Abstract
Background:
Patients on continuous flow ventricular assist devices (CF-VADs) are at high risk for development of Acquired von-Willebrand Syndrome (AVWS) and non-surgical bleeding. von Willebrand Factor (vWF) plays an essential role in maintaining hemostasis via platelet binding to the damaged endothelium to facilitate coagulation. In CF-VAD patients, degradation of vWF into low MW multimers that are inefficient in facilitating coagulation occurs and has been primarily attributed to the supraphysiological shear stress associated with the CF-VAD impeller.
Methods:
In this review, we evaluate information from literature regarding unravelling behavior of surface immobilized vWF under pulsatile and continuous flow pertaining to: (A) the process of arterial endothelial vWF production and release into circulation, (B) the critical shear stress required to unravel surface bound vs. soluble vWF which leads to degradation, and (C) the role of pulsatility in on the production and degradation of vWF.
Results and Conclusion:
When taken together, this data suggests that the loss of pulsatility and its impact on arterial endothelial cells plays an important role in production, release, unravelling and proteolytic degradation of vWF into low MW multimers contributing to development of AVWS and that restoration of pulsatility can potentially mitigate this issue by preventing AVWS and minimizing risk of non-surgical bleeding.
Left Ventricular assist devices (LVADs) are standard therapy for patients with advanced heart failure (HF) refractory to medical therapy.1 Categorized based on the type of flow, both pulsatile flow VADs (PF-VADs) and continuous flow VADs (CF-VADs) provide hemodynamic support and improve the quality of life and functional capacity of patients.2,3 Clinically, CF-VAD implantations are preferred over PF-VADs owing to advantages in size, reliability, energy efficiency, and ease of implant. However, CF-VADs provide non-physiological stimuli that are associated with pump thrombosis (due to platelet activation as a result of supraphysiological shear 4,5) and non-surgical bleeding, particularly in the gastrointestinal (GI) tract.6,7 While still major challenges, hemolysis and pump thrombosis have been mitigated through pump designs which reduce the shear and residence times, proper positioning of the outflow cannula, and anticoagulant therapies.8 Non-surgical bleeding remains a frequent complication in patients supported with CF-VADs.9 Despite the fact that bleeding is a multifactorial event, evidence links GI bleeding and development of acquired von Willebrand syndrome (AVWS), however it is important to note that not all patients that develop AVWS experience non-surgical bleeding.10,11 In AVWS, the large globular plasma glycoprotein vWF, which plays a critical role in facilitating coagulation through platelet adhesion, undergoes cleavage into low molecular weight (MW) multimers, which bind poorly to platelets and sub-endothelial collagen.12 Quantitative evaluation of vWF multimers shows that CF-VAD patients often display a significant reduction in plasma vWF levels, and increased vWF degradation.13 While decreased circulating high MW vWF multimers does not always lead to GI bleeding, patients experiencing GI bleeds have significantly lower levels of vWF.10 The requisite anticoagulation therapy CF-VAD patients undergo complicates this pathophysiology. However, a clinical trial reported high levels of bleeding in CF-VAD patients not receiving systemic anticoagulation.14 Despite the complex nature of this process, vWF degradation appears to play an important role in the development of non-surgical bleeding.
vWF degradation into low MW multimers requires shear mediated unravelling, resulting in the exposure of the scissile bond located within the A2 domain for proteolytic cleavage via metalloprotease A Disintegrin-like and Metalloprotease ThromboSpondin-13 (ADAMTS13).15-17 Studies using mock flow loops and vortex mixers confirm that exposure of vWF to supraphysiological shear rates in the presence of ADAMTS13 (> 10,000 s−1) results in unraveling, proteolytic degradation, and breakdown of vWF into low MW multimers.18 While considering shear stress, it is also important to consider the type of flow regime. High shear stress associated with turbulence in the CF-VAD promotes vWF unravelling and degradation19 whereas; high supraphysiological shear stress associated with CF-VADs using microfluidic devices where the flow is laminar did not result in excessive cleavage or decreased function of von Willebrand factor in the presence of ADAMTS13.20 While AVWS development has primarily been considered in the context of these supraphysiological shear rates observed within the CF-VADs,21 there are reasons to believe that the exposure to supraphysiological shear may not be the sole contributor. The short (30-200 milliseconds) residence time for vWF within the high shear stress areas of the CF-VAD, even when the flow is turbulent, may be insufficient for the unraveling of vWF required for degradation. Further, high supraphysiological shear within CF-VADs is limited to localized regions (< 25% of CF-VAD volume)22 and newer designs have reduced impeller shear rates and pump residence time.23 Notably, such improvements to the Heartmate III device (Abbott Laboratories, Chicago, IL) resulted in an increased proportion of patients with intact high MW vWF multimers (from 9% to 25%). However, nearly 75% of Heartmate III patients still developed AVWS.24
Pulsatility is an intrinsic aspect of arterial circulation. Arterial endothelial cells transduce pulsatile pressure, shear, and stretch to maintain normal endothelial function, and changes in pulsatility affects arterial endothelial cell function.25 Further, arterial endothelial cells produce and secrete vWF and represent the primary location of exposure of vWF to shear associated with blood flow.26 Early LVAD design improvements focused on minimizing hemolysis and thrombosis, to improve patient safety and survival with CF-VADs despite the loss of pulsatility. Frequent incidences of non-surgical bleeding with CF-VAD usage have renewed efforts to understand the consequences of loss of physiologic pulsatility and identification of mechanisms that possibly contribute to AVWS. Based on what we know from the process of arterial endothelial vWF production and release into circulation, theoretical and experimental studies establishing minimum shear thresholds for unravelling of surface bound vs. soluble vWF, the relationship between level of pulsatility and vWF degradation,27 and our recent study evaluating the unravelling behavior of surface immobilized vWF under pulsatile and continuous flow;28 we postulate that the loss of pulsatility experienced by the arterial endothelium is highly significant in the context of vWF production, release, unravelling, and proteolytic degradation into low MW multimers. A detailed discussion of each of these factors follows.
