Abstract
Background
Identifying COVID-19 correlates of protection and immunity thresholds is important for policy makers and vaccine development. We aimed to identify correlates of protection of BNT162b2 (Pfizer–BioNTech) vaccination against COVID-19.
Methods
In this prospective cohort study, households within a radius of 40 km of the Sheba Medical Center in Israel in which a new SARS-CoV-2 infection (defined as the index case) was detected within the previous 24 h were approached between July 25 and Nov 15, 2021. We included adults (aged >18 years) who had received one or two vaccine doses, had an initial negative SARS-CoV-2 PCR and no previous infection reported, and had a valid IgG and neutralising antibody result. The exposure of interest was baseline immune status, including IgG antibody concentration, neutralising antibody titre, and T-cell activation. The outcomes of interest were PCR-positive SARS-CoV-2 infection between day 2 and day 21 of follow-up and intensity of disease symptoms (self-reported via a telephone questionnaire) among participants who had a confirmed infection. Multivariable logistic and ordered logit ordinal regressions were used for the adjusted analysis. To identify immunological thresholds for clinical protection, we estimated the conditional probability of infection and moderate or severe disease for individuals with pre-exposure IgG and neutralising antibody concentrations above each value observed in the study data.
Findings
From 16 675 detected index cases in the study region, 5718 household members agreed to participate, 1461 of whom were eligible to be included in our study. 333 (22·8%) of 1461 household members who were not infected with SARS-CoV-2 at baseline were infected within 21 days of follow-up. The baseline (pre-exposure) IgG and neutralising antibodies were higher in participants who remained uninfected than in those who became infected (geometric mean IgG antibody concentration 168·2 binding antibody units [BAU] per mL [95% CI 158·3–178·7] vs 130·5 BAU/mL [118·3–143·8] and geometric mean neutralising antibody titre 197·5 [181·9–214·4] vs 136 ·7 [120·3–155·4]). Increasing IgG and neutralising antibody concentrations were also significantly associated with a reduced probability of increasing disease severity. Odds of infection were significantly reduced each time baseline IgG antibody concentration increased by a factor of ten (odds ratio [OR] 0·43 [95% CI 0·26–0·70]) and each time baseline neutralising antibody titre increased by a factor of two (0·82 [0·74–0·92]). In our cohort, the probability of infection if IgG antibody concentrations were higher than 500 BAU/mL was 11% and the probability of moderate disease severity was 1%; the probability of infection if neutralising antibody titres were above or equal to 1024 was 8% and the probability of moderate disease severity was 2%. T-cell activation rates were not significantly associated with reduced probability of infection (OR 1·04, 95% CI 0·83–1·30).
Interpretation
Both IgG and neutralising antibodies are correlates of protection against SARS-CoV-2 infection. Our data suggest that IgG concentrations higher than 500 BAU/mL and neutralising antibody titres of 1024 or more are thresholds for immunological protection from SARS-CoV-2 delta variant infection. Potentially, updated protective thresholds against emerging variants of concern could be calculated, which could support decision makers on administration of new vaccination strategies and on the optimal period between vaccine doses.
Funding
Israeli Ministry of Health.
Introduction
SARS-CoV-2 was first identified in December, 2019, in Wuhan, China, and subsequently spread globally.1 On March 11, 2020, WHO declared COVID-19 a global pandemic. In December, 2020, emergency-use authorisation was given for the BNT162b2 (Pfizer–BioNTech) vaccine, followed by ten additional vaccines.2 The early initial effectiveness of these COVID-19 vaccines quickly became evident. 1 month following vaccination, vaccine effectiveness of mRNA vaccines was approximately 95%, in both clinical studies and nationwide surveillance data.3, 4, 5 However, 5–6 months following vaccination, a rapid decline in vaccine effectiveness was recorded in parallel with waning of the humoral immune response.6, 7 In 2021 and 2022, population-based8 and individual-based9, 10 studies showed that higher neutralising antibody and IgG antibody concentrations are correlated with lower breakthrough infection rates following a second dose of BNT162b2 than lower concentrations, suggesting that the amount of IgG antibodies and neutralising antibodies could indicate the degree of protection from SARS-CoV-2 infection. Identifying COVID-19 correlates of protection and immunity thresholds is important for informing vaccine policy, targeting at-risk populations for vaccination, deciding on boosting strategies, and aiding in the development of new vaccines.
Research in context.
Evidence before this study
The BNT162b2 (Pfizer–BioNTech) vaccine was authorised for emergency use against COVID-19 by the US Food and Drug Administration on Dec 11, 2020. Despite vaccination programmes being rolled out worldwide, at least five variants of concern have caused large COVID-19 surges. Because indoor environments in which individuals have sustained close contact increase the risk of SARS-CoV-2 infection, within-household transmission is particularly important to study. In this prospective cohort study, we investigate the immune correlates of protection in the household environment. Before undertaking the study, we searched PubMed, and the medRxiv and bioRxiv preprint servers, for articles published from December, 2020 to July 1, 2021 (ie, just before the study began) using the keywords “coronavirus disease 2019” or “COVID-19” or “SARS-CoV-2” and “correlates of protection” and “antibody” and “household”. We used no language restrictions. We found six studies, but only two were done in the household setting. Most of the studies assessed seroprevalence after a household case was identified. Two studies compared viral load kinetics of infection. Only one study attempted to correlate pre-infection immune status with risk of infection. Yet, this study was conducted in the pre-vaccine era in Nicaragua. The authors reported higher protection among seropositive (previously infected) household members with 69·2% (95% CI 60·7–75·9) protection from any infection, and 79·4% (64·9–87·9) protection against moderate or severe infection. No studies reported antibody titres before infection. Therefore, no studies evaluated the correlates of protection of vaccine-induced immune responses in the household setting.
Added value of this study
To our knowlege, this is the first prospective cohort study across different ages, sexes, and comorbidities that evaluated immune correlates of protection from SARS-CoV-2 infection in a vaccinated population in the household setting. In this study, we examined the association between immune status of household members before exposure to a household member with confirmed SARS-CoV-2 infection, and their subsequent risk of infection and disease severity after exposure. To identify protective immunological thresholds, we estimated the conditional probability of infection and moderate to severe disease for individuals with pre-exposure IgG antibody and neutralising antibody concentrations above each value observed in the study data. We found that baseline antibody concentrations were higher in those who remained uninfected, the odds of infection were significantly reduced each time the baseline IgG antibody concentration increased by a factor of ten (odds ratio 0·43, 95% CI 0·26–0·70), and the odds of infection were significantly reduced each time the baseline neutralising antibody titre increased by a factor of two (0·82, 0·74–0·92). The probability of being infected, among highly exposed household members with undetectable IgG antibody concentrations was 64% and decreased linearly—with increasing IgG antibody concentrations—to 17% in those with concentrations above 500 BAU/mL. Similarly, the probability of being infected decreased to 7% for individuals with baseline neutralising antibody titres of at least 1024.
