Abstract
This study seeks to investigate challenges to combined antiretroviral therapy (cART) treatment adherence and treatment outcomes in Kampala, Uganda. Data was collected from a survey administered to two cohorts of patients with human immunodeficiency virus type 1 (HIV-1) receiving care and cART from the Joint Clinical Research Centre (JCRC) in Kampala. Cohort I consisted of 93 individuals successfully treated on cART for a period of three years, while Cohort II consisted of 56 individuals who have experienced treatment failure with first-line cART within two years. We hypothesize that distance to the treatment facility would be a predictor of poor adherence and thus treatment failure. However, results suggested otherwise, whereby participants living more than two hours away from their treatment facility were actually less likely to miss their daily dose of cART (OR = 0.33, p < 0.05), compared to those living in proximity to the treatment center. Further, high-income employment (OR = 3.82, p < 0.05) and partnered relationship status (OR = 4.28, p < 0.05) were predicted to increase the probability of missing doses. These findings may be explained by the deep-seated stigma which has remained pervasive in the lives of HIV-positive population in Kampala, even 30 years after the peak of the HIV/AIDS epidemic.
The introduction of combined antiretroviral therapy (cART) has made it possible to slow the progression of the human immunodeficiency virus type 1 (HIV-1), thereby improving the health and well-being of millions of HIV-positive individuals (Reda & Biadgilign, 2012). Sub-Saharan Africa shoulders the majority of the burden of the HIV-1 epidemic, with 1.4 million people living with HIV-1 (PLHIV) in Uganda alone (Lange et al., 2004; UNAIDS, 2018). Within the last decades, treatment programs have become widespread across the continent with the assistance of financing organizations such as the US President’s Emergency Plan For AIDS Relief, Global Fund to Fight AIDS, Tuberculosis and Malaria, as well as strategic partnerships with pharmaceutical companies and foundations (Hardon et al., 2006; Reda & Biadgilign, 2012). As a result, it was estimated that 72% of people diagnosed with HIV-1 in Uganda were on treatment as of 2018 (World Bank, 2018).
cART consists of several drugs taken in combination to target the virus at various stages of its life-cycle, in order to prevent replication (Zolopa, 2010). Patients who are successfully treated on cART are considered to be virally supressed, with an undetectable viral load (i.e. less than 20 copies of the virus). However, for cART to be most effective, patients must adhere to strict medication regimens, ideally without missing a single dose (i.e. perfect adherence) (Hardon et al., 2006). While no standard definition exists for adherence, it generally refers to whether or not a medication has been taken in a timely manner and at the prescribed dosage. High levels of adherence (≥95%) to cART are necessary for treatment success as well as the prevention of the development of drug-resistant strains of HIV-1 (Hardon et al., 2006; Tuller et al., 2010). While drug resistance has stabilized in high-income countries, low- to middle-income countries are experiencing increased prevalence of circulating and transmitted HIV-1 drug resistant strains. Frequency of treatment failure based on a rebound of circulating HIV-1 and the emergence of drug resistant HIV-1 following the initiation of first line cART remains higher in Uganda (5% failure/year of cART) than observed in high income countries, despite Uganda being one of the first East African countries to implement the scale-up of antiretroviral programs (Author, 2013; Author et al., 2013; Pham, Wilson, Law, Kelleher, & Zhang, 2014; Author et al., 2019).
While cART adherence reported in African countries has often been shown to surpass levels of adherence in North American countries, study results tend to vary greatly depending on the research context. In Uganda, estimates of the proportion of patients who reported optimal cART adherence ranged from 68% to 98% (Mills et al., 2006). Adherence rates have also been shown to decline over time in long-standing treatment programs (Nachega et al., 2010). Those who miss more than one dose of their medication in a week are already at risk of treatment failure, and thus the development of drug resistance (Hardon et al., 2006). Patients generally begin on first-line cART (the most effective and low-cost medication), which is often available through many treatment programs in low-income countries such as Uganda, free of charge. However, drug-resistant HIV may require treatment with second- or third-line cART which can be costly, require more treatment monitoring, have greater adverse events, and involve more complicated treatment regimens (Bacha et al., 2012). Patients experiencing virological failure in Uganda may therefore have limited access to second- and third-line cART, highlighting the need for the identification of predictors of treatment failure and barriers to adherence (Boender et al., 2015; Hardon et al., 2006).
Adherence to treatment may be challenging due to a range of cultural, social, and financial factors (Byakika-Tusiime et al., 2005; Hardon et al., 2006; Weiser et al., 2003). One important factor that can affect adherence to treatment is the distance to the clinic. Commonly referred to as the distance-decay effect, research observes a consistent spatial pattern—people living closer to health care facilities being more likely use them (Hunter & Shannon, 1985). The distance-decay effect is a persistent problem in some sub-Saharan African countries where health care resources are spatially concentrated in favour of urban, high-income areas (Iyer et al., 2020). For example, McLaren et al. (2014) find in South Africa that people who live more than 2km from nearest clinics are less likely to consult health professionals and have doctors or nurses present at birth for children. Research also points to longer distance and travelling time to health facilities as the most important barrier to health care utilization in the Ahafo-Ano South district, Ghana (Buor, 2003). It is also found in Nigeria and Zambia that a long distance to the facility is strongly associated with difficulty in accessing HIV clinics (Adelekan et al., 2019; Cope et al., 2016). In sub-Saharan African countries, this distance-decay relationship is often considered reflective of structural problems such as lack of public transportation, high travel cost, poor road condition, and long travel time (Antabe et al., 2020; Atuoye et al., 2015; Geleto et al., 2018). In Uganda, its has been argued that sub-optimal adherence can be attributed to structural barriers accessing the medication including long distance to health care clinics, rather than behavioural barriers such as people’s inability to follow the drug regimen (Crane et al., 2006; Mukherjee et al., 2016). Thus, it is possible that the distance-decay effect plays an important role in this context; longer travel time may be associated with lower odds of adhering to HIV-1 treatment.
