Abstract
Introduction
This study investigated the anatomic feasibility of a new surgical therapy option for radial head arthrosis using an autologous vascularized bone graft of the second metatarsal and proximal fibula to recreate the proximal radiohumeral joint.
Materials and methods
Upper and lower extremities of eleven body donors were evaluated using CT prior to anatomic dissection. Several distinct anatomic parameters were measured on the ipsi- and contralateral radial and fibular head and the second metatarsal base: bone diameter, articular surface diameter, head height, metaphyseal (neck) diameter, articular surface radius, total articular surface area, and angulation of the articular surfaces (facet). Each dissection phase was photographed in a standardized fashion and all measurements were repeated by direct caliper-measurements.
Results
When comparing the proximal radius and fibula to search for anatomic similarities, similar values were found in the maximum articular surface diameter and minimum and maximum measures of the neck diameter. Comparing the proximal radius and the second metatarsal, statistically similar values were found in the maximum neck diameter performing direct measurements and CT evaluation, the maximum head diameter in CT evaluation and the articular facet angulation.
Conclusions
Neither the proximal fibula nor the base of the second metatarsal are ideal bone grafts for replacement of the head of the radius. The base of the second metatarsal might be a bit more suitable as a potential donor since the angulation of the proximal articular facet is similar to that of the radius.
Level of evidence
Level IV, anatomic study.
Keywords: Radial head, Radial head fracture, Osteoarthritis, Radial head dislocation, Elbow prosthesis, Fibular autograft, Second metatarsal
Introduction
Proximal radius fractures in children and adolescents account for around 4–21% of all pediatric elbow injuries (1, 2). Although pediatric radial head/neck fracture treatment can generally be considered successful if initiated promptly, delayed or missed treatment can cause significant morbidity due to the precarious vascularization of the head of the radius and the associated potential for necrosis and remodeling defects (3–5). In this regard, severe proximal radius malalignment may cause progressive radial head dislocation and disruption of the proximal radioulnar joint integrity (6). The head of the radius eventually loses its native concavity and degenerates over time. Thus, the causes for pediatric radial head degeneration and early arthrosis are mainly posttraumatic reasons as well as—more rarely—avascular necrosis (7).
While prosthetic treatment of radial head arthrosis in adults is an established procedure, no such opportunity exists in the pediatric age group (8). Secondary corrective osteotomy techniques are usually effective in reliably realigning the proximal radius since the head of the radius has usually lost its anatomic orientation and congruency. Hence, radial head resection might be the only solution to reduce pain and improve function in this patient cohort (9–12). However, such procedure is not without risks due to the potential occurrence of lateral elbow instability and longitudinal forearm instability (12, 13). The latter may cause a vicious circle of consecutive ulnocarpal impaction syndrome with a need of ulnar shortening procedures due to the proximal radial migration (14). To prevent such occurrence, multiple interposition arthroplasty techniques have already been described for the pediatric population (9, 11).
Given the above-mentioned circumstances and obstacles, native radial head replacement with autologous tissue would be desirable for children, adolescents and young adults. This, however, is a quite difficult task to fulfill, as the elbow joint with its three articular parts is a quite complex joint. Only the interaction between humerus and ulna, humerus and radius as well as radius and ulna allow the unhampered movability and stability of the joint. In this, the morphology of the humeroradial and radioulnar joint surfaces and thus of the proximal radius itself play a large role. The size and the orientation of the superior articular surface of the head of the radius are important for its articulation with the humeral capitulum, the articular surface of the radial circumference and the size of the radial neck are important for its articulation with the radial notch of the ulna and the interaction with the annular ligament of the radius (1, 15–18). Thus, the donor bone should not only involve a bone whose function is sufficiently compensated biomechanically, but which also adequately accomplishes the required tasks in the complex elbow joint.
Based on these anatomic considerations, we hypothesized that the second metatarsal bone and the fibular head would be suitable donors for a replacement of the proximal radius. This study thus investigated the feasibility of a new surgical therapy option using an autologous vascularized bone graft of the second metatarsal and proximal fibula to recreate the radiohumeral joint. Anatomic features and landmarks were evaluated to determine whether these bones would be potential donors for such degenerative indications.
Materials and methods
For the purpose of this study, specimens from eleven body donors were used. The donors gave their consent during life for their body to be used for scientific and teaching purposes to the Center for Anatomy and Cell Biology of the Medical University of Vienna. Additionally, the study was approved by the ethical committee of the Medical University of Vienna (1314/2017). Of these eleven body donors, both upper and lower extremities were included, resulting in a total of 22 upper and lower extremities, respectively. The specimens were used fresh and were only stored at 4 °C prior to imaging and dissection. Inclusion criteria for this study were the absence of any signs of previous pathologies (e.g., injuries, tumors), previous surgery (e.g., endoprosthetic replacement, plating), advanced osteoarthritis of the joint and congenital malformations in the specimens.
Evaluation
First, CT evaluation was performed on the entire upper and lower limbs prior to anatomic dissection to preserve anatomic distances in and around the joint area. All CT scans were conducted on the same scanner (Siemens SOMATOM FORCE; Siemens Healthcare AG, Erlangen, Germany) with a slice thickness of 1 mm, 80 kV and activated tube current modulation. A sharp bone kernel (Br69d) was used for reconstruction. The radius and fibula were placed longitudinally along the Z-axis of the scanner. For the second metatarsal bone, the foot was fixed in a 90°-angle to the Z-axis. Multiplanar reconstructions were aligned in a 90°-angle to and along the long axis of the respective bones. Morphological measurements were taken in accordance with the measurements on anatomical specimens. For CT evaluations, Osirix® (www.osirix-viewer.com) was used.
