Abstract
The field of neuroscience has largely overlooked the impact of motherhood on brain function outside the context of responses to infant stimuli. Here, we apply spectral dynamic causal modelling (spDCM) to resting-state fMRI data to investigate differences in brain function between a group of 40 first-time mothers at 1-year postpartum and 39 age- and education-matched women who have never been pregnant. Using spDCM, we investigate the directionality (top–down vs. bottom–up) and valence (inhibition vs excitation) of functional connections between six key left hemisphere brain regions implicated in motherhood: the dorsomedial prefrontal cortex, ventromedial prefrontal cortex, posterior cingulate cortex, parahippocampal gyrus, amygdala, and nucleus accumbens. We show a selective modulation of inhibitory pathways related to differences between (1) mothers and non-mothers, (2) the interactions between group and cognitive performance and (3) group and social cognition, and (4) differences related to maternal caregiving behaviour. Across analyses, we show consistent disinhibition between cognitive and affective regions suggesting more efficient, flexible, and responsive behaviour, subserving cognitive performance, social cognition, and maternal caregiving. Together our results support the interpretation of these key regions as constituting a parental caregiving network. The nucleus accumbens and the parahippocampal gyrus emerging as ‘hub’ regions of this network, highlighting the global importance of the affective limbic network for maternal caregiving, social cognition, and cognitive performance in the postpartum period.
Subject terms: Cognitive neuroscience, Psychology
The transition to motherhood is associated with both structural and functional neural plasticity across pregnancy and the postpartum period1–7, with brain changes that are long-lasting8,9 and may persist across the lifespan10–14 (reviewed in15). Studies of brain function in motherhood have largely focused on maternal neural responses to infant stimuli16–18, with meta-analyses finding several brain regions showing consistent activation in response to infant cues19–21. As these regions have been commonly found in functional neuroimaging studies of motherhood, they have been proposed to form a putative ‘parental caregiving brain network’18. Regional activation of the parental caregiving network in response to infant stimuli is associated with better maternal attachment and positive caregiving behaviours18,22,23, whereas aberrant activation of these regions is associated with postpartum depression24–27, substance use28,29, and trauma30,31. This suggests that activation of the parental caregiving network may support the initiation and maintenance of complex and sensitive maternal behaviours18. While each region of the putative parental caregiving network is consistently implicated in maternal behaviour, the network itself has not been theoretically formalised, and it is unknown how regions within the network influence each other to facilitate maternal caregiving.
The field of parental neuroscience has largely overlooked the impact of motherhood on brain function outside the context of responses to infant stimuli (e.g., during a resting-state). Resting-state connectivity is particularly interesting, as it is considered an index of ‘intrinsic’ brain function, and provides the neural framework for the individual’s full range of extrinsic responses32,33. Changes in resting-state brain function associated with motherhood may uncover global, generalised changes in brain function, with broader impacts for maternal behaviour15, beyond responses to infant stimuli.
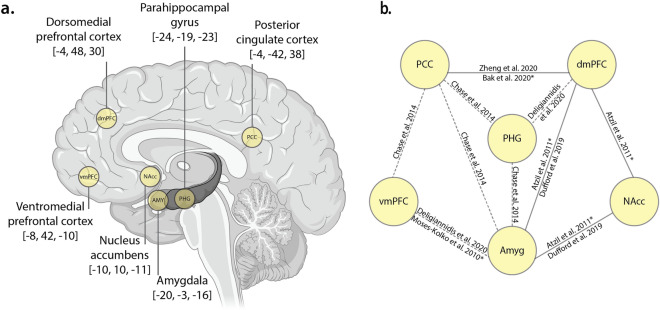
A small number of studies have measured functional connectivity in relation to motherhood, and these studies have shown connectivity differences between six key regions of the putative parental caregiving network: the dorsomedial prefrontal cortex, ventromedial prefrontal cortex, parahippocampal gyrus, posterior cingulate cortex, amygdala, and nucleus accumbens3,24,27,30,34–36 (Fig. 1a).
Figure 1.
(a.) Regions of interest. Six left-hemisphere Regions of Interest (ROIs), with MNI coordinates (x,y,z). Brain image created with BioRender. (b.) Previously reported differences in functional connectivity. Schematic diagram showing the extant functional connectivity literature in motherhood for selected regions of the parental caregiving brain network. Dashed lines represent connections which differ between mothers with and without postpartum depression, studies using task-based fMRI scans to derive connectivity are denoted with an asterisk. These known connections were used to formulate the study hypotheses. Abbreviations: PCC, posterior cingulate cortex; vmPFC, ventromedial prefrontal cortex; dmPFC, dorsomedial prefrontal cortex; PHG, parahippocampal gyrus; Amyg, amygdala; NAcc, nucleus accumbens.
Functional connectivity between the amygdala, nucleus accumbens, and dorsomedial prefrontal cortex are related to positive maternal care3,30. For example, increased functional connectivity between the amygdala, nucleus accumbens and dorsomedial prefrontal cortex at rest is associated with improved ability to scaffold appropriate mother–child interactions3. While watching a mother–child interaction, mothers with higher behavioural mother–child synchrony (the coordination of gaze, touch and vocalisations between mother and child) showed higher connectivity between the nucleus accumbens and medial prefrontal cortex, compared to mothers with lower maternal-child synchrony30. Additionally, in non-depressed mothers, there is a positive relationship between cognitive performance and functional connectivity between two higher-order association areas, the dorsomedial prefrontal cortex and the posterior cingulate cortex, both during task35 and at rest34, as illustrated in Fig. 1b.
Beyond these studies of functional connectivity in healthy mothers, much of what is known about maternal brain connectivity is from studies comparing mothers with and without postpartum depression. Mothers with postpartum depression show aberrant connectivity between the posterior cingulate cortex, parahippocampal gyrus, amygdala, ventromedial prefrontal cortex, and dorsomedial prefrontal cortex24,36,37, suggesting these regions are important for maternal caregiving, and are sensitive to, or involved in, postpartum mood disorders. Very little is known about the differences in resting state brain function between non-mothers and mothers without mood disorders.
The investigation of maternal brain function outside of the context of postpartum depression, and reactivity to infant stimuli, is important in order to characterise the ‘normative’ functional neural adaptations to motherhood. Importantly, while the parental caregiving network has been proposed to account for commonly activated regions found in neuroimaging studies of motherhood, no study has explicitly tested the network outside of infant-centric stimuli. Furthermore, an important step towards understanding the functional adaptations to motherhood is to examine the direction and valence of information flow throughout the parental caregiving brain network. Functional connectivity analyses indicate coherence of timeseries between spatially distinct regions, but do not provide information on the directionality of the connections. By contrast, effective connectivity analyses indicate the direction of connections (e.g., whether they represent top-down, lateral, or bottom-up information flow, as understood within the predictive coding framework38) as well as the valence of the connections (whether they are excitatory or inhibitory). Understanding the dynamics of information flow within the parental caregiving brain network is an important step to understanding the functional adaptations in early motherhood.
We investigated differences between mothers and non-mothers in effective connectivity between regions of the putative parental caregiving network, using spectral dynamic causal modelling (spDCM)39. SpDCM can map specific circuits that influence functional brain differences between mothers and non-mothers and enables the investigation of effective brain connectivity in maternal caregiving behaviour, cognitive performance, and social cognition.
Six regions of the parental caregiving network18 consistently emerge across functional connectivity studies in motherhood, showing relationships with maternal caregiving, cognition, and postpartum depression. Other regions are also implicated, but less consistently3,24,27,30,34–36. These regions are also more broadly implicated in social cognition outside of the context of motherhood40,41, including theory of mind, empathy, and mentalising abilities. Informed by this understanding, we tested four hypotheses. First, to map the connectivity of the parental caregiving network, we hypothesised that mothers and non-mothers would show differences in effective connectivity between the dorsal and ventral medial prefrontal cortices, posterior cingulate cortex, parahippocampal gyrus, amygdala, and nucleus accumbens. Second and third, we hypothesised that an increased influence of higher-order regions, including dorsomedial prefrontal cortex, ventromedial prefrontal cortex, posterior cingulate cortex, and parahippocampal gyrus42–44, would be related to both cognitive performance (verbal memory, working memory, and processing speed); and social cognition (empathy and theory of mind). Fourth, we hypothesise that maternal caregiving (attachment and self-efficacy) would be related to an increased influence of the amygdala and nucleus accumbens, consistent with functional connectivity studies of maternal caregiving behaviour3,30.
