Abstract
Various reports of neurological manifestations of SARS-COV-2 infection after the virus outbreak are available, including anosmia, seizures, acute flaccid myelitis, Guillain-Barré syndrome (GBS), and encephalitis. Most of the literature has focused on the respiratory manifestation of SARS-CoV-2 infection in adults, but recent evidence showed that it is not confined to the respiratory tract. This report is about a rare variant of GBS acute motor axonal neuropathy (AMAN) in a child due to COVID-19 infection
An 11 years old boy was referred to the hospital with a history of three-day lasting mild fever, and gastroenteritis, two weeks before starting symptoms. He was presented with progressive ascending weakness, paresthesia, and areflexia in four limbs four days ago. Nasopharyngeal swab polymerase chain reaction (PCR) was positive for SARS-CoV-2. The electrodiagnostic finding was compatible with acute generalized axonal motor neuropathy, and imaging revealed thoracolumbar syrinx and nerve root enhancement in lumbosacral MRI. Other lab tests were normal.
GBS and its variant are one of the manifestations of SARS-CoV-2 in children. Children with an unexplained neurological process should be tested for SARS-CoV-2.
Key Words: Guillain-Barré syndrome (GBS), Acute Axonal Motor Neuropathy (AMAN), COVID-19, Children
Introduction
Multiple reports announced details of symptoms and outcomes of severe acute respiratory syndrome coronavirus 2 (SARS‑CoV‑2) infection with a primary focus on respiratory complications since the outbreak of this virus. However, reports point out this virus’s effect on various organs, including the nervous system [1].
As demonstrated, the virus can infect several organ systems, including the nervous, renal, pulmonary, and cardiovascular systems. Infection of the nervous system with SARS-CoV-2 may develop Guillain– Barre´ syndrome (GBS) [2].
GBS refers to a group of immunological disorders, often characterized by acute or subacute weakness in limbs or cranial nerve innervated muscles [3].
Epstein–Barr virus, Campylobacter jejune, Cytomegalovirus, Influenza A virus, Hemophilus influenza, and Mycoplasma pneumoniae are the most common microorganisms involved in developing axonal and demyelinating subtypes of GBS. Previously discovered types of coronaviruses, SARS-COV, Middle East respiratory syndrome (MERS) and Zika virus have been announced as associative viruses with GBS as well [4].
There is a belief that coronaviruses can cause GBS in certain patients, either directly through neuro-invasive capacity angiotensin-converting enzyme 2 (ACE2) receptors on neuronal tissues) or indirectly through the response of the immune system (an inflammatory mechanism) [5].
The beginning of GBS is with numbness in the lower limbs and weakness in the same distribution, and this process often develops over days. The symptoms can progress rapidly, particularly weakness, resulting in quadriplegia within a few days. Approximately 50% of patients achieve maximum weakness by two weeks, 80% by three weeks, and 90% by four weeks [6].
Three main subtypes of GBS are acute inflammatory demyelinating polyneuropathy (AIDP), acute motor axonal neuropathy (AMAN), and acute motor sensory axonal neuropathy (AMSAN), according to electrophysiological features. The most common manifestation of COVID-19-associated GBS has features of classic post-infectious GBS (AIDP subtype), and possibly they have the same immune-mediated pathogenetic mechanisms [7].
Among GBS subtypes, AMAN diagnosis is based on the decreased amplitude of compound muscle action potentials (CMAPs) with no evidence of demyelination [8].
Here is a report of a case of AMAN type of GBS associated with COVID-19.
Case report
An 11 years old boy was presented complaining of generalized ascending muscle weakness for four days before referring to the hospital. There were no complaints of paresthesia and vertigo, dysphonia and dysphagia, bowel and bladder dysfunction, blurred vision, or diplopia.
He had a history of nausea, vomiting, and diarrhea two weeks ago. At that time, his parent had upper respiratory tracks infection, and after a survey, their COVID-polymerase chain reaction (PCR) test was positive.
In an examination, the patient was alert and conscious. His vital signs were normal (blood pressure (BP): 110/80 mmHg/pulse rate (PR): 81 beats per minute / body temperature (BT): 37.1°C / respiratory rate (RR): 15 breaths per minute / Oxygen saturation (O2Sat):97%), with no orthostatic hypotension. Cranial nerves were normal, and muscle limbs strength was 1/5 in distal and 3/5 in proximal lower limbs and 4/5 in distal and 5/5 in proximal upper limbs. The deep tendon reflex was 2+ in the upper limbs and 1+ in the knee, and Achill’s reflex was absent. His gag was normal. The sensation was intact to light touch.
