Abstract
Objective
To report the long-term outcomes of anti-vascular endothelial growth factor (VEGF) treatment in eyes with peripapillary choroidal neovascularisation (PPCNV) associated with age-related macular degeneration (AMD).
Methods
A retrospective cohort study included patients with AMD-related PPCNV. Eyes were treated with anti-VEGF according to pro re nata regimen. Inactivation index was calculated as the proportion of disease inactivity from the total follow up time.
Results
Sixty-seven eyes of 66 consecutive patients were included in the study; mean follow-up time was 53.2 months. Best corrected visual acuity (BCVA) remained stable for the first four years of follow up, with a significant deterioration in BCVA thereafter. Baseline BCVA was a significant predictor of final BCVA (p < 0.001). The mean inactivation index was 0.38 ± 0.23. Subretinal fluid (SRF) at presentation was significantly associated with decreased inactivation index (p < 0.05). Worse baseline BCVA, SRF and pigment epithelium detachment (PED), male sex, and younger patient age were associated with increased risk for recurrence after first inactivation (p < 0.05).
Conclusion
The use of anti-VEGF agents in the treatment of AMD-related PPCNV managed to preserve BCVA in the first four years of follow-up. Male sex, SRF and PED at presentation and baseline BCVA are associated with increased risk for PPCNV recurrence after the first inactivation, and should prompt careful follow-up in these patients.
Subject terms: Retinal diseases, Macular degeneration, Vision disorders
Introduction
Peripapillary choroidal neovascularisation (PPCNV) is a rare subtype of choroidal neovascular membrane (CNVM), accounting for about 10% of all CNVMs [1]. In the BEAP study, the prevalence of PPCNV in an elderly UK population aged ≥65 years was 0.29% with a female predilection [2]. Because of its extrafoveal location, PPCNV often produces minimal or no symptoms. However, if left untreated, the natural history of PPCNV is unfavourable, with up to 25% of patients deteriorating to best-corrected visual acuity (BCVA) of 20/500 or worse after 3 years [1]. Treatment options for PPCNV include laser photocoagulation, photodynamic therapy, surgery and anti-vascular endothelial growth factor (VEGF) intravitreal injections [1, 3]. Previous studies obtained favourable results with anti-VEGF injections, with up to 85% of eyes achieving resolution of subretinal fluid, and improvement in visual acuity. However, most investigated PPCNV of different aetiologies, did not use bevacizumab, and had a relatively small sample size or follow-up [4–9]. In this study, we present 67 eyes of 66 patients with age-related macular degeneration (AMD) PPCNV in a retinal clinic in a public, tertiary-care hospital. We report our experience with AMD-related PPCNV treated with anti-VEGF injections in real-life circumstances over a long follow-up period.
To the best of our knowledge this is the largest cohort of AMD-related PPCNV with the longest follow-up time reported so far.
Materials and methods
All data for the study were collected and analysed in accordance with the policies of the Institutional Review Board of Meir Medical Centre, and the tenets of the Declaration of Helsinki. This study was approved by the Institutional Review Board of Meir Medical Centre.
Study participants
We reviewed the clinical records of consecutive patients with AMD-related PPCNV who visited our retinal clinic from January 2003 to May 2020.
Inclusion criteria were treatment-naïve patients with AMD-related PPCNV located around the optic nerve (excluding the nasal side) treated with intravitreal anti-VEGF injections, and with at least 6 months of follow-up. Exclusion criteria were myopia (more than 6 dioptres), younger than 50 years, polypoidal choroidal vasculopathy, no or unreadable optical coherence tomography (OCT) and fluorescein or indocyanine green angiography (FA/ICG) imaging, and patients treated with photodynamic therapy.
Data collection
Each patient routinely underwent Snellen chart BCVA measurements and a complete ophthalmic exam as part of routine clinical care. Retinal OCT imaging (Stratus OCT, Carl Zeiss, USA or Heidelberg OCT, Heidelberg, Germany) was performed at baseline and at each follow-up visit. FA and ICG were performed at baseline.
