Abstract
Background
Sweetened beverage intake may play a role in non-alcoholic fatty liver disease (NAFLD) development, but scientific evidence on their role is limited. This study examined associations between sugar-sweetened beverages (SSB), low/no-calorie beverages (LNCB) and fruit juice (FJ) intakes and NAFLD in four European studies.
Methods
Data for 42,024 participants of Lifelines Cohort, NQPlus, PREDIMED-Plus and Alpha Omega Cohort were cross-sectionally analysed. NAFLD was assessed using Fatty Liver Index (FLI) (≥60). Restricted cubic spline analyses were used to visualize dose–response associations in Lifelines Cohort. Cox proportional hazard regression analyses with robust variance were performed for associations in individual cohorts; data were pooled using random effects meta-analysis. Models were adjusted for demographic, lifestyle, and other dietary factors.
Results
Each additional serving of SSB per day was associated with a 7% higher FLI-defined NAFLD prevalence (95%CI 1.03–1.11). For LNCB, restricted cubic spline analysis showed a nonlinear association with FLI-defined NAFLD, with the association getting stronger when consuming ≤1 serving/day and levelling off at higher intake levels. Pooled Cox analysis showed that intake of >2 LNCB servings/week was positively associated with FLI-defined NAFLD (PR 1.38, 95% CI 1.15–1.61; reference: non-consumers). An inverse association was observed for FJ intake of ≤2 servings/week (PR 0.92, 95% CI: 0.88–0.97; reference: non-consumers), but not at higher intake levels. Theoretical replacement of SSB with FJ showed no significant association with FLI-defined NAFLD prevalence (PR 0.97, 95% CI 0.95–1.00), whereas an adverse association was observed when SSB was replaced with LNCB (PR 1.12, 95% CI 1.03–1.21).
Conclusions
Pooling results of this study showed that SSB and LNCB were positively associated with FLI-defined NAFLD prevalence. Theoretical replacement of SSB with LNCB was associated with higher FLI-defined NAFLD prevalence. An inverse association was observed between moderate intake of FJ and FLI-defined NAFLD. Our results should be interpreted with caution as reverse causality cannot be ruled out.
Subject terms: Metabolic syndrome, Diabetes
Introduction
Non-alcoholic fatty liver disease (NAFLD) is a general term that refers to a broad range of liver disorders, including benign macrovesicular hepatic steatosis (>5% of hepatocytes), non-alcoholic steatohepatitis, hepatic fibrosis, and liver cirrhosis, which constitutes the third most common cause of hepatocellular carcinoma [1]. Globally, NAFLD prevalence is estimated to be around 25%, which has steadily increased and currently coincides with obesity, type 2 diabetes, and metabolic syndrome epidemic [2].
Major determinants of NAFLD include sedentary lifestyles and poor-quality diets, namely high sugar intake [3–5]. A recent meta-analysis of 12 studies (nine cross-sectional, two case-control and one cohort study) showed that sugar-sweetened beverages (SSB) intake was associated with a 39% (95% CI 1.29–1.50) higher NAFLD risk [6]. However, studies on the association between SSB intake and NAFLD were mainly conducted in relatively small studies using liver imaging techniques or biopsies to define NAFLD, which are often not feasible in large population studies [6, 7]. Surrogate formulas—employing serum biomarkers—have been developed, validated and widely used in recent decades, including the Fatty Liver Index (FLI) by Bedogni et al. [8–10], to facilitate non-invasive and simple NAFLD assessment in larger studies. However, evidence on the association between SSB and NAFLD detected with FLI in larger studies is still limited [3, 11, 12].
Low/no-calorie sweeteners have been increasingly used as an alternative to sugar. However, evidence on the association between low/no calorie beverages (LNCB) and NAFLD is limited and generally shows no association [7, 13, 14]. Besides LNCB, fruit juice (FJ) is also often considered an alternative to SSB, although some argue that FJ contains high amounts of sugar apart from its vitamin and polyphenol content. Population-based studies showed beneficial associations with cardiometabolic health at moderate doses of FJ consumption [15–17], but evidence specifically on NAFLD is lacking.
Given all the aforementioned considerations, we aimed to investigate the association between SSB, LNCB, and FJ intakes and FLI-defined NAFLD in four European studies. Additionally, we investigated whether LNCB and FJ could be used as an alternative to SSB by means of substitution analyses which is currently lacking in the literature.
Methods
Study design and population
The SWEET project is a European Union-funded project that aims to investigate public health and safety, obesity, and sustainability risks and benefits of replacing sugar with sweeteners and sweetness enhancers (www.sweetproject.eu). The results presented in this manuscript are based on harmonized data of the Lifelines Cohort study (Lifelines; The Netherlands), the Nutrition Questionnaire Plus study (NQPlus; The Netherlands), the PREDIMED-Plus study (PREDIMED-Plus; Spain), and the Alpha Omega Cohort (AOC; The Netherlands). All participants in all cohorts gave written informed consent before participating.
Lifelines
Lifelines is a multidisciplinary prospective population-based cohort study examining in a unique three-generation design the health and health-related behaviours of 167,729 persons living in the North of The Netherlands [18, 19]. It employs a broad range of investigative procedures in assessing biomedical, sociodemographic, behavioural, physical and psychological factors that contribute to the health and disease of the general population, with a special focus on multi-morbidity and complex genetics. Between 2006 and 2013, participants aged 0–93 yr were recruited to undergo baseline measurements. For present analysis, data for 152,728 participants aged ≥18 yr were included. After consecutive exclusion of those with missing dietary data (n = 8633), implausible energy intake (<800 or >4000 kcal/day for men or <500 or >3500 kcal/day for women; n = 15,483) [20], missing FLI data (n = 86,137), history of hepatitis (n = 31), excessive alcohol consumption (>20 g/day for women and >30 g/day for men; n = 1825), or missing covariates (n = 3679), n = 36,940 remained for current analyses (Supplemental Table 1). Lifelines has been approved by the Medical Ethical Review Committee of the University Medical Center in Groningen.
NQPlus
NQPlus is a prospective cohort study involving mainly Caucasian Dutch adults aged 20–70 yr from the central part of the Netherlands (Wageningen, Ede, Renkum, Arnhem, Barneveld, Veenendaal) [21]. NQPlus’ objectives were to establish a database for national dietary assessment reference to produce and validate food frequency questionnaires (FFQs), as well as to provide a database for longitudinal study in dietary factors and health-related outcomes. Extensive data collection was performed on habitual dietary intake and various health outcomes, including cardiometabolic health parameters. Between 2011 and 2013, 2048 participants were included in the study. After participants with missing dietary data (n = 401), implausible energy intake (n = 20), missing FLI data (n = 56), history of hepatitis (n = 32) or excessive alcohol consumption (n = 184) were consecutively excluded, n = 1341 remained (Supplemental Table 1). Due to the high percentages of missing covariates (n physical activity = 110 [8%], n sedentary behaviour = 111 [8%], n smoking status = 126 [9%]), multiple imputations were applied using “mice” package in R and five datasets were produced [22]. NQPlus was approved by the ethical committee of Wageningen University and Research.
PREDIMED-Plus
PREDIMED-Plus is a multicentre randomized controlled trial which was conducted based on the idea of the previous study, the PREDIMED (in Spanish: PREvención con DIeta MEDiterránea) trial [23]. PREDIMED-Plus aims to examine the effect of a traditional Mediterranean diet with energy restriction and increased physical activity on weight reduction and cardiovascular incidence and mortality. PREDIMED-Plus was conducted in 23 study centres across Spain. From September 2013 to November 2016, PREDIMED-plus recruited men aged 55–75 yr and women aged 60–75 yr with overweight or obesity who met at least three metabolic syndrome criteria according to the International Diabetes Federation and the AHA/National Heart, Lung and Blood Institute [24]. For present analysis, data from the Canary Island study centre were used, which included information for 266 participants with complete dietary data. Participants with implausible energy intake (n = 3), missing FLI (n = 10), and excessive alcohol consumption (n = 1) were consecutively excluded, and n = 250 remained for the current analyses (Supplemental Table 1). PREDIMED-Plus was registered at the International Standard Randomized Controlled Trial database (ISRCTN; 89898870) and the protocol was approved by local Research Ethics Committees.
