Abstract
Background
To report the clinical manifestations of non-arteritic anterior ischemic optic neuropathy (NAION) cases after coronavirus disease 2019 (COVID-19) vaccination in Korea.
Methods
This multicenter retrospective study included patients diagnosed with NAION within 42 days of COVID-19 vaccination. We collected data on vaccinations, demographic features, presence of vascular risk factors, ocular findings, and visual outcomes of patients with NAION.
Results
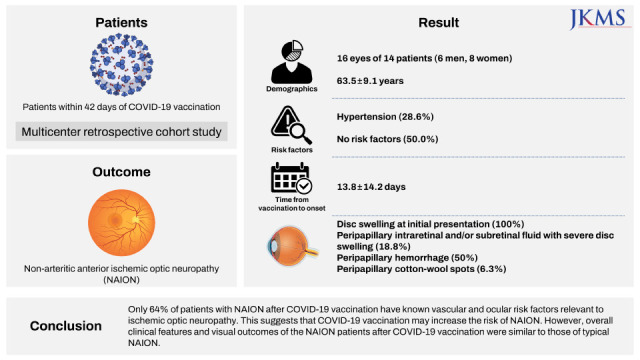
The study included 16 eyes of 14 patients (6 men, 8 women) with a mean age of 63.5 ± 9.1 (range, 43–77) years. The most common underlying disease was hypertension, accounting for 28.6% of patients with NAION. Seven patients (50.0%) had no vascular risk factors for NAION. The mean time from vaccination to onset was 13.8 ± 14.2 (range, 1–41) days. All 16 eyes had disc swelling at initial presentation, and 3 of them (18.8%) had peripapillary intraretinal and/or subretinal fluid with severe disc swelling. Peripapillary hemorrhage was found in 50% of the patients, and one (6.3%) patient had peripapillary cotton-wool spots. In eight fellow eyes for which we were able to review the fundus photographs, the horizontal cup/disc ratio was less than 0.25 in four eyes (50.0%). The mean visual acuity was logMAR 0.6 ± 0.7 at the initial presentation and logMAR 0.7 ± 0.8 at the final visit.
Conclusion
Only 64% of patients with NAION after COVID-19 vaccination have known vascular and ocular risk factors relevant to ischemic optic neuropathy. This suggests that COVID-19 vaccination may increase the risk of NAION. However, overall clinical features and visual outcomes of the NAION patients after COVID-19 vaccination were similar to those of typical NAION.
Keywords: Coronavirus Disease 2019 (COVID-19), Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2), Vaccination, Non-Arteritic Anterior Ischemic Optic Neuropathy
Graphical Abstract
INTRODUCTION
Coronavirus disease 2019 (COVID-19), caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), has become one of the major global health problems to date.1,2 After its efficacy and safety were reported in clinical trials, the Food and Drug Administration approved the first vaccine against SARS-CoV-2 in December 2020. Since then, several vaccines have received emergency authorizations for use in many countries worldwide.3,4
In the midst of the global pandemic, the safety and efficacy of COVID-19 vaccines are of special interest.5,6,7 Several ocular complications following COVID-19 vaccination have also been reported.8,9,10 Side effects of COVID-19 vaccination include ocular inflammation, vascular lesions, and cranial nerve palsy, including Bell’s palsy. However, the mechanism of events after COVID-19 vaccination remains unclear.
In Korea, where two recombinant mRNA vaccines (BNT162b2, Pfizer-BioNTech and mRNA-1273, Moderna) and two adenovirus vector-based vaccines (ChAdOx1 nCoV-19, Oxford-AstraZeneca and Ad26.COV2 Janssen, Johnson & Johnson) have been mainly distributed, a few cases of ocular disease after COVID-19 vaccination have been reported.11,12,13,14 However, with the widespread distribution of vaccines, the need for a nationwide survey of ocular disease after COVID-19 vaccination has been raised.
Therefore, the Korean Neuro-Ophthalmology Society (KNOS) has performed a nationwide survey for neuro-ophthalmologic diseases following COVID-19 vaccinations. This study aimed to report the clinical features of non-arteritic anterior ischemic optic neuropathy (NAION) after COVID-19 vaccination.
