Abstract
The prevalence of obesity has been increasing worldwide, and its pathogenesis is closely related to preadipo-cyte differentiation. Because the presence of obesity increases the risk of chronic disease, it is important to decrease exces-sive body fat accumulation. This study aimed to demonstrate the anti-adipogenesis and anti-obesity effects of gongmi tea and gongmi so extract. The 3T3-L1 preadipocyte cell line was stained with Oil red O, and the expression levels of peroxi-some proliferator-activated receptor-γ (PPARγ), adiponectin, and fatty acid-binding protein 4 (FABP4) were evaluated via Western blot analysis. A mouse model of obesity was developed by feeding C57BL/6 male mice a high-fat diet (HFD). Gongmi tea or gongmi so extract was orally administered at a dose of 200 mg/kg for 6 weeks. The mouse body weight was measured weekly during the study period, and the epididymal adipose tissue weight and blood serum were analyzed at the end of the study period. The gongmi tea and gongmi so extract did not exhibit toxicity in mice. Oil red O staining showed that gongmi tea significantly decreased excessive body fat accumulation. In addition, gongmi tea (300 μg/mL) significantly downregulated adipogenic transcription factors, such as PPARγ, adiponectin, and FABP4. In vivo tests indicated that oral administration of gongmi tea or gongmi so extract to C57BL/6 mice with HFD-induced obesity effectively decreased their body weight and epididymal adipose tissue. Gongmi tea and gongmi so extract have potent in vitro anti-adipogenic effects in 3T3-L1 cells and in vivo anti-obesity effects in mice with HFD-induced obesity.
Keywords: herb medicine, high-fat diet, metabolic syndrome, obesity
INTRODUCTION
Obesity is a result of energy imbalance caused by changes in diet and physical activity as well as excessive body fat accumulation (Kang et al., 2016; Chen and Lin, 2021). Previous studies have demonstrated that obesity is associated with an increased risk of cardiovascular disease, type 2 diabetes, hypertension, and fatty liver disease as well as increased mortality (Hotamisligil, 2006; Saltiel and Olefsky, 2017). Lipid accumulation is caused by an increase in triglyceride production and adipocyte differentiation during adipogenesis (Kim et al., 2017). This lipid accumulation process controls the lipid droplet size as the cell number or proliferation increases (Lee and Kang, 2021). In vitro studies of adipogenesis frequently use the mouse embryonic fibroblast 3T3-L1 preadipocyte cell line (Nam et al., 2019). This cell line can be stimulated using 3-isobutyl-1-methylxanthine (IBMX), dexamethasone (Dex), and insulin to differentiate into adipocyte-like cells. Such stimulants activate complex signaling pathways and various transcription factors (Seo et al., 2018; Choi et al., 2020). The C57BL/6 mouse model of high-fat diet (HFD)-induced obesity is widely used for in vivo studies, and the anti-obesity effect can be confirmed by measuring the body and organ weights. The mechanism underlying adipogenesis is regulated by the expression of adipogenic transcription factors, such as CCAAT/enhancer-binding protein (C/EBP)α, C/EBPβ, nuclear receptor peroxisome proliferator-activated receptor-γ (PPARγ), and sterol regulatory element-binding transcription factor 1 (SREBP-1) (Moseti et al., 2016; Seo et al., 2018; Kang et al., 2021). The expression of PPARγ is activated by adipogenic genes such as C/EBPα and SREBP-1 in the early stages of adipocyte proliferation and is involved in promoting and regulating adipogenesis (Kim et al., 2017; Li et al., 2020). Based on these findings, many studies have been conducted with a focus on adipogenesis to control obesity (Moseti et al., 2016; Gire et al., 2021; Papathanasiou et al., 2021).
Gongmi tea is a water extract made of 11 herbs (Table 1), whereas the gongmi so extract contains 14 herbs (Table 2). The dried and mature pericarp of Citrus unshiu Marco. exhibits anti-hepatic injury (Wu et al., 2021) and antioxidant (Wang et al., 2020) effects. In addition, Phyllostachys spp. and Zea mays L. exert antioxidant and antimicrobial effects (D’Amato et al., 2019; Mullen et al., 2019; Xiao et al., 2020). Poria cocos (Fr.) Wolf, Atractylodes ovata (Thunb.) DC, Cinnamomum cassia Presl, and Astragalus membranaceus Bunge var. membranaceus are widely known for their anti-inflammatory properties (Gruenwald et al., 2010; Huang et al., 2014; Auyeung et al., 2016; Hossen et al., 2021; Bao et al., 2022). Furthermore, Rehmannia glutinosa (Gaertn.) Libosch. Ex Steud has anti-diabetic effects (Gong et al., 2019), and the alkaloids from Nelumbo nucifera Gaertner have anticancer effects (Manogaran et al., 2019). To date, gongmi tea and gongmi so extract are used to relieve swelling and constipation. However, their effect on the differentiation of 3T3-L1 cells and mice with HFD-induced obesity remains unclear.
