1. WHAT IS NEW OR DIFFERENT
Inclusion of continuous glucose monitoring (CGM) targets for children, adolescents, and young adults <25 years.
Emphasis on individualized care plans that make use of effective educational strategies to achieve glucose targets that are person‐centered and designed to empower young people and caregivers. These plans should incorporate cognitive behavioral techniques that encompass:
problem‐solving
goal setting
communication skills
motivational interviewing
family conflict resolution
coping skills, and stress management
Adoption of a unified fingerstick capillary glucose (SMBG) target of between 4 and 10 mmol/L (70–180 mg/dl), which aligns with the target CGM time in range (TIR), while emphasizing a tighter fasting target range of 4–8 mmol/L (70–144 mg/dl).
Recognition that disparities in the social determinants of health (SDOH) and inequitable access to modern diabetes therapies represent significant barriers to achieving glucose targets and optimizing clinical outcomes. Health stakeholders are responsible for addressing this disparity.
2. EXECUTIVE SUMMARY AND RECOMMENDATIONS
- Achieving target glucose levels assessed through CGM, HbA1c, and/or SMBG:
- Reduces risks of acute and chronic complications of diabetes A
- Minimizes the detrimental effects of hypoglycemia and hyperglycemia on brain development, cognitive function, mood and quality of life B
- Target HbA1c for young people with diabetes should be <53 mmol/mol (<7.0%) A
- An HbA1c target of <48 mmol/mol (6.5%) is recommended for the remission phase or early stage 3 diabetes “honeymoon” period and in populations with access to advanced technology combined with a highly skilled specialized health care professional service adept in diabetes education E
- HbA1c assessments are recommended every 3 months E
- CGM metrics, recorded over a 14‐day period, should have time spent as follows: B
- >70% between 3.9 and 10 mmol/L (70–180 mg/dl)
- <4%: <3.9 mmol/L (70 mg/dl)
- <1%: <3.0 mmol/L (54 mg/dl)
- <25%: >10 mmol/L (180 mg/dl)
- <5%: >13.9 mmol/L (250 mg/dl)
- Glycemic variability (coefficient of variation, [%CV]) target ≤36%
SMBG should be assessed at least 6 times a day for a person with diabetes taking insulin B
Recommended target glucose values are between 4 and 10 mmol (70–180 mg/dl), with a narrower fasting target range of 4–8 mmol/L (70–144 mg/dl). E
- Less stringent HbA1c, CGM, or SMBG targets are only advisable when achieving the standard target is assessed as being detrimental to the overall well‐being of the person with diabetes or their caregivers. Factors to consider when setting a less stringent target include (but are not limited to):
- access to insulin analogs, advanced insulin delivery technology (for example automated insulin delivery), supplies needed to regularly check capillary blood glucose levels, or CGM needed to safely achieve targets E
- Underlying significant psychosocial health concerns exacerbated by efforts to achieve target glucose levels E
A multidisciplinary education team should clearly and collectively communicate recommended glycemic targets; sharing the same philosophy and goals and speaking with “one voice” has beneficial effects on glycemic and psychosocial outcomes. B
Individualized care plans are recommended to help a person with diabetes achieve glycemic targets. E
Data collection and between‐center benchmarking can improve the proportion of people with diabetes reaching glycemic targets. B
Addressing social determinants of health, and improving access to the healthcare team, insulin, and technologies increases the proportion of people reaching glycemic targets. A
3. THE IMPORTANCE OF SETTING GLYCEMIC TARGETS
Glycemic targets for young people with diabetes are needed as optimizing glycemia reduces short and long‐term complications. 1 , 2 In addition to protecting against micro‐ and macrovascular complications, of particular importance in pediatrics is the negative association of hypoglycemia and hyperglycemia on cognition and brain structure, 3 especially in individuals with early onset diabetes. 4 Further, the wider impact of diabetes on healthcare systems and health economics is an important driver to target better glycemic outcomes to prevent future complications. 5 , 6
Diabetes registries have shown steady improvement in median HbA1c levels in recent decades, yet only a minority of young people attain current glycemic targets. 7 The improvements that have been demonstrated can be attributed to multiple factors, including how healthcare teams set and communicate glycemic targets, improved therapeutics (insulin analogs, CGM), highly skilled and knowledgeable workforce, and, recently, the use of automated insulin delivery systems. Nevertheless, social determinants of health, pediatric diabetes workforce constraints, and access to improved therapeutics remain significant barriers preventing more young people from reaching target glycemia, and further, drives health inequity. 8 , 9
