Abstract
Nonalcoholic fatty liver disease continues to be one of the major health challenges facing the world, with estimates of non-alcoholic steatohepatitis (NASH) prevalence in over 25 percent of the world’s population. NASH represents a spectrum of disease that may lead to hepatic fibrosis and eventual cirrhosis, with the risk of cirrhosis decompensation, and hepatocellular carcinoma. New therapies are desperately needed for NASH, especially for later stages of fibrosis and cirrhosis. Galectin-3 inhibition is being explored as a new liver antifibrotic therapy. This concise review will outline the state of the art of this new therapeutic target.
Keywords: Galectin-3 inhibition, Non-alcoholic fatty liver disease, Fibrosis, Macrophage
Core Tip: Galectin-3 inhibition is being advanced as a therapy for liver fibrosis and cirrhosis. Clinicians need to understand the rationale behind this new advance. This minireview will highlight the basic science, as well as recent advances in the field, including the concept of the “galectin-3 fibrosome” and the galectin-3 positive macrophage that enters the liver from the peripheral circulation in the setting of nonalcoholic fatty liver disease. Galectin-3 appears to be central to the non-alcoholic steatohepatitis fibrosis process, and inhibition of galectin-3 is imperative to curtail liver fibrosis.
INTRODUCTION
Non-alcoholic steatohepatitis (NASH) is an aggressive form of nonalcoholic fatty liver disease characterized by hepatic steatosis, ballooning hepatocytes, inflammation of hepatic lobules, and excessive deposition of fibrotic tissue. If left untreated, NASH may progress to cirrhosis and hepatocellular carcinoma (HCC), which are major causes of morbidity and mortality[1,2]. The risks for HCC are particularly worrisome in the subpopulation of NASH with diabetes, obesity[3], hypertension and dyslipidemia[4]. Thus far, there are no approved pharmacotherapeutics for the treatment of NASH and the only curative treatment for cirrhosis and early-stage HCC is a liver transplant.
NASH has a complex pathogenesis that is triggered by multiple metabolic factors, including insulin resistance, genetic factors, and lifestyle issues such as unbalanced excessive caloric intake and lack of exercise[5]. Classically, the disease has been divided into early and late stages, and investigative pharmacotherapeutics target different pathogenic metabolic pathways to gain resolution of steatohepatitis or regression of fibrosis, or ideally both processes. Regardless of the etiology or the pathway, the changeover from nonalcoholic fatty liver disease (NAFLD) to NASH leads to liver fibrosis and cirrhosis, via the transformation of the hepatic stellate cell (HSC) to an activated myofibroblast that lays down collagen. In liver fibrosis, the interaction of HSCs with other cells is complex. Liver sinusoidal endothelial cells modulate HSCs quiescence as well as fibrosis regression in the homeostatic state[6]. In the fibrotic process, apoptotic hepatocytes increase the inflammatory response and activate macrophages. Chronic liver injury leads to continuous HSCs activation, first via the resident liver macrophage, the Kupffer cell, and then via myeloid derived liver macrophages which then promote extracellular matrix (ECM) accumulation and tissue structure remodeling and resulting in progressive liver fibrosis[7].
The transforming growth factor (TGF)-β1 has been viewed as the major profibrogenic cytokine released by the liver cell upon injury, turning the HSC into a myofibroblast. A comprehensive review of liver fibrosis has recently been published[8], as well as a review of the signaling pathways and drugs targeting the various pathways in non-alcoholic steatohepatitis[9].
There are many disease processes where the galectin-3 protein has been implicated[10]. Recent data has shown galectin-3 as a direct causative agent in diverse diseases such as endometriosis[11], cardiac fibrosis and atrial fibrillation[12], and Alzheimer’s disease[13]. Galectin-3 plays a leading role in cancer progression and in the tumor microenvironment[14]. In HCC, overexpression of galectin-1 and galectin-3 have been noted[15], and galectin-3 favors tumor metastases via activation of β-catenin signaling[16].
In cirrhosis, galectin-3 has been proven to be a biomarker, in combination with other scores, to discriminate advanced cirrhosis and predict post-transplant infectious complications[17]. High tissue expression of galectin-3 was also associated with the risk of chronic liver disease and worse overall survival[18]. Blood levels of galectin-3 have not correlated as a biomarker in NASH, since other background diseases such as heart disease can raise galectin-3 levels on their own[19]. This review will focus on the role of galectin-3 in the liver fibrosis associated with NASH.
