Abstract
What Is Known and Objective
Traumatic brain injury (TBI) is a major cause of disability, and it has been associated with agitation and aggression. In a previous study, we reviewed the literature to identify evidence‐based pharmacological agents for treatment of agitation in TBI. Based on the results of our previous study that summarizes the findings of several systematic reviews, the use of haloperidol and benzodiazepines is not supported by the available evidence while the use of amantadine, beta blockers, antiepileptics and methylphenidate is supported by the limited available evidence. In this study, we describe the psycho‐pharmacological agents that were administered to patients with agitation and/or aggression in the context of TBI in inpatient facilities of a private, non‐profit health care system in southwest Virginia. We will also compare the psycho‐pharmacological agents ordered before and after psychiatric consultation.
Methods
Adult patients who were admitted to Carilion Clinic's inpatient facilities from March 30, 2013, to March 30, 2018, had a diagnosis of TBI, and received psychiatric consultation for agitation and/or aggression were enrolled in this study. A retrospective review of electronic medical records was conducted by researchers and data were collected on the following measures: ordered psycho‐pharmacological agents, frequency, dosing and duration of orders, whether each administered psycho‐pharmacological agent was started before or after psychiatric consultation, and psycho‐pharmacological agents prescribed upon discharge.
Results and Discussion
About 68% of patients were started on benzodiazepines and/or typical antipsychotics and 23% of patients were subsequently discharged on these medication categories. Only 23% of patients were ordered to receive medications supported by the evidence such as amantadine, beta blockers or antiepileptics. The percentage of patient‐days with an order to receive typical antipsychotics significantly decreased following psychiatric consultation (p = 0.0056), but the percentage of patient‐days with an order to receive benzodiazepines significantly increased following psychiatric consultation (p = 0.0001). This finding remained statistically significant after excluding patients with active or unclear alcohol/benzodiazepine withdrawal (p < 0.0001).
What Is New and Conclusion
This study demonstrates the widespread use of typical antipsychotics and benzodiazepines in the management of agitation in TBI and the importance of multidisciplinary collaboration, research and education of providers to improve patient care.
Keywords: agitation, traumatic brain injury
Traumatic brain injury (TBI) is a major cause of disability, and it has been associated with agitation and aggression. In this study, we examined the psycho‐pharmacological agents that were administered to patients with agitation and/or aggression in the context of TBI in inpatient facilities of a private, non‐profit health care system in Southwest Virginia. About 68% of patients were started on benzodiazepines and/or typical antipsychotics and 23% of patients were subsequently discharged on these medication categories. Only 23% of patients were ordered to receive medications supported by the evidence. Percentage of patients who received each group of psycho‐pharmacological agents. Non‐recommended = Patients who received benzodiazepines OR typical antipsychotics. Recommended = Patients who received amantadine OR beta blockers (propranolol and pindolol) OR methylphenidate OR valproic acid OR carbamazepine. Atypical antipsychotics = Patients who received atypical antipsychotics that can be considered an acceptable alternative for management of acute agitation in TBI based on available evidence. Multiple medication groups = Patients who received all mentioned medication groups. Did not receive = Patients who did not receive any of the medications of interest.

1. WHAT IS KNOWN AND OBJECTIVE
According to the Centres for Disease Control, traumatic brain injury (TBI) is broadly defined as a disruption in the normal function of the brain that can be caused by a bump, blow or jolt to the head, or penetrating head injury. 1 There are ~50 million new cases of TBI worldwide every year and about 3.5 million in the United States alone. 2 Behavioural disorders often ensue following a TBI which are often devastating for both the individual and their family system. For instance, individuals with TBI can become irritable, agitated and aggressive leading to possible loss of employment, relationships, domestic violence and incarceration in its more severe form. 1 , 3 , 4 Mental health professionals often encounter TBI patients in the hospital setting and are commonly called upon to guide treatment of their behavioural disturbances. While addressing potential organic etiologies of agitation and behavioural interventions are considered the first line of treatment for agitation or aggression in the context of TBI, acute pharmacological management is often necessary to ensure safety for both the patient and health care team. However, there are limited guidelines for this treatment.
