curr. Diabetes Rev. 2018;14:427–433. doi: 10.2174/1573399813666170705161039. [DOI] [PubMed] [Google Scholar]
- 67.Fu Q.-Y., Li Q.-S., Lin X.-M., Qiao R.-Y., Yang R., Li X.-M., Dong Z.-B., Xiang L.-P., Zheng X.-Q., Lu J.-L., et al. Antidiabetic Effects of Tea. Molecules. 2017;22:849. doi: 10.3390/molecules22050849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Munir K.M., Chandrasekaran S., Gao F., Quon M.J. Mechanisms for Food Polyphenols to Ameliorate Insulin Resistance and Endothelial Dysfunction: Therapeutic Implications for Diabetes and Its Cardiovascular Complications. Am. J. Physiol. Endocrinol. Metab. 2013;305:E679–E686. doi: 10.1152/ajpendo.00377.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Huang J., Wang Y., Xie Z., Zhou Y., Zhang Y., Wan X. The Anti-Obesity Effects of Green Tea in Human Intervention and Basic Molecular Studies. Eur. J. Clin. Nutr. 2014;68:1075–1087. doi: 10.1038/ejcn.2014.143. [DOI] [PubMed] [Google Scholar]
- 70.Siriwardhana N., Kalupahana N.S., Cekanova M., LeMieux M., Greer B., Moustaid-Moussa N. Modulation of Adipose Tissue Inflammation by Bioactive Food Compounds. J. Nutr. Biochem. 2013;24:613–623. doi: 10.1016/j.jnutbio.2012.12.013. [DOI] [PubMed] [Google Scholar]
- 71.Suzuki T., Pervin M., Goto S., Isemura M., Nakamura Y. Beneficial Effects of Tea and the Green Tea Catechin Epigallocatechin-3-Gallate on Obesity. Molecules. 2016;21:1305. doi: 10.3390/molecules21101305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Araghizadeh A., Kohanteb J., Fani M.M. Inhibitory Activity of Green Tea (Camellia sinensis) Extract on Some Clinically Isolated Cariogenic and Periodontopathic Bacteria. Med. Princ. Pract. 2013;22:368–372. doi: 10.1159/000348299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Aylıkcı B.U., Colak H. Halitosis: From Diagnosis to Management. J. Nat. Sci. Biol. Med. 2013;4:14–23. doi: 10.4103/0976-9668.107255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.De Moraes M.D.R., Carneiro J.R.M., Passos V.F., Santiago S.L. Effect of Green Tea as a Protective Measure against Dental Erosion in Coronary Dentine. Braz. Oral Res. 2016;30:S1806-83242016000100213. doi: 10.1590/1807-3107BOR-2016.vol30.0013. [DOI] [PubMed] [Google Scholar]
- 75.Kushiyama M., Shimazaki Y., Murakami M., Yamashita Y. Relationship between Intake of Green Tea and Periodontal Disease. J. Periodontol. 2009;80:372–377. doi: 10.1902/jop.2009.080510. [DOI] [PubMed] [Google Scholar]
- 76.Narotzki B., Levy Y., Aizenbud D., Reznick A.Z. Green Tea and Its Major Polyphenol EGCG Increase the Activity of Oral Peroxidases. Adv. Exp. Med. Biol. 2013;756:99–104. doi: 10.1007/978-94-007-4549-0_13. [DOI] [PubMed] [Google Scholar]
- 77.Song M., Teng Z., Li M., Niu X., Wang J., Deng X. Epigallocatechin Gallate Inhibits Streptococcus Pneumoniae Virulence by Simultaneously Targeting Pneumolysin and Sortase A. J. Cell. Mol. Med. 2017;21:2586–2598. doi: 10.1111/jcmm.13179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Gopal J., Muthu M., Paul D., Kim D.-H., Chun S. Bactericidal Activity of Green Tea Extracts: The Importance of Catechin Containing Nano Particles. Sci. Rep. 2016;6:19710. doi: 10.1038/srep19710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Cho Y.S., Schiller N.L., Kahng H.Y., Oh K.H. Cellular Responses and Proteomic Analysis of Escherichia coli Exposed to Green Tea Polyphenols. Curr. Microbiol. 2007;55:501–506. doi: 10.1007/s00284-007-9021-8. [DOI] [PubMed] [Google Scholar]
- 80.Sirk T.W., Brown E.F., Friedman M., Sum A.K. Molecular Binding of Catechins to Biomembranes: Relationship to Biological Activity. J. Agric. Food Chem. 2009;57:6720–6728. doi: 10.1021/jf900951w. [DOI] [PubMed] [Google Scholar]
- 81.Sirk T.W., Brown E.F., Sum A.K., Friedman M. Molecular Dynamics Study on the Biophysical Interactions of Seven Green Tea Catechins with Lipid Bilayers of Cell Membranes. J. Agric. Food Chem. 2008;56:7750–7758. doi: 10.1021/jf8013298. [DOI] [PubMed] [Google Scholar]
- 82.Yoshizawa S., Horiuchi T., Suganuma M., Nishiwaki S., Yatsunami J., Okabe S., Okuda T., Muto Y., Frenkel K., Troll W., et al. Phenolic Compounds in Food and Their Effects on Health II. Volume 507. American Chemical Society; Washington, DC, USA: 1992. Penta-O-Galloyl-β-D-Glucose and (−)-Epigallocatechin Gallate; pp. 316–325. (ACS Symposium Series). [Google Scholar]
- 83.Ann Beltz L., Kay Bayer D., Lynn Moss A., Mitchell Simet I. Mechanisms of Cancer Prevention by Green and Black Tea Polyphenols. Anticancer Agents Med. Chem. 2006;6:389–406. doi: 10.2174/187152006778226468. [DOI] [PubMed] [Google Scholar]