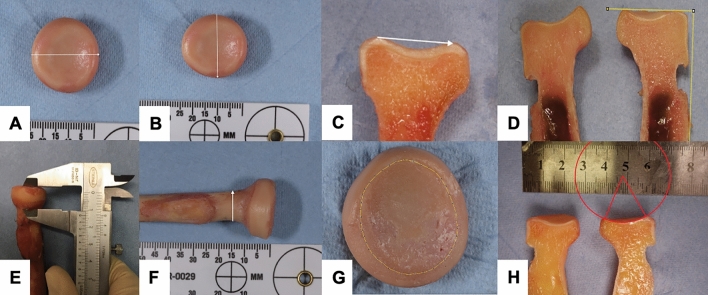
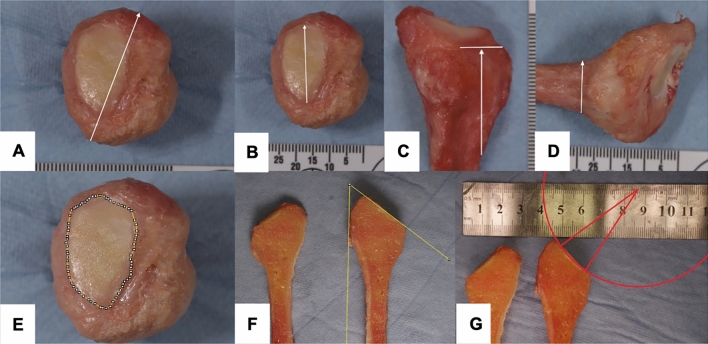
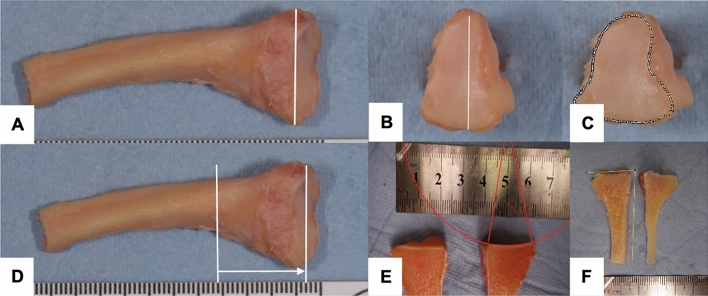
The following parameters were measured on the ipsi- and contralateral radial head (Fig. 1), fibular head (Fig. 2) and the second metatarsal base (Fig. 3) using appropriately selected axial, sagittal and coronar images: bone diameter (minimum, maximum); articular surface diameter (minimum, maximum), head height, metaphyseal (neck) diameter (minimum, maximum), and articular surface radius.
Fig. 1.
The anatomic radial head measurements are shown: bone diameter (A, B), articular surface diameter (C), angulation of the articular surface (D), head height (E), neck diameter (F), articular surface area (G), and articular surface radius (H)
Fig. 2.
The anatomic fibular head measurements are shown: bone diameter (A), articular surface diameter (B), head height (C), neck diameter (D), articular surface area (E), angulation of the articular surface (F), and articular surface radius (G)
Fig. 3.
The anatomic second metatarsal base measurements are shown: bone diameter (A), articular surface diameter (B), articular surface area (C), head height (D), articular surface radius (E), and angulation of the articular surface (F)
Afterwards, all soft tissue was removed from the distal humerus and the proximal radius of the upper extremities as well as from the proximal fibula and the second metatarsal bone of the lower extremities. Thereafter, the extracted bones were cut in half following a longitudinal plane through the axis along the maximum articular surface diameter for evaluation of the articular surface radius. Each dissection phase was photographed in a standardized fashion for further evaluation. All measurements previously performed in the CT images were repeated by direct caliper-measurements (accuracy 0.05 mm) of all specimens. Total articular surface area, articular surface radius, and angulation of the articular surfaces (facet) to the longitudinal axis were measured digitally on the photographs using ImageJ (https://imagej.nih.gov/ij/index.html).
Statistical analysis
The different specimens were compared among each other with regards to their anatomic morphometrics. Mean, standard deviation, minimum and maximum values were computed for each metric variable. A paired t-test was applied for all metric variables in comparisons between the three bones. An unpaired t-test was applied to detect side differences. Pearson’s correlation coefficient was used to evaluate anatomical measurements and CT evaluation. R-values were interpreted following Cohen’s definition (19). R-values smaller than 0.1 show no correlation, between 0.1 and 0.3 weak correlations and higher than 0.5 strong correlations. P-values smaller than 0.05 were deemed significant. Bonferroni-correction was applied due to multiple testing; the p value was thus adjusted to 0.0021 (24 tests). A second rater evaluated an initial series of photographic and CT measurements, and an ICC was established. Following the definition of Cicchetti (20), ICC was interpreted thus: 0.75–1.00 excellent; 0.60–0.74 good; 0.40–0.59 fair; < 0.40 poor agreement.
Results
Upper and lower extremities of four female and seven male body donors were used (age 78.5 ± 11.0 years). No significant differences between sides were detected (p = 0.075–0.991), thus no distinction between right or left side was made for all further comparisons. Descriptive data of all measurements may be found in Table 1.
Table 1.