However, not all analyses of health care utilization report distance as a major barrier to access (Nemet & Bailey, 2000). cART users frequently cite their inability to pay for long-distance transport costs as a reason for non-adherence, yet it is often shown that there is no substantial association between treatment outcomes and geographical distance to treatment facilities (Billioux et al., 2018; Byakika-Tusiime et al., 2005; Hardon et al., 2006). This inconsistent pattern points to the potential importance of understanding the interactions between health and geographical concepts such as location, place, space, and distance as socially constructed (Jones & Moon, 1993; Kearns, 1993). Therefore, rather than to define distance with a linear measure, it may be critical to contextualize what distance can mean to people who are engaged with HIV-1 care and treatment in Uganda (see (Simandan, 2016)). For example, essential antiretroviral medicines for HIV-1 treatment is only available in 27% of district hospitals and 40% of lower-level health facilities in Uganda (Windisch et al., 2011), with the insufficient and irregular supply of medicines (Muyinda & Mugisha, 2015). In Uganda, the Joint Clinical Research Centre (JCRC), the facility where we conducted this study, is considered to be a high-quality treatment facility, which guarantees a constant supply of drugs and minimal waiting times. In this context, it is possible that people who plan to get their medication from the JCRC by travelling hours to get there could be more invested in treatment adherence. In addition, research shows that HIV-related stigma and discrimination can serve as a major barrier to the uptake of specialized HIV care (Gesesew et al., 2017; Rao et al., 2012). Due to fear of social exclusion, some PLHIV cannot choose treatment facilities in their communities to maintain anonymity, even with extra time and cost (McMahon et al., 2017). In this case, clinics farther away from households are seen as places for safer access to care (Billioux et al., 2018). Hence, we recognized that it is also plausible that longer travel time may be associated with higher odds of adhering to HIV-1 treatment.
As shown above, the debate on whether geographical barriers such as distance and travelling time to health facilities and should be understood as geometrically or socially constructed continues in the literature in health geography. To contribute to this debate, we aim to understand whether travel time as a proxy of geographical barrier has any significant impact on HIV-1 treatment (cART) outcomes in Kampala, Uganda. In addition, we also explore other predictors of cART outcomes. We hypothesized that those with a greater burden of mobility (i.e. longer travel times) would experience lower levels of adherence and therefore be more susceptible to treatment failure, compared to those who travel minimally, after controlling for demographic characteristics.
Methods
Study Site
This project was conducted in partnership with the Joint Clinical Research center (JCRC) in the greater Kampala region, who have provided HIV-1 treatment services for over 200,000 patients in Uganda, in addition to other services such as tuberculosis care and treatment. Antiretrovirals are available at the JCRC free of charge. The treatment facility is located in the Wakiso district, approximately 10 kilometers away from Kampala’s city center (see Figure 1). Kampala is Uganda’s capital city, located in central region of Uganda bordering Lake Victoria (Vermeiren et al., 2012). As Kampala is found within the Buganda Kingdom, the most commonly spoken language is Luganda (Brisset-Foucault, 2013). However, the city’s rapid urban expansion and increased commercial activity has drawn settlers from various regions of Uganda, with a variety of native languages. Paved roads in Kampala funnel traffic through the densely populated city, resulting in severe traffic jams (Vermeiren et al., 2012).
Figure 1:
Map of Kampala, Uganda and the location of the JCRC
Study Design
Data was collected from patients receiving routine care and treatment from the JCRC, under an agreement of confidentiality. In-country ethics approval was obtained from the Institutional Review Board/Research Ethics Committee (IRB/REC) in addition to approval from the Research Ethics Board (REB) at the University of Western Ontario in Canada. From the JCRC’s HIV-1 treatment department, eligible patients who arrived for their regular clinic visits were asked if they were interested in participating in the study. Patients who expressed interest were given a consent form in their preferred language (either English or Luganda). Patients who agreed to participate completed a structured questionnaire specifying demographic, geographic and behavioral characteristics. Upon completion of the questionnaire, participants were compensated for their time and transportation costs.
In order to evaluate predictors of divergent treatment outcomes, the study included two cohorts of patients: those who had been successfully treated on first line cART for a period of three years and those who have experienced treatment failure on first-line cART within two years. All patients receiving cART pass through the same intake department, no matter which line of treatment they are on, therefore both cohorts were recruited from the same pool of patients. As treatment failure represents the least frequent treatment outcome, the treatment success cohort consisted of 93 individuals, while the treatment failure cohort consisted of 56 individuals. Treatment ‘failure’ was determined based on clinical parameters (i.e. detectable viral load) obtained from the JCRC patient database, and adherence to cART was assessed with respect to missed appointments and discontinuity of drug regimen (using self-reported data & JCRC patient logs). Patients’ treatment regimen (i.e. first-line, second-line, etc.) was recorded using the patient database.
Measurement
The focal dependant variable was treatment failure (failure = 1 and success = 0), in addition to indicators of treatment adherence such as missed doses (yes/missed = 1 and no/never missed = 0). Adherence was assessed from when the patient was initiated on first-line treatment. To measure adherence, participants were asked if they have ever 1) missed a dose of their cART medication, 2) missed an appointment, 3) stopped taking their medication intentionally, and 4) unable to refill their medication. All four variables were coded as either 1 = yes or 0 = no. All demographic information was drawn from the study questionnaire, with the exception of age which was recorded from the patient database. Age was categorized into intervals of approximately ten years, aggregating the upper and lower limits to ensure the relative equality of group sizes. Only patients over the age of 18 qualified for the study. Gender and parenthood (i.e. whether they have children) were also determined based on the participants responses to the questionnaire. Participants were then asked to identify which income range they correspond to. Income figures correspond to the amount of Ugandan shillings (UGX) earned per month. As salaries in Uganda tend to be inconsistent, participants’ responses represent estimates of their average income (Hardon et al., 2006). Participants originally selected one of five ranges of income; however, the three higher income ranges were merged due to the small number of individuals in each group. An income of 100,000 UGX corresponds to approximately $36.00 CAD, and 350,000 UGX corresponds to $126.00 CAD. Participants were categorized as either below 100,000 UGX per month, over 350,000 UGX, or between 100,000 and 350,000 UGX. An income of 350,000 UGX is considered to be relatively high in the Ugandan context, accounting for purchasing power (Wanyenze et al., 2006). Those who identified themselves as unemployed were aggregated into the category of under 100,000 UGX.