GALECTIN-3: BASIC FEATURES
Galectins are proteins that are modified in homoeostasis or under pathological conditions by adding glycans to their peptide chains, which in turn modulates their function. These proteins are known as glycoproteins, and the addition of a carbohydrate molecule to a protein molecule is known as glycosylation.
Glycosylation is critical for both cellular and extracellular activities. ‘Lectins’ are glycan binding proteins capable of recognizing distinct sugar residues, that in turn signal a cascade of molecular events. “S type” lectins, or galectins, selectively bind β- galactosides. Galectins can be found in the nucleus and the cytoplasm, as well as on the cell membrane and in the extracellular space and the ECM[20].
There are fifteen mammalian galectins that have been identified, all of them sharing a structure sequence of 130 amino acids and at least one carbohydrate recognition domain (CRD). Galectins are expressed in practically all immune cells in a constitutive or induced fashion. Galectins 1 and 3 are secreted into extracellular space[21].
Galectin-3 is a 30 kDa protein encoded by a single gene, LGALS3, located on chromosome 14, locus q21-q22. It was initially identified as MAC2 protein and is constitutively expressed on macrophages. Galectin-3 is a ‘chimera’ type of galectin, presenting one CRD, with a non CRD section of 30 N terminal amino acids, followed by 80 amino acids of tandem rich proline, tyrosine, and glycine[22].
Reviewing the galectin-3 medical literature presents challenges. The original name for galectin-3 was MAC2, and that is still being utilized. At times one also sees ‘LGALS3’ used, which is the gene that codes for galectin-3. Another term seen in the literature regarding this versatile protein is the galectin-3 binding protein, also known as MAC2 binding protein, or LGALS3BP. This 90 kD multifocal glycoprotein is a receptor ligand for galectin-3 and is present in human body fluids and appears to have a prognostic and functional role in cancer[23]. The term also seen in the literature is MAC2BPGi, the MAC2 binding protein glycan isomer that is now being used as a serum biomarker for assessing liver fibrosis in various liver diseases[24].
GALECTIN-3 ROLE IN LIVER FIBROSIS AND CIRRHOSIS, THE GALECTIN-3 FIBROSOME
The role of galectin-3 in liver fibrosis was introduced with the discovery that upregulated galectin-3 expression was temporally and spatially related to the induction of hepatic fibrosis and that disruption of the galectin-3 gene blocks myofibroblast activation and procollagen (I) expression[25]. Further confirmation of upregulation of galectin-3 expression has been obtained in other preclinical models of hepatic fibrosis[26], NASH[27] and cholestatic liver diseases[28].
In liver fibrosis, galectin-3, once secreted by several cells, including monocytes and macrophages, help activate quiescent fibroblasts to become myofibroblasts, which is the hallmark event in tissue fibrosis formation[25]. The function of galectins in fibrosis is based on the formation of oligomeric structures that lead to cross linking and lattice like structures. These lattices form a supramolecular assembly and activate different signaling pathways on the cell surface[29].
This fibrosis lattice process appears to be occurring in many disease states such as kidney fibrosis, cardiac fibrosis, and pulmonary fibrosis. In the lung fibrosis associated with coronavirus disease 2019, a macromolecular assembly on the surface of epithelial and mesenchymal cells that clusters pro-fibrotic factors has been discovered[30]. The researchers coined the term the ‘galectin-3 fibrosome’ to describe how galectin-3 oligomerizes via its N-terminal domain and binds modified glycan chains on glycoproteins on cell surfaces. The galectin-3 interactions anchor two complexes, TGF-βRII and the (CD98hc): β1-integrin complex that mediate inflammatory and fibrotic cellular responses to extracellular stimuli. TGF-βRII is a key receptor for the profibrotic cytokine TGF-β1. The CD98 heavy chain (CD98hc): β1-integrin complex mediates inflammatory cytokine responses to extracellular factors[31]. This discovery was made in idiopathic pulmonary fibrosis, and needs to be proven in hepatic fibrosis, but disruption of this ‘gal-3-fibrosome’ appears to be a promising target for new anti-fibrotic therapies.