Patient demographics, Snellen chart BCVA [converted to logarithm of minimal angle of resolution (logMAR)], slit lamp examination, OCT findings and type of treatment given were extracted from the medical records. We defined OCT findings of subretinal fluid (SRF), intraretinal fluid (IRF), pigment epithelial detachment (PED), and subretinal hyperreflective material (SHRM) as in previous studies by our group [10, 11].
Anti-VEGF injections
The protocol of treatments in our practice was to start with 3 consecutive monthly intravitreal injections of bevacizumab 1.25 mg/0.05 ml and continue with administration pro re nata [12]. The patients were evaluated 1 month after the third bevacizumab injection, and in case of no response to treatment the patients were switched to ranibizumab 0.5 mg/0.05 ml (Lucentis, Novartis) or aflibercept 2 mg/0.05 ml (Eylea, Bayer). All intravitreal injections were performed with a 30-gauge needle through the pars plana under aseptic conditions and local anaesthesia.
Disease activity, inactivity and recurrence
PPCNV activity was defined based on the findings of dilated fundoscopic examination, new onset of retinal haemorrhage, presence of IRF, SRF or SHRM in OCT imaging, and/or leakage in FA or ICG. In case of signs of PPCNV activity, patients received anti-VEGF injections. No response to treatment was considered as non-improvement of these signs despite receiving anti-VEGF treatment. Recurrence was defined as signs of disease activity in eyes that previously had complete resolution of these signs. We calculated disease inactivity time as the period of follow-up in which no activity was observed clinically or on imaging, and during which the patient was not given any treatment.
To compare patients with different follow-up periods, an inactivity index was calculated as the proportion of inactivity from the total follow-up time. Inactivity index = Inactivity time/Total follow-up time.
The primary outcome for this retrospective study was BCVA at 1 month after the first 3 bevacizumab injections. Secondary outcomes were BCVA at 6, 12, 24, 36, 48, 60 months after baseline and at the final visit, time to recurrence after the first inactivation and inactivity index.
Statistical analysis
All data collected in the study were entered into a Microsoft Excel 2007 worksheet (Microsoft Corp., Redmond, WA). Statistical analyses were performed using SPSS-25 (IBM, Armonk, NY, USA). Results are expressed as mean ± SD, median (range) or N (%). Pearson correlations were used to find relations between baseline demographics and OCT measurements and delta BCVA. Baseline OCT characteristics were compared with the same parameters measured later using paired t-test or Wilcoxon test for nonparametric variables, as appropriate. Chi-square or Fisher’s exact test were used as indicated, for analysis of categorical variables. One-way analysis of variance was used to compare multiple group averages. Linear regression was used to predict final BCVA and Inactivity Index. Kaplan–Meier and Cox Regression, each when appropriate, were used to compare time curves in two or more groups, analysing the effect of several risk-factors on time-to-recurrence. p-values <0.05 were considered statistically significant and represent results for two-sided tests.
Results
We identified 101 eyes in 100 patients with AMD-related PPCNV in our records. Fifteen eyes were excluded due to PPCNV located nasally to the optic nerve, 17 eyes were excluded due to low quality of the retinal imaging or incomplete documentation, and 2 eyes were excluded due to insufficient follow-up time.
A total of 67 eyes of 66 patients with PPCNV were included in the study. The mean age of the patients was 80.5 ± 7.6 years and 58% were women. Mean duration of follow up was 53 months (range 7.5–131 months) (Table 1). The BCVA (mean ± SD, logMAR) at baseline and one month after the third bevacizumab injection were 0.53 ± 0.54 (Snellen ≈ 20/70) and 0.43 ± 0.41 (≈20/53), respectively (p = 0.27). At the final follow-up examination, the BCVA was significantly decreased from baseline [0.72 ± 0.59 (Snellen ≈ 20/100), p < 0.01]. There was a statistically significant deterioration in BCVA with respect to baseline beginning in the fourth year of follow-up (Table 1).
Table 1.
