Ureteroscopy is a commonly performed endourological procedure. Raised pelvicalyceal pressure (PCP) is an unavoidable consequence of this procedure, as a degree of distension is required to visualize the ureter and collecting system. This raised pressure is achieved primarily by saline irrigation through the working channel of the ureteroscope. The adverse effects of such raised pressures are known to include pyelovenous reflux, which is the movement of substances from the renal pelvis into the vascular compartment of the kidney and commonly includes contrast agents and pathogens [1]. Normal PCP is reported to be approximately 10 cmH20, with the threshold for pyelovenous reflux thought to be approximately 30 mmHg [2]. Previous studies have reported very high PCPs, with average pressures >50 mmHg during stone management and pressure peaks of up to 328 mmHg [3]. Whilst observational studies are possible during clinically indicated ureterorenoscopy and have been conducted to evaluate the efficacy of equipment designed to minimize pressure within the collecting system [4], experimental models are desirable to thoroughly evaluate the effects of such raised pressure on the kidney and examine interventions intended to decrease these pressures. Previous studies have been performed using animal models, including with anaesthetized pigs [5], and non‐perfused kidneys from animals slaughtered for food production [6]. Here, we report the first use of an ex vivo normothermic machine perfusion (NMP) system for evaluation of the effect of raised pressures on the kidney during ureteroscopy.
The purpose of the study was to adapt our existing NMP system to allow ureterorenoscopy to be performed and physiologically relevant variables, including PCP, to be continuously monitored during experimental manipulation. Three pairs of porcine kidneys were retrieved after the slaughter of pigs for food production at a local abattoir, as reported previously [7]. Following standard Home Office procedures, the pigs were stunned and exsanguinated. The thoracic and abdominal viscera were removed en bloc by trained slaughtermen. The intact viscera were then handed to the investigator (R.D.), who isolated and cannulated the renal arteries before administering a cold saline flush. The warm ischaemic time (time of death to cold flush) was under 15 min in all cases, which is comparable to that experienced by human donor kidneys retrieved for transplant. Both kidneys were then removed from the viscera and attached to a hypothermic machine perfusion system (KidneyAssist transporter; XVIVO, Gothenburg, Sweden) similar to those used clinically for organ preservation. In the first case, the kidneys were resected en bloc with intact ureters, bladder and short section of urethra; in the second and third cases, the bladder was removed and ureters kept long (>20 cm). Autologous blood was collected directly into citrate‐phosphate‐dextrose solution‐containing Macopharma blood collection sets and filtered to remove leucocytes.
Two KidneyAssist (XVIVO) ex vivo NMP devices were set up with perfusion circuits including a centrifugal blood pump, hollow‐fibre oxygenator, and custom 3D‐printed organ chamber, venous reservoir and ureteric gantry (Fig. 1D). The technique is based on cardiopulmonary bypass technology and involves pumping warmed, oxygenated blood through a cannula in the renal artery. Blood then circulates through the kidney and drains via the renal vein into a reservoir and back to the pump. We used 600 mL autologous leucocyte‐depleted blood diluted with Ringer's solution to a haematocrit of 25% with heparin (5000 IU), co‐amoxiclav (1200 mg) and calcium gluconate. Perfusion pressure was fixed at a mean of 80 mmHg and temperature was kept at 37°C. The ureter of the control kidney was cannulated prior to connection of the kidney to the circuit; the ureter of the kidney to be subject to ureteroscopy (‘intervention’) was extended from the perfusion chamber and secured to a gantry to facilitate insertion of a flexible ureteroscope. Renal blood flow (RBF), arterial pressure and arterial temperature were continuously monitored. After a 1‐h period of NMP, allowing RBF to stabilize, a single‐use digital flexible ureteroscope (Lithovue™; Boston Scientific, Marlborough, MA, USA) was inserted via the bladder (Case 1) or distal ureter (Cases 2 and 3), with the tip positioned in the centre of the renal pelvis. A pressure sensor was inserted via the working channel of the ureteroscope to permit continuous measurement of PCP. Saline irrigation was pressurized by means of a manual pressure bag. Creatinine was added to the irrigation fluid to attain a concentration of 1000 μM, in order that the rate of appearance in the perfusate could be measured to quantify pyelovenous reflux. PCP was increased through the course of each experiment in a stepwise fashion, with target pressures up to 200 mmHg.
