Abstract
Background and study aims Pancreatic necrosis is an independent predictor of morbidity and mortality among patients with acute pancreatitis. We compared the safety and outcomes of three techniques including endoscopic necrosectomy, fluoroscopy-guided percutaneous necrosectomy by an interventional radiologist, and surgical necrosectomy.
Patients and methods Using the Nationwide Readmissions Database, we identified hospitalized patients who underwent pancreatic necrosectomy from 2016 to 2019. They were identified using the International Classification of Diseases, 10th Revision, Procedure Coding System.
Results: Of the 2,281 patients meeting the selection criteria, the method of pancreatic necrosectomy was as follows: endoscopy (n = 672), percutaneous (n = 1,338), and surgery (n = 271). Compared to surgery, the rate of mortality was lowest for endoscopy (hazard ratio (HR) 0.27; 95 % CI 0.08–0.90; P = 0.033) followed by percutaneous (HR 0.44; 95 % CI, 0.20–0.98; P = 0.045). Endoscopy was associated with less post-procedure bleeding compared to percutaneous and surgical necrosectomy ( P < 0.001), as well as lower rates of post-procedure renal failure ( P < 0.001) and respiratory failure ( P = 0.002). Endoscopy was associated with average shorter lengths of stay and total hospital costs when compared with percutaneous and surgical approaches, respectively (20.1 vs 25.8 vs 38.3 days; P < 0.001) and ($ 57K vs $ 76K vs $ 123K; P < 0.001).
Conclusions Endoscopic necrosectomy is associated with significantly lower risk of inpatient mortality, adverse events, length of stay, and cost when compared to percutaneous and surgical approaches.
Introduction
In the United States, acute pancreatitis is the third most common hospital-related gastrointestinal disorder. Acute pancreatitis is a considerable health care burden with an estimated annual cost of $ 2.2 billion 1 2 . Necrotizing disease develops in approximately 15 % to 20 % of patients with pancreatitis and carries a mortality rate as high as 39 % 3 4 . The 2020 American Gastroenterological Association (AGA) guidelines recommend drainage and/or debridement of pancreatic necrosis with symptoms or associated complications such as obstruction, recurrent pancreatitis, or fistula formation 5 .
Traditionally, open necrosectomy was the gold standard approach in managing necrotizing pancreatitis 6 . However, earlier surgical debridement techniques were associated with significant morbidity and mortality 7 8 9 10 11 . The current standard of care involves a minimally invasive “step-up” approach involving endoscopic methods of drainage and/or debridement and has largely made direct surgical necrosectomy a thing of the past 5 12 13 14 . Furthermore, current evidence suggest that endoscopy may offer long-term advantages over the percutaneous approach 15 . This paradigm shift is supported by the literature with studies reporting similar or improved mortality and reduced complication rates including but not limited to organ failure, pancreatic fistula, incisional hernia, and pancreatic insufficiency with endoscopy 16 17 18 19 .
Nevertheless, nationwide studies evaluating the effect of surgical, percutaneous, and endoscopic intervention on clinical outcomes is not well described in the literature. Thus, we aimed to examine the effect of each of these interventions on 30-day readmission rates, inpatient mortality, and healthcare utilization using a nationwide inpatient database.
Patients and methods
Data source
A national retrospective cohort study was conducted using the Healthcare Cost and Utilization Project (HCUP) Nationwide Readmissions Database (NRD) from 2016 to 2019. The NRD is a database of inpatient admissions and readmissions representing about 60 % of all-payer hospitalizations and the United States population 20 . International Classification of Diseases, Tenth Revision (ICD-10) codes were used to identify diagnoses and procedures. Patients were included in the study if they were at least 18 years of age with a non-elective admission diagnosis of pancreatic necrosis (ICD-10-CM codes K85.9, K85.01, K85.02, K85.11, K85.12, K85.21, K85.22, K85.31, K85.32, K85.81, K85.82) and further stratified according to endoscopic (ICD-10-CM code 0F9G8), percutaneous (by an interventional radiologist) (ICD-10-CM code 0F9G3), and surgical necrosectomy (ICD-10-CM codes 0F9G0) from January to November; patients were excluded if admitted in December to track 30-day readmission rates. Readmissions were also excluded if related to a traumatic injury.
