Abstract
Background
Right ventricle dysfunction (RVD) at echocardiography predicts mortality in patients with acute pulmonary embolism (PE), but heterogeneous definitions of RVD have been used. We performed a meta-analysis to assess the role of different definitions of RVD and of individual parameters of RVD as predictors of death.
Methods
A systematic search for studies including patients with confirmed PE reporting on right ventricle (RV) assessment at echocardiography and death in the acute phase was performed. The primary study outcome was death in-hospital or at 30 days.
Results
RVD at echocardiography, regardless of its definition, was associated with increased risk of death (risk ratio 1.49, 95% CI 1.24–1.79, I2=64%) and PE-related death (risk ratio 3.77, 95% CI 1.61–8.80, I2=0%) in all-comers with PE, and with death in haemodynamically stable patients (risk ratio 1.52, 95% CI 1.15–2.00, I2=73%). The association with death was confirmed for RVD defined as the presence of at least one criterion or at least two criteria for RV overload. In all-comers with PE, increased RV/left ventricle (LV) ratio (risk ratio 1.61, 95% CI 1.90–2.39) and abnormal tricuspid annular plane systolic excursion (TAPSE) (risk ratio 2.29 CI 1.45–3.59) but not increased RV diameter were associated with death; in haemodynamically stable patients, neither RV/LV ratio (risk ratio 1.11, 95% CI 0.91–1.35) nor TAPSE (risk ratio 2.29, 95% CI 0.97–5.44) were significantly associated with death.
Conclusion
Echocardiography showing RVD is a useful tool for risk stratification in all-comers with acute PE and in haemodynamically stable patients. The prognostic value of individual parameters of RVD in haemodynamically stable patients remains controversial.
Short abstract
Echocardiography is a useful tool for risk stratification in all-comers with acute PE but standardisation is required https://bit.ly/3HR8rdw
Introduction
Pulmonary embolism (PE) is a life-threatening disease [1]. Death is related to prevalent comorbidities in a variable proportion of patients; however, in the acute phase, PE itself accounts for the majority of deaths [2]. Right ventricle (RV) failure plays a major role in mortality due to PE, and its assessment could be used to identify patients at increased risk for death [3, 4, 5]. Current international guidelines recognise an essential role of RV dysfunction (RVD) at echocardiography in the risk stratification process of patients with acute PE [6, 7]. Risk stratification is crucial for decision making in patients with acute PE concerning disposition, treatment and early discharge [8].
However, the definition of RVD at echocardiography differed across studies in patients with acute PE, and this may have influenced study results. Several qualitative and quantitative findings of RVD at echocardiography have been reported in clinical studies and are used in daily clinical practice to diagnose RVD [9]. The most commonly reported parameters are RV hypokinesis, McConnell's sign, RV end-diastolic diameter, pulmonary artery pressure (PAP), tricuspid annular plane systolic excursion (TAPSE) and right ventricle/left ventricle (RV/LV) diameter ratio. Previous studies comparing different measures of RVD suggested that all of these were associated with an increased risk of death, but with different accuracy of the individual findings [10, 11].
This critical review and meta-analysis aimed at assessing the accuracy of different parameters of RVD at echocardiography for the prediction of short-term all-cause mortality, PE-related death and adverse outcome.
Methods
The study protocol for the meta-analysis was registered in PROSPERO (Prospero ID: CRD42021266948) and the findings were reported according to the PRISMA guidelines. Firstly, a critical review was conducted to assess the available evidence on RVD as combination of findings as well as on individual parameters of RV overload at echocardiography and short-term death. Quantitative meta-analyses were subsequently performed on the association between individual echocardiographic parameters and short-term death, whenever data were available.
MEDLINE and EMBASE databases were searched from inception to October 2021 (Supplementary Appendix 1).
Studies meeting the following criteria were eligible for inclusion in the critical review: 1) inclusion of patients with acute PE; 2) echocardiography performed in the early phase of PE; 3) availability of data on all-cause in-hospital death or death at 30 days; and 4) availability of data on in-hospital adverse outcome or at 30 days.
The following parameters at echocardiography were considered: RV diameter, RV/LV, RV hypokinesis, PAP, TAPSE and RVD as a comprehensive definition based on the presence of pre-specified numbers of criteria at echocardiography.
Studies were excluded in case of: 1) case reports; 2) case series <50 patients; 3) thrombus in transit studies; and 4) language other than English.
To be included in the meta-analysis, studies had to report on the number of patients dying/surviving among those with a specific finding at echocardiography. Study authors were contacted whenever data for meta-analysis could not be extrapolated from the text; as an alternative, study data available in previous meta-analyses were reported. In case provision of unpublished data was not possible, risk measurements (odds ratios (ORs), hazard ratios (HRs)) with 95% confidence interval (CI) were used for the meta-analyses, if available.
Study selection and data extraction
Three investigators (L.A. Cimini, M. Candeloro and M. Phywaczewska) independently reviewed the search list by title and abstract and determined study eligibility. Candidate records were then reviewed and selected for data retrieval. Two authors (L.A. Cimini and G. Maraziti) independently reviewed each study for inclusion in the critical review and in the meta-analysis and performed quality assessment and data extraction using standardised extraction forms. Disagreements were resolved through revision by an additional reviewer (M. Candeloro) and by discussion. Extracted data included information on study design (retrospective, prospective, date of publication), patient features (mean age, sex, severity of PE, etc.), definitions of RVD, prevalence of RVD, all-cause mortality, PE-related mortality and adverse outcome.
The Quality Assessment of Diagnostic Accuracy Studies-2 (QUADAS-2) risk of bias tool was used to assess the quality of included studies [12]. QUADAS-2 is the currently recommended tool to evaluate the risk of bias and applicability of primary diagnostic accuracy studies. QUADAS-2 consists of four key domains: patient selection, index test, reference standard, and flow and timing. Each is assessed in terms of risk of bias and the first three in terms of concerns regarding applicability.
Study outcome and measurements
The primary outcome of the meta-analysis was short-term death, defined as death occurring in-hospital or within 30 days since PE diagnosis. Secondary outcomes were: 1) short-term PE-related death; and 2) short-term adverse outcome, according to the definition used in the individual studies.
For each RV parameter at echocardiography, the following measurements were collected: number of patients with/without study outcome event based on presence/absence of the parameter; risk measurements (OR, HR) with 95% CI.
Statistical analysis
For each echocardiographic parameter, sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), positive likelihood ratio, negative likelihood ratio, prediction intervals and 95% CI were calculated [13, 14]. For the meta-analysis, different statistics were used based on the type of available data (see supplementary material) [15, 16, 17]. We planned separate analyses for: 1) all-cause short-term death; 2) PE-related death; and 3) short-term adverse outcome.
The appropriateness of pooling data across studies was assessed using the Cochran's Chi-squared test and the I2 test for heterogeneity [18]. I2 statistic over 50% identified significant heterogeneity. We examined potential sources of heterogeneity by conducting sensitivity analyses in the following categories: 1) studies including only patients with echocardiography available; 2) studies including >100 patients; 3) studies published after 2010; 4) number of criteria at echocardiography used to define RVD (e.g., at least one, at least two, etc.); 5) different echocardiographic cut-offs; 6) haemodynamically stable patients; and 7) prospective studies. The exclusion of outlier studies from the analysis was also used to address significant heterogeneity.