METHODS
We retrospectively reviewed the medical records of all patients diagnosed with NAION after COVID-19 vaccination between February 2021 and November 2021. Only patients who developed symptoms within 42 days of COVID-19 vaccination were included in this study.15,16 The diagnosis of NAION was based on the standard clinical criteria including: 1) sudden onset of visual symptoms; 2) relative afferent pupillary defect; 3) swollen optic disc; and 4) any visual field (VF) defect.17 Patients likely to have optic neuritis were excluded by the following criteria: pain on eye movement, a history of any central nervous system inflammatory demyelinating disease, or seropositivity for aquaporin-4 or myelin oligodendrocyte glycoprotein antibody. Patients likely to have arteritis or inflammatory disease were also excluded by the following criteria: 1) any symptoms or signs of temporal arteritis; 2) an erythrocyte sedimentation rate higher than 50 mm/hr; 3) a history of vasculitis or other autoimmune diseases; and 4) any evidence of ocular inflammation such as iritis, vitritis, or vitreous hemorrhage. We finally obtained reports of 14 patients (16 eyes) from 9 centers, 1 of which had already been published in the literature.12
We obtained the following data based on COVID-19 vaccinations: the type of vaccine, the number of vaccine doses, and the time from vaccination to onset of symptoms. In addition, clinical data obtained with case report forms included basic demographic information (age, sex, and laterality of the involved eyes) and past medical history, including medication, initial symptoms, and treatment methods.
We also collected ocular imaging data at the initial presentation, including fundus photographs and optical coherence tomography images, as well as examination data for visual functions, such as visual acuity (VA) and VF test. Finally, we reviewed the VA at the final visit for analysis of visual outcomes. The VA was converted to logMAR for the analysis. Poor vision, classified as count finger, hand motion, light perception, and no light perception, was converted to logMAR 1.7, 2.0, 2.3, and 3.0, respectively.18 The VF test was conducted using the Humphrey perimetry Swedish Interactive Threshold Algorithm 24-2 or 30-2 (Carl Zeiss Meditec, Inc., Dublin, CA, USA). Mean deviation (MD) measurements were used in the final analysis.
Continuous data are summarized and presented as mean ± standard deviation and range, while categorical data are presented as proportions and percentages. The Wilcoxon signed-rank test was performed to compare the initial and final visual functions. All statistical analyses were performed using SPSS version 23.0 (SPSS Inc., Chicago, IL, USA). Statistical significance was established at P < 0.05 for all calculations.
Ethics statement
This multicenter retrospective study was conducted by the KNOS. The study protocol was reviewed and approved by the Institutional Review Board of each author’s institution (Institutional Review Board of Seoul National University Hospital (No. H-2112-036-1279) and Ethics Committee at Konkuk University Medical Center (No. 2021-12-029)). The study was conducted in accordance with the tenets of the Declaration of Helsinki. The requirement for informed consent was waived because of the retrospective study design.
RESULTS
Baseline demographics of the study population
This study included 14 patients with NAION, 8 (57.1%) of whom were women. The mean age of the patients was 63.5 ± 9.1 (range, 43–77) years. The mean time from vaccination to symptom onset was 13.8 ± 14.2 (range, 1–41) days. Bilateral NAION was present in 2 cases and occurred simultaneously on the same day. The most common underlying disease was hypertension, accounting for 28.6% of patients with NAION. Seven patients (50.0%) had no vascular risk factors for NAION. The demographic data of the study population are summarized in Table 1.
Table 1. Baseline demographic data of the study population.
| Demographic characteristics | Non-arteritic anterior ischemic optic neuropathy (n = 14) | |
|---|---|---|
| Age, yr | 63.5 ± 9.1 (43–77) | |
| Sex, male:female | 6:8 | |
| Time from vaccination to onset of symptoms, days | 13.8 ± 14.2 (1–41) | |
| Past medical history, No. (%) | ||
| Diabetes | 1 (7.1) | |
| Hypertension | 4 (28.6) | |
| Hyperlipidemia | 2 (14.3) | |
| Cardiovascular disease | 2 (14.3) | |
| Cerebrovascular disease | 1 (7.1) | |
Types of vaccines
In six patients, NAION occurred after vaccination with the recombinant mRNA BNT162b2 vaccine (Pfizer-BioNTech), and the other six patients were administered the adenovirus vector ChAdOx1 nCoV-19 vaccine (Oxford-AstraZeneca). The types of vaccines used in the remaining two cases were unknown.
