Abstract
Background
To investigate the risk factors of pancreatitis after endoscopic retrograde cholangiopancreatography (ERCP) in patients with biliary tract diseases.
Methods
We retrospectively analyzed the clinical data of 480 patients who underwent ERCP for biliary tract diseases at the Affiliated Zhongshan Hospital of Dalian University from October 2011 to October 2016. The patients were divided into a study group (n = 75, with PEP) and a control group (n = 405, without PEP) based on whether they developed post-ERCP pancreatitis (PEP), and their clinical baseline data and intraoperative conditions were retrieved and compared. Then, factors associated with PEP were analyzed using logistic regression model, based on which a nomogram prediction model was constructed. The receiver operating characteristic (ROC) curve and calibration curve were used to evaluate the performance of the prediction model.
Results
Significant differences in age, sex, history of pancreatitis, history of choledocholithiasis, pancreatic duct imaging, pancreatic sphincterotomy, difficult cannulation, multiple cannulation attempts and juxtapapillary duodenal diverticula were observed between the two groups. Multivariate logistic regression analysis showed that age less than 60 years (OR, 0.477; 95% CI, 0.26–0.855), female sex (OR, 2.162; 95% CI, 1.220–3.831), history of pancreatitis (OR, 2.567; 95% CI, 1.218–5.410), history of choledocholithiasis (OR, 2.062; 95% CI, 1.162–3.658), pancreatic sphincterotomy (OR, 2.387; 95% CI, 1.298–4.390), pancreatic duct imaging (OR, 4.429; 95% CI, 1.481–13.242), multiple cannulation attempts (OR, 2.327; 95% CI, 1.205–4.493), difficult cannulation (OR, 2.421; 95% CI, 1.143–5.128), and JPD (OR, 2.002; 95% CI, 1.125–3.564) were independent risk factors for PEP. The nomogram for predicting the occurrence of PEP demonstrated an area under the ROC curve (AUC) of 0.787, and the calibration curves of the model showed good consistency between the predicted and actual probability of PEP.
Conclusion
Our results showed that age less than 60 years, female sex, history of pancreatitis, history of choledocholithiasis, pancreatic sphincterotomy, pancreatic duct imaging, multiple cannulation attempts, difficult cannulation and juxtapapillary duodenal diverticula were independent risk factors for PEP. In addition, the established nomogram demonstrated promising clinical efficacy in predicting PEP risk in patients who underwent ERCP for biliary tract diseases.
Keywords: Biliary tract disease, Endoscopic retrograde cholangiopancreatography, Risk factors, Postoperative pancreatitis
Background
Endoscopic retrograde cholangiopancreatography (ERCP) was first performed by McCune et al. in 1968 [1], and the first successful case in China was reported in 1973 [2]. This procedure is now commonly used to diagnose and treat biliary tract and pancreatic diseases [3]. Although ERCP is considered safe and effective in treating biliary tract diseases [4, 5], its rate of complications and mortality of ERCP over a decade was reported to be 10-12% and 0.4-1.4%, respectively [6–8].
The main complications after ERCP are pancreatitis, bleeding, cholecystitis, infection and intestinal perforation. Among them, post-ERCP pancreatitis (PEP) is the most common, with an incidence between 1.6 and 15% [9, 10]. Most cases of PEP are mild or moderate, and only a few present with severe conditions. Epidemiological studies showed that about 1.5% of PEP cases were moderate to severe, and the overall mortality rate of PEP was approximately 3% [9]. In severe cases, PEP can lead to multiple organ dysfunction, thereby increasing the risk of treatment-related mortality. The treatment of PEP requires substantial healthcare resources and usually needs prolonged hospitalization, increasing the psychosocial burden of endoscopists [11, 12]. Thus, timely identifying related risk factors could be critical for preventing and reducing the incidence of PEP.
In this study, we used univariate and multivariate analysis to identify the influence of patient baseline data and intraoperative conditions on the incidence of PEP and screened out independent risk factors to build a predicting score model that clinicians could use for targeted treatment and interventions.
Materials and methods
General information
This study was approved by the Ethics Committee of the Affiliated Zhongshan Hospital of Dalian University, following the clinical data of patients who underwent ERCP for biliary tract diseases from October 2011 to October 2016 were retrospectively analyzed. The study inclusion criteria were [13]: (1) aged 18–80 years old; (2) the diagnosis of PEP was made following the criteria developed by Cotton et al. in 1991 [14]: occurrence of persistent pancreatitis-related symptoms (i.e., new or aggravated abdominal pain) after ERCP, accompanied by an increase in serum amylase level by more than 3 times the normal limit within 24 h after surgery, and requiring hospitalization for more than 1 day; (3) patients with normal serum amylase or < 3 times higher than the normal limit before ERCP; and (4) patients with complete preoperative biochemical examinations for blood and urine, liver and kidney function tests, hematuria amylase, blood lipids and blood glucose, as well as electrocardiogram, abdominal color ultrasound or computed tomography, and postoperative examinations of blood biochemistry, serum amylase, liver function and others. The exclusion criteria were: (1) patients with benign and malignant tumors of the biliopancreatic system; (2) the presence of benign and malignant duodenal stenosis; (3) non-native papilla (including post-sphincterotomy, post-papillectomy, post-papillary balloon dilatation, and post-choledochojejunostomy); and (4) missing indicators that could have affected the main statistical analysis of this study.
