Abstract
Background
Little is known about the association between respiratory events prior to diagnosis of chronic obstructive pulmonary disease (COPD) and future clinical outcomes in Japan. We investigated the association between pre-COPD diagnosis respiratory events and the incidence of exacerbations in a cohort of newly diagnosed COPD patients in Japan.
Patients and Methods
Data were retrieved from the JMDC claims database. Patients ≥40 years old with a first COPD diagnosis (≥1 hospitalization or ≥2 outpatient claims for COPD) between 2010 and 2016 were included. The incidence rate (IR) of exacerbations in patients with or without any respiratory event (including lower respiratory tract infection and respiratory failure) in the year preceding diagnosis was compared. A negative binomial model explored the association between pre-diagnosis respiratory event and IR ratio (IRR) of exacerbations.
Results
A total of 20,212 patients newly diagnosed with COPD were identified: 61% male, mean age 55 years (SD 9); of these, 955 (4.7%) had experienced ≥1 respiratory event in the year preceding diagnosis. Median duration of follow-up was 3.3 years during which the IR of exacerbations was 0.31 per patient-year (95% confidence interval [CI] 0.29–0.33) in patients with respiratory event, and 0.11 (95% CI 0.10–0.13) in patients without. The IR for severe exacerbation was nearly 10 times greater in patients with respiratory event versus without. Experiencing respiratory event pre-diagnosis was independently associated with an increased IRR of future moderate-to-severe exacerbation (adjusted IRR, 2.7; 95% CI 2.3–3.1).
Conclusion
Patients experiencing respiratory events in the year preceding COPD diagnosis should be considered at-risk of worse clinical COPD outcomes.
Keywords: COPD diagnosis, lower respiratory tract infection, exacerbation, outcome
Introduction
Acute exacerbations of chronic obstructive pulmonary disease (COPD; AECOPD) are periods of acute worsening of COPD symptoms (eg, breathlessness, cough, increased sputum volume) that extend beyond typical daily symptom variation of COPD and require a modification of treatment.1 Approximately 40% to 60% of patients with COPD experience at least one AECOPD per year.2 It was recently evidenced that even a single moderate AECOPD over a 12-month period is associated with an increased risk of future AECOPD.3–5 AECOPD are also associated with lung function decline and increased risk of mortality.6 The presence of respiratory events, namely lower respiratory tract infections (LRTIs), in patients prior to COPD diagnosis is important from a clinical standpoint. Upon initial diagnosis of COPD, one-third of patients will have experienced ≥2 major respiratory events in the past 2 years, which is substantially more frequent than in the general population.7 The Global Initiative for Chronic Obstructive Lung Disease (GOLD) recommends that the presence of frequent winter bronchitis be considered a key indicator for considering a diagnosis of COPD.1 Moreover, LRTIs may be associated with higher risk of airflow limitation8 and clinical presentation of COPD.9 However, few studies have assessed the impact of LRTIs on the pathway to exacerbation following diagnosis.10 In Japan, very few cohort studies have described the characteristics and outcomes of patients after a new diagnosis of COPD.
The objectives of the present study were (1) to describe the frequency and incidence of exacerbations (including respiratory infections) in a cohort of Japanese patients newly diagnosed with COPD and (2) to explore the association between an LRTI or acute respiratory failure in the year preceding the first recorded COPD diagnosis and the number of exacerbations post-COPD diagnosis.
Materials and Methods
This observational cohort study analyzed data from the JMDC® Claims Database in Japan, which includes information on medical claims covered by private health insurance systems since 2005.11,12 At the time of this study, data were available from 7.2 million salaried workers and their families. Information included inpatient claims (hospitalizations with duration and motive for the hospitalization, tests and procedures performed), outpatient claims (medical visits and diagnoses), and pharmacy claims (dispensed drugs and dates).
Patient-level data were provided after anonymization. The need for informed consent was waived, as data available in the database were standardized and anonymized. The study was approved by the Research Institute of Healthcare Data Science (Approval No. RI2020010).
