Abstract
Light chain cast nephropathy (LCCN) is a leading cause of acute kidney injury (AKI) in patients with multiple myeloma (MM) and is now defined as a myeloma defining event. While the long-term prognosis has improved with novel agents, short-term mortality remains significantly higher in patients with LCCN especially if the renal failure is not reversed. Recovery of renal function requires a rapid and significant reduction of the involved serum free light chain. Therefore, proper treatment of these patients is of the utmost importance. In this paper, we provide an algorithm for treatment of MM patients who present with biopsy-proven LCCN or in those where other causes of AKI have been ruled out. The algorithm is based on data from randomized trial whenever possible. When trial data is not available, our recommendations is based on non-randomized data and expert opinions on best practices. We recommend that all patients should enroll in a clinical trial if available prior to resorting to the treatment algorithm we outlined.
Subject terms: Myeloma, Therapeutics
Introduction
Acute kidney injury (AKI) caused by light chain cast nephropathy (LCCN) is one of the major complications from multiple myeloma (MM). It is most commonly seen at initial MM diagnosis but can also develop later in the course of disease during relapse. The incidence AKI at diagnosis is 16–31% when measured by serum creatinine (Scr) concentration >1.4 mg/dl, and 16–22% when defined using Scr >2 mg/dl [1, 2]. By estimated glomerular filtration rate (eGFR), 36–45% have an eGFR < 60 ml/min/1.73 m2, while 12–17% have an eGFR <30 ml/min/1.73 m2 [2–4]. A large French study of 1038 patients with MM found that ~25% met the 2014 International Myeloma Working Group (IMWG) renal impairment criterion (Scr > 2 mg/dl or eGFR < 40 ml/min/1.73 m2), with 12.9% requiring dialysis [5]. Other have reported dialysis dependence in 6–8% of patients during the clinical course of MM [4, 6].
Of the four myeloma defining events (MDEs) (hyperCalcemia, Renal impairment, Anemia and Bone lytic lesions), renal impairment imposes the greatest impact on overall survival (OS) even after adjusting for other cofactors and comorbidities [2–6]. This was especially evident in the alkylator era [6, 7]. Although this effect is diminished by novel agents particularly bortezomib in clinical trial settings [8, 9], real world data still reflect that AKI at the time of myeloma diagnosis imposes a negative impact on mortality particularly in the first 6 months of therapy [4, 5, 10]. Fortunately, recovery of kidney function reverses the negative impact on OS [5–7, 10]. But while long-term survival has improved, short-term mortality remains higher in patients without kidney recovery regardless of the treatment regimen [8].
Mechanisms of renal impairment
In 2014, the IMWG clarified that only AKI secondary to LCCN qualifies as a MDE [11]. LCCN occurs when the overproduced monoclonal free light chain (FLC) by the myeloma cells interacts with the Tamm Horsfall protein in the loop of Henle to form light chain casts [12]. The light chain casts obstruct the tubules causing rupture which induces an immune response further injuring the tubules [13]. Injury is also mediated by hydrogen peroxide produced by the FLC and FLC activation of the Nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) and Apoptosis signal-regulating kinase 1 (ASK1) or Janus kinases, signal transducer and activator of transcription proteins (STATs) pathway [13–15]. These pathways induce apoptosis, inflammation and promote tubulointerstitial fibrosis that further the damage beyond tubular objection.
Serum FLC concentrations are predictive of the development of AKI and its recovery. AKI is rare when serum FLC concentration is <50 mg/dl but increases significantly when the concentration exceeds 80–200 mg/dl [16, 17]. A high serum FLC concentration alone may not be sufficient as a high urinary FLC excretion appears to be necessary for AKI to occur [7, 18, 19]. Conversely, a rapid reduction of serum FLC concentration is the key to reversing the kidney injury. A minimum reduction of 50–60% of serum FLC has been found to be associated with renal recovery in LCCN [20, 21]. One study finds that fewer patients recovered kidney function with the same degree of FLC reduction achieved at day 21 as compared to day 12 [22]. Finally, a serum FLC concentration of <50 mg/dl at the end of cycle 1 of chemotherapy is associated with better renal recovery in a recent clinical trial using a high cutoff (HCO) dialyzer [23].
Diagnosis
Renal impairment in MM is currently defined by an eGFR of <40 ml/min/1.73 m2 or a Scr >2 mg/dl [11]. For diagnostic and management purposes, the etiology of AKI in MM must be established. Only LCCN qualifies as a MDE, and its management is different from the management needs for AKI due to other causes. Thus, in patients with MM presenting with AKI, other causes of renal failure such as dehydration, hypercalcemia, drug-induced nephritis, unrelated diabetes or hypertension, and other monoclonal gammopathy related renal pathology must all be excluded. There are a number of renal disorders than are caused by monoclonal proteins besides LCCN that are collectively incorporated in the diagnostic umbrella now termed monoclonal gammopathy of renal significance (MGRS) [24]. Kidney biopsy is the gold standard for differentiating between LCCN, MGRS lesions, and other unrelated causes of AKI [25]. Unfortunately, a kidney biopsy is not always feasible for various reasons, especially in the acute setting. Once dehydration, hypercalcemia, and other causes of AKI are excluded based on clinical presentation, the main differential is between LCCN and MGRS renal lesions. Since most MGRS kidney lesions affect the glomeruli, they cause a high degree of albuminuria. In contrast, LCCN is associated with mainly Bence Jones proteinuria often with <10% albuminuria [26]. Therefore, in patients with >1 g/d proteinuria with <10% albuminuria, and a serum FLC concentration >150 mg/dl, the probability of LCCN is high enough that a kidney biopsy can be omitted [7, 26, 27]. On the other hand, patients with high degree of albuminuria or lower serum FLC, or where there is any uncertainty about the etiology of AKI should undergo a kidney biopsy.
Current frontline treatment options
Proteasome inhibitors (Table 1)
Table 1.