The clinical characteristics according to the type of vaccines are presented in Table 2. On average, NAION occurred 2.8 ± 1.3 (range, 1–4) days after BNT162b2 vaccination and 22.5 ± 14.2 (range, 4–41) days after ChAdOx1 nCoV-19 vaccination. Five of six patients who received BNT162b2 vaccination experienced NAION after the first dose, while three patients experienced NAION after the first dose of ChAdOX1 nCoV-19 vaccination.
Table 2. Clinical characteristics of patients with non-arteritic anterior ischemic optic neuropathy according to the type of coronavirus disease 2019 vaccines.
| Clinical characteristics | Recombinant mRNA BNT162b2 vaccine (Pfizer-BioNTech) (n = 6) | Adenovirus vector ChAdOx1 nCoV-19 vaccine (Oxford-AstraZeneca) (n = 6) | |
|---|---|---|---|
| Age, yr | 59.7 ± 11.8 (43–77) | 64.4 ± 5.4 (54–69) | |
| Sex, male:female | 3:3 | 2:4 | |
| Time from vaccination to onset of symptoms, daysa | 2.8 ± 1.3 (1–4) | 22.5 ± 14.2 (4–41) | |
| No. of vaccine doses, 1st dose:2nd dose:3rd dose | 5:1:0 | 3:3:0 | |
| Past medical history, No. (%) | |||
| Diabetes | 1 (16.7) | 0 (0.0) | |
| Hypertension | 3 (50.0) | 1 (16.7) | |
| Hyperlipidemia | 1 (7.1) | 0 (0.0) | |
| Cardiovascular disease | 0 (0.0) | 0 (0.0) | |
| Cerebrovascular disease | 1 (16.7) | 0 (0.0) | |
| Visual acuity at the initial presentation, logMAR | 0.6 ± 0.6 (0.2–1.7) | 0.9 ± 0.8 (0.0–1.7) | |
| Visual acuity at the final presentation, logMAR | 0.5 ± 0.4 (0.1–1.0) | 1.2 ± 1.0 (0.0–2.3) | |
aData of two patients were missing.
Initial presentation of NAION
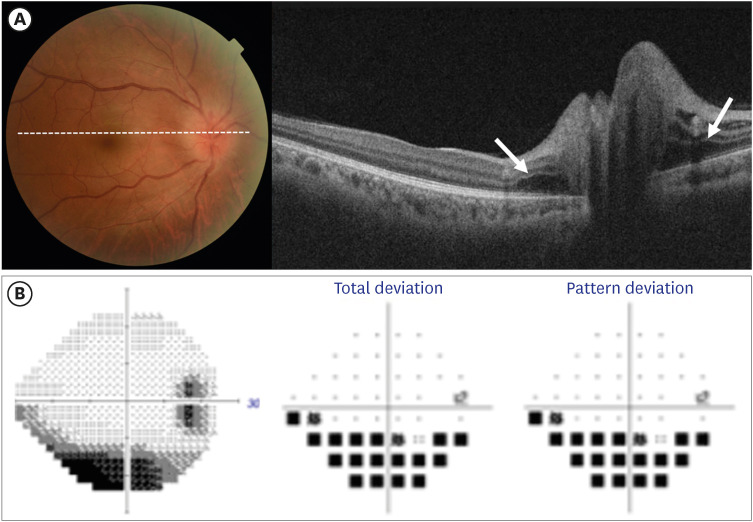
All patients complained of visual symptoms such as decreased vision or VF defects, and only one patient reported ocular pain. Of the 14 patients, 2 (14.3%) showed bilateral involvement of the disease; therefore, a total of 16 eyes with NAION were included in this study. All 16 eyes had disc swelling at the initial presentation, and 3 (18.8%) of them had peripapillary intraretinal and/or subretinal fluid with severe disc swelling (Fig. 1). Peripapillary hemorrhage was found in 50% of the patients, and 1 (6.3%) patient had peripapillary cotton-wool spots. We also obtained fundus photographs of the fellow eye of patients with unilateral NAION. In eight fellow eyes for which we were able to review the fundus photographs, the horizontal cup/disc (C/D) ratio was less than 0.25 in four eyes (50.0%, group 1), while the fellow eyes of the other four cases (group 2) had horizontal C/D ratios greater than 0.25. Age, sex, and vaccination type did not differ between the two groups. However, none of the patients in group 2 had vascular risk factors, which were found in 50% of the cases in group 1. In group 2, NAION occurred in all cases after the second dose of vaccination, whereas in group 1, NAION occurred after the first dose.