AOC
AOC is a prospective cohort study of 4837 Dutch patients aged 60–80 yr who had experienced a myocardial infarction (MI) within 10 yr before study enrolment [25, 26]. The cohort is continuously followed for cause-specific mortality. Baseline measurements took place in 2002 and 2006 during which patients filled in questionnaires on demographics, lifestyle, diet, medication use and medical history, and underwent physical examination by trained research nurses. For current analyses, patients with missing dietary data (n = 453) or implausible dietary intake (n = 24), missing FLI (n = 179), excessive alcohol consumption (n = 647), or missing covariates (n = 41) were excluded, resulting in a total of n = 3493 remaining for analysis (Supplemental Table 1). AOC was registered at ClinicalTrials.gov (NCT03192410) and was approved by a central Medical Ethics Committee in the Netherlands.
Dietary assessment
Dietary intake data were collected using a newly developed and validated 110-item FFQ [27] and 183-item semi-quantitative FFQ [21, 28] in Lifelines and NQPlus, respectively, which covered all major food groups. These FFQs were created based on the Dutch consumption survey and were validated for energy and a range of nutrients as well as intake biomarkers. Average daily nutrient intake was calculated by multiplying consumption intake frequency by portion size and nutrient content in grams as indicated in the Dutch Food Composition Table (NEVO) 2011. In PREDIMED-Plus, dietary intake was collected using a 143-item FFQ [23, 29, 30], based on usual food consumption in Spain and designed to assess overall diet. This FFQ has been validated in different studies using four 3- or 4-day food records. Average daily nutrient intake was calculated by multiplying consumption intake frequency by portion size and nutrient content in the Spanish food composition tables. Within AOC, dietary data were collected using a semi-quantitative 203-item FFQ, a modified and extended version of the validated FFQ that was specifically designed to estimate fatty acids and cholesterol intake [31]. The Dutch Food Composition (NEVO) 2006 was used to convert food intake to energy and nutrient intakes in AOC. All population studies include intake measures of SSB, LNCB, and FJ, except for AOC where LNCB was combined with water, which therefore could not be analysed individually. Definition of SSB and LNCB covers soft drinks or lemonade sweetened with sugar or sugar substitutes. Coffee or tea sweetened with sugar or sweeteners as well as sweetened dairy drinks were not included in the definition of SSB and LNCB. FJ was defined as fruit juice i.e., apple juice, orange juice and mixed fruit juice, which were mainly pasteurized. SSB, LNCB, and FJ intakes were presented as servings of 150 ml.
Assessment and definition of NAFLD
Blood samples were collected after an overnight fast in all studies, except for AOC where overnight fasting blood samples were collected in 36% of all participants. Blood lipids including total cholesterol and triglycerides (TG) as well as liver enzymes including gamma-glutamyl transferase (GGT) were measured using routine procedures on a Roche Modular P800 chemistry analyser in Lifelines, Dimension Vista 1500 automated analyser or a Roche Modular P800 chemistry analyser in NQplus, and Cobas 8000 c 702 Module Roche Diagnostics in PREDIMED-Plus. In AOC, TG was analysed using Roche Hitachi 912, whereas liver enzymes were determined using automated analyses Abbott Architect ci8200. Anthropometry measurements (weight, height and waist circumference) were performed by well-trained staff. Body weight and height were measured without shoes. Body mass index (BMI) was calculated as weight divided by squared height (kg/m2). NAFLD was defined using FLI ≥ 60, which has been established by Bedogni et al. (2006) [8]. FLI is calculated based on serum concentrations of TG and GGT, BMI, and waist circumference, applying the following formula:
FLI = (eL/1 + eL)×100, where L = 0.953 × log(TG) + 0.139 × BMI + 0.718 × log(GGT) + 0.053 × waist circumference−15.745.
Assessment of covariates
Information on sociodemographic, lifestyle, and disease history was obtained from self- or interviewer-administered questionnaires. Educational levels was categorized into low (less than secondary qualification), medium (from secondary qualification up to university with no bachelor’s degree) or high (Bachelor’s, Master’s, or Ph.D. degree). Smoking status was categorized as non-smoker, former smoker, or current smoker. In Lifelines and NQplus, physical activity and sedentary behaviours (TV-watching) was assessed using the Short Questionnaire to Assess Health (SQUASH) [32] and the Activity Questionnaire for Adults and Adolescents (AQuAA) [33], respectively. In PREDIMED-Plus, physical activity was assessed using the Physical Activity Readiness Questionnaire (PAR-Q) as well as the Rapid Assessment of Physical Activity questionnaires 1 and 2 (RAPA-1 and RAPA-2) [34]. Sedentary activity in PREDIMED-Plus was assessed by using the Nurses’ Health Study physical activity questionnaire validated for the Spanish population [35]. Physical activity in Lifelines, NQPlus and PREDIMED-Plus was reported in Metabolic equivalent (MET)-min/week for moderate-level activity and in min/week for sedentary behaviour. In AOC, physical activity was assessed by using validated the Physical Activity Scale for the Elderly [36], and categorized as low (no activity/light activity, ≤3 METs), moderate (0–5 day/week moderately or vigorously active, >3 METs) or high (≥5 day/week moderately or vigorously active, >3 METs) physical activity. Alcohol intake was assessed using FFQ from which ethanol intake was calculated and categorized as 0, >0–≤10, >10–≤20, or >20 g/day.
Statistical analysis
Baseline characteristics were presented as mean with standard deviation for normally distributed continuous variables or as a median and interquartile range for variables with skewed distribution. Categorical variables were shown as numbers and percentages. Dose–response associations between SSB, LNCB, and FJ intakes and FLI-defined NAFLD prevalence were first analysed using restricted cubic spline analyses (3 knots) [37]. To ensure adequate power and precision, restricted cubic spline analyses were only performed for Lifelines. The fit of the spline model was examined against a linear model with the likelihood-ratio test. Cox proportional hazard regression with robust variance estimate was used to investigate the associations of SSB, LNCB and FJ intakes with FLI-defined NAFLD prevalence with a 95% confidence interval (PR [95% CI]) in each cohort, whereas theoretical substitution analyses were conducted using the leave-one-out model, where the model included SSB, LNCB, and FJ (in servings/day) as one variable followed by beverage defined as a replacement. Results of individual cohorts were subsequently pooled using random effect meta-analyses. To explore the potential of reverse causality, sensitivity analyses were conducted in Lifelines after excluding those with self-reported medical conditions (i.e., diabetes, CVD, hypercholesterolemia, or hypertension). All analyses were adjusted for age, sex (Model 1), educational levels (low, medium, or high), lifestyle factors including moderate physical activity (MET-min/week or in categories as in AOC), sedentary behaviour (min/week), smoking status (never, former, or current smoker), and alcohol use (0, >0–≤10, >10–≤20, or >20 g/day) (Model 2), intakes of other food groups including grains (g/day), potatoes (g/day), vegetables (g/day), fruit (g/day), meat and processed meat (g/day), dairy (g/d), coffee (ml/day), tea (ml/day), legumes (g/day), nuts (g/day), oils and fats (g/day), sugary foods (candy, sweet snack i.e., cookies, milk chocolate, any kind of toppings and popsicle) (g/day), and mutual adjustment for other beverages (SSB, LNCB, and FJ in g/day) as well as total energy intake (kcal/d) (Model 3). Statistical analysis was performed using R 4.0.2 and RStudio 1.3.959 for NQplus, PREDIMED-Plus, and AOC, and RStudio 2022.02.0 for Lifelines.
Results
General characteristics of participants
The mean (SD) age ranged from 45 (7) yr in Lifelines to 69 (6) yr in AOC (Table 1). More than half of the participants in Lifelines (61%), NQPlus (52%) and PREDIMED-Plus (66%) were women, whereas the opposite was observed in AOC (women 23%). Most participants reported low/moderate education, except for NQPlus participants of whom 54% reported higher education levels. Most participants never smoked, except for AOC participants of whom 66% were former smokers. Median (IQR) SSB intake ranged from 0.0 (0.0–0.1) serving/day in NQPlus and PREDIMED-Plus, to 0.1 (0.0–0.6) serving/day in Lifelines. Similar patterns were observed for LNCB. Median (IQR) FJ intake ranged from 0.1 (0.0–0.6) serving/day in NQPlus to 0.4 (0.0–1.1) serving/day in PREDIMED-Plus. FLI-defined NAFLD prevalence was 22% in Lifelines and NQplus, 60% in AOC and 78% in PREDIMED-Plus (Table 2).
Table 1.