We also performed specific analyses for each echocardiographic parameter, whenever possible.
The statistical analyses, forest plots and publication bias analyses were performed using R version 4.0.5.
The study was performed without any external support. The authors had full access to and take full responsibility for the integrity of all the data. All authors have read and agreed to the manuscript as it is written.
Results
Critical review
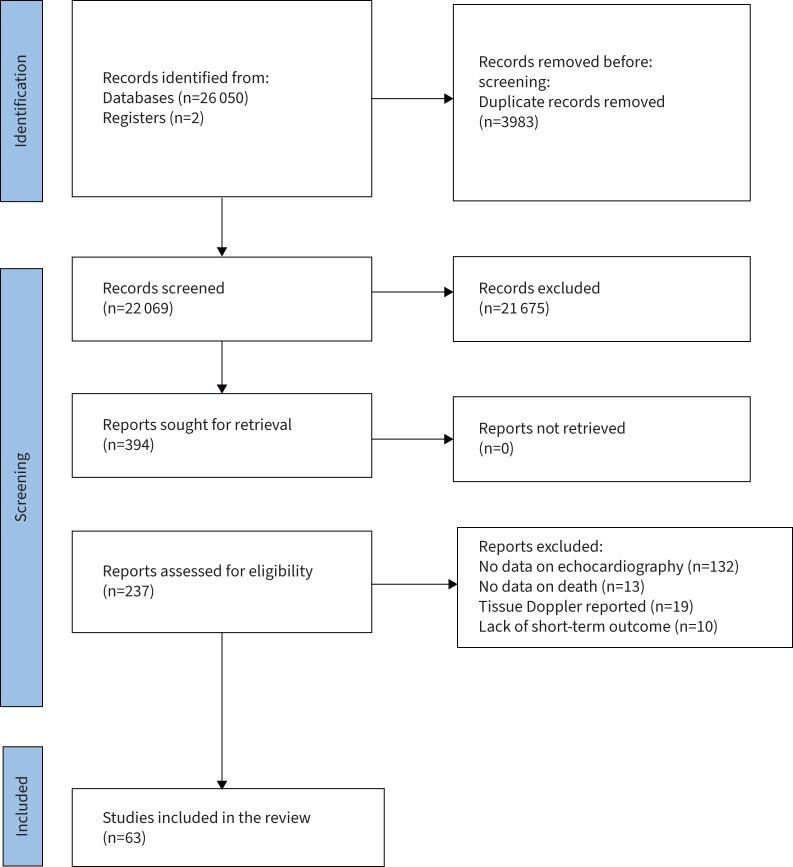
Our search identified 26 052 records. After removal of 21 675 papers that did not fulfil the inclusion criteria, 265 records were retrieved for full-text examination (figure 1). 63 studies were finally retained for the systematic review.
FIGURE 1.
Prisma diagram of the study.
We identified seven parameters at echocardiography for which data on mortality were reported in at least three studies: 1) RVD as a variable combination of parameters of RV overload; 2) RV/LV ratio; 3) TAPSE; 4) RV hypokinesis; 5) RV diameter; 6) PAP and 7) McConnell's sign (table 1).
TABLE 1.
Characteristics of studies according to different echocardiographic parameters
| Parameter at echocardiography | Studies | Overall patients | Retrospective | Single-centre | Found an association with mortality | Found an association with adverse outcome |
| RVD | 41 | 12 209 | 10 | 17 | NA | NA |
| Hypokinesis | 6 | 17 510 | 3 | 3 | 3 | 3 |
| McConnell's sign | 4 | 1078 | 2 | 4 | 3 | NA |
| PAP | 6 | 1104 | 3 | 6 | 6 | NA |
| RV diameter | 10 | 2091 | 4 | 5 | 7 | NA |
| RV/LV ratio | 16 | 6391 | 8 | 6 | 6 | 5 |
| TAPSE | 14 | 5697 | 7 | 8 | 9 | 1 |
NA: not applicable; RVD: right ventricular dysfunction as comprehensive definition; PAP: pulmonary artery pressure; RV/LV ratio: right ventricle/left ventricle diameter ratio; TAPSE: tricuspid annular plane systolic excursion.
Overall, 41 studies, all observational, reported on RVD at echocardiography and mortality (supplementary table S1) [19–59].
16 different definitions of RVD were used across these studies, and in particular the presence of a single criterion of RV overload was used in 16 studies, the presence of at least two criteria in eight studies and the presence of at least three parameters of RV overload were required in one study only. The most commonly used criteria of RV overload used for definition of RVD were: 1) RV dilatation; 2) RV/LV ratio; 3) paradox septal systolic motion; 4) pulmonary hypertension; 5) hypokinesis of the RV free wall; 6) tricuspid regurgitation systolic velocity over 2.6 m·s−1 from the apical or subcostal four-chamber view; 7) absence of the inspiratory collapse of the inferior vena cava; 8) McConnell's sign; and 9) TAPSE. The majority of the studies (58% of examined studies) described an association between RVD and short-term death. A meta-analysis on the association between RVD at echocardiography and clinical outcome is reported below.
The role of RV/LV ratio as predictor of clinical course in patients with acute PE was reported in 16 studies [10, 11, 23, 24, 28, 45, 60–69] (supplementary table S2).
Three different cut-off values were used across the individual studies: 0.6, 0.9 and 1.0. Controversial results were reported across studies on the association between increased RV/LV ratio and the risk of death. Published and unpublished data were pooled in a meta-analysis to assess the role of RV/LV ratio as predictor of death, and the results are reported below.
14 observational studies assessed the role of TAPSE in the prediction of short-term death [10, 11, 45, 60, 62–65, 70–75] (supplementary table S3). A formal meta-analysis on the association between TAPSE and clinical outcome is reported below.
Among the studies reporting on TAPSE, two also described the role of TRPG/TAPSE (tricuspid regurgitation peak gradient/TAPSE) (890 patients, one retrospective, one prospective) in predicting all-cause short-term death. In both the studies, TRPG/TAPSE was associated with increased risk of all-cause short-term death. A meta-analysis was not possible for this parameter.
Six studies (17 510 patients), all observational, reported on the association between RV hypokinesis and clinical outcome [39, 60, 70, 76–78] (supplementary table S4). In all these studies, RV hypokinesis was associated with increased risk of death or adverse outcome. A meta-analysis of available data is reported below.
Four studies reported on the association between McConnell's sign and clinical outcome [10, 61, 62, 70] (supplementary table S5). All these studies, except for one, found a significant association between McConnell's sign and death.
Six observational studies (1104 patients; three prospective, three retrospectives) reported on the association between PAP and clinical outcome [48, 61, 63, 71, 79, 80] (supplementary table S6) and found an increased risk of short-term death in patients with increased PAPs; however, a meta-analysis on this parameter was not possible.
10 studies (2091 patients) assessed the association between RV diameter and clinical course in patients with acute PE [10, 11, 20, 26, 45, 48, 61, 62, 79, 80] (supplementary table S7). A meta-analysis on the prognostic role of RV diameter is reported below.