Fig. 1. Images of the fundus and visual field maps. (A) Fundus photograph and corresponding horizontal cross-sectional optical coherence tomography scan in a patient with non-arteritic anterior ischemic optic neuropathy following coronavirus disease 2019 vaccination. Severe optic disc edema and peripapillary sub-retinal fluid (white arrow) are seen. (B) Visual field test results (grayscale map, total deviation probability map, and pattern deviation probability map) of the patients show the inferior altitudinal defect.
Visual outcomes
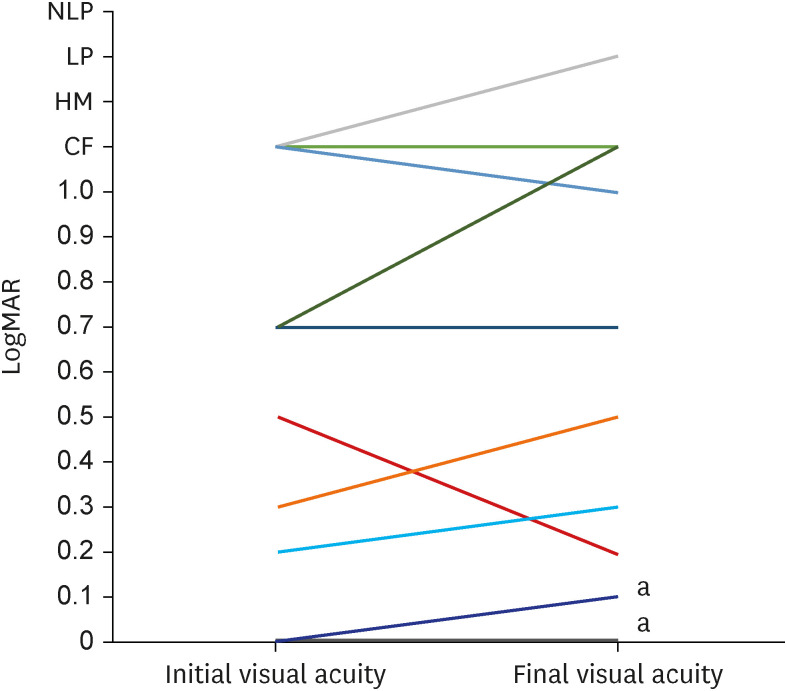
Of the 16 eyes, initial and final VA data were obtained from 14 and 13 eyes, respectively. The mean initial VA was logMAR 0.6 ± 0.7 (range, 1.7–0.0). After a mean follow-up period of 5.9 ± 2.7 months, the mean final VA was logMAR 0.7 ± 0.8 (range, 2.3–0.0), and five cases (38.5%) had a final VA of logMAR 1.0. Of the 12 eyes that had data of both initial and final VA, 2 (16.7%) had visual improvement ≥ logMAR 0.3, and the other two (16.7%) showed progression of visual impairment ≥ logMAR 0.3. No significant difference was observed between the initial and final VA values (P = 0.453) (Fig. 2).
Fig. 2. Line chart showing the changes of visual acuity.
NLP = no light perception, LP = light perception, HM = hand motion, CF = count finger.
aTwo eyes showed the same changes.
The VF test was performed at the initial and final visits in 13 and 12 eyes, respectively. At the initial presentation, seven eyes (53.8%) showed an altitudinal VF defect. Total defects were detected in four eyes (30.8%) and non-specific focal defects in two eyes (15.4%). To evaluate VF defects quantitatively, we collected the MD values of the VF test using Humphrey perimetry. The initial MD was −12.3 ± 10.3 dB (range, −32.79–1.05 dB), and the final MD was −15.1 ± 10.7 dB (range, −31.23–0.74 dB). There was no significant difference between the initial and final MD values (P = 0.375).