Setting glycemic targets has been standard practice for diabetes organizations, including ISPAD, the American Diabetes Association (ADA), and the National Institute of Clinical Excellence (NICE) in the United Kingdom for 20 years, and have been regularly updated when evidence has supported change. 10 For example, when different stakeholders published divergent HbA1c targets, and lower HbA1c targets were shown not to increase the rates of severe hypoglycemia, 11 lower targets were adopted. It is important to recognize that setting targets contributes to improving glycemia as shown by the observation that a combination of setting a lower target HbA1c and consistency among members of teams within centers is associated with lower center HbA1c levels. 11 , 12 It is essential that target setting is a collaborative discussion with the person with diabetes (including caregivers) and health care professionals. Furthermore, prospective audit activity, involvement in data registries, and clinical benchmarking, including quality improvement implementation, are also associated with overall improvements in glycemic outcomes. 13 , 14
Health care professionals and people with diabetes now have a wide array of tools to assess glycemia, including self‐monitored capillary blood glucose (SMBG) values, HbA1c, and CGM. While traditionally HbA1c has been the gold standard, there are limitations to this measurement as discussed later. Correspondingly, with rapidly increasing adoption of CGM, which arguably avoids these limitations, CGM metric reporting has been standardized and CGM metrics are included in this chapter. The recent COVID‐19 global pandemic, and increased opportunities for the use of video or phone appointments between a person with diabetes and/or their carer and the health care professional, has highlighted the utility of CGM metrics to assess glycemia when laboratory measurement of HbA1c level is not available. While disparities also exist for accessing telemedicine including implicit bias, well‐developed work plans can expand the population who can benefit from this health delivery method. 15 Nevertheless, not all young people can access CGM and are reliant on SMBG and/or HbA1c measurement. Using all available forms of glycemic data, in combination if available, will give the most accurate account of glycemia to help guide therapy.
Individualized glycemic target setting above the stated HbA1c target has been emphasized in recent consensus statements. 16 , 17 This was included to address concerns that for some young people with diabetes, particularly in limited resource settings, (see ISPAD 2022 Consensus Guidelines Chapter 25 on Managing Diabetes in children and adolescents in Limited Resource Setting) stringent HbA1c targets may increase the risk of severe hypoglycemia, or cause psychological distress (for the person with diabetes and/or their caregivers) through treatment burden that outweighs the long‐term benefit of a lower HbA1c. Although historically lower HbA1c was considered a risk factor for severe hypoglycemia, this association is no longer observed with contemporary intensive management. 18 For example, data registries have demonstrated that the overall incidence of severe hypoglycemia has decreased at the same time overall HbA1c has improved. 19 Access to diabetes technology, including CGM with or without automation of insulin delivery, can further reduce the risk of severe hypoglycemia while making it possible to achieve target glycemia (see ISPAD 2022 Consensus Guidelines Chapter 16 Diabetes Technologies: Glucose monitoring, and Chapter 17 Diabetes Technologies: Insulin Delivery). Therefore, outside of limited resource settings, risk of severe hypoglycemia can no longer be justified as a reason for a higher HbA1c target in the majority of cases. However, if setting stringent glycemic targets is considered to have an overall negative impact on psychological wellness (either for the person with diabetes and/or their caregivers), which may include severe anxiety that outweighs the long‐term benefit of optimizing glucose values, a higher glycemic target may be appropriate in combination with efforts to address the barriers to healthier glycemia. Other exceptions occur in certain situations, for example, in a person with diabetes and a limited lifespan or neonatal diabetes, and in situations where stringent glycemic targets are unattainable and will add management burden over any improvement in short or long‐term morbidity and mortality.
In ISPAD 2022 Consensus Guidelines Chapter 6 on Diabetes Education in Children and Adolescents, we highlight the importance of the multidisciplinary education team sharing the same philosophy and goals and speaking with “one voice,” with beneficial effects on metabolic and psychosocial outcomes. Education should be person‐centered, with a personalized diabetes educational approach being an integral part of the psychosocial support for young people with diabetes and their families. (See ISPAD 2022 Consensus Guideline Chapter 15 on Psychological Care of children and adolescents with type 1 diabetes). Therefore, for the majority of young people with diabetes, the priority of the multidisciplinary team is to develop (in consultation with the person with diabetes and their caregivers) an individualized care plan to achieve the ISPAD recommended targets, rather than individualizing the glycemic target itself.
4. MEASURES OF GLYCEMIA AND TARGETS
4.1. Glycated Hemoglobin
4.1.1. Target
A target of <53 mmol/mol (<7.0%) is recommended for all young people with diabetes (Figure 1). Individualized care plans should be a collaboration between the young people with diabetes, their caregivers, and the multidisciplinary team. Where barriers exist to achieving this target (for example access to insulin analogs, advanced technologies like CGM and automated insulin delivery, psychological distress), individualized targets may be selected.
FIGURE 1.
