Abstract
We investigated myocardial performance concerning obstructive sleep apnea (OSA) severity and the benefits of continuous positive airway pressure (CPAP) therapy. In this randomized sham-controlled trial, 52 patients (mean age, 49 years; 92%, males; mean AHI, 59) with severe OSA were randomly assigned to receive either CPAP or sham treatment for 3 months. The severity of OSA was determined using the apnea/hypopnea index (AHI), oxygen desaturation index (ODI), percentage of sleep time below 90% oxygen saturation (T90), and average O2 saturation during sleep (mean SpO2). We compared the changes in myocardial work after 3 months of CPAP (n = 26) versus the sham group (n = 26) at rest and during an exercise stress test. Unlike AHI or ODI, indices of hypoxemia including T90 and mean SpO2 were significantly correlated with global constructive work, as defined by work of left ventricle (LV) that contributes to LV ejection during systole (T90, β = 0.393, p = 0.012; mean SpO2, β = 0.331, p = 0.048), and global wasted work (GWW), as defined by work of LV that does not contribute to LV ejection (T90, β = 0.363, p = 0.015; mean SpO2, β = − 0.370, p = 0.019). After 3 months, GWW decreased (80.0 ± 49.2 to 60.8 ± 26.3, p = 0.009) and global work efficiency increased (94.0 ± 4.5 to 95.7 ± 2.0, p = 0.008) in the CPAP group compared to those in the sham group. At the 3-month follow-up exercise stress echocardiography, worsening of GWW during exercise was significantly decreased in the CPAP group compared to that in the sham group (p = 0.045 at 50 W). Hypoxemia indices were closely associated with myocardial performance in patients with severe OSA. CPAP treatment for 3 months improved left ventricular myocardial performance by decreasing wasted work and increasing work efficacy compared to the sham treatment.
Subject terms: Cardiology, Echocardiography
Introduction
Obstructive sleep apnea (OSA) is a common sleep disorder characterized by repetitive upper airway obstruction during sleep1,2. Patients with OSA are at an increased risk of cardiovascular diseases3. OSA leads to decreased myocardial oxygenation, increased sympathetic activation, and increased afterload, which in turn have adverse effects on both left (LV) and right ventricular (RV) functions4,5. Continuous positive airway pressure (CPAP) therapy improves myocardial oxygenation, attenuates sympathetic activation, and reduces the LV afterload6–8. CPAP therapy for 3 months improved systolic functions of LV and RV and the diastolic function of LV9,10. LV global longitudinal strain (LVGLS) offers an advantage in evaluating subclinical dysfunction, particularly in heart failure with preserved ejection fraction11. However, LVGLS is also affected by the afterload12. Thus, decreased LVGLS may be driven by an increased afterload rather than impaired myocardial contractility. While the LV pressure–volume (P–V) loop is the ideal way to evaluate LV function, it is not practical because of its invasive nature13–15. Myocardial work is a new echocardiography-based imaging tool that quantifies LV performance based on the pressure–strain (P–S) loop and has been validated by invasively derived P–V measurements in various cardiac conditions. Previous studies reported that the LV P–S loop sufficiently reflects the LV P–V loop, and the LV P–S loop using 2D speckle tracking echocardiography can be an alternative method to evaluate LV function accounting for LV afterload16,17. In this study, we tested the hypotheses that the severity of OSA is associated with myocardial performance calculated by myocardial work and that CPAP treatment improves myocardial performance at rest and during exercise.
Results
Baseline clinical characteristics
Twenty-six participants from each group were analyzed. Table 1 shows the baseline characteristics of the two groups. There were no significant differences in baseline characteristics between the two groups, except for body mass index, history of dyslipidemia, and proportion of statin users. Baseline PSG characteristics, such as mean apnea/hypopnea index (AHI), mean oxygen saturation (mean SpO2) during sleep, percentage of sleep time below 90% oxygen saturation(T90), average heart rate during sleep, and oxygen desaturation index(ODI), which was calculated the average number of desaturation episodes per hour, were not significantly different.
Table 1.
Baseline characteristics of the study population.