Results
Mothers and non-mothers did not differ in age or education45. Compared to non-mothers, mothers had poorer sleep and higher depression (Table 1). Mothers also scored higher than non-mothers on the wellbeing component, which combines depression, anxiety, and sleep, indicating poorer wellbeing in mothers. Mothers and non-mothers did not differ on the cognitive performance component. Mothers scored higher than non-mothers on the social cognition component, indicating better social cognition. The means and standard deviations for the cognition and social cognition variables within the principal components, and significance tests, are reported in Supplementary Information 3. Time since postpartum (age of infant) also did not correlate with any cognitive, social cognitive, caregiving or wellbeing measure (Supplementary Information 4).
Table 1.
Demographic information for Mothers and Non-Mothers, including independent samples t-test comparisons between groups for age, education, sleep (Pittsburgh Sleep Quality Index), depression (Beck Depression Inventory), and anxiety (Beck Anxiety Index).
| Mothers (N = 40) | Non-Mothers (N = 39) | t-value | Cohen’s d | p-value | |||
|---|---|---|---|---|---|---|---|
| Mean (SD) | Range | Mean (SD) | Range | ||||
| Age (years) | 32.5 (3.3) | 25–40 | 31.3 (4.0) | 20–39 | 1.36 | 0.31 | .18 |
| Education (years) | 18.2 (2.5) | 13–24.5 | 17.6 (2.7) | 11–24 | 1.03 | 0.23 | .30 |
| Sleep (PSQI) | 7.3 (3.1) | 2–17 | 4.7 (2.6) | 0–10 | 4.17 | 0.94 | < .001 |
| Depression (BDI) | 8.6 (5.6) | 0–24 | 5.0 (4.3) | 0–17 | 3.20 | 0.72 | .002 |
| Anxiety (BAI) | 10.7 (8.6) | 0–32 | 8.6 (6.63) | 0–29 | 1.30 | 0.29 | .20 |
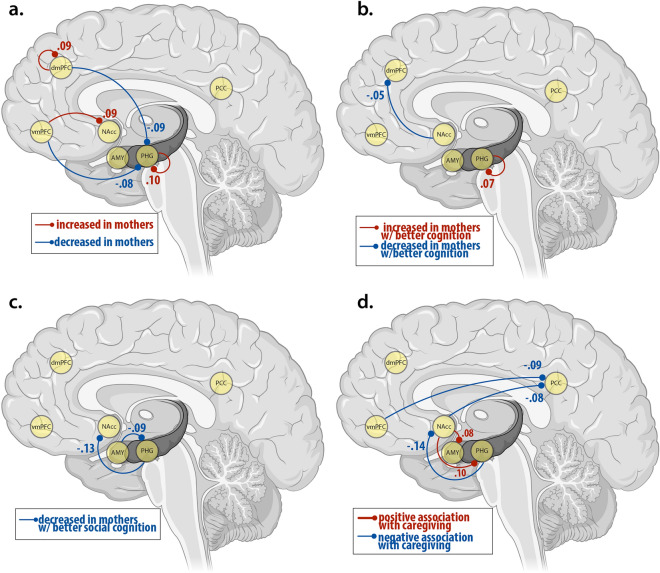
At the subject-level, diagnostic statistics ensured first-level model inversion had converged. The average variance explained across subject-level DCM inversion was very high (> 85, mothers: M = 87.6, SD = 1.6, range = 82.4–91.4; non-mothers: M = 87.9, SD = 2.4, range = 79.9–92.3). The results of each of the four models are shown in Fig. 2. All significant connections in all four models were inhibitory connections (Tables 2, 3, 4, 5).
Figure 2.
Schematic diagram showing spDCM results for (a.) the main effect of group (mothers vs. non-mothers), (b.) the interaction effect of group-by-cognition, (c.) the interaction effect of group-by-social cognition, and (d.) the main effect of maternal caregiving in mothers only. Effect sizes, directionality and valence of all connections are reported in Tables 2, 3, 4, 5. All connections are inhibitory. Values represent effect sizes (see Tables 2, 3, 4, 5) in Hz. Blue lines depict decreased inhibition and red lines depict increased inhibition. Dashed lines represent connections that are attenuated when the model adjusts for wellbeing, and solid lines are connections which are present whether or not wellbeing is adjusted for. Abbreviations: PCC, posterior cingulate cortex; vmPFC, ventromedial prefrontal cortex; dmPFC, dorsomedial prefrontal cortex; PHG, parahippocampal gyrus; Amyg, amygdala; NAcc, nucleus accumbens.
Table 2.
Effect size, valence and posterior probabilities for each connection showing a main effect of group (mothers > non-mothers).
| Connection | Valence | Mothers versus non-mothers | |
|---|---|---|---|
| Effect size in Hz [95% CI] | Posterior probability | ||
| dmPFC→ dmPFC | Inhibition | 0.09 [0,0.16] | 0.97 |
| dmPFC → PHG | Inhibition | − 0.09 [− 0.19,0] | 0.97 |
| vmPFC → PHG | Inhibition | − 0.08 [− 0.16,0] | 096 |
| vmPFC → NAcc | Inhibition | 0.09 [0.01,0.17 | 0.96 |
| PHG → PHG | Inhibition | 0.10 [0.18,0.01] | 0.98 |
Hz Hertz, CI Confidence Interval, dmPFC dorsomedial prefrontal cortex, vmPFC ventromedial prefrontal cortex, PHG parahippocampal gyrus, Amyg amygdala, NAcc nucleus accumbens.
Table 3.
Effect size, valence and posterior probabilities for each connection showing an interaction effect of group-by-cognition.
| Connection | Valence | Group-by-cognition | |
|---|---|---|---|
| Effect size in Hz [95% CI] | Posterior probability | ||
| PHG → PHG | Inhibition | 0.07 [0.01, 0.12] | 0.98 |
| NAcc → dmPFC | Inhibition | − 0.05 [− 0.11, − 0.00] | 0.95 |
Hz Hertz, CI Confidence Interval, dmPFC dorsomedial prefrontal cortex, PHG parahippocampal gyrus, NAcc nucleus accumbens.
Table 4.
Effect size, valence and posterior probabilities for each connection showing an interaction effect of group-by-social cognition, with and without controlling for wellbeing.
| Connection | Valence | Group-by-social cognition | |
|---|---|---|---|
| Effect size in Hz [95% CI] | Posterior probability | ||
| PHG → NAcc | Inhibition | − 0.13 [− 0.21, − 0.05] | 1.00 |
| Amyg → PHG | Inhibition | − 0.09 [− 0.18, − 0.01] | 0.97 |
Hz Hertz, CI Confidence Interval, dmPFC dorsomedial prefrontal cortex, vmPFC ventromedial prefrontal cortex, PHG parahippocampal gyrus, Amyg amygdala, NAcc nucleus accumbens.
Table 5.
Main effect of maternal caregiving, with and without controlling for wellbeing.
| Connection | Valence | Maternal caregiving | |
|---|---|---|---|
| Effect size in Hz [95% CI] | Posterior probability | ||
| vmPFC → PCC | Inhibition | − 0.09 [− 0.17, − 0.01] | 0.97 |
| PHG → NAcc | Inhibition | − 0.14 [− 0.22, − 0.06] | 1.00 |
| Amyg → NAcc | Inhibition | − 0.13 [− 0.20, − 0.05] | 1.00 |
| NAcc → PCC | Inhibition | − 0.08 [− 0.16, − 0.01] | 0.97 |
| NAcc → PHG | Inhibition | 0.1 [0.02, 0.18] | 0.98 |
Hz Hertz, CI Confidence Interval, dmPFC dorsomedial prefrontal cortex, vmPFC ventromedial prefrontal cortex, PHG parahippocampal gyrus, PCC posterior cingulate cortex, Amyg amygdala, NAcc nucleus accumbens.
Compared to non-mothers, mothers showed decreased top-down inhibition of the parahippocampal gyrus from the dorsomedial prefrontal cortex, and the ventromedial prefrontal cortex, and increased top-down inhibition of the nucleus accumbens from the ventral medial prefrontal cortex (Table 2, Fig. 2a). The dorsomedial prefrontal cortex and parahippocampal gyrus showed increased self-inhibition in mothers compared to non-mothers. Mothers with better cognitive performance showed decreased bottom-up inhibition from the nucleus accumbens to the dorsomedial prefrontal cortex, and increased self-inhibition of the parahippocampal gyrus (Table 3, Fig. 2b). Mothers with better social cognition showed decreased top-down inhibition of the nucleus accumbens from the parahippocampal gyrus, and increased bottom-up inhibition from the amygdala to the parahippocampal gyrus (Table 4, Fig. 2c). Within the mother group, higher maternal caregiving was associated with increased bottom-up inhibition from the nucleus accumbens to the parahippocampal gyrus, decreased inhibition from the nucleus accumbens and the ventromedial prefrontal cortex to the posterior cingulate cortex, and decreased inhibition to the nucleus accumbens from the amygdala and parahippocampal gyrus (Table 5, Fig. 2d).