Participants indicated their current relationship status. Those who reported that they are single (i.e. never married) were grouped into one category. Those who indicated that they are currently married or have a boyfriend/girlfriend were grouped into the category “with partner,” and those who indicated that they were previously married (now either widowed or separated/divorced) were grouped together. This categorization of relationship status follows the general method employed by Wanyenze et al. (2006), adjusted slightly to assess the impact of partnership on adherence, rather than marital status. Finally, patients indicated the time they spent in transit on the way to the clinic where they receive HIV-1 treatment from the JCRC. Original categories were as follows: 1) less than 10 minutes, 2) 10 to 30 minutes, 3) 30 minutes to one hour, 4) one to two hours, 5) greater than two hours. This variable was re-categorized to 1) less than one hour, 2) one to two hours, and 3) greater than two hours, to ensure the adequacy of group sizes per category for the regression analysis. Accounting for the urban context of Kampala, travel times were taken into consideration rather than travel distances, as movement through the city is often restricted due to traffic rather than kilometer distance (Vermeiren et al., 2012).
Statistical Analysis
The statistical analysis was conducted in various stages. All survey data was initially entered into IBM SPSS Statistics 25, and then analyzed using the statistical software package Stata 16. Descriptive statistics were first calculated for all theoretically relevant demographic and clinical variables, indicating counts and sample percentages for categorical variables (seen in Table 1 and Table 4). The mean and standard deviation of viral load was computed, as this measure is a continuous variable. In Table 2 and Table 5, bivariate logistic regression was used to assess the influence of each independent variable, including transportation time, on the key dependent variables (treatment failure and missing a dose). Lastly, multivariate logistic regression was conducted (Table 2 and Table 5) to determine the relative influence of transit time when controlling for demographic variables. We evaluated potential demographic variables that may influence adherence including family composition, gender, wealth and age. Results were presented with odds ratios (OR) derived from regression coefficients.
Table 1:
Demographic and health characteristics of treatment failure and treatment success cohorts
| Covariates | Treatment Success Cohort (n=93) | Treatment Failure Cohort (n=56) |
|---|---|---|
|
| ||
| Time to Clinic | ||
| Less than one hour | 41 (44.09%) | 23 (41.07%) |
| One to two hours | 20 (21.51%) | 20 (35.71%) |
| Greater than two hours | 32 (34.41%) | 13 (23.21%) |
| Viral Load (copies/ml) | <20 (SD 0) | 260,525.93 (SD 840102.75) |
| Age | ||
| 18–30 | 14 (15.05%) | 17 (30.36%) |
| 31–40 | 22 (23.66%) | 17 (30.36%) |
| 41–50 | 34 (36.56%) | 8 (14.29%) |
| 51+ | 23 (24.73%) | 14 (25.00%) |
| Gender | ||
| Female | 60 (64.52%) | 36 (64.29%) |
| Male | 33 (35.48%) | 20 (35.71%) |
| Income (UGX) | ||
| <100,000 (including unemployed) | 40 (43.01%) | 24 (42.86%) |
| 100,000–350,000 | 33 (35.48%) | 21 (37.50%) |
| > 350,000 | 20 (21.51%) | 11 (19.64%) |
| Relationship Status | ||
| Single | 16 (17.20%) | 13 (23.21%) |
| With Partner | 56 (60.22%) | 31 (55.36%) |
| Separated or Widowed | 21 (22.58%) | 12 (21.43%) |
| Children | ||
| Has children | 80 (86.02%) | 43 (76.79%) |
| Does not have children | 13 (13.98%) | 13 (23.21%) |
Mean (SD) or n (%), Sample size = 149.
Table 4:
Demographic and Health Characteristics of HIV-positive individuals with self-reported optimal and sub-optimal adherence to ART
| Self-reported missed doses (n=63) | Self-reported never missed doses (n=86) | |
|---|---|---|
|
| ||
| Time to Clinic | ||
| Less than one hour | 35 (55.56%) | 29 (33.72%) |
| One to two hours | 16 (25.40%) | 24 (27.91%) |
| Greater than two hours | 12 (19.05%) | 33 (38.37%) |
| Time to Failure (days) | 255.25 (SD 306.51) | 147.55 (SD 255.63) |
| Viral Load | 44,845.91 (SD | 136,814.19 (SD |
| (copies/ml) | 178,132.32) | 676,623.40) |
| Age | ||
| 18–30 | 20 (31.75%) | 11 (12.79%) |
| 31–40 | 15 (23.81%) | 24 (27.91%) |
| 41–50 | 12 (19.05%) | 30 (34.88%) |
| 51+ | 16 (25.40%) | 21 (24.42%) |
| Gender | ||
| Female | 39 (61.90%) | 57 (66.28%) |
| Male | 24 (38.10%) | 29 (33.72%) |
| Income (UGX) | ||
| <100,000 (including unemployed) | 23 (36.51%) | 41 (47.67%) |
| 100,000–350,000 | 23 (36.51%) | 31 (36.05%) |
| > 350,000 | 17 (26.98%) | 14 (16.28%) |
| Relationship Status | ||
| Single | 8 (12.70%) | 21 (24.42%) |
| With partner | 38 (60.32%) | 49 (56.98%) |
| Separated or widowed | 17 (26.98%) | 16 (18.60%) |
| Children | ||
| Has children | 46 (73.02%) | 77 (89.53%) |
| Does not have children | 17 (26.98%) | 9 (10.47%) |
Mean (SD) or n (%), Sample size = 149.