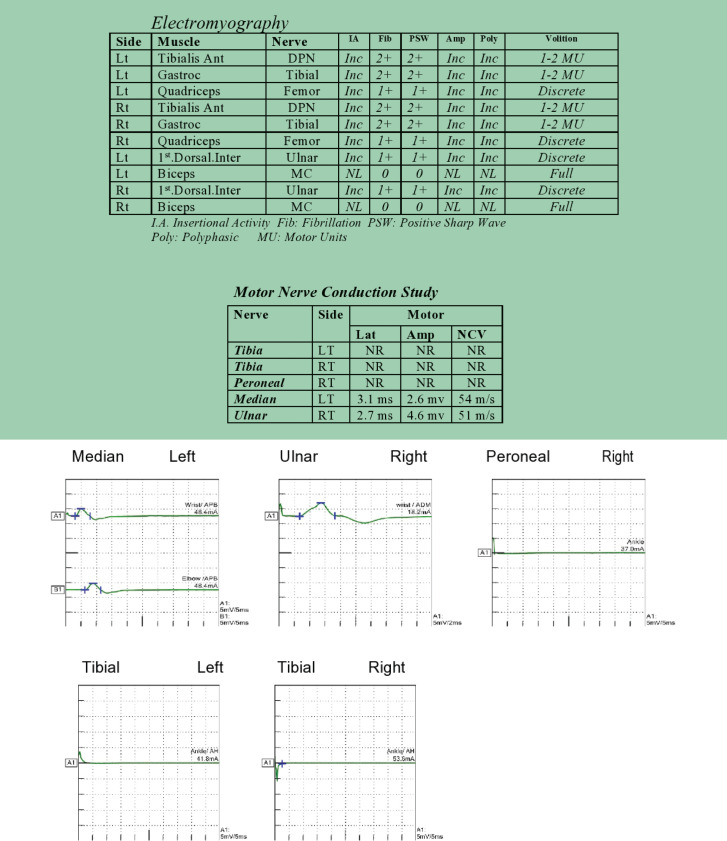
The patient was admitted to the hospital and treated with IVIG. First Electromyography - Nerve Conduction Study (EMG-NCS) was performed five days after the onset of weakness. All sensory tests were normal. In motor tests, the authors observed a decrease in the amplitude of bilateral deep peroneal nerve (DPN) and tibial CMAPs and partial conduction block in DPN CMAP (ankle–fibular stimulation). All distal motor latencies and F waves were normal in the upper limbs and absent in the lower limbs. During needle EMG, neurogenic motor unit action potential (MUAPs) [i.e., high amplitude and polyphasic MUAPs] were seen without significant spontaneous activity.
The second EMG-NCS was performed on the 40th day of the onset of weakness. All sensory tests were normal, and lower limb CMAPs and F waves were absent. The amplitude of upper limb CMAPs was decreased with normal distal motor latency. Upper F waves were latent (min: 28.5 – max: 33) (Figure 1). In needle EMG, the authors observed neurogenic findings in upper and lower limbs; Neurogenic MUAP and decreased recruitment (1-2 MUAP in Lower limbs) and positive sharp waves and fibrillation, more prominent in lower limbs (distal >proximal). These findings are compatible with acute generalized motor axonal neuropathy (AMAN) with evidence of active axonal loss without sign of significant regeneration.
Figure 1.
Detailed second EDX study of our patient
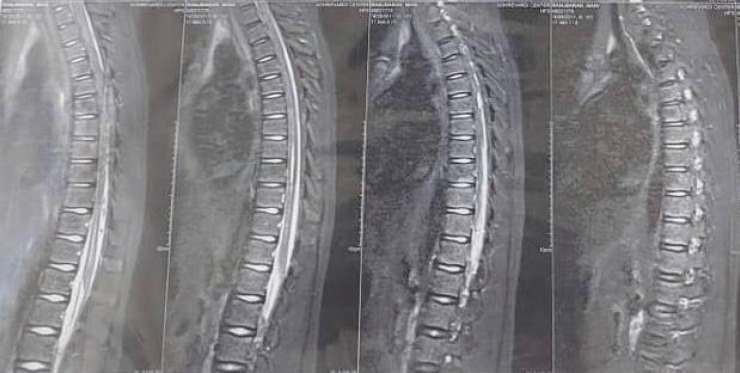
Because of the presence of an upward right plantar reflex (right Babinski sign) in the second visit, Magnetic resonance imaging (MRI) of the brain and cervical and thoracolumbosacral spine were done (after the second EMG-NCS and AMAN diagnosis). They revealed thoracolumbar syrinx and nerve root enhancement in the lumbosacral region (Figure 2).
Figure 2.