DISCUSSION
There have been few reports on post-vaccination NAION. Only three cases following influenza vaccination have been reported, and one of them was taking a phosphodiesterase-5 inhibitor, which is one of the risk factors for NAION.19,20 In Korea, free influenza vaccination is provided to people over the age of 65 under the National Immunization Program, but there have been no NAION case reports after influenza vaccination. However, NAION after receiving the COVID-19 vaccine has been reported more frequently than after other vaccines. The number of vaccine doses administered during the same period in Korea was 49,736,449 for BNT162b2 and 20,309,089 for ChAdOx1 nCoV-19. Accordingly, the incidence of NAION following COVID-19 vaccination, calculated by dividing the number of events by the number of vaccine doses, was 0.12 cases per million doses after BNT162b2 and 0.30 cases per million doses after ChAdOx1 nCoV-19, which was similar to those reported in other countries in population-based pharmacovigilance surveillance systems.9 This may be due in part to the recent high interest in COVID-19 pandemic and its vaccines. Despite this, COVID-19 vaccination may have a significant association with the risk of NAION.
Transient nonperfusion or hypoperfusion of the optic nerve head (ONH) is by far the commonest etiology of NAION.21 Nachbor et al.22 suggested immune-mediated microangiopathy by the vaccine-induced inflammatory cascade in NAION associated with COVID-19 vaccination. Triggering the inflammatory cascade itself might adversely affect vascular function.23,24 Furthermore, transient endothelial dysfunction induced by the mRNA vaccine against COVID-19 was reported.25 This can partially explain the perfusion insufficiency in NAION following COVID-19 vaccination.
Although embolic lesions of the arteries/arterioles feeding the ONH are only an occasional cause of NAION, they should be considered as one of the mechanisms of NAION following COVID-19 vaccination. Recently, vaccine-induced immune thrombotic thrombocytopenia (VITT) has been reported following the administration of COVID-19 adenovirus vector-based vaccines and recombinant mRNA vaccines.26 VITT may contribute to the development of NAION by inducing arterial thrombosis or thromboembolism in the posterior ciliary artery, particularly in patients with NAION combined with other ocular ischemic lesions such as retinal arterial or venous occlusion and cotton-wool spots.27
In our case series, NAION occurred within four days of the BNT162b2 vaccination, whereas it occurred on an average of three weeks after the ChAdOx1 nCoV-19 vaccination. The previous report on optic neuropathy after COVID-19 vaccination presented cases with various periods between the vaccination and symptom onset.28 Due to the very low incidence of NAION after COVID-19 vaccination, it is difficult to statistically compare and evaluate the significant difference in the clinical course of post-vaccination NAION. In addition, the clinical characteristics including the risk of vascular diseases may be different according to the type of vaccine as the patients over 75 years of age had been given the BNT162b2 vaccination for the first and second shots. However, differences in the clinical course according to the type of vaccination should be considered in the future.
The appearance of the affected optic disc in our patients was not different from that previously reported. Peripapillary hemorrhage was frequently noticed, and peripapillary intra- and sub-retinal fluid was also found in approximately 20% of patients, which was comparable to previous reports.29,30 Meanwhile, a crowded optic disc, which is a well-known risk factor for NAION, was found in half of the fellow unaffected eyes. Although statistical comparison with previous studies is difficult due to the small number of subjects in the current study, the proportion of crowded optic discs is considered to be lower than that in previous studies.31,32 Interestingly, all patients without a crowded optic disc in the fellow eye had experienced NAION after the second dose of the COVID-19 vaccine. Compared with the first dose, the second dose of the vaccine caused a prominent increase in inflammatory markers, resulting in transient deterioration of endothelial function.25 This implies that post-vaccination inflammation and consequent endothelial dysfunction may have contributed to the development of NAION after COVID-19 vaccination, although there was no clinical evidence of vasculitis in those patients. Taken together, our cases suggest that COVID-19 vaccination may increase the incidence of NAION in patients with no risk factors for ischemic optic neuropathy.
The visual prognosis was similar to the clinical course of NAION reported in a previous randomized control study.33 In the current survey, there were two cases (16.7%) with a visual improvement of logMAR 0.3 or higher and five cases (38.5%) with a final VA of logMAR 1.0 or worse. Therefore, the clinical course and visual outcomes of NAION after COVID-19 vaccination may be in accordance with those of general NAION.