General characteristics of the study participants*.
| Variable | |
|---|---|
| Age (Years, median (range)) | 82 (57–96) |
| Sex (Female, n (%)) | 39 (58.2) |
| Eye (Right, n (%)) | 36 (53.7) |
| BCVA (LogMAR, mean ± SD (Snellen))** | |
| Baseline (n = 67) | 0.53 ± 0.55 (≈20/70) |
| After 3 injections (n = 67) | 0.43 ± 0.41 (≈20/53) |
| 6 Months (n = 67) | 0.46 ± 0.47 (≈20/60) |
| 12 Months (n = 62) | 0.53 ± 0.6 (≈20/70) |
| 24 Months (n = 52) | 0.52 ± 0.51 (≈20/65) |
| 36 Months (n = 35) | 0.52 ± 0.45 (≈20/65) |
| 48 Months (n = 30) | 0.63 ± 0.55 (≈20/85)a |
| 60 Months (n = 24) | 0.62 ± 0.51 (≈20/85)a |
| Final visit (n = 67) | 0.72 ± 0.59 (≈20/100)a |
| Follow-up time (Months, median (range)) | 44 (7.5–131.8) |
| Anti-VEGF injections (N, median (Range)) | 14 (3–74) |
*n = 67 unless specified otherwise.
**All times were compared against visual acuity at baseline with Mann–Whitney test.
ap < 0.01.
BCVA best-corrected visual acuity, LogMAR logarithm of the minimum angle of resolution, VEGF vascular endothelial growth factor.
Among the 67 eyes included in the study, 59 (88%) reached disease inactivity during follow-up, PPCNV became inactive in 29 eyes (49%) after the first treatment cycle of 3 bevacizumab injections. Twenty eyes were switched to ranibizumab during follow-up; 11 eyes were switched to aflibercept. The mean inactivity index was 0.38 ± 0.23. The presence of SRF at presentation was associated with significantly lower Inactivity Index (p < 0.05 ).
Of the eyes that reached disease inactivity, 50 (85%) had a recurrence during follow-up (Fig. 1). The mean time to recurrence was 9 ± 18 months, while 35 eyes had recurrence less than 3 months after the first inactivation (Estimated probability of recurrence was 0.59). This subgroup of patients had male preponderance (p < 0.01), and was significantly younger than patients with longer inactivation [median age 79 (range 57–89) vs. 84 (range 69–96) years, p < 0.05].
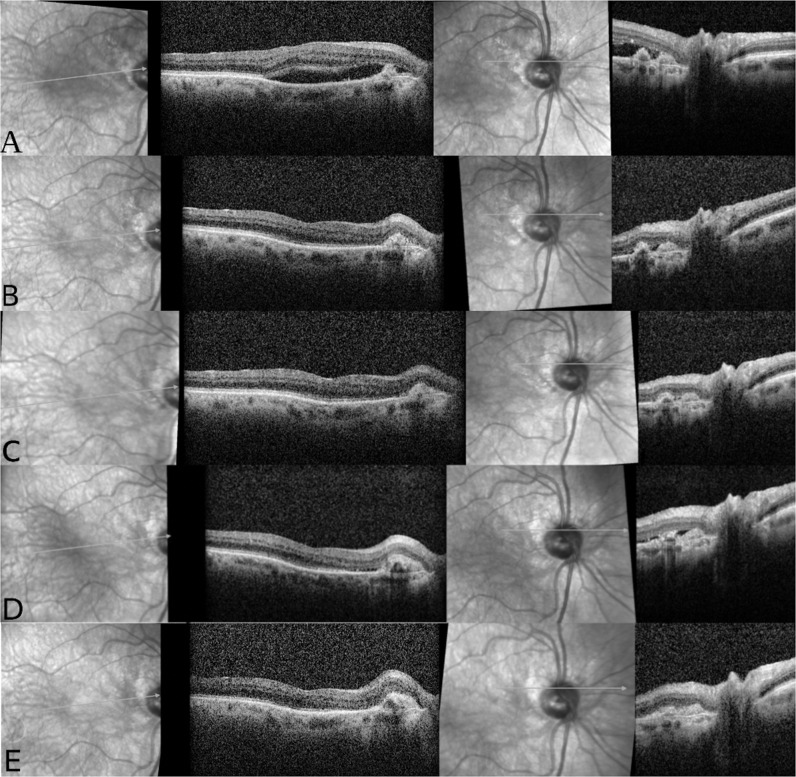
Fig. 1. Sequential OCT imaging of the right eye of an 88-year-old man with AMD related PPCNV.