ERCP procedure
All ERCP procedures in this study were performed by experienced teams, which included an endoscopist with at least 100 successfully completed ERCP per year and 10 or more years of experience, an endoscopy nurse adequately skilled in the technique, and a physician. The patients were orally anesthetized using a 10 mL dyclonine capsule for throat mucosa anesthesia and intravenous combined anesthesia with 3–5 mg/kg per hours propofol pump for those intolerant. Fujitcan’s duodenoscope system ED-530 XT electronic endoscopic system was used, tentative insertion along the oesophagus, enter the duodenal bulb, incorporating the duodenal papilla into the center of the field of view, observed the morphology and structure of the nipple in detail. The selection of bile and pancreatic duct cannulation was according to the size and morphology of the duodenal papilla, opening and wrinkle, local presence or absence of diverticula and the relationship between the duodenal papilla and diverticulum. In cases of difficult intubation, the precut sphincterotomy or needle knife was employed to gain access to the common bile duct (CBD) when standard methods using catheters, cannulatomes and guidewires have failed. After successful intubation, bile and pancreatic fluid was withdrawn and then appropriate contrast agent was injected. According to the patient’s condition, endoscopic sphincterotomy (EST), duodenal papillary balloon dilatation, net basket or balloon stone extraction, pancreatic duct stent placement or bile duct stent placement was performed. All included patients were treated with prophylactic 100 mg rectal indomethacin before the procedure.
Baseline indicators
Baseline data were collected from all patients, including age, sex, history of pancreatitis, history of diabetes, history of hypertension, history of previous ERCP, history of previous cholecystectomy, history of choledocholithiasis, total bilirubin, and triglyceride levels. Whether patients had the following intraoperative conditions were observed and recorded, including common bile duct dilatation, pancreatic duct imaging (pancreatic duct can be observed below X ray after contrast injection), EST {the sphincterotomy is extended to expose the biliary lumen and the biliary duct can be cannulated, including pancreatic sphincterotomy, biliary sphincterotomy, duodenal minor papilla sphincterotomy (use a needle-knife or wire-assisted access sphincterotomy to dorsal pancreatic duct drainage when patients with recurrent acute or chronic pancreatitis who have complete or incomplete pancreas divisum)}, duodenal papillary balloon dilatation (a 8-mm diameter balloon was placed in the papilla to dilate, used as an alternative to EST for extracting CBD stones < 8 mm in patients, choledocholithiasis is the only indication), difficult cannulation (According to the European Digestive Endoscopy Association (ESGE) guidelines [15], defined as more than five contacts with the papilla prior to successful cannulation; more than 5 min of cannulation time following visualization of the papilla; and more than one inadvertent pancreatic duct (PD) cannulation or opacification), multiple cannulation attempts (≥ 3 times), operation time more than 60 min, and juxtapapillary duodenal diverticula.
Statistical analysis
Data analysis was performed using the SPSS software (version 26.0). Count data are presented as n (%), and measurement data as mean ± SD. Comparisons between groups were performed using the χ2 test or Fisher’s exact test. Univariate and multivariate logistic regression models were used to analyze the relationship between clinical data and PEP to determine the independent risk factors for PEP. The obtained independent risk factors were used to construct a nomogram model using the “rms” package of the R software, which could be used to assess the risk of PEP. Additionally, a calibration curve and receiver operating characteristic (ROC) curve were drawn to evaluate the performance of the nomogram. P < 0.05 was used to indicate a significant difference.
Results
Comparison of baseline characteristics between the groups
Based on the inclusion and exclusion criteria, a total of 480 patients were found eligible for this study. Those who developed PEP were assigned to the study group (n = 75), while those who did not develop PEP were assigned to the control group (n = 405). The clinical baseline data of all patients are shown in Table 1. There were significant differences between the study group and control group in age (< 60 years; 45.3% vs. 31.6%), sex (female; 60.0% vs. 37.3%), history of pancreatitis (yes; 26.7% vs. 7.9%), and history of choledocholithiasis (yes; 42.7% vs. 28.9%) (all P < 0.05). However, no marked differences were found between the two groups in terms of history of diabetes, hypertension, ERCP and its indications, cholecystectomy, total bilirubin and triglyceride (P > 0.05).
Table 1.
Comparison of baseline data between the two groups
| Baseline data | Control group (n = 405) |
Study group (n = 75) |
X2 | P |
|---|---|---|---|---|
| Age (%) | 5.334 | 0.021 | ||
| < 60 | 128(31.6) | 34(45.3) | ||
| ≥ 60 | 277(68.4) | 41(54.7) | ||
| Sex (%) | 13.516 | <0.001 | ||
| Male | 254(62.7) | 30(40.0) | ||
| Female | 151(37.3) | 45(60.0) | ||
| History of pancreatitis (%) | 23.069 | <0.001 | ||
| No | 373(92.1) | 55(73.3) | ||
| Yes | 32(7.9) | 20(26.7) | ||
| History of diabetes (%) | 0.889 | 0.346 | ||
| No | 321(79.3) | 63(84.0) | ||
| Yes | 84(20.7) | 12(16.0) | ||
| History of hypertension (%) | 0.026 | 0.872 | ||
| No | 277(68.4) | 52(69.3) | ||
| Yes | 128(31.6) | 23(30.7) | ||
| History of ERCP (%) | 1.504 | 0.220 | ||
| No | 285(70.4) | 58(77.3) | ||
| Yes | 120(29.6) | 17(22.7) | ||
| Indication for ERCP (%) | ||||
| Choledocholithiasis | 203(50.1) | 46(61.3) | 3.185 | 0.074 |
| biliary stricture | 0(0) | 1(1.3) | - | 0.156 |
| pancreatitis | 39(9.6) | 12(16.0) | 2.704 | 0.100 |
| cholangeitis | 216(53.3) | 45(60.0) | 1.134 | 0.287 |
| Others | 110(27.2) | 17(22.7) | 0.657 | 0.418 |
| History of cholecystectomy (%) | 0.558 | 0.455 | ||
| No | 358(88.4) | 64(85.3) | ||
| Yes | 47(11.6) | 11(14.7) | ||
| History of choledocholithiasis (%) | 5.612 | 0.018 | ||
| No | 288(71.1) | 43(57.3) | ||
| Yes | 117(28.9) | 32(42.7) | ||
| Total bilirubin | 78.93 ± 95.77 | 65.85 ± 92.76 | 1.091 | 0.276 |
| Triglyceride | 1.52 ± 0.85 | 1.45 ± 0.68 | 0.751 | 0.453 |
Count data are expressed as n (%), and measurement data as mean ± SD. ERCP, Endoscopic retrograde cholangiopancreatography; study group, patients developed pancreatitis after ERCP; control group, patients did not develop pancreatitis after ERCP.
