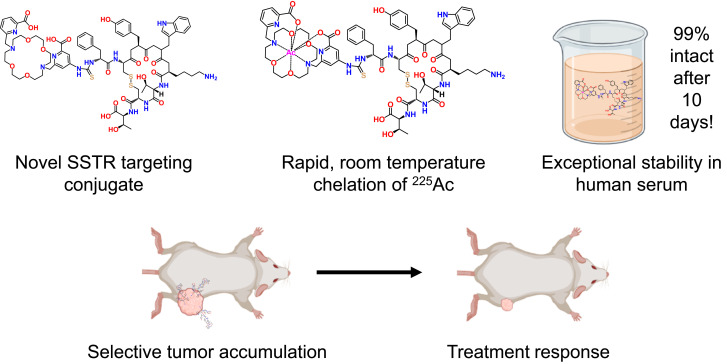
Visual Abstract
Keywords: oncology, actinium, targeted α-therapy, neuroendocrine tumors, octreotate, somatostatin
Abstract
Neuroendocrine tumors (NETs) express somatostatin receptors (SSTRs) 2 and 5. Modified variants of somatostatin, the cognate ligand for SSTR2 and SSTR5, are used in treatment for metastatic and locoregional disease. Peptide receptor radionuclide therapy with 177Lu-DOTATATE (DOTA-octreotate), a β-particle–emitting somatostatin derivative, has demonstrated survival benefit in patients with SSTR-positive NETs. Despite excellent results, a subset of patients has tumors that are resistant to treatment, and alternative agents are needed. Targeted α-particle therapy has been shown to kill tumors that are resistant to targeted β-particle therapy, suggesting that targeted α-particle therapy may offer a promising treatment option for patients with 177Lu-DOTATATE–resistant disease. Although DOTATATE can chelate the clinically relevant α-particle–emitting radionuclide 225Ac, the labeling reaction requires high temperatures, and the resulting radioconjugate has suboptimal stability. Methods: We designed and synthesized MACROPATATE (MACROPA-octreotate), a novel radioconjugate capable of chelating 225Ac at room temperature, and assessed its in vitro and in vivo performance. Results: MACROPATATE demonstrated comparable affinity to DOTATATE (dissociation constant, 21 nM) in U2-OS-SSTR2, a SSTR2-positive transfected cell line. 225Ac-MACROPATATE demonstrated superior serum stability at 37°C over time compared with 225Ac-DOTATATE. Biodistribution studies demonstrated higher tumor uptake of 225Ac-MACROPATATE than of 225Ac-DOTATATE in mice engrafted with subcutaneous H69 NETs. Therapy studies showed that 225Ac-MACROPATATE exhibits significant antitumor and survival benefit compared with saline control in mice engrafted with SSTR-positive tumors. However, the increased accumulation of 225Ac-MACROPATATE in liver and kidneys and subsequent toxicity to these organs decreased its therapeutic index compared with 225Ac-DOTATATE. Conclusion: 225Ac-MACROPATATE and 225Ac-DOTATATE exhibit favorable therapeutic efficacy in animal models. Because of elevated liver and kidney accumulation and lower administered activity for dose-limiting toxicity of 225Ac-MACROPATATE, 225Ac-DOTATATE was deemed the superior agent for targeted α-particle peptide receptor radionuclide therapy.
Neuroendocrine tumors (NETs) are a heterogeneous family of neoplasms originating in cells within the endocrine and nervous systems that reside in the gastrointestinal tract, lungs, pancreas, thyroid, and gonads (1,2). Many NETs overexpress somatostatin receptors (SSTRs) (3). This high receptor expression offers a targetable vulnerability in NETs, which has long been exploited for therapy.
Somatostatin-like derivatives have been used as drugs themselves or as scaffolds to deliver radioisotopes for peptide receptor radionuclide therapy (PRRT). One of the most successful of these is the pairing of Tyr3-octreotate with the chelator DOTA, yielding DOTATATE (4,5). Radiolabeled DOTATATE has been successfully used for both PET imaging (6) (68Ga, 64Cu) and therapeutic (177Lu) purposes. The phase 3 randomized controlled clinical trial NETTER-1 showed that patients with treatment-refractory NETs who received 177Lu-DOTATATE had significantly better progression-free survival than patients receiving somatostatin analogs (7). The results of this trial led to FDA approval of 177Lu-DOTATATE (Lutathera; Advanced Accelerator Applications) in January 2018 for the treatment of SSTR-positive gastroenteropancreatic NETs (8).
Although these results made PRRT a first-in-class treatment option for patients with NETs, many are ab initio resistant to, or develop resistance after treatment with, β-particle–emitting 177Lu-DOTATATE. α-particle–emitting radionuclides are an attractive alternative to β-particle–emitting radionuclides because of their short range, which can mitigate off-target effects, and the high energy deposited by these particles over that short range (also known as high linear energy transfer, or LET) (9,10). The α-particle–emitting nuclide 225Ac has been coupled to prostate-specific membrane antigen (PSMA)–targeting ligands to successfully treat prostate cancers refractory to treatments with androgen deprivation, taxanes, and 177Lu-PSMA-617 (Pluvicto; Advanced Accelerator Applications), which is approved for treatment of patients with PSMA-positive metastatic castration-resistant prostate cancer in the United States (11–14). Recently, a phase I clinical trial of patients with gastroenteropancreatic NETs previously treated with 177Lu-DOTATATE and receiving 225Ac-DOTATATE therapy showed stable disease or a partial response in 82% of patients (15). Similarly, another study with 225Ac-DOTATATE found it to have efficacy in patients with SSTR-positive paraganglioma (16). α-emitting PRRT with 213Bi (half-life, 45 min) and 212Pb (half-life, 10.6 h) have shown promising clinical results as well (17,18).
