Abstract
Immunotherapy has revolutionized the treatment of cancer. In particular, immune checkpoint blockade, bispecific antibodies, and adoptive T-cell transfer have yielded unprecedented clinical results in hematological malignancies and solid cancers. While T cell-based immunotherapies have multiple mechanisms of action, their ultimate goal is achieving apoptosis of cancer cells. Unsurprisingly, apoptosis evasion is a key feature of cancer biology. Therefore, enhancing cancer cells’ sensitivity to apoptosis represents a key strategy to improve clinical outcomes in cancer immunotherapy. Indeed, cancer cells are characterized by several intrinsic mechanisms to resist apoptosis, in addition to features to promote apoptosis in T cells and evade therapy. However, apoptosis is double-faced: when it occurs in T cells, it represents a critical mechanism of failure for immunotherapies. This review will summarize the recent efforts to enhance T cell-based immunotherapies by increasing apoptosis susceptibility in cancer cells and discuss the role of apoptosis in modulating the survival of cytotoxic T lymphocytes in the tumor microenvironment and potential strategies to overcome this issue.
Keywords: immunotherapy, adoptive; cytotoxicity, immunologic
The dual role of apoptosis in cancer immunotherapy
Cancer immunotherapies exploit the immune system to combat cancers and thus have revolutionized the field of immuno-oncology, leading to unprecedented outcomes in relapsed and refractory patients.1 Especially, modulation of T cell’s anticancer activity through immune checkpoint blockade (ICB) (eg, anti-programmed cell death protein-1 (PD-1)/programmed death ligand-1 or anti-cytotoxic T-lymphocytes-associated protein 4 (CTLA-4) antibodies, online supplemental box 1) showed a significant clinical response in a subset of solid and hematologic malignancies. Bispecific antibodies (online supplemental box 1) represent another strategy triggering cancer recognition by T cells.2 The anti-CD19/CD3 bispecific T-cell engager blinatumomab was approved in 2014 for B-acute lymphoblastic leukemia (B-ALL) and several anti-CD20/CD3, and anti-BCMA/CD3 antibodies are in advanced clinical development (online supplemental box 1).3–6 Although checkpoint inhibitors are currently the treatment backbone for several cancer types, many patients eventually develop secondary resistance and progressive disease in the end.7 Chimeric antigen receptor T-cell (CAR-T) therapy, a form of adoptive cell transfer (ACT),8 has also demonstrated substantial anticancer efficacy in treating relapsed or refractory B-cell leukemias, lymphomas, and multiple myeloma, which resulted in the approval of multiple CAR-T products by the US Food and Drug Administration (FDA) (online supplemental box 1).9–14 Nevertheless, approximately 50% of pediatric B-ALL and up to 70% of patients with B-cell lymphoma still do not respond or eventually relapse to the CAR-T therapy.10 12 13 15 Therefore, improving the potency of T cell-based immunotherapies is critical for improving the clinical outcomes of patients with cancer.
jitc-2022-005967supp001.pdf (21.8KB, pdf)
The ultimate goal of anticancer therapy, including T cell-based immunotherapies, is to eliminate cancer cells, mainly by efficiently inducing apoptosis in cancer cells. Apoptosis, a programmed cellular mechanism leading to cell death, is a complex biological process involving a vast array of tightly controlled cellular components.16 The acquisition of resistance to programmed cellular death (eg, apoptosis) is a key feature of cancer progression.17 For instance, genetic alteration of the anti-apoptotic regulator (eg, translocation and/or gain of B-cell lymphoma 2 (BCL-2)) has been well characterized as a key biological marker in multiple lymphomas, including follicular B-cell non-Hodgkin’s lymphoma, diffuse large B-cell lymphoma, and B-cell chronic lymphocytic leukemia (CLL).18 High levels of BCL-2 expression protect these fast-growing lymphomas against apoptosis, allowing malignant B cells to survive under various stress factors, such as cytokine deprivation. The critical role of apoptosis in cancer development has been further identified during the transformation of premalignant cells into malignant cells. While MYC expression in premalignant cells increases sensitivity to apoptosis, a similar expression of MYC in malignant cells provides a strong proliferative advantage without inducing apoptosis. This proliferative advantage of MYC expression can be attributed to the co-expression of anti-apoptotic regulators (ie, BCL-2) in malignant cells, indicating that acquiring resistance to apoptosis by increasing expression of anti-apoptotic regulator (ie, BCL-2) during malignant transformation is an important checkpoint in cancer development.18 19 Considering the critical role of apoptotic resistance in cancer development, this resistance may also provide a strong protective mechanism against T cell-based immunotherapies. Therefore, it is essential to understand not only the general molecular mechanisms of apoptosis but also the evasion mechanism of cancer cells to enhance the anticancer activity of T cell-based immunotherapies.
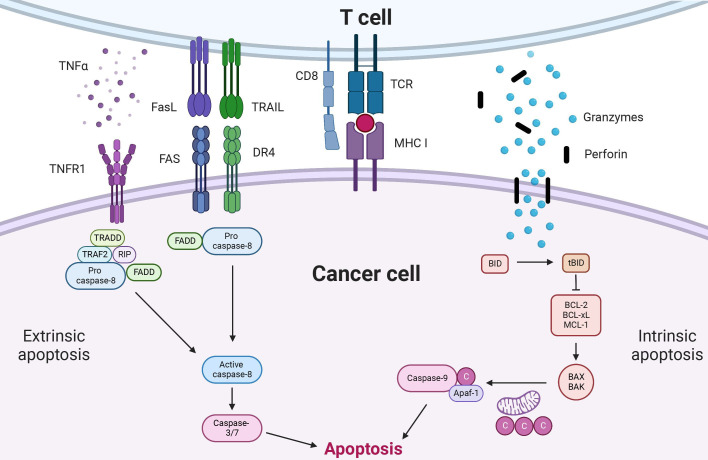
Multiple cellular insults and external stimuli, broadly categorized as intrinsic or extrinsic, can promote apoptosis. Intrinsic apoptosis is triggered by DNA damage, excessive reactive oxygen species (ROS), hypoxia, or cellular/metabolic stress.20 In contrast, extrinsic apoptosis is initiated by the so-called ‘death ligands,’ such as Fas ligand (FasL or CD95L), TRAIL (TNF-related apoptosis-inducing ligand), and tumor necrosis factors (TNFs).21 Immune cells, particularly T cells, use both pathways to activate apoptosis in cancer cells (figure 1). On T-cell receptor (TCR) engagement, T cells release cytolytic granules containing granzymes and perforin in the immune synaptic space to initiate the intrinsic apoptotic pathway. Perforins are pore-forming proteins that diffuse across immunological synapses and oligomerize to form pores on the target cell membrane, facilitating the entry of granzymes into the target cell.22 Granzymes generally enter target cells through pores formed by perforin; it has also been described that granzymes can pass the cell membrane of target cells without requiring perforin, via pinocytosis.23 Four subsets of granzymes (granzyme A, B, K, and M) have been identified in human T cells; particularly granzyme B plays an essential role in promoting T cell-induced apoptosis in target cells.24 On entry into the target cell, granzyme B cleaves BH3 Interacting Domain Death Agonist (BID), a BCL-2 homology (BH3)-only pro-apoptotic protein that plays an essential role in promoting apoptosis. Cleavage of BID results in the generation of the active form of BID (truncated BID/tBID). Subsequently, tBID activates pro-apoptotic effector proteins, such as BAX and BAK, by interfering with their interaction with BCL-2. This activation of effector proteins leads to the induction of instability of the mitochondrial membrane potential and the release of cytochrome c from the mitochondria to the cytoplasm. Cytochrome c leads to the formation of apoptosome complexes (cytochrome c:APAF-1:Caspase-9), which activate effectors, Caspase-3 and Caspase-7, promoting downstream apoptotic signaling cascades.25 However, to trigger the extrinsic apoptotic pathway, the engagement of T cell-derived death ligands and their associated receptors in target cells is necessary. When T cells are activated, it leads to an increase in the expression of death ligands, such as FasL, TNF-α, and TRAIL. These death ligands bind to their respective death receptors (eg, FasL-Fas (CD95), TNF-α-TNFRSF1A (TNFR1), and TRAIL-TNFRSF10A (DR4)) on target cells, triggering the formation of death-inducing signaling complexes (DISCs), comprizing adaptor proteins (eg, Fas-associated death domains (FADD) or TNF receptor type 1-associated death protein (TRADD)) and initiator Caspase-8. DISCs eventually initiate downstream apoptotic signaling cascades to induce cellular apoptosis.25
Figure 1.
Apoptosis induced by T cells. T cells can induce apoptosis in cancer by both extrinsic and intrinsic pathways. To promote intrinsic apoptosis in cancer, granzymes is first transported into cancer cell via perforin, and granzymes cleave BID to generate truncated BID (tBID). tBID inhibits anti-apoptotic regulators (eg, BCL-2, BCL-xL, MCL-1), leading to the formation of homodimer or heterodimer of BAX and BAK on the membrane of mitochondria. These BAK/BAX dimerization releases cytochrome C and Apaf-1 from mitochondria to the cytoplasm. Together with Caspase-9, cytochrome C and Apaf-1 form apoptosomes that can cause apoptosis in cancer cells. FasL, TRAIL, and TNF-α expressed by T cells engage with their corresponding receptor in cancer cells to initiate extrinsic apoptosis. On engagement, death receptor complexes are formed and activate Caspase-8. Finally, activated Caspase-8 stimulates the activity of Caspase-3/7, resulting in the induction of apoptosis in the cancer cell. C: cytochrome C, BCL-2, B-cell lymphoma 2; FADD, Fas-associated death domains; FasL, Fas ligand; MHC, major histocompatibility complex; TCR, T-cell receptor; TNF, tumor necrosis factor; TRADD, TNF receptor type 1-associated death protein; TRAIL, TNF-related apoptosis-inducing ligand.
