Abstract
Background
KRAS mutations occur frequently in advanced non-small cell lung cancer (aNSCLC); the G12C mutation is the most prevalent. Alterations in STK11 or KEAP1 commonly co-occur with KRAS mutations in aNSCLC. Using real-world data, we assessed the effect of KRAS G12C mutation with or without STK11 and/or KEAP1 mutations on overall survival (OS) in patients with aNSCLC receiving cancer immunotherapy (CIT), chemotherapy, or both in first line (1L) and second line (2L).
Methods
Patients diagnosed with aNSCLC between January 2011 and March 2020 in a clinico-genomic database were included. Cox proportional hazards models adjusted for left truncation, baseline demographics and clinical characteristics were used to analyze the effect of STK11 and/or KEAP1 co-mutational status on OS in patients with KRAS wild-type (WT) or G12C mutation.
Results
Of 2715 patients with aNSCLC without other actionable driver mutations, 1344 (49.5%) had KRAS WT cancer, and 454 (16.7%) had KRAS G12C–positive cancer. At 1L treatment start, significantly more patients with KRAS G12C–positive cancer were female, smokers, and had non-squamous histology, a higher prevalence of metastasis and programmed death-ligand 1 positivity than those with KRAS WT cancer. Median OS was comparable between patients with KRAS G12C–positive and KRAS WT cancer when receiving chemotherapy or combination CIT and chemotherapy in the 1L or 2L. Median OS was numerically longer in patients with KRAS G12C vs KRAS WT cancer treated with 1L CIT (30.2 vs 10.6 months, respectively) or 2L CIT (11.3 vs 7.6 months, respectively). Co-mutation of STK11 and KEAP1 was associated with significantly shorter OS in patients receiving any type of 1L therapy, regardless of KRAS G12C mutational status.
Conclusions
This real-world study showed that patients with KRAS G12C–positive or KRAS WT cancer have similar OS in the 1L or 2L when treated with chemotherapy or combination CIT and chemotherapy. In contrast to aNSCLC patients with EGFR or ALK driver mutations, patients with KRAS G12C–positive cancer may benefit from CIT monotherapy. Co-mutation of STK11 and KEAP1 was associated with significantly shorter survival, independent of KRAS G12C mutational status, reflecting the poor prognosis and high unmet need in this patient population.
Keywords: Non-small cell lung cancer, KRAS G12C, STK11, KEAP1, Metastasis, Immunotherapy, Chemotherapy
Background
Non-small cell lung cancer (NSCLC) accounts for approximately 85% of all lung cancers [1]. NSCLC is a complex disease consisting of numerous molecular subtypes [2]. Biomarker testing of NSCLC has a direct impact on treatment decisions, and screening tumors for actionable driver mutations is essential before initiating treatment [3]. In general, recommended treatment for patients include targeted therapies for patients with advanced NSCLC (aNSCLC) bearing alterations in driver oncogenes (EGFR, ALK, ROS1, BRAF, NTRK, RET, or MET), cancer immunotherapy (CIT) with immune checkpoint inhibitors alone or in combination with chemotherapy [3].
Mutations in Kirsten Rat Sarcoma Viral Oncogene Homolog (KRAS) are prevalent in NSCLC, occurring in approximately 25 to 40% of patients (≈5–10% in Asian patients) [4–7]. In particular, the KRAS glycine 12 to cysteine (G12C) activating mutation has the highest prevalence (≈40% of all KRAS mutations in NSCLC) [8, 9]. Tumors that harbor KRAS mutations are usually among the most aggressive and refractory to treatment [8]. Recently, sotorasib, a mutation-specific inhibitor of KRAS G12C has received marketing authorization in KRAS G12C-mutated aNSCLC patients who have received at least one prior systemic therapy [10, 11]. While KRAS G12C inhibitors provide an exciting new second-line or later (2L+) treatment option for patients with KRAS G12C–positive tumors, the standard-of-care therapies for first-line (1L) KRAS G12C-mutated aNSCLC patients currently remain CIT targeting programmed cell death protein 1 (PD-1) or programmed cell death 1 ligand 1 (PD-L1) alone or combined with chemotherapy.
Patients with tumors that harbor KRAS mutations have shown a longer median overall survival (OS) when treated with CIT alone vs chemotherapy alone (28 months (95% CI: 23-NR) vs 11 months (95% CI: 7–25)) [10] or when treated with combination therapy (CIT and chemotherapy) vs chemotherapy alone (21 months (95% CI: 16-NR) vs 14 months (95% CI: 8-NR)) [10]. While patients with KRAS G12C–positive cancer may benefit from treatment with CIT alone or in combination with chemotherapy [12, 13], numbers of patients with KRAS G12C-positive cancer were low in these previous studies, and it remains unclear how the benefit compares with that seen in patients with KRAS wild-type (WT) cancer. Furthermore, somatic genomic alterations in serine/threonine kinase 11 (STK11) or kelch like ECH associated protein 1 (KEAP1) commonly co-occur with KRAS mutations in NSCLC (25–30%) [14], and co-mutation of KRAS with STK11 or KEAP1 is associated with significantly worse survival [14–16]. It is unknown whether co-occurring mutations affect prognosis and whether differential responses to treatment and consequent effects on survival outcomes exist in these patient populations. This study used real-world data to assess the effect of KRAS G12C mutational status on OS in patients with aNSCLC with or without STK11 and/or KEAP1 co-mutations who received CIT, chemotherapy, or both in the 1L and 2L using real-world data.
Methods
Data source
This study used the nationwide (US-based) deidentified Flatiron Health-Foundation Medicine NSCLC clinico-genomic database (FH-FMI CGDB). Retrospective longitudinal clinical data were derived from electronic health record (EHR) data, comprising patient-level structured and unstructured data, curated via technology-enabled abstraction, and were linked to genomic data derived from FMI comprehensive genomic profiling (CGP) tests in the CGDB by de-identified, deterministic matching [17]. Genomic alterations were identified via CGP of > 300 cancer-related genes on FMI’s next-generation sequencing (NGS) test (FMI sequencing platform[s]: FoundationOne®CDx, FoundationOne®, FoundationOne®Liquid, or FoundationOne®Liquid CDx) [18–20]. Both liquid and solid assays were used in this study. As liquid assays may not detect alterations if shedding of circulating DNA is low, only solid assays were used to define WT KRAS, KEAP1, and STK11.
Patient population
Eligibility criteria included: (1) aged ≥18 years with aNSCLC newly diagnosed between January 1, 2011, and March 31, 2020; (2) had structured activity within 90 days after aNSCLC diagnosis and had 6 months of follow-up after treatment initiation; (3) did not have functional or likely functional driver alterations (short variants, copy number alterations, or fusions) in EGFR, ALK, ROS1, BRAF, ERBB2, MET, or RET; (4) received treatment with CIT (eg, immune checkpoint inhibitors) alone, combination CIT and chemotherapy, or chemotherapy alone and did not receive targeted therapies for driver mutations or nonapproved monotherapy CIT for aNSCLC; (5) displayed no evidence of being diagnosed with other cancers in the database; and (6) had ≥1 definitive (ie, positive or negative) molecular test, including a test for the KRAS gene, with results before or after 1L treatment initiation. If a patient had several specimen collections, the closest to initiation of 1L treatment was used. Patients were categorized by KRAS mutational status (G12C vs WT). The KRAS WT group excluded patients with any KRAS alteration.