| Sham (n = 26) | CPAP (n = 26) | p Value | |
|---|---|---|---|
| Age, year | 48.8 ± 10.7 | 49.1 ± 11.4 | 0.920 |
| Male sex, n (%) | 24 (92) | 24 (92) | 1.000 |
| Height, cm | 169.9 ± 7.3 | 170.9 ± 6.8 | 0.583 |
| Weight, kg | 76.2 ± 12.6 | 81.7 ± 11.7 | 0.108 |
| Body mass index, kg/m2 | 26.2 ± 3.0 | 27.8 ± 2.8 | 0.047 |
| Body surface area, m2 | 1.90 ± 0.18 | 1.96 ± 0.18 | 0.234 |
| Systolic BP mmHg | 127.8 ± 16.0 | 126 ± 19.6 | 0.722 |
| Diastolic BP mmHg | 77.3 ± 10.6 | 75.8 ± 13.7 | 0.662 |
| Hypertension, n (%) | 15 (58) | 17 (65) | 0.569 |
| Diabetes mellitus, n (%) | 1 (4) | 4 (15) | 0.158 |
| Dyslipidemia, n (%) | 2 (7) | 8 (31) | 0.035 |
| Current smokers, n (%) | 6 (23) | 10 (39) | 0.229 |
| Medications | |||
| Calcium channel blockers, n (%) | 8 (31) | 10 (39) | 0.560 |
| RAAS blockers, n (%) | 8 (31) | 9 (35) | 0.768 |
| Beta-blockers, n (%) | 4 (15) | 4 (15) | 1.000 |
| Diuretics, n (%) | 0 (0) | 0 (0) | 1.000 |
| Statins, n (%) | 2 (7) | 8 (31) | 0.035 |
| Polysomnographic characteristics | |||
| AHI, events/hour | 53.4 ± 20.5 | 64.2 ± 20.5 | 0.063 |
| Mean oxygen saturation, % | 94.5 ± 1.6 | 93.9 ± 1.7 | 0.238 |
| T90, % | 7.1 ± 7.3 | 10.5 ± 11.6 | 0.210 |
| Average heart rate during sleep, bpm | 62.3 ± 6.3 | 63.3 ± 6.8 | 0.576 |
| Oxygen desaturation index, events/hour | 40.8 ± 20.5 | 47.7 ± 19.6 | 0.239 |
Values are n (%) or mean ± standard deviation.
CPAP continuous positive airway pressure, AHI apnea/hypopnea index, BP blood pressure, RAAS renin–angiotensin–aldosterone system, T90 percentage of sleep time below 90% oxygen saturation.
Association of severity of OSA with myocardial work
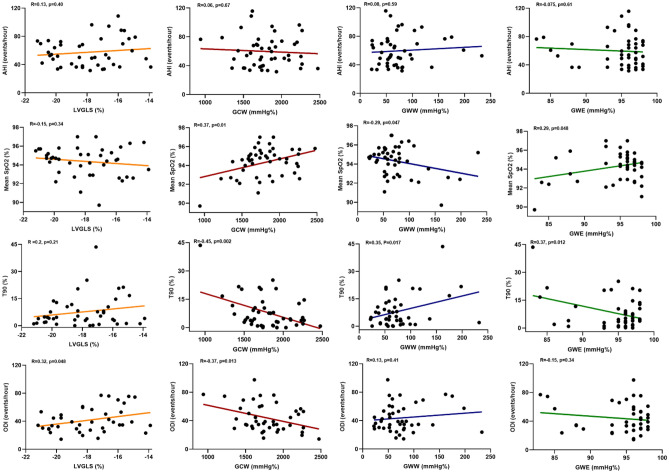
Indices of hypoxemia, such as T90 and mean SpO2, were significantly correlated with all myocardial work parameters, except for LVGLS (Fig. 1). T90 was significantly correlated with GWW (r = 0.35, p = 0.017), GWE (r = − 0.37, p = 0.012), and GCW (r = − 0.45, p = 0.002). The mean SpO2 also correlated with GWW (r = − 0.29, p = 0.047), GWE (r = 0.29, p = 0.048), and GCW (r = 0.37, p = 0.01). However, AHI was not correlated with either LVGLS or myocardial work parameters, but ODI was correlated with LVGLS (r = 0.32, p = 0.048) and GCW (r = − 0.37, p = 0.013). In a multivariate regression analysis including age, sex, body mass index, history of hypertension, and smoking, both T90 and mean SpO2 showed significant association with GCW and GWW, but no significant association with AHI or ODI (Table 2).
Figure 1.
The correlation between AHI and hypoxemia indices (T90, mean SpO2, and ODI) with LVGLS and myocardial work. AHI, apnea/hypopnea index; LVGLS, left ventricular global longitudinal strain; GCW, global constructive work; GWE, global work efficiency; and GWW, global wasted work; Mean SpO2, mean oxygen saturation; ODI, oxygen desaturation index; T90, percentage of sleep time below 90% oxygen saturation.
Table 2.
Multivariate linear regression analysis* for LVGLS and myocardial work according to AHI and hypoxemia indices (T90, mean SpO2, and ODI).
| Variables | LVGLS (%) | GCW (mmHg) | GWW (mmHg) | GWE (mmHg) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Beta | t | p Value | Beta | t | p Value | Beta | t | p Value | Beta | t | p Value | |
| AHI (events/hour) | 0.051 | 0.297 | 0.768 | 0.028 | 0.156 | 0.877 | − 0.053 | − 0.313 | 0.756 | 0.082 | 0.508 | 0.614 |
| T90 (%) | 0.113 | 0.705 | 0.485 | − 0.393 | − 2.631 | 0.012 | 0.363 | 2.548 | 0.015 | − 0.352 | − 2.559 | 0.014 |
| Mean SpO2 (%) | − 0.128 | − 0.757 | 0.454 | 0.331 | 2.040 | 0.048 | − 0.370 | − 2.454 | 0.019 | 0.282 | 1.883 | 0.067 |
| ODI (events/hour) | 0.193 | 1.019 | 0.316 | − 0.323 | − 1.08 | 0.096 | 0.128 | 0.688 | 0.496 | − 0.112 | − 0.622 | 0.538 |
*Adjusted by age, sex, body mass index, history of hypertension, and smoking.