Cancers have developed several strategies to evade immune cell-mediated apoptosis. For instance, mutation of TP53, a key tumor suppressor gene that confers resistance to apoptosis is strongly associated with decreased immune function genes (eg, granzymes and perforin) in patients with gastric cancer.26 27 This observation suggests that aberrant TP53 activity could affect the anticancer immune response. Modulation of anti-apoptotic (eg, BCL-2, CFLAR, and BIRC2) and pro-apoptotic proteins (FAS, FADD, TNFRSF10B (death receptor 5—DR5), BID, and Caspase-8) is another important mechanism used by cancer cells to blunt cancer immunotherapy’s anticancer efficacy.28–33 Maruyama et al reported that >40% of patients with metastatic renal cell cancer with no response or progressive disease were positive for immunohistochemical staining of BCL-2, while patients with complete or partial responses were negative for BCL-2 during the treatment course of immune-stimulatory treatments (eg, interferon (IFN)-α, IFN-γ, and interleukin-2).28 Furthermore, we performed a retrospective analysis of the clinical response of patients with lymphoma treated with anti-CD19 CAR-T therapy and showed that patients with genetic alterations in BCL-2 (ie, gain or translocation of BCL-2) show significantly lower response and overall survival to CAR-T treatments than patients without genetic alterations of BCL-2,33 implicating that genetic alteration of BCL-2 plays a crucial role in the anticancer efficacy of CAR-T therapy. In addition to the effect of altered intrinsic regulators of apoptosis on cancer immunotherapy, our group also demonstrated that CAR-T cells’ anticancer efficacy is significantly reduced when leukemic cells display decreased expression of positive regulators of apoptosis, especially in the death receptor pathway (FasL, TRAIL, and TNF-α).34 By using unbiased genome-wide CRISPR knock-out (KO) screening, we identified that the deletion of anti-apoptotic regulators (eg, BIRC2, CFLAR, and TRAF2) in the B-ALL cell line NALM-6 led to significant enhancement of the anticancer activity of CAR-T cells, while KO of pro-apoptotic regulators (eg, FADD, Caspase-8, BID, and TNFRSF10B) resulted in a decrease of anticancer activity of CAR-T cells. Further validation with clinical data revealed that the downregulation of pro-apoptotic regulators was significantly associated with a poor clinical response in patients with B-ALL treated with anti-CD19 CAR-T cells. Likewise, Upadhyay et al showed that Fas-FasL-mediated cancer killing plays a crucial role in T cell-based immunotherapy, and the expression of Fas in cancer strongly correlates with the clinical outcome of CAR-T therapy.29 Importantly, these studies suggest that mechanisms conferring resistance to apoptosis in some cancer cells can also drive T-cell dysfunction, leading to poor clinical outcomes of T cell-based immunotherapy.
Given that resistance to apoptosis in cancers could be a critical factor associated with poor clinical outcomes of T-cell mediated immunotherapy by causing dysfunction of T cells, this review will highlight several novel therapeutic strategies designed to augment T cell-mediated cancer apoptosis. Furthermore, it discusses tumor-derived or tumor microenvironment (TME)-derived factors that govern T-cell apoptosis and rational strategies to prevent it.
Strategies to enhance T-cell mediated cancer apoptosis
Despite remarkable clinical outcomes of T cell-based immunotherapies, a substantial number of patients do not benefit from these approaches.9–15 Considering the importance of cancer apoptosis susceptibility in the cytolytic activity of T-cell therapy, several interesting therapeutic approaches have been investigated to overcome apoptosis resistance in cancer cells (figure 2 and table 1).
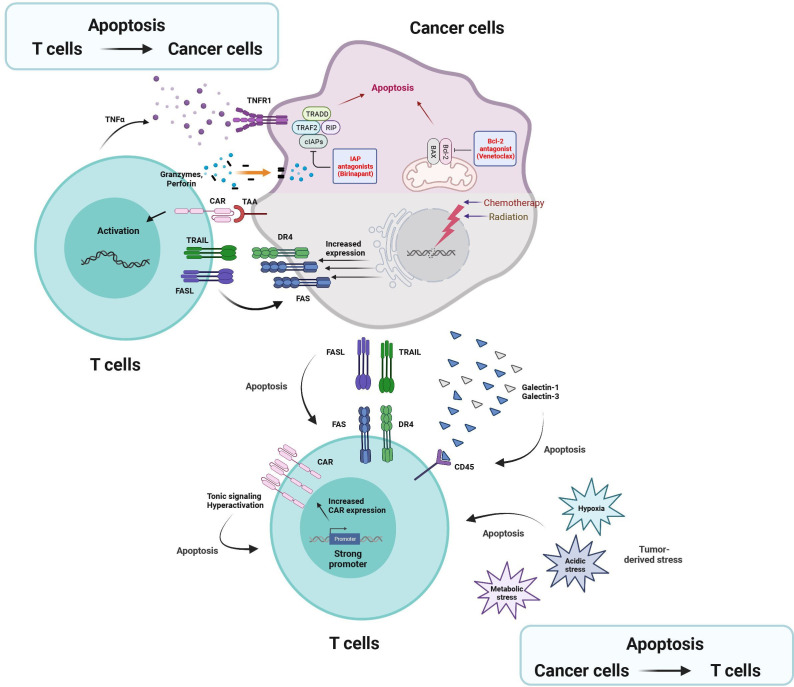
Figure 2.
Dual effects of apoptosis in T cell-based immunotherapy. On the cancer cells side, chemotherapy and radiation therapy induces the expression of DR4 and Fas in cancer cells, sensitizing cancer cell to TRAIL-mediated and FasL-mediated apoptosis. In addition, treatment of small molecules that can specifically inhibit anti-apoptotic regulators (eg, IAP and BCL-2) leads to the enhancement of cancer apoptosis mediated by TNF-α or granzymes/perforin. On the T-cell side, multiple factors (eg, FasL, TRAIL, and galectin) and stress (eg, hypoxia, metabolic alteration, and acidification) derived from cancer cell and tumor microenvironment promote apoptosis in T cells. Hyperactivation and tonic signaling of CAR-T cells by increased CAR expression on the surface induce apoptosis in T cell. BCL-2, B-cell lymphoma 2; CAR, chimeric antigen receptor; cIAP, cellular IAP; DR4, death receptor 4; FasL, Fas ligand; IAP, inhibitor of apoptosis proteins; TAA, Tumor associated antigen; TNF, tumor necrosis factor; TRADD, TNF receptor type 1-associated death protein; TRAIL, TNF-related apoptosis-inducing ligand.
Table 1.
Summary of the preclinical combinations of pro-apoptotic drugs and T cell-based immunotherapies
| Class | Drug | Target | Combination | Cancer type | Effect | Ref |
| Chemo therapy | Cisplatin, etoposide | DNA damage | NKT cells | NSCLC cell lines CRC cell lines PC cell lines BC cell lines |
Sensitization to TRAIL-mediated and FasL-mediated apoptosis | 37 |
| Chemo therapy | Doxorubicin, 5-fluorouracyl |
DNA damage | Vγ9Vδ2 T cells | CRC cell lines | Sensitization to TRAIL-mediated apoptosis | 38 |
| Chemo therapy | Doxorubicin | DNA damage | NK or T cells | Melanoma and bladder cancer cell lines | Sensitization to TRAIL-mediated apoptosis | 39 |
| Radiation | Sublethal irradiation | – | antitumor CTLs and NK cells | CRC cell lines | Sensitization to TRAIL-mediated and FasL-mediated apoptosis | 40 |
| Radiation | Sublethal irradiation | – | CEA-specific HLA-A2-restricted CD8(+) CTLs | 23 human carcinoma cell lines (12 colons, 7 lungs, and 4 prostate) | Sensitization to FasL-mediated apoptosis | 41 |
| SMAC mimetic | Birinapant | Inhibition of XIAP and cIAP1/2 | anti-CD19 CAR T cells | B-ALL | Increase of CAR T cell-mediated apoptosis | 34 |
| SMAC mimetic | Birinapant | Inhibition of IAPs | anti-HER2 CAR T cells | HER2+patient-derived colorectal tumoroids | Sensitization to TNF-α-mediated apoptosis | 56 |
| SMAC mimetic | Birinapant | Inhibition of IAPs | anti-CD19 CAR T cells | B-ALL | Sensitization to TNF-α-mediated apoptosis | 32 |
| SMAC mimetic | ASTX660 | Inhibition of IAPs | cytotoxic TIL | HNSCC | Enhanced immunogenic cell death | 59 |
| BH3 mimetic | ABT737 | Bcl-2 family anti-apoptotic proteins | anti-CD19 CAR T cells | Patients with childhood precursor-B ALL | Increase of CAR T cell-mediated apoptosis | 65 |
| BH3 mimetic | Venetoclax | Bcl-2 family anti-apoptotic proteins | anti-CD19 CAR T cells | B-ALL and B-lymphoma cell lines | Increase of CAR T cell-mediated apoptosis | 67 |
B-ALL, B-cell acute lymphoblastic leukemia; BC, breast cancer; BCL-2, B-cell lymphoma 2; CAR, chimeric antigen receptor; CRC, colorectal cancer; CTL, cytotoxic T cell; FasL, Fas ligand; HER2, human epidermal growth factor receptor-2; HNSCC, head and neck cancer; IAP, inhibitor of apoptosis proteins; NK, natural killer; NKT, natural killer T; NSCLC, non-small cell lung cancer; PC, prostate cancer; SMAC, second mitochondria-derived activator of caspase; TIL, tumor-infiltrating lymphocyte; TNF, tumor necrosis factor; TRAIL, TNF-related apoptosis-inducing ligand.