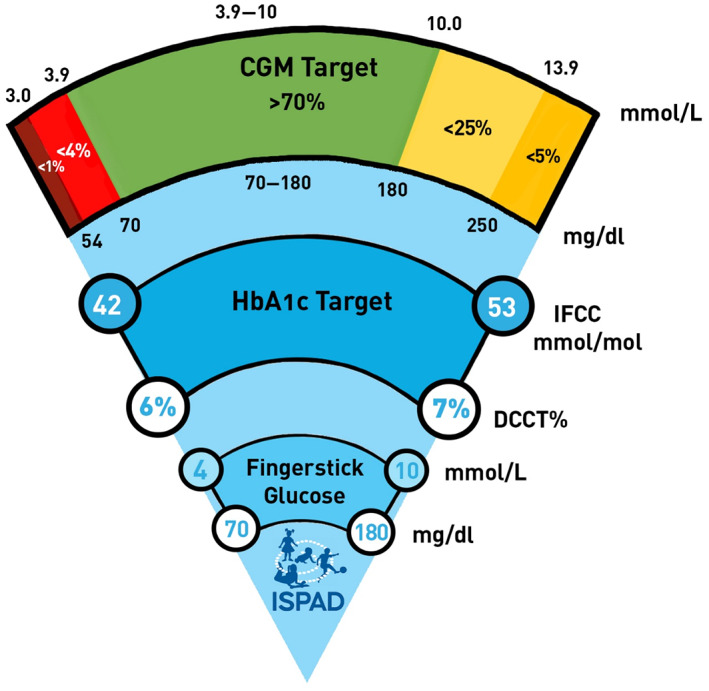
Glycemic targets are dependent on the measures available; finger stick capillary glucose (SMBG) levels, HbA1c, and CGM values. The term “finger stick” glucose is used instead of SMBG in the figure, which is designed to be easily interpreted by people with diabetes. The different modes of measuring glycemia are closely related, but are not equivalent, and the image is intended as an educational aid. SMBG targets align with the CGM optimal range; however, fasting SMBG levels are recommended to fall between 4 and 8 mmol/L (70–144 mg/dl)
The target of <53 mmol/mol (<7.0%) is chosen with the aim of avoiding long‐term microvascular and macrovascular complications. The curvilinear relationship of HbA1c and the development of microvascular and macrovascular complications indicates that HbA1c values that approach 42 mmol/mol (6%) may continue to yield risk reduction, but that the relative gains are less as compared with reducing HbA1c levels to the upper limit of the target (53 mmol/mol [7%]). 20 , 21 An HbA1c target of <48 mmol/mol mmol/mol (6.5%) is recommended during the remission phase or early stage 3 diabetes “honeymoon” and when using contemporary treatment such as continuous glucose monitoring, or automated insulin delivery in combination with a highly skilled specialized workforce adept at diabetes education. This would apply to most young people with diabetes who are not living in a limited resource setting. Other groups recommend this lower HbA1c target of 48 mmol/mol (6.5%) to the diabetes population that they represent (e.g., the 2020 NICE guidelines available at [www.nice.org.uk/guidance/NG18], and Sweden), however, these reflect health care settings where access to the aforementioned technology and workforce are available for the majority of people with diabetes. ISPAD has retained an HbA1c target range between 6% and <7% largely because it represents many populations of people with diabetes around the world who do not have this equity in access.
4.1.2. Laboratory and practical considerations
Glycated hemoglobin (HbA1c), continues to have a central role in setting glycemic targets, by virtue of several factors; (i) Definitive evidence of the association between HbA1c and the development of diabetes complications, 1 , 20 (ii) a standardized reference method and procedure set by the IFCC and endorsed by all the major stakeholders, 22 (iii) availability of point‐of‐care measurement in clinic and in outreach or remote settings, and (iv) barriers to universal access to CGM (and associated glycemic metrics). Every young person with diabetes should have a minimum of four HbA1c measurements per year (at ~3‐month intervals). It is recommended that centers regularly audit HbA1c levels, benchmark their data against consensus statements and, if possible, contribute their data to registries and quality improvement initiatives.