General characteristics of Lifelines, NQPlus, PREDIMED-Plus and AOC participants.
| Characteristicsa | Lifelines | NQPlusb | PREDIMED-Plus | AOC |
|---|---|---|---|---|
| n | 36,940 | 1341 | 250 | 3493 |
| Age, yr | 45 ± 13 | 52 ± 12 | 65 ± 4 | 69 ± 6 |
| Women | 22,504 (61) | 657 (49) | 166 (66) | 786 (23) |
| Education | ||||
| Low | 1618 (4) | 9 (1) | 175 (70) | 2037 (58) |
| Moderate | 23,978 (65) | 608 (45) | 64 (26) | 1074 (31) |
| High | 11,344 (31) | 724 (54) | 11 (4) | 382 (11) |
| Smoking status | ||||
| Never | 17,053 (46) | 668 (55) | 171 (68) | 631 (18) |
| Former | 12,401 (34) | 461 (38) | 69 (28) | 2300 (66) |
| Current | 7476 (20) | 86 (7) | 10 (4) | 562 (16) |
| Moderate physical activity, MET-min/week | 1680 [818, 2988] | 798 [210, 1680] | 350 [0, 1399] | 731 (21)c |
| Sedentary behaviour, min/week | 840 [630, 1260] | 1800 [1200, 2700] | 1842 ± 789 | NA |
| Alcohol use | ||||
| 0 g/day | 1008 (3) | 76 (6) | 109 (44) | 211 (6) |
| >0–≤10 g/day | 27,177 (74) | 845 (63) | 109 (44) | 2202 (63) |
| >10–≤20 g/day | 7519 (20) | 306 (23) | 28 (11) | 766 (22) |
| >20 g/day | 1236 (3) | 114 (9) | 4 (2) | 314 (9) |
| Prevalent diabetes | 882 (2) | 45 (3) | 79 (32) | 592 (17) |
| Hypertension | 7948 (22) | 329 (25) | 225 (90) | 1719 (49) |
| History of CVD | 890 (2) | 40 (3) | All | All |
| Metabolic markers | ||||
| FLI | 26.9 [11.1, 55.7] | 27.0 [10.6, 55.9] | 78.6 [63.7, 88.8] | 67.3 [47.8, 83.5] |
| TG, mg/dL | 104 ± 69 | 91 [67, 129] | 132 [97, 169] | 145 [106, 204] |
| GGT, U/L | 20.0 [15.0–29.0] | 18.3 [13.3, 27.0] | 22.0 [16.0, 30.0] | 32.0 [24.0, 46.0] |
| Waist circumference, cm | 90.3 ± 12.3 | 91.2 ± 12.7 | 107.0 ± 9.9 | 101.7 ± 10.5 |
| BMI, kg/m2 | 26.0 ± 4.3 | 25.9 ± 4.1 | 32.5 ± 3.5 | 27.7 ± 3.8 |
| BMI ≥25 | 20,373 (55) | 721 (54) | 250 (100) | 2687 (77) |
| ALT | 19.0 [14.0, 27.0] | 24.1 [18.9, 30.7] | 18.0 [13.8, 24.0] | 16.0 [13.0, 21.0] |
| AST | 23.0 [19.0–27.00] | 21.4 [18.0, 27.0] | 18.0 [16.8, 24.0] | 27.0 [24.0, 32.0] |
| Dietary intakes | ||||
| SSB, servings/day | 0.1 [0.0, 0.6] | 0.0 [0.0, 0.1] | 0.0 [0.0, 0.1] | 0.1 [0.0, 0.5] |
| LNCB, servings/day | 0.1 [0.0, 0.6] | 0.0 [0.0, 0.1] | 0.0 [0.0, 0.0]d | NA |
| FJ, servings/day | 0.2 [0.0, 0.7] | 0.1 [0.0, 0.6] | 0.4 [0.0, 1.1] | 0.4 [0.1, 1.0] |
| Total energy, kcal/day | 2028 ± 566 | 2013 ± 555 | 2071 ± 532 | 1875 ± 509 |
| Grains, g/day | 191 ± 80 | 195 ± 87 | 122 ± 57 | 169 ± 59 |
| Potatoes, g/day | 92 ± 54 | 67 [37,97] | 50 [39,96] | 94 ± 45 |
| Vegetables, g/day | 103 ± 57 | 156 ± 86 | 269 ± 110 | 71 [52,94] |
| Fruits, g/day | 110 [42, 220] | 210 [83, 238] | 404 ± 179 | 116 [54, 262] |
| Meat and processed meat, g/day | 77 ± 36 | 70 ± 41 | 101 ± 50 | 77 [44, 107] |
| Dairy, g/day | 270 [167, 401] | 299 ± 182 | 460 ± 244 | 245 [162, 380] |
| Coffee, ml/day | 424 ± 277 | 406 [174, 638] | 50 [50, 125] | 375 [375, 562] |
| Tea, ml/day | 232 [45, 348] | 174 [67, 406] | 0 [0, 3] | 150 [45, 450] |
| Nuts, g/day | 7 [3,16] | 12 [6,21] | 13 [4,30] | 3 [1,7] |
| Legumes, g/day | 11 [0, 28] | 37 [19,75] | 59 ± 29 | 21 [12,37] |
| Oils and fats, g/day | 22 [12,32] | 27 ± 17 | 21 ± 10 | 37 ± 3 |
| Sugary food, g/day | 63 [39,95] | 51 ± 35 | 46 [24,80] | 64 [34, 101] |
AOC Alpha Omega Cohort, ALT alanine transaminase, AST aspartate aminotransferase, CVD cardiovascular disease, FJ fruit juice, FLI Fatty Liver Index, GGT gamma-glutamyl transferase, LNCB low/no-calorie sweetened beverages, MET metabolic equivalent task, NQPlus Nutrition Questionnaire Plus, SSB sugar-sweetened beverages, TG triglycerides.
aValue are mean ± SD, median [25th, 75th percentile], or n (%) as indicated.
bn physical activity = 1231, n sedentary activity = 1230, n smoking status = 1251.
cIn categories: no/light activity n = 1465 (42%), moderate physical activity is presented in the table, high physical activity n = 1297 (37%).
dIntake ranged from 0.00 to 3.33 serving/day.
Table 2.
Associations of SSB, LNCB, and FJ intakes with FLI-defined NAFLD prevalence for each serving per day increment in all cohorts.
| PR (95% CI) | ||||||
|---|---|---|---|---|---|---|
| Lifelines | NQPlusa | PREDIMED-Plus | AOC | Pooled | ||
| n total | 36,940 | 1341 | 250 | 3493 | PR (95% CI) | I2, p-value |
| n (%) cases | 8200 (22) | 290 (22) | 195 (78) | 2092 (60) | ||
| SSB | ||||||
| Model 1b | 1.11 (1.09–1.13) | 1.02 (0.82–1.26) | 1.01 (0.79–1.29) | 1.05 (1.03–1.07) | 1.07 (1.02–1.13) | 86%, <0.01 |
| Model 2c | 1.06 (1.04–1.08) | 0.99 (0.81–1.22) | 1.01 (0.77–1.31) | 1.04 (1.02–1.07) | 1.05 (1.04–1.07) | 0%, 0.55 |
| Model 3d | 1.09 (1.07–1.11) | 0.94 (0.76–1.17) | 1.01 (0.74–1.37) | 1.05 (1.02–1.08) | 1.07 (1.03–1.11) | 58%, 0.07 |
| LNCB | ||||||
| Model 1b | 1.24 (1.22–1.26) | 1.35 (1.19–1.53) | 1.08 (0.93–1.25) | NA | 1.23 (1.12–1.34) | 73%, 0.02 |
| Model 2c | 1.21 (1.19–1.23) | 1.29 (1.14–1.45) | 1.08 (0.94–1.25) | NA | 1.20 (1.12–1.29) | 58%, 0.09 |
| Model 3d | 1.20 (1.18–1.22) | 1.22 (1.08–1.37) | 1.12 (0.96–1.31) | NA | 1.20 (1.18–1.22) | 0%, 0.57 |
| FJ | ||||||
| Model 1b | 1.05 (1.02–1.08) | 0.99 (0.81–1.20) | 1.03 (0.96–1.11) | 0.99 (0.96–1.02) | 1.02 (0.98–1.06) | 63%, 0.04 |
| Model 2c | 1.05 (1.02–1.08) | 0.96 (0.80–1.16) | 1.03 (0.96–1.10) | 1.00 (0.97–1.03) | 1.02 (0.99–1.06) | 51%, 0.11 |
| Model 3d | 1.07 (1.04–1.10) | 0.96 (0.81–1.14) | 1.03 (0.94–1.12) | 1.01 (0.98–1.04) | 1.03 (0.99–1.08) | 67%, 0.03 |
AOC Alpha Omega Cohort, CI confidence interval, FJ fruit juice, LNCB low/no-calorie sweetened beverages, NQPlus Nutrition Questionnaire Plus, PR prevalence ratio, SSB sugar-sweetened beverages.
aImputed with multiple imputation method.
bModel 1: adjusted for age and sex.
cModel 2: model 1 with additional adjustment for education levels, moderate physical activity, sedentary behaviour, smoking status, alcohol intake.
dModel 3: model 2 with additional adjustment for grains, potatoes, vegetable, fruit, meat and processed meat, dairy, coffee, tea, legumes, nuts, oils and fats, sugary foods, mutual adjustment for other beverages (SSB, LNCB, or FJ, except for AOC where LNCB consumption data was not available), and energy intake.