Descriptive data of all measurements including statistical correlation between direct measurements and CT evaluation (Bonferroni-correction to 0.0021 was applied)
| Minimum | R (p-value) | Maximum | R (p-value) | ||||
|---|---|---|---|---|---|---|---|
| Direct measurement | CT | Direct measurement | CT | ||||
| Radius head diameter (mm) | 23.2 ± 2.1 (19–26.5) | 23.1 ± 2.2 (18.7–26.7) | 0.926 (< 0.001) | 24.9 ± 2.1 (20.5–28) | 24.3 ± 2.1 (19.6–27.7) | 0.974 (< 0.001) | |
| Fibula head diameter (mm) | 28.9 ± 3.1 (24–35) | 29.3 ± 2.6 (25.4–33.6) | 0.921 (< 0.001) | 32.1 ± 3.1 (25–37) | 29.6 ± 3.4 (21.9–36.6) | 0.852 (< 0.001) | |
| 2nd metatarsal base diameter (mm) | 17.9 ± 2.1 (15–22.5) | 17.8 ± 1.7 (14.8–22.3) | 0.857 (< 0.001) | 22.9 ± 2.1 (19–27) | 23.8 ± 2.0 (19.8–27.75) | 0.841 (< 0.001) | |
| Radius head articular surface diameter (mm) | 17.1 ± 1.9 (13.5–21) | 16.9 ± 1.6 (14.3–20.1) | 0.848 (< 0.001) | 18.6 ± 1.9 (14–21) | 18.0 ± 1.4 (15–20.6) | 0.760 (< 0.001) | |
| Fibula head articular surface diameter (mm) | 14.7 ± 1.4 (12–17) | 13.9 ± 2.1 (10.6–18.5) | 0.508 (0.022)* | 17.6 ± 2.1 (15–22) | 17.6 ± 2.7 (11.2–21.4) | 0.625 (0.002) | |
| 2nd metatarsal articular surface diameter (mm) | 8.4 ± 0.9 (6.5–10) | 8.1 ± 1.4 (5.71–10.41) | 0.846 (< 0.001) | 20.4 ± 1.9 (16.5–25) | 20.8 ± 1.9 (16.3–24.6) | 0.870 (< 0.001) | |
| Radius neck diameter (mm) | 13.9 ± 1.4 (11–16) | 13.8 ± 1.4 (10.6–16.2) | 0.841 (< 0.001) | 13.7 ± 1.6 (10.5–16) | 14.3 ± 1.6 (10.8–16.5) | 0.794 (< 0.001) | |
| Fibula neck diameter (mm) | 13.0 ± 2.0 (10–19) | 13.3 ± 1.7 (10.5–17.3) | 0.171 (0.448)* | 12.8 ± 1.8 (10–16) | 13.1 ± 2.1 (10.2–17.3) | 0.614 (0.003)* | |
| 2nd metatarsal proximal metaphysis diameter (mm) | 8.8 ± 1.3 (6–11) | 8.9 ± 1.1 (7.2–11.4) | 0.720 (< 0.001) | 9.3 ± 1.3 (8–13) | 10.0 ± 1.1 (8.43–12.4) | 0.242 (0.279)* | |
| Direct measurement | CT | R (p value) | |||||
| Radius head height (mm) | 12.3 ± 0.9 (11–14) | 13.2 ± 0.9 (11.6–15) | 0.484 (0.022)* | ||||
| Fibula head height (mm) | 17.7 ± 1.4 (15–20) | 23.6 ± 2.2 (19.1–27.7) | 0.483 (0.031)* | ||||
| 2nd metatarsal base height (mm) | 17.1 ± 1.9 (13.5–20) | 19.1 ± 2.5 (13.2–23.6) | 0.729 (< 0.001) | ||||
| Photograph-evaluation | CT | R (p value) | |||||
| Radius articular facet radius (mm) | 21.1 ± 3.7 (15.5–30.4) | 18.7 ± 3.9 (14.5–30.35) | 0.805 (< 0.001) | ||||
| Fibula articular facet radius (mm) | 33.4 ± 10.9 (16.65–57.55) | 28.5 ± 8.9 (14.0–44.6) | 0.936 (< 0.001) | ||||
| 2nd metatarsal articular facet radius (mm) | 44.4 ± 14.9 (24.5–79.5) | 39.8 ± 12.4 (17.45–61.5) | 0.755 (< 0.001) | ||||
| Photograph-evaluation | |||||||
| Radius proximal articular surface area (mm2) | 458.4 ± 83.5 (299.6–591.2) | ||||||
| Fibula articular surface area (mm2) | 196.0 ± 44.0 (132.4–261.9) | ||||||
| 2nd metatarsal base articular surface area (mm2) | 194.4 ± 36.2 (115.1–253) | ||||||
| Radius articular facet angulation (°) | 93.0 ± 4.0 (86.26–100.7) | ||||||
| Fibula articular facet angulation (°) | 61.1 ± 8.7 (44.5–80.7) | ||||||
| 2nd metatarsal articular facet angulation (°) | 88.4 ± 4.1 (81.1–98.3) | ||||||
*No statistically significant correlation
Results were grouped into three morphologically important aspects: Facet angulation, morphology of the articular facet and morphology of the neck. A summary of all statistical comparisons including mean differences between the morphometrics of the three bones can be found in Table 2.
Table 2.