In a previous study, 5 we reviewed the literature to identify evidence‐based pharmacological agents for the treatment of agitation in TBI. Based on the results of our previous study that summarizes the findings of several systematic reviews, 6 , 7 , 8 , 9 , 10 , 11 , 12 , 13 , 14 , 15 the use of haloperidol and benzodiazepines is not supported by the available evidence while the use of dopaminergic agents (amantadine and methylphenidate), beta blockers (propranolol and pindolol) and antiepileptics (valproic acid) is supported by the limited available evidence. Our findings were in line with the recommendations of the international group of researchers and clinicians (known as INCOG) against the use of haloperidol for agitation in the context of TBI 16 and preclinical data suggesting that haloperidol can hinder neurocognitive recovery following TBI. 17 , 18 , 19 , 20 One clinical question that may follow this information, would be “What agent(s) can be used for acute agitation in the context of TBI?”. Currently, there is one small study conducted in the outpatient setting that supports the use of olanzapine for the treatment of agitation in the context of TBI. 21 In the absence of better evidence, we recommended atypical antipsychotics (and particularly olanzapine) as a practical alternative(s) to typical antipsychotics and benzodiazepines for as‐needed management of acute agitation.
Despite this data, several studies 22 , 23 , 24 indicate that typical antipsychotics with strong dopamine antagonism (such as haloperidol) and benzodiazepines can be frequently used in the management of agitation and/or aggression in patients with TBI. Moreover, decisions regarding the management of agitation in TBI are frequently made in an interdisciplinary environment and there is limited information on how different disciplines affect patient care.
The primary objective of this study is to describe the psycho‐pharmacological agents administered to patients with agitation and/or aggression in the context of TBI in inpatient units of Carilion Clinic. Secondary objectives include evaluating whether there is a difference in prescription patterns before and after psychiatric consultation, evaluating whether the length of stay is different based on whether a patient receives an evidence‐based medication for agitation in TBI or not, and evaluating whether the frequency of agitated days is different before and after starting an evidence‐based medication.
2. METHODS
2.1. Study population
This study included adult patients admitted to Carilion Clinic's inpatient facilities from March 30, 2013, to March 30, 2018, with a diagnosis of TBI who received psychiatric consultation for agitation, aggression or behavioural disturbance during admission. Children (individuals <18‐years‐old) were excluded and there was no maximum age for inclusion in this study. Carilion Clinic is a private, non‐profit health care system located in southwest Virginia and mostly serves Central and southwestern Virginia and Southern West Virginia. Clarion Clinic's inpatient facilities provide acute inpatient care and inpatient rehabilitation and include teaching and non‐teaching hospitals.
To identify relevant encounters, inpatient hospitalizations with the following characteristics were selected: (1) an admission and/or discharge diagnosis of TBI and (2) at least one psychiatric consultation during the course of admission. The ‘reason for consult’ section of psychiatric consult orders were searched for agit*, aggress*, behav* and/or angry. The results were reviewed independently by two researchers for meeting the inclusion/exclusion criteria. Disagreements were resolved by discussion and consensus. All patients that met the inclusion criteria and lacked the exclusion criteria were included in the study. This study was not funded.
2.2. Data collection/extraction
Researchers reviewed the medical records of the included patients retrospectively to extract the following information: age, gender, race/ethnicity, time since index trauma, severity of TBI, length of admission, comorbid conditions, acute alcohol or benzodiazepine withdrawal, timing and frequency of agitation, ordered psycho‐pharmacological agents, frequency, dosing and duration of orders, whether each administered psychopharmacological agent was prescribed before admission, psycho‐pharmacological agents prescribed upon discharge and timing and frequency of psychiatric consultation and follow up.
Agitated days were defined by at least one report of agitation during the day in the medical records. The severity of TBI was defined based on the classification of Veterans Affairs/Department of Defence clinical practice guidelines for management of concussion/mild TBI. 25 The presence/absence of acute alcohol or benzodiazepine withdrawal was determined based on results of psychiatric evaluations and/or documentation of recent alcohol/benzodiazepine use in medical records. The cases without clear documentation of recent alcohol/benzodiazepine use were classified as unclear. Data were divided in half and assigned to two researchers for extraction. Data points without ambiguity were not extracted in duplicates. Questions about data points were resolved through discussion and consensus.
Data were extracted and managed using REDCap electronic data capture software which is approved by Carilion Clinic's institutional review board (IRB). 26 , 27 This software is firewall protected, and compliant with the health insurance portability and accountability act (HIPAA). De‐identified data were then exported from this software into a secure network directory for statistical analysis with SAS. All procedures of this study were approved by Carilion Clinic's IRB.