Multiple myeloma trials with renal impaired patients.
| Trial | Regimen | Patients | Inclusion criteria | Hematologic and Renal Outcomes | Toxicity |
|---|---|---|---|---|---|
| Phase III VISTA | MP vs VMP |
NDMM 450 with CrCl >50 ml/min 227 CrCl ≤ 50 ml/min |
Scr <2 mg/dl |
HR in patients with CrCl <50 ml/min ORR: MP = 46% vs VMP = 68% CR: MP = 5% vs VMP = 31% Renal response Overall: MP = 34% vs VMP = 44% CrCl < 30 ml/min: MP = 7% vs VMP = 37% CrCl 30 - < 50 ml/min: MP = 39% vs VMP = 46% |
Grade 4 hematologic AEs (42%) and SAEs (63%) were higher in patients with CrCl ≤ 30 ml/min but discontinuation rates were similar. Patients who achieved reversal of RI had less grade 5 AEs (8% vs 15%), SAEs (43% vs 60%) and discontinuation (6% vs 24%) than patients without reversible RI. |
| Phase II study Ludwig et al. | BDD |
68 NDMM and RMM |
eGFR < 50 ml/min |
Hematologic response Renal response CR/nCR = 38% CRrenal = 31% VGPR = 15% PRrenal = 7% PR = 13% MRrenal = 24% MR = 6% |
Grade 3/4 AEs Anemia - 50% Neutropenia - 14.7% Thrombocytopenia -14.7% Infections: Grade 4 - 19.1%, Grade 3 - 48.5% |
| Phase III Hovon-65/ HMMG-HD4 | PAD/ASCT/Bortezomib maintenance vs VAD/ASCT/thalidomide maintenance |
NDMM 81 with Scr ≥ 2 mg/dl 746 with <2 mg/dl |
No exclusion for kidney function |
HR in patients with Scr > 2 mg/dl ORR: PAD 75% vs VAD 36% >VGPR: PAD 33% vs VAD 9% Renal response ORR: PAD = 81% vs VAD = 63% CRrenal: PAD = 58% vs VAD = 43% PRrenal: PAD = 0% vs VAD = 3% MRrenal: PAD = 23% vs VAD = 17% |
Grade 4 AEs Anemia: VAD (7%) vs PAD (8%) Neutropenia: VAD (1%) vs PAD (3%) Thrombocytopenia: VAD (5%) vs PAD (10%)* Infections: VAD (21%) vs PAD (26%) Nonhematologic AE GI symptoms: VAD (7%) vs PAD (11%)# PN: VAD (10%) vs PAD (24%)# * = p < 0.01, # = p < 0.001 |
| Phase III MYRE |
BD: bortezomib 1.3 mg/m2, dex 20 mg Days (1,2,4,5, 8,9,11,12) cycle 1, cycle 2 is 28 days for age >70 years vs CBD: BD + iv CTX 750 mg/m2 Day 1 |
184 NDMM with LCCN |
Scr > 1.92 mg/dl or eGFR < 40 ml/min/1.73m2 |
Hematologic response at 3 months ORR: BD = 78.3% vs CBD = 77.2% CR/VGPR: BD 39.1% vs CBD = 51.1% Renal response at 3 months ORR: BD = 44.6% vs CBD = 51.1% |
Serious AEs BD – 32.6% vs CBD – 43.5% Grade ≥ 3 Cytopenia: BD (5.4%) vs CBD (9.8%) Sepsis/pneumonia: BD (2.2%) vs CBD (5.4%) None were statistically significant |
| Tosi et al. |
T or TD Thalidomide 100–400 mg daily Dex- 40 mg days 1–4 |
20 RRMM |
Scr > 1.5 mg/dl or CrCl < 60 ml/min |
Hematologic response at 3 months ORR: 75% PR: 45% Renal response (Scr < 1.5 mg/dl) ORR: 80% |
Constipation – 25% Lethargy – 25% Leukopenia – 5% 50% of patients on 400 mg dose could not tolerate the dose due to Grade ≥ 2 toxicity. |
| Analysis of the phase III MM-009 and MM-010 Dimopoulos et al. |
Lenalidomide 25 mg days 1–21 Dex 40 mg Days 1–4, 9–12, 17–21 |
341 RRMM |
Scr ≥ 2.0 mg/dl |
Hematologic response CR: mild RI 16%, mod RI 16%. sRI 6% VGPR: mild RI 19%, mod RI 11%, sRI 31% PR: mild RI 30%, mod RI 29%, sRI 13% Improvement of renal function by 1 level was experienced by 72% |
Neutropenia: 32–48%. Thrombocytopenia: 38% severe RI, 22% moderate RI and 9% mild/no RI. Anemia: 44% severe RI, 21% moderate RI, 5% mild/no RI Pneumonia: 25% severe RI, 9% moderate RI, 7% mild/no RI. Dehydration: 13% severe RI, 2% moderate RI and 0.8% mild/no RI |
| Phase II study Ludwig et al. |
Lenalidomide dosed based on CrCl Dex 40 mg Days 1–4, 9–12, 17–21 cycle 1 and weekly afterwards |
35 NDMM RRMM LCCN with 15 biopsy confirmed |
Scr > 2 mg/dl or CrCl < 50 ml/min |
Hematologic and Renal response CR: 20% VGPR: 8% PR: 40% CRrenal: 14.2% PRrenal: 11.4% MRrenal: 20% |
Grade 3/4 toxicities Anemia: 43% Thrombocytopenia: 23% Leukopenia: 12% Neutropenia: 15% 3 patients withdrew due to AE and 4 deaths during the first 2 cycles |
| Phase II study Bridoux et al. |
Lenalidomide (dose based on kidney function) Dex 40 mg weekly |
38 RRMM |
No Scr cutoff but renal function needed to be stable |
Hematologic response ORR: 76% CR: 0% VGPR: 26% PR: 50% ≥50 ml/min: 75% vs <50 ml/min: 78% |
Nonserious AEs Thrombocytopenia: normal (40%) vs sRI/HD (63.6%) Leukopenia: normal (20%) vs sRI/HD (36.4%) Anemia: normal (0%) vs sRI/HD (27.3%) Fatigue: normal (20%) vs sRI/HD (45.5%) |
| PrE1003 |
Lenalidomide dose escalation Dex 40 mg weekly |
63 RRMM |
Hematologic response at 3 months CrCl 30–60 ml/min: 60% CrCl < 30 ml/min not on dialysis: 60% CrCl < 30 ml/min on dialysis: 20% |
Grade 3/4 AEs 45% Most common AEs were anemia, decreased appetite, muscle weakness/fatigue Pneumonia: 19.3% |
|
| Phase II MM-013 |
Pomalidomide 4 mg days 1–21 Dex 40 mg weekly if <75 years of age or 20 mg weekly if >75 years of age |
81 RRMM |
eGFR < 45 ml/min/1.73 m2 |
HR ORR/VGPR or better A: 39.4%/18.2% B: 32.4%/8/8% C: 14.3%/7.1% Renal response A: 18.2%; complete RR in 18.2$% B: 35.3% C: 7.1% |
Grade 3/4 AEs (A/B/C) Neutropenia - 60.6%/44.1%/57.1% Anemia - 27.3%/35.3%/57.1% Thrombocytopenia – 27.3%/17.6%/50% Infections – 39.4%/26.5%/28.6% Pneumonia – 12.1%/5.9%/7.1% Hyperkalemia – 0%/8.8%/14.3% |
AE adverse event, BDD and PAD bortezomib doxorubicin dexamethasone, CBD cyclophosphamide bortezomib dexamethasone, CR complete response, CrCl – creatinine clearance by Cockcroft Gault method, dex dexamethasone, eGFR estimated glomerular filtration rate by Modification of Diet in Renal Disease (MDRD) method, Group A eGFR 30–<45 ml/min/1.73 m2, Group B eGFR < 30 ml/min/1.73 m2 not dialysis dependent, Group C dialysis dependent, HR hematologic response, LCCN light chain cast nephropathy, MP melphalan prednisone, nCR – near complete response, NDMM newly diagnosed multiple myeloma, ORR overall response rate, PR partial response, RI renal impairment (mild = CrCl ≥ 60 ml/min, moderate = ≥30 to <60 ml/min, severe =<30 ml/min), RR renal response, RRMM relapse or refractory multiple myeloma, SAE severe adverse event, Scr serum creatinine, sRI severe renal impairment with CrCl <30 ml/min, VGPR very good partial response, VMP bortezomib melphalan prednisone.