A Anti-VEGF (Bevacizumab) therapy was initiated due to SRF involving the fovea on 02/2017. B After 3 monthly injections of intravitreal bevacizumab, the retina was completely dry. C 13 months follow-up – retina is completely dry, PEDs nasal to the macula. D Appearance of sub-retinal fluid adjacent to the nasal macular PEDs, the patient was referred to 3 monthly injections of intravitreal bevacizumab. E After 26 months of follow up the retina is completely dry. OCT optical coherence tomography; AMD age-related macular degeneration; PPCNV peripapillary choroidal neovascularisation; VEGF vascular endothelial growth factor; SRF subretinal fluid, PED pigment epithelium detachment.
To investigate the effect of duration of initial inactivity to recurrence, Cox regression analysis was used to determine the effect of several variables, including demographics, OCT, and fundoscopic parameters at presentation. Kaplan–Meier survival analysis demonstrated that the variables age <80 years and male sex were associated with a shorter time to recurrence (Figs. 2 and 3). In univariate analysis, sex (p = 0.011), age (p = 0.048), presence of SRF and pigment epithelial detachment (PED, p = 0.006), and BCVA at baseline (p = 0.04) were statistically significant for recurrence. In multivariate Cox analysis, sex, combined SRF and PED, and BCVA at baseline were significant predictors of recurrence. Briefly, worse BCVA, combined SRF and PED, and male sex were associated with a higher probability of recurrence after the initial inactivation period. Univariate linear regression was used to investigate the effect of various variables on final BCVA. Multivariate linear regression showed that baseline BCVA was the only significant predictor (p < 0.001) of the BCVA at the final follow-up visit. Thus, a better baseline BCVA predicted a better BCVA at the final visit. There were no statistically significant differences in demographic or OCT parameters and BCVA at baseline and during follow-up between the 6 eyes (8%) that were treated with laser photocoagulation and those that were not. Univariate linear regression found significant correlations between the Inactivity Index and baseline BCVA, SRF, and PED (isolated and combined). In multivariate analysis, SRF at presentation was the only significant variable to predict a lower Inactivity Index (p = 0.004). Briefly, eyes with SRF at presentation were associated with a higher treatment burden and refractory disease.
Fig. 2.

Kaplan–Meier curve for the survival time to recurrence after inactivation in patients above 80 years of age and below it.
Fig. 3.

Kaplan–Meier curve for the survival time to recurrence after inactivation in males and females.
Discussion
This study describes our experience with 67 eyes with AMD-related PPCNV treated with anti-VEGF agents in real-life settings. The mean follow-up time was 53 months. To the best of our knowledge this is the largest retrospective cohort study of AMD-related PPCNV.
The mean BCVA after the first three bevacizumab injections and the mean BCVA at 6 months were better than baseline, but this did not reach statistical significance. An initial improvement of BCVA after treatment with anti-VEGF agents was previously described [5, 9]. An analysis of the change in average BCVA in 12-month intervals revealed preservation of vision during the first few years of follow-up, and significant deterioration in BCVA with respect to baseline beginning in the fourth year (Table 1). This is the first study to report that after 4 or more years of follow-up, the mean BCVA deteriorated significantly. Our results are in contrast with a study by Lin et al. [9] who reported a cohort of 23 eyes with AMD-related PPCNV treated with anti-VEGF agents, and found no statistically significant change at the end of follow-up. Perhaps the reason was the relatively small cohort. Also, in our study the follow-up time was recorded from the initial treatment and thereafter. In the study by Lin et al., follow-up commenced from the initial diagnosis, and treatment began after an estimated average of 16 months; only nine eyes received treatment from the beginning of follow-up. Therefore, the actual follow-up time is not similar.