Outcomes and analysis
The primary outcome was OS in patients with KRAS WT or KRAS G12C–positive aNSCLC, sorted by the following factors: (1) treatment line (1L or 2L); (2) treatment ([a] CIT alone or in combination with other CIT or with nonchemotherapy; [b] combination CIT and chemotherapy; or [c] chemotherapy alone); 3) presence of mutated STK11 (mSTK11) and/or mutated KEAP1 (mKEAP1) vs STK11 WT and KEAP1 WT, including any functional status (unknown, likely and known). OS is defined as time from an index date to the date of death for individual patients who have died. Patients without a death date are censored at the last evidence of them being alive, e.g., structured activity in the database.
All statistical analyses were performed with R. Kaplan-Meier (KM) curves, associated medians, and 95% confidence intervals were estimated for survival outcomes. Cox proportional hazards models adjusted for baseline demographics and clinical characteristics (age, sex, race, cancer type [de novo or recurrent], PD-L1 status, any metastasis, tumor mutational burden, histology, and 1L treatment [for 2L analysis only]) were used to analyze the effect of mutational status on OS in patients receiving CIT alone, combination CIT and chemotherapy, or chemotherapy alone. A separate category within a variable was created for the missing values. Adjustments to account for left truncation and immortal bias were applied to the KM analysis and the Cox regression model.
Results
Baseline demographics and clinical characteristics
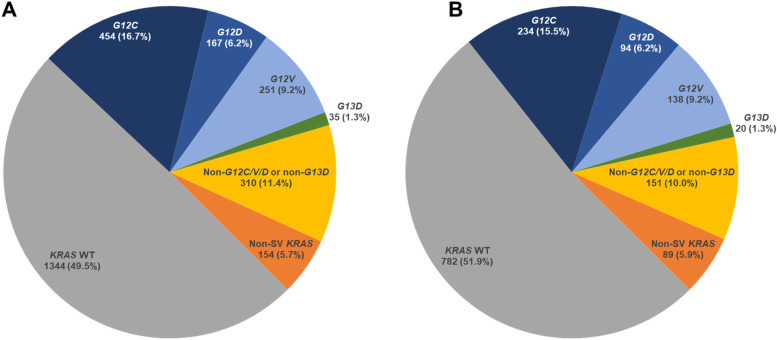
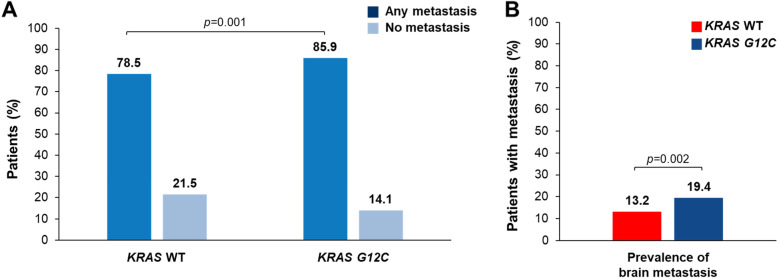
Of the 2715 aNSCLC patients without actionable driver mutations in the 1L setting, 1344 (49.5%) were KRAS WT, 454 (16.7%) had tumors with the KRAS G12C mutation, 251 (9.2%) had tumors with the KRAS G12V mutation, 167 (6.2%) had tumors with the KRAS G12D mutation, 35 (1.3%) had tumors with the KRAS G13D mutation, 310 (11.4%) had a non-G12C/D/V or non-G13D KRAS mutation, and 154 (5.7%) had alterations other than short variants such as copy number variations or rearrangements (Fig. 1A). mSTK11-mKEAP1 was more prevalent in patients with KRAS G12C–positive cancer vs KRAS WT cancer (14.3% vs 9.9%; p = 0.012) (Table 1). Interestingly, while mSTK11-KEAP1 WT was also more prevalent in patients with KRAS G12C-positive cancer vs KRAS WT cancer (13.0% vs 9.0%; p = 0.05), STK11 WT-mKEAP1 was more prevalent in patients with KRAS WT cancer vs KRAS G12C-positive cancer (15.0% vs 7.0%; p < 0.001) (Table 1). For 1L treatment, patients with KRAS WT cancer received, in order of frequency, chemotherapy alone (69.7%), combination CIT and chemotherapy (19.5%), or CIT alone (10.8%); similarly, the majority of patients with KRAS G12C–positive cancer received chemotherapy alone (61.9%), combination CIT and chemotherapy (22.2%), and CIT alone (15.9%) (Table 1). At 1L initiation, patients with KRAS G12C–positive cancer had a significantly higher prevalence of metastasis than patients with KRAS WT cancer (85.9% vs 78.5%; p = 0.001; Fig. 2A). In patients with metastasis, those with KRAS G12C–positive cancer had a significantly higher prevalence of brain metastasis than patients with KRAS WT cancer (19.4% vs 13.2%; p = 0.002; Fig. 2B). In all patients at 1L initiation, 10.4% of patients with KRAS WT cancer and 16.7% of patients with KRAS G12C-positive cancer had brain metastasis.
Fig. 1.
KRAS Mutational Status in (A) the 1L and (B) the 2L Settings. SV, short variant; WT, wild type
Table 1.