Statistical comparison between the morphometrics of radius, fibula and second metatarsal (Bonferroni-correction to 0.0021 was applied)
| Radius vs | Articular facet angulation (°) | Minimum head diameter (mm) | Maximum head diameter (mm) | Head height (mm) | Minimum articular surface diameter (mm) | Maximum articular surface diameter (mm) | Articular facet radius (mm) | Proximal articular surfaces area (mm2) | Minimum neck diameter (mm) | Maximum neck diameter (mm) |
|---|---|---|---|---|---|---|---|---|---|---|
| Photograph | Direct | Direct | Direct | Direct | Direct | Photograph | Photograph | Direct | Direct | |
| Fibula | ||||||||||
| Mean difference ± standard deviation | 31.9 ± 8.9 | − 5.8 ± 2.2 | − 7.2 ± 2.2 | − 5.3 ± 1.3 | 2.4 ± 2.5 | 1.0 ± 3.2 | − 11.9 ± 12.6 | 262.4 ± 89.0 | 0.9 ± 2.4 | 0.9 ± 2.3 |
| p value | < 0.001 | < 0.001 | < 0.001 | < 0.001 | < 0.001 | 0.151 | < 0.001 | < 0.001 | 0.076 | 0.120 |
| CT | CT | CT | CT | CT | CT | CT | CT | |||
| Mean difference ± standard deviation | − 6.2 ± 2.4 | − 5.3 ± 2.3 | − 10.5 ± 2.1 | 2.9 ± 2.1 | 0.4 ± 2.6 | − 9.8 ± 10.2 | 0.5 ± 2.1 | 1.2 ± 2.4 | ||
| p value | < 0.001 | < 0.001 | < 0.001 | < 0.001 | 0.490 | < 0.001 | 0.276 | 0.023 | ||
| 2nd metatarsal | ||||||||||
| Mean difference ± standard deviation | 4.7 ± 6.6 | 5.2 ± 2.3 | 2.1 ± 2.5 | − 4.8 ± 1.8 | 3.2 ± 1.7 | − 1.8 ± 1.7 | − 23.0 ± 15.0 | 264.0 ± 63.8 | 3.0 ± 1.4 | 0.2 ± 2.3 |
| p value | 0.005 | < 0.001 | 0.001 | < 0.001 | < 0.001 | < 0.001 | < 0.001 | < 0.001 | < 0.001 | 0.745 |
| CT | CT | CT | CT | CT | CT | CT | CT | |||
| Mean difference ± standard deviation | 5.3 ± 1.5 | 0.5 ± 1.9 | − 5.9 ± 2.3 | 3.8 ± 1.8 | − 2.7 ± 1.8 | − 21.1 ± 12.6 | 2.3 ± 1.3 | 0.1 ± 1.8 | ||
| p value | < 0.001 | 0.198 | < 0.001 | < 0.001 | < 0.001 | < 0.001 | < 0.001 | 0.761 | ||
Marked in italics are the non-significant results
Articular facet angulation
The facet angulation of the proximal radius and the proximal fibula differed significantly (p < 0.001), whereas the comparison of the facet angulation between the proximal radius and the second metatarsal base showed statistically similar values (p = 0.005).
Morphology of the head and articular facet
Looking at the metrics of the radial head, only the comparison of the maximum head diameter in CT evaluation of the proximal radius and the second base metatarsal provided statistically similar values (p = 0.198). The minimum head diameter of the proximal second metatarsal and both measures of the fibular head as well as the height of the head in all bones differed significantly (p values all < 0.001).
Comparing the articular facets of the three bones, statistically similar values were found in the maximum articular surface diameter (direct, p = 0.151; CT, p = 0.490) performing direct measurements as well as CT evaluation in the proximal radius and the proximal fibula. The minimum articular surface diameter as well as facet radius and proximal articular surface area differed significantly (p values all < 0.001). The comparison between proximal radial and proximal second metatarsal articular facet radius provided statistically significant differences (p < 0.001).
Morphology of the neck
Comparing the proximal radius and the proximal fibula, statistically similar values were found in the maximum (direct, p = 0.120; CT, p = 0.023) as well as minimum (direct, p = 0.076; CT, p = 0.276) measures of the neck diameter performing direct measurements as well as CT evaluation. Comparing the proximal radius and the proximal second metatarsal, statistically similar values were only found in the maximum neck diameter performing direct measurements (p = 0.745) as well as CT evaluation (p = 0.761).
Interrater evaluation
Interrater evaluation showed an excellent agreement between the two raters (0.989–0.997).
Discussion
The aim of this study was to compare anatomic details of the fibular head and second metatarsal as potential donors for an autologous radial head replacement. Introducing a vascularized autologous bone graft would be beneficial for the patients’ outcome as it would be an option to maximize usage of the elbow joint with minimal joint alteration. However, as the results of this study confirm (Table 3), no ideal autologous replacement of the proximal radius exist yet (21–23) and the challenge in this quest is rather to find a graft with the largest overlap in the most important morphometric parameters.
Table 3.
