Abstract
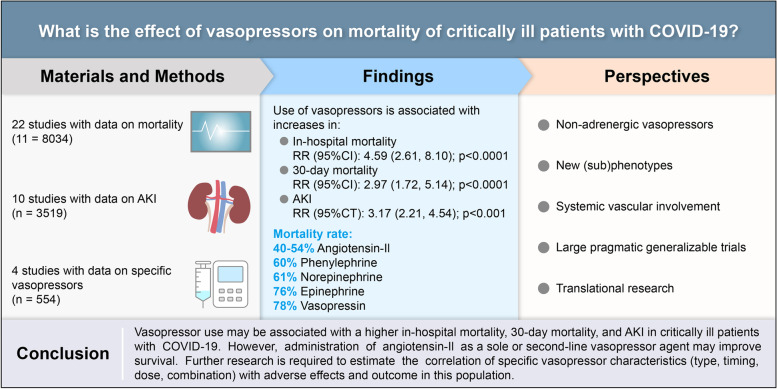
Exogenous catecholamines may have pronounced side effects and affect physiological cascades. The aim of this study was to investigate the effect of vasopressors on mortality of critically ill patients with coronavirus disease 2019 (COVID-19). A systematic search of PubMed, Scopus, and ClinicalTrials.gov was conducted for relevant articles until December 2022. Eligibility criteria were randomized controlled and non-randomized trials. The primary outcome was in-hospital and 30-day mortality. The quality of studies was assessed using the Methodological Index for Non-Randomized Studies (MINORS) tool, while paired meta-analysis was used to estimate the pooled risk ratios (RR) along with their 95% Confidence Interval (95% CI). Analyses of 22 studies (n = 8034) revealed that vasopressor use is associated with mortality compared to no vasopressor therapy [RR (95%CI): 4.30 (3.21, 5.75); p < 0.001]. In-hospital and 30-day mortality are significantly higher in patients who receive vasopressors [RR (95%CI): 4.60 (2.47, 8.55); p < 0.001 and RR (95%CI): 2.97 (1.72, 5.14); p < 0.001, respectively]. Also, analyses of data from 10 studies (n = 3519) revealed that vasopressor use is associated with acute kidney injury [RR (95%CI): 3.17 (2.21, 4.54); p < 0.001]. In conclusion, current use of vasopressors in critically ill patients with COVID-19 may be associated with higher in-hospital mortality, 30-day mortality, and incidence rate of acute kidney injury. Further research is required to estimate the correlation of specific vasopressor characteristics (type, timing, dose, combination) with adverse effects and mortality in this population.
Graphical Abstract
Supplementary Information
The online version contains supplementary material available at 10.1007/s44254-023-00013-7.
Keywords: Covid-19, Critically ill, Hemodynamics, Vasopressor, Intensive care, Mortality
Introduction
Mounting evidence suggest that coronavirus disease 2019 (COVID-19) should be perceived as a new entity with its own characteristics and distinct pathophysiology, including complex immuno-inflammatory, thrombotic, and parenchymal derangements [1]. The cytokine storm and the dysregulation of host response are more severe in COVID-19-related acute respiratory distress syndrome (ARDS) than in ARDS of other causes [2–4]. SARS-CoV-2 not only infects the respiratory tract, but also injures the vascular endothelium and epithelium [5, 6].
Most critically ill patients with COVID-19 need hemodynamic support that may still be guided by the current, non-covid, surviving sepsis campaign guidelines recommending the use of vasopressors to optimize mean arterial pressure (MAP) and cardiac output and provide adequate organ perfusion [7–9]. Most of these medications improve the hemodynamic function through enhancement of the adrenergic pathway; however, they may have important side-effects due to excessive adrenergic stimulation [10–12]. Of note, exogenous catecholamines can have a pronounced impact on inflammation and immunosuppression, metabolism, endothelial lesion, platelet activation, and coagulation [13]. As critically ill patients with COVID-19 are characterized by a similar pathophysiology, exogenous vasopressors could further dysregulate their physiological cascades and aggravate outcome [14].
We therefore performed a systematic review and meta-analysis to investigate the effect of vasopressors on mortality of critically ill patients with COVID-19.
Material and methods
The protocol was registered in the PROSPERO international prospective register of systematic reviews on 13 December 2021 (CRD42021297595). This systematic review and meta-analysis was designed according to the preferred reporting items for systematic reviews and meta-analyses (PRISMA) checklist (Additional file 1: Appendix A) [15].
Inclusion and exclusion criteria
The inclusion criteria of the current systematic review and meta-analysis were: (1) randomized controlled trials (RCTs) and observational studies; (2) critically ill patients admitted to the intensive care (ICU) or high dependency unit (HDU), including patients admitted through the Emergency Department (ED); (3) adults (≥ 18 years old) hospitalized primarily for COVID-19; (4) SARS-CoV-2 infection confirmed by reverse transcription polymerase chain reaction test of nasopharyngeal or oropharyngeal samples; and (5) vasopressor vs. no vasopressor administration. We excluded animal studies, case reports, review papers, editorials, abstracts, white papers, and non-English literature. We also excluded studies about pediatric patients and non-ICU/HDU/ED patients.
Outcomes of interest and data extraction
The primary outcome was in-hospital and 30-day mortality. Secondary outcome was to investigate (1) the hemodynamic profiles of patients at first measuring point and after six hours [heart rate, MAP, central venous pressure (CVP), urinary output, blood lactate levels, cardiac output or cardiac index, systemic vascular resistance index, central venous oxygen saturation, oxygen delivery index, and oxygen consumption index]; (2) the number of participants who achieved the target MAP; (3) time to achieve the target MAP; (4) adverse events including arrhythmia, acute myocardial infarction, cardiac arrest, acute mesenteric ischemia, digital ischemia, acute kidney injury (AKI); (5) vasopressor-free days; (6) ICU or HDU length of stay; (7) duration of mechanical ventilation; (8) ventilator free days; (9) hospital length of stay; and (10) all-cause mortality at 90-days.
The data from each study were extracted by two independent authors (MM, GM) with a customized format. Any disagreements between the two independent authors were resolved by four other authors (EL, IP, NP, AC). Publication details (authors, year), study information (design, population, department of admission, follow-up, inclusion–exclusion criteria, number of cases/cohort-size, and subgroups), hemodynamic profile (heart rate, MAP, CVP, urinary output, blood lactate levels, cardiac output or cardiac index, systemic vascular resistance index, central venous oxygen saturation, oxygen delivery index, oxygen consumption index) at first measuring point and six hours after vasopressor use, the number of participants who achieved the target MAP and time to achieve the target MAP, adverse events, vasopressor-free days, ICU length of stay, hospital length of stay, duration of mechanical ventilation, ventilator-free days, all-cause mortality in all groups at 28 or 30 days, and all-cause mortality at 90 days were extracted in a pre-designed excel spreadsheet. The definition used for AKI and the mortality follow-up timepoints for each study are presented in Additional file 3: Appendix C1. Authors of studies with missing data were contacted in an attempt to obtain relevant data.
Search strategy
The search strategy was intended to explore all available published and unpublished studies from January 2020 to January 2022. A comprehensive initial search was employed in PubMed (MEDLINE), Scopus, and ClinicalTrials.gov databases by two independent investigators (MM, GM) followed by an analysis of the text words contained in Title/Abstract and indexed terms. A second search was conducted by combining free text words (vasopressor, epinephrine, norepinephrine, phenylephrine, vasopressin, dopamine, angiotensin-II, covid-19, critically ill, intensive care) and indexed terms with Boolean operators. Finally, a third search was conducted with the reference lists of all identified reports and articles for additional studies. After the initial data was compiled, a refresh repeat search until December 31, 2022 was performed. Additional file 2 (Appendix B) presents the exact search algorithm used for all databases.