Study Population
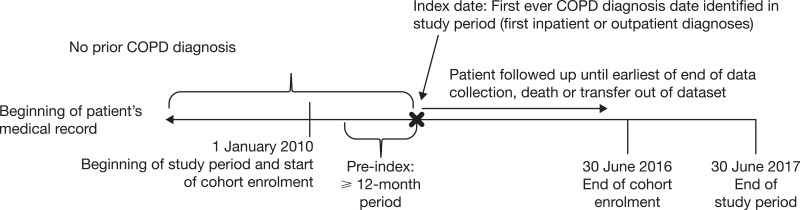
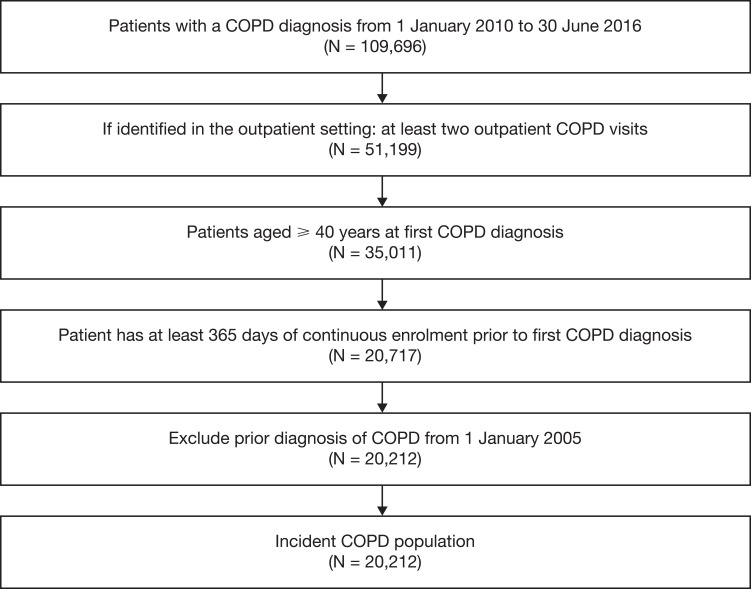
Patients with a new diagnosis of COPD between 1 January 2010 and 30 June 2016 were included in the cohort. As this study used secondary data, which are not collected directly for medical and research purposes, a combination of criteria was used. The diagnosis of COPD was approximated by the presence of (1) healthcare claims for COPD care with either ≥1 hospitalization claim or ≥2 outpatient claims with a code for COPD using the International Classification of Diseases, 10th edition (ICD-10 codes J41-J44 as primary or secondary diagnosis); the date of the first identified COPD diagnosis was the index date; and (2) age ≥40 years at index date. In turn, patients were considered as newly diagnosed with COPD if they had no ICD-10 diagnostic code for COPD any time prior to index date (going back in time since the start of data availability). Patients with <365 days of continuous data availability in the database prior to index date were excluded.
The baseline period was the 365-day period preceding index date. The follow-up period started on the day after index date and lasted until 30 June 2017 (administrative censoring), death or transfer out of the database, whichever occurred first. Figure 1 shows a schematic of the study design.
Figure 1.
Study design.
Abbreviation: COPD, chronic obstructive pulmonary disease.
Respiratory Events Before Diagnosis
The occurrence of a respiratory event during baseline was defined as having ≥1 outpatient visit or inpatient admission for (a) an LRTI (using ICD-10 codes for “influenza and pneumonia” [J09–J18, J68.0, J85.0, J85.1, J85.2, J85.3, A48.1, B01.2, B05.2, B25.0], “other acute lower respiratory infections” [J20–J22] and “status asthmaticus” [J46]) or (b) an acute respiratory failure (J80, J96) including pulmonary arrest (R09.2), in the absence of decompensated heart failure (I50), pneumothorax (J93) or myocardial infarction (I21) for the same admission, as these may mimic symptoms of an LRTI.
Outcomes
The outcome of interest was the occurrence of AECOPD (any severity, or categorized into moderate or severe) during the follow-up period. Moderate AECOPD were defined by an outpatient visit with a diagnostic code for COPD (J41–J44) and a claim for either a systemic corticosteroid or an antibiotic commonly used in treating respiratory infections with a dispensation within 7 days of the outpatient visit. Severe AECOPD were broadly defined as a hospitalization due to a respiratory event (AECOPD, LRTI, or acute respiratory failure in the absence of decompensated heart failure, pneumothorax or myocardial infarction and using the codes listed above). The definition of strictly-speaking exacerbation was extended to “respiratory infection” to minimize the risk of missing severe exacerbation events that would be coded as the co-occurring respiratory infection, if any.
Baseline Covariates
Baseline covariates included age at index date, sex, history of hospitalizations and outpatient visit (for any reason), comorbidities and respiratory medication. The presence of each comorbidity (yes/no) namely current asthma, gastroesophageal reflux disease, ulcer, lung cancer, and several cardiovascular diseases was defined as ≥1 inpatient or ≥2 outpatient claims codes during baseline. Respiratory medication use (yes/no) was approximated by the presence of ≥1 dispensation claim during baseline. These included COPD inhaled relievers (short-acting β2-agonists [SABA] or short-acting muscarinic antagonists [SAMA]), maintenance therapy (inhaled corticosteroids, long-acting β2-agonists [LABA], long-acting muscarinic antagonists [LAMA], slow-release theophylline) and expectorants.
Statistical Analyses
Statistical analyses were conducted using SAS® version 9.4 (SAS Institute Inc., Cary, North Carolina, USA). Analyses were conducted in all patients, and then separately in patients with or without a baseline respiratory event. Descriptive analyses were conducted on patients’ characteristics at baseline. To address objective 1, the number of patients experiencing at least one AECOPD (of any type, or separately for moderate and severe) during follow-up was described. The incidence rates (IRs) of AECOPD per patient-years of follow-up and 95% confidence intervals (CIs) were estimated. To address objective 2, IR ratios (IRRs) and 95% CIs for the number of AECOPD of any type during follow-up (dependent variable) in association with having ≥1 baseline respiratory event (vs none, independent variable) were estimated using negative binomial models considering overdispersion of count data; follow-up time was used as an offset term. Univariable models were built for baseline respiratory events with each potential confounder (age, sex, and baseline comorbidities) as the independent variable. Variables associated with the number of AECOPD during follow-up with a p value <0.25 were included in a multivariable model. The final multivariable model was fitted using a stepwise backward selection method, which included variables associated with the number of AECOPD during follow-up with a p value <0.05.
