Abstract
Imaging hemodynamics is crucial for the diagnosis, treatment, and prevention of vascular diseases. However, current imaging techniques are limited due to the use of ionizing radiation or contrast agents, short penetration depth, or complex and expensive data acquisition systems. Photoacoustic tomography shows promise as a solution to these issues. However, existing photoacoustic tomography methods collect signals either sequentially or through numerous detector elements, leading to either low imaging speed or high system complexity and cost. To address these issues, here we introduce a method to capture a 3D photoacoustic image of vasculature using a single laser pulse and a single-element detector that functions as 6,400 virtual ones. Our method enables ultrafast volumetric imaging of hemodynamics in the human body at up to 1 kHz and requires only a single calibration for different objects and for long-term operations. We demonstrate 3D imaging of hemodynamics at depth in humans and small animals, capturing the variability in blood flow speeds. This concept can inspire other imaging technologies and find applications such as home-care monitoring, biometrics, point-of-care testing, and wearable monitoring.
Introduction
Vascular diseases, including atherosclerosis, thrombosis, and aneurysms, pose serious health risks such as heart attack, stroke, and organ failure1. The early detection of these diseases through imaging of hemodynamics is crucial, enabling prompt intervention and treatment2. The efficacy of pharmacological therapies and surgical interventions, such as angioplasty and stenting, can also be evaluated through monitoring changes in blood flow and velocity3. Additionally, assessing an individual’s risk of developing vascular diseases is possible by measuring factors such as blood flow velocity, facilitating preventive measures. In biomedical research, imaging of hemodynamics can provide insights into the physiology and pathology of blood vessels, aiding in the development of new treatments for diseases such as hypertension, diabetes, and cancer. Overall, imaging of hemodynamics plays a vital role in disease diagnosis, treatment, and prevention, ultimately improving patient outcomes and advancing medical research1.
There are several techniques available to image hemodynamics in the human body, each with its own strengths and limitations. Magnetic resonance imaging (MRI), computed tomography (CT) angiography, and positron emission tomography (PET) are all capable of producing high-resolution images of the vascular system and blood flow dynamics, but they require the use of ionizing radiation and the injection of contrast agents, which can have adverse health effects4–6. Moreover, relying on strong ionizing sources and numerous detector elements, these techniques are bulky and expensive, making them inaccessible to mobile clinics or small healthcare facilities. Optical imaging techniques, such as fluorescence imaging and optical coherence tomography (OCT), offer noninvasive visualization of hemodynamics, but their penetration depths are constrained by the optical diffusion limit (~1–2 mm) and do not have sufficient specificity to hemoglobin7,8. Doppler ultrasound is another option, providing real-time measurement of blood flow velocity and direction. However, even with recent improvements in minimizing ultrasound probes, state-of-the-art ultrasound imaging techniques still require burdensome and costly data acquisition systems, e.g., Verasonics, due to hundreds to thousands of detector elements9,10.
Photoacoustic tomography (PAT), also known as optoacoustic tomography, offers a promising solution to the limitations faced by other imaging techniques for hemodynamic imaging11–13. Unlike other techniques, PAT utilizes the photoacoustic (PA) effect to absorb the energy of incident photons by optical absorbers, such as hemoglobin, in biological tissue and re-emit them as ultrasonic waves (PA waves) to generate optical contrast tomographic images14–17. As a result, PAT does not rely on ionizing radiation or contrast agents. Moreover, due to the weak scattering of ultrasound in biological tissue, PAT provides a depth-to-resolution ratio of approximately 200, enabling high spatial resolution at depths up to several centimeters11. Two primary forms of PAT are photoacoustic microscopy (PAM)17–19, which requires sequential scanning of the probing beam, and photoacoustic computed tomography (PACT)15,20,21, which captures a 3D image using one or a few pulses of the probing beam. PAM utilizes a single-element detector, which requires a simple data acquisition system but suffers from low imaging speed. In contrast, PACT offers higher imaging speed but requires numerous detection elements and the corresponding data acquisition systems, making it complex, expensive, and bulky.
To overcome the challenges of complexity, cost, and size in PACT systems, researchers have been working on ways to reduce the number of detector elements needed to reconstruct a 3D image. One promising approach utilizes the principles of compressive sensing and single-pixel imaging22–25, which use acoustic scatterers to achieve PA or ultrasound tomography with just a single detector element26–29. However, these techniques are time-consuming, as they require a sequence of measurements with different mask configurations, limiting their speed. To address this issue, researchers have developed novel methods that take advantage of the spatiotemporal encoding of an ergodic relay (ER) or a chaotic cavity30,31. These techniques can produce high-quality single-shot images while using fewer detector elements32–36. However, they have only been demonstrated for 2D imaging and require recalibration for different objects, which can be time-consuming. Additionally, they may not be suitable for long-term imaging in unstable environments due to their sensitivity to boundary conditions.
Here, we present photoacoustic computed tomography through an ergodic relay (PACTER), a PACT system that simultaneously addresses the challenges faced by other imaging techniques. PACTER provides a highly accessible and efficient solution, paving the way for noninvasive, label-free, and ultrafast 3D imaging of hemodynamics at depth in humans. With PACTER, a single-element detector encodes information equivalent to that of 6,400 virtual ones, enabling the reconstruction of a tomographic image of vasculature in 3D with just a single laser pulse. The system achieves ultrafast volumetric imaging at kilohertz rates, making it possible to capture fast hemodynamics in the human body in real-time. We demonstrate PACTER’s capability in monitoring vital signs in small animals and visualizing human hemodynamics in response to cuffing, capturing the variability in blood flow speeds. Because PACTER signals are unaffected by the boundary conditions of the object, the system only needs to be calibrated once and is suitable for long-term imaging in an unstable environment. PACTER’s single-element detector design makes it convenient, affordable, and compact, thus translatable to clinical applications such as home-care monitoring37,38, biometrics, point-of-care testing39, and noninvasive hemodynamic monitoring in intensive care units40. The single-element detector concept in PACTER can also be generalized to other imaging technologies, such as ultrasonography41, sonar42, and radar43.
Results
PACTER system
The PACTER system requires calibration only once prior to its utilization for a complete series of imaging. (Methods). In the calibration procedure (Fig. 1a), the laser beam was transmitted through the ER and focused on a uniform optical absorber placed on top of the ER. We used bovine blood as our calibration target (Supplementary Fig. 1). Using two motorized stages, we controlled the positions of a pair of mirrors to steer the focused laser beam across the field of view (FOV) in the x-y plane and recorded the PACTER signals at each scanning position. After calibration, the uniform optical absorber could be removed, and the system was ready for imaging. In the imaging procedure (Fig. 1b), the focusing lens was replaced by a fly’s eye homogenizer (Supplementary Fig. 2), which converted the incident laser beam into a widefield, homogenized illumination pattern that had the same shape and width as that of the calibration FOV (Supplementary Note 1). To acquire imaging data, the object was directly placed on top of the ER using ultrasound gel as the coupling medium, and we recorded the PACTER signal generated by the object following each laser pulse.