2.3. Statistical analysis
Data were analysed using SAS 9.4 software. Measures of central tendency and variation were used to describe numeric variables and frequencies and percentages were used for categorical variables. Means and standard deviations (SD) were reported for variables with normal distribution and medians and interquartile ranges (IQR) were reported for variables with skewed distribution.
Psycho‐pharmacological agents were summarized into the following groups: benzodiazepines (except for midazolam as this medication is often used as anaesthetic for procedures), typical antipsychotics, atypical antipsychotics, valproic acid (including valproate, sodium valproate and divalproex), carbamazepine, amantadine, beta blockers (including only propranolol and pindolol that are used for management of agitation in TBI) and methylphenidate. Based on the available evidence 6 , 7 , 8 , 9 , 10 , 11 , 12 , 13 , 14 , 15 and the researchers' previous study, 5 psycho‐pharmacological agents were further summarized into three larger groups: non‐recommended for as‐needed management of acute agitation in TBI (typical antipsychotics and benzodiazepines), recommend for as‐needed management of acute TBI (atypical antipsychotics), and recommended for scheduled (preventative) management of agitation in TBI (amantadine, beta blockers, methylphenidate, valproic acid and carbamazepine). These medication groupings are summarized in Table 1. Data were reported for psychopharmacological agents that were started during admission. Percentages of patients that were started on each category of medications and percentages of patients who were discharged on each category of psychopharmacological agents were reported. The percentage of admission days that each patient was ordered to receive each category of medications was calculated. McNemar's test was used to evaluate whether there was a significant difference in the percentage of admission days each medication group was ordered before and after psychiatric consultation. Paired sample t tests were used to compare the frequency of agitated days before and after initiation of recommended scheduled medications. Patients were divided into two groups based on whether they had received recommended medications (for scheduled and/or as‐needed use) at any point during admission and a Mann–Whitney U test was used to compare length of stay between the two groups.
TABLE 1.
Summary of medication groupings
| As‐needed management of acute agitation in TBI | |
| Non‐recommended | Typical antipsychotics, benzodiazepines |
| Recommended a | Atypical antipsychotics (especially olanzapine) as practical alternatives |
| Scheduled management of agitation in TBI | |
| Recommended | Amantadine (especially in the context of chronic TBI), propranolol (particularly in the context of acute TBI), methylphenidate and antiepileptics |
Note: Please see researchers' previous work 5 for data that supports the strength of each recommendation.
As practical alternative(s) based on limited evidence.
3. RESULTS
The initial electronic record search based on admission/discharge diagnosis of TBI and psychiatric consultation identified 298 visits for 289 unique patients. Following the examination of the ‘reason for consult’ section of psychiatric consult orders and review by researchers for meeting inclusion/exclusion criteria, a total of 31 patients were identified.
The baseline characteristics of the patients are summarized in Table 2. As demonstrated in this table, the average age of our sample was 50.13 years (SD = 17.21), and the majority of patients were male (71%) and White (97%). Median length of stay was 10.99 (IQR = 4.89, 26.12) and 77% of the patients included in this study, were admitted within 20 days of their latest TBI while only 13% presented more than a year after their TBI. Patients who received recommended medications, were more likely to be male (79% vs. 58%), and have moderate–severe TBI (74% vs. 42%). These differences were clinically significant although analytical statistical tests were not performed as they were not included in the study's original protocol.
TABLE 2.
Baseline characteristics
| Variable | All patients | Patients receiving recommended medications | Patients not receiving recommended medications |
|---|---|---|---|
| Sample Size | 31 | 19 | 12 |
| Age | Mean (SD) = 50.13 (17.21) | Mean (SD) = 51.21 (16.54) | Mean (SD) = 48.42 (18.85) |
| Gender | 9 Female, 22 male | 4 Female, 15 male | 5 Female, 7 male |
| Race | 30 Caucasian, 1 African American | 18 Caucasian, 1 African American | 12 Caucasian |
| LOS | Median (IQR) = 10.99 (4.89, 26.12) | Median (IQR) = 17.07 (7.09, 26.77) | Median (IQR) = 7.93 (3.38, 17.50) |
| TBI severity | 8 Mild, 13 moderate, 6 severe, 4 unknown | 3 Mild, 9 moderate, 5 severe, 2 unknown | 5 Mild, 4 moderate, 1 severe, 2 unknown |
| Time since most recent TBI (days) | Median (IQR) = 1 (0, 13) | Median (IQR) = 1 (0, 13) | Median (IQR) = 1 (0, 8) |
| Alcohol/benzodiazepine withdrawal | 2 Active, 25 ruled out, 4 unclear | 2 Active, 14 ruled out, 3 unclear | 11 Ruled out, 1 unclear |
Note: Recommended medications = medications recommended for an‐needed and/or preventative use, TBI severity = defined based on VA/DOD guidelines. 25
Abbreviations: IQR, interquartile range; LOA, length of stay; SD, standard deviation.