Implications of all the available evidence
Identifying COVID-19 correlates of protection and immunity thresholds is important for policy makers and vaccine development, and can help estimate the optimal period between each vaccine dose to help prevent surges of new COVID-19 variants, especially for clinically vulnerable populations. This study indicates that both IgG and neutralising antibodies are correlates of protection and that IgG antibody concentrations above 500 BAU or neutralising antibody titres of at least 1024 largely protect against SARS-CoV-2 delta variant infection. Although the current dominant variants are omicron subvariants, this study provides evidence for the correlation of antibody concentrations with disease severity and a proof of concept that would probably hold true for other variants of concern including omicron and its subvariants in light of the cross-protection between SARS-CoV-2 variants.
In this prospective household cohort study, conducted during the delta SARS-CoV-2 variant surge in Israel, we followed up vaccinated and previously uninfected household contacts of a verified index case and aimed to estimate the association between their baseline immune status (ie, IgG antibody concentrations, neutralising antibody titres, and cell-mediated immunity) and the risk of subsequent infection.
Methods
Study design and participants
The Israeli COVID-19 family study (ICoFS) was a prospective cohort study done during the delta (B.1.617.2) SARS-CoV-2 variant surge of the COVID-19 pandemic in Israel (from July 25 to Nov 15, 2021). Households whose residence was within a radius of 40 km of the Sheba Medical Center (Ramat-Gan, Israel), in which a new SARS-CoV-2 infection (defined as the index case) was detected within the previous 24 h, were approached. All household members older than 18 years who signed an informed consent form underwent a SARS-CoV-2 nasopharyngeal swab, PCR, and serology testing (appendix pp 3–4). Households included any individuals that were living in the same house.
We only included participants with an initial negative SARS-CoV-2 PCR who had not been previously infected. Other inclusion criteria were a valid IgG and neutralising antibody result and receipt of one or two doses of the BNT162b2 vaccine. Accordingly, the index case within each household was not included in the study, but rather only served to ensure a substantial amount of exposure within the household. At the time of the study, PCR testing was free and widely available. Additionally, individuals were highly motivated to be tested, because those who had been infected with SARS-CoV-2 received a green pass that allowed participation in social activities regardless of vaccination status.
The protocol (SMC-8130-21) was approved by the Institutional Review Board of the Sheba Medical Center and written informed consent was obtained from all study participants.
Procedures
The day on which the index infection was diagnosed was defined as day 0 of follow-up. Data and samples were collected in four phases. In phase 1 (pre-study data), SARS-CoV-2 PCR results of all participants were retrieved from the national registry between day –14 and day 0. In phase 2 (baseline data), between day 0 and day 2 a research team, which included a nurse and a paramedic, visited the households, received written informed consent, and obtained nasopharyngeal and blood samples from all index cases and household members who were older than 18 years. In phase 3 (continuous active surveillance), between day 2 and day 21, household members who had a negative PCR test in phase 1 were offered a second PCR test by a research team at their home or at a nearby clinic regardless of symptoms. In phase 4 (infection data collection), between day 20 and day 60, a follow-up telephone questionnaire was administered by study coordinators to all participants to collect data on past vaccination, isolation practices, baseline medical conditions and symptoms, and severity of disease if infected anywhere in Israel. Data on gender were self-reported, with three available options (ie, male, female, or other).
The units of observation in the study were individuals who were not infected with SARS-CoV-2 at baseline, residing within the household of at least one individual with SARS-CoV-2 infection that had been confirmed in the previous 24 h. The exposure of interest was the baseline immune status (designated as pre-exposure measures), including IgG antibody concentration, neutralising antibody titre, and T-cell activation. The outcomes of interest were PCR-positive SARS-CoV-2 infection between day 2 and 21 of follow-up and intensity of disease symptoms among those who had a confirmed infection. To ensure more complete detection of the outcome, the data we collected were linked with national COVID-19 data, which include all COVID-19 PCR tests conducted anywhere in Israel.
Information about symptoms was collected from participants infected during the study (appendix pp 5–7). The intensity of disease symptoms was categorised as negligible, mild, moderate, or severe. Moderate and severe infections were defined following WHO criteria.11 Negligible infection was defined as the presentation of up to three symptoms (excluding fever, shortness of breath, need for oxygen support, and admission to hospital). Mild infection was defined as the presentation of four to nine symptoms (excluding fever above 37·5°C if fever lasted for more than 48 h, need for oxygen support, and admission to hospital). Moderate infection was defined as the presentation of at least ten symptoms or if fever was above 37·5°C for more than 48 h. Severe infection was defined as a requirement for oxygen support or admission to hospital.
Additional variables collected and used for adjustment of the statistical models include the number of vaccine doses, time since the last vaccine dose, number of comorbidities, BMI, and quarantine practices within the household (ie, whether individuals with COVID-19 were quarantined from the exposed household member). We defined full quarantine if the index case answered yes to four questions on sleeping, eating, using the toilet, and wearing masks (questions 38–41 in questionnaire; appendix p 7). Partial quarantine was defined if yes was answered to at least one question. No quarantine was defined if no was answered to all four questions.
Nasopharyngeal swabs were obtained, and SARS-CoV-2 mRNA was identified by quantitative real-time PCR via the Allplex 2019-nCoV platform (Seegene, South Korea). Blood samples were tested using the SARS-CoV-2 IgG II Quant (Abbott, IL, USA) test according to the manufacturer's instructions and expressed as binding antibody units (BAU) per the WHO standard. SARS-CoV-2 pseudovirus neutralisation assay against the SARS-CoV-2 parental strain was performed as described using a green fluorescent protein reporter-based pseudotyped virus with a vesicular stomatitis virus backbone coated with SARS-CoV-2 spike protein.12 Values above 23 BAU/mL and above a titre of 8 were considered positive for IgG and neutralising antibodies, respectively. Of all samples, 15% were randomly selected and assessed for T-cell activation; enzyme-linked immune absorbent spot was used to measure antigen-specific T cells that secrete IFN-γ. Due to high costs and requirement for rapid shipment of whole blood samples to the laboratory, 15% of samples were randomly selected on the basis of the time of sampling. A stimulation index lower than or equal to 2 was considered negative, whereas a stimulation index higher than 2 was considered positive. Additional information about the laboratory tests is provided in the appendix (pp 3–4).
Statistical analysis
Details of the sample size calculation performed during the design of the study are included in the appendix (p 2). Exposures were log-transformed, using base 10 for IgG antibody concentrations and base 2 for neutralising antibody titres and T-cell activity. The distribution of variables among the study population was described as appropriate for the variable types.