Fig. 1. PACTER system.
a,b, Schematic of the PACTER system in the calibration (a) and imaging (b) procedures. HWP, half-wave plate; PBS, polarizing beamsplitter; BT, beam trap; DAQ, data acquisition unit. The differences between the two modes are highlighted in the black dotted boxes. c, Schematic of the single-element ultrasonic transducer fabricated on the ER. d, 1D (t) PACTER signals detected by the ultrasonic transducer at time instances t0, t1, and tN. e, Reconstruction of a 4D (xyzt) image of human palmar vessels from the signals in d. Norm., normalized. Scale bar, 1 mm.
To enhance the detection sensitivity and improve the stability of the system, we fabricated a single-element ultrasonic transducer on the hypotenuse surface of the prism of the ER (Fig. 1c). The transducer was based on a lead magnesium niobate–lead titanate (PMN-PT) single crystal (Supplementary Figs. 3 and 4), which achieved exceptional piezoelectric performance, such as high piezoelectric constant (d33) and electromechanical coupling coefficient (kt)44–46. Further, because one of the gold electrodes of the transducer was directly sputtered on the surface of the ER, the PACTER signals inside the ER could reach the transducer with the maximum transmission, and the transducer and the ER became a whole piece that facilitated stable data acquisition.
Conventionally, a single-element ultrasonic transducer can only acquire a 1D signal in the time domain (Fig. 1d). However, with the ER, PACTER can use the single-element transducer to encode spatiotemporal information equivalent to those captured by 6,400 detectors (Supplementary Video 1), which can then be used to reconstruct a 3D map of the optical absorbers in the imaging volume. With a kilohertz laser repetition rate, PACTER can use the 1D (t) signals to generate a thousand 3D (xyz) volumes per second, leading to a high-speed 4D (xyzt) image of optical absorption in, e.g., human palmar vessels (Fig. 1e).
PACTER signal and reconstruction
PACTER needs only a one-time universal calibration despite its more stringent requirement for 3D imaging (Supplementary Note 2, Supplementary Fig. 5, Supplementary Video 2). In the calibration procedure, the focused laser beam was scanned across the FOV in 80 by 80 steps with a step size of 0.1 mm (Fig. 2a). To ensure that the PACTER signal acquired at each calibration pixel was distinct from others, we chose the step size to be about a half of the full width at half maximum (FWHM) of a line profile along the cross-correlation map, i.e., ~0.21 mm (Supplementary Fig. 6). Although the calibration signals were obtained by scanning the laser beam across a 2D plane, they could be used as 80 × 80 = 6,400 virtual transducers for 3D reconstruction (Fig. 2b) because (1) the PACTER signals were object-independent, and (2) the calibration signals were generated at the bottom of the 3D imaging volume. When source points in the 3D volume (, m = 1, 2, …) were illuminated by a laser pulse, the PA signals they generated would propagate to the calibrated virtual transducers (rn, n = 1, 2, …) after time , where c is the speed of the sound in the medium. Then, these signals would follow the same acoustic path inside the ER to the ultrasonic transducer as that of the calibration signals. From the transducer’s perspective, compared with the calibration signal kn(t) acquired at rn (Fig. 2c), the signal from the source point relayed through rn is proportional to kn(t) delayed by tm,n, i.e., (Fig. 2d). The signal is modulated by both p0,m, the initial pressure at , and a weighting factor dependent on the angle and distance. Accordingly, we developed an algorithm to reconstruct the initial pressure in the 3D volume (Supplementary Note 3). Because the reconstruction is prohibitively computationally intensive, we reformulated the forward model through temporal convolution and implemented it using the fast Fourier transformation, increasing the computational efficiency by 48,000 times.
Fig. 2. Single-shot 3D reconstruction in PACTER.
a, Illustration of the calibration procedure of PACTER. Focused laser beams for calibration are shown in green. Calibration pixels are highlighted as orange dots. Calibration step size is 0.1 mm. The calibration pixels (80 × 80) become 6,400 virtual transducers. r1, r2, r3, are the positions of three calibrated virtual transducers. b, Illustration of PACTER of human palmar vessels. The homogenized beam for widefield illumination is shown in green. and are the positions of two source points in the vessels. Blue and green spheres denote the PA waves generated by the source points. The calibrated virtual transducers capture the PA signals from and with different delays, indicated by the thick blue and green lines. c, PACTER signals, k1(t), k2(t), k3(t), of the calibrated virtual transducers corresponding to r1, r2, r3, respectively. d, PACTER signal from the widefield imaging consists of PA signals from and , which are essentially k1(t), k2(t), k3(t), delayed according to the distance between the calibrated virtual transducer and the source point.
Spatiotemporal characterization of PACTER
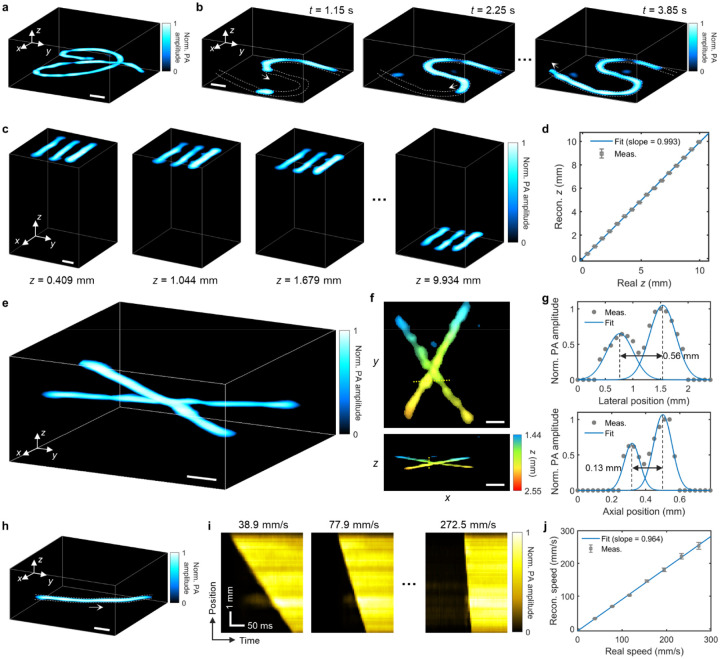
Using the signals acquired by the single detector, PACTER can image the 3D structure of a curved black wire with a single laser pulse (Fig. 3a) and the 4D dynamics of bovine blood flushing through an S-shaped tube when the tube was illuminated by multiple laser pulses (Fig. 3b, Supplementary Video 3). To evaluate whether the 3D volumes reconstructed by PACTER were correct measurements of the objects, we first compared the perspective views of the 3D volumes reconstructed by PACTER with the ground-truth projection images formed by raster-scanning the laser beam across the objects (Supplementary Fig. 7). Despite a lower spatial resolution compared with the ground truth, the comparison demonstrates that PACTER can correctly reconstruct the 3D objects in the lateral (x-y) directions. Second, we imaged a thin object in water, whose z positions were precisely controlled and measured by a linear translation stage. We imaged the object at multiple z positions, reconstructed the 3D volumes (Fig. 3c), and compared the z positions in the reconstructed volumes with the real ones. As shown in Fig. 3d, the reconstructed and real z positions were linearly related (R2 = 1.000) with a slope (0.993) close to 1, demonstrating that PACTER can accurately reconstruct the 3D objects in the axial (z) direction.