The nucleus accumbens and the parahippocampal gyrus emerged as ‘hub’ regions of the maternal network. Both the nucleus accumbens and the parahippocampal gyrus show significant connections in every model tested (main effect of group, main effect of maternal caregiving, interaction effect of group-by-cognition, and interaction effect of group-by-social cognition). When interpreting the results from our four models together, these two key regions show the highest nodal degree (number of connections), with 29% of all connections to or from the nucleus accumbens (9/31) and 26% of all connections to or from the parahippocampal gyrus (8/31). There were only three connections that did not include either the parahippocampal gyrus or the nucleus accumbens. For mothers versus non-mothers this included self inhibition of the dorsomedial prefrontal cortex; and in mothers with higher maternal caregiving, reduced inhibition of the posterior cingulate cortex from the ventromedial prefrontal cortex, and from the dorsomedial prefrontal cortex to the amygdala was observed. The strongest connections (effect size in Hz > 0.1) between these hub regions were from the parahippocampal gyrus to the nucleus accumbens (− 0.15 Hz), self-connections from the parahippocampal gyrus to itself (0.11 Hz), and from the nucleus accumbens to the parahippocampal gyrus (0.10 Hz). Other connections stronger than 0.10 Hz were from the amygdala to the nucleus accumbens (− 0.13 Hz; maternal caregiving) and from the dorsomedial prefrontal cortex to the parahippocampal gyrus (− 0.10 Hz; mothers vs non-mothers).
Discussion
We investigated the causal interactions between six key left-hemisphere regions of the parental caregiving network (dorsomedial prefrontal cortex, ventromedial prefrontal cortex, posterior cingulate cortex, parahippocampal gyrus, amygdala, and nucleus accumbens), using spectral dynamic causal modelling (spDCM). Our results are the first experimental evidence to confirm that this network, previously referred to as the ‘parental caregiving network’18 is highly implicated in maternal caregiving. We showed that the dynamics of this network differ between non-mothers and mothers at 1-year postpartum. Specifically, we show consistent patterns of disinhibition between cognitive and affective regions across all analyses, suggesting (1) a more responsive, flexible, and efficient emotion-regulation system in mothers compared to non-mothers, (2) increased selectivity for goal-directed information in mothers who perform better on cognitive tasks, (3) disinhibition between cognitive and affective mentalising networks related to both cognition and social cognition, and (4) a more selective affective limbic network in mothers with higher maternal caregiving. The results indicated that the NAcc and PHG are ‘hub’ regions of the maternal caregiving brain network, (discussed further in Supplementary Information 5).
Effective connectivity of the parental caregiving network in mothers versus non-mothers
Relative to non-mothers, mothers showed increased self-inhibition of the dmPFC and the PHG. Self-connections regulate the excitatory and inhibitory balance of the network; controlling the gain (or excitatory/inhibitory balance) of the region to extrinsic connections46. In predictive coding theory, which is core to understanding the results of DCM analysis, the brain infers the cause of its sensory inputs by minimising prediction errors throughout the hierarchical cortical network47,48. Importantly, as self-connections regulate the gain or excitability of superficial pyramidal cells within a region, they also regulate the precision of the prediction error. Thus, the finding that mothers show increased self-inhibition of the dmPFC and PHG is consistent with the interpretation that motherhood is associated with increased network efficiency (e.g., ref 49) and more precise prediction error48,50 of the parental caregiving network relative to non-mothers. This improved precision for motherhood contrasts with pathologies like schizophrenia, which is associated with reduced self-connections and aberrant prediction error throughout the cortical hierarchy48,50.
Mothers also showed decreased top-down inhibition from the dmPFC and vmPFC to the PHG; and from the vmPFC to NAcc, compared to non-mothers. Together, the dmPFC, vmPFC, NAcc and PHG form key regions of the ventromedial emotion regulatory system, which is implicated in early processing and encoding of the emotional significance of a stimulus51. This system is involved in early appraisal, encoding, and automatic regulation of emotion. The connection between dmPFC to PHG, and vmPFC to PHG was reduced in mothers relative to non-mothers, indicating a less inhibited top-down circuit for mothers relative to non-mothers. A less inhibited network for mothers implies the network is more easily excitable compared to non-mothers; in other words, less input is required to reach the ‘threshold’ for activation. Together with the increased self-connection of the dmPFC and PHG, this pattern of results indicates a more responsive, flexible and efficient emotion regulation system in mothers than non-mothers51.
Differences in effective connectivity between mothers and non-mothers related to cognition
Behaviourally, mothers and non-mothers showed no significant differences in cognitive performance (verbal memory, working memory, and processing speed)45. The absence of group differences in cognitive performance may partly explain the low dimensionality of the effective connectivity network found in the group-by-cognition analysis.
Our spDCM results show that mothers with better cognitive performance showed decreased bottom-up inhibition from the NAcc to the dmPFC, and increased self-inhibition of the PHG. As noted above, self-inhibition within DCM reflects the excitation/inhibition balance of superficial pyramidal cells within a cortical region46,48. The PHG is involved in memory consolidation52 and mediates communication between the cortex and the hippocampus53. The PHG may be considered a filter, or a gate, which selectively controls information transfer between the neocortex and the hippocampus through intrinsic inhibitory mechanisms42,54. Increased self-inhibition of the PHG in mothers with better cognitive performance is consistent with memory consolidation and retrieval functions which require selective and precise integration of information to and from the hippocampus42. These findings suggest that mothers in the peripartum period may experience increased information selectivity by the PHG, which may be related to cognitive performance in the peripartum period.
In addition, mothers with better cognitive performance showed decreased bottom-up inhibition of the dmPFC from the NAcc. This connection was no longer significant once accounting for differences in wellbeing (Supplementary Information 2), indicating that this connection is affected by the individual’s level of depression, anxiety, and sleep quality. The dmPFC and NAcc form core regions of the cortico-basal ganglia-thalamic network that underlies goal-directed motor behaviour to motivationally salient events44,55,56. The dmPFC in particular is implicated in the control of actions within this network, and the input from the NAcc to the dmPFC provides the emotional and motivational context to drive the appropriate motor response44. Importantly, the connection between the NAcc and dmPFC is known to be mediated by sleep44. A key part of memory consolidation, ‘replay’, occurs during sleep and neurons within these regions show spike patterns consistent with replay57,58. Taken together, our results indicate that the bottom-up inhibitory connection from the NAcc to the dmPFC may underlie wellbeing-mediated differences in cognition between mothers and non-mothers45.
Differences in effective connectivity between mothers and non-mothers related to social cognition
Compared to non-mothers, mothers had significantly higher scores on measures of social cognition (theory of mind, and empathy; Supplementary Information 3). Mothers with higher social cognition scores had decreased inhibition from the amygdala to the parahippocampal gyrus, and from the PHG to the NAcc, relative to non-mothers. The NAcc-Amyg-PHG network forms the affective limbic network that is implicated in social connectedness, and its converse, loneliness59. This network overlaps with the subcortical components of the ventromedial emotion regulatory system, which is implicated in early appraisal, encoding, and automatic regulation of emotion51. The limbic network is particularly involved in affective aspects of theory of mind, pertaining to making inferences about emotions43, as well as assigning motivational value to goals on the basis of internal states and environmental stimuli60. That mothers with better social cognition showed decreased inhibition within this network indicates that the network is relatively disinhibited relative to non-mothers. In other words, the network can be more responsive, i.e., requires lower levels of input to activate, in mothers with improved social cognition. Consistent with our other analyses, mothers show a more responsive limbic network which supports their responsiveness to affective stimuli that underlie self-referential and other-referential action43,59,60.