Characteristics
Baseline patient characteristics included demographical variables (age and gender), length of hospital stay for index admission, elective admission, day of admission, hospital teaching status, number of beds, type of insurance (Medicare, Medicaid, private, self-pay, other), rural-urban district code, household income, discharge outcome (death or survival), and 30-day readmission status. We calculated the Elixhauser comorbidity index to adjust for multiple comorbidities, which accounts for 29 clinical variables 21 . Elixhauser comorbidity software by Quan et al. is a user-available program utilized to classify comorbidities 22 23 .
Outcomes
Outcomes included readmission rates, length of stay, cost, adverse events, and mortality. Readmissions were identified using the unique identifier “nrd_visitlink”. Readmissions were included if they occurred after an index admission, met the inclusion criteria, and were within 30 days. Inherently, patients who died during the index hospitalization were excluded. Patients with multiple readmissions had their primary and subsequent readmissions identified and separated for analysis. Time to readmission was calculated from the day of index admission discharge to the day of readmission. Total cost was provided per individual patient within the NRD database and extracted for analysis. We calculated and compared median and interquartile costs for each treatment modality.
Statistical analysis
Missing data were examined quantitatively and plotted for visualization. Little’s test was used to determine if data were missing completely at random (MCAR) with significance at P < 0.05. Data were also analyzed using the covariate-dependent missingness (CDM) assumption, an extension of Little’s test, accounting for covariates and unequal variances 24 . Variables with more than 2 % missing data that failed Little’s MCAR and CDM testing underwent multiple imputations (25 datasets) for sensitivity analysis 25 .
Data was analyzed by descriptive statistics; categorical values were represented as percentages, while continuous variables were represented by median and interquartile ranges. Pearson χ2 tests were used to compare categorical variables. Kaplan Meier estimations were used to illustrate differences in 30-day mortality among each modality. We used multivariable regression analysis to evaluate the effect of each treatment modality on outcome variables; cox proportion regression analysis was used to determine the risk of mortality. All statistical analyses were performed with Stata 17 (College Station, Texas, United States). The study was determined to be exempt from Institutional Review Board review because human subjects were not involved. Data was acquired from a de-identified registry publicly accessible.
Results
Study characteristics
Between 2016 and 2019, 2,281 patients met the selection criteria of which 34 % (n = 786) were females. The method of pancreatic necrosectomy (as well as average age with standard deviation) was as follows: endoscopy, 672 (52.1 ± 12.01 years); percutaneous, 1,338 (53.2 ± 11.6 years); and surgery, 271 (52.0 ± 12.01 years). The number of necrosectomies consistently increased each year with 86 procedures performed in 2016, 665 in 2017, 723 in 2018, and 806 in 2019 ( Table 1 ).
Table 1. Study characteristics of patients undergoing surgical, percutaneous, and endoscopic necrosectomy.
| Variables | Surgical necrosectomy | Percutaneous necrosectomy | Endoscopic necrosectomy | Obs, n | P value | |||
| % | SE | % | SE | % | SE | |||
| Hospital procedure volume | ||||||||
|
1.05 | (0.75) | 2.61 | (0.54) | 1.01 | (0.46) | 45 | 0.000 |
|
5.04 | (1.83) | 4.75 | (0.80) | 0.99 | (0.50) | 84 | |
|
11.93 | (2.86) | 7.30 | (1.07) | 4.37 | (1.21) | 159 | |
|