Table 2:
Bivariate (Model 1) and multivariate (Model 2) and regression of treatment failure (OR)
| Covariates | Model 1: OR (SE) | Model 2: OR (SE) |
|---|---|---|
|
| ||
| Time to Clinic | ||
| Less than one hour | (reference) | |
| One to two hours | 1.78 (.73) | 1.76 (.77) |
| Greater than two hours | .72 (.30) | .79 (.37) |
| Age | ||
| 18–30 | (reference) | |
| 31–40 | .64 (.30) | .66 (.34) |
| 41–50 | .19 (.10)** | .18 (.11)** |
| 51 + | .50 (.25) | .50 (.27) |
| Female | .99 (.35) | .50 (.32) |
| Relationship Status | ||
| Single | (reference) | |
| With Partner | .68 (.30) | .65 (.32) |
| Separated or Widowed | .70 (.36) | .80 (.45) |
| Have Children | .54 (.23) | .83 (.44) |
| Income (UGX) | ||
| <100,000 | (reference) | |
| 100,000–350,000 | 1.06 (.40) | .99 (.42) |
| >350,000 | .92 (.42) | 1.02 (.44) |
p < .01. Sample size = 149. SE: Standard Error.
Model 2 Maximum Log Likelihood: −91.02Model 1 Maximum Log Likelihood: −96.57
Table 5:
Bivariate (Model 1) and multivariate (Model 2) and regression of missing doses (OR)
| Covariates | Model 1: OR (SE) | Model 2: OR (SE) |
|---|---|---|
|
| ||
| Time to Clinic | ||
| Less than one hour | (reference) | |
| One to two hours | .55 (.23) | .40 (.20) † |
| Greater than two hours | .30 (.13)** | .33 (.16)* |
| Age | ||
| 18–30 | (reference) | |
| 31–40 | .34 (.17)* | .20 (.12)** |
| 41–50 | .22 (.11)** | .18 (.11)** |
| 51+ | .42 (.21) † | .40 (.23) |
| Female | .83 (.29) | 1.42 (.71) |
| Relationship Status | ||
| Single | (reference) | |
| With Partner | 2.04 (.95) | 4.28 (2.60)* |
| Separated or Widowed | 2.79 (1.51)† | 8.28 (5.76)** |
| Have Children | 3.16 (1.43)* | 3.64 (2.24)* |
| Income (UGX) | ||
| <100,000 (including unemployed) | (reference) | |
| 100,000–350,000 | 1.32 (.50) | 1.53 (.70) |
| >350,000 | 2.16 (.96) † | 3.82 (2.42)* |
p < .10.
p < .05.
p < .01.
Sample size = 149. SE: Standard Error.
Model 1 Maximum Log Likelihood: −97.10
Model 2 Maximum Log Likelihood: −83.62
Logistic regression was the most appropriate choice for analysis, as the data satisfies key assumptions. Both outcome variables are binary (whether or not one failed on treatment, and whether or not one missed a dose), observations are independent of one-another, and there is no multicollinearity between independent variables. The study’s sample size approaches the suggested minimum observation-predictor ratio: A minimum of ten cases representing the least probable outcome should be included per independent variable (Peng et al., 2002). The least probable outcome was treatment failure, with a probability of 0.38. With a total of six independent variables included in statistical models, the suggested minimum sample size is 158.
Results
As shown in Table 1, all participants in the treatment success cohort have an undetectable viral load (below 20 copies of viral RNA per ml of plasma) and therefore standard deviation was zero. This was expected, as anyone who is successful on cART will have suppressed the virus to the point where it is undetected by standard viral load tests. The mean viral load for the treatment failure cohort was approximately 260,500 copies of RNA/ml of plasma, with a detectable viral load classified as >1000 copies/ml. However standard deviation was comparatively high, at approximately 840,000.
In the sample, the largest proportion of participants fell into the 41 to 50 age range. The mean age for the success cohort was approximately 43 and the mean for the failure cohort was 40. Although mean ages are similar, the distribution across age categories differs. In the treatment failure cohort, the 41–50 age range contained the smallest proportion of participants. However, the largest proportion of the treatment success cohort fell between ages 41–50. The proportion of males and females is relatively equal in both groups, although the majority of the sample were women. This was expected as women are both more likely to seek out treatment and more vulnerable to HIV-1 due to biological and cultural factors (Türmen, 2003; Mills, Beyrer, Birungi, & Dybul, 2012).
In Table 2, Model 1 assesses the bivariate association between transit time to clinic and treatment failure, in addition to an analysis of the relationship between demographic variables (age, gender, income, parenthood and relationship status) and treatment failure. Model 2 in Table 2 assesses the association between transit time and treatment failure when controlling for demographic variables. In terms of the variables of interest, the treatment failure and success cohorts seem remarkably similar. The maximum log likelihood for Model 2 (−91.02) barely increases from the baseline of −98.64 which was calculated for the intercept-only model, with no covariates (Peng et al., 2002). A substantial increase in the value of log likelihood would have hinted at the fit of our model’s coefficients. Both models are weak predictors of treatment failure, as the only significant association is seen in the 40–50 age range, suggesting that compared to patients aged 18–30, patients aged 40–50 had a 61% decrease in their probability of treatment failure.
A bivariate analysis of adherence indicators was conducted to gauge their importance as predictors of treatment failure (Table 3). Expectedly, a lack of adherence was a significant predicator of treatment failure. Those who miss a dose were more than twice as likely to fail (OR = 2.10, p < .05). Similarly, those who reported an inability to refill their medication were approximately 3.7 times more likely to fail treatment (OR = 3.71, p < .05). Those who indicated that they intentionally stopped taking their medication were also more likely to fail, although this association was noted with little statistical significance and should thus be interpreted with caution (OR = 2.93, p < .10). In total, 42% of participants reported having missed at least one dose of their medication (n=63), the majority of which reflected on periods of low adherence rather than simply missing one dose. Compared to other measures of adherence, missing a dose was the most statistically significant predictor of treatment failure (p = .03), and the most frequently occurring indicator of sub-optimal adherence. This measure was further examined in a separate logistic regression in order to gauge the influence of our predictor variables on the likelihood of missing doses.