Comparison of intraoperative conditions between the two groups
The intraoperative conditions of patients are shown in Table 2. Compared with the control group, the study group had a significantly higher proportion in pancreatic duct imaging, pancreatic sphincterotomy, pancreatic stents, difficult cannulation, multiple cannulation attempts, and juxtapapillary duodenal diverticula (all P < 0.05). However, there was no significant difference between the two groups in intraoperative common bile duct dilatation, biliary sphincterotomy, duodenal papillary sphincterotomy, duodenal papillary balloon dilatation, and operation time exceeding 60 min (all P > 0.05).
Table 2.
Comparison of intraoperative conditions of patients between the two groups
| Intraoperative conditions | Control group (n = 405) |
Study group (n = 75) |
X2 | P |
|---|---|---|---|---|
| Common bile duct dilatation (%) | 0.382 | 0.537 | ||
| No | 356(87.9) | 64(85.3) | ||
| Yes | 49(12.1) | 11(14.7) | ||
| Pancreatic duct imaging (%) | 16.083 | <0.001 | ||
| No | 395(97.5) | 65(86.7) | ||
| Yes | 10(2.5) | 10(13.3) | ||
| Pancreatic sphincterotomy (%) | 19.079 | <0.001 | ||
| No | 322(79.5) | 42(56.0) | ||
| Yes | 83(20.5) | 33(44.0) | ||
| Biliary sphincterotomy (%) | 1.757 | 0.185 | ||
| No | 290(71.6) | 48(64.0) | ||
| Yes | 115(28.4) | 27(36.0) | ||
| Duodenal minor papilla sphincterotomy (%) | 1.023 | 0.312 | ||
| No | 109(26.9) | 16(21.3) | ||
| Yes | 296(73.1) | 59(78.7) | ||
| Duodenal papillary balloon dilatation (%) | 0.745 | 0.388 | ||
| No | 243(60.0) | 41(54.7) | ||
| Yes | 162(40.0) | 34(45.3) | ||
| Pancreatic stents (%) | 7.255 | 0.007 | ||
| No | 335(82.7) | 52(69.3) | ||
| Yes | 70(17.3) | 23(30.7) | ||
| Difficult cannulation (%) | 14.294 | <0.001 | ||
| No | 372(91.9) | 58(77.3) | ||
| Yes | 33(8.1) | 17(22.7) | ||
| Multiple cannulation attempts (%) | 10.268 | 0.001 | ||
| < 3 times | 347(85.7) | 53(70.7) | ||
| ≥ 3 times | 58(14.3) | 22(29.3) | ||
| Operation time (%) | 1.583 | 0.208 | ||
| <60 min | 347(85.7) | 60(80.0) | ||
| ≥ 60 min | 58(14.3) | 15(20.0) | ||
| Juxtapapillary duodenal diverticula (%) | 15.477 | <0.001 | ||
| No | 279(68.9) | 34(45.3) | ||
| Yes | 126(31.1) | 41(54.7) |
Count data are expressed as n (%)
Risk factors for post-ERCP pancreatitis
Univariate analysis of the general data and intraoperative conditions of the included patients showed that age less than 60, female sex, history of pancreatitis, history of choledocholithiasis, pancreatic sphincterotomy, pancreatic duct imaging, multiple cannulation attempts, difficult cannulation and juxtapapillary duodenal diverticula were significantly associated with PEP (P < 0.05), while no significant difference was observed for history of diabetes, hypertension, ERCP and cholecystectomy, total bilirubin, triglyceride, common bile duct dilatation, biliary sphincterotomy, duodenal minor papilla sphincterotomy, duodenal papillary balloon dilatation, pancreatic stents and operation time more than 60 min (P > 0.05) (Table 3).
Table 3.
Univariate analysis of risk factors for post-ERCP pancreatitis
| Variables | B | S.E. | Wald | P | OR | 95%CI | |
|---|---|---|---|---|---|---|---|
| Lower limit | Upper limit | ||||||
| Age (< 60) | -0.585 | 0.255 | 5.243 | 0.022 | 0.557 | 0.338 | 0.919 |
| Sex (female) | 0.926 | 0.257 | 12.956 | <0.001 | 2.523 | 1.524 | 4.177 |
| History of pancreatitis | 1.444 | 0.320 | 20.427 | <0.001 | 4.239 | 2.266 | 7.929 |
| History of diabetes | -0.318 | 0.338 | 0.883 | 0.347 | 0.728 | 0.375 | 1.412 |
| History of hypertension | -0.044 | 0.272 | 0.026 | 0.872 | 0.957 | 0.561 | 1.632 |
| History of ERCP | -0.362 | 0.296 | 1.493 | 0.222 | 0.696 | 0.389 | 1.245 |