Baseline Characteristics of Patients With aNSCLC by KRAS Mutational Status and Treatment Line
| 1L | 2L | |||||
|---|---|---|---|---|---|---|
| Characteristic, n (%) | KRAS WT n = 1344 | KRAS G12C n = 454 | p | KRAS WT n = 782 | KRAS G12C n = 234 | p |
| Sex | ||||||
| Female | 488 (36.3) | 281 (61.9) | < 0.001 | 296 (37.9) | 143 (61.1) | < 0.001 |
| Male | 856 (63.7) | 173 (38.1) | 486 (62.1) | 91 (38.9) | ||
| Age at line start | ||||||
| 18–64 years | 479 (35.6) | 169 (37.2) | 0.572 | 292 (37.3) | 92 (39.3) | 0.591 |
| ≥ 65 years | 865 (64.4) | 285 (62.8) | 490 (62.6) | 142 (60.7) | ||
| Race | ||||||
| White | 955 (71.1) | 327 (72.0) | 0.108 | 546 (69.8) | 167 (71.4) | 0.598 |
| Asian | 12 (0.9) | ≤5 (≤1.1) | 9 (1.2) | ≤5 (≤2.1) | ||
| Black or African American | 107 (8.0) | 25 (5.5) | 63 (8.1) | 17 (7.3) | ||
| Hispanic or Latino | ≤5 (≤1.1) | |||||
| Other | 167 (12.4) | 61 (13.4) | 113 (14.5) | 29 (12.4) | ||
| Missing | 103 (7.7) | 35 (7.7) | 51 (6.5) | ≥16 (≥6.8) | ||
| Smoking status | ||||||
| Previous or current | 1255 (93.4) | 445 (98.0) | < 0.001 | 727 (93.0) | 228 (97.4) | 0.018 |
| Stage at initial diagnosis before progressing to advanced disease | ||||||
| Stage I at initial diagnosis | 112 (8.3) | 52 (11.5) | 0.005 | 65 (8.3) | 34 (14.5) | 0.001 |
| Stage II at initial diagnosis | 80 (6.0) | ≥30 (≥6.6) | 46 (5.9) | ≥7 (≥2.9) | ||
| Stage III at initial diagnosis | 326 (24.3) | 74 (16.3) | 221 (28.3) | 41 (17.5) | ||
| Stage IV at initial diagnosis | 808 (60.1) | 293 (64.5) | 440 (56.3) | 147 (62.8) | ||
| Unknown stage at initial diagnosis | 18 (1.3) | ≤5 (≤1.1) | 10 (1.3) | ≤5 (≤2.1) | ||
| Histology | ||||||
| Non-squamous | 745 (55.4) | 415 (91.4) | < 0.001 | 411 (52.6) | 214 (91.5) | < 0.001 |
| Squamous | 532 (39.6) | 19 (4.2) | 337 (43.1) | 9 (3.8) | ||
| NOS | 67 (5.0) | 20 (4.4) | 34 (4.3) | 11 (4.7) | ||
| TMBa | ||||||
| High | 638 (47.5) | 174 (38.3) | < 0.001 | 171 (21.9) | 38 (16.2) | < 0.001 |
| Low | 689 (51.3) | 235 (51.8) | 599 (76.6) | 171 (73.1) | ||
| Missing or unknown | 17 (1.3) | 45 (9.9) | 12 (1.5) | 25 (10.7) | ||
| PD-L1b | ||||||
| High (≥50%) | 139 (10.3) | 83 (18.3) | < 0.001 | 73 (9.3) | 35 (15.0) | 0.003 |
| Low (1–49%) | 208 (15.5) | 69 (15.2) | 116 (14.8) | 40 (17.1) | ||
| Negative (< 1%) | 228 (17.0) | 46 (10.1) | 134 (17.1) | 21 (9.0) | ||
| Missing or unknown | 769 (57.2) | 256 (56.4) | 459 (58.7) | 138 (59.0) | ||
| Cancer type | ||||||
| De novo | 809 (60.2) | 294 (64.8) | 0.095 | 441 (56.4) | 147 (62.8) | 0.095 |
| Recurrent | 535 (39.8) | 160 (35.2) | 341 (43.6) | 87 (37.2) | ||
| ECOG PS (Eastern Cooperative Oncology Group performance status) | ||||||
| 0 | 326 (24.3) | 110 (24.2) | 0.231 | 149 (19.1) | 49 (20.9) | 0.560 |
| 1 | 544 (40.5) | 163 (35.9) | 371 (47.4) | 106 (45.3) | ||
| ≥ 2 | 193 (14.3) | 76 (16.8) | 143 (18.3) | 45 (19.2) | ||
| Missing or unknown | 361 (20.5) | 105 (23.1) | 119 (15.2) | 34 (14.5) | ||
| Drug category | ||||||
| Chemotherapy | 937 (69.7) | 281 (61.9) | 0.003 | 272 (34.8) | 83 (35.5) | 0.379 |
| CIT | 145 (10.8) | 72 (15.9) | 445 (56.9) | 138 (59.0) | ||
| Combination CIT and chemotherapy | 262 (19.5) | 101 (22.2) | 65 (8.3) | 13 (5.6) | ||
| STK11 and/or KEAP1 mutational status | ||||||
| STK11 WT-KEAP1 WT | 881 (65.6) | 240 (52.9) | < 0.001 | 515 (65.9) | 125 (53.4) | 0.022 |
| mSTK11-mKEAP1 | 133 (9.9) | 65 (14.3) | 0.012 | 78 (10.0) | 31 (13.2) | 0.194 |
| mSTK11-KEAP1 WT | 129 (9.0) | 59 (13.0) | 0.05 | 72 (9.2) | 30 (12.8) | 0.136 |
| STK11 WT-mKEAP1 | 201 (15.0) | 32 (7.0) | < 0.001 | 117 (15.0) | 19 (8.1) | 0.010 |
| Treatment Start Year Group | ||||||
| 2009–2015 | 134 (10.0) | 54 (11.9) | 0.176 | 31 (4.0) | 15 (6.4) | 0.238 |
| 2016–2017 | 377 (28.1) | 137 (30.2) | 215 (27.5) | 73 (31.2) | ||
| 2018–2020 | 833 (62.0) | 263 (57.9) | 536 (68.5) | 146 (62.4) | ||
1L first line; 2L second line; aNSCLC advanced non-small cell lung cancer; CIT cancer immunotherapy; ECOG PS Eastern Cooperative Oncology Group performance status; mt mutation; NOS not otherwise specified; PD-L1 programmed death-ligand 1; TMB tumor mutational burden; WT wild type
aTMB was categorized as high or low using a different threshold (mutations [mut]/megabase[Mb]) for each treatment line; TMB high was defined as ≥10 mut/Mb in the 1L and ≥ 16 mut/Mb in the 2L.
bPD-L1high was defined as Tumor Proportion Score ≥ 50%, low 1–49% and negative < 1%
Fig. 2.
Prevalence of Metastasis in Patients With aNSCLC Treated in the 1L. 1L, first line; aNSCLC, advanced non-small cell lung cancer; WT, wild type
Of the 1745 patients in the 2L setting, 782 (51.9%) were KRAS WT, 234 (15.5%) had KRAS G12C–positive tumors, 138 (9.2%) had tumors with a KRAS G12V mutation, 94 (6.2%) had tumors with a KRAS G12D mutation, 20 (1.3%) had tumors with a KRAS G13D mutation, 151 (10.0%) had a non-G12C/D/V or non-G13D KRAS mutation, and 89 (5.9%) had alterations other than short variants (Fig. 1B). STK11 WT-mKEAP1 and STK11 WT-KEAP1 WT were more prevalent in patients with KRAS WT cancer, than those with KRAS G12C-positive cancer (15.0% vs 8.1%; p = 0.01 and 65.9% vs 53.4%, p = 0.02, respectively) (Table 1). Patients with KRAS WT cancer received for 2L treatment, in order of frequency, CIT alone (56.9%), chemotherapy alone (34.8%), and combination CIT and chemotherapy (8.3%); this pattern of results was similar in the 2L setting in patients with KRAS G12C–positive cancer (59.0, 35.5, and 5.6%, respectively; Table 1).