First, researchers have tested whether conventional anticancer therapeutics, such as chemotherapy or radiotherapy, can enhance cancer apoptosis during immunotherapy. Both, chemotherapeutic agents (eg, alkylating agents, anthracyclines, vinca alkaloids, and antimetabolites) and radiation (eg, X-ray), potently promote intrinsic apoptosis in cancer cells by inducing DNA damage and/or inhibiting cell cycle.35 36 Moreover, treatment with selected chemotherapies (eg, etoposide, doxorubicin, and 5-fluorouracil) and radiation can also modulate death receptor-mediated extrinsic apoptosis in cancer cells by affecting the transcriptional activity of death receptors (eg, Fas and TNFRSF10 families).37–41 On exposure to etoposide, DR5 and Fas expression increased in human lung, colorectal, prostate, bladder, and breast cancer cell lines, leading to improved immune cell (natural killer T cell-mediated killing).37 Similarly, sublethal doses of doxorubicin upregulate TRAIL receptors on cancer cells (eg, MAR and JOHW colorectal carcinoma cell lines), promoting natural killer and tumor-infiltrating lymphocyte (TIL) cytotoxicity.39
Interestingly, the authors also observed that doxorubicin treatment reduced the expression of intracellular FLICE inhibitory protein (c-FLIP), a key anti-apoptotic inhibitor of death receptor-mediated apoptosis. This finding suggests that doxorubicin can sensitize cancer cells to immune cell-mediated extrinsic apoptosis by modulating both pro-apoptotic and anti-apoptotic regulators. Another evidence of chemotherapy-induced immune killing reported that 5-fluorouracil treatment upregulates the expression of DR5 in colon cancer-initiating cells in vitro, leading to enhancement of T cell-mediated cytotoxicity.38 In addition to chemotherapeutic agents, irradiation, which causes DNA damage and intracellular stress, can also lead to the induction of death receptor-mediated cancer cell apoptosis. For instance, a sublethal dose of irradiation can upregulate the expression of Fas and DR5 in colorectal carcinoma cell lines, making them susceptible to TRAIL-induced and Fas-induced apoptosis.40 Further investigation identified that a sublethal dose of irradiation can increase Fas expression in over 40% of the cancer cell lines, including colon, lung, and prostate cancers.41 This suggests that radiation may be an effective strategy in combination with T cell-based immunotherapy to enhance the sensitivity of extrinsic apoptosis in cancer cells. In addition to the modulation of cytolytic activity of endogenous T cells in radiation therapy and chemotherapy, the use of recombinant anti-Fas and anti-DR4/5 agonists along with radiation and chemotherapeutic agents has been investigated. Treatment with recombinant TRAIL combined with bortezomib, vorinostat (SAHA), and valproic acid significantly induced cancer cell apoptosis by sensitizing cancer cells to extrinsic apoptosis signal.42–45
Finally, with ample preclinical evidence that chemotherapy and radiation therapy can increase the sensitivity of cancer cells to T cell-mediated apoptosis, various clinical trials exploiting the combination of immunotherapy and chemo/radiation have been registered and conducted (see online supplemental table 1). First, the combination of pembrolizumab (online supplemental box 1) and chemotherapy (ie, carboplatin and either paclitaxel or nanoparticle albumin-bound-paclitaxel) significantly improved overall survival and progression-free survival in patients with metastatic squamous non-small cell lung cancer (NSCLC) as compared with chemotherapy only treated patients.46 Another clinical investigation using an immunotherapeutic combination (nivolumab and ipilimumab, online supplemental box 1) with chemotherapy (carboplatin, paclitaxel, pemetrexed, and cisplatin) showed similar results.47 These two independent clinical trials eventually led to FDA approval of chemotherapy in combination with checkpoint blockade for first-line metastatic squamous NSCLC treatment. Despite the substantial synergy between immune checkpoint inhibitors and chemo/radiation therapy, one caveat remains: apoptosis of T cells can also be increased by chemotherapy and radiation due to the lack of ability of chemotherapy and radiation to distinguish target cells (ie, cancer cells) and effector cells (ie, T cells). The resistance mechanisms of T cells to chemotherapy-induced and radiation-induced apoptosis remain largely unknown. Several studies have highlighted that memory T cells can escape apoptosis triggered by chemotherapy and irradiation, implicating the potential role of memory T cells in synergy strategies.48 49 Considering the resistance of memory T cells to apoptosis, one possible explanation is that a high level of BCL-250 and low level of Bcl-2-like 11 (BIM)51 expression in memory T cells may increase the threshold of apoptotic sensitivity, allowing them to evade chemotherapy-induced and radiation-induced apoptosis. However, further investigations are required to fully understand survival mechanisms of T cells during chemotherapy and radiation.
jitc-2022-005967supp002.pdf (129.6KB, pdf)
While chemotherapy and/or radiation increase the sensitivity of cancer cells to apoptosis, direct inhibition of apoptotic regulators has also been observed in combination with T cell-based immunotherapy. One example is the inhibitor of apoptosis proteins (IAPs) that are overexpressed in many cancers52 and include several members, such as cellular IAP1 (cIAP1), cIAP2, X-linked IAP (xIAP), neuronal apoptosis inhibitory protein (NAIP), livin, and survivin.53 cIAP1 plays a critical role in inhibiting TNF-α-mediated apoptosis by preventing the formation of the apoptotic complex (FADD/RIPK1/Caspase-8). Moreover, xIAP inhibits apoptosis by blocking Caspase-3 and Caspase-7 by directly binding to them.54 Given the importance of IAPs in inhibiting apoptosis in cancer, multiple agents have been investigated to promote IAP degradation, thereby sensitizing cancer cells to apoptosis. Second mitochondria-derived activator of caspase (SMAC) mimetics are small synthetic molecules whose structural and functional features are similar to SMAC, which are endogenous antagonists of IAPs. Several SMAC mimetics (eg, birinapant, LCL-161, ASTX660, Debio1143, BV-6, GDC-0152, CUCD-427, HGS1029, and AT-406) have been developed (online supplemental box 2).55 Particularly, birinapant has been extensively evaluated for its anticancer properties, including in combination with T cell-based immunotherapies such as immune checkpoint inhibitors, such as anti-PD-1 and anti-CTLA-4 antibodies, driving significantly enhanced TNF-α-mediated cancer cell apoptosis and increase in survival in a murine glioblastoma (GBM) model.32 34 56 57 Birinapant also improved the anticancer efficacy of CAR-T therapy in murine models. Treatment with birinapant enhanced anti-human epidermal growth factor receptor-2 CAR-T cells-mediated cancer killing by sensitizing cancer cells to TNF-mediated apoptosis.56 Likewise, Song et al also found that treatment of birinapant enhances CAR-T cell-mediated tumor killing in GBM model.58 Using a CRISPR Cas9 KO library, key mediators of synergy between CAR-T cells and birinapant such as RIPK1, FADD, and TNFRSF10B in cancer cells are identified.32 Our group also demonstrated that birinapant treatment improved CAR-T cells ability to eliminate B-ALL, which otherwise lacks sensitivity to extrinsic apoptosis34; however, these results were obtained in vitro and in vivo validation is required to exclude toxicity on CAR T cells. In addition to birinapant, Ye et al studied the combinatorial efficacy of cytotoxic TILs and ASTX660, another antagonist of cIAP1/2 and xIAP, in a preclinical model with head and neck squamous cell carcinoma (HNSCC).59 The authors found that ASTX660 treatment induced—in the presence of TNF-α—calreticulin (CRT) expression, heat shock proteins 70/90, and high mobility group protein on the surface of human HNSCC cell lines (eg, UMSCC-46 and UMSCC-47). These are key molecular signatures for immunogenic cell death (ICD), suggesting that ASTX660 and TNF-α promoted ICD in HNSCC cell lines. ASTX660-mediated induction of ICD was further confirmed in syngeneic murine cancer models (HNSCC) when combined with radiation. Interestingly, the authors identified that ASTX660 treatment plus TNF-α led to clonal expansion of antigen-specific T-cell clones. This might be due to the enhancement of the antigen-processing machinery in cancer cells, as evidenced by the upregulation of critical components of the antigen-processing machinery (eg, human leukocyte antigen (HLA)-A, HLA-B, HLA-C, ERp57, CRT (intracellular), Transporter associated with antigen processing 1 (TAP1), and TAP2) in human HNSCC cell lines after exposure to ASTX660 and TNF-α.
Members of the BCL-2 family, such as BCL-2, BCL-XL, MCL-1, BAX, and BAK, play a critical role in regulating intrinsic apoptosis by modulating the permeabilization of the mitochondrial membrane.60 As previously discussed, upregulation of BCL-2 activity via overexpression or translocation is one of the key features of various cancers.61 Several BCL-2 inhibitors have been developed, including obatoclax, AT101, ABT737, S-055746, S65487, PNT-2258, navitoclax, and venetoclax (online supplemental box 2).62 In particular, venetoclax, an orally available small-molecule inhibitor with high specificity to BCL-2, has demonstrated substantial anticancer efficacy in treating CLL, other lymphomas, and acute myeloid leukemia, leading to its FDA approval in these settings.63 Recently, Kohlhapp et al reported that venetoclax could enhance the anticancer efficacy of anti-PD-1 antibody (MDX-1106) treatment.64 Interestingly, the authors found that venetoclax treatment increased tumor infiltrating effector memory T cells, which could also explain the potential role of memory T cells in synergy with pro-apoptotic drugs. The beneficial effects of venetoclax in T cell-based immunotherapy were further identified by our group. Using a combination of venetoclax and CAR-T therapy, we demonstrated a significant improvement in CAR-T cells’ anticancer activity against various lymphoma and leukemia xenograft models (eg, OCI-Ly18, MINO, NALM6, KG-1, and MOLM-14).33 In addition to venetoclax, the combinatory effect of different BCL-2 inhibitors (ie, ABT737) with CAR-T therapy was tested, and it was found that adding ABT737 to CART19 resulted in an increase in Caspase 3/7 activity in cancer cells, leading to cancer killing enhancement.65
While, increasing cancer cell sensitivity to apoptosis using aforementioned pro-apoptotic molecules results in the enhancement of the anticancer response of T cell-based immunotherapies in some models, one potential concern of this approach is the unintended toxicity of these agents on effector immune cells such as T cells, which could be critical for the long-term efficacy of combination immunotherapy. A study showed that SMAC mimetic (ie, LBW242) treatment significantly inhibited virus-specific CD8+ T-cell expansion in vivo by inducing T-cell apoptosis, ultimately leading to the failure of virus replication.66 Moreover, although Lee et al and Kohlhapp et al demonstrated that venetoclax augmented the anticancer response of CAR-T therapy and anti-PD-1 treatment, they also found that co-culture of venetoclax with genetically non-modified T cells and CAR-T cells potently reduced their viability.33 64 These observations strongly suggest that careful design of combination therapies and the sequence of administration are required to avoid T-cell toxicity and ensure long-term therapeutic efficacy. One possible administration strategy to avoid bystander effects on T cells is to pretreat the cancer cells with cytotoxic drugs. Recently we reported that patients with lymphoma receiving venetoclax during bridging therapy prior CAR-T cell infusion achieved significant improvement in clinical response compared with patients treated with no venetoclax-included bridging therapy.33 In line with our clinical observations, pretreatment of cancer cells with venetoclax enhanced CAR-T cell-mediated anticancer activity in vitro.67 These preclinical and clinical data strongly suggest that pre-sensitizing cancer cells with anti-apoptotic inhibitors could enhance the anticancer effect of T cell-based immunotherapy while reducing toxicity to T cells.
T-cell apoptosis limits anticancer immunity in the TME
Apoptosis in cancer therapy could induce both cancer cells to die and result in T-cell death. T-cell apoptosis is an indirect result of multiple immunosuppressive mechanisms in cancer genesis. For example, T-cell dysfunction, such as exhaustion, is a physiological state in which T cells lose their effector functions while maintaining viability. Prolonged exhaustion ultimately leads to T cells undergoing cellular apoptosis.68 Furthermore, the immunosuppressive TME, including immunosuppressive immune cells (eg, T regulatory cells, tumor-associated macrophages, and myeloid-derived suppressor cells)69 70 and lack of key nutrients (ie, low arginine and changes in available metabolites)71–73 also have substantial effects on the proliferation and survival of cytotoxic T cells in the TME. Because there are already extensive revisions of the literature on T-cell exhaustion74–76 and other immunosuppressive factors,77–79 we focused on the mechanisms of immune evasion that directly trigger apoptosis in T-cells.