Table 1.
Composition of gongmi tea
| Botanical name | Amount (g) |
|---|---|
| Phyllostachys spp. | 212 |
| Triticum aestivum L. | 160 |
| Rehmannia glutinosa (Gaertn.) Libosch. Ex Steud. | 120 |
| Zea mays L. | 120 |
| Poria cocos (Fr.) Wolf | 120 |
| Nelumbo nucifera Gaertner | 44 |
| Phragmites communis Trin. | 44 |
| Astragalus membranaceus Bunge var. membranaceus | 24 |
| Cinnamomum cassia Prel | 24 |
| Citrus unshiu Marcov. | 24 |
| Atractylodes ovata (Thunb.) DC | 24 |
| Total | 916 |
Table 2.
Composition of gongmi so extract
| Botanical name | Amount (g) |
|---|---|
| Hordeum vulgare L. | 485 |
| Atractylodes ovata (Thunb.) DC | 485 |
| Citrus unshiu Marcov. | 485 |
| Glycyrrhiza uralensis Fisch. | 320 |
| Syzygium aromaticum Merrill & Perry | 320 |
| Zingiber officinale Roscoe | 320 |
| Cinnamomum cassia Presl | 320 |
| Citrus aurantium var. daidai | 240 |
| Raphanus sativus L. | 240 |
| Amomum villosum Loureiro var. xanthioides T. L. Wu et Senjen | 160 |
| Poria cocos (Fr.) Wolf | 100 |
| Mentha canadensis L. | 100 |
| Foeniculum vulgare Mill. | 100 |
| Myristica fragrans Houtt. | 100 |
| Total | 3,775 |
In this study, we investigated the effects of gongmi tea and gongmi so extract on adipocyte differentiation and adipogenesis in 3T3-L1 cells. Furthermore, we evaluated their effects on mouse body weight, epididymal adipose tissue weight, and kidney and liver damage in a mouse model of HFD-induced obesity.
MATERIALS AND METHODS
Chemicals and reagents
Oil red O, Dex, insulin, IBMX, and SB203580 (a p38 inhibitor, a positive control) were purchased from Sigma-Aldrich Chemical Co.. Dulbecco’s modified Eagle’s medium (DMEM) and phosphate-buffered saline (PBS) were purchased from HiMedia Laboratories Pvt. Ltd.. Roswell Park Memorial Institute (RPMI) 1640 was purchased from Welgene. Penicillin-streptomycin and fetal bovine serum (FBS) were obtained from Thermo Fisher Scientific and 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxy-methoxyphen-yl)-2-(4-sulfophenyl)-2H-tetrazolium (MTS) reagent from Promega Corporation. Adiponectin, fatty acid-binding protein 4 (FABP4), PPARγ, and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) were obtained from Santa Cruz Biotechnology, Inc. and horseradish peroxidase (HRP)-conjugated secondary antibodies from ABclonal Technology.
Preparation of gongmi tea and gongmi so extract
Gongmi tea and gongmi so extract were prepared by mixing the 11 and 14 herbs listed in Table 1 and 2, respectively, in the appropriate ratios. The two mixtures were extracted once using a 10-fold water solvent at 100°C for 4 h and then filtered. The gongmi tea extract solution and gongmi so extract were concentrated using a continuous vacuum evaporator at around 40°C and 600 mmHg.
Cell experiments
The 3T3-L1 preadipocyte cell line was purchased from the American Type Culture Collection. The cells were cultured in DMEM containing 10% FBS and penicillin-streptomycin and then incubated under a 5% CO2 atmosphere at 37°C. Subsequently, the cells were seeded in 6-well plates to full confluence for 2 days for adipocyte differentiation and then cultured in IBMX, Dex, and insulin (MDI) differentiation medium (0.5 mM IBMX, 1 μM Dex, and 10 μg/mL insulin). After 2 days, the medium was replaced with DMEM containing 10 μg/mL insulin. On day 8, the differentiation was completed (Fig. 1).
Fig. 1.
Schematic of 3T3-L1 preadipocyte differentiation. Fully cultured 3T3-L1 cells were treated with 0.5 mM 3-isobutyl-1-methylxanthine, 1 μM dexamethasone, and 10 μg/mL insulin (MDI) for 2 days. The cell culture medium was changed with new 10 μg/mL of insulin-containing Dulbecco’s modified Eagle’s medium every 2 days. To investigate the effects of gongmi tea or gongmi so extract on adipogenesis, differentiated 3T3-L1 cells were treated every 2 days. After 8 days of differentiation, Oil red O staining and Western blot analysis were conducted.