AHI apnea/hypopnea index, LVGLS left ventricular global longitudinal strain, GCW global constructive work, GWE global work efficiency and GWW global wasted work, Mean SpO2 mean oxygen saturation, ODI oxygen desaturation index, T90 percentage of sleep time below 90% oxygen saturation.
CPAP effects on myocardial work
Table 3 shows the effects of the sham and CPAP treatment on LV function using echocardiographic parameters. The LV dimension, LV mass index, left atrial volume index, and LV ejection fraction did not significantly different between the baseline and post-treatment. The E’ velocity, an early marker of diastolic dysfunction and LVGLS, was positively changed in the CPAP treatment group after 3 months (change of E’ velocity: 0.65 ± 1.70 cm/s vs. − 0.61 ± 1.85 cm/s, p = 0.014). There was no changes in GWI and GCW before or after treatment. However, GWW was decreased (p = 0.015) and GWE was improved (p = 0.027) after 3 months in the CPAP treatment group but not in the sham treatment group. The GWW and GWE from baseline to the 3-month follow-up significantly differed between the two groups (GWW, p = 0.003; GWE, p = 0.003) (Supplementary Fig. S1). The changes in GWW and GWE in the subgroups are described in Supplementary Table S1. In patients with hypertension, CPAP treatment decreased GWW and increased GWE. In addition, a subgroup analysis of AHI, mean SpO2, and ODI showed that CPAP treatment decreased GWW and increased GWE significantly in patients with very severe OSA (AHI > 50/h, mean SpO2 ≤ 95%, or ODI > 39/h). However, CPAP treatment significantly reduced GWW in the case of T90 ≤ 10% and increased GWE in the case of T90 > 10%, but did not significantly reduce GWW.
Table 3.
Echocardiographic parameters of the study population.
| Sham (n = 26) | CPAP (n = 26) | |||
|---|---|---|---|---|
| Baseline | Follow-up | Baseline | Follow-up | |
| Traditional echocardiography parameter | ||||
| LVEDV, mL/m2 | 59.3 ± 6.3 | 57.4 ± 6.2 | 60.4 ± 9.0 | 59.5 ± 9.1 |
| LVESV, mL/m2 | 21.3 ± 3.6 | 20.5 ± 2.7 | 20.8 ± 4.7 | 21.4 ± 3.8 |
| LV mass index, g/m2 | 85.7 ± 12.8 | 83.9 ± 14.1 | 92.0 ± 15.0 | 91.2 ± 11.9 |
| LA volume index, mL/m2 | 25.9 ± 5.3 | 23.8 ± 5.3 | 26.5 ± 5.8 | 26.6 ± 5.2 |
| LVEF, % | 64 ± 6 | 64 ± 6 | 66 ± 5 | 65 ± 6 |
| S′ velocity, cm/sec | 8.4 ± 1.6 | 9.1 ± 2.0 | 8.7 ± 1.4 | 8.9 ± 1.6 |
| e′ velocity, cm/sec | 9.4 ± 2.7 | 8.7 ± 2.9 | 7.7 ± 2.2* | 8.3 ± 2.3 |
| E/e′ | 7.6 ± 1.5 | 7.9 ± 1.8 | 8.9 ± 2.4* | 8.9 ± 2.4 |
| 2D speckle-tracking indices (LVGLS and Myocardial work) | ||||
| LVGLS, % | − 18.0 ± 1.9 | − 18.0 ± 2.5 | − 17.8 ± 2.1 | − 20.0 ± 2.1*,† |
| GWI, mmHg% | 1556.8 ± 257.7 | 1505.0 ± 305.5 | 1606.8 ± 322.5 | 1586.1 ± 208.4 |
| GCW, mmHg% | 1746.3 ± 258.8 | 1700.8 ± 354.2 | 1840.5 ± 347.0 | 1789.7 ± 212.4 |
| GWW, mmHg% | 70.3 ± 39.2 | 87.4 ± 71.9 | 80.0 ± 49.2 | 60.8 ± 26.3† |
| GWE, % | 95.3 ± 3.2 | 94.0 ± 4.3† | 94.0 ± 4.5 | 95.7 ± 2.0† |
*Student’s t-test, p < 0.05, compared with the corresponding sham group.
†Paired t-test, p < 0.05, compared to baseline values within the group.
GCW global constructive work, GWE global work efficiency, GWI global work index, GWW global wasted work, LA left atrium, LV left ventricle, LVEDV left ventricular end diastolic volume, LVEF left ventricular ejection fraction, LVESV left ventricular end-systolic volume, LVGLS left ventricular global longitudinal strain.