Results
Overall, 20,212 patients newly diagnosed with COPD were identified in the JMDC database (Figure 2). More than 90% of patients were identified with COPD using the presence of outpatient visits claims, as only 10% had a hospitalization (for COPD or not) during baseline (Table 1). Females represented 38.9% of the cohort; mean age at index date was 55.0 (SD 8.8) years. Nearly half of patients had no respiratory medication at baseline; the most frequently used respiratory medications were expectorants in 9751 (48.2%) patients and SABA in 3295 (16.3%) patients. The most frequently observed comorbidities (Table 2) were gastritis and duodenitis (44.8%), history of asthma (34.3%), gastroesophageal reflux disease (27.8%), gastric ulcer (25.6%), lung cancer (15.1%), stroke (14.1%), congestive heart failure (14.1%), and type 2 diabetes (11.1%). Less than one-third of patients had current asthma. During the baseline period, 955 (4.7%) patients had at least one respiratory event. Patients with a baseline respiratory event were older and had more comorbidities than those without. They were more frequently using respiratory medication during the baseline period, be it reliever (23% vs 16%) or maintenance medication (16% vs 13%).
Figure 2.
Patient selection and flow chart.
Abbreviation: COPD, chronic obstructive pulmonary disease.
Table 1.
Baseline Sociodemographics and Respiratory Medication in All Patients Newly Diagnosed with COPD (N = 20,212), and Patients Categorized According to Whether They Had at Least One Severe Respiratory Event During Baseline (N = 955) or Not (N = 19,257)
| Overall | No Respiratory Eventa | 1+ Respiratory Event | |
|---|---|---|---|
| N = 20,212 | N = 19,257 | N = 955 | |
| n (%) | n (%) | n (%) | |
| Sex, n (%) | |||
| Female | 7859 (38.9) | 7563 (39.3) | 296 (31.0) |
| Male | 12,353 (61.1) | 11,694 (60.7) | 659 (69.0) |
| Age at index date, years | |||
| Mean (SD) | 55.0 (8.8) | 54.9 (8.8) | 58.0 (8.8) |
| Median (IQR) | 55 (48, 62) | 55 (48, 61) | 58 (51, 65) |
| Age at index date, years | |||
| 40–49 | 6145 (30.4) | 5961 (31.0) | 184 (19.3) |
| 50–59 | 7414 (36.7) | 7065 (36.7) | 349 (36.5) |
| 60–69 | 5329 (26.4) | 5019 (26.1) | 310 (32.5) |
| ≥ 70 | 1324 (6.6) | 1212 (6.3) | 112 (11.7) |
| Respiratory medication useb | |||
| None | 9511 (47.1) | 9152 (47.5) | 359 (37.6) |
| Reliever medication | |||
| None | 16,916 (83.7) | 16,183 (84.0) | 733 (76.8) |
| SABA | 3295 (16.3) | 3073 (16.0) | 222 (23.2) |
| SAMA | 2 (0.010) | 2 (0.01) | 0 (0.0) |
| Maintenance therapy | |||
| None | 17,626 (87.2) | 16,824 (87.4) | 802 (84.0) |
| ICS+LABA | 2103 (10.4) | 1983 (10.3) | 120 (12.6) |
| ICS monotherapy | 654 (3.2) | 626 (3.3) | 28 (2.9) |
| LAMA monotherapy | 147 (0.7) | 114 (0.6) | 33 (3.5) |
| LABA monotherapy | 82 (0.4) | 75 (0.4) | 7 (0.7) |
| ICS+LABA+LAMA | 69 (0.3) | 53 (0.3) | 16 (1.7) |
| LABA+LAMA | 19 (0.09) | 13 (0.07) | 6 (0.6) |
| ICS+LAMA | 6 (0.03) | 5 (0.03) | 1 (0.1) |
| Expectorant (R05 or R05F) | 9751 (48.2) | 9215 (47.9) | 536 (56.1) |
| Slow-release theophylline | 1077 (5.3) | 1002 (5.2) | 75 (7.9) |
| History of hospitalization | |||
| 1 | 2013 (10.0) | 1705 (8.9) | 308 (32.3) |
| ≥ 2 | 1781 (8.8) | 1274 (6.6) | 507 (53.1) |
| History of outpatient visits | |||
| 1 | 801 (4.0) | 767 (4.0) | 34 (3.6) |
| ≥ 2 | 18,999 (94.0) | 18,119 (94.1) | 880 (92.1) |
Notes: aRespiratory events during baseline included lower respiratory tract infection and acute respiratory failure; bDefined as at least one dispensation claim recorded in the 365 days prior to index date.
Abbreviations: COPD, chronic obstructive pulmonary disease; ICS, inhaled corticosteroids; IQR, interquartile range; LABA, long-acting β2-agonists; LAMA, long-acting muscarinic antagonists; SABA, short-acting β2-agonists; SAMA, short-acting muscarinic antagonists; SD, standard deviation.
Table 2.