In most cohorts, participants with a higher SSB intake were more likely to be younger, men and less physically active. The opposite trends were observed in AOC. In all cohorts, SSB consumers were more likely to be diagnosed with diabetes, had a higher intake of meat, sugary food and total energy, and lower intake of vegetables and fruit (Supplemental Table 2). Participants with higher LNCB intake were more likely to be younger, less physically active, to be diagnosed with diabetes, hypercholesterolemia or hypertension, have a BMI ≥25, higher meat intake, and lower intake of vegetables and fruit (Supplemental Table 3). For FJ, participants with a higher intake were more likely to have BMI <25 and higher sugary food consumption (Supplemental Table 4).
Association between SSB, LNCB and FJ intakes and FLI-defined NAFLD prevalence
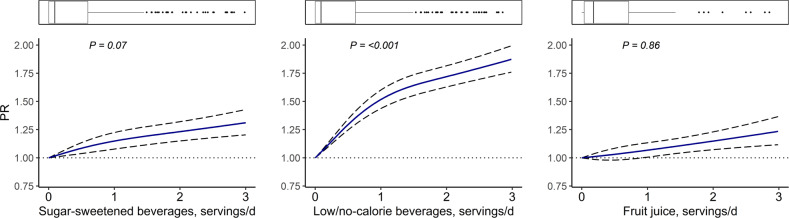
Dose–response analysis in Lifelines did not provide strong evidence of a non-linear association between SSB and FLI-defined NAFLD (P non-linearity = 0.07) (Fig. 1). Pooled results of four studies showed an association between each additional serving/day of SSB and higher prevalence of FLI-defined NAFLD (PR 1.07, 95% CI 1.03–1.11) (Table 2). Although there was evidence of interaction between SSB and age categories, results from a stratified analysis in Lifelines by age as well as by sex did not indicate significant differences (Supplemental Table 6). Sensitivity analysis by excluding those with self-reported disease in Lifelines did not alter the associations (Supplemental Table 7).
Fig. 1. Dose–response associations of SSB, LNCB and FJ intakes with FLI-defined NAFLD in Lifelines.
Three knots with 0 g/day as a reference value were placed and model was adjusted for age, sex, education levels, moderate physical activity, sedentary behaviour, smoking status, alcohol intake, grains, potatoes, vegetable, fruit, meat and processed meat, dairy, coffee, tea, legumes, nuts, oils and fats, sugary foods, mutual adjustment for other beverages (SSB, LNCB, or FJ), and energy intake. CI confidence interval, FJ fruit juices, LNCB low/no-calorie sweetened beverages, PR prevalence ratio, SSB sugar-sweetened beverages.
Dose–response analysis in Lifelines showed strong evidence of a non-linear association between LNCB and FLI-defined NAFLD prevalence (P non-linearity = <0.001) (Fig. 1), with the association getting stronger when consuming ≤1 serving/day and levelling off at higher intake levels. Subsequent Cox regression showed that, compared to no intake, LNCB intake of ≤2 servings/week was associated with a PR of 1.13 (95% CI 1.00–1.26), whereas intake of >2 servings/week was associated with a PR of 1.38 (95% CI 1.15–1.61) (Table 3). In Lifelines, stratified analysis by sex showed a stronger association in women (PR 1.65, 95% CI 1.54–1.77) than in men (PR 1.36, 95% CI 1.29–1.43) when comparing those with LNCB intake >2 servings/week with non-consumers (Supplemental Table 6). Excluding participants with self-reported disease did not alter the main findings (Supplemental Table 7). Theoretical replacement of SSB with the same amount of LNCB was associated with a higher prevalence of FLI-defined NAFLD (PR 1.12, 95% CI 1.03–1.21) (Table 4).
Table 3.
Associations of SSB, LNCB, and FJ intakes with FLI-defined NAFLD prevalence for categories of beverage consumption in all cohorts.
| PR (95% CI) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| SSB | LNCB | FJ | |||||||
| No intake | ≤2 servings/week | >2 servings/week | No intake | ≤2 servings/week | >2 servings/week | No intake | ≤2 servings/week | >2 servings/week | |
| Lifelines | |||||||||
| N total | 13949 | 9262 | 14,122 | 14,476 | 7929 | 14,305 | 8154 | 11,747 | 17,039 |
| N(%) cases | 3325 (24) | 1438 (18) | 3781 (27) | 2897 (20) | 1454 (18) | 3849 (27) | 2180 (27) | 2322 (20) | 3698 (22) |
| Model 1b | 1 (ref) | 0.75 (0.71–0.79) | 1.08 (0.99–1.09) | 1 (ref) | 0.95 (0.90–1.01) | 1.51 (1.45–1.58) | 1 (ref) | 0.78 (0.75–0.82) | 0.87 (0.83–0.90) |
| Model 2c | 1 (ref) | 0.77 (0.73–1.04) | 0.99 (0.95–1.04) | 1 (ref) | 1.00 (0.95–1.06) | 1.50 (1.44–1.56) | 1 (ref) | 0.85 (0.81–0.90) | 0.94 (0.90–0.98) |
| Model 3d | 1 (ref) | 0.90 (0.86–0.95) | 1.08 (1.03–1.13) | 1 (ref) | 1.05 (0.99–1.11) | 1.48 (1.42–1.54) | 1 (ref) | 0.92 (0.87–0.96) | 1.01 (0.97–1.06) |
| NQPlusa | |||||||||
| N total | 737 | 412 | 192 | 883 | 268 | 190 | 292 | 493 | 556 |
| N(%) cases | 155 (21) | 90 (22) | 45 (23) | 160 (18) | 65 (24) | 65 (34) | 66 (23) | 109 (22) | 115 (21) |
| Model 1b | 1 (ref) | 0.95 (0.76–1.20) | 1.03 (0.76–1.39) | 1 (ref) | 1.28 (1.00–1.64) | 1.78 (1.40–2.26) | 1 (ref) | 0.97 (0.74–1.26) | 0.91 (0.70–1.18) |
| Model 2c | 1 (ref) | 0.97 (0.77–1.22) | 1.01 (0.74–1.36) | 1 (ref) | 1.22 (0.96–1.57) | 1.67 (1.30–2.13) | 1 (ref) | 0.99 (0.76–1.28) | 0.93 (0.71–1.20) |
| Model 3d | 1 (ref) | 1.06 (0.84–1.34) | 1.02 (0.74–1.39) | 1 (ref) | 1.17 (0.91–1.51) | 1.50 (1.18–1.91) | 1 (ref) | 1.08 (0.83–1.41) | 1.02 (0.79–1.32) |
| PREDIMED-Plus | |||||||||
| N total | 180 | 49 | 21 | 189 | 45 | 16 | 77 | 41 | 132 |
| N(%) cases | 139 (77) | 10 (80) | 17 (81) | 142 (75) | 40 (89) | 13 (81) | 58 (75) | 28 (68) | 109 (83) |
| Model 1b | 1 (ref) | 1.02 (0.87–1.21) | 1.00 (0.79–1.26) | 1 (ref) | 1.20 (1.04–1.37) | 1.08 (0.85–1.37) | 1 (ref) | 0.92 (0.72–1.18) | 1.10 (0.95–1.28) |
| Model 2c | 1 (ref) | 1.00 (0.85–1.17) | 0.97 (0.78–1.22) | 1 (ref) | 1.22 (1.07–1.40) | 1.07 (0.85–1.34) | 1 (ref) | 0.92 (0.73–1.16) | 1.10 (0.94–1.28) |
| Model 3d | 1 (ref) | 0.97 (0.83–1.14) | 0.96 (0.75–1.24) | 1 (ref) | 1.23 (1.07–1.43) | 1.12 (0.89–1.42) | 1 (ref) | 0.95 (0.76–1.20) | 1.13 (0.96–1.32) |
| AOC | |||||||||
| N total | 1577 | 721 | 1195 | NA | NA | NA | 873 | 630 | 1990 |
| N(%) cases | 903 (57) | 426 (59) | 761 (64) | 567 (65) | 362 (58) | 1163 (58) | |||
| Model 1b | 1 (ref) | 1.02 (0.94–1.10) | 1.09 (1.02–1.15) | NA | NA | NA | 1 (ref) | 0.89 (0.82–0.96) | 0.91 (0.85–0.96) |
| Model 2c | 1 (ref) | 1.03 (0.95–1.10) | 1.08 (1.02–1.15) | NA | NA | NA | 1 (ref) | 0.90 (0.83–0.98) | 0.94 (0.89–1.00) |
| Model 3d | 1 (ref) | 1.03 (0.96–1.11) | 1.09 (1.02–1.16) | NA | NA | NA | 1 (ref) | 0.92 (0.85–1.00) | 0.97 (0.91–1.03) |
| Pooled | |||||||||
| Model 3d | 0.97 (0.89–1.05) | 1.08 (1.04–1.12) | 1.13 (1.00–1.26) | 1.38 (1.15–1.61) | 0.92 (0.88–0.97) | 1.00 (0.97–1.04) | |||
| I2, p value | 75%, <0.01 | 0%, 0.78 | 72%, 0.03 | 78%, 0.01 | 0%, 0.74 | 18%, 0.30 | |||
AOC Alpha Omega Cohort, CI confidence interval, FJ fruit juice, LNCB low/no-calorie sweetened beverages, NQPlus Nutrition Questionnaire Plus, PR prevalence ratio, SSB sugar-sweetened beverages.