Of all included studies, four reported long-term outcomes (605 patients): two studies assessed the association between RV/LV and long-term course of PE patients, two RVD as a combination of findings and long-term course [41, 41, 61, 63]. RV/LV >1 was associated with an increased risk of long-term death (two studies, 331 patients) [61, 62]. In one study, RVD was not associated with an increased risk of long-term death [40]. However, the heterogeneity on data reporting did not allow a formal meta-analysis on the long-term value of RVD.
Quality assessment and risk of bias
Results of QUADAS-2 assessments are reported in supplementary tables S8 and S9. Several studies were judged to be at low or uncertain risk of bias. The main reasons were un-blinding of echocardiography with respect to clinical status and other risk stratification strategies, and extrapolation of PE patients with echocardiography performed from PE populations for whom echocardiography was not mandatory; the latter could have led to selection bias. Furthermore, the timing of echocardiography was not uniformly defined.
Meta-analysis: RVD and short-term death, PE-related death or adverse outcome
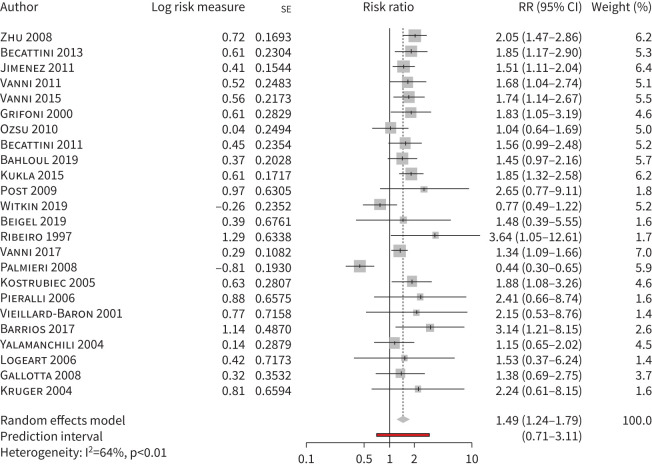
Out of 41 studies reporting on RVD and clinical outcome, one was excluded from the meta-analysis for lack of data [57]. Of the remaining studies, 24 (8121 patients) had data available on short-term death (in four studies after direct contact with the author) and were included in a meta-analysis [19–30, 44, 46–56, 59]. RVD was associated with increased risk of short-term death in all-comers with PE, with the presence of significant heterogeneity (risk ratio 1.49, 95% CI 1.24–1.79, I2=64%) (figure 2, table 2). Heterogeneity decreased after excluding studies with <100 patients or studies published beyond 2010 (table 3); heterogeneity also decreased after excluding the study by Palmieri et al. (23 studies, 8032 patients, risk ratio 1.56, 95% CI 1.38–1.77, I2=14%) [49]. In this single-centre study, rates of death and of haemodynamic deterioration were particularly high (13.3 and 37%, respectively) and RVD was not associated with short-term death. Among the 24 studies, RVD was associated with short-term death in studies defining RVD as the presence of at least one criterion of RV overload and in studies requiring at least two criteria but not in the single study requiring at least three criteria (one study, 89 patients, risk ratio 0.44, 95% CI 0.30–0.65) (table 3, supplementary figure S1A). When the analysis focused on studies only including haemodynamically stable patients, RVD was associated with short-term death (15 studies, 5453 patients, risk ratio 1.52, 95% CI 1.15–2.00, I2=73%) (table 4, supplementary figure S1B).
FIGURE 2.
Short-term all-cause mortality in pulmonary embolism patients with versus without right ventricle dysfunction defined as a combination of findings at echocardiography. RR: risk ratio.
TABLE 2.
Meta-analyses on the role of RVD or individual findings at echocardiography in predicting all-cause death in all-comers with acute pulmonary embolism
| Parameter at echocardiography | Studies/patients n | Risk ratio (95% CI) |
| RVD | 24/8121 | 1.49 (1.24–1.79) |
| RV diameter | 5/944 | 0.58# (0.05–1.21) |
| RV/LV | 12/4019 | 1.61 (1.09–2.39) |
| TAPSE | 11/4095 | 2.29 (1.45–3.59) |
| Hypokinesis¶ | 6/17 510 | 1.60 (1.14–2.25) |
RVD: right ventricular dysfunction as comprehensive definition; CI: confidence interval; RV/LV ratio: right ventricle/left ventricle diameter ratio; TAPSE: tricuspid annular plane systolic excursion. #: standardised mean difference. ¶: the outcome of this analysis is short-term adverse outcome.
TABLE 3.
Sensitivity analyses
| Studies/patients n | Risk ratio (95% CI), I2 (%) |
Haemodynamically stable patients only
risk ratio (95% CI), I2 (%) |
|
| RVD | |||
| Number of criteria for RVD definition: | |||
|
≥1 echo criterion ≥2 echo criteria ≥3 echo criteria |
16/5488 7/2633 1/89 | 1.50 (1.28–1.87), 24 1.76 (1.46–2.12), 0 0.44 (0.30–0.65)# | 1.52 (1.15–2.00), 73 |
| Studies including >100 patients | 16/7502 | 1.59 (1.36–1.87), 36 | - |
| Studies published beyond 2010 | 12/6318 | 1.47 (1.24–1.74), 33 | - |
| RV/LV | |||
| Different cut-offs: | |||
| ≥1 | 9/2079 | 1.57 (0.93–2.65), 35 | 1.11 (0.91–1.35), 0 |
| >1 | 2/990 | 1.09 (0.91–1.35), 0 | |
| >0.6 | 1/950 | 3.47 (1.72–6.98)# | |
| TAPSE | |||
| Different cut-offs: | |||
| ≥15 mm | 3/828 | 3.23 (1.45–7.18), 0 | 2.29 (0.97–5.44), 71 |
| >16 mm | 6/1159 | 2.19 (0.80–6.03), 66 | |
| >20 mm | 1/2108 | 1.68 (0.78–3.64), 0 |
CI: confidence interval; RVD: right ventricular dysfunction as comprehensive definition; RV/LV ratio: right ventricle/left ventricle diameter ratio; TAPSE: tricuspid annular plane systolic excursion. #: the result is obtained for one study only, thus I2 is not calculated.
TABLE 4.
Accuracy of different echocardiographic parameters in predicting prognosis
| Echocardiographic parameter | Sensitivity | Specificity | PPV | NPV | Positive likelihood ratio | Negative likelihood ratio |
| RVD | 0.74 (0.6–0.9) | 0.51 (0.4–0.6) | 0.09 (0.06–0.12) | 0.97 (0.94–0.98) | 1.51 | 0.51 |
| RV/LV | 0.44 (0.3–0.5) | 0.64 (0.6–0.7) | 0.08 (0.05–0.12) | 0.64 (0.57–0.71) | 1.22 | 0.88 |
| TAPSE | 0.57 (0.5–0.6) | 0.65 (0.5–0.8) | 0.06 (0.04–0.1) | 0.65 (0.51–0.76) | 1.63 | 0.66 |
Data calculated from studies reporting number of patients with/without study outcome event based on presence/absence of the parameter. PPV: positive predictive value; NPV: negative predictive value; RVD: right ventricular dysfunction as comprehensive definition; RV/LV ratio: right ventricle/left ventricle diameter ratio; TAPSE: tricuspid annular plane systolic excursion.