- 84.Graham H.N. Green Tea Composition, Consumption, and Polyphenol Chemistry. Prev. Med. 1992;21:334–350. doi: 10.1016/0091-7435(92)90041-F. [DOI] [PubMed] [Google Scholar]
- 85.Komatsu Y., Suematsu S., Hisanobu Y., Saigo H., Matsuda R., Hara K. Effects of PH and Temperature on Reaction Kinetics of Catechins in Green Tea Infusion. Biosci. Biotechnol. Biochem. 1993;57:907–910. doi: 10.1271/bbb.57.907. [DOI] [Google Scholar]
- 86.Singh B.N., Shankar S., Srivastava R.K. Green Tea Catechin, Epigallocatechin-3-Gallate (EGCG): Mechanisms, Perspectives and Clinical Applications. Biochem. Pharmacol. 2011;82:1807–1821. doi: 10.1016/j.bcp.2011.07.093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Fujiki H., Sueoka E., Watanabe T., Suganuma M. Synergistic Enhancement of Anticancer Effects on Numerous Human Cancer Cell Lines Treated with the Combination of EGCG, Other Green Tea Catechins, and Anticancer Compounds. J. Cancer Res. Clin. Oncol. 2015;141:1511–1522. doi: 10.1007/s00432-014-1899-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Vidak M., Rozman D., Komel R. Effects of Flavonoids from Food and Dietary Supplements on Glial and Glioblastoma Multiforme Cells. Molecules. 2015;20:19406–19432. doi: 10.3390/molecules201019406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Lambert J.D., Elias R.J. The Antioxidant and Pro-Oxidant Activities of Green Tea Polyphenols: A Role in Cancer Prevention. Arch. Biochem. Biophys. 2010;501:65–72. doi: 10.1016/j.abb.2010.06.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Zaveri N.T. Green Tea and Its Polyphenolic Catechins: Medicinal Uses in Cancer and Noncancer Applications. Life Sci. 2006;78:2073–2080. doi: 10.1016/j.lfs.2005.12.006. [DOI] [PubMed] [Google Scholar]
- 91.Mitra S., Dash R. Natural Products for the Management and Prevention of Breast Cancer. Evid. Based. Complement. Alternat. Med. 2018;2018:8324696. doi: 10.1155/2018/8324696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Shankar E., Kanwal R., Candamo M., Gupta S. Dietary Phytochemicals as Epigenetic Modifiers in Cancer: Promise and Challenges. Semin. Cancer Biol. 2016;40–41:82–99. doi: 10.1016/j.semcancer.2016.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.De Cicco P., Catani M.V., Gasperi V., Sibilano M., Quaglietta M., Savini I. Nutrition and Breast Cancer: A Literature Review on Prevention, Treatment and Recurrence. Nutrients. 2019;11:1514. doi: 10.3390/nu11071514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Li M., Tse L.A., Chan W.-C., Kwok C.-H., Leung S.-L., Wu C., Yu W.-C., Yu I.T.-S., Yu C.H.-T., Wang F., et al. Evaluation of Breast Cancer Risk Associated with Tea Consumption by Menopausal and Estrogen Receptor Status among Chinese Women in Hong Kong. Cancer Epidemiol. 2016;40:73–78. doi: 10.1016/j.canep.2015.11.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Yuan J.-M., Sun C., Butler L.M. Tea and Cancer Prevention: Epidemiological Studies. Pharmacol. Res. 2011;64:123–135. doi: 10.1016/j.phrs.2011.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Zhou P., Li J.-P., Zhang C. Green Tea Consumption and Breast Cancer Risk: Three Recent Meta-Analyses. Breast Cancer Res. Treat. 2011;127:581–583. doi: 10.1007/s10549-010-1338-5. [DOI] [PubMed] [Google Scholar]
- 97.Rathore K., Choudhary S., Odoi A., Wang H.-C.R. Green Tea Catechin Intervention of Reactive Oxygen Species-Mediated ERK Pathway Activation and Chronically Induced Breast Cell Carcinogenesis. Carcinogenesis. 2012;33:174–183. doi: 10.1093/carcin/bgr244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Ruch R.J., Cheng S.J., Klaunig J.E. Prevention of Cytotoxicity and Inhibition of Intercellular Communication by Antioxidant Catechins Isolated from Chinese Green Tea. Carcinogenesis. 1989;10:1003–1008. doi: 10.1093/carcin/10.6.1003. [DOI] [PubMed] [Google Scholar]
- 99.Moradzadeh M., Hosseini A., Erfanian S., Rezaei H. Epigallocatechin-3-Gallate Promotes Apoptosis in Human Breast Cancer T47D Cells through down-Regulation of PI3K/AKT and Telomerase. Pharmacol. Rep. 2017;69:924–928. doi: 10.1016/j.pharep.2017.04.008. [DOI] [PubMed] [Google Scholar]
