Abstract
Mutagens are chemical molecules that have the ability to damage DNA. Mutagens can enter into our body upon consumption of improperly cooked or processed food products such as high temperature or prolonged cooking duration. Mutagens are found in the food products can be classified into N-nitroso derivatives, polycyclic aromatic hydrocarbons, and heterocyclic aromatic amines. Food products with high fat and protein content are more prone to mutagenic formation. Microorganisms were found to be a potent weapon in the fight against various mutagens through biotransformation. Therefore, searching for the microorganisms which have the ability to transform mutagens and the development of techniques for the identification as well as detection of mutagens in food products is much needed. In the future, methods for the identification and detection of these mutagens as well as the identification of new and more potent microorganisms which can transform mutagens into non-mutagens are much needed.
Keywords: Heterocyclic aromatic amines, Microbial transformation, Mutagens, N-nitroso derivatives, Polycyclic aromatic hydrocarbons, Processed foods
Introduction
The occurrence of mutagens in heat-processed or canned foods is known but its source of origin, disposition, and concentration dependent effects were studied only to a certain level. However, the application of microbes that results in the metamorphosis of the mutagens to non-toxic molecules is yet to be fully explored from molecular to organism level. Since the 1970s, it was evident that the presence of dietary supplements in processed or canned foods could cause cancers (Bjeldanes et al., 1982a; 1982b; Commoner et al., 1978; Diallo et al., 2018; Fiolet et al., 2018; Kim et al., 2019; Nagao et al., 1977; Tikkanen et al., 1993). Mutagens are chemical molecules that can alter DNA arrangement by reacting with purines and pyrimidines as an electrophilic attacker. This can further cause damage to DNA structure through base alteration, frame-shift mutation and finally results in the collapse of DNA repair mechanisms. Mutagens can also cause chromosomal aberration and exchange of sister chromatids and thus occurrence of mutagens in foods can cause carcinogenesis (Miller and Miller, 1986; Rencüzogullari et al., 2001). This carcinogenic effect of mutagens which enters into our body through food products has been a matter of serious concern for the scientific community because the survival of humankind and the entire living species is solely dependent upon the food supply.
The occurrence of mutagens in food products can be natural or due to cooking or processing conditions. Several chemical components are involved in the processing of food products such as preservatives (like nitrites when used in the form of sodium as well as potassium salts) which can give rise to mutagens. Preservatives are well known to decelerate some chemical reactions of food thereby enhancing antimicrobial possessions (Wójciak et al., 2014). But circumstantially the presence of nitrites as food preservatives can result in the formation of mutagens (Herrmann et al., 2015; Herraiz and Galisteo, 2018; Molognoni et al., 2019). This has led to the scientific debate over the use of preservatives in the processing of food products. Therefore, such preservatives should be either used in minimal concentrations or in combination with other preservatives such that they won’t cause harm upon consumption. Thus understanding the proper concentration and combination of preservatives used can not only yield positive outcomes but also known to reduce the load of harmful microorganisms in the food products such as Clostridium botulinum, which is a known harmful pathogen for causing food poisoning (Sofos et al., 1979; Sofos, 1985; Zamora and Zaritzky, 1987). Improper concentrations and combinations could lead to the formation of mutagenic compounds such as Ethylnitrolic Acid (ENA) and 2-Methyl-1,4-Dinitro pyrrole (DNMP) (Kito et al., 1978; Namiki et al., 1980; Pérez-Prior et al., 2009). Several studies have reported the increased risk of various cancers upon the consumption of improperly cooked or processed foods, mainly meat (Bartkiene et al., 2017; Herrmann et al., 2015; IARC, 2015; Molognoni et al., 2019). These mutagens (ENA, DNMP, and others) showed the various degree of reactivity and proved to have mutagenic or carcinogenic effect when tested against TA98 and TA100 strains of Salmonella typhimurium with or without the use of activation by metabolism (Hansen et al., 1981; Motta et al., 2020; Namiki and Kada, 1975; Pérez-Prior et al., 2009, 2010). Several food flavoring agents which can cause mutagenic or carcinogenic effects were in use in the form of beef stock extracts and meat flavors. The production of such flavoring agents is dependent on the meat variety, plant materials and ultimately processing conditions (Liao et al., 2010; Shabbir et al., 2014; Stavric et al., 1997).
Microbes, a microscopic world present amongst us along with the plants have made our life fruitful with their diverse characteristics. Microbes support our existence by providing us with various food products, medicines, vaccines and also safeguard our environment by working as an environment cleanser. Even though microbes are used in several research areas from food products to oil spillage degradation, very few have reported the application of microbes in the transformation of mutagens to non-mutagens in the processed foods. Fermentation, which is a necessary step in the processing of food products that helps to get better food products in terms of safety, enhanced aroma, and also has widespread acceptability. Since its inception in food processing, it has helped consumers to consume spoiled free food products. In meat processing food products it was found that the addition of microorganisms such as lactic acid bacteria aids in controlling the fermentation process. This provided the foundation for the application of microorganisms in the food processing industry to control several processes including mutagens in processed or canned foods (De Dea Lindner, 2017). Fermentation is known to manipulate the mutagenic substances present in the food products; however, the mechanisms involved are yet to be fully explored (Haskard et al., 2000). Many such chemical substances were used in the making of food products since ancient times but their mutagenic effects were not in focus. Several such chemical substances used as food supplements and preservatives were still not proved to show any positive results in the Ames test. But several studies have reported that the various chemicals involved in the processed foods react amongst each other under various conditions and circumstances to give rise to several mutagens or carcinogens (Binstok et al., 1998; Molognoni et al., 2019; Shu et al., 1991).
Few research groups have reported the mechanism involved in the conversion of precursor mutagens into mutagens as well as the conversion of harmful mutagens into harmless compounds by using microorganisms (Binstok et al., 1998; Kito et al., 1978; Namiki et al., 1980). Conversions of these harmful/toxic mutagenic substances present in the food products to a harmless/non-toxic form which will be non-mutagenic with the help of microorganisms are termed as “biotransformation”, which literally means conversion of one form to another form of similar appearance. But another similar term is “bioconversion”, which means conversion of one substance into another through multiple steps (Lilly, 1994). Employing natural or engineered microorganisms for bioconversion of one harmful product into another harmless product needs a complete assessment of metabolic pathways involved in the conversion. In such an approach, a method known as “One strain-Many compound'' was applied to get more than 100 compounds from only 6 strains of microorganisms, which have the potential of bioconversion (Bode et al., 2002). Bioconversion of any substance occurs due to any of the following processes: redox reaction, isomer formation, condensation, and lyase reaction (Parkinson et al., 2013; Smitha et al., 2017). In comparison to the chemical approach, biotransformation is way more effective due to higher surface to volume ratio, increased metabolic rate, and much safer to use (Hegazy et al., 2015). One such study was reported by the application of Lactobacillus bulgaricus LB-UFSC 001 strain for the bioconversion of mutagens such as ENA and DNMP present in processed meat (Molognoni et al., 2019). In general, the substrate involved and the product formed in biotransformation bear a structural resemblance. Sometimes either a single microorganism or combinations of microorganisms are used in the biotransformation of one substance into another substance (Ray and Bera, 2014).
This review paper focuses on the (i) various classes of mutagens that are most commonly found in processed foods, (ii) their metabolic assessment, (iii) techniques involved in the detection of mutagens, (iv) analysis of mutagens, and finally (v) microbial conversion of mutagen into non-mutagenic semblance.
Mutagens in foods
Mutagens of Processed foods
During the food processing, several mutagens make their way into the final food product either intentionally or accidentally. The presence of these mutagens in food products is a matter of grave concern because it results in mutagenesis followed by carcinogenesis. Mutagens can be classified into the following categories:
N-Nitroso derivatives
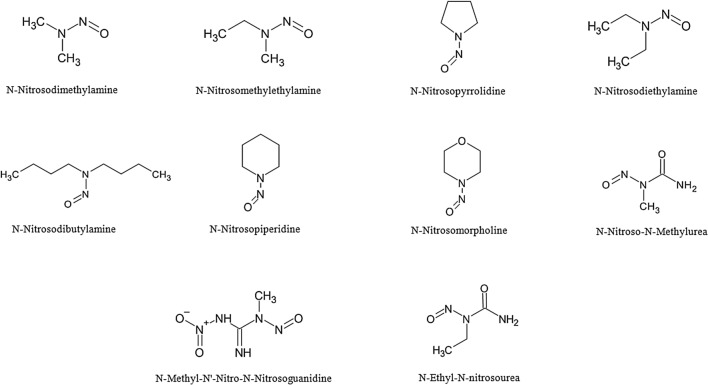
N-Nitroso derivatives were proven to be efficient mutagens when converted to their amide form because they can then act as robust alkylating agents. Amide form of N-Nitroso derivatives can be formed by reaction between nitrate and amines or amides of food products which are formed by catabolism of proteins or any other food components. N-Nitrosodimethylamine is a commonly found N-Nitroso derivative in cheese, beer, and nitrate-nitrite-preserved meats (Table 1) along with other potent mutagens such as N-nitrosopyrrolidine and N-nitrosopiperidine (Miller and Miller, 1986). Nitrosamines are present in several agricultural products, seafood, milk and milk products, meat and meat products, processed meats, oils, margarine, soybean paste, soy sauce, seasoning, sauce, and alcoholic beverages (Park et al., 2015). Figure 1. presents the schematic representation of structures of N-Nitroso derivatives. The formation of these mutagens in processed foods was greatly reduced by the addition of ascorbic acid (Ohshima and Bartsch, 1981). Few studies have demonstrated the induction of colorectal cancer due to the presence of N-Nitroso derivatives (Bogovski and Bogovski, 1981; Clinton et al., 1988; Cross and Sinha, 2004). These derivatives act as alkylating factors thereby altering the DNA which ultimately results in cancer (Saffhill et al., 1985). In general, these N-Nitroso derivatives are found in processed smoky or fire-dried meat because at high temperature nitrogen present in the surrounding atmosphere gets converted into nitrogen oxide followed by reacting with amines present in meat. Table 2 depicts the list of specific mutagens found from specific processed foods under particular temperature conditions. The human body absorbs nearly 36–140 μg of N-Nitroso derivatives mainly through bacon and beer. One study reported the formation of “Nitrosoazetidine-2-carboxylic acid” in the stomach if the diet contains more nitrites and free amino acids. It is a well-known nitrosamine derivative that was found to be carcinogenic (Lijinsky et al., 1970). So, these N-Nitroso derivatives that are known to cause cancer or mutation can be controlled by limiting the conditions favorable for its formation in food.
Table 1.
List of various mutagens found in varying processed foods
| Chemicals | Example | Type of processed food | References |
|---|---|---|---|
| N-Nitroso derivatives | NDMA | Cheese, beer and nitrate-nitrite-preserved meats | Miller and Miller (1986) |
| NPYR and NPIP | Nitrate-nitrite-preserved meats | Scanlan (2003) | |
| Polycyclic aromatic hydrocarbons | Benzopyrene | Charcoal grilled food (meat, fish) | Sugimura et al. (1979) |
| Heterocyclic aromatic amines | Trp-P-1 | Burned and charred foods (meat, fish) | Hosaka et al. (1981), Matsukura et al. (1981), Ohgaki et al. (1984), Takayama et al. (1984; 1985) |
| Trp-P-2 | |||
| Glu-P-1 | |||
| Glu-P-2 | |||
| Imidazo[4,5-F]Quinoxaline derivatives | Fried beef | Sugimura (1986) | |
| Food additives | AF-2 | Preservative | Miller and Miller (1986) |
| Saccharin | |||
| ENA and DNMP | Meat, beef, pork | Namiki et al. (1981) |
Fig. 1.
Structures of various mutagenic N-Nitroso derivatives and its structures (Bogovski and Bogovski, 1981; Park et al., 2015)
Table 2.
