Abstract
Purpose
Various blood cell parameters have been identified as predictive markers of tumor responses and the survival of patients with cancer treated with immune checkpoint inhibitors. The purpose of this study is to assess the ability of various blood cell parameters to predict therapeutic effects and survival in patients with esophageal squamous cell carcinoma (ESCC) treated with nivolumab monotherapy.
Patients and Methods
We evaluated neutrophil-to-lymphocyte (NLR), platelet-to-lymphocyte (PLR) and lymphocyte-to-monocyte (LMR) ratios as predictive markers of patients’ survival and effects of nivolumab monotherapy after one or more prior chemotherapies for unresectable advanced or recurrent ESCC.
Results
The objective response and disease control rates were 20.3% and 47.5%, respectively. The LMRs before, and 14 and 28 days after nivolumab initiation were significantly higher in patients with complete response (CR)/partial response (PR)/stable disease (SD) than those with progressive disease (PD). The NLRs at 14 and 28 days after nivolumab initiation were significantly lower in patients with CR/PR/SD than with PD. The optimal cutoffs for these parameters significantly discriminated patients with CR/PR/SD and PD. Univariate and multivariate analyses identified pretreatment NLRs as a significant independent factor for progression-free and overall survival (hazard ratio [HR]: 1.19, 95% confidence interval [CI]: 1.07–1.32, and HR 1.23, 95% CI: 1.11–1.37, respectively; p ≤ 0.001 for both).
Conclusion
The pretreatment LMRs, and NLR and LMR at 14 and 28 days after starting nivolumab monotherapy were significantly associated with the clinical therapeutic effect. The pretreatment NLR was significantly associated with patients’ survival. These blood cell parameters before and during the early days of nivolumab monotherapy can help to identify patients with ESCC who would most likely benefit from nivolumab monotherapy.
Keywords: biomarkers, cancer, esophagus, immunotherapy, response, survival
Introduction
The results of the ATTRACTION-3 trial revealed that nivolumab is associated with a significant improvement in overall survival and a favorable safety profile compared with taxane therapy in patients with unresectable advanced or recurrent esophageal squamous cell carcinoma (ESCC), who were previously refractory or intolerant to a previous course of fluoropyrimidine-based and platinum-based chemotherapy.1 Furthermore, nivolumab significantly improved the survival of patients with esophageal cancer in the postoperative adjuvant setting and as first-line therapy for unresectable advanced or recurrent ESCC.2,3 Hence, it is currently being applied to treat esophageal cancer.
Although nivolumab is a key drug for treating ESCC, the response rate to nivolumab monotherapy is relatively low at ~ 20%.1,4 Rapid tumor progression after initiating immune checkpoint inhibitors (ICIs) in defined proportions is generally a concern for various types of cancer.5,6 Although the expression of programmed death molecule ligand 1 (PD-L1), such as tumor proportion (TPS) and combined positive (CPS) scores, provide some degree of indication regarding therapeutic effects and survival,7–10 its clinical application remains insufficient. Therefore, further exploration of predictive biomarkers of immunotherapy response and survival is crucially important.
Blood cell parameters such as neutrophil-to-lymphocyte, platelet-to-lymphocyte and lymphocyte-to-monocyte ratios (NLRs, PLRs and LMRs, respectively) are predictive markers of therapeutic effects and survival rates among patients with various types of cancer treated with ICIs.11–18 However, to the best of our knowledge, correlations among blood parameters, therapeutic effects and survival have not been fully evaluated in patients with ESCC under nivolumab monotherapy. Here, we evaluated the ability of various clinical factors, including NLR, PLR and LMR, to predict therapeutic effects and survival in patients with unresectable advanced or recurrent ESCC treated with second- or later-line nivolumab monotherapy.
Patients and Methods
Patients
We reviewed 64 consecutive patients with ESCC who underwent nivolumab monotherapy after at least one prior chemotherapeutic regimen at our institution between June 2016 and December 2021. Histological tumor types were diagnosed as ESCC in biopsy samples obtained before treatment or during resection. Seven patients had participated in the ATTRACTION-3 trial.1 We excluded four patients who received only a single dose of nivolumab and in whom its clinical effects could not be evaluated due to rapid cancer progression that led to significant deterioration of their general condition, and one with concurrent advanced rectal cancer. Therefore, we assessed factors associated with therapeutic effect and survival in 59 patients. The Institutional Review Board at Hiroshima University approved this study (Approval no. E-2225), which complied with the Declaration of Helsinki (2013). The requirement for informed consent was waived because this study was a retrospective review of a patient database and all data were anonymized and are presented in aggregates.
Nivolumab Therapy and Clinical Response Assessment
We intravenously administered 53 patients with nivolumab (240 mg) over a period of 30 min at 2-week intervals for 6 weeks. Six patients preferred to receive nivolumab (480 mg) every 4 weeks. Clinical responses to nivolumab were assessed in all patients by computed tomography (CT) imaging during the three courses. Responses were evaluated by CT sooner for patients who had large tumor volumes or poor overall condition. Patients with worsening symptoms were also examined at the discretion of the attending physician. Clinical tumor responses to nivolumab were judged as complete response (CR), partial response (PR), stable disease (SD) and progressive disease (PD) according to the Response Evaluation Criteria in Solid Tumors (RECIST).19 All patients had measurable target regions such as lymph node and/or organ metastasis.