CPAP effect on myocardial work during exercise
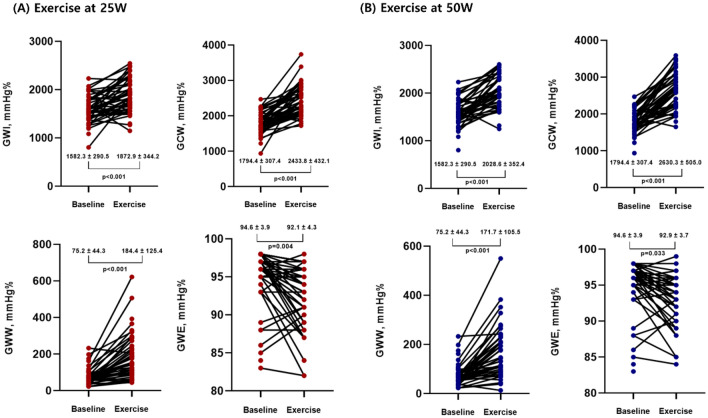
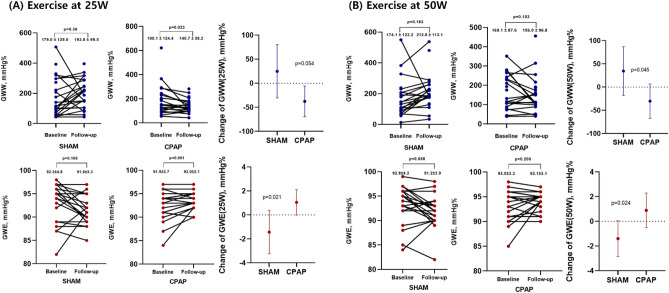
In all patients, GWI, GCW, and GWW were significantly increased during exercise compared to those at baseline for both 25 W and 50 W exercise. However, GWE was decreased during exercise compared to the baseline value (Fig. 2, Supplementary Table S2). During 25 W exercise, there was no difference in myocardial work parameters between the sham and CPAP group before treatment; however, after 3 months of treatment, the GWW of the CPAP group was significantly decreased compared with that at the baseline (190.1 ± 124.4 vs. 140.7 ± 58.2, p = 0.023), and it was significantly lower than that of sham group (193.8 ± 99.5 vs. 140.7 ± 58.2, p = 0.035). There was no difference in GWE before and after treatment in both groups, but the amount of change was significantly different (p = 0.021) (Fig. 3, Supplementary Table S3). After the 50 W exercise, there was no change in pre- and post-treatment myocardial work parameters between the sham and CPAP groups, but a difference of change in GWW (p = 0.045) and GWE (p = 0.024) was confirmed between these two groups (Fig. 3).
Figure 2.
Comparison of myocardial work parameters between baseline and 25 W exercise (A) or 50 W exercise (B). CPAP, continuous positive airway pressure; GCW, global constructive work; GWE, global work efficiency; GWI, global work index; and GWW, global wasted work.
Figure 3.
Changes in GWW and GWE in the two study groups according to exercise. (A) Changes at 25 W and (B) at 50 W. CPAP, continuous positive airway pressure; GWE, global work efficiency; and GWW, global wasted work.
Discussion
This randomized, sham-controlled clinical trial demonstrated the association between hypoxemia severity and myocardial work using the LV P-S loop and assessed the benefits of CPAP therapy in patients with severe OSA. The key findings were as follows: (1) in patients with severe OSA, the severity of hypoxemia was more closely associated with myocardial performance than the most commonly used OSA metric, AHI; (2) CPAP treatment for 3 months improved LV myocardial performance by decreasing LV wasted work and increasing work efficacy; (3) GWW increased and GWE decreased during exercise in patients with severe OSA, and CPAP treatment reduced GWW and increased GWE in these patients.
Hypoxemia leads to hypoxia, which causes endothelial dysfunction by causing oxidative stress and inflammation in endothelial cells and mitochondria by altering various intracellular molecules18. Endothelial dysfunction is a major cause of cardiovascular disease. OSA is associated with intermittent hypoxia, which increases proangiogenic and pro-inflammatory molecules in cellular model and elevates blood pressure clinically, implying that it increases the LV afterload19,20. Thus, intermittent hypoxia in patients with OSA is a major factor that induces endothelial dysfunction and inflammation, ultimately affecting left ventricular diastolic and systolic function.
AHI is the most commonly used index for diagnosis and determining OSA severity (mild: 5–14/h, moderate: 15–29/h, severe: > 30/h)2. Although AHI mostly correlates with hypoxemia severity, in some patients whose AHI is mostly attributed to arousal-based hypopneas (without much desaturation), the correlation between the two can be poor. Several studies have reported that hypoxemia severity is more closely related to cardiovascular disease than AHI; in particular, T90 and mean SpO2, which are part of the conventional OSA metrics, have been identified as important predictors of sudden cardiac death and mortality in patients with chronic heart failure21,22. Similarly, myocardial work was more correlated with hypoxic indices in this study than AHI within the severe OSA category. This suggests that hypoxemia severity may be an important indicator of myocardial dysfunction in patients with severe OSA. Therefore, it is important to evaluate hypoxemia severity, in addition to AHI alone, when examining the relationship between the severity of OSA and the risk of end-organ damage. Subgroup analysis showed a significant difference in the changes in GWW and GWE between the CPAP and sham groups in patients with mean SpO2 < 95%. However, in the case of T90 ≥ 10%, GWE increased significantly after CPAP treatment. Although there was no significant difference in any subgroups based on hypoxemia severity, GWW tended to decrease and GWE tended to increase in the CPAP group. Thus, hypoxemia appears to play a major mechanism in the negative effect of OSA on myocardial mechanics, and the severity of the hypoxemia is a predictor of the CPAP effect.