For the crude analysis, the geometric mean and 95% CIs of each immunological marker at baseline were compared by infection (yes or no) and by disease severity (no infection, negligible infection, mild infection, or moderate infection). For the adjusted analysis, the association between baseline (pre-exposure or pre-infection) immune status and infection was estimated using a multivariable logistic regression model. The association between baseline immune status and disease severity (no infection, negligible infection, mild infection, or moderate infection) was estimated using an ordered logit ordinal regression. All models were adjusted for age, sex, BMI, the existence of comorbidities (yes or no), quarantine within the household (no, partial, or full), days since last vaccine dose, and household sizes. Days since vaccination and household size were continuous variables. The models were clustered by households by including a random effect. For each exposure (ie, IgG concentrations, neutralising antibody titres, and T-cell activity), a separate model was fit among individuals for whom data regarding that particular immunological marker were available. Missing data in other variables were multiply imputed five times, with the analysis performed on each imputed dataset and the results pooled using Rubin's rules.13
To identify immunological thresholds for clinical protection, we estimated the conditional probability of infection and substantial disease for individuals with baseline IgG antibody and neutralising antibody concentrations above each value observed in the study data. This probability was calculated for the study population as a whole and separately for households in which the index patient was not isolated to ensure a substantial exposure. Using the observed thresholds to discretise the different exposures, we then fit a logistic regression model, similar to the one used for the adjusted analysis, with the lowest antibody value as the baseline. We report the odds ratio (OR) as the exponentiated coefficient.
Role of the funding source
The funder was involved in data collection and in the proofreading of the manuscript; they had no role in study design, data analysis, or data interpretation.
Results
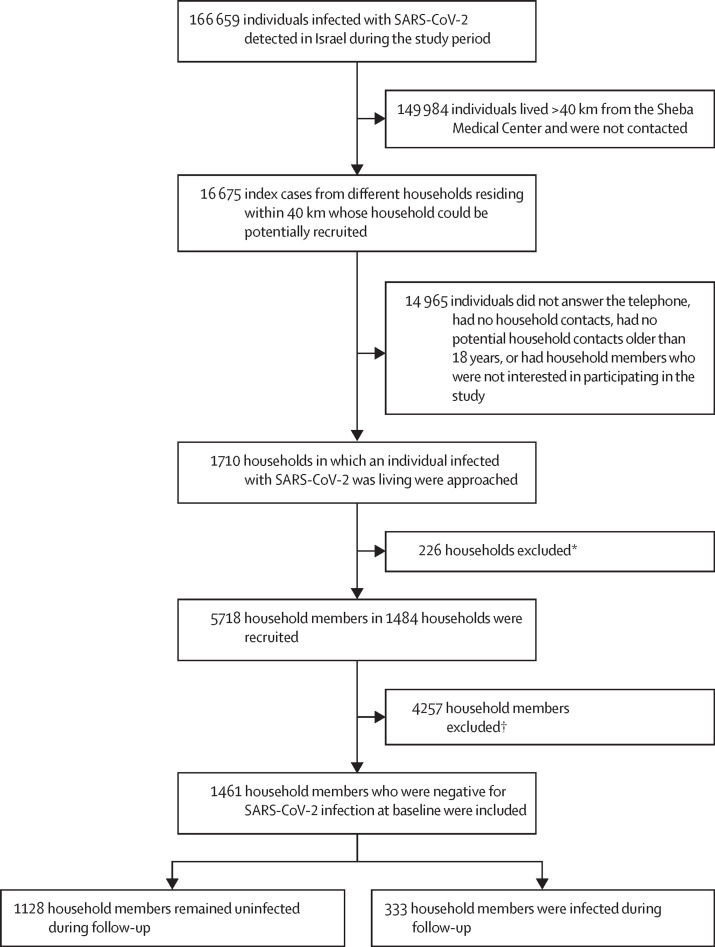
Between July 25 and Nov 15, 2021, 166 659 individuals were infected with SARS-CoV-2 in Israel (figure 1 ). We contacted 16 675 of these individuals (ie, index cases) by telephone. A total of 1710 households agreed to participate. 1484 were eventually recruited, with a total of 5718 household members. 1461 of these household members were included in the study (figure 1; table 1 ). Because only five individuals with confirmed SARS-CoV-2 infection had a severe infection, they were categorised together with participants who had moderate disease. Therefore, only three categories were used in the final analysis: negligible, mild, and moderately severe infection.
Figure 1.
Study design flow chart
*226 households were approached by telephone and agreed to participate but were not recruited due to technical difficulties. †The total of 4257 household members excluded in the current study include: individuals who were positive for SARS-CoV-2 infection at baseline (n=926), individuals who were 18 years or younger (n=1787), individuals who had received a third vaccine dose (n=306), individuals who were unvaccinated (n=66), and individuals who were not available for serology (n=1372).
Table 1.
Baseline characteristics of study population
| Individuals not infected with SARS-CoV-2 (n=1128) | Individuals infected with SARS-CoV-2 (n=333) | p value | ||
|---|---|---|---|---|
| Age, years | 42 (34–49) | 41 (36–48) | 0·84 | |
| Sex | .. | .. | 0·33 | |
| Female | 603 (53·5%) | 188 (56·5%) | .. | |
| Male | 525 (46·5%) | 145 (43·5%) | .. | |
| BMI, kg/m2* | 24·5 (22·0–27·6) | 24·9 (22·5–28·3) | 0·052 | |
| Number of vaccine inoculations | .. | .. | <0·0001 | |
| One dose | 53 (4·7%) | 0 | .. | |
| Two doses | 1075 (95·0%) | 333 (100·0%) | .. | |
| Time since last vaccine dose, days | 177 (162–193) | 180 (164–194) | 0·089 | |
| Number of comorbidities† | .. | .. | 0·036 | |
| None | 812/979 (83·0%) | 259 (77·5%) | .. | |
| One or more | 167/979 (17·0%) | 74 (22·5%) | .. | |
| Quarantine of index case during illness‡ | .. | .. | <0·0001 | |
| No quarantine | 287/1007 (29·0%) | 196/328 (60·0%) | .. | |
| Partial quarantine | 196/1007 (19·0%) | 42/328 (13·0%) | .. | |
| Full quarantine | 524/1007 (52·0%) | 90/328 (27·0%) | .. | |
| Number of people in household | 4 (3–5) | 4 (3–5) | 0·81 | |
| Number of index cases in household | 1·09 (0·35) | 1·18 (0·52) | <0·0001 | |
Data are n (%), median (IQR), or mean (SD).
BMI data not available for 176 individuals without SARS-CoV-2 infection and four individuals with SARS-CoV-2 infection.
Data on comorbidities not available for 149 individuals without SARS-CoV-2 infection.