Table 3 summarizes the percentage of patients who were started on each medication group during the course of admission. As depicted in this table, ~68% of patients were ordered to receive the non‐recommended medication groups (benzodiazepines and typical antipsychotics) and 23% of patients were subsequently discharged on these medications. Considering atypical antipsychotics as practical alternatives for as‐needed management of acute agitation in TBI, 61% of patients were started on this medication group and 32% of patients were discharged on them. Lastly, only 23% of patients were ordered to receive preventative medications and 10% of patients continued these medications at discharge. Please note that each patient could have received more than one group of medications during the course of admission.
TABLE 3.
Medication groups and percentage of patients started and discharged on each medication group
| Medication group | Percentage of patients who were started on it | Percentage of patients who were discharged on it |
|---|---|---|
| Non‐Recommended | 21/31 = 68% | 7/31 = 23% |
| As‐needed management | ||
| Benzodiazepines (except Midazolam) | 17/31 = 55% | 6/31 = 19% |
| Typical antipsychotics | 14/31 = 45% | 2/31 = 6% |
| Recommended | 19/31 = 61% | 10/31 = 32% |
| As‐needed management | ||
| Atypical antipsychotics a | 19/31 = 61% | 10/31 = 32% |
| Scheduled management | ||
| All combined | 7/31 = 23% | 3/31 = 10% |
| Valproic acid—DVV | 6/31 = 19% | 4/31 = 13% |
| Carbamazepine | 0/31 = 0% | 0/31 = 0% |
| Amantadine | 3/31 = 10% | 2/31 = 6% |
| Beta blockers | 1/31 = 3% | 0/31 = 0% |
| Methylphenidate | 1/31 = 3% | 1/31 = 3% |
Recommended as practical alternative(s) based on limited evidence.
Figure 1 depicts the percentage of patients who received recommended medications, non‐recommended medications and/or atypical antipsychotics during their inpatient treatment. This graph includes both patients who were started on each medication group, as well as those who were on these medications prior to admission and continued to receive them during their hospitalization. As depicted in this graph, the majority of the patients (84%) received non‐recommended medications either alone or in a combination with other medications.
FIGURE 1.
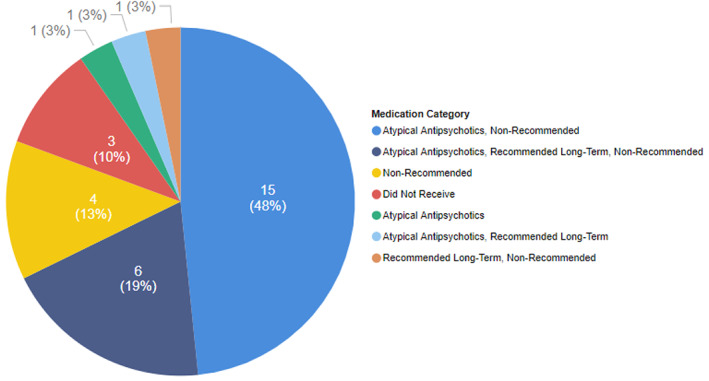
Percentage of patients who received each group of psycho‐pharmacological agents. Non‐recommended = Patients who received benzodiazepines OR typical antipsychotics. Recommended = Patients who received amantadine OR beta blockers (propranolol and pindolol) OR methylphenidate OR valproic acid OR carbamazepine. Atypical antipsychotics = Patients who received atypical antipsychotics that can be considered an acceptable alternative for management of acute agitation in TBI based on available evidence. Multiple medication groups = Patients who received all mentioned medication groups. Did not receive = Patients who did not receive any of the medications of interest.