List of specific mutagens found from specific processed foods under particular temperature condition
| Mutagens | Food kind | Example | Condition | Concentration | References |
|---|---|---|---|---|---|
| MeIQx; PhIP; DiMeIQx | Thermally processed commercial finished foods | Grilled chicken and pork, flame-broiled fish, meat patties | Grilling condition: 80–90 min, 180–200 °C; Broiled condition: gas flame, 10–15 min |
MeIQx: 0.04–0.4 ng/g PhIP: 0.5–3.8 ng/g DiMeIQx: 0.03–0.2 ng/g |
Tikkanen et al. (1993) |
| NDMA | Thermally processed and fermented food | Smoky and fire-dried meat, bacon, beer | – | 36–140 µg | Cross and Sinha (2004), Gangolli et al. (1994), Mirvish, (1995) |
| Benzopyrene | Thermally processed and fermented food | Grilled meat, hamburgers and chicken | – | 25–300 µg/day | Cross and Sinha (2004), Kazerouni et al. (2001), Phillips (1999) |
| DMIP; IQ; MeIQ; MeIQx; 4,8-DiMeIQx; 4,7,8-TriMeIQx; PhIP; Trp-P-1; Trp-P-2; AαC; MeAαC; Harman; Norharman | Offal foods | Raw beef liver, lamb kidney, beef tongue | 220–225 °C, 5 min | 0.25 ng/g | Khan et al. (2009) |
Polycyclic aromatic hydrocarbons
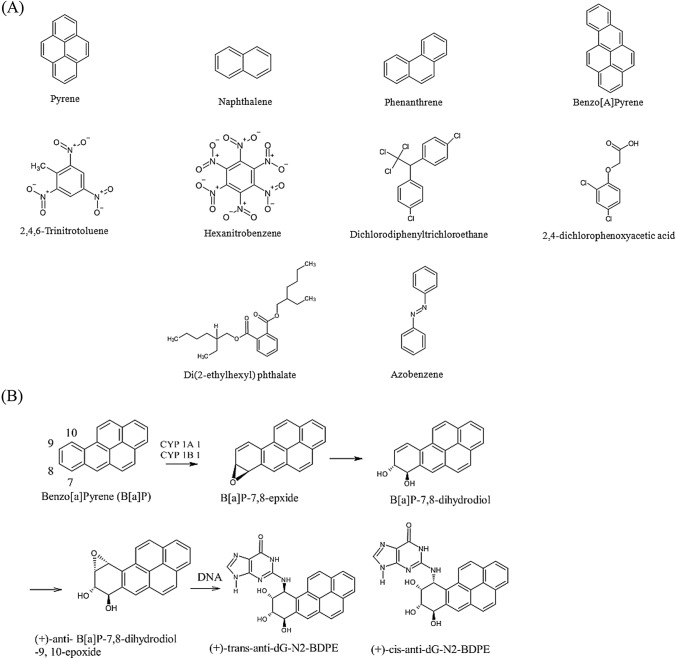
The polycyclic aromatic hydrocarbons (PAHs) occur in food products that are processed through charcoal grilling because it can melt the fat present in the food (such as meat or fish) (Table 1). Deposition of smoke particles generated due to partial burning (Pyrolysis) can also produce PAH Compounds. Even the food additives like smoke-flavoring products can generate a potential amount of PAH (Hamidi et al., 2016). It was also reported that due to high temperature some organic contents of food fragments generate free radicals, which in turn can produce a significant amount of PAH to food (Wang et al., 2019). The use of edible oils can also add PAH to food materials which reflect the method of its formation (Ingenbleek et al., 2019). It can also get added to the final food product due to the pyrolysis of oil, wood, garbage and coal (Kim et al., 2021). Direct addition through air, water and soil is observed (Göker et al., 2021). Structures of a few PAHs were illustrated in Fig. 2A. Due to high temperature the PAH gets converted into benzopyrene which can affect the DNA by forming adducts with it (Fig. 2B) (Sugimura et al., 1979). Benzopyrene is formed due to the partial burning of organic components (Kazerouni et al., 2001; Phillips, 1999). The occurrence of benzopyrene in processed foods in terms of concentration is low (1–10 ng/g) (Miller and Miller 1986). However, it was reported that charcoal-broiled beef can contain up to 8–50 µg/kg (Krone and Iwaoka, 1983; Lijinsky and Shubik, 1964). Hence, the attempt should be either finding an alternative for burning and grilling methods or finding a way out to convert these mutagenic compounds to non-mutagenic compounds, which is further explored in the review.
Fig. 2.
(A) Structures of various mutagenic Polycyclic aromatic hydrocarbons (Shukla et al., 2014). (B) DNA Adducts of Polycyclic aromatic hydrocarbons Benzo [a] Pyrene (Basu, 2018)
Heterocyclic aromatic amines
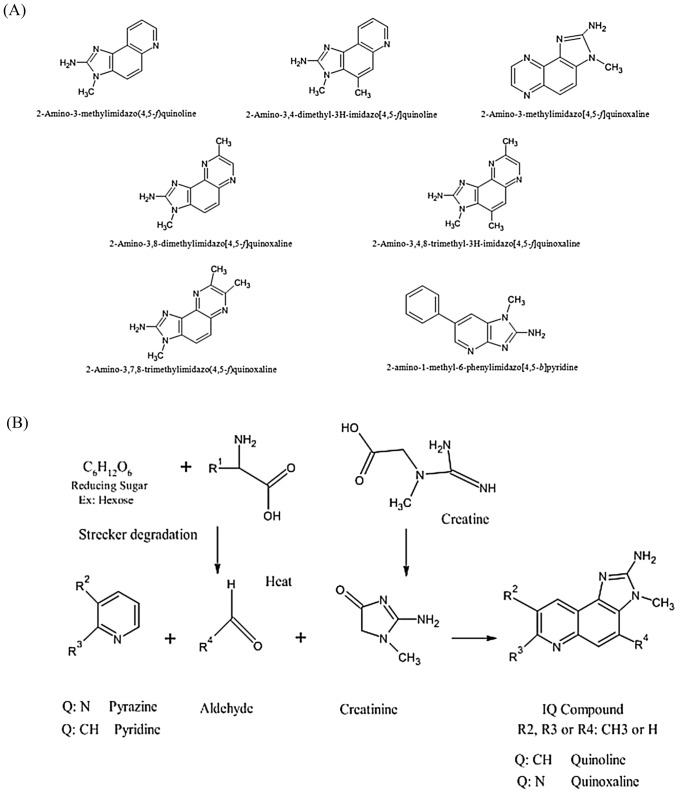
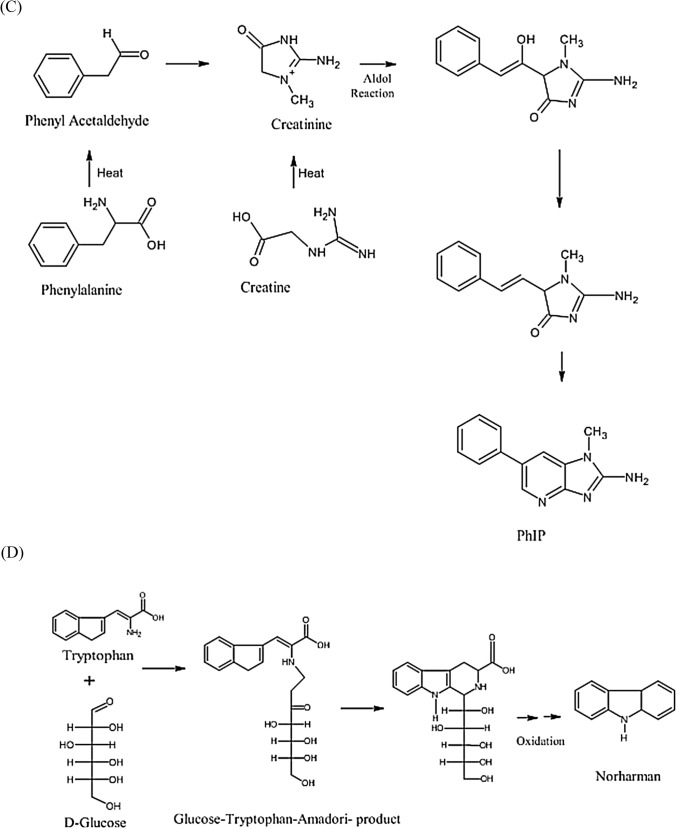
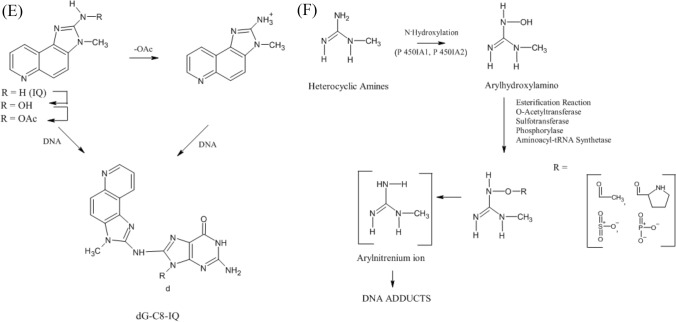
The most notable mutagens are heterocyclic aromatic amines. These are further classified into two categories (a) IQ type and (b) non-IQ type. IQ type is those whose amino functional groups are resistant to nitrite treatment, whereas non-IQ type is those whose amino functional groups are non-resistant to nitrite treatment (Tsuda et al., 1985). The mutagens belonging to this category are highly stable; therefore they remain in the food products for a very long time (Wakabayashi et al., 1992). Amino methyl imidazo furo pyridine, a well-known heterocyclic amine, consists of oxygen and can occur in 4 isomers due to the positioning of oxygen and N-methyl group (Knize et al., 1990). The structures of various mutagenic heterocyclic amines were illustrated in Fig. 3A. These compounds were produced due to the reaction between amino acids (such as creatine and creatinine) (Fig. 3B–D) that are present in meat and sugars at high temperature (Gibis, 2016; Jägerstad et al., 1991; Khan et al., 2009; Milić et al., 1993; Pearson et al., 1992). The compounds such as creatine, amino acids, and sugars react with each other at elevated temperatures of 150–300 °C due to the Maillard reaction (Dong et al., 2020). Although an elevated temperature do not have any direct effect, the duration of applied temperature is responsible for forming HAA. Water activity and pH are also equally responsible for HAA formation during the cooking procedure (Oz, 2021). A study also revealed that the polar types of HAA like imidazoquinoline & imidazoquinoxaline produced at a temperature around 150–250 °C. At 250 °C due to pyrolysis of amino acid non-polar type of HAA (dipyridoimidazole or pyridoindole) are formed because of pyrolysis of amino acid (Barzegar et al., 2019). A temperature between 100 and 300 °C IQ type (Thermic HAA) are formed and temperature above 300 °C Non-IQ type (Pyrolytic HAA) are formed. For example, a study revealed that more HAA is formed on the surface of meat than the internal part. The possible reason could be the transportation of water and dissolved substances to the surface due to capillary action (Alaejos and Afonso, 2011). HAA formation can be controlled by increasing the sugar contents of food (Meurillon and Engel, 2016). These heterocyclic aromatic amines also affect the DNA by forming adducts with it (Fig. 3E and F). Proper research is needed to restrict the formation of DNA-Adducts, which is otherwise a major challenge for a healthy life.
Fig. 3.
(A) Structures of various mutagenic heterocyclic amines (Wakabayashi et al., 1992). (B) Formation of Heterocyclic aromatic amines—IQ Compounds (Gibis, 2016). (C) Formation of Heterocyclic aromatic amines—PhIP (Gibis, 2016). (D) Formation of Heterocyclic aromatic amines—Norharman as Beta carboline (Gibis, 2016). (E) DNA Adducts of Heterocyclic aromatic amines—IQ DNA (Bose et al., 2015). (F) DNA Adducts of Heterocyclic aromatic amines—DNA (Schut and Snyderwine, 1999)
These mutagens were also found to be present in processed foods that went through charring (Table 1). Due to heat, pyrolysis occurs resulting in the formation of heterocyclic aromatic amines. Amino acids such as tryptophan, glutamic acid and phenylalanine give rise to strong mutagens upon pyrolysis. Trp-P-1(3-amino-1,4-dimethyl-5H-pyrido[4,3-b]indole), Trp-P-2(3-amino-1-methyl-5H-pyrido[4,3]indole), Glu-P-1(2-amino-6-methyldipyrido-[1,2-a:3’,2’d] imidazole), and Glu-P-2 (2-aminodipyrido-[1,2-a:3’,2’d]imidazole) are mutagens formed via pyrolysis of tryptophan and glutamic acid respectively (Hosaka et al., 1981; Matsukura et al., 1981; Ohgaki et al., 1984; Takayama et al., 1984, 1985). Imidazo[4,5-F]Quinoxaline derivatives were also demonstrated to be potent mutagens obtained from fried beef (Table 1) (Sugimura, 1986; Yamamoto et al., 1978). Amino-α-carboline (AαC) and methylamino-α-carboline (MeAαC) are the mutagens that are produced during pyrolysis of soybean globulin (Yoshida et al., 1978).
Most common heterocyclic amines in cooked meat are 2-amino-3,8-dimethylimidazo [4,5-f]-quinoxaline (MeIQx); 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine (PhIP); DiMeIQx (Combination of 2-amino-3,4,8-trimethylimidazo[4,5-f]quinoxaline (4,8-DiMeIQx) and 2-amino-3,7,8-trimethylimidazo[4,5-f]quinoxaline (7,8-DiMeIQx)) (Kasai et al., 1980; Krone and Iwaoka, 1983; Lynch et al., 1992; Sugimura et al., 1979; Tikkanen et al., 1993; Yamamoto et al., 1978). It was found that the daily consumption of PhIP is 43–110 ng/day and that of MeIQx is 14–47 ng/day (Cantwell et al., 2004; Cross and Sinha, 2004; Delfino et al., 2000; Sinha et al., 2000a; 2000b; 2001). These substances are very much responsible for the carcinogenesis of intestinal cancer (Ito et al., 1997; Shirai et al., 1995). Heterocyclic aromatic amines are also generated from meat flavors and beef stock extracts which are known as ‘processed food flavors’ (Stavric et al., 1997).