Production and Release of Endothelial vWF into Circulation
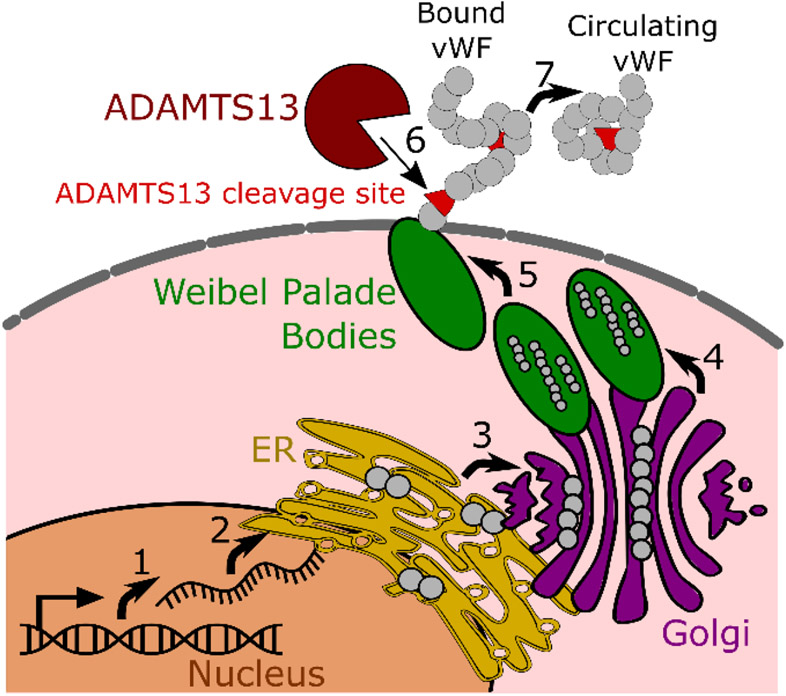
vWF is a plasma glycoprotein, synthesized by endothelial cells, stored in Weibel-Palade bodies (WPBs) or by activated platelets, and released into circulation.29 Endothelial cells ensure basal levels of vWF via constitutive, stimulation-independent secretion of vWF from WPBs (Fig. 1, 1-6) and an increase in circulating vWF occurs in response to stimuli like vascular injury.30 Newly released vWF from WPBs is in the form of ultra-large multimers, which remain anchored to the endothelial surface and unravels under shear stress until being proteolytically released by ADAMTS13 (Fig. 1 6-7).15-17 While ADAMTS13 can bind to globular vWF, it cannot cleave it, as the scissile bond located on the A2 domain and other potential cleavage sites are hidden in its folded, globular form. Therefore fluid shear stress from blood flow is an important mediator of arterial homeostasis and maintenance of basal vWF levels. Change from normal pulsatile flow to continuous flow or ‘static’ no-flow conditions results in a sharp decrease in vWF release from endothelial cells.27,31 While it is not entirely clear if pulsatile flow increases transcriptionally regulated production of vWF, pulsatile stretch may enhance release of vWF stored in WPBs.32
Figure 1:
Schematic representation of high MW vWF production and secretion. Transcription (1) and translation (2) of vWF monomers occurs in the nucleus. Dimerization (3) occurs in the endoplasmic reticulum (ER), followed by multimerization in the Golgi. vWF multimers are packaged into Weibel Palade bodies (4), from which vWF is secreted at the cell membrane (5). High MW VWF is cleaved from endothelial cells by ADAMTS-13 (6) leading to circulation (7).
In normal physiology, binding and proteolytic cleavage of vWF by ADAMTS13 occurs at three distinct locations: (1) Surface of the arterial endothelium: the endothelial cells that make up the arterial vessel walls and are an important source of vWF. vWF secreted from WPBs that are produced within the arterial endothelial cells remains anchored to the arterial endothelium and requires proteolytic cleavage for release as ultra large vWF molecules into circulation, (2) In the blood stream: Globular vWF molecules in free circulation or in CF-VAD may unravel under elevated shear, exposing binding sites and the scissile bond resulting in cleavage, and (3) Sites of vascular injury: following rupture of any vessel within the body, large vWF multimers present in the blood stream bind to activated platelets and sub-endothelial collagen beneath the damaged endothelial layer and unravel in response to shear from blood flow. Unravelling exposes both the A2 domain and additional binding exosites and cleavage locations, resulting in degradation of vWF into smaller fibrils with significantly reduced adhesive potential to prevent whole vessel occlusion.