In both the 1L and 2L settings, a higher proportion of patients with KRAS G12C–positive tumors were female, were former or current smokers, and had tumors with a higher proportion of PD-L1–high and non-squamous histology than patients with KRAS WT cancer (Table 1). When limiting to patients with known PD-L1 status, 41.9% (83/198) of KRAS G12C-positive patients were PD-L1-high as compare to 24.2% (139/575) of KRAS WT patients (p < 0.001).
Effect of KRAS G12C mutational status on OS by treatment type
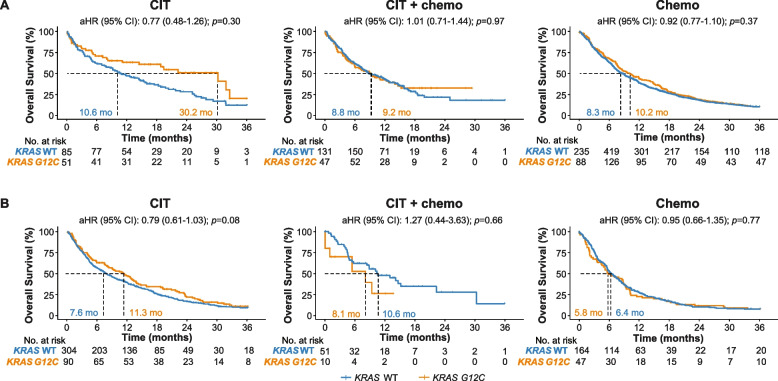
In the 1L setting, patients with KRAS G12C–positive cancer treated with CIT alone demonstrated numerical, but not statistically significant, longer OS (median, 30.2 months; 95% CI, 14.5-not reached [NR]) vs patients with KRAS WT cancer (median, 10.6 months; 95% CI, 7.9–15.6; adjusted hazard ratio [aHR], 0.77; 95% CI, 0.48–1.26; p = 0.30) (Fig. 3A). The median (95% CI) OS in patients with KRAS G12C-positive vs KRAS WT cancer treated with chemotherapy alone was 10.2 (8.0-12.5) months vs 8.3 (7.4-9.4) months (aHR, 0.92; 95% CI, 0.77–1.10; p = 0.37). In patients treated with combination CIT and chemotherapy, median OS was 9.2 (5.9-14.0) months in patients with KRAS G12C-positive tumors vs. 8.8 (7.4-11.9) months in patients with KRAS WT tumors (aHR, 1.01; 95% CI, 0.71–1.44; p = 0.97) (Fig. 3A).
Fig. 3.
KRAS Mutational Status on OS by Treatment in (A) the 1L* and (B) the 2L†. 1L, first line; 2L, second line; aHR, adjusted hazard ratio; CIT, cancer immunotherapy; ns, non-significant; OS, overall survival; PD-L1, programmed cell death 1 ligand 1; TMB, tumor mutational burden; WT, wild type. * n = 1798; adjusted by age, sex, race, cancer type, PD-L1, any metastasis, TMB, and histology in the 1L. † n = 1016; adjusted by age, sex, race, cancer type, PD-L1, any metastasis, TMB, histology, and drug category in the 1L
It is interesting that for patients receiving 1L CIT alone, median OS for patients with KRAS G12C-positive cancer is numerically longer than patients with KRAS WT cancer. To assess the potential effect of different PD-L1 expression level, we calculated median OS in patients with high (≥50%) PD-L1 expression level, and observed a similar trend of longer OS in patients with KRAS G12C-positive cancer treated with CIT alone (median, 32.6 months; 95% CI, 19.4-NR) vs patients with KRAS WT cancer (median, 10.0 months; 95% CI, 5.2–17.5; aHR, 0.57; 95% CI, 0.28–1.14, p = 0.11) (Table 2).
Table 2.
OS in patients with KRAS G12C or wild-type tumors with high PD-L1 (≥50%) and receiving 1L CIT alone
| N | Death | Median OS (95% CI) | Adjusted* HR (95% CI) p-value |
|
|---|---|---|---|---|
| KRAS wild-type | 65 | 41 | 10.0 (5.2–17.5) | Reference |
| KRAS G12C | 43 | 13 | 32.6 (19.4-NR) |
0.57 (0.28–1.14) p = 0.11 |
*Adjusted for age, sex, de novo or recurrent disease, metastasis at baseline, TMB status and histology
It is also interesting that for patients with KRAS G12C-positive cancer, median OS for 1L CIT alone is numerically longer than 1L combination CIT and chemotherapy (30.2 months (14.5-NR) vs. 9.2 (5.9–14.0)), we postulate that one key difference in the tumor PD-L1 expression level might at least partially explain this observation. While 1L combination chemotherapy and CIT was approved for patients with NSCLC tumors with any PD-L1 levels, 1L CIT alone was approved only for patients with NSCLC tumors expressing PD-L1. Therefore, patients treated with CIT alone may have tumors with overall higher PD-L1 expression, compared to those treated with the combination chemotherapy and CIT. Indeed, in our study, 59.7% of KRAS G12C-positive patients treated with 1L CIT alone vs 17.8% of patients treated with 1L combination chemotherapy and CIT have high PD-L1 expression (Table 3). When limiting to patients with known PD-L1 status, 78.2% of KRAS G12C-positive patients treated with 1L CIT alone vs 28.6% of patients treated with 1L combination chemotherapy and CIT have high PD-L1 expression (Table 3). Higher PD-L1 expression might be one reason that the KRAS G12C-positive patients receiving 1L CIT alone tend to have longer median OS than patients receiving 1L combination chemotherapy and CIT.
Table 3.