DOTA and its derivatives are used to chelate 225Ac and many of the previously mentioned radionuclides. However, to chelate 225Ac with DOTA to yield high-specific-activity radioconjugates, temperatures above 70°C are typically required. Even if these temperatures are used, the resulting complex’s thermodynamic stability (19) and labeling kinetics are suboptimal (20). Thiele et al. showed that MACROPA, an 18-membered macrocycle, is capable of chelating 225Ac at room temperature more quickly and at lower concentrations than DOTA (21). The 225Ac-MACROPA complex showed comparable stability (8 d) to 225Ac-DOTA in human serum and in C57BL6 mice. Further preclinical studies have demonstrated the suitability of 225Ac-labeled MACROPA-containing radioconjugates for targeted α-therapy with both small-molecule and antibody conjugates (22,23).
In this work, we synthesized and characterized MACROPATATE, consisting of MACROPA coupled to Tyr3-octreotate, and compared its performance with that of DOTATATE with respect to 225Ac labeling efficiency, serum stability, target engagement, and therapeutic efficacy. We found that MACROPATATE exhibits improved stability over DOTATATE when complexed to 225Ac, maintains high SSTR binding affinity, demonstrates favorable in vivo target localization, and has significant antitumor activity.
MATERIALS AND METHODS
Synthesis and Radiolabeling of MACROPATATE and DOTATATE
MACROPATATE and DOTATATE were prepared by conjugating isothiocyanate-activated MACROPA and DO3A-tri-tert-butyl ester, respectively, to immobilized octreotate (21,24,25). After synthesis and deprotection, the products were characterized for purity and identity by high-performance liquid chromatography and liquid chromatography–mass spectrometry, respectively. Full synthetic details for both conjugates are reported in Supplemental Figure 1 (supplemental materials are available at http://jnm.snmjournals.org). Radiolabeling of MACROPATATE and DOTATATE with 225Ac was performed at room temperature or 70°C, respectively, in NH4OAc (pH 5.5), and the products were characterized using instant thin-layer chromatography (ITLC). Full radiolabeling and characterization details are provided in the supplemental information.
Cell Culture and In Vitro Assays
A panel of SSTR2- and SSTR5-expressing cell lines were cultured, and their SSTR2 and SSTR5 expression levels were evaluated using flow cytometry. The highly positive U2OS-SSTR2 cell line was used to confirm the binding affinity of radiolabeled 225Ac-MACROPATATE in a saturation binding assay. Full cell culture details and experimental procedures for flow cytometry and saturation assays are reported in the supplemental information.
Serum Stability Studies
225Ac-MACROPATATE and 225Ac-DOTATATE were evaluated for stability in human serum (EMD Millipore) at 37°C and pH 7.4. Radiochelate was diluted to 370 kBq in 1 mL of human serum and placed on an Eppendorf ThermoMixer set to 37°C. At fixed intervals, aliquots were removed from the reactions and analyzed by ITLC as described in the supplemental information.
Murine Subcutaneous Xenograft Models
All procedures and animal studies followed a protocol approved by the National Institutes of Health Institutional Animal Care and Use Committee (protocol ROB104). Female athymic homozygous nude mice (NCI Athymic NCr-nu/nu strain 553; Charles River Laboratories), 8–10 wk old, were subcutaneously engrafted with 8 × 106 H69 cells in 200 μL of ice-cold phosphate-buffered saline. Treatment of tumors with radiotracers for biodistribution or therapy studies was performed once palpable tumors developed, approximately 1 mo after inoculation. The biodistributions of both 225Ac-MACROPATATE and 225Ac-DOTATATE were evaluated in H69 subcutaneous tumor models, both with and without d-lysine pretreatment (26). Full experimental details for the biodistribution experiments are reported in the supplemental information.
Dose-Finding Study for 225Ac-MACROPATATE
To evaluate the therapeutic potential of 225Ac-MACROPATATE, we performed a dose-finding study on mice. Mice (n ≥ 3) bearing H69 tumor xenografts were first injected with d-lysine hydrochloride (35 mg/mouse) and then treated with 148, 93.3, 46.3, or 23.1 kBq of 225Ac-MACROPATATE; their body weights and tumor growth were monitored over several weeks. The highest tested dose of 225Ac-MACROPATATE was based on a recent report of therapy using 225Ac-DOTATATE, which found that 148 kBq was well tolerated in mice (27).
Head-to-Head Therapy Study with 225Ac-MACROPATATE or 225Ac-DOTATATE
For our therapy study, we wished to identify the highest administered activities that exhibited acceptable toxicity as measured by mouse weight loss and survival, and we found 46.3 kBq of 225Ac-MACROPATATE and 148 kBq of 225Ac-DOTATATE to be suitable. Animals engrafted with H69 cells were treated with either of the 2 radioconjugates or saline (n = 8–10, each). Local control and survival were the primary outcomes. All groups were tracked for humane endpoints including, but not limited to, tumors larger than 2,000 mm3 and weight loss greater than 20%.
Statistical Analysis
Statistical analysis was performed using Prism (version 9.0; GraphPad Software). Statistical analysis of survival curves was performed using the log-rank test. Comparisons of organ uptake, tumor volume, and stability were performed using the Student t test.