Table 3:
Logistic regression of treatment outcome (bivariate)
| Covariates | OR (SE) |
|---|---|
|
| |
| Missing doses (unintentionally) | 2.10 (.72)* |
| Missing appointments | 1.01 (0.40) |
| Stopped taking medication intentionally | 2.93 (1.75) † |
| Unable to refill medication | 3.71 (2.37)* |
p < .10.
p < .05. Sample size = 149. SE: Standard Error.
In Table 5, Model 1 presents the association between our predictor variables and the likelihood of missing doses. Surprisingly, participants who traveled over two hours to get to the JCRC were approximately 70% less likely to miss a dose relative to those who traveled less than one hour, both before (OR = 0.30, p < .01) and after (OR = 0.33, p < .05) controlling for demographic variables (Table 5, Model 2). Controlling for demographic variables, significance begins to emerge for other categories of transit time: Those who traveled one to two hours to get to the clinic were also 60% less likely to miss doses compared to those who traveled less than one hour (OR = 0.40, p < .10). Therefore, the trend of decreasing probability of missing doses in those with longer travel times is consistent across transit time categories.
The association between treatment failure and the 41–50 age range holds in the second analysis predicting missed doses. In the bivariate analysis, those between the ages of 41 and 50 are least likely to miss a dose, compared to the 18–30 age range (OR = 0.18, p < .05). Further, those in the 31–40 age range are 80% less likely to miss a dose (OR = 0.20, p < .05). Gender remained unrelated to adherence throughout the analysis. However, relationship status became increasingly significant when controlling for demographic variables. Those who have a partner were approximately 4.3 times more likely to miss a dose, compared to their single counterparts (OR = 4.28, p < .05). This has been previously reported and may be associated with partner stigma and concerns in sharing HIV-1 infection status (Dlamini et al., 2009). Further, those who reported having children were more than three times more likely to miss their medication, both before and after controlling for demographic variables (OR = 3.64, p < .05). Finally, those who corresponded to the higher-income category of >350,000 were more likely to miss doses compared to those who make less than 100,000 (including those who are unemployed). The bivariate regression suggests that high-income individuals are over twice as likely to miss a dose (OR = 2.16, p < .10), and the association is amplified after controlling for demographic variables in Table 5. In Table 5, Model 2, high-income patients were nearly 4 times more likely to miss their medication (OR = 3.82, p < .05).
Model 2 appears to be the best fit, with a maximum log likelihood of −83.62, compared to the baseline value of −101.50 for the intercept-only model. Akaike’s Information Criterion (AIC), and Schwarz’s Bayesian Information Criterion (BIC) provide a further indication of the fit of the model (Table 6). AIC decreases by approximately 14, suggesting that Model 2 is a better fit than the null model. The BIC, however, is more resistant to change, and suggests a negative difference. It should be noted that these metrics are a function of the number of model parameters and the sample size. For moderate sample sizes, the BIC tends to point to the model which is most parsimonious, therefore this metric may not be appropriate for this study (Burnham & Anderson, 2004).
Table 6:
Akaike’s Information Criterion (AIC) and Schwarz’s Bayesian Information Criterion (BIC)
| Model | df | AIC | BIC |
|---|---|---|---|
|
| |||
| Null Model | 1 | 204.99 | 208.00 |
| Model 2 | 12 | 191.24 | 227.28 |
Model 2 found in Table 5. df: Degrees of freedom
Discussion
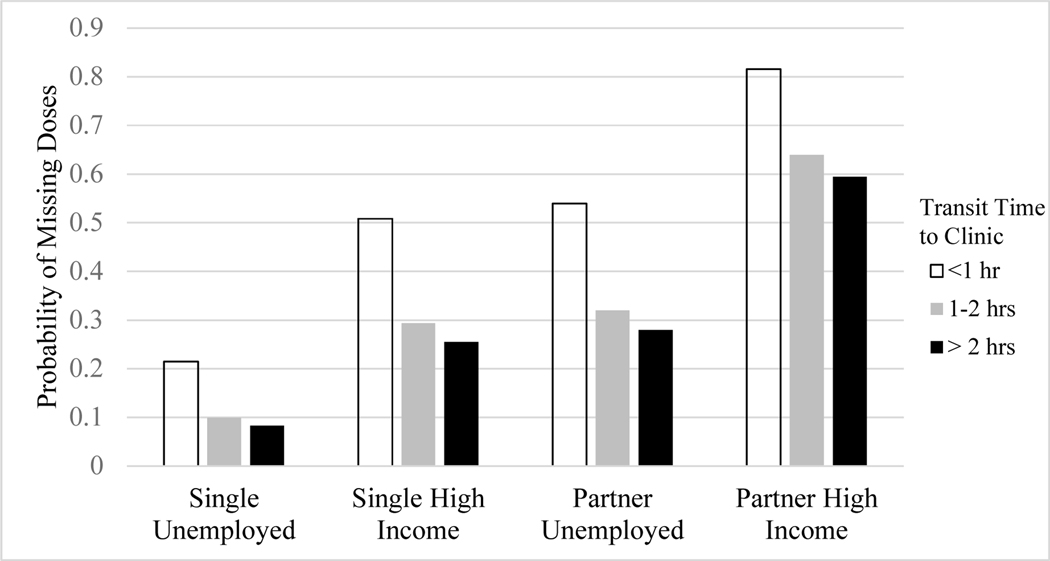
The key findings of this study are portrayed in Figure 1, which illustrates the relative predicted probability that a range of individuals would miss a dose of their medication. We find the person with the lowest probability of missing a dose is predicted to be single, unemployed (or low income), and live over two hours away from the treatment facility. Conversely, someone who has a high paying job, a partner, and lives close to the facility is predicted to have an 80% chance of missing their medication.