Cell viability
Mouse splenocytes were prepared as described previously (Park et al., 2018). Briefly, BALB/c female mice were sacrificed via cervical dislocation. From the isolated mouse spleen, a single-cell suspension was prepared using a 50 μm mesh strainer (BD Falcon). To remove red blood cells, the cell pellet was incubated in red blood cell lysis buffer (BioLegend) at room temperature for 5 min and resuspended in RPMI 1640 media. Mouse splenocytes and 3T3-L1 cells were seeded in 96-well plates (SPL Life Sciences Co.) and treated with garcinia, gongmi tea, or gongmi so extract at different concentrations (100, 200, and 300 μg/mL) for 48 h. Cell proliferation was assayed using MTS reagent according to the manufacturer’s protocols. Absorbance at 490 nm was measured using an enzyme-linked immunosorbent assay (ELISA) microplate reader (ELx808, BioTek Instruments Inc.).
Oil red O staining
First, 3T3-L1 cells seeded in 6-well plates (SPL Life Sciences Co.) were cultured with gongmi tea or gongmi so extract at different concentrations (100 and 300 μg/mL). The culture media were changed with fresh media every 2 days. On day 8 of differentiation, the 3T3-L1 cells were washed twice with PBS and then fixed with 10% formalin for 1 h. The adipocytes were stained with Oil red O in the dark for 1 h. The stained lipid droplets were dissolved in 100% isopropanol, and absorbance at 490 nm was measured using an ELISA microplate reader. Visualization images were obtained using light microscopy and photography (DS-Ri2 microscopy camera, Nikon Corp.).
Western blot analysis
First, 3T3-L1 cells seeded in 6-well plates were treated with MDI differentiation medium and gongmi tea or gongmi so extract at different concentrations (100 and 300 μg/mL). The cell supernatant was changed to insulin-containing medium every 2 days. SB203580 (10 μM) and garcinia (200 μg/mL) were used as positive controls. Using cell culture lysis buffer (Promega Corp.), differentiated 3T3-L1 cells were harvested. Subsequently, equal amounts of protein lysates resuspended in the sample buffer were boiled at 100°C for 10 min and electrophoresed using 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis and then transferred to polyvinylidene difluoride (PVDF) membranes (Millipore). The PVDF membranes were incubated with primary antibodies against adiponectin, FABP4, PPARγ, and GAPDH at 4°C overnight, washed, and then incubated with HRP-conjugated anti-rabbit or anti-mouse immunoglobulin secondary antibodies. With the use of Western BrightTM electrochemiluminescence reagents and a C300 chemiluminescence imager (Azure Biosystems Inc.), protein bands were detected.
Animal experiments
We followed the guidelines of the Animal Care and Use Committee of Chonnam National University (no. CNU IACUC-YB-2021-152) in all animal procedures. Five-week-old C57BL/6 male mice (Damool Science) were maintained in an animal room with controlled temperature and a 12/12-h light/dark cycle. After a 1-week acclimation, the mice were randomly divided into five groups. The control group (n=6) was fed a chow diet (DBL Co., Ltd.), whereas the HFD group (n=24) was fed a diet with 60% kcal fat (Research Diets Inc.). The other three groups were orally administered with garcinia, gongmi tea, and gongmi so extract at a dose of 200 mg/kg every day to HFD group. At the end of the experiment, all mice were euthanized using isoflurane. To determine kidney and liver damage, serum was collected from the heart and analyzed using a serological analyzer (Fujifilm Corp.). The serum uric acid (UA), creatinine (CRE), and blood urea nitrogen (BUN) levels were determined to evaluate nephrotoxicity, whereas the serum glutamic oxaloacetic transaminase (GOT) and glutamic pyruvic transaminase (GPT) levels were determined to evaluate hepatotoxicity.
Statistical analysis
All data were acquired in quadruplicate, and the experiments were repeated at least three times. The results obtained from the representative experiments are expressed as mean±standard error of the mean. Statistical differences were evaluated via one-way analysis of variance for multigroup comparisons, followed by Tukey’s post hoc test; P<0.05 was considered statistically significant.
RESULTS
Safety of gongmi tea and gongmi so extract
The effects of gongmi tea and gongmi so extract on cell viability were evaluated via MTS assay. Briefly, mice splenocytes and 3T3-L1 cells were treated with gongmi tea or gongmi so extract for 48 h. Gongmi tea and gongmi so extract at concentrations of 100, 200, and 300 μg/mL did not exhibit any cytotoxicity on splenocytes (Fig. 2A) and 3T3-L1 cells (Fig. 2B and 2C) without or with MDI.
Fig. 2.
Effects of gongmi tea and gongmi so extract on cell viability. (A) Mouse splenocytes, (B) 3T3-L1 preadipocytes, and (C) MDI-treated 3T3-L1 preadipocytes were seeded in 96-well plates. The cells were treated with garcinia, gongmi tea, or gongmi so extract (100, 200, and 300 μg/mL) for 48 h. Cell viability was tested via MTS assay, and absorbance was measured at 490 nm. Gongmi tea and gongmi so extract did not exhibit cytotoxicity at concentrations of 100, 200, and 300 μg/mL. Data are expressed as mean±standard error of the mean. MDI, 3-isobutyl-1-methylxanthine, dexamethasone, and insulin; MTS, 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxy-methoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium.