21.40 | (3.78) | 12.99 | (1.54) | 13.33 | (2.16) | 321 | |
|
60.58 | (4.89) | 72.35 | (2.22) | 80.30 | (2.64) | 1,672 | |
|
100.00 | 100.00 | 100.00 | 2,281 | ||||
| Indicator of sex | ||||||||
|
66.96 | (4.82) | 66.75 | (1.81) | 62.51 | (2.79) | 1,494 | 0.463 |
|
33.04 | (4.82) | 33.25 | (1.81) | 37.49 | (2.79) | 786 | |
|
100.00 | 100.00 | 100.00 | 2,281 | ||||
| Calendar year | ||||||||
|
6.34 | (1.92) | 5.16 | (0.98) | 0.00 | (0.00) | 86 | 0.000 |
|
35.16 | (5.26) | 39.83 | (3.05) | 5.43 | (1.45) | 665 | |
|
30.57 | (4.77) | 26.57 | (2.49) | 42.43 | (4.27) | 723 | |
|
27.93 | (4.73) | 28.44 | (2.73) | 52.13 | (4.29) | 806 | |
|
100.00 | 100.00 | 100.00 | 2,281 | ||||
| Insurance carrier | ||||||||
|
30.07 | (4.26) | 31.48 | (1.98) | 29.69 | (2.90) | 671 | 0.923 |
|
22.16 | (3.76) | 19.55 | (1.75) | 17.75 | (2.27) | 421 | |
|
44.27 | (5.36) | 44.28 | (2.08) | 47.63 | (2.78) | 986 | |
|
3.51 | (1.56) | 4.69 | (0.87) | 4.94 | (1.49) | 101 | |
|
100.00 | 100.00 | 100.00 | 2,179 | ||||
| Median household income | ||||||||
|
22.69 | (4.08) | 22.06 | (1.63) | 24.31 | (2.85) | 516 | 0.663 |
|
30.27 | (4.16) | 31.21 | (2.03) | 26.12 | (2.45) | 669 | |
|
31.06 | (3.59) | 27.93 | (1.84) | 28.32 | (2.78) | 643 | |
|
15.98 | (3.09) | 18.80 | (1.66) | 21.25 | (2.52) | 434 | |
|
100.00 | 100.00 | 100.00 | 2,262 | ||||
| Bed size of hospital | ||||||||
|
5.27 | (1.89) | 5.41 | (1.00) | 2.89 | (1.29) | 106 | 0.274 |
|
21.52 | (4.61) | 15.13 | (1.84) | 14.72 | (2.60) | 360 | |
|
73.21 | (4.78) | 79.45 | (2.07) | 82.38 | (2.85) | 1,815 | |
|
100.00 | 100.00 | 100.00 | 2,281 | ||||
| Hospital urban-rural designation | ||||||||
|
59.92 | (5.21) | 65.75 | (2.91) | 70.11 | (3.72) | 1,513 | 0.197 |
|
40.08 | (5.21) | 33.42 | (2.89) | 29.89 | (3.72) | 756 | |
|
0.00 | (0.00) | 0.83 | (0.34) | 0.00 | (0.00) | 11 | |
|
100.00 | 100.00 | 100.00 | 2,281 | ||||
| Teaching status | ||||||||
|
10.23 | (2.70) | 7.86 | (1.11) | 3.42 | (0.98) | 156 | 0.022 |
|
89.77 | (2.70) | 91.31 | (1.17) | 96.58 | (0.98) | 2,114 | |
|
0.00 | (0.00) | 0.83 | (0.34) | 0.00 | (0.00) | 11 | |
|
100.00 | 100.00 | 100.00 | 2,281 | ||||
| Patient Location | ||||||||
|
21.89 | (3.61) | 25.80 | (1.97) | 25.53 | (2.91) | 574 | 0.426 |
|
24.72 | (4.53) | 21.84 | (1.67) | 26.47 | (3.13) | 535 | |
|
30.00 | (4.21) | 22.86 | (1.83) | 20.91 | (2.67) | 526 | |
|
7.91 | (2.47) | 14.09 | (1.55) | 11.45 | (1.92) | 286 | |
|
10.62 | (2.68) | 8.76 | (1.30) | 8.21 | (1.78) | 201 | |
|
4.85 | (1.99) | 6.66 | (1.20) | 7.44 | (1.55) | 152 | |
|
100.00 | 100.00 | 100.00 | 2,274 | ||||
| Admission day is a weekend | ||||||||
|
72.90 | (3.91) | 72.90 | (1.75) | 77.02 | (2.39) | 1,690 | 0.344 |
|
27.10 | (3.91) | 27.10 | (1.75) | 22.98 | (2.39) | 590 | |
|
100.00 | 100.00 | 100.00 | 2,281 | ||||
Hospital practices
The majority of necrosectomies were performed at high-volume centers (75 %, n = 1,672), with a large bed capacity (79.6 %, n = 1,815), and located typically in the central counties of large metropolitan areas with a population exceeding 1 million people (66.3 %, n = 1,513). Clinical centers were likely to be metropolitan teaching hospitals (92.7 %, = 2,114), and the most common payer for the inpatients was private insurance (45 %, n = 986), followed by Medicare (30.8 %, n = 671).
Readmissions
From the total cohort of patients, approximately 24.2 % (n = 552) of patients who underwent necrosectomy experienced 30-day hospital readmission. Using multivariable cox regression analysis, compared to surgery, percutaneous necrosectomy had a mildly higher risk for readmission followed by endoscopy, however, this was not statistically significant (percutaneous hazard ratio (HR): 1.48, 95 % CI 0.86 – 2.53, P = 0.156; endoscopy HR: 1.43, 95 % CI 0.78 – 2.63, P = 0.247) ( Table 2 ).