Mothers with higher social cognition also showed reduced inhibition from the dmPFC to the NAcc. However, this connection was only significant once accounting for wellbeing. Importantly, while the NAcc-Amyg-PHG limbic network subserves affective theory of mind and empathy, the dmPFC is involved in a separate but parallel cognitive theory of mind/empathy network43. This indicates that improved social cognition in motherhood is associated with reduced top-down inhibition of the affective theory of mind network from the cognitive theory of mind network. Interestingly, the link between the dmPFC and NAcc in the group-by-cognition and group-by-social cognition models were reciprocal. In the cognition model, the information flow was top-down (dmPFC to NAcc) and in the social cognition model, the information flow was bottom-up (NAcc to dmPFC). Both analyses indicate that improved cognition is associated with disinhibition of the links between more cognitively-oriented regions (dmPFC) and the reward-based and affective limbic system (NAcc-Amyg-PHG). The dmPFC-NAcc connection in both models was mediated by wellbeing (depression, anxiety and sleep), indicating that disinhibition or increased responsiveness is affected by an individual’s daily functioning. The dependence of the dmPFC-NAcc connection on wellbeing in these models (Supplementary Information 2) is compatible with the known physiology of this connection in sleep, memory, cognition57,58,61.
Influence of maternal caregiving on effective connectivity within the parental caregiving network
Strikingly, every node within the studied network was influenced by maternal caregiving. These regions, previously hypothesised to form a parental caregiving network18, do indeed appear to function together as a network related to maternal caregiving in mothers (dmPFC was also influenced by caregiving, but only when controlling for wellbeing; see Supplementary Information 2). Specifically, the affective limbic network was particularly implicated in maternal caregiving, with reciprocal increases and decreases in inhibition with positive maternal care. Interestingly, information transfer within the affective limbic network occurred via the NAcc. Models of the affective limbic system posit that the NAcc is the key ‘nexus’ or hub region within this network, and plays a major role in action selection in goal-based behaviour60. This system facilitates action selection so that the individual can rapidly and flexibly switch between goals, with the NAcc integrating multiple sources of information from the PHG and amygdala to assign motivational value to those goals. This result suggests that positive maternal caregiving behaviour is highly dependent on information transfer within the affective limbic network, with the NAcc serving as the hub. The motivational salience of caregiving behaviour is highlighted by the fact that the amygdala and NAcc are consistently activated in response to infant cues in task-based fMRI studies18,30,62–66. Experiencing increased pleasure and reward in response to an infant increases the salience of infant cues, and promotes increased attention and attachment, which serves as a positive feedback loop to maintain sensitive caregiving behaviours67,68.
Inhibitory connection from the dmPFC to the affective limbic network (this time to the amygdala) was implicated in maternal caregiving, but only when controlling for wellbeing (Supplementary Information 2). The vmPFC, which is more directly implicated in affective mentalising and theory of mind43 was not implicated. Intriguingly, interaction of the cognitive and affective mentalising networks may be implicated in maternal caregiving, rather than specifically affective or cognitive aspects of the mentalising network. Top-down connection from the dmPFC to the limbic network was more disinhibited (i.e., more excitable) with increased positive maternal caregiving, and modulated by wellbeing, suggesting that the interaction between cognitive and affective networks is most affected by wellbeing, and associated with mood disorders in the postpartum period.
While a more disinhibited network may subserve more flexible and responsive behaviour, in the extreme it may lead to hyperexcitability, and predispose the individual to pathologies like mood and anxiety disorders69,70. Inhibitory connections in particular are important in the pathogenesis of anxiety and fear disorders69,71. Activation of the amygdala and increased salience of infant cues is part of a healthy adaptation to motherhood, giving rise to increased vigilance, protectiveness, threat detection, and preoccupation with the infant72,73. However, the brain circuits important for adaptive ‘checking and worrying’ behaviours, overlap with those that are hyperactive in anxiety disorders67. Whilst plasticity in salience networks is a healthy part of threat detection and harm avoidance74, aberrant functional adaptations – abnormally decreased or excessive worry – may be related to postpartum psychopathology67. Importantly, as the participants in this study were healthy and did not meet the criteria for depression or anxiety (see Results) the results represent variability within the healthy postpartum experience.
Mothers with higher maternal caregiving also showed decreased inhibition of the PCC from the vmPFC, and NAcc, indicating an important role of the PCC in positive maternal care34. Within the ‘dynamic systems’ model of PCC function, increased activity of the dorsal posterior cingulate allows increased whole-brain meta-stability, allowing efficient and rapid transitions between neural states, and more efficient cognition. We found that increased caregiving is associated with directional reductions of dorsal PCC inhibition from the vmPFC and NAcc, consistent with the interpretation that those with higher caregiving scores can recruit more efficient and flexible cognitive system in response to salient, behaviourally-rewarding stimuli (i.e., the infant). This result complements earlier findings in mothers with postpartum depression, whereby the PCC showed reduced resting state functional connectivity with the PHG, vmPFC, and amygdala24. Postpartum depression affects the quality of maternal caregiving75, further underlying the importance of vmPFC/limbic inhibitory connections with the posterior cingulate in positive maternal caregiving.
Study context & future directions
The study findings should be interpreted within the context of the design and limitations. We specifically examined mothers around 1 year postpartum (10–14 months, mean 12 months). Given that the postpartum period is associated with highly plastic changes in the brain, body, and environment more generally15, effective connectivity in mothers may differ at other points in the postpartum period (see also Supplementary Information 4). The cross-sectional study design limits the results to differences between groups, and not changes across the peripartum period76,77. Furthermore, this study design does not rule out the possibility that group differences may exist between mothers and non-mothers in this sample based on biological and/or psychosocial differences contributing to self-selection into motherhood, potentially preceding conception. Future studies could use longitudinal samples to examine how these networks develop across the transition to motherhood. The study included a modest sample size of 40 mothers and 39 non-mothers. Whilst this is considered a large sample size for conducting spDCM analyses78, and in studies of parental neuroscience2,4,6,8, larger samples may be able to detect more subtle effects. Our results indicate a specific influence of motherhood on inhibitory effective connectivity, which may indicate that the primary inhibitory neurotransmitter, GABA, is influenced by motherhood (Supplementary Information 7). It is not possible to explore this hypothesis based on the current data; future studies should examine this possibility using appropriate methods (e.g., magnetic resonance spectroscopy, hormone testing). The dimensionality of the spDCM model, while large in comparison to previous spDCM models (e.g.,79), is probably low with respect to the known widespread effects of parenthood on the brain (e.g.,10,18,66). spDCM is a relatively new but highly robust analytical approach to investigate effective connectivity between regions. As it is computationally demanding, spDCM is most suited to models with low dimensionality, and hypothesis-driven seeds. For this reason, we chose to reduce the dimensionality of the model space by examining left-lateralised regions only. We had no strong hypotheses regarding the laterality of the effects, and the studies that informed our selection of ROIs (Fig. 1b) showed a tendency towards left- than right-lateralised results. While it is typical in network neuroimaging to reduce the model dimensionality to aid computation and interpretation of the final results, the results of spDCM are specific to the regions of interest used in the model, and studies using different seed regions may yield different effects. As more studies investigate resting state brain connectivity in motherhood, different brain regions may emerge as those most relevant for understanding motherhood and caregiving behaviour.
Conclusions
In summary, the observed patterns of disinhibition indicate a maternal brain that is more efficient, flexible and responsive at rest. The consistency of this pattern across analyses suggests that these differences generalise across diverse behaviours in the postpartum period, including those that are related to maternal caregiving, as well as non-maternal behaviours, i.e., cognition and social cognition. Together our results support the interpretation of these key left-hemisphere regions as constituting a parental caregiving network, with the nucleus accumbens and the parahippocampal gyrus emerging as ‘hub’ regions, highlighting the global importance of the affective limbic network for maternal caregiving, social cognition, and cognitive performance in the postpartum period.
Methods
The study protocol was approved by Monash University Human Research Ethics Committee (Project ID: 19,455) and conducted in accordance with the Australian National Statement on Ethical Conduct in Human Research (2007).
Participants
Our sample comprised 86 women, 43 first-time mothers approximately 1-year postpartum (11.7 ± 1.7 months) and 43 women who had never been pregnant. All participants were aged over 18 years and right-handed. Inclusion criteria for the study included fluency in English, not currently pregnant, no personal or family history of psychiatric disorder except depression and anxiety, no history of head injury, neurological disorder, epilepsy or stroke, and completion of an MRI safety survey for subsequent MRI scanning (e.g., presence of metal in the body, past surgeries, and claustrophobia). Seven participants were excluded due to incomplete or insufficient fMRI scans: two participants had incomplete scans, one had an insufficient imaging field of view issue, and four had poor image segmentation results. Our final sample included 40 mothers and 39 non-mothers.