Summary of anatomic criteria met by investigated donor bones (minimum and maximum measures were summarized as one characteristic)
| Proximal fibula | Proximal second metatarsal | |
|---|---|---|
| Characteristics important for humeroradial articulation (4): articular surface diameter, articular surface area, facet radius, facet angulation | 1/4 | 1/4 |
| Characteristics important for radioulnar articulation (3): head diameter, head height, neck diameter | 1/3 | 2/3 |
| Summary | 2/7 | 3/7 |
Apart from the medial collateral ligament, the radius is a major restraint against valgus stress, and—together with the coronoid process—also stabilizes against posterior rotational instability, which is why especially in the adolescent population, the preservation of the biomechanical role of the radius plays a large role in the therapy of radial head/neck fractures. (13, 16, 17, 24–27). In cases of radial head degeneration after trauma, resection of the head or implant replacement are the most frequent reported options left. However, resection of the head of the radius leads to an isolated force distribution through the ulna, which then may lead to the development of progressive ulnohumeral osteoarthritis despite a good clinical outcome (12). This is most often combined with a proximal migration of the radius as well as increased ulnar variance, an associated proximal radioulnar impingement and valgus as well as posterior rotational instability associated with persistent overall elbow instability and redislocations of the joint. (12, 18, 28–31) But also implant replacement of the head of the radius is not a viable option as long-term survival of the implant—which is an important factor in treating adolescents—is short compared to other joints and complications, such as loosening, wear and associated capitellar cartilage erosion, occur often (32–35). Recently, Schnetzke et al. (8) reported a rather high complication and revision rate in patients with a monopolar radial head replacement with an implant survival rate of 75.1% at 18 years after surgery with the highest failure rate in first the postoperative year.
Proximal fibular bone graft
The fibula is a widely used bone for autologous grafts in defects throughout the body. Most often, the diaphyseal part is used and but in some cases, also proximal fibular bone grafts are described for repair of lateral malleolus or the distal radius (36, 37).
Our measurements revealed that, although in some parameters differences (neck diameter, articular surface diameter) were subjectively rather small (approx. 1 mm), the majority of values differed significantly between the radius and fibular head (>2.5 mm) (Table 2). This may be a first argument against a possible autologous transplantation of the fibular head replacing the resected radial head. Another point that may be detrimental for such a transplantation is the fact that the articular surface of the fibular head is not entirely covered by cartilage, thus leading to a large bone-cartilage interface especially between the fibular head and the radial notch of the proximal ulna. Additionally, the angulation of the articular surface is an important factor to include. The angle between the proximal articular surface of the radius and the longitudinal axis of the radius is approximately 90° (93.05±4.01), the angle between the proximal articular surface of the fibula and the longitudinal axis of the fibula is approximately 60° (61.07±8.65), thus showing a difference in angulation of 30° which leads to a biomechanically unstable interaction between the humerus and the radius with a potential of dislocation depending on the alignment of the graft. Furthermore, the variability of the angulation of the proximal articular facet of the fibula is quite high with possible angles between 10° and 90° (38).
Second metatarsal base bone graft
The second metatarsal bone has also been described as a free bone graft for a long time and especially the distal part has since been used as a graft for reconstruction of the temporomandibular joint, the mandible, the ulnar head, the lateral malleolus and the scaphoid bone (39–44). Less frequent, the metatarsal base has been used. Del Piñal et al. (45) have described a surgical technique for reconstruction of the distal radius using the base of the second or third metatarsal bone.
With regards to transplantation of the second metatarsal, the maximum neck diameter and articular surface angulation would be arguments for such an autologous surgical procedure. However, several other measurements do definitely provide not enough similarities to make this bone a clear, considerable option, although the differences, contrary to the proximal fibula, are rather small in comparison (see Table 2). Opposed to the fibular head, a lateral (intermetatarsal) articular surface of the second metatarsal exists. But it is not long and rounded enough to provide sufficient support for the proximal radioulnar articulation. Moreover, the shape of the second metatarsal base is rather different compared to the radius. Despite these largely statistically significant differences, this does not necessarily mean that an autologous transplantation is to be completely rejected because minor (significant) differences might still be clinically acceptable. In addition, the angulation of the proximal articular surface of the second metatarsal bone is similar to the angulation of the radius with less variability (see Table 1), leading to a possibly more stable articulation with the humeral capitulum.
Apart from morphometric disadvantages of the second metatarsal base bone graft, one has also to keep in mind some further disadvantages for its use, e.g., one creates an isolated iatrogenic Lisfranc ligament injury when harvesting the bone, which consequence remains unclear until now (46–48).
Graft choice for radial head replacement
A comparison of our data with the current literature and thus an objective graft recommendation is very difficult because only one article proposing an autologous bone graft for radial head replacement currently exists to the best of our knowledge. Han et al. (48) described an autologous second metatarsal transfer to the head of the radius in five patients. The authors used 1.5–2-cm non-vascularized second metatarsal base grafts to reconstruct the proximal radius. The presented postoperative results were good to excellent in the short-term (mean follow-up 44.8 months) with regards to elbow range-of-motion and donor site problems. The authors facilitated transplantation by shaving parts of the second metatarsal base to accommodate its placement into this joint area and imitate the shape of the head of the radius. However, objective parameters for graft selection are missing.
As described before, two important articulations have to be considered when replacing the proximal radius. In the humeroradial joint, the articular contact area and the lateral collateral ligament have to be taut similar to the anatomical situation to maximize valgus and posterior rotational stability of the elbow joint. Here, graft length, size of the proximal articular facet and facet angulation would be important parameters to consider. Graft length depends on the defect to repair and is easily facilitated through both the proximal fibula and the proximal second metatarsal. The size of the articular facet is in both the fibula and the second metatarsal significantly smaller than the facet of the radius and concerning the facet angulation, only the angulation of the second metatarsal bone is rather similar to the radius (see Table 2). In the proximal radioulnar joint, the size of the head and neck play an important role in the interaction with the annular ligament and the radial notch of the ulna to facilitate pronation-supination movement. Here, the head or base size, respectively, of the proposed grafts do not seem to meet the criteria but the size of both necks seem to. However, both grafts are no perfect fit for the radial notch of the ulna (see Table 3).