Table 2. Multivariable cox regression showing the risk of readmission for patients undergoing surgical, percutaneous, and endoscopic necrosectomy.
| Variables | Hazard ratio | P value | 95 % CI |
| Necrosectomy | |||
|
1.00 | ||
|
1.48 | 0.156 | [0.86–2.53] |
|
1.43 | 0.247 | [0.78–2.63] |
| Hospital procedure volume | |||
|
1.00 | ||
|
1.00 | 0.998 | [0.35–2.86] |
|
0.73 | 0.567 | [0.24–2.17] |
|
1.02 | 0.964 | [0.39–2.68] |
|
0.92 | 0.866 | [0.36–2.34] |
|
1.00 | 0.522 | [0.98–1.01] |
| Indicator of sex | |||
|
1.00 | ||
|
1.00 | 0.999 | [0.71–1.42] |
| Calendar year | |||
|
1.00 | ||
|
1.32 | 0.472 | [0.62–2.81] |
|
1.40 | 0.368 | [0.67–2.94] |
|
1.72 | 0.150 | [0.82–3.62] |
| Insurance carrier | |||
|
1.00 | ||
|
1.01 | 0.956 | [0.62–1.65] |
|
1.11 | 0.574 | [0.77–1.61] |
|
0.84 | 0.724 | [0.32–2.21] |
| Median household income | |||
|
1.00 | ||
|
1.16 | 0.498 | [0.76–1.77] |
|
1.30 | 0.277 | [0.81–2.07] |
|
1.33 | 0.295 | [0.78–2.25] |
| Bed size of hospital | |||
|
1.00 | ||
|
1.15 | 0.758 | [0.47–2.79] |
|
1.30 | 0.531 | [0.57–2.98] |
| Hospital urban-rural designation | |||
|
1.00 | ||
|
1.52 | 0.081 | [0.95–2.43] |
|
1.17 | 0.780 | [0.38–3.63] |
| Teaching status | |||
|
1.00 | ||
|
0.97 | 0.934 | [0.53–1.80] |
|
1.00 | ||
| Patient location | |||
|
1.00 | ||
|
0.96 | 0.851 | [0.63–1.47] |
|
1.01 | 0.975 | [0.55–1.84] |
|
1.02 | 0.955 | [0.57–1.83] |
|
1.00 | 0.999 | [0.45–2.23] |
|
1.18 | 0.679 | [0.54–2.60] |
|
0.80 | 0.233 | [0.55–1.16] |
|
1.10 | 0.013 | [1.02–1.18] |
Mortality and adverse events
Kaplan Meier survival analysis comparing endoscopy, percutaneous, and surgical necrosectomy revealed that endoscopy was associated with the lowest 30-day mortality outcome (P = 0.0356) ( Fig. 1 ). Using adjusted Cox proportion regression models, compared to surgery, the risks of mortality was lowest for endoscopy (HR: 0.27; 95 % CI 0.08–0.90; P = 0.033) followed by percutaneous (HR: 0.44; 95 % CI, 0.20–0.98; P = 0.045) ( Table 3 ). Endoscopy was associated with less post-procedure bleeding and need for transfusion of blood products compared to percutaneous and surgical necrosectomy (n = 74 vs n = 195 vs n = 102; P < 0.001 and n = 41 vs n = 177 vs n = 51; P < 0.001, respectively). Endoscopy and surgery had similar rates of acute kidney injury (AKI); however, the rate of AKI was lower when compared to percutaneous necrosectomy (n = 134, vs n = 449, vs n = 133; P < 0.001). Endoscopic necrosectomy was least likely to be associated with respiratory failure when compared to percutaneous and surgical approaches (n < 10, vs n = 18, vs n = 13; P = 0.002), see Supplementary Table 1 . Due to HCUP guidelines, data with counts < 10 could not be presented.
Fig. 1.