Procedure
After providing informed consent, all participants completed a pregnancy test, and then completed a psychosocial and cognitive battery, including paper and computer-based tasks and questionnaires. Demographic information and medical and obstetric history (including information about pregnancy, birth, breast-feeding, and time spent in primary care) was collected via clinical interview. Cognitive measures included the Hopkins Verbal Learning Test (HVLT; verbal memory)80, digit span forward and backward (working memory maintenance and manipulation, respectively)81, Symbol-Digit Modalities Task (SDMT; processing speed)82; Reading the Mind in Films Task (RMFT; Theory of Mind)83, Toronto Empathy Questionnaire84, and the Prospective and Retrospective Memory Questionnaire (PRMQ; subjective memory)85. Psychosocial measures included the Pittsburgh Sleep Quality Index (PSQI)86, Beck Depression Inventory (BDI)87, Beck Anxiety Inventory (BAI)88, Maternal Postnatal Attachment Scale (MPAS)89, and Maternal Self-Efficacy Questionnaire (MEQ)90. Participants completed the PRMQ, BDI, BAI, MPAS, and MEQ questionnaires at a time of their convenience on their own devices (e.g., laptop, phone) via a Qualtrics survey, before attending their testing session. The relationships between cognitive performance, self-perception of memory ability, and wellbeing (sleep, depression and anxiety) in this sample are discussed in detail in Orchard et al.45.
MRI acquisition
All MRI scans were obtained on a Siemens 3 T Skyra MR scanner (Erlangen, Germany) with a 32-channel head and neck coil at Monash Biomedical Imaging, Melbourne, Australia. T1-weighted magnetization-prepared rapid gradient-echo (MP2RAGE) images were acquired (TR = 5000 ms, TE = 2.98 ms, TI 1 = 700 ms, TI 2 = 2500 ms, matrix size = 256 × 240 × 172, bandwidth = 240 Hz/pixel) and used for anatomical reference. T2-weighted images (TR = 3200 ms, TE = 452 ms, matrix size = 206 × 206 × 128, bandwidth = 698 Hz/pixel) were also used for anatomical reference. Resting state fMRI (rsfMRI) data were obtained using a multiband, multi-echo planar imaging (EPI) sequence (TR = 910 ms; multiband acceleration factor = 4; matrix size = 64 × 64; number of slices = 40; TE 1 = 12.60 ms; TE 2 = 29.23 ms; TE 3 = 45.86; TE 4 = 62.49; number of time points = 767; bandwidth = 2520 Hz/pixel; 3.2 mm isotropic voxel). Following functional data acquisition, a reverse-phase scan was acquired to correct the rsfMRI scan for geometric distortions.
MRI processing and denoising
All images were first processed using a standardised pipeline, fmriprep91. Briefly, the pipeline included slice time correction, susceptibility distortion correction using gradient field-maps, co-registration to the corresponding T1w using boundary-based registration with six degrees of freedom. Motion correcting transformations, BOLD-to-T1w transformation and T1w-to-template (MNI) warp were concatenated and applied in a single step. No participants exceeded a previously established head motion exclusion criteria92 (mean framewise displacement > 0.55 mm). For each scan, we then applied an automated TE-dependent ICA-based multi-echo de-noising pipeline, tedana93, resulting in an optimally combined fMRI image94. Images were further denoised by regressing out averaged signals from the white matter and cerebral spinal fluid voxel wise, prior to high-pass filtering (f > 0.005 Hz) and linear detrending. Quality control metrics, functional connectivity matrices and carpet plots were visualised to ensure the pre-processing and denoising steps achieved the desired effects of reducing noise and association between head motion and functional connectivity92. The full quality control protocol and report can be found at: https://github.com/sidchop/NAPPY_DCM.
Data analysis
Behavioural data
Full behavioural methods and results are reported in Orchard et al.45. Briefly, we used principal components analyses (PCA) to compute summary scores using z-scored measures of cognitive performance (Hopkins Verbal Learning Test, Digit Span Forward, Digit Span Backward, and Symbol Digit Modalities Task), social cognition (Reading the Mind in the Films Task, Toronto Empathy Questionnaire), maternal caregiving (Maternal Attachment Questionnaire and Maternal Self-Efficacy Questionnaire), and wellbeing (Pittsburgh Sleep Quality Index, Beck Depression Inventory and Beck Anxiety Inventory). PCs with eigenvalues > 1 were retained95 for further use within the spectral dynamic causal modelling (spDCM) analyses.
Regions of interest
Six regions of interest (ROIs) were identified as key nodes for subsequent spDCM analysis based on the available functional and effective connectivity literature in motherhood (Fig. 1)3,24,27,30,34–36, and compared against the relevant Brodmann areas mapped in MNI space to ensure consistency between region labelling96. To assist this process, we compared the stereotactic coordinates from peak voxels, as well as visually inspected the reported clusters. We ensured none of the identified spheres overlapped with one another, and compared their co-ordinates against the MNI 2009 atlas, taking known grey-matter and white-matter boundaries into consideration. The identified neural network comprised the left dorsomedial prefrontal cortex, ventromedial prefrontal cortex, posterior cingulate cortex, parahippocampal gyrus, amygdala, and nucleus accumbens (Fig. 1a). As spDCM is computationally demanding, it is most suited to models with low dimensionality and hypothesis-driven seeds. Furthermore, there was no evidence to suggest laterality of the effects and studies that informed our selection of ROIs (Fig. 1b) showed a slight tendency towards left- than right-lateralised results. We therefore examined only left-lateralised regions to reduce the model space. To extract BOLD time series corresponding to the six ROIs, we selected the MNI coordinates as the centre of a 5-mm (dorsomedial prefrontal cortex, ventromedial prefrontal cortex, posterior cingulate cortex, and parahippocampal gyrus) or 3-mm sphere (amygdala and nucleus accumbens), which were then used to compute the subject-specific principal eigenvariate and correct for confounds.
Neural network modelling: Spectral dynamic causal modelling (spDCM)
The spDCM analyses were performed using the functions of DCM12.5 (revision 7497) implemented in SPM12 (Supplementary Information 1). In order to assess our main hypothesis, we focused on spDCM analyses that assessed differences in the effective connectivity within the parental caregiving network (1) for mothers versus non-mothers, (2) modulated by cognition and (3) social cognition in mothers versus non-mothers, (4) modulated by maternal caregiving in mothers.
At the first-level, a fully-connected model was created for each participant. Using spDCM, we estimated the DCMs, fitted the complex cross-spectral density using a parameterised power-law model of endogenous neural fluctuations39. The analysis allowed for the measurement of causal interactions between regions, as well as the amplitude and exponent of endogenous neural fluctuations within each region39. Model inversion was based on standard variational Laplace procedures97. This method of Bayesian inference uses Free Energy as a proxy for (log) model evidence, while optimising the posterior density under a Laplace approximation of model parameters.
To characterise how group differences in neural circuitry were modulated by maternal caregiving, cognition, and social cognition, hierarchical models over the parameters were specified within a hierarchical Parametric Empirical Bayes (PEB) framework for DCM98. We based these models on our hypotheses. For each model, all continuous behavioural regressors (cognition, social cognition, and maternal caregiving) were mean centred so that the intercept of each model was interpretable as the group mean connectivity99,100. Group factor was modelled as the main regressor of interest as a vector consisting of 1 (mothers) and − 1 (non-mothers). The interaction terms were created by first centring the continuous variables (cognition, social cognition) and then creating an element-by-element product of the newly centred variables with the categorical grouping variable. Maternal caregiving scores were modelled as main regressor of interest, whereas the remaining scores were modelled as regressors of no interest in all models. For our model of group differences (mothers vs. non-mothers), and for our model of maternal caregiving, we included cognition and social cognition as regressors of no interest. Bayesian model reduction was used to test all reduced models within each parent PEB model, assuming that a different combination of connections could exist98 and ‘pruning’ redundant model parameters. Parameters of the best pruned models in the last Occam’s window were averaged and weighted by their evidence (i.e., Bayesian Model Averaging) to generate final estimates of connection parameters. To identify important effects, i.e., changes in directed connectivity, we compared models using log Bayesian model evidence to ensure the optimal balance between model complexity and accuracy. Models were compared with and without each effect and the posterior probability was calculated for each model using the softmax function of the log Bayes factor. We treat effects (i.e., connection strengths and their changes) with a posterior probability > 0.95 (i.e., strong evidence) as significant for reporting purposes.