Percentage of patients with a high (≥50%), low (1–49%) or negative (< 1%) PD-L1 status by treatment type among aNSCLC patients (A) or those with known PD-L1 status (B) receiving 1L CIT alone, combination chemotherapy and CIT, or chemotherapy
| A. | ||||||
| PD-L1a category | KRAS G12C | KRAS WT | ||||
|
1L CIT alone patients (N = 72) |
1L combination chemotherapy + CIT patients (N = 101) |
1L Chemotherapy patients (N = 281) |
1L CIT alone patients (N = 145) |
1L combination chemotherapy + CIT patients (N = 262) |
1L Chemotherapy patients (N = 937) | |
| High (≥50%) | 43 (59.7) | 18 (17.8) | 22 (7.8) | 65 (44.8) | 27 (10.3) | 47 (5.0) |
| Low (1–49%) | 10 (13.9) | 24 (23.8) | 35 (12.5) | 23 (15.9) | 63 (24.0) | 122 (13.0) |
| Negative (< 1%) | 2 (2.8) | 21 (20.8) | 23 (8.2) | 7 (4.8) | 74 (28.2) | 147 (15.7) |
| Missing/unknown | 17 (23.6) | 38 (37.6) | 201 (71.5) | 50 (34.5) | 98 (37.4) | 621 (66.3) |
| B. | ||||||
| PD-L1a category excluding unknown | KRAS G12C | KRAS WT | ||||
|
1L CIT alone patients (N = 55) |
1L combination chemotherapy + CIT patients (N = 63) |
1L chemotherapy patients (N = 80) |
1L CIT alone patients (N = 95) |
1L combination chemotherapy + CIT patients (N = 164) |
1L chemotherapy patients (N = 316) | |
| High (≥50%) | 43 (78.2) | 18 (28.6) | 22 (27.5) | 65 (68.4) | 27 (16.5) | 47 (14.9) |
| Low (1–49%) | 10 (18.2) | 24 (38.1) | 35 (43.8) | 23 (24.2) | 63 (38.4) | 122 (38.6) |
| Negative (< 1%) | 2 (3.6) | 21 (33.3) | 23 (28.8) | 7 (7.4) | 74 (45.1) | 147 (46.5) |
1L first line; aNSCLC advanced non-small cell lung cancer; CIT cancer immunotherapy; PD-L1 programmed death-ligand 1; WT wild type
aPD-L1 high was defined as Tumor Proportion Score ≥ 50%, low 1–49% and negative < 1%
In the 2L setting, median OS in patients with KRAS G12C–positive vs KRAS WT cancer treated with CIT alone was 11.3 (8.1–13.8) months vs 7.6 (6.3–9.3) months (aHR, 0.79; 95% CI, 0.61–1.03; p = 0.08). Median OS in patients treated with chemotherapy alone was 5.8 (4.4–9.3) months vs 6.4 (5.6–7.9) months (aHR, 0.95; 95% CI, 0.66–1.35; p = 0.77), and median OS in patients treated with combination CIT and chemotherapy was 8.1 (1.0-NR) months vs 10.6 (8.5-NR) months (aHR, 1.27; 95% CI, 0.44–3.63; p = 0.66) (Fig. 3B).
Effect of KRAS G12C mutational status and co-occurring STK11 and/or KEAP1 mutations on OS by treatment line
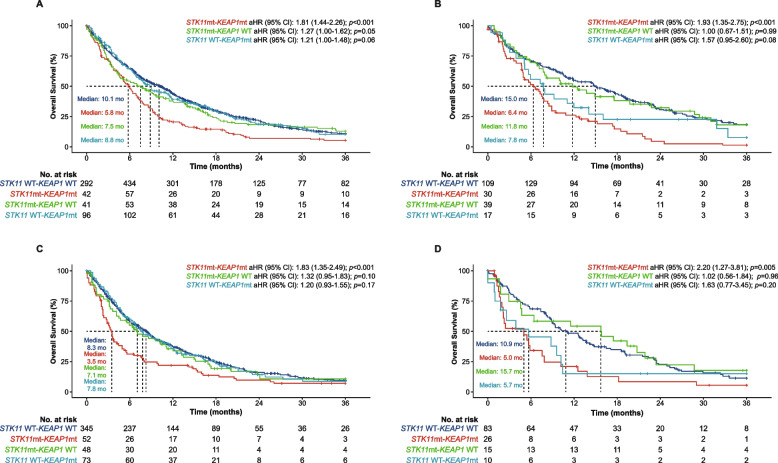
For patients with KRAS WT cancer across all treatment types in the 1L setting, significantly shorter median OS was observed in patients with mSTK11-mKEAP1 vs STK11 WT-KEAP1 WT (5.8 vs 10.1 months; aHR, 1.81; 95% CI, 1.44–2.26; p < 0.001) (Fig. 4A). Numerically shorter median OS was also observed in patients with mSTK11-KEAP1 WT vs STK11 WT-KEAP1 WT (7.5 vs 10.1 months; aHR, 1.27; 95% CI, 1.00–1.62; p = 0.05) and with mKEAP1-STK11 WT vs STK11 WT-KEAP1 WT (8.8 vs 10.1 months; aHR, 1.21; 95% CI, 1.00–1.48; p = 0.06). A similar pattern of results was observed in patients with KRAS G12C–positive cancer: median OS was significantly shorter in patients with mSTK11-mKEAP1 vs STK11 WT-KEAP1 WT (6.4 vs 15.0 months; aHR, 1.93; 95% CI, 1.35–2.75; p < 0.001) (Fig. 4B). Median OS was also numerically shorter in patients with mKEAP1-STK11 WT vs STK11 WT-KEAP1 WT (7.8 vs 15.0 months; aHR, 1.57; 95% CI, 0.95–2.60; p = 0.08). Patients with mSTK11-KEAP1 WT had numerically shorter median OS vs STK11 WT-KEAP1 WT, although this difference was not statistically significant (11.8 vs 15.0 months; aHR, 1.00; 95% CI, 0.67–1.51; p = 0.99).
Fig. 4.
Effect of STK11 and/or- KEAP1 Mutations on OS in Patients With aNSCLC in the 1L* With (A) KRAS WT or (B) KRAS G12C and in the 2L† With (C) KRAS WT or (D) KRAS G12C. 1L, first line; 2L, second line; aHR, adjusted hazard ratio; aNSCLC, advanced non-small cell lung cancer; ns, non-significant; OS, overall survival; PD-L1, programmed cell death 1 ligand 1; TMB, tumor mutational burden; WT, wildtype. * Adjusted by age, sex, race, cancer type, PD-L1, any metastasis, TMB, and histology. † Adjusted by age, sex, race, cancer type, PD-L1, any metastasis, TMB, histology, and drug category in the 1L. ‡ HR comparing STK11 WT-KEAP1 WT vs mSTK11 and/or mKEAP1
For patients with KRAS WT cancer across all treatment types in the 2L setting, significantly shorter OS was observed in patients with mSTK11-mKEAP1 vs STK11 WT-KEAP1 WT (3.5 vs 8.3 months; aHR, 1.83; 95% CI, 1.35–2.49; p < 0.001) (Fig. 4C). Similarly, median OS was significantly shorter in patients with KRAS G12C–positive cancer with mSTK11-mKEAP1 vs STK11 WT-KEAP1 WT (5.0 vs 10.9 months; aHR, 2.20; 95% CI, 1.27–3.81; p = 0.005) (Fig. 4D). Differences in OS in other mutational groups compared with STK11 WT-KEAP1 WT were non-significant.
Discussion
This retrospective real-world study used an EHR-linked CGDB to assess OS in patients with KRAS WT and KRAS G12C–positive aNSCLC by treatment line, treatment type, and co-mutations in STK11 and/or KEAP1.
Patients with aNSCLC with KRAS G12C–positive tumors were found to have comparable OS relative to patients with KRAS WT tumors when receiving combination CIT and chemotherapy or chemotherapy alone in the 1L or 2L; this finding is consistent with majority of the previous literatures using other datasets [12, 13, 21, 22].