Assessment of methodological quality
Articles identified for retrieval were assessed by two independent authors (MM, GM) for methodological quality before inclusion in the review using standardized critical appraisal tools. The quality of the included observational studies was assessed using the Methodological Index for Non-Randomized Studies (MINORS) tool [16], while the Risk of Bias 2.0 (RoB 2.0) tool was used for RCTs [17]. Any disagreements between the authors appraising the articles were resolved through discussion with the other authors.
Data analysis and synthesis
A paired meta-analysis was used to estimate the pooled risk ratios (RR) along with their 95% Confidence Interval (95% CI). Based on the presence of statistical heterogeneity, the meta-analysis was conducted according to fixed- or random effect models. The statistical heterogeneity was estimated by the use of the Cochran’s Q and I2 indices. When I2 > 50% and/or PQ < 0.10, the random effects model was used, otherwise the fixed effects model was implemented [18]. Funnel plots as well as the Begg’s test were used to determine the existence of publication bias [19, 20]. The statistical significance was set at p < 0.05. All statistical analyses were performed in Review Manager (Rev-Man) [Computer program], Version 5.3. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Results
Altogether, 1495 relevant citations were identified and screened, while 93 studies were included in our final assessment for possible data extraction (Fig. 1). In total, data extraction was possible in 34 studies [21–54].
Fig. 1.
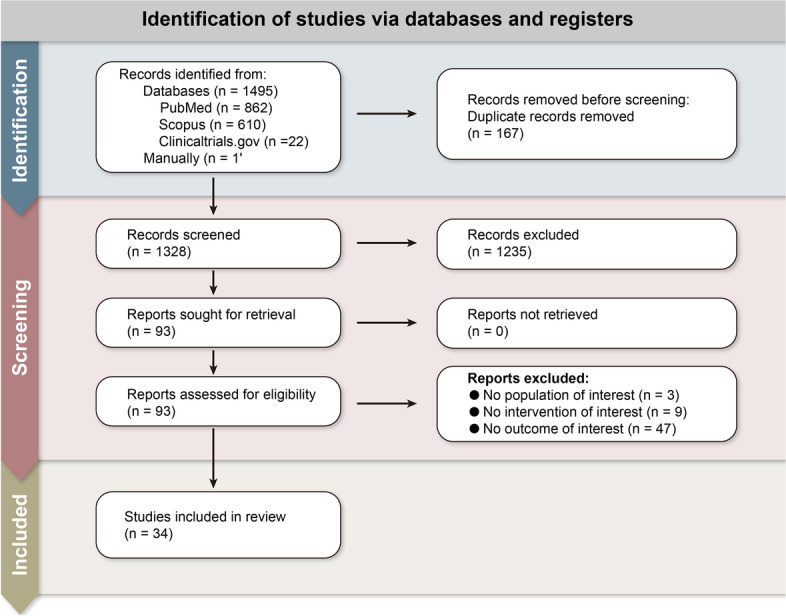
Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) diagram
Study characteristics
All the 34 included studies were observational in their design [21–54]. Twenty studies included only patients admitted to the ICU [21, 23, 25, 26, 28, 31–33, 36–40, 44–48, 53, 54], five studies included patients admitted to a COVID-19-dedicated HDU [29, 30, 34, 35, 43], eight studies included patients who were admitted to both HDU and ICU [22, 24, 27, 41, 42, 49, 50, 52], and one study included Emergency Department patients who were later admitted either to ICU [51]. Thirty-one studies included data about patients who received vs. patients who did not receive vasopressors [21–38, 40–49, 51, 53, 54] and were included in review. Moreover, three studies included patients who received angiotensin-II [39, 50, 52] and, out of those, two compared the use of angiotensin-II with other vasopressors [50, 52] (Additional file 3: Appendix C2). Table 1 summarizes the main characteristics of the included studies.
Table 1.
Main characteristics of the included studies
| Authors Country, YOP Study Design | Department | Number of patients | Αge Mean ± SD / Median (IQR) | Μale (%) / Female (%) | |
|---|---|---|---|---|---|
| Received vasopressors | No vasopressors | ||||
| Ionescu et al. USA, 2021 Retrospective [21] | ICU | 191 | 90 | 61 ± 13.9 | 154 (54.8%) / 127 (45.2%) |
| Pelayo et al. USA, 2020 Retrospective [22] | ICU + HDU | 42 | 181 | 65.91 ± 14.95 | 116 (52%) / 107 (48%) |
| Lowe et al. UK, 2021 Retrospective [23] | ICU | 49 | 32 | 57 ± 18 | 50 (61.7%) / 31 (38.3%) |
| Hansrivijit et al. USA, 2021 Retrospective [24] | ICU + HDU | 53 | 230 | 64.1 ± 15.9 | 159 (56.2%) / 124 (43.8%) |
| Mesquida et al. Spain/Mexico/Brazil, 2021 Prospective [25] | ICU + IRCU | 12 | 59 | 59 ± 13 | 51 (69.9%) / 22 (30.1%) |
| Ghosn et al. UAE, 2021 Retrospective [26] | ICU | 66 | 44 | 50 (40–59) | 98 (89.1%) / 12 (10.9%) |
| Farooqui et al. Saudi Arabia, 2021 Retrospective [27] | ICU + HDU | 249 | 776 | 55.8 ± 18.52 | 582 (56.8%) / 443 (43.2%) |
| Neves et al. Brazil, 2021 Retrospective [28] | ICU | 54 | 41 | 64.9 ± 15.1 | 61 (64.2%) / 34 (35.8%) |
| Bernardo et al. Portugal, 2021 Retrospective [29] | HDU | 18 | 526 | 68.9 ± 17.9 | 298 (54.8%) / 246 (45.2%) |
| Hardenberg et al. Germany, 2021 Retrospective [30] | HDU | 95 | 128 | 62 (51–75) | 147 (65.9%) / 76 (34.1%) |
| Geri et al. France, 2021 Retrospective [31] | ICU | 165 | 214 | 62 (53.69) | 291 (76.8%) / 88 (23.2%) |
| Namendys-Silva et al. Mexico, 2021 Retrospective [32] | ICU | 139 | 25 | 57.3 ± 13.7 | 114 (69.5%) / 50 (30.5%) |
| Auld et al. USA, 2021 Retrospective [33] | ICU | 143 | 74 | 64 (54–73) | 119 (54.8%) / 98 (45.2%) |
| Nabors et al. USA, 2021 Retrospective [34] | HDU | 23 | 64 | 86 (80–105) | 48 (55.2%) / 39 (44.8%) |
| Salacup et al. USA, 2021 Retrospective [35] | HDU | 49 | 193 | 66 (58–76) | 123 (50.8%) / 119 (49.2%) |
| Nasrulah et al. USA, 2021 Retrospective [36] | ICU | 24 | 34 | 62 (54–73) | 37 (63.8%) / 21 (36.2%) |
| Sjostorm et al. Sweden, 2021 Prospective [37] | ICU | 40 | 13 | 59 (33–76) | 39 (73.6%) / 14 (26.4%) |
| Ismail et al. UAE, 2021 Retrospective [38] | ICU | 176 | 195 | 53 ± 13 | 314 (84.6%) / 57 (15.4%) |
| Osofu-Barko et al.a USA, 2021 Retrospective [39] | ICU | 10 | Not available | 64.5 ± 6.15 | 9 (90%) / 1 (10%) |
| Ramkumar et al. India, 2021 Prospective [40] | ICU | 29 | 31 | 50 (37.5–63) | 42 (70%) / 18 (30%) |
| Biccard et al. (ACCCOS) Africab, 2021 Prospective [41] | ICU + HDU | 931 | 2155 | 56 ± 16.11 | 1890 (60.6%) / 1228 (39.4%) |
| Mammen et al. India, 2021 Secondary analysis of RCT [42] | ICU + HDU | 18 | 433 | 51 ± 12.4 | 346 (76.7%) / 105 (23.3%) |