Transport time and cost have been identified in the literature as an important barrier for adherence, although the degree of influence of transit time on cART adherence and treatment outcomes is not agreed upon (Ammon et al., 2018; Hardon et al., 2006). Our results suggest that transportation time may actually have a positive relationship with treatment adherence. The findings show that those further away from the clinic (at least two hours) were actually less likely to report missing their medication. This observation may support the importance of understanding distance as socially constructed rather than geometrically constructed (Jones & Moon, 1993; Kearns, 1993). In other words, PLHIV may have unique reasons to choose the JCRC beyond its close proximity from their households. This trend is consistent with previous research in Uganda (Billioux et al., 2018), identifying that 57% of individuals travelled further than their nearest facility for HIV-1 treatment. Similarly, Akullian et al. (2016) show that PLHIV travel longer distance for care, potentially due to limited availability of specialized HIV-1 services and preference of higher tiered facilities. Another explanation could be that people prefer to go to clinics further away because of the deep-seated stigma associated with HIV/AIDS in Uganda (Hardon et al., 2006). cART users in Uganda tend to choose treatment facilities which are isolated from their home community, in order to avoid contact with friends, family and acquaintances (Hardon et al., 2006). Such encounters put them at a risk of publicizing their HIV-1 status and thus facing stigmatization and discrimination. Based on findings from our research and previous studies (Billioux et al., 2018), we argue that the utilization of HIV care may not always be determined by ease of access to treatment locations, but may tend to be influenced by people’s sense of safety within their neighbourhoods. Hence, if people feel unsafe in terms of the potential of stigmatization, they would prefer to travel long distances to receive care in safer locations, especially when such locations are perceived to be providing specialized, higher-tier and more advanced HIV-1 care.
Further, while it was expected that stable relationships and employment would be positive predictors of adherence, this study’s results show otherwise. This may be attributed to the influence of stigma in both their work and family lives. Due to the high prevalence and importance of HIV/AIDS in Uganda, it is assumed that someone who is taking daily medication must be taking antiretrovirals and is therefore HIV-positive. cART users have expressed the fear of losing their jobs if they are seen taking medication at work (Hardon et al., 2006). They mention that their employers believe that someone with HIV-1 has a low life-expectancy and is therefore not worth the investment of resources such as time and money. The fear of stigma in the workplace pressures individuals to take their medication in secret, which may prevent someone from taking their medication at the correct time, which is necessary for cART to be effective (Hardon et al., 2006). This might be even more so the case for those with high-paying employment, and thus a higher social status. In these cases, there may be an increased perception of risk if someone is seen taking their medication.
We expected family and partner support to improve cART adherence; however, our results suggest the reverse. At the family level, a lack of disclosure of one’s HIV-1 status may result in poor adherence. Status disclosure often results in abandonment and poor treatment by one’s spouse, thus prompting many cART users to secretively take their medication (Hardon et al., 2006). Those who were either separated or widowed were also more likely to miss doses compared to their single counterparts. As this study is retrospective, it is possible that at that the time of failure, these participants were still in a relationship, making them vulnerable to stigma. Those with children were also more likely to miss doses, which may be attributed to busier schedules and/or non-disclosure of HIV-1 status.
In general, the degree of discomfort that cART users feel when taking their medication in shared spaces may have a substantial influence on their adherence. The effect of this discomfort may even be additive if someone experiences stigma or discrimination at home as well as at work. In the context of health geography, Kearns (1993) refers to space as “the local context of health, disease and social process” (p. 140). By using this approach to health research, we can understand the interactions between cART users and the places they frequent. The level of stigma experienced at home, at work and around health facilities may have a stronger influence on adherence than the time spent in transit to the clinic. If the fear of status disclosure is pervasive in the lives of those on cART, structural interventions such as increasing the number of treatment facilities will not be effective in improving treatment outcomes. In order to improve the utilization of treatment services, interventions must consider the experiences of the HIV-1-positive population within their local contexts, as well as the healthcare system itself (Andersen, 1995).
Limitations
Variability in measurements of adherence may produce divergent results. For instance, participants were asked if they have ever missed doses on treatment. However, if a participant has been on cART for years, it is probable that they have missed at least one dose. As previously mentioned, those who reported missing doses tended to have experienced a prolonged period of low adherence, rather than a single dose. Still, self-reported measures of adherence have been shown to be consistent with more objective measures of adherence (i.e. electronic drug monitoring) (Author et al., 2004). Regardless, the development of standardized measurements of adherence will facilitate future analyses. Quantitative studies should take into consideration the variability of the lived experiences of cART users. To conduct a comprehensive evaluation of the relationship between geospatial variables and treatment adherence, quantitative measures of geographic mobility should be analyzed in conjunction with qualitative measures (i.e. via semi-structured interviews with participants). These interviews were conducted with participants as part of this research project and results will be analyzed in a forthcoming study.
In addition, we did not control for education in this study. The JCRC houses patient’s administrative data including information such as age and education. Unfortunately, a substantial amount of the information regarding patient’s education was missing. As a result, we were not able to account for education in our analysis. This omission may be a limitation as previous studies show that the association between HIV treatment adherence and education is statistically significant (Heestermans et al., 2016; Ridgeway et al., 2018). It is important for future research to consider the role of patients’ level of education on HIV-1 treatment adherence.
Finally, the study’s moderate sample size must be considered in the interpretation of results. The sample may not be representative of all those in Uganda who are on HIV-1 treatment. Rather, the study should be considered within the context of the JCRC. Our results suggest a degree of internal validity as our measures of adherence were negatively correlated with treatment success, which is consistent with academic literature (Hardon et al., 2006; Lange et al., 2004). The study’s sample size of 149 may present certain limitations in terms of potential statistical significance, however, related studies have used similar sample sizes and produced meaningful analyses (Mills et al., 2006; Siedner et al., 2013). Addressing this limitation, future studies should collect data from multiple treatment centres in Uganda, which allows more advanced techniques such as multilevel analysis to be employed for understanding the effect of individual- and facility-level factors at the national level.
Conclusion
Adherence greater than 95% to cART is necessary for treatment success, and high rates of adherence are necessary to minimize drug resistance. Although cART users are highly motivated to follow their treatment plan, challenges such as stigma undermine their best efforts to do so. It is therefore crucial that factors influencing adherence are identified and evaluated. Resource-constrained regions require a context-specific approach to maximize treatment adherence and combat drug resistance. The identification of barriers to ARV treatment adherence will provide guidance for future public health initiatives targeting the HIV/AIDS epidemic in Uganda.
