Abstract
Background
Postpartum psychiatric disorders (PPD) are common complications of childbirth. A common explanation for their development is that the psychological, hormonal, and immune changes associated with pregnancy and parturition may trigger psychiatric symptoms postpartum. Rheumatoid arthritis (RA) is characterized by abnormalities in the activity of the hypothalamic–pituitary–adrenal axis and of the immune system, but its association with PPD is unknown. We analyzed whether women with RA before childbirth have an increased risk of PPD.
Methods
We conducted a large population-based cohort study including mothers of singleton births in the Danish (1995–2015), Finnish (1997–2013), and Swedish Medical Birth Registers (2001–2013) (N = 3,516,849). We linked data from the Medical Birth Registers with data from several national socioeconomic and health registers. Exposure was defined as having a diagnosis of RA before childbirth, while the main outcome was a clinical diagnosis of psychiatric disorders 90 days postpartum. We analyzed the association between RA and PPD using Cox proportional hazard models, stratified by a personal history of psychiatric disorders.
Results
Among women without a history of psychiatric disorders, the PPD incidence rate was 32.2 in the exposed and 19.5 per 1000 person-years in the unexposed group; women with RA had a higher risk of overall PPD than their unexposed counterparts [adjusted hazard ratio (HR) = 1.52, 95% confidence intervals (CI) 1.17 to 1.98]. Similar associations were also observed for postpartum depression (HR = 1.65, 95% CI 1.09 to 2.48) and other PPD (HR = 1.59, 95% CI 1.13 to 2.24). Among women with a history of psychiatric disorders, the incidence rate of overall PPD was 339.6 in the exposed and 346.6 per 1000 person-years in the unexposed group; RA was not associated with PPD. We observed similar associations between preclinical RA (RA diagnosed after childbirth) and PPD to those corresponding to clinical RA.
Conclusions
Rheumatoid arthritis was associated with an increased PPD risk in women without, but not in those with a psychiatric history. If our findings are confirmed in future studies, new mothers with RA may benefit from increased surveillance for new-onset psychiatric disorders postpartum.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12916-023-02837-3.
Keywords: Postpartum psychiatric disorders, Postpartum depression, Rheumatoid arthritis, Cohort study
Background
Postpartum psychiatric disorders (PPD), with an overall prevalence of 11–15%, are among the most common complications that women experience in relation to childbirth [1]. They may disrupt the developing attachment between the mother and the child and may have important social and health-related consequences for affected women and their families (e.g., increased risks of subsequent chronic medical conditions, premature death, self-harm or suicide, and infanticide) [2, 3]. Postpartum depression is one of the most common PPDs with an estimated prevalence of 5–15% [4, 5]. A number of risk factors for PPD have been identified, including genetic factors, history of psychiatric disorders, primiparity, and pregnancy complications [6, 7]. However, the etiology of PPD is considered very complex and to a large extent unknown.
Immunological mechanisms have been implicated in the pathogenesis of psychiatric disorders both in general population and in postpartum women [8, 9]. One of the immune-related disorders, rheumatoid arthritis (RA), has a prevalence of 1–2% in the general population and affects mostly women aged 30 or older [10]. A large number of studies based on general population samples have documented an increased prevalence of psychiatric disorders in patients with RA [8, 11]. RA is favorably influenced by pregnancy due to the suppression of autoimmunity during pregnancy [12]. After delivery, the relatively immunologically suppressed state of pregnancy shows a rebound and there is a shift from an anti- to a pro-inflammatory state [12]. This may contribute to the exacerbation of the activity of RA. Moreover, women without adequately suppressed immune function and/or prolonged and exaggerated inflammatory response postpartum may be particularly sensitive to difficulties in emotional regulation during the postpartum period [9]. Therefore, a link between RA and new-onset PPD or postpartum relapse in patients with a history of psychiatric disorders is plausible. Furthermore, women with RA before childbirth have an increased risk of pregnancy complications and adverse birth outcomes [13], which may also increase PPD risk. A large cohort study has also shown that inflammatory bowel disease, an immune-mediated inflammatory disease similar to RA, is associated with an increased risk of new-onset PPD [5]. In contrast, another study reported a lower risk of new-onset depression in the year after birth in women with juvenile idiopathic arthritis than in their unexposed counterparts [14]. To our knowledge, no previous study investigated the association between RA before childbirth and the risk of PPD.
In this large cohort study including women from three Nordic countries, we investigated whether women with RA before childbirth had an increased risk of PPD. Since women with previous psychiatric disorders have an increased risk of a relapse postpartum and a more heterogeneous PPD phenotype than women without such a history [15], we also analyzed whether the association differs according to a history of psychiatric disorders.
Methods
Study population and design
This population-based cohort study used data from several nationwide registers in Denmark, Finland, and Sweden, where every resident has a unique personal identification number, allowing linkage between nationwide registers [16]. The registers used for this study are described in detail in Additional file 1: Table S1. Our study population consisted of women with singleton births in the nationwide Medical Birth Registers in Denmark during 1995–2015 [17], in Finland during 1997–2013 [18], and in Sweden during 2001–2013 [19]. We first identified all births in the Medical Birth Registers for the Danish and Swedish cohorts, while for the Finnish cohort, due to the data protection rules of the data holder authority, we could include a randomly selected 90% of the births from the register. The study periods were chosen based on considerations regarding the years when information on both inpatient care and specialized outpatient care was available in the country’s patient register. Our study population consisted of mothers of the abovementioned 3,516,849 singleton births.
Measures
Rheumatoid arthritis
Information on the diagnoses of RA was obtained from the Danish Hospital Register during 1977–2016 [20], the Finnish Hospital Discharge Register during 1987–2015 [21], the Swedish Patient Register during 1968–2014 [22], and the Swedish Medical Birth Register during 2001–2013 [19]. The diagnosis of RA was identified by International Statistical Classification of Diseases and Related Health Problems (ICD) codes (Additional file 1: Table S2). We classified women as exposed to RA if they were diagnosed with either a primary or a secondary diagnosis of RA prior to giving birth. Women without a record of RA before childbirth were considered unexposed. The date of the first hospitalization or the first visit to the outpatient clinic associated with the RA diagnosis was considered the date of the clinical diagnosis of RA.
Prior to the development of RA, there is a period with an increase in levels of disease-related biomarkers similar to that of clinical RA, including autoimmune antibodies and a wide range of inflammatory cytokines and chemokines (termed as “preclinical RA”) [23]. Thus, we also defined exposure to clinical RA (i.e., RA diagnosed before childbirth) and preclinical RA (i.e., RA first diagnosed after childbirth) using the date of first RA diagnosis to disentangle their effects on PPD [24].
Postpartum psychiatric disorders and a history of psychiatric disorders
Information on psychiatric diagnoses was retrieved from the Danish Hospital Register, the Danish Psychiatric Central Research Register [25], the Finnish Hospital Discharge Register, and the Swedish Patient Registers (Additional file 1: Table S1). Our primary outcome was a first diagnosis of a psychiatric disorder (ICD 10: F00-F99) given as primary or secondary diagnosis in hospital inpatient or outpatient care in the first 90 days after delivery. We chose this period as it is the most vulnerable time after birth [26]. To address concerns about statistical power and to consider that there was likely a lag between onset of symptoms and timing of diagnoses, we conducted analyses with a more widely defined postpartum period, i.e., the first 365 days postpartum as our secondary outcome. Each woman was followed from the date of delivery until a PPD, death, emigration, or 90/365 days postpartum, whichever came first. We further categorized PPDs as (1) postpartum depression (given its clinical relevance and prevalence) and (2) other PPD.