Quarantine data not available for 121 individuals without SARS-CoV-2 infection and five individuals with SARS-CoV-2 infection.
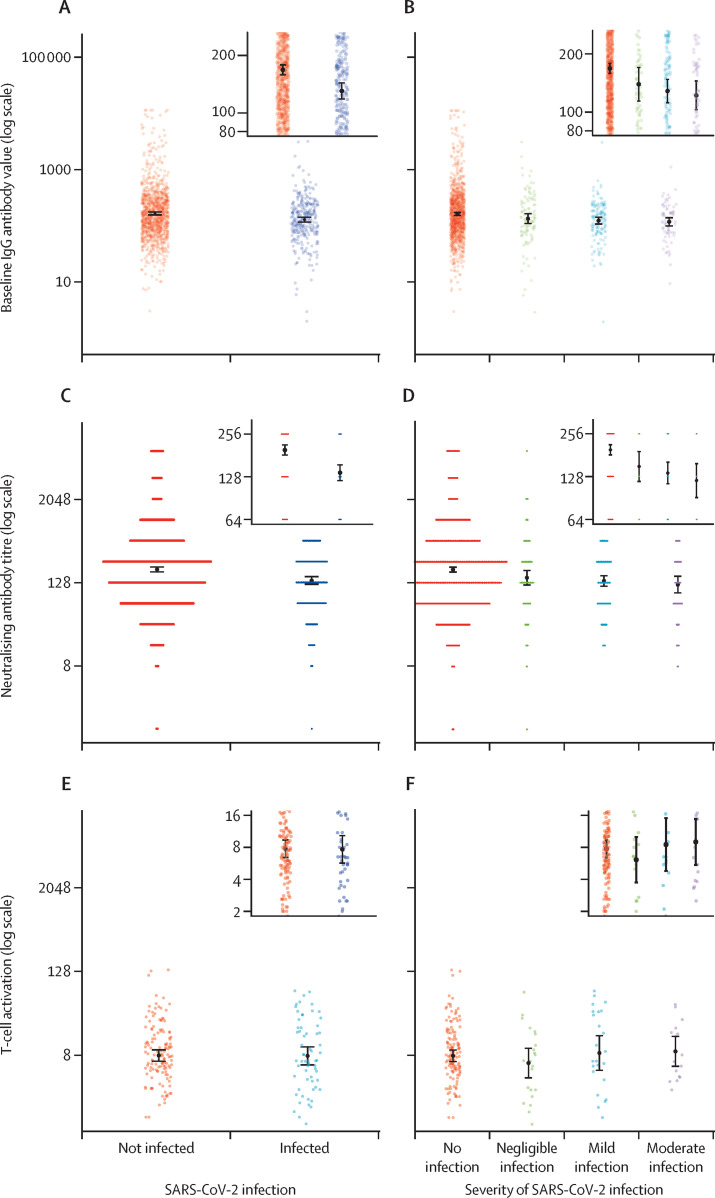
Geometric mean antibody concentrations at baseline were higher in participants who did not become infected with SARS-CoV-2 than those who were infected during the follow-up period of 21 days (IgG antibodies 168·2 BAU/mL [95% CI 158·3–178·7] vs 130·5 BAU/mL [118·3–143·8] and neutralising antibody titres 197·5 [181·9–214·4] vs 136·7 [120·3–155·4]). Moreover, baseline antibody titres were inversely correlated with disease severity. Participants with negligible disease had IgG antibody titres of 139·2 BAU/mL (113·9–170·0) compared with 122·1 BAU/mL (102·9–144·8) in those with moderate disease, and neutralising antibody titres of 150·8 (118·4–192·20) versus 120·1 (91·2–158·1) in those with moderate disease. T-cell activation rates did not differ between participants who did not become infected with SARS-CoV-2 (stimulation index of 7·8 [95% CI 6·4–9·4]) and those who were infected during follow-up (7·7 [5·7–10·3]). T-cell activation was also not correlated with disease severity (stimulation index of 6·1 [3·7–9·9] for negligible infection vs 9·0 [5·5–14·8] for moderate infection; figure 2E, F ; appendix pp 12–13).
Figure 2.
Baseline immune markers by infection outcome
(A) IgG antibody concentrations in individuals without and with SARS-CoV-2 infection. (B) IgG antibody concentrations by severity of disease. (C) Neutralising antibody titres in individuals without and with SARS-CoV-2 infection. (D) Neutralising antibody titres by severity of disease. (E) T-cell activation in individuals without and with SARS-CoV-2 infection. (F) T-cell activation by severity of disease. Geometric means are represented by black dots, and the 95% CI is represented by the bars.
Multivariable analyses estimated that higher IgG antibody concentration and higher neutralising antibody titres were significantly associated with a reduced probability of infection (table 2 ). For IgG antibody concentrations, the OR of infection each time antibody concentrations increased by a factor of ten was 0·43 (95% CI 0·26–0·70). For neutralising antibody titres, the OR of infection each time the antibody titre increased by a factor of two was 0·82 (0·74–0·92). T-cell activation rates were not significantly associated with reduced probability of infection (1·04, 0·83–1·30; table 2)
Table 2.
Multivariable analysis for prediction of infection
| OR (95% CI) | p value | ||
|---|---|---|---|
| Prediction by IgG antibody titre | |||
| IgG values, log10 | 0·43 (0·26–0·70) | <0·0006 | |
| Age | 0·94 (0·76–1·16) | 0·58 | |
| Sex | |||
| Male | 1 (ref) | .. | |
| Female | 1·39 (0·97–1·98) | 0·072 | |
| BMI | 1·17 (0·96–1·43) | 0·12 | |
| Comorbidities* | |||
| None | 1 (ref) | .. | |
| One or more | 1·37 (0·85–2·19) | 0·20 | |
| Quarantine within the household | |||
| No quarantine | 1 (ref) | .. | |
| Partial quarantine | 0·17 (0·09–0·33) | <0·0001 | |
| Full quarantine | 0·13 (0·07–0·23) | <0·0001 | |
| Household size | 0·95 (0·78–1·15) | 0·60 | |
| Days from second vaccine to infection day† | 1·12 (0·89–1·40) | 0·33 | |
| Prediction by neutralising antibody titre | |||
| Neutralising antibody titres, log2 | 0·82 (0·74–0·92) | <0·0004 | |
| Age | 0·99 (0·81–1·24) | 0·97 | |
| Sex | |||
| Male | 1 (ref) | .. | |
| Female | 1·48 (1·03–2·13) | 0·036 | |
| BMI | 1·19 (0·97–1·46) | 0·10 | |
| Comorbidities* | |||
| None | 1 (ref) | .. | |
| One or more | 1·30 (0·81–2·09) | 0·28 | |
| Quarantine within the household | |||
| No quarantine | 1 (ref) | .. | |
| Partial quarantine | 0·17 (0·09–0·33) | <0·0001 | |
| Full quarantine | 0·14 (0·08–0·24) | <0·0001 | |
| Household size | 0·95 (0·78–1·16) | 0·62 | |
| Days from second vaccine to infection day† | 1·10 (0·88–1·38) | 0·40 | |
| Prediction by T-cell activation | |||
| T-cell activation, log10 | 1·04 (0·3–1·0) | 0·72 | |
| Age | 0·72 (0·43–1·21) | 0·22 | |
| Sex | |||
| Male | 1 (ref) | .. | |
| Female | 1·74 (0·77–3·92) | 0·18 | |
| BMI | 1·30 (0·74–2·31) | 0·34 | |
| Comorbidities* | |||
| None | 1 (ref) | .. | |
| One or more | 1·43 (0·53–3·91) | 0·48 | |
| Quarantine within the household | |||
| No quarantine | 1 (ref) | .. | |
| Partial quarantine | 0·16 (0·04–0·61) | 0·0079 | |
| Full quarantine | 0·14 (0·06–0·34) | <0·0001 | |
| Household size | 1·03 (0·74–1·45) | 0·86 | |
| Days from second vaccine to infection day† | 1·22 (0·77–1·91) | 0·39 | |
OR=odds ratio.