NLR, PLR, and LMR Data
We assessed hematological toxicity before administering each cycle of nivolumab. We evaluated blood cell parameters immediately before and 14 and 28 days after starting nivolumab therapy. We could not obtain data at 2 weeks from the six patients who receive nivolumab (480 mg) every 4 weeks. Therefore, blood cell makers on day 14 were evaluated in 53 patients. The blood cell markers NLR, PLR, and LMR were defined as the absolute neutrophil count divided by the absolute lymphocyte count, the absolute platelet count divided by the absolute lymphocyte count, and the absolute lymphocyte count divided by the absolute monocyte count, respectively.
Statistical Analysis
Categorical variables were analyzed using χ2 tests, and clinical therapeutic effects were compared using continuous variables analyzed by unpaired t-tests. The optimal cutoffs of each blood cell parameter for predicting therapeutic effects were determined from receiver operating characteristic (ROC) curves. The optimal cutoff of the pretreatment NLR for survival was also determined by ROC analysis of life-and death predictions at one-year after the patients started on nivolumab.
Survival outcomes were evaluated in November 2022. Progression-free survival (PFS) was defined as the interval from starting nivolumab until the first adverse event (tumor progression or death from any cause) or the most recent followup. Overall survival (OS) was defined as the interval between starting nivolumab until death due to any cause or the most recent follow-up. Survival data were analyzed using Kaplan-Meier curves and compared using Log rank tests. The effects of various clinicopathological and blood cell parameters on survival were evaluated using univariate analyses, and independent predictors of survival were determined using multivariate Cox proportional hazards analyses with forward stepwise selection. Statistical significance was set at p < 0.05. All data were statistically analyzed using SPSS version 27 (IBM Corp., Armonk, NY, USA).
Results
Characteristics of the Patients
Table 1 shows the characteristics of the patients at the time of nivolumab initiation (male, n = 52; female, n = 7; mean age, 69.4 ± 8.3 years) and best clinical responses to nivolumab. The clinical responses were CR in 1 (1.7%), PR in 11 (18.6%), SD in 16 (27.1%) and PD in 31 (52.5%) patients. Therefore, the objective response (CR+PR; ORR) and disease control (CR+PR+SD, DCR) rates were 20.3% and 47.5%, respectively.
Table 1.
Characteristics of Patients
| Parameters | n = 59 |
|---|---|
| Mean age (± standard deviation, y) | 69.4 ± 8.3 |
| Gender | |
| Male | 52 (88.1%) |
| Female | 7 (11.9%) |
| Performance statusa | |
| 0 | 32 (54.2%) |
| 1 | 24 (40.7%) |
| 2 | 3 (5.1%) |
| Body mass index | 19.5 ± 3.4 |
| Smoking history | |
| + | 49 (83.1%) |
| – | 10 (16.9%) |
| Primary tumor location | |
| Cervical | 13 (22.0%) |
| Upper third | 8 (13.6%) |
| Middle third | 26 (44.1%) |
| Lower third | 12 (20.3%) |
| Tumor differentiation | |
| Well differentiated | 5 (8.5%) |
| Moderate differentiated | 28 (47.5%) |
| Poorly differentiated | 16 (27.1%) |
| SCC (not assessable) | 10 (16.9%) |
| Diseases status | |
| Postoperative recurrence | 23 (39.0%) |
| Unresectable advanced | 36 (61.0%) |
| Prior radiation therapy | |
| + | 49 (83.1%) |
| – | 10 (16.9%) |
| Number of previous chemotherapiesb | |
| 1 | 46 (78.0%) |
| 2 | 9 (15.3%) |
| ≥3 | 4 (6.8%) |
| Number of organs with metastasisc | |
| 1 | 24 (40.7%) |
| 2 | 24 (40.7%) |
| ≥3 | 11 (18.6%) |
| Best clinical responsed | |
| CR | 1 (1.7%) |
| PR | 11 (18.6%) |
| SD | 16 (27.1%) |
| PD | 31 (52.5%) |
Notes: All values except age are shown as n (%). aEastern Cooperative Oncology Group performance status. bNumber of treatment regimens from diagnosis of unresectable advanced or recurrent ESCC until starting nivolumab. cAt the time of nivolumab initiation. dAccording to the Response Evaluation Criteria in Solid Tumors.
Abbreviations: CR, complete response; PD, progressive disease; PR, partial response; SCC, squamous cell carcinoma; SD, stable disease.
Blood Cell Parameters
Supplementary Table 1 shows the results of blood tests before (day 0) and days 14 and 28 after nivolumab administration. The values (mean ± SD) of neutrophils, lymphocytes, monocytes and platelet counts at each time of administration were determined, and NLR, PLR, and the LMR were calculated based on these measured values.