Comparative effects of gongmi tea and gongmi so extract on adipogenesis inhibition
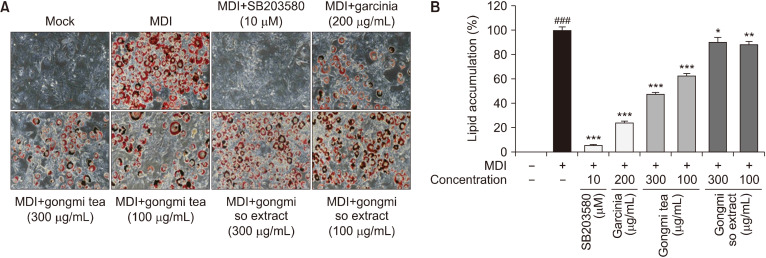
Briefly, 3T3-L1 cells treated with gongmi tea or gongmi so extract were incubated with MDI. Oil red O staining was used to determine the extent of adipocyte differentiation. The number and size of differentiated adipocytes increased after the MDI treatment, and gongmi tea significantly decreased lipid accumulation in adipocytes (Fig. 3A).
Fig. 3.
Effects of gongmi tea and gongmi so extract on lipid accumulation in adipocytes. The 3T3-L1 cell line was seeded in 6-well plates and grown to full confluence for 2 days. Then, the cells were treated with gongmi tea or gongmi so extract and differentiated into adipocytes via the MDI treatment. The differentiated adipocytes were treated with gongmi tea or gongmi so extract (100 and 300 μg/mL), and SB203580 and garcinia were used as positive controls. (A) Intracellular lipids were stained with Oil red O and then quantified. The administration of gongmi tea and gongmi so extract decreased lipid accumulation. (B) Stained lipid droplets were dissolved in 100% isopropanol, and absorbance was measured at 490 nm. Data are expressed as mean±standard error of the mean. ###P<0.001 compared with the non-treated group. *P<0.05, **P<0.01, and ***P<0.001 compared with the MDI-treated group. MDI, 3-isobutyl-1-methylxanthine, dexamethasone, and insulin.
SB203580 and garcinia were used as positive controls. Lipid droplets were dissolved in 100% isopropanol. To quantify intracellular lipid accumulation, absorbance of the dissolved lipid droplets was measured at 490 nm. Gongmi tea at concentrations of 100 and 300 μg/mL decreased lipid accumulation by 40% and 50%, respectively (Fig. 3B). SB203580 and garcinia significantly inhibited adipogenesis in 3T3-L1 cells.
Effects of gongmi tea and gongmi so extract on the protein expression of adipogenic transcription factors
The activation of adipogenic transcription factors, such as PPARγ, adiponectin, and FABP4, led to adipogenesis. To investigate the mechanism underlying adipogenesis inhibition, the protein expressions of PPARγ, adiponectin, and FABP4 were detected via Western blot analysis. Gongmi tea at a concentration of 300 μg/mL significantly decreased the expressions of PPARγ, adiponectin, and FABP4 (Fig. 4). On the other hand, gongmi so extract at concentrations of 100 and 300 μg/mL slightly decreased the expressions of PPARγ and adiponectin.
Fig. 4.
Effects of gongmi tea and gongmi so extract on the protein expression of adipogenic transcription factors. The 3T3-L1 cell line was seeded in 6-well plates and grown to full confluence for 2 days. Adipocytes were treated with gongmi tea or gongmi so extract during differentiation. SB203580 and garcinia were used as positive controls. Cell lysates were isolated, and the protein expressions of peroxisome proliferator-activated receptor-γ (PPARγ), adiponectin, and fatty acid-binding protein 4 (FABP4) were determined via Western blot analysis. Glyceral-dehyde 3-phosphate dehydrogenase (GAPDH) was used as a loading control. MDI, 3-isobutyl-1-methylxanthine, dexamethasone, and insulin.
Effects of gongmi tea and gongmi so extract on body weight and epididymal adipose tissue weight in C57BL/6 mice
The mouse body weights were measured weekly to determine the effect of gongmi tea or gongmi so extract on obesity (Fig. 5). A significant increase in the body weight of the HFD group was observed compared with that of the control group (Fig. 6A). Oral administration of 200 mg/kg of gongmi tea or gongmi so extract to the HFD group decreased weight gain by 10% and 4%, respectively. The garcinia group (200 mg/kg of garcinia used as a positive control) had the same body weight as the control group (Table 3). Furthermore, the epididymal adipose tissue mass was lower in the gongmi tea group than in the HFD group (Fig. 6B and 6C).
Fig. 5.
Experimental design of a mouse model with high-fat diet (HFD)-induced obesity. Five-week-old C57BL/6 male mice were divided into five groups. The control group was fed a chow diet (n=6), whereas the experimental group was fed an HFD (60% kcal, n=24). The mice were orally administered gongmi tea or gongmi so extract (200 mg/kg) for 6 weeks, and their body weight and food and water consumption were monitored every week. After the study period, the mice were euthanized for analysis.