The maximum lifespan of erythrocytes is ~100–120 days with an average age at any given time ranging from 40 to 60 days. 23 , 24 HbA1c reflects average blood glucose concentration in the preceding 8–12 weeks. 25 More recent plasma glucose concentrations contribute proportionately more to the HbA1c concentration—estimated to be 50% contribution from the previous 30 days, with 40% and 10% contributions from the previous 31–90 days and 91–120 days, respectively. 26
4.1.3. Limitations of HbA1c
Clinical states associated with altered rates of hemoglobin turnover or erythrocyte survival will affect HbA1c measurements and therefore clinical utility (Table 1). As HbA1c directly reflects average glucose levels, highly variable glucose levels with fluctuating hypo‐ and hyperglycemia can result in the same HbA1c measurement as an individual with stable glucose levels. This is important as glycemic variability predicts severe hypoglycemia, and there is a growing body of evidence that glycemic variability is an independent risk factor for short ‐ and long‐term complications. 27 , 28 Arguably, CGM, by virtue of providing metrics for both average glucose, glucose out of target range and glycemic variability, as well as having a very high correlation with HbA1c, provides a better reflection of overall glycemia. CGM offers an alternative proxy for HbA1c [the Glucose Management Index (GMI)], 29 however, there is some discordance between GMI and laboratory HbA1c, and hence, the term “estimated” HbA1c should be avoided. 30 Evidence supports the association of diabetes complications and CGM derived measures, particularly time in range. 31 However as widespread CGM uptake has been a more recent phenomenon, it will take time for large registry data to definitively connect CGM metrics with the development of micro‐ and macrovascular complications. However, where CGM data are not available, evaluation of fructosamine and/or 1,5‐anhydroglucitol (1,5‐AG) may be the only alternative when HbA1c is not truly reflective of glycemia (Table 1).
TABLE 1.
Clinical states affecting erythrocyte turnover and their effects on HbA1c
| Increased erythrocyte turnover resulting in lower HbA1c | Reduced erythrocyte turnover resulting in higher HbA1c |
|---|---|
|
Recovery from iron, vitamin B12, and folate deficiency Pregnancy: second trimester Chronic kidney disease: Erythropoietin treatment and dialysis Acute blood loss Hemolysis (e.g., Sickle cell trait/disease, thalassemia, G6PD) Cystic fibrosis Chemotherapy |
Iron, vitamin B12, and folate deficiency Pregnancy: third trimester Chronic kidney disease: uremia |
Fructosamine is the generic term for plasma protein ketoamines or 1‐amino‐1‐deoxy‐D‐fructose, 32 and more specifically is the measurement of the total stable irreversible serum glycated proteins at any given time. The half‐life of serum proteins is significantly shorter than that of erythrocytes, and the degree of glycation is therefore more reflective of shorter‐term alterations in plasma glucose concentrations that is estimated to be 2–3 weeks, which is consistent with the half‐life of albumin (20 days) which comprises 80% of total serum proteins. 33 , 34 1,5‐AG has been proposed in the assessment of glycemic variability. 35 Low 1,5‐AG values are indicative of both high circulating plasma glucose concentration, as well as fluctuations in plasma glucose concentrations (hyperglycemic excursions). 1,5‐AG concentration reflects plasma glucose concentrations over the preceding 2–14 days.
4.2. Continuous glucose monitoring
4.2.1. CGM targets
ISPAD endorses previously published standards for time spent in each glycemic band 36 (Figure 1). These are time spent:
>70% between 3.9 and 10 mmol/L (70–180 mg/dl),
<4% <3.9 mmol/L (70 mg/dl),
<1% <3.0 mmol/L (54 mg/dl),
<25% >10 mmol/L (180 mg/dl),
<5% >13.9 mmol/L (250 mg/dl)
Glycemic variability (%CV) target ≤36%
Average sensor glucose by virtue of a strong correlation between mean sensor glucose and HbA1c, and association with the risk of microvascular complications, 37 and measures of glycemic variability (as a predictor of hypoglycemia), are included. These metrics are all reported as part of standardized CGM reports, termed the ambulatory glucose profile (AGP). When available, CGM targets should be used in conjunction with HbA1c targets (Figure 1). On rare occasions, as discussed above, less stringent time in range goals may be applied where efforts to reach the target may be detrimental to overall wellbeing.
4.2.2. Practical considerations for continuous glucose monitoring
The evidence and best practice for the use of CGM in improving glycemia and psychosocial burden is reviewed in ISPAD 2022 Consensus Guidelines Chapter 16 Diabetes Technologies: Glucose monitoring. This includes appropriate expectation setting and education. Early adoption of CGM from diagnosis is associated with long‐term benefits to HbA1c. 38 , 39 Unfortunately, access to CGM is not universal and can depend on geographic location, local health care funding policy, and socioeconomic status (including insurance). Further, there is racial‐ethnic and insurance‐mediated bias in recommending CGM by health care providers. 40
Skin irritation is a significant negative aspect of CGM, 41 and is the commonest reason for discontinuation. 42 Various strategies have been developed to address this issue 43 and are discussed further in ISPAD 2022 Consensus Guidelines Chapter 16 Diabetes Technologies: Glucose monitoring and Chapter 19 Other complications and associated conditions in children and adolescents with type 1 diabetes. Alarm fatigue can also contribute to CGM discontinuation, and as such, a person centered approach should be used when introducing CGM alarms. 44
CGM accuracy is an important consideration, especially in the hypoglycemic range. According to the consensus statement, the maximal allowable time spent <3.9 mmol/L (70 mg /dl) is 4%, however people without diabetes may spend 3.2% of their time in this zone, but rarely <3.0 mmol/L (54 mg/dl), depending on the accuracy of the sensor used. 45 , 46 Therefore, reducing time spent in the very low <3.0 mmol/L (54 mg/dl) is most important. Fortunately, each subsequent generation of CGM has improved accuracy to the point that several CGM and intermittently scanned CGM (isCGM) systems are approved to be used non‐adjunctively. Confirmation of hypoglycemia using a SMBG is recommended. SMBG confirmation should also occur when there is a discrepancy between symptoms of hyperglycemia or hypoglycemia and an apparently normal sensor glucose value.