RESULTS
MACROPATATE Forms Stable Complex with 225Ac at Room Temperature
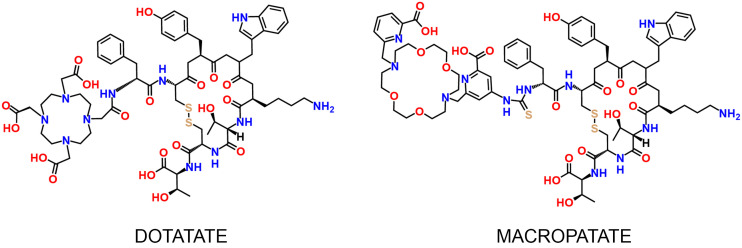
We successfully synthesized DOTATATE and MACROPATATE with good yields and high purity, characterizing the identity and purity of both molecules via high-performance liquid chromatography and mass spectrometry (Fig. 1; Supplemental Fig. 1). After synthesis, we labeled both DOTATATE and MACROPATATE with 225Ac. Radiolabeling was conducted in mildly acidic (pH 5.5) NH4OAc buffer (0.1 M). We found that MACROPATATE could be radiolabeled quantitatively after 1 h of incubation at room temperature (18–20°C), whereas DOTATATE required heating at 70°C for 1 h to achieve comparable purity and yield. Typical specific activities for both radioconjugates were approximately 185 GBq/mmol. A representative instant thin-layer chromatogram of 225Ac-MACROPATATE is shown in Figure 2A and Supplemental Figure 2, and an instant thin-layer chromatogram of 225Ac-DOTATATE is shown in Supplemental Figure 3.
FIGURE 1.
Structures of DOTATATE and MACROPATATE.
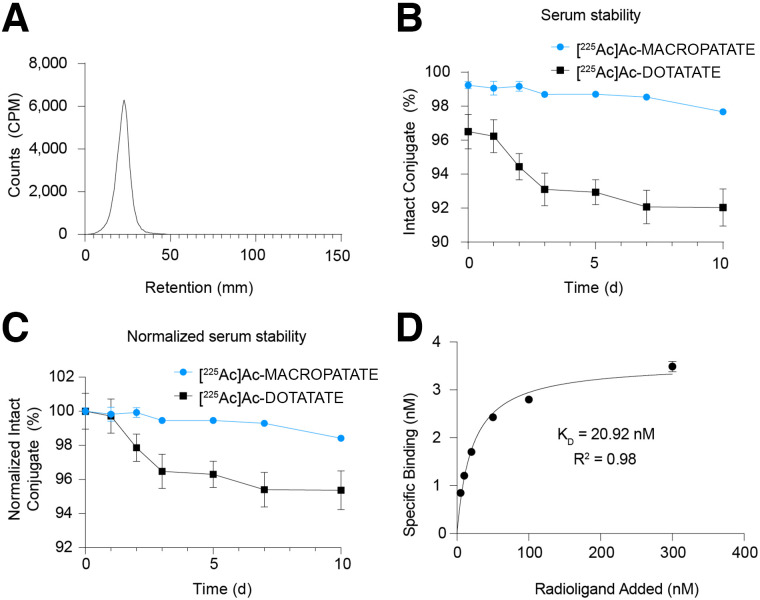
FIGURE 2.
MACROPATATE stably chelates 225Ac and binds to SSTR. (A) Representative ITLC chromatogram of 225Ac-MACROPATATE. (B) Intact conjugate remaining over time of 225Ac-MACROPATATE and 225Ac-DOTATATE in human serum incubated at 37°C, as measured by ITLC. (C) Percentage of initial intact conjugate remaining over time, normalized to starting amount, after incubation in human serum at 37°C. (D) Assessment of SSTR2 binding affinity of 225Ac-MACROPATATE in U2-OS SSTR2 cells using saturation binding assay.
We evaluated the stability of both molecules in human serum at 37°C using ITLC (Figs. 2B and 2C; Supplemental Figs. 4–7). 225Ac-MACROPATATE and 225Ac-DOTATATE were stable to 10 d. Stability experiments conducted in human serum indicated that 225Ac-MACROPATATE has significantly greater stability than 225Ac-DOTATATE (98% vs. 95%, P = 0.0097), and our results compared well with a recent stability investigation of 225Ac-DOTATATE reported by others, which found 90% intact 225Ac-DOTATATE after 10 d (27). Thus, 225Ac-MACROPATATE exhibited a modest yet significant stability advantage over 225Ac-DOTATATE.
225Ac-MACROPATATE Retains Affinity for SSTR
After evaluating the purity and stability of the 225Ac-MACROPATATE conjugate, we sought to confirm its binding affinity using SSTR2-expressing cells in vitro (28–30). Saturation binding assays showed a binding affinity of 21 nM (Fig. 2D), comparable to that reported for Eu-DOTATATE (22 nM), which has been used as a surrogate for 225Ac-labeled DOTATATE (27). This value is also in a similar range to other studies examining radiopeptide somatostatin derivatives (31,32). These results confirmed that MACROPA conjugation to octreotate does not adversely affect SSTR binding.