Comorbidities included hypertension, dyslipidaemia, diabetes, heart disease, lung disease, coagulation disorder, liver disease, renal failure, autoimmune disease, and immunosuppression. The full questionnaire and definitions of comorbidities are in the appendix (pp 5–7).
This analysis was only done in participants who had two doses.
Additionally, ordered logistic regression for prediction of severity of disease estimated that higher IgG antibody concentrations and neutralising antibody titres were significantly associated with a reduced probability of increasing disease severity (table 3 ). For IgG antibody concentrations, the OR of increasing disease severity each time the antibody concentration increased by a factor of ten was 0·45 (95% CI 0·29–0·71). For neutralising antibody titres, the OR of increasing disease severity each time antibody titre increased by a factor of two was 0·83 (0·75–0·91).
Table 3.
Ordered logistic ordinal regression for prediction of severity of disease
| OR (95% CI) | p value | ||
|---|---|---|---|
| Prediction by IgG antibody titre | |||
| IgG values, log10 | 0·45 (0·29–0·71) | <0·005 | |
| Age | 0·91 (0·75–1·12) | 0·38 | |
| Sex | |||
| Male | 1 (ref) | 1 (ref) | |
| Female | 1·48 (1·06–2·06) | 0·022 | |
| BMI | 1·21 (1·00–1·46) | 0·045 | |
| Comorbidities* | |||
| None | 1 (ref) | 1 (ref) | |
| One or more | 1·38 (0·89–2·14) | 0·16 | |
| Quarantine within the household | |||
| No quarantine | 1 (ref) | 1 (ref) | |
| Partial quarantine | 0·18 (0·10–0·33) | <0·0001 | |
| Full quarantine | 0·14 (0·09–0·23) | <0·0001 | |
| Prediction by neutralising antibody titre | |||
| Neutralising antibody titres, log2 | 0·83 (0·75–0·91) | <0·0002 | |
| Age | 0·96 (0·78–1·17) | 0·69 | |
| Sex | |||
| Male | 1 (ref) | 1 (ref) | |
| Female | 1·57 (1·11–2·21) | 0·0099 | |
| BMI | 1·23 (1·01–1·50) | 0·0035 | |
| Comorbidities* | |||
| None | 1 (ref) | 1 (ref) | |
| One or more | 1·31 (0·84–2·05) | 0·23 | |
| Quarantine within the household | |||
| No quarantine | 1 (ref) | 1 (ref) | |
| Partial quarantine | 0·18 (0·10–0·32) | <0·0001 | |
| Full quarantine | 0·15 (0·09–0·24) | <0·0001 | |
OR=odds ratio.
Comorbidities included hypertension, dyslipidaemia, diabetes, heart disease, lung disease, coagulation disorder, liver disease, renal failure, autoimmune disease, and immunosuppression. The full questionnaire and definitions of comorbidities are in the appendix (pp 5–7).
Another variable that was found to be significantly associated with infection and disease severity among exposed household members was the index household member not being quarantined from the rest of the household (Table 2, Table 3).
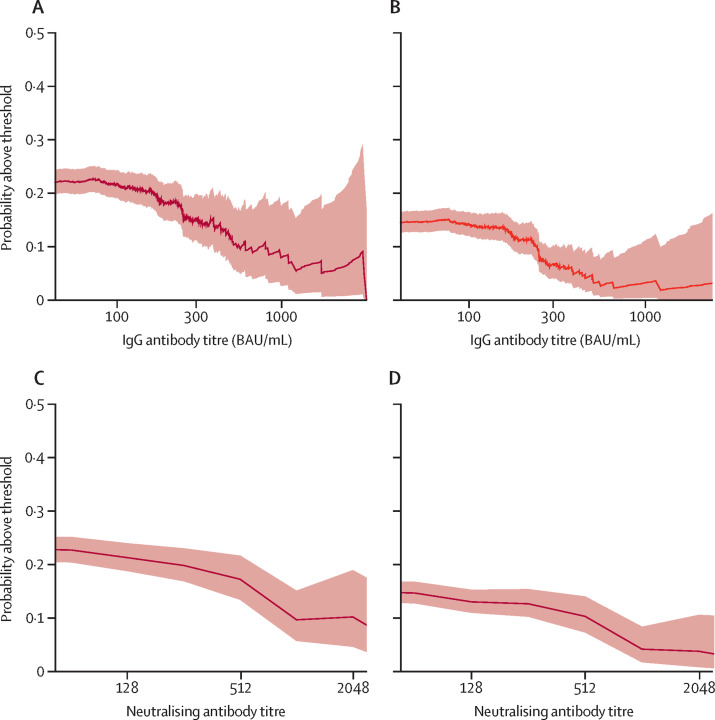
The probability of a household contact becoming infected among all exposed household members given undetected baseline IgG antibody concentrations (<23 BAU/mL) was 38% (13/34), and the probability among only those who were exposed to index cases who were not isolating given undetected baseline IgG antibody concentrations (<23 BAU/mL) was 64% (9/14). This probability decreased with increasing IgG antibody concentrations. If the contact's baseline IgG antibody concentration was higher than 500 BAU/mL, the probability of that contact becoming infected among all exposed household members dropped to 11% (14/128) and the probability among household members exposed to non-isolated index cases in the household dropped to 17% (8/48). Similarly, the probability of having moderate symptomatic disease decreased with increasing IgG antibody concentrations, dropping to 4% (7/169) for all exposed participants with baseline IgG concentrations between 300 BAU/mL and 500 BAU/mL, and dropping to 9% (5/57) for those exposed to non-isolating index cases (ie, in households with no quarantine) with baseline IgG concentrations between 300 BAU/mL and 500 BAU/mL. For individuals with baseline IgG concentrations higher than 500 BAU/mL, the probability of having moderate symptomatic disease further decreased to 1% (1/128) for all participants and to 0% for those exposed to non-isolating index cases (figure 3 ; appendix pp 9, 12–13). Similarly, for baseline neutralising antibody titres of up to 512, the probability of a household contact becoming infected among all participants was 24% (320/1336) and the probability among only those who were exposed to index cases who were not isolating was approximately 43% (189/438). If baseline neutralising antibody titres were higher than or equal to 1024, the probability of a household contact becoming infected among all participants dropped to 8% (5/60) and the probability among only those who were exposed to index cases who were not isolating dropped to 7% (2/27). Among contacts with neutralising antibody titres of up to 512, the probability for moderate symptomatic disease was 6% (77/1336) among all participants and 11% (50/438) among only those who were exposed to index cases who were not isolating. Among contacts with baseline neutralising antibody titres higher than or equal to 512, the probability of moderate disease severity among all exposed household members dropped to 2% (1/60) and the probability among only those who were exposed to index cases who were isolating dropped to 0% (figure 3; appendix pp 9, 12–13).