Although not statistically significant, potentially due to limited patient counts, patients with KRAS G12C–positive cancer treated with 1L CIT showed a trend toward longer survival vs patients with KRAS WT cancer. Similar results were observed in patients with high (≥50%) PD-L1 expression level. These results with significantly larger sample size are consistent with previous reports of improved survival with immune checkpoint inhibitors in KRAS-mutant aNSCLC [12]. Consistent with the findings of this study, KRAS mutations are associated with increased PD-L1 expression in patients with aNSCLC [23] and contribute to immunosuppression [24]. A recent study [25] also found a strong association between mutated KRAS and immune biomarkers linked to response to immune checkpoint inhibition [26]. Together these results suggest that patients with aNSCLC whose tumors harbor KRAS mutations may have particularly favorable outcomes with CIT [26], which is distinct from EGFR-mutated NSCLC [26, 27]. Also, since the single agent KRAS G12C inhibitors appear to have a lower overall response rate compared to the target-specific tyrosine kinase inhibitors [26, 28–30], it is possible that the KRAS G12C inhibitors need to be combined with CIT moving into 1L. Further analyses are needed to evaluate KRAS mutational status in patients with aNSCLC treated with CIT alone and to explore the effect of different KRAS variants (e.g., G12D and G12V) on survival relative to G12C.
In this study, patients with aNSCLC and concurrent double mutations in STK11 and KEAP1 had significantly shorter OS vs patients with STK11 WT and KEAP1 WT receiving any type of 1L or 2L therapy regardless of KRAS G12C mutational status. This is consistent with recent retrospective analyses conducted using an EHR-linked CGDB that found that mutations in STK11 and KEAP1 in aNSCLC were associated with worse outcomes (shorter progression-free survival and OS) in patients treated with anti–PD-L1/PD-1 therapies and platinum-based chemotherapy [31]. Our results build on these previous findings and further underscore the poor prognosis and high unmet medical need that exists in patients with aNSCLC with co-occurring mutations in STK11 and KEAP1, with either KRAS WT cancer or KRAS G12C–positive cancer. If KRAS G12C-positive patients with STK11 co-mutations are sensitive to the KRAS G12C inhibitors, this may serve as the rationale of expediting the exploration of single-agent KRAS G12C inhibitors in certain biomarker-selected patient sub-population in 1L.
Initial studies of KRAS G12C inhibitors excluded patients with active brain metastasis [11]. Our study showed that KRAS G12C-positive patients have a higher prevalence of brain metastasis as compared to patients with KRAS WT tumors. It is therefore critical to evaluate whether KRAS G12C inhibitors can be beneficial for patients with brain metastasis and brain-penetration may be a key consideration for future generations of KRAS G12C inhibitors.
This study has several limitations. CGDB data are generated from real-world clinical practice; thus, some data may have been miscoded or may be subject to errors encountered in an oncology clinic. The data do not capture information about patients’ history or treatment outside of the specific cancer care site, which may lead to underreporting or missing data. Limited data exist from patients attending or beginning treatment elsewhere, such as academic medical centers, as the CGDB largely reflects community oncology treatment information. The study population was comprised of patients who received treatment in the United States, and the results may not be generalizable to patients treated globally. For specimen collection, patients were categorized as having KRAS G12C or KRAS WT cancer based on the closest specimen collection to index date (1L or 2L treatment initiation). With this approach, patients may have developed a KRAS mutation after receiving therapy; however, sensitivity analyses were conducted to evaluate selection bias. Analyses were rerun in patients with a specimen collection date within 90 days of the index date (1L or 2L treatment initiation), and results were found to be consistent. We categorized patients with KRAS G12C, KEAP1, and STK11 mutational status using both solid and liquid assays; patients with cancer that was categorized as WT were assigned based on solid assays only. To avoid selection bias, we further restricted and subsequently reperformed our analyses of patients categorized using solid assays only. The results remained consistent; however, to ensure a larger sample size, patients whose mutational status was determined using liquid assays were included in the final analysis. Due to the entry selection rules, the CGDB is inherently left truncated. For inclusion in the cohort, patients were required to have undergone NGS testing by FMI, and, therefore, must have been alive until the date of the NGS test. The analyses were adjusted for left truncation to control for this potential immortal bias. Eastern Cooperative Oncology Group performance statuses were missing in 14 to 23% of patients and not adjusted for due to this high missingness. A higher proportion of patients with KRAS G12C–positive tumors had non-squamous histology than patients with KRAS WT tumors. Although squamous, NOS vs non-squamous histology was adjusted for, no further details such as adenocarcinoma was available in the CGDB. This study included patients with locally advanced (stage IIIB and IIIC) and metastatic (stage IV) diseases, which could have also progressed from an initial diagnosis at early stages (stage I, II and IIIA). Although stage at initial diagnosis was not adjusted for, cancer type (de novo vs recurrent) and presence of metastasis at baseline were included in the model to adjust for heterogeneity in patient population. Finally, this study included a high percentage of patients with missing PD-L1 data; PD-L1 status was unknown for > 50% of patients in each group. Despite these limitations, these findings highlight the importance of evaluating genomic alterations in clinical practice to better understand how selection of treatment and therapy type affect survival in patients with tumors bearing genomic alterations. This study offers a unique design advantage to the published literature to date. Other studies have assessed the effect of STK11 and/or KEAP1 mutations either in patients whose tumors harbor KRAS mutations only, or in a mixed patient population whose tumors could harbor either KRAS WT or KRAS mutations [32–35]. This study evaluated survival benefit with different 1L and 2L therapies in patients with KRAS G12C or WT cancer with or without STK11 and/or KEAP1 mutations, providing additional insights into these frequently co-occurring mutations. Further analyses with a larger sample size evaluating the interplay between KRAS and STK11 and/or KEAP1 by select treatment types are warranted.
Results from this study may inform personalized treatment for patients with KRAS-mutated NSCLC, as certain combinations of mutations in KRAS and other genes may generate biological diversity that may respond to tailored treatment [36]. Together, these results may enable personalized care and help optimize patient outcomes.
Conclusions
Patients with aNSCLC with KRAS G12C mutations who were treated with 1L and 2L chemotherapy, and combination CIT and chemotherapy had similar OS compared with patients with KRAS WT cancer. Patients with KRAS G12C–positive cancer showed a (non-significant) numerically longer OS when treated with CIT monotherapy. Further validation with independent datasets is warranted. Co-occurring mutations in STK11 and KEAP1 were associated with significantly shorter OS in the 1L and 2L settings, regardless of KRAS G12C or WT status. This study highlights the importance of evaluating genomic alterations in clinical practice to better understand the interplay between treatment type and survival.
Acknowledgements
We thank Melanie Huntley and Emily Casey for their valuable contributions and Pragati Sharma for her assistance with this work.