Pooled sensitivity, specificity, PPV and NPV of RVD for short-term death are reported in table 4.
26 studies reported on the association between RVD and secondary outcomes and allowed specific meta-analyses (supplementary table S1) [19, 21, 25–28, 30–44, 49, 55, 56, 58]. Different definitions of adverse outcome were used across the included studies (supplementary table S7). RVD at echocardiography was associated with increased risk of PE-related death both in all-comers (11 studies, 3909 patients; 5.3% versus 2.1%, risk ratio 3.31, 95% CI 1.84–5.97, I2 = 0%) and in haemodynamically stable patients (eight studies, 2656 patients; 4.1% versus 2.2%, risk ratio 3.16, 95% CI 1.51–6.60) (supplementary figure S2A and SB), and of adverse outcome (20 studies, 5264 patients; risk ratio 1.68, 95% CI 1.40–2.02, I2=55%) (supplementary figure S3).
Meta-analyses on individual parameters of RV overload and clinical outcome
Among 16 studies reporting on RV/LV diameter ratio, four were excluded from the meta-analysis due to lack of data that was not resolved after contact with the authors; finally, 12 studies were included in the meta-analysis (supplementary table S2). Increased RV/LV diameter ratio was associated with increased risk of short-term death in all-comers (4019 patients; 8.3% versus 5.7%, risk ratio 1.61, 95% CI 1.09–2.39, I2=46%) (supplementary figure S4, supplementary table S2). Sensitivity analyses were performed in studies including haemodynamically stable patients (table 3, supplementary figure S5A) and by grouping for different cut-offs (table 3, supplementary figure S5B). Pooled sensitivity, specificity, PPV and NPV of RV/LV diameter ratio for short-term death are reported in table 4.
Overall, 14 studies reported on the prognostic role of TAPSE. After exclusion of four studies due to lack of data, 11 were included in the meta-analysis (supplementary table S3). The risk of short-term death was higher in all-comers with reduced versus normal TAPSE (11 studies, 4095 patients; 9.7% versus 5.2%, risk ratio 2.29, 95% CI 1.45–3.59, I2=49%) (table 2, supplementary figure S6). This result was consistent for different TAPSE cut-offs (>15 mm and ≤15 mm) (table 3, supplementary figure 7A). When the analysis was restricted to six studies including haemodynamically stable patients only, a marginally significant association between TAPSE and short-term death was observed, with significant heterogeneity (3240 patients; risk ratio 2.29, 95% CI 0.97–5.44, I2=71%) (table 3, supplementary figure S7B). In haemodynamically stable patients, TAPSE was not associated with PE-related death (OR 1.55, 95% CI 0.82–2.59, I2=90%) (supplementary figure S7C). Pooled sensitivity, specificity, PPV and NPV are reported in table 4.
Six studies reported on the association between RV hypokinesis at echocardiography and short-term outcomes and were included in the meta-analysis [39, 60, 70, 76–78]. A meta-analysis on death was not possible. Hypokinesis was associated with an increased risk of short-term adverse outcome with significant heterogeneity (17 510 patients, risk ratio 1.60, 95% CI 1.14–2.25, I2=79%) (table 2, supplementary figure S8). The study by Bikdeli et al. [76] excluded from the analysis over 50% of patients who did not undergo echocardiography. After exclusion of this study from the current analysis, heterogeneity was significantly reduced and the association was confirmed (risk ratio 1.42, 95% CI 1.0–2.02, I2=53%).
Overall, five studies reported on the association between mean RV diameter and clinical outcome and were included in the meta-analysis [10, 11, 48, 61, 62]. One study had adverse outcome and one had PE-related death as primary outcome [10, 11]. An increased RV diameter was not associated with increased risk of death or adverse outcome (five studies, 944 patients; standardised mean difference 0.58, 95% CI 0.05–1.21, I2=63%) (table 2, supplementary figure S9).
Discussion
Our study shows that RVD, defined as a combination of findings of RV overload at echocardiography, is a predictor of short-term death in all-comers with acute PE and in haemodynamically stable patients. Among individual parameters of RVD, increased RV/LV ratio and abnormal TAPSE but not RV diameter were associated with increased risk of short-term death in all-comers with PE. In haemodynamically stable patients, neither TAPSE nor increased RV/LV ratio showed a significant association with death. RV hypokinesis was associated with the risk for short-term adverse events in all-comers. In addition, the study shows relevant heterogeneity in the definition of RVD at echocardiography in studies in patients with acute PE. This leads to a high degree of heterogeneity in almost all the estimates on the prognostic value of RVD and claims for a standardisation in the definition of RVD in patients with acute PE.
PE is a life-threatening disease, with death mainly related to acute RV failure [33], and, consequently, the identification of RV overload is considered an essential step of risk stratification for early death [3]. Echocardiography has been consistently reported as a tool for identification of RV overload and risk stratification for short-term death. However, echocardiography is an operator-dependent technique and lacks standardisation for the definition of acute RVD. The guidelines of the European Society of Cardiology report that, due to the peculiar RV geometry, no individual echocardiographic parameter can provide fast and reliable information on RV size or function [6]. Our critical review shows that 16 different definitions of RVD were used across 41 studies that reported on the prognostic role of RVD in patients with acute PE. These definitions differed for the number and type of signs of RV overload that were required for the diagnosis of RVD. Indeed, nine parameters of RV overload were described at echocardiography. In this meta-analysis we found that RVD, defined as the presence of at least one sign of RV overload or as two or more signs, is associated with ∼50% increase in the risk of short-term death in patients with acute PE. This wide range of definitions of RVD could have caused the significant heterogeneity observed in the meta-analysis of RVD in all-comers and in haemodynamically stable patients with acute PE and the wide prediction interval for this parameter. Our findings call for a standardisation in the definition of RVD at echocardiography in patients with acute PE in order to use RVD efficiently for decision making concerning patient management. Whether standardisation should refer to the role of specific signs of RV overload or to implementation of technical accuracy (e.g., echocardiography with ECG trace) or to the skills of the operator (e.g., differential diagnosis between chronic and acute RV overload, accuracy of measurements) remains to be defined.
Our critical review shows a correlation with death for several individual findings of RV overload at echocardiography. We were able to run formal meta-analyses for four of these findings. An association with short-term death was confirmed in all-comers with acute PE for increased RV/LV ratio, abnormal TAPSE and RV hypokinesis but not RV diameter. The lack of association between RV diameter and death is not surprising as a combined assessment of the right and left ventricle is necessary to make a final judgement. Moreover, we did not observe a significant association with death when the single parameters were evaluated in haemodynamically stable patients. The absence of significant association could be due to underpower for TAPSE, while it seems questionable for RV/LV ratio. In a recent meta-analysis, increasing RV/LV was a predictor of all-cause short-term death in haemodynamically stable patients (three studies, meta-analysis of continuous values) [81]. In our study, RV/LV was not a predictor of death in haemodynamically stable patients (six studies, meta-analysis of proportions). It is conceivable that different study methodologies led to different results; this difference again reinforces the concept of the need for standardisation of echocardiographic assessment in patients with acute PE.