4.3. Capillary glucose measurements (SMBG)
4.3.1. SMBG targets
SMBG targets should be 4–10 mmol (70–180 mg/dl). SMBG levels should be targeted to correspond to an HbA1c <53 mmol/mol (7%). This aligns with the CGM time in range target of >70% between 3.9 and 10 mmol (70–180 mg/dl), and the strong correlation of CGM metrics with HbA1c reviewed earlier. Tighter fasting target range of 4–8 mmol/L (70–144 mg/dl) are recommended in order to achieve the above stated HbA1c target. Previous ISPAD guidelines 16 and current ADA and NICE guidelines have recommended a variety of glucose value ranges depending on time of day and relationship to meals. 16 Without empiric evidence that such specific targeting reduces hyperglycemia or hypoglycemia, combined with the potential for healthcare professionals to send mixed messages and overly detailed education causing confusion, the newly defined SMBG targets offer a pragmatic solution. SMBG glucose level target prior to bed above 3.9 mmol/L (70 mg/dl) are appropriate, however caregivers may have more confidence with higher levels within the 4–10 mmol/L (70–180 mg/dl) range in certain scenarios; for example, if there has been preceding hypoglycemia, peri‐exercise, hypoglycemia unawareness, or no access to CGM with hypoglycemia threshold alarms. Ideal glucose levels prior to and during exercise are dependent on many factors including type and duration of exercise, insulin regimen, and CGM use, and are detailed in ISPAD 2022 Consensus Guidelines Chapter 14 for Exercise in Children and Adolescents with Diabetes. The evidence that SMBG has a healthy impact on glycemia in young people with T2D is limited. The potential benefit vs. cost of CGM in this population also remains unclear.
5. A DEVELOPMENTAL PERSPECTIVE FOR GLYCEMIC TARGET SETTING
While glucose targets outlined above can be applied to all young people with diabetes, a challenging time for the individual and their caregivers can occur as the honeymoon period wanes due to diminishing residual endogenous insulin secretion. Beyond the honeymoon, there may be a requirement for more intensive management and associated burden to maintain glycemic targets. The long‐term HbA1c trajectory is strongly predicted early after diagnosis, which highlights the importance of attaining target glucose levels early in the life course. 47 , 48 , 49 As outlined in ISPAD 2022 Clinical Practice Guidelines Chapter 6 on Diabetes Education in Children and Adolescents, it is important that glucose targets be addressed and reinforced during the post honeymoon phase when HbA1c increases and TIR decreases.
The developmental age of the person with diabetes is associated with unique challenges to achieving the aforementioned glucose targets. For example, management in pre‐school children can be particularly difficult due to unpredictable eating and activity levels and associated higher glycemic variability. 50 See ISPAD 2022 Consensus Guidelines Chapter 23 on Managing Diabetes In Preschoolers. At school age, young people are beginning independent care. There is some evidence that focused age‐appropriate educational interventions are effective in children and families (See ISPAD 2022 Clinical Practice Guidelines Chapter 6 on Diabetes Education in Children and Adolescents). Further, adolescence is a critical period of independence and physiologic changes associated with increasing insulin resistance, with an increase in HbA1c seen in multiple international registries. 51 Adolescent and culturally appropriate education tools are needed to reinforce individualized care plans that aim to meet glycemic targets while balancing lifestyle and psychological factors (See ISPAD 2022 Consensus Guidelines Chapter 21 Diabetes in Adolescence).
6. HEALTH CARE PRIORITIES AND FUTURE DIRECTIONS
The social determinants of health, encompassing “the conditions in which people are born, grow, work, live, and age, and the wider set of forces and systems shaping the conditions of daily life (WHO),” strongly predict the likelihood of an individual achieving recommended or optimal glycemic targets. 9 , 52 ISPAD recognizes that these disparities represent significant barriers to optimal care, and collective efforts are needed to understand and address systemic inequities including medical racism and societal policies that entrench generational poverty. As such, there is a responsibility for health care professionals to advocate on behalf of young people with diabetes who have limited access to healthcare, including technology. Indeed, health providers are known to have implicit bias with respect to offering diabetes technology, which drives inequity. 40 , 53 Specifically, healthcare reimbursement policies and wider government policy that drives socioeconomic disparities are essential to improve health equity. For the person with diabetes, this should translate to equity in accessing an appropriately resourced multi‐disciplinary care team (including dietetic, nursing, psychology, social work, and medical expertise), access to technologies such as CGM and automated insulin delivery, and modern insulin analogs.