We defined a personal history of psychiatric disorders as having at least one record of psychiatric disorders in inpatient or outpatient care prior to delivery; the corresponding ICD codes used are shown in Additional file 1: Table S2. A family history of psychiatric disorders was also identified using the same strategy (i.e., based on biological parents identified in the Danish Civil Registration System and the Swedish Multi-Generation Register).
Other covariates
From the Medical Birth Registers, we obtained information on maternal age at delivery, maternal body mass index (BMI) in early pregnancy (available in Sweden during the whole study period and in Denmark since 2004), calendar year of childbirth, gestational age, birth weight, parity, and stillbirth. Small for gestational age (SGA) was defined as being below the 10th percentile of the birth weight for gestational age distribution based on the Swedish sex-specific reference curve for normal fetal growth [27]. We identified maternal highest education at the time of delivery from the Danish Integrated Database for Longitudinal Labor Market Research and the Education Register in Sweden and Finland, and maternal marital status at delivery from the Danish Civil Registry System, the Swedish Total Population Register, and the Finnish Medical Birth Register. The Danish Integrated Database for Longitudinal Labor Market Research and the Swedish Medical Birth Register provided data on the mothers’ country of origin.
Information on maternal diabetes before or during pregnancy, pre-eclampsia, and placental abruption during the index pregnancy was obtained from the Danish Hospital Register, the Finnish Hospital Discharge Register, and the Swedish Medical Birth Register. A full list of ICD codes used to define the above medical conditions is presented in Additional file 1: Table S2. Information on psychiatric medications was obtained from the Swedish Prescribed Drug Register (since 2005) and the Finnish Prescription Register (since 1995) using the Anatomical Therapeutic Classification codes N05 and N06A [28, 29].
Statistical analyses
We performed Cox proportional hazard models to estimate crude and adjusted hazard ratios (HRs) with 95% confidence intervals (CIs) for the associations between maternal RA and the risk of PPD. Visual inspection of the log–log curves suggested no evidence of violation of the assumption of proportional hazards. Since we found evidence that a history of psychiatric disorders modified the association between RA and PPD, we also presented results stratified by personal history of psychiatric disorders before childbirth. In the main multivariable model, we adjusted for a priori selected confounders which were available in all three countries, i.e., factors associated with both RA and PPD, but not on the causal pathway between RA and PPD: maternal age at delivery (< 25, 25–29, 30–34, or ≥ 35 years), calendar year of childbirth (in 5-year intervals during 1995–2015), parity (primiparous or multiparous), maternal highest education (primary and lower secondary, upper secondary, or university), and marital status at delivery (married/registered partnership vs. not). Since information on maternal BMI in early pregnancy (< 18.5 kg/m2 for underweight, 18.5–24.9 kg/m2 for normal weight, 25.0–29.9 kg/m2 for overweight, and ≥ 30.0 kg/m2 for obesity), family history of psychiatric disorders (yes or no), and country of origin (Denmark/Sweden or other countries) was not available for all study participants, we adjusted for these characteristics in separate models among those with available data on these variables.
We performed separate analyses for exposure to clinical RA and to preclinical RA; these analyses aimed to discriminate the effect of the potential treatment and care from the effect of RA alone because we assumed that women with preclinical RA did not receive specific RA treatment or care before childbirth.
We performed a number of sensitivity analyses. To control for confounding from shared familial factors, we performed an analysis with a sibling design involving women and their sisters from the Danish and Swedish Medical Birth Registers. The sister pairs were identified based on the same ID of their biological mother and were included if both had at least one singleton delivery during the study period. We ran stratified Cox proportional hazards models in a subsample of 666,794 maternal-sister pairs, with a separate stratum for each maternal-sister pair. Only maternal-sister pairs discordant for RA and PPD were informative and contributed to the risk estimates. We additionally adjusted one by one for pre-eclampsia, placental abruption, SGA [27], preterm birth (gestational age < 37 weeks), and stillbirth to investigate whether pregnancy complications and adverse delivery outcomes contributed to the association of interest. Besides, to address potential biases related to selective fertility [30], i.e., that women with a previous PPD or other pregnancy complications or adverse birth outcomes would refrain from engaging in a new pregnancy, we performed analyses restricted to primiparous women. Since diabetes and pre-eclampsia may be regarded as immune-related diseases and were associated with increased risks of PPD [31, 32], we performed stratified analyses and tests of interaction between RA and preeclampsia, and tests of interaction between RA and diabetes to examine the potential effect modification by these conditions. To consider differences over time in the care of pregnant women with RA or in the prevalence of PPD (Additional file 1: Fig. S1), we also conducted a propensity score-matched (PSM) analysis to further balance the exposure groups on potential confounders. The propensity scores were estimated using logistic regression as the probability of exposure to RA given baseline characteristics such as calendar year of childbirth, parity, maternal age at delivery, and study country. Each woman exposed to RA was matched to an unexposed woman on the propensity scores by using greedy match algorithms with a ratio of 1:1. We then repeated the analyses in the PSM sub-cohort with additional adjustments for the woman’s highest education and marital status at delivery. To increase the specificity of the definition of preclinical RA, we also performed analyses with a narrowly defined preclinical RA, i.e., RA diagnoses in the year after delivery. To account for the effect of use of psychiatric medication among women with a history of psychiatric disorders, we analyzed in this sub-cohort the association between RA and PPD after stratifying by a history of psychiatric medication use. Finally, we repeated our main analyses with our secondary outcome, i.e., PPD in the 365 days after delivery.
Statistical analyses were performed using SAS version 9.2. A two-tailed p-value of < 0.05 was considered statistically significant.
Results
Of the 3,516,849 childbirths of women included in our study, 293,371 (8.34%) had and 3,223,478 (91.66%) did not have a history of psychiatric disorders. The incidence rates of PPD in the first 90 days postpartum for the two groups were 346.5/1000 person-years and 19.5/1000 person-years, respectively (Additional file 1: Table S3). Altogether, 8407 (0.24%) births were exposed to maternal RA and 3,508,442 (99.76%) were unexposed (Table 1). Compared to women without RA, women with RA were more likely to be older and better educated and to have higher BMI in early pregnancy and less likely to be married or in a registered partnership at the time of delivery. They were also more likely to have a personal and a family history of psychiatric disorders, as well as pre-eclampsia and placental abruption during the index birth. In addition, exposed mothers were more likely to have preterm births, SGA births, and stillbirths compared to those in the unexposed group (Table 1). Characteristics of women exposed to preclinical RA relative to that of women with clinical RA or unexposed women are shown in Additional file 1: Table S4.
Table 1.