| History of cholecystectomy | 0.269 | 0.361 | 0.556 | 0.456 | 1.309 | 0.645 | 2.658 |
| History of choledocholithiasis | 0.605 | 0.258 | 5.508 | 0.019 | 1.832 | 1.105 | 3.037 |
| Total bilirubin | -0.002 | 0.001 | 1.181 | 0.277 | 0.998 | 0.995 | 1.001 |
| Triglyceride | -0.126 | 0.167 | 0.565 | 0.452 | 0.882 | 0.635 | 1.224 |
| Common bile duct dilatation | 0.222 | 0.360 | 0.380 | 0.537 | 1.249 | 0.616 | 2.530 |
| Pancreatic duct imaging | 1.804 | 0.467 | 14.943 | <0.001 | 6.077 | 2.434 | 15.172 |
| Pancreatic sphincterotomy | 1.115 | 0.263 | 17.934 | <0.001 | 3.048 | 1.820 | 5.106 |
| Biliary sphincterotomy | 0.350 | 0.265 | 1.745 | 0.186 | 1.418 | 0.844 | 2.383 |
| Duodenal minor papilla sphincterotomy | 0.306 | 0.303 | 1.017 | 0.313 | 1.358 | 0.749 | 2.461 |
| Duodenal papillary balloon dilatation | 0.218 | 0.253 | 0.743 | 0.389 | 1.244 | 0.757 | 2.043 |
| Pancreatic stents | 0.750 | 0.283 | 7.031 | 0.008 | 2.117 | 1.216 | 3.685 |
| Difficult cannulation | 1.195 | 0.330 | 13.098 | <0.001 | 3.304 | 1.730 | 6.312 |
| Multiple cannulation attempts (≥ 3 times) | 0.910 | 0.291 | 9.798 | 0.002 | 2.483 | 1.405 | 4.389 |
| Operation time (≥ 60 min) | 0.403 | 0.322 | 1.567 | 0.211 | 1.496 | 0.796 | 2.809 |
| Juxtapapillary duodenal diverticulum | 0.982 | 0.256 | 14.767 | <0.001 | 2.670 | 1.618 | 4.406 |
Multivariate logistic regression analysis (Table 4) was then performed to identify independent factors associated with the risk of developing PEP. The results showed that age less than 60 years (OR, 0.477; 95% CI, 0.26–0.855), female sex (OR, 2.162; 95% CI, 1.220–3.831), history of pancreatitis (OR, 2.567; 95% CI, 1.218–5.410), history of choledocholithiasis (OR, 2.062; 95% CI, 1.162–3.658), pancreatic sphincterotomy (OR, 2.387; 95% CI, 1.298–4.390), pancreatic duct imaging (OR, 4.429; 95% CI, 1.481–13.242), multiple cannulation attempts (OR, 2.327; 95% CI, 1.205–4.493), difficult cannulation (OR, 2.421; 95% CI, 1.143–5.128) and juxtapapillary duodenal diverticula (OR, 2.002; 95% CI, 1.481–13.242) 1.125–3.564) were independent risk factors for PEP (P < 0.05).
Table 4.
Multivariate analysis of risk factors for post-ERCP pancreatitis
| Variables | B | S.E. | Wald | P | OR | 95%CI | |
|---|---|---|---|---|---|---|---|
| Lower limit | Upper limit | ||||||
| Age (< 60) | -0.740 | 0.298 | 6.180 | 0.013 | 0.477 | 0.266 | 0.855 |
| Sex (female) | 0.771 | 0.292 | 6.983 | 0.008 | 2.162 | 1.220 | 3.831 |
| History of pancreatitis | 0.943 | 0.380 | 6.146 | 0.013 | 2.567 | 1.218 | 5.410 |
| History of choledocholithiasis | 0.724 | 0.293 | 6.115 | 0.013 | 2.062 | 1.162 | 3.658 |
| Pancreatic duct imaging | 1.488 | 0.559 | 7.090 | 0.008 | 4.429 | 1.481 | 13.242 |
| Pancreatic sphincterotomy | 0.870 | 0.311 | 7.831 | 0.005 | 2.387 | 1.298 | 4.390 |
| Difficult cannulation | 0.884 | 0.383 | 5.330 | 0.021 | 2.421 | 1.143 | 5.128 |
| Multiple cannulation attempts (≥ 3 times) | 0.845 | 0.336 | 6.328 | 0.012 | 2.327 | 1.205 | 4.493 |
| Juxtapapillary duodenal diverticulum | 0.694 | 0.294 | 5.570 | 0.018 | 2.002 | 1.125 | 3.564 |
Establishment and validation of a predicting model for estimating PEP risk
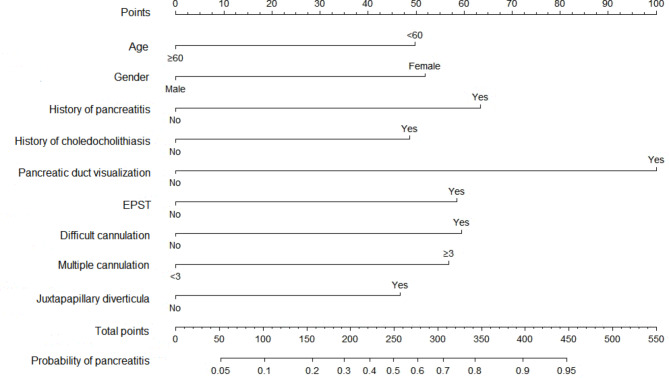
A nomogram comprising nine covariates was constructed based on the results of the multivariate logistic regression model (Fig. 1). Each risk factor was assigned a score, and the probability of PEP was inferred by summing the scores of all factors. As shown in Fig. 2, the standard curve fitted well with the calibrated prediction curve, indicating satisfactory consistency between the predicted and the actual incidence of PEP in our study cohort.
Fig. 1.
Nomogram for predicting the probability of post-ERCP pancreatitis
Abbreviations: EPST, endoscopic pancreatic sphincterotomy.
Fig. 2.