To compensate for the suboptimal fit of the second metatarsal base to the radial notch of the ulna, an additional anconeus interposition (49), Achilles tendon interposition (9) or corium interposition (11) could be options to optimize the radioulnar movability. Alternatively, a costochondral graft has been described especially for bone replacement in children. It could have benefits due to its compatibility, applicableness, processability and rarity of additional functional damage to the patient. Additionally, this graft has shown the potential of growth, which is of additional advantage in children. However, this growth potential may also be of disadvantage as unpredictable growth with graft hyperplasia has been reported (50).
Some limitations have to be taken into account when interpreting the presented results. As common in anatomical studies, the age of the body donors is significantly higher than the intended target group. As the morphometric comparison was applied between the different characteristics of bones of the same donors, the significance of differences is still applicable, although the exact size is not. Also, some parameters may be influenced by high anatomical variability, which would need a larger number of specimens to meet the necessary sample size. Nonetheless, the presented results reflect a viable cross section sufficient to answer the initial research query. Further limitations may be attributed to the clinical applicability. As pediatric radial head fractures may lead to post-traumatic axial deviation, replacement of the radial head with an autologous graft may therefore only be considered in selected cases. It must also be considered whether one accepts the donor morbidity for a questionable gain in function at the elbow.
In summary, neither the proximal fibula nor the base of the second metatarsal are an ideal bone graft for replacement of the head of the radius. Both bones do have several unsuitable parameters, while the remainder is somewhat moderately useful. While the fibular head does exhibit increased absolute values compared to the proximal radius, those of the second metatarsal are overall smaller than the head of the radius. Considering our predefined, most relevant anatomic criteria, the base of the second metatarsal might be a bit more suitable as a potential donor, especially as the angulation of the proximal articular facet is similar to the angulation of the radius. To gain further knowledge into this topic, biomechanical studies are needed.
Acknowledgements
The authors acknowledge the human donors of this study and are grateful for their commitment to medicine and anatomic science. The authors of this study have no conflicts of interest to declare.
Author contributions
SF conceived the study and wrote the first draft of the manuscript; JA collected the data and performed statistical analysis. LH performed statistical analysis and wrote and edited the manuscript. SR and MF collected the data. All authors approved the final manuscript.
Funding
Open access funding provided by Medical University of Vienna.
Declarations
Conflict of interest
All authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical standards
This retrospective study involving human cadavers was in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Nicholson LT, Skaggs DL. Proximal radius fractures in children. J Am Acad Orthop Surg. 2019;27:e876–e886. doi: 10.5435/JAAOS-D-18-00204. [DOI] [PubMed] [Google Scholar]
- 2.Kang S, Park SS. Predisposing effect of elbow alignment on the elbow fracture type in children. J Orthop Trauma. 2015;29:e253–258. doi: 10.1097/BOT.0000000000000322. [DOI] [PubMed] [Google Scholar]
- 3.Ekdahl M, Baar A, Larrain C, et al. Severe joint cartilage degeneration after minimally displaced fracture of proximal radius in children: a report of 2 cases. JSES Int. 2020;4:1006–1010. doi: 10.1016/j.jseint.2020.08.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dietzel M, Scherer S, Esser M, et al. Fractures of the proximal radius in children: management and results of 100 consecutive cases. Arch Orthop Trauma Surg. 2021 doi: 10.1007/s00402-021-03917-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Qiao F, Jiang F. Closed reduction of severely displaced radial neck fractures in children. BMC Musculoskelet Disord. 2019;20:567. doi: 10.1186/s12891-019-2947-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Van Zeeland NL, Bae DS, Goldfarb CA. Intra-articular radial head fracture in the skeletally immature patient: progressive radial head subluxation and rapid radiocapitellar degeneration. J Pediatr Orthop. 2011;31:124–129. doi: 10.1097/BPO.0b013e31820742de. [DOI] [PubMed] [Google Scholar]
- 7.Falciglia F, Giordano M, Aulisa AG, et al. Radial neck fractures in children: results when open reduction is indicated. J Pediatr Orthop. 2014;34:756–762. doi: 10.1097/BPO.0000000000000299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Schnetzke M, Jung MK, Groetzner-Schmidt C, et al. Long-term outcome and survival rate of monopolar radial head replacement. J Shoulder Elbow Surg. 2021;30:e361–e369. doi: 10.1016/j.jse.2020.11.031. [DOI] [PubMed] [Google Scholar]