GALECTIN-3 MACROPHAGE ACTIVATION AND LIVER FIBROSIS
The tissue-resident liver macrophages, termed Kupffer cells, represent key phagocytes that closely interact with local parenchymal, interstitial, and other immunological cells in the liver to maintain homeostasis and tolerance against harmless antigens[32]. Upon liver injury, the pool of hepatic macrophages expands dramatically by infiltrating bone marrow/monocyte-derived macrophages. The interplay of the injured microenvironment and altered macrophage pool skews the subsequent course of the liver injury[33].
Liver macrophages are laden with galectin-3[34]. The activated macrophages that enter the liver from the peripheral circulation in the setting of injury are shown to be markedly stained with galectin-3 on immunohistochemistry, and this immunohistochemistry staining is clearly different from normal liver tissue[35] (see accompanying Figure 1). Macrophage plasticity allows changes from an M1 to an M2 subtype, and that subtype performs a protective role in liver injury[36]. CD68 is the pan macrophage marker, and CD 206 is a marker for the M2 macrophage. In a Pediatric NAFLD immunofluorescence histology study, different subpopulations of hepatocytes and galectin-3 positive macrophages were correlated with distinct stages of the NAFLD to NASH disease spectrum. Researchers found that the number of α-SMA/Gal-3+ cells was significantly increased in the NASH fibrosis stage. The data reinforced a direct correlation between an increased fibrosis score and α-SMA/Gal-3+ cells in NAFLD children and supported the profibrogenic role of galectin-3[37].
Figure 1.
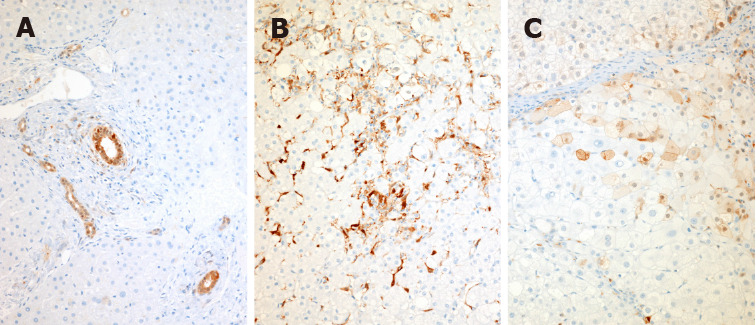
Immunohistochemical staining of normal, non-alcoholic steatohepatitis, and non-alcoholic steatohepatitis cirrhosis liver tissue with a galectin-3 antibody. A: Normal liver: In the normal liver, immunoreactive galectin-3 is present in epithelial cells of all large and small bile ducts and in periportal ductules; B: Non-alcoholic steatohepatitis (NASH) liver: In livers with inflammatory disease, staining appears in activated macrophages both in the parenchyma and in the portal tracts as well as in lymphoid germinal centers when present in chronic hepatitis. Notably there is no galectin-3 positivity in hepatocytes in pre-cirrhotic inflammatory disease, even in the presence of ballooning degeneration, Mallory-Denk bodies or features of apoptosis; C: NASH cirrhosis: Galectin-3 appears in hepatocytes and can be focal or widespread with both cytoplasmic and nuclear activity. A-C: Citation: Goodman Z, Lowe E, Boudes P. Hepatic Expression of Galectin-3, a Pro-fibrotic and Pro-inflammatory Marker: An Immunohistochemical Survey. Proceedings of the American Association for the Study of Liver Diseases meetings; 2022 Nov; Washington, US. Copyright© The Authors 2020. The authors have obtained the permission for figure using from the authors (Supplementary material).
Reading this literature also presents difficulties with the older terms M1 and M2 macrophage still being used, along with newer terms for macrophages described by their cluster of differentiation (CD 68, CD 206) as well as even newer terms depicting liver macrophages as NAM’s; NASH associated macrophages, including a subset that may be triggering receptor expressed on myeloid cells 2 positive (TREM-2+), and may promote the emergence of restorative macrophages during recovery from liver damage[38]. More recent literature involving single cell RNA sequencing has defined a subset of NAM’s that are TREM2+ and CD9+ positive and are a macrophage population that expands in fibrosis and differentiates from circulating monocytes. These macrophages are similar to ‘LAM’s; lipid associated macrophages, which surround adipose tissue and form ‘crown-like macrophages’[39].
In addition to the galectin-3 macrophage literature, further proof of the role of galectin-3 in liver fibrosis has come from live cell to cell mapping that has captured galectin-3 glycan interactions amongst live hepatic cells, the macrophage, and the stellate cells[40]. Yet another confirmation has recently been obtained from genomic studies, where a swine model of NASH had elevated levels of the LGALS3 gene that codes for galectin-3. Most interestingly only LGALS3 was associated with lipid droplet areas, suggesting a role for galectin-3 in the transition of NAFLD to NASH[41].