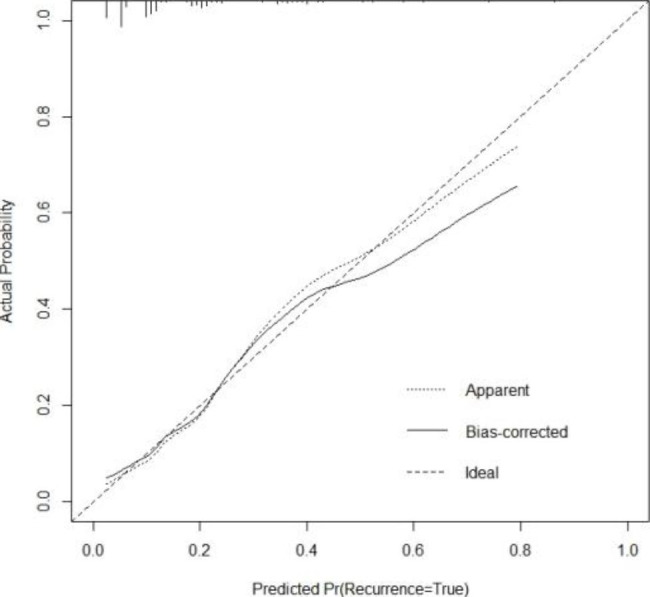
Calibration plot of the constructed nomogram
Note: Apparent indicates the prediction curve without calibration; Bias-corrected indicates the calibrated prediction curve; Ideal represents the standard curve; predicted Pr is the predicted incidence of post-ERCP pancreatitis; and Actual Probability indicates the actual incidence of post-ERCP pancreatitis
Receiver operator characteristic (ROC) curve
The performance of the nomogram model in predicting the risk of PEP was assessed by plotting the ROC curve, which showed an AUC of 0.787 and a 95% CI ranging between 0.726 and 0.847, indicating a high predictive value of the prediction model (P < 0.05) (Fig. 3).
Fig. 3.
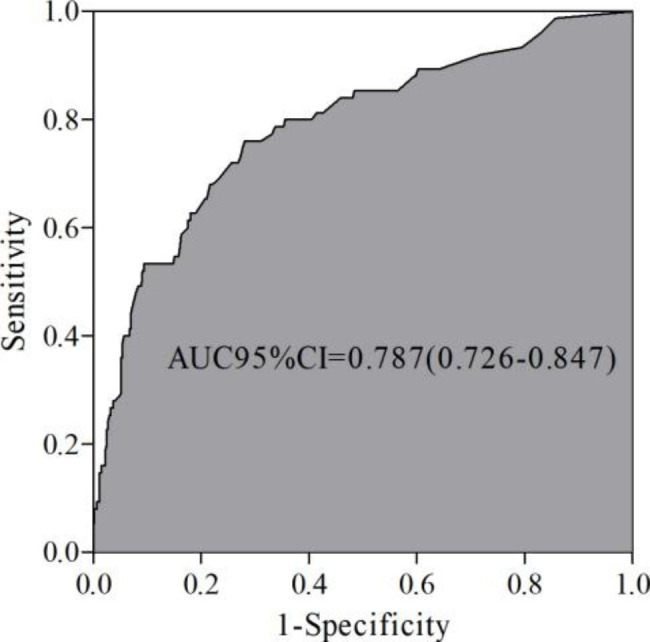
Receiver operator characteristic (ROC) curve
Discussion
PEP, a common complication of ERCP, can significantly increase the risk of mortality of the patients and healthcare-associated costs [16], indicating the need to strategize newer strategies to reduce the incidence of post-ERCP complications. However, before developing specific interventions, it is important to understand the risk factors for PEP. More and more influencing factors have been found with the deepening of research, including individual differences of patients, the experience level of endoscopists, and many clinical factors [17]. In this present study, we analyzed the risk factors associated with PEP, based on which we constructed a scoring model that could be used as a reference for the targeted prevention of PEP, thereby reducing its incidence and additional medical costs and improving therapeutic outcomes.
The results showed that there were significant differences between the study group and control group in age, sex, history of pancreatitis, history of choledocholithiasis, pancreatic duct imaging, pancreatic sphincterotomy, pancreatic stents, difficult cannulation, multiple cannulation attempts, and juxtapapillary duodenal diverticula. Further univariate and multivariate analysis confirmed that age (< 60 years), sex (female), history of pancreatitis, history of choledocholithiasis, pancreatic sphincterotomy, pancreatic duct imaging, multiple cannulation attempts, difficult cannulation and juxtapapillary duodenal diverticula were associated with PEP and were independent risk factors for PEP, which was consistent with previous findings [17, 18]. Studies have shown that the prevalence of PEP is significantly higher in patients younger than 60 years of age than in elderly patients [19, 20], possibly due to the decrease in the exocrine function of the pancreas with increasing age. When the pancreas is mildly injured, the body is stressed, leading to a sudden increase in pancreatic juice and further aggravating pancreatic injury. Gender differences can cause different body structures, functions, and hormone secretion levels, resulting in a higher probability of PEP in women than in men [21]. In addition, studies have reported that patients with a history of pancreatitis, choledocholithiasis, intraoperative pancreatic sphincterotomy, and intraoperative pancreatic duct imaging are independent risk factors for PEP [22]. This is because such patients might have an underlying impaired pancreatic juice excretion, making them more likely to suffer from acute pancreatitis after ERCP-caused irritation to the pancreas [23].