| Andrade et al. USA, 2021 Retrospective [43] | HDU | 63 | 221 | 67 ± 14.5 | 155 (54.6%) / 129 (45.4%) |
| Chand et al. USA, 2020 Retrospective [44] | ICU | 233 | 67 | 58.2 ± 12.6 | 182 (60.7%) / 118 (39.3%) |
| Bezzera et al. Brazil, 2021 Retrospective [45] | ICU | 325 | 99 | 66.42 ± 13.79 | 251 (59.2%) / 173 (40.8%) |
| Dang et al. USA, 2021 Retrospective [46] | ICU | 63 | 26 | 65 (57–70) | 52 (58.4%) / 37 (41.6%) |
| Gundogan et al. Turkey, 2021 Retrospective [47] | ICU | 173 | 248 | 67 (57–76) | 251 (59.6%) / 170 (40.4%) |
| Estella et al. Spain, 2021 Prospective [48] | ICU | 419 | 3 | 63 (54–71) | 305 (72.3%) / 117 (27.7%) |
| Gadhiya et al. USA, 2021 Retrospective [49] | ICU + HDU | 53 | 230 | 64.1 ± 15.9 | 159 (56.2%) / 124 (43.8%) |
| Serpa Neto et al.a Multicentric study, 2022 Prospective [50] | ICU + HDU | 65 | 67 | 61 (53–67) | 105 (79.5%) / 27 (20.5%) |
| Brandão Neto et al. Brazil, 2021 Prospective [51] | ED and then transferred to ICU + HDU | 179 | 327 | 60.1 ± 15.1 | 290 (57.3%) / 216 (42.7%) |
| Leisman et al.a USA, 2020 Retrospective [52] | ICU + HDU | 10 | 19 | 56 ± 14 | 19 (65.5%) / 10 (34.5%) |
| Burrell et al. Australia, 2021 Prospective [53] | ICU | 111 | 93 | 63.5 (53–72) | 140 (68.6%) / 64 (31.4%) |
| Ramos et al. Multicentric study, 2022 Retrospective [54] | ICU | 337 | 308 | 61.4 ± 16.6 | 387 (60%) / 258 (40%) |
YOP Year of Publication, SD Standard Deviation, IQR Interquartile Range, USA United States of America, ICU Intensive Care Unit, HDU High Dependency Unit, UK United Kingdom, IR3CU Intensive Respiratory Care Unit, UAE United Arab Emirates, ED Emergency Department
aThese studies included patients who received angiotensin-II
b10 African Countries
Synthesis including all patients
Primary outcome
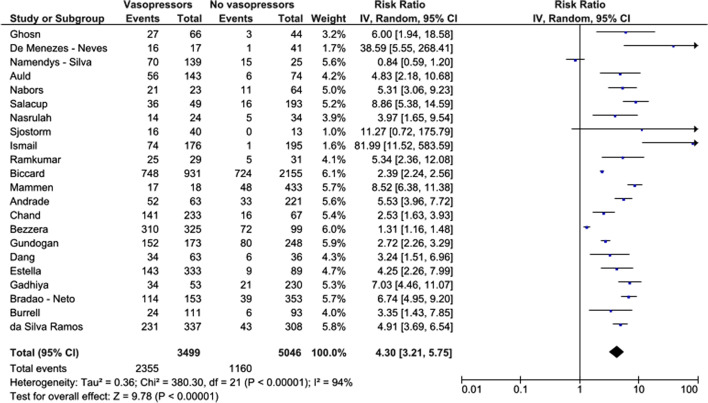
Twenty-two out of the 34 included studies provided data on in-hospital mortality in patients who received vs. patients who did not receive vasopressors, resulting in a total population of 8034 individuals [26, 28, 32–38, 40–49, 51, 53, 54]. Due to high heterogeneity (I2: 94%, PQ < 0.001), the random-effects model was implemented. Vasopressor use is associated with mortality in patients who received vasopressors compared to those who did not receive vasopressor therapy [RR (95%CI): 4.30 (3.21, 5.75); p < 0.0001] (Fig. 2). Visual inspection of the funnel plot (Additional file 4: Appendix D1) and Begg’s test (p = 0.93) did not reveal significant publication bias.
Fig. 2.
Effect of vasopressor use on mortality of critically ill patients with COVID-19
We performed subgroup analyses based on the department of admission. The results for all three subgroups, namely ICU [RR (95%CI): 3.64 (2.44, 5.44); p < 0.0001], HDU [RR (95%CI): 6.25 (4.63, 8.44); p < 0.0001], and ICU + HDU [RR (95%CI): 5.52 (2.51, 12.15); p < 0.0001], remained statistically significant for higher mortality rates in patients who received vasopressors.
Subgroup analyses were also performed based on the mortality follow-up timepoints. Only the in-hospital and 30-day mortality subgroups had three or more studies that allowed data extraction and analysis. The in-hospital and 30-day mortality were significantly higher in patients who received vasopressors [RR (95%CI): 4.59 (2.61, 8.10); p < 0.0001 and RR (95%CI): 2.97 (1.72, 5.14); p < 0.0001, respectively].
Effect of major vasopressors on mortality
Four studies provided data on mortality based on the specific vasopressor(s) administered [39, 44, 50, 52]. The highest mortality rate was observed in patients treated with vasopressin or epinephrine (78% and 76%, respectively) [44]. Three of those studies investigated the role of angiotensin-II as a sole or second-, third-, fourth-, or fifth-line vasopressor agent [39, 50, 52]. These studies showed the lowest mortality rate. The relevant data are depicted in Table 2.
Table 2.
Data on mortality based on major vasopressors
| Authors Country, YOP Study Design | Intervention Group: Deaths / All (%) | Comparator Group: Deaths / All (%) |
|---|---|---|
| Chand et al. USA, 2020 Retrospective [44] |
Any vasopressor support: 141 / 233 (61%) Norepinephrine ± other vasopressors: 138 / 226 (61%) Phenylephrine ± other vasopressors: 53 / 89 (60%) Vasopressin ± other vasopressors: 81 / 104 (78%) Epinephrine ± other vasopressors: 19 / 25 (76%) |
No vasopressor support: 16 / 67 (24%) |
| Leisman et al. USA, 2020 Retrospective [52] | Angiotensin-II ± other vasopressors: 4 / 10 (40%) | Other vasopressors: 10 / 19 (53%) |
| Serpa Neto et al. Multicentric study, 2022 Prospective [50] | Angiotensin-II ± norepinephrine: 35 / 65 (54%) | Other vasopressors: 27 / 67 (40%) |
| Ofosu-Barko et al. USA, 2021 Retrospective [39] | Angiotensin-II + other vasopressors: 4 / 10 (40%) | - |
Secondary outcomes – acute kidney injury
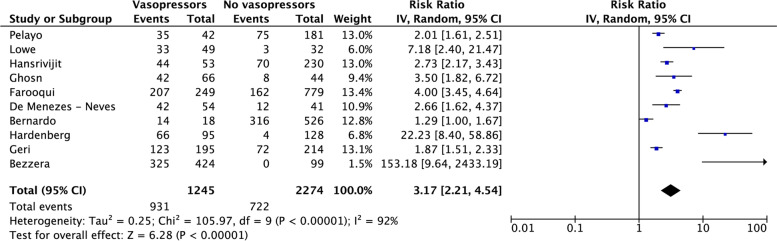
Ten studies provided data on AKI in patients who received vs. patients who did not receive vasopressors, resulting in a total population of 3519 individuals [22–24, 26–31, 45]. Due to high heterogeneity (I2: 92%, PQ < 0.001), the random-effects model was implemented. Vasopressor use is associated with AKI in patients who receive vasopressors compared to those who do not receive vasopressor therapy [RR (95%CI): 3.17 (2.21, 4.54); p < 0.001] (Fig. 3).