GALECTIN-3 INHIBITION CURRENT THERAPEUTIC APPROACHES
The rationale to block galectin-3 in hepatic fibrosis has been laid out. The conserved homology between galectins makes treatment strategy difficult, and the ubiquity of galectins in our body as well as the need to have normal galectin intracellular function, have confounded galectin inhibition therapy approaches. The role of intracellular galectin-3 in the context of fibrosis development is less well understood and the available data suggest the extracellular component is likely the main driver of its pro-fibrotic effects[42]. Although the best target area of inhibition remains unclear, blocking galectin-3 in the extracellular space and avoiding intracellular galectin-3 inhibition might indeed be the best approach[43], especially for late-stage liver disease patients who are tenuous from their liver disease and other comorbidities.
Galectin-3 inhibition involves complex sugar organic chemistry. Galectin-3 inhibitors employ either small molecules that can be given orally, or large molecules that are administered parenterally[44]. The traditional approach has been to block the CRD, as there are specific structural features in small molecule oligosaccharides that promote stronger binding to the CRD[45]. Additionally, small molecule inhibitors offer another potential advantage to be engineered to selectively bind each of the known fifteen galectins[43].
Large polysaccharide molecules can bind not only to the galectin-3 CRD, but also the N-terminal[46]. Studies have suggested that inhibiting the galectin-3 CRD alone might be inadequate, and several researchers have contended that both the N- and C- terminus of galectin-3 should be targeted to combat fibrosis[47]. Thus far there does not appear to be any safety or tolerability issues in humans associated with inhibiting galectin-3 both extracellularly and intracellularly[44]. A recent hepatic impairment study of galectin-3 inhibition revealed no safety concerns, even when administered in late-stage cirrhosis[48].
Galectin blockers are now a focus of intensive research. Studies are now integrating galectin research with a transdisciplinary approach that includes the disciple of complex sugar chemistry known as ‘Glycobiology’, along with material science, and a variety of galectin targeted biomaterials. These studies remain preclinical and have been recently reviewed[49]. For now, galectin-3 inhibition for hepatic fibrosis is moving forward in trials with several agents.
GALECTIN-3 INHIBITORS CURRENTLY IN TRIALS
The most advanced inhibitors currently in trials are belapectin and GB1107. Belapectin is a large molecule galactoarabino-rhamnogalacturonan polysaccharide inhibitor derived from natural sources. Post hoc analysis of a phase 2 belapectin study in compensated cirrhosis showed that belapectin prevented esophageal varices formation in a subgroup analysis of patients without esophageal varices at baseline, and reduced hepatic venous pressure gradient after 52 weeks of therapy[50]. A follow up international adaptive P2b/3 trial is now ongoing using the clinical endpoint of preventing esophageal varices based on endoscopic evaluation[51].
GB1107 is a small molecule thiogalactoside oral inhibitor targeting the CRD. It is advancing in P2 with a trial in cirrhotics and a first in human study with GB1211, an analogue of GB1107, is proceeding into a P2 study with cirrhosis of all etiologies[52].
CONCLUSION
This review centered around the evolving role of the galectin-3 and the hepatic Gal3+ macrophage at the center of the liver fibrotic pathway. Cell to cell interactions between the hepatocyte, the macrophage, and the stellate cells initiate the transformation of the stellate cell into a myofibroblast that lays down collagen in the ECM. Genomics, transcriptomics, proteomics, immunohistochemistry staining, and live cell to cell mapping have confirmed the vital role of galectin-3 in liver fibrosis. The concept of the ‘galectin-3 fibrosome’ has been illuminated, and the role of the galectin-3 positive macrophage in liver fibrosis continues to evolve. The picture is filling in but is by no means complete.
A confounding factor for those researching this topic is that the medical literature is confused by older terms still being employed for the same process, both for galectin-3, and for the hepatic Gal3+ macrophage. The author believes an international consensus needs to be achieved on nomenclature as this field moves forward. It is apparent that galectin-3 inhibition for liver fibrosis and cirrhosis will continue to be a fertile target of clinical research. Given galectin-3’s role in HCC and HCC metastatic spread, it is intriguing to speculate that galectin-3 inhibition might have protective effects against HCC development in cirrhosis, as well as a potential future role in adjunctive HCC therapy. In the next few years, data from upcoming galectin-3 inhibition trials will determine whether the future of NASH therapy includes this promising antifibrotic approach.