aImputed with multiple imputation method.
bModel 1: adjusted for age and sex.
cModel 2: model 1 with additional adjustment for education levels, moderate physical activity, sedentary behaviour, smoking status, alcohol intake.
dModel 3: model 2 with additional adjustment for grains, potatoes, vegetable, fruit, meat and processed meat, dairy, coffee, tea, legumes, nuts, oils and fats, sugary foods, mutual adjustment for other beverages (SSB, LNCB, or FJ, except for AOC where LNCB consumption data was not available), and energy intake.
Table 4.
Adjusted associations of SSB, LNCB, and FJ consumption and FLI-defined NAFLD prevalence when replacing SSB with the same amount of LNCB or FJ.
| PR (95%CI) | ||||||
|---|---|---|---|---|---|---|
| Lifelines | NQPlusa | PREDIMED-Plus | AOC | Pooled | ||
| n total | 36,940 | 1341 | 250 | 3493 | PR | I2, p-value |
| n (%) cases | 8200 (22) | 290 (22) | 195 (78) | 2092 (60) | ||
| SSB by LNCB | ||||||
| Model 1b | 1.11 (1.08–1.14) | 1.36 (1.06–1.73) | 1.08 (0.82–1.42) | NA | 1.16 (1.02–1.30) | 50%, 0.13 |
| Model 2c | 1.13 (1.10–1.15) | 1.32 (1.04–1.69) | 1.08 (0.80–1.44) | NA | 1.15 (1.06–1.23) | 19%, 0.29 |
| Model 3d | 1.13 (1.11–1.16) | 1.30 (1.02–1.65) | 1.10 (0.81–1.51) | NA | 1.14 (1.09–1.18) | 0%, 0.38 |
| Model 4e | 1.10 (1.07–1.13) | 1.28 (1.00–1.63) | 1.11 (0.80–1.53) | NA | 1.12 (1.03–1.21) | 3%, 0.36 |
| SSB by FJ | ||||||
| Model 1b | 0.92 (0.88–0.95) | 0.97 (0.72–1.29) | 1.03 (0.78–1.35) | 0.94 (0.91–0.98) | 0.93 (0.91–0.96) | 0%, 0.77 |
| Model 2c | 0.97 (0.93–1.00) | 0.96 (0.71–1.29) | 1.02 (0.76–1.36) | 0.96 (0.92–0.99) | 0.97 (0.94–0.99) | 0%, 0.96 |
| Model 3d | 0.99 (0.96–1.03) | 1.02 (0.76–1.38) | 1.01 (0.74–1.39) | 0.96 (0.93–1.00) | 0.98 (0.95–1.00) | 0%, 0.69 |
| Model 4e | 0.98 (0.95–1.02) | 1.00 (0.74–1.36) | 1.02 (0.74–1.39) | 0.96 (0.93–1.00) | 0.97 (0.95–1.00) | 0%, 0.87 |
AOC Alpha Omega Cohort, CI confidence interval, FJ fruit juice, LNCB low/no-calorie sweetened beverages, NQPlus Nutrition Questionnaire Plus, PR prevalence ratio, SSB sugar-sweetened beverages.
aImputed with multiple imputation method.
bModel 1: adjusted for age and sex.
cModel 2: model 1 with additional adjustment for education levels, moderate physical activity, sedentary behaviour, smoking status, alcohol intake.
dModel 3: model 2 with additional adjustment for grains, potatoes, vegetable, fruit, meat and processed meat, dairy, coffee, tea, legumes, nuts, oils and fats, sugary foods, and mutual adjustment for other beverages (SSB, LNCB, or FJ, except for AOC where LNCB consumption data was not available).
eModel 4: model 3 with additional adjustment for energy intake.
Dose–response analysis in Lifelines did not suggest evidence of a nonlinear association between FJ and FLI-defined NAFLD prevalence (P non-linearity = 0.86) (Fig. 1). However, pooled analysis by categories showed an inverse association between FJ and FLI-defined NAFLD at intake levels of ≤2 servings/week (PR 0.92, 95% CI 0.88–0.97), but no association was seen at higher intake levels (PR 1.00, 95% CI 0.97–1.04) when compared to no intake (Table 3). After excluding those with self-reported disease, the associations did not change significantly (Supplemental Table 6). Finally, there was no evidence for an association between theoretical replacement of SSB with the same amount of FJ and FLI-defined NAFLD (PR 0.97, 95% CI: 0.95–1.00) (Table 4).
Discussion
Harmonized data analyses of four European studies encompassing both general population and cardiometabolic subjects showed positive associations between SSB and LNCB intakes and FLI-defined NAFLD prevalence. Dose-response analyses in Lifelines showed a non-linear positive association between LNCB and FLI-defined NAFLD with the stronger association at intake levels of <1 serving/day and levelling off at higher intake levels. A beneficial association was observed for moderate FJ intake ranging from ≤2 servings/day when compared to no intake. Theoretical replacement of SSB with the same amount of LNCB showed a positive association with FLI-defined NAFLD, whereas no significant association was observed when replacing SSB with FJ.
Overall, our findings on the association between SSB and FLI-defined NAFLD prevalence are consistent with previous research [6, 7, 11, 38, 39]. A meta-analysis of 12 studies (nine cross-sectional, two case-control and one cohort study) showed a 39% (95% CI 1.29–1.50) higher NAFLD risk—as assessed using liver imaging or biopsy—when comparing highest SSB intake group with non-consumer [6]. Furthermore, a recent study by Zhang et al. in 14,845 participants showed a 47% (HR 95% CI 1.25–1.23) and 59% (HR 95% CI 1.07–2.37) higher prevalence of NAFLD when NAFLD was assessed using abdominal ultrasonography and serum biomarkers (hepatic steatosis index) [11]. In addition, results from short-term trials, experimental studies, and meta-analyses of RCT evaluating individual metabolic markers further confirm our findings [40–44]. The biological explanation of the adverse association between SSB intake and NAFLD can be explained by several biological mechanisms. Its high fructose content may induce hepatic de novo lipogenesis and possibly insulin resistance [45, 46]. Moreover, lack of energy compensation after liquid calorie intake affects energy balance disruption, which contributes to weight gain, an important factor in NAFLD development [47].