Bortezomib is a reversible proteasome inhibitor that is not renally cleared nor nephrotoxic [28]. In the VISTA trial, the addition of bortezomib (V) to melphalan and prednisone (MP) significantly increased the overall response rate (ORR) from 46 to 68%, and the complete response (CR) rate from 5 to 31%, respectively in patients with eGFR < 50 ml/min/1.73 m2 [9]. Renal recovery rate in those with an eGFR < 30 ml/min/1.73 m2 was 37% in the VMP arm vs 7% in the MP treated patients. Bortezomib was combined with doxorubicin and dexamethasone (BDD) in a phase II study in both newly diagnosed (NDMM) and relapsed myeloma patients with a median eGFR of 20.5 (3.7–49.9) ml/min. BDD produced an ORR of 72%, with a very good partial response (VGPR) or better rate of 52% [29]. Improvement in eGFR correlated with the depth of hematologic response (HR) with the median posttreatment eGFR of 59.6 ml/min, 38.9 ml/min and 16.8 ml/min in patients who achieved >VGPR, partial response (PR) or minimal response (MR), and stable disease or less, respectively. Median progression free survival (PFS) of this study was 12.1 months and OS had not been reached after a median follow-up of 22.4 months. In the phase III Hovon-65/HMMG-HD4 trial, BDD was compared to vincristine Adriamycin and dexamethasone (VAD) induction followed by autologous stem cell transplant (ASCT) followed by thalidomide maintenance in the VAD group and bortezomib maintenance in the BDD group [30]. Despite a significantly higher prevalence of high risk cytogenetics [del17p and t(4;14)] in the patient with Scr >2 mg/dl, the OS of patients treated with BDD was similar to those with baseline creatinine ≤2 mg/dl [8]. BDD achieved a significantly higher ORR and >VGPR rate (75% and 33%, respectively) in the patients with a baseline creatinine >2 mg/dl as compared to VAD (36% and 9%, respectively). Patients with a baseline creatinine ≤2 mg/dl had similar OS regardless of treatment but those with Scr >2 mg/dl treated with VAD had an inferior OS. Overall renal response was 81% (58% renal CR) in the BDD treated patients vs 63% (43% renal CR) in the VAD treated patients. A randomized trial comparing bortezomib plus dexamethasone (BD) to bortezomib, cyclophosphamide, dexamethasone (CBD) found similar HR (78.3% in BD vs 77.2% in CBD, p = 1.00) and >VGPR rate (39.1% in BD and 51.1% in CBD, p = 0.14) in NDMM with renal impairment not requiring dialysis at 3 months [31]. There was no difference in the overall renal response (44.6% in BD vs 51.1% in CBD, p = 0.46) but recovery of patients with AKI stage 3 (creatinine increased >3 times of baseline) trended toward CBD therapy (23.2% in BD vs 46.7% in CBD, p = 0.07).
Limited data exist in the frontline therapy of severe renally impaired MM patients treated with the other proteasome inhibitors. A phase I/Ib study was conducted with ixazomib in severe renally impaired patients including 7 patients on dialysis [32]. A dose of 3 mg on Days 1, 8, 15 on a 28-days cycle was established for safety and pharmacokinetics standpoint but no efficacy data was provided. A study of carfilzomib in severe renally impaired patients including 10 patients on dialysis recommended the doses of 27 and 56 mg/m2 [33]. In a phase II trial of carfilzomib dexamethasone in renally impaired relapsed refractory (RRMM) patients using the 15 and 27 mg/m2 dosing, HR was similar in patients with varying degree of renal impairment including patients on dialysis [34]. They achieved an ORR of 25.5%, with all being PR or less. Adverse effects were also similar except for AKI in the 3 of 10 patients with moderate renal impairment (creatinine clearance between 30 and 49 ml/min) [34]. Unfortunately, the risk of renal toxicity including thrombotic microangiopathy makes carfilzomib an unattractive choice in MM patients with AKI [35, 36]. Currently, neither ixazomib nor carfilzomib is approved for frontline treatment of MM in the United States.
Immunomodulators (Table 1)
Immunomodulators have been used in MM patients with renal impairment. Despite a similar mechanism of actions, they possess different pharmacologic properties that give each a unique pharmacokinetics and different side effects. Thalidomide is hydrolyzed by all body fluids and probably has limited renal clearance. Pharmacokinetic studies however showed clearance with dialysis, thus it should be given after dialysis [37]. Lenalidomide is renally cleared and dialyzable. It requires dose adjustment according to GFR and dialysis status [38]. Pomalidomide is renally secreted, but it is metabolized by the liver resulting in only mild extension of its half-life even in severe renal impairment making dosage adjustment for renal function unnecessary but should be given post dialysis [39].
There are only two small studies of thalidomide in renal failure patients with MM. The first is a 7 patient study where thalidomide was dosed between 100 mg and 400 mg daily in patients with creatinine clearance (CrCl) between 47 ml/min and dialysis dependence [40]. Three patients achieved a CR that lasted 5–8 months and 1 patient achieved a PR for 1 year. Adverse effects included constipation, peripheral neuropathy and hyperkalemia were noted in the dialysis dependent patient. In a separate study of 20 RRMM patients with renal impairment treated with thalidomide or thalidomide dexamethasone, 75% achieved a HR with 45% achieving a PR or better after stem cell transplant [41]. Dose reduction to 200 mg daily was required in 50% of patients treated with the 400 mg daily dose due to adverse effects.