Taken together our results seem to support the statement of the European Society of Cardiology (ESC) guidelines on the need for more than one abnormality at echocardiography to predict prognosis accurately. Good-quality studies suggested that a clearly increased RV/LV (RV/LV >1) combined with TAPSE <16 mm could be used to identify a group of normotensive patients with acute PE at increased risk for death [45].
PPVs and NPVs were suboptimal for the use of echocardiography as a stand-alone test for decision making. Recently, a systematic review and meta-analysis showed that RVD can be detected by computed tomography (CT) imaging; the diagnostic accuracy of CT compared with echocardiography varies depending on specific findings [82]. The assessment of multiple CT findings of RVD seems to have improved diagnostic accuracy compared to stand-alone findings. This finding is in keeping with the results of our meta-analysis, showing that the assessment of combined parameters at echocardiography has improved prognostic value in comparison to stand-alone findings.
This information is crucial for clinicians dealing with patients with acute PE as these patients may have a wide range of causes of death, and not all of them are related to RV overload. In fact, echocardiography can detect acute RVD in ∼25% of all PE patients. In haemodynamically unstable patients, bedside echocardiography is the first imaging method for the initial assessment and to guide further management including life-saving therapies [6]. In haemodynamically stable patients with suspected PE, echocardiography provides important information regarding prognosis and differential diagnosis. However, clinical assessment and laboratory markers should be integrated with echocardiographic findings for risk stratification.
Our study has some limitations. First, pooling the results from different studies resulted in significant heterogeneity that was assessed through several methods and sensitivity analyses. Only few studies reported data on more than one finding of RV overload. As a consequence, results on different findings were often obtained from different combinations of studies. Overall, 35.8% of the studies included in our meta-analyses had an uncertain risk of bias, which was due to the lack of information on several domains. We excluded all studies dealing with Doppler tissue imaging in patients with acute PE. This technique is promising, but not commonly available in the urgent setting.
Our study also has some strengths. By including over 40 studies, this is one of the most comprehensive meta-analyses on the prognostic role of RVD at echocardiography in patients with acute PE. By providing several sensitivities analyses we were able to describe the specific prognostic value of different findings and of different cut-offs.
In conclusion, echocardiography is a useful tool for risk stratification in all-comers and in haemodynamically stable patients with acute PE, but standardisation is required. The prognostic value of individual parameters of RVD in haemodynamically stable patients remains controversial indicating comprehensive assessment of RV function These results can inform future studies, guidelines and clinicians dealing with management of patients with acute PE.
Supplementary material
Please note: supplementary material is not edited by the Editorial Office, and is uploaded as it has been supplied by the author.
Supplementary material 00641-2022.SUPPLEMENT (1.3MB, pdf)
Footnotes
Provenance: Submitted article, peer reviewed.
This study is registered at https://www.crd.york.ac.uk/prospero/ with identifier number CRD42021266948.
Conflict of interest: None declared.
References
- 1.Barco S, Valerio L, Gallo A, et al. Global reporting of pulmonary embolism-related deaths in the World Health Organization mortality database: vital registration data from 123 countries. Res Pract Thromb Haemost 2021; 5: e12520. doi: 10.1002/rth2.12520 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Goldhaber SZ, Visani L, De Rosa M. Acute pulmonary embolism: clinical outcomes in the International Cooperative Pulmonary Embolism Registry (ICOPER). Lancet 1999; 353: 1386–1389. doi: 10.1016/S0140-6736(98)07534-5 [DOI] [PubMed] [Google Scholar]
- 3.Barco S, Mahmoudpour SH, Planquette B, et al. Prognostic value of right ventricular dysfunction or elevated cardiac biomarkers in patients with low-risk pulmonary embolism: a systematic review and meta-analysis. Eur Heart J 2019; 40: 902–910. doi: 10.1093/eurheartj/ehy873 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Becattini C, Maraziti G, Vinson DR, et al. Right ventricle assessment in patients with pulmonary embolism at low risk for death based on clinical models: an individual patient data meta-analysis. Eur Heart J 2021; 42: 3190–3199. [DOI] [PubMed] [Google Scholar]
- 5.Maraziti G, Vinson DR, Becattini C. Echocardiography for risk stratification in patients with pulmonary embolism at low risk of death: a response. Eur Heart J 2021; 43: 86–87. doi: 10.1093/eurheartj/ehab779 [DOI] [PubMed] [Google Scholar]
- 6.Konstantinides SV, Meyer G, Becattini C, et al. 2019 ESC Guidelines for the diagnosis and management of acute pulmonary embolism developed in collaboration with the European Respiratory Society (ERS): The Task Force for the diagnosis and management of acute pulmonary embolism of the European Society of Cardiology (ESC). Eur Respir J 2019; 54: 1901647. doi: 10.1183/13993003.01647-2019 [DOI] [PubMed] [Google Scholar]
- 7.Giri J, Sista AK, Weinberg I, et al. Interventional therapies for acute pulmonary embolism: current status and principles for the development of novel evidence: a scientific statement from the American Heart Association. Circulation 2019; 140: e774–e801. doi: 10.1161/CIR.0000000000000707 [DOI] [PubMed] [Google Scholar]
- 8.Becattini C, Agnelli G. Acute treatment of venous thromboembolism. Blood 2020; 135: 305–316. doi: 10.1182/blood.2019001881 [DOI] [PubMed] [Google Scholar]
- 9.Goldhaber SZ. Echocardiography in the management of pulmonary embolism. Ann Intern Med 2002; 136: 691–700. doi: 10.7326/0003-4819-136-9-200205070-00012 [DOI] [PubMed] [Google Scholar]
- 10.Pruszczyk P, Goliszek S, Lichodziejewska B. Prognostic value of echocardiography in normotensive patients with acute pulmonary embolism. JACC Cardiovasc Imaging 2014; 7: 553–560. doi: 10.1016/j.jcmg.2013.11.004 [DOI] [PubMed] [Google Scholar]
- 11.Paczyńska M, Sobieraj P, Burzyński Ł,et al. Tricuspid annulus plane systolic excursion (TAPSE) has superior predictive value compared to right ventricular to left ventricular ratio in normotensive patients with acute pulmonary embolism. Arch Med Sci 2016; 12: 1008–1014. doi: 10.5114/aoms.2016.57678 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Whiting PF, Rutjes AW, Westwood ME, et al. QUADAS-2: a revised tool for the quality assessment of diagnostic accuracy studies. Ann Intern Med 2011; 155: 529–536. doi: 10.7326/0003-4819-155-8-201110180-00009 [DOI] [PubMed] [Google Scholar]
- 13.IntHout J, Ioannidis JP, Rovers MM, et al. Plea for routinely presenting prediction intervals in meta-analysis. BMJ Open 2016; 6: e010247. doi: 10.1136/bmjopen-2015-010247 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Shim SR, Kim SJ, Lee J. Diagnostic test accuracy: application and practice using R software. Epidemiol Health 2019; 41: e2019007. doi: 10.4178/epih.e2019007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chang BH, Hoaglin DC. Meta-analysis of odds ratios: current good practices. Med Care 2017; 55: 328–335. doi: 10.1097/MLR.0000000000000696 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Higgins JPT, Green S, eds. Cochrane Handbook for Systematic Reviews of Interventions. Version 5.1.0. The Cochrane Collaboration; 2011. https://handbook-5-1.cochrane.org Date last updated: March 2011.