ENA and DNMP
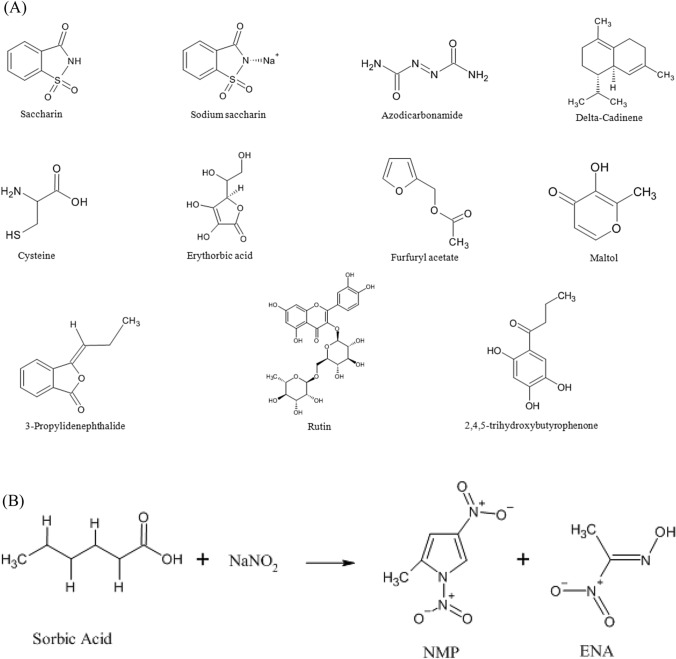
One study reported the experiment on ENA and DNMP, to evaluate the generation of these compounds in the laboratory under various cooking conditions. For the production of ENA and DNMP mutagens, nitrite and sorbate were mixed in 8:1 and 2:1 proportions respectively. This reaction was performed in four batches under different conditions. First batch was done by adding sodium nitrite and potassium sorbate in the ratio of 2:1 without sodium erythorbate, whereas the second batch was supplemented with 0.1% of sodium erythorbate. Third batch had sodium nitrite and potassium sorbate in the ratio of 8:1 without sodium erythorbate, whereas the second batch was supplemented with 0.1% of sodium erythorbate. The food samples used were pork meat and boneless poultry mortadella. Meat (50 g) from all the batches was used for evaluation. To evaluate the meat samples were heated at temperature 60 ± 5 °C, whereas the pH was maintained at 5.0 ± 0.1.ENA (5 µg/kg) and DNMP (12 µg/kg) of mortadella meat samples were used as controls. Based on the results, it was observed that the samples with sodium nitrite and potassium sorbate ratio of 8:1 had generated higher ENA and DNMP load. Sodium erythorbate, an antioxidant which is known to reduce the formation of mutagens in food products, proved to be ineffective in doing so. Hence based on the results it was evident that the addition of sodium erythorbate did not have any effect on reducing ENA formation. However, it was observed to control DNMP concentration. One study reported that the concentration of ENA level is proportional to nitrite concentration. It was also reported that at 170 °C, the level of ENA decreased by ~ 37% (Molognoni et al., 2019).
Another study reported the laboratory preparation of DNMP by dissolving 2 g of potassium sorbate with 20 g of sodium nitrite in water at pH 3.5. Later, it was heated to 60 °C for 30 min under constant stirring. A standard Y was extracted by using DCM at 25 °C and pH 3.5. It was dried with anhydrous sodium sulfate followed by purification through chromatography. Using toluene and ethyl acetate as mobile phase and ethyl acetate as elution buffer. The obtained filtrate was then analyzed using TLC. DNMP was identified as a yellow spot with Rf value of 0.6. This fraction was then separated and crystallized using a combination of ethyl acetate and hexane (50:50). Later, the compound was identified using q-Mass 910 spectrometer (Perkin-Elmer (USA)). In addition, ENA was also prepared by the reaction between nitroethane and sodium nitrite. The compound form was identified with the help of GC–MS, NMR and IR spectrometer. Anti-oxidants such as ascorbic acid, vegetable juices and cysteine can be used to avoid the formation of such mutagens (Binstok et al., 1998). Cysteine prevents the formation of DNMP by attacking DNMP aromatic ring structure and thereafter removing the C-nitro group (Osawa et al., 1986). It was also observed that higher concentrations of sodium nitrite compared to potassium sorbate yields more ENA and DNMP formation (Molognoni et al., 2019).
Food additives
Food additives were proved as potent and efficient mutagens. The Food and Drug Administration is working tirelessly to control the use of such mutagenic substances in food processing as food additives. Sorbic acid and sodium nitrite were well-known and most widely used food preservatives. Several organic chemicals are used in food preparations such as Azodicarbonamide, Cardinene, Cysteine, Erythorbic Acid, Furfuryl Acetate, Maltol, 3-Propylidenephthalide, Rutin, and 2,4,5-Trihydroxybutyrophenone, which are weak mutagenic compounds. Saccharine, aspartame, and flavoring agents like chilly sauce, soy sauce, vanilla essence, rose syrup, and ice cream were tested by employing Ames Salmonella microsome assay and their structures were presented in Fig. 4A. Sorbic acid was used in cooked food to increase their shelf life, to protect the food from microbial contamination, and to increase the quality of food (Robach and Sofos, 1982). But later it was found that sorbic acid reacts with sodium nitrite resulting in the formation of mutagenic compounds such as ENA and NMP (Fig. 4B), mostly around pH 3.5–4.2 (Hartman, 1983; Namiki et al., 1981). These two compounds, ENA and DNMP, were proved to be strong alkylating agents resulting in damaging the DNA (Pérez-Prior et al., 2010). Food additives are substances that found their way into food products by intentional activities of humans. Food additives are used for several reasons: to increase food flavor, to preserve food products for a long duration, and to enhance food texture. A well-known and widely used food additive is “2-(2-Furyl)-3-(5-nitro -2-furyl)acrylamide (AF 2)”, a highly mutagenic compound that has been used in food products as a preservative (Table 1). Few studies also reported the mutagenic and carcinogenic effect of saccharin on the urinary bladder, another well-known food preservative.
Fig. 4.
(A) Structures of various mutagenic Food additives (Kramers, 1975; Zeiger, 1993). (B) Schematic representation of ENA (Pérez-Prior et al., 2009)
Mutagens of prepackaged foods
Several compounds/ingredients that were essential and integral in food processing were in fact found to be mutagenic as well as carcinogenic. In most cases, these ingredients are non-toxic when present individually and in normal conditions but can be harmful (mutagenic/ carcinogenic) when combined with other ingredients or unfavorable cooking or processing conditions, which results in the formation of mutagenic forms. One such mutagenic compound that was recognized as carcinogenic in foods that are already cooked (fried) was heterocyclic amines (specifically aminoimidazoazaarene (AIA) compounds that comprise quinolines and quinoxalines along with pyridines) (Felton et al., 1992; Tikkanen et al., 1993; Turesky, 1990; Wakabayashi et al., 1992). One study also reported that caramel, which was used as a coloring agent in beverages, gravy mixes, soup, etc., also possesses mutagenic activity (Krone and Iwaoka, 1983). The presence of fatty acids in food when heated along with creatine (an amino acid) proved to increase the mutagenicity of food products (Yoshida and Okamoto, 1980). It was also found that lipids and their degraded intermediates present in the food products contribute to more mutagenesis than any other component of food nutrients. Similarly, it was also reported that creatine phosphate and tryptophan were also responsible for accelerating the mutagenic effect (Taylor et al., 1985). In the same way, the addition of ammonium ions in food products having high carbohydrate content also raises mutagenicity (Iwaoka et al., 1981). Therefore it was reported that the level of carbonyl, as well as nitrogenous components in the food sample, was responsible for mutagenicity level in final food products (Krone et al., 1986). Therefore, it was concluded that all the major nutrients of foods viz. carbohydrates, amino acids, fats, and lipids may generates mutagenic compounds when reacts with each other in any specific condition. In the case of amino acids mainly creatinine and tryptophan are involved in mutagenic formation. One study reported the unintentional entry of harmful substances because of plastic wrapping used in food products for a long time (Nerín et al., 2016). Research is also needed in finding such components and reaction conditions that can indulge in forming mutagenic compounds.
Factors affecting mutagenicity
Few studies have reported that both time and temperature have an effect on mutagenicity. Under frying temperature (150–300 °C) mutagenicity increases and then becomes stable. Temperature above 140 °C was found to be responsible for enhancing mutagenicity in the case of fried ground beef (Bjeldanes et al., 1983; Krone et al., 1986; Pariza et al., 1979). The presence of mutagens was tested in grilled chicken and pork, flame-broiled fish, and meat patties at an ambiance of 180–200 °C for 80–90 min (grilling conditions) and gas flame for 10–15 min (broiled fish) (Tikkanen et al., 1993). These mutagens were formed due to the pyrolysis of proteins present in food. 2-amino-1-methyl-6-phenylimidazo [4,5-b]pyridine (PhIP) average estimated uptake was found to be approximately 0.4-16 µg/day (Wakabayashi et al., 1992). Several studies have reported that heating increases the mutagenicity of food products (Krone and Iwaoka, 1983). One study reported that when food samples were washed with distilled water it reduced the risk of mutagenicity with every subsequent washing. Hence it was evident that the precursors of the mutagens can be extracted by washing with distilled water (Krone et al., 1986). When two samples of fish were tested (steamed and non-steamed), it was observed that steamed fish showed more mutagenicity. This result implies that precursors of mutagens in fish were converting into a more responsive form, like conversion of creatine into creatinine at high temperature (Jägerstad et al., 1983).
Time is another major factor that also affects the level of mutagenicity in food products. The boiled beef extract showed increased mutagenicity at less than 30 h of boiling time. In the fried sole system it was found that heating at a temperature of 125 °C for 18 min enhanced mutagenicity. Both time and temperature were found to be responsible for mutagenicity but the effect of temperature was found to be more dangerous compared to time (Bjeldanes et al., 1983; Krone et al., 1986).
Metabolic assessment of mutagens
Among the mutagens and carcinogens studied to understand the metabolic pathways involved most of them belonged to heterocyclic amines. These mutagens and carcinogens can cause serious harm to DNA leading to disruption in the metabolic structure of the body. Therefore, it is essential to study and understand the metabolic pathways involved in mutagenesis and carcinogenesis. It also helps us to know how it will affect and cause impairment in the human system. Formation of mutagens by metabolism can be identified by specialized method in AMES test i.e. pre incubation method. In this method, the food material is pre incubated with Salmonella strains in the presence of liver enzymes (rat liver microsome preparation) which allows the formation of any mutagenic metabolites from the parent food material. Some compounds may be detected more efficiently using the pre-incubation method. These compounds belong to chemical classes that include short chain aliphatic nitrosamines, divalent metals, aldehydes, azo-dyes and diazo compounds, pyrollizidine alkaloids, allyl compounds and nitro compounds.
Heterocyclic amines were found to be turned on with the help of cytochrome P450s. Cytochrome P450s help heterocyclic amines to get converted into hydroxyl amino group from amino group. Several studies have reported that cytochrome P450IA2 is the most efficient enhancer of heterocyclic amines (Boobis et al., 1994; Cross and Sinha, 2004; Turesky et al., 1991). This is followed by the conversion of hydroxylamine to esters by acetic acid, proline, and last but not least sulphuric acid. Finally, these mutagens will act as DNA contaminants. This very property of heterocyclic amine, that it can enter and bind to DNA molecules, was elucidated by 32P-postlabeling method (Wakabayashi et al., 1992).
Activation of heterocyclic amines also occurs due to one-electron oxidation that results in the formation of a free-radical substance. This happens due to extra-hepatic tissue and catalyzation with the help of prostaglandin H (Wild and Degen, 1987; Cross and Sinha, 2004). Trp-P-2, a heterocyclic amine, was also found to be activated with 12-O-tetra-decanoylphorbol- 13-acetate. N-nitrosoamine derivatives which are also proved to be potent mutagens were also activated with the help of cytochrome P-450 (Crespi et al., 1991). Few compounds function as a mutagenic precursor for example 1-methyl-1,2,3,4 -tetrahydro-β-carboline-3-carboxylic acid. This mutagenic precursor is produced by the combination of L-typtophan and acetaldehyde. Tyramine is another such compound that converts into mutagens after nitrite treatment (Ochiai et al., 1984). Mutagenic precursors get converted into mutagens under acidic conditions that arise due to conversion of nitrate to nitrite (Sugimura, 1986). Research is still needed in the field that helps us to recognize any such more pathways responsible for the formation of mutagens.
Detection of Mutagens
Identification of mutagens from food samples
Several methods were employed in the extraction of mutagens from the food products. One study reported the extraction of mutagens by mixing the canned foods with methanol followed by filtration using Whatman filter paper No.1. Later, methanol was removed from the filtrate using a rotary evaporator and mixed with distilled water, four times the weight of the food sample. The obtained mixture was then partitioned with the help of Dichloromethane (DCM-CH2Cl2) after adjusting the pH to 2.5. The pH of the aqueous layer was adjusted to 10 and finally, it was extracted with DCM. After extraction, it was dried, evaporated, and finally dissolved in Dimethyl sulfoxide (DMSO-(CH3)2SO) (Krone and Iwaoka, 1983).
The fat can be eliminated from the boneless food product by homogenization in acetone followed by hexane extraction. This process was followed by DCM to separate the alkaline substances. The obtained product was further concentrated through evaporation using nitrogen steam and later was stored in methanol (Tikkanen et al. 1993). One study reported the extraction of mutagens from the food samples. With the help of 1 M NaOH and later mixed with diatomaceous earth. It was then added to the empty column (propylsulfonic acid (PRS) cartridge) and eluted with ethyl acetate to the PRS cartridge followed by rinsing with methanol: water (4:6 v/v). PRS cartridge then coupled with C8 cartridge and then absorbed by ammonium acetate, finally rinsed with water and eluted out by methanol: ammonia (9:1 v/v). The solvent was then evaporated and the resultant was dissolved in 4,7,8-TriMeIQx in methanol (Khan et al., 2009). Another study reported the use of an aqueous extraction method for unsheathing of mutagens from the food sample. In this method, frozen food products were mixed with distilled water followed by dissolving in sodium sulfate (Na2SO4) by adjusting the pH to 2.5 followed by filtration. Later the obtained filtrate was then extracted using DCM by maintaining the pH of the solution. The final products were dissolved in DMSO (Krone and Iwaoka, 1983). Extraction of mutagens by performing a liquid–liquid extraction process was also reported. In this process, the sample was mixed with HCl followed by its extraction using DCM. After separation, two layers of DCM were discarded and the further process was carried out using an aqueous layer. To this aqueous layer, fresh DCM was added after adjusting the pH to 13 with the help of NaOH. The obtained product was dried completely for further use (Stavric et al., 1997). Mutagens were also extracted using solutions with acetonitrile and formic acid. The solution was centrifuged to get the supernatant to which magnesium sulfate and activated charcoal were added respectively and again centrifuged. Finally, the entire solution mixture was dried under nitrogen and stored at 70% methanol supplemented with 0.1% formic acid. Though there are several methods reported for the extraction of mutagens, a method/procedure should be optimized and standardized properly for successful extraction of mutagens from any specific food products.