On activation, T cells enhance the expression of pro-apoptotic proteins (eg, FasL, TRAILs, and TNF), potentially promoting the death of target cells as well as death receptors on their surface (Fas, TRAIL receptors, TNF receptor). This upregulation of death receptors increases the susceptibility of activated T cells to apoptosis.80 81 This process is called activation-induced cell death (AICD) and plays a vital role in maintaining peripheral immune tolerance and preventing autoimmune disease development.82 Cancer cells can take advantage of this T-cell liability by using it as a potential immunoevasion strategy. Reports in the late 1990s demonstrated that FasL expression in several malignancies (melanoma, colon, head/neck, liver, and lung) serves as a mechanism of cancer evasion.83–86 This phenomenon, coupled with evidence that T cells increase the expression of Fas on activation, highlights that cancers can induce apoptosis in T cells via the extrinsic pathway to evade immune surveillance. There has also been evidence of upregulation of TRAIL in a few malignancies (melanoma, liver, breast, and lung), although its correlation with the clinical outcome has been controversial.87–90 While Bron et al found no correlation with prognosis in patients with melanoma,87 Cross et al observed a negative association between TRAIL expression in breast cancers and the clinical outcome of patients with breast cancer.88 Moreover, heterogeneity exists in the ubiquity of FasL and TRAIL expression across cancers. Another captivating aspect of this mechanism is the observation that cancer can secrete exosomes expressing FasL and independently induce T-cell apoptosis.91–93 Such observations amplify the potency of FasL-mediated apoptosis of T cells directed by cancer cells, which may cause peripheral T-cell dysfunction.94
This cancer-induced, death receptor-mediated T-cell apoptosis has been proven to directly hinder responses to immunotherapy.95–99 Zhu et al used a novel autochthonous melanoma mouse model to demonstrate that FasL-mediated T-cell apoptosis facilitates cancer resistance to anti-CTLA-4 antibody, anti-PD-1 antibody, and ACT.98 Similar to TCR-mediated activation, CAR-driven T-cell activation also increases the susceptibility of CAR-T cells to apoptosis by upregulating death receptors and associated ligands on their surface. Hyperstimulation of CAR-T cells by incorporating two co-stimulatory domains (CD28 and 4-1BB) also increases Fas and DR5 expression and promoted CAR-T cells apoptosis.96 97 Lastly, tonic signaling of 4-1BB co-stimulation due to greater anti-CD19 CAR expression driven by a strong promoter, such as retroviral long terminal repeat, increases levels of FasL, leading to apoptosis of CAR-T cells on activation.99 While introducing multiple co-stimulatory domains into CAR construct was intended to enhance activation,95 these data present a potential concern of overstimulation suggesting a need of ‘modulating’ CAR-activation in T cells rather than just boosting it.
Cancer and TME cells also secrete factors that can directly trigger T-cell apoptosis.100 101 Galectins are a family of proteins produced and secreted by various cells, including cancer and immune cells.102 Galectins bind to β-galactosides on glycoproteins and glycolipids via a conserved carbohydrate recognition domain, thereby regulating miscellaneous biological events, including apoptosis.100 101 Many studies have demonstrated that cancer-secreted galectin-3 (Gal-3) can induce T-cell apoptosis in various cancers, including melanoma, lung, and colorectal cancer, on binding to their target TCRs, such as CD7, CD29, CD45, and CD71.103–108 Mechanistically, cancer-secreted Gal-3 binds to CD45, activating independent pathways involving protein kinase C and ROS, resulting in sustained ERK 1/2 phosphorylation, Caspase-9 activation, cytochrome c release, and Caspase-3 activation to induce apoptosis.109 Besides the function of Gal-3, secreted Gal-1 in the TME also correlated with increased cancer progression (following ICB therapy), which could be due to T-cell apoptosis, likely mediated through a CD45-binding dependent mechanism.110–116 However, this correlation is not consistent across cancers. While elevated Gal-1 correlates with T-cell apoptosis in pancreatic117 and lung110 cancer cell lines, it was not confirmed in a melanoma cell line108 or in vitro against activated primary T cells,118 suggesting that its effects may be heterogeneous across malignancies.
Similarly, gangliosides and sialic acid-containing glycosphingolipids found on outer plasma membranes are overexpressed in cancers and shed into the TME.119 Although the apoptotic effects of gangliosides and their expression in different cancers have not been investigated as extensively as galectins, Finke and Tannenbaum have elucidated their general effect on T-cell apoptosis through a series of studies. Finke et al demonstrate in both a GBM and a renal cell carcinoma model that cancer gangliosides are responsible for inducing T-cell apoptosis.120 121 Moreover, Bharti and Singh show the induction of bone marrow cell apoptosis through T-cell lymphoma-derived gangliosides.122 Regarding the mechanism of ganglioside-mediated T-cell apoptosis, gangliosides have been shown to be internalized by activated T cells, resulting in ROS production, cytochrome c release, and Caspases-8 and Caspase-9 activation.123 This implies that gangliosides may promote both intrinsic and extrinsic apoptosis. Notably, gangliosides facilitate the intrinsic pathway of apoptosis, as evidenced by the induction of ROS, cytochrome c release, Caspase-9 activation, and downregulation of anti-apoptotic BCL genes, such as BCL-XL and BCL-2.124
Metabolic pathways and associated enzymes may also play important roles in T-cell apoptosis. For instance, glucose deprivation can reduce the proliferation of Jurkat cells and primary human T cells in vitro.125 This reduction might be linked to the increase in intrinsic apoptosis since the knockdown of pro-apoptotic BH-3-only protein (ie, Noxa) improves the survival of T cells when limited glucose is available. Considering that T cells encounter significant competition in the uptake of glucose by cancer cells in TME,126 the lack of glucose in T cells may increase the susceptibility of T cells to apoptosis, leading to impairment of the anticancer activity of T cells. In addition to the glycolytic pathway, fatty acid metabolism is critical for T cell-mediated anticancer activity. While T cells use fatty acid oxidation to form and maintain the memory phenotype,127 inhibiting fatty acid synthase potentially reduces the expression of FasLs, preventing T cells from restimulation-induced cell death.128 In addition to the intrinsic alteration of T-cell metabolism in inducing apoptosis, metabolites from cancer cells may also promote apoptosis in T cells. For example, kynurenine, a metabolite of tryptophan by indoleamine 2,3-dioxygenase in cancer cells, can induce apoptosis in thymocytes and terminally differentiated T helper cells.129
The last well-documented secretion-based methods of direct cancer-induced T-cell apoptosis are the acidic and hypoxic stress found in the TME. Acidity is caused by the ‘Warburg effect’, whereby cancer cells preferentially engage in aerobic glycolysis rather than oxidative phosphorylation metabolism of glucose.130 Consequently, they increase their glucose intake to meet their energy demands, producing excess lactate acid, which is secreted into the microenvironment, causing acidification of the extracellular space.131 Long-term exposure (>3 days) to acidic pH in the TME (pH 6.5) caused permanent damage and T-cell apoptosis in C57BL-murine B16-melanoma TILs.132 Under extreme conditions, acidic stress (pH 3.3 for 25 min at 37°C) induces intrinsic apoptosis in Jurkat T cells by increasing cell cycle arrest.133 Although in vitro studies demonstrated that acidic stress can alter apoptosis in T cells, the effect of acidic conditions in vivo remains unknown and requires careful validation. Along with increased acidity, hypoxia in the TME can also be a critical factor affecting T-cell apoptosis. Kiang et al found hypoxia-induced apoptosis in the Jurkat cell line.134 The authors attributed apoptosis to increase NO production due to the upregulation of NO synthase, subsequently increasing Caspase-9 activation, cytochrome c levels, and Caspase-3 activation. In addition, hypoxia (1% O2) induces apoptosis in primary T cells from healthy donors, hypothesizing it to result from a buildup of endogenous adenosine in the extracellular medium. The authors found that T cells had an upregulation of the adenosine receptor A2aR. The downstream effects of these receptors in inducing apoptosis have not been characterized.135
Strategies to avoid T-cell apoptosis in the TME
Long-term survival and functionality of T cells are critical to ensure the anticancer efficacy of cancer immunotherapies.136 Several strategies have been developed to prevent T-cell apoptosis. Yamamoto et al established a novel CAR-T cell that inhibits FasL-mediated T-cell apoptosis by truncating the intracellular death domain of Fas or introducing a point mutation (I246N) in the Fas death domain.137 These modifications allow CAR-T cells to become resistant to cancer-induced FasL-mediated apoptosis by inhibiting the recruitment of FADD into the apoptotic complex and preventing DISC formation. The failure of DISC formation enhanced CAR-T cell persistence and anticancer activity in a murine B16 melanoma cancer model. Importantly, Fas-engineered T cells did not show uncontrolled proliferation, at least in in-vivo models, suggesting that modulation of T-cell extrinsic apoptosis may be a safe and feasible strategy.137 Similarly, another study reported that CRISPR-mediated KO of Fas reduced the AICD of anti-CD19 CAR-T cells during chronic exposure to target cells, which led to increased T-cell expansion.138 In addition to modifying extrinsic apoptosis in T cells, Charo et al generated murine T cells that overexpress BCL-2 and tested whether this modification leads to enhanced anticancer activity of cytolytic T cells by preventing apoptosis.35 The authors identified that BCL-2 overexpressing T cells show superior anticancer activity compared with wild-type T cells by improving long-term survival in the absence of a survival signal. Recently, another study revealed that constitutive overexpression of BCL-2 in CAR-T cells improves CAR-T cells proliferation and reduces AICD in CAR-T cells.139 Our group further demonstrates that higher levels of BCL-2 expression in CAR-T cells of patients with lymphoma significantly correlate with enhanced clinical response (ie, CAR-T persistence and overall survival) of CAR-T therapy, suggesting that modulating intrinsic apoptosis in T cells is an important strategy to enhance CAR-T therapy.33 In addition to BCL-2, there are other critical anti-apoptotic regulators (ie, MCL-1 and BCL-xL) affecting T-cell survival and differentiation. Studies using transgenic expression of these anti-apoptotic regulators have suggested their potential implications in T cell-based immunotherapy. For instance, constitutive expression of BCL-xL rescued activation-induced cell death of CD8+ T cells in a viral infectious model.140 Enhanced expression of MCL-1 promotes long-term memory formation in the acute phase of vaccinia virus infections.141 Despite the beneficial effect of BCL-2 family overexpression in T cells, altering the BCL-2 signal in T cells requires additional attention, as the constitutive expression of BCL-2 in murine T cells promoted T-cell lymphoma development (ie, 18 of 68 BCL-2 transgenic mice developed T-cell lymphoma).142
Finally, developing strategies to avoid T-cell apoptosis would be beneficial for preventing potential apoptosis of T cells when combining immunotherapies with pro-apoptotic drugs. Our group recently reported a novel strategy to overcome venetoclax-mediated CAR-T cell toxicity by developing venetoclax-resistant CAR-T cells (ven-CAR-T).33 In ven-CAR-T, we introduced a mutant form of BCL-2 containing a point mutation at the 104 amino acid residue (Phe104Leu or F104L) located in the binding pocket of venetoclax. Accordingly, venetoclax cannot bind to BCL-2(F104L) and loses its inhibitory function.143 144 Therefore, by overexpressing BCL-2(F104L) in ven-CAR-T, ven-CAR-T showed strong resistance to venetoclax, leading to a significant enhancement of CAR-T cells and venetoclax combination effects.