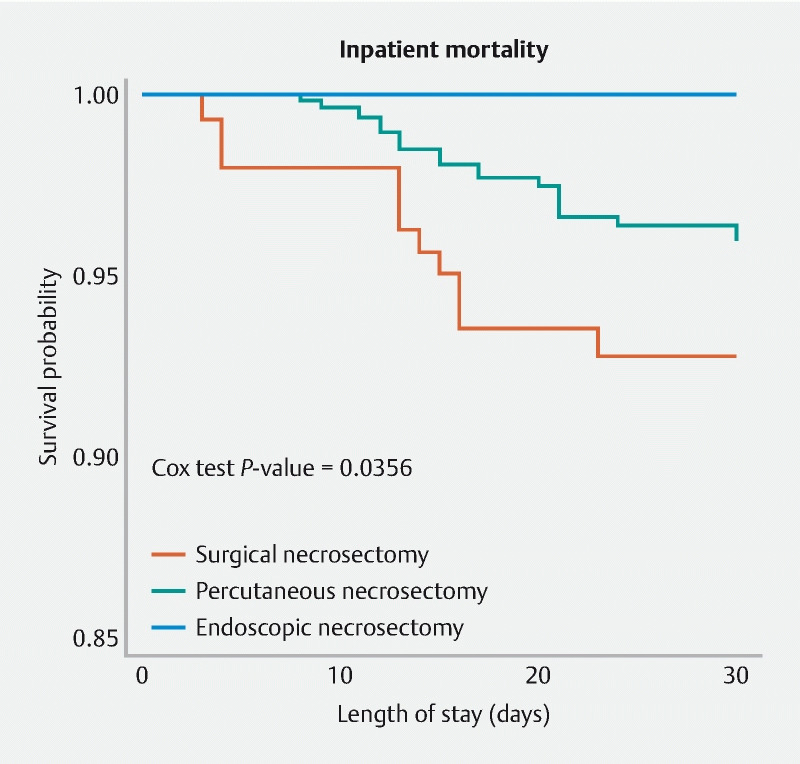
Survival curve comparing endoscopic, percutaneous, and surgical necrosectomy using cox regression analysis.
Table 3. Multivariable cox regression showing mortality risk.
| Variables | Hazard Ratio | P value | 95 % CI |
| Necrosectomy | |||
|
1.00 | ||
|
0.44 | 0.045 | [0.20–0.98] |
|
0.27 | 0.033 | [0.08–0.90] |
| Hospital procedure volume | |||
|
1.00 | ||
|
586303537.04 | ||
|
637016451.05 | 0.000 | [7.3e + 07–5.5e + 09] |
|
1172755835.53 | 0.000 | [8.9e + 07–1.5e + 10] |
|
1036563885.57 | 0.000 | [6.8e + 07–1.6e + 10] |
|
1.05 | 0.004 | [1.02–1.09] |
| Indicator of sex | |||
|
1.00 | ||
|
0.68 | 0.411 | [0.27–1.70] |
| Calendar year | |||
|
1.00 | ||
|
1.53 | 0.514 | [0.43–5.45] |
|
3.36 | 0.010 | [1.33–8.47] |
|
3.66 | 0.010 | [1.36–9.82] |
| Insurance carrier | |||
|
1.00 | ||
|
0.67 | 0.663 | [0.11–3.98] |
|
1.33 | 0.504 | [0.58–3.06] |
|
1.03 | 0.959 | [0.29–3.64] |
| Median household income | |||
|
1.00 | ||
|
1.52 | 0.469 | [0.49–4.78] |
|
0.93 | 0.907 | [0.28–3.09] |
|
0.62 | 0.461 | [0.17–2.22] |
| Bed size of hospital | |||
|
1.00 | ||
|
3.00 | 0.499 | [0.12 –73.47] |
|
1.71 | 0.761 | [0.05 –53.50] |
| Hospital urban-rural designation | |||
|
1.00 | ||
|
1.40 | 0.586 | [0.41–4.76] |
|
0.00 | ||
| Teaching status of urban hospitals | |||
|
1.00 | ||
|
1.86 | 0.563 | [0.23 –15.09] |
|
1.00 | ||
| Patient Location: NCHS Urban-Rural Code | |||
|
1.00 | ||
|
0.98 | 0.963 | [0.38–2.49] |
|
1.07 | 0.924 | [0.26–4.47] |
|
0.92 | 0.903 | [0.22–3.82] |
|
0.38 | 0.355 | [0.05–2.98] |
|
0.70 | 0.758 | [0.07–6.82] |
|
0.73 | 0.497 | [0.30–1.79] |
|
0.99 | 0.956 | [0.80–1.23] |
Length of stay and cost
Endoscopy was associated with an average shorter length of stay of 20.1 days compared to 25.8 days for percutaneous necrosectomy and 38.3 days for surgical necrosectomy ( P < 0.001). Total hospital costs were lowest for endoscopy compared to percutaneous and surgical necrosectomies, respectively ($57K vs $76K vs $123K, P < 0.001).
Discussion
To our knowledge, this is the first nationwide study to assess outcomes and healthcare utilization among patients undergoing three therapeutic modalities for pancreatic necrosectomy using endoscopic, percutaneous, and surgical approaches. This study includes several novel observations.