Figure 3.
Probability of a household contracting SARS-CoV-2 infection with or without moderate symptoms
The probability of getting infected (A) or having moderate symptoms (B) based on pre-infection IgG concentrations. The probability of getting infected (C) or having moderate symptoms (D) based on pre-infection neutralising antibody titres. The solid line denotes the calculated probability (odds ratio) given the increasing concentrations and the shaded areas denote the 95% CI. BAU=binding antibody units.
Adjusted models show that for individuals with baseline IgG antibody concentrations higher than 500 BAU/mL, the odds of infection are 5·6 lower than for individuals with IgG antibody concentrations below baseline concentration (ie, <24 BAU/mL; OR 0·18, 95% CI 0·05–0·70; appendix pp 10–11). For individuals with neutralising antibody titres of at least 1024, the odds of infection are 9·1 times lower than for individuals with neutralising antibody titres of 64 and lower (0·11, 0·03–0·48; appendix pp 10–11).
Discussion
In this prospective cohort study, we found that IgG antibody concentrations and neutralising antibody titres are significantly associated with the risk of SARS-CoV-2 infection and could accordingly serve as correlates of protection for infection and intensity of symptomatic disease. We did not find a correlation between T-cell activation and SARS-CoV-2 infection; however, we cannot exclude the possibility that the study was underpowered to identify this association. Moreover, we identified potential antibody thresholds that might indicate a substantially lower probability of being infected by the SARS-CoV-2 delta variant, even after exposure to a non-isolated household member who was infected with SARS-CoV-2.
It has been previously reported that IgG antibodies and neutralising antibodies are correlated with the risk of SARS-CoV-2 infection.8, 9, 10, 14, 15, 16 However, the strength of the current study is the availability of a large cohort of highly exposed individuals (with an index case in the household) who were followed up for 21 days, for whom blood samples were obtained shortly before infection. Moreover, data on symptoms of those who were infected also allowed us to assess the correlation with symptomatic disease. Furthermore, previous studies have mostly described the correlation of antibody concentrations with infection within the first 2 months after vaccination. In our study, most of the population was vaccinated by a second BNT162b2 dose approximately 6 months before the study, allowing assessment of correlates of protection during a period of waning of the immune response.
Our crude analysis showed that participants who did not get infected had higher antibody concentrations than those who did get infected, and that a reduction in antibody concentrations increases the risk of infection. The correlation of both IgG and neutralising antibody concentrations with disease intensity shows that even if antibody concentrations are not high enough to protect from infection, they can reduce the severity of disease. Overall, the results of this study, together with the shown waning of humoral response following the second vaccine dose,6, 17 highlight the importance of high antibody concentrations for protection against SARS-CoV-2 infection. Booster doses—which have been shown to be effective for increasing antibody concentrations,18, 19—are currently the most important course of action for increased protection against infection. Using adjusted models, we investigated the effect of antibody concentrations on the probability of infection beyond other factors such as BMI, time from infection, age, sex, comorbidities, and quarantine measures. Isolation was a secondary factor used to control for confounding of the main exposures. However, isolation was significantly associated with protection from infection. In household settings, isolation of the index case has been reported to protect other household members from infection.20 Although the overall probability of infection was greater when the household member infected with SARS-CoV-2 did not isolate than when they did isolate, the association between increased antibody titres and a lowered probability of infection also persisted when isolation did not occur.
We did not find a significant correlation between T-cell activation and protection from infection, although our study might have been underpowered to detect this effect due to the small sample size of the T-cell cohort. Studies published between 2021 and 2022 showed high SARS-CoV-2-specific T-cell activation following severe disease21 and impaired protection from infection in rhesus macaques (Macaca mulatta) following CD8 T-cell depletion,22 suggesting that cellular immunity can contribute to protection against SARS-CoV-2. It was proposed that memory T cells might protect populations from severe infections,23, 24 perhaps when antibody titres are waning. In our study, only five participants with confirmed SARS-CoV-2 infection had severe disease and T-cell activation was measured in only one of these individuals, precluding analysis of a correlation between T-cell activation and severe disease. Future studies in older adults or in those who are immunocompromised should assess the role of cellular response in this regard.
Our results show that among individuals with baseline (ie, pre-exposure) IgG antibody concentrations higher than 500 BAU/mL and neutralising antibody titres higher than 512, the probability of infection is substantially lower than the probability for individuals with lower concentrations, and the probability of having substantial symptoms is extremely low. These results suggest that individuals with IgG or neutralising antibody concentrations above these thresholds could be largely protected from symptomatic disease. Indeed, in our cohort, IgG concentrations of at least 500 BAU/mL or neutralising antibody titres of above 512 served as an 89% protective threshold from moderately severe disease. However, due to broad 95% CIs, we were not able to estimate a 90% population protective threshold. Instead, we showed that individuals with IgG antibody concentrations above 500 BAU/mL are 5·6 times more protected than individuals with concentrations less than 23 BAU/mL, and that individuals with neutralising antibody titres of 1024 or greater are 9·1 times more protected than individuals with titres less than 64. Because neutralisation is a functional assay, we believe this threshold is true for every infection, including infection by the omicron SARS-CoV-2 variant of concern. Unfortunately, because neutralising antibodies against omicron are approximately eight to ten times lower than neutralising antibodies against the ancestral strain even after a third vaccine dose,25 these thresholds are not easily achieved, and therefore many symptomatic infections occur and protection is largely against severe disease and death.26 As shown by a study of health-care workers who received a second booster vaccine (four vaccine doses), the higher the antibody concentrations the greater the protection against mild symptomatic disease.27 Regarding IgG antibodies, different variants of concern would probably have different thresholds. However, the IgG antibody threshold established in our study against the SARS-CoV-2 delta variant, in combination with serological studies assessing the correlation between neutralising and binding antibodies following different vaccine regimens and past infections,28, 29, 30, 31 might be used as a basis to calculate the threshold needed against other variants of concern.