ACKNOWLEDGEMENTS
The author acknowledges Dr. Pol Boudes for reviewing this manuscript and Dr. Zachary Goodman for providing the galectin-3 antibody histological images.
Footnotes
Conflict-of-interest statement: The author reports no relevant conflicts of interest for this article.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Corresponding Author’s Membership in Professional Societies: American College of Gastroenterology, American Association for the Study of Liver Diseases.
Peer-review started: October 6, 2022
First decision: December 12, 2022
Article in press: February 8, 2023
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: United States
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Mijailović NR, Serbia; Xing HC, China S-Editor: Wang JJ L-Editor: A P-Editor: Wang JJ
References
- 1.Said A, Ghufran A. Epidemic of non-alcoholic fatty liver disease and hepatocellular carcinoma. World J Clin Oncol. 2017;8:429–436. doi: 10.5306/wjco.v8.i6.429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.White DL, Kanwal F, El-Serag HB. Association between nonalcoholic fatty liver disease and risk for hepatocellular cancer, based on systematic review. Clin Gastroenterol Hepatol. 2012;10:1342–1359.e2. doi: 10.1016/j.cgh.2012.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cholankeril G, Patel R, Khurana S, Satapathy SK. Hepatocellular carcinoma in non-alcoholic steatohepatitis: Current knowledge and implications for management. World J Hepatol. 2017;9:533–543. doi: 10.4254/wjh.v9.i11.533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Takuma Y, Nouso K. Nonalcoholic steatohepatitis-associated hepatocellular carcinoma: our case series and literature review. World J Gastroenterol. 2010;16:1436–1441. doi: 10.3748/wjg.v16.i12.1436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lonardo A, Leoni S, Alswat KA, Fouad Y. History of Nonalcoholic Fatty Liver Disease. Int J Mol Sci. 2020;21 doi: 10.3390/ijms21165888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Furuta K, Guo Q, Hirsova P, Ibrahim SH. Emerging Roles of Liver Sinusoidal Endothelial Cells in Nonalcoholic Steatohepatitis. Biology (Basel) 2020;9 doi: 10.3390/biology9110395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Thibaut R, Gage MC, Pineda-Torra I, Chabrier G, Venteclef N, Alzaid F. Liver macrophages and inflammation in physiology and physiopathology of non-alcoholic fatty liver disease. FEBS J. 2022;289:3024–3057. doi: 10.1111/febs.15877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Friedman SL, Pinzani M. Hepatic fibrosis 2022: Unmet needs and a blueprint for the future. Hepatology. 2022;75:473–488. doi: 10.1002/hep.32285. [DOI] [PubMed] [Google Scholar]
- 9.Yang YY, Xie L, Zhang NP, Zhou D, Liu TT, Wu J. Updates on novel pharmacotherapeutics for the treatment of nonalcoholic steatohepatitis. Acta Pharmacol Sin. 2022;43:1180–1190. doi: 10.1038/s41401-022-00860-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sciacchitano S, Lavra L, Morgante A, Ulivieri A, Magi F, De Francesco GP, Bellotti C, Salehi LB, Ricci A. Galectin-3: One Molecule for an Alphabet of Diseases, from A to Z. Int J Mol Sci. 2018;19 doi: 10.3390/ijms19020379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Yamashita S, Hashimoto K, Sawada I, Ogawa M, Nakatsuka E, Kawano M, Kinose Y, Kodama M, Sawada K, Kimura T. Endometrial galectin-3 causes endometriosis by supporting eutopic endometrial cell survival and engraftment in the peritoneal cavity. Am J Reprod Immunol. 2022;87:e13533. doi: 10.1111/aji.13533. [DOI] [PubMed] [Google Scholar]