Combined ventricular systolic and arterial stiffening is considered to be an important factor in the pathophysiology of heart failure with preserved ejection fraction23. In our previous study, we demonstrated that 3 months of CPAP treatment improved pulse wave velocity, arterial elastance, and ventricular–vascular coupling index9. Myocardial work is considered to be the ideal parameter to reflect LV function by mirroring the pressure loading of the LV13. In this respect, the improvement of GWE observed in the study is a result of the reduction of LV afterload and activation of the sympathetic nervous system caused by CPAP treatment and the increase in stroke volume and cardiac function24. In the subgroup analysis, the effect of GWE and GWW improvement between the CPAP and sham groups was significant in patients with hypertension. This additionally implies that improvement in arterial stiffness is an important factor in the improvement of myocardial work in patients with sleep apnea.
Few studies have evaluated myocardial work during exercise stress echocardiography in healthy populations. In the healthy control individuals in these studies, GWI, GCW, and GWW increased, but there was no change in GWE25,26. However, patients with heart failure with preserved ejection fraction showed that the reduced GWE during effort was mainly determined by increased GWW during exercise27. In our study, consistent with previous literature, GWI, GCW, and GWW increased during exercise and, as a result, GWE decreased compared to that at baseline. When comparing myocardial work during exercise before and after treatment in each group, there was no significant difference in GWE. However, because GWW was decreased in the CPAP treatment group at 25 W exercise, it was expected that CPAP could improve myocardial function in patients with severe OSA during exercise. However, the underlying mechanism remains unclear. A possible explanation is that OSA is independently associated with clinical and subclinical myocardial ischemia and GWW sensitively reflects myocardial injury. While GWW represents work performed by the LV, it does not contribute to LV ejection, but provides information on myocardial contractile reserve in viable myocardium. Clinically or sub-clinically damaged myocardium has dyssynchronous contractions and post-systolic shortening, which contribute to changes in GWW28. Moreover, GWW is related to increased myocardial wall stress against a higher afterload; thus, the damaged myocardium may have been more affected by increased afterload during exercise29.
This study had some limitations. First is the innate limitation of the method used for calculating myocardial work. Myocardial work was calculated assuming that peripheral blood pressure values were equal to central blood pressure and the myocardial strain was equal to volume change. Second, patients with structural heart disease, coronary heart disease, arrhythmia, and uncontrolled BP were excluded. It is not easy to generalize our results across all populations or clinical situations. However, we found that CPAP treatment improved LV mechanics even in patients with subclinical myocardial dysfunction. Third, since the effect of CPAP treatment was compared over a relatively short period of 3 months, the long-term effects are unknown. However, importantly, CPAP therapy improved myocardial performance, even over the short study period. Fourth, the study population was small. Thus, further studies are needed to verify the results using a greater number of patients. However, given the difficulty of conducting a randomized sham-controlled trial of CPAP on a large number of patients, the results of this study are still valuable. Finally, no comparison was made with a healthy population. Therefore, it is difficult to generalize the value of myocardial work in this study to the characteristics of patients with OSA. Nevertheless, this study is meaningful as it confirmed the correlation between the severity of hypoxemia and LV performance using myocardial work in patients with severe OSA.
In conclusion, indices of hypoxic burden are more closely associated with myocardial performance than AHI in patients with severe OSA. 3-months CPAP treatment improved LV myocardial performance by decreasing LV wasted work and increasing work efficacy compared to the sham treatment.
Methods
Study population and design
This study was conducted as a sub-study of the previous trial: The Continuous Positive Airway Pressure Therapy on Left Ventricular Diastolic Function in Patients with Severe Obstructive Sleep Apnea trial was designed to investigate the effects of 3 months of CPAP on LV diastolic function. It was a prospective, single-center, randomized, blinded, sham-controlled trial of parallel groups. The design and primary outcomes of the trial have been previously reported9. Briefly, a total of 60 patients with severe OSA (AHI > 30/h) by screening polysomnography (PSG) were enrolled2. Exclusion criteria included LV ejection fraction < 50%, uncontrolled blood pressure (BP) (≥ 180/110 mmHg), prior history of coronary artery disease, significant valvular dysfunction (of a moderate or greater degree), arrhythmia, an estimated glomerular filtration rate of less than 60 mL/min/1.73 m2 of body surface area, and treatment with anxiolytics or sedatives. The final 56 patients were randomly divided into a sham treatment group (n = 28) and a CPAP treatment group (n = 28) through 1:1 randomization (Supplementary Fig. S2). All patients were masked to the treatment. Over the 3-months treatment period, two patients in each group discontinued interventions; thus, 26 patients in each group were included in the analysis. The data on study populations had been used to assess diastolic function and speckle tracking echocardiography in a previous study9,10 and were re-analyzed to calculate myocardial work parameters in the current study. The original trial is registered with www.ClinicalTrials.gov (NCT01854398). This study was approved by the Institutional Review Board of the Yonsei University Health System (approval number: 1-2013-0021), and the investigation conforms with the principles outlined in the Declaration of Helsinki. All participants provided informed consent.