Characteristics of the study population according to maternal rheumatoid arthritis before childbirth
| Births of women | ||||
|---|---|---|---|---|
| With RA (N = 8407, 0.24%) |
Without RA (N = 3,508,442, 99.76%) |
|||
| No | % | No | % | |
| Maternal characteristics | ||||
| Study country | ||||
| Denmark | 4297 | 51.11 | 1,291,985 | 36.83 |
| Finland | 215 | 2.56 | 912,370 | 26.00 |
| Sweden | 3895 | 46.33 | 1,304,087 | 37.17 |
| Maternal age at delivery (years) | ||||
| < 25 | 735 | 8.74 | 539,085 | 15.37 |
| 25–29 | 2271 | 27.02 | 1,116,203 | 31.81 |
| 30–34 | 3163 | 37.62 | 1,188,931 | 33.89 |
| ≥ 35 | 2238 | 26.62 | 664,223 | 18.93 |
| Maternal country of origin a | ||||
| Denmark/Sweden | 4262 | 52.03 | 1,494,124 | 57.55 |
| Other country | 3929 | 47.96 | 1,099,794 | 42.37 |
| Unknown | < 5 | 0.01 | 2154 | 0.08 |
| Maternal BMI in early pregnancy (kg/m2)a | ||||
| < 18.5 | 204 | 2.49 | 57,841 | 2.23 |
| 18.5–24.9 | 3728 | 45.51 | 1,084,402 | 41.76 |
| 25.0–29.9 | 1654 | 20.19 | 475,472 | 18.32 |
| ≥ 30.0 | 876 | 10.69 | 235,133 | 9.06 |
| Unknown | 1730 | 21.12 | 743,224 | 28.63 |
| Marital status at delivery | ||||
| Single, widowed or divorced | 4710 | 56.03 | 1,761,697 | 50.21 |
| Married or in registered partnership | 3690 | 43.89 | 1,712,614 | 48.82 |
| Unknown | 7 | 0.08 | 34,131 | 0.97 |
| Maternal highest education at delivery | ||||
| Primary and lower secondary | 1226 | 14.58 | 792,049 | 22.58 |
| Upper secondary | 3449 | 41.03 | 1,235,290 | 35.21 |
| University | 3668 | 43.63 | 1,334,893 | 38.05 |
| Unknown | 64 | 0.76 | 146,210 | 4.16 |
| Maternal smoking in early pregnancy | ||||
| No | 7122 | 84.72 | 2,954,987 | 84.23 |
| Yes | 957 | 11.38 | 421,187 | 12.00 |
| Unknown | 328 | 3.90 | 132,268 | 3.77 |
| Parity | ||||
| Primiparous | 3533 | 42.02 | 1,441,227 | 41.08 |
| Multiparous | 4874 | 57.98 | 2,066,8347 (58. 90) | 58. 90 |
| Unknown | 0 | 0.00 | 868 | 0.02 |
| Psychiatric disorders before childbirth | ||||
| No | 7221 | 85.89 | 3,216,257 | 91.67 |
| Yes | 1186 | 14.11 | 292,185 | 8.33 |
| Family history of psychiatric disordersa | ||||
| No | 6631 | 80.94 | 1,960,464 | 75.52 |
| Yes | 1040 | 12.70 | 240,350 | 9.26 |
| Unknown b | 521 | 6.36 | 395,258 | 15.24 |
| Diabetes | ||||
| No | 7984 | 94.97 | 3,349,807 | 95.48 |
| Yes | 423 | 5.03 | 158,735 | 4.52 |
| Pre-eclampsia | ||||
| No | 7996 | 95.11 | 3,410,056 | 97.20 |
| Yes | 411 | 4.89 | 98,386 | 2.80 |
| Placental abruption | ||||
| No | 8363 | 99.48 | 3,496,287 | 99.65 |
| Yes | 44 | 0.52 | 12,155 | 0.35 |
| Characteristics of the birth | ||||
| Calendar year of birth | ||||
| 1995–2000 | 759 | 9.03 | 566,190 | 16.14 |
| 2001–2005 | 2077 | 24.70 | 1,048,250 | 29.88 |
| 2006–2010 | 3017 | 35.89 | 1,119,437 | 31.90 |
| 2011–2015 | 2554 | 30.38 | 774,565 | 22.08 |
| Small for gestational age | ||||
| No | 7280 | 86.60 | 3,115,135 | 88.79 |
| Yes | 1009 | 12.00 | 346,001 | 9.86 |
| Unknown | 118 | 1.40 | 47,036 | 1.35 |
| Preterm birth (gestational < 37 weeks) | ||||
| No | 7675 | 91.30 | 3,307,208 | 94.26 |
| Yes | 667 | 7.93 | 170,470 | 4.86 |
| Unknown c | 65 | 0.77 | 30,764 | 0.88 |
| Stillbirth | ||||
| No | 8367 | 99.52 | 3,499,154 | 99.74 |
| Yes | 40 | 0.48 | 9288 | 0.26 |
RA rheumatoid arthritis, No. number, BMI body-mass index
aAvailable only in Sweden and Denmark
bThere was no register link to the biological mother or father
cIncludes the missing or unlikely short (< 22 weeks in Denmark and in Sweden), or long gestational ages (> 45 weeks in Denmark; > 46 weeks in Sweden)
Main analyses
Among women exposed to RA, 1.8% had a PPD, 0.8% postpartum depression, and 1.0% other PPD, while corresponding figures among the unexposed women were 1.1% for overall PPD, 0.4% for postpartum depression, and 0.6% for other PPD, respectively. Compared to women without RA, women with RA had higher risks of overall PPD (HR = 1.39, 95% CI 1.18 to 1.64), postpartum depression (HR = 1.37, 95% CI 1.07 to 1.77), and other PPD (HR = 1.66, 95% CI 1.34 to 2.05). In analyses stratified by a history of psychiatric disorders before childbirth, we observed increased risks of new-onset overall PPD (HR = 1.52, 95% CI 1.17 to 1.98), postpartum depression (HR = 1.65, 95% CI 1.09 to 2.48), and other PPD (HR = 1.59, 95% CI 1.13 to 2.24) in women with RA relative to their unexposed counterparts. There were no associations between RA and PPD among women with a history of psychiatric disorders before childbirth (Table 2).
Table 2.