Fig. 3.
Effect of vasopressor use on the incidence of acute kidney injury
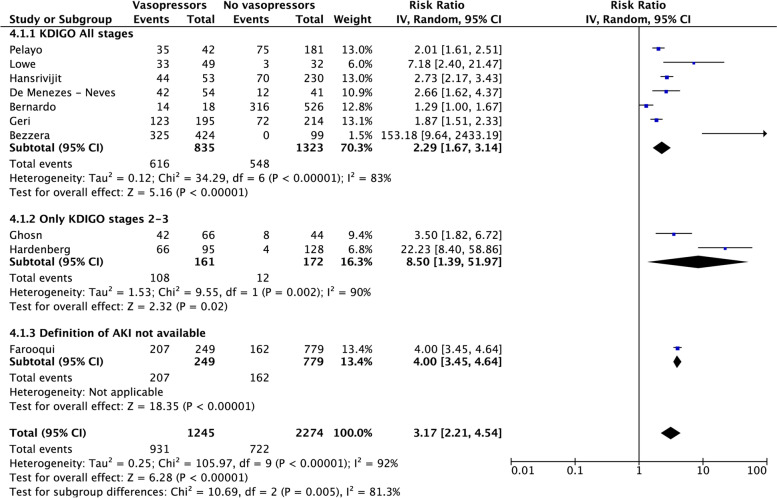
Subgroup analyses were performed based on the definition of AKI that was used in the included studies. Only the subgroup with patients at all KDINGO stages included more than three studies, allowing for meta-analysis to be performed. Specifically, vasopressor use is associated with AKI in patients who receive vasopressors compared to those who do not receive vasopressor therapy [RR (95%CI): 2.29 (1.67–3.14); p < 0.001] (Fig. 4).
Fig. 4.
Subgroup analyses based on the definition of acute kidney injury
Other secondary outcomes
No data were identified for the remaining secondary outcomes.
Sensitivity analysis
A sensitivity analysis was performed for both outcomes based on the implemented meta-analyses model (fixed vs. random effect); in both cases, the sensitivity analysis confirmed the robustness of the findings. The synthesized results of the present systematic review and meta-analysis together with the results of the sensitivity analysis are depicted in Table 3.
Table 3.
Synthesis of the results with a sensitivity analysis (fixed vs. random effects)
| Outcomes | RR (CI); p value Model used |
Sensitivity analysis |
|---|---|---|
| Alternate model | ||
| Mortality (all timepoints) |
4.30 (3.21, 5.75); < 0.0001 Random effects |
2.51 (2.39, 2.64); < 0.0001 Fixed effects |
| Mortality: subgroups based on department | ||
| ICU |
3.64 (2.44, 5.44); < 0.0001 Random effects |
1.94 (1.78, 2.12); < 0.0001 Fixed effects |
| HDU |
6.25 (4.63, 8.44); < 0.0001 Random effects |
6.16 (4.81, 7.90); < 0.0001 Fixed effects |
| ICU & HDU |
5.52 (2.51, 12.15); < 0.0001 Random effects |
2.71 (2.55, 2.89); < 0.0001 Fixed effects |
| Mortality: subgroups based on mortality timepoints | ||
| In hospital |
4.59 (2.61, 8.10); < 0.0001 Random effects |
2.30 (2.10, 2.52); < 0.0001 Fixed effects |
| In ICU |
4.07 (2.28, 7.28); < 0.0001 Random effects |
4.07 (2.28, 7.28); < 0.0001 Fixed effects |
| 30-day |
2.97 (1.72, 5.14); < 0.0001 Random effects |
2.40 (2.25, 2.57); < 0.0001 Fixed effects |
| 90-day |
2.78 (1.98, 3.91); < 0.0001 Random effects |
2.74 (2.27, 3.31); < 0.0001 Fixed effects |
| 6-weeks |
4.25 (2.26, 7.99); < 0.0001 Random effects |
4.25 (2.26, 7.99); < 0.0001 Fixed effects |
| Not available |
7.94 (5.72, 311.02); < 0.0001 Random effects |
8.09 (6.16, 10.62); < 0.0001 Fixed effects |
| AKI (all timepoints) |
3.17 (2.21, 4.54); < 0.0001 Random effects |
2.64 (2.42, 2.88); < 0.0001 Fixed effects |
| AKI: subgroups based on KDIGO stage | ||
| KDIGO all stages |
2.29 (1.67, 3.14); < 0.0001 Random effects |
2.02 (1.81, 2.26); < 0.0001 Fixed effects |
| KDIGO stages 2–3 |
8.50 (1.39, 51.97); 0.02 Random effects |
6.21 (3.61, 10.68); < 0.0001 Fixed effects |
| Not available KDIGO |
4.00 (3.45, 4.64); < 0.0001 Random effects |
4.00 (3.45, 4.64); < 0.0001 Fixed effects |
ICU Intensive Care Unit, HDU High Dependency Unit, AKI Acute Kidney Injury
Risk of bias, quality of evidence
The overall quality of the studies, as assessed by the MINORS tool, ranged between moderate and high. The exact score for each study is available in Additional file 3: Appendix C3. In addition, visual inspection of the funnel plot (Additional file 4: Appendix D2) and the Begg’s test (p = 0.18) did not reveal significant publication bias for the studies included in the AKI analysis. All included studies were observational non-randomized studies with high heterogeneity that does not allow to derive an estimate of overall effect. According to GRADE criteria, the quality of evidence provided by the studies was low.
Discussion
Many high-quality RCTs have addressed the effect of vasopressors on the outcomes of non-covid patients, yet their impact in patients with COVID-19 had not been studied so far. The most important finding of this systematic review and meta-analysis is the association between vasopressor therapy and in-hospital mortality, 30-day mortality, and incidence rate of AKI as compared to no vasopressor therapy in critically ill patients with COVID-19. Although these results are based on non-randomized evidence, they raise significant concerns for the routine management of these individuals.
The mortality of critically ill patients with COVID-19 remains high [55, 56]. A main cause is the characteristics of the SARS-CoV-2 infection, which can rapidly affect many organs including the cardiovascular system [57]. Although administration of vasopressors is a fundamental treatment of hypotension, the traditional (non-covid) hemodynamic management and the adverse effects of vasoactive agents may be associated with complications and poor outcome in patients with COVID-19. Indeed, the present analysis revealed an association between vasopressor use and mortality. This association may be coincidental due to the severity of critical illness. However, catecholamines exert numerous biological effects including effects on the immune and hematological systems, the renin–angiotensin–aldosterone system, the cardiovascular and respiratory systems, and others [9, 14]. This complex interplay that may have a detrimental effect on survival.