First, the number of necrosectomies identified in this nationwide database increased each year and were primarily performed at academic teaching hospitals. It is unclear what is driving this increased number of pancreatic necrosectomies and may be multifactorial. However, anecdotally, since the widespread adoption of lumen apposing metal stents (LAMS) into clinical practice as well as the adoption of a ‘step-up’ approach, the number of necrosectomies has shifted towards endoscopy from percutaneous and surgical approaches. However, this may not fully explain the rise observed in this study and may reflect a limitation of the database pertaining to a change in coding from ICD-9 to ICD-10.
Readmission rates were similar for all three methods. Our data is in-line with a single center UK study comparing endoscopic, minimal access retroperitoneal (MARPN), and open necrosectomy which reported similar readmission rates among these modalities ( P = 0.850) 26 . A US study by Bang et al. compared endoscopic and surgical approaches, which also reported similar readmission rates between the two cohorts ( P = 0.145) 27 .
Second, endoscopy was associated with significantly less mortality (despite controlling for the severity of illness across multiple parameters using Elixhauser comorbidity scores). When compared to surgery, patients undergoing endoscopic necrosectomy were 73 % less likely to experience mortality, while the percutaneous approach was associated with 54 % less mortality. To this end, a meta-analysis comparing endoscopic and surgical drainage for necrotizing pancreatitis comprising of 11,807 patients showed that the pooled rate of mortality following surgical necrosectomy was 22 % (95 % confidence interval [CI]: 19 %-26 %) and 3 % (95 %CI:2 %-4 %) in the endoscopic drainage cohort 28 . Khan et al. performed a meta-analysis comparing endoscopic and percutaneous drainage of pancreatic fluid collections which included seven studies with 490 patients 29 . The study reported that five deaths occurred during 30-day follow-up in the percutaneous group, while no deaths occurred in the endoscopic group ( P = 0.16). Our study looked at real-world outcomes using a readmission claims database to provide nationally representative and comparative data across all three modalities; our analysis demonstrated that endoscopy was least likely to be associated with inpatient mortality as well as less blood transfusion requirements and incidence of respiratory failure.
Third, endoscopy had the shortest average length of hospital stay. On average, patients stayed a total of 20 days if they had an endoscopic necrosectomy compared to 38 days with percutaneous necrosectomy and 38.3 days with surgical necrosectomy. The randomized trial by Bang et al. showed that patients who had endoscopic necrosectomy spent on average 16.5 (± 12.2) days in the hospital compared with patients who had surgery spent on average 23.3 (17.5) days in the hospital 27 . However, this was not statistically significant ( P = 0.57) likely due to the small sample size (n = 66). However, a trend towards statistical significance ( P = 0.57) together with the findings in our study provides more evidence that patients who undergo endoscopic interventions for treating pancreatic necrosis are likely to experience shorter hospital lengths of stay.
When compared to the cost of surgery, endoscopic necrosectomy, on average, was associated with $66K less cost per patient. Patients who underwent percutaneous necrosectomy experienced an average reduction of $47K compared to the cost associated with surgery. These findings are consistent with prior observations 27 . Prasath et al. performed a cost-effectiveness analysis comparing endoscopic and surgical necrosectomy 30 . The study showed that using an endoscopic step-up approach was the dominant economic strategy with 7.92 QALYs for $90,864.09. Using a surgical step-up approach resulted in a decrease of 0.09 QALYs and a cost increase of $10,067.89 while open necrosectomy resulted in a decrease of 0.4 QALYs and an increased cost of $18,407.52.
Our study has limitations. First, admissions were identified using only the primary diagnosis, consistent with best used methodologies provided by HCUP. This accepted protocol ensures that only patients with pancreatic necrosis are identified. Nonetheless, patients may be missed if their disease led to admission but was not listed as the first diagnosis or if they were treated as outpatients. Second, due to the retrospective observational nature of this study, residual confounders may be missed, and causality cannot be established. Third, while we were able to control for severity of illness, we were unable to account for why patients were treated by a specific modality. In theory, percutaneous and/or surgical approaches could be used for more complex cases and thereby influence outcomes. Furthermore, despite controlling for severity of illness, mortality in severe acute pancreatitis can be due to several other systemic factors including the presence of infected necrosis not fully accounted for in our study. Fourth, the NRD does not provide medications, laboratory, or imaging results. Finally, as comorbidities are identified using the presence of specific ICD-10 codes, the potential for coding errors or missed coding may skew the results.