Several phase II trials had been performed with lenalidomide dexamethasone (Rd) in renally impaired MM patients. The dosing schedule used was: 10 mg daily for CrCl >30 but ≤50 ml/min, 15 mg q48 hours for CrCl <30 ml/min but not on dialysis, and 5 mg daily after dialysis in patients on dialysis for 21 days on a 28-days cycle in one study [42]. In a small study of 35 NDMM and relapsed patients with high dose dexamethasone, 4 died during the first 2 cycles and 5 withdrew from the study, 3 were for adverse events. On an intention to treat analysis, 20% had a CR and 8% had a VGPR. Sixteen (45.7%) of the 35 patients had a renal response, with 14.2% achieving a renal CR, 11.4% with renal PR and 20% in renal MR. Another phase II trial of Rd using the same lenalidomide dosing but weekly dexamethasone achieved an ORR of 76% with 50% PR and 26% VGPR [43]. The ORR was similar between patients with normal kidney function and mild renal impairment vs patients with moderate renal impairment to ESRD; however, only 1 patient with severe renal impairment (eGFR < 30 ml/min/1.73 m2 or ESRD) achieved a VGPR. A dose escalation trial in renally impaired RRMM patients used a similar dosing schedule for dialysis independent patients dosed lenalidomide at 15 mg 3 times a week after dialysis for dialysis dependent patients [44]. The highest dosing achieved in the study was 25 mg daily for patients with CrCl 30–59 ml/min, 15 mg daily for those with CrCl < 30 ml/min regardless of dialysis status. The ORR was 54.3% with the poorest response seen in the dialysis group (20%). Twenty-eight patients experienced grade 3/4 adverse events with 1 death in a dialysis dependent patient from lung infection, sepsis and multiorgan failure attributed to therapy while 5 others died of unrelated deaths due to cirrhosis, intra-abdominal hemorrhage, sudden death and 2 ESRD. Median PFS was 12.6 months and OS was 20.0 months.
A phase II trial was conducted with pomalidomide and low dose dexamethasone in RRMM patients [45]. All 81 patients had an eGFR <45 ml/min/1.73 m2 with 14 dialysis dependent patients. Pomalidomide was dosed at 4 mg days 1–21 on a 28-days cycle. ORR was 39.4% in group A (patients with eGFR between 30 and 45 ml/min/1.73 m2), 32.4% in group B (patients with <30 ml/min/1.73 m2 not on dialysis) and 14.3% in group C (patients on dialysis). No CR was recorded but VGPR was noted in 18.2% of group A, 8.8% of group B and 7.1% of group C patients. Median PFS correlated with baseline kidney function [6.5 m (A) vs 4.2 m (B) vs 2.4 m (C)] and OS [16.4 m (A) vs 11.8 m (B) vs 5.2 m (C)] was the shortest among dialysis dependent patients. Leukopenia was more than twice as common in dialysis dependent patients but infection rates were similar. Thrombocytopenia was also most severe in the dialysis dependent patients. Dose reduction and discontinuation rates were similar amongst the 3 groups. Pomalidomide is currently approved for relapsed MM with 2 prior therapies including lenalidomide and a proteasome inhibitor.
Anti-CD38 monoclonal antibodies
Currently, 3 randomized trials have been conducted with daratumumab in the upfront setting in MM. The addition of daratumumab to the backbone of Rd was compared in MAIA, bortezomib thalidomide dexamethasone (VTD) in CASSIOPEIA and bortezomib lenalidomide dexamethasone (VRD) in the GRIFFIN trial [46–48]. Unfortunately, all 3 trials excluded patients with an eGFR <30 ml/min/1.73 m2 so data on severe renal impairment or dialysis are limited to case reports for daratumumab [49]. A study comparing isatuximab, carfilzomib, dexamethasone vs carfilzomib plus dexamethasone has been conducted in RRMM with renal impairment (IKEMA trial) [50]. Unfortunately, in this study, only 2.7% and 2.4% of the patients had severe renal impairment.
Extracorporeal therapies
Plasmapheresis (PLEX) for the treatment of AKI in MM was first reported in 1976 [51]. Since then, three randomized trials had been performed with differing outcomes. Zucchelli et al. randomized 15 patients to daily PLEX with hemodialysis vs 14 patients to peritoneal dialysis (PD) [52]. Patients who underwent PLEX had greater reduction of Bence Jones proteinuria than PD (p < 0.001). Eleven of 13 dialysis dependent patients treated with PLEX became dialysis independent but only 2 of 11 PD patients recovered kidney function. Although positive, this study was criticized for high early mortality (35.7%) in the PD group vs 6.7% in the PLEX treated patients. Johnson et al. randomized 21 patients to hemodialysis vs hemodialysis plus thrice weekly PLEX. Dialysis was required for 7 of 11 PLEX and 5 of 10 hemodialysis patients [53]. Kidney function improved in 63.6% of the PLEX patients vs 50% of the hemodialysis patients (p = NS), but of the dialysis dependent patients, all 3 who recovered had received PLEX. Clark et al. randomized 58 patients to 5–7 PLEX over the first 10 days and 39 patients to the control group. Hemodialysis was required for 25.9% of the PLEX patients vs 36% of the controls [54]. At the end of the study, dialysis requirement and death were noted in 17.9% and 33.3% of the controls and 8.6% and 32.8% of the PLEX patients respectively, (p = NS). The primary endpoint (a composite of death, dialysis dependence and eGFR < 29 ml/min/1.73 m2) was documented in 57.9% of PLEX patients vs 69.2% of the controls, p = 0.36.
Although some felt that PLEX was ineffective based on the Clark study, it is important to point out significant differences among these studies. LCCN was confirmed by kidney biopsy in majority of the patients in the Zucchelli and Johnson studies, but few biopsies were performed in the Clark study [52–54]. Dialysis requirement was the threefold higher in positive Zucchelli study than the negative Clark study. Investigators in the Johnson study also felt that PLEX was more beneficial in patients with the more severe AKI. Finally, patients in the Zucchelli study received daily PLEX while the Johnson and Clark studies were limited to every other day [52–54].