Comparison of Clinical Data According to Therapeutic Effect
We compared various clinical parameters among the patients with CR/PR/SD (n = 28, 47.5%) and PD (n = 31, 52.5%) (Supplementary Table 2). None of these clinical parameters, significantly differed.
Comparison of Blood Cell Parameters According to Therapeutic Effect
Table 2 shows a comparison of blood cell parameters between patients with CR/PR/SD and PD. The NLRs at 14 and 28 days after nivolumab initiation were significantly lower in patients with CR/PR/SD than with PD (4.5 ± 2.8 versus [vs] 6.8 ± 3.9, p = 0.019 and 3.9 ± 1.9 vs 8.1 ± 5.8, p = 0.001, respectively). The LMR before, at 14 and 28 days after nivolumab initiation were significantly higher in patients with CR/PR/SD than those with PD (3.1 ± 1.6 vs 2.3 ± 1.0, p = 0.021; 2.9 ± 1.8 vs 2.0 ± 0.7, p = 0.019 and 3.0 ± 1.4 vs 2.0 ± 0.9, p = 0.001, respectively).
Table 2.
Comparison of Blood Cell Parameters According to Therapeutic Effects
| Pretreatment | |||
|---|---|---|---|
| Parameters | CR/PR/SD (n = 28) | PD (n = 31) | p |
| NLR | 4.1± 2.3 | 5.6 ± 3.6 | 0.056 |
| LMR | 3.1 ± 1.6 | 2.3 ± 1.0 | 0.021 |
| PLR | 269 ± 126 | 316 ± 197 | 0.280 |
| CEA (ng/mL) | 4.0 ± 3.0 | 26.9 ± 116.1 | 0.302 |
| SCC (ng/mL) | 2.4 ± 3.3 | 5.6 ± 11.5 | 0.146 |
| 14 days after nivolumab initiation | |||
| Parameters | CR/PR/SD (n = 26) | PD (n = 27) | p |
| NLR | 4.5 ± 2.8 | 6.8 ± 3.9 | 0.019 |
| LMR | 2.9 ± 1.8 | 2.0 ± 0.7 | 0.019 |
| PLR | 286 ± 159 | 288 ± 147 | 0.962 |
| 28 days after nivolumab initiation | |||
| Parameters | CR/PR/SD (n = 28) | PD (n = 31) | p |
| NLR | 3.9 ± 1.9 | 8.1 ± 5.8 | 0.001 |
| LMR | 3.0 ± 1.4 | 2.0 ± 0.9 | 0.001 |
| PLR | 270 ± 141 | 372 ± 354 | 0.160 |
Note: All values are shown as means ± standard deviation.
Abbreviations: CR, complete response; LMR, lymphocyte-to-monocyte ratio; NLR, neutrophil-to-lymphocyte ratio; PD, progressive disease; PLR, platelet-to-lymphocyte ratio; PR, partial response; SD, stable disease.
Optimal Cutoffs to Predict Therapeutic Effects Using Blood Cell Parameters
We plotted the sensitivity and specificity of predicting clinical therapeutic effects (CR/PR/SD vs PD) at the levels of the above significant NLRs and LMRs (Supplementary Figure 1). Based on the ROC curves, the optimal cutoff for pretreatment LMR to predict PD was 3.5 (Supplementary Figure 1A). The optimal cutoff points of NLR and LMR on day 14 after initiating nivolumab were 5.8 (Supplementary Figure 1B) and 2.8 (Supplementary Figure 1C), respectively. The optimal cutoffs for NLR and LMR 28 days after starting nivolumab were 5.6 (Supplementary Figure 1D) and 2.9 (Supplementary Figure 1E), respectively.
Clinical Tumor Response According to Optimal Cutoff Values of NLR and LMR
Table 3 shows the clinical therapeutic effects according to above optimal cutoffs of NLR and LMR. Pretreatment LMR was a significant predictive marker of therapeutic effect, as 40.4% and 75.0% of patients with pretreatment LMR < 3.5 vs ≥ 3.5, respectively achieved CR/PR/SD (p = 0.03).
Table 3.
Clinical Therapeutic Effects According to Optimal Cutoffs
| Parameters | Optimal Cutoff | CR/PR/SD n = 28 | PD n = 31 | p |
| Pretreatment LMR | < 3.5 (n = 47, %) | 19 (40.4%) | 28 (59.6) | 0.03 |
| ≥ 3.5 (n = 12, %) | 9 (75.0%) | 3 (25.0%) | ||
| Parametersa | Optimal cutoff | CR/PR/SD n = 26 | PD n = 27 | p |
| NLR day 14 | < 5.8 (n = 32, %) | 20 (62.5%) | 12 (37.5%) | 0.02 |
| ≥ 5.8 (n = 21, %) | 6 (28.6%) | 15 (71.4%) | ||
| LMR day 14 | < 2.8 (n = 39, %) | 15 (38.5%) | 24 (61.5%) | 0.01 |
| ≥ 2.8 (n = 14, %) | 11 (78.6%) | 3 (21.4%) | ||
| Parametersa | Optimal cutoff | CR/PR/SD n = 28 | PD n = 31 | p |
| NLR day 28 | < 5.6 (n = 38, %) | 25 (65.8%) | 13 (34.2%) | 0.0001 |
| ≥ 5.6 (n = 21, %) | 3 (14.3%) | 18 (85.7%) | ||
| LMR day 28 | < 2.9 (n = 40, %) | 13 (32.5%) | 27 (67.5%) | 0.001 |
| ≥ 2.9 (n = 19, %) | 15 (78.9%) | 4 (21.1%) |
Note: aDays after starting nivolumab monotherapy.