Of the 34 studies included in this systematic review and meta-analysis, only four studies provided data on specific vasopressors. The highest mortality rate was observed in patients receiving vasopressin or epinephrine. Although one can appraise that these patients had severe shock necessitating second- and third-line vasopressors [7], these observations merit further discussion. Epinephrine is well-known for its adverse effects in non-covid and COVID-19 patients [58], but our observations regarding vasopressin are quite interesting. Although vasopressin infusion reduces total norepinephrine-equivalent dose requirements and may be renal and pulmonary vasculature sparing [59], there is evidence showing a pronounced activation of the vasopressin system in COVID-19 patients and that molecular complexes form between the SARS-CoV-2 spike protein, soluble angiotensin-converting enzyme-2 (ACE2), and vasopressin, facilitating cellular infection and aggravating outcome [60, 61]. However, data from a small clinical cohort did not show a clinically relevant effect of vasopressin infusion on viral mRNA levels in critically ill patients with COVID-19 who were not treated with corticosteroids or interleukin-6 antagonists [59]. Considering the high heterogeneity of the extracted data in the present study and that vasopressin is suggested as a second-line vasopressor in the latest international guidelines [7, 9], further research is required to establish the therapeutic efficiency of vasopressin in critically ill patients with COVID-19.
Another intriguing finding is the low mortality rate in patients receiving angiotensin-II as a sole or second-line vasopressor agent. Serpa Neto et al. showed a potentially positive effect of angiotensin-II on blood pressure and fraction of inspired oxygen in COVID-19 patients, but they did not collect data regarding treatment with steroids or other drugs, which may have affected their results [50]. Ofosu-Barko et al. and Leisman et al. reported that angiotensin-II treatment was associated with rapid improvement in multiple physiologic indices [39, 52]. The rationale for angiotensin-II therapy is based on decreasing the expression of the ACE2 receptors, which facilitate the entry of SARS-CoV-2 into cells [62, 63]. Of note, the progressive loss of ACE2 in COVID-19 shifts the system to an overall higher angiotensin level due to the impaired ability of ACE2 to degrade it, which may explain the hemodynamic stability during the initial stages of the disease [64]. Moreover, recent experimental evidence suggests that angiotensin-II administration is associated with a similar level of cardiovascular resuscitation, less myocardial oxygen consumption, and less inflammation compared to norepinephrine [65]. Taking into consideration the characteristics of angiotensin-II, more research is needed to evaluate its potent effects in COVID-19-related shock.
A meta-analysis of RCTs with non-covid patients reported that vasopressor therapy is not associated with differences in mortality in the overall population, while prophylactic administration in patients with vasodilatory shock may improve survival [66]. In addition, a Cochrane systematic review found no evidence of substantial differences in total mortality between several vasopressors [67]. Nevertheless, vasopressors are a heterogeneous class of drugs with powerful and immediate hemodynamic effects, and each drug has advantages and disadvantages. These characteristics are particularly important in patients with COVID-19 who are characterized by unique pathophysiological disturbances and different hemodynamic phenotypes that necessitate a thorough understanding of the underlying complex pathophysiology and careful selection and administration of vasoactive agents.
In COVID-19, the progressive hypoxemia initially increases cardiac output and capillary recruitment, which maintain microcirculatory oxygen-extraction capacity by increasing red blood cell availability (silent hypoxia) [68–70]. However, microcirculatory flow decreases proportionally to the increasing inflammation, hypercoagulation, and thrombosis, eventually resulting in multi-organ failure [68, 71–73]. In the study by Mesquida et al., patients had important microcirculatory alterations, and the degree of these alterations correlated with the severity of the respiratory disease [25]. The relationship between MAP and organ blood flow may be different in critically ill patients with COVID-19 and improving only macrocirculation might be inadequate to maintain tissue perfusion. In these patients, vasopressor use can overwhelm endogenous receptor-mediated vessel regulation, further contributing to hemodynamic incoherence [71, 74], and therefore, hemodynamic management should focus on optimizing microcirculatory perfusion and oxygen delivery instead of attaining a predefined MAP target.
Limitations
This meta-analysis was based on observational studies, while the results are subject to confounding by indication. In addition, due to the lack of RCTs, the synthesis of all the available knowledge on the specific outcomes was difficult. This is an inherent problem to observational studies, and not least considering the difficulties of collecting data during the periods of surges across the globe. Moreover, we could not obtain individual data to reach a minimal level of evidence that could result on relevant findings. Therefore, the effects of different vasopressors may reflect differences in severity and/or practices.
Furthermore, most of the included studies were published before November 2021 and thus, we were not able to analyze the data according to COVID-19 surge. In addition, the level of heterogeneity was high; possible reasons are the baseline status of patients, comorbidities, severity of COVID-19, and hospital department, i.e., HDU, ICU, and ED. There were no data for adjusting the resulting odds ratios according to age, comorbidities, the presence of septic shock, or other known factors that affect ICU mortality. Also, most of the secondary outcomes could not be assessed. Another limitation is the heterogeneity of definitions of AKI that were used across different studies. Finally, non-English publications were not included. Therefore, the results of this systematic review and meta-analysis must be interpreted with caution. International registries should collect uniform data to evaluate the effect of vasopressors on mortality and other outcomes in critically ill patients with COVID-19.
Conclusions
Current use of vasopressors in critically ill patients with COVID-19 may be associated with higher in-hospital mortality, 30-day mortality, and incidence rate of AKI. The lower mortality rate in patients receiving angiotensin-II as a sole or second-line vasopressor agent is worth noting. Of note, the included studies were observational with high heterogeneity, which does not allow to derive an estimate of overall effect. Randomized controlled trials and translational research are required to estimate the correlation of specific vasopressor characteristics (type, timing, dose, combination) with adverse effects and mortality in this population.
Perspectives
The results of the present systematic review and meta-analysis suggest for early administration of low-dose vasopressors, with or without inodilator agents, in an effort to avoid excessive doses that could have detrimental effect on survival, especially at later disease stages. An alternative second-line vasopressor may be angiotensin-II. However, further immediate research is recommended to elucidate the effects of angiotensin-II and other vasopressors acting through pathways other than the adrenergic. These agents may be associated with a significant increase in survival.
A possible explanation for the association of vasopressors with mortality may lie in the microcirculation [68–77]. The physiological pulsatile shear stress from normal laminar flow has a pivotal role in maintaining normal endothelial function and the expression of ACE2s and other anticoagulant/antithrombotic or antioxidant substances [78]. However, dysfunctional endothelium resulting from turbulent flow displays a hypercoagulant/prothrombotic and pro‐oxidant state that impairs microcirculatory reactivity and flow [79]. Therefore, therapeutic approaches should consider the systemic vascular involvement, allowing an individualized, physiology-guided management. It is almost certain that there are distinct COVID-19 phenotypes/subphenotypes that include impairment of microvasculature as key feature, and their identification will have important therapeutic implications [80]. Of note, a high CVP in critically ill patients with COVID-19 impairs venous return and retrogradely increases post-capillary venular pressure which, together with the excessive vasopressor doses, impair capillary perfusion and increase the oxygen diffusion distance [81, 82]. Consequently, optimizing fluid administration is also crucial for improving tissue perfusion in this population.
The present systematic review and meta-analysis included data from observational studies. Further research and well-designed trials are necessary to investigate the effect of the type (catecholamine vs. non-catecholamine), timingof initiation, and infusion rates of vasopressors in order to develop more specific treatment strategies and integrate a more individualized approach in patients with COVID-19. Although designing and conducting RCTs on vasopressors may be difficult during a disease outbreak, the need for assessing their effect on outcomes of critically ill patients with COVID-19 is imperative. We recommend the use of animal models and the integration of translational research to aid in the identification of the most suitable vasopressor in this population and to better define homogenous target (sub)populations for trials [83–85]. Large pragmatic RCTs with very broad inclusion criteria can help improving the generalizability of our findings.