Abbreviations
- 1L
First line
- 2L
Second line
- aHR
Adjusted hazard ratio
- aNSCLC
Advanced non-small cell lung cancer
- CGBD
Clinico-genomic database
- CGP
Comprehensive genomic profiling
- CIT
Cancer immunotherapy
- EHR
Electronic health record
- FH-FMI
Flatiron Health-Foundation Medicine
- IRB
Institutional Review Board
- KEAP1
kelch like ECH associated protein 1
- KM
Kaplan-Meier
- KRAS
Kirsten Rat Sarcoma Viral Oncogene Homolog
- NGS
Next-generation sequencing
- NOS
Not otherwise specified
- ns
Non-significant
- NR
Not reached
- NSCLC
Non-small cell lung cancer
- OS
Overall survival
- p
p-value
- PD-1
Programmed cell death protein 1
- PD-L1
Programmed cell death 1 ligand 1
- STK11
Serine/threonine kinase 11
- SV
Short variant
- TMB
Tumor mutational burden
- WT
Wild type
Authors’ contributions
CJ, ME, HP, ZS, and QZ were responsible for conceptualization. CJ, NP, AG, and QZ contributed to methodology, validation, and project administration. CJ, AG, QZ, NP, and PL conducted formal analysis and data curation. All authors contributed substantially to the interpretation and writing of the manuscript. The author(s) read and approved the final manuscript.
Funding
This work was sponsored by F. Hoffmann-La Roche Ltd., and Genentech, Inc. Support for third-party writing assistance, furnished by Sarah Nordquist, PhD, of Health Interactions, Inc., was provided by F. Hoffmann-La Roche Ltd.
Availability of data and materials
The data that support the findings of this study originated from Flatiron Health, Inc., and Foundation Medicine, Inc. These deidentified data may be made available upon request and are subject to a license agreement with Flatiron Health and Foundation Medicine, Inc.; interested researchers should contact Flatiron Health at n to determine licensing terms.
Declarations
Ethics approval and consent to participate
No human subjects were involved in this study. This study utilized a de-identified database, which according to the US Code of Federal Regulations (45 CFR 46.102(e) (1)) is not considered human subjects research. Data used for this study were licensed for use from Flatiron Health, Inc. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines were used to guide the reporting of this observational study, and all methods were carried out in accordance to relevant guideline and regulation. Institutional Review Board (IRB) approval of the study protocol, including a waiver of informed consent, was obtained prior to study conduct, from the WCG IRB. Please note that WCG is the full name of the IRB, not an abbreviation.
Consent for publication
Not applicable.
Competing interests
CJ, NP, AG, ME, HP, PL, ZS, and QZ are employees of Genentech, Inc., and shareholders of F. Hoffmann-La Roche Ltd. QZ also owns stock in Regeneron, BMS, Pfizer, BioNTech, AC Immune, and AbbVie.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Zhen Shi and Qing Zhang contributed equally to this work.
Contributor Information
Zhen Shi, Email: shi.zhen@gene.com.
Qing Zhang, Email: zhangq47@gene.com.
References
- 1.Herbst RS, Morgensztern D, Boshoff C. The biology and management of non-small cell lung cancer. Nature. 2018;553(7689):446–454. doi: 10.1038/nature25183. [DOI] [PubMed] [Google Scholar]
- 2.Chu QS. Targeting non-small cell lung cancer: driver mutation beyond epidermal growth factor mutation and anaplastic lymphoma kinase fusion. Ther Adv Med Oncol. 2020;12:1758835919895756. doi: 10.1177/1758835919895756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.NCCN guidelines non-small cell lung cancer, version 3.2020. 2020.
- 4.Adderley H, Blackhall FH, Lindsay CR. KRAS-mutant non-small cell lung cancer: converging small molecules and immune checkpoint inhibition. EBioMedicine. 2019;41:711–716. doi: 10.1016/j.ebiom.2019.02.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Yang H, Liang S-Q, Schmid RA, Peng R-W. New horizons in KRAS-mutant lung cancer: dawn after darkness. Front Oncol. 2019;9:953. doi: 10.3389/fonc.2019.00953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Skoulidis F, Heymach JV. Co-occurring genomic alterations in non-small-cell lung cancer biology and therapy. Nat Rev Cancer. 2019;19(9):495–509. doi: 10.1038/s41568-019-0179-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dearden S, Stevens J, Wu YL, Blowers D. Mutation incidence and coincidence in non small-cell lung cancer: meta-analyses by ethnicity and histology (mutMap) Ann Oncol. 2013;24(9):2371–2376. doi: 10.1093/annonc/mdt205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Uras IZ, Moll HP, Casanova E. Targeting KRAS mutant non-small-cell lung cancer: past, present and future. Int J Mol Sci. 2020;21(12) [DOI] [PMC free article] [PubMed]
- 9.Bar-Sagi D, Knelson EH, Sequist LV. A bright future for KRAS inhibitors. Nat Cancer. 2020;1(1):25–27. doi: 10.1038/s43018-019-0016-8. [DOI] [PubMed] [Google Scholar]
- 10.Herdeis L, Gerlach D, McConnell DB, Kessler D. Stopping the beating heart of cancer: KRAS reviewed. Curr Opin Struct Biol. 2021;71:136–147. doi: 10.1016/j.sbi.2021.06.013. [DOI] [PubMed] [Google Scholar]
- 11.Skoulidis F, Li BT, Dy GK, Price TJ, Falchook GS, Wolf J, et al. Sotorasib for lung cancers with KRAS p.G12C mutation. N Engl J Med. 2021;384(25):2371–2381. doi: 10.1056/NEJMoa2103695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Herbst RS, Lopes G, Kowalski DM, Kasahara K, editors. Association of KRAS mutational status with response to pembrolizumab monotherapy given as first-line therapy for PD-L1-positive advanced nonsquamous NSCLC in KEYNOTE-042. Geneva: European Society of Medical Oncology Immuno-Oncology Congress; 2019. [Google Scholar]
- 13.Gadgeel S, Rodriguez-Abreu D, Felip E, Esteban E, editors. KRAS mutational status and efficacy in KEYNOTE-189: pembrolizumab (pembro) plus chemotherapy (chemo) vs placebo plus chemo as first-line therapy for metastatic non-squamous NSCLC. Geneva: European Society of Medical Oncology Immuno-Oncology Congress; 2019. [Google Scholar]
- 14.Arbour KC, Jordan E, Kim HR, Dienstag J, Yu HA, Sanchez-Vega F, et al. Effects of co-occurring genomic alterations on outcomes in patients with KRAS-mutant non-small cell lung cancer. Clin Cancer Res. 2018;24(2):334–340. doi: 10.1158/1078-0432.CCR-17-1841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bange E, Marmarelis ME, Hwang W-T, Yang Y-X, Thompson JC, Rosenbaum J, et al. Impact of KRAS and TP53 co-mutations on outcomes after first-line systemic therapy among patients with STK11-mutated advanced non-small-cell lung cancer. JCO precis. Oncol. 2019:3. [DOI] [PMC free article] [PubMed]