Fig. 1.
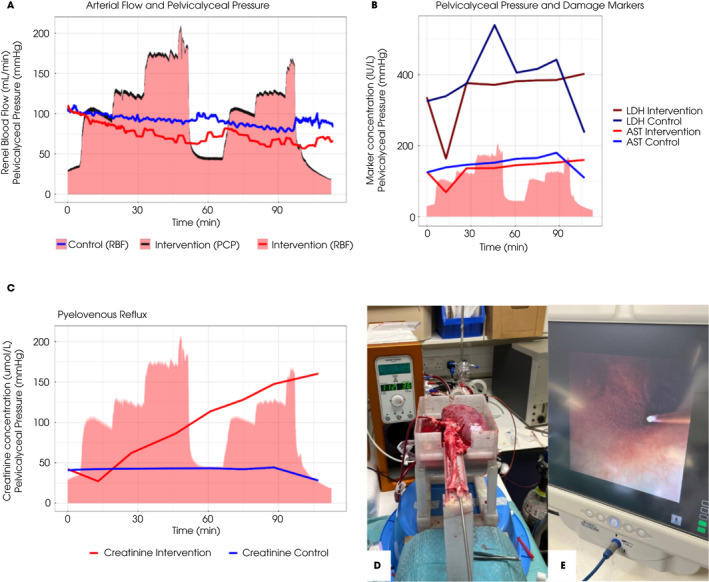
Ureteroscopy during ex vivo normothermic kidney perfusion. (A) Renal blood flow (RBF) for the intervention kidney (red line) and control kidney (blue line), overlaid on pelvicalyceal pressure (PCP) exerted on the intervention kidney (black line, red area). The adverse effect of raised pressures on RBF can be seen. (B) Damage markers (lactate dehydrogenase [LDH] and aspartate aminotransferase [AST]) overlaid on the same PCP curve. There was no significant difference between the two kidneys. (C) Appearance of creatinine in the perfusate of the kidney undergoing intervention indicates pyelovenous reflux, which can be quantified. (D) Image showing ex vivo perfused kidney and experimental set‐up. (E) Endoscopic image showing the pressure wire tip positioned in the collecting system. All images and data were derived from the third of three reported pilot experiments.
The first experiment demonstrated that urological intervention with PCP monitoring during ex vivo normothermic kidney perfusion was possible. Pressure sensing was sufficiently accurate to discern regular vermiculation in the perfused segment of ureter. Cases 2 and 3 showed that the experimental set‐up and results are consistent and reproducible. Representative results from the third experiment are shown in Fig. 1. PCPs typical of those seen during clinical ureteroscopy were achieved using a manual pressure bag with the saline irrigation (red area in Fig. 1A–C). Increasing PCP resulted in attenuation of RBF in the kidney undergoing intervention, with temporary recovery between experimental runs (red line in Fig. 1A). Despite impairment of RBF, we did not detect a difference in markers indicative of renal injury (aspartate aminotransferase and lactate dehydrogenase; Fig. 1B); however, we did see clear evidence of pyelovenous reflux with progressive appearance of the creatinine that had been spiked in the irrigation fluid in perfusate (Fig. 1C). Figure 1D shows the macroscopic appearance of an ex vivo, instrumented kidney during NMP; Fig. 1E shows an endoscopic view of the collecting system with the pressure sensor wire positioned centrally.