Chemical derivatization
In order to detect the presence of mutagens in any food sample, it should be converted to a form that can be easily detectable by HPLC or LC/GC–MS. The presence of mutagens MeIQx, PhIP, and DiMeIQx in the processed foods (namely grilled chicken and pork, flame-broiled fish, and meat patties) were detected in the form of 3,5-Bis(trifluoromethyl)benzyl bromide. 100 μL of 3,5-Bis (trifluoromethyl)benzyl bromide was added to 2 mL of acetonitrile (reference sample) and 0.5 mL of diisopropylethylamine (test sample) to get its derivatives. All the chemicals, reference, and test samples were extracted in their derivative form with the help of hexane. The mutagens in the food samples were also detected in the form of heptafluorobutyric anhydride derivatives. To get this derivative, food samples were mixed with 5 mL of hexane along with 50 μL of heptafluorobutyric anhydride. Among the two it was found that heptafluorobutyric anhydride derivatives gave the best result in GC–MS and samples were found to get detected at a concentration of 5 mg/L. 3,5-Bis(trifluoromethyl)-benzyl bromide derivatives were found to be satisfactory for sensitive mutagen detection and gave better results in terms of reproducibility. 7,8-DiMeIQx, 4,8-DiMeIQx, and MeIQx mutagens were detected at as low as 10 µL concentration and PhIP at 70 µg/L concentration in the form of 3,5-Bis(trifluoromethyl)-benzyl bromide derivative (Tikkanen et al., 1993).
Analysis of mutagens
Food samples can be analyzed using analytical techniques like HPLC or LC/GC–MS for the detection and evaluation of specific mutagens. All the mutagenic heterocyclic amines like., DMIP, MeIQx, 4,8-DiMeIQx and PhOQ were identified using LC–MS/MS with turbo ion spray source operating positive mode, C8 symmetry LC column (150 mm × 2.1 mm I.D, 5 µm particle size). 3,5-bis(trifluoromethyl)benzyl bromide derivatives and hepta fluorobutyric anhydride derivatives were identified using the gas chromatography-mass spectrometry (GC–MS/MS) with negative ionization and run time 75 min, 25 m non-polar ULTRA-2 column 0.2 mm I.D, 0.11 µm film thickness and the temperature was held at 270 °C for 3 min and then increased to 320 °C at 10 °C/minute, helium was used as the carrier gas, the split less time was 0.35 min and the injection volume of the sample was 1.0 µL (Tikkanen et al., 1993). Mutagens formed by sorbic acid-sodium nitrite reactions was identified using TLC and high performance liquid chromatography (HPLC) with UV light region methods, n-hexan: acetic acid: ethyl acetate: chloroform (45:6::10:1) mobile phase was used with µBondpeak column, diluent was water: methanol (85:15), products were detected at 254 nm at a flow rate of 1.5 mL/min (Namiki et al., 1981). The use of HPLC /LC–MS technique was also reported in the identification of mutagens for the food samples (Khan et al., 2009). The electrochemical technique is another approach that was also found to be effective in the determination of heterocyclic amine (Wakabayashi et al., 1992). The use of HPLC in combination with electrochemical detection was also reported in the detection of mutagens (Takahashi et al., 1985). Only LC was also used for the determination of ENA mutagen with µBondapak C18 column. For the mobile phase, water: methanol in the ratio of 85:15 was used at a flow rate of 1.5 mL/min. Detection of mutagens was done using a UV spectrophotometer at 254 nm (Namiki et al., 1981).
One study reported the application of LC–MS for the determination of mutagens, in which 20% acetonitrile + 80% acetic acid was used for preparing the stock solution. Prior to LC–MS, the sample was purified using a solid phase extraction (SPE) cartridge bound to HEMA (2-Hydroxyethyl methacrylate) resin, a cationic exchanger. A sample mixed with 0.06% formic acid and 20% acetonitrile was loaded onto the column. Later, the column was washed with 0.1 N HCl followed by 20% methyl alcohol in 0.1 N HCl + 1 mL water. Mutagens (heterocyclic aromatic amines) bound to the column were eluted out by using 40% acetonitrile + 0.5 N ammonium acetate-ammonium hydroxide buffer of pH 9.4. The eluate was then dried for further work (Stavric et al., 1997). Another study reported the application of PRS cation exchange with the C18 column. In this method, the sample was loaded onto the column with 20% methyl alcohol + 80% 0.1 N HCl. For elution, 40% acetonitrile + 60% 0.5 M ammonium acetate of pH 8.5 was used. The eluate obtained was concentrated by keeping it at 80 °C. It was then applied to C18 column by using 0.5 M ammonium acetate as a mobile phase. Mutagen was eluted through the column by using 90% methyl hydroxide in a 10% aqueous ammonia solution. Finally, the obtained eluate was dried for further use. For the detection of mutagens using LC–MS, a polymer-based PLRP-S column with LC-600 pump and SPD-10A UV detector was used. The flow rate was maintained at 0.7 mL/min. Two different mobile phases were used: phase A with 0.05% formic acid + 5 mM ammonium acetate in water at pH 3.2, whereas phase B with 0.06% formic acid + 49.82% acetonitrile + 3 mM ammonium acetate in water at pH 3.8. The mobile phase was used in the gradient system starting with 18% of phase B for 1 min then shifted to 25% of phase B at a rate of 14% per min for 3.4 min. Then the gradient was increased to 50% at a rate of 1.9% per min for 7 min and finally to 18% for 28 min. While using the TSK-GEL ODS-80TM column, the flow rate was maintained at 0.6 mL/min and a gradient mobile phase system was used. Mobile phase A contains 0.15% formic acid + 15 mM ammonium acetate at pH 3.1, whereas phase B contains 0.15% formic acid + 15 mM ammonium acetate + 50% acetonitrile. Initially, 12% of phase B for 1 min was used then 51% for 14 min, and finally 70% for 5 min (Stavric et al., 1997).
One study reported the application of GC–MS with a 70 eV electronic impact detector. Methyl siloxane was used as a solvent system, whereas helium was used as carrier gas. For the detection of DNMP mutagen the conditions were 100 °C initial temperature which was increased to 250 °C at a rate of 8 °C per min. Flow rate used was 1 mL/min with the injection volume of 1 μL along with ethyl acetate. Retention time was 10.68 min. For the detection of ENA mutagen, initial temperature was set to 40 °C and the temperature was increased to 250 °C at 10 °C per min. Flow rate was maintained at 1 mL/min. Injection volume was 1 μL whereas retention time was observed as 2.18 min. Quantitatively ENA was determined using HPLC with automatic injector, integrator and spectrophotometric detector of variable wavelength. Mobile phase of water: methanol (85:15) was used for performing HPLC at a flow rate of 1 mL/min with 1 μL of injection volume. Detection of mutagen was achieved at 254 nm and retention time was recorded at 6.5 min (Binstok et al., 1998).
Detection of mutagens like DNMP and NMAP was done by HPLC technique using methanol–water (1:9) mobile phase. Flow rate was maintained at 2 mL/min with approximately 15 psi pressure. Detection of mutagens was done at 254 nm (Shu et al., 1991). One study reported the use of HPLC–DAD (diode-array detector), AC-200F NMR (Nuclear Magnetic Resonance) spectrometer, 5500 QTRP hybrid quadrupole-linear ion trap-mass spectrophotometer with electro spray ionizer and 1290 infinity HPLC-QqLIT-ESI/MS/MS, 7100 capillary electrophoresis (CE) with DAD. For HPLC, the stationary phase used was di-isopropyl-3-aminopropyl silane attached with hydroxylated silica, phenyl-hexyl, or C18 (Molognoni et al., 2019).
Testing of mutagenicity
In 1966, Ames and Whitfield proposed histidine auxotroph strains for screening chemicals for mutagens using spot test method that was previously used by Szybalski and Iyer and Szybalski for mutagen screening with an E. coli strain. The spot test uses selective agar medium plate seeded with the test organism (Mortelmans and Zeiger, 2000). Later, in 1973, (Ames et al., 1975) with his colleagues developed the more sensitive plate incorporation assay procedure. This method greatly enhanced the ability of laboratories to test chemicals for mutagenicity (Ames et al., 1975). Later, the same Bruce N. Ames along with his colleague, (Maron and Ames, 1983) introduced additional target strains such as frameshift strain (TA97) and a strain carrying an ochre mutation on a multicopy plasmid (TA102) into the method which was practiced then (Maron and Ames, 1983). The Salmonella test was scientifically validated in a study of 300 known chemicals by 1977 (Maron and Ames, 1983). It was subsequently validated globally and usage has been started in various Chemical Industries.
Mutagenicity of any compound was tested through Salmonella mutagenicity testing method. Plate incorporation method was used to determine the mutagenicity of the suspected mutagens such as MeIQx, PhIP and DiMeIQx, whereas mutagenic activity was determined by calculating revertants/g (de Meester, 1989; Maron and Ames, 1983). Salmonella typhimurium tester strain TA98 was used for testing mutagens. Maximum mutagenic activity was observed in processed fish products compared to other food products such as meat patties, grilled chicken and pork. Some of the fish samples even showed highest concentration of PhIP with 0.5–5.5 ng/g of fish but the factor causing strong mutagenic activity of fish is still unclear (Tikkanen et al., 1993). To accelerate the process of mutagenicity testing metabolic activation of mutagens were added. For example, addition of fortified liver microsomes enhances metabolic activation. One study reported the use of Ames test to determine the mutagenicity of the food products through employing Salmonella typhimurium strain 1538, TA98, and TA100 as test organisms (Krone and Iwaoka, 1983). Food samples were tested in the presence and absence of Aroclor (80 μL) induced rat liver microsome preparation. Among the studied food samples, French fried fish sticks were found to be more mutagenic with mutagenic activity ratio (MAR) of 1.8 for acidic acid, whereas 2.2 for basic extract. However, this was also considered as non-mutagenic because the criteria used by them indicated positive response up to MAR of 2.5. Additionally, it was also observed that if the food samples were heated at high temperature for a long duration it increases the risk of mutagenicity. Beef and associated food products (beef broth with MAR 13) were found to be more mutagenic compared to other food products such as pink salmon (with MAR 17.6). In the case of canned food it was observed that steam and freezing pre-treatment are harmful for human health as it enhances the tendency of mutagenicity. Similar results were reported upon performing bioassay by using Salmonella typhimurium TA98 strain with Aroclor induced S-9 rat liver homogenate to induce metabolic activation and results were reported as revertant colonies (Stavric et al., 1997).
Food chemicals that are positive by AMES test are generally carcinogens, but the correlation is not definitive. There are some carcinogenic chemicals formed from food processing that are not detected by Salmonella AMES test because they act through various other, non-genotoxic mechanisms or mechanisms absent in this bacterial species. It is also identified that some special mutagens which are not always detected by plate incorporation /pre-incubation method and requires alternative procedures (OECD 2020).
Microbial transformation of mutagens
Several research groups have reported the bioconversion of mutagens found in the food products, however the complete mechanism is yet to be fully explored. For the biotransformation of food mutagens (such as ENA and DNMP) formed due to reaction between nitrite and sorbate, the microbial strain used was Lactobacillus bulgaricus LB-UFSC 001. This strain was preferred due to the use of the microbe as a starter culture in the fermentation process of meat (Molognoni et al., 2019). Lactobacillus bulgaricus LB-UFSC 001 strain used for the study was reactivated a night before the experiment followed by centrifugation at 3800 g-force at 4 °C for 10 min. The obtained pellet was washed with 0.1 mol/L HEPES (4-(2-hydroxyethyl)-1- piperazineethanesulfonic acid) buffer. Cell density was evaluated with the help of a spectrophotometer through calculating the transmission or absorbance of light to get ~ 108 cells/mL. 1 mL of HEPES buffer was used for preparing cell suspension. To investigate the microbial transformation capacity of Lactobacillus bulgaricus LB-UFSC 001 strain, the culture developed by the above mentioned method was inoculated with the test mutagenic sample (ENA and DNMP). The test samples used for mutagenic determination were of two types: one was chemically produced in laboratory and other is from mortadella. By analyzing results it was evident that Lactobacillus bulgaricus LB-UFSC 001 strain was capable of degrading potent mutagens (such as ENA and DNMP), which were produced by the reaction between nitrite and sorbate. Lactobacillus bulgaricus LB-UFSC 001 strain was also reported for the degradation of polycyclic aromatic hydrocarbons as well as biogenic amines (Bartkiene et al., 2017). Additionally, it was also reported for the production of nitroreductase, an enzyme which has the ability to act as catalyst in electron transfer reaction (NADH ↔ NAD+ + H+ + 2e−). This reaction helps to convert the C-nitro group of the compound into C-nitrous form till substitute amines are formed. The result was evaluated on the basis of NMAP detection in the reaction medium. Additionally, the test sample also reported the loss of mutagenic activity of DNMP upon mutagenicity testing. The results indicated that Lactobacillus bulgaricus LB-UFSC 001 strain can degrade ~ 80% of mutagenic content of the food samples under 5 min, which was based on the kinetic analysis. Additionally it was reported that when 100 µg/kg of mutagens were added externally, Lactobacillus bulgaricus LB-UFSC 001 strain can convert 100% of mutagens under 20 min (Molognoni et al., 2019). Motta et al. (2020) reported the function of oxygen insensitive nitroreductase type I B enzyme for bioconversion of DNMP to NMAP. Type I signifies that the enzyme can reduce nitro group either in the presence or absence of oxygen and B represents that the enzyme uses NADH and NADPH as electron source. The organisms found responsible for the activity were Staphylococcus xylosus LYOCARNI SXH-01, Lactobacillus fermentum LB-UFSC 0017, and Lactobacillus casei LB-UFSC 0019.