- 16.Spira AI, Tu H, Aggarwal S, Hsu H, Carrigan G, Wang X, et al. A retrospective observational study of the natural history of advanced non–small-cell lung cancer in patients with KRAS p.G12C mutated or wild-type disease. Lung Cancer. 2021;159:1–9. doi: 10.1016/j.lungcan.2021.05.026. [DOI] [PubMed] [Google Scholar]
- 17.Singal G, Miller PG, Agarwala V, Li G, Kaushik G, Backenroth D, et al. Association of patient characteristics and tumor genomics with clinical outcomes among patients with non–small cell lung cancer using a clinicogenomic database. JAMA. 2019;321(14):1391–1399. doi: 10.1001/jama.2019.3241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Frampton GM, Fichtenholtz A, Otto GA, Wang K, Downing SR, He J, et al. Development and validation of a clinical cancer genomic profiling test based on massively parallel DNA sequencing. Nat Biotechnol. 2013;31(11):1023–1031. doi: 10.1038/nbt.2696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.He J, Abdel-Wahab O, Nahas MK, Wang K, Rampal RK, Intlekofer AM, et al. Integrated genomic DNA/RNA profiling of hematologic malignancies in the clinical setting. Blood. 2016;127(24):3004–3014. doi: 10.1182/blood-2015-08-664649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Woodhouse R, Li M, Hughes J, Delfosse D, Skoletsky J, Ma P, et al. Clinical and analytical validation of FoundationOne liquid CDx, a novel 324-gene cfDNA-based comprehensive genomic profiling assay for cancers of solid tumor origin. PLoS One. 2020;15(9):e0237802. doi: 10.1371/journal.pone.0237802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Cui W, Franchini F, Alexander M, Officer A, Wong H-L, Ijzerman M, et al. Real world outcomes in KRAS G12C mutation positive non-small cell lung cancer. Lung Cancer. 2020;146:310–317. doi: 10.1016/j.lungcan.2020.06.030. [DOI] [PubMed] [Google Scholar]
- 22.Sebastian M, Eberhardt WEE, Hoffknecht P, Metzenmacher M, Wehler T, Kokowski K, et al. KRAS G12C-mutated advanced non-small cell lung cancer: a real-world cohort from the German prospective, observational, nation-wide CRISP registry (AIO-TRK-0315) Lung Cancer. 2021;154:51–61. doi: 10.1016/j.lungcan.2021.02.005. [DOI] [PubMed] [Google Scholar]
- 23.D'Incecco A, Andreozzi M, Ludovini V, Rossi E, Capodanno A, Landi L, et al. PD-1 and PD-L1 expression in molecularly selected non-small-cell lung cancer patients. Br J Cancer. 2015;112(1):95–102. doi: 10.1038/bjc.2014.555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kalvala A, Wallet P, Yang L, Wang C, Li H, Nam A, et al. Phenotypic switching of naïve t cells to immune-suppressive treg-like cells by mutant KRAS. J Clin Med. 2019;8(10) [DOI] [PMC free article] [PubMed]
- 25.Liu C, Zheng S, Jin R, Wang X, Wang F, Zang R, et al. The superior efficacy of anti-PD-1/PD-L1 immunotherapy in KRAS-mutant non-small cell lung cancer that correlates with an inflammatory phenotype and increased immunogenicity. Cancer Lett. 2020;470:95–105. doi: 10.1016/j.canlet.2019.10.027. [DOI] [PubMed] [Google Scholar]
- 26.Salgia R, Pharaon R, Mambetsariev I, Nam A, Sattler M. The improbable targeted therapy: KRAS as an emerging target in non-small cell lung cancer (NSCLC) Cell Rep Med. 2021;2(1):100186. doi: 10.1016/j.xcrm.2020.100186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lisberg A, Cummings A, Goldman JW, Bornazyan K, Reese N, Wang T, et al. A phase II study of Pembrolizumab in EGFR-mutant, PD-L1+, tyrosine kinase inhibitor Naïve patients with advanced NSCLC. J Thorac Oncol. 2018;13(8):1138–1145. doi: 10.1016/j.jtho.2018.03.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Mok TS, Wu YL, Ahn MJ, Garassino MC, Kim HR, Ramalingam SS, et al. Osimertinib or platinum-Pemetrexed in EGFR T790M-positive lung Cancer. N Engl J Med. 2017;376(7):629–640. doi: 10.1056/NEJMoa1612674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ou SH, Ahn JS, De Petris L, Govindan R, Yang JC, Hughes B, et al. Alectinib in Crizotinib-refractory ALK-rearranged non-small-cell lung Cancer: a phase II global study. J Clin Oncol. 2016;34(7):661–668. doi: 10.1200/JCO.2015.63.9443. [DOI] [PubMed] [Google Scholar]
- 30.Drilon A, Oxnard GR, Tan DSW, Loong HHF, Johnson M, Gainor J, et al. Efficacy of Selpercatinib in. N Engl J Med. 2020;383(9):813–824. doi: 10.1056/NEJMoa2005653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Papillon-Cavanagh S, Doshi P, Dobrin R, Szustakowski J, Walsh AM. STK11 and KEAP1 mutations as prognostic biomarkers in an observational real-world lung adenocarcinoma cohort. ESMO Open. 2020;5(2) [DOI] [PMC free article] [PubMed]
- 32.Nie W, Gan L, Wang X, Gu K, Qian F-F, Hu M-J, et al. Atezolizumab prolongs overall survival over docetaxel in advanced non-small-cell lung cancer patients harboring STK11 or KEAP1 mutation. OncoImmunology. 2021;10(1):1865670. doi: 10.1080/2162402X.2020.1865670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Cho BC, Lopes G, Kowalski DM, Kasahara K, editors. Relationship between STK11 and KEAP1 mutational status and efficacy in KEYNOTE-042: pembrolizumab monotherapy versus platinum-based chemotherapy as first-line therapy for PD-L1-positive advanced NSCLC. American Association for Cancer Research Virtual Annual Meeting II; 2020. [Google Scholar]
- 34.Skoulidis F, Goldberg ME, Greenawalt DM, Hellmann MD, Awad MM, Gainor JF, et al. STK11/LKB1 mutations and PD-1 inhibitor resistance in KRAS-mutant lung adenocarcinoma. Cancer Discov. 2018;8(7):822–835. doi: 10.1158/2159-8290.CD-18-0099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ricciuti B, Recondo G, Umeton R, Nishino M, Sholl LM, Awad MM. Impact of KRAS allele subtypes and concurrent genomic alterations on clinical outcomes to programmed death 1 axis blockade in non-small cell lung cancer. JCO. 2019;37(15_suppl):9082. doi: 10.1200/JCO.2019.37.15_suppl.9082. [DOI] [Google Scholar]
- 36.Skoulidis F, Byers LA, Diao L, Papadimitrakopoulou VA, Tong P, Izzo J, et al. Co-occurring genomic alterations define major subsets of KRAS-mutant lung adenocarcinoma with distinct biology, immune profiles, and therapeutic vulnerabilities. Cancer Discov. 2015;5(8):860–877. doi: 10.1158/2159-8290.CD-14-1236. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data that support the findings of this study originated from Flatiron Health, Inc., and Foundation Medicine, Inc. These deidentified data may be made available upon request and are subject to a license agreement with Flatiron Health and Foundation Medicine, Inc.; interested researchers should contact Flatiron Health at n to determine licensing terms.