Cell Lines Were Selected for In Vivo Studies
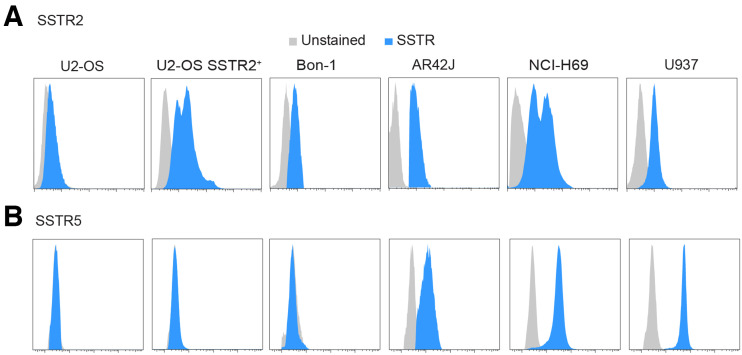
To find a suitable model for our murine subcutaneous xenograft models, we assessed SSTR2 and SSTR5 expression by flow cytometry in several cell lines (U2-OS, U2-OS-SSTR2, AR42J, H69, Bon-1, and U937) (Fig. 3). US-OS exhibited low to negligible levels of both SSTR2 and SSTR5. All other cell lines were SSTR2-positive, with expression decreasing in the order U2OS-SSTR2 > H69 > AR42J > U937 > Bon-1. The cell lines H69, AR42J, and U937 also displayed moderate expression of SSTR5. The H69 cell line was chosen for in vivo experiments because of its high expression of SSTR2 and SSTR5 and its established history as a model system for investigating SSTR-targeting radioconjugates (33,34).
FIGURE 3.
Flow cytometry assessment of SSTR2 (A) and SSTR5 (B) in panel of SSTR-expressing cell lines.
225Ac-MACROPATATE Demonstrates Target Engagement In Vivo
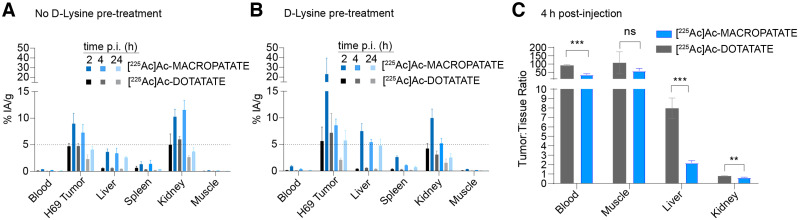
After confirming the stability and SSTR binding of 225Ac-MACROPATATE in vitro, we evaluated its biodistribution and compared it with that of 225Ac-DOTATATE in mice bearing SSTR-positive H69 tumor xenografts. Both tracers showed favorable tumor uptake, with percentage injected activity per gram of tissue of 9% and 5% at 2 h and 4% and 2% at 24 h for 225Ac-MACROPATATE and 225Ac-DOTATATE, respectively (Figs. 4A and 4B; Supplemental Figs. 8–11). Although both tracers displayed excellent tumor-to-muscle ratios of more than 50:1 at 4 h (Fig. 4C), they also had high renal uptake, as is typical of SSTR-targeting peptides (35,36). 225Ac-MACROPATATE displayed significantly higher tumor accumulation at 2 and 24 h than did 225Ac-DOTATATE (P < 0.05). However, the liver and kidney uptake of 225Ac-MACROPATATE were also higher, possibly because the higher hydrophobicity of the conjugate could slow clearance, leading to increased liver uptake. The liver accumulation of 225Ac-MACROPATATE was 2–3 times higher than that of 225Ac-DOTATATE at all time points investigated.
FIGURE 4.
225Ac-labeled MACROPATATE and DOTATATE bind to SSTR-positive tumors. (A and B) Selected-organ biodistribution (n = 3) of 225Ac-MACROPATATE and 225Ac-DOTATATE (37 kBq) without (A) or with (B) preadministration of d-lysine (35 mg/mouse). (C) Corresponding tumor-to-tissue ratios for selected organs with lysine pretreatment. Full 12-organ biodistribution data are reported in Supplemental Figures 8–11. Error bars represent SD. ***P < 0.005. *P < 0.01. %IA/g = percentage injected activity per gram of tissue.
The high kidney accumulation of peptide radioconjugates is routinely lowered by preadministration of d-lysine (26). Accordingly, we observed a lower kidney percentage injected activity per gram of tissue for both 225Ac-MACROPATATE and 225Ac-DOTATATE after d-lysine administration than in kidneys of animals not receiving d-lysine. This lowered kidney accumulation of the radiotracers was most pronounced at 4 h after injection, with the kidney signal for 225Ac-MACROPATATE changing from 11.5% to 5.2% (P = 0.0057) and 225Ac-DOTATATE decreasing from 6.0% to 3.1% (P = 0.0039).
225Ac-MACROPATATE and 225Ac-DOTATATE Delay Tumor Growth and Improve Survival of Mice Bearing NET Xenografts
The promising biodistribution profile of 225Ac-MACROPATATE led us to investigate its therapeutic efficacy. As a preliminary investigation, we evaluated a series of treatment activities of 225Ac-MACROPATATE ranging from 23.1 to 148 kBq in mice bearing H69 tumor xenografts. All mice in the 148-kBq treatment group (10/10) and 1 of 3 mice in the 92.3-kBq group were euthanized within 10 d of treatment because of substantial weight loss (>20%). All other mice displayed minimal weight loss, and a clear dose-dependent reduction in tumor volume was evident (Supplemental Figs. 12–14). On the basis of these results, 46.3 kBq of 225Ac-MACROPATATE was selected as the appropriate dose for further investigation.