Baseline Comorbidities in All Patients Newly Diagnosed with COPD (N = 20,212), and Patients Categorized According to Whether They Had at Least One Severe Respiratory Event During Baseline (N = 955) or Not (N = 19,257)
| Overall | No Respiratory Eventa | 1+ Respiratory Event | |
|---|---|---|---|
| N = 20,212 | N = 19,257 | N = 955 | |
| n (%) | n (%) | n (%) | |
| Comorbidities | |||
| History of asthma | 6935 (34.3) | 6595 (34.2) | 340 (35.6) |
| Gastritis and duodenitis | 9047 (44.8) | 8575 (44.5) | 472 (49.4) |
| Gastroesophageal reflux disease | 5624 (27.8) | 5182 (26.9) | 442 (46.3) |
| Gastric ulcer | 5175 (25.6) | 4770 (24.8) | 405 (42.4) |
| Duodenal ulcer | 534 (2.6) | 491 (2.5) | 43 (4.5) |
| Peptic ulcer | 112 (0.6) | 98 (0.5) | 14 (1.5) |
| Bronchiectasis | 318 (1.6) | 294 (1.5) | 24 (2.5) |
| Lung fibrosis | 222 (1.1) | 187 (1.0) | 35 (3.7) |
| Lung cancer | 3046 (15.1) | 2677 (13.9) | 369 (38.6) |
| Stroke | 2850 (14.1) | 2604 (13.5) | 246 (25.8) |
| Congestive heart failure | 2831 (14.0) | 2490 (12.9) | 341 (35.7) |
| Acute myocardial infarction | 702 (3.5) | 593 (3.1) | 109 (11.4) |
| Atrial fibrillation or flutter | 616 (3.0) | 543 (2.8) | 73 (7.6) |
| Chronic ischemic heart disease | 603 (3.0) | 543 (2.8) | 60 (6.3) |
| Type 2 diabetes | 2237 (11.1) | 2025 (10.5) | 212 (22.2) |
| Depression | 1916 (9.5) | 1806 (9.4) | 110 (11.5) |
| Anxiety disorder | 1658 (8.2) | 1549 (8.0) | 109 (11.4) |
| Malignant neoplasm of pancreas | 788 (3.9) | 715 (3.7) | 73 (7.6) |
| Malignant neoplasm of breast | 479 (2.4) | 459 (2.4) | 20 (2.1) |
| Hepatic fibrosis | 405 (2.0) | 349 (1.8) | 56 (5.9) |
| Malignant neoplasm of bladder | 400 (2.0) | 380 (2.0) | 20 (2.1) |
| Malignant neoplasm of esophagus | 215 (1.1) | 179 (0.9) | 36 (3.8) |
| Current asthma | |||
| No | 14,401 (71.2) | 13,747 (71.4) | 654 (68.5) |
| Yes | 5811 (28.8) | 5510 (28.6) | 301 (31.5) |
Note: aRespiratory events during baseline included lower respiratory tract infection and acute respiratory failure.
Abbreviation: COPD, chronic obstructive pulmonary disease.
The median duration of follow-up was 3.3 years (interquartile range, IQR 2.4–4.5) in the entire population and was shorter in patients with a respiratory event at baseline (median, 2.6 years; IQR 0.9–3.8) than in patients without (median, 3.3 years; IQR 2.4–4.5). During follow-up, 4839 (24%) patients in the entire population had ≥1 AECOPD of any severity (Table 3). The percentage of patients with ≥1 AECOPD of any severity was approximately 2 times greater in patients with a baseline history of a respiratory event compared with patients without (43% vs 23%). When differentiating exacerbations by severity, the percentage of patients with moderate AECOPD during follow-up was found to be similar in patients with and without baseline respiratory events (21% vs 20%), whereas the percentage of patients with severe AECOPD was 6 times greater in patients with baseline respiratory event versus without (31% vs 5%). The IR of severe exacerbation was 10 times greater in patients with baseline respiratory event (IR = 0.20 per patient-year [95% CI 0.18–0.22]) than in patients without (IR = 0.02 [95% CI 0.02–0.02]). In the subgroup of patients with a baseline respiratory event, the IR of severe AECOPD was nearly 2 times greater than that of moderate exacerbations, with IR = 0.20 per patient-year (95% CI 0.18–0.22) and IR = 0.11 per patient-year (95% CI 0.10–0.13), respectively.
Table 3.
Frequency and Incidence of AECOPDa (Any, Moderate and Severe) During Follow-Up in All Patients (N = 20,212) and Patients Categorized According to Whether They Had at Least One Respiratory Eventb During Baseline (N = 955) or Not (N = 19,257)
| Patients with ≥ 1 AECOPD, n (%) | Total Number of Events | Patient-Years of Follow-Up | Incidence Rate of AECOPD per Patient-Year (95% CI) | |
|---|---|---|---|---|
| AECOPD of any severity | ||||
| All patients | 4839 (23.9) | 8269 | 68,611 | 0.121 (0.118–0.123) |
| No respiratory event at baseline | 4427 (23.0) | 7490 | 66,105 | 0.113 (0.111–0.116) |
| 1+ respiratory event at baseline | 412 (43.1) | 779 | 2506 | 0.311 (0.290–0.333) |
| Moderate AECOPD | ||||
| All patients | 4004 (19.8) | 6313 | 68,611 | 0.092 (0.090–0.094) |
| No respiratory event at baseline | 3804 (19.8) | 6032 | 66,105 | 0.091 (0.089–0.094) |
| 1+ respiratory event at baseline | 200 (20.9) | 281 | 2506 | 0.112 (0.100–0.126) |
| Severe AECOPD | ||||
| All patients | 1301 (6.4) | 1956 | 68,611 | 0.029 (0.027–0.030) |
| No respiratory event at baseline | 1007 (5.2) | 1458 | 66,105 | 0.022 (0.021–0.023) |
| 1+ respiratory event at baseline | 294 (30.8) | 498 | 2506 | 0.199 (0.182–0.217) |
Notes: aAECOPD during follow-up were defined as moderate (managed in the outpatient setting) or severe (defined as hospitalization for COPD exacerbation, lower respiratory tract infection or acute respiratory failure); bRespiratory events during baseline included lower respiratory tract infection and acute respiratory failure.