Supplementary Information
Additional file 1: Appendix A. PRISMA checklist.
Additional file 2: Appendix B. Algorithms used for all databases.
Additional file 3: Appendix C1. Definitions used for AKI and mortality follow-up time points. Appendix C2. Angiotensin-related data. Appendix C3. MINORS results for each study.
Additional file 4: Appendix D1. Funnel plot for mortality meta-analysis. Appendix D2. Funnel plot for AKI meta-analysis.
Acknowledgements
We would like to thank Dr. George L. Anesi, University of Pennsylvania Perelman School of Medicine, for responding to our request for additional study data.
Abbreviations
- COVID-19
Coronavirus disease 2019
- RR
Risk ratios
- CI
Confidence interval
- ARDS
Acute respiratory distress syndrome
- MAP
Mean arterial pressure
- RCT
Randomized controlled trial
- ICU
Intensive care unit
- HDU
High dependency unit
- ED
Emergency department
- CVP
Central venous pressure
- AKI
Acute kidney injury
- MINORS
Methodological index fornon-randomized studies
- ACE2
Angiotensin-converting enzyme 2
Authors’ contributions
Conceptualization: AC. Data curation: MM, GM. Formal analysis: MM, GM. Methodology: AC, MM, GM, EL, NP, IP. Project administration: AC. Visualization: MM, GM, EL, NP, IP, AC. Writing—original draft: AC. Writing—review & editing: AC, MM, GM, EL, NP, IP. The author(s) read and approved the final manuscript.
Funding
No funding received.
Availability of data and materials
Data can be made available upon request after publication through a collaborative process. Researchers should provide a methodically sound proposal with specific objectives in an approval proposal. Please contact the corresponding author for additional information.
Declarations
Ethics approval and consent to participate
Non-applicable.
Consent for publication
Non-applicable.
Competing interests
The author Prof. Dr. Athanasios Chalkias is a member of the Editorial Board in Anesthesiology and Perioperative Science. The paper was handled by the other Editor and had undergone rigorous peer review process. Prof. Dr. Athanasios Chalkias was not involved in the journal's whole peer review procedure of this manuscript. The other authors declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Maria Mermiri and Georgios Mavrovounis contributed equally and share first authorship.
References
- 1.Osuchowski MF, Winkler MS, Skirecki T, Cajander S, Shankar-Hari M, Lachmann G, et al. The COVID-19 puzzle: deciphering pathophysiology and phenotypes of a new disease entity. Lancet Respir Med. 2021;9:622–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Sinha P, Calfee CS, Cherian S, Brealey D, Cutler S, King C, et al. Prevalence of phenotypes of acute respiratory distress syndrome in critically ill patients with COVID-19: a prospective observational study. Lancet Respir Med. 2020;8:1209–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Puelles VG, Lütgehetmann M, Lindenmeyer MT, Sperhake JP, Wong MN, Allweiss L, et al. Multiorgan and renal tropism of SARS-CoV-2. N Engl J Med. 2020;383:590–2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wichmann D, Sperhake JP, Lütgehetmann M, Steurer S, Edler C, Heinemann A, et al. Autopsy findings and venous thromboembolism in patients with COVID-19: a prospective cohort study. Ann Intern Med. 2020;173:268–77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Varga Z, Flammer AJ, Steiger P, Haberecker M, Andermatt R, Zinkernagel AS, et al. Endothelial cell infection and endotheliitis in COVID-19. Lancet. 2020;395:1417–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gupta A, Madhavan MV, Sehgal K, Nair N, Mahajan S, Sehrawat TS, et al. Extrapulmonary manifestations of COVID-19. Nat Med. 2020;26:1017–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Evans L, Rhodes A, Alhazzani W, Antonelli M, Coopersmith CM, French C, et al. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock 2021. Intensive Care Med. 2021;47:1181–247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Botta M, Tsonas AM, Pillay J, Boers LS, Algera AG, Bos LDJ, et al. Ventilation management and clinical outcomes in invasively ventilated patients with COVID-19 (PRoVENT-COVID): a national, multicentre, observational cohort study. Lancet Respir Med. 2021;9(2):139–48. [DOI] [PMC free article] [PubMed]
- 9.Alhazzani W, Evans L, Alshamsi F, Møller MH, Ostermann M, Prescott HC, et al. Surviving sepsis campaign guidelines on the management of adults with coronavirus disease 2019 (COVID-19) in the ICU: first update. Crit Care Med. 2021;49:e219–34. [DOI] [PubMed] [Google Scholar]
- 10.Dünser MW, Hasibeder WR. Sympathetic overstimulation during critical illness: adverse effects of adrenergic stress. J Intensive Care Med. 2009;24:293–316. [DOI] [PubMed] [Google Scholar]
- 11.Schmittinger CA, Torgersen C, Luckner G, Schröder DC, Lorenz I, Dünser MW. Adverse cardiac events during catecholamine vasopressor therapy: a prospective observational study. Intensive Care Med. 2012;38:950–8. [DOI] [PubMed] [Google Scholar]
- 12.Bangash MN, Kong ML, Pearse RM. Use of inotropes and vasopressor agents in critically ill patients. Br J Pharmacol. 2012;165:2015–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hartmann C, Radermacher P, Wepler M, Nußbaum B. Non-hemodynamic effects of catecholamines. Shock. 2017;48:390–400. [DOI] [PubMed] [Google Scholar]
- 14.Gubbi S, Nazari MA, Taieb D, Klubo-Gwiezdzinska J, Pacak K. Catecholamine physiology and its implications in patients with COVID-19. Lancet Diabetes Endocrinol. 2020;8:978–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sterne JA, Hernán MA, Reeves BC, Savović J, Berkman ND, Viswanathan M, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ. 2016;355:i4919. [DOI] [PMC free article] [PubMed]
- 17.Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898. [DOI] [PubMed]
- 18.Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002;21:1539–58. [DOI] [PubMed] [Google Scholar]
- 19.Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics. 1994;50:1088–101. [PubMed] [Google Scholar]
- 20.Mavridis D, Salanti G. How to assess publication bias: funnel plot, trim-and-fill method and selection models. Evid Based Ment Health. 2014;17:30. [DOI] [PubMed] [Google Scholar]
- 21.Ionescu F, Zimmer MS, Petrescu I, Castillo E, Bozyk P, Abbas A, et al. Extubation failure in critically Ill COVID-19 patients: risk factors and impact on in-hospital mortality. J Intensive Care Med. 2021;36:1018–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Pelayo J, Lo KB, Bhargav R, Gul F, Peterson E, DeJoy IR, et al. Clinical characteristics and outcomes of community- and hospital-acquired acute kidney injury with COVID-19 in a US Inner City Hospital system. Cardiorenal Med. 2020;10:223–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lowe R, Ferrari M, Nasim-Mohi M, Jackson A, Beecham R, Veighey K, et al. Clinical characteristics and outcome of critically ill COVID-19 patients with acute kidney injury: a single Centre cohort study. BMC Nephrol. 2021;22(1):92. [DOI] [PMC free article] [PubMed]
- 24.Hansrivijit P, Gadhiya KP, Gangireddy M, Goldman JD. Risk factors, clinical characteristics, and prognosis of acute kidney injury in hospitalized COVID-19 patients: a retrospective cohort study. Medicines (Basel). 2021;8:4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mesquida J, Caballer A, Cortese L, Vila C, Karadeniz U, Pagliazzi M, et al. Peripheral microcirculatory alterations are associated with the severity of acute respiratory distress syndrome in COVID-19 patients admitted to intermediate respiratory and intensive care units. Crit Care. 2021;25(1):381. [DOI] [PMC free article] [PubMed]