One study reported the conversion capacity of various microbes under aerobic and anaerobic conditions of DNMP to NMAP. Microorganisms used for this study were of intestinal origin hence collected from feces. Sample was diluted to 25-fold in 0.1 M HEPES buffer at pH 7.6. To this 10 mg of DNMP dissolved in 0.5 mL of DMSO was added. Later the prepared samples were kept in anaerobic condition for 24 h followed by extraction with the help of ethyl acetate. Results indicated that Bacillus thetaiotaomicron VPI, major human intestinal bacteria has the ability to metabolize DNMP. This was determined using thin layer chromatography (TLC), Rf value was found to be 0.26 for DNMP. The formation of NMAP was determined through CI mass spectra, NMR and UV spectral data. Intestinal bacterial species such as Actinomyces, Bacteroides, Clostridium, Eubacterium, Fusobacterium and Peptostreptococcus sp. showed positive results for DNMP metabolism. It was also reported that within 10–20 min Bacillus thetaiotaomicron VPI strain started the conversion of DNMP to NMAP and it reached a steady state in 4 h (Shu et al., 1991). PhIP, one of the abundant heterocyclic aromatic amines found in cooked-meat, can be degraded with the help of human gut microbiota. One such efficient microorganism is Eubacterium hallii. It was found that in proximal colon E. hallii along with other microflora can convert PhIP to PhIP-M1 with 300-fold increased efficiency (Fekry et al., 2016; Zhang et al., 2019a, 2019b). Beer et al. (2019) reported the conversion of other heterocyclic aromatic compounds like AαC, Trp-P-1, harman, norharman, PhIP, MeIQx, and MeIQ with the help of human fecal microflora. The search for similar microorganisms is needed that can help to degrade mutagenic compounds into non-mutagenic compounds efficiently and in less time. Gene level study can also help to identify the product responsible for such conversion. It can also help in the enhancement of the process.
Pathway of microbial conversion
Few studies have reported that microbial transformation of mutagens occurs through certain metabolic pathways. The pathway consists of electrons backflow due to the omission of C-nitro group and presence of N-nitro group in NMAP. This reaction involved consequent shifting of two electrons, whereas shifting of only one electron can give rise to superoxide radicals which can reform DNMP. Lactobacillus bulgaricus LB-UFSC 001 strain took only 30 min for the transformation of DNMP under all experimental conditions. The nitroreductase from this strain can catalyze electron transfer from NAD(P)H for the generation of components like NMAP, NPYR, etc. (Molognoni et al., 2019).
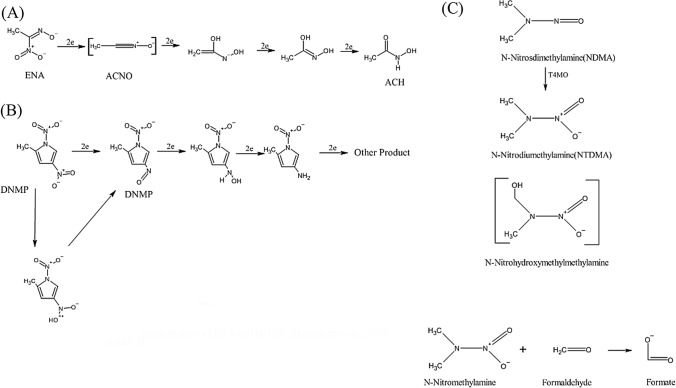
Similar to DNMP, ENA was also reduced to forms that are having less or no mutagenic activity by Lactobacillus bulgaricus LB-UFSC 001 strain using nitroreductase and this became evident by the presence of NO2−, NO3− and some nitrile intermediates Acetone Oxide (ACNO) (Fig. 5A and B). Analytical studies have revealed that the conversion of ENA occurs due to nucleophilic attack through carbon and not by nitrogen of ACNO. The stable intermediates formed in the reaction have strong C = O bond, hence capable of forming end product acetylcholine (ACH). This observation suggested that along with nitroreductase, nitrile hydralases also worked for bioconversion of ENA. The end product ACH is non-mutagenic, thus used as a clinical product for urease inhibition. NDMA (N-Nitrosodimethylamine), a potent carcinogen found in drinking water, can be metabolized through the T4MO pathway. This pathway converts NDMA to NTDMA (N-Nitrodimethylamine) and then to formaldehyde and N-nitromethyl amine. These results were obtained using Pseudomonas mendocina (Fournier et al., 2006) (Fig. 5C). More such pathways & organisms are yet to be explored for better results in the near future.
Fig. 5.
(A) and (B) Microbial Transformation of ENA and DNMP (Molognoni et al., 2019). (C) Microbial Transformation of NDMA (Fournier et al., 2006)
A heterocyclic aromatic amine, can be converted to PhIP-M1 with the help of a gut microflora called E. hallii. The conversion occurs via glycerol metabolism pathway. The glycerol is metabolized in the gut to 3-hydroxypropionaldehyde (3-HPA) with the help of organisms like Lactobacillus reuteri and Enterococcus species. The intermediate 3-HPA then react with PhIP to form PhIP-M1. The pathway of bioconversion requires glycerol/diol dehydratase (GDH) (Fekry et al., 2016). Zhang et al., (2019a, 2019b) elaborated the pathway and reported the action of two enzymes for conversion of PhIP to PhIP-M1. They reported that PhIP is found in the intestine in the form of PhIP-G (PhIP-glucuronide) which is metabolized to PhIP with the help of an enzyme beta-glucuronidase (B-GUS), secreted by Faecalibacterium prausnitzii. PhIP is then converted to PhIP-M1 after reacting with 3-HPA, which is formed from glycerol with the help of another enzyme glycerol/diol dehydratase (GDH), secreted by E. hallii, Flavonifractor plautii, Blautia obeum, and Lactobacillus reuteri. It is the association of B-GUS and GDH producing microbiome that helps in bioconversion of a potential mutagen PhIP. Beer et al. (2019) provided a new dimension to the already known metabolism of glycerol pathway for degradation of PhIP. They reported the degradation of various heterocyclic aromatic amines like AαC, Trp-P-1, harman, norharman, PhIP, MeIQx, and MeIQ with the involvement of reuterin. When glycerol metabolized in the intestine, with the help of gut microorganisms, it forms 3-HPA, acrolein, and reuterin. Reuterin reacts with these compounds and leads to their bioconversion. This process involves dehydration, reduction, oxidation, microbial reduction, and microbial hydroxylation. A brief summary of common mutagens and their degradation pathway has been presented in tabulated form (Table 3).
Table 3.
Details about the pathway of bioconversion of some important mutagens of food
| Mutagens | Pathway | End-product | Microorganism | References |
|---|---|---|---|---|
| PhIP | Metabolism of glycerol to 3-HPA | PhIP-M1 | Eubacterium hallii | Fekry et al. (2016) |
| DNMP | Nitroreductase activity | NMAP | Lactobacillus bulgaricus LB-UFSC 001 | Molognoni et al. (2019) |
| PhIP-G | Metabolism of glycerol to acrolein | PhIP-M1 | Faecalibacterium prausnitzii, E. hallii, Flavonifractor plautii, Blautia obeum, and Lactobacillus reuteri | Zhang et al. (2019a) |
| AαC, Trp-P-1, harman, norharman, PhIP, MeIQx, and MeIQ | Metabolism of glycerol to reuterin | PhIP-M1,4–5, AαC-M6-11,38, MeIQx-M1-8, | Lactobacillus reuteri | Beer et al. (2019) |
| MeIQx | Metabolism of glycerol to acrolein | MeIQx-M1 | Gut microbiota | Zhang et al. (2019b) |
| DNMP | Oxygen insensitive nitroreductase type I B | NMAP | Staphylococcus xylosus LYOCARNI SXH-01, Lactobacillus fermentum LB-UFSC 0017, and Lactobacillus casei LB-UFSC 0019 | Motta et al. (2020) |
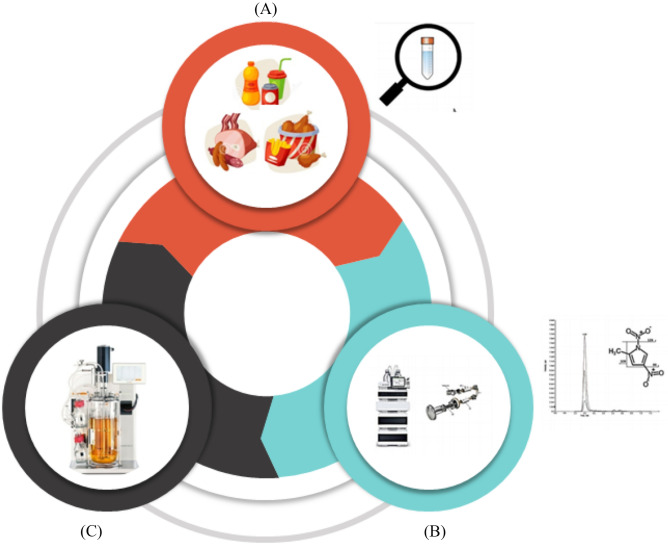
By assessment of various food products, it was evident that several chemicals such as N-nitroso, polycyclic aromatic hydrocarbons, heterocyclic aromatic amines, phenolic compounds, flavonoids, food additives (ENA and DNMP), quinines and their derivatives can enter into food products either intentionally or unintentionally and can cause great harm to our metabolic system. Still, many such chemicals are present in the food products whose concentration is too low for detection or quantification. These mutagens are mainly formed at high temperatures and acidic conditions. These mutagens can be extracted and analyzed by employing analytical techniques such as HPLC, LC–MS, and GC–MS. Presence of these chemicals in the food products has become a potent threat to our lives, hence the use of these chemicals should be restricted or monitored to a certain amount that makes them ineffective or tolerant. Apart from ENA and DNMP, other mutagens should also be degraded in order to make the food products safer to edible. To overcome this problem, choice of food and cooking or processing conditions are to be understood and modified wherever required. Hence, identification of such mutagenic chemicals along with their source is necessary because repeated consumption of such food products can increase the chance of mutagenicity. Additionally, metabolic pathways involved in the conversion of mutagens to non-mutagens are needed to be explored fully. As microorganisms were found to be a potent weapon to fight against various mutagens through biotransformation, therefore search for such microorganisms are required that have the ability to transform food mutagens into non mutagenic form. This can be a boon for mankind because life is dependent on food. Techniques for the identification of mutagens present if food products are also required to get better output. In this regard, mutagenic determination of food products and biotransformation using microorganisms by employing a bioreactor can be very useful. Bioreactors will provide controlled conditions which are required for proper growth of microorganisms and proper reactions to take place. Hence, application of bioreactor for the conversion of mutagenic to non-mutagenic form will become an important tool with respect to efficient conversion and in delivering promising results. Figure 6. presents the self-descriptive and comprehensive representation on degradation of mutagens by microorganisms with the application of bioreactor.
Fig. 6.
Mutagenic determination in processed food and conversion by microorganisms with the use of bioreactor. (A) Selection of processed food and sample preparation; (B) identification of mutagenic/ carcinogenic compound in the processed food; C selection of microorganism for biotransformation & its reactor studies
Acknowledgements
The corresponding author, Dr. Sarat Babu Imandi, acknowledges financial assistance from the Science for Equity Empowerment and Development (SEED) Division, Department of Science & Technology, Government of India, New Delhi under the S&T for Women Scheme (File No. SEED/WS/017/2015/G) for carrying out this study. The authors profusely thank Gandhi Institute of Technology and Management (GITAM) Deemed to be University for the facilities offered.