Although our study did not assess the correlation between antibody concentrations and protection from infection following hybrid immunity (ie, natural infection and vaccination), it is noteworthy that hybrid immunity resulting from previous infection and three vaccine doses was shown to provide the best protection from symptomatic infection and severity of disease from omicron subvariants BA.1 and BA.2 when compared with mRNA vaccination or previous infection.32 This finding could suggest that infection and vaccination provide a combined immune effect. Because cumulative infection rate increased dramatically with the emergence of omicron variants, the implications of hybrid immunity for public health might be substantial and should be investigated in this context in the future.
Our study has several limitations. First, the study was done during a period in which the delta SARS-CoV-2 variant of concern was predominant and included individuals who received a maximum of two doses of the BNT162b2 vaccine. These factors render interpretation of the study a challenge in the current reality (ie, one in which the dominant variant is omicron and many individuals have been vaccinated with more than two doses). Second, the study did not include individuals with a known previous infection, or individuals who were 18 years or younger, limiting the generalisability of our findings. Third, we could not assess correlates of protection from severe disease or death because the number of participants with these outcomes was low and a much larger cohort would be required; therefore, assessing the clinical significance of correlates of protection in reducing admissions to hospital and death is not possible. Fourth, despite the adjustment performed, residual confounding is possible, for example, due to physical characteristics within the household that are not well captured in our data. Fifth, outcome misclassification is possible, particularly for asymptomatic infections, although active surveillance was performed, and the study was performed in a period when a positive result was certified by a green pass allowing societal advantages. An additional limitation of this study is that the effect estimates of a given individual also depend on the exposure of other individuals in the same household (ie, interference), because infection of additional household members raises the probability of other household members also becoming infected.
In conclusion, our results indicate that IgG antibodies and neutralising antibodies are correlates of protection and that IgG antibody concentrations above 500 BAU/mL or neutralising antibody titres of 1024 or higher largely protect against infection with the delta SARS-CoV-2 variant of concern. Despite reduced neutralising antibody concentrations of ancestor BNT162b2 vaccine recipients,33 there is substantial cross-protection against omicron variants following booster vaccination.34 Therefore, this study provides evidence for the correlation of antibody concentrations with disease severity and a proof of concept that would probably hold true for other variants of concern, including omicron and its subvariants.
Data sharing
The data and study protocol will be shared after publication, upon request. Only deidentified, relevant, individual participant data will be available. Data will be available by email from the corresponding author.
Declaration of interests
GRY served as a member of an advisory board for Moderna, and received consulting fees from Medison and speaking fees from Teva, MSD, Pfizer, AstraZeneca, and Medison. YL received a grant from Pfizer outside the submitted work. All other authors declare no competing interests.
Acknowledgments
Acknowledgments
This study was funded by the Israeli Ministry of Health. We acknowledge Vered Roa and Limor Ben-David and their teams for coordinating the study and comprehensive data and sample collection, the Ministry of Health team for retrieving data and for full cooperation on this study, and the Sheba Management team for assisting in the logistics. We thank Gert Zimmer (Institute of Virology and Immunology, Mittelhäusern, Switzerland) for the replication-competent pseudotyped virus with a vesicular stomatitis virus backbone coated with SARS-CoV-2 spike protein. We also thank the volunteers who agreed to participate, donated blood, and replied to the telephone questionnaires. YL is supported by the Nehemia Rubin Excellence in Biomedical Research: the TELEM Program of Chaim Sheba Medical Center.
Contributors
GR-Y, YL, MG, SA-P, and YK contributed to study conception and design. GJ, IG, VI, OH, SK-L, TL, YK, KA, SA, RK, MM, NHT, LM, RD, and EM contributed to data collection. GR-Y, YL, GJ, MG, NB, CR, and AZ contributed to data analysis and interpretation of results. GR-Y, YL, GJ, and NB contributed to preparation of the draft manuscript. GJ, CR, NT, MG, and GR-Y accessed and verified the data used in this study. All authors had permission to access the raw data. All authors reviewed the results and approved the final manuscript. All authors accept responsibility to submit the manuscript for publication.
Supplementary Material
References
- 1.Wu F, Zhao S, Yu B, et al. A new coronavirus associated with human respiratory disease in China. Nature. 2020;579:265–269. doi: 10.1038/s41586-020-2008-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Zieneldien T, Kim J, Cao J, Cao C. COVID-19 vaccines: current conditions and future prospects. Biology (Basel) 2021;10:960. doi: 10.3390/biology10100960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Amit S, Regev-Yochay G, Afek A, Kreiss Y, Leshem E. Early rate reductions of SARS-CoV-2 infection and COVID-19 in BNT162b2 vaccine recipients. Lancet. 2021;397:875–877. doi: 10.1016/S0140-6736(21)00448-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Jackson LA, Anderson EJ, Rouphael NG, et al. An mRNA vaccine against SARS-CoV-2—preliminary report. N Engl J Med. 2020;383:1920–1931. doi: 10.1056/NEJMoa2022483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Haas EJ, Angulo FJ, McLaughlin JM, et al. Impact and effectiveness of mRNA BNT162b2 vaccine against SARS-CoV-2 infections and COVID-19 cases, hospitalisations, and deaths following a nationwide vaccination campaign in Israel: an observational study using national surveillance data. Lancet. 2021;397:1819–1829. doi: 10.1016/S0140-6736(21)00947-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Levin EG, Lustig Y, Cohen C, et al. Waning immune humoral response to BNT162b2 COVID-19 vaccine over 6 months. N Engl J Med. 2021;385:e84. doi: 10.1056/NEJMoa2114583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Goldberg Y, Mandel M, Bar-On YM, et al. Waning immunity after the BNT162b2 vaccine in Israel. N Engl J Med. 2021;385:e85. doi: 10.1056/NEJMoa2114228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Khoury DS, Cromer D, Reynaldi A, et al. Neutralizing antibody levels are highly predictive of immune protection from symptomatic SARS-CoV-2 infection. Nat Med. 2021;27:1205–1211. doi: 10.1038/s41591-021-01377-8. [DOI] [PubMed] [Google Scholar]