Fig. 3. Spatiotemporal characterization of PACTER.
a, 3D PACTER image of a curved black wire. Norm., normalized. b, Snapshots of 4D PACTER showing bovine blood flushing through an S-shaped tube. c, 3D PACTER images of three bars printed with black ink on a transparent film. In each image, the object was placed at a different z position. d, Reconstructed versus real z positions of the objects in c. The measurement results are plotted as means ± standard errors of the means (n = 1,980). The blue curve represents a linear fit. e, 3D PACTER image of two crossing human hairs in agarose. f, Maximum z- (top) and y-projections (bottom) of the 3D volume in e. The z positions of the object are color-encoded. g, Profiles along the yellow dashed lines in f denoted by gray dots. The blue curves represent two-term Gaussian fits. Black arrows denote the minimum distances that can separate the two hairs. h, 3D PACTER image of bovine blood flushing through a tube. The white arrow indicates the flushing direction. i, PA amplitudes along the tube in h versus time, when the blood flushes through the tube at different speeds. j, Speeds of the blood flow quantified from the reconstructed images versus the real speeds in i. The measurement results are plotted as means ± standard errors of the means (n = 74). The blue curve represents a linear fit. Scale bars, 1 mm.
To quantify the resolution of PACTER, we imaged two human hairs embedded in an agarose block (Fig. 3e). The hairs were intentionally positioned such that they were in close contact with each other, forming a crossing pattern that could be seen in both z- and y-projections (Fig. 3f). Defining the spatial resolution as the minimum distance that can distinguish the peaks of the two hairs, we found the lateral and axial resolutions of PACTER to be 0.56 mm and 0.13 mm, respectively (Fig. 3g). The anisotropic spatial resolutions along the lateral and axial directions were related to the image formation process in PACTER (Supplementary Notes 4 and 5, Supplementary Figs. 8 and 9). The coarser lateral resolution was due to the acoustic impedance mismatch between the object and the ER (Supplementary Note 4) while both resolutions are limited by the frequency-dependent acoustic attenuation (Supplementary Fig. 10).
To evaluate whether PACTER could be reliably used to image 4D dynamics, i.e., time-lapse movements of 3D objects, we captured 4D images of bovine blood flushing through a tube at different speeds precisely controlled by a syringe pump (Fig. 3h, Supplementary Video 4). Based on the reconstructed 4D images, we plotted the PA amplitudes along the tube (1D images) over time (Fig. 3i), calculated the speeds of the blood flow (Supplementary Fig. 11), and compared them with the real speeds set by the syringe pump (Fig. 3j). A linear relationship (R2 = 0.999) between the reconstructed and real speeds with a slope (0.964) close to 1 can be observed, demonstrating that PACTER is capable of 4D imaging, faithfully reconstructing the dynamics of 3D objects over time. Empowered by the imaging speed of up to a thousand volumes per second, PACTER could resolve the high-speed dynamics of the blood flushing through the tube at 272.5 mm/s in 3D, with a temporal resolution of 1 ms (Supplementary Video 5).
4D in vivo imaging of mouse hemodynamics with PACTER
Enabled by the capability of noninvasive, label-free, and ultrafast 3D imaging, PACTER is expected to be suitable for monitoring hemodynamics in vivo. Here, we evaluated PACTER’s capability in monitoring vital signs in small animals. We imaged the hemodynamics of the abdominal regions of mice (Fig. 4a). With a single laser pulse, PACTER could reconstruct the abdominal vasculature in 3D (Fig. 4b,c). When multiple laser pulses were used, PACTER revealed the 4D dynamics of the blood vessels (Supplementary Videos 6 and 7). Based on the 4D PACTER datasets, we isolated individual blood vessels from the cross sections of the 3D volumes (Fig. 4d,e) and visualized their motions and structural changes (Fig. 4f,g).
Fig. 4. PACTER of mouse hemodynamics in vivo.
a, Schematic of the mouse imaging experiment. b,c, 3D PACTER images of the abdominal vasculature of mouse 1 (b) and mouse 2 (c). Norm., normalized. d,e, Cross-sectional 2D images corresponding to the yellow rectangle in b (d) and the magenta rectangle in c (e) at four different time instances from the 4D PACTER datasets. t0 = 0.28 s, t1 = 0.26 s. White solid curves represent the Gaussian fits of the vessels’ profile denoted by the yellow (d) and magenta (e) dashed lines. Differences from the first image are highlighted. f,g, PA amplitudes along the yellow dashed line (1D images) in d (f) and the magenta dashed line in e (g) versus time, where the time instances in d and e are labeled with vertical gray lines. h,i, Center positions (blue solid curves) and widths (orange dash-dotted curves) of the vessels versus time, based on the fits in d (h) and e (i). The shaded areas denote the standard deviations (n = 5). j,k, Fourier transforms of the center positions and widths of the vessels in h (j) and i (k), showing the respiratory frequency from the vessel center positions only (j) or both the vessel center positions and widths (k). Scale bars, 1 mm.
By recording the time-lapse changes of the center positions and widths of the blood vessels, the respiratory motion could be tracked and identified (Fig. 4h,i). Using Fourier analysis, we found that the center position of the blood vessel of mouse 1 fluctuated periodically, exhibiting a respiratory frequency of 1.8 Hz (Fig. 4j), whereas the width of the vessel was relatively stable. In comparison, a respiratory frequency of 1.4 Hz could be observed from both the center position and width of the blood vessel of mouse 2 (Fig. 4k). Further, when we imaged the third mouse (Supplementary Video 8), we observed a respiratory frequency of 1.9 Hz from the width, not the center position, of the blood vessel (Supplementary Fig. 12). The distinct 4D hemodynamics of the blood vessels from the three mice demonstrated that PACTER could be a practical tool in monitoring vital signs, such as breathing, in small animals.