Other extracorporeal devices used in the treatment of LCCN includes the high cut-off (HCO) dialyzers. HCO dialyzers are dialyzers with pore size up to 50 kd (vs 5 kd in normal dialyzers) which can reduce serum FLC levels by >70% [55]. Two randomized trials have been performed to date. MYRE was the first trial to report enrolled 98 dialysis dependent patients in France from 2011 to 2016 [23]. Patient were treated with BD on a 21-day cycle and cyclophosphamide could be added after cycle 3 if response was insufficient. Eight 5 h HCO dialysis were performed within the first 10 days while the control group received the same with a regular dialyzer. HCO dialyzer treatment produced a higher ORR (78.3% vs 60.4%, p 0.06) and significantly higher >VGPR rate (69.6% vs 47.9% respectively, OR - 2.37, p = 0.03) as compared to control. HCO hemodialysis resulted in a significantly higher renal recovery at 6 months (secondary endpoint, 56.4% vs 35.4% respectively, odds ratio – 2.37, p = 0.04) and 12 months (60.9% of the HCO vs 37.5% of control, OR 2.59, p = 0.02) but did not reach significance at 3 months (primary endpoint, 41.3% vs 33.3% respectively, p = 0.42). There was no difference in OS.
The EuLITE study enrolled 90 dialysis dependent patients mainly in the United Kingdom from 2008 to 2013 used BDD as the chemotherapy [56]. The HCO dialysis was scheduled for 6 h the first day followed by another seven 8 h sessions over 10 days. Control patients received 4 h dialysis thrice weekly. Despite similar single session FLC reductions (-77% for κ, -72% for λ) as MYRE, patients in EuLITE treated with the HCO dialyzer had a lower CR (14% vs 30%) and VGPR (23% vs 32%) at 6 months as compared to control patients [23, 56]. Moreover, the HCO group had an inferior age adjusted OS (hazard ratio – 2.63, p = 0.03) as compared to control despite similar renal recovery rates at 90 days (56% HCO vs 51% control, p - 0.81) [56]. Higher infectious complications especially pulmonary infection in the HCO group vs control (31% vs 9% respectively) may have played a role.
Recommendations
Given the importance of renal recovery and the requirement for rapid serum FLC reduction, an aggressive therapeutic approach is justified in patients MM patients with AKI. Regimens used should have a high and rapid response rate; the drugs used should not require modifications for renal function and should be readily available for immediate administration. The goal should be to reduce circulating serum FLCs as quickly as possible, including the use of PLEX (Fig. 1). In NDMM, based on the activity of various drugs and treatment regimens used as initial therapy so far, we prefer daratumumab combined with VCD (bortezomib cyclophosphamide dexamethasone) or VD as initial therapy. We have found high activity with VCD/VD in the past [57], but in our clinical experience and trial data from Andromeda [58], the addition of daratumumab hastens the response and limits the number of days extracorporeal light chain removal that is needed. In countries where IMiDs can be easily obtained for hospitalized patients, daratumumab plus VTD such as the regimen used in CASSIOPEIA could be an option instead of daratumumab plus VCD [48]. Lenalidomide should probably be avoid in the upfront setting due to requirement for dose adjustment.
Fig. 1. Treatment algorithm for newly diagnosed multiple myeloma patients with acute kidney injury.
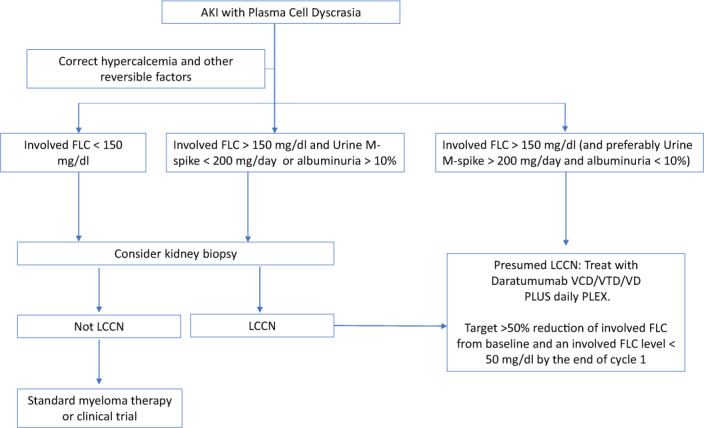
AKI acute kidney injury, FLC free light chains, M monoclonal, LCCN light chain cast nephropathy, VCD bortezomib, cyclophosphamide, dexamethasone, VTD bortezomib, thalidomide, dexamethasone, PLEX plasma exchange.
Extracorporeal light chain removal with PLEX should started as soon as possible to help reduce the serum FLC concentration more rapidly. Even though data on PLEX is controversial, the procedure has low risk, and in our opinion provides patients with the best chance at renal recovery [20, 57]. PLEX should be performed daily until the involved FLC is below 150 mg/dl or >60% reduction from baseline, if possible. Since PLEX could potentially remove daratumumab, daratumumab should be given after PLEX. In countries where HCO dialyzers are available, these could be an option instead of PLEX, although the 5 h sessions used in the MYRE trial should be performed.
The situation is more complicated with RRMM patients since many of the relapsed medications have not been tested in severe renal impaired patients. In these patients, renal recovery is equally important since eligibility of CAR-T cell therapy and clinical trials require an eGFR >40–50 ml/min/1.73 m2. If the patient did not relapse on an anti-CD38 antibody drug, daratumumab or isatuximab could be used. Combination therapy based on bortezomib, dexamethasone, thalidomide and a 4-day continuous infusion of cisplatin, doxorubicin, cyclophosphamide, and etoposide (VDT-PACE) can be used in renally impaired patients. In these patients, cisplatin is usually omitted and cyclophosphamide should be dose adjusted for kidney function. PLEX should be offered especially in patients with options of potential future therapies.
Acknowledgements
Supported in part by grants CA 168762 and CA186781 from the National Cancer Institute, Rockville, MD, USA, and the Marvin Family Grant.
Author contributions
All of the authors collectively conceived of the paper, researched the literature, and wrote the paper.
Data availability
This is a current treatment algorithm. There are no new data generated for this paper and data sharing is not applicable.