AUTHOR CONTRIBUTIONS
All authors reveiwed and summarized literature on glycemic targets. All authors reviewed and edited manuscript drafts. MDB coordinated revisions of the manuscript based on input from ISPAD membership, the co‐authors, and ISPAD leadership.
CONFLICT OF INTEREST
None of the authors has any conflicts of interest relevant to the subject matter of the article.
ACKNOWLEDGEMENT
Open access publishing facilitated by University of Otago, as part of the Wiley ‐ University of Otago agreement via the Council of Australian University Librarians.
de Bock M, Codner E, Craig ME, et al. ISPAD Clinical Practice Consensus Guidelines 2022: Glycemic targets and glucose monitoring for children, adolescents, and young people with diabetes. Pediatr Diabetes. 2022;23(8):1270‐1276. doi: 10.1111/pedi.13455
DATA AVAILABILITY STATEMENT
This article is an invited review/consensus statement. Data sharing is not applicable.
REFERENCES
- 1. Diabetes Control Complications Trial/Epidemiology of Diabetes Interventions Complications Study Research Group . Intensive diabetes treatment and cardiovascular disease in patients with type 1 diabetes. N Engl J Med. 2005;353(25):2643‐2653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Nathan DM, Genuth S, Lachin J, et al. The effect of intensive treatment of diabetes on the development and progression of long‐term complications in insulin‐dependent diabetes mellitus. N Engl J Med. 1993;329(14):977‐986. doi: 10.1056/nejm199309303291401 [DOI] [PubMed] [Google Scholar]
- 3. Nevo‐Shenker M, Shalitin S. The impact of hypo‐and hyperglycemia on cognition and brain development in young children with type 1 diabetes. Horm Res Paediatr. 2021;94(3–4):115‐123. [DOI] [PubMed] [Google Scholar]
- 4. Ferguson SC, Blane A, Wardlaw J, et al. Influence of an early‐onset age of type 1 diabetes on cerebral structure and cognitive function. Diabetes Care. 2005;28(6):1431‐1437. [DOI] [PubMed] [Google Scholar]
- 5. Sørensen J, Ploug UJ. The cost of diabetes‐related complications: registry‐based analysis of days absent from work. Econ Res Int. 2013;2013:1‐8. [Google Scholar]
- 6. Tao B, Pietropaolo M, Atkinson M, Schatz D, Taylor D. Estimating the cost of type 1 diabetes in the US: a propensity score matching method. PLoS One. 2010;5(7):e11501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Bak JC, Serné EH, Kramer MH, Nieuwdorp M, Verheugt CL. National diabetes registries: do they make a difference? Acta Diabetol. 2021;58(3):267‐278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Agarwal S, Schechter C, Gonzalez J, Long JA. Racial–ethnic disparities in diabetes technology use among young adults with type 1 diabetes. Diabetes Technol Ther. 2021;23(4):306‐313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Lipman TH, Hawkes CP. Racial and socioeconomic disparities in pediatric type 1 diabetes: time for a paradigm shift in approach. Diabetes Care. 2021;44(1):14‐16. [DOI] [PubMed] [Google Scholar]
- 10. Redondo MJ, Libman I, Maahs DM, et al. The evolution of hemoglobin A1c targets for youth with type 1 diabetes: rationale and supporting evidence. Diabetes Care. 2021;44(2):301‐312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Maahs DM, Hermann JM, DuBose SN, et al. Contrasting the clinical care and outcomes of 2,622 children with type 1 diabetes less than 6 years of age in the United States T1D exchange and German/Austrian DPV registries. Diabetologia. 2014;57(8):1578‐1585. [DOI] [PubMed] [Google Scholar]
- 12. Swift PG, Skinner K, De Beaufort C, et al. Target setting in intensive insulin management is associated with metabolic control: the Hvidoere childhood diabetes study group Centre differences study 2005. Pediatr Diabetes. 2010;11(4):271‐278. [DOI] [PubMed] [Google Scholar]
- 13. Samuelsson U, Åkesson K, Peterson A, Hanas R, Hanberger L. Continued improvement of metabolic control in Swedish pediatric diabetes care. Pediatr Diabetes. 2018;19(1):150‐157. [DOI] [PubMed] [Google Scholar]
- 14. Alonso GT, Corathers S, Shah A, et al. Establishment of the T1D exchange quality improvement collaborative (T1DX‐QI). Clin Diabetes. 2020;38(2):141‐151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Prahalad P, Leverenz B, Freeman A, et al. Closing disparities in pediatric diabetes telehealth care: lessons from telehealth necessity during the COVID‐19 pandemic. Clin Diabetes. 2022;40(2):153‐157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. DiMeglio LA, Acerini CL, Codner E, et al. ISPAD clinical practice consensus guidelines 2018: glycemic control targets and glucose monitoring for children, adolescents, and young adults with diabetes. Pediatr Diabetes. 2018;19(Suppl 27):105‐114. doi: 10.1111/pedi.12737 [DOI] [PubMed] [Google Scholar]