Our results agree with those of long-term studies of sub-foveal AMD treated with anti-VEGF agents in real-world settings [13–15]. In a multivariate linear regression model, we found that baseline BCVA was the only significant predictor of final BCVA. The only study similar to ours that included long follow-up was that of Lin et al., but in that study results of regression models regarding final BCVA were not reported.
To gain insight into the factors associated with response to treatment, and compare eyes with different follow-up periods, we created an Inactivity Index for each eye in the study. We found a significant association between SRF at presentation and a lower inactivity index that remained significant in multivariate regression. The meaning of this finding is that eyes with SRF at presentation had a more refractory disease, with persistent signs of PPCNV activity.
The estimated probability of recurrence at 3 months after the first inactivation was 0.59, which is lower than the estimated probability of recurrence found by Lin et al [9]. We found that male sex was associated with a shorter time to recurrence (HR = 2.08, p < 0.05; Fig. 3). After reviewing the current literature regarding PPCNV, we believe this is the first time that patient sex was correlated with a shorter time to recurrence in eyes with PPCNV. It seems that male sex is a strong predictor of earlier recurrence in AMD-related PPCNV. Thus, men could benefit from closer follow-up and a more intense treatment protocol. This relationship was also demonstrated by Kuroda et al. [16] for macular neovascular AMD. Therefore, it would be interesting to know the sex distribution of the patients with early recurrence in the study of Lin et al. and see whether the higher probability of recurrence in their study is associated with a higher proportion of male patients.
Younger patients were found to have a shorter time to recurrence (Fig. 2). This finding could indicate that younger patients with AMD-related PPCNV have more aggressive CNVM. Reviewing the current literature, we did not find information regarding this point for PPCNV. The current literature dealing with macular neovascular AMD has conflicting findings regarding patient age and response to anti-VEGF treatment. Ersoy et al. [17] reported that younger patient age was associated with non-response to anti-VEGF treatment in neovascular AMD. In contrast, other studies on neovascular AMD reported an association between older age and the probability of recurrence [16, 18]. Other baseline patient factors associated with recurrence were combined SRF and PED on OCT imaging and worse initial BCVA. We did not find a similar association between these factors and PPCNV recurrence in the literature. However, our findings are supported by studies conducted in eyes with macular neovascular AMD. Ersoy et al. [17] showed that SRF and PED had a poor response to treatment with anti-VEGF agents, and Golbaz et al. [19] demonstrated that eyes with SRF were more prone to recurrence. These findings suggest a more aggressive disease attributed to SRF, and are also consistent with our finding of a lower Inactivity Index associated with SRF.
Previous studies on PPCNV pathophysiology demonstrated that the majority of PPCNV grew through breaks in Bruch’s membrane in a similar manner to macular neovascular AMD. It is important to mention, however, that a minority of PPCNVs originate from choroidal vessels that grow around the peripapillary border of Bruch’s membrane [3, 20]. These occurrences, however, are usually located on the nasal side of the optic nerve, and are usually small and asymptomatic [20].
This study had several limitations, the first of which is its retrospective design. Second, our data included OCT imaging performed with two different OCT machines, including time domain OCT. Therefore, we did not include quantitative parameters such as central macular thickness and SRF dimensions. Third, the use of different anti-VEGF agents was according to the treating physician’s discretion, and as often happens in real-world studies, the decision to switch could have been affected by the cost of the anti-VEGF agent and insurance status of the patient. Fourth, we used ‘final BCVA’ as one of the outcomes of our study. We are aware that ‘final BCVA’ is not an optimal outcome in a cohort of patients with different follow-up periods. However, the median follow-up time of our cohort was 44 months, and we decided to include it in our analyses in a similar manner to other previously published papers on PPCNV [9, 21, 22].
In summary, to the best of our knowledge our study describes the largest cohort so far of patients with AMD-related PPCNV using real-world data. Our study is unique for its long average follow-up time for this population. Male sex and a younger patient age were associated with a shorter time to recurrence of AMD-related PPCNV after disease inactivation. Moreover, the presence of SRF at presentation was associated with a lower inactivity index and might require a more aggressive treatment strategy. Since we showed that after four years of follow-up, AMD-related PPCNV had a deleterious effect on BCVA despite PRN treatment with anti-VEGF agents, future studies should investigate the effect of other treatment protocols such as treat-and-extend on BCVA and PPCNV progression.