Abbreviations: CR, complete response; LMR, lymphocyte-to-monocyte ratio; NLR, neutrophil-to-lymphocyte ratio; PD, progressive disease; PLR, platelet-to-lymphocyte ratio; PR, partial response; SD, stable disease.
The NLR on day 14 was also a significant predictor of therapeutic effects, and 62.5% vs 28.6% of patients with NLR of < 5.8 and ≥ 5.8 on day 14 respectively achieved CR/PR/SD (p = 0.02). The LMR day 14 was also a significant predictive marker for therapeutic effect, and 38.5% vs 78.6% of patients on day 14 had LMRs of < 2.8 and ≥ 2.8, respectively, achieved CR/PR/SD (p = 0.01).
The NLR and LMR on day 28 were significant predictive markers of a therapeutic effect; 65.8% vs 14.3% of patients with NLRs < 5.6 and ≥ 5.6, respectively (p = 0.0001), and 32.5% vs 78.9% of those with LMRs < 2.9 and ≥ 2.9, respectively, (p = 0.001) achieved CR/PR/SD.
Univariate and Multivariate Analyses of Survival Predictors
Univariate analysis of PFS (Supplementary Table 3) showed that serum carcinoembryonic antigen (CEA) and squamous cell carcinoma (SCC) antigen values, liver metastasis, NLR, LMR and PLR were significant prognostic factors associated with PFS. Furthermore, multivariate analysis identified SCC (hazard ratio [HR]: 1.04, 95% confidence interval [CI]: 1.01–1.07, p = 0.02), liver metastasis (HR: 2.44, 95% CI: 1.23–4.84, p = 0.01) and NLR (HR: 1.19, 95% CI: 1.07–1.32, p = 0.001) as significant independent covariates for PFS.
Univariate analysis of OS (Table 4) revealed that PS, CEA and SCC values, liver metastasis, NLR, LMR and PLR were significant prognostic factors associated with OS. Furthermore, multivariate analysis subsequently identified SCC level (HR: 1.05, 95% CI: 1.02–1.09, p = 0.002) and NLR (HR: 1.23, 95% CI: 1.11–1.37, p < 0.001) as significant independent covariates for OS.
Table 4.
Univariate and Multivariate Analyses of Overall Survival
| Variables | Univariate | Multivariate | ||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p | HR | 95% CI | p | |
| Mean age (± SD, y) | 1.01 | 0.98–1.04 | 0.62 | |||
| Gender | ||||||
| Male | 1 | |||||
| Female | 0.74 | 0.29–1.87 | 0.52 | |||
| Performance statusa | ||||||
| 0 | 1 | |||||
| 1, 2 | 2.11 | 1.18–3.80 | 0.01 | |||
| Body mass index (continuous) | 1.00 | 0.91–1.09 | 0.93 | |||
| Smoking history | ||||||
| – | 1 | |||||
| + | 1.23 | 0.55–2.75 | 0.61 | |||
| Primary tumor location | ||||||
| Cervical and Upper third | 1 | |||||
| Middle and lower third | 0.91 | 0.50–1.66 | 0.76 | |||
| Diseases status | ||||||
| Postoperative recurrence | 1 | |||||
| Unresectable advanced | 1.12 | 0.62–2.02 | 0.72 | |||
| Previous radiation therapy | ||||||
| – | 1 | |||||
| + | 1.18 | 0.53–2.64 | 0.69 | |||
| Pretreatment CEA (continuous) | 1.004 | 1.001–1.01 | 0.01 | |||
| Pretreatment SCC (continuous) | 1.06 | 1.02–1.09 | 0.001 | 1.05 | 1.02–1.09 | 0.002 |
| Tumor differentiation | ||||||
| Others | 1 | |||||
| Poorly differentiated | 1.01 | 0.53–1.93 | 0.98 | |||
| Number of previous chemotherapiesb | ||||||
| 1 | 1 | |||||
| ≥ 2 | 0.48 | 0.23–1.01 | 0.054 | |||
| Number of organs with metastasisc | ||||||
| 1 | 1 | |||||
| 2 | 1.42 | 0.78–2.60 | 0.25 | |||
| Lymph node metastasis | ||||||
| – | 1 | |||||
| + | 0.83 | 0.43–1.60 | 0.58 | |||
| Lung metastasis | ||||||
| – | 1 | |||||
| + | 0.86 | 0.48–1.53 | 0.61 | |||
| Liver metastasis | ||||||
| – | 1 | |||||
| + | 2.31 | 1.18–4.52 | 0.02 | |||
| Bone metastasis | ||||||
| – | 1 | |||||
| + | 1.24 | 0.55–2.77 | 0.61 | |||
| Pretreatment blood cell markers | ||||||
| NLR (continuous) | 1.23 | 1.11–1.37 | < 0.001 | 1.23 | 1.11–1.37 | < 0.001 |
| LMR (continuous) | 0.70 | 0.54–0.91 | 0.01 | |||
| PLR (continuous) | 1.003 | 1.001–1.01 | 0.01 | |||
Notes: aEastern Cooperative Oncology Group performance status. bNumber of treatments from the diagnosis of unresectable advanced or recurrent ESCC until starting nivolumab. cAt the time of nivolumab initiation.