- 100.Huang C.-Y., Han Z., Li X., Xie H.-H., Zhu S.-S. Mechanism of EGCG Promoting Apoptosis of MCF-7 Cell Line in Human Breast Cancer. Oncol. Lett. 2017;14:3623–3627. doi: 10.3892/ol.2017.6641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Li Y., Yuan Y.-Y., Meeran S.M., Tollefsbol T.O. Synergistic Epigenetic Reactivation of Estrogen Receptor-α (ERα) by Combined Green Tea Polyphenol and Histone Deacetylase Inhibitor in ERα-Negative Breast Cancer Cells. Mol. Cancer. 2010;9:274. doi: 10.1186/1476-4598-9-274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Farabegoli F., Govoni M., Spisni E., Papi A. EGFR Inhibition by (−)-Epigallocatechin-3-Gallate and IIF Treatments Reduces Breast Cancer Cell Invasion. Biosci. Rep. 2017;37:BSR20170168. doi: 10.1042/BSR20170168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Aller G.S.V., Carson J.D., Tang W., Peng H., Zhao L. Biochemical and Biophysical Research Communications Epigallocatechin gallate (EGCG), a major component of green tea, is a dual phosphoinositide-3-kinase/mTOR inhibitor. Biochem. Biophys. Res. Commun. 2011;406:194–199. doi: 10.1016/j.bbrc.2011.02.010. [DOI] [PubMed] [Google Scholar]
- 104.Baker K.M., Bauer A.C. Green Tea Catechin, EGCG, Suppresses PCB 102-Induced Proliferation in Estrogen-Sensitive Breast Cancer Cells. Int. J. Breast Cancer. 2015;2015:163591. doi: 10.1155/2015/163591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Sheng J., Shi W., Guo H., Long W., Wang Y., Qi J., Liu J., Xu Y. The Inhibitory Effect of (−)-Epigallocatechin-3-Gallate on Breast Cancer Progression via Reducing SCUBE2 Methylation and DNMT Activity. Molecules. 2019;24:2899. doi: 10.3390/molecules24162899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Deb G., Thakur V.S., Limaye A.M., Gupta S. Epigenetic Induction of Tissue Inhibitor of Matrix Metalloproteinase-3 by Green Tea Polyphenols in Breast Cancer Cells. Mol. Carcinog. 2015;54:485–499. doi: 10.1002/mc.22121. [DOI] [PubMed] [Google Scholar]
- 107.De Amicis F., Russo A., Avena P., Santoro M., Vivacqua A., Bonofiglio D., Mauro L., Aquila S., Tramontano D., Fuqua S.A.W., et al. In Vitro Mechanism for Downregulation of ER-α Expression by Epigallocatechin Gallate in ER+/PR+ Human Breast Cancer Cells. Mol. Nutr. Food Res. 2013;57:840–853. doi: 10.1002/mnfr.201200560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Jang J.-Y., Lee J.-K., Jeon Y.-K., Kim C.-W. Exosome Derived from Epigallocatechin Gallate Treated Breast Cancer Cells Suppresses Tumor Growth by Inhibiting Tumor-Associated Macrophage Infiltration and M2 Polarization. BMC Cancer. 2013;13:421. doi: 10.1186/1471-2407-13-421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Yang H., Wang M., Sun H., Zhu S., Jin J. Synergetic Effect of EP1 Receptor Antagonist and (−)-Epigallocatechin-3-Gallate in Hepatocellular Carcinoma. Pharmacology. 2019;104:267–275. doi: 10.1159/000502076. [DOI] [PubMed] [Google Scholar]
- 110.Shimizu M., Shirakami Y., Sakai H., Kubota M., Kochi T., Ideta T., Miyazaki T., Moriwaki H. Chemopreventive Potential of Green Tea Catechins in Hepatocellular Carcinoma. Int. J. Mol. Sci. 2015;16:6124–6139. doi: 10.3390/ijms16036124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Larsen C.A., Dashwood R.H., Bisson W.H. Tea Catechins as Inhibitors of Receptor Tyrosine Kinases: Mechanistic Insights and Human Relevance. Pharmacol. Res. 2010;62:457–464. doi: 10.1016/j.phrs.2010.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Sur S., Pal D., Mandal S., Roy A., Panda C.K. Tea Polyphenols Epigallocatechin Gallete and Theaflavin Restrict Mouse Liver Carcinogenesis through Modulation of Self-Renewal Wnt and Hedgehog Pathways. J. Nutr. Biochem. 2016;27:32–42. doi: 10.1016/j.jnutbio.2015.08.016. [DOI] [PubMed] [Google Scholar]
- 113.Shimizu M., Shirakami Y., Sakai H., Adachi S., Hata K., Hirose Y., Tsurumi H., Tanaka T., Moriwaki H. (−)-Epigallocatechin Gallate Suppresses Azoxymethane-Induced Colonic Premalignant Lesions in Male C57BL/KsJ-Db/Db Mice. Cancer Prev. Res. 2008;1:298–304. doi: 10.1158/1940-6207.CAPR-08-0045. [DOI] [PubMed] [Google Scholar]
- 114.Shirakami Y., Shimizu M., Adachi S., Sakai H., Nakagawa T., Yasuda Y., Tsurumi H., Hara Y., Moriwaki H. (−)-Epigallocatechin Gallate Suppresses the Growth of Human Hepatocellular Carcinoma Cells by Inhibiting Activation of the Vascular Endothelial Growth Factor-Vascular Endothelial Growth Factor Receptor Axis. Cancer Sci. 2009;100:1957–1962. doi: 10.1111/j.1349-7006.2009.01241.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Iranikhah M., Stricker S., Freeman M.K. Future of Bisphosphonates and Denosumab for Men with Advanced Prostate Cancer. Cancer Manag. Res. 2014;6:217–224. doi: 10.2147/CMAR.S40151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.McLarty J., Bigelow R.L.H., Smith M., Elmajian D., Ankem M., Cardelli J.A. Tea polyphenols decrease serum levels of prostate-specific antigen, hepatocyte growth factor, and vascular endothelial growth factor in prostate cancer patients and inhibit production of hepatocyte growth factor and vascular endothelial growth factor in vitro. Cancer Prev. Res. 2009;2:673–682. doi: 10.1158/1940-6207.CAPR-08-0167. [DOI] [PubMed] [Google Scholar]