Thoracolumbosacral MRI – sagittal view
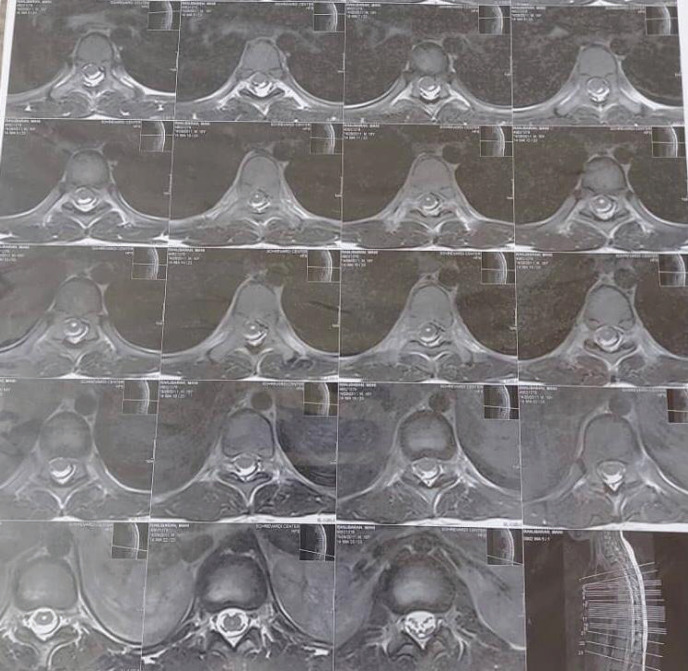
Evaluation for a trigger of the GBS was done. Blood, urine, and stool cultures were negative. The patient’s SARS-CoV-2 nucleic acid amplification was positive. (Figure 3).
Figure 3.
Thoracic MRI – axial view
Other lab tests: White blood cell (WBCs): 4500 per microliter; Hemoglobin (HB): 11.5 gm/dL; Mean corpuscular volume (MCV): 84 fl; platelet (PLT): 249000 per microliter; Erythrocyte Sedimentation Rate (ESR): 8 mm/hr; C-reactive protein (CRP): 0.3 mg/L; Alkaline Phosphatase (ALP): 498 IU/L; Creatine phosphokinase (CPK): 111 mcg/L); Electrolytes, hepatic function panel, fibrinogen, and ferritin were normal. His chest radiographs patchy bilateral areas of consolidation, nodular opacities, bronchial wall thickening, and small pleural effusions, compatible with a viral lower respiratory tract infection. (Figure 4).
Figure 4.
Lumbosacral MRI – axial view
Due to the early onset of IVIG treatment and low diagnostic value in the first week, and observation of patient improvement after treatment with IVIG (improved muscle strength and initiation of walking), lumbar puncture (LP) was not performed.
Discussion
In the recent case reports of COVID-19, there was evidence of the association between COVID-19 and GBS. They have described adults with a broad spectrum of GBS variants, including demyelinating, axonal, and Miller-Fisher, in connection with COVID-19 [9].
A recently published systematic review on neurological manifestations in patients with COVID-19 has shown data ranging from common, non-specific symptoms to more complex and life-threatening conditions, such as cerebrovascular diseases and encephalopathies [10].
In 19 reports, variants of GBS associated with SARS-CoV-2 are demonstrated in one presenting five cases from Italy. The first reported child case of SARS-CoV-2 infection associated with the AMAN variant of GBS was by Frank et al. [2]. In this study, the authors reported a 15-year-old male patient presenting with ascending lower limb weakness and pain, spreading to upper limbs. Antibody test and PCR were positive for SARS-CoV-2. Other lab tests, including blood and cerebral spinal fluid (CSF) analysis, were normal. EMG-NCS findings were compatible with the AMAN variant of GBS. Another report was described by Krishnakumar et al. about an adolescent male presented with progressive proximal weakness of the bilateral lower limbs without bladder or bowel involvement with the diagnosis of AMAN [11]. In another case reported by Nihal Akçay [12], a six‐year‐old male was presented with progressive ascending symmetric paralysis over a four‐day course and two days of fever (positive PCR for COVID-19). He had severe weakness in the respiratory muscles, requiring invasive mechanical ventilation, severe bilateral lower and upper limb flaccid weakness (with a manual muscle test of 1/5), and absent deep tendon reflexes. CSF showed high protein without pleocytosis. The EMG-NCS was suggestive of AMAN (GBS associated with SARS‐CoV‐2 infection).
In this case, an 11 years old boy with ascending paralysis two weeks after gastroenteritis and positive PCR for COVID-19 with evidence of AMAN due to COVID-19 is presented. The detailed comparison of the findings of this patient with other studies describing the AMAN variant of GBS following SARS-COV-2 infection is demonstrated in Table 1.