Shear Stress: Surface-bound vs. Globular vWF in Bulk Flow
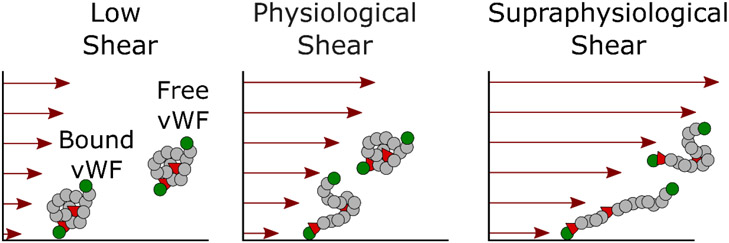
Vessel shear stress varies as a function of the radial distance from the center of the vessel, with surface immobilized molecules experiencing significantly greater shear than molecules suspended in the fluid. Simulation and experimental studies confirm that vWF can unfold in response to shear both in solution and when immobilized on substrates. However, the shear threshold for unravelling of surface-bound vs. soluble vWF may differ significantly (Fig. 2). Molecular simulations suggest that the critical shear rate threshold for unravelling of vWF in solution (unbound) can be ~ 50-fold greater than for vWF immobilized to a surface.33 Experimental studies using fluorescence microscopy,34 small-angle neutron scattering (SANS)35 and fluorescence spectroscopy36 confirm that the critical shear rate for vWF unravelling was > 5000 s−1 in solution.
Figure 2:
Shear mediated unravelling behavior of surface bound and free vWF under low, physiological and supraphysiological flow.
The critical shear rates for unravelling of surface bound vWF has been experimentally measured in the range of 1000 to 3500 s−1 based on the method of immobilization. 37,38 Dong et. al. showed that newly released vWF from endothelial cells undergo elongation and string formation at significantly lower shear rates (< 500 s−1) than vWF molecules immobilized with different chemistries.39 This study also found that under flow conditions, endothelial bound vWF was cleaved within seconds to minutes in the presence of normal plasma (~ 100% ADAMTS-13 activity) in contrast to plasma from patients with thrombotic thrombocytopenic purpura (TTP) (~ 0% to 10% ADAMTS-13 activity) that resulted in endothelial bound strings of vWF. These results demonstrate vWF unravelling occurs at significantly lower shear rates for endothelial bound vWF compared to surface bound vWF and vWF in bulk flow.
Unravelling Behavior of Surface Immobilized vWF under Continuous and Pulsatile Flow
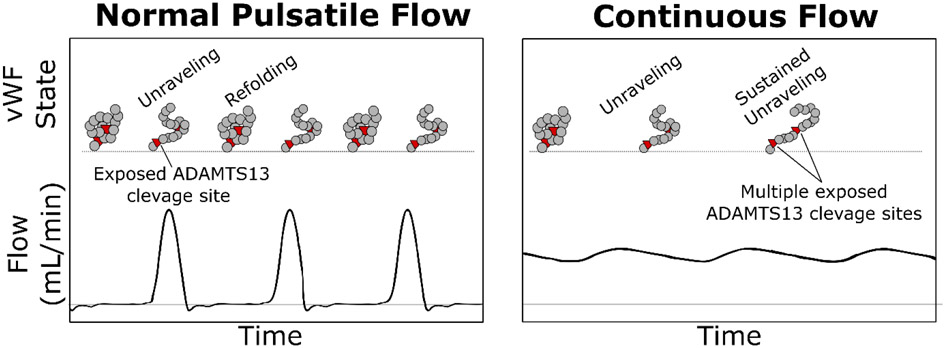
Normal pulsatile flow as seen in arterial vessels is associated with higher peak shear stress but also provides a period of zero/low shear. Continuous flow is associated with a constant shear stress albeit at a lower magnitude than the peak seen with pulsatile flow. In-vitro, unravelling of vWF has been almost exclusively studied under conditions of continuous flow. Our group was the first to evaluate the unravelling behavior of vWF molecules immobilized using avidin-biotin chemistry under conditions of pulsatile flow. Our results demonstrated that the unravelling lengths of vWF are significantly reduced under conditions of pulsatile flow than with continuous flow. Increasing the magnitude of pulsatility up to physiological levels reduces unravelling lengths of vWF molecules, and the frequency of pulsatility (20-60 cycles/min) does not have a major effect on the maximum or minimum unravelling lengths.28 These direct vWF molecular observations suggest that vWF conformation changes related to recoiling are on the order of approximately 1 second. Despite the higher peak shear stress, pulsatile flow does not result in significant unraveling of surface bound vWF, which minimizes exposure of the scissile bond within the A2 domain for proteolytic cleavage by ADAMTS13. Conversely, with continuous flow, the constant exposure to shear stress causes extensive and sustained unravelling of vWF leading to greater unraveling lengths, potentially increasing exposure of the scissile bond within the A2 domain for ADAMTS13 mediated proteolytic degradation of vWF into low MW multimers (Fig. 3).
Figure 3:
Unravelling and recoiling of surface-bound vWF under pulsatile and continued unravelling of vWF under continuous flow.
Significance of the Arterial Endothelium in vWF Production and Degradation
In summary, data from published studies by others and our recent work strongly indicate that the arterial endothelium is a critical location where vWF degradation occurs and is a major contributor to the development of AVWS. Specific evidence includes the following 6 observations: (1) Newly released ultra large endothelial vWF is bound to the endothelium prior to release in circulation and requires ADAMTS13 for cleavage from the endothelial surface and release into circulation; this event precedes interaction of vWF in circulation with the CF-VAD impeller.15-17 (2) The critical shear rate for unravelling of newly released endothelial-bound vWF is < 500 s−1 , which is significantly lower than shear rates at which globular soluble vWF unravels.39 (3) Continuous flow results in greater unravelling of surface-bound vWF compared to pulsatile flow. Increase in the magnitude of pulsatile flow to normal pulsatility results in decreased vWF unravelling lengths which suggests that vWF at the endothelial surface undergoes extensive and sustained unravelling under minimally pulsatile flow, exposing additional ADAMTS13 binding sites.28 (4) Recent studies with mock flow loops and swine models show a dose-response relationship between degree of pulsatility and vWF; both high MW multimers and vWF collagen-binding activity were boosted with increased levels of pulsatility (but with similar average shear stress), with a strong direct correlation between pulse pressure and high MW multimers.27 (5) In-vitro studies suggest that endothelial production and/or release of vWF is suppressed under continuous flow, and basal levels of vWF are restored following return to normal pulsatile flow.27 (6) Published studies and our patient data show that circulating vWF levels decrease by > 40% in patients 1-3 months following CF-VAD placement.