Hazard ratios with 95% confidence intervals for postpartum psychiatric disorders within 90 days after delivery according to rheumatoid arthritis before childbirth, and stratified by a personal history of psychiatric disorders
| Outcome | Exposure | No | % | Incidence rate | Crude model | Adjusted model a | P-value for the interaction b |
|---|---|---|---|---|---|---|---|
| (per 1000 person-years) | HRs (95% CI) | HRs (95% CI) | |||||
| All births | |||||||
| Overall PPD | Unexposed to RA | 38,790 | 1.1 | 45.3 | 1.00 (Ref.) | 1.00 (Ref.) | < 0.001 |
| Exposed to RA | 150 | 1.8 | 73.5 | 1.62 (1.38, 1.90) | 1.39 (1.18, 1.64) | ||
| Postpartum depression | Unexposed to RA | 15,548 | 0.4 | 18 | 1.00 (Ref.) | 1.00 (Ref.) | 0.02 |
| Exposed to RA | 63 | 0.8 | 30.6 | 1.69 (1.32, 2.17) | 1.37 (1.07, 1.77) | ||
| Other psychiatric disorders postpartum | Unexposed to RA | 19,396 | 0.6 | 22.6 | 1.00 (Ref.) | 1.00 (Ref.) | 0.18 |
| Exposed to RA | 87 | 1 | 42.1 | 1.86 (1.50, 2.30) | 1.66 (1.34, 2.05) | ||
| Births by women without a history of psychiatric disorders | |||||||
| Overall PPD | Unexposed to RA | 15,374 | 0.5 | 19.5 | 1.00 (Ref.) | 1.00 (Ref.) | |
| Exposed to RA | 57 | 0.8 | 32.2 | 1.66 (1.28, 2.15) | 1.52 (1.17, 1.98) | ||
| Postpartum depression | Unexposed to RA | 5678 | 0.2 | 7.2 | 1.00 (Ref.) | 1.00 (Ref.) | |
| Exposed to RA | 23 | 0.3 | 13 | 1.81 (1.20, 2.72) | 1.65 (1.09, 2.48) | ||
| Other psychiatric disorders postpartum | Unexposed to RA | 8675 | 0.3 | 11 | 1.00 (Ref.) | 1.00 (Ref.) | |
| Exposed to RA | 34 | 0,5 | 19.2 | 1.75 (1.25, 2.45) | 1.59 (1.13, 2.24) | ||
| Births by women with a history of psychiatric disorders | |||||||
| Overall PPD | Unexposed to RA | 23,416 | 8 | 346.6 | 1.00 (Ref.) | 1.00 (Ref.) | |
| Exposed to RA | 93 | 7.8 | 339.6 | 0.98 (0.80, 1.20) | 0.88 (0.72, 1.09) | ||
| Postpartum depression | Unexposed to RA | 9870 | 3.4 | 140.7 | 1.00 (Ref.) | 1.00 (Ref.) | |
| Exposed to RA | 40 | 3.4 | 140.5 | 1 (0.73, 1.36) | 0.84 (0.60, 1.15) | ||
| Other psychiatric disorders postpartum | Unexposed to RA | 10,721 | 3.7 | 158.7 | 1.00 (Ref.) | 1.00 (Ref.) | |
| Exposed to RA | 53 | 4.4 | 190 | 1.2 (0.91, 1.57) | 1.15 (0.88, 1.51) | ||
HRs hazard ratios, CI confidence interval, No. number, PPD postpartum psychiatric disorders, RA rheumatoid arthritis, Ref. reference
aAdjusted for maternal age at delivery, calendar year of childbirth, parity, maternal highest education levels at delivery, and maternal marital status at delivery
bP-values correspond to the interaction between RA and a history of psychiatric disorders
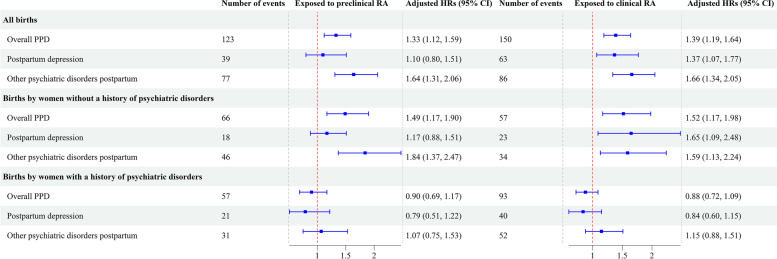
Both women with clinical RA and preclinical RA had increased risks of overall PPD and other psychiatric disorders during the first 90 days postpartum (Fig. 1). We found an increased risk of postpartum depression during the first 90 days postpartum only among women with clinical RA. However, when we analyzed postpartum depression during the first 365 days after delivery as the outcome, we observed increased risks of postpartum depression both in women with preclinical RA and clinical RA compared to women unexposed to clinical and preclinical RA (HR = 1.23, 95% CI 1.00 to 1.50 for clinical RA; HR = 1.27, 95% CI 1.03 to 1.56 for preclinical RA, Additional file 1: Fig. S2). In analyses stratified by a history of psychiatric disorders, preclinical RA was associated with increased risks of overall PPD, postpartum depression, and other PPD among women without a history of psychiatric disorders, but not among those with such a history (Fig. 1).
Fig. 1.
Hazard ratios and 95% confidence intervals for postpartum psychiatric disorders within 90 days after childbirth according to clinical rheumatoid arthritis (rheumatoid arthritis diagnosed before childbirth) and preclinical rheumatoid arthritis (rheumatoid arthritis diagnosed after childbirth). Abbreviations: HRs, hazard ratios; CI, confidence interval; PPD, postpartum psychiatric disorders; RA, rheumatoid arthritis. Adjusted for maternal age at delivery, calendar year of childbirth, parity, maternal highest education levels at delivery, and maternal marital status at delivery. The reference group consists of births by women who unexposed to clinical RA and preclinical RA
Sensitivity analyses
Further adjustment for maternal BMI in early pregnancy, country of origin, and a family history of psychiatric disorders among those with such data did not change the results substantially (Additional file 1: Table S5 and S6). In the sibling analyses, the association between RA and PPD disappeared, but we observed a trend towards an association between RA and an increased risk of new-onset postpartum depression among women without a psychiatric history (Additional file 1: Table S7). Adjusting for SGA, preterm birth, stillbirth, pre-eclampsia, or placental abruption one by one did not substantially alter the association between RA and the risk of PPD (Additional file 1: Table S8). The point estimates corresponding to the association between RA and PPD, postpartum depression, and other PPD among women without a history of psychiatric disorders before childbirth were somewhat higher when we restricted the analyses to primiparous women compared with those observed in the main analyses (Additional file 1: Table S9). The association of RA with the risk of new-onset PPD was stronger in diabetic women than among non-diabetic women (P-value for interaction = 0.02, Additional file 1: Table S10). The associations did not differ between women with and without pre-eclampsia.
After performing a PSM analysis, the exposed (N = 8407) and unexposed (N = 8407) cohorts were comparable in terms of most of the baseline characteristics at birth (Additional file 1: Table S11). The patterns of the associations were similar to those observed in the main analyses (Additional file 1: Table S12). In analyses restricted to women with a history of psychiatric disorders, we found that RA tended to be associated with an increased risk of PPD in women without a history of psychiatric medications, but not in those with a history of psychiatric medications (Additional file 1: Table S13). When we restricted the definition of preclinical RA to having the first diagnosis of RA 1 year after childbirth (Additional file 1: Fig. S3) or performed analyses with our secondary outcome, i.e., PPD in the first 365 days, results were similar to those obtained in the primary analyses (Additional file 1: Fig. S4).
Discussion
We found that RA was associated with increased risks of new-onset PPDs, including postpartum depression, and other PPD among women without a history of psychiatric disorders, but not among women with a history of psychiatric disorders. We also observed largely similar associations between preclinical RA (i.e., RA diagnosed after childbirth) and PPD to those corresponding to clinical RA.
Several population-based cohort studies examined the associations of immune-related disorders, e.g., pre-eclampsia, autoimmune thyroid disorders, inflammatory bowel disease, and atopic diseases, with PPD risk [5, 32–34]. Only Feldman and associates studied the association between some type of rheumatic disease and the risk of PPD; they found that women with juvenile idiopathic arthritis had a lower risk of postpartum depression than their unexposed counterparts [14]. Earlier studies regarding the association between different types of rheumatic disease and the risk of psychiatric disorders in general population samples have also been inconsistent [35–37]. Previous studies regarding immune-related disorders and PPD risk generally excluded multiparous women or women with a history of psychiatric disorders before delivery. We are the first to examine the associations between RA before childbirth and PPD risk, as well as to stratify analyses concerning immune-related diseases and PPD risk by women’s history of psychiatric disorders.