- 26.Ghosn M, Attallah N, Badr M, Abdallah K, De Oliveira B, Nadeem A, et al. Severe acute kidney injury in critically ill patients with COVID-19 admitted to ICU: incidence, risk factors, and outcomes. J Clin Med. 2021;10:1217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Farooqui MA, Almegren A, Binrushud SR, Alnuwaiser FA, Almegren NM, Alhamied NA, et al. Incidence and outcome of acute kidney injury in patients hospitalized with coronavirus disease-19 at a tertiary care medical center in Saudi Arabia. Cureus. 2021;13:e18927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Neves PDMM, Sato VAH, Mohrbacher S, Ferreira BMC, Oliveira ÉS, Pereira LVB, et al. Acute kidney injury due to COVID-19 in intensive care unit: an analysis from a Latin-American center. Front Med (Lausanne). 2021;8:620050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Bernardo J, Gonçalves J, Gameiro J, Oliveira J, Marques F, Duarte I, et al. The impact of transient and persistent acute kidney injury in hospital mortality in COVID-19 patients. J Bras Nefrol. 2021. In press. 10.1590/2175-8239-JBN-2021-0123. [DOI] [PMC free article] [PubMed]
- 30.Hardenberg JB, Stockmann H, Aigner A, Gotthardt I, Enghard P, Hinze C, et al. Critical illness and systemic inflammation are key risk factors of severe acute kidney injury in patients with COVID-19. Kidney Int Rep. 2021;6:905–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Geri G, Darmon M, Zafrani L, Fartoukh M, Voiriot G, Le Marec J, et al. Acute kidney injury in SARS-CoV2-related pneumonia ICU patients: a retrospective multicenter study. Ann Intensive Care. 2021;11:86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ñamendys-Silva SA, Alvarado-Ávila PE, Domínguez-Cherit G, Rivero-Sigarroa E, Sánchez-Hurtado LA, Gutiérrez-Villaseñor A, et al. Outcomes of patients with COVID-19 in the intensive care unit in Mexico: a multicenter observational study. Heart Lung. 2021;50(1):28–32. [DOI] [PMC free article] [PubMed]
- 33.Auld SC, Caridi-Scheible M, Blum JM, Robichaux C, Kraft C, Jacob JT, et al. ICU and ventilator mortality among critically ill adults with coronavirus disease 2019. Crit Care Med. 2020;48(9):e799–804. [DOI] [PMC free article] [PubMed]
- 34.Nabors C, Sridhar A, Hooda U, Lobo SA, Levine A, Frishman WH, et al. Characteristics and outcomes of patients 80 years and older hospitalized with coronavirus disease 2019 (COVID-19). Cardiol Rev. 2021;29:39–42. [DOI] [PubMed] [Google Scholar]
- 35.Salacup G, Lo KB, Gul F, Peterson E, De Joy R, Bhargav R, et al. Characteristics and clinical outcomes of COVID-19 patients in an underserved-inner city population: a single tertiary center cohort. J Med Virol. 2021;93:416–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Nasrullah A, Jayakrishnan T, Wedgeworth P, Mosley M, Vashistha K, Haag A, et al. Characteristics and outcomes of COVID-19 patients admitted to intensive care units in a large health system in Western Pennsylvania. Cureus. 2021;13:e16552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sjöström B, Månsson E, ViklundKamienny J, Östberg E. Characteristics and definitive outcomes of COVID-19 patients admitted to a secondary hospital intensive care unit in Sweden. Health Sci Rep. 2021;4:e446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ismail K, Bensasi H, Taha A, Nazir A, Abdelkhalek M, Mohamed W, et al. Characteristics and outcome of critically ill patients with coronavirus disease-2019 (COVID-19) pneumonia admitted to a tertiary care center in the United Arab Emirates during the first wave of the SARS-CoV-2 pandemic. A retrospective analysis. PLoS One. 2021;16:e0251687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ofosu-Barko K, Liu Y, AlkhatibTamimi F, Al Husami W, Ganatra S, Resnic F, et al. Angiotensin II Administration in patients With COVID-19 shock. Crit Pathw Cardiol. 2021;20:100–2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ramkumar R, Rani D, Bhattacharjee S, Aggarwal R, Soni KD, Aravindan A, et al. Epidemiology and clinical characteristics of COVID- 19 patients requiring critical care in a Tertiary care teaching hospital. J Anaesthesiol Clin Pharmacol. 2021;37:366–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.African COVID-19 Critical Care Outcomes Study (ACCCOS) Investigators. Patient care and clinical outcomes for patients with COVID-19 infection admitted to African high-care or intensive care units (ACCCOS): a multicentre, prospective, observational cohort study. Lancet. 2021;397(10288):1885–94. [DOI] [PMC free article] [PubMed]
- 42.Mammen JJ, Kumar S, Thomas L, Kumar G, Zachariah A, Jeyaseelan L, et al. Factors associated with mortality among moderate and severe patients with COVID-19 in India: a secondary analysis of a randomised controlled trial. BMJ Open. 2021;11:e050571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Andrade JA, Muzykovsky K, Truong J. Risk factors for mortality in COVID-19 patients in a community teaching hospital. J Med Virol. 2021;93:3184–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Chand S, Kapoor S, Orsi D, Fazzari MJ, Tanner TG, Umeh GC, et al. COVID-19-associated critical illness-report of the first 300 patients admitted to intensive care units at a New York City Medical Center. J Intensive Care Med. 2020;35:963–70. [DOI] [PubMed] [Google Scholar]
- 45.Bezerra R, Teles F, Mendonca PB, Damte T, Likaka A, Ferrer-Miranda E, et al. Outcomes of critically ill patients with acute kidney injury in COVID-19 infection: an observational study. Ren Fail. 2021;43:911–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Dang MKM, Bhatt I, Dulu AO, Zhang H, Kostelecky N, Pastores SM. Clinical characteristics, management, and outcomes of cancer patients with coronavirus disease 2019 Admitted to the ICU. Crit Care Explor. 2021;3:e0535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Gündoğan K, Akbudak İH, Hancı P, Halaçlı B, Temel Ş, Güllü Z, et al. Clinical outcomes and independent risk factors for 90-day mortality in critically ill patients with respiratory failure infected with SARS-CoV-2: a multicenter study in Turkish intensive care units. Balkan Med J. 2021;38:296–303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Estella Á, Garcia Garmendia JL, de la Fuente C, Machado Casas JF, Yuste ME, Amaya Villar R, et al. Predictive factors of six-week mortality in critically ill patients with SARS-CoV-2: a multicenter prospective study. Med Intensiva (Engl Ed). 2021;46(4):179–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Gadhiya KP, Hansrivijit P, Gangireddy M, Goldman JD. Clinical characteristics of hospitalised patients with COVID-19 and the impact on mortality: a single-network, retrospective cohort study from Pennsylvania state. BMJ Open. 2021;11:e042549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Serpa Neto A, Landoni G, Ostermann M, Lumlertgul N, Forni L, Alvarez-Belon L, et al. Angiotensin II infusion in COVID-19: an international, multicenter, registry-based study. J Med Virol. 2022;94:2079–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Brandão Neto RA, Marchini JF, Marino LO, Alencar JCG, Lazar Neto F, Ribeiro S, et al. Mortality and other outcomes of patients with coronavirus disease pneumonia admitted to the emergency department: a prospective observational Brazilian study. PLoS One. 2021;16(1):e0244532. [DOI] [PMC free article] [PubMed]