Summary Table
What was known before
AMD related PPCNV responds to treatment with anti-VEGF agents.
The BCVA of patients with AMD related PPCNV treated with anti-VEGF agents remains stable for the first two years of follow-up.
What this study adds
The BCVA of patients with AMD related PPCNV treated with anti-VEGF agents remains stable for 4 years and deteriorates thereafter.
Younger men with AMD related PPCNV are more likely to have a shorter time to recurrence after disease inactivation.
This is the largest cohort of eyes with AMD related PPCNV, approximately three times larger than the previously largest cohort, with the longest follow-up duration so far.
Author contributions
NS was responsible for the data collection and analysis and wrote the manuscript. AF was responsible for data analysis and critical review of the manuscript. EG was responsible for data collection and drafting the manuscript. AN, YK, AO, GR, AYN, NG were also responsible for parts of data collection and critical review of the manuscript. OS was responsible for data collection, drafting and critical revision of the manuscript. All authors contributed to conceptualisation and approved the final version of the manuscript.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Jutley G, Jutley G, Tah V, Lindfield D, Menon G. Treating peripapillary choroidal neovascular membranes: a review of the evidence. Eye. 2011;25:675–81. doi: 10.1038/eye.2011.24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Wilde C, Poostchi A, Mehta RL, Hillman JG, MacNab HK, Messina M, et al. Prevalence of peripapillary choroidal neovascular membranes (PPCNV) in an elderly UK population—the Bridlington eye assessment project (BEAP): a cross-sectional study (2002–2006) Eye. 2019;33:451–8. doi: 10.1038/s41433-018-0232-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lopez PF, Green WR. Peripapillary subretinal neovascularization. A review. Retina. 1992;12:147–71. doi: 10.1097/00006982-199212020-00012. [DOI] [PubMed] [Google Scholar]
- 4.Davis AS, Folk JC, Russell SR, Sohn EH, Boldt HC, Stone EM et al. Intravitreal bevacizumab for peripapillary choroidal neovascular membranes. Arch Ophthalmol. 2012. 10.1001/archophthalmol.2012.465. [DOI] [PubMed]
- 5.Singh SR, Fung AT, Fraser-Bell S, Lupidi M, Mohan S, Gabrielle PH, et al. One-year outcomes of anti-vascular endothelial growth factor therapy in peripapillary choroidal neovascularisation. Br J Ophthalmol. 2020;104:678–83. doi: 10.1136/bjophthalmol-2019-314542. [DOI] [PubMed] [Google Scholar]
- 6.Saffra NA, Reinherz BJ. Peripapillary choroidal neovascularization associated with optic nerve head drusen treated with anti-VEGF agents. Case Rep. Ophthalmol. 2015;6:51–55. doi: 10.1159/000375480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Anastasilakis K, Symeonidis C, Kaprinis K, Mataftsi A, Tzamalis A, Dimitrakos SA. Peripapillary neovascular membrane in a young pregnant woman and prompt response to ranibizumab injections following uneventful delivery. Case Rep Ophthalmol. 2011;2:129–33. doi: 10.1159/000328385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hamoudi H, Sørensen TL. Effect of intravitreal ranibizumab in the treatment of peripapillary choroidal neovascularisation. J Ophthalmol. 2011. 10.1155/2011/602729. [DOI] [PMC free article] [PubMed]
- 9.Lin T, Dans K, Meshi A, Muftuoglu IK, Amador-Patarroyo MJ, Chen KC, et al. Age-related macular degeneration–associated peripapillary choroidal neovascularization in the era of anti–vascular endothelial growth factor therapy. Retina. 2019;39:1936–44. doi: 10.1097/IAE.0000000000002272. [DOI] [PubMed] [Google Scholar]