Abbreviations: AECOPD, acute exacerbation of chronic obstructive pulmonary disease; CI, confidence interval; COPD, chronic obstructive pulmonary disease.
Table 4 displays the crude and adjusted IRR of AECOPD (any severity) in relation to having a respiratory event at baseline (vs not). The presence of a baseline respiratory event was associated with a 2.7-times increased rate of AECOPD (95% CI 2.3–3.1), independent of older age, sex and baseline comorbidities. Sex did not appear to be a confounder in the association between the number of AECOPD during follow-up and occurrence of a respiratory event at baseline. Comorbidities independently associated with increased IRR of AECOPD included lung cancer, bronchiectasis, and lung fibrosis.
Table 4.
Risk Ratio of AECOPDa (Any Severity) During Follow-Up in Association with Respiratory Eventb (vs None) Prior to a First COPD Diagnosis, Estimated by Negative Binomial Models in a Cohort of 20,212 Newly Diagnosed COPD Patients
| Crude RR (95% CI) | Final Adjustedc RR (95% CI) | |
|---|---|---|
| Respiratory event during baseline (n = 955), versus none (n = 19,257, reference) | 3.55 (3.08–4.10) | 2.70 (2.33–3.12) |
| Age category 40–49 years (n = 6145) | 1 (reference) | 1 (reference) |
| 50–59 (n = 7414) | 0.92 (0.86–0.99) | 1.06 (0.98–1.15) |
| 60–69 (n = 5329) | 1.30 (1.20–1.40) | 1.29 (1.18–1.41) |
| ≥ 70 (n = 1324) | 1.72 (1.51–1.98) | 1.60 (1.38–1.85) |
| Male (vs female) | 1.03 (0.96–1.10) | – |
| Comorbidity at baseline (yes/no) | ||
| History of asthma (n = 6935) | 1.18 (1.10–1.27) | 1.18 (1.10–1.26) |
| Gastritis and duodenitis (n = 9047) | 1.19 (1.11–1.27) | 1.07 (1.00–1.15) |
| Gastroesophageal reflux disease (n = 5624) | 1.41 (1.31–1.52) | 1.12 (1.04–1.21) |
| Gastric ulcer (n = 5175) | 1.43 (1.32–1.54) | 1.17 (1.09–1.27) |
| Duodenal ulcer (n = 534) | 1.12 (0.91–1.39) | – |
| Peptic ulcer (n = 112) | 1.14 (0.72–1.81) | – |
| Bronchiectasis (n = 318) | 1.58 (1.24–2.02) | 1.41 (1.11–1.78) |
| Lung fibrosis (n = 222) | 3.03 (2.27–4.04) | 1.93 (1.45–2.56) |
| Lung cancer (n = 3046) | 1.85 (1.69–2.03) | 1.45 (1.32–1.59) |
| Stroke (n = 2850) | 1.23 (1.12–1.36) | – |
| Congestive heart failure (n = 2831) | 1.56 (1.42–1.72) | 1.11 (1.00–1.22) |
| Acute myocardial infarction (n = 702) | 1.48 (1.24–1.77) | – |
| Atrial fibrillation and flutter (n = 616) | 1.24 (1.02–1.50) | – |
| Chronic ischemic heart disease (n = 603) | 1.21 (0.99–1.48) | – |
| Type 2 diabetes (n = 2237) | 1.37 (1.23–1.52) | – |
| Depression (n = 1916) | 1.13 (1.01–1.26) | – |
| Anxiety disorder (n = 1658) | 1.12 (0.99–1.26) | – |
| Malignant neoplasm of pancreas (n = 788) | 1.26 (1.06–1.50) | – |
| Malignant neoplasm of breast (n = 479) | 0.95 (0.76–1.19) | – |
| Hepatic fibrosis (n = 405) | 1.59 (1.24–2.03) | – |
| Malignant neoplasm of bladder (n = 400) | 0.86 (0.67–1.11) | 0.74 (0.58–0.94) |
| Malignant neoplasm of esophagus (n = 215) | 2.29 (1.66–3.16) | 1.46 (1.07–2.00) |
Notes: aAECOPD during follow-up were defined as moderate (managed in the outpatient setting) or severe (defined as hospitalization for COPD exacerbation, lower respiratory tract infection or acute respiratory failure); bRespiratory events during baseline included lower respiratory tract infection and acute respiratory failure; cCovariates for the multivariable model were chosen using stepwise backward selection (p < 0.05).