The lack of an association among women with a history of psychiatric disorders in our study may be somewhat unexpected, given the well-documented increased risk of postpartum psychiatric relapse in this group [38]. One possible explanation is that the diagnosis of PPD among women with a history of psychiatric disorders is very likely to be a regular visit for their pre-existing psychiatric disorder. If the PPD only accounts for a minor part of all psychiatric disorders in women with a history of psychiatric disorder, the competing risk from other psychiatric disorders may have attenuated the association between RA and PPD in this group. Second, in anticipation of their potentially difficult postpartum period arising from a high psychiatric vulnerability, increased RA activity, sleep deprivation, and the psychological demands of caring for a newborn, women with a history of psychiatric disorders may be more likely to receive prophylactic antidepressants or anti-psychotic medications. When we further performed stratified analyses according to their prescriptions of psychiatric medications, we found a trend towards an association between RA and an increased risk of PPD only among women without a history of psychiatric medications. This result partially supports that intake of psychiatric medications might contribute to the explanation of our lack of an association between RA and PPD among women with a history of psychiatric disorders. However, we did not have information on psychiatric medication for a sufficiently large part of the cohort to detect significant differences in this respect. Third, since only psychiatric disorders diagnosed in specialized outpatient care and inpatient care are included in the Nordic registers, there is likely to be an underreporting with respect to the history of psychiatric disorders [25]; this misclassification may have limited statistical power to detect modest associations between RA and PPD in this group.
A concern to address in studies regarding the association between RA and the risk of psychiatric disorders is the separation of the inflammation-related effects of RA from the effects of RA treatment and care. We tried to disentangle the role of treatment and care for RA from the RA per se by examining the risk of PPD among women with preclinical RA, given that the preclinical RA involves increased inflammation typically associated with active RA, but without severe symptoms, taking any treatment and care yet [23]. Increased risks of postpartum depression during the first 365 days after birth were observed irrespective of whether the women were exposed to clinical or preclinical RA, although the risks were less consistent for postpartum depression during the first 90 days due to limited power in the group with preclinical RA. These findings may point towards an effect of the disease rather than the effects of RA-specific treatment or care or the effects of limitations of life activities resulting from severe RA symptoms. Another potential explanation for this finding is that women with PPD might also have a higher overall risk of subsequent RA, indicating a bidirectional association between RA and PPD, as suggested in a previous study [39, 40].
The potential biological mechanisms underlying the association between RA and PPD are unclear. The role of inflammation in the association between RA and depression outside the pregnancy is well-known [8] and may also account for the association between RA and postpartum depression. Upregulation of major proinflammatory cytokines, such as inter-leukin-1β and tumor necrosis factor α, play an important role in the pathogenesis of RA [12]. Similarly, the activation of the inflammatory response is involved in the pathophysiology of postpartum depression [41]. Potential mechanisms linking inflammation due to RA to changes in the activity of the central nervous system have been identified in several animal models [42–44]. Immune-mediated inflammation can modulate neurotransmission, neurogenesis, neuroendocrine activity, and neuroplasticity and affect corticolimbic circuits involved in emotion and stress regulation [8]. Additionally, the cytokines could induce alterations in HPA axis and glucocorticoid function, which are well known to be associated with PPD [8]. We found that in women without a history of psychiatric disorders, the association between RA and overall PPD was strengthened by the presence of diabetes. Given that alterations in dysregulation of the HPA axis and cytokine-mediated inflammatory responses can be induced by either RA or diabetes [45, 46], an interaction between shared mechanistic pathways might be biologically plausible. Further studies are needed to explore the mechanisms underlying the observed associations.
The main strength of our study is the use of large-scale register-based data from Denmark, Finland, and Sweden. The study includes nearly all births in these countries during up to two decades and prospectively collects information on exposure, outcome, and covariates, which reduces the risks of selection and recall bias. Some limitations of the study need to be taken into consideration when interpreting our findings. First, as our study was register-based, we could identify only the most severe psychiatric disorders, but not patients who sought care only in primary care [25], and thus, we may have underestimated the incidence of PPD and that of personal and family history of psychiatric disorders. Validation studies of the RA diagnoses in the National Patient Register reported a positive predictive value of 59% in Danish data [47], 82% in Finnish data [48], and 91% in Swedish data [49]. However, these potential misclassifications of exposure and outcome would most likely be non-differential and thus might bias the risk estimates towards the null. Second, although we adjusted for a wide range of potential confounders, residual confounding cannot be ruled out, given that some important information on treatment of RA, RA disease activity, and available informal care and social support of RA was not available. However, we expect the potential impact of RA treatment and care to be modest because of the similar associations we observed between preclinical RA and PPD. Third, although we have a large cohort from three countries, the number of events is small in some sub-analyses. Fourth, we used sibling design to control for unmeasured genetic and environmental factors shared by families and observed a trend towards an increased risk of postpartum depression in women with RA. However, caution is needed when interpreting these findings given the very limited number of siblings discordant for exposure and outcome. Additionally, sibling comparisons are, by design, associated with some limitations, such as residual confounding not shared by siblings [50].
Conclusions
In this large population-based cohort study from three Nordic countries, we found that among women without a history of psychiatric disorders, those with RA had a higher risk of PPD, especially postpartum depression, than their unexposed counterparts. There were no associations between RA and PPD among women with a history of psychiatric disorders. If our findings are confirmed in future studies, new mothers with RA may benefit from increased surveillance for new-onset psychiatric disorders postpartum.
Supplementary Information
Additional file 1: Table S1. Description of the registers included in the study. Table S2. The International Classification of Diseases codes used to define medical conditions. Table S3. Incidence proportion and incidence rates of postpartum psychiatric disorders within 90 days after childbirth in the study cohort. Table S4. Characteristics of the study population according to clinical rheumatoid arthritis and preclinical rheumatoid arthritis. Table S5. Hazard ratios and 95% confidence intervals for postpartum psychiatric disorders within 90 days after childbirth according to rheumatoid arthritis before childbirth, after additional adjustment for maternal body-mass index during early pregnancy. Table S6. Hazard ratios and 95% confidence intervals for postpartum psychiatric disorders within 90 days after childbirth according to rheumatoid arthritis before childbirth, after additional adjustment for maternal country origin and family history of psychiatric disorders. Table S7. Hazard ratios and 95% confidence intervals for postpartum psychiatric disorders within 90 days after childbirth according to rheumatoid arthritis before childbirth in a sibling design. Table S8. Hazard ratios and 95% confidence intervals for postpartum psychiatric disorders within 90 days after childbirth according to rheumatoid arthritis before childbirth after adjusting for potential explanatory factors. Table S9. Hazard ratios and 95% confidence intervals for postpartum psychiatric disorders within 90 days after childbirth according to rheumatoid arthritis before childbirth, in analyses restricted to primiparous women. Table S10. Hazard ratios and 95% confidence intervals for postpartum psychiatric disorders within 90 days after childbirth according to rheumatoid arthritis before childbirth in stratified analyses. Table S11. Baseline characteristics of the propensity score matching sub-cohort. Table S12. Hazard ratios and 95% confidence intervals for postpartum psychiatric disorders within 90 days after childbirth according to rheumatoid arthritis before childbirth in the propensity-matched sub-cohort. Table S13. Hazard ratios and 95% confidence intervals for the associations between rheumatoid arthritis before childbirth and postpartum psychiatric disorders within 90 days after childbirth among women with a personal history of psychiatric disorders, further stratified by previous use of psychiatric medication. Fig S1. The time trend of the postpartum psychiatric disorder rates over the study period in the exposed and the unexposed group. Fig S2. Hazard ratios and 95% confidence intervals for postpartum psychiatric disorders within 365 days after childbirth according to clinical rheumatoid arthritis (rheumatoid arthritis diagnosed before childbirth) and preclinical rheumatoid arthritis (rheumatoid arthritis diagnosed after childbirth). Fig S3. Hazard ratios and 95% confidence intervals for postpartum psychiatric disorders within 365 days after childbirth according to clinical rheumatoid arthritis (rheumatoid arthritis diagnosed before childbirth) and preclinical rheumatoid arthritis (rheumatoid arthritis diagnosed in the first year after childbirth). Fig S4. Hazard ratios and 95% confidence intervals for the associations between rheumatoid arthritis before childbirth and postpartum psychiatric disorders within the first 365 days after delivery, stratified by a personal history of psychiatric disorders.