Conclusions
As immunotherapy is ready to make its next steps and advances as a line of therapy for patients, a critical factor is the development of strategies to overcome the current limitations that preclude responses in a significant subset of patients. This review discussed the dual role of apoptosis in T cell-based immunotherapy, from cancer (ie, resistance to apoptosis) as well as a T-cell side (ie, apoptotic death).
Most cancers are characterized by resistance to apoptosis through several mechanisms, including neutralizing pro-apoptotic signals by either increasing expression of anti-apoptotic molecules such as IAPs and BCL-2 or decreasing positive regulators of the death receptor-mediated apoptosis. Therefore, increasing the sensitivity of cancer cells to apoptosis should be considered a vital strategy to improve the anticancer activity of T cell-based immunotherapies. Combining pro-apoptotic drugs may be an appealing approach for sensitizing cancer cells to T cell-mediated death; for instance, inhibiting key anti-apoptotic regulators (IAPs and BCL-2) by targeted small molecules (SMAC mimetics and ABT737) enhanced CAR-T cell-mediated anticancer activities. However, because such drugs may also induce T-cell apoptosis, careful consideration of the administration timing/dose of pro-apoptotic drugs or apoptosis-sensitizing treatments must be made to determine the optimal therapeutic regimens.
Regarding T-cell apoptosis, cancer evades immunotherapy by secreting pro-apoptotic inducers against cytolytic T cells and developing a hostile TME. Thus, there is a clear need for combinations that can prevent these evasion mechanisms. CAR-T cell therapy presents a versatile option not only for combination strategies but also for the possibility of performing genetic engineering (eg, Fas KO, mutant Fas, or constitutive overexpression of BCL-2). However, as a consequence of enhancing T-cell survival/expansion by aforementioned modulations, safety concerns such as abnormal lymphoproliferation and tumorigenesis of modified T cells appear. Therefore, it is critical to include safety switches in these models to maximize safety in clinical use (eg, the anti-inducible Caspase-9 system and antibody-mediated cellular cytotoxicity using a truncated epidermal growth factor receptor/anti-epidermal growth factor receptor antibody).
In conclusion, apoptosis is a crucial player in T cell-based immunotherapy. Deep knowledge of mechanisms of apoptosis resistance in cancer and T-cell biology is necessary to promote cancer cell apoptosis and prevent T-cell death. Several novel agents being developed together with the most recent advances in bioengineering will pave the way for the success of next-generation therapeutic combinations.
Footnotes
Twitter: @PuneethGuru_, @MarcoRuella
Contributors: YGL and MR wrote and revised the manuscript. NY, IC, PP, AC, LP, CTS, PG, and RP revised the manuscript. MR and YGL funded the project. MR supervised the project. All authors reviewed and approved the manuscript.
Funding: This research was supported by the Mark Foundation (MR), the Upenn TAPITMAT grant (MR), the Lymphoma Research Foundation (YGL), Basic Science Research Program through the national Research Foundatio of Korea (2022R1C1C1011603, YGL), NCI 1K99CA212302 and NCI R00CA212302 (MR), Laffey McHugh Foundation (MR), Parker Institute for Cancer Immunotherapy (MR), and Berman and Maguire Funds for Lymphoma Research at Penn (MR).
Competing interests: M. Ruella reports grants from the Mark Foundation, Upenn TAPITMAT, the Lymphoma Research Foundation, the NCI (1K99CA212302 and R00CA212302), the Parker Institute for Cancer Immunotherapy, and the Berman and Maguire Funds for Lymphoma Research at the University of Pennsylvania during the conduct of the study; grants from Novartis outside the submitted work; a patent for BCL-2 and CART pending; is listed as an inventor of CART technologies, University of Pennsylvania, partly licensed to Novartis, Tmunity, and viTToria Biotherapeutics; research funding from AbClon, Beckman Coulter, Lumicks, and ONI; consultancy for/honoraria from NanoString Technologies Inc. and GLG; advisory boards for AbClon, Bayer, Sana, Bristol Myers Squibb, GSK, and viTToria Biotherapeutics; and is a scientific founder of viTToria Biotherapeutics. No disclosures were reported by the other authors.
Provenance and peer review: Commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Ethics statements
Patient consent for publication
Not applicable.
References
- 1.Farkona S, Diamandis EP, Blasutig IM. Cancer immunotherapy: the beginning of the end of cancer? BMC Med 2016;14:73. 10.1186/s12916-016-0623-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Labrijn AF, Janmaat ML, Reichert JM, et al. Bispecific antibodies: a mechanistic review of the pipeline. Nat Rev Drug Discov 2019;18:585–608. 10.1038/s41573-019-0028-1 [DOI] [PubMed] [Google Scholar]
- 3.Esfandiari A, Cassidy S, Webster RM. Bispecific antibodies in oncology. Nat Rev Drug Discov 2022;21:411–2. 10.1038/d41573-022-00040-2 [DOI] [PubMed] [Google Scholar]
- 4.Ma J, Mo Y, Tang M, et al. Bispecific antibodies: from research to clinical application. Front Immunol 2021;12:626616. 10.3389/fimmu.2021.626616 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Przepiorka D, Ko C-W, Deisseroth A, et al. Fda approval: blinatumomab. Clin Cancer Res 2015;21:4035–9. 10.1158/1078-0432.CCR-15-0612 [DOI] [PubMed] [Google Scholar]
- 6.Ruella M, Gill S. How to train your T cell: genetically engineered chimeric antigen receptor T cells versus bispecific T-cell engagers to target CD19 in B acute lymphoblastic leukemia. Expert Opin Biol Ther 2015;15:761–6. 10.1517/14712598.2015.1009888 [DOI] [PubMed] [Google Scholar]
- 7.Pitt JM, Vétizou M, Daillère R, et al. Resistance mechanisms to immune-checkpoint blockade in cancer: tumor-intrinsic and -extrinsic factors. Immunity 2016;44:1255–69. 10.1016/j.immuni.2016.06.001 [DOI] [PubMed] [Google Scholar]
- 8.Cerrano M, Ruella M, Perales M-A, et al. The advent of CAR T-cell therapy for lymphoproliferative neoplasms: integrating research into clinical practice. Front Immunol 2020;11:888. 10.3389/fimmu.2020.00888 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Berdeja JG, Madduri D, Usmani SZ, et al. Ciltacabtagene autoleucel, a B-cell maturation antigen-directed chimeric antigen receptor T-cell therapy in patients with relapsed or refractory multiple myeloma (CARTITUDE-1): a phase 1b/2 open-label study. Lancet 2021;398:314–24. 10.1016/S0140-6736(21)00933-8 [DOI] [PubMed] [Google Scholar]
- 10.Maude SL, Laetsch TW, Buechner J, et al. Tisagenlecleucel in children and young adults with B-cell lymphoblastic leukemia. N Engl J Med 2018;378:439–48. 10.1056/NEJMoa1709866 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Munshi NC, Anderson LD, Shah N, et al. Idecabtagene vicleucel in relapsed and refractory multiple myeloma. N Engl J Med 2021;384:705–16. 10.1056/NEJMoa2024850 [DOI] [PubMed] [Google Scholar]
- 12.Neelapu SS, Locke FL, Bartlett NL, et al. Axicabtagene ciloleucel CAR T-cell therapy in refractory large B-cell lymphoma. N Engl J Med 2017;377:2531–44. 10.1056/NEJMoa1707447 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Schuster SJ, Bishop MR, Tam CS, et al. Tisagenlecleucel in adult relapsed or refractory diffuse large B-cell lymphoma. N Engl J Med 2019;380:45–56. 10.1056/NEJMoa1804980 [DOI] [PubMed] [Google Scholar]
- 14.Wang M, Munoz J, Goy A, et al. KTE-X19 CAR T-cell therapy in relapsed or refractory mantle-cell lymphoma. N Engl J Med 2020;382:1331–42. 10.1056/NEJMoa1914347 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chong EA, Ruella M, Schuster SJ, et al. Five-Year outcomes for refractory B-cell lymphomas with CAR T-cell therapy. N Engl J Med 2021;384:673–4. 10.1056/NEJMc2030164 [DOI] [PubMed] [Google Scholar]
- 16.Strasser A, O’Connor L, Dixit VM. Apoptosis signaling. Annu Rev Biochem 2000;69:217–45. 10.1146/annurev.biochem.69.1.217 [DOI] [PubMed] [Google Scholar]
- 17.Fulda S. Tumor resistance to apoptosis. Int J Cancer 2009;124:511–5. 10.1002/ijc.24064 [DOI] [PubMed] [Google Scholar]
- 18.Fairlie WD, Lee EF. Co-Operativity between Myc and Bcl-2 pro-survival proteins in cancer. Int J Mol Sci 2021;22:2841. 10.3390/ijms22062841 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.McMahon SB. Myc and the control of apoptosis. Cold Spring Harb Perspect Med 2014;4:a014407. 10.1101/cshperspect.a014407 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Voskoboinik I, Whisstock JC, Trapani JA. Perforin and granzymes: function, dysfunction and human pathology. Nat Rev Immunol 2015;15:388–400. 10.1038/nri3839 [DOI] [PubMed] [Google Scholar]
- 21.Schulze-Osthoff K, Ferrari D, Los M, et al. Apoptosis signaling by death receptors. Eur J Biochem 1998;254:439–59. 10.1046/j.1432-1327.1998.2540439.x [DOI] [PubMed] [Google Scholar]
- 22.Liu C-C, Walsh CM, Young J-E. Perforin: structure and function. Immunol Today 1995;16:194–201. 10.1016/0167-5699(95)80121-9 [DOI] [PubMed] [Google Scholar]
- 23.Froelich CJ, Orth K, Turbov J, et al. New paradigm for lymphocyte granule-mediated cytotoxicity. target cells bind and internalize granzyme B, but an endosomolytic agent is necessary for cytosolic delivery and subsequent apoptosis. J Biol Chem 1996;271:29073–9. 10.1074/jbc.271.46.29073 [DOI] [PubMed] [Google Scholar]