Supplementary Material
Figure 2:
Probability of missing doses for individuals with and without a partner and with and without employment
Highlights:
cART users who were in a relationship had lower adherence than those who were single
Those with high-income employment were more likely to miss a dose versus low-income
Those traveling long distances to HIV treatment facilities were highly adherent
Treatment adherence may be affected by stigma within households as well as workplaces
Acknowledgements
This research was made possible through the generous support and guidance of the JCRC staff including Christine Mukasa, Erinah Abaasa, Sandra Rwambuya and many others. I would also like to acknowledge the study’s participants for their willingness to share their experiences with us. Funding was provided to Author by the NIH/NIAID grant AI49170. Author also received funding for this study as a Canada Research Chair Tier I with the title, CRC in HIV-1 Pathogenesis and Viral Control.
Footnotes
Declarations of interest: Dr. Kityo is the executive director of the JCRC. Miriam Namasinga and Ritah Katasi are also employed by the clinic.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References:
- Adelekan B.… Aliyu A.(2019). Social barriers in accessing care by clients who returned to HIV care after transient loss to follow-up. AIDS Research and Therapy, 16(1), 17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Akullian AN … Babigumira JB (2016). People living with HIV travel farther to access healthcare: a population‐based geographic analysis from rural Uganda. Journal of the International AIDS Society, 19(1), 20171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ammon N.… Corkery JM (2018). Factors impacting antiretroviral therapy adherence among human immunodeficiency virus–positive adolescents in Sub-Saharan Africa: a systematic review. Public Health, 157, 20–31. 10.1016/j.puhe.2017.12.010 [DOI] [PubMed] [Google Scholar]
- Andersen RM (1995). Revisiting the Behavioral Model and Access to Medical Care : Does it Matter ? Journal of Health and Social Behavior, 36(1), 1–10. 10.1007/s [DOI] [PubMed] [Google Scholar]
- Antabe R.… Galaa Y(2020). Utilization of breast cancer screening in Kenya: what are the determinants? BMC Health Services Research, 20(1), 1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Atuoye KN. … Luginaah I.(2015). Can she make it? Transportation barriers to accessing maternal and child health care services in rural Ghana. BMC Health Services Research, 15(1), 333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bacha T.… Worku A.(2012). Predictors of treatment failure and time to detection and switching in HIV-infected Ethiopian children receiving first line anti-retroviral therapy. BMC Infectious Diseases, 12(197), 1–8. 10.1186/1471-2334-12-197 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Billioux VG … Chang LW (2018). HIV viral suppression and geospatial patterns of HIV antiretroviral therapy treatment facility use in Rakai, Uganda. Aids, 32(6), 819–824. 10.1097/QAD.0000000000001761 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boender TS … Bertagnolio S.(2015). Long-term virological outcomes of first-line antiretroviral therapy for HIV-1 in low- and middle-income countries: A systematic review and meta-analysis. Clinical Infectious Diseases, 61(9), 1453–1461. 10.1093/cid/civ556 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brisset-Foucault F.(2013). A citizenship of distinction in the open radio debates of Kampala. Africa, 83(2), 227–250. 10.1017/S0001972013000028 [DOI] [Google Scholar]
- Buor D.(2003). Analysing the primacy of distance in the utilization of health services in the Ahafo‐Ano South district, Ghana. The International Journal of Health Planning and Management, 18(4), 293–311. [DOI] [PubMed] [Google Scholar]
- Burnham KP, & Anderson DR (2004). Multimodel inference: Understanding AIC and BIC in model selection. Sociological Methods and Research, 33(2), 261–304. 10.1177/0049124104268644 [DOI] [Google Scholar]
- Byakika-Tusiime J.… Bangsberg DR (2005). Adherence to HIV antiretroviral therapy in HIV+ Ugandan patients purchasing therapy. International Journal of STD and AIDS, 16(1), 38–41. 10.1258/0956462052932548 [DOI] [PubMed] [Google Scholar]
- Cope AB … Miller WC (2016). Distance to testing sites and its association with timing of HIV diagnosis. AIDS Care, 28(11), 1423–1427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crane JT … Bangsberg DR (2006). The Price of Adherence : Qualitative Findings From HIV Positive Individuals Purchasing Fixed-Dose Combination Generic HIV Antiretroviral Therapy in Kampala, Uganda. 10(4). 10.1007/s10461-006-9080-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dlamini PS … Holzemer WL (2009). HIV Stigma and Missed Medications in HIV-Positive People in Five African Countries. AIDS Patient Care and STDs, 23(5), 377–387. 10.1089/apc.2008.0164 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geleto A.… Loxton D.(2018). Barriers to access and utilization of emergency obstetric care at health facilities in sub-Saharan Africa: a systematic review of literature. Systematic Reviews, 7(1), 183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gesesew HA … Mwanri L.(2017). Significant association between perceived HIV related stigma and late presentation for HIV/AIDS care in low and middle-income countries: A systematic review and meta-analysis. PloS One, 12(3), e0173928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Author 2013. [details removed for peer review]
- Hardon A.… Organization, W. H. (2006). From access to adherence: The challenges of antiretroviral treatment: Studies from Botswana, Tanzania and Uganda 2006. [Google Scholar]
- Heestermans T. … Klipstein-Grobusch K.(2016). Determinants of adherence to antiretroviral therapy among HIV-positive adults in sub-Saharan Africa: a systematic review. BMJ Global Health, 1(4). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hunter JM, & Shannon GW (1985). Jarvis revisited: distance decay in service areas of mid-19th century asylums. The Professional Geographer, 37(3), 296–302. [DOI] [PubMed] [Google Scholar]