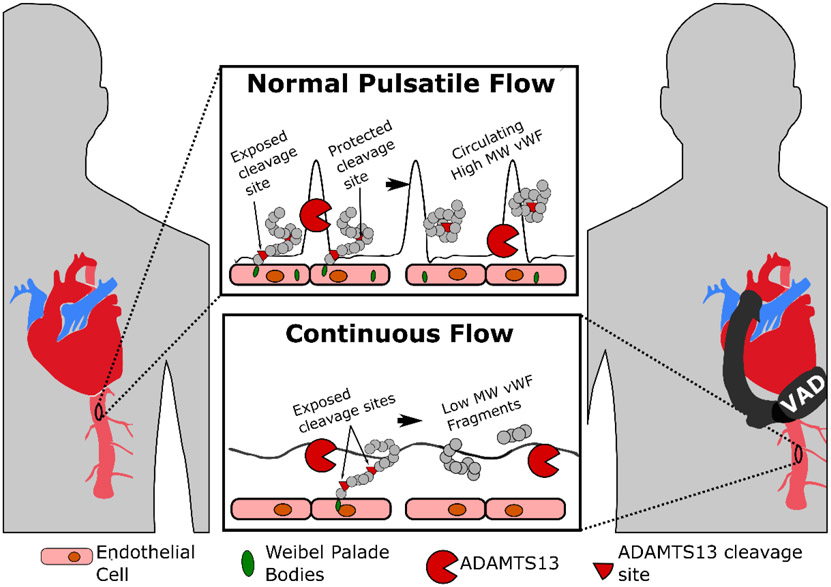
These observations strongly suggest that the loss or marked decrease in pulsatility associated with CF-VAD support directly affects both the degradation of newly released ultra large vWF from endothelial cells and the production/release of endothelial vWF. Specifically, newly released ultra large vWF attached to the surface of the arterial endothelial cells undergoes only partial unravelling and refolding with normal pulsatile flow and is released following cleavage by ADAMTS13 as high MW multimers into circulation. The partial unravelling behavior potentially protects the scissile bond within the vWF A2 domain, preserving vWF in its high MW form. However, under continuous flow associated with CF-VAD support, endothelial bound vWF undergoes extensive unravelling, exposing the scissile bond within the vWF A2 domain and potentially additional cleavage sites for ADAMTS13 to facilitate proteolytic degradation into low MW vWF multimers released into circulation and are inefficient in preventing bleeding (Fig. 4).
Figure 4: Proposed Mechanism of AVWS development:
Endothelial vWF requires cleavage by ADAMTS13 for vWF release into circulation. Under pulsatile flow, the unraveling is limited, resulting in release of high MW vWF whereas under continuous flow, sustained, unravelling results in exposure of the scissile bond in the A2 domain resulting in ADAMTS13 mediated proteolytic degradation into low MW multimers.
Additionally, the change from pulsatile to continuous flow either directly or indirectly has an inhibitory effect on endothelial release of vWF, which can be restored to basal levels following reintroduction of pulsatility. Based on published results that suggest that a pulse pressure < 35 mm Hg is a major risk factor for development of non-surgical bleeding,40 we suspect that restoration pulse pressure ≥ 35 mmHg is essential.
Implications for Current CF-VADs and the Design of the Next Generation of CF-VADs
CF-VADs continuously unload the native ventricle and significantly diminishes arterial pressure pulsatility. Flow modulation is a potential option to reintroduce pulsatility and been proposed both in literature41-44 and introduced into clinical practice to augment pulsatility (Lavare cycle in HeartWare HVAD),45,46 facilitate opening of the aortic valve (intermittent low speed in Jarvik 2000)47 or to improve washing (intrinsic pulsatility mechanism with Heartmate 3, (i.e.) 1 pulse every 2 seconds when operating above 4000 rpm).48 While Heartmate 3, currently the only FDA approved CF-VAD, is capable of intermittent speed modulation to provide artificial pulsatility49,50 the intermittent speed reduction is not representative of physiological pulsatility and clinical studies show limited benefit in preventing non-surgical bleeding.24 In addition to Heartmate 3, there are two other pumps, the CorWave LVAD, which relies on a proprietary ‘wave membrane’ technology and the EVAHEART®2 CF-VAD, which uses a hydraulically levitated “open vane” impeller. Restoration of near-physiological levels of pulsatility is possible with the Heartmate 3 and the EVAHEART®2 via rapid changed in pump speed51-53 and in the CorWave Membrane LVAD with changes in the frequency of actuation of the membrane.54 Unlike the native ventricle which can generate peak flows of 30-40 L/min, these devices are limited to peak flow rates of ~ 10 L/min which limits the level of pulsatility that can be restored. Therefore, pulsatility can be restored in two ways: (1) Synchronous Flow Modulation (SYN): where changes in pump speed are accomplished in synchrony with native myocardial contractions.55-57 The only variable that can be modulated is the pulse pressure. These devices (Heartmate 3) are limited to peak flow rates of ~10 L/min, which limits the pulse pressure that can be generated with synchronous flow modulation to < 20 mmHg.58 Pulsatile devices in development like the VentriFlo and NuPulseCV iVAS generally produce higher levels of arterial pulsatility than CF-VADs at native heart rates (> 60 bpm), and are capable of faster displacement, which can be used to enhance arterial pressure pulsatility and reduce vWF degradation.(2) Asynchronous Flow Modulation (ASYN): where changes in pump speed are accomplished independent of the native heart rate.59,60 Both pulse pressure and frequency can be modulated to achieve desired levels of pulsatility. Asynchronous modulation at lower VAD flow modulation rates (≤ 25 bpm) can generate near physiological pulse pressures (~40 mmHg) even with peak flow rates of 10 L/min.58 While all three CF-VADs can restore some level of pulsatility, given their peak flow rate limitations, it is unclear as to the levels of pulse pressure and pulse frequency that need to be restored to prevent vWF dysregulation.