Hypothesis 1
To investigate the effect of mothers versus non-mothers on the intrinsic effective connectivity of the parental caregiving network, the following hypothesis was tested within the PEB framework:
Hypothesis 2
To investigate the interaction effect between the mother group and cognition, the following hypothesis was tested within the PEB framework:
Hypothesis 3
To investigate the interaction effect between the mother group and social cognition, the following hypothesis was tested within the PEB framework:
Hypothesis 4
To investigate the effect of maternal caregiving in mothers on the intrinsic effective connectivity of the parental caregiving network, the following hypothesis was tested within the PEB framework:
Finally, following our earlier result45 where we found that differences between mothers and non-mothers in cognition were accounted for by differences in wellbeing, we hypothesised that connectivity in the four comparisons will be affected by poorer wellbeing (sleep, depression, and anxiety). In general, the effect of wellbeing on effective connectivity was modest, and results are provided in Supplementary Information 2.
Supplementary Information
Acknowledgements
The authors are supported by the Australian Research Council (ARC) Centre of Excellence for Integrative Brain Function (CE140100007), and this study received support from an early career researcher project grant from the Centre to ERO. SDJ is supported by an Australian National Health and Medical Research Council Fellowship (APP1174164).
Author contributions
The study was conceptualised by E.R.O., S.D.J., P.G.D.W. & G.F.E. Study funding was sourced by E.R.O. & S.D.J. Data was collected by E.R.O. Data was analysed by E.R.O., K.V., S.C. & T.T. The first draft of the manuscript was written by E.R.O., S.D.J. & K.V. with edits by S.C., T.T., P.G.D.W. & G.F.E. All authors have read and approved the final version of the manuscript.
Data availability
All data supporting the results of this study are presented in the tables and supplements of the article. For access to raw data please contact the corresponding author.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-023-31696-4.
References
- 1.Dufford, A. J., Salzwedel, A. P., Gilmore, J. H., Gao, W. & Kim, P. Maternal trait anxiety symptoms, frontolimbic resting‐state functional connectivity, and cognitive development in infancy. Dev. Psychobiol. (2021). [DOI] [PMC free article] [PubMed]
- 2.Kim P, Dufford AJ, Tribble RC. Cortical thickness variation of the maternal brain in the first 6 months postpartum: associations with parental self-efficacy. Brain Struct. Funct. 2018;223:3267–3277. doi: 10.1007/s00429-018-1688-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Dufford AJ, Erhart A, Kim P. Maternal brain resting-state connectivity in the postpartum period. J. Neuroendocrinol. 2019;31:e12737. doi: 10.1111/jne.12737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kim P, et al. The plasticity of human maternal brain: longitudinal changes in brain anatomy during the early postpartum period. Behav. Neurosci. 2010;124:695–700. doi: 10.1037/a0020884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kim P, et al. Associations between stress exposure and new mothers’ brain responses to infant cry sounds. NeuroImage. 2020;223:117360. doi: 10.1016/j.neuroimage.2020.117360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Luders E, et al. Potential brain age reversal after pregnancy: younger brains at 4–6weeks postpartum. Neuroscience. 2018;386:309–314. doi: 10.1016/j.neuroscience.2018.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Luders E, et al. From baby brain to mommy brain: widespread gray matter gain after giving birth. Cortex. 2020;126:334–342. doi: 10.1016/j.cortex.2019.12.029. [DOI] [PubMed] [Google Scholar]
- 8.Hoekzema E, et al. Pregnancy leads to long-lasting changes in human brain structure. Nat. Neurosci. 2017;20:287–296. doi: 10.1038/nn.4458. [DOI] [PubMed] [Google Scholar]
- 9.Martínez-García M, et al. Do pregnancy-induced brain changes reverse? The brain of a mother 6 years after parturition. Brain Sci. 2021;11:168. doi: 10.3390/brainsci11020168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Orchard ER, et al. Neuroprotective effects of motherhood on brain function in late life: a resting-state fMRI study. Cereb. Cortex. 2021;31:1270–1283. doi: 10.1093/cercor/bhaa293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Orchard ER, et al. Relationship between parenthood and cortical thickness in late adulthood. PLoS One. 2020;15:e0236031. doi: 10.1371/journal.pone.0236031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.de Lange A-MG, et al. History of childbirths relates to region-specific brain aging patterns in middle and older-aged women. BioRxiv. 2020;166(10):400. [Google Scholar]
- 13.de Lange A-MG, et al. Population-based neuroimaging reveals traces of childbirth in the maternal brain. Proc. Natl. Acad. Sci. 2019;116:22341–22346. doi: 10.1073/pnas.1910666116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Voldsbekk I, et al. Women with a history of previous childbirths show less evident white matter brain ageing. bioRxiv. 2020;223:3267. [Google Scholar]
- 15.Orchard ER, Rutherford HJ, Holmes AJ, Jamadar SD. Matrescence: lifetime impact of motherhood on cognition and the brain. Trends Cogn. Sci. 2023;27(3):302–316. doi: 10.1016/j.tics.2022.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cardenas EF, Kujawa A, Humphreys KL. Neurobiological changes during the peripartum period: implications for health and behavior. Soc. Cogn. Affect. Neurosci. 2019 doi: 10.1093/scan/nsz091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Duan C, Cosgrove J, Deligiannidis KM. Understanding peripartum depression through neuroimaging: a review of structural and functional connectivity and molecular imaging research. Curr. Psychiatry Rep. 2017;19:70. doi: 10.1007/s11920-017-0824-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Feldman R. The adaptive human parental brain: implications for children's social development. Trends Neurosci. 2015;38:387–399. doi: 10.1016/j.tins.2015.04.004. [DOI] [PubMed] [Google Scholar]
- 19.Rigo P, et al. Brain processes in mothers and nulliparous women in response to cry in different situational contexts: a default mode network study. Parenting. 2019;19:69–85. doi: 10.1080/15295192.2019.1555430. [DOI] [Google Scholar]
- 20.Witteman J, et al. Towards a neural model of infant cry perception. Neurosci. Biobehav. Rev. 2019;99:23–32. doi: 10.1016/j.neubiorev.2019.01.026. [DOI] [PubMed] [Google Scholar]
- 21.Paul S, et al. Neural pathways of maternal responding: systematic review and meta-analysis. Arch. Womens Ment. Health. 2019;22:179–187. doi: 10.1007/s00737-018-0878-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Strathearn L, Fonagy P, Amico J, Montague PR. Adult attachment predicts maternal brain and oxytocin response to infant cues. Neuropsychopharmacology. 2009;34:2655–2666. doi: 10.1038/npp.2009.103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Noriuchi M, Kikuchi Y, Senoo A. The functional neuroanatomy of maternal love: mother’s response to infant’s attachment behaviors. Biol. Psychiat. 2008;63:415–423. doi: 10.1016/j.biopsych.2007.05.018. [DOI] [PubMed] [Google Scholar]