Competing interests
Dr NL reports Stock and Other Ownership Interests at Senseonics, AbbVie, Verrica, and research funding from Omeros, all outside the submitted work. Dr SVR reports grants from NIH, outside the submitted work.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Knudsen LM, Hippe E, Hjorth M, Holmberg E, Westin J. Renal function in newly diagnosed multiple myeloma—a demographic study of 1353 patients. The Nordic Myeloma Study Group. Eur J Haematol. 1994;53:207–12. doi: 10.1111/j.1600-0609.1994.tb00190.x. [DOI] [PubMed] [Google Scholar]
- 2.Terpos E, Christoulas D, Kastritis E, Katodritou E, Pouli A, Michalis E, et al. The Chronic Kidney Disease Epidemiology Collaboration Cystatin-C (CKD-EPI-CysC) Equation Has an Independent Prognostic Value for Overall Survival in Newly-Diagnosed Patients with Symptomatic Multiple Myeloma; Is It Time to Change from MDRD to CKD-EPI-CysC equations? Eur J Haematol. 2013;91:347–55. doi: 10.1111/ejh.12164. [DOI] [PubMed] [Google Scholar]
- 3.Kleber M, Ihorst G, Deschler B, Jakob C, Liebisch P, Koch B, et al. Detection of renal impairment as one specific comorbidity factor in multiple myeloma: multicenter study in 198 consecutive patients. Eur J Haematol. 2009;83:519–27. doi: 10.1111/j.1600-0609.2009.01318.x. [DOI] [PubMed] [Google Scholar]
- 4.Ho PJ, Moore EM, McQuilten ZK, Wellard C, Bergin K, Augustson B, et al. Renal Impairment at Diagnosis in Myeloma: Patient Characteristics, Treatment, and Impact on Outcomes. Results From the Australia and New Zealand Myeloma and Related Diseases Registry. Clin Lymphoma Myeloma Leuk. 2019;19:e415–e24. doi: 10.1016/j.clml.2019.05.010. [DOI] [PubMed] [Google Scholar]
- 5.Courant M, Orazio S, Monnereau A, Preterre J, Combe C, Rigothier C. Incidence, prognostic impact and clinical outcomes of renal impairment in patients with multiple myeloma: a population-based registry. Nephrol Dial Transpl. 2021;36:482–90. doi: 10.1093/ndt/gfz211. [DOI] [PubMed] [Google Scholar]
- 6.Blade J, Fernandez-Llama P, Bosch F, Montoliu J, Lens XM, Montoto S, et al. Renal failure in multiple myeloma: presenting features and predictors of outcome in 94 patients from a single institution. Arch Intern Med. 1998;158:1889–93. doi: 10.1001/archinte.158.17.1889. [DOI] [PubMed] [Google Scholar]
- 7.Knudsen LM, Hjorth M, Hippe E. Renal failure in multiple myeloma: reversibility and impact on the prognosis. Nordic Myeloma Study Group. Eur J Haematol. 2000;65:175–81. doi: 10.1034/j.1600-0609.2000.90221.x. [DOI] [PubMed] [Google Scholar]
- 8.Scheid C, Sonneveld P, Schmidt-Wolf IG, van der Holt B, el Jarari L, Bertsch U, et al. Bortezomib before and after autologous stem cell transplantation overcomes the negative prognostic impact of renal impairment in newly diagnosed multiple myeloma: a subgroup analysis from the HOVON-65/GMMG-HD4 trial. Haematologica. 2014;99:148–54. doi: 10.3324/haematol.2013.087585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dimopoulos MA, Richardson PG, Schlag R, Khuageva NK, Shpilberg O, Kastritis E, et al. VMP (Bortezomib, Melphalan, and Prednisone) is active and well tolerated in newly diagnosed patients with multiple myeloma with moderately impaired renal function, and results in reversal of renal impairment: cohort analysis of the phase III VISTA study. J Clin Oncol. 2009;27:6086–93. doi: 10.1200/JCO.2009.22.2232. [DOI] [PubMed] [Google Scholar]
- 10.Gonsalves WI, Leung N, Rajkumar SV, Dispenzieri A, Lacy MQ, Hayman SR, et al. Improvement in renal function and its impact on survival in patients with newly diagnosed multiple myeloma. Blood Cancer J. 2015;5:e296. doi: 10.1038/bcj.2015.20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Rajkumar SV, Dimopoulos MA, Palumbo A, Blade J, Merlini G, Mateos MV, et al. International Myeloma Working Group updated criteria for the diagnosis of multiple myeloma. Lancet Oncol. 2014;15:e538–48. doi: 10.1016/S1470-2045(14)70442-5. [DOI] [PubMed] [Google Scholar]
- 12.Sanders PW, Booker BB. Pathobiology of cast nephropathy from human Bence Jones proteins. J Clin Investig. 1992;89:630–9. doi: 10.1172/JCI115629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sanders PW. Mechanisms of light chain injury along the tubular nephron. J Am Soc Nephrol. 2012;23:1777–81. doi: 10.1681/ASN.2012040388. [DOI] [PubMed] [Google Scholar]
- 14.Ying WZ, Li X, Rangarajan S, Feng W, Curtis LM, Sanders PW. Immunoglobulin light chains generate proinflammatory and profibrotic kidney injury. J Clin Investig. 2019;129:2792–806. doi: 10.1172/JCI125517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ying WZ, Wang PX, Aaron KJ, Basnayake K, Sanders PW. Immunoglobulin light chains activate nuclear factor-kappaB in renal epithelial cells through a Src-dependent mechanism. Blood. 2011;117:1301–7. doi: 10.1182/blood-2010-08-302505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Yadav P, Sathick IJ, Leung N, Brown EE, Cook M, Sanders PW, et al. Serum free light chain level at diagnosis in myeloma cast nephropathy-a multicentre study. Blood Cancer J. 2020;10:28. doi: 10.1038/s41408-020-0295-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yadav P, Cockwell P, Cook M, Pinney J, Giles H, Aung YS, et al. Serum free light chain levels and renal function at diagnosis in patients with multiple myeloma. BMC Nephrol. 2018;19:178. doi: 10.1186/s12882-018-0962-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Visram A, Rajkumar SV, Kapoor P, Dispenzieri A, Lacy MQ, Gertz MA, et al. Monoclonal proteinuria predicts progression risk in asymptomatic multiple myeloma with a free light chain ratio >/=100. Leukemia. 2022;36:1429–31. doi: 10.1038/s41375-022-01529-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Drayson M, Begum G, Basu S, Makkuni S, Dunn J, Barth N, et al. Effects of paraprotein heavy and light chain types and free light chain load on survival in myeloma: an analysis of patients receiving conventional-dose chemotherapy in Medical Research Council UK multiple myeloma trials. Blood. 2006;108:2013–9. doi: 10.1182/blood-2006-03-008953. [DOI] [PubMed] [Google Scholar]