- 9.Factor S, Rotman D, Pritsch T, et al. Radial head excision and Achilles allograft interposition arthroplasty for the treatment of chronic pediatric radiocapitellar pathologies: a report of four cases. Shoulder Elbow. 2021;13:213–222. doi: 10.1177/1758573219897859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wegmann H, Heider S, Novak M, et al. Outcome following excision of the radial head in children with open physes for impaired elbow motion. J Shoulder Elbow Surg. 2019;28:525–529. doi: 10.1016/j.jse.2018.08.045. [DOI] [PubMed] [Google Scholar]
- 11.Farr S, Abualruz MM, Girsch W. Radial head resection and corium interposition arthroplasty in skeletally immature patients with isolated posttraumatic radial head arthrosis. Tech Hand Up Extrem Surg. 2020;24:37–42. doi: 10.1097/BTH.0000000000000259. [DOI] [PubMed] [Google Scholar]
- 12.Antuna SA, Sanchez-Marquez JM, Barco R. Long-term results of radial head resection following isolated radial head fractures in patients younger than forty years old. J Bone Joint Surg Am. 2010;92:558–566. doi: 10.2106/JBJS.I.00332. [DOI] [PubMed] [Google Scholar]
- 13.Smith AM, Urbanosky LR, Castle JA, et al. Radius pull test: predictor of longitudinal forearm instability. J Bone Joint Surg Am. 2002;84:1970–1976. doi: 10.2106/00004623-200211000-00010. [DOI] [PubMed] [Google Scholar]
- 14.Nunez FA, Jr, Evans PJ. Chronic longitudinal instability of the forearm treated with a combination of ulnar shortening osteotomy, pronator teres transfer, and tightrope technique. Tech Hand Up Extrem Surg. 2018;22:99–103. doi: 10.1097/BTH.0000000000000200. [DOI] [PubMed] [Google Scholar]
- 15.Hotchkiss RN, Weiland AJ. Valgus stability of the elbow. J Orthop Res. 1987;5:372–377. doi: 10.1002/jor.1100050309. [DOI] [PubMed] [Google Scholar]
- 16.Morrey BF, An KN, Stormont TJ. Force transmission through the radial head. J Bone Joint Surg Am. 1988;70:250–256. doi: 10.2106/00004623-198870020-00014. [DOI] [PubMed] [Google Scholar]
- 17.Beingessner DM, Dunning CE, Gordon KD, et al. The effect of radial head excision and arthroplasty on elbow kinematics and stability. J Bone Joint Surg Am. 2004;86:1730–1739. doi: 10.2106/00004623-200408000-00018. [DOI] [PubMed] [Google Scholar]
- 18.Ring D, Jupiter JB, Zilberfarb J. Posterior dislocation of the elbow with fractures of the radial head and coronoid. J Bone Joint Surg Am. 2002;84:547–551. doi: 10.2106/00004623-200204000-00006. [DOI] [PubMed] [Google Scholar]
- 19.Cohen J. Set correlation and contingency tables. Appl Psychol Meas. 1988;12:425–434. doi: 10.1177/014662168801200410. [DOI] [Google Scholar]
- 20.Cicchetti DV. Guidelines, criteria and rules of thumb for evaluating normed and standardized assessment instruments in psychology. Psychol Assessment. 1994;6:284–290. doi: 10.1037/1040-3590.6.4.284. [DOI] [Google Scholar]
- 21.van Riet RP, Van Glabbeek F, Neale PG, et al. The noncircular shape of the radial head. J Hand Surg. 2003;28:972–978. doi: 10.1016/S0363-5023(03)00426-X. [DOI] [PubMed] [Google Scholar]
- 22.King GJ, Zarzour ZD, Patterson SD, et al. An anthropometric study of the radial head: implications in the design of a prosthesis. J Arthroplasty. 2001;16:112–116. doi: 10.1054/arth.2001.16499. [DOI] [PubMed] [Google Scholar]
- 23.Beredjiklian PK, Nalbantoglu U, Potter HG, et al. Prosthetic radial head components and proximal radial morphology: a mismatch. J Shoulder Elbow Surg. 1999;8:471–475. doi: 10.1016/S1058-2746(99)90079-4. [DOI] [PubMed] [Google Scholar]
- 24.Morrey BF, An KN. Articular and ligamentous contributions to the stability of the elbow joint. Am J Sports Med. 1983;11:315–319. doi: 10.1177/036354658301100506. [DOI] [PubMed] [Google Scholar]
- 25.Damiani M, King GJ. Coronoid and radial head reconstruction in chronic posttraumatic elbow subluxation. Instr Course Lect. 2009;58:481–493. [PubMed] [Google Scholar]
- 26.Burkhart KJ, Gruszka D, Frohn S, et al. Locking plate osteosynthesis of the radial head fractures: clinical and radiological results. Unfallchirurg. 2015;118:949–956. doi: 10.1007/s00113-014-2562-y. [DOI] [PubMed] [Google Scholar]
- 27.Businger A, Ruedi TP, Sommer C. On-table reconstruction of comminuted fractures of the radial head. Injury. 2010;41:583–588. doi: 10.1016/j.injury.2009.10.026. [DOI] [PubMed] [Google Scholar]
- 28.Mikic ZD, Vukadinovic SM. Late results in fractures of the radial head treated by excision. Clin Orthop Relat Res. 1983;181:220–228. doi: 10.1097/00003086-198312000-00034. [DOI] [PubMed] [Google Scholar]
- 29.Heim U. Combined fractures of the radius and the ulna at the elbow level in the adult. Analysis of 120 cases after more than 1 year. Rev Chir Orthop Reparatrice Appar Mot. 1998;84:142–153. [PubMed] [Google Scholar]