Abbreviations: CEA, Carcinoembryonic antigen; LMR, lymphocyte-to-monocyte ratio; NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio; SCC, squamous cell carcinoma-related antigen.
Survival Rates of Patients Under Nivolumab Monotherapy
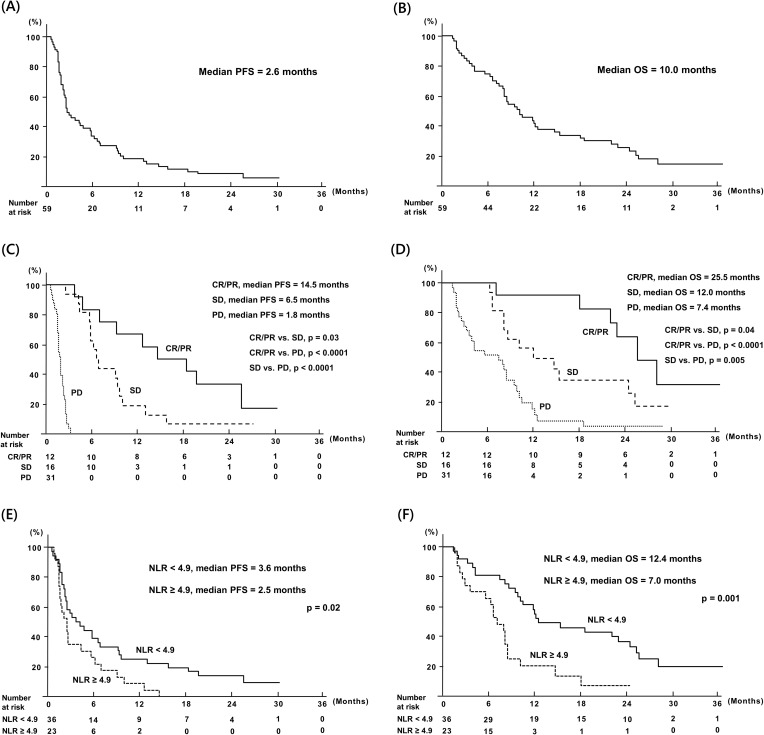
The 1- and 2-year PFS and OS rates of all patients were 18.6% and 8.5%, respectively, and 41.2% and 25.2%, respectively (Figure 1A and B).
Figure 1.
Survival rates of patients under nivolumab monotherapy. (A) PFS of all patients. (B) OS of all patients. (C) PFS according to tumor responses. (D) OS according to tumor responses. (E) PFS according to according to optimal cutoff of pretreatment NLR. (F) OS according to according to optimal cutoff of pretreatment NLR.
Abbreviations: NLR, neutrophil-to-lymphocyte; OS, overall survival; PFS, progression-free survival.
The 1- and 2-year PFS rates of patients with CR/PR and with SD were 66.7% and 33.3%, respectively, and 18.8% and 6.3%, respectively. The 1-year PFS rate of patients with PD was 0.0% (CR/PR vs SD, p = 0.03; CR/PR vs PD, p < 0.0001; SD vs PD, p < 0.0001; Figure 1C). Furthermore, the 1- and 2-year OS rates of patients with CR/PR, SD, and PD were 91.7% and 64.2%, 56.3% and 35.2%, and 15.6% and 3.9%, respectively (CR/PR vs SD, p = 0.04; CR/PR vs PD, p < 0.0001; SD vs PD, p = 0.005; Figure 1D).
Survival Rates According to Pretreatment NLR
The optimal pretreatment NLR cutoff for predicting life and death at one year after nivolumab initiation was determined by ROC analysis, because pretreatment NLR was a significant independent blood cell parameter for PFS and OS. The optimal cutoff for pretreatment NLR was 4.9 (AUC = 0.75; 95% CI, 0.63–0.88; p = 0.001), and the value was similar to the mean value of pretreatment NLR.
The 1- and 2-year PFS rates of patients with pretreatment NLRs < 4.9 and ≥ 4.9 were 25.0% and 13.9%, and 8.7% and 0.0%, respectively (p = 0.02; Figure 1E). Furthermore, 1- and 2-year OS rates of patients with pretreatment NLRs < 4.9 and ≥ 4.9 were 57.7% and 36.0%, and 19.6% and 6.5%, respectively (p = 0.001, Figure 1F). The PFS and OS significantly differed between patients with pretreatment NLRs < 4.9 and ≥ 4.9.