Current flow modulation approaches like Lavare cycle augment pulsatility but is sporadic with prolonged periods of diminished pulsatility that can potentially cause vWF degradation and affect the physiologic release of vWF from endothelial cells. Restoration of normal levels of pulse pressures (>35 mmHg) on a consistent basis can only be accomplished at lower pulse frequencies (≤ 25 bpm). A recent publication (Previous AO publication) demonstrated that pulsatility delivered at lower pulse frequencies (20 bpm) did not adversely affect vWF unraveling (similar vWF lengths) compared to normal frequencies. Flow modulation with CF-VADs can cause increases in peak shear rates but previous studies have not demonstrated differences in time averaged hemolysis or cf_LVAD induced vWF degradation44,61 compared to constant pump speed operation. Thus, flow modulation approaches that consistently provide physiologic levels of pulsatility may not only minimize vWF destruction but also maintain physiologic release. Additionally, Lavare cycle and flow modulation with HeartMate 3 have been demonstrated to increase ventricular intracavity washing and washout of the aortic root,45,62 potentially reducing thrombisis risk. Flow modulation to produce physiologic levels of pulsatility can not only further augment intraventricular and aortic washout, but has the potential to improve intrapump washing. However, blood pumps need to be designed to avoid areas of blood stasis especially during low flow rates during flow modulation. Studies evaluating the effects of various combinations of pulse pressure and pulse frequency on vWF production and degradation can potentially provide important insights into minimal levels of pulsatility required and optimal pulsatility profiles that can minimize surface bound vWF degradation and restore basal vWF production.
Conclusions
In summary, current literature on endothelial vWF production and release, suggests a critical role for the arterial endothelium, especially following the loss of pulsatility, in the development of AVWS. Further studies are necessary to elucidate the exact mechanisms through which the complete or partial loss of pulsatility affects various events associated with the production and degradation of vWF. Knowledge gained from these studies will provide insights that can help identify critical levels of pulsatility (pulse pressure, pulse frequency) necessary to maintain normal levels of vWF production and minimize degradation of endothelial bound vWF. These findings can potentially inform viable strategies to reintroduce pulsatility using current CF-VADs and guide the development of the next generation VADs to prevent AVWS, which is a major contributing factor for the development of non-surgical bleeding.
ACKNOWLEDGEMENTS
The authors would like to acknowledge funding support from the National Institute of Health (Grant # R01HL151663) and the National Science Foundation (Grant # 2004475). Ian C. Berg was supported by a T32 training grant from the National Institute of Health (Grant # T32HL129948)
Non-Standard Abbreviations
- ADAMTS13
A Disintegrin-like and Metalloprotease ThromboSpondin-13
- ASYN
asynchronous flow modulation
- AVWS
acquired von-willebrand syndrome
- CF-VADs
continuous flow ventricular assist devices
- ER
endoplasmic reticulum
- GI
gastrointestinal
- HF
heart failure
- LVADs
left ventricular assist devices
- MW
molecular weight
- PF-VADs
pulsatile-flow ventricular assist devices
- SYN
synchronous flow modulation
- WPBs
weibel-palade bodies
- vWF
von Willebrand Factor
Footnotes
CONFLICTS OF INTEREST
None Declared.