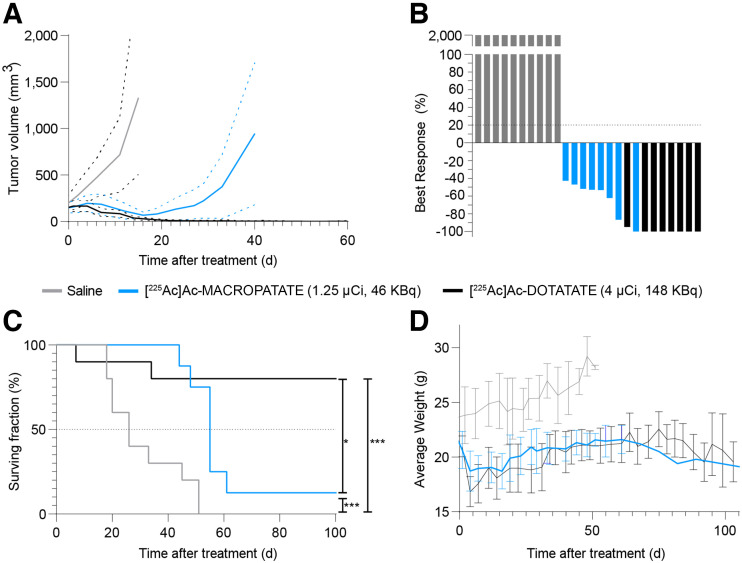
Animals treated with 225Ac-MACROPATATE and 225Ac-DOTATATE demonstrated a significant tumor growth delay and improvements in survival compared with saline-treated controls (Figs. 5A–5C; Supplemental Figs. 15–17). Mice treated with 225Ac-MACROPATATE exhibited an initial reduction in tumor volume lasting approximately 3 wk after treatment. However, the tumors subsequently relapsed in most mice (7 of 8). Conversely, 225Ac-DOTATATE treatment resulted in complete, durable tumor remission for all mice. However, 2 mice in the 225Ac-DOTATATE treatment group were euthanized because of weight loss. Although mice in the MACROPATATE treatment group also displayed some weight loss immediately after treatment, their weights stabilized within 2 wk (Fig. 5D). 225Ac-MACROPATATE significantly improved median survival relative to the vehicle control (55 d vs. 26 d; log rank, P = 0.0006), whereas 8 of 10 mice (80%) treated with 225Ac-DOTATATE survived the full 100-d duration of the study. Overall, 225Ac-MACROPATATE exhibited favorable local control and a survival benefit over saline-treated animals. However, mice treated with 225Ac-DOTATATE showed significantly better local control and overall survival (P < 0.02).
FIGURE 5.
Therapeutic response of mice bearing H69 lung NET xenografts treated with 225Ac-MACROPATATE (46 kBq), 225Ac-DOTATATE (148 kBq), or vehicle control shows that targeted α-therapy with 225Ac-MACROPATATE and 225Ac-DOTATATE is effective. (A) Tumor volume measurements over time. Solid lines represent average volume, and dashed lines represent 95% CI. Plots for each dataset are discontinued after first mouse death due to excessive tumor volume. Full tumor volume measurements for study duration for each mouse are reported in Supplemental Figures 15–17. (B) Maximal response to treatment (tumor volume growth percentage) of individual mice. Nonresponding mice are represented as full tumor growth (2,000% increase). (C) Mouse survival over time. Endpoint was defined as tumor volume > 2,000 mm3 or weight loss over 20% of starting weight. (D) Average mouse body weights over time for each treatment group. Error bars represent SD. ***P < 0.005. *P < 0.05.
DISCUSSION
PRRT with 177Lu-DOTATATE represents a significant advance for patients with SSTR-expressing NETs. Nevertheless, treatment options for tumors refractory to 177Lu-based PRRT are needed. By exploiting the unique properties of α-particles, namely high energy deposition over a short path, we may be able to overcome resistance to 177Lu-based PRRT (16,27,37,38). However, the DOTA chelator used in these and other studies is suboptimal for chelation of the large Ac3+ ion (19). With the goal of achieving a more stable SSTR-targeting radioconjugate for 225Ac targeted α-particle therapy, we designed, synthesized, and characterized the conjugate MACROPATATE, wherein we replace the DOTA of DOTATATE with the expanded macrocyclic chelator MACROPA, which has been shown to more stably chelate 225Ac3+ than DOTA can (21).
We confirmed that 225Ac-MACROPATATE displayed a high tumor accumulation, tumor growth delay, and survival benefit in xenograft models of NETs. However, our radioconjugate also exhibited a narrow therapeutic index as evinced by toxicity at lower injected activities than is the case with 225Ac-DOTATATE. As such, the head-to-head therapy study was performed with a 3-fold higher injected activity in animals receiving 225Ac-DOTATATE than in those receiving 225Ac-MACROPATATE. Biodistribution studies indicated a relatively high liver accumulation for 225Ac-MACROPATATE. Notably, unlike most other organs, this liver signal did not appear to diminish over time. This persistent accumulation of 225Ac may arise from metabolism of the radioconjugate and may be responsible for the observed higher toxicity of 225Ac-MACROPATATE. Previous investigations of 177Lu-DOTATATE have indicated significant degradation of the targeting octreotate portion of the tracer, likely because of metabolism (39). Such metabolism may significantly impact the biodistribution of the 225Ac radioconjugates explored in this work. Thus, the disparate off-target uptake of 225Ac-MACROPATATE and 225Ac-DOTATATE despite their similar tumor accumulation may also reflect accumulation of fragmented species in nontarget organs. Although 225Ac-MACROPATATE demonstrates significant antitumor activity in SSTR-expressing models of NET, 225Ac-MACROPATATE remains inferior to 225Ac-DOTATATE in vivo. Therefore, 225Ac-MACROPATATE requires significant optimization to decrease off-target accumulation and associated toxicity. For instance, variation of the specific activity or molar amount of 225Ac-MACROPATATE injected may provide decreased background accumulation while preserving tumor uptake, for such optimization has been shown to greatly improve the pharmacokinetic profile of 177Lu-DOTATATE (40).