4D in vivo imaging of hemodynamics in human hands with PACTER
To demonstrate PACTER’s effectiveness in monitoring hemodynamics in humans, we imaged the hand vasculature of two participants. Different regions of the hand, e.g., fingers, proximal phalanx, and thenar regions, were imaged independently as the participants moved their hands to align those regions with the ER (Supplementary Fig. 13). In the following study, we focused on imaging the participants’ thenar vasculature and their responses to cuffing, which was induced by a sphygmomanometer wrapped around the participants’ upper arm (Fig. 5a). Using PACTER, we imaged the thenar vasculature in 3D with single laser pulses (Fig. 5b,c) and reconstructed the 4D dynamics of the blood vessels in response to cuffing (Supplementary Videos 9 and 10). As shown in the maximum amplitude projections of the 4D datasets (Fig. 5d,e), whereas some blood vessels exhibited a relatively stable PA amplitude throughout the experiment, the other vessels showed a decreased PA amplitude after cuffing due to the occlusion of blood flows; when the cuffing was released, the blood flows recovered, and the PA amplitude was rapidly restored (Fig. 5f,g). The different hemodynamics of these two types of blood vessels in response to cuffing may indicate their distinct roles in the circulatory system47: the blood vessels with stable and changing PA amplitudes could be venous and arterial, respectively, agreeing with the observations reported in other cuffing-based studies48–50. With the capability to simultaneously image both arterial and venous blood in vivo, PACTER provides additional benefits over conventional pulse oximetry, which can only monitor arterial blood without spatial resolution51.
Fig. 5. PACTER of human hand hemodynamics in vivo.
a, Schematic of the human hand imaging experiment. b,c, 3D PACTER images of the thenar vasculature of participant 1 (b) and participant 2 (c). Norm., normalized. d,e, Maximum amplitude projections of the 3D volumes from the 4D PACTER datasets along the z axis in b (d) and c (e) at the time instances before, during, and after cuffing. t0 = 17.44 s, t1 = 19.02 s. The solid lines flank the vessels under investigation. Differences from the images during cuffing are highlighted. f,g, PA amplitudes along the vessels (1D images) in d (f) and e (g) versus time, where the time instances in d and e are labeled with vertical gray lines. The blue and orange arrows indicate peak responses in the occlusion and recovery phases, respectively. h, Positions (solid circles) of the blood front along the blood vessel during the occlusion (left) and recovery (right) phases in f. The blue curve is an exponential fit with an occlusion rate of 1.3 ± 0.1 m/s, and the orange curve is a linear fit showing the blood flow speed of 16.5 ± 2.8 m/s. i, Comparison between the durations of the occlusion and recovery phases in f. ***P < 0.001, calculated by the two-sample t-test. j, Positions (solid circles) of the blood front along the blood vessel during the occlusion (left) and recovery (right) phases in g. The blue curve is an exponential fit with an occlusion rate of 2.4 ± 0.3 m/s, and the orange curve is a linear fit showing the blood flow speed of 32.9 ± 6.5 m/s. k, Comparison between the durations of the occlusion and recovery phases in g. ***P < 0.001, calculated by the two-sample t-test. l, Comparison between the blood flow speeds during recovery in f and g. *P < 0.05. Scale bars, 1 mm.
Because PA amplitudes have 100% sensitivity to optical absorption11, the 4D hemodynamics imaged by PACTER revealed the real-time changes in the blood vessels in response to cuffing, and the linear position of the blood front during the recovery phase could be used to measure the blood flow speed50. For participant 1, the occlusion rate of the vessel was found to be 1.3 ± 0.1 m/s, significantly slower than the blood flow speed of 16.5 ± 2.8 m/s extracted from the recovery phase (Fig. 5h,i). For participant 2, the occlusion rate and the blood flow speed of the vessel were found to be 2.4 ± 0.3 m/s and 32.9 ± 6.5 m/s, respectively (Fig. 5j,k), exhibiting a greater blood flow speed compared with participant 1 (Fig. 5l). Immediately after an imaging session, we asked participant 1 to slightly move their hand and used PACTER to image a different area of the thenar region (Supplementary Video 11). Using the same analysis on a different blood vessel, the occlusion rate and the blood flow speed were found to be 0.6 ± 0.1 m/s and 22.4 ± 6.4 m/s, respectively (Supplementary Fig. 14). Taken together, we demonstrated that PACTER could monitor the hemodynamics in human, including the consistent responses of thenar vasculature to cuffing, and capture the variability in blood flow speeds.
Discussion
Although the current implementation of PACTER requires motorized stages for calibration, a pulsed laser for illumination, and a DAQ card for data acquisition, these requirements could be fulfilled using cheaper and more compact alternatives. Owing to the universal calibration capability, the ER in PACTER could be pre-calibrated, eliminating the need for motor-based calibration. The pulsed laser and the DAQ card could be replaced with cost-effective light-emitting diodes (LEDs)52 and microcontrollers53, respectively, which could further enhance the portability of the system. Additionally, mass production of the ER and the single-element ultrasonic transducer could substantially lower the cost of the system, making PACTER more accessible to users or researchers in low-resource settings, further lowering the barriers to clinical translation.
Due to the large dimensions of the ER compared with the acoustic wavelength, the PA waves need to propagate a long distance inside the ER; therefore, a slight change in the speed of sound due to temperature fluctuations54 would cause large differences in the measured PACTER signal (Supplementary Video 12). To address this problem, we built a temperature stabilizing box to maintain the temperature of the ER (Supplementary Fig. 15), which stabilizes the temperature of the ER at a set temperature, e.g., 30 °C, at all times, guaranteeing a constant speed of sound throughout the experiments. In addition, the penetration depth of our PACTER system was limited to 3.6 mm in vivo due to the strong attenuation of 532 nm light by endogenous chromophores in biological tissue55. Changing the wavelength to 1064 nm could increase the penetration depth to several centimeters15,20. Another limitation of PACTER is the relatively small FOV (8 mm × 8 mm) constrained by the diameter of the glass rod in the ER. We believe that the design of the ER could be optimized further to achieve a larger FOV, enabling new applications such as vascular biometrics56. Finally, the spatial resolution of PACTER is currently limited by the acoustic impedance mismatch between the object and the ER, which could be addressed in the future by adding an impedance matching layer on top of the ER.