- 17.Sherwood MW, Melloni C, Jones WS, et al. Individual proton pump inhibitors and outcomes in patients with coronary artery disease on dual antiplatelet therapy: a systematic review. J Am Heart Assoc 2015; 4: e002245. doi: 10.1161/JAHA.115.002245 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med 2002; 21: 1539–1558. doi: 10.1002/sim.1186 [DOI] [PubMed] [Google Scholar]
- 19.Ozsu S, Karaman K, Mentese A, et al. Combined risk stratification with computerized tomography/echocardiography and biomarkers in patients with normotensive pulmonary embolism. Thromb Res 2010; 126: 486–492. doi: 10.1016/j.thromres.2010.08.021 [DOI] [PubMed] [Google Scholar]
- 20.Bahloul M, Regaieg K, Dlela M, et al. Pulmonary embolism in intensive care units: more frequent or more known? Prospective study of 75 cases. Clin Respir J 2019; 13: 513–520. doi: 10.1111/crj.13053 [DOI] [PubMed] [Google Scholar]
- 21.Jiménez D, Aujesky D, Moores L, et al. Combinations of prognostic tools for identification of high-risk normotensive patients with acute symptomatic pulmonary embolism. Thorax 2011; 66: 75–81. doi: 10.1136/thx.2010.150656 [DOI] [PubMed] [Google Scholar]
- 22.Becattini C, Agnelli G, Vedovati MC, et al. Multidetector computed tomography for acute pulmonary embolism: diagnosis and risk stratification in a single test. Eur Heart J 2011; 32: 1657–1663. doi: 10.1093/eurheartj/ehr108 [DOI] [PubMed] [Google Scholar]
- 23.Vanni S, Socci F, Pepe G, et al. High plasma lactate levels are associated with increased risk of in-hospital mortality in patients with pulmonary embolism. Acad Emerg Med 2011; 18: 830–835. doi: 10.1111/j.1553-2712.2011.01128.x [DOI] [PubMed] [Google Scholar]
- 24.Vanni S, Jiménez D, Nazerian P, et al. Short-term clinical outcome of normotensive patients with acute PE and high plasma lactate. Thorax 2015; 70: 333–338. doi: 10.1136/thoraxjnl-2014-206300 [DOI] [PubMed] [Google Scholar]
- 25.Vanni S, Polidori G, Vergara R, et al. Prognostic value of ECG among patients with acute pulmonary embolism and normal blood pressure. Am J Med 2009; 122: 257–264. doi: 10.1016/j.amjmed.2008.08.031 [DOI] [PubMed] [Google Scholar]
- 26.Grifoni S, Olivotto I, Cecchini P, et al. Short-term clinical outcome of patients with acute pulmonary embolism, normal blood pressure, and echocardiographic right ventricular dysfunction. Circulation 2000; 101: 2817–2822. doi: 10.1161/01.CIR.101.24.2817 [DOI] [PubMed] [Google Scholar]
- 27.Becattini C, Casazza F, Forgione C, et al. Acute pulmonary embolism: external validation of an integrated risk stratification model. Chest 2013; 144: 1539–1545. doi: 10.1378/chest.12-2938 [DOI] [PubMed] [Google Scholar]
- 28.Zhu L, Yang Y, Wu Y, et al. Value of right ventricular dysfunction for prognosis in pulmonary embolism. Int J Cardiol 2008; 127: 40–45. doi: 10.1016/j.ijcard.2007.06.093 [DOI] [PubMed] [Google Scholar]
- 29.Krüger S, Graf J, Merx MW, et al. Brain natriuretic peptide predicts right heart failure in patients with acute pulmonary embolism. Am Heart J 2004; 147: 60–65. doi: 10.1016/S0002-8703(03)00528-3 [DOI] [PubMed] [Google Scholar]
- 30.Post F, Mertens D, Sinning C, et al. Decision for aggressive therapy in acute pulmonary embolism: implication of elevated troponin T. Clin Res Cardiol 2009; 98: 401–408. doi: 10.1007/s00392-009-0017-1 [DOI] [PubMed] [Google Scholar]
- 31.George E, Kumamaru KK, Ghosh N, et al. Computed tomography and echocardiography in patients with acute pulmonary embolism: part 2: prognostic value. J Thorac Imaging 2014; 29: W7–W12. doi: 10.1097/RTI.0000000000000048 [DOI] [PubMed] [Google Scholar]
- 32.Kumamaru KK, George E, Ghosh N, et al. Normal ventricular diameter ratio on CT provides adequate assessment for critical right ventricular strain among patients with acute pulmonary embolism. Int J Cardiovasc Imaging 2016; 32: 1153–1161. doi: 10.1007/s10554-016-0887-z [DOI] [PubMed] [Google Scholar]
- 33.Lankeit M, Friesen D, Aschoff J, et al. Highly sensitive troponin T assay in normotensive patients with acute pulmonary embolism. Eur Heart J 2010; 31: 1836–1844. doi: 10.1093/eurheartj/ehq234 [DOI] [PubMed] [Google Scholar]
- 34.Weekes AJ, Johnson AK, Troha D, et al. Prognostic value of right ventricular dysfunction markers for serious adverse events in acute normotensive pulmonary embolism. J Emerg Med 2017; 52: 137–150. doi: 10.1016/j.jemermed.2016.09.002 [DOI] [PubMed] [Google Scholar]
- 35.Binder L, Pieske B, Olschewski M, et al. N-terminal pro-brain natriuretic peptide or troponin testing followed by echocardiography for risk stratification of acute pulmonary embolism. Circulation 2005; 112: 1573–1579. doi: 10.1161/CIRCULATIONAHA.105.552216 [DOI] [PubMed] [Google Scholar]
- 36.Taylor RA, Davis J, Liu R, et al. Point-of-care focused cardiac ultrasound for prediction of pulmonary embolism adverse outcomes. J Emerg Med 2013; 45: 392–399. doi: 10.1016/j.jemermed.2013.04.014 [DOI] [PubMed] [Google Scholar]
- 37.Dudzinski DM, Hariharan P, Parry BA, et al. Assessment of right ventricular strain by computed tomography versus echocardiography in acute pulmonary embolism. Acad Emerg Med 2017; 24: 337–343. doi: 10.1111/acem.13108 [DOI] [PubMed] [Google Scholar]
- 38.Ohigashi H, Haraguchi G, Yoshikawa S, et al. Comparison of biomarkers for predicting disease severity and long-term respiratory prognosis in patients with acute pulmonary embolism. Int Heart J 2010; 51: 416–420. doi: 10.1536/ihj.51.416 [DOI] [PubMed] [Google Scholar]
- 39.Zhu L, Yang YH, Wu YF, et al. Value of transthoracic echocardiography combined with cardiac troponin I in risk stratification in acute pulmonary thromboembolism. Chin Med J (Engl) 2007; 120: 17–21. doi: 10.1097/00029330-200701010-00004 [DOI] [PubMed] [Google Scholar]
- 40.Dellas C, Puls M, Lankeit M, et al. Elevated heart-type fatty acid-binding protein levels on admission predict an adverse outcome in normotensive patients with acute pulmonary embolism. J Am Coll Cardiol 2010; 55: 2150–2157. doi: 10.1016/j.jacc.2009.10.078 [DOI] [PubMed] [Google Scholar]