We observed positive associations between LNCB, as well as between theoretical replacement of SSB with LNCB and FLI-defined NAFLD prevalence. To date, evidence on LNCB and NAFLD is still limited [7, 13, 14], especially from large studies. In line with our findings, prospective analysis among 1636 women participating in the Framingham Heart Study showed a 48% higher risk of NAFLD and LNCB—when NAFLD was detected using computed liver attenuation measurements [7]. However, this association disappeared after further adjustment for BMI (OR 1.11, 95% CI 0.80–1.56). A prospective study in women with a history of gestational diabetes also showed no association between LNCB and the majority of NAFLD markers when the models were adjusted by various covariates, particularly pre-pregnancy BMI, as well as when performing sensitivity analyses excluding those with chronic diseases - indicating that reverse causality may be the cause [14]. Within our study, participants at higher intake levels of LNCB tended to have a BMI ≥ 25, which might also indicate the presence of reverse causality. Participants with NAFLD or elevated NAFLD parameters may have switched from SSB to LNCB to control their health, e.g., weight regulation. Evidence from experimental studies was summarized in a meta-analysis of 12 RCTs involving 601 adults with overweight or obesity showing that replacing SSB with LNCB reduced body weight, body fat, BMI, intrahepatocellular lipid, but not other clinical parameters including liver enzymes i.e. alanine transaminase and aspartate aminotransferase [41]. A biological explanation for the association between LNCB with health outcomes is inconclusive. LNCB has been suggested to disturb reward systems, activate the cephalic phase insulin response, and induce gut microbiota dysbiosis, which can further lead to insulin resistance and the development of type 2 diabetes [48, 49]. However, more studies including experimental studies in humans as well as long-term evaluations in large cohorts are still needed to further investigate these hypotheses [7, 50].
To date, only a few studies specifically explored associations of FJ with health outcomes. Our results suggest an inverse association when consuming ≤2 servings/day compared to no intake, which has also been observed in a dose–response meta-analysis of two cohorts investigating the association between FJ intake (75–150 ml/day) and MetS incidence [15]. Khan et al. also demonstrated an inverse association between FJ intake of ~150 ml and CVD incidence, but not at higher intake levels [51]. A recent and extensive meta-analysis by D’Elia also suggested a non-linear association between low to moderate 100% FJ intake (<80 ml/day) with incidence rates of stroke and CVD [17]. This inverse association among participants with moderate intake levels might be due to overall healthier behaviours of participants within this intake group. However, this notion is not clearly seen in our study populations as the distribution of characteristics was comparable across groups in all studies. Possible underlying mechanisms of the inverse associations between moderate intake of FJ and NAFLD include the beneficial composition of FJ, which may contain a certain amount of antioxidants (i.e polyphenols) and other bioactive components (i.e vitamins, fibre and minerals) that enhance metabolic profile [15, 17, 51, 52]. However, its high caloric content and the presence of fructose may be counterproductive to the benefits of consuming higher quantities of FJ.
Strengths of our study include the large sample size derived from several European population-based studies, the opportunity to control for a wide range of relevant confounders and to perform stratified analysis particularly in Lifelines which comprised the largest cohort. We conducted the harmonized statistical analyses across cohorts including substitution analyses which to date, are often lacking in recent studies. This is the first meta-analysis focusing on NAFLD assessed by FLI, which facilitated non-invasive and simple assessment in larger studies. There are also several limitations to this study. Dietary intake was self-reported, hence bias due to social desirability cannot be excluded. Although the FFQs used in current study were extensively evaluated for a range of nutrients and foods, they were not specifically designed to examine SSB, LNCB, and specific sweeteners intakes. Moreover, SSB, LNCB, and FJ intakes in our study were relatively low, which may explain the lower risk estimates in our study when compared to some previous ones i.e from the US [6, 7, 13]. SSB intake in our Dutch study populations was lower than that reported in the Dutch National Food Consumption Survey, which may be because SSB was grouped together with carbonated/soft/isotonic drinks and diluted syrups. We used FLI as a proxy for NAFLD, which has limitations including its inability to distinguish moderate to severe steatosis from mild steatosis [9, 53]. The gold standard for NAFLD diagnosis is liver biopsy which is an invasive and costly procedure. To avoid these consequences, non-invasive imaging procedures have been introduced, i.e ultrasound or magnetic resonance. However, it also has limitations associated with availability and cost for large-scale studies, therefore use of serum biomarkers is preferred. FLI has been externally validated in several European and Asian populations [10, 54–57], and has been endorsed to be the best-validated tool to detect the presence of steatosis [58, 59]. Despite adjustment for a wide range of confounders, residual confounding may still be present. However, additional sensitivity analysis using the E-value [60] showed that any unmeasured confounder would need to be quite strongly associated with both exposure and outcome to explain away the association (data not shown), which is not very likely based on the results for the confounders included. Finally, as always in a cross-sectional study, reverse causality cannot be excluded.
In conclusion, we observed adverse associations between SSB and LNCB intakes and FLI-defined NAFLD prevalence, as well as between replacement of SSB with the same amount of LNCB and FLI-defined NAFLD, which may partly be explained by reverse causality. Our findings suggest a beneficial association between moderate intake of FJ and FLI-defined NAFLD at intake level of ≤2 servings/day when compared to no intake. These findings provide additional information on the potential adverse impact of sweetened beverages on health. Longer-term prospective studies with objective methods determining the intake of sugar and sweeteners are warranted to further substantiate our findings.
Supplementary information
Acknowledgements
This EU project under the acronym “SWEET” has received funding from the European Union’s Horizon 2020 research and innovation programme under grant agreement No. 774293. The funders had no role in the study design, data collection, data analysis and interpretation, writing the report, or the decision to submit the article for publication. The authors wish to acknowledge the assistance of the research team of all cohorts included in this study and all the study participants.
Author contributions
JAH, JCGH, and AR are coordinators of the SWEET EU project and together with EJMF initiated the research question. NDN analysed the data. NDN, JN, and EMB-B interpreted the results and drafted the manuscript. NDN, JN, EMB-B, MECB, SSS-M, CP-R, JMG, LS-M and EJMF discussed the results and reviewed the draft manuscript. All authors critically revised the manuscript and approved the final version of the manuscripts.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request and after approval from the relevant partners.
Competing interests
SSS-M has received recent research funding for epidemiological studies on dairy products and cardiometabolic diseases from the Dutch Dairy Association and the Danish Dairy Research Foundation. JCGH is a member of the International Sweeteners and Mars Scientific Advisory Boards and has received honorariums from the International Sweeteners Association together with AR. JCGH and JAH are also conducting the SWITCH trial funded by the American Beverage Association. AR has received an honorarium from Unilever and Nestlé. In the past, EJMF has received an unrestricted grant from Friesland Campina and European Beer Institute and conducted a study on added sugar and individual sugars partly funded by Kenniscentrum Suiker en Gezondheid (2011). Other authors have no competing interests to be disclosed.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
The online version contains supplementary material available at 10.1038/s41387-023-00237-3.