Conclusions
Our study is the first to compare endoscopic, percutaneous, and surgical necrosectomy across a validated nationwide inpatient database. This study highlights the safety and economic impact of using endoscopy as the preferred method for necrosectomy in patients with pancreatic necrosis.
Footnotes
Competing interests Dr. Douglas G. Adler is a consultant for Boston Scientific and Micro Tech. All other authors have no relevant conflict of interests.
Supplementary material :
References
- 1.Shaheen N J, Hansen R A, Morgan D R et al. The burden of gastrointestinal and liver diseases, 2006. Am J Gastroenterol. 2006;101:2128–2138. doi: 10.1111/j.1572-0241.2006.00723.x. [DOI] [PubMed] [Google Scholar]
- 2.Fagenholz P J, Fernández-del Castillo C, Harris N S et al. Direct medical costs of acute pancreatitis hospitalizations in the United States. Pancreas. 2007;35:302–307. doi: 10.1097/MPA.0b013e3180cac24b. [DOI] [PubMed] [Google Scholar]
- 3.Beger H G, Rau B, Mayer J et al. Natural course of acute pancreatitis. World J Surg. 1997;21:130–135. doi: 10.1007/s002689900204. [DOI] [PubMed] [Google Scholar]
- 4.Practice Parameters Committee of the American College of Gastroenterology . Banks P A, Freeman M L. Practice guidelines in acute pancreatitis. Am J Gastroenterol. 2006;101:2379–2400. doi: 10.1111/j.1572-0241.2006.00856.x. [DOI] [PubMed] [Google Scholar]
- 5.Baron T H, DiMaio C J, Wang A Y et al. American Gastroenterological Association Clinical Practice Update: Management of Pancreatic Necrosis. Gastroenterology. 2020;158:67–750. doi: 10.1053/j.gastro.2019.07.064. [DOI] [PubMed] [Google Scholar]
- 6.Traverso L W, Kozarek R A. Pancreatic necrosectomy: definitions and technique. J Gastrointest Surg. 2005;9:436–439. doi: 10.1016/j.gassur.2004.05.013. [DOI] [PubMed] [Google Scholar]
- 7.Tsiotos G G, Luque-de León E, Sarr M G. Long-term outcome of necrotizing pancreatitis treated by necrosectomy. Br J Surg. 1998;85:1650–1653. doi: 10.1046/j.1365-2168.1998.00950.x. [DOI] [PubMed] [Google Scholar]
- 8.Rau B, Bothe A, Beger H G. Surgical treatment of necrotizing pancreatitis by necrosectomy and closed lavage: changing patient characteristics and outcome in a 19-year, single-center series. Surgery. 2005;138:28–39. doi: 10.1016/j.surg.2005.03.010. [DOI] [PubMed] [Google Scholar]
- 9.Connor S, Alexakis N, Raraty M G et al. Early and late complications after pancreatic necrosectomy. Surgery. 2005;137:499–505. doi: 10.1016/j.surg.2005.01.003. [DOI] [PubMed] [Google Scholar]
- 10.Howard T J, Patel J B, Zyromski N et al. Declining morbidity and mortality rates in the surgical management of pancreatic necrosis. J Gastrointest Surg. 2007;11:43–49. doi: 10.1007/s11605-007-0112-4. [DOI] [PubMed] [Google Scholar]
- 11.Haghshenasskashani A, Laurence J M, Kwan V et al. Endoscopic necrosectomy of pancreatic necrosis: a systematic review. Surg Endosc. 2011;25:3724–3730. doi: 10.1007/s00464-011-1795-x. [DOI] [PubMed] [Google Scholar]
- 12.Working Group IAP/APA Acute Pancreatitis Guidelines . IAP/APA evidence-based guidelines for the management of acute pancreatitis. Pancreatology. 2013;13:e1–e15. doi: 10.1016/j.pan.2013.07.063. [DOI] [PubMed] [Google Scholar]
- 13.Arvanitakis M, Dumonceau J M, Albert J et al. Endoscopic management of acute necrotizing pancreatitis: European Society of Gastrointestinal Endoscopy (ESGE) evidence-based multidisciplinary guidelines. Endoscopy. 2018;50:524–546. doi: 10.1055/a-0588-5365. [DOI] [PubMed] [Google Scholar]
- 14.Ramai D, Enofe I, Deliwala S S et al. Early (<4 Weeks) Versus Standard (≥ 4 Weeks) Endoscopic Drainage of Pancreatic Walled-Off Fluid Collections: A Systematic Review and Meta-analysis [published online ahead of print, 2022 Nov 14] Gastrointest Endosc. 2022 doi: 10.1016/j.gie.2022.11.003. [DOI] [PubMed] [Google Scholar]