- Iyer HS … Adebamowo C.(2020). Neighborhood Greenness and Burden of Non-communicable Diseases in Sub-Saharan Africa: A Multi-country Cross-sectional Study. Environmental Research, 110397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones K, & Moon G.(1993). Medical geography: taking space seriously. Progress in Human Geography, 17(4), 515–524. [Google Scholar]
- Kearns RA (1993). Place and health: towards a reformed medical geography. The Professional Geographer, 45(2), 139–147. [Google Scholar]
- Author 2013. [details removed for peer review]
- Lange JMA … Zewdie D.(2004). What policymakers should know about drug resistance and adherence in the context of scaling-up treatment of HIV infection. Aids, 18(SUPPL. 3), 69–74. 10.1097/00002030-200406003-00013 [DOI] [PubMed] [Google Scholar]
- McLaren ZM … Leibbrandt M.(2014). Distance decay and persistent health care disparities in South Africa. BMC Health Services Research, 14(1), 541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McMahon SA … Kilewo C.(2017). Stigma, facility constraints, and personal disbelief: why women disengage from HIV care during and after pregnancy in Morogoro Region, Tanzania. AIDS and Behavior, 21(1), 317–329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mills EJ … Dybul MR (2012). Engaging Men in Prevention and Care for HIV/AIDS in Africa. PLoS Medicine, 9(2), 1–4. 10.1371/journal.pmed.1001167 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mills EJ … Bangsberg DR (2006). Adherence to antiretroviral therapy in sub-Saharan Africa and North America: A meta-analysis. Journal of the American Medical Association, 296(6), 679–690. 10.1001/jama.296.6.679 [DOI] [PubMed] [Google Scholar]
- Mukherjee JS … Farmer PE (2016). Community-based ART programs: sustaining adherence and follow-up. Current Hiv/Aids Reports, 13(6), 359–366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muyinda H, & Mugisha J.(2015). Stock-outs, uncertainty and improvisation in access to healthcare in war-torn Northern Uganda. Social Science & Medicine, 146, 316–323. [DOI] [PubMed] [Google Scholar]
- Nachega JB. … Schechter M.(2010). Antiretroviral therapy adherence and retention in care in middle-income and low-income countries: current status of knowledge and research priorities. Current Opinion in HIV and AIDS, 5(1), 70–77. 10.1097/COH.0b013e328333ad61 [DOI] [PubMed] [Google Scholar]
- Nemet GF, & Bailey AJ (2000). Distance and health care utilization among the rural elderly. Social Science & Medicine, 50(9), 1197–1208. [DOI] [PubMed] [Google Scholar]
- Peng CYJ … Ingersoll GM (2002). An introduction to logistic regression analysis and reporting. Journal of Educational Research, 96(1), 3–14. 10.1080/00220670209598786 [DOI] [Google Scholar]
- Pham QD … Zhang L.(2014). Global burden of transmitted HIV drug resistance and HIV-exposure categories. AIDS, 28(18), 2751–2762. 10.1097/QAD.0000000000000494 [DOI] [PubMed] [Google Scholar]
- Author 2019. [details removed for peer review]
- Rao D.… Crane HM (2012). A structural equation model of HIV-related stigma, depressive symptoms, and medication adherence. AIDS and Behavior, 16(3), 711–716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reda AA, & Biadgilign S.(2012). Determinants of Adherence to Antiretroviral Therapy among HIV-Infected Patients in Africa. AIDS Research and Treatment, 1–8. 10.1155/2012/574656 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ridgeway K.… McCarraher DR (2018). Interventions to improve antiretroviral therapy adherence among adolescents in low-and middle-income countries: A systematic review of the literature. PloS One, 13(1), e0189770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siedner MJ … Bangsberg DR (2013). GPS-measured distance to clinic, but not self-reported transportation factors, are associated with missed HIV clinic visits in rural Uganda. AIDS, 27(9), 1503–1508. 10.1097/QAD.0b013e32835fd873.GPS-measured [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simandan D.(2016). Proximity, subjectivity, and space: Rethinking distance in human geography. Geoforum, 75, 249–252. [Google Scholar]
- Tuller DM … Weiser SD (2010). Transportation costs impede sustained adherence and access to HAART in a clinic population in Southwestern Uganda: A qualitative study. AIDS and Behavior, 14(4), 778–784. 10.1007/s10461-009-9533-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turmen T.(2003). Gender and HIV/AIDS. International Journal of Gynaecology and Obstetrics: The Official Organ of the International Federation of Gynaecology and Obstetrics, 82, 411–418. 10.1016/S0020-7292Z03.00202-9 [DOI] [PubMed] [Google Scholar]
- UNAIDS. (2018). Country Fact Sheets: Uganda, 2018. HIV and AIDS Estimates. https://www.unaids.org/en/regionscountries/countries/uganda [Google Scholar]
- Vermeiren K.… Mukwaya P.(2012). Urban growth of Kampala, Uganda: Pattern analysis and scenario development. Landscape and Urban Planning, 106(2), 199–206. 10.1016/j.landurbplan.2012.03.006 [DOI] [Google Scholar]
- Wanyenze R.… Bangsberg DR (2006). HIV counseling and testing practices at an urban hospital in Kampala, Uganda. AIDS and Behavior, 10(4), 361–367. 10.1007/s10461-005-9035-9 [DOI] [PubMed] [Google Scholar]
- Weise SD. … Richard M.(2003). Barriers to Antiretroviral Adherence for Patients Liing with HIV Infection and AIDS in Botswana. Journal of Acquired Immune Deficiency Syndromes, 34(3), 281–288. [DOI] [PubMed] [Google Scholar]
- Windisch R.… de Savigny D.(2011). Scaling up antiretroviral therapy in Uganda: using supply chain management to appraise health systems strengthening. Globalization and Health, 7(1), 25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- World Bank. (2018). Antiretroviral therapy coverage (% of people living with HIV). https://data.worldbank.org/indicator/SH.HIV.ARTC.ZS?end=2017&locations=UG&start=2 000&view=chart
- Zolopa AR (2010). The evolution of HIV treatment guidelines: Current state-of-the-art of ART. Antiviral Research, 85(1), 241–244. 10.1016/j.antiviral.2009.10.018 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.