References
- 1.Chalasani N, Younossi Z, Lavine JE, Charlton M, Cusi K, Rinella M, et al. The diagnosis and management of nonalcoholic fatty liver disease: practice guidance from the American Association for the Study of Liver Diseases. Hepatology. 2018;67:328–57. doi: 10.1002/hep.29367. [DOI] [PubMed] [Google Scholar]
- 2.Younossi ZM, Koenig AB, Abdelatif D, Fazel Y, Henry L, Wymer M. Global epidemiology of nonalcoholic fatty liver disease-meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology. 2016;64:73–84. doi: 10.1002/hep.28431. [DOI] [PubMed] [Google Scholar]
- 3.Rietman A, Sluik D, Feskens EJM, Kok FJ, Mensink M. Associations between dietary factors and markers of NAFLD in a general Dutch adult population. Eur J Clin Nutr. 2018;72:117–23. doi: 10.1038/ejcn.2017.148. [DOI] [PubMed] [Google Scholar]
- 4.van den Berg EH, Amini M, Schreuder TCMA, Dullaart RPF, Faber KN, Alizadeh BZ, et al. Prevalence and determinants of non-alcoholic fatty liver disease in lifelines: a large Dutch population cohort. PLoS ONE. 2017;12:e0171502.. doi: 10.1371/journal.pone.0171502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bullón-Vela V, Abete I, Tur JA, Pintó X, Corbella E, Martínez-González MA, et al. Influence of lifestyle factors and staple foods from the Mediterranean diet on non-alcoholic fatty liver disease among older individuals with metabolic syndrome features. Nutrition. 2020;71:110620. doi: 10.1016/j.nut.2019.110620. [DOI] [PubMed] [Google Scholar]
- 6.Chen H, Wang J, Li Z, Lam CWK, Xiao Y, Wu Q, et al. Consumption of Sugar-sweetened beverages has a dose-dependent effect on the risk of non-alcoholic fatty liver disease: an updated systematic review and dose-response meta-analysis. Int J Environ Res Public Health. 2019;16:2192.. doi: 10.3390/ijerph16122192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Park WY, Yiannakou I, Petersen JM, Hoffmann U, Ma J, Long MT. Sugar-sweetened beverage, diet soda, and nonalcoholic fatty liver disease over 6 years: the Framingham Heart Study. Clin Gastroenterol Hepatol. 2021;21:01178–2. doi: 10.1016/j.cgh.2021.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bedogni G, Bellentani S, Miglioli L, Masutti F, Passalacqua M, Castiglione A, et al. The Fatty Liver Index: a simple and accurate predictor of hepatic steatosis in the general population. BMC Gastroenterol. 2006;6:33. doi: 10.1186/1471-230X-6-33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Castera L, Friedrich-Rust M, Loomba R. Noninvasive assessment of liver disease in patients with nonalcoholic fatty liver disease. Gastroenterology. 2019;156:1264–81.e4. doi: 10.1053/j.gastro.2018.12.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Koehler EM, Schouten JN, Hansen BE, Hofman A, Stricker BH, Janssen HL. External validation of the fatty liver index for identifying nonalcoholic fatty liver disease in a population-based study. Clin Gastroenterol Hepatol. 2013;11:1201–4. doi: 10.1016/j.cgh.2012.12.031. [DOI] [PubMed] [Google Scholar]
- 11.Zhang S, Gu Y, Bian S, Lu Z, Zhang Q, Liu L, et al. Soft drink consumption and risk of nonalcoholic fatty liver disease: results from the Tianjin Chronic Low-Grade Systemic Inflammation and Health (TCLSIH) cohort study. Am J Clin Nutr. 2021;113:1265–74. doi: 10.1093/ajcn/nqaa380. [DOI] [PubMed] [Google Scholar]
- 12.Cantoral A, Contreras-Manzano A, Luna-Villa L, Batis C, Roldán-Valadez EA, Ettinger AS, et al. Dietary sources of fructose and its association with fatty liver in Mexican young adults. Nutrients. 2019;11:522.. doi: 10.3390/nu11030522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ma J, Fox CS, Jacques PF, Speliotes EK, Hoffmann U, Smith CE, et al. Sugar-sweetened beverage, diet soda, and fatty liver disease in the Framingham Heart Study cohorts. J Hepatol. 2015;63:462–9. doi: 10.1016/j.jhep.2015.03.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hinkle SN, Rawal S, Bjerregaard AA, Halldorsson TI, Li M, Ley SH, et al. A prospective study of artificially sweetened beverage intake and cardiometabolic health among women at high risk. Am J Clin Nutr. 2019;110:221–32. doi: 10.1093/ajcn/nqz094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Semnani-Azad Z, Khan TA, Blanco Mejia S, de Souza RJ, Leiter LA, Kendall CWC, et al. Association of major food sources of fructose-containing sugars with incident metabolic syndrome: a systematic review and meta-analysis. JAMA Netw Open. 2020;3:e209993–e209993. doi: 10.1001/jamanetworkopen.2020.9993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Khan TA, Chiavaroli L, Zurbau A, Sievenpiper JL. A lack of consideration of a dose–response relationship can lead to erroneous conclusions regarding 100% fruit juice and the risk of cardiometabolic disease. Eur J Clin Nutr. 2019;73:1556–60. doi: 10.1038/s41430-019-0514-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.D’Elia L, Dinu M, Sofi F, Volpe M, Strazzullo P. 100% Fruit juice intake and cardiovascular risk: a systematic review and meta-analysis of prospective and randomised controlled studies. Eur J Nutr. 2021;60:2449–67. doi: 10.1007/s00394-020-02426-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Klijs B, Scholtens S, Mandemakers JJ, Snieder H, Stolk RP, Smidt N. Representativeness of the LifeLines Cohort Study. PLoS ONE. 2015;10:e0137203.. doi: 10.1371/journal.pone.0137203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sijtsma A, Rienks J, van der Harst P, Navis G, Rosmalen JGM, Dotinga A. Cohort Profile Update: lifelines, a three-generation cohort study and biobank. Int J Epidemiol. 2021;51:e295–e302. doi: 10.1093/ije/dyab257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Willett, W (2013) Nutritional epidemiology. New York: Oxford University Press.
- 21.Brouwer-Brolsma EM, van Lee L, Streppel MT, Sluik D, van de Wiel AM, de Vries JHM, et al. Nutrition Questionnaires plus (NQplus) study, a prospective study on dietary determinants and cardiometabolic health in Dutch adults. BMJ Open. 2018;8:e020228.. doi: 10.1136/bmjopen-2017-020228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Sterne JA, White IR, Carlin JB, Spratt M, Royston P, Kenward MG, et al. Multiple imputation for missing data in epidemiological and clinical research: potential and pitfalls. BMJ. 2009;338:b2393. doi: 10.1136/bmj.b2393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Martinez-Gonzalez MA, Buil-Cosiales P, Corella D, Bullo M, Fito M, Vioque J, et al. Cohort Profile: design and methods of the PREDIMED-Plus randomized trial. Int J Epidemiol. 2019;48:387–388o. doi: 10.1093/ije/dyy225. [DOI] [PubMed] [Google Scholar]
- 24.Alberti KG, Eckel RH, Grundy SM, Zimmet PZ, Cleeman JI, Donato KA, et al. Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation. 2009;120:1640–5. doi: 10.1161/CIRCULATIONAHA.109.192644. [DOI] [PubMed] [Google Scholar]
- 25.Kromhout D, Giltay EJ, Geleijnse JM. n-3 Fatty acids and cardiovascular events after myocardial infarction. N Engl J Med. 2010;363:2015–26. doi: 10.1056/NEJMoa1003603. [DOI] [PubMed] [Google Scholar]
- 26.Geleijnse JM, Giltay EJ, Schouten EG, de Goede J, Oude Griep LM, Teitsma-Jansen AM, et al. Effect of low doses of n-3 fatty acids on cardiovascular diseases in 4,837 post-myocardial infarction patients: design and baseline characteristics of the Alpha Omega Trial. Am Heart J. 2010;159:539–546.e2. doi: 10.1016/j.ahj.2009.12.033. [DOI] [PubMed] [Google Scholar]
- 27.Brouwer-Brolsma EM, Perenboom C, Sluik D, van de Wiel A, Geelen A, Feskens EJ, et al. Development and external validation of the ‘Flower-FFQ’: a FFQ designed for the Lifelines Cohort Study. Public Health Nutr. 2021;25:225–36. doi: 10.1017/s1368980021002111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Streppel MT, de Vries JH, Meijboom S, Beekman M, de Craen AJ, Slagboom PE, et al. Relative validity of the food frequency questionnaire used to assess dietary intake in the Leiden Longevity Study. Nutr J. 2013;12:75.. doi: 10.1186/1475-2891-12-75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Fernández-Ballart JD, Piñol JL, Zazpe I, Corella D, Carrasco P, Toledo E, et al. Relative validity of a semi-quantitative food-frequency questionnaire in an elderly Mediterranean population of Spain. Br J Nutr. 2010;103:1808–16. doi: 10.1017/s0007114509993837. [DOI] [PubMed] [Google Scholar]
- 30.de la Fuente-Arrillaga C, Ruiz ZV, Bes-Rastrollo M, Sampson L, Martinez-González MA. Reproducibility of an FFQ validated in Spain. Public Health Nutr. 2010;13:1364–72. doi: 10.1017/s1368980009993065. [DOI] [PubMed] [Google Scholar]