REFERENCES
- 1.McLarty A Mechanical Circulatory Support and the Role of LVADs in Heart Failure Therapy. Clinical Medicine Insights. Cardiology 9, 1–5, doi: 10.4137/CMC.s19694 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Thohan V et al. Cellular and hemodynamics responses of failing myocardium to continuous flow mechanical circulatory support using the DeBakey-Noon left ventricular assist device: a comparative analysis with pulsatile-type devices. The Journal of heart and lung transplantation : the official publication of the International Society for Heart Transplantation 24, 566–575, doi: 10.1016/j.healun.2004.02.017 (2005). [DOI] [PubMed] [Google Scholar]
- 3.Slaughter MS et al. Advanced heart failure treated with continuous-flow left ventricular assist device. The New England journal of medicine 361, 2241–2251, doi: 10.1056/NEJMoa0909938 (2009). [DOI] [PubMed] [Google Scholar]
- 4.Chen Z et al. Flow features and device-induced blood trauma in CF-VADs under a pulsatile blood flow condition: A CFD comparative study. Int J Numer Method Biomed Eng 34, 10.1002/cnm.2924, doi: 10.1002/cnm.2924 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Fraser KH, Zhang T, Taskin ME, Griffith BP & Wu ZJ A quantitative comparison of mechanical blood damage parameters in rotary ventricular assist devices: shear stress, exposure time and hemolysis index. J Biomech Eng 134, 081002–081002, doi: 10.1115/1.4007092 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chen Z, Sun A, Wang H, Fan Y & Deng X Non-physiological shear stress-induced blood damage in ventricular assist device. Medicine in Novel Technology and Devices 3, 100024, doi: 10.1016/j.medntd.2019.100024 (2019). [DOI] [Google Scholar]
- 7.Cushing K & Kushnir V Gastrointestinal Bleeding Following LVAD Placement from Top to Bottom. Digestive diseases and sciences 61, 1440–1447, doi: 10.1007/s10620-016-4123-4 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Fraser KH, Zhang T, Taskin ME, Griffith BP & Wu ZJ A quantitative comparison of mechanical blood damage parameters in rotary ventricular assist devices: shear stress, exposure time and hemolysis index. J Biomech Eng 134, 081002, doi: 10.1115/1.4007092 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Eckman PM & John R Bleeding and thrombosis in patients with continuous-flow ventricular assist devices. Circulation 125, 3038–3047, doi: 10.1161/circulationaha.111.040246 (2012). [DOI] [PubMed] [Google Scholar]
- 10.Crow S et al. Acquired von Willebrand syndrome in continuous-flow ventricular assist device recipients. Ann Thorac Surg 90, 1263–1269; discussion 1269, doi: 10.1016/j.athoracsur.2010.04.099 (2010). [DOI] [PubMed] [Google Scholar]
- 11.Uriel N et al. Acquired von Willebrand Syndrome After Continuous-Flow Mechanical Device Support Contributes to a High Prevalence of Bleeding During Long-Term Support and at the Time of Transplantation. Journal of the American College of Cardiology 56, 1207–1213, doi: 10.1016/j.jacc.2010.05.016 (2010). [DOI] [PubMed] [Google Scholar]
- 12.Franchini M & Mannucci PM Acquired von Willebrand syndrome: focused for hematologists. Haematologica 105, 2032–2037, doi: 10.3324/haematol.2020.255117 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bartoli CR Pathologic von Willebrand factor degradation is a major contributor to left ventricular assist device-associated bleeding: pathophysiology and evolving clinical management. Annals of Cardiothoracic Surgery 10, 389–392 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Katz JN et al. Safety of reduced anti-thrombotic strategies in HeartMate II patients: A one-year analysis of the US-TRACE Study. The Journal of Heart and Lung Transplantation 34, 1542–1548, doi: 10.1016/j.healun.2015.06.018 (2015). [DOI] [PubMed] [Google Scholar]
- 15.Dong JF et al. ADAMTS-13 metalloprotease interacts with the endothelial cell-derived ultra-large von Willebrand factor. J Biol Chem 278, 29633–29639, doi: 10.1074/jbc.M301385200 (2003). [DOI] [PubMed] [Google Scholar]
- 16.Dong JF et al. ADAMTS-13 rapidly cleaves newly secreted ultralarge von Willebrand factor multimers on the endothelial surface under flowing conditions. Blood 100, 4033–4039, doi: 10.1182/blood-2002-05-1401 (2002). [DOI] [PubMed] [Google Scholar]
- 17.Jin S et al. Proteolytic Cleavage of Endothelial Cell-Bound Von Willebrand Factor Polymers by ADAMTS13 in the Absence of Flow Shear Stress. Blood 112, 3913, doi: 10.1182/blood.V112.11.3913.3913 (2008). [DOI] [Google Scholar]
- 18.Bartoli CR et al. Inhibition of ADAMTS-13 by Doxycycline Reduces von Willebrand Factor Degradation During Supraphysiological Shear Stress: Therapeutic Implications for Left Ventricular Assist Device-Associated Bleeding. JACC Heart Fail 3, 860–869, doi: 10.1016/j.jchf.2015.06.016 (2015). [DOI] [PubMed] [Google Scholar]