- 117.Bettuzzi S., Brausi M., Rizzi F., Castagnetti G., Peracchia G., Corti A. Chemoprevention of Human Prostate Cancer by Oral Administration of Green Tea Catechins in Volunteers with High-Grade Prostate Intraepithelial Neoplasia: A Preliminary Report from a One-Year Proof-of-Principle Study. Cancer Res. 2006;66:1234–1240. doi: 10.1158/0008-5472.CAN-05-1145. [DOI] [PubMed] [Google Scholar]
- 118.Schroeder A.C., Xiao H., Zhu Z., Li Q., Bai Q., Wakefield M.R., Mann J.D., Fang Y. A Potential Role for Green Tea as a Radiation Sensitizer for Prostate Cancer. Pathol. Oncol. Res. 2019;25:263–268. doi: 10.1007/s12253-017-0358-4. [DOI] [PubMed] [Google Scholar]
- 119.Chuu C.-P., Chen R.-Y., Kokontis J.M., Hiipakka R.A., Liao S. Suppression of Androgen Receptor Signaling and Prostate Specific Antigen Expression by (−)-Epigallocatechin-3-Gallate in Different Progression Stages of LNCaP Prostate Cancer Cells. Cancer Lett. 2009;275:86–92. doi: 10.1016/j.canlet.2008.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Khan N., Mukhtar H. Tea and Health: Studies in Humans. Curr. Pharm. Des. 2013;19:6141–6147. doi: 10.2174/1381612811319340008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Gupta S., Ahmad N., Nieminen A.L., Mukhtar H. Growth Inhibition, Cell-Cycle Dysregulation, and Induction of Apoptosis by Green Tea Constituent (−)-Epigallocatechin-3-Gallate in Androgen-Sensitive and Androgen-Insensitive Human Prostate Carcinoma Cells. Toxicol. Appl. Pharmacol. 2000;164:82–90. doi: 10.1006/taap.1999.8885. [DOI] [PubMed] [Google Scholar]
- 122.Duhon D., Bigelow R.L.H., Coleman D.T., Steffan J.J., Yu C., Langston W., Kevil C.G., Cardelli J.A. The Polyphenol Epigallocatechin-3-Gallate Affects Lipid Rafts to Block Activation of the c-Met Receptor in Prostate Cancer Cells. Mol. Carcinog. 2010;49:739–749. doi: 10.1002/mc.20649. [DOI] [PubMed] [Google Scholar]
- 123.Hagen R.M., Chedea V.S., Mintoff C.P., Bowler E., Morse H.R., Ladomery M.R. Epigallocatechin-3-Gallate Promotes Apoptosis and Expression of the Caspase 9a Splice Variant in PC3 Prostate Cancer Cells. Int. J. Oncol. 2013;43:194–200. doi: 10.3892/ijo.2013.1920. [DOI] [PubMed] [Google Scholar]
- 124.Hastak K., Agarwal M.K., Mukhtar H., Agarwal M.L. Ablation of Either P21 or Bax Prevents P53-Dependent Apoptosis Induced by Green Tea Polyphenol Epigallocatechin-3-Gallate. FASEB J. 2005;19:789–791. doi: 10.1096/fj.04-2226fje. [DOI] [PubMed] [Google Scholar]
- 125.Luo K.L., Luo J.-H., Yu Y.P. (−)-Epigallocatechin-3-Gallate Induces Du145 Prostate Cancer Cell Death via Downregulation of Inhibitor of DNA Binding 2, a Dominant Negative Helix-Loop-Helix Protein. Cancer Sci. 2010;101:707–712. doi: 10.1111/j.1349-7006.2009.01425.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Brown M.D., Gilmore P.E., Hart C.A., Samuel J.D., Ramani V.A.C., George N.J., Clarke N.W. Characterization of Benign and Malignant Prostate Epithelial Hoechst 33342 Side Populations. Prostate. 2007;67:1384–1396. doi: 10.1002/pros.20620. [DOI] [PubMed] [Google Scholar]
- 127.Moses M.A., Henry E.C., Ricke W.A., Gasiewicz T.A. The Heat Shock Protein 90 Inhibitor, (−)-Epigallocatechin Gallate, Has Anticancer Activity in a Novel Human Prostate Cancer Progression Model. Cancer Prev. Res. 2015;8:249–257. doi: 10.1158/1940-6207.CAPR-14-0224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Dizdar Ö., Kılıçkap S. Global Epidemiology of Gastrointestinal Cancers. In: Yalcin S., Philip P.A., editors. Textbook of Gastrointestinal Oncology. Springer International Publishing; Cham, Switwerland: 2019. pp. 1–12. [Google Scholar]
- 129.Suganuma M., Takahashi A., Watanabe T., Iida K., Matsuzaki T., Yoshikawa H.Y., Fujiki H. Biophysical Approach to Mechanisms of Cancer Prevention and Treatment with Green Tea Catechins. Molecules. 2016;21:1566. doi: 10.3390/molecules21111566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Zheng J.-S., Yang J., Fu Y.-Q., Huang T., Huang Y.-J., Li D. Effects of Green Tea, Black Tea, and Coffee Consumption on the Risk of Esophageal Cancer: A Systematic Review and Meta-Analysis of Observational Studies. Nutr. Cancer. 2013;65:1–16. doi: 10.1080/01635581.2013.741762. [DOI] [PubMed] [Google Scholar]