Acknowledgements
Not applicable.
Abbreviations
- BMI
Body mass index
- CIs
Confidence intervals
- HRs
Hazard ratios
- ICD
International Statistical Classification of Diseases and Related Health Problems
- PPD
Postpartum psychiatric disorders
- PSM
Propensity score-matched
- RA
Rheumatoid arthritis
- SGA
Small for gestational age
Authors’ contributions
KL conceived and designed the study and supervised and directed the conduct of the study. KL, ML, FY, MM, and WY developed the initial analysis plan. ML performed the data management and statistical analyses and drafted the manuscript. JL, MG, and KL requested the register data from their respective countries. FY also contributed to the actual data management and statistical analyses. JL and KL provided funding. ML, FY, MM, WY, MG, EA, DL, JL, and KL contributed to the interpretation of the results and critically revised the manuscript for important intellectual content. All authors read and approved the final version of the manuscript and agree to be accountable for their work. The corresponding author attests that all listed authors meet the authorship criteria.
Funding
Open access funding provided by Karolinska Institute. This study was supported by the China Scholarship Council (202006100013), the Swedish Council for Working Life and Social Research (2015–00837), and National Natural Science Foundation of China (82073570). Jiong Li is supported by the Novo Nordisk Foundation (NNF18OC0052029); Nordic Cancer Union (R275-A15770 and R278-A15877); Danish Council for Independent Research (DFF-6110-00019B, 9039-00010B, and 1030-00012B); and Karen Elise Jensens Fond (2016). Maohua Miao is supported by the Innovation-oriented Science and Technology Grant from NHC Key Lab of Reproduction Regulation (CX2022-04). Krisztina László is supported by the Swedish Heart and Lung Foundation (20180306).
Availability of data and materials
Data used for this study are not openly available because the law and the ethical permit does not allow to share them. The data were analyzed on a secure server at Statistics Denmark run by Aarhus University.
Declarations
Ethics approval and consent to participate
According to Danish, Finish, and Swedish legislation, register-based studies do not require informed consent. The study was approved by the Danish Data Protection Agency (No. 2013–41-2569) and the Research Ethics Committee at Karolinska Institute in Stockholm (No. 2016/288–31/1, 2021–03315). The requirement for ethical approval was waived in Finland, since the data were anonymous and the registered individuals were not contacted.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Meltzer-Brody S, Howard LM, Bergink V, Vigod S, Jones I, Munk-Olsen T, Honikman S, Milgrom J. Postpartum psychiatric disorders. Nat Rev Dis Primers. 2018;4:18022. doi: 10.1038/nrdp.2018.22. [DOI] [PubMed] [Google Scholar]
- 2.Johannsen BMW, Laursen TM, Bech BH, Munk-Olsen T. General medical conditions and mortality in women with postpartum psychiatric disorders. Acta Psychiatr Scand. 2020;142(6):467–475. doi: 10.1111/acps.13232. [DOI] [PubMed] [Google Scholar]
- 3.Valdimarsdóttir U, Hultman CM, Harlow B, Cnattingius S, Sparén P. Psychotic illness in first-time mothers with no previous psychiatric hospitalizations: a population-based study. PLoS Med. 2009;6(2):e13. doi: 10.1371/journal.pmed.1000013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Gaynes BN, Gavin N, Meltzer-Brody S, Lohr KN, Swinson T, Gartlehner G, Brody S, Miller WC. Perinatal depression: prevalence, screening accuracy, and screening outcomes. Evid Rep Technol Assess (Summ) 2005;119:1–8. doi: 10.1037/e439372005-001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Vigod SN, Kurdyak P, Brown HK, Nguyen GC, Targownik LE, Seow CH, Kuenzig ME, Benchimol EI. Inflammatory bowel disease and new-onset psychiatric disorders in pregnancy and post partum: a population-based cohort study. Gut. 2019;68(9):1597–1605. doi: 10.1136/gutjnl-2018-317610. [DOI] [PubMed] [Google Scholar]
- 6.Bauer AE, Maegbaek ML, Liu X, Wray NR, Sullivan PF, Miller WC, Meltzer-Brody S, Munk-Olsen T. Familiality of psychiatric disorders and risk of postpartum psychiatric episodes: a population-based cohort study. Am J Psychiatry. 2018;175(8):783–791. doi: 10.1176/appi.ajp.2018.17111184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Meltzer-Brody S, Maegbaek ML, Medland SE, Miller WC, Sullivan P, Munk-Olsen T. Obstetrical, pregnancy and socio-economic predictors for new-onset severe postpartum psychiatric disorders in primiparous women. Psychol Med. 2017;47(8):1427–1441. doi: 10.1017/S0033291716003020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Nerurkar L, Siebert S, McInnes IB, Cavanagh J. Rheumatoid arthritis and depression: an inflammatory perspective. Lancet Psychiatry. 2019;6(2):164–173. doi: 10.1016/S2215-0366(18)30255-4. [DOI] [PubMed] [Google Scholar]
- 9.Corwin EJ, Pajer K. The psychoneuroimmunology of postpartum depression. J Womens Health (Larchmt) 2008;17(9):1529–1534. doi: 10.1089/jwh.2007.0725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Symmons DP. Epidemiology of rheumatoid arthritis: determinants of onset, persistence and outcome. Best Pract Res Clin Rheumatol. 2002;16(5):707–722. doi: 10.1053/berh.2002.0257. [DOI] [PubMed] [Google Scholar]
- 11.Marrie RA, Bernstein CN. Psychiatric comorbidity in immune-mediated inflammatory diseases. World Psychiatry. 2021;20(2):298–299. doi: 10.1002/wps.20873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ostensen M, Villiger PM. The remission of rheumatoid arthritis during pregnancy. Semin Immunopathol. 2007;29(2):185–191. doi: 10.1007/s00281-007-0072-5. [DOI] [PubMed] [Google Scholar]
- 13.Littlejohn EA. Pregnancy and rheumatoid arthritis. Best Pract Res Clin Obstet Gynaecol. 2020;64:52–58. doi: 10.1016/j.bpobgyn.2019.09.005. [DOI] [PubMed] [Google Scholar]
- 14.Ehrmann Feldman D, Vinet É, Sylvestre MP, Hazel B, Duffy C, Bérard A, Meshefedjian G, Bernatsky S. Postpartum complications in new mothers with juvenile idiopathic arthritis: a population-based cohort study. Rheumatology (Oxford) 2017;56(8):1378–1385. doi: 10.1093/rheumatology/kex168. [DOI] [PubMed] [Google Scholar]
- 15.Rasmussen MH, Strøm M, Wohlfahrt J, Videbech P, Melbye M. Risk, treatment duration, and recurrence risk of postpartum affective disorder in women with no prior psychiatric history: a population-based cohort study. PLoS Med. 2017;14(9):e1002392. doi: 10.1371/journal.pmed.1002392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Li J, Vestergaard M, Obel C, Cnattingus S, Gissler M, Olsen J. Cohort profile: the Nordic Perinatal Bereavement Cohort. Int J Epidemiol. 2011;40(5):1161–1167. doi: 10.1093/ije/dyq127. [DOI] [PubMed] [Google Scholar]
- 17.Bliddal M, Broe A, Pottegård A, Olsen J, Langhoff-Roos J. The Danish Medical Birth Register. Eur J Epidemiol. 2018;33(1):27–36. doi: 10.1007/s10654-018-0356-1. [DOI] [PubMed] [Google Scholar]
- 18.Statistics Finland, the Finnish Medical Birth Register, https://thl.fi/en/web/thlfi-en/statistics-and-data/data-and-services/register-descriptions/newborns. Accessed 10 Nov 2022.