This study has the inherent limitations of the case series: retrospective design, impossibility to establish a cause-effect relationship, and lack of ability to generalize. First, owing to the retrospective nature of the study, we were unable to collect clinical data from all subjects. We could not fully obtain the data on sleep apnea syndrome, which is well known to be associated with NAION.34 In addition, we did not have laboratory data such as complete blood count, coagulation test, D-dimer, and fibrinogen, which can aid in the detection of thrombophilia or VITT. Second, we included all patients with NAION who developed symptoms within 42 days of the COVID-19 vaccination. We confirmed a temporal relationship between vaccination and NAION, not causality. However, it should be noted that the vaccine adverse event reporting system is based on the time relationship between vaccination and adverse effects. Last, a nationwide survey should be performed to confirm the incidence and evaluate the risk of NAION after COVID-19 in total population. However, the current study is the largest multicenter survey on NAION after the COVID-19 vaccine in Korea, and has its strength in that the diagnosis of NAION in all patients was confirmed by a neuro-ophthalmologist.
In conclusion, although there is no doubt that vaccination against SARS-CoV-2 is essential to end the global COVID-19 pandemic, adverse events should also be monitored, considering that vaccines are administered to a large number of healthy people. Clinicians should be aware of the possibility of NAION occurrence after COVID-19 vaccination, even in cases without crowded optic discs and vascular risk factors. Considering the postulated mechanisms, clinicians may consider the evaluation for hypercoagulopathy or systemic inflammation. The majority of the clinical features and visual outcomes were similar to those of typical NAION. Further studies are required to determine the causality between NAION and COVID-19 vaccination.
Footnotes
Disclosure: The authors have no potential conflicts of interest to disclose.
- Conceptualization: Jung JH, Shin HJ, Choi DG.
- Data curation: Moon Y.
- Formal analysis: Moon Y.
- Investigation: Moon Y, Jung JH, Shin HJ, Choi DG, Park KA, Jeon H, Lee BJ, Kim SJ, Oh SY, Ahn H, Chung SA, Kim US, Lee HJ, Lee JY, Choi YJ.
- Validation: Moon Y, Jung JH, Shin HJ, Choi DG, Park KA, Jeon H, Lee BJ, Kim SJ, Oh SY, Ahn H, Chung SA, Kim US, Lee HJ, Lee JY, Choi YJ.
- Visualization: Moon Y, Jung JH, Shin HJ.
- Writing - original draft: Moon Y.
- Writing - review & editing: Moon Y, Jung JH, Shin HJ, Choi DG, Park KA, Jeon H, Lee BJ, Kim SJ, Oh SY, Ahn H, Chung SA, Kim US, Lee HJ, Lee JY, Choi YJ.
References
- 1.Wiersinga WJ, Rhodes A, Cheng AC, Peacock SJ, Prescott HC. Pathophysiology, transmission, diagnosis, and treatment of coronavirus disease 2019 (COVID-19): a review. JAMA. 2020;324(8):782–793. doi: 10.1001/jama.2020.12839. [DOI] [PubMed] [Google Scholar]
- 2.Woolf SH, Masters RK, Aron LY. Effect of the COVID-19 pandemic in 2020 on life expectancy across populations in the USA and other high income countries: simulations of provisional mortality data. BMJ. 2021;373:n1343. doi: 10.1136/bmj.n1343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Alderson J, Batchelor V, O'Hanlon M, Cifuentes L, Richter FC, Kopycinski J, et al. Overview of approved and upcoming vaccines for SARS-CoV-2: a living review. Oxf Open Immunol. 2021;22(1):iqab010. doi: 10.1093/oxfimm/iqab010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cai C, Peng Y, Shen E, Huang Q, Chen Y, Liu P, et al. A comprehensive analysis of the efficacy and safety of COVID-19 vaccines. Mol Ther. 2021;29(9):2794–2805. doi: 10.1016/j.ymthe.2021.