In summary, PACTER, a noninvasive, label-free, and ultrafast imaging technique, enables 4D imaging of hemodynamics in humans using the 1D signal captured by a single detector, achieving an imaging speed of up to a thousand volumes per second. We have demonstrated PACTER’s capability to visualize the 4D hemodynamics in humans and small animals. We have also shown the convenience of using PACTER to image different objects, including human hands and mouse abdomens, without the need for recalibration. PACTER’s high imaging speed allows for immediate intervention in case of abnormal hemodynamic changes. Additionally, PACTER’s low cost and compact form factor are ideal for point-of-care testing, facilitating quick and easy assessment of hemodynamic parameters at the bedside or in remote locations. We envision that PACTER will have profound impacts on a wide range of applications in biomedical research and clinical settings, including home care of diabetic-foot ulcers37 or carotid-artery disease38, point-of-care screening for hypertension39, and simultaneous oximetry of both arterial and venous blood in intensive care units40. Furthermore, PACTER’s single-shot 3D imaging concept using a single-element detector can extend beyond optical imaging, aiding fields such as medical ultrasonography41, underwater sonar42, and airborne radar43.
Methods
Experimental setup
In the PACTER system, the power of a 5-ns pulsed laser beam at 532 nm (INNOSLAB IS8II-DE, EdgeWave; 1 kHz pulse repetition rate) was controlled by a half-wave plate (WPH10M-532, Thorlabs) and a polarizing beam splitter (PBS25–532-HP, Thorlabs). The beam reflected by the PBS was sent to a beam trap (BT610, Thorlabs). The beam transmitted through the PBS was expanded by a beam expander consisting of two lenses (ACN254–050-A and AC254–100-A, Thorlabs). During calibration (Fig. 1a), the expanded beam was steered by the mirrors mounted on two motorized linear translation stages (PLS-85, PI), which were controlled by two motor drivers (CW215, Circuit Specialists). The beam diameter was adjusted to be 2 mm by an iris (SM1D12, Thorlabs), and the beam was sent through a lens (AC254–300-A, Thorlabs) and focused on top of the ER. A container with a window at its bottom sealed with an optically and ultrasonically transparent disposable polyethylene membrane was filled with bovine blood, which was used as a uniform optical absorber for calibration (Fig. 2a). The container was placed on top of the ER, where ultrasound gel (Aquasonic 100, ParkerLabs) was applied between the polyethylene membrane and the ER surface to facilitate acoustic coupling. During imaging (Fig. 1b), the beam diameter was adjusted to be 6 mm by the iris, and the beam was sent through a fly’s eye homogenizer (Supplementary Note 1) consisting of two microlens arrays (#64–480, Edmund Optics) and a lens (AC254–250-A, Thorlabs), which homogenized the beam in the imaging volume. Ultrasound gel was applied between the object and the top of the ER to facilitate acoustic coupling. The PACTER signals detected by the ultrasonic transducer were amplified by two low-noise amplifiers (ZKL-1R5+, Mini-Circuits), filtered by a low-pass filter (BLP-70+, Mini-Circuits), and digitized by a data acquisition card (ATS9350, AlazarTech) installed on a desktop computer. A multifunctional input/output (I/O) device (PCIe-6321, National Instruments) was used to control the laser, the motorized stages, and the data acquisition card.
Universally calibratable ER
The ER in PACTER consists of a right-angle prism (PS611, Thorlabs; 25 mm right-angle edge length) and a customized optical rod (VY Optoelectronics; 18 mm diameter, 175 mm length, top and bottom surfaces polished to 60–40 surface quality). Both components were made of ultraviolet (UV) fused silica, which has good optical transparency and low acoustic attenuation. To enhance ergodicity57, the edges of the prism were ground by a diamond saw (SYJ-150, MTI Co.) following a sawtooth pattern to obtain chaotic boundaries36. The prism and the rod were glued by UV-curing optical adhesive (NOA68, Norland Products), following exposure under UV light for 12 hr. To avoid the change of speed of sound due to temperature fluctuations during experiments, the whole ER was sealed in a temperature stabilizing box (Supplementary Fig. 15) regulated by a thermocouple (SA1-E, Omega Engineering), a heating pad (SRFG-303/10, Omega Engineering), and a temperature controller (Dwyer 32B-33, Cole-Parmer).
Fabrication of the ultrasonic transducer
The fabrication process of the ultrasonic transducer is as follows. First, PMN-PT piezoelectric single crystal (CTS Corporation) was chosen as the core component for acoustic-electrical conversion due to the excellent piezoelectric coefficient and high permittivity, which is suitable for high-frequency transducers with small aperture sizes because of the general electrical impedance matching (50 ohms). Second, based on the material parameters, a transducer modeling software (PiezoCAD) based on Krimboltz, Leedom, and Mattaei (KLM) equivalent circuit model was employed to simulate and optimize the design of the transducer. Hence, a 40 MHz PMN-PT transducer with a small active aperture size of 0.4 × 0.4 mm2 was designed and obtained. The piezoelectric element exhibits a central frequency of 40 MHz. Third, the piezoelectric crystal was lapped to the required thickness (40-μm), gold electrodes were sputtered on both sides, and then a layer of conductive silver paste (E-solder 3022) was deposited onto the piezoelectric sheet as a backing layer. Fourth, the acoustic stack was diced into designed elements size. Fifth, using Kapton tapes as a mask, a gold electrode was sputtered on the corner of the hypotenuse surface of the prism of the ER (Supplementary Fig. 3). The piezoelectric element was then affixed directly to the electrode on the ER by using a thin layer of conductive silver paste. The wires were connected out to read the signals. Last, a thin parylene layer as the protective layer was deposited onto the device.
Data acquisition
A custom-written LabVIEW (National Instruments) program was used to trigger the pulsed laser, drive the motorized stages during calibration, and acquire the data. The PACTER signals were acquired at a sampling rate of 250 megasamples per second, and a sampling length of 65,532 data points per acquisition. Due to the distance between the object and the ultrasonic transducer, a 28-μs delay was added to the data acquisition following the laser trigger. During calibration, to improve the signal-to-noise ratio (SNR) of the signal, we repeated the acquisition 500 times at each calibrated virtual transducer and used the averaged signal for PACTER reconstruction. To prevent motor backlash, the data were acquired only when the motor was moving forward; the acquisition stopped when the motor returned. During imaging, to improve the temporal resolution of the system, no signal averaging was used, and motor scanning was disabled.