Fig. 6.
Effects of oral administration of gongmi tea and gongmi so extract on body weight and epididymal adipose tissue weight in C57BL/6 mice fed high fat diet. Gongmi tea and gongmi so extract decreased adiposity in mice with high-fat diet (HFD)-induced obesity. (A) Body weight was measured once a week. (B, C) The epididymal adipose tissue was removed immediately and weighed. Data are expressed as mean±standard error of the mean (n=6). #P<0.05 and ##P<0.01 compared with the control group. *P<0.05 and ***P<0.001 compared with the HFD group.
Table 3.
Effects of oral administration of gongmi tea or gongmi so extract on body weight in C57BL/6 mice fed high-fat diet (unit: g)
| 1 week | 2 weeks | 3 weeks | 4 weeks | 5 weeks | 6 weeks | 7 weeks | |
|---|---|---|---|---|---|---|---|
| Control | 19.26±0.12 | 22.04±0.46 | 23.50±0.58 | 24.90±0.58 | 24.67±0.95 | 26.43±0.43 | 27.34±0.38 |
| High-fat diet | 21.75±0.27 | 24.34±0.31 | 26.26±0.26 | 26.83±0.46 | 29.16±0.75 | 29.82±0.93 | 31.28±0.95 |
| Garcinia (200 mg/kg) | 20.46±0.38 | 23.17±0.53 | 23.50±0.58 | 24.85±0.86 | 25.7±0.98 | 26.66±1.15 | 27.65±1.38 |
| Gongmi tea (200 mg/kg) | 20.61±0.33 | 22.64±0.37 | 23.59±0.38 | 24.89±0.60 | 25.94±0.67 | 27.18±0.71 | 28.05±0.74 |
| Gongmi so extract (200 mg/kg) | 21.31±0.28 | 23.89±0.30 | 25.13±0.34 | 25.98±0.48 | 27.45±0.67 | 28.85±0.70 | 29.79±0.68 |
Garcinia was used as a positive control. Data are expressed as mean±standard error of the mean.
Effects of gongmi tea and gongmi so extract on kidney and liver function in mice with HFD-induced obesity
Whole-blood element analysis is essential to index toxicity during the development of medical substances. In this study, the serum UA, CRE, and BUN levels were analyzed to evaluate nephrotoxicity, whereas the GOT and GPT levels were analyzed to evaluate hepatotoxicity. As presented in Fig. 7, oral administration of garcinia, gongmi tea, and gongmi so extract did not exhibit hepato- and nephrotoxicities.
Fig. 7.
Effects of oral administration of gongmi tea and gongmi so extract on kidney and liver damage in C57BL/6 mice fed high-fat diet (HFD). To evaluate nephro- and hepatotoxicity, the serum levels of (A) uric acid (UA), (B) creatinine (CRE), (C) blood urea nitrogen (BUN), (D) glutamic oxaloacetic transaminase (GOT), and (E) glutamic pyruvic transaminase (GPT) were analyzed after oral administration of gongmi tea or gongmi so extract for 6 weeks. Data are expressed as mean±standard error of the mean (n=6). #P<0.05 and ##P<0.01 compared with the control group. *P<0.05 and **P<0.01 compared with the HFD group.
DISCUSSION
Obesity is related to numerous diseases, including type 2 diabetes, atherosclerosis, hypertension, and cardiovascular disease (Ding et al., 2017; Kim et al., 2019). The anti-obesity therapy drugs that are currently commercially available are orlistat and liraglutide (Jee et al., 2021). Orlistat was reported to inhibit pancreatic lipase and to break down dietary fat into absorbable free fatty acids (Gorgojo-Martinez et al., 2019; Heck et al., 2000). Liraglutide suppresses appetite by regulating insulin secretion (Ladenheim, 2015; Kelly et al., 2020). However, these drugs have side effects, such as diarrhea, abdominal pain, irregular menstruation, hypertension, and steatorrhea (Siebenhofer et al., 2016; Cha et al., 2018). Thus, novel alternative drugs with fewer side effects need to be developed. Herbal medicines have been studied as attractive anti-obesity therapy agents without side effects. In this study, we evaluated the anti-adipogenic and anti-obesity effects of gongmi tea and gongmi so extract in MDI-treated 3T3-L1 preadipocytes and a mouse model of HFD-induced obesity. The mice were used for analysis under in vivo conditions as their physiology and biochemistry are similar to those of humans. Furthermore, the 3T3-L1 cell line was used under in vitro conditions (Cros et al., 2020; Karunakaran et al., 2021). First, gongmi tea and gongmi so extract at concentrations of 100, 200, and 300 mg/mL did not exhibit any cytotoxicity on 3T3-L1 cells and mouse splenocytes (Fig. 2). Therefore, we used concentrations of 100 and 300 μg/mL in the cell experiments. The number of adipocytes stained with Oil red O significantly decreased in the gongmi tea group compared with the MDI-treated group (Fig. 3A), as well as intracellular lipid accumulation (Fig. 3B). Next, we confirmed the expression of adipogenic transcription factors (PPARγ, adiponectin, and FABP4). As presented in Fig. 4, gongmi tea significantly inhibited the expressions of PPARγ, adiponectin, and FABP4. The downregulation of transcription factors plays a significant role in the inhibition of adipocyte differentiation. The C57BL/6 mice were fed a diet with 60% kcal fat to establish an obesity mice model. Simultaneously, gongmi tea and gongmi so extract were orally administered. As a result, the HFD-induced increase in mouse body weight was significantly decreased by the oral administration of gongmi tea. In addition, gongmi tea did not exhibit hepato- and nephro-toxicities in mice with HFD-induced obesity.