Targeted α-particle therapy agents directed at NETs, such as 225Ac-DOTATATE, have demonstrated promising results in small clinical studies and warrant further investigation. Other α-particle–emitting radionuclides being investigated include 213Bi and 212Pb. Recently, a phase I study with the α-particle–emitting PRRT agent 212Pb-DOTAMTATE, which has a lead-optimized chelator, has shown good tolerability and overall response rates of 80% in patients naïve to PRRT (18). The phase II study (NCT05153772) is open and recruiting. These studies indicate that the intrinsic properties of α-emitters can elicit responses in tumors otherwise refractory to β-emitting PRRT (41,42).
More broadly, several strategies aiming to improve the efficacy of 177Lu-PRRT in NETs are being investigated and may apply to targeted α-particle therapy PRRT as well. For instance, deploying epigenetic modulators has been shown to increase the membrane expression of SSTR and subsequent accumulation of PRRT agent (43–46). Combinatorial approaches exploiting inhibitors of DNA damage repair are also being explored (NCT04086485, NCT04375267, and NCT03958045).
CONCLUSION
We have successfully synthesized MACROPATATE, a novel SSTR2- and SSTR5-targeting PRRT agent tailored to deliver 225Ac to NETs. Importantly, we showed that MACROPATATE is able to chelate 225Ac at room temperature and that this complex has 4-fold lower susceptibility to degradation than 225Ac-DOTATATE in human serum. Both 225Ac-MACROPATATE and 225Ac-DOTATATE demonstrated excellent in vivo target engagement in NET xenografts and exhibited local control and survival superior to saline. However, although 225Ac-MACROPATATE had in vitro stability superior to 225Ac-DOTATATE, it underperformed compared with 225Ac-DOTATATE in vivo. Optimization is therefore needed before further translation.
DISCLOSURE
This work was supported in part by the National Institutes of Biomedical Imaging and Bioengineering of the National Institutes of Health (awards R21EB027282 and R01EB029259), as well as by the National Cancer Institute from Intramural Research Program funds ZIA BC 011800 and ZIA BC 010891. This project was funded in whole or in part by the National Cancer Institute, National Institutes of Health, under contract 75N91019D00024. The content of this publication does not necessarily reflect the views or policies of the Department of Health and Human Services, nor does mention of trade names, commercial products, or organizations imply endorsement by the U.S. government. Justin Wilson and Nikki Thiele are authors of a patent for the use of MACROPA as a chelator for 225Ac chelation. No other potential conflict of interest relevant to this article was reported.
ACKNOWLEDGMENTS
We thank Dr. Julie Nonnekens for providing the U2OS-SSTR2 cell line and Dr. Mark Hellmitch for providing the Bon-1 cell line. The graphical abstract figure was created using BioRender.com.
KEY POINTS
QUESTION: Can chemical conjugation of the MACROPA chelator to SSTR-targeting octreotate result in an SSTR-targeting radioconjugate for targeted α-therapy superior to DOTATATE?
PERTINENT FINDINGS: 225Ac-MACROPATATE demonstrated in vitro SSTR2 affinity comparable to that of the standard 225Ac-DOTATATE, as well as higher in vivo uptake in SSTR-positive xenografts. However, its efficacy is limited by a poor therapeutic index, highlighting a need for further optimization before translation.
IMPLICATIONS FOR PATIENT CARE: We confirm that targeted α-therapies for NETs demonstrate high efficacy in preclinical studies. Such agents warrant further clinical investigation to offer a therapeutic option for patients with disease refractory to β-particle PRRT.