- 24.Chase HW, Moses-Kolko EL, Zevallos C, Wisner KL, Phillips ML. Disrupted posterior cingulate–amygdala connectivity in postpartum depressed women as measured with resting BOLD fMRI. Soc. Cogn. Affect. Neurosci. 2014;9:1069–1075. doi: 10.1093/scan/nst083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Laurent HK, Ablow JC. A cry in the dark: depressed mothers show reduced neural activation to their own infant’s cry. Soc. Cogn. Affect. Neurosci. 2012;7:125–134. doi: 10.1093/scan/nsq091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Musser ED, Kaiser-Laurent H, Ablow JC. The neural correlates of maternal sensitivity: an fMRI study. Dev. Cogn. Neurosci. 2012;2:428–436. doi: 10.1016/j.dcn.2012.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Moses-Kolko EL, et al. Abnormally reduced dorsomedial prefrontal cortical activity and effective connectivity with amygdala in response to negative emotional faces in postpartum depression. Am. J. Psychiatry. 2010;167:1373–1380. doi: 10.1176/appi.ajp.2010.09081235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Swain JE, Ho SS. Early postpartum resting-state functional connectivity for mothers receiving buprenorphine treatment for opioid use disorder: a pilot study. J. Neuroendocrinol. 2019 doi: 10.1111/jne.12770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Landi N, et al. Maternal neural responses to infant cries and faces: relationships with substance use. Front. Psych. 2011;2:32. doi: 10.3389/fpsyt.2011.00032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Atzil S, Hendler T, Feldman R. Specifying the neurobiological basis of human attachment: brain, hormones, and behavior in synchronous and intrusive mothers. Neuropsychopharmacology. 2011;36:2603–2615. doi: 10.1038/npp.2011.172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Schechter DS, et al. An fMRI study of the brain responses of traumatized mothers to viewing their toddlers during separation and play. Soc. Cogn. Affect. Neurosci. 2012;7:969–979. doi: 10.1093/scan/nsr069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Raichle ME. The restless brain. Brain Connect. 2011;1:3–12. doi: 10.1089/brain.2011.0019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Jamadar S, Egan GF, Calhoun VD, Johnson B, Fielding J. Intrinsic connectivity provides the baseline framework for variability in motor performance: a multivariate fusion analysis of low-and high-frequency resting-state oscillations and antisaccade performance. Brain Connect. 2016;6:505–517. doi: 10.1089/brain.2015.0411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zheng J-X, et al. Disruption within brain default mode network in postpartum women without depression. Medicine. 2020;99:e20045. doi: 10.1097/MD.0000000000020045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Bak Y, Nah Y, Han S, Lee S-K, Shin N-Y. Altered neural substrates within cognitive networks of postpartum women during working memory process and resting-state. Sci. Rep. 2020;10:1–12. doi: 10.1038/s41598-020-66058-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Deligiannidis KM, et al. GABAergic neuroactive steroids and resting-state functional connectivity in postpartum depression: a preliminary study. J. Psychiatr. Res. 2013;47:816–828. doi: 10.1016/j.jpsychires.2013.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Zhang S, et al. Aberrant resting-state interhemispheric functional connectivity in patients with postpartum depression. Behav. Brain Res. 2020;382:112483. doi: 10.1016/j.bbr.2020.112483. [DOI] [PubMed] [Google Scholar]
- 38.Bastos AM, et al. Canonical microcircuits for predictive coding. Neuron. 2012;76:695–711. doi: 10.1016/j.neuron.2012.10.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Razi A, Kahan J, Rees G, Friston KJ. Construct validation of a DCM for resting state fMRI. Neuroimage. 2015;106:1–14. doi: 10.1016/j.neuroimage.2014.11.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Schurz M, Radua J, Aichhorn M, Richlan F, Perner J. Fractionating theory of mind: a meta-analysis of functional brain imaging studies. Neurosci. Biobehav. Rev. 2014;42:9–34. doi: 10.1016/j.neubiorev.2014.01.009. [DOI] [PubMed] [Google Scholar]
- 41.Ferrari C, et al. The dorsomedial prefrontal cortex plays a causal role in integrating social impressions from faces and verbal descriptions. Cereb. Cortex. 2016;26:156–165. doi: 10.1093/cercor/bhu186. [DOI] [PubMed] [Google Scholar]
- 42.De Curtis M, Paré D. The rhinal cortices: a wall of inhibition between the neocortex and the hippocampus. Prog. Neurobiol. 2004;74:101–110. doi: 10.1016/j.pneurobio.2004.08.005. [DOI] [PubMed] [Google Scholar]
- 43.Abu-Akel A, Shamay-Tsoory S. Neuroanatomical and neurochemical bases of theory of mind. Neuropsychologia. 2011;49:2971–2984. doi: 10.1016/j.neuropsychologia.2011.07.012. [DOI] [PubMed] [Google Scholar]
- 44.Euston DR, Gruber AJ, McNaughton BL. The role of medial prefrontal cortex in memory and decision making. Neuron. 2012;76:1057–1070. doi: 10.1016/j.neuron.2012.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Orchard ER, Ward PG, Egan GF, Jamadar SD. Baby-brain phenomena is a subjective experience: absence of evidence for cognitive deficit in new mothers at 1-year postpartum. bioRxiv. 2021;20(2):158. [Google Scholar]
- 46.Snyder, A. D., Ma, L., Steinberg, J. L., Woisard, K. & Moeller, F. G. Dynamic causal modeling self-connectivity findings in the functional magnetic resonance imaging neuropsychiatric literature. Front. Neurosci.15 (2021). [DOI] [PMC free article] [PubMed]
- 47.Friston K. Hierarchical models in the brain. PLoS Comput. Biol. 2008;4:e1000211. doi: 10.1371/journal.pcbi.1000211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ranlund S, et al. Impaired prefrontal synaptic gain in people with psychosis and their relatives during the mismatch negativity. Hum. Brain Mapp. 2016;37:351–365. doi: 10.1002/hbm.23035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Benhamou E, et al. The neurophysiological architecture of semantic dementia: spectral dynamic causal modelling of a neurodegenerative proteinopathy. Sci. Rep. 2020;10:1–13. doi: 10.1038/s41598-020-72847-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zhou Y, et al. Altered intrinsic and extrinsic connectivity in schizophrenia. NeuroImage Clin. 2018;17:704–716. doi: 10.1016/j.nicl.2017.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Almeida JR, et al. Abnormally increased effective connectivity between parahippocampal gyrus and ventromedial prefrontal regions during emotion labeling in bipolar disorder. Psychiatry Res. Neuroimag. 2009;174:195–201. doi: 10.1016/j.pscychresns.2009.04.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Köhler S, et al. Memory impairments associated with hippocampal versus parahippocampal-gyrus atrophy: an MR volumetry study in Alzheimer’s disease. Neuropsychologia. 1998;36:901–914. doi: 10.1016/S0028-3932(98)00017-7. [DOI] [PubMed] [Google Scholar]
- 53.Burgmans S, et al. The posterior parahippocampal gyrus is preferentially affected in age-related memory decline. Neurobiol. Aging. 2011;32:1572–1578. doi: 10.1016/j.neurobiolaging.2009.09.008. [DOI] [PubMed] [Google Scholar]
- 54.Bunce JG, Barbas H. Prefrontal pathways target excitatory and inhibitory systems in memory-related medial temporal cortices. Neuroimage. 2011;55:1461–1474. doi: 10.1016/j.neuroimage.2011.01.064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Alexander GE, DeLong MR, Strick PL. Parallel organization of functionally segregated circuits linking basal ganglia and cortex. Annu. Rev. Neurosci. 1986;9:357–381. doi: 10.1146/annurev.ne.09.030186.002041. [DOI] [PubMed] [Google Scholar]
- 56.Alexander GE, Crutcher MD. Functional architecture of basal ganglia circuits: neural substrates of parallel processing. Trends Neurosci. 1990;13:266–271. doi: 10.1016/0166-2236(90)90107-L. [DOI] [PubMed] [Google Scholar]
- 57.Euston DR, Tatsuno M, McNaughton BL. Fast-forward playback of recent memory sequences in prefrontal cortex during sleep. Science. 2007;318:1147–1150. doi: 10.1126/science.1148979. [DOI] [PubMed] [Google Scholar]
- 58.Lansink CS, Goltstein PM, Lankelma JV, McNaughton BL, Pennartz CM. Hippocampus leads ventral striatum in replay of place-reward information. PLoS Biol. 2009;7:e1000173. doi: 10.1371/journal.pbio.1000173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Vitale, E. M. & Smith, A. S. neurobiology of loneliness, isolation, and loss: integrating human and animal perspectives. Front. Behav. Neurosci. 85 (2022). [DOI] [PMC free article] [PubMed]
- 60.Mannella F, Gurney K, Baldassarre G. The nucleus accumbens as a nexus between values and goals in goal-directed behavior: a review and a new hypothesis. Front. Behav. Neurosci. 2013;7:135. doi: 10.3389/fnbeh.2013.00135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Hikida T, et al. Nucleus accumbens pathways control cell-specific gene expression in the medial prefrontal cortex. Sci. Rep. 2020;10:1–9. doi: 10.1038/s41598-020-58711-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Barrett J, et al. Maternal affect and quality of parenting experiences are related to amygdala response to infant faces. Soc. Neurosci. 2012;7:252–268. doi: 10.1080/17470919.2011.609907. [DOI] [PubMed] [Google Scholar]
- 63.Seifritz E, et al. Differential sex-independent amygdala response to infant crying and laughing in parents versus nonparents. Biol. Psychiat. 2003;54:1367–1375. doi: 10.1016/S0006-3223(03)00697-8. [DOI] [PubMed] [Google Scholar]