Table 4 compares and contrasts ordered medications before and after psychiatric consultation. As shown in this table, the percentage of patient‐days with orders to receive benzodiazepines significantly increased following psychiatric consultation (p = 0.0001) and the percentage of patient‐days with orders to receive typical antipsychotics significantly decreased following psychiatric consultation (p = 0.0056). There was not a statistically significant difference between the percentage of days recommended medications were ordered before and after psychiatric consultation (p ≈ 1). Active alcohol/benzodiazepine withdrawal occurred at a frequency of 6%, was ruled out for 81% of the population and was unclear for the remaining 13% of patients. Benzodiazepines can be considered appropriate/necessary for the treatment of patients with alcohol/benzodiazepine withdrawal. As a result, the analysis of patient‐days with orders to receive benzodiazepines before and after psychiatric consultation was repeated after excluding patients with active or unclear alcohol/benzodiazepine withdrawal. The increases in percentage of patient‐days with a benzodiazepine order following psychiatric consultation remained statistically significant (p < 0.0001).
TABLE 4.
Number of patient‐days each medication group was ordered before and after psychiatric consultation
| Medication group | Number of patient‐days this medication was ordered | Number of patient‐days this medication was ordered before psych consult | Number of patient‐days this medication was ordered after psych consult | p value |
|---|---|---|---|---|
| Non‐Recommended | 215/591 = 36% | 63/222 = 28% | 152/369 = 41% | 0.0023 |
| As‐needed management | ||||
| Benzodiazepines (except Midazolam) | 182/591 = 31% | 47/222 = 21% | 135/369 = 37% | 0.0001 |
| Typical antipsychotics | 58/591 = 10% | 32/222 = 14% | 26/369 = 7% | 0.0056 |
| Recommended | 212/591 = 36% | 83/222 = 37% | 129/369 = 35% | 0.6118 |
| As‐needed management | ||||
| Atypical antipsychotics a | 200/591 = 34% | 78/222 = 35% | 122/369 = 33% | 0.6701 |
| Scheduled management | ||||
| All combined | 102/591 = 17% | 37/222 = 17% | 65/369 = 18% | 0.8547 |
| Valproic acid | 83/591 = 14% | 27/222 = 12% | 56/369 = 15% | 0.3686 |
| Carbamazepine | 0/591 = 0% | 0/222 = 0% | 0/369 = 0% | 1 |
| Amantadine | 31/591 = 5% | 15/222 = 7% | 16/369 = 4% | 0.2767 |
| Beta blockers | 15/591 = 3% | 8/222 = 4% | 7/369 = 2% | 0.3137 |
| Methylphenidate | 4/591 = 1% | 0/222 = 0% | 4/369 = 1% | 1 |
Recommended as practical alternative(s) based on limited evidence.
The most commonly prescribed benzodiazepine was lorazepam, and the most commonly prescribed atypical antipsychotic was haloperidol. The average dose of lorazepam administered per day of admission for patients who received this medication was 1.05 mg/day and the average dose of haloperidol administered per day of admission for patients who received this medication was 2.96 mg/day. There was not a statistically significant difference in length of stay between patients who received recommended medications and those who did not receive recommended medications [Median (interquartile range): 17.07 (7.09–26.77) vs. 7.93 (3.37–17.50), respectively, p = 0.1492]. Among seven patients who were started on preventative recommended medications, the frequency of days of agitation was not statistically different before versus after starting preventative recommended medications (33% agitated days before vs. 34% agitated days after starting recommended medications, p = 0.999).
4. DISCUSSION
In our study, benzodiazepines and/or typical antipsychotics were ordered for more than two‐thirds of the patients with TBI during the course of admission. Approximately a quarter of the patients were discharged from the hospital on a benzodiazepine and/or a typical antipsychotic. Meanwhile, less than a quarter of the patients were started on medications supported by current evidence for scheduled preventative use. Our data also demonstrate a decrease in the number of days typical antipsychotics were ordered after psychiatric consultation and an increase in the number of days benzodiazepines were ordered after psychiatric consultation. This study did not detect a statistical difference in length of stay between patients who were ordered to receive recommended medications and those who were not ordered to receive them. The frequency of agitated days was not statistically different before vs. after starting recommended preventative medications.
Our results are consistent with other observational studies suggesting that antipsychotics and benzodiazepines are often used for management of agitation in the context of TBI, 22 , 23 , 24 especially by non‐experts. 23 , 24 These findings are in contrast with the current lack of evidence for, and the existence of evidence against, the use of benzodiazepines and typical antipsychotics for management of agitation in TBI. Preclinical 17 , 18 , 19 , 20 and clinical 28 , 29 , 30 studies have suggested that benzodiazepines and antipsychotics may be harmful for patients with TBI as they can interfere with neurocognitive recovery, increase length of stay, and increase the duration of post‐traumatic amnesia. However, there are instances in which benzodiazepines are indicated for agitation. For instance, in cases of agitation due to alcohol or benzodiazepine withdrawal, administering benzodiazepines may be necessary to prevent withdrawal seizures and/or delirium tremens. However, in our sample, the increase in the percentage of patient‐days with an order to receive benzodiazepines remained statistically significant after excluding patients with active or unclear alcohol/benzodiazepine withdrawal. It is unclear whether psychiatry recommended an increase in the use of benzodiazepines for patients or whether other factors led to this increase. This finding and the interactions between psychiatrists and primary providers that could have contributed to this increase will need to be further studied.