REFERENCES
- 1. Oronsky B, Ma PC, Morgensztern D, Carter CA. Nothing but NET: a review of neuroendocrine tumors and carcinomas. Neoplasia. 2017;19:991–1002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Chauhan A, Yu Q, Ray N, et al. Global burden of neuroendocrine tumors and changing incidence in Kentucky. Oncotarget. 2018;9:19245–19254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Alonso-Gordoa T, Capdevila J, Grande E. GEP-NETs update: biotherapy for neuroendocrine tumours. Eur J Endocrinol. 2015;172:R31–R46. [DOI] [PubMed] [Google Scholar]
- 4. Pauwels E, Cleeren F, Bormans G, Deroose CM. Somatostatin receptor PET ligands: the next generation for clinical practice. Am J Nucl Med Mol Imaging. 2018;8:311–331. [PMC free article] [PubMed] [Google Scholar]
- 5. Reubi JC, Schar JC, Waser B, et al. Affinity profiles for human somatostatin receptor subtypes SST1-SST5 of somatostatin radiotracers selected for scintigraphic and radiotherapeutic use. Eur J Nucl Med. 2000;27:273–282. [DOI] [PubMed] [Google Scholar]
- 6. Wild D, Bomanji JB, Benkert P, et al. Comparison of 68Ga-DOTANOC and 68Ga-DOTATATE PET/CT within patients with gastroenteropancreatic neuroendocrine tumors. J Nucl Med. 2013;54:364–372. [DOI] [PubMed] [Google Scholar]
- 7. Strosberg J, El-Haddad G, Wolin E, et al. Phase 3 trial of 177Lu-Dotatate for midgut neuroendocrine tumors. N Engl J Med. 2017;376:125–135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Mittra ES. Neuroendocrine tumor therapy: 177Lu-DOTATATE. AJR. 2018;211:278–285. [DOI] [PubMed] [Google Scholar]
- 9. Wild D, Frischknecht M, Zhang H, et al. Alpha- versus beta-particle radiopeptide therapy in a human prostate cancer model (213Bi-DOTA-PESIN and 213Bi-AMBA versus 177Lu-DOTA-PESIN). Cancer Res. 2011;71:1009–1018. [DOI] [PubMed] [Google Scholar]
- 10. King AP, Lin FI, Escorcia FE. Why bother with alpha particles? Eur J Nucl Med Mol Imaging. 2021;49:7–17. [DOI] [PubMed] [Google Scholar]
- 11. Kojima S, Cuttler JM, Shimura N, Koga H, Murata A, Kawashima A. Present and future prospects of radiation therapy using alpha-emitting nuclides. Dose Response. 2018;16:1559325817747387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Kratochwil C, Bruchertseifer F, Rathke H, et al. Targeted alpha-therapy of metastatic castration-resistant prostate cancer with 225Ac-PSMA-617: swimmer-plot analysis suggests efficacy regarding duration of tumor control. J Nucl Med. 2018;59:795–802. [DOI] [PubMed] [Google Scholar]
- 13. Sathekge M, Bruchertseifer F, Knoesen O, et al. 225Ac-PSMA-617 in chemotherapy-naive patients with advanced prostate cancer: a pilot study. Eur J Nucl Med Mol Imaging. 2019;46:129–138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Sartor O, de Bono J, Chi KN, et al. Lutetium-177-PSMA-617 for metastatic castration-resistant prostate cancer. N Engl J Med. 2021;385:1091–1103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Ballal S, Yadav MP, Bal C, Sahoo RK, Tripathi M. Broadening horizons with 225Ac-DOTATATE targeted alpha therapy for gastroenteropancreatic neuroendocrine tumour patients stable or refractory to 177Lu-DOTATATE PRRT: first clinical experience on the efficacy and safety. Eur J Nucl Med Mol Imaging. 2020;47:934–946. [DOI] [PubMed] [Google Scholar]
- 16. Yadav MP, Ballal S, Sahoo RK, Bal C. Efficacy and safety of 225Ac-DOTATATE targeted alpha therapy in metastatic paragangliomas: a pilot study. Eur J Nucl Med Mol Imaging. 2022;49:1595–1606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Kratochwil C, Giesel FL, Bruchertseifer F, et al. 213Bi-DOTATOC receptor-targeted alpha-radionuclide therapy induces remission in neuroendocrine tumours refractory to beta radiation: a first-in-human experience. Eur J Nucl Med Mol Imaging. 2014;41:2106–2119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Delpassand ES, Tworowska I, Esfandiari R, et al. Targeted alpha-emitter therapy with 212Pb-DOTAMTATE for the treatment of metastatic SSTR-expressing neuroendocrine tumors: first-in-human, dose-escalation clinical trial. J Nucl Med. 2022;63:1326–1333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Deal KA, Davis IA, Mirzadeh S, Kennel SJ, Brechbiel MW. Improved in vivo stability of actinium-225 macrocyclic complexes. J Med Chem. 1999;42:2988–2992. [DOI] [PubMed] [Google Scholar]
- 20. Yang H, Wilson JJ, Orvig C, et al. Harnessing alpha-emitting radionuclides for therapy: radiolabeling method review. J Nucl Med. 2022;63:5–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Thiele NA, Brown V, Kelly JM, et al. An eighteen-membered macrocyclic ligand for actinium-225 targeted alpha therapy. Angew Chem Int Ed Engl. 2017;56:14712–14717. [DOI] [PubMed] [Google Scholar]
- 22. Kelly JM, Amor-Coarasa A, Ponnala S, et al. A single dose of 225Ac-RPS-074 induces a complete tumor response in an LNCaP xenograft model. J Nucl Med. 2019;60:649–655. [DOI] [PubMed] [Google Scholar]
- 23. Bell MM, Gutsche NT, King AP, et al. Glypican-3-targeted alpha particle therapy for hepatocellular carcinoma. Molecules. 2020;26:4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Axelsson O, Olsson A, inventors; GE Healthcare AS, assignee. Synthesis of cyclen derivatives. U.S. patent 8,138,332 (B2). March 20, 2012.