- 15.Onnekink A M, Boxhoorn L, Timmerhuis H C et al. Endoscopic Versus Surgical Step-Up Approach for Infected Necrotizing Pancreatitis (ExTENSION): long-term follow-up of a randomized trial. Gastroenterology. 2022;163:712–7.22E16. doi: 10.1053/j.gastro.2022.05.015. [DOI] [PubMed] [Google Scholar]
- 16.van Santvoort H C, Besselink M G, Bakker O J et al. A step-up approach or open necrosectomy for necrotizing pancreatitis. N Engl J Med. 2010;362:1491–1502. doi: 10.1056/NEJMoa0908821. [DOI] [PubMed] [Google Scholar]
- 17.van Brunschot S, van Grinsven J, van Santvoort H C et al. Endoscopic or surgical step-up approach for infected necrotising pancreatitis: a multicentre randomised trial. Lancet. 2018;391:51–58. doi: 10.1016/S0140-6736(17)32404-2. [DOI] [PubMed] [Google Scholar]
- 18.Hollemans R A, Bakker O J, Boermeester M A et al. Superiority of step-up approach vs open necrosectomy in long-term follow-up of patients with necrotizing pancreatitis. Gastroenterology. 2019;156:1016–1026. doi: 10.1053/j.gastro.2018.10.045. [DOI] [PubMed] [Google Scholar]
- 19.Ricci C, Pagano N, Ingaldi C et al. Treatment for infected pancreatic necrosis should be delayed, possibly avoiding an open surgical approach: a systematic review and network meta-analysis. Ann Surg. 2021;273:251–257. doi: 10.1097/SLA.0000000000003767. [DOI] [PubMed] [Google Scholar]
- 20.HCUP Nationwide Readmissions Database (NRD) . Rockville, MD: Agency for Healthcare Research and Quality; 2019. Healthcare Cost and Utilization Project (HCUP) https://www.hcup-us.ahrq.gov/nrdoverview.jsp
- 21.Moore B J, White S, Washington R et al. Identifying increased risk of readmission and in-hospital mortality using hospital administrative data: The AHRQ Elixhauser Comorbidity index. Med Care. 2017;55:698–705. doi: 10.1097/MLR.0000000000000735. [DOI] [PubMed] [Google Scholar]
- 22.Quan H, Sundararajan V, Halfon P et al. Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Med Care. 2005;43:1130–1139. doi: 10.1097/01.mlr.0000182534.19832.83. [DOI] [PubMed] [Google Scholar]
- 23.van Walraven C, Austin P C, Jennings A et al. A modification of the Elixhauser comorbidity measures into a point system for hospital death using administrative data. Med Care. 2009;47:626–633. doi: 10.1097/MLR.0b013e31819432e5. [DOI] [PubMed] [Google Scholar]
- 24.Li C. Little’s test of missing completely at random. Stata J. 2013;13:795–809. [Google Scholar]
- 25.Royston P. Multiple imputation of missing values. Stata J. 2004;4:227–241. [Google Scholar]
- 26.Saunders R, Hughes F E, Evans J C et al. Cost analysis and outcomes of endoscopic, minimal access and open pancreatic necrosectomy. Ann Surg Open. 2021;2:e068. doi: 10.1097/AS9.0000000000000068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bang J Y, Arnoletti J P, Holt B A et al. An endoscopic transluminal approach, compared with minimally invasive surgery, reduces complications and costs for patients with necrotizing pancreatitis. Gastroenterology. 2019;156:1027–1040. doi: 10.1053/j.gastro.2018.11.031. [DOI] [PubMed] [Google Scholar]
- 28.Mohamadnejad M, Anushiravani A, Kasaeian A et al. Endoscopic or surgical treatment for necrotizing pancreatitis: Comprehensive systematic review and meta-analysis. Endosc Int Open. 2022;10:E420–E428. doi: 10.1055/a-1783-9229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Khan M A, Hammad T, Khan Z. Endoscopic versus percutaneous management for symptomatic pancreatic fluid collections: a systematic review and meta-analysis. Endosc Int Open. 2018;6:E474–E483. doi: 10.1055/s-0044-102299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Prasath V, Quinn P L, Oliver J B et al. Cost-effectiveness analysis of infected necrotizing pancreatitis management in an academic setting. Pancreatology. 2022;22:185–193. doi: 10.1016/j.pan.2021.11.011. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