- 64.Swain JE, et al. Maternal brain response to own baby-cry is affected by cesarean section delivery. J. Child Psychol. Psychiatry. 2008;49:1042–1052. doi: 10.1111/j.1469-7610.2008.01963.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Barrett J, Fleming AS. Annual Research review: all mothers are not created equal: neural and psychobiological perspectives on mothering and the importance of individual differences. J. Child Psychol. Psychiatry. 2011;52:368–397. doi: 10.1111/j.1469-7610.2010.02306.x. [DOI] [PubMed] [Google Scholar]
- 66.Swain JE, Lorberbaum JP, Kose S, Strathearn L. Brain basis of early parent-infant interactions: psychology, physiology, and in vivo functional neuroimaging studies. J. Child Psychol. Psychiatry. 2007;48:262–287. doi: 10.1111/j.1469-7610.2007.01731.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Kim P, Strathearn L, Swain JE. The maternal brain and its plasticity in humans. Horm. Behav. 2016;77:113–123. doi: 10.1016/j.yhbeh.2015.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Strathearn L, Li J, Fonagy P, Montague PR. What's in a Smile? Maternal brain responses to infant facial cues. Pediatrics. 2008;122:40–51. doi: 10.1542/peds.2007-1566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Babaev O, Piletti Chatain C, Krueger-Burg D. Inhibition in the amygdala anxiety circuitry. Exp. Mol. Med. 2018;50:1–16. doi: 10.1038/s12276-018-0063-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Clancy K, Ding M, Bernat E, Schmidt NB, Li W. Restless ‘rest’: intrinsic sensory hyperactivity and disinhibition in post-traumatic stress disorder. Brain. 2017;140:2041–2050. doi: 10.1093/brain/awx116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Wang J, Tian Y, Zeng L-H, Xu H. Prefrontal disinhibition in social fear: a vital action of somatostatin interneurons. Front. Cell. Neurosci. 2020;14:611732. doi: 10.3389/fncel.2020.611732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Leckman JF, Mayes LC. Preoccupations and behaviors associated with romantic and parental love: perspectives on the origin of obsessive-compulsive disorder. Child Adolesc. Psychiatr. Clin. N. Am. 1999;8:635–665. doi: 10.1016/S1056-4993(18)30172-X. [DOI] [PubMed] [Google Scholar]
- 73.Lonstein JS, Levy F, Fleming AS. Common and divergent psychobiological mechanisms underlying maternal behaviors in non-human and human mammals. Horm. Behav. 2015;73:156–185. doi: 10.1016/j.yhbeh.2015.06.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Feygin DL, Swain JE, Leckman JF. The normalcy of neurosis: evolutionary origins of obsessive–compulsive disorder and related behaviors. Prog. Neuropsychopharmacol. Biol. Psychiatry. 2006;30:854–864. doi: 10.1016/j.pnpbp.2006.01.009. [DOI] [PubMed] [Google Scholar]
- 75.Field T. Postpartum depression effects on early interactions, parenting, and safety practices: a review. Infant Behav. Dev. 2010;33:1–6. doi: 10.1016/j.infbeh.2009.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.King DR, de Chastelaine M, Elward RL, Wang TH, Rugg MD. Recollection-related increases in functional connectivity predict individual differences in memory accuracy. J. Neurosci. 2015;35:1763–1772. doi: 10.1523/JNEUROSCI.3219-14.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Cabeza R, et al. Maintenance, reserve and compensation: the cognitive neuroscience of healthy ageing. Nat. Rev. Neurosci. 2018;19:701–710. doi: 10.1038/s41583-018-0068-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Goulden N, et al. Sample size estimation for comparing parameters using dynamic causal modeling. Brain Connect. 2012;2:80–90. doi: 10.1089/brain.2011.0057. [DOI] [PubMed] [Google Scholar]
- 79.Voigt K, Razi A, Harding IH, Andrews ZB, Verdejo-Garcia A. Neural network modelling reveals changes in directional connectivity between cortical and hypothalamic regions with increased BMI. Int. J. Obes. 2021;45:2447–2454. doi: 10.1038/s41366-021-00918-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Brandt J. The Hopkins verbal learning test: development of a new memory test with six equivalent forms. Clin. Neuropsychol. 1991;5:125–142. doi: 10.1080/13854049108403297. [DOI] [Google Scholar]
- 81.Woods DL, et al. Improving digit span assessment of short-term verbal memory. J. Clin. Exp. Neuropsychol. 2011;33:101–111. doi: 10.1080/13803395.2010.493149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Smith, A. The symbol-digit modalities test: a neuropsychologic test of learning and other cerebral disorders. Learn. Disord. 83–91 (1968).
- 83.Golan O, Baron-Cohen S, Hill JJ, Golan Y. The, “reading the mind in films” task: complex emotion recognition in adults with and without autism spectrum conditions. Soc. Neurosci. 2006;1:111–123. doi: 10.1080/17470910600980986. [DOI] [PubMed] [Google Scholar]
- 84.Spreng RN, McKinnon MC, Mar RA, Levine B. The Toronto empathy questionnaire: scale development and initial validation of a factor-analytic solution to multiple empathy measures. J. Person. Assess. 2009;91:62–71. doi: 10.1080/00223890802484381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Smith G, Del Sala S, Logie RH, Maylor EA. Prospective and retrospective memory in normal ageing and dementia: a questionnaire study. Memory. 2000;8:311–321. doi: 10.1080/09658210050117735. [DOI] [PubMed] [Google Scholar]
- 86.Buysse DJ, Reynolds CF, III, Monk TH, Berman SR, Kupfer DJ. The Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiatry Res. 1989;28:193–213. doi: 10.1016/0165-1781(89)90047-4. [DOI] [PubMed] [Google Scholar]
- 87.Beck AT, Ward CH, Mendelson M, Mock J, Erbaugh J. An inventory for measuring depression. Arch. Gen. Psychiatry. 1961;4:561–571. doi: 10.1001/archpsyc.1961.01710120031004. [DOI] [PubMed] [Google Scholar]
- 88.Beck AT, Epstein N, Brown G, Steer RA. An inventory for measuring clinical anxiety: psychometric properties. J. Consult. Clin. Psychol. 1988;56:893. doi: 10.1037/0022-006X.56.6.893. [DOI] [PubMed] [Google Scholar]
- 89.Condon, J. T. Maternal Postnatal Attachment Scale [measurement instrument]. 10.25957/5dc34bb22f714 (2015).
- 90.Teti DM, Gelfand DM. Behavioral competence among mothers of infants in the first year: the mediational role of maternal self-efficacy. Child Dev. 1991;62:918–929. doi: 10.2307/1131143. [DOI] [PubMed] [Google Scholar]
- 91.Esteban O, et al. fMRIPrep: a robust preprocessing pipeline for functional MRI. Nat. Methods. 2019;16:111–116. doi: 10.1038/s41592-018-0235-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Parkes L, Fulcher B, Yucel M, Fornito A. An evaluation of the efficacy, reliability, and sensitivity of motion correction strategies for resting-state functional MRI. Neuroimage. 2018;171:415–436. doi: 10.1016/j.neuroimage.2017.12.073. [DOI] [PubMed] [Google Scholar]
- 93.Kundu P, Inati SJ, Evans JW, Luh W-M, Bandettini PA. Differentiating BOLD and non-BOLD signals in fMRI time series using multi-echo EPI. Neuroimage. 2012;60:1759–1770. doi: 10.1016/j.neuroimage.2011.12.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Ahmed, Z. et al.https://zenodo.org/record/6461353#.YxEopezMI6461350Q.
- 95.Field, A. Discovering Statistics Using IBM SPSS Statistics. (sage, 2013).
- 96.Project, Y. B. S. W.
- 97.Friston K, Mattout J, Trujillo-Barreto N, Ashburner J, Penny W. Variational free energy and the Laplace approximation. Neuroimage. 2007;34:220–234. doi: 10.1016/j.neuroimage.2006.08.035. [DOI] [PubMed] [Google Scholar]
- 98.Friston K, et al. Bayesian model reduction and empirical Bayes for group (DCM) studies. Neuroimage. 2016;128:413–431. doi: 10.1016/j.neuroimage.2015.11.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Zeidman P, et al. A guide to group effective connectivity analysis, part 1: first level analysis with DCM for fMRI. Neuroimage. 2019;200:174–190. doi: 10.1016/j.neuroimage.2019.06.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Zeidman P, et al. A guide to group effective connectivity analysis, part 2: second level analysis with PEB. Neuroimage. 2019;200:12–25. doi: 10.1016/j.neuroimage.2019.06.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data supporting the results of this study are presented in the tables and supplements of the article. For access to raw data please contact the corresponding author.