- 19.Axelsson O. The Swedish medical birth register. Acta Obstet Gynecol Scand. 2003;82(6):491–492. doi: 10.1034/j.1600-0412.2003.00172.x. [DOI] [PubMed] [Google Scholar]
- 20.Andersen TF, Madsen M, Jørgensen J, Mellemkjoer L, Olsen JH. The Danish National Hospital Register. A valuable source of data for modern health sciences. Dan Med Bull. 1999;46(3):263–268. [PubMed] [Google Scholar]
- 21.Sund R. Quality of the Finnish Hospital discharge register: a systematic review. Scand J Public Health. 2012;40(6):505–515. doi: 10.1177/1403494812456637. [DOI] [PubMed] [Google Scholar]
- 22.Waldenlind K, Eriksson JK, Grewin B, Askling J. Validation of the rheumatoid arthritis diagnosis in the Swedish National Patient Register: a cohort study from Stockholm County. BMC Musculoskelet Disord. 2014;15:432. doi: 10.1186/1471-2474-15-432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Deane KD. Preclinical rheumatoid arthritis (autoantibodies): an updated review. Curr Rheumatol Rep. 2014;16(5):419. doi: 10.1007/s11926-014-0419-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rom AL, Wu CS, Olsen J, Jawaheer D, Hetland ML, Christensen J, Ottesen B, Morch LS. Parental rheumatoid arthritis and childhood epilepsy: a nationwide cohort study. Neurology. 2016;87(24):2510–2516. doi: 10.1212/WNL.0000000000003424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mors O, Perto GP, Mortensen PB. The Danish Psychiatric Central Research Register. Scand J Public Health. 2011;39(7 Suppl):54–57. doi: 10.1177/1403494810395825. [DOI] [PubMed] [Google Scholar]
- 26.Munk-Olsen T, Laursen TM, Pedersen CB, Mors O, Mortensen PB. New parents and mental disorders: a population-based register study. JAMA. 2006;296(21):2582–2589. doi: 10.1001/jama.296.21.2582. [DOI] [PubMed] [Google Scholar]
- 27.Marsal K, Persson PH, Larsen T, Lilja H, Selbing A, Sultan B. Intrauterine growth curves based on ultrasonically estimated foetal weights. Acta Paediatr. 1996;85(7):843–848. doi: 10.1111/j.1651-2227.1996.tb14164.x. [DOI] [PubMed] [Google Scholar]
- 28.Wettermark B, Hammar N, Fored CM, Leimanis A, OtterbladOlausson P, Bergman U, Persson I, Sundström A, Westerholm B, Rosén M. The new Swedish Prescribed Drug Register–opportunities for pharmacoepidemiological research and experience from the first six months. Pharmacoepidemiol Drug Saf. 2007;16(7):726–735. doi: 10.1002/pds.1294. [DOI] [PubMed] [Google Scholar]
- 29.Rikala M, Hartikainen S, Sulkava R, Korhonen MJ. Validity of the Finnish Prescription Register for measuring psychotropic drug exposures among elderly finns: a population-based intervention study. Drugs Aging. 2010;27(4):337–349. doi: 10.2165/11315960-000000000-00000. [DOI] [PubMed] [Google Scholar]
- 30.Dolman C, Jones IR, Howard LM. Women with bipolar disorder and pregnancy: factors influencing their decision-making. BJPsych Open. 2016;2(5):294–300. doi: 10.1192/bjpo.bp.116.003079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ruohomaki A, Toffol E, Upadhyaya S, Keski-Nisula L, Pekkanen J, Lampi J, Voutilainen S, Tuomainen TP, Heinonen S, Kumpulainen K, et al. The association between gestational diabetes mellitus and postpartum depressive symptomatology: a prospective cohort study. J Affect Disorders. 2018;241:263–268. doi: 10.1016/j.jad.2018.08.070. [DOI] [PubMed] [Google Scholar]
- 32.Bergink V, Laursen TM, Johannsen BM, Kushner SA, Meltzer-Brody S, Munk-Olsen T. Pre-eclampsia and first-onset postpartum psychiatric episodes: a Danish population-based cohort study. Psychol Med. 2015;45(16):3481–3489. doi: 10.1017/S0033291715001385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Bergink V, Pop VJM, Nielsen PR, Agerbo E, Munk-Olsen T, Liu X. Comorbidity of autoimmune thyroid disorders and psychiatric disorders during the postpartum period: a Danish nationwide register-based cohort study. Psychol Med. 2018;48(8):1291–1298. doi: 10.1017/S0033291717002732. [DOI] [PubMed] [Google Scholar]
- 34.Ren T, Chen J, Yu Y, He H, Zhang J, Li F, Svendsen K, Obel C, Wang H, Li J. The association of asthma, atopic dermatitis, and allergic rhinitis with peripartum mental disorders. Clin Transl Allergy. 2021;11(10):e12082. doi: 10.1002/clt2.12082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.van't Land H, Verdurmen J, Ten Have M, van Dorsselaer S, Beekman A, de Graaf R. The association between arthritis and psychiatric disorders; results from a longitudinal population-based study. J Psychosom Res. 2010;68(2):187–193. doi: 10.1016/j.jpsychores.2009.05.011. [DOI] [PubMed] [Google Scholar]
- 36.Packham JC, Hall MA, Pimm TJ. Long-term follow-up of 246 adults with juvenile idiopathic arthritis: predictive factors for mood and pain. Rheumatology (Oxford) 2002;41(12):1444–1449. doi: 10.1093/rheumatology/41.12.1444. [DOI] [PubMed] [Google Scholar]
- 37.Shen CC, Hu LY, Yang AC, Kuo BI, Chiang YY, Tsai SJ. Risk of psychiatric disorders following ankylosing spondylitis: a nationwide population-based retrospective cohort study. J Rheumatol. 2016;43(3):625–631. doi: 10.3899/jrheum.150388. [DOI] [PubMed] [Google Scholar]
- 38.Harlow BL, Vitonis AF, Sparen P, Cnattingius S, Joffe H, Hultman CM. Incidence of hospitalization for postpartum psychotic and bipolar episodes in women with and without prior prepregnancy or prenatal psychiatric hospitalizations. Arch Gen Psychiatry. 2007;64(1):42–48. doi: 10.1001/archpsyc.64.1.42. [DOI] [PubMed] [Google Scholar]
- 39.Lu MC, Guo HR, Lin MC, Livneh H, Lai NS, Tsai TY. Bidirectional associations between rheumatoid arthritis and depression: a nationwide longitudinal study. Sci Rep. 2016;6:20647. doi: 10.1038/srep20647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lin CY, Li CK, Liu JM, Hsu RJ, Chuang HC, Chang FW. Postpartum depression and subsequent autoimmune diseases in Taiwan. Int J Environ Res Public Health. 2018;15(8):1783. doi: 10.3390/ijerph15081783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Silverman ME, Smith L, Lichtenstein P, Reichenberg A, Sandin S. The association between body mass index and postpartum depression: a population-based study. J Affect Disord. 2018;240:193–198. doi: 10.1016/j.jad.2018.07.063. [DOI] [PubMed] [Google Scholar]