- 10.Segal O, Barayev E, Nemet AY, Mimouni M. Predicting response of exudative age-related macular degeneration to Bevacizumab based on spectralis optical coherence tomography. Retina. 2016;36:259–63. doi: 10.1097/IAE.0000000000000690. [DOI] [PubMed] [Google Scholar]
- 11.Pokroy R, Mimouni M, Barayev E, Segev F, Geffen N, Nemet AY, et al. Prognostic value of subretinal hyperreflective material in neovascular age-related macular degeneration treated with Bevacizumab. Retina. 2018;38:1485–91. doi: 10.1097/IAE.0000000000001748. [DOI] [PubMed] [Google Scholar]
- 12.Melamud A, Stinnett S, Fekrat S. Treatment of neovascular age-related macular degeneration with intravitreal bevacizumab: efficacy of three consecutive monthly injections. Am J Ophthalmol. 2008. 10.1016/j.ajo.2008.03.014. [DOI] [PubMed]
- 13.Holz FG, Tadayoni R, Beatty S, Berger A, Cereda MG, Cortez R, et al. Multi-country real-life experience of anti-vascular endothelial growth factor therapy for wet age-related macular degeneration. Br J Ophthalmol. 2015;99:220–6. doi: 10.1136/bjophthalmol-2014-305327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Khanani AM, Skelly A, Bezlyak V, Griner R, Torres LR, Sagkriotis A. SIERRA-AMD: A retrospective, real-world evidence study of patients with neovascular age-related macular degeneration in the United States. Ophthalmol Retin. 2020;4:122–33. doi: 10.1016/j.oret.2019.09.009. [DOI] [PubMed] [Google Scholar]
- 15.Singer MA, Awh CC, Sadda S, Freeman WR, Antoszyk AN, Wong P, et al. HORIZON: An open-label extension trial of ranibizumab for choroidal neovascularization secondary to age-related macular degeneration. Ophthalmology. 2012;119:1175–83. doi: 10.1016/j.ophtha.2011.12.016. [DOI] [PubMed] [Google Scholar]
- 16.Kuroda Y, Yamashiro K, Miyake M, Yoshikawa M, Nakanishi H, Oishi A, et al. Factors associated with recurrence of age-related macular degeneration after anti-vascular endothelial growth factor treatment: a retrospective cohort study. Ophthalmology. 2015;122:2303–10. doi: 10.1016/j.ophtha.2015.06.053. [DOI] [PubMed] [Google Scholar]
- 17.Ersoy L, Ristau T, Kirchhof B, Liakopoulos S. Response to anti-VEGF therapy in patients with subretinal fluid and pigment epithelial detachment on spectral-domain optical coherence tomography. Graefe’s Arch Clin Exp Ophthalmol. 2014;252:889–97. doi: 10.1007/s00417-013-2519-9. [DOI] [PubMed] [Google Scholar]
- 18.Kikushima W, Sakurada Y, Yoneyama S, Sugiyama A, Tanabe N, Kume A, et al. Incidence and risk factors of retreatment after three-monthly aflibercept therapy for exudative age-related macular degeneration. Sci Rep. 2017;7:1–8. doi: 10.1038/srep44020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Golbaz I, Ahlers C, Stock G, Schütze C, Schriefl S, Schlanitz F, et al. Quantification of the therapeutic response of intraretinal, subretinal, and subpigment epithelial compartments in exudative AMD during anti-VEGF therapy. Investig Ophthalmol Vis Sci. 2011;52:1599–605. doi: 10.1167/iovs.09-5018. [DOI] [PubMed] [Google Scholar]
- 20.Sarks SH. New vessel formation beneath the retinal pigment epithelium in senile eyes. Br J Ophthalmol. 1973;57:951–65. doi: 10.1136/bjo.57.12.951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Figueroa MS, Noval S, Contreras I. Treatment of peripapillary choroidal neovascular membranes with intravitreal bevacizumab. Br J Ophthalmol. 2008;92:1244–7. doi: 10.1136/bjo.2008.144196. [DOI] [PubMed] [Google Scholar]
- 22.Adrean SD, Grant S.Chaili S, Bevacizumab (Avastin) and thermal laser combination therapy for peripapillary choroidal neovascular membranes. J Ophthalmol. 2017: 10.1155/2017/4802690. [DOI] [PMC free article] [PubMed]