- 19.Bortot M et al. Turbulent Flow Promotes Cleavage of VWF (von Willebrand Factor) by ADAMTS13 (A Disintegrin and Metalloproteinase With a Thrombospondin Type-1 Motif, Member 13). Arteriosclerosis, Thrombosis, and Vascular Biology 39, 1831–1842, doi:doi: 10.1161/ATVBAHA.119.312814 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bortot M et al. Pathologic Shear and Elongation Rates Do Not Cause Cleavage of Von Willebrand Factor by ADAMTS13 in a Purified System. Cell Mol Bioeng 13, 379–390, doi: 10.1007/s12195-020-00631-2 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bartoli CR, Restle DJ, Zhang DM, Acker MA & Atluri P Pathologic von Willebrand factor degradation with a left ventricular assist device occurs via two distinct mechanisms: mechanical demolition and enzymatic cleavage. J Thorac Cardiovasc Surg 149, 281–289, doi: 10.1016/j.jtcvs.2014.09.031 (2015). [DOI] [PubMed] [Google Scholar]
- 22.Berk ZBK et al. Evaluation of in vitro hemolysis and platelet activation of a newly developed maglev LVAD and two clinically used LVADs with human blood. Artif Organs 43, 870–879, doi: 10.1111/aor.13471 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kang J et al. Reduced continuous-flow left ventricular assist device speed does not decrease von Willebrand factor degradation. The Journal of Thoracic and Cardiovascular Surgery 151, 1747–1754.e1741. doi: 10.1016/j.jtcvs.2016.01.031 (2016). [DOI] [PubMed] [Google Scholar]
- 24.Geisen U et al. Improved Values for Acquired Von Willebrand Syndrome and Better Clinical Outcome in Patients with Heartmate III Compared to Heartmate II. Blood 128, 2582–2582 (2016). [Google Scholar]
- 25.McEniery CM et al. Endothelial function is associated with pulse pressure, pulse wave velocity, and augmentation index in healthy humans. Hypertension 48, 602–608, doi: 10.1161/01.HYP.0000239206.64270.5f (2006). [DOI] [PubMed] [Google Scholar]
- 26.Turner NA, Nolasco L, Ruggeri ZM & Moake JL Endothelial cell ADAMTS-13 and VWF: production, release, and VWF string cleavage. Blood 114, 5102–5111, doi: 10.1182/blood-2009-07-231597 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Vincent F et al. Arterial Pulsatility and Circulating von Willebrand Factor in Patients on Mechanical Circulatory Support. J Am Coll Cardiol 71, 2106–2118, doi: 10.1016/j.jacc.2018.02.075 (2018). [DOI] [PubMed] [Google Scholar]
- 28.Wang Y et al. Effect of Pulsatility on Shear-induced Extensional Behavior of Von Willebrand Factor. Artif Organs, doi: 10.1111/aor.14133 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lenting PJ, Christophe OD & Denis CV von Willebrand factor biosynthesis, secretion, and clearance: connecting the far ends. Blood 125, 2019–2028, doi: 10.1182/blood-2014-06-528406 (2015). [DOI] [PubMed] [Google Scholar]
- 30.Rusu L & Minshall R in Endothelial Dysfunction—Old Concepts and New Challenges (In Tech, 2018). [Google Scholar]
- 31.Galbusera M et al. Fluid shear stress modulates von Willebrand factor release from human vascular endothelium. Blood 90, 1558–1564 (1997). [PubMed] [Google Scholar]
- 32.Xiong Y et al. Hypertensive stretch regulates endothelial exocytosis of Weibel-Palade bodies through VEGF receptor 2 signaling pathways. Cell Research 23, 820–834, doi: 10.1038/cr.2013.56 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Schwarzl R & Netz RR Hydrodynamic Shear Effects on Grafted and Non-Grafted Collapsed Polymers. Polymers 10, 926 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Schneider SW et al. Shear-induced unfolding triggers adhesion of von Willebrand factor fibers. Proc Natl Acad Sci U S A 104, 7899–7903, doi: 10.1073/pnas.0608422104 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Singh I, Themistou E, Porcar L & Neelamegham S Fluid shear induces conformation change in human blood protein von Willebrand factor in solution. Biophys J 96, 2313–2320, doi: 10.1016/j.bpj.2008.12.3900 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Themistou E et al. Application of fluorescence spectroscopy to quantify shear-induced protein conformation change. Biophysical journal 97, 2567–2576, doi: 10.1016/j.bpj.2009.08.023 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Siedlecki CA et al. Shear-dependent changes in the three-dimensional structure of human von Willebrand factor. Blood 88, 2939–2950 (1996). [PubMed] [Google Scholar]
- 38.Wang Y et al. Shear-Induced Extensional Response Behaviors of Tethered von Willebrand Factor. Biophysical journal. 116, 2092–2102, doi: 10.1016/j.bpj.2019.04.025 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Dong J.-f. et al. ADAMTS-13 rapidly cleaves newly secreted ultralarge von Willebrand factor multimers on the endothelial surface under flowing conditions. Blood 100, 4033–4039, doi: 10.1182/blood-2002-05-1401 (2002). [DOI] [PubMed] [Google Scholar]
- 40.Joly JM et al. High Right Atrial Pressure and Low Pulse Pressure Predict Gastrointestinal Bleeding in Patients With Left Ventricular Assist Device. J Card Fail 24, 487–493, doi: 10.1016/j.cardfail.2018.03.003 (2018). [DOI] [PubMed] [Google Scholar]
- 41.Ising M et al. Flow Modulation Algorithms for Continuous Flow Left Ventricular Assist Devices to Increase Vascular Pulsatility: A Computer Simulation Study. Cardiovascular Engineering and Technology 2, 90, doi: 10.1007/s13239-011-0042-x (2011). [DOI] [Google Scholar]
- 42.Soucy KG et al. Rotary pump speed modulation for generating pulsatile flow and phasic left ventricular volume unloading in a bovine model of chronic ischemic heart failure. The Journal of Heart and Lung Transplantation 34, 122–131, doi: 10.1016/j.healun.2014.09.017 (2015). [DOI] [PubMed] [Google Scholar]