- 131.Shimizu M., Deguchi A., Lim J.T.E., Moriwaki H., Kopelovich L., Weinstein I.B. (−)-Epigallocatechin gallate and polyphenon E inhibit growth and activation of the epidermal growth factor receptor and human epidermal growth factor receptor-2 signaling pathways in human colon cancer cells. Clin. Cancer Res. 2005;11:2735–2746. doi: 10.1158/1078-0432.CCR-04-2014. [DOI] [PubMed] [Google Scholar]
- 132.Oh S., Gwak J., Park S., Yang C.S. Green Tea Polyphenol EGCG Suppresses Wnt/β-Catenin Signaling by Promoting GSK-3β- and PP2A-Independent β-Catenin Phosphorylation/Degradation. Biofactors. 2014;40:586–595. doi: 10.1002/biof.1185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Cerezo-Guisado M.I., Zur R., Lorenzo M.J., Risco A., Martín-Serrano M.A., Alvarez-Barrientos A., Cuenda A., Centeno F. Implication of Akt, ERK1/2 and Alternative P38MAPK Signalling Pathways in Human Colon Cancer Cell Apoptosis Induced by Green Tea EGCG. Food Chem. Toxicol. 2015;84:125–132. doi: 10.1016/j.fct.2015.08.017. [DOI] [PubMed] [Google Scholar]
- 134.Navarro-Perán E., Cabezas-Herrera J., Sánchez-del-Campo L., García-Cánovas F., Rodríguez-López J.N. The Anti-Inflammatory and Anti-Cancer Properties of Epigallocatechin-3-Gallate Are Mediated by Folate Cycle Disruption, Adenosine Release and NF-ΚB Suppression. Inflamm. Res. 2008;57:472–478. doi: 10.1007/s00011-008-8013-x. [DOI] [PubMed] [Google Scholar]
- 135.Umeda D., Yano S., Yamada K., Tachibana H. Involvement of 67-KDa Laminin Receptor-Mediated Myosin Phosphatase Activation in Antiproliferative Effect of Epigallocatechin-3-O-Gallate at a Physiological Concentration on Caco-2 Colon Cancer Cells. Biochem. Biophys. Res. Commun. 2008;371:172–176. doi: 10.1016/j.bbrc.2008.04.041. [DOI] [PubMed] [Google Scholar]
- 136.Adachi S., Nagao T., Ingolfsson H.I., Maxfield F.R., Andersen O.S., Kopelovich L., Weinstein I.B. The Inhibitory Effect of (−)-Epigallocatechin Gallate on Activation of the Epidermal Growth Factor Receptor Is Associated with Altered Lipid Order in HT29 Colon Cancer Cells. Cancer Res. 2007;67:6493–6501. doi: 10.1158/0008-5472.CAN-07-0411. [DOI] [PubMed] [Google Scholar]
- 137.Adachi S., Shimizu M., Shirakami Y., Yamauchi J., Natsume H., Matsushima-Nishiwaki R., To S., Weinstein I.B., Moriwaki H., Kozawa O. (−)-Epigallocatechin Gallate Downregulates EGF Receptor via Phosphorylation at Ser1046/1047 by P38 MAPK in Colon Cancer Cells. Carcinogenesis. 2009;30:1544–1552. doi: 10.1093/carcin/bgp166. [DOI] [PubMed] [Google Scholar]
- 138.Liu L., Ju Y., Wang J., Zhou R. Epigallocatechin-3-Gallate Promotes Apoptosis and Reversal of Multidrug Resistance in Esophageal Cancer Cells. Pathol. Res. Pract. 2017;213:1242–1250. doi: 10.1016/j.prp.2017.09.006. [DOI] [PubMed] [Google Scholar]
- 139.Zhu B.-H., Zhan W.-H., Li Z.-R., Wang Z., He Y.-L., Peng J.-S., Cai S.-R., Ma J.-P., Zhang C.-H. (−)-Epigallocatechin-3-Gallate Inhibits Growth of Gastric Cancer by Reducing VEGF Production and Angiogenesis. World J. Gastroenterol. 2007;13:1162–1169. doi: 10.3748/wjg.v13.i8.1162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Morris J., Moseley V.R., Cabang A.B., Coleman K., Wei W., Garrett-Mayer E., Wargovich M.J. Reduction in Promotor Methylation Utilizing EGCG (Epigallocatechin-3-Gallate) Restores RXRα Expression in Human Colon Cancer Cells. Oncotarget. 2016;7:35313–35326. doi: 10.18632/oncotarget.9204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Li Z.G., Shimada Y., Sato F., Maeda M., Itami A., Kaganoi J., Komoto I., Kawabe A., Imamura M. Inhibitory Effects of Epigallocatechin-3-Gallate on N-Nitrosomethylbenzylamine-Induced Esophageal Tumorigenesis in F344 Rats. Int. J. Oncol. 2002;21:1275–1283. doi: 10.3892/ijo.21.6.1275. [DOI] [PubMed] [Google Scholar]
- 142.Wu H., Xin Y., Xiao Y., Zhao J. Low-Dose Docetaxel Combined with (−)-Epigallocatechin-3-Gallate Inhibits Angiogenesis and Tumor Growth in Nude Mice with Gastric Cancer Xenografts. Cancer Biother. Radiopharm. 2012;27:204–209. doi: 10.1089/cbr.2011.1103. [DOI] [PubMed] [Google Scholar]
- 143.Shimizu M., Shirakami Y., Sakai H., Yasuda Y., Kubota M., Adachi S., Tsurumi H., Hara Y., Moriwaki H. (−)-Epigallocatechin Gallate Inhibits Growth and Activation of the VEGF/VEGFR Axis in Human Colorectal Cancer Cells. Chem. Biol. Interact. 2010;185:247–252. doi: 10.1016/j.cbi.2010.03.036. [DOI] [PubMed] [Google Scholar]
- 144.American Cancer Society Key Statistics for Lung Cancer. [(accessed on 2 January 2023)]. Available online: https://www.cancer.org/cancer/lung-cancer/about/key-statistics.html.