One of the secondary objectives of this study was to detect any potential benefit in length of stay or frequency of agitated days for patients who received the recommended medications. No statistically significant difference was detected between length of stay in patients who received the recommended medications and those who did not. Having said that, potential improvements in length of stay may have been masked by differences in confounding demographic characteristics. For instance, patients who received recommended medications were more likely to be male and have moderate to severe TBI. These differences could have led to higher frequency/severity of agitation or other complications of TBI that might have prolonged length of stay. Moreover, as depicted in Figure 1, many patients who received recommended medications also received non‐recommended medications that might have hindered their recovery. The frequency of agitated days was also not statistically different before versus after starting recommended scheduled medications. It is noteworthy, however, that only a small group of patients were started on these medications during admission. This small sample size might have been insufficient to detect improvement. Moreover, this retrospective chart review was not able to use standardized measures to detect slight changes in agitation. Further prospective studies with larger sample sizes and the use of standardized scales may be required to replicate the effects of these medications.
The strengths of our study include a detailed chart review of pharmacological management of agitation and/or aggression in patients with TBI incorporating a comparison of practice patterns before and after psychiatric consult and medications prescribed upon discharge. The limitations of our study include a small sample size and reliance on a retrospective review of medical records which may be subject to human error and missing information. Data extraction was not performed in duplicates which further exposes the data to human error. The classification of medication groups in this study is based on the currently available evidence and this classification may change with the advent of more robust evidence. The researchers' literature review and design of this study were performed prior to the popularity of alpha 2 agonists for the treatment of agitated delirium. 31 , 32 As a result, researchers did not collect information on this group of medications which is another limitation of this study.
It is also important to highlight that most of our sample were White males. In general, men account for approximately two‐thirds of TBI‐related medical encounters 1 which is consistent with our sample. This study's patient population was also predominantly White (97%) which is consistent with southwest Virginia's population demographics (93% White 33 ). However, Carilion Clinic also serves several other regions of Virginia (Valley, Southside and Central) and several neighbouring states with lower percentages of White people. As a result, the possibility of underutilization of psychiatric consultation for management of agitation for non‐White patients may need to be considered. Regardless of the reason, the demographic characteristics of our sample, limit the generalizability of the findings to non‐Caucasian populations.
5. WHAT IS NEW AND CONCLUSION
This study demonstrates the widespread use of typical antipsychotics and benzodiazepines in management of agitation in the context of TBI and a significant reduction in the use of typical antipsychotics following psychiatric consultation. These findings suggest that educating providers about the management of agitation in TBI and further multidisciplinary collaboration may improve the care of patients with TBI.
CONFLICT OF INTEREST
The authors declare no conflicts of interest.
PATIENT CONSENT STATEMENT
This research is a retrospective observational study. Direct patient consent was not obtained. This study was approved by Carilion Clinic's Institutional Review Board (IRB).
Rahmani E, Lemelle T, Sharp H, Smarbafzadeh E, Kablinger A. A descriptive analysis of pharmacological management of aggression and/or agitation in patients with traumatic brain injury in a Southwest Virginia inpatient population. J Clin Pharm Ther. 2022;47(12):2083‐2090. doi: 10.1111/jcpt.13754
DATA AVAILABILITY STATEMENT
The data that support the findings of this study are available from Carilion Clinic. Restrictions apply to the availability of these data, which were used under licence for this study. Data are available with the permission of Carilion Clinic.