Discussion
The numbers of patients with metastatic ESCC that is currently being clinically treated with ICIs are increasing. Nivolumab significantly improved the survival of patients with esophageal cancer in the postoperative adjuvant setting and as therapy for unresectable advanced or recurrent tumors.1–3 We evaluated the significance of NLRs, PLRs and LMRs as predictive markers of therapeutic effects and the survival of patients with unresectable advanced or recurrent ESCC who underwent second- or later-line nivolumab monotherapy. Although various pretreatment clinical factors were not associated with therapeutic effect, pretreatment LMRs, and NLR and LMR at 14 and 28 days after starting nivolumab monotherapy were significantly associated with the clinical therapeutic effect. Furthermore, pretreatment NLR was significantly associated with PFS and OS.
The ORR and DCR in the present study were 20.3% and 47.5%, respectively, and median PFS and OS were 2.6 and 10.0 months, respectively. The ORR and DCR in the ATTRACTION-3 trial were 19.3% and 37.4%, respectively, and the median PFS and OS were 1.7 and 10.9 months.1 Furthermore, the ORR and DCR were 22.4% and 41.1%, respectively, and median PFS and OS were 2.7 and 13.4 months in the Japanese subpopulation from the ATTRACTION-3 trial.20 Some patients included in the present study had a poor PS (PS 2) or had undergone 2 or ≥ 3 chemotherapy regimens before nivolumab administration, might have had correlations among a poor prognosis, poor general status and a potentially poor response to treatment. However, the overall tumor response and prognosis in the present study were relatively in line with those of the ATTRACTION-3 trial.
Neutrophils play essential roles in tumor development and progression. They can be mechanistically recruited to the tumor microenvironment, secrete proliferative factors, and suppress T-lymphocyte activity, thus promoting tumor angiogenesis, invasion, and metastasis.21–24 Lymphocytes comprise a major component of the immune system. They can inhibit tumor proliferation and metastasis via cytotoxicity, thereby participating in tumor surveillance and defense.25 Monocytes are also key immune cells, as they can change the tumor microenvironment (TME) through local immunosuppression and facilitating angiogenesis, thereby promoting the progression of cancer.26 Therefore, the NLR and LMR are systemic inflammation indicators of the balance between an antitumor immune response and protumor inflammation, and are indicators of a tumor response and the prognosis of various types of cancers treated by ICIs.11–15,18 We also found that the NLR and LMR before and during the early days of nivolumab monotherapy were significantly associated with the clinical therapeutic effect and survival for unresectable advanced or recurrent ESCC.
The various pretreatment clinical factors were not associated with clinical therapeutic effect. However, we found that pretreatment LMR, as well as NLR and LMR on days 14 and 28 after starting nivolumab monotherapy were significantly associated with the therapeutic effect. The optimal cutoffs for these blood parameters clearly discriminated patients with CR/PR/SD from those with PD. These results suggested that nivolumab tends to be effective for patients with a high LMR before starting treatment. Therefore, pretreatment LMR might be a useful marker for using nivolumab to treat ESCC. Furthermore, nivolumab was relatively ineffective for patients with high NLRs or low LMRs on days 14 and 28 after nivolumab initiation. Therefore, early image assessment should be considered for patients with high NLR or low LMR at 14 and 28 days after starting nivolumab to avoid unnecessary additional administration to those with a potentially poor response.
Pretreatment NLR is a prognostic indicator for various types of cancer treated by ICIs. The reported cutoffs of pretreatment NLRs required to significantly stratify survival range from 3 to 7 in these studies,11–13,18 and 3.84, 5.0 and 6.0 for unresectable advanced and recurrent esophageal cancer, although the reports11–13 included patients with esophageal adenocarcinoma treated by some types of ICI or by combination chemotherapies.27–29 The optimal cutoff for pretreatment NLRs required to significantly stratify the survival of patients with ESCC who underwent nivolumab monotherapy in the present study was 4.9. Our cutoff value of NLR was within the range for those of esophageal and other types of cancer.11–13,18,27–29
We found here that PLRs before, and on 14 and 28 days after treatment were not significant predictors of a clinical therapeutic effect and survival. However, others have shown that PLRs are important predictive markers of an ESCC tumor response to a PD-1 inhibitor and survival.29,30 These studies combined the same anti-PD-1 antibody (camrelizumab) with chemotherapy for some patients, and administered it in a neoadjuvant setting for resectable ESCC or as treatment for advanced metastatic ESCC. Not only the PLR but also other parameters, such as NLR, LMR and the systemic immune-inflammation index (PLT multiplied by the NLR), have also been identified as potentially effective predictors of tumor response and survival.29,30 Optimal blood parameters and cutoff values might differ somewhat depending on the type of ICI, the treatment setting or the presence or absence of combination chemotherapy.