Multiple imagings of the pancreatic duct were found to increase the incidence of PEP [24]. Pancreatography increases pressure in the pancreatic duct, resulting in pancreatic juice reflux and increasing the risk of acute pancreatitis post-ERCP. Electrocoagulation performed during pancreatic sphincterotomy can further damage the pancreatic duct, easily leading to acute pancreatitis after ERCP [25]. Therefore, appropriate clinical treatment and intervention can be given to patients with a history of pancreatitis, choledocholithiasis, pancreatic duct imaging, and pancreatic sphincterotomy to reduce the risk of PEP. Difficult cannulation and multiple cannulation attempts are also risk factors for PEP. Repeated attempts at cannulating the duodenal papilla can lead to papillary injury, papillary swelling, sphincter relaxation and contraction dysfunction, hindering the discharge of pancreatic juice from the pancreatic duct and resulting in the accumulation of juice in the pancreatic duct. As a result, the pressure in the pancreatic duct is increased, causing damage to pancreatic duct epithelial cells and acini, activation of pancreatic enzymes, and induction of PEP [26]. Further, it is known that the juxtapapillary duodenal diverticula is a contributor to PEP. Residual food in the duodenal diverticulum or huge diverticulum compresses the biliary and pancreatic sphincters, inducing contraction and cramps of the biliary sphincter and poor bile excretion. Bile and bacteria may remain in the diverticulum, leading to duodenal papillitis, triggering dyskinesia of the sphincter, and then inducing pancreatitis [27].Timely risk assessment, optimally at the onset of acute pancreatitis, is crucial to maximize treatment efficacy, provide optimal management and identify those at high risk of developing complications such as infected necrosis or organ failure, which might ultimately lead to unfavorable treatment and survival outcomes [28–31]. Previously, multiple scoring systems, such as the Harmless acute pancreatitis Score (HAPS) [32], Ranson criteria [33], Balthazar score [34], bedside index for severity of acute pancreatitis (BISAP) [35], PANC3 score [36] and others, were proposed for the early phase of acute pancreatitis to predict the disease course and further outcomes. However, their accuracy tended to vary based on the population selected, institutions performed, etc. [37, 38]. Additionally, there is a lack of scoring model systems, a user-friendly scoring system using clinically readily available data, for estimating the risk of PEP. In this regard, a recent study by Fu et al. were the first to build and validate a risk prediction model, especially for PEP after biliary stent placement due to malignant biliary obstruction (MBO), which demonstrated an AUC of 0.810 and 0.781 in their developmental and validation cohort [39]. However, their study focused only on MBO cases, thereby affecting the generalizability of their results for other cases undergoing ERCP. Zheng et al. recently proposed a scoring system for predicting the risk of PEP, but their study was limited by a lack of cases, external validation, and a limited number of variables in their scoring model [40]. Thus, considering the lack of scoring systems and limitations in the current literature, we undertook this present study to determine the potential use of a scoring model to assist clinicians in identifying high-risk patients and help implement preventive measures more promptly. Our proposed prediction model for PEP demonstrated good agreement with the actual observed values after calibration. The ROC curve (AUC = 0.787) also indicated the promising predictive value of our proposed nomogram.
Despite the interesting findings described, our study had several limitations. First, this was a single-center retrospective study with a relatively small sample size, limiting the generalizability of our proposed nomogram, thus, urging the need for further verification in large-scale, multicenter prospective studies. Second, due to the limited data available, we could not validate the nomogram and hope that in the future, these findings could be validated in populations outside of China. Third, due to the issues in data retrieval, some data, such as operator experience or papilla type, were not investigated.
Conclusion
In summary, age less than 60 years, sex being female, history of pancreatitis, history of choledocholithiasis, pancreatic sphincterotomy, pancreatic duct imaging, multiple cannulation attempts, difficult cannulation, and juxtapapillary duodenal diverticula are independent risk factors for PEP. Therefore, clinicians need to evaluate these indicators before ERCP to effectively prevent PEP, which could help reduce the incidence of PEP, patients’ mortality and pain and medical resources. Thus, our proposed model could be used as a reference for estimating the risk of these patients to develop PEP, based on which a personalized treatment approach could be used to improve the treatment and survival outcomes of these sufferers.
Acknowledgements
Not applicable.
Author Contribution
Jin-yuan Chi and Lin-ya Ma designed the study and wrote the manuscript. Jia-cheng Zou and Yue-feng Ma collected and analyzed data. All authors have read and approved the submitted paper.
Funding
This project is supported by the Dalian Deng Feng Program: key medical specialties in construction funded by the People’s Government of Dalian Municipality ([2021] 243)
Data Availability
The datasets used and/or analyzed during the current study can be made available from the corresponding author on reasonable request.
Declarations
Ethics approval and consent to participate