Abbreviations: AECOPD, acute exacerbation of COPD; CI, confidence interval; COPD, chronic obstructive pulmonary disease; RR, risk ratio.
Discussion
This longitudinal observational cohort study included 20,212 patients newly diagnosed with COPD covered by private health insurance in Japan. We estimated the rate of AECOPD during follow-up and explored the association between a history of a respiratory event (LRTI or respiratory failure) prior to COPD diagnosis and the IRR of future AECOPD.
Characteristics of Newly Diagnosed Patients with COPD
In our cohort, patients were young (55 years old on average) due to recruitment of patients with a new diagnosis who were identified from an employees’ private health insurance database. Surprisingly, the prevalence of females in the present study is much higher than what was observed in hospital cohorts, which show that only 5% to 7% of Japanese COPD patients are female.13–15 Patients with current asthma were not excluded from our cohort and accounted for 29% of the cohort. Although high, this proportion is in line with recently published prospective results from Hashimoto et al, in which Japanese outpatients aged ≥ 40 years with COPD were included.16 The authors found that 36% of patients had a diagnosis of asthma made by the physician, including 25.5% with a confirmed diagnosis of asthma-COPD overlap as per the Japanese Respiratory Society diagnostic criteria.16 Gastritis and duodenitis, gastroesophageal reflux disease, and gastric ulcers were among the most commonly reported comorbidities in our cohort, and were present in more than one-half of patients. Although the prevalence of lung cancer is high in Japan – it is the third-most-frequent cancer type17,18 – the observed proportion of patients with lung cancer in our study (15%) is much higher than expected.15,19,20 It is possible that in many patients in the cohort, COPD was diagnosed upon the exploration or the treatment of lung cancer, namely because spirometry is recommended before bronchoscopy or thoracic surgery.21
Incidence of Exacerbations
During a median follow-up duration of 3.3 years, 24% of newly diagnosed patients experienced 1+ AECOPD, namely of moderate severity. As a comparison, in a cohort study conducted in the UK, more than 75% of newly diagnosed COPD patients experienced 1+ exacerbation over an average follow-up duration of 5 years.6 The IR of exacerbation of any severity observed in our study was low, with IR = 0.12 (95% CI 0.12–0.12) exacerbation per patient-year of follow-up. Although it is well known that exacerbation rates are lower in the Japanese population compared with Western countries’ populations (eg, due to genetic, ethnicity-related, and healthcare system-related factors), this rate is lower than in previous Japanese studies, which reported rates of exacerbations between 0.24 and 0.80 events per year in COPD populations, irrespective of patients’ prior history of exacerbations.22 Several reasons may explain the lower incidence of exacerbations found in the present study. First, COPD diagnosis relied on IDC-10 codes and age (≥40 years old), but not on spirometry, which was unavailable in the database. It is possible that non-COPD patients were included, thus leading to underestimation of the incidence of exacerbations. Second, only newly diagnosed, younger patients were included, and the incidence of exacerbations may be lower at an early stage of the disease. Finally, data for our study population were retrieved from an employees’ private health insurance database, which led to inclusion of individuals benefiting from better access to healthcare.
Frequency of Pre-Diagnosis Respiratory Events and Future Outcomes
Among the newly diagnosed patients, fewer than 5% had a respiratory event in the year preceding the first COPD diagnosis. These patients were more likely male and were older than patients who did not have any event. The level of cardiovascular comorbidity was markedly higher than in patients without a respiratory event, namely stroke (26% vs 14%), heart failure (36% vs 13%), acute myocardial infarction (11% vs 3%), and atrial fibrillation (8% vs 3%). Altogether, the presence of a respiratory event and of a high level of cardiovascular comorbidity may indicate more severe clinical status of COPD upon diagnosis. Moreover, the older age of this subgroup suggests that the diagnosis was made at a later stage of the disease. Because severe comorbid diseases may be associated with respiratory events, including COPD exacerbations, these characteristics were included as possible confounders in multivariable regression models. The observed association between pre-diagnosis respiratory events and subsequent exacerbations is not explained by the imbalance between the two groups at baseline, owing to these variables. However, residual confounding cannot be ruled out either.
The presence of a respiratory event in the year preceding COPD diagnosis (vs none) was infrequent but was associated with an increased rate of subsequent exacerbations. More importantly, these patients were at much higher risk of severe exacerbations, and the incidence of severe exacerbations was greater than the incidence of moderate exacerbations. The presence of a pre-diagnosis respiratory event was associated with a 2.7-fold increase in future exacerbation, independent of age and comorbidities. This result is in line with recent findings using the Clinical Practice Research Datalink database in the UK, where severe LRTI in the year preceding diagnosis was found to increase the risk of future exacerbations (IRR = 1.6; 95% CI 1.5–1.6).10
Study Limitations
In claims databases, the sensitivity and precision of identifying patients diagnosed with chronic diseases may vary. Moreover, routine clinical care diagnosis may also not always align with guidelines recommended criteria for diagnosis of COPD. Regarding COPD, the database does not contain information on spirometry, GOLD stage, or smoking status. The identification of COPD patients thus relied on a combination of diagnoses made by prescribers for claims purposes. The use of a minimum age (40 years old) and of several codes in the inclusion criteria aimed to minimize the risk of including patients that do not have GOLD-defined COPD.