- 12.Lee KN, Kim DY, Boo KY, Kim YG, Roh SY, Baek YS, Kim DH, Lee DI, Shim J, Choi JI, Hwang GS, Kim YH. Therapeutic implications of galectin-3 in patients with atrial fibrillation. Sci Rep. 2022;12:784. doi: 10.1038/s41598-022-04894-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Boza-Serrano A, Vrillon A, Minta K, Paulus A, Camprubí-Ferrer L, Garcia M, Andreasson U, Antonell A, Wennström M, Gouras G, Dumurgier J, Cognat E, Molina-Porcel L, Balasa M, Vitorica J, Sánchez-Valle R, Paquet C, Venero JL, Blennow K, Deierborg T. Galectin-3 is elevated in CSF and is associated with Aβ deposits and tau aggregates in brain tissue in Alzheimer's disease. Acta Neuropathol. 2022;144:843–859. doi: 10.1007/s00401-022-02469-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ephraim R, Fraser S, Nurgali K, Apostolopoulos V. Checkpoint Markers and Tumor Microenvironment: What Do We Know? Cancers (Basel) 2022;14 doi: 10.3390/cancers14153788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Setayesh T, Colquhoun SD, Wan YY. Overexpression of Galectin-1 and Galectin-3 in hepatocellular carcinoma. Liver Res. 2020;4:173–179. doi: 10.1016/j.livres.2020.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Song M, Pan Q, Yang J, He J, Zeng J, Cheng S, Huang Y, Zhou ZQ, Zhu Q, Yang C, Han Y, Tang Y, Chen H, Weng DS, Xia JC. Galectin-3 favours tumour metastasis via the activation of β-catenin signalling in hepatocellular carcinoma. Br J Cancer. 2020;123:1521–1534. doi: 10.1038/s41416-020-1022-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.An Y, Xu S, Liu Y, Xu X, Philips CA, Chen J, Méndez-Sánchez N, Guo X, Qi X. Role of Galectins in the Liver Diseases: A Systematic Review and Meta-Analysis. Front Med (Lausanne) 2021;8:744518. doi: 10.3389/fmed.2021.744518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Cervantes-Alvarez E, Limon-de la Rosa N, Vilatoba M, Pérez-Monter C, Hurtado-Gomez S, Martinez-Cabrera C, Argemi J, Alatorre-Arenas E, Yarza-Regalado S, Tejeda-Dominguez F, Lizardo-Thiebaud MJ, Mendez-Guerrero O, Gamboa-Dominguez A, Aguilar-Salinas CA, Huang CA, Kershenobich D, Bataller R, Torre A, Navarro-Alvarez N. Galectin-3 is overexpressed in advanced cirrhosis and predicts post-liver transplant infectious complications. Liver Int. 2022;42:2260–2273. doi: 10.1111/liv.15326. [DOI] [PubMed] [Google Scholar]
- 19.Chen H, Chen C, Fang J, Wang R, Nie W. Circulating galectin-3 on admission and prognosis in acute heart failure patients: a meta-analysis. Heart Fail Rev. 2020;25:331–341. doi: 10.1007/s10741-019-09858-2. [DOI] [PubMed] [Google Scholar]
- 20.Radhakrishnan A, Park K, Kwak IS, Jaabir M, Sivakamavalli J. Classification of Lectins. In: Elumalai P, Lakshmi S. Lectins. Singapore: Springer, 2021. [Google Scholar]
- 21.Johannes L, Jacob R, Leffler H. Galectins at a glance. J Cell Sci. 2018;131:jcs208884. doi: 10.1242/jcs.208884. [DOI] [PubMed] [Google Scholar]
- 22.Sun MJ, Cao ZQ, Leng P. The roles of galectins in hepatic diseases. J Mol Histol. 2020:51: 473–484. doi: 10.1007/s10735-020-09898-1. [DOI] [PubMed] [Google Scholar]
- 23.Capone E, Iacobelli S, Sala G. Role of galectin 3 binding protein in cancer progression: a potential novel therapeutic target. J Transl Med. 2021;19:405. doi: 10.1186/s12967-021-03085-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Behairy OG, El-Gendy SA, Ibrahim DY, Mansour AI, El-Shimi OS. Mac-2 binding protein glycan isomer as noninvasive tool to assess liver fibrosis in children with chronic liver disease. Hepatol Res. 2021;51:277–283. doi: 10.1111/hepr.13608. [DOI] [PubMed] [Google Scholar]