- 24.Trapani JA. Granzymes: a family of lymphocyte granule serine proteases. Genome Biol 2001;2:1–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Martínez-Lostao L, Anel A, Pardo J. How do cytotoxic lymphocytes kill cancer cells? Clin Cancer Res 2015;21:5047–56. 10.1158/1078-0432.CCR-15-0685 [DOI] [PubMed] [Google Scholar]
- 26.Ozaki T, Nakagawara A. Role of p53 in cell death and human cancers. Cancers (Basel) 2011;3:994–1013. 10.3390/cancers3010994 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Jiang Z, Liu Z, Li M, et al. Immunogenomics analysis reveals that TP53 mutations inhibit tumor immunity in gastric cancer. Transl Oncol 2018;11:1171–87. 10.1016/j.tranon.2018.07.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Maruyama R, Yamana K, Itoi T, et al. Absence of Bcl-2 and FAS/CD95/APO-1 predicts the response to immunotherapy in metastatic renal cell carcinoma. Br J Cancer 2006;95:1244–9. 10.1038/sj.bjc.6603359 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Upadhyay R, Boiarsky JA, Pantsulaia G, et al. A critical role for fas-mediated off-target tumor killing in T-cell immunotherapyfas mediates bystander tumor killing by T cells. Cancer Discov 2021;11:599–613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kelly JD, Dai J, Eschwege P, et al. Downregulation of Bcl-2 sensitises interferon-resistant renal cancer cells to Fas. Br J Cancer 2004;91:164–70. 10.1038/sj.bjc.6601895 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Fulda S, Meyer E, Debatin K-M. Inhibition of TRAIL-induced apoptosis by Bcl-2 overexpression. Oncogene 2002;21:2283–94. 10.1038/sj.onc.1205258 [DOI] [PubMed] [Google Scholar]
- 32.Dufva O, Koski J, Maliniemi P, et al. Integrated drug profiling and CRISPR screening identify essential pathways for CAR T-cell cytotoxicity. Blood 2020;135:597–609. 10.1182/blood.2019002121 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Lee YG, Guruprasad P, Ghilardi G, et al. Modulation of Bcl-2 in both T cells and tumor cells to enhance chimeric antigen receptor T-cell immunotherapy against cancer. Cancer Discov 2022;12:2372–91. 10.1158/2159-8290.CD-21-1026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Singh N, Lee YG, Shestova O, et al. Impaired death receptor signaling in leukemia causes antigen-independent resistance by inducing CAR T-cell dysfunction. Cancer Discov 2020;10:552–67. 10.1158/2159-8290.CD-19-0813 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Charo J, Finkelstein SE, Grewal N, et al. Bcl-2 overexpression enhances tumor-specific T-cell survival. Cancer Res 2005;65:2001–8. 10.1158/0008-5472.CAN-04-2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ricci MS, Zong W-X. Chemotherapeutic approaches for targeting cell death pathways. Oncologist 2006;11:342–57. 10.1634/theoncologist.11-4-342 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Mattarollo SR, Kenna T, Nieda M, et al. Chemotherapy pretreatment sensitizes solid tumor-derived cell lines to V alpha 24+ NKT cell-mediated cytotoxicity. Int J Cancer 2006;119:1630–7. 10.1002/ijc.22019 [DOI] [PubMed] [Google Scholar]
- 38.Todaro M, Orlando V, Cicero G, et al. Chemotherapy sensitizes colon cancer initiating cells to Vγ9Vδ2 T cell-mediated cytotoxicity. PLoS One 2013;8:e65145. 10.1371/journal.pone.0065145 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Wennerberg E, Sarhan D, Carlsten M, et al. Doxorubicin sensitizes human tumor cells to NK cell- and T-cell-mediated killing by augmented TRAIL receptor signaling. Int J Cancer 2013;133:1643–52. 10.1002/ijc.28163 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ifeadi V, Garnett-Benson C. Sub-lethal irradiation of human colorectal tumor cells imparts enhanced and sustained susceptibility to multiple death receptor signaling pathways. PLoS One 2012;7:e31762. 10.1371/journal.pone.0031762 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Garnett CT, Palena C, Chakarborty M, et al. Sublethal irradiation of human tumor cells modulates phenotype resulting in enhanced killing by cytotoxic T lymphocytes. Cancer Res 2004;64:7985–94. 10.1158/0008-5472.CAN-04-1525 [DOI] [PubMed] [Google Scholar]
- 42.Brooks AD, Jacobsen KM, Li W, et al. Bortezomib sensitizes human renal cell carcinomas to TRAIL apoptosis through increased activation of caspase-8 in the death-inducing signaling complex. Mol Cancer Res 2010;8:729–38. 10.1158/1541-7786.MCR-10-0022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kim H-B, Kim M-J, Lee S-H, et al. Amurensin G, a novel SIRT1 inhibitor, sensitizes TRAIL-resistant human leukemic K562 cells to TRAIL-induced apoptosis. Biochemical Pharmacology 2012;84:402–10. 10.1016/j.bcp.2012.03.014 [DOI] [PubMed] [Google Scholar]
- 44.Koschny R, Boehm C, Sprick MR, et al. Bortezomib sensitizes primary meningioma cells to TRAIL-induced apoptosis by enhancing formation of the death-inducing signaling complex. J Neuropathol Exp Neurol 2014;73:1034–46. 10.1097/NEN.0000000000000129 [DOI] [PubMed] [Google Scholar]
- 45.Seol D-W. P53-independent up-regulation of a TRAIL receptor DR5 by proteasome inhibitors: a mechanism for proteasome inhibitor-enhanced TRAIL-induced apoptosis. Biochem Biophys Res Commun 2011;416:222–5. 10.1016/j.bbrc.2011.11.053 [DOI] [PubMed] [Google Scholar]
- 46.Paz-Ares L, Luft A, Vicente D, et al. Pembrolizumab plus chemotherapy for squamous non-small-cell lung cancer. N Engl J Med 2018;379:2040–51. 10.1056/NEJMoa1810865 [DOI] [PubMed] [Google Scholar]
- 47.Reck M, Ciuleanu T-E, Dols MC, et al. Nivolumab (nivo) + ipilimumab (IPI) + 2 cycles of platinum-doublet chemotherapy (chemo) vs 4 cycles chemo as first-line (1L) treatment (tx) for stage iv/recurrent non-small cell lung cancer (NSCLC): checkmate 9la. JCO 2020;38:9501. 10.1200/JCO.2020.38.15_suppl.9501 [DOI] [Google Scholar]
- 48.Arina A, Beckett M, Fernandez C, et al. Tumor-reprogrammed resident T cells resist radiation to control tumors. Nat Commun 2019;10:3959. 10.1038/s41467-019-11906-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Shibayama Y, Tsukahara T, Emori M, et al. Implication of chemo-resistant memory T cells for immune surveillance in patients with sarcoma receiving chemotherapy. Cancer Sci 2017;108:1739–45. 10.1111/cas.13319 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Grayson JM, Zajac AJ, Altman JD, et al. Cutting edge: increased expression of Bcl-2 in antigen-specific memory CD8+ T cells. J Immunol 2000;164:3950–4. 10.4049/jimmunol.164.8.3950 [DOI] [PubMed] [Google Scholar]
- 51.Sabbagh L, Srokowski CC, Pulle G, et al. A critical role for TNF receptor-associated factor 1 and Bim down-regulation in CD8 memory T cell survival. Proc Natl Acad Sci USA 2006;103:18703–8. 10.1073/pnas.0602919103 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Berthelet J, Dubrez L. Regulation of apoptosis by inhibitors of apoptosis (IAPs). Cells 2013;2:163–87. 10.3390/cells2010163 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Fulda S, Vucic D. Targeting IAP proteins for therapeutic intervention in cancer. Nat Rev Drug Discov 2012;11:109–24. 10.1038/nrd3627 [DOI] [PubMed] [Google Scholar]
- 54.Scott FL, Denault J-B, Riedl SJ, et al. Xiap inhibits caspase-3 and -7 using two binding sites: evolutionarily conserved mechanism of IAPs. EMBO J 2005;24:645–55. 10.1038/sj.emboj.7600544 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Morrish E, Brumatti G, Silke J. Future therapeutic directions for Smac-mimetics. Cells 2020;9:406. 10.3390/cells9020406 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Michie J, Beavis PA, Freeman AJ, et al. Antagonism of IAPs enhances CAR T-cell efficacy. Cancer Immunol Res 2019;7:183–92. 10.1158/2326-6066.CIR-18-0428 [DOI] [PubMed] [Google Scholar]
- 57.Beug ST, Beauregard CE, Healy C, et al. Smac mimetics synergize with immune checkpoint inhibitors to promote tumour immunity against glioblastoma. Nat Commun 2017;8:1–15. 10.1038/ncomms14278 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Song EZ, Wang X, Philipson BI, et al. The IAP antagonist birinapant enhances chimeric antigen receptor T cell therapy for glioblastoma by overcoming antigen heterogeneity. Mol Ther Oncolytics 2022;27:288–304. 10.1016/j.omto.2022.11.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Ye W, Gunti S, Allen CT, et al. ASTX660, an antagonist of cIAP1/2 and XIAP, increases antigen processing machinery and can enhance radiation-induced immunogenic cell death in preclinical models of head and neck cancer. OncoImmunology 2020;9. 10.1080/2162402X.2019.1710398 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Adams JM, Cory S. The bcl-2 apoptotic switch in cancer development and therapy. Oncogene 2007;26:1324–37. 10.1038/sj.onc.1210220 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Correia C, Schneider PA, Dai H, et al. Bcl2 mutations are associated with increased risk of transformation and shortened survival in follicular lymphoma. Blood 2015;125:658–67. 10.1182/blood-2014-04-571786 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Perini GF, Ribeiro GN, Pinto Neto JV, et al. Bcl-2 as therapeutic target for hematological malignancies. J Hematol Oncol 2018;11:65. 10.1186/s13045-018-0608-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Souers AJ, Leverson JD, Boghaert ER, et al. Abt-199, a potent and selective Bcl-2 inhibitor, achieves antitumor activity while sparing platelets. Nat Med 2013;19:202–8. 10.1038/nm.3048 [DOI] [PubMed] [Google Scholar]