The present study has some limitations, one of which is the single-center, retrospective design. Others are that some patients were administered nivolumab (480 mg) every 4 weeks, and blood data were not collected from these patients on day 14. However, our findings are important for selecting the appropriate treatment with which to manage individual patients with unresectable advanced or recurrent ESCC who might benefit from nivolumab monotherapy. Furthermore, blood parameters are clinically very useful, because they are uniformly available, recursive, inexpensive, and minimally invasive. Larger, prospective, multicenter studies are needed to confirm our findings and establish an optimal approach to using NLR and LMR to accurately predict responses and the survival of patients with ESCC who will undergo nivolumab therapy in the future.
Conclusion
In conclusion, pretreatment LMR, and NLR and LMR at 14 and 28 days after starting nivolumab monotherapy were significantly associated with a clinical therapeutic effect. Baseline NLRs were significantly associated with survival in patients with unresectable advanced or recurrent ESCC who received nivolumab monotherapy. Therefore, these blood cell parameters might help to identify patients who would most likely benefit from ICI.
Acknowledgments
The Authors thank all the staff at the Department of Surgical Oncology, Hiroshima University, for providing valuable advice regarding the preparation of this article.
Disclosure
All authors report no conflicts of interest in this work.
References
- 1.Kato K, Cho BC, Takahashi M, et al. Nivolumab versus chemotherapy in patients with advanced oesophageal squamous cell carcinoma refractory or intolerant to previous chemotherapy (ATTRACTION-3): a multicentre, randomised, open-label, Phase 3 trial. Lancet Oncol. 2019;20:1506–1517. doi: 10.1016/S1470-2045(19)30626-6 [DOI] [PubMed] [Google Scholar]
- 2.Kelly RJ, Ajani JA, Kuzdzal J, et al. Adjuvant nivolumab in resected esophageal or gastroesophageal junction cancer. N Engl J Med. 2021;384:1191–1203. doi: 10.1056/NEJMoa2032125 [DOI] [PubMed] [Google Scholar]
- 3.Doki Y, Ajani JA, Kato K, et al. Nivolumab combination therapy in advanced esophageal squamous-cell carcinoma. N Engl J Med. 2022;386:449–462. doi: 10.1056/NEJMoa2111380 [DOI] [PubMed] [Google Scholar]
- 4.Kudo T, Hamamoto Y, Kato K, et al. Nivolumab treatment for oesophageal squamous-cell carcinoma: an open-label, multicentre, Phase 2 trial. Lancet Oncol. 2017;18:631–639. doi: 10.1016/S1470-2045(17)30181-X [DOI] [PubMed] [Google Scholar]
- 5.Kim JY, Lee KH, Kang J, et al. Hyperprogressive disease during anti-PD-1 (PDCD1)/PD-L1 (CD274) therapy: a systematic review and meta-analysis. Cancers. 2019;11:1699. doi: 10.3390/cancers11111699 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Toki MI, Syrigos N, Syrigos K. Hyperprogressive disease: a distinct pattern of progression to immune checkpoint inhibitors. Int J Cancer. 2021;149:277–286. doi: 10.1002/ijc.33429 [DOI] [PubMed] [Google Scholar]
- 7.Leone AG, Petrelli F, Ghidini A, Raimondi A, Smyth EC, Pietrantonio F. Efficacy and activity of PD-1 blockade in patients with advanced esophageal squamous cell carcinoma: a systematic review and meta-analysis with focus on the value of PD-L1 combined positive score. ESMO Open. 2022;7:100380. doi: 10.1016/j.esmoop.2021.100380 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Yoon HH, Jin Z, Kour O, et al. Association of PD-L1 expression and other variables with benefit from immune checkpoint inhibition in advanced gastroesophageal cancer: systematic review and meta-analysis of 17 phase 3 randomized clinical trials. JAMA Oncol. 2022;8:1456–1465. doi: 10.1001/jamaoncol.2022.3707 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Shiraishi T, Toyozumi T, Sakata H, et al. Soluble PD-L1 concentration is proportional to the expression of PD-L1 in tissue and is associated with a poor prognosis in esophageal squamous cell carcinoma. Oncology. 2022;100:39–47. doi: 10.1159/000518740 [DOI] [PubMed] [Google Scholar]
- 10.Okadome K, Baba Y, Nomoto D, et al. Prognostic and clinical impact of PD-L2 and PD-L1 expression in a cohort of 437 oesophageal cancers. Br J Cancer. 2020;122:1535–1543. doi: 10.1038/s41416-020-0811-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ota Y, Takahari D, Suzuki T, et al. Changes in the neutrophil-to-lymphocyte ratio during nivolumab monotherapy are associated with gastric cancer survival. Cancer Chemother Pharmacol. 2020;85:265–272. doi: 10.1007/s00280-019-04023-w [DOI] [PubMed] [Google Scholar]