- 20.Leung N, Gertz MA, Zeldenrust SR, Rajkumar SV, Dispenzieri A, Fervenza FC, et al. Improvement of cast nephropathy with plasma exchange depends on the diagnosis and on reduction of serum free light chains. Kidney Int. 2008;73:1282–8. doi: 10.1038/ki.2008.108. [DOI] [PubMed] [Google Scholar]
- 21.Hutchison CA, Bradwell AR, Cook M, Basnayake K, Basu S, Harding S, et al. Treatment of acute renal failure secondary to multiple myeloma with chemotherapy and extended high cut-off hemodialysis. Clin J Am Soc Nephrol. 2009;4:745–54. doi: 10.2215/CJN.04590908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hutchison CA, Cockwell P, Stringer S, Bradwell A, Cook M, Gertz MA, et al. Early reduction of serum-free light chains associates with renal recovery in myeloma kidney. J Am Soc Nephrol. 2011;22:1129–36. doi: 10.1681/ASN.2010080857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bridoux F, Carron PL, Pegourie B, Alamartine E, Augeul-Meunier K, Karras A, et al. Effect of High-Cutoff Hemodialysis vs Conventional Hemodialysis on Hemodialysis Independence Among Patients With Myeloma Cast Nephropathy: A Randomized Clinical Trial. Jama. 2017;318:2099–110. doi: 10.1001/jama.2017.17924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Leung N, Bridoux F, Nasr SH. Monoclonal Gammopathy of Renal Significance. N Engl J Med. 2021;384:1931–41. doi: 10.1056/NEJMra1810907. [DOI] [PubMed] [Google Scholar]
- 25.Royal V, Leung N, Troyanov S, Nasr S, Ecotiere L, LeBlanc R, et al. Clinicopathologic predictors of renal outcomes in light chain cast nephropathy: a multicenter retrospective study. Blood. 2020;135:1833–46. doi: 10.1182/blood.2019003807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Leung N, Gertz M, Kyle RA, Fervenza FC, Irazabal MV, Eirin A, et al. Urinary albumin excretion patterns of patients with cast nephropathy and other monoclonal gammopathy-related kidney diseases. Clin J Am Soc Nephrol. 2012;7:1964–8. doi: 10.2215/CJN.11161111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Dimopoulos MA, Terpos E, Chanan-Khan A, Leung N, Ludwig H, Jagannath S, et al. Renal impairment in patients with multiple myeloma: a consensus statement on behalf of the International Myeloma Working Group. J Clin Oncol. 2010;28:4976–84. doi: 10.1200/JCO.2010.30.8791. [DOI] [PubMed] [Google Scholar]
- 28.Jagannath S, Barlogie B, Berenson JR, Singhal S, Alexanian R, Srkalovic G, et al. Bortezomib in recurrent and/or refractory multiple myeloma. Initial clinical experience in patients with impared renal function. Cancer. 2005;103:1195–200. doi: 10.1002/cncr.20888. [DOI] [PubMed] [Google Scholar]
- 29.Ludwig H, Adam Z, Hajek R, Greil R, Tothova E, Keil F, et al. Light chain-induced acute renal failure can be reversed by bortezomib-doxorubicin-dexamethasone in multiple myeloma: results of a phase II study. J Clin Oncol. 2010;28:4635–41. doi: 10.1200/JCO.2010.28.1238. [DOI] [PubMed] [Google Scholar]
- 30.Sonneveld P, Schmidt-Wolf IG, van der Holt B, El Jarari L, Bertsch U, Salwender H, et al. Bortezomib induction and maintenance treatment in patients with newly diagnosed multiple myeloma: results of the randomized phase III HOVON-65/ GMMG-HD4 trial. J Clin Oncol. 2012;30:2946–55. doi: 10.1200/JCO.2011.39.6820. [DOI] [PubMed] [Google Scholar]
- 31.Bridoux F, Arnulf B, Karlin L, Blin N, Rabot N, Macro M, et al. Randomized Trial Comparing Double Versus Triple Bortezomib-Based Regimen in Patients With Multiple Myeloma and Acute Kidney Injury Due to Cast Nephropathy. J Clin Oncol. 2020;38:2647–57. doi: 10.1200/JCO.20.00298. [DOI] [PubMed] [Google Scholar]
- 32.Gupta N, Hanley MJ, Harvey RD, Badros A, Lipe B, Kukreti V, et al. A pharmacokinetics and safety phase 1/1b study of oral ixazomib in patients with multiple myeloma and severe renal impairment or end-stage renal disease requiring haemodialysis. Br J Haematol. 2016;174:748–59. doi: 10.1111/bjh.14125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Quach H, White D, Spencer A, Ho PJ, Bhutani D, White M, et al. Pharmacokinetics and safety of carfilzomib in patients with relapsed multiple myeloma and end-stage renal disease (ESRD): an open-label, single-arm, phase I study. Cancer Chemother Pharm. 2017;79:1067–76. doi: 10.1007/s00280-017-3287-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Badros AZ, Vij R, Martin T, Zonder JA, Kunkel L, Wang Z, et al. Carfilzomib in multiple myeloma patients with renal impairment: pharmacokinetics and safety. Leukemia. 2013;27:1707–14. doi: 10.1038/leu.2013.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yui JC, Van Keer J, Weiss BM, Waxman AJ, Palmer MB, D’Agati VD, et al. Proteasome inhibitor associated thrombotic microangiopathy. Am J Hematol. 2016;91:E348–52. doi: 10.1002/ajh.24447. [DOI] [PubMed] [Google Scholar]
- 36.Fotiou D, Roussou M, Gakiopoulou C, Psimenou E, Gavriatopoulou M, Migkou M, et al. Carfilzomib-associated renal toxicity is common and unpredictable: a comprehensive analysis of 114 multiple myeloma patients. Blood Cancer J. 2020;10:109. doi: 10.1038/s41408-020-00381-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Eriksson T, Hoglund P, Turesson I, Waage A, Don BR, Vu J, et al. Pharmacokinetics of thalidomide in patients with impaired renal function and while on and off dialysis. J Pharm Pharm. 2003;55:1701–6. doi: 10.1211/0022357022241. [DOI] [PubMed] [Google Scholar]
- 38.Dimopoulos MA, Terpos E, Goldschmidt H, Alegre A, Mark T, Niesvizky R. Treatment with lenalidomide and dexamethasone in patients with multiple myeloma and renal impairment. Cancer Treat Rev. 2012;38:1012–9. doi: 10.1016/j.ctrv.2012.02.009. [DOI] [PubMed] [Google Scholar]