Imaging protocols
All human and animal imaging experiments were performed in accordance with relevant guidelines and regulations. The human imaging experiments followed the protocol approved by the Institutional Review Board (IRB) of the California Institute of Technology. Two healthy adult participants were recruited for this study. Written informed consent was obtained from both participants according to the protocol. The PACTER system was pre-calibrated using bovine blood, and the participants were enrolled for the imaging procedure only. For imaging the vasculature in human hands, after applying ultrasound gel, the participants were instructed to place their hands on top of the ER. For imaging the human hand hemodynamics in response to cuffing, a sphygmomanometer was wrapped around the participants’ upper arm (Fig. 5a). To induce blood vessel occlusion, the sphygmomanometer was inflated to high pressure (200 mgHg), maintained for a short time (~15 s), and then quickly released; the total imaging time was 30 s. The animal imaging experiments followed the protocol approved by the Institutional Animal Care and Use Committee (IACUC) of the California Institute of Technology. IRB and IACUC were aware of both protocols before approval. The fluence of the laser beam for imaging (5 mJ/cm2) was within the American National Standards Institutes (ANSI) safety limit for laser exposure (20 mJ/cm2 at 532 nm)58.
Animal preparation
Female athymic nude mice (Hsd: Athymic Nude-Foxn1nu, Envigo; 15–20 g and 4–5 weeks old) were used for the animal imaging experiments. Before imaging, the mouse was placed in a small chamber with 5% vaporized isoflurane mixed with air for anesthesia induction. It was then transferred to a customized animal mount, which has a hole at the bottom such that the abdomen of the mouse can be imaged by PACTER. Ultrasound gel was applied on top of the ER, and then the animal mount was lowered until the mouse abdomen was in contact with the ER. Throughout the imaging session, the mouse was kept anesthetized with a continuous supply of 1.5% vaporized isoflurane, its head was fixed to the stereotaxic frame of the mount, and its body temperature was maintained at ~38 °C by a heating pad.
Sample preparation
For calibration, refrigerated bovine blood (#910, Quad Five) was restored to room temperature and transferred to the customized container (Fig. 2a) for acquiring the calibration signals for PACTER reconstruction. Besides bovine blood, black tape (6132-BA-10, 3M), black rubber (5508T44, McMaster-Carr), and black ink (X-1, Tamiya) were also used to test their performance as uniform optical absorbers for calibration. During the test, black tape and black rubber were cut into 10 mm × 10 mm sheets and placed on top of the ER, with ultrasound gel as the coupling medium; black ink was stored in the customized container. For imaging, black wires (8251T9, McMaster-Carr) were bent into curved shapes and placed in the customized container filled with water. Bar patterns were printed with black ink on a transparent film (ITF-30, Octago), cut into 10 mm × 10 mm sheets, and placed in the water-filled customized container; a wooden stick mounted on a manual linear translation stage (PT1, Thorlabs) was glued to the back of the film to control its z position in the imaging volume. Human hairs were embedded in a 4% agarose block (A-204–25, GoldBio), and the block was placed in the water-filled customized container during imaging. Polyurethane tubes (MRE025, Braintree Scientific; 0.012″ inside diameter) were first placed in the water-filled customized container in straight or curved shapes, and a syringe was used to flush bovine blood through the tubes; the speed of the blood flow was controlled by a syringe pump (NE-300, New Era).
PACTER reconstruction
In PACTER, the signal s(t) detected by the ultrasonic transducer at time t in a homogeneous medium is expressed as (Supplementary Note 3)
| #(1) |
Here, M and N are the numbers of the source points and the calibrated virtual transducers, respectively; kn(t) is the normalized impulse response from the calibration at the n-the virtual transducer; and rn are the locations of the m-th source point and the n-th virtual transducer, respectively; p0,m, is a value proportional to the initial pressure at ; θm,n denotes the incidence angle satisfying with n being the normal vector of the calibration plane; θ1is the critical angle the ultrasonic refraction from water to fused silica; , represents the indicator function defined in Eq. (S6); c is the speed of sound in the homogeneous medium; δ(t) denotes the delta function.
Discretizing Eq. (1), we obtain the forward model
| #(2) |
where s, represents a vector of length L, p0 denotes a vector of length M(=M1M2M3) which consists of all voxels in a 3D image of size M1 × M2 × M3, and H is the system matrix of size L × M. This forward model has a computational complexity of max{O(MN), O(NLlog2L)}. To obtain an image from the signals s, we invert the forward model by solving the regularized optimization problem
| #(3) |
Here, ∣p0∣TV denotes the total variation of the 3D image corresponding to p0, and λ is the regularization parameter. Numerically, we solve this optimization problem through a Fast Iterative Shrinkage-Thresholding Algorithm (FISTA)59.
Image processing
The reconstructed images were first denoised using a 3D median filter (in the 3-by-3-by-3 neighborhood) and smoothed using a 3D Gaussian filter (with a 0.1-by-0.1-by-2 standard deviation kernel). We then applied a Hessian-matrix-based vesselness filter60 to the denoised images to improve the contrast of vascular structures in 3D. Finally, we added the vesselness-enhanced images (self-normalized) with a weighting factor of 0.8 back to the filtered images with a weighting factor of 0.2 and obtained the presented images. The images were rendered in 3D or 4D (time-lapse 3D) using the Imaris (Bitplane) software. The speeds of bovine blood flushing through the tube in Fig. 3 were calculated by differentiating the PA amplitudes along the tube and fitting the relationship between the traveling distance of the blood front versus time. The vessels’ profiles in Fig. 4 were fitted as a Gaussian function, exp(−(x − x0)2/w2), where the center positions and widths of the vessels were estimated from x0 and , respectively. The vessels’ profiles in Supplementary Fig. 12 were fitted as a two-term Gaussian function, where the center positions and widths were estimated from the first term. The positions of the blood front along the blood vessels in Fig. 5 and Supplementary Fig. 14 were obtained through thresholding the PA amplitude profiles. Denoting the total length of the blood vessel profile as Lp, we fitted the positions of the blood front along the blood vessels during the occlusion and recovery phases with do(t) = a exp(−vot/Lp) and dr(t) = −vrt + b, respectively, where a and b were constants, vo was the occlusion rate, and vr was the blood flow speed. The durations of the occlusion and recovery phases were estimated by to = 3Lp/vo and tr = 0.95Lp/vr, respectively, which were the time it took for the blood front to propagate 95%, i.e., 1 − exp(−3), of Lp. To determine if the differences between to and tr were significant, we applied a Welch’s (unequal variances) t-test to determine the P values under the null hypothesis that the mean values of to are not different from those of tr. The same t-test was also performed to determine if the difference between the blood flow speeds in Fig. 5l was significant.
Supplementary Material
Acknowledgments
We thank Yanyu Zhao for contributing to the universal calibration. This work was supported in part by National Institutes of Health grants R01 EB028277, U01 EB029823, and R35 CA220436 (Outstanding Investigator Award). The computations presented here were conducted in the Resnick High Performance Computing Center, a facility supported by Resnick Sustainability Institute at the California Institute of Technology
Competing interests
L.V.W. has a financial interest in Microphotoacoustics Inc., CalPACT LLC, and Union Photoacoustic Technologies Ltd., which, however, did not support this work. K.M. has a financial interest in Microphotoacoustics, Inc. The other authors declare no competing interests.