- 25.Henderson NC, Mackinnon AC, Farnworth SL, Poirier F, Russo FP, Iredale JP, Haslett C, Simpson KJ, Sethi T. Galectin-3 regulates myofibroblast activation and hepatic fibrosis. Proc Natl Acad Sci U S A. 2006;103:5060–5065. doi: 10.1073/pnas.0511167103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Jeftic I, Jovicic N, Pantic J, Arsenijevic N, Lukic ML, Pejnovic N. Galectin-3 Ablation Enhances Liver Steatosis, but Attenuates Inflammation and IL-33-Dependent Fibrosis in Obesogenic Mouse Model of Nonalcoholic Steatohepatitis. Mol Med. 2015;21:453–465. doi: 10.2119/molmed.2014.00178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Pejnovic N, Jeftic I, Jovicic N, Arsenijevic N, Lukic ML. Galectin-3 and IL-33/ST2 axis roles and interplay in diet-induced steatohepatitis. World J Gastroenterol. 2016;22:9706–9717. doi: 10.3748/wjg.v22.i44.9706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Tian J, Yang G, Chen HY, Hsu DK, Tomilov A, Olson KA, Dehnad A, Fish SR, Cortopassi G, Zhao B, Liu FT, Gershwin ME, Török NJ, Jiang JX. Galectin-3 regulates inflammasome activation in cholestatic liver injury. FASEB J. 2016;30:4202–4213. doi: 10.1096/fj.201600392RR. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Nabi IR, Shankar J, Dennis JW. The galectin lattice at a glance. J Cell Sci. 2015;128:2213–2219. doi: 10.1242/jcs.151159. [DOI] [PubMed] [Google Scholar]
- 30.Professor Bibek Gooptu. Leicester Institute of Structural & Chemical Biology. [cited 11 August 2020]. Available from: https://m.youtube.com/watch?v=PXfZr1KXjP8 .
- 31.Miah A, Stylianou P, Tongue P, Roach KM, Bradding P, Gooptu B. S89 Ex vivo studies of the gal-3-fibrosome hypothesis in IPF and non-fibrotic control lung tissue and myofibroblasts. Thorax. 2019;74:A57.1–A57. [Google Scholar]
- 32.Li H, Zhou Y, Wang H, Zhang M, Qiu P, Zhang R, Zhao Q, Liu J. Crosstalk Between Liver Macrophages and Surrounding Cells in Nonalcoholic Steatohepatitis. Front Immunol. 2020;11:1169. doi: 10.3389/fimmu.2020.01169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wen Y, Lambrecht J, Ju C, Tacke F. Hepatic macrophages in liver homeostasis and diseases-diversity, plasticity and therapeutic opportunities. Cell Mol Immunol. 2021;18:45–56. doi: 10.1038/s41423-020-00558-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Traber PG, Zomer E. Therapy of experimental NASH and fibrosis with galectin inhibitors. PLoS One. 2013;8:e83481. doi: 10.1371/journal.pone.0083481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Goodman Z, Lowe E, Boudes P. Hepatic Expression of Galectin-3, a Pro-fibrotic and Pro-inflammatory Marker: An Immunohistochemical Survey. Proceedings of the American Association for the Study of Liver Diseases meetings; 2022 Nov; Washington, US. [Google Scholar]
- 36.Barreby E, Chen P, Aouadi M. Macrophage functional diversity in NAFLD - more than inflammation. Nat Rev Endocrinol. 2022;18:461–472. doi: 10.1038/s41574-022-00675-6. [DOI] [PubMed] [Google Scholar]
- 37.de Oliveira FL, Panera N, De Stefanis C, Mosca A, D'Oria V, Crudele A, De Vito R, Nobili V, Alisi A. The Number of Liver Galectin-3 Positive Cells Is Dually Correlated with NAFLD Severity in Children. Int J Mol Sci. 2019;20 doi: 10.3390/ijms20143460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Coelho I, Duarte N, Barros A, Macedo MP, Penha-Gonçalves C. Trem-2 Promotes Emergence of Restorative Macrophages and Endothelial Cells During Recovery From Hepatic Tissue Damage. Front Immunol. 2020;11:616044. doi: 10.3389/fimmu.2020.616044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Hundertmark J, Berger H, Tacke F. Single Cell RNA Sequencing in NASH. Methods Mol Biol. 2022;2455:181–202. doi: 10.1007/978-1-0716-2128-8_15. [DOI] [PubMed] [Google Scholar]