- 17. Lee S, Ooi L, Lai Y. Children and adolescents: standards of medical care in diabetes—2021. Diabetes Care. 2021;44(S1):180‐199. [Google Scholar]
- 18. Johnson SR, Holmes‐Walker DJ, Chee M, Earnest A, Jones TW, Group: AS . Universal subsidized continuous glucose monitoring funding for young people with type 1 diabetes: uptake and outcomes over 2 years, a population‐based study. Diabetes Care. 2022;45(2):391‐397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Karges B, Rosenbauer J, Kapellen T, et al. Hemoglobin A1c levels and risk of severe hypoglycemia in children and young adults with type 1 diabetes from Germany and Austria: a trend analysis in a cohort of 37,539 patients between 1995 and 2012. PLoS Med. 2014;11(10):e1001742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Reichard P, Nilsson B‐Y, Rosenqvist U. The effect of long‐term intensified insulin treatment on the development of microvascular complications of diabetes mellitus. N Engl J Med. 1993;329(5):304‐309. [DOI] [PubMed] [Google Scholar]
- 21. The absence of a glycemic threshold for the development of long‐term complications: the perspective of the diabetes control and complications trial. Diabetes. 1996;45(10):1289‐1298. [PubMed] [Google Scholar]
- 22. Hanas R, John G. Committee IHcC. 2010 consensus statement on the worldwide standardization of the hemoglobin A1C measurement. Diabetes Care. 2010;33(8):1903‐1904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Cohen RM, Franco RS, Khera PK, et al. Red cell life span heterogeneity in hematologically normal people is sufficient to alter HbA1c. Blood. 2008;112(10):4284‐4291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Fitzgibbons JF, Koler RD, Jones RT. Red cell age‐related changes of hemoglobins AIa+ b and AIc in normal and diabetic subjects. J Clin Invest. 1976;58(4):820‐824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Nathan DM, Singer DE, Hurxthal K, Goodson JD. The clinical information value of the glycosylated hemoglobin assay. N Engl J Med. 1984;310(6):341‐346. [DOI] [PubMed] [Google Scholar]
- 26. Tahara Y, Shima K. Kinetics of HbA1c, glycated albumin, and fructosamine and analysis of their weight functions against preceding plasma glucose level. Diabetes Care. 1995;18(4):440‐447. [DOI] [PubMed] [Google Scholar]
- 27. Rama Chandran S, Tay WL, Lye WK, et al. Beyond HbA1c: comparing glycemic variability and glycemic indices in predicting hypoglycemia in type 1 and type 2 diabetes. Diabetes Technol Ther. 2018;20(5):353‐362. [DOI] [PubMed] [Google Scholar]
- 28. Ceriello A, Monnier L, Owens D. Glycaemic variability in diabetes: clinical and therapeutic implications. Lancet Diabetes Endocrinol. 2019;7(3):221‐230. [DOI] [PubMed] [Google Scholar]
- 29. Riddlesworth TD, Beck RW, Gal RL, et al. Optimal sampling duration for continuous glucose monitoring to determine long‐term glycemic control. Diabetes Technol Ther. 2018;20(4):314‐316. [DOI] [PubMed] [Google Scholar]
- 30. Perlman JE, Gooley TA, McNulty B, Meyers J, Hirsch IB. HbA1c and glucose management indicator discordance: a real‐world analysis. Diabetes Technol Ther. 2021;23(4):253‐258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Yapanis M, James S, Craig ME, O'Neal D, Ekinci EI. Complications of diabetes and metrics of glycemic management derived from continuous glucose monitoring. J Clin Endocrinol Metab. 2022;107(6):e2221‐e2236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Armbruster DA. Fructosamine: structure, analysis, and clinical usefulness. Clin Chem. 1987;33(12):2153‐2163. [PubMed] [Google Scholar]
- 33. Anguizola J, Matsuda R, Barnaby OS, et al. Review: glycation of human serum albumin. Clin Chim Acta. 2013;425:64‐76. doi: 10.1016/j.cca.2013.07.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Anguizola J, Matsuda R, Barnaby OS, et al. Glycation of human serum albumin. Clin Chim Acta. 2013;425:64‐76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Dungan KM. 1, 5‐anhydroglucitol (GlycoMark™) as a marker of short‐term glycemic control and glycemic excursions. Expert Rev Mol Diagn. 2008;8(1):9‐19. [DOI] [PubMed] [Google Scholar]