- 41.Kaeberich A, Seeber V, Jiménez D, et al. Age-adjusted high-sensitivity troponin T cut-off value for risk stratification of pulmonary embolism. Eur Respir J 2015; 45: 1323–1331. doi: 10.1183/09031936.00174514 [DOI] [PubMed] [Google Scholar]
- 42.Lankeit M, Kempf T, Dellas C, et al. Growth differentiation factor-15 for prognostic assessment of patients with acute pulmonary embolism. Am J Respir Crit Care Med 2008; 177: 1018–1025. doi: 10.1164/rccm.200712-1786OC [DOI] [PubMed] [Google Scholar]
- 43.Keller K, Rappold L, Gerhold-Ay A, et al. Sex-specific differences in pulmonary embolism. Thromb Res 2019; 178: 173–181. doi: 10.1016/j.thromres.2019.04.020 [DOI] [PubMed] [Google Scholar]
- 44.Beigel R, Mazin I, Goitein O, et al. Intermediate-risk pulmonary embolism: aiming to improve patient stratification. Eur J Intern Med 2019; 65: 32–36. doi: 10.1016/j.ejim.2019.04.018 [DOI] [PubMed] [Google Scholar]
- 45.Pruszczyk P, Kurnicka K, Ciurzyński M, et al. Defining right ventricular dysfunction by echocardiography in normotensive patients with pulmonary embolism. Pol Arch Intern Med 2020; 130: 741–747. [DOI] [PubMed] [Google Scholar]
- 46.Vanni S, Nazerian P, Bova C, et al. Comparison of clinical scores for identification of patients with pulmonary embolism at intermediate-high risk of adverse clinical outcome: the prognostic role of plasma lactate. Intern Emerg Med 2017; 12: 657–665. doi: 10.1007/s11739-016-1487-6 [DOI] [PubMed] [Google Scholar]
- 47.Witkin A, Wilcox SR, Chang Y, et al. Impact of chronic right ventricular pressure overload in short-term outcomes of acute pulmonary embolism: a retrospective analysis. J Crit Care 2019; 51: 1–5. doi: 10.1016/j.jcrc.2019.01.007 [DOI] [PubMed] [Google Scholar]
- 48.Ribeiro A, Lindmarker P, Juhlin-Dannfelt A, et al. Echocardiography Doppler in pulmonary embolism: right ventricular dysfunction as a predictor of mortality rate. Am Heart J 1997; 134: 479–487. doi: 10.1016/S0002-8703(97)70085-1 [DOI] [PubMed] [Google Scholar]
- 49.Palmieri V, Gallotta G, Rendina D, et al. Troponin I and right ventricular dysfunction for risk assessment in patients with nonmassive pulmonary embolism in the Emergency Department in combination with clinically based risk score. Intern Emerg Med 2008; 3: 131–138. doi: 10.1007/s11739-008-0134-2 [DOI] [PubMed] [Google Scholar]
- 50.Kostrubiec M, Łabyk A, Pedowska-Włoszek J, et al. Assessment of renal dysfunction improves troponin-based short-term prognosis in patients with acute symptomatic pulmonary embolism. J Thromb Haemost 2010; 8: 651–658. doi: 10.1111/j.1538-7836.2010.03762.x [DOI] [PubMed] [Google Scholar]
- 51.Pieralli F, Olivotto I, Vanni S, et al. Usefulness of bedside testing for brain natriuretic peptide to identify right ventricular dysfunction and outcome in normotensive patients with acute pulmonary embolism. Am J Cardiol 2006; 97: 1386–1390. doi: 10.1016/j.amjcard.2005.11.075 [DOI] [PubMed] [Google Scholar]
- 52.Vieillard-Baron A, Page B, Augarde R, et al. Acute cor pulmonale in massive pulmonary embolism: incidence, echocardiographic pattern, clinical implications and recovery rate. Intensive Care Med 2001; 27: 1481–1486. doi: 10.1007/s001340101032 [DOI] [PubMed] [Google Scholar]
- 53.Barrios D, Morillo R, Lobo JL, et al. Assessment of right ventricular function in acute pulmonary embolism. Am Heart J 2017; 185: 123–129. doi: 10.1016/j.ahj.2016.12.009 [DOI] [PubMed] [Google Scholar]
- 54.Yalamanchili K, Sukhija R, Aronow WS, et al. Prevalence of increased cardiac troponin I levels in patients with and without acute pulmonary embolism and relation of increased cardiac troponin I levels with in-hospital mortality in patients with acute pulmonary embolism. Am J Cardiol 2004; 93: 263–264. doi: 10.1016/j.amjcard.2003.09.058 [DOI] [PubMed] [Google Scholar]
- 55.Logeart D, Lecuyer L, Thabut G, et al. Biomarker-based strategy for screening right ventricular dysfunction in patients with non-massive pulmonary embolism. Intensive Care Med 2007; 33: 286–292. doi: 10.1007/s00134-006-0482-1 [DOI] [PubMed] [Google Scholar]
- 56.Gallotta G, Palmieri V, Piedimonte V, et al. Increased troponin I predicts in-hospital occurrence of hemodynamic instability in patients with sub-massive or non-massive pulmonary embolism independent to clinical, echocardiographic and laboratory information. Int J Cardiol 2008; 124: 351–357. doi: 10.1016/j.ijcard.2006.03.096 [DOI] [PubMed] [Google Scholar]
- 57.Samaranayake CB, Royle G, Jackson S, et al. Right ventricular dysfunction and pulmonary hypertension following sub-massive pulmonary embolism. Clin Respir J 2017; 11: 867–874. doi: 10.1111/crj.12429 [DOI] [PubMed] [Google Scholar]
- 58.Jiménez D, Kopecna D, Tapson V, et al. Derivation and validation of multimarker prognostication for normotensive patients with acute symptomatic pulmonary embolism. Am J Respir Crit Care Med 2014; 189: 718–726. doi: 10.1164/rccm.201311-2040OC [DOI] [PubMed] [Google Scholar]
- 59.Kukla P, McIntyre WF, Koracevic G, et al. Relation of atrial fibrillation and right-sided cardiac thrombus to outcomes in patients with acute pulmonary embolism. Am J Cardiol 2015; 115: 825–830. doi: 10.1016/j.amjcard.2014.12.049 [DOI] [PubMed] [Google Scholar]
- 60.Dahhan T, Siddiqui I, Tapson VF, et al. Clinical and echocardiographic predictors of mortality in acute pulmonary embolism. Cardiovasc Ultrasound 2016; 14: 44. doi: 10.1186/s12947-016-0087-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Khemasuwan D, Yingchoncharoen T, Tunsupon P, et al. Right ventricular echocardiographic parameters are associated with mortality after acute pulmonary embolism. J Am Soc Echocardiogr 2015; 28: 355–362. doi: 10.1016/j.echo.2014.11.012 [DOI] [PubMed] [Google Scholar]