- 64.Kohlhapp FJ, Haribhai D, Mathew R, et al. Venetoclax increases intratumoral effector T cells and antitumor efficacy in combination with immune checkpoint blockade. Cancer Discov 2021;11:68–79. 10.1158/2159-8290.CD-19-0759 [DOI] [PubMed] [Google Scholar]
- 65.Karlsson H, Lindqvist AC, Fransson M, et al. Combining CAR T cells and the Bcl-2 family apoptosis inhibitor ABT-737 for treating B-cell malignancy. Cancer Gene Ther 2013;20:386–93. 10.1038/cgt.2013.35 [DOI] [PubMed] [Google Scholar]
- 66.Gentle IE, Moelter I, Lechler N, et al. Inhibitors of apoptosis proteins (IAPs) are required for effective T-cell expansion/survival during antiviral immunity in mice. Blood 2014;123:659–68. 10.1182/blood-2013-01-479543 [DOI] [PubMed] [Google Scholar]
- 67.Yang M, Wang L, Ni M, et al. Pre-sensitization of malignant B cells through venetoclax significantly improves the cytotoxic efficacy of CD19.CAR-T cells. Front Immunol 2020;11:608167. 10.3389/fimmu.2020.608167 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Yi JS, Cox MA, Zajac AJ. T-Cell exhaustion: characteristics, causes and conversion. Immunology 2010;129:474–81. 10.1111/j.1365-2567.2010.03255.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Bates GJ, Fox SB, Han C, et al. Quantification of regulatory T cells enables the identification of high-risk breast cancer patients and those at risk of late relapse. J Clin Oncol 2006;24:5373–80. 10.1200/JCO.2006.05.9584 [DOI] [PubMed] [Google Scholar]
- 70.Curiel TJ, Coukos G, Zou L, et al. Specific recruitment of regulatory T cells in ovarian carcinoma fosters immune privilege and predicts reduced survival. Nat Med 2004;10:942–9. 10.1038/nm1093 [DOI] [PubMed] [Google Scholar]
- 71.Rolf J, Zarrouk M, Finlay DK, et al. Ampkα1: a glucose sensor that controls CD8 T-cell memory. Eur J Immunol 2013;43:889–96. 10.1002/eji.201243008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Sinclair LV, Rolf J, Emslie E, et al. Control of amino-acid transport by antigen receptors coordinates the metabolic reprogramming essential for T cell differentiation. Nat Immunol 2013;14:500–8. 10.1038/ni.2556 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Wang R, Dillon CP, Shi LZ, et al. The transcription factor Myc controls metabolic reprogramming upon T lymphocyte activation. Immunity 2011;35:871–82. 10.1016/j.immuni.2011.09.021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Jiang Y, Li Y, Zhu B. T-Cell exhaustion in the tumor microenvironment. Cell Death Dis 2015;6:e1792. 10.1038/cddis.2015.162 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Lu B, Finn OJ. T-Cell death and cancer immune tolerance. Cell Death Differ 2008;15:70–9. 10.1038/sj.cdd.4402274 [DOI] [PubMed] [Google Scholar]
- 76.Pauken KE, Wherry EJ. Overcoming T cell exhaustion in infection and cancer. Trends Immunol 2015;36:265–76. 10.1016/j.it.2015.02.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Beatty GL, Moon EK. Chimeric antigen receptor T cells are vulnerable to immunosuppressive mechanisms present within the tumor microenvironment. Oncoimmunology 2014;3:e970027. 10.4161/21624011.2014.970027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Jiang X, Wang J, Deng X, et al. Role of the tumor microenvironment in PD-L1/PD-1-mediated tumor immune escape. Mol Cancer 2019;18:10. 10.1186/s12943-018-0928-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Yeku OO, Purdon TJ, Koneru M, et al. Armored CAR T cells enhance antitumor efficacy and overcome the tumor microenvironment. Sci Rep 2017;7:10541. 10.1038/s41598-017-10940-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Croft M. The role of TNF superfamily members in T-cell function and diseases. Nat Rev Immunol 2009;9:271–85. 10.1038/nri2526 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Volpe E, Sambucci M, Battistini L, et al. Fas-fas ligand: checkpoint of T cell functions in multiple sclerosis. Front Immunol 2016;7:382. 10.3389/fimmu.2016.00382 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Green DR, Droin N, Pinkoski M. Activation-Induced cell death in T cells. Immunol Rev 2003;193:70–81. 10.1034/j.1600-065x.2003.00051.x [DOI] [PubMed] [Google Scholar]
- 83.Hahne M, Rimoldi D, Schröter M, et al. Melanoma cell expression of Fas (APO-1/CD95) ligand: implications for tumor immune escape. Science 1996;274:1363–6. 10.1126/science.274.5291.1363 [DOI] [PubMed] [Google Scholar]
- 84.Niehans GA, Brunner T, Frizelle SP, et al. Human lung carcinomas express fas ligand. Cancer Res 1997;57:1007–12. [PubMed] [Google Scholar]
- 85.O’Connell J, O’Sullivan GC, Collins JK, et al. The Fas counterattack: Fas-mediated T cell killing by colon cancer cells expressing Fas ligand. J Exp Med 1996;184:1075–82. 10.1084/jem.184.3.1075 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Strand S, Hofmann WJ, Hug H, et al. Lymphocyte apoptosis induced by CD95 (APO-1/Fas) ligand-expressing tumor cells -- a mechanism of immune evasion? Nat Med 1996;2:1361–6. 10.1038/nm1296-1361 [DOI] [PubMed] [Google Scholar]
- 87.Bron LP, Scolyer RA, Thompson JF, et al. Histological expression of tumour necrosis factor-related apoptosis-inducing ligand (TRAIL) in human primary melanoma. Pathology 2004;36:561–5. 10.1080/00313020400011268 [DOI] [PubMed] [Google Scholar]
- 88.Cross SS, Harrison RF, Balasubramanian SP, et al. Expression of receptor activator of nuclear factor kappabeta ligand (RANKL) and tumour necrosis factor related, apoptosis inducing ligand (TRAIL) in breast cancer, and their relations with osteoprotegerin, oestrogen receptor, and clinicopathological variables. J Clin Pathol 2006;59:716–20. 10.1136/jcp.2005.030031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Shiraki K, Yamanaka T, Inoue H, et al. Expression of TNF-related apoptosis-inducing ligand in human hepatocellular carcinoma. Int J Oncol 2005;26:1273–81. 10.3892/ijo.26.5.1273 [DOI] [PubMed] [Google Scholar]
- 90.Spierings DCJ, de Vries EGE, Timens W, et al. Expression of TRAIL and TRAIL death receptors in stage III non-small cell lung cancer tumors. Clin Cancer Res 2003;9:3397–405. [PubMed] [Google Scholar]
- 91.Andreola G, Rivoltini L, Castelli C, et al. Induction of lymphocyte apoptosis by tumor cell secretion of fasl-bearing microvesicles. J Exp Med 2002;195:1303–16. 10.1084/jem.20011624 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Kim JW, Wieckowski E, Taylor DD, et al. Fas ligand-positive membranous vesicles isolated from sera of patients with oral cancer induce apoptosis of activated T lymphocytes. Clin Cancer Res 2005;11:1010–20. 10.1158/1078-0432.1010.11.3 [DOI] [PubMed] [Google Scholar]
- 93.Taylor DD, Gerçel-Taylor C, Lyons KS, et al. T-Cell apoptosis and suppression of T-cell receptor/CD3-zeta by Fas ligand-containing membrane vesicles shed from ovarian tumors. Clin Cancer Res 2003;9:5113–9. [PubMed] [Google Scholar]
- 94.Taylor DD, Gerçel-Taylor C. Tumour-Derived exosomes and their role in cancer-associated T-cell signalling defects. Br J Cancer 2005;92:305–11. 10.1038/sj.bjc.6602316 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.June CH, O’Connor RS, Kawalekar OU, et al. Car T cell immunotherapy for human cancer. Science 2018;359:1361–5. 10.1126/science.aar6711 [DOI] [PubMed] [Google Scholar]
- 96.Tschumi BO, Dumauthioz N, Marti B, et al. Cart cells are prone to Fas- and DR5-mediated cell death. J Immunother Cancer 2018;6:71. 10.1186/s40425-018-0385-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Künkele A, Johnson AJ, Rolczynski LS, et al. Functional tuning of cars reveals signaling threshold above which CD8+ CTL antitumor potency is attenuated due to cell fas-fasl-dependent AICD. Cancer Immunol Res 2015;3:368–79. 10.1158/2326-6066.CIR-14-0200 [DOI] [PubMed] [Google Scholar]
- 98.Zhu J, Powis de Tenbossche CG, Cané S, et al. Resistance to cancer immunotherapy mediated by apoptosis of tumor-infiltrating lymphocytes. Nat Commun 2017;8:1404. 10.1038/s41467-017-00784-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Gomes-Silva D, Mukherjee M, Srinivasan M, et al. Tonic 4-1BB costimulation in chimeric antigen receptors impedes T cell survival and is vector-dependent. Cell Rep 2017;21:17–26. 10.1016/j.celrep.2017.09.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Liu F-T, Rabinovich GA. Galectins as modulators of tumour progression. Nat Rev Cancer 2005;5:29–41. 10.1038/nrc1527 [DOI] [PubMed] [Google Scholar]
- 101.Leffler H, Carlsson S, Hedlund M, et al. Introduction to galectins. Glycoconj J 2002;19:433–40. 10.1023/B:GLYC.0000014072.34840.04 [DOI] [PubMed] [Google Scholar]
- 102.Yang R-Y, Rabinovich GA, Liu F-T. Galectins: structure, function and therapeutic potential. Expert Rev Mol Med 2008;10:e17. 10.1017/S1462399408000719 [DOI] [PubMed] [Google Scholar]
- 103.Dumic J, Dabelic S, Flögel M. Galectin-3: an open-ended story. Biochim Biophys Acta 2006;1760:616–35. 10.1016/j.bbagen.2005.12.020 [DOI] [PubMed] [Google Scholar]