Footnotes
Code availability
The reconstruction code, the system control software, and the data collection software are proprietary and used in licensed technologies, yet they are available from the corresponding author upon reasonable request.
Data availability
The main data supporting the results in this study are available within the paper and its Supplementary Information. Other data are too large to be publicly shared, yet they are available for research purposes from the corresponding author on reasonable request.
References
- 1.Sanz J. & Fayad Z. A. Imaging of atherosclerotic cardiovascular disease. Nature 451, 953–957 (2008). [DOI] [PubMed] [Google Scholar]
- 2.Kadem M., Garber L., Abdelkhalek M., Al-Khazraji B. K. & Keshavarz-Motamed Z. Hemodynamic Modeling, Medical Imaging, and Machine Learning and Their Applications to Cardiovascular Interventions. IEEE Rev. Biomed. Eng. 16, 403–423 (2023). [DOI] [PubMed] [Google Scholar]
- 3.Davies P. F. Hemodynamic shear stress and the endothelium in cardiovascular pathophysiology. Nat. Clin. Pract. Cardiovasc. Med. 6, 16–26 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Fox M. D. & Raichle M. E. Spontaneous fluctuations in brain activity observed with functional magnetic resonance imaging. Nat. Rev. Neurosci. 8, 700–711 (2007). [DOI] [PubMed] [Google Scholar]
- 5.Maurovich-Horvat P., Ferencik M., Voros S., Merkely B. & Hoffmann U. Comprehensive plaque assessment by coronary CT angiography. Nat. Rev. Cardiol. 11, 390–402 (2014). [DOI] [PubMed] [Google Scholar]
- 6.Provost J. et al. Simultaneous positron emission tomography and ultrafast ultrasound for hybrid molecular, anatomical and functional imaging. Nat. Biomed. Eng. 2, 85–94 (2018). [DOI] [PubMed] [Google Scholar]
- 7.Fan J. L. et al. High-speed volumetric two-photon fluorescence imaging of neurovascular dynamics. Nat. Commun. 11, 6020 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Shu X., Beckmann L. & Zhang H. F. Visible-light optical coherence tomography: a review. J. Biomed. Opt. 22, 1 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wang C. et al. Continuous monitoring of deep-tissue haemodynamics with stretchable ultrasonic phased arrays. Nat. Biomed. Eng. 5, 749–758 (2021). [DOI] [PubMed] [Google Scholar]
- 10.Hu H. et al. A wearable cardiac ultrasound imager. Nature 613, 667–675 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wang L. V. & Hu S. Photoacoustic Tomography: In Vivo Imaging from Organelles to Organs. Science 335, 1458–1462 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wang L. V. & Yao J. A practical guide to photoacoustic tomography in the life sciences. Nat. Methods 13, 627–638 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Weber J., Beard P. C. & Bohndiek S. E. Contrast agents for molecular photoacoustic imaging. Nat. Methods 13, 639–650 (2016). [DOI] [PubMed] [Google Scholar]
- 14.Zhang H. F., Maslov K. & Wang L. V. In vivo imaging of subcutaneous structures using functional photoacoustic microscopy. Nat. Protoc. 2, 797–804 (2007). [DOI] [PubMed] [Google Scholar]
- 15.Li L. et al. Single-impulse panoramic photoacoustic computed tomography of small-animal whole-body dynamics at high spatiotemporal resolution. Nat. Biomed. Eng. 1, 0071 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Xu M. & Wang L. V. Universal back-projection algorithm for photoacoustic computed tomography. Phys. Rev. E 71, 016706 (2005). [DOI] [PubMed] [Google Scholar]
- 17.Yao J. et al. High-speed label-free functional photoacoustic microscopy of mouse brain in action. Nat. Methods 12, 407–410 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Cao R. et al. Label-free intraoperative histology of bone tissue via deep-learning-assisted ultraviolet photoacoustic microscopy. Nat. Biomed. Eng. (2022) doi: 10.1038/s41551-022-00940-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Shi J. et al. High-resolution, high-contrast mid-infrared imaging of fresh biological samples with ultraviolet-localized photoacoustic microscopy. Nat. Photonics 13, 609–615 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lin L. et al. Single-breath-hold photoacoustic computed tomography of the breast. Nat. Commun. 9, 2352 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Na S. et al. Massively parallel functional photoacoustic computed tomography of the human brain. Nat. Biomed. Eng. 6, 584–592 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Duarte M. F. et al. Single-pixel imaging via compressive sampling. IEEE Signal Process. Mag. 25, 83–91 (2008). [Google Scholar]
- 23.Sun B. et al. 3D Computational Imaging with Single-Pixel Detectors. Science 340, 844–847 (2013). [DOI] [PubMed] [Google Scholar]
- 24.Sun M.-J. et al. Single-pixel three-dimensional imaging with time-based depth resolution. Nat. Commun. 7, 12010 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Stellinga D. et al. Time-of-flight 3D imaging through multimode optical fibers. Science 374, 1395–1399 (2021). [DOI] [PubMed] [Google Scholar]
- 26.Kruizinga P. et al. Compressive 3D ultrasound imaging using a single sensor. Sci. Adv. 3, (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Luís Dean-Ben X. & Razansky D. Localization optoacoustic tomography. Light Sci. Appl. 7, 18004–18004 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Deán-Ben X. L., Özbek A., López-Schier H. & Razansky D. Acoustic Scattering Mediated Single Detector Optoacoustic Tomography. Phys. Rev. Lett. 123, 174301 (2019). [DOI] [PubMed] [Google Scholar]
- 29.Hahamovich E., Monin S., Levi A., Hazan Y. & Rosenthal A. Single-detector 3D optoacoustic tomography via coded spatial acoustic modulation. Commun. Eng. 1, 25 (2022). [Google Scholar]
- 30.Montaldo G., Palacio D., Tanter M. & Fink M. Time reversal kaleidoscope: A smart transducer for three-dimensional ultrasonic imaging. Appl. Phys. Lett. 84, 3879–3881 (2004). [Google Scholar]
- 31.Montaldo G., Palacio D., Tanter M. & Fink M. Building three-dimensional images using a time-reversal chaotic cavity. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 52, 1489–1497 (2005). [DOI] [PubMed] [Google Scholar]