- 62.Zanobetti M, Converti C, Conti A, et al. Prognostic value of emergency physician performed echocardiography in patients with acute pulmonary thromboembolism. West J Emerg Med 2013; 14: 509–517. doi: 10.5811/westjem.2013.4.12690 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Kanar BG, Şahin A, Göl G, et al. Timing and magnitude of regional right ventricular function and their relationship with early hospital mortality in patients with acute pulmonary embolism. Anatol J Cardiol 2019; 22: 26–32. doi: 10.14744/AnatolJCardiol.2019.38906 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Ciurzyński M, Kurnicka K, Lichodziejewska B, et al. Tricuspid regurgitation peak gradient (TRPG)/tricuspid annulus plane systolic excursion (TAPSE) – a novel parameter for stepwise echocardiographic risk stratification in normotensive patients with acute pulmonary embolism. Circ J 2018; 82: 1179–1185. doi: 10.1253/circj.CJ-17-0940 [DOI] [PubMed] [Google Scholar]
- 65.Stein PD, Matta F, Janjua M, et al. Outcome in stable patients with acute pulmonary embolism who had right ventricular enlargement and/or elevated levels of troponin I. Am J Cardiol 2010; 106: 558–563. doi: 10.1016/j.amjcard.2010.03.071 [DOI] [PubMed] [Google Scholar]
- 66.Frémont B, Pacouret G, Jacobi D, et al. Prognostic value of echocardiographic right/left ventricular end-diastolic diameter ratio in patients with acute pulmonary embolism: results from a monocenter registry of 1,416 patients. Chest 2008; 133: 358–362. doi: 10.1378/chest.07-1231 [DOI] [PubMed] [Google Scholar]
- 67.Sanchez O, Trinquart L, Caille V, et al. Prognostic factors for pulmonary embolism: the prep study, a prospective multicenter cohort study. Am J Respir Crit Care Med 2010; 181: 168–173. doi: 10.1164/rccm.200906-0970OC [DOI] [PubMed] [Google Scholar]
- 68.Sanchez O, Trinquart L, Planquette B, et al. Echocardiography and pulmonary embolism severity index have independent prognostic roles in pulmonary embolism. Eur Respir J 2013; 42: 681–688. doi: 10.1183/09031936.00097512 [DOI] [PubMed] [Google Scholar]
- 69.Pruszczyk P, Bochowicz A, Torbicki A, et al. Cardiac troponin T monitoring identifies high-risk group of normotensive patients with acute pulmonary embolism. Chest 2003; 123: 1947–1952. doi: 10.1378/chest.123.6.1947 [DOI] [PubMed] [Google Scholar]
- 70.Carroll BJ, Heidinger BH, Dabreo DC, et al. Multimodality assessment of right ventricular strain in patients with acute pulmonary embolism. Am J Cardiol 2018; 122: 175–181. doi: 10.1016/j.amjcard.2018.03.013 [DOI] [PubMed] [Google Scholar]
- 71.Tatlisu MA, Kaya A, Keskin M, et al. The association of blood urea nitrogen levels with mortality in acute pulmonary embolism. J Crit Care 2017; 39: 248–253. doi: 10.1016/j.jcrc.2016.12.019 [DOI] [PubMed] [Google Scholar]
- 72.Lobo JL, Holley A, Tapson V, et al. Prognostic significance of tricuspid annular displacement in normotensive patients with acute symptomatic pulmonary embolism. J Thromb Haemost 2014; 12: 1020–1027. doi: 10.1111/jth.12589 [DOI] [PubMed] [Google Scholar]
- 73.Yuriditsky E, Mitchell OJ, Sibley RA, et al. Low left ventricular outflow tract velocity time integral is associated with poor outcomes in acute pulmonary embolism. Vasc Med 2020; 25: 133–140. doi: 10.1177/1358863X19880268 [DOI] [PubMed] [Google Scholar]
- 74.Lee K, Kwon O, Lee EJ, et al. Prognostic value of echocardiographic parameters for right ventricular function in patients with acute non-massive pulmonary embolism. Heart Vessels 2019; 34: 1187–1195. doi: 10.1007/s00380-019-01340-1 [DOI] [PubMed] [Google Scholar]
- 75.Ösken A, Yelgeç NS, Şekerci SS, et al. Differences in clinical and echocardiographic variables and mortality predictors among older patients with pulmonary embolism. Aging Clin Exp Res 2021; 33: 2223–2230. doi: 10.1007/s40520-021-01882-3 [DOI] [PubMed] [Google Scholar]
- 76.Bikdeli B, Lobo JL, Jiménez D, et al. Early use of echocardiography in patients with acute pulmonary embolism: findings from the RIETE registry. J Am Heart Assoc 2018; 7: e009042. doi: 10.1161/JAHA.118.009042 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Kucher N, Rossi E, De Rosa M, et al. Prognostic role of echocardiography among patients with acute pulmonary embolism and a systolic arterial pressure of 90 mm Hg or higher. Arch Intern Med 2005; 165: 1777–1781. doi: 10.1001/archinte.165.15.1777 [DOI] [PubMed] [Google Scholar]
- 78.Park JR, Chang SA, Jang SY, et al. Evaluation of right ventricular dysfunction and prediction of clinical outcomes in acute pulmonary embolism by chest computed tomography: comparisons with echocardiography. Int J Cardiovasc Imaging 2012; 28: 979–987. doi: 10.1007/s10554-011-9912-4 [DOI] [PubMed] [Google Scholar]
- 79.Sukhija R, Aronow WS, Lee J, et al. Association of right ventricular dysfunction with in-hospital mortality in patients with acute pulmonary embolism and reduction in mortality in patients with right ventricular dysfunction by pulmonary embolectomy. Am J Cardiol 2005; 95: 695–696. doi: 10.1016/j.amjcard.2004.10.055 [DOI] [PubMed] [Google Scholar]
- 80.Meneveau N, Ming LP, Séronde MF, et al. In-hospital and long-term outcome after sub-massive and massive pulmonary embolism submitted to thrombolytic therapy. Eur Heart J 2003; 24: 1447–1454. doi: 10.1016/S0195-668X(03)00307-5 [DOI] [PubMed] [Google Scholar]
- 81.Prosperi-Porta G, Ronksley P, Kiamanesh O, et al. Prognostic value of echocardiography-derived right ventricular dysfunction in haemodynamically stable pulmonary embolism: a systematic review and meta-analysis. Eur Respir Rev 2022; 31: 220120. doi: 10.1183/16000617.0120-2022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Chornenki NLJ, Poorzargar K, Shanjer M, et al. Detection of right ventricular dysfunction in acute pulmonary embolism by computed tomography or echocardiography: a systematic review and meta-analysis. J Thromb Haemost 2021; 19: 2504–2513. doi: 10.1111/jth.15453 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Please note: supplementary material is not edited by the Editorial Office, and is uploaded as it has been supplied by the author.
Supplementary material 00641-2022.SUPPLEMENT (1.3MB, pdf)