Abbreviations
- AαC
2-amino-9H-pyrido[2,3-b]indole
- ACH
Acetylcholine
- ACNO
Acetone oxide
- B-GUS
Beta-glucuronidase
- DCM
Dichloromethane
- DiMeIQx
(Combination of 2-amino-3,4,8-trimethylimidazo [4,5-f]quinoxaline (4,8-DiMeIQx) and 2-amino-3,7,8-trimethylimidazo[4,5-f]quinoxaline (7,8-DiMeIQx))
- DMSO
Dimethyl sulfoxide
- DNMP
2-methyl-1,4-dinitro pyrrole
- ENA
Ethylnitrolic acid
- g-force
Gravitational force
- GC–MS
Gas chromatography–mass spectrometry
- GDH
Glycerol/dioldehydratase
- HEPES
4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid
- HPLC
High-performance liquid chromatography
- HPA
2-hydroxypropyl acrylate
- IR
Infrared
- LC–MS
Liquid chromatography–mass spectrometry
- MAR
Mutagenic activity ratio
- MeIQ/MeIQx
2-amino-3,8-dimethylimidazo[4,5-f]-quinoxaline
- MNNG
N-methyl-N'-nitro-N-nitrosoguanidine
- NDBA
N-Nitrosodibutylamine
- NDEA
N-Nitrosodiethylamine
- NDMA
N-Nitrosodimethylamine
- NMAP
Nitrilotri(methyl phosphonic acid)
- NMOR
N-Nitrosomorpholine
- NMP
N-Methylpyrrolidone
- NMR
Nuclear magnetic resonance
- NMU
N-Nitroso-N-methylurea
- NPYR
N-Nitrosopyrrolidine
- NTDMA
N-Nitrodimethylamine
- PAHs
Polycyclic aromatic hydrocarbons
- PhIP
2-Amino-1-methyl-6-phenylimidazo [4,5-b]pyridine
- PRS
Propylsulfonic acid
- Trp-P-1
3-amino-1,4-dimethyl-5H-pyrido[4,3-b]indole
Author contributions
NKM, SM and TC carried out literature mining and drafted the manuscript. SKK and SBI extensively modified, edited, and reorganized the manuscript. All authors read and approved the final manuscript.
Funding
Science for Equity Empowerment and Development Division, SEED/WS/017/2015/G, Imandi Sarat Babu.
Declarations
Conflict of interest
Narendra Kishore Merugu, Saikumar Manapuram, Tanushree Chakraborty, Sita Kumari Karanam, Sarat Babu Imandi declares that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Narendra Kishore Merugu, Email: mnkbio@gmail.com.
Saikumar Manapuram, Email: sai.kumar@vipragen.com.
Tanushree Chakraborty, Email: contacttanushree@gmail.com.
Sita Kumari Karanam, Email: sitakaranam@gmail.com.
Sarat Babu Imandi, Email: simandi@gitam.edu.
References
- Alaejos MS, Afonso AM. Factors that affect the content of heterocyclic aromatic amines in foods. Comprehensive Reviews in Food Science and Food Safety. 2011;10:52–108. doi: 10.1111/j.1541-4337.2010.00141.x. [DOI] [Google Scholar]
- Ames BN, McCann J, Yamasaki E. Methods for detecting carcinogens and mutagens with the salmonella/mammalian-microsome mutagenicity test. Mutation Research. 1975;31:347–364. doi: 10.1016/0165-1161(75)90046-1. [DOI] [PubMed] [Google Scholar]
- Bartkiene E, Bartkevics V, Mozuriene E, Krungleviciute V, Novoslavskij A, Santini A, Rozentale I, Juodeikiene G, Cizeikiene D. The impact of lactic acid bacteria with antimicrobial properties on biodegradation of polycyclic aromatic hydrocarbons and biogenic amines in cold smoked pork sausages. Food Control. 2017;71:285–292. doi: 10.1016/j.foodcont.2016.07.010. [DOI] [Google Scholar]
- Barzegar F, Kamankesh M, Mohammadi A. Heterocyclic aromatic amines in cooked food: a review on formation, health risk-toxicology and their analytical techniques. Food Control. 2019;280:240–254. doi: 10.1016/j.foodchem.2018.12.058. [DOI] [PubMed] [Google Scholar]
- Basu AK. DNA damage, mutagenesis and cancer. International Journal of Molecular Sciences. 2018;19:970. doi: 10.3390/ijms19040970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beer F, Urbat F, Franz CM, Huch M, Kulling SE, Bunzel M, Bunzel D. The human fecal microbiota metabolizes foodborne heterocyclic aromatic amines by reuterin conjugation and further transformations. Molecular Nutrition & Food Research. 2019;63:1801177. doi: 10.1002/mnfr.201801177. [DOI] [PubMed] [Google Scholar]
- Binstok G, Campos C, Varela O, Gerschenson LN. Sorbate-nitrite reactions in meat products. Food Research International. 1998;31:581–585. doi: 10.1016/S0963-9969(99)00031-9. [DOI] [Google Scholar]
- Bjeldanes LF, Grose KR, Davis PH, Stuermer DH, Healy SK, Felton JS. An XAD-2 resin method for efficient extraction of mutagens from fried ground beef. Mutation Research 1982;105:43–49. doi: 10.1016/0165-7992(82)90206-8. [DOI] [PubMed] [Google Scholar]
- Bjeldanes LF, Morris MM, Felton JS, Healy S, Stuermer D, Berry P, Timourian H, Hatch FT. Mutagens from the cooking of food. II. Survey by Ames/Salmonella test of mutagen formation in the major protein-rich foods of the American diet. Food and Chemical Toxicology. 1982;20:357–363. doi: 10.1016/S0278-6915(82)80099-9. [DOI] [PubMed] [Google Scholar]
- Bjeldanes LF, Morris MM, Timourian H, Hatch FT. Effects of meat composition and cooking conditions on mutagen formation in fried ground beef. Journal of Agricultural and Food Chemistry. 1983;31:18–21. doi: 10.1021/jf00115a005. [DOI] [PubMed] [Google Scholar]
- Bode HB, Bethe B, Höfs R, Zeeck A. Big effects from small changes: possible ways to explore nature’s chemical diversity. ChemBioChem. 2002;3:619–627. doi: 10.1002/1439-7633(20020703)3:7<619::AID-CBIC619>3.0.CO;2-9. [DOI] [PubMed] [Google Scholar]
- Bogovski P, Bogovski S. Special report animal species in which n-nitroso compounds induce cancer. International Journal of Cancer. 1981;27:471–474. doi: 10.1002/ijc.2910270408. [DOI] [PubMed] [Google Scholar]
- Boobis AR, Lynch AM, Murray S, de la Torre R, Solans A, Farré M, Segura J, Gooderham NJ, Davies DS. CYP1A2-catalyzed conversion of dietary heterocyclic amines to their proximate carcinogens is their major route of metabolism in humans. Cancer Research. 1994;54:89–94. [PubMed] [Google Scholar]
- Bose A, Pande P, Jasti VP, Millsap AD, Hawkins EK, Rizzo CJ, Basu AK. DNA polymerases κ and ζ cooperatively perform mutagenic translesion synthesis of the C8–2'-deoxyguanosine adduct of the dietary mutagen IQ in human cells. Nucleic Acids Research. 2015;43:8340–8351. doi: 10.1093/nar/gkv750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cantwell M, Mittl B, Curtin J, Carroll R, Potischman N, Caporaso N, Sinha R. Relative validity of a food frequency questionnaire with a meat-cooking and heterocyclic amine module. Cancer Epidemiology, Biomarkers & Prevention. 2004;13:293–298. doi: 10.1158/1055-9965.EPI-270-2. [DOI] [PubMed] [Google Scholar]
- Clinton SK, Bostwick DG, Olson LM, Mangian HJ, Visek WJ. Effects of ammonium acetate and sodium cholate on N-methyl-N'-nitro-N-nitrosoguanidine-induced colon carcinogenesis of rats. Cancer Research. 1988;48:3035–3039. [PubMed] [Google Scholar]
- Commoner B, Vithayathil AJ, Dolara P, Nair S, Madyastha P, Cuca GC. Formation of mutagens in beef and beef extract during cooking. Science. 1978;201:913–916. doi: 10.1126/science.567374. [DOI] [PubMed] [Google Scholar]
- Crespi CL, Penman BW, Gelboin HV, Gonzalez FJ. A tobacco smoke-derived nitrosamine, 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone, is activated by multiple human cytochrome P450s including the polymorphic human cytochrome P4502D6. Carcinogenesis. 1991;12:1197–1201. doi: 10.1093/carcin/12.7.1197. [DOI] [PubMed] [Google Scholar]
- Cross AJ, Sinha R. Meat-related mutagens/carcinogens in the etiology of colorectal cancer. Environmental and Molecular Mutagenesis. 2004;44:44–55. doi: 10.1002/em.20030. [DOI] [PubMed] [Google Scholar]
- De Dea Lindner J. Characteristics and production of microbial cultures. In: Penna ALB, Nero LA, Todorov SD, editors. Fermented foods of Latin America: From traditional knowledge to innovative applications. Boca Raton, FL: CRC Press, Inc.; 2017. pp. 269–296. [Google Scholar]
- de Meester C. Bacterial mutagenicity of heterocyclic amines found in heat-processed food. Mutation Research. 1989;221:235–262. doi: 10.1016/0165-1110(89)90038-9. [DOI] [PubMed] [Google Scholar]
- Delfino RJ, Sinha R, Smith C, West J, White E, Lin HJ, Liao S-Y, Gim JSY, Ma HL, Butler J, Anton-Culver H. Breast cancer, heterocyclic aromatic amines from meat and N-acetyltransferase 2 genotype. Carcinogenesis. 2000;21:607–615. doi: 10.1093/carcin/21.4.607. [DOI] [PubMed] [Google Scholar]
- Diallo A, Deschasaux M, Latino-Martel P, Hercberg S, Galan P, Fassier P, Allès B, Guéraud F, Pierre FH, Touvier M. Red and processed meat intake and cancer risk: results from the prospective NutriNet-Santé cohort study. International Journal of Cancer. 2018;142:230–237. doi: 10.1002/ijc.31046. [DOI] [PubMed] [Google Scholar]
- Dong H, Xian Y, Li H, Bai W, Zeng X. Potential carcinogenic heterocyclic aromatic amines (HAAs) in foodstuffs: formation, extraction, analytical methods, and mitigation strategies. Comprehensive Reviews in Food Science and Food Safety. 2020;19:365–404. doi: 10.1111/1541-4337.12527. [DOI] [PubMed] [Google Scholar]
- Fekry MI, Engels C, Zhang J, Schwab C, Lacroix C, Sturla SJ, Chassard C. The strict anaerobic gut microbe Eubacterium hallii transforms the carcinogenic dietary heterocyclic amine 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine (PhIP) Environmental Microbiology Reports. 2016;8:201–209. doi: 10.1111/1758-2229.12369. [DOI] [PubMed] [Google Scholar]
- Felton JS, Knize MG, Roper M, Fultz E, Shen NH, Turteltaub KW. Chemical analysis, prevention, and low-level dosimetry of heterocyclic amines from cooked food. Cancer Research. 1992;52:2103s–2107s. [PubMed] [Google Scholar]
- Fiolet T, Srour B, Sellem L, Kesse-Guyot E, Allès B, Méjean C, Deschasaux M, Fassier P, Latino-Martel P, Beslay M, Hercberg S, Lavalette C, Monteiro CA, Julia C, Touvier M. Consumption of ultra-processed foods and cancer risk: results from NutriNet-Santé prospective cohort. British Medical Journal. 2018;360:k322. doi: 10.1136/bmj.k322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fournier D, Hawari J, Streger SH, McClay K, Hatzinger PB. Biotransformation of N-Nitrosodimethylamine by Pseudomonas mendocina KR1. Applied and Environmental Microbiology. 2006;72:6693–6698. doi: 10.1128/AEM.01535-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gangolli SD, van den Brandt PA, Feron VJ, Janzowsky C, Koeman JH, Speijers GJA, Spiegelhalder B, Walker R, Wishnok JS. Nitrate, nitrite and N-nitroso compounds. European Journal of Pharmacology: Environmental Toxicology and Pharmacology. 1994;292:1–38. doi: 10.1016/0926-6917(94)90022-1. [DOI] [PubMed] [Google Scholar]
- Gibis M. Heterocyclic aromatic amines in cooked meat products: causes, formation, occurrence, and risk assessment. Comprehensive Reviews in Food Science and Food Safety. 2016;15:269–302. doi: 10.1111/1541-4337.12186. [DOI] [PubMed] [Google Scholar]
- Göker G, Kıralan S, Tekin A, Erdoğdu F. Formation kinetics of polycyclic aromatic hydrocarbons (PAHs) during drying process of olive pomace. Food Chemistry. 2021;345:128856. doi: 10.1016/j.foodchem.2020.128856. [DOI] [PubMed] [Google Scholar]
- Hamidi EN, Hajeb P, Selamat J, Abdull Razis AF. Polycyclic aromatic hydrocarbons (PAHs) and their bioaccessibility in meat: a tool for assessing human cancer risk. Asian Pacific Journal of Cancer Prevention. 2016;17:15–23. doi: 10.7314/APJCP.2016.17.1.15. [DOI] [PubMed] [Google Scholar]
- Hansen TJ, Tannenbaum SR, Archer MC. Identification of nonenylnitrolic acid in corn treated with nitrous acid. Journal of Agricultural and Food Chemistry. 1981;29:1008–1011. doi: 10.1021/jf00107a031. [DOI] [PubMed] [Google Scholar]