Measures of obstructive sleep apnea severity
The standard channels recommended by the American Academy of Sleep Medicine (AASM) were used to perform overnight PSG, and data were processed using the Embletta X100 system (Natus Embla Systems, Oakville, ON, Canada). AHI was defined as the number of apnea and hypopnea events divided by total sleep time, expressed as events per hour. Apnea was defined as a reduction in airflow greater than 90% of the pre‐event baseline and occurring for ≥ 10 s measured using a thermocouple signal. Hypopnea events were defined as a reduction in airflow > 30% of the pre‐event baseline and occurring for ≥ 10 s in association with at least a 3% oxygen desaturation or arousal. Hypopnea was considered obstructive if any of the following were present: snoring during the event, an increase in the flattening of the nasal pressure flow, or paradoxical breathing. Obstructive AHI ≥ 15 events/h was considered clinically significant for OSA. Central apneas were defined as apneas occurring without associated respiratory effort lasting ≥ 10 s.
Severe OSA was defined as an AHI > 30/h, and very severe OSA was defined as an AHI > 50/h in this study30,31. Measures of PSG-derived hypoxemia metrics, including mean SpO2 during sleep, T90, and ODI, were used to further classify patients with severe OSA into those with and without severe hypoxemia.
Transthoracic echocardiography and exercise echocardiography
All patients underwent transthoracic echocardiography at baseline and after 3 months in both the CPAP and sham treatment groups using a 2.5-MHz transducer ultrasound machine (Vivid 7 or Vivid E9 system, GE Healthcare). Conventional 2D, M-mode, and Doppler images were obtained and measured according to the current recommendations32. 2D speckle tracking echocardiography was performed using customized software (EchoPAC PC, version 113; GE Medical Systems). We manually traced the endocardial border of the LV in the acquired apical four-chamber, apical three-chamber, and apical two-chamber views to obtain the LVGLS. The LVGLS was calculated by averaging the peak strain value of the three apical views. All data were measured by two experienced cardiologists who were blinded to the data analysis.
Exercise stress echocardiography was performed for all patients at baseline and the 3-month follow-up. Symptom-limited supine bicycle exercise stress echocardiography was performed according to current guidelines using a cycle ergometer33. The participants started pedaling at a workload of 25 W, which was increased by 25 W every 3 min up to peak exercise and stopped if the patient complained of symptoms. Three standard apical views were acquired at each exercise stage. Echocardiographic images at 25 W and 50 W were used for analysis, as the measurement results were unreliable due to the poor quality of speckle tracking in tachycardia conditions (almost ≥ 75 W images).
Myocardial work evaluation
Myocardial work was calculated as a combination of LV strain and the estimated LV pressure curve. In the software, the LV strain provided the P–S loop, which was calculated using 2D speckle tracking echocardiography, and peripheral systolic blood pressure was measured non-invasively under the assumption that the peak LV pressure was equal to the peak systolic arterial pressure. The isovolumic and ejection phases were determined through the opening and closing times of the mitral and aortic valves16. The calculated myocardial work parameters were as follows:
Global work index (GWI): total work of LV, that is, the area under the curve of the P–S loop.
Global constructive work (GCW): work of LV that contributes to LV ejection during systole, that is, myocardial shortening during systole (positive segmental work) and lengthening during isovolumic relaxation (negative segmental work).
Global wasted work (GWW): work of LV that does not contribute to LV ejection, specifically, myocardial lengthening during systole (negative segmental work) and shortening during isovolumic relaxation (positive segmental work).
Global work efficiency (GWE): the ratio of constructive work to total work (GCW/[GCW + GWW]).
The BP of all patients was measured in the supine position after a 5-min rest just before echocardiography. Supplementary Fig. S3 illustrates the measurement of the myocardial work parameters using the software.
Statistical analysis
As described previously, the sample size was calculated using an a-value of 0.05, a beta value of 0.1, and a power of 90% based on previous research. Assuming a dropout rate of 20%, the sample size was calculated as 30 patients per group, resulting in a target sample size of 60 patients7. Continuous variables are presented as mean ± standard deviation, and categorical variables are presented as frequencies (%). Between-group comparisons were analyzed using a two-sample t-test. Before and after treatment comparisons were performed using paired t-tests. Pearson’s correlation analysis and multivariable regression analysis were used to analyze the correlation between OSA severity and LV performance. Subgroup analysis was performed by dividing patients based on the presence or absence of hypertension and by the median value of OSA severity indices, such as AHI, mean SpO2, T90, and ODI. Statistical significance was set at a two-sided p-value < 0.05. Statistical analyses were conducted using SPSS (version 22.0; IBM, Armonk, NY, USA) and R statistical software (version 4.0.2; R Foundation for Statistical Computing, Vienna, Austria).