- 42.Muller N, Schwarz MJ. The immune-mediated alteration of serotonin and glutamate: towards an integrated view of depression. Mol Psychiatry. 2007;12(11):988–1000. doi: 10.1038/sj.mp.4002006. [DOI] [PubMed] [Google Scholar]
- 43.Krishnadas R, Nicol A, Sassarini J, Puri N, Burden AD, Leman J, Combet E, Pimlott S, Hadley D, McInnes IB, et al. Circulating tumour necrosis factor is highly correlated with brainstem serotonin transporter availability in humans. Brain Behav Immun. 2016;51:29–38. doi: 10.1016/j.bbi.2015.08.005. [DOI] [PubMed] [Google Scholar]
- 44.Calabrese F, Rossetti AC, Racagni G, Gass P, Riva MA, Molteni R. Brain-derived neurotrophic factor: a bridge between inflammation and neuroplasticity. Front Cell Neurosci. 2014;8:430. doi: 10.3389/fncel.2014.00430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Moulton CD, Pickup JC, Ismail K. The link between depression and diabetes: the search for shared mechanisms. Lancet Diabetes Endocrinol. 2015;3(6):461–471. doi: 10.1016/S2213-8587(15)00134-5. [DOI] [PubMed] [Google Scholar]
- 46.Fasshauer M, Bluher M, Stumvoll M. Adipokines in gestational diabetes. Lancet Diabetes Endo. 2014;2(6):488–499. doi: 10.1016/S2213-8587(13)70176-1. [DOI] [PubMed] [Google Scholar]
- 47.Linauskas A, Overvad K, Johansen MB, Stengaard-Pedersen K, de Thurah A. Positive predictive value of first-time rheumatoid arthritis diagnoses and their serological subtypes in the Danish National Patient Registry. Clin Epidemiol. 2018;10:1709–1720. doi: 10.2147/CLEP.S175406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Paltta J, Heikkilä HK, Pirilä L, Eklund KK, Huhtakangas J, Isomäki P, et al. The validity of rheumatoid arthritis diagnoses in Finnish biobanks. Scand J Rheumatol. 2021;52(1):1–9. doi: 10.1080/03009742.2021.1967047. [DOI] [PubMed] [Google Scholar]
- 49.Molander V, Bower H, Askling J. Validation and characterization of venous thromboembolism diagnoses in the Swedish National Patient Register among patients with rheumatoid arthritis. Scand J Rheumatol. 2022;52(2):1–7. doi: 10.1080/03009742.2021.2001907. [DOI] [PubMed] [Google Scholar]
- 50.Frisell T, Oberg S, Kuja-Halkola R, Sjolander A. Sibling comparison designs: bias from non-shared confounders and measurement error. Epidemiology. 2012;23(5):713–720. doi: 10.1097/EDE.0b013e31825fa230. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Table S1. Description of the registers included in the study. Table S2. The International Classification of Diseases codes used to define medical conditions. Table S3. Incidence proportion and incidence rates of postpartum psychiatric disorders within 90 days after childbirth in the study cohort. Table S4. Characteristics of the study population according to clinical rheumatoid arthritis and preclinical rheumatoid arthritis. Table S5. Hazard ratios and 95% confidence intervals for postpartum psychiatric disorders within 90 days after childbirth according to rheumatoid arthritis before childbirth, after additional adjustment for maternal body-mass index during early pregnancy. Table S6. Hazard ratios and 95% confidence intervals for postpartum psychiatric disorders within 90 days after childbirth according to rheumatoid arthritis before childbirth, after additional adjustment for maternal country origin and family history of psychiatric disorders. Table S7. Hazard ratios and 95% confidence intervals for postpartum psychiatric disorders within 90 days after childbirth according to rheumatoid arthritis before childbirth in a sibling design. Table S8. Hazard ratios and 95% confidence intervals for postpartum psychiatric disorders within 90 days after childbirth according to rheumatoid arthritis before childbirth after adjusting for potential explanatory factors. Table S9. Hazard ratios and 95% confidence intervals for postpartum psychiatric disorders within 90 days after childbirth according to rheumatoid arthritis before childbirth, in analyses restricted to primiparous women. Table S10. Hazard ratios and 95% confidence intervals for postpartum psychiatric disorders within 90 days after childbirth according to rheumatoid arthritis before childbirth in stratified analyses. Table S11. Baseline characteristics of the propensity score matching sub-cohort. Table S12. Hazard ratios and 95% confidence intervals for postpartum psychiatric disorders within 90 days after childbirth according to rheumatoid arthritis before childbirth in the propensity-matched sub-cohort. Table S13. Hazard ratios and 95% confidence intervals for the associations between rheumatoid arthritis before childbirth and postpartum psychiatric disorders within 90 days after childbirth among women with a personal history of psychiatric disorders, further stratified by previous use of psychiatric medication. Fig S1. The time trend of the postpartum psychiatric disorder rates over the study period in the exposed and the unexposed group. Fig S2. Hazard ratios and 95% confidence intervals for postpartum psychiatric disorders within 365 days after childbirth according to clinical rheumatoid arthritis (rheumatoid arthritis diagnosed before childbirth) and preclinical rheumatoid arthritis (rheumatoid arthritis diagnosed after childbirth). Fig S3. Hazard ratios and 95% confidence intervals for postpartum psychiatric disorders within 365 days after childbirth according to clinical rheumatoid arthritis (rheumatoid arthritis diagnosed before childbirth) and preclinical rheumatoid arthritis (rheumatoid arthritis diagnosed in the first year after childbirth). Fig S4. Hazard ratios and 95% confidence intervals for the associations between rheumatoid arthritis before childbirth and postpartum psychiatric disorders within the first 365 days after delivery, stratified by a personal history of psychiatric disorders.
Data Availability Statement
Data used for this study are not openly available because the law and the ethical permit does not allow to share them. The data were analyzed on a secure server at Statistics Denmark run by Aarhus University.