REFERENCES
- 1. Centers for Disease Control and Prevention . Report to congress on traumatic brain injury in the United States: epidemiology and rehabilitation. National Center for Injury Prevention and Control; Division of Unintentional Injury Prevention; 2015. [DOI] [PubMed] [Google Scholar]
- 2. Maas AIR, Menon DK, Adelson PD, et al. Traumatic brain injury: integrated approaches to improve prevention, clinical care, and research. Lancet Neurol. 2017;16:987‐1048. [DOI] [PubMed] [Google Scholar]
- 3. Vaishnavi S, Rao V, Fann JR. Neuropsychiatric problems after traumatic brain injury: unraveling the silent epidemic. Psychosomatics. 2009;50:198‐205. [DOI] [PubMed] [Google Scholar]
- 4. Lauterbach MD, Notarangelo PL, Nichols SJ, Lane KS, Koliatsos VE. Diagnostic and treatment challenges in traumatic brain injury patients with severe neuropsychiatric symptoms: insights into psychiatric practice. Neuropsychiatr Dis Treat. 2015;11:1601‐1607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Rahmani E, Lemelle TM, Samarbafzadeh E, Kablinger AS. Pharmacological treatment of agitation and/or aggression in patients with traumatic brain injury: a systematic review of reviews. J Head Trauma Rehabil. 2021;36(4):E262‐E283. doi: 10.1097/HTR.0000000000000656 [DOI] [PubMed] [Google Scholar]
- 6. Luauté J, Plantier D, Wiart L, Tell L, SOFMER group . Care management of the agitation or aggressiveness crisis in patients with TBI. Systematic review of the literature and practice recommendations. Ann Phys Rehabil Med. 2016;59(1):58‐67. doi: 10.1016/j.rehab.2015.11.001 [DOI] [PubMed] [Google Scholar]
- 7. Hicks AJ, Clay FJ, Hopwood M, et al. The efficacy and harms of pharmacological interventions for aggression after traumatic brain injury‐systematic review. Front Neurol. 2019;10:1169. doi: 10.3389/fneur.2019.01169 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Mehta S, McIntyre A, Janzen S, Iruthayarajah J, Bateman A, Teasell R. Pharmacological management of agitation among individuals with moderate to severe acquired brain injury: a systematic review. Brain Inj. 2018;32(3):287‐296. doi: 10.1080/02699052.2017.1419377 [DOI] [PubMed] [Google Scholar]
- 9. Mohamed Ali O, Faruqui RA. Efficacy and tolerability of antipsychotics in treatment of agitation and aggression following traumatic brain injury (Tbi): systematic review of clinical trials. Eur Psychiatry. 2015;30(1):28‐31. [Google Scholar]
- 10. Ter Mors BJ, Backx APM, Spauwen P, Ponds RWHM, Van Harten PN, Van Heugten CM. Efficacy of amantadine on behavioural problems due to acquired brain injury: a systematic review. Brain Inj. 2019;33(9):1137‐1150. doi: 10.1080/02699052.2019.1631482 [DOI] [PubMed] [Google Scholar]
- 11. Nash RP, Weinberg MS, Laughon SL, McCall RC, Bateman JR, Rosenstein DL. Acute pharmacological management of behavioral and emotional dysregulation following a traumatic brain injury: a systematic review of the literature. Psychosomatics. 2019;60(2):139‐152. doi: 10.1016/j.psym.2018.11.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Sami MB, Faruqui R. The effectiveness of dopamine agonists for treatment of neuropsychiatric symptoms post brain injury and stroke. Acta Neuropsychiatr. 2015;27(6):317‐326. doi: 10.1017/neu.2015.17 [DOI] [PubMed] [Google Scholar]
- 13. Stelmaschuk S, Will MC, Meyers T. Amantadine to treat cognitive dysfunction in moderate to severe traumatic brain injury. J Trauma Nurs. 2015;22(4):194‐E2. doi: 10.1097/JTN.0000000000000138 [DOI] [PubMed] [Google Scholar]
- 14. Wheaton P, Mathias JL, Vink R. Impact of pharmacological treatments on cognitive and behavioral outcome in the postacute stages of adult traumatic brain injury: a meta‐analysis. J Clin Psychopharmacol. 2011;31(6):745‐757. doi: 10.1097/JCP.0b013e318235f4ac [DOI] [PubMed] [Google Scholar]
- 15. Williamson D, Frenette AJ, Burry LD, et al. Pharmacological interventions for agitated behaviours in patients with traumatic brain injury: a systematic review. BMJ Open. 2019;9(7):e029604. doi: 10.1136/bmjopen-2019-029604 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Ponsford J, Janzen S, McIntyre A, et al. INCOG recommendations for management of cognition following traumatic brain injury, part I. J Head Trauma Rehabil. 2014;29(4):307‐320. doi: 10.1097/HTR.0000000000000074 [DOI] [PubMed] [Google Scholar]