- 104.Fukumori T, Takenaka Y, Yoshii T, et al. Cd29 and CD7 mediate galectin-3-induced type II T-cell apoptosis. Cancer Res 2003;63:8302–11. [PubMed] [Google Scholar]
- 105.Li W, Jian-jun W, Xue-Feng Z, et al. CD133(+) human pulmonary adenocarcinoma cells induce apoptosis of CD8(+) T cells by highly expressed galectin-3. Clin Invest Med 2010;33:E44–53. 10.25011/cim.v33i1.11837 [DOI] [PubMed] [Google Scholar]
- 106.Peng W, Wang HY, Miyahara Y, et al. Tumor-Associated galectin-3 modulates the function of tumor-reactive T cells. Cancer Res 2008;68:7228–36. 10.1158/0008-5472.CAN-08-1245 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Stillman BN, Hsu DK, Pang M, et al. Galectin-3 and galectin-1 bind distinct cell surface glycoprotein receptors to induce T cell death. J Immunol 2006;176:778–89. 10.4049/jimmunol.176.2.778 [DOI] [PubMed] [Google Scholar]
- 108.Zubieta MR, Furman D, Barrio M, et al. Galectin-3 expression correlates with apoptosis of tumor-associated lymphocytes in human melanoma biopsies. Am J Pathol 2006;168:1666–75. 10.2353/ajpath.2006.050971 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Xue H, Liu L, Zhao Z, et al. The N-terminal tail coordinates with carbohydrate recognition domain to mediate galectin-3 induced apoptosis in T cells. Oncotarget 2017;8:49824–38. 10.18632/oncotarget.17760 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Banh A, Zhang J, Cao H, et al. Tumor galectin-1 mediates tumor growth and metastasis through regulation of T-cell apoptosis. Cancer Res 2011;71:4423–31. 10.1158/0008-5472.CAN-10-4157 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Huflejt ME, Leffler H. Galectin-4 in normal tissues and cancer. Glycoconj J 2003;20:247–55. 10.1023/B:GLYC.0000025819.54723.a0 [DOI] [PubMed] [Google Scholar]
- 112.Lu L-H, Nakagawa R, Kashio Y, et al. Characterization of galectin-9-induced death of Jurkat T cells. J Biochem 2007;141:157–72. 10.1093/jb/mvm019 [DOI] [PubMed] [Google Scholar]
- 113.Nambiar DK, Aguilera T, Cao H, et al. Galectin-1-driven T cell exclusion in the tumor endothelium promotes immunotherapy resistance. J Clin Invest 2019;129:5553–67. 10.1172/JCI129025 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Norambuena A, Metz C, Vicuña L, et al. Galectin-8 induces apoptosis in Jurkat T cells by phosphatidic acid-mediated ERK1/2 activation supported by protein kinase A down-regulation. J Biol Chem 2009;284:12670–9. 10.1074/jbc.M808949200 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Perillo NL, Pace KE, Seilhamer JJ, et al. Apoptosis of T cells mediated by galectin-1. Nature 1995;378:736–9. 10.1038/378736a0 [DOI] [PubMed] [Google Scholar]
- 116.Sturm A, Lensch M, André S, et al. Human galectin-2: novel inducer of T cell apoptosis with distinct profile of caspase activation. The Journal of Immunology 2004;173:3825–37. 10.4049/jimmunol.173.6.3825 [DOI] [PubMed] [Google Scholar]
- 117.Tang D, Gao J, Wang S, et al. Apoptosis and anergy of T cell induced by pancreatic stellate cells-derived galectin-1 in pancreatic cancer. Tumour Biol 2015;36:5617–26. 10.1007/s13277-015-3233-5 [DOI] [PubMed] [Google Scholar]
- 118.Stowell SR, Qian Y, Karmakar S, et al. Differential roles of galectin-1 and galectin-3 in regulating leukocyte viability and cytokine secretion. The Journal of Immunology 2008;180:3091–102. 10.4049/jimmunol.180.5.3091 [DOI] [PubMed] [Google Scholar]
- 119.Chang F, Li R, Ladisch S. Shedding of gangliosides by human medulloblastoma cells. Experimental Cell Research 1997;234:341–6. 10.1006/excr.1997.3619 [DOI] [PubMed] [Google Scholar]
- 120.Chahlavi A, Rayman P, Richmond AL, et al. Glioblastomas induce T-lymphocyte death by two distinct pathways involving gangliosides and CD70. Cancer Res 2005;65:5428–38. 10.1158/0008-5472.CAN-04-4395 [DOI] [PubMed] [Google Scholar]
- 121.Kudo D, Rayman P, Horton C, et al. Gangliosides expressed by the renal cell carcinoma cell line SK-RC-45 are involved in tumor-induced apoptosis of T cells. Cancer Res 2003;63:1676–83. [PubMed] [Google Scholar]
- 122.Bharti AC, Singh SM. Induction of apoptosis in bone marrow cells by gangliosides produced by a T cell lymphoma. Immunol Lett 2000;72:39–48. 10.1016/s0165-2478(00)00156-5 [DOI] [PubMed] [Google Scholar]
- 123.Sa G, Das T, Moon C, et al. Gd3, an overexpressed tumor-derived ganglioside, mediates the apoptosis of activated but not resting T cells. Cancer Res 2009;69:3095–104. 10.1158/0008-5472.CAN-08-3776 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Sa G. Tumor gangliosides and T cells a deadly encounter. Front Biosci 2012;S4:502–19. 10.2741/s281 [DOI] [PubMed] [Google Scholar]
- 125.Alves NL, Derks IAM, Berk E, et al. The noxa/mcl-1 axis regulates susceptibility to apoptosis under glucose limitation in dividing T cells. Immunity 2006;24:703–16. 10.1016/j.immuni.2006.03.018 [DOI] [PubMed] [Google Scholar]
- 126.Chang C-H, Qiu J, O’Sullivan D, et al. Metabolic competition in the tumor microenvironment is a driver of cancer progression. Cell 2015;162:1229–41. 10.1016/j.cell.2015.08.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.O’Neill LAJ, Kishton RJ, Rathmell J. A guide to immunometabolism for immunologists. Nat Rev Immunol 2016;16:553–65. 10.1038/nri.2016.70 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Voss K, Luthers CR, Pohida K, et al. Fatty acid synthase contributes to restimulation-induced cell death of human CD4 T cells. Front Mol Biosci 2019;6:106. 10.3389/fmolb.2019.00106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Fallarino F, Grohmann U, Vacca C, et al. T cell apoptosis by tryptophan catabolism. Cell Death Differ 2002;9:1069–77. 10.1038/sj.cdd.4401073 [DOI] [PubMed] [Google Scholar]
- 130.Liberti MV, Locasale JW. The warburg effect: how does it benefit cancer cells? Trends Biochem Sci 2016;41:211–8. 10.1016/j.tibs.2015.12.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Gatenby RA, Gillies RJ. Why do cancers have high aerobic glycolysis? Nat Rev Cancer 2004;4:891–9. 10.1038/nrc1478 [DOI] [PubMed] [Google Scholar]
- 132.Calcinotto A, Filipazzi P, Grioni M, et al. Modulation of microenvironment acidity reverses anergy in human and murine tumor-infiltrating T lymphocytes. Cancer Res 2012;72:2746–56. 10.1158/0008-5472.CAN-11-1272 [DOI] [PubMed] [Google Scholar]
- 133.Kim JY, Cheng X, Wölfl S. Acidic stress induced G1 cell cycle arrest and intrinsic apoptotic pathway in jurkat T-lymphocytes. Exp Cell Res 2017;350:140–6. 10.1016/j.yexcr.2016.11.015 [DOI] [PubMed] [Google Scholar]
- 134.Kiang JG, Krishnan S, Lu X, et al. Inhibition of inducible nitric-oxide synthase protects human T cells from hypoxia-induced apoptosis. Mol Pharmacol 2008;73:738–47. 10.1124/mol.107.041079 [DOI] [PubMed] [Google Scholar]
- 135.Sun J, Zhang Y, Yang M, et al. Hypoxia induces T-cell apoptosis by inhibiting chemokine C receptor 7 expression: the role of adenosine receptor A (2). Cell Mol Immunol 2010;7:77–82. 10.1038/cmi.2009.105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Fraietta JA, Lacey SF, Orlando EJ, et al. Determinants of response and resistance to CD19 chimeric antigen receptor (CAR) T cell therapy of chronic lymphocytic leukemia. Nat Med 2018;24:563–71. 10.1038/s41591-018-0010-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Yamamoto TN, Lee P-H, Vodnala SK, et al. T cells genetically engineered to overcome death signaling enhance adoptive cancer immunotherapy. J Clin Invest 2019;129:1551–65. 10.1172/JCI121491 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Ren J, Zhang X, Liu X, et al. A versatile system for rapid multiplex genome-edited CAR T cell generation. Oncotarget 2017;8:17002–11. 10.18632/oncotarget.15218 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Wang H, Han P, Qi X, et al. Bcl-2 enhances chimeric antigen receptor T cell persistence by reducing activation-induced apoptosis. Cancers (Basel) 2021;13:197. 10.3390/cancers13020197 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Petschner F, Zimmerman C, Strasser A, et al. Constitutive expression of Bcl-xL or Bcl-2 prevents peptide antigen-induced T cell deletion but does not influence T cell homeostasis after a viral infection. Eur J Immunol 1998;28:560–9. [DOI] [PubMed] [Google Scholar]
- 141.Gui J, Hu Z, Tsai C-Y, et al. MCL1 enhances the survival of CD8+ memory T cells after viral infection. J Virol 2015;89:2405–14. 10.1128/JVI.02480-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Linette G, Hess J, Sentman C, et al. Peripheral T-cell lymphoma in lckpr-bcl-2 transgenic mice. Blood 1995;86:1255–60. 10.1182/blood.V86.4.1255.bloodjournal8641255 [DOI] [PubMed] [Google Scholar]
- 143.Tahir SK, Smith ML, Hessler P, et al. Potential mechanisms of resistance to venetoclax and strategies to circumvent it. BMC Cancer 2017;17:399. 10.1186/s12885-017-3383-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Birkinshaw RW, Gong J-N, Luo CS, et al. Structures of Bcl-2 in complex with venetoclax reveal the molecular basis of resistance mutations. Nat Commun 2019;10:2385. 10.1038/s41467-019-10363-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
jitc-2022-005967supp001.pdf (21.8KB, pdf)
jitc-2022-005967supp002.pdf (129.6KB, pdf)