Supplementary Information
Author contributions
S.-E.K. and J.S..: study design, data acquisition, data analysis, data interpretation, and manuscript drafting and review; Y.K., I.C., C.Y.S., J.-W.H.: data acquisition, interpretation, and manuscript review; G.-R.H.: study concept and design, data analysis, data interpretation, manuscript review and revision, and study supervision.
Data availability
Data of this study are available from the corresponding author on request due to privacy/ethical restrictions.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors contributed equally: Se-Eun Kim and Jiwon Seo.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-023-32274-4.
References
- 1.Jun JC, Chopra S, Schwartz AR. Sleep apnoea. Eur. Respir. Rev. 2016;25:12–18. doi: 10.1183/16000617.0077-2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kapur VK, et al. Clinical practice guideline for diagnostic testing for adult obstructive sleep apnea: An American Academy of Sleep Medicine clinical practice guideline. J. Clin. Sleep Med. 2017;13:479–504. doi: 10.5664/jcsm.6506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sánchez-de-la-Torre M, Campos-Rodriguez F, Barbé F. Obstructive sleep apnoea and cardiovascular disease. Lancet Respir. Med. 2013;1:61–72. doi: 10.1016/s2213-2600(12)70051-6. [DOI] [PubMed] [Google Scholar]
- 4.Parati G, et al. Heart failure and sleep disorders. Nat. Rev. Cardiol. 2016;13:389–403. doi: 10.1038/nrcardio.2016.71. [DOI] [PubMed] [Google Scholar]
- 5.Cowie MR, Gallagher AM. Sleep disordered breathing and heart failure: what does the future hold? JACC Heart Fail. 2017;5:715–723. doi: 10.1016/j.jchf.2017.06.016. [DOI] [PubMed] [Google Scholar]
- 6.Akar Bayram N, et al. Effects of continuous positive airway pressure therapy on left ventricular function assessed by tissue Doppler imaging in patients with obstructive sleep apnoea syndrome. Eur. J. Echocardiogr. 2009;10:376–382. doi: 10.1093/ejechocard/jen257. [DOI] [PubMed] [Google Scholar]
- 7.Butt M, Dwivedi G, Shantsila A, Khair OA, Lip GY. Left ventricular systolic and diastolic function in obstructive sleep apnea: Impact of continuous positive airway pressure therapy. Circ. Heart Fail. 2012;5:226–233. doi: 10.1161/circheartfailure.111.964106. [DOI] [PubMed] [Google Scholar]
- 8.Arias MA, et al. Obstructive sleep apnea syndrome affects left ventricular diastolic function: Effects of nasal continuous positive airway pressure in men. Circulation. 2005;112:375–383. doi: 10.1161/circulationaha.104.501841. [DOI] [PubMed] [Google Scholar]
- 9.Shim CY, et al. Effects of continuous positive airway pressure therapy on left ventricular diastolic function: A randomised, sham-controlled clinical trial. Eur. Respir. J. 2018 doi: 10.1183/13993003.01774-2017. [DOI] [PubMed] [Google Scholar]
- 10.Kim D, et al. Continuous positive airway pressure therapy restores cardiac mechanical function in patients with severe obstructive sleep apnea: A randomized, sham-controlled study. J. Am. Soc. Echocardiogr. 2019;32:826–835. doi: 10.1016/j.echo.2019.03.020. [DOI] [PubMed] [Google Scholar]
- 11.Tschöpe C, Senni M. Usefulness and clinical relevance of left ventricular global longitudinal systolic strain in patients with heart failure with preserved ejection fraction. Heart Fail. Rev. 2020;25:67–73. doi: 10.1007/s10741-019-09853-7. [DOI] [PubMed] [Google Scholar]
- 12.Hubert A, et al. Estimation of myocardial work from pressure-strain loops analysis: An experimental evaluation. Eur. Heart J. Cardiovasc. Imaging. 2018;19:1372–1379. doi: 10.1093/ehjci/jey024. [DOI] [PubMed] [Google Scholar]
- 13.Abawi D, et al. The non-invasive assessment of myocardial work by pressure-strain analysis: Clinical applications. Heart Fail. Rev. 2021 doi: 10.1007/s10741-021-10119-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Burkhoff D, Mirsky I, Suga H. Assessment of systolic and diastolic ventricular properties via pressure–volume analysis: A guide for clinical, translational, and basic researchers. Am. J. Physiol. Heart Circ. Physiol. 2005;289:H501–512. doi: 10.1152/ajpheart.00138.2005. [DOI] [PubMed] [Google Scholar]
- 15.Foëx P, Leone BJ. Pressure–volume loops: A dynamic approach to the assessment of ventricular function. J. Cardiothorac. Vasc. Anesth. 1994;8:84–96. doi: 10.1016/1053-0770(94)90020-5. [DOI] [PubMed] [Google Scholar]