- 145.Fu H., He J., Mei F., Zhang Q., Hara Y., Ryota S., Lubet R.A., Chen R., Chen D., You M. Lung Cancer Inhibitory Effect of Epigallocatechin-3-Gallate Is Dependent on Its Presence in a Complex Mixture (Polyphenon E) Cancer Prev. Res. 2009;2:531–537. doi: 10.1158/1940-6207.CAPR-08-0185. [DOI] [PubMed] [Google Scholar]
- 146.Manna S., Mukherjee S., Roy A., Das S., Panda C.K. Tea Polyphenols Can Restrict Benzo[a]Pyrene-Induced Lung Carcinogenesis by Altered Expression of P53-Associated Genes and H-Ras, c-Myc and Cyclin D1. J. Nutr. Biochem. 2009;20:337–349. doi: 10.1016/j.jnutbio.2008.04.001. [DOI] [PubMed] [Google Scholar]
- 147.Manna S., Banerjee S., Mukherjee S., Das S., Panda C.K. Epigallocatechin Gallate Induced Apoptosis in Sarcoma180 Cells In Vivo: Mediated by P53 Pathway and Inhibition in U1B, U4-U6 UsnRNAs Expression. Apoptosis. 2006;11:2267–2276. doi: 10.1007/s10495-006-0198-2. [DOI] [PubMed] [Google Scholar]
- 148.Lu G., Liao J., Yang G., Reuhl K.R., Hao X., Yang C.S. Inhibition of Adenoma Progression to Adenocarcinoma in a 4-(Methylnitrosamino)-1-(3-Pyridyl)-1-Butanone-Induced Lung Tumorigenesis Model in A/J Mice by Tea Polyphenols and Caffeine. Cancer Res. 2006;66:11494–11501. doi: 10.1158/0008-5472.CAN-06-1497. [DOI] [PubMed] [Google Scholar]
- 149.Liu J.D., Chen S.H., Lin C.L., Tsai S.H., Liang Y.C. Inhibition of Melanoma Growth and Metastasis by Combination with (−)-Epigallocatechin-3-Gallate and Dacarbazine in Mice. J. Cell. Biochem. 2001;83:631–642. doi: 10.1002/jcb.1261. [DOI] [PubMed] [Google Scholar]
- 150.Takahashi A., Watanabe T., Mondal A., Suzuki K., Kurusu-Kanno M., Li Z., Yamazaki T., Fujiki H., Suganuma M. Mechanism-Based Inhibition of Cancer Metastasis with (−)-Epigallocatechin Gallate. Biochem. Biophys. Res. Commun. 2014;443:1–6. doi: 10.1016/j.bbrc.2013.10.094. [DOI] [PubMed] [Google Scholar]
- 151.Takahashi N., Kobayashi M., Ogura J., Yamaguchi H., Satoh T., Watanabe K., Iseki K. Immunoprotective Effect of Epigallocatechin-3-Gallate on Oral Anticancer Drug-Induced α-Defensin Reduction in Caco-2 Cells. Biol. Pharm. Bull. 2014;37:490–492. doi: 10.1248/bpb.b13-00700. [DOI] [PubMed] [Google Scholar]
- 152.Li G.-X., Chen Y.-K., Hou Z., Xiao H., Jin H., Lu G., Lee M.-J., Liu B., Guan F., Yang Z., et al. Pro-Oxidative Activities and Dose–Response Relationship of (−)-Epigallocatechin-3-Gallate in the Inhibition of Lung Cancer Cell Growth: A Comparative Study in Vivo and in Vitro. Carcinogenesis. 2010;31:902–910. doi: 10.1093/carcin/bgq039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Shim J.-H., Su Z.-Y., Chae J.-I., Kim D.J., Zhu F., Ma W.-Y., Bode A.M., Yang C.S., Dong Z. Epigallocatechin Gallate Suppresses Lung Cancer Cell Growth through Ras-GTPase-Activating Protein SH3 Domain-Binding Protein 1. Cancer Prev. Res. 2010;3:670–679. doi: 10.1158/1940-6207.CAPR-09-0185. [DOI] [PubMed] [Google Scholar]
- 154.Milligan S.A., Burke P., Coleman D.T., Bigelow R.L., Steffan J.J., Carroll J.L., Williams B.J., Cardelli J.A. The Green Tea Polyphenol EGCG Potentiates the Antiproliferative Activity of C-Met and Epidermal Growth Factor Receptor Inhibitors in Non-Small Cell Lung Cancer Cells. Clin. Cancer Res. 2009;15:4885–4894. doi: 10.1158/1078-0432.CCR-09-0109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Shi J., Liu F., Zhang W., Liu X., Lin B., Tang X. Epigallocatechin-3-Gallate Inhibits Nicotine-Induced Migration and Invasion by the Suppression of Angiogenesis and Epithelial-Mesenchymal Transition in Non-Small Cell Lung Cancer Cells. Oncol. Rep. 2015;33:2972–2980. doi: 10.3892/or.2015.3889. [DOI] [PubMed] [Google Scholar]
- 156.Sadava D., Whitlock E., Kane S.E. The Green Tea Polyphenol, Epigallocatechin-3-Gallate Inhibits Telomerase and Induces Apoptosis in Drug-Resistant Lung Cancer Cells. Biochem. Biophys. Res. Commun. 2007;360:233–237. doi: 10.1016/j.bbrc.2007.06.030. [DOI] [PubMed] [Google Scholar]
- 157.Eom D.-W., Lee J.H., Kim Y.-J., Hwang G.S., Kim S.-N., Kwak J.H., Cheon G.J., Kim K.H., Jang H.-J., Ham J., et al. Synergistic Effect of Curcumin on Epigallocatechin Gallate-Induced Anticancer Action in PC3 Prostate Cancer Cells. BMB Rep. 2015;48:461–466. doi: 10.5483/BMBRep.2015.48.8.216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Khafif A., Schantz S.P., Chou T.C., Edelstein D., Sacks P.G. Quantitation of Chemopreventive Synergism between (−)-Epigallocatechin-3-Gallate and Curcumin in Normal, Premalignant and Malignant Human Oral Epithelial Cells. Carcinogenesis. 1998;19:419–424. doi: 10.1093/carcin/19.3.419. [DOI] [PubMed] [Google Scholar]
- 159.Somers-Edgar T.J., Scandlyn M.J., Stuart E.C., Le Nedelec M.J., Valentine S.P., Rosengren R.J. The Combination of Epigallocatechin Gallate and Curcumin Suppresses ER Alpha-Breast Cancer Cell Growth in Vitro and in Vivo. Int. J. Cancer. 2008;122:1966–1971. doi: 10.1002/ijc.23328. [DOI] [PubMed] [Google Scholar]