In conclusion, this study demonstrated that gongmi tea and gongmi so extract exert inhibitory effects on adipogenesis in 3T3-L1 preadipocytes. Furthermore, oral administration of gongmi tea and gongmi so extract decreased body weight and epididymal adipose tissue size in mice with HFD-induced obesity. Gongmi tea and gongmi so extract also do not exhibit hepato- and nephrotoxicities in mice. In addition, as a result of measuring the dietary intake of mice, it was confirmed that gongmi tea and gongmi so extract inhibited dietary intake (data not shown). Hence, our results suggest that gongmi tea and gongmi so extract are novel therapeutic candidates for the treatment of obesity-related metabolic diseases.
Footnotes
FUNDING
This research was supported by the National Research Foundation of Korea (grant no. NRF-2021CG027010114).
AUTHOR DISCLOSURE STATEMENT
The authors declare no conflict of interest.
AUTHOR CONTRIBUTIONS
Concept and design: HJ, YRK. Analysis and interpreta-tion: YG, RJ. Data collection: SJJ. Writing the article: SJJ. Critical revision of the article: YG, RJ. Final approval of the article: all authors. Statistical analysis: YG, RJ. Ob-tained funding: HJ. Overall responsibility: YRK.
References
- Auyeung KK, Han QB, Ko JK. Astragalus membranaceus: a review of its protection against inflammation and gastrointestinal cancers. Am J Chin Med. 2016;44:1–22. doi: 10.1142/S0192415X16500014. [DOI] [PubMed] [Google Scholar]
- Bao TR, Long GQ, Wang Y, Wang Q, Liu XL, Hu GS, et al. New lanostane-type triterpenes with anti-inflammatory activity from the epidermis of Wolfiporia cocos. J Agric Food Chem. 2022;70:4418–4433. doi: 10.1021/acs.jafc.2c00823. [DOI] [PubMed] [Google Scholar]
- Cha JY, Nepali S, Lee HY, Hwang SW, Choi SY, Yeon JM, et al. Chrysanthemum indicum L. ethanol extract reduces high-fat diet-induced obesity in mice. Exp Ther Med. 2018;15:5070–5076. doi: 10.3892/etm.2018.6042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen WY, Lin FH. Oxidized hyaluronic acid hydrogels as a carrier for constant-release clenbuterol against high-fat diet-induced obesity in mice. Front Endocrinol. 2021;12:572690. doi: 10.3389/fendo.2021.572690. https://doi.org/10.3389/fendo.2021.572690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choi DH, Han JH, Yu KH, Hong M, Lee SY, Park KH, et al. Anti-oxidant and anti-obesity activities of Polygonum cuspidatum extract through alleviation of lipid accumulation on 3T3-L1 adipocytes. J Microbiol Biotechnol. 2020;30:21–30. doi: 10.4014/jmb.1910.10040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cros J, Bidlingmeyer L, Rosset R, Seyssel K, Crézé C, Stefanoni N, et al. Effect of nutritive and non-nutritive sweeteners on hemodynamic responses to acute stress: a randomized crossover trial in healthy women. Nutr Diabetes. 2020;10:1. doi: 10.1038/s41387-019-0104-y. https://doi.org/10.1038/s41387-019-0104-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- D'Amato R, De Feudis M, Guiducci M, Businelli D. Zea mays L. grain: increase in nutraceutical and antioxidant properties due to Se fortification in low and high water regimes. J Agric Food Chem. 2019;67:7050–7059. doi: 10.1021/acs.jafc.9b02446. [DOI] [PubMed] [Google Scholar]
- Ding Y, Wang Y, Jeon BT, Moon SH, Lee SH. Enzymatic hydrolysate from velvet antler suppresses adipogenesis in 3T3-L1 cells and attenuates obesity in high-fat diet-fed mice. EXCLI J. 2017;16:328–339. doi: 10.17179/excli2016-638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gire D, Acharya J, Malik S, Inamdar S, Ghaskadbi S. Molecular mechanism of anti-adipogenic effect of vitexin in differentiating hMSCs. Phytother Res. 2021;35:6462–6471. doi: 10.1002/ptr.7300. [DOI] [PubMed] [Google Scholar]