- 43.Naito N et al. Rotational speed modulation used with continuous-flow left ventricular assist device provides good pulsatility†. Interactive CardioVascular and Thoracic Surgery 26, 119–123, doi: 10.1093/icvts/ivx236 (2017). [DOI] [PubMed] [Google Scholar]
- 44.Naito N et al. Influence of a Rotational Speed Modulation System Used With an Implantable Continuous-Flow Left Ventricular Assist Device on von Willebrand Factor Dynamics. Artificial Organs 40, 877–883, doi: 10.1111/aor.12666 (2016). [DOI] [PubMed] [Google Scholar]
- 45.Kumar J, Elhassan A, Dimitrova G & Essandoh M The Lavare Cycle: A Novel Pulsatile Feature of the HVAD Continuous-Flow Left Ventricular Assist Device. Journal of Cardiothoracic and Vascular Anesthesia 33, 1170–1171, doi: 10.1053/j.jvca.2018.11.029 (2019). [DOI] [PubMed] [Google Scholar]
- 46.Saeed D et al. Gastrointestinal Bleeding in Patients with HeartWare Ventricular Assist Device: Does the Activation of the Lavare Cycle Make a Difference? ASAIO Journal 64 (2018). [DOI] [PubMed] [Google Scholar]
- 47.Kohno H et al. Can the intermittent low-speed function of left ventricular assist device prevent aortic insufficiency? Journal of Artijicial Organs 24, 191–198, doi: 10.1007/s10047-020-01234-4 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Bourque K et al. Design Rationale and Preclinical Evaluation of the HeartMate 3 Left Ventricular Assist System for Hemocompatibility. ASAIO Journal 62 (2016). [DOI] [PubMed] [Google Scholar]
- 49.Bourque K et al. In Vivo Assessment of a Rotary Left Ventricular Assist Device-induced Artificial Pulse in the Proximal and Distal Aorta. Artificial Organs 30, 638–642, doi: 10.1111/j.1525-1594.2006.00276.x (2006). [DOI] [PubMed] [Google Scholar]
- 50.Pirbodaghi T, Cotter C & Bourque K Power Consumption of Rotary Blood Pumps: Pulsatile Versus Constant-Speed Mode. Artificial Organs 38, 1024–1028, doi: 10.1111/aor.12323 (2014). [DOI] [PubMed] [Google Scholar]
- 51.Pirbodaghi T, Axiak S, Weber A, Gempp T & Vandenberghe S Pulsatile control of rotary blood pumps: Does the modulation waveform matter? The Journal of thoracic and cardiovascular surgery 144, 970–977, doi: 10.1016/j.jtcvs.2012.02.015 (2012). [DOI] [PubMed] [Google Scholar]
- 52.Pirbodaghi T, Weber A, Axiak S, Carrel T & Vandenberghe S Asymmetric speed modulation of a rotary blood pump affects ventricular unloading. European journal of cardio-thoracic surgery : official journal of the European Association for Cardio-thoracic Surgery 43, 383–388, doi: 10.1093/ejcts/ezs299 (2013). [DOI] [PubMed] [Google Scholar]
- 53.Saito S et al. Post-approval study of a highly pulsed, low-shear-rate, continuous-flow, left ventricular assist device, EVAHEART: A Japanese multicenter study using J-MACS. The Journal of Heart and Lung Transplantation 33, 599–608, doi: 10.1016/j.healun.2014.02.015 (2014). [DOI] [PubMed] [Google Scholar]
- 54.Botterbusch C et al. Progress in the Development of the Pulsatile CorWave LVAD. The Journal of Heart and Lung Transplantation 40, S104, doi: 10.1016/j.healun.2021.01.338 (2021). [DOI] [Google Scholar]
- 55.Cox LG, Loerakker S, Rutten MC, de Mol BA & van de Vosse FN A mathematical model to evaluate control strategies for mechanical circulatory support. Artificial organs 33, 593–603, doi: 10.1111/j.1525-1594.2009.00755.x (2009). [DOI] [PubMed] [Google Scholar]
- 56.Letsou GV et al. Improved left ventricular unloading and circulatory support with synchronized pulsatile left ventricular assistance compared with continuous-flow left ventricular assistance in an acute porcine left ventricular failure model. The Journal of thoracic and cardiovascular surgery 140, 1181–1188, doi: 10.1016/j.jtcvs.2010.03.043 (2010). [DOI] [PubMed] [Google Scholar]
- 57.Shi Y, Lawford PV & Hose DR Numerical modeling of hemodynamics with pulsatile impeller pump support. Annals of biomedical engineering 38, 2621–2634, doi: 10.1007/s10439-010-0001-y (2010). [DOI] [PubMed] [Google Scholar]
- 58.Ising MS, Sobieski MA, Slaughter MS, Koenig SC & Giridharan GA Feasibility of Pump Speed Modulation for Restoring Vascular Pulsatility with Rotary Blood Pumps. Asaio j 61, 526–532, doi: 10.1097/mat.0000000000000262 (2015). [DOI] [PubMed] [Google Scholar]
- 59.Bearnson GB et al. Pulsatile operation of a centrifugal ventricular assist device with magnetic bearings. ASAIO journal (American Society for Artificial Internal Organs : 1992) 42, M620–624 (1996). [DOI] [PubMed] [Google Scholar]
- 60.Qian KX Pulsatile impeller heart: a viable alternative to a problematic diaphragm heart. Medical engineering & physics 18, 57–66 (1996). [DOI] [PubMed] [Google Scholar]
- 61.Chen Z et al. Shear stress and blood trauma under constant and pulse-modulated speed CF-VAD operations: CFD analysis of the HVAD. Medical & Biological Engineering & Computing 57, 807–818, doi: 10.1007/s11517-018-1922-0 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Ortiz S, Vu V, Montes R & May-Newman K Left Ventricular Flow Dynamics with the HeartMate3 Left Ventricular Assist Device: Effect of Inflow Cannula Position and Speed Modulation. ASAIO Journal 67, 1301–1311, doi: 10.1097/mat.0000000000001523 (2021). [DOI] [PubMed] [Google Scholar]