This study was approved by the Ethics Committee of the Affiliated Zhongshan Hospital of Dalian University. Informed consent was obtained from all subjects. All methods were carried out in accordance with relevant guidelines and regulations.
Consent for publication
Not applicable.
Competing interests
The authors have no competing interests to declare.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.McCune WS, Shorb PE, Moscovitz H. Endoscopic cannulation of the ampulla of vater: a preliminary report. Ann Surg. 1968 May;167(5):752–6. 10.1097/00000658-196805000-00013. PMID: 5646296; PMCID: PMC1387128. [DOI] [PMC free article] [PubMed]
- 2.Dickinson PB, Belsito AA. Diagnostic value of Endoscopic Retrograde Cholangiopancreatography in the Jaundiced patient. Ann Intern Med. 1973;78(5):817. doi: 10.7326/0003-4819-78-5-817_2. [DOI] [Google Scholar]
- 3.Johnson KD, Perisetti A, Tharian B, Thandassery R, Jamidar P, Goyal H, Inamdar S. Endoscopic Retrograde Cholangiopancreatography-Related Complications and their management strategies: a “Scoping. Literature Rev Dig Dis Sci. 2020;65(2):361–75. doi: 10.1007/s10620-019-05970-3. [DOI] [PubMed] [Google Scholar]
- 4.Seo DY, Lee SR, Heo JW, No MH, Rhee BD, Ko KS, Kwak HB, Han J. Ursolic acid in health and disease. Korean J Physiol Pharmacol. 2018;22(3):235–48. doi: 10.4196/kjpp.2018.22.3.235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Galeazzi M, Mazzola P, Valcarcel B, Bellelli G, Dinelli M, Pasinetti GM, Annoni G. Endoscopic retrograde cholangiopancreatography in the elderly: results of a retrospective study and a geriatricians’ point of view. BMC Gastroenterol. 2018;18(1):38. doi: 10.1186/s12876-018-0764-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Siiki A, Tamminen A, Tomminen T, Kuusanmaki P. ERCP procedures in a finnish community hospital: a retrospective analysis of 1207 cases. Scand J Surg. 2012;101(1):45–50. doi: 10.1177/145749691210100109. [DOI] [PubMed] [Google Scholar]
- 7.Kapral C, Muhlberger A, Wewalka F, Duller C, Knoflach P, Schreiber F, Working Groups Quality A. Endoscopy of Austrian Society of G, Hepatology. Quality assessment of endoscopic retrograde cholangiopancreatography: results of a running nationwide austrian benchmarking project after 5 years of implementation. Eur J Gastroenterol Hepatol. 2012;24(12):1447–54. doi: 10.1097/MEG.0b013e3283583c6f. [DOI] [PubMed] [Google Scholar]
- 8.Glomsaker T, Hoff G, Kvaloy JT, Soreide K, Aabakken L, Soreide JA, Norwegian Gastronet EG. Patterns and predictive factors of complications after endoscopic retrograde cholangiopancreatography. Br J Surg. 2013;100(3):373–80. doi: 10.1002/bjs.8992. [DOI] [PubMed] [Google Scholar]
- 9.Kochar B, Akshintala VS, Afghani E, Elmunzer BJ, Kim KJ, Lennon AM, Khashab MA, Kalloo AN, Singh VK. Incidence, severity, and mortality of post-ERCP pancreatitis: a systematic review by using randomized, controlled trials. Gastrointest Endosc. 2015;81(1):143–9. doi: 10.1016/j.gie.2014.06.045. [DOI] [PubMed] [Google Scholar]
- 10.Perisetti A, Goyal H, Sharma N. Clinical safety and outcomes of glucagon use during endoscopic retrograde cholangiopancreatography (ERCP) Endosc Int Open. 2022;10(4):E558–E61. doi: 10.1055/a-1747-3242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Committee ASoP, Chandrasekhara V, Khashab MA, Muthusamy VR, Acosta RD, Agrawal D, Bruining DH, Eloubeidi MA, Fanelli RD, Faulx AL, et al. Adverse events associated with ERCP. Gastrointest Endosc. 2017;85(1):32–47. doi: 10.1016/j.gie.2016.06.051. [DOI] [PubMed] [Google Scholar]
- 12.Elmunzer BJ. Reducing the risk of post-endoscopic retrograde cholangiopancreatography pancreatitis. Dig Endosc. 2017;29(7):749–57. doi: 10.1111/den.12908. [DOI] [PubMed] [Google Scholar]
- 13.Muhammedoglu B, Kale IT. Comparison of the safety and efficacy of single-stage endoscopic retrograde cholangiopancreatography plus laparoscopic cholecystectomy versus two-stage ERCP followed by laparoscopic cholecystectomy six-to-eight weeks later: a randomized controlled trial. Int J Surg. 2020;76:37–44. doi: 10.1016/j.ijsu.2020.02.021. [DOI] [PubMed] [Google Scholar]
- 14.Cotton PB, Lehman G, Vennes J, Geenen JE, Russell RC, Meyers WC, Liguory C, Nickl N. Endoscopic sphincterotomy complications and their management: an attempt at consensus. Gastrointest Endosc. 1991;37(3):383–93. doi: 10.1016/S0016-5107(91)70740-2. [DOI] [PubMed] [Google Scholar]
- 15.Testoni PA, Mariani A, Aabakken L, Arvanitakis M, Bories E, Costamagna G, Devière J, Dinis-Ribeiro M, Dumonceau JM, Giovannini M, Gyokeres T, Hafner M, Halttunen J, Hassan C, Lopes L, Papanikolaou IS, Tham TC, Tringali A, van Hooft J, Williams EJ. Papillary cannulation and sphincterotomy techniques at ERCP: european Society of Gastrointestinal Endoscopy (ESGE) Clinical Guideline. Endoscopy. 2016;48(7):657–83. doi: 10.1055/s-0042-108641. [DOI] [PubMed] [Google Scholar]
- 16.Freeman ML, Nelson DB, Sherman S, Haber GB, Herman ME, Dorsher PJ, Moore JP, Fennerty MB, Ryan ME, Shaw MJ, et al. Complications of endoscopic biliary sphincterotomy. N Engl J Med. 1996;335(13):909–18. doi: 10.1056/NEJM199609263351301. [DOI] [PubMed] [Google Scholar]
- 17.Zhu Y, Jia P, Wang ML. Effects of predictive nursing on the postoperative complications after endoscopic retrograde cholangio-pancreatography. Chin J Mod Nurs. 2018;24(26):4. [Google Scholar]
- 18.Chen XF, Xu YF, Zhou ZH, Xiang ZG, Tang XL, Yang SL, Zheng Y. An analysis for the risk factors of postoperative complications in patients with liver cirrhosis after endoscopic retrograde cholangiopancreatography. Chin Hepatol. 2021;26(3):259–6165. [Google Scholar]
- 19.Lin Y, Liu X, Cao DQ, Tang JH, Wen JJ, Li TF, Liu PF, Xia TS. Analysis of risk factors and prevention strategies of post-ERCP pancreatitis. Eur Rev Med Pharmacol Sci. 2017;21(22):5185–90. doi: 10.26355/eurrev_201711_13838. [DOI] [PubMed] [Google Scholar]