- Gong W, Zhang N, Cheng G, Zhang Q, He Y, Shen Y, et al. Rehmannia glutinosa Libosch extracts prevent bone loss and architectural deterioration and enhance osteoblastic bone formation by regulating the IGF-1/PI3K/mTOR pathway in streptozotocin-induced diabetic rats. Int J Mol Sci. 2019;20:3964. doi: 10.3390/ijms20163964. https://doi.org/10.3390/ijms20163964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gorgojo-Martínez JJ, Basagoiti-Carreño B, Sanz-Velasco A, Serrano-Moreno C, Almodóvar-Ruiz F. Effectiveness and tolerability of orlistat and liraglutide in patients with obesity in a real-world setting: The XENSOR Study. Int J Clin Pract. 2019;73:e13399. doi: 10.1111/ijcp.13399. https://doi.org/10.1111/ijcp.13399. [DOI] [PubMed] [Google Scholar]
- Gruenwald J, Freder J, Armbruester N. Cinnamon and health. Crit Rev Food Sci Nutr. 2010;50:822–834. doi: 10.1080/10408390902773052. [DOI] [PubMed] [Google Scholar]
- Heck AM, Yanovski JA, Calis KA. Orlistat, a new lipase inhibitor for the management of obesity. Pharmacotherapy. 2000;20:270–279. doi: 10.1592/phco.20.4.270.34882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hossen MJ, Amin A, Fu XQ, Chou JY, Wu JY, Wang XQ, et al. The anti-inflammatory effects of an ethanolic extract of the rhizome of Atractylodes lancea, involves Akt/NF-κB signaling pathway inhibition. J Ethnopharmacol. 2021;277:114183. doi: 10.1016/j.jep.2021.114183. https://doi.org/10.1016/j.jep.2021.114183. [DOI] [PubMed] [Google Scholar]
- Hotamisligil GS. Inflammation and metabolic disorders. Nature. 2006;444:860–867. doi: 10.1038/nature05485. [DOI] [PubMed] [Google Scholar]
- Huang JM, Zhang GN, Shi Y, Zha X, Zhu Y, Wang MM, et al. Atractylenolide-I sensitizes human ovarian cancer cells to paclitaxel by blocking activation of TLR4/MyD88-dependent pathway. Sci Rep. 2014;4:3840. doi: 10.1038/srep03840. https://doi.org/10.1038/srep03840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jee W, Lee SH, Ko HM, Jung JH, Chung WS, Jang HJ. Anti-obesity effect of polygalin C isolated from Polygala japonica Houtt. via suppression of the adipogenic and lipogenic factors in 3T3-L1 adipocytes. Int J Mol Sci. 2021;22:10405. doi: 10.3390/ijms221910405. https://doi.org/10.3390/ijms221910405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kang MC, Kang N, Ko SC, Kim YB, Jeon YJ. Anti-obesity effects of seaweeds of Jeju Island on the differentiation of 3T3-L1 preadipocytes and obese mice fed a high-fat diet. Food Chem Toxicol. 2016;90:36–44. doi: 10.1016/j.fct.2016.01.023. [DOI] [PubMed] [Google Scholar]
- Kang YM, Kang HA, Cominguez DC, Kim SH, An HJ. Papain ameliorates lipid accumulation and inflammation in high-fat diet-induced obesity mice and 3T3-L1 adipocytes via AMPK activation. Int J Mol Sci. 2021;22:9885. doi: 10.3390/ijms22189885. https://doi.org/10.3390/ijms22189885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karunakaran RS, Lokanatha O, Muni Swamy G, Venkataramaiah C, Muni Kesavulu M, Appa Rao C, et al. Anti-obesity and lipid lowering activity of bauhiniastatin-1 is me-diated through PPAR-g/AMPK expressions in diet-induced obese rat model. Front Pharmacol. 2021;12:704074. doi: 10.3389/fphar.2021.704074. https://doi.org/10.3389/fphar.2021.704074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kelly AS, Auerbach P, Barrientos-Perez M, Gies I, Hale PM, Marcus C, et al. NN8022-4180 Trial Investigators, author. A randomized, controlled trial of liraglutide for adolescents with obesity. N Engl J Med. 2020;382:2117–2128. doi: 10.1056/NEJMoa1916038. [DOI] [PubMed] [Google Scholar]
- Kim BM, Cho BO, Jang SI. Anti-obesity effects of Diospyros lotus leaf extract in mice with high-fat diet-induced obesity. Int J Mol Med. 2019;43:603–613. doi: 10.3892/ijmm.2018.3941. [DOI] [PubMed] [Google Scholar]