Despite the evidence of mild lower respiratory tract infection in CXR, the patient indicated a chest CT scan. However, it was not done due to the lack of any respiratory symptoms and normal O2 saturation in the examination.
Our patient received 2g/kg intravenous immunoglobulin over three days and responded well. Antiviral treatment was not performed due to the passage of time, more than three weeks after the onset of symptoms, and lack of any respiratory symptoms.
There is controversy about the starting of corticosteroids. If the patient does not respond well to IVIG, corticosteroids should be started or started at first because of an underlying disease.
Due to the lack of vaccination in children, they are exposed to this disease and its complication. It is vital to have a protocol for treatment and determining the role of the drug regime in GBS and its variant following SARS-CoV-2. More than 70% of patients showed a good prognosis, mainly after treatment with intravenous immunoglobulin. Following previous findings regarding classic GBS and COVID-19, patients with significantly older ages have less favorable outcomes [7]. Children with AMAN seem to have higher short-term morbidity, more severe clinical course, and slower recovery than those with AIDP [13].
Children with an unexplained neurological process should be tested for SARS-CoV-2. Because of the poor sensitivity of the SARS-CoV-2 PCR testing, repeating PCR or antibody testing may also be required. If COVID-19 is not diagnosed, there may be a missed opportunity for antiviral treatment, and there will be an increased risk of exposure and infection for hospital staff [9].
Authors’ contribution
Alireza Nateghian and Katayoun Moradi contributed to the design of the study. Mozhde Mohammadpour collected the data, followed the patient, and contributed to writing the manuscript’s content. Naseh Yousefi and Mohammad Sadegh Khabbaz reviewed the manuscript. Alireza Nateghian, Katayoun Moradi, and Mozhde Mohammadpour drafted the final manuscript. All authors read and approved the final version of the manuscript.
Conflict of interest
No conflicts are declared.
Acknowledgment
This study did not receive any financial support.
References
- 1.Whittaker A M. Neurological manifestations of COVID‐19: a systematic review and current update. . Acta Neurologica Scandinavica. 2020;142(1):14–22. doi: 10.1111/ane.13266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Frank C.H.M, et al. Guillain–Barré syndrome associated with SARS-CoV-2 infection in a pediatric patient. Journal of tropical pediatrics, 2020 doi: 10.1093/tropej/fmaa044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lowery, M.M, et al. Atypical variant of Guillain Barre syndrome in a patient with COVID-19. The Journal of Critical Care Medicine. 2020:6–4. doi: 10.2478/jccm-2020-0038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Willison, H.J., B.C. Guillain-barre syndrome. The Lancet. 2016;388(10045):717–727. doi: 10.1016/S0140-6736(16)00339-1. [DOI] [PubMed] [Google Scholar]
- 5.Rahimi K. Guillain-Barre syndrome during COVID-19 pandemic: an overview of the reports. Neurological Sciences. 2020 :1–8. doi: 10.1007/s10072-020-04693-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Donofrio, P.D. Guillain-Barré Syndrome. CONTINUUM: Lifelong Learning in Neurology. 2017;23(5): 1295–1309. doi: 10.1212/CON.0000000000000513. [DOI] [PubMed] [Google Scholar]
- 7.Abu-Rumeileh, S, et al. Guillain–Barré syndrome spectrum associated with COVID-19: an up-to-date systematic review of 73 cases. Journal of neurology. 2020:1–38. doi: 10.1007/s00415-020-10124-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Petrelli, C, et al. Acute Motor Axonal Neuropathy Related to COVID-19 Infection: A New Diagnostic Overview. Journal of Clinical Neuromuscular Disease. 2020;22(2):120–121. doi: 10.1097/CND.0000000000000322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Curtis, M, et al. Guillain-Barré syndrome in a child with COVID-19 infection. Pediatrics. 2021;147(4) doi: 10.1542/peds.2020-015115. [DOI] [PubMed] [Google Scholar]
- 10.Munhoz, R.P, et al. Neurological complications in patients with SARS-CoV-2 infection: a systematic review. Arquivos de Neuro-Psiquiatria. 2020;78(5):290–300. doi: 10.1590/0004-282x20200051. [DOI] [PubMed] [Google Scholar]
- 11.Krishnakumar, A, et al. Guillain–Barré Syndrome with Preserved Reflexes in a Child after COVID-19 Infection. Indian Journal of Pediatrics. 2021:1–2. doi: 10.1007/s12098-021-03792-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gupta, P.K., et al. How different is AMAN from AIDP in childhood GBS? A prospective study from North India. The Indian Journal of Pediatrics. 2019;86(4):329–334. doi: 10.1007/s12098-018-2835-5. [DOI] [PubMed] [Google Scholar]