- 160.Wang S., Chen R., Zhong Z., Shi Z., Chen M., Wang Y. Epigallocatechin-3-Gallate Potentiates the Effect of Curcumin in Inducing Growth Inhibition and Apoptosis of Resistant Breast Cancer Cells. Am. J. Chin. Med. 2014;42:1279–1300. doi: 10.1142/S0192415X14500803. [DOI] [PubMed] [Google Scholar]
- 161.Ye F., Zhang G.-H., Guan B.-X., Xu X.-C. Suppression of Esophageal Cancer Cell Growth Using Curcumin, (−)-Epigallocatechin-3-Gallate and Lovastatin. World J. Gastroenterol. 2012;18:126–135. doi: 10.3748/wjg.v18.i2.126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Zhou D.-H., Wang X., Yang M., Shi X., Huang W., Feng Q. Combination of Low Concentration of (−)-Epigallocatechin Gallate (EGCG) and Curcumin Strongly Suppresses the Growth of Non-Small Cell Lung Cancer in Vitro and in Vivo through Causing Cell Cycle Arrest. Int. J. Mol. Sci. 2013;14:12023–12036. doi: 10.3390/ijms140612023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Du G.-J., Wang C.-Z., Qi L.-W., Zhang Z.-Y., Calway T., He T.-C., Du W., Yuan C.-S. The Synergistic Apoptotic Interaction of Panaxadiol and Epigallocatechin Gallate in Human Colorectal Cancer Cells. Phytother. Res. 2013;27:272–277. doi: 10.1002/ptr.4707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Kostin S.F., McDonald D.E., McFadden D.W. Inhibitory Effects of (−)-Epigallocatechin-3-Gallate and Pterostilbene on Pancreatic Cancer Growth in Vitro. J. Surg. Res. 2012;177:255–262. doi: 10.1016/j.jss.2012.04.023. [DOI] [PubMed] [Google Scholar]
- 165.Nair S., Barve A., Khor T.-O., Shen G.-X., Lin W., Chan J.Y., Cai L., Kong A.-N. Regulation of Nrf2- and AP-1-Mediated Gene Expression by Epigallocatechin-3-Gallate and Sulforaphane in Prostate of Nrf2-Knockout or C57BL/6J Mice and PC-3 AP-1 Human Prostate Cancer Cells. Acta Pharmacol. Sin. 2010;31:1223–1240. doi: 10.1038/aps.2010.147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Rahman A.A., Makpol S., Jamal R., Harun R., Mokhtar N., Ngah W.Z.W. Tocotrienol-Rich Fraction, [6]-Gingerol and Epigallocatechin Gallate Inhibit Proliferation and Induce Apoptosis of Glioma Cancer Cells. Molecules. 2014;19:14528–14541. doi: 10.3390/molecules190914528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Lambert J.D., Hong J., Kim D.H., Mishin V.M., Yang C.S. Piperine Enhances the Bioavailability of the Tea Polyphenol (−)-Epigallocatechin-3-Gallate in Mice. J. Nutr. 2004;134:1948–1952. doi: 10.1093/jn/134.8.1948. [DOI] [PubMed] [Google Scholar]
- 168.Lambert J.D., Kwon S.-J., Ju J., Bose M., Lee M.-J., Hong J., Hao X., Yang C.S. Effect of Genistein on the Bioavailability and Intestinal Cancer Chemopreventive Activity of (−)-Epigallocatechin-3-Gallate. Carcinogenesis. 2008;29:2019–2024. doi: 10.1093/carcin/bgn182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Peters C.M., Green R.J., Janle E.M., Ferruzzi M.G. Formulation with Ascorbic Acid and Sucrose Modulates Catechin Bioavailability from Green Tea. Food Res. Int. 2010;43:95–102. doi: 10.1016/j.foodres.2009.08.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Shim S.-M., Yoo S.-H., Ra C.-S., Kim Y.-K., Chung J.-O., Lee S.-J. Digestive Stability and Absorption of Green Tea Polyphenols: Influence of Acid and Xylitol Addition. Food Res. Int. 2012;45:204–210. doi: 10.1016/j.foodres.2011.10.016. [DOI] [Google Scholar]
- 171.Amin A.R.M.R., Wang D., Zhang H., Peng S., Shin H.J.C., Brandes J.C., Tighiouart M., Khuri F.R., Chen Z.G., Shin D.M. Enhanced Anti-Tumor Activity by the Combination of the Natural Compounds (−)-Epigallocatechin-3-Gallate and Luteolin: Potential Role of P53. J. Biol. Chem. 2010;285:34557–34565. doi: 10.1074/jbc.M110.141135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Papi A., Farabegoli F., Iori R., Orlandi M., De Nicola G.R., Bagatta M., Angelino D., Gennari L., Ninfali P. Vitexin-2-O-Xyloside, Raphasatin and (−)-Epigallocatechin-3-Gallate Synergistically Affect Cell Growth and Apoptosis of Colon Cancer Cells. Food Chem. 2013;138:1521–1530. doi: 10.1016/j.foodchem.2012.11.112. [DOI] [PubMed] [Google Scholar]
- 173.Mirzaaghaei S., Foroughmand A.M., Saki G., Shafiei M. Combination of Epigallocatechin-3-Gallate and Silibinin: A Novel Approach for Targeting Both Tumor and Endothelial Cells. ACS Omega. 2019;4:8421–8430. doi: 10.1021/acsomega.9b00224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Papież M.A., Krzyściak W. Epicatechin Acts Synergistically with Curcumin-Induced Cytogenotoxic Effect in Acute Promyelocytic Leukemia HL-60 Cell Line. J. Unexplored Med. Data. 2017;2:52. doi: 10.20517/2572-8180.2017.12. [DOI] [Google Scholar]
- 175.Luo T., Wang J., Yin Y., Hua H., Jing J., Sun X., Li M., Zhang Y., Jiang Y. (−)-Epigallocatechin Gallate Sensitizes Breast Cancer Cells to Paclitaxel in a Murine Model of Breast Carcinoma. Breast Cancer Res. 2010;12:R8. doi: 10.1186/bcr2473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Stearns M.E., Wang M. Synergistic Effects of the Green Tea Extract Epigallocatechin-3-Gallate and Taxane in Eradication of Malignant Human Prostate Tumors. Transl. Oncol. 2011;4:147–156. doi: 10.1593/tlo.10286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Rodriguez M., Du G.-J., Wang C.-Z., Yuan C.-S. Letter to the Editor: Panaxadiol’s Anticancer Activity Is Enhanced by Epicatechin. Am. J. Chin. Med. 2010;38:1233–1235. doi: 10.1142/S0192415X10008597. [DOI] [PubMed] [Google Scholar]