- 31.Feunekes GI, Van Staveren WA, De Vries JH, Burema J, Hautvast JG. Relative and biomarker-based validity of a food-frequency questionnaire estimating intake of fats and cholesterol. Am J Clin Nutr. 1993;58:489–96. doi: 10.1093/ajcn/58.4.489. [DOI] [PubMed] [Google Scholar]
- 32.Wendel-Vos GC, Schuit AJ, Saris WH, Kromhout D. Reproducibility and relative validity of the short questionnaire to assess health-enhancing physical activity. J Clin Epidemiol. 2003;56:1163–9. doi: 10.1016/s0895-4356(03)00220-8. [DOI] [PubMed] [Google Scholar]
- 33.Chinapaw MJM, Slootmaker SM, Schuit AJ, van Zuidam, M, and van Mechelen W. Reliability and validity of the Activity Questionnaire for Adults and Adolescents (AQuAA) BMC Med Res Methodol. 2009;9:58.. doi: 10.1186/1471-2288-9-58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Topolski TD, LoGerfo J, Patrick DL, Williams B, Walwick J, Patrick MB. The rapid assessment of physical activity (RAPA) among older adults. Prev Chronic Dis. 2006;3:A118. [PMC free article] [PubMed] [Google Scholar]
- 35.Martínez-González MA, López-Fontana C, Varo JJ, Sánchez-Villegas A, Martinez JA. Validation of the Spanish version of the physical activity questionnaire used in the Nurses’ Health Study and the Health Professionals’ Follow-up Study. Public Health Nutr. 2005;8:920–7. doi: 10.1079/phn2005745. [DOI] [PubMed] [Google Scholar]
- 36.Schuit AJ, Schouten EG, Westerterp KR, Saris WH. Validity of the physical activity scale for the elderly (PASE): according to energy expenditure assessed by the doubly labeled water method. J Clin Epidemiol. 1997;50:541–6. doi: 10.1016/s0895-4356(97)00010-3. [DOI] [PubMed] [Google Scholar]
- 37.Desquilbet L, Mariotti F. Dose-response analyses using restricted cubic spline functions in public health research. Stat Med. 2010;29:1037–57. doi: 10.1002/sim.3841. [DOI] [PubMed] [Google Scholar]
- 38.Asgari-Taee F, Zerafati-Shoae N, Dehghani M, Sadeghi M, Baradaran HR, Jazayeri S. Association of sugar sweetened beverages consumption with non-alcoholic fatty liver disease: a systematic review and meta-analysis. Eur J Nutr. 2019;58:1759–69. doi: 10.1007/s00394-018-1711-4. [DOI] [PubMed] [Google Scholar]
- 39.Sekkarie A, Welsh JA, Northstone K, Stein AD, Ramakrishnan U, Vos MB. Associations between free sugar and sugary beverage intake in early childhood and adult NAFLD in a population-based UK Cohort. Children (Basel, Switzerland) 2021;8:290. doi: 10.3390/children8040290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Aeberli I, Gerber PA, Hochuli M, Kohler S, Haile SR, Gouni-Berthold I, et al. Low to moderate sugar-sweetened beverage consumption impairs glucose and lipid metabolism and promotes inflammation in healthy young men: a randomized controlled trial. Am J Clin Nutr. 2011;94:479–85. doi: 10.3945/ajcn.111.013540. [DOI] [PubMed] [Google Scholar]
- 41.McGlynn ND, Khan TA, Wang L, Zhang R, Chiavaroli L, Au-Yeung F, et al. Association of low- and no-calorie sweetened beverages as a replacement for sugar-sweetened beverages with body weight and cardiometabolic risk: a systematic review and meta-analysis. JAMA Netw Open. 2022;5:e222092.. doi: 10.1001/jamanetworkopen.2022.2092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Te Morenga LA, Howatson AJ, Jones RM, Mann J. Dietary sugars and cardiometabolic risk: systematic review and meta-analyses of randomized controlled trials of the effects on blood pressure and lipids. Am J Clin Nutr. 2014;100:65–79. doi: 10.3945/ajcn.113.081521. [DOI] [PubMed] [Google Scholar]
- 43.Stanhope KL, Medici V, Bremer AA, Lee V, Lam HD, Nunez MV, et al. A dose-response study of consuming high-fructose corn syrup-sweetened beverages on lipid/lipoprotein risk factors for cardiovascular disease in young adults. Am J Clin Nutr. 2015;101:1144–54. doi: 10.3945/ajcn.114.100461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Raben A, Møller BK, Flint A, Vasilaris TH, Christina Møller A, Juul Holst J, et al. Increased postprandial glycaemia, insulinemia, and lipidemia after 10 weeks’ sucrose-rich diet compared to an artificially sweetened diet: a randomised controlled trial. Food Nutr Res. 2011;55: 10.3402/fnr.v55i0.5961 [DOI] [PMC free article] [PubMed]
- 45.Zhang C, Li L, Zhang Y, Zeng C. Recent advances in fructose intake and risk of hyperuricemia. Biomed Pharmacother. 2020;131:110795. doi: 10.1016/j.biopha.2020.110795. [DOI] [PubMed] [Google Scholar]
- 46.Jensen T, Abdelmalek MF, Sullivan S, Nadeau KJ, Green M, Roncal C, et al. Fructose and sugar: a major mediator of non-alcoholic fatty liver disease. J Hepatol. 2018;68:1063–75. doi: 10.1016/j.jhep.2018.01.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Sylvetsky AC, Rother KI. Trends in the consumption of low-calorie sweeteners. Physiol Behav. 2016;164:446–50. doi: 10.1016/j.physbeh.2016.03.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Liauchonak I, Qorri B, Dawoud F, Riat Y, Szewczuk MR. Non-nutritive sweeteners and their implications on the development of metabolic syndrome. Nutrients. 2019;11:644.. doi: 10.3390/nu11030644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Malik VS, Hu FB. The role of sugar-sweetened beverages in the global epidemics of obesity and chronic diseases. Nat Rev Endocrinol. 2022;18:205–18. doi: 10.1038/s41574-021-00627-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.O’Connor D, Pang M, Castelnuovo G, Finlayson G, Blaak E, Gibbons C, et al. A rational review on the effects of sweeteners and sweetness enhancers on appetite, food reward and metabolic/adiposity outcomes in adults. Food Funct. 2021;12:442–65. doi: 10.1039/d0fo02424d. [DOI] [PubMed] [Google Scholar]
- 51.Khan TA, Sievenpiper JL. Controversies about sugars: results from systematic reviews and meta-analyses on obesity, cardiometabolic disease and diabetes. Eur J Nutr. 2016;55:25–43. doi: 10.1007/s00394-016-1345-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Crowe-White K, Parrott JS, Stote KS, Gutschall M, Benson-Davies S, Droke E, et al. Metabolic impact of 100% fruit juice consumption on antioxidant/oxidant status and lipid profiles of adults: an evidence-based review. Crit Rev Food Sci Nutr. 2017;57:152–62. doi: 10.1080/10408398.2015.1102861. [DOI] [PubMed] [Google Scholar]
- 53.Fedchuk L, Nascimbeni F, Pais R, Charlotte F, Housset C, Ratziu V, et al. Performance and limitations of steatosis biomarkers in patients with nonalcoholic fatty liver disease. Aliment Pharmacol Ther. 2014;40:1209–22. doi: 10.1111/apt.12963. [DOI] [PubMed] [Google Scholar]
- 54.Sviklāne L, Olmane E, Dzērve Z, Kupčs K, Pīrāgs V, Sokolovska J. Fatty liver index and hepatic steatosis index for prediction of non-alcoholic fatty liver disease in type 1 diabetes. J Gastroenterol Hepatol. 2018;33:270–6. doi: 10.1111/jgh.13814. [DOI] [PubMed] [Google Scholar]
- 55.Huang X, Xu M, Chen Y, Peng K, Huang Y, Wang P, et al. Validation of the fatty liver index for nonalcoholic fatty liver disease in middle-aged and elderly Chinese. Medicine. 2015;94:e1682. doi: 10.1097/md.0000000000001682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Yang B-L, Wu W-C, Fang K-C, Wang Y-C, Huo T-I, Huang Y-H, et al. External validation of fatty liver index for identifying ultrasonographic fatty liver in a large-scale cross-sectional study in Taiwan. PLoS ONE. 2015;10:e0120443.. doi: 10.1371/journal.pone.0120443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Zhu J, He M, Zhang Y, Li T, Liu Y, Xu Z, et al. Validation of simple indexes for nonalcoholic fatty liver disease in western China: a retrospective cross-sectional study. Endocr J. 2018;65:373–81. doi: 10.1507/endocrj.EJ17-0466. [DOI] [PubMed] [Google Scholar]
- 58.European Association for the Study of the Liver (EASL), European Association for the Study of Diabetes (EASD) and European Association for the Study of Obesity (EASO) EASL-EASD-EASO Clinical Practice Guidelines for the management of non-alcoholic fatty liver disease. J Hepatol. 2016;64:1388–402. 10.1016/j.jhep.2015.11.004 [DOI] [PubMed]
- 59.Eslam M, Sarin SK, Wong VW-S, Fan J-G, Kawaguchi T, Ahn SH, et al. The Asian Pacific Association for the Study of the Liver clinical practice guidelines for the diagnosis and management of metabolic associated fatty liver disease. Hepatol Int. 2020;14:889–919. doi: 10.1007/s12072-020-10094-2. [DOI] [PubMed] [Google Scholar]
- 60.VanderWeele TJ, Ding P. Sensitivity analysis in observational research: introducing the E-value. Ann Intern Med. 2017;167:268–74. doi: 10.7326/m16-2607. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request and after approval from the relevant partners.