- 17. Hoffman AN, Cheng JP, Zafonte RD, Kline AE. Administration of haloperidol and risperidone after neurobehavioral testing hinders the recovery of traumatic brain injury‐induced deficits. Life Sci. 2008;83:602‐607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Kline AE, Hoffman AN, Cheng JP, Zafonte RD, Massucci JL. Chronic administration of antipsychotics impede behavioral recovery after experimental traumatic brain injury. Neurosci Lett. 2008;448:263‐267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Kline AE, Massucci JL, Zafonte RD, Dixon CE, DeFeo JR, Rogers EH. Differential effects of single versus multiple administrations of haloperidol and risperidone on functional outcome after experimental brain trauma. Crit Care Med. 2007;35:919‐924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Phelps TI, Bondi CO, Ahmed RH, Olugbade YT, Kline AE. Divergent long‐term consequences of chronic treatment with haloperidol, risperidone, and bromocriptine on traumatic brain injury‐induced cognitive deficits. J Neurotrauma. 2015;32:590‐597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Maturana Waidele R, Maturana Rodillo R. Control de la agresivi‐dad con olanzapina en pacientes post tec [aggressiveness control using olanzapine in post‐TBI patients]. Cienc Trab. 2009;31:22‐24. [Google Scholar]
- 22. Pisa FE, Cosano G, Giangreco M, et al. Prescribing practice and off‐label use of psychotropic medications in post‐acute brain injury rehabilitation centres: a cross‐sectional survey. Brain Inj. 2015;29(4):508‐516. doi: 10.3109/02699052.2014.992474 [DOI] [PubMed] [Google Scholar]
- 23. Fugate LP, Spacek LA, Kresty LA, Levy CE, Johnson JC, Mysiw WJ. Measurement and treatment of agitation following traumatic brain injury: II. A survey of the brain injury special interest Group of the American Academy of physical medicine and rehabilitation. Arch Phys Med Rehabil. 1997;78(9):924‐928. doi: 10.1016/s0003-9993(97)90051-4 [DOI] [PubMed] [Google Scholar]
- 24. Francisco GE, Walker WC, Zasler ND, Bouffard MH. Pharmacological management of neurobehavioural sequelae of traumatic brain injury: a survey of current physiatric practice. Brain Inj. 2007;21(10):1007‐1014. doi: 10.1080/02699050701559558 [DOI] [PubMed] [Google Scholar]
- 25. Management of Concussion/mTBI Working Group . VA/DoD clinical practice guideline for management of concussion/mild traumatic brain injury. J Rehabil Res Dev. 2009;46(6):CP1, 1059‐CP68, 1068. [PubMed] [Google Scholar]
- 26. Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG. Research electronic data capture (REDCap)—a metadata‐driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42(2):377‐381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Harris RT, Minor BL, Elliott V, et al. REDCap consortium, the REDCap consortium: building an international community of software partners. J Biomed Inform. 2019;95:103208. doi: 10.1016/j.jbi.2019.103208 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Anderson RL, Birrer KL, DeRyke XL. Haloperidol use in acute traumatic brain injury: a safety analysis. J Intensive Crit Care. 2016;02:1‐6. [Google Scholar]
- 29. Mysiw WJ, Bogner JA, Corrigan JD, Fugate LP, Clinchot DM, Kadyan V. The impact of acute care medications on rehabilitation outcome after traumatic brain injury. Brain Inj. 2006;20:905‐911. [DOI] [PubMed] [Google Scholar]
- 30. Rao N, Jellinek HM, Woolston DC. Agitation in closed head injury: haloperidol effects on rehabilitation outcome. Arch Phys Med Rehabil. 1985;66:30‐34. [PubMed] [Google Scholar]
- 31. Carrasco G, Baeza N, Cabré L. Dexmedetomidine, agitated delirium, and “off‐label” drugs. J Thorac Dis. 2016;8(10):E1432‐E1434. doi: 10.21037/jtd.2016.10.02 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Mo Y, Zimmermann AE. Role of dexmedetomidine for the prevention and treatment of delirium in intensive care unit patients. Ann Pharmacother. 2013;47(6):869‐876. doi: 10.1345/aph.1AR708 [DOI] [PubMed] [Google Scholar]
- 33. Demographics Research Group at Weldon Cooper Center for Public Service. University of Virginia; 2014:1‐19. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data that support the findings of this study are available from Carilion Clinic. Restrictions apply to the availability of these data, which were used under licence for this study. Data are available with the permission of Carilion Clinic.