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Klein NP, Lewis N, Goddard K, Fireman B, Zerbo O, Hanson KE, et al. Surveillance for adverse events after COVID-19 mRNA vaccination. JAMA. 2021;326(14):1390–1399. doi: 10.1001/jama.2021.15072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Li X, Ostropolets A, Makadia R, Shaoibi A, Rao G, Sena AG, et al. Characterising the background incidence rates of adverse events of special interest for COVID-19 vaccines in eight countries: multinational network cohort study. BMJ. 2021;373:n1435. doi: 10.1136/bmj.n1435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gee J, Marquez P, Su J, Calvert GM, Liu R, Myers T, et al. First month of COVID-19 vaccine safety monitoring - United States, December 14, 2020-January 13, 2021. MMWR Morb Mortal Wkly Rep. 2021;70(8):283–288. doi: 10.15585/mmwr.mm7008e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Haseeb AA, Solyman O, Abushanab MM, Abo Obaia AS, Elhusseiny AM. Ocular complications following vaccination for COVID-19: a one-year retrospective. Vaccines (Basel) 2022;10(2):342. doi: 10.3390/vaccines10020342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wang MT, Niederer RL, McGhee CN, Danesh-Meyer HV. COVID-19 vaccination and the eye. Am J Ophthalmol. 2022;240:79–98. doi: 10.1016/j.ajo.2022.02.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ng XL, Betzler BK, Ng S, Chee SP, Rajamani L, Singhal A, et al. The eye of the storm: COVID-19 vaccination and the eye. Ophthalmol Ther. 2022;11(1):81–100. doi: 10.1007/s40123-021-00415-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Park HS, Byun Y, Byeon SH, Kim SS, Kim YJ, Lee CS. Retinal hemorrhage after SARS-CoV-2 vaccination. J Clin Med. 2021;10(23):5705. doi: 10.3390/jcm10235705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Chung SA, Yeo S, Sohn SY. Non-arteritic anterior ischemic optic neuropathy following COVID-19 vaccination. Korean J Ophthalmol. 2022;36(2):168–170. doi: 10.3341/kjo.2021.0128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Song MY, Koh KM, Hwang KY, Kwon YA, Kim KY. Relapsed disciform stromal herpetic keratitis following mRNA COVID-19 vaccination: a case report. Korean J Ophthalmol. 2022;36(1):80–82. doi: 10.3341/kjo.2021.0150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kim SY, Kang MS, Kwon HJ. Bilateral panuveitis mimicking Vogt-Koyanagi-Harada disease following the first dose of ChAdOx1 nCoV-19 vaccine. Ocul Immunol Inflamm. 2022;30(5):1218–1221. doi: 10.1080/09273948.2022.2026410. [DOI] [PubMed] [Google Scholar]
- 15.Frontera JA, Tamborska AA, Doheim MF, Garcia-Azorin D, Gezegen H, Guekht A, et al. Neurological events reported after COVID-19 vaccines: an analysis of VAERS. Ann Neurol. 2022;91(6):756–771. doi: 10.1002/ana.26339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Barda N, Dagan N, Ben-Shlomo Y, Kepten E, Waxman J, Ohana R, et al. Safety of the BNT162b2 mRNA COVID-19 vaccine in a nationwide setting. N Engl J Med. 2021;385(12):1078–1090. doi: 10.1056/NEJMoa2110475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.The IONDT Research Group. The ischemic optic neuropathy decompression trial (IONDT): design and methods. Control Clin Trials. 1998;19(3):276–296. doi: 10.1016/s0197-2456(98)00003-8. [DOI] [PubMed] [Google Scholar]
- 18.Lee JW, Lai JS, Yick DW, Tse RK. Retrospective case series on the long-term visual and intraocular pressure outcomes of phacomorphic glaucoma. Eye (Lond) 2010;24(11):1675–1680. doi: 10.1038/eye.2010.108. [DOI] [PubMed] [Google Scholar]
- 19.Kawasaki A, Purvin VA, Tang R. Bilateral anterior ischemic optic neuropathy following influenza vaccination. J Neuroophthalmol. 1998;18(1):56–59. [PubMed] [Google Scholar]