- 178.Mayr C., Wagner A., Neureiter D., Pichler M., Jakab M., Illig R., Berr F., Kiesslich T. The Green Tea Catechin Epigallocatechin Gallate Induces Cell Cycle Arrest and Shows Potential Synergism with Cisplatin in Biliary Tract Cancer Cells. BMC Complement. Altern. Med. 2015;15:194. doi: 10.1186/s12906-015-0721-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Shervington A., Pawar V., Menon S., Thakkar D., Patel R. The Sensitization of Glioma Cells to Cisplatin and Tamoxifen by the Use of Catechin. Mol. Biol. Rep. 2009;36:1181–1186. doi: 10.1007/s11033-008-9295-3. [DOI] [PubMed] [Google Scholar]
- 180.Qian F., Ye C.L., Wei D.Z., Lu Y.H., Yang S.L. In Vitro and in Vivo Reversal of Cancer Cell Multidrug Resistance by 2′,4′-Dihydroxy-6′-Methoxy-3′,5′-Dimethylchalcone. J. Chemother. 2005;17:309–314. doi: 10.1179/joc.2005.17.3.309. [DOI] [PubMed] [Google Scholar]
- 181.Zhang Q., Wei D., Liu J. In Vivo Reversal of Doxorubicin Resistance by (−)-Epigallocatechin Gallate in a Solid Human Carcinoma Xenograft. Cancer Lett. 2004;208:179–186. doi: 10.1016/j.canlet.2004.01.033. [DOI] [PubMed] [Google Scholar]
- 182.Liang G., Tang A., Lin X., Li L., Zhang S., Huang Z., Tang H., Li Q.Q. Green Tea Catechins Augment the Antitumor Activity of Doxorubicin in an in Vivo Mouse Model for Chemoresistant Liver Cancer. Int. J. Oncol. 2010;37:111–123. [PubMed] [Google Scholar]
- 183.Farhan M., Zafar A., Chibber S., Khan H.Y., Arif H., Hadi S.M. Mobilization of copper ions in human peripheral lymphocytes by catechins leading to oxidative DNA breakage: A structure activity study. Arch. Biochem. Biophys. 2015;580:31–40. doi: 10.1016/j.abb.2015.06.019. [DOI] [PubMed] [Google Scholar]
- 184.Farhan M., Khan H.Y., Oves M., Al-Harrasi A., Rehmani N., Arif H., Hadi S.M., Ahmad A. Cancer therapy by catechins involves redox cycling of copper ions and generation of reactive oxygen species. Toxins. 2016;8:37. doi: 10.3390/toxins8020037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Botten D., Fugallo G., Fraternali F., Molteni C. Structural Properties of Green Tea Catechins. J. Phys. Chem. B. 2015;119:12860–12867. doi: 10.1021/acs.jpcb.5b08737. [DOI] [PubMed] [Google Scholar]
- 186.Jigisha A., Nishant R., Navin K. Green tea: A magical herb with miraculous outcomes. Int. Res. J. Pharm. 2012;3:139–148. [Google Scholar]
- 187.Koch W., Kukula-Koch W., Komsta Ł., Marzec Z., Szwerc W., Głowniak K. Green tea quality evaluation based on its catechins and metals composition in combination with chemometric analysis. Molecules. 2018;23:1689. doi: 10.3390/molecules23071689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Barbosa D.S. Green tea polyphenolic compounds and human health. J. Verbr. Lebensm. 2007;2:407–413. doi: 10.1007/s00003-007-0246-z. [DOI] [Google Scholar]
- 189.Ambigaipalan P., Young W., Shahidi F. Epigallocatechin (EGC) esters as potential sources of antioxidants. Food Chem. 2020;309:125609. doi: 10.1016/j.foodchem.2019.125609. [DOI] [PubMed] [Google Scholar]
- 190.Lawless M.W., O’Byrne K.J., Gray S.G. Targeting oxidative stress in cancer. Expert Opin. Ther. Targets. 2010;14:1225–1245. doi: 10.1517/14728222.2010.526933. [DOI] [PubMed] [Google Scholar]
- 191.Kellogg J.J., Graf T.N., Paine M.F., McCune J.S., Kvalheim O.M., Oberlies N.H., Cech N.B. Comparison of Metabolomics Approaches for Evaluating the Variability of Complex Botanical Preparations: Green Tea (Camellia sinensis) as a Case Study. J. Nat. Prod. 2017;80:1457–1466. doi: 10.1021/acs.jnatprod.6b01156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Bonuccelli G., Sotgia F., Lisanti M.P. Matcha green tea (MGT) inhibits the propagation of cancer stem cells (CSCs), by targeting mitochondrial metabolism, glycolysis, and multiple cell signaling pathways. Aging. 2018;10:1867–1883. doi: 10.18632/aging.101483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Schulze J., Melzer L., Smith L., Teschke R. Green Tea and Its Extracts in Cancer Prevention and Treatment. Beverages. 2017;3:17. doi: 10.3390/beverages3010017. [DOI] [Google Scholar]
- 194.Hadi S.M., Asad S.F., Singh S., Ahmad A. A putative mechanism for anticancer and apoptosis inducing properties of plant-derived polyphenolic compounds. IUBMB Life. 2000;50:167–171. doi: 10.1080/152165400300001471. [DOI] [PubMed] [Google Scholar]
- 195.Hadi S.M., Bhat S.H., Azmi A.S., Hanif S., Shamim U., Ullah M.F. Oxidative breakage of cellular DNA by plant polyphenols: A putative mechanism for anticancer properties. Semin. Cancer Biol. 2007;17:370–376. doi: 10.1016/j.semcancer.2007.04.002. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.