- 12.Sacdalan DB, Lucero JA, Sacdalan DL. Prognostic utility of baseline neutrophil-to-lymphocyte ratio in patients receiving immune checkpoint inhibitors: a review and meta-analysis. Onco Targets Ther. 2018;11:955–965. doi: 10.2147/OTT.S153290 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Takenaka Y, Oya R, Takemoto N, Inohara H. Neutrophil-to-lymphocyte ratio as a prognostic marker for head and neck squamous cell carcinoma treated with immune checkpoint inhibitors: meta-analysis. Head Neck. 2022;44:1237–1245. doi: 10.1002/hed.26997 [DOI] [PubMed] [Google Scholar]
- 14.Sekine K, Kanda S, Goto Y, et al. Change in the lymphocyte-to-monocyte ratio is an early surrogate marker of the efficacy of nivolumab monotherapy in advanced non-small-cell lung cancer. Lung Cancer. 2018;124:179–188. doi: 10.1016/j.lungcan.2018.08.012 [DOI] [PubMed] [Google Scholar]
- 15.Xiao L, Li L, Chen G, Zhang Y, Gao Q. The lymphocyte-to-monocyte ratio could predict the efficacy of PD-1 inhibitors in patients with advanced cancer. Transl Cancer Res. 2020;9:4111–4120. doi: 10.21037/tcr-20-1451 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhou K, Cao J, Lin H, et al. Prognostic role of the platelet to lymphocyte ratio (PLR) in the clinical outcomes of patients with advanced lung cancer receiving immunotherapy: a systematic review and meta-analysis. Front Oncol. 2022;12:962173. doi: 10.3389/fonc.2022.962173 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kurashina R, Ando K, Inoue M, et al. Platelet-to-lymphocyte ratio predicts the efficacy of pembrolizumab in patients with urothelial carcinoma. Anticancer Res. 2022;42:1131–1136. doi: 10.21873/anticanres.15576 [DOI] [PubMed] [Google Scholar]
- 18.Cheng M, Li G, Liu Z, Yang Q, Jiang Y. Pretreatment neutrophil-to-lymphocyte ratio and lactate dehydrogenase predict the prognosis of metastatic cervical cancer treated with combination immunotherapy. J Oncol. 2022;2022:1828473. doi: 10.1155/2022/1828473 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45:228–247. doi: 10.1016/j.ejca.2008.10.026 [DOI] [PubMed] [Google Scholar]
- 20.Takahashi M, Kato K, Okada M, et al. Nivolumab versus chemotherapy in Japanese patients with advanced esophageal squamous cell carcinoma: a subgroup analysis of a multicenter, randomized, open-label, phase 3 trial (ATTRACTION-3). Esophagus. 2021;18:90–99. doi: 10.1007/s10388-020-00794-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bonavita E, Galdiero MR, Jaillon S, Mantovani A. Phagocytes as corrupted policemen in cancer-related inflammation. Adv Cancer Res. 2015;128:141–171. [DOI] [PubMed] [Google Scholar]
- 22.Kusumanto YH, Dam WA, Hospers GA, Meijer C, Mulder NH. Platelets and granulocytes, in particular the neutrophils, form important compartments for circulating vascular endothelial growth factor. Angiogenesis. 2003;6:283–287. doi: 10.1023/B:AGEN.0000029415.62384.ba [DOI] [PubMed] [Google Scholar]
- 23.Uribe-Querol E, Rosales C. Neutrophils in cancer: two sides of the same coin. J Immunol Res. 2015;2015:983698. doi: 10.1155/2015/983698 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Xiong S, Dong L, Cheng L. Neutrophils in cancer carcinogenesis and metastasis. J Hematol Oncol. 2021;14:173. doi: 10.1186/s13045-021-01187-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mantovani A, Allavena P, Sica A, Balkwill F. Cancer-related inflammation. Nature. 2008;454:436–444. doi: 10.1038/nature07205 [DOI] [PubMed] [Google Scholar]
- 26.Chanmee T, Ontong P, Konno K, Itano N. Tumor-associated macrophages as major players in the tumor microenvironment. Cancers. 2014;6:1670–1690. doi: 10.3390/cancers6031670 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Gao Y, Zhang Z, Li Y, et al. Pretreatment neutrophil-to-lymphocyte ratio as a prognostic biomarker in unresectable or metastatic esophageal cancer patients with anti-PD-1 therapy. Front Oncol. 2022;12:834564. doi: 10.3389/fonc.2022.834564 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Booka E, Kikuchi H, Haneda R, et al. Neutrophil-to-lymphocyte ratio to predict the efficacy of immune checkpoint inhibitor in upper gastrointestinal cancer. Anticancer Res. 2022;42:2977–2987. doi: 10.21873/anticanres.15781 [DOI] [PubMed] [Google Scholar]
- 29.Liu J, Gao D, Li J, Hu G, Liu J, Liu D. The predictive value of systemic inflammatory factors in advanced, metastatic esophageal squamous cell carcinoma patients treated with camrelizumab. Onco Targets Ther. 2022;15:1161–1170. doi: 10.2147/OTT.S382967 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Zhang X, Gari A, Li M, et al. Combining serum inflammation indexes at baseline and post treatment could predict pathological efficacy to anti‑PD‑1 combined with neoadjuvant chemotherapy in esophageal squamous cell carcinoma. J Transl Med. 2022;20:61. doi: 10.1186/s12967-022-03252-7 [DOI] [PMC free article] [PubMed] [Google Scholar]