- Hartman PE. Review: Putative mutagens and carcinogens in foods. II: Sorbate and sorbate-nitrite interactions. Environmental Mutagenesis. 1983;5:217–222. doi: 10.1002/em.2860050209. [DOI] [PubMed] [Google Scholar]
- Haskard C, Binnion C, Ahokas J. Factors affecting the sequestration of aflatoxin by Lactobacillusrhamnosus strain GG. Chemico-Biological Interactions. 2000;128:39–49. doi: 10.1016/S0009-2797(00)00186-1. [DOI] [PubMed] [Google Scholar]
- Hegazy MEF, Mohamed TA, ElShamy AI, Mohamed AEHH, Mahalel UA, Reda EH, Shaheen AM, Tawfik WA, Shahat AA, Shams KA, Abdel-Azim NS, Hammouda FM. Microbial biotransformation as a tool for drug development based on natural products from mevalonic acid pathway: a review. Journal of Advanced Research. 2015;6:17–33. doi: 10.1016/j.jare.2014.11.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herraiz T, Galisteo J. Nitrosative deamination of 2′-deoxyguanosine and DNA by nitrite, and antinitrosating activity of β-carboline alkaloids and antioxidants. Food and Chemical Toxicology. 2018;112:282–289. doi: 10.1016/j.fct.2017.12.042. [DOI] [PubMed] [Google Scholar]
- Herrmann SS, Granby K, Duedahl-Olesen L. Formation and mitigation of N-nitrosamines in nitrite preserved cooked sausages. Food Chemistry. 2015;174:516–526. doi: 10.1016/j.foodchem.2014.11.101. [DOI] [PubMed] [Google Scholar]
- Hosaka S, Matsushima T, Hirono I, Sugimura T. Carcinogenic activity of 3-amino-1-methyl-5H-pyrido [4,3-b]indole (Trp-P-2), a pyrolysis product of tryptophan. Cancer Letters. 1981;13:23–28. doi: 10.1016/0304-3835(81)90082-3. [DOI] [PubMed] [Google Scholar]
- IARC. Red Meat and Processed Meat. Volume 114, Published by the International Agency for Research on Cancer, France (2015)
- Ingenbleek L, Veyrand B, Adegboye A, Hossou SE, Koné AZ, Oyedele AD, Kisito CSKJ, Dembélé YK, Eyangoh S, Verger P, Leblanc JC, Durand S, Venisseau A, Marchand P, Bizec BL. Polycyclic aromatic hydrocarbons in foods from the first regional total diet study in Sub-Saharan Africa: contamination profile and occurrence data. Food Control. 2019;103:133–144. doi: 10.1016/j.foodcont.2019.04.006. [DOI] [Google Scholar]
- Ito N, Hasegawa R, Imaida K, Tamano S, Hagiwara A, Hirose M, Shirai T. Carcinogenicity of 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine (PhIP) in the rat. Mutation Research. 1997;376:107–114. doi: 10.1016/S0027-5107(97)00032-8. [DOI] [PubMed] [Google Scholar]
- Iwaoka WT, Krone CA, Sullivan JJ, Meaker EH, Johnson CA, Miyasato LS. A source of error in mutagen testing of foods. Cancer Letters. 1981;11:225–230. doi: 10.1016/0304-3835(81)90112-9. [DOI] [PubMed] [Google Scholar]
- Jägerstad M, Reuterswärd AL, Öste R, Dahlqvist A, Grivas S, Olsson K, Nyhammar T. Creatinine and Maillard reaction products as precursors of mutagenic compounds formed in fried beef. In: Waller GR, Feather MS, editors. The maillard reaction in foods and nutrition. Washington, DC: American Chemical Society; 1983. pp. 507–519. [Google Scholar]
- Jägerstad M, Skog K, Grivas S, Olsson K. Formation of heterocyclic amines using model systems. Mutation Research. 1991;259:219–233. doi: 10.1016/0165-1218(91)90119-7. [DOI] [PubMed] [Google Scholar]
- Kasai H, Yamaizumi Z, Wakabayashi K, Nagao M, Sugimura T, Yokoyama S, Miyazawa T, Spingarn NE, Weisburger JH, Nishimura S. Potent novel mutagens produced by broiling fish under normal conditions. Proceedings of the Japan Academy, Series B. 1980;56:278–283. doi: 10.2183/pjab.56.278. [DOI] [Google Scholar]
- Kazerouni N, Sinha R, Hsu C-H, Greenberg A, Rothman N. Analysis of 200 food items for benzo[a]pyrene and estimation of its intake in an epidemiologic study. Food and Chemical Toxicology. 2001;39:423–436. doi: 10.1016/S0278-6915(00)00158-7. [DOI] [PubMed] [Google Scholar]
- Khan MR, Bertus LM, Busquets R, Puignou L. Mutagenic heterocyclic amine content in thermally processed offal products. Food Chemistry. 2009;112:838–843. doi: 10.1016/j.foodchem.2008.06.045. [DOI] [Google Scholar]
- Kim SR, Kim K, Lee SA, Kwon SO, Lee JK, Keum N, Park SM. Effect of red, processed, and white meat consumption on the risk of gastric cancer: an overall and dose-response meta-analysis. Nutrients. 2019;11:826. doi: 10.3390/nu11040826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim H-J, Cho J, Jang A. Effect of charcoal type on the formation of polycyclic aromatic hydrocarbons in grilled meats. Food Chemistry. 2021;343:128453. doi: 10.1016/j.foodchem.2020.128453. [DOI] [PubMed] [Google Scholar]
- Kito Y, Namiki M, Tsuji K. A new n-nitropyrrole: 1,4-Dinitro-2-methylpyrrole, formed by the reaction of sorbic acid with sodium nitrite. Tetrahedron. 1978;34:505–508. doi: 10.1016/0040-4020(78)80043-X. [DOI] [Google Scholar]
- Knize MG, Roper M, Shen NH, Felton JS. Proposed Structures for an amino-dimethylimidazofuropyridine mutagen in cooked meats. Carcinogenesis. 1990;11:2259–2262. doi: 10.1093/carcin/11.12.2259. [DOI] [PubMed] [Google Scholar]
- Kramers PGN. The mutagenicity of saccharin. Mutation Research. 1975;32:81–92. doi: 10.1016/0165-1110(75)90012-3. [DOI] [PubMed] [Google Scholar]
- Krone CA, Iwaoka WT. Mutagen formation in processed foods. In: Finley JW, Schwass DE, editors. Xenobiotics in foods and feeds. Washington, DC: American Chemical Society; 1983. pp. 117–127. [Google Scholar]
- Krone CA, Yeh SM, Iwaoka WT. Mutagen formation during commercial processing of foods. Environmental Health Perspectives. 1986;67:75–88. doi: 10.1289/ehp.866775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liao GZ, Wang GY, Xu XL, Zhou GH. Effect of cooking methods on the formation of heterocyclic aromatic amines in chicken and duck breast. Meat Science. 2010;85:149–154. doi: 10.1016/j.meatsci.2009.12.018. [DOI] [PubMed] [Google Scholar]
- Lijinsky W, Shubik P. Benzo(a)pyrene and other polynuclear hydrocarbons in charcoal-broiled meat. Science. 1964;145:53–55. doi: 10.1126/science.145.3627.53. [DOI] [PubMed] [Google Scholar]
- Lijinsky W, Keefer L, Loo J. The preparation and properties of some nitrosamino acids. Tetrahedron. 1970;26:5137–5153. doi: 10.1016/S0040-4020(01)93166-7. [DOI] [PubMed] [Google Scholar]
- Lilly MD. Eighth P. V. Danckwerts memorial lecture presented at Glaziers' Hall, London, U.K. 13 May 1993: Advances in biotransformation processes. Chemical Engineering Science. 49: 151-159 (1994)
- Lynch AM, Knize MG, Boobis AR, Gooderham NJ, Davies DS, Murray S. Intra- and interindividual variability in systemic exposure in humans to 2-amino-3,8-dimethylimidazo[4,5-f]quinoxaline and 2-amino-1-methyl- 6-phenylimidazo[4,5-b]pyridine, carcinogens present in cooked beef. Cancer Research. 1992;52:6216–6223. [PubMed] [Google Scholar]
- Maron DM, Ames BN. Revised methods for the Salmonella mutagenicity test. Mutation Research. 1983;113:173–215. doi: 10.1016/0165-1161(83)90010-9. [DOI] [PubMed] [Google Scholar]
- Matsukura N, Kawachi T, Morino K, Ohgaki H, Sugimura T, Takayama S. Carcinogenicity in mice of mutagenic compounds from a tryptophan pyrolyzate. Science. 1981;213:346–347. doi: 10.1126/science.7244619. [DOI] [PubMed] [Google Scholar]
- Meurillon M, Engel E. Mitigation strategies to reduce the impact of heterocyclic aromatic amines in proteinaceous foods. Trends in Food Science & Technology. 2016;50:70–84. doi: 10.1016/j.tifs.2016.01.007. [DOI] [Google Scholar]
- Milić BLJ, Djilas SM, Čanadanović-Brunet JM. Synthesis of some heterocyclic aminoimidazoazarenes. Food Chemistry. 1993;46:273–276. doi: 10.1016/0308-8146(93)90118-Y. [DOI] [Google Scholar]
- Miller EC, Miller JA. Carcinogens and mutagens that may occur in foods. Cancer. 1986;58:1795–1803. doi: 10.1002/1097-0142(19861015)58:8+<1795::AID-CNCR2820581403>3.0.CO;2-H. [DOI] [PubMed] [Google Scholar]
- Mirvish SS. Role of N-nitroso compounds (NOC) and N-nitrosation in etiology of gastric, esophageal, nasopharyngeal and bladder cancer and contribution to cancer of known exposures to NOC. Cancer Letters. 1995;93:17–48. doi: 10.1016/0304-3835(95)03786-V. [DOI] [PubMed] [Google Scholar]
- Molognoni L, Motta GE, Daguer H, Lindner JDD. Microbial biotransformation of N-nitro-, C-nitro-, and C-nitrous-type mutagens by Lactobacillus delbrueckii subsp. bulgaricus in meat products. Food and Chemical Toxicology. 2019;136:110964. doi: 10.1016/j.fct.2019.110964. [DOI] [PubMed] [Google Scholar]
- Mortelmans K, Zeiger E. The Ames Salmonella/microsome mutagenicity assay. Mutation Research. 2000;455:29–60. doi: 10.1016/S0027-5107(00)00064-6. [DOI] [PubMed] [Google Scholar]
- Motta GE, Molognoni L, Daguer H, Angonese M, da Silva Correa Lemos AL, Dafre AL, De Dea Lindner J. The potential of bacterial cultures to degrade the mutagen 2-methyl-1,4-dinitro-pyrrole in a processed meat model. Food Research International. 2020;136:109441. doi: 10.1016/j.foodres.2020.109441. [DOI] [PubMed] [Google Scholar]
- Nagao M, Honda M, Seino Y, Yahagi T, Sugimura T. Mutagenicities of smoke condensates and the charred surface of fish and meat. Cancer Letters. 1977;2:221–226. doi: 10.1016/S0304-3835(77)80025-6. [DOI] [PubMed] [Google Scholar]
- Namiki M, Kada T. Formation of ethylnitrolic acid by the reaction of sorbic acid with sodium nitrite. Agricultural and Biological Chemistry. 1975;39:1335–1336. [Google Scholar]
- Namiki M, Udaka S, Osawa T, Tsuji K, Kada T. Formation of mutagens by sorbic acid-nitrite reaction: effects of reaction conditions on biological activities. Mutation Research. 1980;73:21–28. doi: 10.1016/0027-5107(80)90132-3. [DOI] [PubMed] [Google Scholar]
- Namiki M, Osawa T, Ishibashi H, Namiki K, Tsuji K. Chemical aspects of mutagen formation by sorbic acid-sodium nitrite reaction. Journal of Agricultural and Food Chemistry. 1981;29:407–411. doi: 10.1021/jf00104a046. [DOI] [PubMed] [Google Scholar]
- Nerín C, Aznar M, Carrizo D. Food contamination during food process. Trends in Food Science & Technology. 2016;48:63–68. doi: 10.1016/j.tifs.2015.12.004. [DOI] [Google Scholar]
- Ochiai M, Wakabayashi K, Nagao M, Sugimura T. Tyramine is a major mutagen precursor in soy sauce, being convertible to a mutagen by nitrite. Gan. 1984;75:1–3. [PubMed] [Google Scholar]
- OECD. Test No. 471: Bacterial Reverse Mutation Test, OECD Guidelines for the Testing of Chemicals, Section 4, OECD Publishing, Paris. (2020)
- Ohgaki H, Negishi C, Wakabayashi K, Kusama K, Sato S, Sugimura T. Induction of sarcomas in rats by subcutaneous injection of dinitropyrenes. Carcinogenesis. 1984;5:583–585. doi: 10.1093/carcin/5.5.583. [DOI] [PubMed] [Google Scholar]
- Ohshima H, Bartsch H. Quantitative estimation of endogenous nitrosation in humans by monitoring N-nitrosoproline excreted in the urine. Cancer Research. 1981;41:3658–3662. [PubMed] [Google Scholar]
- Osawa T, Ishibashi H, Namiki M, Kada T, Tsuji K. Desmutagenic action of food components on mutagens formed by the sorbic acid/nitrite reaction. Agricultural and Biological Chemistry. 1986;50:1971–1977. [Google Scholar]
- Oz E. The impact of fat content and charcoal types on quality and the development of carcinogenic polycyclic aromatic hydrocarbons and heterocyclic aromatic amines formation of barbecued fish. International Journal of Food Science & Technology. 2021;56:954–964. doi: 10.1111/ijfs.14748. [DOI] [Google Scholar]