- 25. Haug AR, Auernhammer CJ, Wangler B, et al. 68Ga-DOTATATE PET/CT for the early prediction of response to somatostatin receptor-mediated radionuclide therapy in patients with well-differentiated neuroendocrine tumors. J Nucl Med. 2010;51:1349–1356. [DOI] [PubMed] [Google Scholar]
- 26. Behr TM, Sharkey RM, Sgouros G, et al. Overcoming the nephrotoxicity of radiometal-labeled immunoconjugates: improved cancer therapy administered to a nude mouse model in relation to the internal radiation dosimetry. Cancer. 1997;80:2591–2610. [DOI] [PubMed] [Google Scholar]
- 27. Tafreshi NK, Pandya DN, Tichacek CJ, et al. Preclinical evaluation of [225Ac]Ac-DOTA-TATE for treatment of lung neuroendocrine neoplasms. Eur J Nucl Med Mol Imaging. 2021;48:3408–3421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Lin M, Welch MJ, Lapi SE. Effects of chelator modifications on 68Ga-labeled [Tyr3]octreotide conjugates. Mol Imaging Biol. 2013;15:606–613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Fani M, Del Pozzo L, Abiraj K, et al. PET of somatostatin receptor-positive tumors using 64Cu- and 68Ga-somatostatin antagonists: the chelate makes the difference. J Nucl Med. 2011;52:1110–1118. [DOI] [PubMed] [Google Scholar]
- 30. Fani M, Braun F, Waser B, et al. Unexpected sensitivity of sst2 antagonists to N-terminal radiometal modifications. J Nucl Med. 2012;53:1481–1489. [DOI] [PubMed] [Google Scholar]
- 31. Stallons TAR, Saidi A, Tworowska I, Delpassand ES, Torgue JJ. Preclinical investigation of 212Pb-DOTAMTATE for peptide receptor radionuclide therapy in a neuroendocrine tumor model. Mol Cancer Ther. 2019;18:1012–1021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Ullrich M, Bergmann R, Peitzsch M, et al. Multimodal somatostatin receptor theranostics using [64Cu]Cu-/[177Lu]Lu-DOTA-(Tyr3)octreotate and AN-238 in a mouse pheochromocytoma model. Theranostics. 2016;6:650–665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Bogden AE, Taylor JE, Moreau JP, Coy DH, LePage DJ. Response of human lung tumor xenografts to treatment with a somatostatin analogue (Somatuline). Cancer Res. 1990;50:4360–4365. [PubMed] [Google Scholar]
- 34. Isobe T, Onn A, Morgensztern D, et al. Evaluation of novel orthotopic nude mouse models for human small-cell lung cancer. J Thorac Oncol. 2013;8:140–146. [DOI] [PubMed] [Google Scholar]
- 35. Geenen L, Nonnekens J, Konijnenberg M, Baatout S, De Jong M, Aerts A. Overcoming nephrotoxicity in peptide receptor radionuclide therapy using [177Lu]Lu-DOTA-TATE for the treatment of neuroendocrine tumours. Nucl Med Biol. 2021;102–103:1–11. [DOI] [PubMed] [Google Scholar]
- 36. Vegt E, de Jong M, Wetzels JF, et al. Renal toxicity of radiolabeled peptides and antibody fragments: mechanisms, impact on radionuclide therapy, and strategies for prevention. J Nucl Med. 2010;51:1049–1058. [DOI] [PubMed] [Google Scholar]
- 37. Bal C, Yadav M, Ballal S, Tripathi M. Safety and therapeutic efficacy of 225Ac-DOTATATE targeted alpha therapy in metastatic gastroenteropancreatic neuroendocrine tumors stable or refractory to 177Lu-DOTATATE PRRT [abstract]. J Nucl Med. 2020;61(suppl 1):416. [DOI] [PubMed] [Google Scholar]
- 38. Miederer M, Henriksen G, Alke A, et al. Preclinical evaluation of the alpha-particle generator nuclide 225Ac for somatostatin receptor radiotherapy of neuroendocrine tumors. Clin Cancer Res. 2008;14:3555–3561. [DOI] [PubMed] [Google Scholar]
- 39. Lubberink M, Wilking H, Ost A, et al. In vivo instability of 177Lu-DOTATATE during peptide receptor radionuclide therapy. J Nucl Med. 2020;61:1337–1340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Nicolas GP, Mansi R, McDougall L, et al. Biodistribution, pharmacokinetics, and dosimetry of 177Lu-, 90Y-, and 111In-labeled somatostatin receptor antagonist OPS201 in comparison to the agonist 177Lu-DOTATATE: the mass effect. J Nucl Med. 2017;58:1435–1441. [DOI] [PubMed] [Google Scholar]
- 41. Yadav MP, Ballal S, Sahoo RK, Tripathi M, Seth A, Bal C. Efficacy and safety of 225Ac-PSMA-617 targeted alpha therapy in metastatic castration-resistant prostate cancer patients. Theranostics. 2020;10:9364–9377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Kratochwil C, Bruchertseifer F, Giesel FL, et al. 225Ac-PSMA-617 for PSMA-targeted α-radiation therapy of metastatic castration-resistant prostate cancer. J Nucl Med. 2016;57:1941–1944. [DOI] [PubMed] [Google Scholar]
- 43. Sharma R, Earla B, Baidoo K, et al. Upregulation of somatostatin receptor type 2 in a receptor-deficient in vivo pancreatic neuroendocrine tumor model improves tumor response to targeted 177Lu-DOTATATE. bioRxiv website. https://www.biorxiv.org/content/10.1101/2022.04.25.489401v1. Published April 26, 2022. Accessed December 28, 2022.
- 44. Taelman VF, Radojewski P, Marincek N, et al. Upregulation of key molecules for targeted imaging and therapy. J Nucl Med. 2016;57:1805–1810. [DOI] [PubMed] [Google Scholar]
- 45. Guenter R, Aweda T, Carmona Matos DM, et al. Overexpression of somatostatin receptor type 2 in neuroendocrine tumors for improved Ga68-DOTATATE imaging and treatment. Surgery. 2020;167:189–196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Shah RG, Merlin MA, Adant S, Zine-Eddine F, Beauregard JM, Shah GM. Chemotherapy-induced upregulation of somatostatin receptor-2 increases the uptake and efficacy of 177Lu-DOTA-octreotate in neuroendocrine tumor cells. Cancers (Basel). 2021;13:232. [DOI] [PMC free article] [PubMed] [Google Scholar]