- 36. Battelino T, Danne T, Bergenstal RM, et al. Clinical targets for continuous glucose monitoring data interpretation: recommendations from the international consensus on time in range. Diabetes Care. 2019;42(8):1593‐1603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Beck RW, Bergenstal RM, Riddlesworth TD, et al. Validation of time in range as an outcome measure for diabetes clinical trials. Diabetes Care. 2019;42(3):400‐405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Mulinacci G, Alonso GT, Snell‐Bergeon JK, Shah VN. Glycemic outcomes with early initiation of continuous glucose monitoring system in recently diagnosed patients with type 1 diabetes. Diabetes Technol Ther. 2019;21(1):6‐10. [DOI] [PubMed] [Google Scholar]
- 39. Prahalad P, Ding VY, Zaharieva DP, et al. Teamwork, targets, technology, and tight control in newly diagnosed type 1 diabetes: the pilot 4T study. J Clin Endocrinol Metab. 2022;107(4):998‐1008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Odugbesan O, Addala A, Nelson G, et al. Implicit racial‐ethnic and insurance mediated bias to recommending diabetes technology: insights from T1D exchange multi‐center pediatric and adult diabetes provider cohort. Diabetes Technol Ther. 2022;24(9):619‐627. doi: 10.1089/dia.2022.0042 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Pleus S, Ulbrich S, Zschornack E, Kamann S, Haug C, Freckmann G. Documentation of skin‐related issues associated with continuous glucose monitoring use in the scientific literature. Diabetes Technol Ther. 2019;21(10):538‐545. [DOI] [PubMed] [Google Scholar]
- 42. Asarani NAM, Reynolds AN, Boucher SE, de Bock M, Wheeler BJ. Cutaneous complications with continuous or flash glucose monitoring use: systematic review of trials and observational studies. J Diabetes Sci Technol. 2020;14(2):328‐337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Messer LH, Berget C, Beatson C, Polsky S, Forlenza GP. Preserving skin integrity with chronic device use in diabetes. Diabetes Technol Ther. 2018;20:S2‐54‐S2‐64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Miller E, Midyett LK. Just because you can, doesn't mean you should… now. A practical approach to counseling persons with diabetes on use of optional CGM alarms. Diabetes Technol Ther. 2021;23(S3):S‐66‐S‐71. [DOI] [PubMed] [Google Scholar]
- 45. Sofizadeh S, Pehrsson A, Ólafsdóttir AF, Lind M. Evaluation of reference metrics for continuous glucose monitoring in persons without diabetes and prediabetes. J Diabetes Sci Technol. 2020;16(2):373‐382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Shah VN, DuBose SN, Li Z, et al. Continuous glucose monitoring profiles in healthy nondiabetic participants: a multicenter prospective study. J Clin Endocrinol Metab. 2019;104(10):4356‐4364. doi: 10.1210/jc.2018-02763 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Nirantharakumar K, Mohammed N, Toulis KA, Thomas GN, Narendran P. Clinically meaningful and lasting HbA1c improvement rarely occurs after 5 years of type 1 diabetes: an argument for early, targeted and aggressive intervention following diagnosis. Diabetologia. 2018;61(5):1064‐1070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Lachin JM, Bebu I, Nathan DM. The beneficial effects of earlier versus later implementation of intensive therapy in type 1 diabetes. Diabetes Care. 2021;44(10):2225‐2230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Lachin JM, Nathan DM. Understanding metabolic memory: the prolonged influence of glycemia during the diabetes control and complications trial (DCCT) on future risks of complications during the study of the epidemiology of diabetes interventions and complications (EDIC). Diabetes Care. 2021;44(10):2216‐2224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Deeb A. Challenges of diabetes management in toddlers. Diabetes Technol Ther. 2017;19(7):383‐390. [DOI] [PubMed] [Google Scholar]
- 51. Anderzén J, Hermann JM, Samuelsson U, et al. International benchmarking in type 1 diabetes: large difference in childhood HbA1c between eight high‐income countries but similar rise during adolescence—a quality registry study. Pediatr Diabetes. 2020;21(4):621‐627. [DOI] [PubMed] [Google Scholar]
- 52. Hill‐Briggs F, Adler NE, Berkowitz SA, et al. Social determinants of health and diabetes: a scientific review. Diabetes Care. 2020;44:258‐279. doi: 10.2337/dci20-0053 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Addala A, Hanes S, Naranjo D, Maahs DM, Hood KK. Provider implicit bias impacts pediatric type 1 diabetes technology recommendations in the United States: findings from the gatekeeper study. J Diabetes Sci Technol. 2021;15(5):1027‐1033. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
This article is an invited review/consensus statement. Data sharing is not applicable.