- Pariza MW, Ashoor SH, Chu FS, Lund DB. Effects of temperature and time on mutagen formation in pan-fried hamburger. Cancer Letters. 1979;7:63–69. doi: 10.1016/S0304-3835(79)80097-X. [DOI] [PubMed] [Google Scholar]
- Park JE, Seo JE, Lee JY, Kwon H. Distribution of Seven N-Nitrosamines in food. Toxicological Research. 2015;31:279–288. doi: 10.5487/TR.2015.31.3.279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parkinson A, Ogilvie BW, Buckley DB, Kazmi F, Czerwinski M, Parkinson O. Biotransformation of xenobiotics. In: Klaassen CD, editor. Casarett and Doull's toxicology: the basic science of poisons. New York: McGraw Hill; 2013. [Google Scholar]
- Pearson AM, Chen C, Gray JI, Aust SD. Mechanism(s) involved in meat mutagen formation and inhibition. Free Radical Biology and Medicine. 1992;13:161–167. doi: 10.1016/0891-5849(92)90078-U. [DOI] [PubMed] [Google Scholar]
- Pérez-Prior MT, Manso JA, Gómez-Bombarelli R, González-Pérez M, Céspedes IF, García-Santos MP, Calle E, Casado J. Solvent effects in the decomposition reaction of some products formed by the reaction of sorbic acid with sodium nitrite: 1,4-dinitro-2-methylpyrrole and ethylnitrolic acid. Journal of Physical Organic Chemistry. 2009;22:418–424. doi: 10.1002/poc.1477. [DOI] [Google Scholar]
- Pérez-Prior MT, Gómez-Bombarelli R, González-Pérez M, Manso JA, García-Santos MP, Calle E, Casado J. Reactivity of the mutagen 1,4-dinitro-2-methylpyrrole as an alkylating agent. The Journal of Organic Chemistry. 2010;75:1444–1449. doi: 10.1021/jo902329q. [DOI] [PubMed] [Google Scholar]
- Phillips DH. Polycyclic aromatic hydrocarbons in the diet. Mutation Research. 1999;443:139–147. doi: 10.1016/S1383-5742(99)00016-2. [DOI] [PubMed] [Google Scholar]
- Ray L, Bera D. Biotransformation in food processing. In: Bhattacharya S, editor. Conventional and advanced food processing technologies. Hoboken, NJ: Wiley; 2014. pp. 387–410. [Google Scholar]
- Rencüzogullari E, Ila HB, Kayraldiz A, Topaktaş M. Chromosome aberrations and sister chromatid exchanges in cultured human lymphocytes treated with sodium metabisulfite, a food preservative. Mutation Research. 2001;490:107–112. doi: 10.1016/S1383-5718(00)00142-X. [DOI] [PubMed] [Google Scholar]
- Robach MC, Sofos JN. Use of sorbates in meat products, fresh poultry and poultry products: a review. Journal of Food Protection. 1982;45:374–383. doi: 10.4315/0362-028X-45.4.374. [DOI] [PubMed] [Google Scholar]
- Saffhill R, Margison GP, O’Connor PJ. Mechanisms of carcinogenesis induced by alkylating agents. Biochimica Et Biophysica Acta (BBA) Reviews on Cancer. 1985;823:111–145. doi: 10.1016/0304-419X(85)90009-5. [DOI] [PubMed] [Google Scholar]
- Scanlan RA. Nitrosamines. In: Caballero B, editor. Encyclopedia of food sciences and nutrition. Cambridge, MA: Academic Press; 2003. pp. 4142–4147. [Google Scholar]
- Schut HAJ, Snyderwine EG. DNA adducts of heterocyclic amine food mutagens: implications for mutagenesis and carcinogenesis. Carcinogenesis. 1999;20:353–368. doi: 10.1093/carcin/20.3.353. [DOI] [PubMed] [Google Scholar]
- Shabbir MA, Raza A, Anjum FM, Khan MR, Suleria HAR. Effect of thermal treatment on meat proteins with special reference to heterocyclic aromatic amines (HAAs) Critical Reviews in Food Science and Nutrition. 2014;55:82–93. doi: 10.1080/10408398.2011.647122. [DOI] [PubMed] [Google Scholar]
- Shirai T, Tamano S, Sano M, Masui T, Hasegawa R, Ito N. Carcinogenicity of 2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine (PhIP) in rats: dose-response studies. Princess Takamatsu Symposia. 1995;23:232–239. [PubMed] [Google Scholar]
- Shu Y-Z, Kingston DGI, Van Tassell RL, Wilkins TD. Metabolism of 1,4-dinitro-2-methylpyrrole, a mutagen formed by a sorbic acid-nitrite reaction, by intestinal bacteria. Environmental and Molecular Mutagenesis. 1991;17:181–187. doi: 10.1002/em.2850170307. [DOI] [PubMed] [Google Scholar]
- Shukla SK, Mangwani N, Rao TS, Das S. 8 - Biofilm-Mediated bioremediation of polycyclic aromatic hydrocarbons. In: Das S, editor. Microbial biodegradation and bioremediation. Amsterdam: Elsevier; 2014. pp. 203–232. [Google Scholar]
- Sinha R, Kulldorff M, Chow WH, Denobile J, Rothman N. Dietary intake of heterocyclic amines, meat-derived mutagenic activity, and risk of colorectal adenomas. Cancer Epidemiology, Biomarkers & Prevention. 2001;10:559–562. [PubMed] [Google Scholar]
- Sinha R, Gustafson DR, Kulldorff M, Wen WQ, Cerhan JR, Zheng W. 2-Amino-1-methyl-6-phenylimidazo[4,5-b]pyridine, a carcinogen in high- temperature-cooked meat, and breast cancer risk. Journal of the National Cancer Institute. 2000;92:1352–1354. doi: 10.1093/jnci/92.16.1352. [DOI] [PubMed] [Google Scholar]
- Sinha R, Kulldorff M, Swanson CA, Curtin J, Brownson RC, Alavanja MC. Dietary heterocyclic amines and the risk of lung cancer among Missouri women. Cancer Research. 2000;60:3753–3756. [PubMed] [Google Scholar]
- Smitha MS, Singh S, Singh R. Microbial biotransformation: a process for chemical alterations. Journal of Bacteriology & Mycology: Open Access. 2017;4:47–51. [Google Scholar]
- Sofos JN. Improved cooking yields of meat batters formulated with potassium sorbate and reduced levels of NaCl. Journal of Food Science. 1985;50:1571–1575. doi: 10.1111/j.1365-2621.1985.tb10536.x. [DOI] [Google Scholar]
- Sofos JN, Busta FF, Allen CE. Botulism control by nitrite and sorbate in cured meats: a review. Journal of Food Protection. 1979;42:739–770. doi: 10.4315/0362-028X-42.9.739. [DOI] [PubMed] [Google Scholar]
- Stavric B, Lau BPY, Matula TI, Klassen R, Lewis D, Downie RH. Mutagenic heterocyclic aromatic amines (HAAs) in ‘processed food flavour’ samples. Food and Chemical Toxicology. 1997;35:185–197. doi: 10.1016/S0278-6915(96)00119-6. [DOI] [PubMed] [Google Scholar]
- Sugimura T. Past, present, and future of mutagens in cooked foods. Environmental Health Perspectives. 1986;67:5–10. doi: 10.1289/ehp.86675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sugimura T, Nagao M, Weisburger JH. Mutagenic factors in cooked foods. CRC Critical Reviews in Toxicology. 1979;6:189–209. doi: 10.3109/10408447909037483. [DOI] [PubMed] [Google Scholar]
- Takahashi M, Wakabayashi K, Nagao M, Yamamoto M, Masui T, Goto T, Kinae N, Tomita I, Sugimura T. Quantification of 2-amino-3-methylimidazo[4,5-f]quinoline (IQ) and 2-amino-3,8-dimethylimidazo[4,5-f]quinoxaline (MeIQx) in beef extracts by liquid chromatography with electrochemical detection (LCEC) Carcinogenesis. 1985;6:1195–1199. doi: 10.1093/carcin/6.8.1195. [DOI] [PubMed] [Google Scholar]
- Takayama S, Nakatsuru Y, Masuda M, Ohgaki H, Sato S, Sugimura T. Demonstration of carcinogenicity in F344 rats of 2-amino-3-methyl-imidazo[4,5-f]quinoline from broiled sardine, fried beef and beef extract. Gan. 1984;75:467–470. [PubMed] [Google Scholar]
- Takayama S, Ishikawa T, Nakajima H, Sato S. Lung carcinoma induction in Syrian golden hamsters by intratracheal instillation of 1,6-dinitropyrene. Japanese Journal of Cancer Research: Gann. 1985;76:457–461. [PubMed] [Google Scholar]
- Taylor RT, Fultz E, Knize M. Mutagen formation in a model beef boiling system III. Purification and identification of three heterocyclic amine mutagens-carcinogens. Journal of Environmental Science and Health Part a: Environmental Science and Engineering. 1985;20:135–148. [Google Scholar]
- Tikkanen LM, Sauri TM, Latva-Kala KJ. Screening of heat-processed finnish foods for the mutagens 2-amino-3,8-dimethylimidazo[4,5-f]-quinoxaline, 2-amino-3,4,8-trimethylimidazo[4,5-f]- quinoxaline and 2-amino-1-methyl-6-phenylimidazo[4,5-b] pyridine. Food and Chemical Toxicology. 1993;31:717–721. doi: 10.1016/0278-6915(93)90142-L. [DOI] [PubMed] [Google Scholar]
- Tsuda M, Negishi C, Makino R, Sato S, Yamaizumi Z, Hirayama T, Sugimura T. Use of nitrite and hypochlorite treatments in determination of the contributions of IQ-type and non-IQ-type heterocyclic amines to the mutagenicities in crude pyrolyzed materials. Mutation Research. 1985;147:335–341. doi: 10.1016/0165-1161(85)90002-0. [DOI] [PubMed] [Google Scholar]
- Turesky RJ. Metabolism and biodisposition of heterocyclic amines. Progress in Clinical and Biological Research. 1990;347:39–53. [PubMed] [Google Scholar]
- Turesky RJ, Lang NP, Butler MA, Teitel CH, Kadlubar FF. Metabolic activation of carcinogenic heterocyclic aromatic amines by human liver and colon. Carcinogenesis. 1991;12:1839–1845. doi: 10.1093/carcin/12.10.1839. [DOI] [PubMed] [Google Scholar]
- Wakabayashi K, Nagao M, Esumi H, Sugimura T. Food-derived mutagens and carcinogens. Cancer Research. 1992;52:2092s–2098s. [PubMed] [Google Scholar]
- Wang C, Xie Y, Wang H, Bai Y, Dai C, Li C, Xu X, Zhou G. Phenolic compounds in beer inhibit formation of polycyclic aromatic hydrocarbons from charcoal-grilled chicken wings. Food Chemistry. 2019;294:578–586. doi: 10.1016/j.foodchem.2019.05.094. [DOI] [PubMed] [Google Scholar]
- Wild D, Degen GH. Prostaglandin H synthase-dependent mutagenic activation of heterocyclic aromatic amines of the IQ-type. Carcinogenesis. 1987;8:541–545. doi: 10.1093/carcin/8.4.541. [DOI] [PubMed] [Google Scholar]
- Wójciak KM, Karwowska M, Dolatowski ZJ. Use of acid whey and mustard seed to replace nitrites during cooked sausage production. Meat Science. 2014;96:750–756. doi: 10.1016/j.meatsci.2013.09.002. [DOI] [PubMed] [Google Scholar]
- Yamamoto T, Tsuji K, Kosuge T, Okamoto T, Shudo K, Takeda K, Iitaka Y, Yamaguchi K, Seino Y, Yahagi T, Nagao M, Sugimura T. Isolation and structure determination of mutagenic substances in L-glutamic acid pyrolysate. Proceedings of the Japan Academy, Series B. 1978;54:248–250. doi: 10.2183/pjab.54.248. [DOI] [Google Scholar]
- Yoshida D, Okamoto H. Formation of mutagens by heating creatine or amino acids with addition or fatty acids. Agricultural and Biological Chemistry. 1980;44:3025–3027. [Google Scholar]
- Yoshida D, Matsumoto T, Yoshimura R, Matsuzaki T. Mutagenicity of amino-α-carbolines in pyrolysis products of soybean globulin. Biochemical and Biophysical Research Communications. 1978;83:915–920. doi: 10.1016/0006-291X(78)91482-1. [DOI] [PubMed] [Google Scholar]
- Zamora MC, Zaritzky NE. Potassium sorbate inhibition of microorganisms growing on refrigerated packaged beef. Journal of Food Science. 1987;52:257–262. doi: 10.1111/j.1365-2621.1987.tb06587.x. [DOI] [Google Scholar]
- Zeiger E. Mutagenicity of chemicals added to foods. Mutation Research. 1993;290:53–61. doi: 10.1016/0027-5107(93)90032-B. [DOI] [PubMed] [Google Scholar]
- Zhang J, Empl MT, Schneider M, Schröder B, Stadnicka-Michalak J, Breves G, Steinberg P, Sturla SJ. Gut microbial transformation of the dietary mutagen MeIQx may reduce exposure levels without altering intestinal transport. Toxicology in Vitro. 2019;59:238–245. doi: 10.1016/j.tiv.2019.04.004. [DOI] [PubMed] [Google Scholar]
- Zhang J, Lacroix C, Wortmann E, Ruscheweyh HJ, Sunagawa S, Sturla SJ, Schwab C. Gut microbial beta-glucuronidase and glycerol/diol dehydratase activity contribute to dietary heterocyclic amine biotransformation. BMC Microbiology. 2019;19:99. doi: 10.1186/s12866-019-1483-x. [DOI] [PMC free article] [PubMed] [Google Scholar]