- 32.Cox B. T. & Beard P. C. Photoacoustic tomography with a single detector in a reverberant cavity. J. Acoust. Soc. Am. 125, 1426–1436 (2009). [DOI] [PubMed] [Google Scholar]
- 33.Brown M. D., Zhang E. Z., Treeby B. E., Beard P. C. & Cox B. T. Reverberant cavity photoacoustic imaging. Optica 6, 821 (2019). [Google Scholar]
- 34.Li Y. et al. Snapshot photoacoustic topography through an ergodic relay for high-throughput imaging of optical absorption. Nat. Photonics 14, 164–170 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Li Y., Wong T. T. W., Shi J., Hsu H.-C. & Wang L. V. Multifocal photoacoustic microscopy using a single-element ultrasonic transducer through an ergodic relay. Light Sci. Appl. 9, 135 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Li L., Li Y., Zhang Y. & Wang L. V. Snapshot photoacoustic topography through an ergodic relay of optical absorption in vivo. Nat. Protoc. 14, 164–170 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Andersen C. A. Noninvasive assessment of lower extremity hemodynamics in individuals with diabetes mellitus. J. Vasc. Surg. 52, 76S–80S (2010). [DOI] [PubMed] [Google Scholar]
- 38.Sobieszczyk P. & Beckman J. Carotid Artery Disease. Circulation 114, (2006). [DOI] [PubMed] [Google Scholar]
- 39.Vashist S. K., Luppa P. B., Yeo L. Y., Ozcan A. & Luong J. H. T. Emerging Technologies for Next-Generation Point-of-Care Testing. Trends Biotechnol. 33, 692–705 (2015). [DOI] [PubMed] [Google Scholar]
- 40.Marik P. E. & Baram M. Noninvasive Hemodynamic Monitoring in the Intensive Care Unit. Crit. Care Clin. 23, 383–400 (2007). [DOI] [PubMed] [Google Scholar]
- 41.Moore C. L. Point-of-Care Ultrasonography. N. Engl. J. Med. 9 (2011). [DOI] [PubMed] [Google Scholar]
- 42.Trucco A., Palmese M. & Repetto S. Devising an Affordable Sonar System for Underwater 3-D Vision. IEEE Trans. Instrum. Meas. 57, 2348–2354 (2008). [Google Scholar]
- 43.Reigber A. et al. Very-High-Resolution Airborne Synthetic Aperture Radar Imaging: Signal Processing and Applications. Proc. IEEE 101, 759–783 (2013). [Google Scholar]
- 44.Zhou Q., Lau S., Wu D. & Kirk Shung K. Piezoelectric films for high frequency ultrasonic transducers in biomedical applications. Prog. Mater. Sci. 56, 139–174 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Zhou Q., Lam K. H., Zheng H., Qiu W. & Shung K. K. Piezoelectric single crystal ultrasonic transducers for biomedical applications. Prog. Mater. Sci. 66, 87–111 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Zhou Q. et al. PMN-PT single crystal, high-frequency ultrasonic needle transducers for pulsed-wave Doppler application. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 54, 668–675 (2007). [DOI] [PubMed] [Google Scholar]
- 47.Pugsley M. K. & Tabrizchi R. The vascular system An overview of structure and function. J. Pharmacol. Toxicol. Methods 8 (2000). [DOI] [PubMed] [Google Scholar]
- 48.Langham M. C., Floyd T. F., Mohler E. R., Magland J. F. & Wehrli F. W. Evaluation of Cuff-Induced Ischemia in the Lower Extremity by Magnetic Resonance Oximetry. J. Am. Coll. Cardiol. 55, 598–606 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Loenneke J. P. et al. Effect of cuff type on arterial occlusion. Clin. Physiol. Funct. Imaging 33, 325–327 (2013). [DOI] [PubMed] [Google Scholar]
- 50.Zhou Y., Liang J. & Wang L. V. Cuffing-based photoacoustic flowmetry in humans in the optical diffusive regime. J. Biophotonics 9, 208–212 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Sinex J. E. Pulse oximetry: Principles and limitations. Am. J. Emerg. Med. 17, 59–66 (1999). [DOI] [PubMed] [Google Scholar]
- 52.Zhu Y. et al. Light Emitting Diodes based Photoacoustic Imaging and Potential Clinical Applications. Sci. Rep. 8, 9885 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Fatima A. et al. Review of cost reduction methods in photoacoustic computed tomography. Photoacoustics 15, 100137 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Ide J. M. The Velocity of Sound in Rocks and Glasses as a Function of Temperature. J. Geol. 45, 689–716 (1937). [Google Scholar]
- 55.Yao J. & Wang L. V. Photoacoustic microscopy: Photoacoustic microscopy. Laser Photonics Rev. 7, 758–778 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Wang Y. et al. A Robust and Secure Palm Vessel Biometric Sensing System Based on Photoacoustics. IEEE Sens. J. 18, 5993–6000 (2018). [Google Scholar]
- 57.Fink M. & de Rosny J.. Time-reversed acoustics in random media and in chaotic cavities. Nonlinearity 15, R1–R18 (2002). [Google Scholar]
- 58.American National Standards Institute. ANSI Z136.3–2018 American National Standard for Safe Use of Lasers in Health Care. (2018).
- 59.Beck A. & Teboulle M. A fast iterative shrinkage-thresholding algorithm for linear inverse problems. SIAM J. Imaging Sci. 2, 183–202 (2009). [Google Scholar]
- 60.Jerman T., Pernuš F., Likar B. & Špiclin Ž. Beyond Frangi: an improved multiscale vesselness filter. in (eds. Ourselin S. & Styner M. A.) 94132A (2015). doi: 10.1117/12.2081147. [DOI] [Google Scholar]
- 61.Zimmermann M., Lindlein N., Voelkel R. & Weible K. J. Microlens laser beam homogenizer: from theory to application. Laser Beam Shap. VIII 6663, 666302 (2007). [Google Scholar]
- 62.Treeby B. E. & Cox B. T. k-Wave: MATLAB toolbox for the simulation and reconstruction of photoacoustic wave fields. J. Biomed. Opt. 15, 021314 (2010). [DOI] [PubMed] [Google Scholar]
- 63.Saleh B. E. A. & Teich M. C. Fundamentals of Photonics. (Wiley-Interscience, 2007). [Google Scholar]
- 64.Abou-Sayed A. S. & Clifton R. J. Pressure shear waves in fused silica. J. Appl. Phys. 47, 1762–1770 (1976). [Google Scholar]
- 65.Szabo T. L. Diagnostic Ultrasound Imaging: Inside Out. (Elsevier, 2014). [Google Scholar]
- 66.Kino G. S. Acoustic Waves: Devices, Imaging, and Analog Signal Processing. (Prentice-Hall, 1987). [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The main data supporting the results in this study are available within the paper and its Supplementary Information. Other data are too large to be publicly shared, yet they are available for research purposes from the corresponding author on reasonable request.