- 20.Christensen M, Matzen P, Schulze S, Rosenberg J. Complications of ERCP: a prospective study. Gastrointest Endosc. 2004;60(5):721–31. doi: 10.1016/S0016-5107(04)02169-8. [DOI] [PubMed] [Google Scholar]
- 21.Freeman ML. Post-ERCP pancreatitis: patient and technique-related risk factors. JOP. 2002;3(6):169–76. [PubMed] [Google Scholar]
- 22.Buxbaum J, Yan A, Yeh K, Lane C, Nguyen N, Laine L. Aggressive hydration with lactated Ringer’s solution reduces pancreatitis after endoscopic retrograde cholangiopancreatography. Clin Gastroenterol Hepatol. 2014;12(2):303–7e1. doi: 10.1016/j.cgh.2013.07.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kubiliun NM, Adams MA, Akshintala VS, Conte ML, Cote GA, Cotton PB, Dumonceau JM, Elta GH, Fogel EL, Freeman ML, et al. Evaluation of Pharmacologic Prevention of Pancreatitis after Endoscopic Retrograde Cholangiopancreatography: a systematic review. Clin Gastroenterol Hepatol. 2015;13(7):1231–9. doi: 10.1016/j.cgh.2014.11.038. [DOI] [PubMed] [Google Scholar]
- 24.Manoharan D, Srivastava DN, Gupta AK, Madhusudhan KS. Complications of endoscopic retrograde cholangiopancreatography: an imaging review. Abdom Radiol (NY) 2019;44(6):2205–16. doi: 10.1007/s00261-019-01953-0. [DOI] [PubMed] [Google Scholar]
- 25.Davee T, Garcia JA, Baron TH. Precut sphincterotomy for selective biliary duct cannulation during endoscopic retrograde cholangiopancreatography. Ann Gastroenterol. 2012;25(4):291–302. [PMC free article] [PubMed] [Google Scholar]
- 26.Chen M, Wang L, Wang Y, Wei W, Yao YL, Ling TS, Shen YH, Zou XP. Risk factor analysis of post-ERCP cholangitis: a single-center experience. Hepatobiliary Pancreat Dis Int. 2018;17(1):55–8. doi: 10.1016/j.hbpd.2018.01.002. [DOI] [PubMed] [Google Scholar]
- 27.Yildirgan MI, Basoglu M, Yilmaz I, Atamanalp SS, Balik AA, Aydinli B, Ozturk G. Periampullary diverticula causing pancreaticobiliary disease. Dig Dis Sci. 2004;49(11–12):1943–5. doi: 10.1007/s10620-004-9597-9. [DOI] [PubMed] [Google Scholar]
- 28.Mukai S, Itoi T, Tsuchiya T, Ishii K, Tanaka R, Tonozuka R, Sofuni A. Urgent and emergency endoscopic retrograde cholangiopancreatography for gallstone-induced acute cholangitis and pancreatitis. Dig Endosc. 2022. Epub ahead of print. [DOI] [PubMed]
- 29.Weissman S, Ahmed M, Baniqued MR, Ehrlich D, Tabibian JH. Best practices for prevention of post-endoscopic retrograde cholangiopancreatography pancreatitis. World J Gastrointest Endosc. 2021;13(6):161–9. doi: 10.4253/wjge.v13.i6.161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Bhatt H. Post-endoscopic Retrograde Cholangiopancreatography Pancreatitis: an updated review of current preventive strategies. Clin Exp Gastroenterol. 2021;14:27–32. doi: 10.2147/CEG.S276361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Mofidi R, Patil PV, Suttie SA, Parks RW. Risk assessment in acute pancreatitis. Br J Surg. 2009;96(2):137–50. doi: 10.1002/bjs.6431. [DOI] [PubMed] [Google Scholar]
- 32.Lankisch PG, Weber-Dany B, Hebel K, Maisonneuve P, Lowenfels AB. The harmless acute pancreatitis score: a clinical algorithm for rapid initial stratification of nonsevere disease. Clin Gastroenterol Hepatol. 2009;7(6):702–5. doi: 10.1016/j.cgh.2009.02.020. [DOI] [PubMed] [Google Scholar]
- 33.Höckerstedt K. The Ranson criteria for acute pancreatitis. Acta Chir Scand. 1986;152:717. [PubMed] [Google Scholar]
- 34.Chatzicostas C, Roussomoustakaki M, Vardas E, Romanos J, Kouroumalis EA. Balthazar computed tomography severity index is superior to Ranson criteria and APACHE II and III scoring systems in predicting acute pancreatitis outcome. J Clin Gastroenterol. 2003;36(3):253–60. doi: 10.1097/00004836-200303000-00013. [DOI] [PubMed] [Google Scholar]
- 35.Hagjer S, Kumar N. Evaluation of the BISAP scoring system in prognostication of acute pancreatitis - A prospective observational study. Int J Surg. 2018;54(Pt A):76–81. doi: 10.1016/j.ijsu.2018.04.026. [DOI] [PubMed] [Google Scholar]
- 36.Rathnakar SK, Vishnu VH, Muniyappa S, Prasath A. Accuracy and predictability of PANC-3 Scoring System over APACHE II in Acute Pancreatitis: a prospective study. J Clin Diagn Res. 2017;11(2):PC10–3. doi: 10.7860/JCDR/2017/23168.9375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Macedo Silva V, Freitas M, Sousa Magalhães R, Cúrdia Gonçalves T, Boal Carvalho P, Rosa B, Cotter J. Prognostic scores in post-endoscopic retrograde cholangiopancreatography acute pancreatitis: should we use all, some, or none? Eur J Gastroenterol Hepatol. 2022;34(9):905–11. doi: 10.1097/MEG.0000000000002402. [DOI] [PubMed] [Google Scholar]
- 38.Vasudevan S, Goswami P, Sonika U, Thakur B, Sreenivas V, Saraya A. Comparison of various Scoring Systems and biochemical markers in Predicting the Outcome in Acute Pancreatitis. Pancreas. 2018;47(1):65–71. doi: 10.1097/MPA.0000000000000957. [DOI] [PubMed] [Google Scholar]
- 39.Fu Z, Song J, Pi Y, Sun X, Liu M, Xiao Z, Chen J. A Risk Prediction Model for Post-endoscopic Retrograde Cholangiopancreatography Pancreatitis After Stent Insertion for Malignant Biliary Obstruction: Development and Validation. Dig Dis Sci. 2022. Epub ahead of print. [DOI] [PubMed]
- 40.Zheng R, Chen M, Wang X, Li B, He T, Wang L, Xu G, Yao Y, Cao J, Shen Y, Wang Y, Zhu H, Zhang B, Wu H, Zou X, He G. Development and validation of a risk prediction model and scoring system for post-endoscopic retrograde cholangiopancreatography pancreatitis. Ann Transl Med. 2020;8(20):1299. doi: 10.21037/atm-20-5769. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study can be made available from the corresponding author on reasonable request.