- Kim GC, Kim JS, Kim GM, Choi SY. Anti-adipogenic effects of Tropaeolum majus (nasturtium) ethanol extract on 3T3-L1 cells. Food Nutr Res. 2017;61:1339555. doi: 10.1080/16546628.2017.1339555. https://doi.org/10.1080/16546628.2017.1339555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ladenheim EE. Liraglutide and obesity: a review of the data so far. Drug Des Devel Ther. 2015;9:1867–1875. doi: 10.2147/DDDT.S58459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee SG, Kang H. Anti-obesity and lipid metabolism effects of Ulmus davidiana var. japonica in mice fed a high-fat diet. J Microbiol Biotechnol. 2021;31:1011–1021. doi: 10.4014/jmb.2102.02015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li T, Zhang L, Jin C, Xiong Y, Cheng YY, Chen K. Pomegranate flower extract bidirectionally regulates the proliferation, differentiation and apoptosis of 3T3-L1 cells through regulation of PPARγ expression mediated by PI3K-AKT signaling pathway. Biomed Pharmacother. 2020;131:110769. doi: 10.1016/j.biopha.2020.110769. https://doi.org/10.1016/j.biopha.2020.110769. [DOI] [PubMed] [Google Scholar]
- Manogaran P, Beeraka NM, Padma VV. The cytoprotective and anti-cancer potential of bisbenzylisoquinoline alkaloids from Nelumbo nucifera. Curr Top Med Chem. 2019;19:2940–2957. doi: 10.2174/1568026619666191116160908. [DOI] [PubMed] [Google Scholar]
- Moseti D, Regassa A, Kim WK. Molecular regulation of adipogenesis and potential anti-adipogenic bioactive molecules. Int J Mol Sci. 2016;17:124. doi: 10.3390/ijms17010124. https://doi.org/10.3390/ijms17010124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mullen RA, Hurst JJ, Naas KM, Sassoubre LM, Aga DS. Assessing uptake of antimicrobials by Zea mays L. and prevalence of anti-microbial resistance genes in manure-fertilized soil. Sci Total Environ. 2019;646:409–415. doi: 10.1016/j.scitotenv.2018.07.199. [DOI] [PubMed] [Google Scholar]
- Nam W, Nam SH, Kim SP, Levin C, Friedman M. Anti-adipogenic and anti-obesity activities of purpurin in 3T3-L1 preadipocyte cells and in mice fed a high-fat diet. BMC Complement Altern Med. 2019;19:364. doi: 10.1186/s12906-019-2756-5. https://doi.org/10.1186/s12906-019-2756-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Papathanasiou AE, Spyropoulos F, Michael Z, Joung KE, Briana DD, Malamitsi-Puchner A, et al. Adipokines and metabolic regulators in human and experimental pulmonary arterial hypertension. Int J Mol Sci. 2021;22:1435. doi: 10.3390/ijms22031435. https://doi.org/10.3390/ijms22031435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park JU, Kang BY, Kim YR. Ethyl acetate fraction from Dendropanax morbifera leaves increases T cell growth by upregulating NF-AT-mediated IL-2 secretion. Am J Chin Med. 2018;46:453–467. doi: 10.1142/S0192415X18500234. [DOI] [PubMed] [Google Scholar]
- Saltiel AR, Olefsky JM. Inflammatory mechanisms linking obesity and metabolic disease. J Clin Invest. 2017;127:1–4. doi: 10.1172/JCI92035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seo YJ, Kim KJ, Choi J, Koh EJ, Lee BY. Spirulina maxima extract reduces obesity through suppression of adipogenesis and activation of browning in 3T3-L1 cells and high-fat diet-induced obese mice. Nutrients. 2018;10:712. doi: 10.3390/nu10060712. https://doi.org/10.3390/nu10060712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siebenhofer A, Jeitler K, Horvath K, Berghold A, Posch N, Meschik J, et al. Long-term effects of weight-reducing drugs in people with hypertension. Cochrane Database Syst Rev. 2016;3:CD007654. doi: 10.1002/14651858.CD007654. https://doi.org/10.1002/14651858.cd007654.pub4. [DOI] [PubMed] [Google Scholar]
- Wang F, Chen L, Chen S, Chen H, Liu Y. Microbial biotransformation of pericarpium citri reticulatae (PCR) by Aspergillus niger and effects on antioxidant activity. Food Sci Nutr. 2020;9:855–865. doi: 10.1002/fsn3.2049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu J, Ye X, Yang S, Yu H, Zhong L, Gong Q. Systems pharmacology study of the anti-liver injury mechanism of Citri Reticulatae Pericarpium. Front Pharmacol. 2021;12:618846. doi: 10.3389/fphar.2021.618846. https://doi.org/10.3389/fphar.2021.618846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiao Z, Zhang Q, Dai J, Wang X, Yang Q, Cai C, et al. Structural characterization, antioxidant and antimicrobial activity of water-soluble polysaccharides from bamboo (Phyllostachys pubescens Mazel) leaves. Int J Biol Macromol. 2020;142:432–442. doi: 10.1016/j.ijbiomac.2019.09.115. [DOI] [PubMed] [Google Scholar]