- 20.Manasseh G, Donovan D, Shao EH, Taylor SR. Bilateral sequential non-arteritic anterior ischaemic optic neuropathy following repeat influenza vaccination. Case Rep Ophthalmol. 2014;5(2):267–269. doi: 10.1159/000366472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hayreh SS. Ischemic optic neuropathy. Prog Retin Eye Res. 2009;28(1):34–62. doi: 10.1016/j.preteyeres.2008.11.002. [DOI] [PubMed] [Google Scholar]
- 22.Nachbor KM, Naravane AV, Adams OE, Abel AS. Nonarteritic anterior ischemic optic neuropathy associated with COVID-19 vaccination. J Neuroophthalmol. 2021 doi: 10.1097/WNO.0000000000001423. [DOI] [PubMed] [Google Scholar]
- 23.Vlachopoulos C, Dima I, Aznaouridis K, Vasiliadou C, Ioakeimidis N, Aggeli C, et al. Acute systemic inflammation increases arterial stiffness and decreases wave reflections in healthy individuals. Circulation. 2005;112(14):2193–2200. doi: 10.1161/CIRCULATIONAHA.105.535435. [DOI] [PubMed] [Google Scholar]
- 24.Jain S, Khera R, Corrales-Medina VF, Townsend RR, Chirinos JA. “Inflammation and arterial stiffness in humans”. Atherosclerosis. 2014;237(2):381–390. doi: 10.1016/j.atherosclerosis.2014.09.011. [DOI] [PubMed] [Google Scholar]
- 25.Terentes-Printzios D, Gardikioti V, Solomou E, Emmanouil E, Gourgouli I, Xydis P, et al. The effect of an mRNA vaccine against COVID-19 on endothelial function and arterial stiffness. Hypertens Res. 2022;45(5):846–855. doi: 10.1038/s41440-022-00876-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Afshar ZM, Barary M, Babazadeh A, Hosseinzadeh R, Alijanpour A, Miri SR, et al. SARS-CoV-2-related and COVID-19 vaccine-induced thromboembolic events: A comparative review. Rev Med Virol. 2022;32(4):e2327. doi: 10.1002/rmv.2327. [DOI] [PubMed] [Google Scholar]
- 27.Girbardt C, Busch C, Al-Sheikh M, Gunzinger JM, Invernizzi A, Xhepa A, et al. Retinal vascular events after mRNA and adenoviral-vectored COVID-19 vaccines-a case series. Vaccines (Basel) 2021;9(11):1349. doi: 10.3390/vaccines9111349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Elnahry AG, Al-Nawaflh MY, Gamal Eldin AA, Solyman O, Sallam AB, Phillips PH, et al. COVID-19 vaccine-associated optic neuropathy: a systematic review of 45 patients. Vaccines (Basel) 2022;10(10):1758. doi: 10.3390/vaccines10101758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Characteristics of patients with nonarteritic anterior ischemic optic neuropathy eligible for the Ischemic Optic Neuropathy Decompression Trial. Arch Ophthalmol. 1996;114(11):1366–1374. doi: 10.1001/archopht.1996.01100140566007. [DOI] [PubMed] [Google Scholar]
- 30.Kupersmith MJ, Sibony PA, Dave S. Nonarteritic anterior ischemic optic neuropathy induced retinal folds and deformations. Invest Ophthalmol Vis Sci. 2017;58(10):4286–4291. doi: 10.1167/iovs.17-22140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Beck RW, Servais GE, Hayreh SS. Anterior ischemic optic neuropathy. IX. Cup-to-disc ratio and its role in pathogenesis. Ophthalmology. 1987;94(11):1503–1508. [PubMed] [Google Scholar]
- 32.Hayreh SS, Zimmerman MB. Nonarteritic anterior ischemic optic neuropathy: refractive error and its relationship to cup/disc ratio. Ophthalmology. 2008;115(12):2275–2281. doi: 10.1016/j.ophtha.2008.08.007. [DOI] [PubMed] [Google Scholar]
- 33.The Ischemic Optic Neuropathy Decompression Trial Research Group. Optic nerve decompression surgery for nonarteritic anterior ischemic optic neuropathy (NAION) is not effective and may be harmful. JAMA. 1995;273(8):625–632. [PubMed] [Google Scholar]
- 34.Mojon DS, Hedges TR, 3rd, Ehrenberg B, Karam EZ, Goldblum D, Abou-Chebl A, et al. Association between sleep apnea syndrome and nonarteritic anterior ischemic optic neuropathy. Arch Ophthalmol. 2002;120(5):601–605. doi: 10.1001/archopht.120.5.601. [DOI] [PubMed] [Google Scholar]