Normothermic machine perfusion is currently used as a research tool assessing donor organ injury and modification through addition of therapies, and as a preservation method in the clinical setting. We have shown that this technology has the potential to be used more widely and can be adapted for endourological use. Our data demonstrate that ex vivo perfusion of porcine kidneys retrieved from a commercial abattoir may be used for functional assessment of the impact of endourological intervention on the kidney. This model is commensurate with the ‘3Rs’ principle (replacement, reduction and refinement) underlying the conduct of animal experiments, as it obviates the need for an experimental animal. This is associated with ethical benefit and substantial cost savings. Physiological variables may be measured (e.g., RBF) and controlled (e.g., renal perfusion and collecting system pressures). The NMP model is not restricted to evaluating the effects of pressure and provides advantages over non‐perfused bench models for other ureteroscopic experiments. One limitation is the effect of ischaemia‐reperfusion on the kidney; pre‐intervention RBF is lower during NMP than it is in vivo. Despite this, we were able to demonstrate the impact of instrumentation and raised collecting system pressures. Pyelovenous reflux is a known phenomenon which may result clinically in urosepsis, when pathogens and endotoxin are forced into the circulation; the use of creatinine as a marker allows accurate quantification of the extent of this phenomenon, which can only be done in the controlled setting of ex vivo NMP, where the circulating volume is known and fixed. In conclusion, ureterorenoscopy of the ex vivo machine‐perfused porcine kidney, with organs derived from the by‐products of commercial food production, is shown to be a useful and reproducible model for assessment of the impact of endourological procedures on the kidney.
Disclosure of Interests
None of the authors have anything to declare.
Abbreviations
- NMP
normothermic machine perfusion
- PCP
pelvicalyceal pressure
- RBF
renal blood flow
Acknowledgement
This work was supported by a grant from Boston Scientific.
References
- 1. Bapir R, Bhatti KH, Eliwa A et al. Infectious complications of endourological treatment of kidney stones: a meta‐analysis of randomized clinical trials. Arch Ital Urol Androl 2022; 94: 97–106 [DOI] [PubMed] [Google Scholar]
- 2. Tokas T, Herrmann TRW, Skolarikos A, Nagele U. Pressure matters: intrarenal pressures during normal and pathological conditions, and impact of increased values to renal physiology. World J Urol 2019; 37: 125–31 [DOI] [PubMed] [Google Scholar]
- 3. Jung H, Osther PJS. Intraluminal pressure profiles during flexible ureterorenoscopy. SpringerPlus 2015; 4: 373 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Auge BK, Pietrow PK, Lallas CD, Raj GV, Santa‐Cruz RW, Preminger GM. Ureteral access sheath provides protection against elevated renal pressures during routine flexible Ureteroscopic stone manipulation. J Endourol 2004; 18: 33–6 [DOI] [PubMed] [Google Scholar]
- 5. Noureldin YA, Kallidonis P, Ntasiotis P, Adamou C, Zazas E, Liatsikos EN. The effect of irrigation power and ureteral access sheath diameter on the maximal intra‐pelvic pressure during ureteroscopy: in vivo experimental study in a live anesthetized pig. J Endourol 2019; 33: 725–9 [DOI] [PubMed] [Google Scholar]
- 6. Maccraith E, Yap LC, Elamin M, Patterson K, Brady CM, Hennessey DB. Evaluation of the impact of ureteroscope, access sheath, and irrigation system selection on intrarenal pressures in a porcine kidney model. J Endourol 2021; 35: 512–7 [DOI] [PubMed] [Google Scholar]
- 7. Dengu F, Neri F, Ogbemudia E et al. Abdominal multiorgan procurement from slaughterhouse pigs: a bespoke model in organ donation after circulatory death for ex vivo organ perfusion compliant with the 3 Rs (reduction, Replacement & Refinement). Ann Transl Med 2022; 10: 1 [DOI] [PMC free article] [PubMed] [Google Scholar]