- 9.Bergwerk M, Gonen T, Lustig Y, et al. COVID-19 breakthrough infections in vaccinated health care workers. N Engl J Med. 2021;385:1474–1484. doi: 10.1056/NEJMoa2109072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Asamoah-Boaheng M, Goldfarb DM, Karim ME, et al. The relationship between anti-spike SARS-CoV-2 antibody levels and risk of breakthrough COVID-19 among fully vaccinated adults. J Infect Dis. 2022 doi: 10.1093/infdis/jiac403. published online Oct 5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.WHO Coronavirus disease (COVID-19) pandemic. https://www.who.int/emergencies/diseases/novel-coronavirus-2019?adgroupsurvey=adgroupsurvey&gclid=CjwKCAiAxvGfBhB-EiwAMPakqncEmQtLjg-KjLsjXm3JCP_fW-vMmtdapGpw3BDO0KO4T-8RFUcu5hoCL6IQAvD_BwE
- 12.Taddeo A, Veiga IB, Devisme C, et al. Optimized intramuscular immunization with VSV-vectored spike protein triggers a superior immune response to SARS-CoV-2. NPJ Vaccines. 2022;7:82. doi: 10.1038/s41541-022-00508-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Marshall A, Altman DG, Holder RL, Royston P. Combining estimates of interest in prognostic modelling studies after multiple imputation: current practice and guidelines. BMC Med Res Methodol. 2009;9:57. doi: 10.1186/1471-2288-9-57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Vivaldi G, Jolliffe DA, Faustini S, et al. Correlation between post-vaccination anti-Spike antibody titres and protection against breakthrough SARS-CoV-2 infection: a population-based longitudinal study. J Infect Dis. 2022 doi: 10.1093/infdis/jiac321. published online July 30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Maier HE, Balmaseda A, Saborio S, et al. Protection associated with previous SARS-CoV-2 infection in Nicaragua. N Engl J Med. 2022;387:568–570. doi: 10.1056/NEJMc2203985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lustig Y, Gonen T, Meltzer L, et al. Superior immunogenicity and effectiveness of the third compared to the second BNT162b2 vaccine dose. Nat Immunol. 2022;23:940–946. doi: 10.1038/s41590-022-01212-3. [DOI] [PubMed] [Google Scholar]
- 17.Lai A, Caimi B, Franzetti M, et al. Durability of humoral responses after the second dose of mRNA BNT162b2 vaccine in residents of a long term care facility. Vaccines (Basel) 2022;10:446. doi: 10.3390/vaccines10030446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Gilboa M, Mandelboim M, IndenBAU/mLm V, et al. Early immunogenicity and safety of the third dose of BNT162b2 messenger RNA coronavirus disease 2019 vaccine among adults older than 60 years: real-world experience. J Infect Dis. 2022;225:785–792. doi: 10.1093/infdis/jiab584. [DOI] [PubMed] [Google Scholar]
- 19.Bar-On YM, Goldberg Y, Mandel M, et al. Protection of BNT162b2 vaccine booster against COVID-19 in Israel. N Engl J Med. 2021;385:1393–1400. doi: 10.1056/NEJMoa2114255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Talic S, Shah S, Wild H, et al. Effectiveness of public health measures in reducing the incidence of covid-19, SARS-CoV-2 transmission, and covid-19 mortality: systematic review and meta-analysis. BMJ. 2021;375 doi: 10.1136/bmj-2021-068302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lin J, Law R, Korosec CS, et al. Longitudinal assessment of SARS-CoV-2-specific T cell cytokine-producing responses for 1 year reveals persistence of multicytokine proliferative responses, with greater immunity associated with disease severity. J Virol. 2022;96 doi: 10.1128/jvi.00509-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.McMahan K, Yu J, Mercado NB, et al. Correlates of protection against SARS-CoV-2 in rhesus macaques. Nature. 2021;590:630–634. doi: 10.1038/s41586-020-03041-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Young A. T cells in SARS-CoV-2 infection and vaccination. Ther Adv Vaccines Immunother. 2022;10 doi: 10.1177/25151355221115011. 25151355221115011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Moga E, Lynton-Pons E, Domingo P. The robustness of cellular immunity determines the fate of SARS-CoV-2 infection. Front Immunol. 2022;13 doi: 10.3389/fimmu.2022.904686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Nemet I, Kliker L, Lustig Y, et al. Third BNT162b2 vaccination neutralization of SARS-CoV-2 omicron infection. N Engl J Med. 2022;386:492–494. doi: 10.1056/NEJMc2119358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Collie S, Champion J, Moultrie H, Bekker L-G, Gray G. Effectiveness of BNT162b2 vaccine against omicron variant in South Africa. N Engl J Med. 2022;386:494–496. doi: 10.1056/NEJMc2119270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Regev-Yochay G, Gonen T, Gilboa M, et al. Efficacy of a fourth dose of COVID-19 mRNA vaccine against omicron. N Engl J Med. 2022;386:1377–1380. doi: 10.1056/NEJMc2202542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Goldblatt D, Fiore-Gartland A, Johnson M, et al. Towards a population-based threshold of protection for COVID-19 vaccines. Vaccine. 2022;40:306–315. doi: 10.1016/j.vaccine.2021.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zhu K-L, Gao H-X, Yao L, et al. Delta infection following vaccination elicits potent neutralizing immunity against the SARS-CoV-2 omicron. J Infect Dis. 2022 doi: 10.1093/infdis/jiac149. published online April 16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lustig Y, Zuckerman N, Nemet I, et al. Neutralising capacity against delta (B.1.617.2) and other variants of concern following Comirnaty (BNT162b2, BioNTech/Pfizer) vaccination in health care workers, Israel. Euro Surveill. 2021;26 doi: 10.2807/1560-7917.ES.2021.26.26.2100557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lustig Y, Sapir E, Regev-Yochay G, et al. BNT162b2 COVID-19 vaccine and correlates of humoral immune responses and dynamics: a prospective, single-centre, longitudinal cohort study in health-care workers. Lancet Respir Med. 2021;9:999–1009. doi: 10.1016/S2213-2600(21)00220-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Altarawneh HN, Chemaitelly H, Ayoub HH, et al. Effects of previous infection and vaccination on symptomatic omicron infections. N Engl J Med. 2022;387:21–34. doi: 10.1056/NEJMoa2203965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kliker L, Zuckerman N, Atari N, et al. COVID-19 vaccination and BA.1 breakthrough infection induce neutralising antibodies which are less efficient against BA.4 and BA.5 omicron variants, Israel, March to June 2022. Euro Surveill. 2022;27 doi: 10.2807/1560-7917.ES.2022.27.30.2200559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Muecksch F, Wang Z, Cho A, et al. Increased memory B cell potency and breadth after a SARS-CoV-2 mRNA boost. Nature. 2022;607:128–134. doi: 10.1038/s41586-022-04778-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data and study protocol will be shared after publication, upon request. Only deidentified, relevant, individual participant data will be available. Data will be available by email from the corresponding author.