In addition, the duration of COPD was not collected, nor approximated. We included patients at the moment of the first diagnosis indicated by the physician, which does not necessarily correspond to the early stage of the disease. In our study, long-acting bronchodilators were used by 16% of patients with respiratory events during baseline, suggesting that respiratory symptoms were present in the absence of, and prior to, a formal diagnosis being made. However, the period of the diagnosis is important in the course of this chronic disease as it provides the opportunity for physicians to assess the severity of the disease and anticipate future risk. In this perspective, the identification of prior respiratory events that were not identified as exacerbations of COPD is useful in terms of future risk assessment. Our results suggest that respiratory events preceding the period of the diagnosis are indicative of a more severe course of the disease once diagnosed.
Finally, the present study used data on COPD patients managed between 2010 and 2017. It is possible that patterns of COPD diagnosis or management in Japan have changed since then, thus limiting the applicability of our results to a more recent period.
Clinical Implications
The presence of a respiratory event shortly before COPD diagnosis is indicative of a higher risk of future severe exacerbations. The association between a pre-diagnosis respiratory event and subsequent exacerbations may reflect a pattern of susceptibility to severe respiratory events that remains constant in the first 3 or 4 years after diagnosis, irrespective of baseline confounders such as age at diagnosis.2 For instance, LRTIs starting early in life are associated with future subnormal lung function,8 which, in turn, may be one of the several mechanisms behind COPD.23 From this perspective, respiratory events such as LRTIs may be considered as markers of COPD risk and poorer prognosis. However, it should be noted that other potential confounders such as smoking, exposure to air pollutants or eosinophil count24 could not be accounted for in our models because data on them are not collected in the JMDC database.
Conclusions
In conclusion, patients who experience an LRTI or acute respiratory failure in the year preceding the first COPD diagnosis should be considered at higher risk of worse clinical COPD outcomes and could benefit from closer medical monitoring.
Acknowledgments
The authors would like to thank Masato Ishida and Ai Hayashi, former employees of AstraZeneca, for their contributions to the study.
Funding Statement
This study was funded by AstraZeneca.
Abbreviations
AECOPD, acute exacerbations of COPD; CI, confidence interval; COPD, chronic obstructive pulmonary disease; International Classification of Diseases, 10th edition; ICS, inhaled corticosteroids; IQR, interquartile range; IR, incidence rate; IRR, incidence rate ratio; LABA, long-acting β2-agonist; LAMA, long-acting muscarinic antagonists; LRTI, lower respiratory tract infection; RR, risk ratio; SABA, short-acting β2-agonist; SAMA, short-acting muscarinic antagonist; SD, standard deviation.
Data Sharing Statement
Data underlying the findings described in this manuscript may be obtained in accordance with AstraZeneca’s data sharing policy described at https://astrazenecagrouptrials.pharmacm.com/ST/Submission/Disclosure.
Ethics Approval and Informed Consent
Patient-level data were provided after anonymization. The need for informed consent was waived, as data available in the database were standardized and anonymized. The study was approved by the Research Institute of Healthcare Data Science (Approval No. RI2020010).
Consent for Publication
The study used electronic healthcare records routinely collected as part of usual care. No consent for publication was required from patients as the manuscript contains no identifiable details such as photograph(s) and/or videos and/or case history and/or details within the text.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Disclosure
BD, NM, CN, and HM are employees of AstraZeneca and hold stock and/or options in the company. RZ is a former employee of Evidera, which was contracted by AstraZeneca to conduct the study. SG, DL, and SH are employees of Evidera, which was contracted by AstraZeneca to conduct the study. SM has received lecture fees from AstraZeneca, GSK, Nippon Boehringer Ingelheim, Novartis Pharma, Teijin Pharma and grants from ROHTO Pharmaceutical Co., Ltd. The authors report no other conflicts of interest in this work.
References
- 1.Global Initiative for Chronic Obstructive Lung Disease. 2022 GOLD report. Global strategy for the diagnosis, management and prevention of COPD; 2022. Available from: https://goldcopd.org/2022-gold-reports-2/. Accessed February 2, 2022.