- 30.Josefsson PO, Gentz CF, Johnell O, et al. Dislocations of the elbow and intraarticular fractures. Clin Orthop Relat Res. 1989;246:126–130. doi: 10.1097/00003086-198909000-00020. [DOI] [PubMed] [Google Scholar]
- 31.Johnson JA, Beingessner DM, Gordon KD, et al. Kinematics and stability of the fractured and implant-reconstructed radial head. J Shoulder Elbow Surg. 2005;14:195S–201S. doi: 10.1016/j.jse.2004.09.034. [DOI] [PubMed] [Google Scholar]
- 32.Harrington IJ, Sekyi-Otu A, Barrington TW, et al. The functional outcome with metallic radial head implants in the treatment of unstable elbow fractures: a long-term review. J Trauma. 2001;50:46–52. doi: 10.1097/00005373-200101000-00009. [DOI] [PubMed] [Google Scholar]
- 33.Moro JK, Werier J, MacDermid JC, et al. Arthroplasty with a metal radial head for unreconstructible fractures of the radial head. J Bone Joint Surg Am. 2001;83:1201–1211. doi: 10.2106/00004623-200108000-00010. [DOI] [PubMed] [Google Scholar]
- 34.Van Riet RP, Van Glabbeek F, Verborgt O, et al. Capitellar erosion caused by a metal radial head prosthesis. A case report. J Bone Joint Surg Am Vol. 2004;86:1061–1064. doi: 10.2106/00004623-200405000-00028. [DOI] [PubMed] [Google Scholar]
- 35.Popovic N, Lemaire R, Georis P, et al. Midterm results with a bipolar radial head prosthesis: radiographic evidence of loosening at the bone-cement interface. J Bone Joint Surg Am. 2007;89:2469–2476. doi: 10.2106/JBJS.F.00723. [DOI] [PubMed] [Google Scholar]
- 36.Noaman H, Sorour Y, Marzouk A. Wrist arthroplasty for treatment of infected distal radius nonunion using free vascularised proximal fibular bone graft. Injury. 2020 doi: 10.1016/j.injury.2020.11.021. [DOI] [PubMed] [Google Scholar]
- 37.Herring CL, Jr, Hall RL, Goldner JL. Replacement of the lateral malleolus of the ankle joint with a reversed proximal fibular bone graft. Foot Ankle Int. 1997;18:317–323. doi: 10.1177/107110079701800601. [DOI] [PubMed] [Google Scholar]
- 38.Ogden JA (1974) The anatomy and function of the proximal tibiofibular joint. Clin Orthop Relat Res 186–191 [PubMed]
- 39.Bond SE, Saeed NR, Cussons PD, et al. Reconstruction of the temporomandibular joint by the transfer of the free vascularised second metatarsal. Br J Oral Maxillofac Surg. 2004;42:241–245. doi: 10.1016/j.bjoms.2004.01.008. [DOI] [PubMed] [Google Scholar]
- 40.Cavadas PC, Thione A. Skeletal reconstruction of the forearm with a double-barrel fibular flap and second metatarsal head. Injury. 2014;45:2106–2108. doi: 10.1016/j.injury.2014.10.014. [DOI] [PubMed] [Google Scholar]
- 41.Chen H, Yin G, Hou C, et al. Repair of a lateral malleolus defect with a composite pedicled second metatarsal flap. J Int Med Res. 2018;46:5291–5296. doi: 10.1177/0300060518801461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.del Piñal F, Guerrero-Navarro ML, Studer A, et al. Reconstruction of the ulnar head with a vascularized second metatarsal head: case report. J Hand Surg. 2012;37:1568–1573. doi: 10.1016/j.jhsa.2012.04.039. [DOI] [PubMed] [Google Scholar]
- 43.Hu W, Trimaille A, De Vries P, et al. Paediatric distal fibula reconstruction using a pedicled composite second metatarsal flap: a technical note. Injury. 2015;46:2055–2058. doi: 10.1016/j.injury.2015.07.011. [DOI] [PubMed] [Google Scholar]
- 44.Larkins CG, Tannan SC, Burkett AE, et al. Autologous osteoligamentous reconstruction of scaphoid proximal pole with metatarsal head and collateral ligament: cadaver anatomic description of novel surgical technique. Hand (N Y) 2020 doi: 10.1177/1558944719895616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.del Piñal F, Klausmeyer M, Moraleda E, et al. Vascularized graft from the metatarsal base for reconstructing major osteochondral distal radius defects. J Hand Surg. 2013;38:1883–1895. doi: 10.1016/j.jhsa.2013.07.013. [DOI] [PubMed] [Google Scholar]
- 46.Nunley JA, Vertullo CJ. Classification, investigation, and management of midfoot sprains: Lisfranc injuries in the athlete. Am J Sports Med. 2002;30:871–878. doi: 10.1177/03635465020300061901. [DOI] [PubMed] [Google Scholar]
- 47.Lee CA, Birkedal JP, Dickerson EA, et al. Stabilization of Lisfranc joint injuries: a biomechanical study. Foot Ankle Int. 2004;25:365–370. doi: 10.1177/107110070402500515. [DOI] [PubMed] [Google Scholar]
- 48.Han KJ, Oh KS, Chung NS, et al. Radial head arthroplasty using a metatarsal osteochondral autograft. Int Orthop. 2012;36:2501–2506. doi: 10.1007/s00264-012-1666-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Morrey BF, Schneeberger AG. Anconeus arthroplasty: a new technique for reconstruction of the radiocapitellar and/or proximal radioulnar joint. J Bone Joint Surg Am. 2002;84:1960–1969. doi: 10.2106/00004623-200211000-00009. [DOI] [PubMed] [Google Scholar]
- 50.Link JO, Hoffman DC, Laskin DM. Hyperplasia of a costochondral graft in an adult. J Oral Maxillofac Surg. 1993;51:1392–1394. doi: 10.1016/S0278-2391(10)80148-X. [DOI] [PubMed] [Google Scholar]