- 40.Joeh E, O'Leary T, Li W, Hawkins R, Hung JR, Parker CG, Huang ML. Mapping glycan-mediated galectin-3 interactions by live cell proximity labeling. Proc Natl Acad Sci U S A. 2020;117:27329–27338. doi: 10.1073/pnas.2009206117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Herrera-Marcos LV, Martínez-Beamonte R, Macías-Herranz M, Arnal C, Barranquero C, Puente-Lanzarote JJ, Gascón S, Herrero-Continente T, Gonzalo-Romeo G, Alastrué-Vera V, Gutiérrez-Blázquez D, Lou-Bonafonte JM, Surra JC, Rodríguez-Yoldi MJ, García-Gil A, Güemes A, Osada J. Hepatic galectin-3 is associated with lipid droplet area in non-alcoholic steatohepatitis in a new swine model. Sci Rep. 2022;12:1024. doi: 10.1038/s41598-022-04971-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Slack RJ, Mills R, Mackinnon AC. The therapeutic potential of galectin-3 inhibition in fibrotic disease. Int J Biochem Cell Biol. 2021;130:105881. doi: 10.1016/j.biocel.2020.105881. [DOI] [PubMed] [Google Scholar]
- 43.Stegmayr J, Zetterberg F, Carlsson MC, Huang X, Sharma G, Kahl-Knutson B, Schambye H, Nilsson UJ, Oredsson S, Leffler H. Extracellular and intracellular small-molecule galectin-3 inhibitors. Sci Rep. 2019;9:2186. doi: 10.1038/s41598-019-38497-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Al Attar A, Antaramian A, Noureddin M. Review of galectin-3 inhibitors in the treatment of nonalcoholic steatohepatitis. Expert Rev Clin Pharmacol. 2021;14:457–464. doi: 10.1080/17512433.2021.1894127. [DOI] [PubMed] [Google Scholar]
- 45.Dings RPM, Miller MC, Griffin RJ, Mayo KH. Galectins as Molecular Targets for Therapeutic Intervention. Int J Mol Sci. 2018;19 doi: 10.3390/ijms19030905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Miller MC, Zheng Y, Yan J, Zhou Y, Tai G, Mayo KH. Novel polysaccharide binding to the N-terminal tail of galectin-3 is likely modulated by proline isomerization. Glycobiology. 2017;27:1038–1051. doi: 10.1093/glycob/cwx071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Nangia-Makker P, Hogan V, Balan V, Raz A. Chimeric galectin-3 and collagens: Biomarkers and potential therapeutic targets in fibroproliferative diseases. J Biol Chem. 2022;298:102622. doi: 10.1016/j.jbc.2022.102622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Galectin Therapeutics Inc. A Single-dose, Open-label, Pharmacokinetic Study of Belapectin (GR-MD-02) in Subjects With Normal Hepatic Function and Subjects With Varying Degrees of Hepatic Impairment. [accessed 2022 Aug 25]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://www.clinicaltrials.gov/ct2/show/NCT04332432. ClinicalTrials.gov Identifier: NCT04332432.
- 49.Martin-Saldaña S, Chevalier MT, Pandit A. Therapeutic potential of targeting galectins - A biomaterials-focused perspective. Biomaterials. 2022;286:121585. doi: 10.1016/j.biomaterials.2022.121585. [DOI] [PubMed] [Google Scholar]
- 50.Chalasani N, Abdelmalek MF, Garcia-Tsao G, Vuppalanchi R, Alkhouri N, Rinella M, Noureddin M, Pyko M, Shiffman M, Sanyal A, Allgood A, Shlevin H, Horton R, Zomer E, Irish W, Goodman Z, Harrison SA, Traber PG Belapectin (GR-MD-02) Study Investigators. Effects of Belapectin, an Inhibitor of Galectin-3, in Patients With Nonalcoholic Steatohepatitis With Cirrhosis and Portal Hypertension. Gastroenterology. 2020;158:1334–1345.e5. doi: 10.1053/j.gastro.2019.11.296. [DOI] [PubMed] [Google Scholar]
- 51.Galectin Therapeutics Inc. Study Evaluating the Efficacy and Safety of Belapectin for the Prevention of Esophageal Varices in NASH Cirrhosis (NAVIGATE). [accessed 2022 Aug 25]. In: ClinicalTrials.gov [Internet]. Bethesda (MD): U.S. National Library of Medicine. Available from: https://clinicaltrials.gov/ct2/show/NCT04365868. ClinicalTrials.gov Identifier: NCT04365868.
- 52.Galecto Therapeutics Inc. Characterization of the Novel Galectin-3 inhibitor GB1107 on CCL4 induced liver fibrosis in mice. Proceedings of International Liver Congress; 2022 June; Leeds, UK. London. [Google Scholar]