- 2.Hurst JR, Vestbo J, Anzueto A, et al. Susceptibility to exacerbation in chronic obstructive pulmonary disease. N Engl J Med. 2010;363(12):1128–1138. doi: 10.1056/NEJMoa0909883 [DOI] [PubMed] [Google Scholar]
- 3.Vogelmeier CF, Diesing J, Kossack N, Pignot M, Friedrich FW. COPD exacerbation history and impact on future exacerbations - 8-year retrospective observational database cohort study from Germany. Int J Chron Obstruct Pulmon Dis. 2021;16:2407–2417. doi: 10.2147/COPD.S322036 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sethi S, Make BJ, Robinson SB, et al. Relationship of COPD exacerbation severity and frequency on risks for future events and economic burden in the medicare fee-for-service population. Int J Chron Obstruct Pulmon Dis. 2022;17:593–608. doi: 10.2147/COPD.S350248 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Whittaker H, Rubino A, Müllerová H, et al. Frequency and severity of exacerbations of COPD associated with future risk of exacerbations and mortality: a UK routine health care data study. Int J Chron Obstruct Pulmon Dis. 2022, ;17:427–437. doi: 10.2147/COPD.S346591 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rothnie KJ, Müllerová H, Smeeth L, Quint JK. Natural history of chronic obstructive pulmonary disease exacerbations in a general practice-based population with chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2018;198(4):464–471. doi: 10.1164/rccm.201710-2029OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Yawn BP, Wollan P, Rank M. Exacerbations in the pre- and post-COPD diagnosis periods. Pragmat Obs Res. 2013;4:1–6. doi: 10.2147/POR.S41778 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Okyere DO, Bui DS, Washko GR, et al. Predictors of lung function trajectories in population-based studies: a systematic review. Respirology. 2021;26(10):938–959. doi: 10.1111/resp.14142 [DOI] [PubMed] [Google Scholar]
- 9.Sandelowsky H, Ställberg B, Nager A, Hasselström J. The prevalence of undiagnosed chronic obstructive pulmonary disease in a primary care population with respiratory tract infections - A case finding study. BMC Fam Pract. 2011;12(1):122. doi: 10.1186/1471-2296-12-122 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Whittaker H, Nordon C, Rubino A et al. Frequency and severity of respiratory infections prior to COPD diagnosis and risk of subsequent postdiagnosis COPD exacerbations and mortality: EXACOS-UK health care data study. Thorax. 2022,Oct 31. ():Epub ahead of print. doi: 10.1136/thorax-2022-219039 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.JMDC. JMDC claims database; 2022. Available from: https://www.jmdc.co.jp/en/jmdc-claims-database/. Accessed June 7, 2022.
- 12.Wakabayashi Y, Eitoku M, Suganuma N. Characterization and selection of Japanese electronic health record databases used as data sources for non-interventional observational studies. BMC Med Inform Decis Mak. 2021;21(1):167. doi: 10.1186/s12911-021-01526-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Makita H, Suzuki M, Konno S, et al. Unique mortality profile in Japanese patients with COPD: an analysis from the Hokkaido COPD cohort study. Int J Chron Obstruct Pulmon Dis. 2020;15:2081–2090. doi: 10.2147/COPD.S264437 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Haruna A, Muro S, Nakano Y, et al. CT scan findings of emphysema predict mortality in COPD. Chest. 2010;138(3):635–640. doi: 10.1378/chest.09-2836 [DOI] [PubMed] [Google Scholar]
- 15.Ono M, Kobayashi S, Hanagama M, et al. Clinical characteristics of Japanese patients with chronic obstructive pulmonary disease (COPD) with comorbid interstitial lung abnormalities: a cross-sectional study. PLoS One. 2020;15:e0239764. doi: 10.1371/journal.pone.0239764 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hashimoto S, Sorimachi R, Jinnai T, Ichinose M. Asthma and chronic obstructive pulmonary disease overlap according to the Japanese respiratory society diagnostic criteria: the prospective, observational ACO Japan cohort study. Adv Ther. 2021;38(2):1168–1184. doi: 10.1007/s12325-020-01573-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Horinouchi H, Kusumoto M, Yatabe Y, Aokage K, Watanabe S-I, Ishikura S. Lung cancer in Japan. J Thorac Oncol. 2022;17(3):353–361. doi: 10.1016/j.jtho.2021.11.020 [DOI] [PubMed] [Google Scholar]
- 18.The International Agency for Research on Cancer. GLOBOCAN 2020: Japan factsheet; 2020. Available from: https://gco.iarc.fr/today/data/factsheets/populations/392-japan-fact-sheets.pdf. Accessed May 11, 2022.
- 19.Masuda S, Omori H, Onoue A, et al. Comorbidities according to airflow limitation severity: data from comprehensive health examination in Japan. Environ Health Prev Med. 2017;22(1):13. doi: 10.1186/s12199-017-0620-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Nishida Y, Takahashi Y, Tezuka K, et al. A comprehensive analysis of association of medical history with airflow limitation: a cross-sectional study. Int J Chron Obstruct Pulmon Dis. 2017;12:2363–2371. doi: 10.2147/COPD.S138103 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hashimoto N, Matsuzaki A, Okada Y, et al. Clinical impact of prevalence and severity of COPD on the decision-making process for therapeutic management of lung cancer patients. BMC Pulm Med. 2014;14:14. doi: 10.1186/1471-2466-14-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ishii T, Nishimura M, Akimoto A, James MH, Jones P. Understanding low COPD exacerbation rates in Japan: a review and comparison with other countries. Int J Chron Obstruct Pulmon Dis. 2018;13:3459–3471. doi: 10.2147/COPD.S165187 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lange P, Ahmed E, Lahmar ZM, Martinez JF, Bourdin A. Natural history and mechanisms of COPD. Respirology. 2021;26(4):298–321. doi: 10.1111/resp.14007 [DOI] [PubMed] [Google Scholar]
- 24.Tashiro H, Kurihara Y, Takahashi K, et al. Clinical features of Japanese patients with exacerbations of chronic obstructive pulmonary disease. BMC Pulm Med. 2020;20(1):318. doi: 10.1186/s12890-020-01362-w [DOI] [PMC free article] [PubMed] [Google Scholar]