- 39.Gay F, Mina R, Troia R, Bringhen S. Pharmacokinetic evaluation of pomalidomide for the treatment of myeloma. Expert Opin Drug Metab Toxicol. 2013;9:1517–27. doi: 10.1517/17425255.2013.827169. [DOI] [PubMed] [Google Scholar]
- 40.Fakhouri F, Guerraoui H, Presne C, Peltier J, Delarue R, Muret P, et al. Thalidomide in patients with multiple myeloma and renal failure. Br J Haematol. 2004;125:96–7. doi: 10.1111/j.1365-2141.2004.04875.x. [DOI] [PubMed] [Google Scholar]
- 41.Tosi P, Zamagni E, Ronconi S, Benni M, Motta MR, Rizzi S, et al. Safety of autologous hematopoietic stem cell transplantation in patients with multiple myeloma and chronic renal failure. Leukemia. 2000;14:1310–3. doi: 10.1038/sj.leu.2401819. [DOI] [PubMed] [Google Scholar]
- 42.Ludwig H, Rauch E, Kuehr T, Adam Z, Weissmann A, Kasparu H, et al. Lenalidomide and dexamethasone for acute light chain-induced renal failure: a phase II study. Haematologica. 2015;100:385–91. doi: 10.3324/haematol.2014.115204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Bridoux F, Chen N, Moreau S, Arnulf B, Moumas E, Abraham J, et al. Pharmacokinetics, safety, and efficacy of lenalidomide plus dexamethasone in patients with multiple myeloma and renal impairment. Cancer Chemother Pharm. 2016;78:173–82. doi: 10.1007/s00280-016-3068-9. [DOI] [PubMed] [Google Scholar]
- 44.Mikhael J, Manola J, Dueck AC, Hayman S, Oettel K, Kanate AS, et al. Lenalidomide and dexamethasone in patients with relapsed multiple myeloma and impaired renal function: PrE1003, a PrECOG study. Blood Cancer J. 2018;8:86. doi: 10.1038/s41408-018-0110-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Dimopoulos M, Weisel K, van de Donk N, Ramasamy K, Gamberi B, Streetly M, et al. Pomalidomide Plus Low-Dose Dexamethasone in Patients With Relapsed/Refractory Multiple Myeloma and Renal Impairment: Results From a Phase II Trial. J Clin Oncol. 2018;36:2035–43. doi: 10.1200/JCO.2017.76.1742. [DOI] [PubMed] [Google Scholar]
- 46.Voorhees PM, Kaufman JL, Laubach J, Sborov DW, Reeves B, Rodriguez C, et al. Daratumumab, lenalidomide, bortezomib, and dexamethasone for transplant-eligible newly diagnosed multiple myeloma: the GRIFFIN trial. Blood. 2020;136:936–45. doi: 10.1182/blood.2020005288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Facon T, Kumar S, Plesner T, Orlowski RZ, Moreau P, Bahlis N, et al. Daratumumab plus Lenalidomide and Dexamethasone for Untreated Myeloma. N Engl J Med. 2019;380:2104–15. doi: 10.1056/NEJMoa1817249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Moreau P, Attal M, Hulin C, Arnulf B, Belhadj K, Benboubker L, et al. Bortezomib, thalidomide, and dexamethasone with or without daratumumab before and after autologous stem-cell transplantation for newly diagnosed multiple myeloma (CASSIOPEIA): a randomised, open-label, phase 3 study. Lancet. 2019;394:29–38. doi: 10.1016/S0140-6736(19)31240-1. [DOI] [PubMed] [Google Scholar]
- 49.Jeyaraman P, Bhasin A, Dayal N, Pathak S, Naithani R. Daratumumab in dialysis-dependent multiple myeloma. Blood Res. 2020;55:65–7. doi: 10.5045/br.2020.55.1.65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Moreau P, Dimopoulos MA, Mikhael J, Yong K, Capra M, Facon T, et al. Isatuximab, carfilzomib, and dexamethasone in relapsed multiple myeloma (IKEMA): a multicentre, open-label, randomised phase 3 trial. Lancet. 2021;397:2361–71. doi: 10.1016/S0140-6736(21)00592-4. [DOI] [PubMed] [Google Scholar]
- 51.Feest TG, Burge PS, Cohen SL. Successful treatment of myeloma kidney by diuresis and plasmaphoresis. Br Med J. 1976;1:503–4. doi: 10.1136/bmj.1.6008.503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Zucchelli P, Pasquali S, Cagnoli L, Ferrari G. Controlled plasma exchange trial in acute renal failure due to multiple myeloma. Kidney Int. 1988;33:1175–80. doi: 10.1038/ki.1988.127. [DOI] [PubMed] [Google Scholar]
- 53.Johnson WJ, Kyle RA, Pineda AA, O’Brien PC, Holley KE. Treatment of renal failure associated with multiple myeloma. Plasmapheresis, hemodialysis, and chemotherapy. Arch Intern Med. 1990;150:863–9. doi: 10.1001/archinte.1990.00390160111022. [DOI] [PubMed] [Google Scholar]
- 54.Clark WF, Stewart AK, Rock GA, Sternbach M, Sutton DM, Barrett BJ, et al. Plasma exchange when myeloma presents as acute renal failure: a randomized, controlled trial. Ann Intern Med. 2005;143:777–84. doi: 10.7326/0003-4819-143-11-200512060-00005. [DOI] [PubMed] [Google Scholar]
- 55.Hutchison CA, Cockwell P, Reid S, Chandler K, Mead GP, Harrison J, et al. Efficient removal of immunoglobulin free light chains by hemodialysis for multiple myeloma: in vitro and in vivo studies. J Am Soc Nephrol. 2007;18:886–95. doi: 10.1681/ASN.2006080821. [DOI] [PubMed] [Google Scholar]
- 56.Hutchison CA, Cockwell P, Moroz V, Bradwell AR, Fifer L, Gillmore JD, et al. High cutoff versus high-flux haemodialysis for myeloma cast nephropathy in patients receiving bortezomib-based chemotherapy (EuLITE): a phase 2 randomised controlled trial. Lancet Haematol. 2019;6:e217–e228. doi: 10.1016/S2352-3026(19)30014-6. [DOI] [PubMed] [Google Scholar]
- 57.Burnette BL, Leung N, Rajkumar SV. Renal improvement in myeloma with bortezomib plus plasma exchange. N Engl J Med. 2011;364:2365–6. doi: 10.1056/NEJMc1101834. [DOI] [PubMed] [Google Scholar]
- 58.Kastritis E, Palladini G, Minnema MC, Wechalekar AD, Jaccard A, Lee HC, et al. Daratumumab-Based Treatment for Immunoglobulin Light-Chain Amyloidosis. N Engl J Med. 2021;385:46–58. doi: 10.1056/NEJMoa2028631. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
This is a current treatment algorithm. There are no new data generated for this paper and data sharing is not applicable.