- 16.Russell K, et al. A novel clinical method for quantification of regional left ventricular pressure-strain loop area: A non-invasive index of myocardial work. Eur. Heart J. 2012;33:724–733. doi: 10.1093/eurheartj/ehs016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Russell K, et al. Assessment of wasted myocardial work: A novel method to quantify energy loss due to uncoordinated left ventricular contractions. Am. J. Physiol. Heart Circ. Physiol. 2013;305:H996–1003. doi: 10.1152/ajpheart.00191.2013. [DOI] [PubMed] [Google Scholar]
- 18.Janaszak-Jasiecka A, Siekierzycka A, Płoska A, Dobrucki IT, Kalinowski L. Endothelial dysfunction driven by hypoxia—The influence of oxygen deficiency on NO bioavailability. Biomolecules. 2021 doi: 10.3390/biom11070982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Turnbull CD, et al. Effect of OSA on hypoxic and inflammatory markers during CPAP withdrawal: Further evidence from three randomized control trials. Respirology. 2017;22:793–799. doi: 10.1111/resp.12946. [DOI] [PubMed] [Google Scholar]
- 20.Schwarz EI, Schlatzer C, Rossi VA, Stradling JR, Kohler M. Effect of CPAP withdrawal on BP in OSA: Data from three randomized controlled trials. Chest. 2016;150:1202–1210. doi: 10.1016/j.chest.2016.07.012. [DOI] [PubMed] [Google Scholar]
- 21.Gami AS, et al. Obstructive sleep apnea and the risk of sudden cardiac death: A longitudinal study of 10,701 adults. J. Am. Coll. Cardiol. 2013;62:610–616. doi: 10.1016/j.jacc.2013.04.080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Oldenburg O, et al. Nocturnal hypoxaemia is associated with increased mortality in stable heart failure patients. Eur. Heart J. 2016;37:1695–1703. doi: 10.1093/eurheartj/ehv624. [DOI] [PubMed] [Google Scholar]
- 23.Kawaguchi M, Hay I, Fetics B, Kass DA. Combined ventricular systolic and arterial stiffening in patients with heart failure and preserved ejection fraction: Implications for systolic and diastolic reserve limitations. Circulation. 2003;107:714–720. doi: 10.1161/01.cir.0000048123.22359.a0. [DOI] [PubMed] [Google Scholar]
- 24.Kasai T, et al. Overnight effects of obstructive sleep apnea and its treatment on stroke volume in patients with heart failure. Can. J. Cardiol. 2015;31:832–838. doi: 10.1016/j.cjca.2015.01.001. [DOI] [PubMed] [Google Scholar]
- 25.Clemmensen TS, et al. Left ventricular pressure-strain-derived myocardial work at rest and during exercise in patients with cardiac amyloidosis. J. Am. Soc. Echocardiogr. 2020;33:573–582. doi: 10.1016/j.echo.2019.11.018. [DOI] [PubMed] [Google Scholar]
- 26.Borrie A, Goggin C, Ershad S, Robinson W, Sasse A. Noninvasive myocardial work index: Characterizing the normal and ischemic response to exercise. J. Am. Soc. Echocardiogr. 2020;33:1191–1200. doi: 10.1016/j.echo.2020.05.003. [DOI] [PubMed] [Google Scholar]
- 27.D'Andrea A, et al. Impaired myocardial work efficiency in heart failure with preserved ejection fraction. Eur. Heart J. Cardiovasc. Imaging. 2021;22:1312–1320. doi: 10.1093/ehjci/jeab153. [DOI] [PubMed] [Google Scholar]
- 28.Edwards NFA, et al. Global myocardial work is superior to global longitudinal strain to predict significant coronary artery disease in patients with normal left ventricular function and wall motion. J. Am. Soc. Echocardiogr. 2019;32:947–957. doi: 10.1016/j.echo.2019.02.014. [DOI] [PubMed] [Google Scholar]
- 29.Chan J, et al. A new approach to assess myocardial work by non-invasive left ventricular pressure–strain relations in hypertension and dilated cardiomyopathy. Eur. Heart J. Cardiovasc. Imaging. 2019;20:31–39. doi: 10.1093/ehjci/jey131. [DOI] [PubMed] [Google Scholar]
- 30.Maimon N, Hanly PJ. Does snoring intensity correlate with the severity of obstructive sleep apnea? J. Clin. Sleep Med. 2010;6:475–478. doi: 10.5664/jcsm.27938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Brown EC, et al. Respiratory movement of upper airway tissue in obstructive sleep apnea. Sleep. 2013;36:1069–1076. doi: 10.5665/sleep.2812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lang RM, et al. Recommendations for cardiac chamber quantification by echocardiography in adults: An update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J. Am. Soc. Echocardiogr. 2015;28:1–39. doi: 10.1016/j.echo.2014.10.003. [DOI] [PubMed] [Google Scholar]
- 33.Lancellotti P, et al. The clinical use of stress echocardiography in non-ischaemic heart disease: Recommendations from the European Association of cardiovascular imaging and the American Society of echocardiography. J. Am. Soc. Echocardiogr. 2017;30:101–138. doi: 10.1016/j.echo.2016.10.016. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data of this study are available from the corresponding author on request due to privacy/ethical restrictions.