- 52.Leisman DE, Mastroianni F, Fisler G, Shah S, Hasan Z, Narasimhan M, et al. Physiologic response to angiotensin II treatment for coronavirus disease 2019-induced vasodilatory shock: a retrospective matched cohort study. Crit Care Explor. 2020;2:e0230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Burrell AJ, Pellegrini B, Salimi F, Begum H, Broadley T, Campbell LT, et al. Outcomes for patients with COVID-19 admitted to Australian intensive care units during the first four months of the pandemic. Med J Aust. 2021;214:23–30. [DOI] [PubMed] [Google Scholar]
- 54.Ramos FJDS, Atallah FC, Souza MA, Ferreira EM, Machado FR, Freitas FGR. Determinants of death in critically ill COVID-19 patients during the first wave of COVID-19: a multicenter study in Brazil. J Bras Pneumol. 2022;48(5):e20220083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Qian Z, Lu S, Luo X, Chen Y, Liu L. Mortality and clinical interventions in critically ill patient with coronavirus disease 2019: a systematic review and meta-analysis. Front Med (Lausanne). 2021;8:635560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Potere N, Valeriani E, Candeloro M, Tana M, Porreca E, Abbate A, et al. Acute complications and mortality in hospitalized patients with coronavirus disease 2019: a systematic review and meta-analysis. Crit Care. 2020;24:389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Jasiński T, Stefaniak J. COVID-19 and haemodynamic failure: a point of view on mechanisms and treatment. Anaesthesiol Intensive Ther. 2020;52:409–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Luo P, Liu D, Li J. Epinephrine use in COVID-19: friend or foe? Eur J Hosp Pharm. 2021;28:e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Leisman DE, Mehta A, Li Y, Kays KR, Li JZ, Filbin MR, et al. Vasopressin infusion in COVID-19 critical illness is not associated with impaired viral clearance: a pilot study. Br J Anaesth. 2021;127:e146–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Yeung ML, Teng JLL, Jia L, Zhang C, Huang C, Cai JP, et al. Soluble ACE2-mediated cell entry of SARS-CoV-2 via interaction with proteins related to the renin-angiotensin system. Cell. 2021;184:2212–28.e12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Gregoriano C, Molitor A, Haag E, Kutz A, Koch D, Haubitz S, et al. Activation of vasopressin system during COVID-19 is associated with adverse clinical outcomes: an observational study. J Endocr Soc. 2021;5:bvab045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Deshotels MR, Xia H, Sriramula S, Lazartigues E, Filipeanu CM. Angiotensin II mediates angiotensin converting enzyme type 2 internalization and degradation through an angiotensin II type I receptor-dependent mechanism. Hypertension. 2014;64:1368–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Pan M, Vasbinder A, Anderson E, Catalan T, Shadid HR, Berlin H, et al. Angiotensin-converting enzyme inhibitors, angiotensin II receptor blockers, and outcomes in patients hospitalized for COVID-19. J Am Heart Assoc. 2021;10(24):e023535. [DOI] [PMC free article] [PubMed]
- 64.Bendjelid K. Should we use angiotensin II infusion in COVID-19-associated vasoplegic shock? Crit Care. 2020;24:407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Garcia B, Su F, Dewachter L, Favory R, Khaldi A, Moiroux-Sahraoui A, et al. Myocardial effects of angiotensin II compared to norepinephrine in an animal model of septic shock. Crit Care. 2022;26:281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Belletti A, Castro ML, Silvetti S, Greco T, Biondi-Zoccai G, Pasin L, et al. The Effect of inotropes and vasopressors on mortality: a meta-analysis of randomized clinical trials. Br J Anaesth. 2015;115:656–75. [DOI] [PubMed] [Google Scholar]
- 67.Gamper G, Havel C, Arrich J, Losert H, Pace NL, Müllner M, et al. Vasopressors for hypotensive shock. Cochrane Database Syst Rev. 2016;2:CD003709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Favaron E, Ince C, Hilty MP, Ergin B, van der Zee P, Uz Z, et al. Capillary leukocytes, microaggregates, and the response to hypoxemia in the microcirculation of Coronavirus disease 2019 patients. Crit Care Med. 2021;49:661–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Di Dedda U, Ascari A, Fantinato A, Fina D, Baryshnikova E, Ranucci M. Microcirculatory alterations in critically ill patients with COVID-19-associated acute respiratory distress syndrome. J Clin Med. 2022;11:1032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.KanooreEdul VS, Caminos Eguillor JF, Ferrara G, Estenssoro E, Siles DSP, Cesio CE, et al. Microcirculation alterations in severe COVID-19 pneumonia. J Crit Care. 2021;61:73–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Çalışkan M, Baycan ÖF, Çelik FB, Güvenç TS, Atıcı A, Çağ Y, et al. Coronary microvascular dysfunction is common in patients hospitalized with COVID-19 infection. Microcirculation. 2022;18:e12757. [DOI] [PMC free article] [PubMed]
- 72.Rovas A, Osiaevi I, Buscher K, Sackarnd J, Tepasse PR, Fobker M, et al. Microvascular dysfunction in COVID-19: the MYSTIC study. Angiogenesis. 2021;24:145–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.do Espírito Santo DA, Lemos ACB, Miranda CH. In vivo demonstration of microvascular thrombosis in severe COVID-19. J Thromb Thrombolysis. 2020;50:790–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Dubin A, Pozo MO, Casabella CA, Pálizas F Jr, Murias G, Moseinco MC, et al. Increasing arterial blood pressure with norepinephrine does not improve microcirculatory blood flow: a prospective study. Crit Care. 2009;13:R92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Chalkias A, Xenos M. Relationship of effective circulating volume with sublingual red blood cell velocity and microvessel pressure difference: a clinical investigation and computational fluid dynamics modeling. J Clin Med. 2022;11:4885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Chalkias A, Laou E, Mermiri M, Michou A, Ntalarizou N, Koutsona S, et al. Microcirculation-guided treatment improves tissue perfusion and hemodynamic coherence in surgical patients with septic shock. Eur J Trauma Emerg Surg. 2022;48:4699–711. 10.1007/s00068-022-01991-2. Epub ahead of print. [DOI] [PubMed] [Google Scholar]
- 77.Chalkias A. Increasing stress volume vs. increasing tissue perfusion in septic patients. Eur J Anaesthesiol. 2022;39:390–1. [DOI] [PubMed] [Google Scholar]
- 78.Dekker RJ, van Thienen JV, Rohlena J, de Jager SC, Elderkamp YW, Seppen J, et al. Endothelial KLF2 links local arterial shear stress levels to the expression of vascular tone-regulating genes. Am J Pathol. 2005;167:609–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Lupu F, Kinasewitz G, Dormer K. The role of endothelial shear stress on haemodynamics, inflammation, coagulation and glycocalyx during sepsis. J Cell Mol Med. 2020;24:12258–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Mori M, Krumholz HM, Allore HG. Using latent class analysis to identify hidden clinical phenotypes. JAMA. 2020;324:700–1. [DOI] [PubMed] [Google Scholar]
- 81.Chalkias A, Xanthos T, Papageorgiou E, Anania A, Beloukas A, Pavlopoulos F. Intraoperative initiation of a modified ARDSNet protocol increases survival of septic patients with severe acute respiratory distress syndrome. Heart Lung. 2018;47:616–21. [DOI] [PubMed] [Google Scholar]
- 82.Marik PE. Iatrogenic salt water drowning and the hazards of a high central venous pressure. Ann Intensive Care. 2014;4:21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Matthay MA, McAuley DF, Ware LB. Clinical trials in acute respiratory distress syndrome: challenges and opportunities. Lancet Respir Med. 2017;5:524–34. [DOI] [PubMed] [Google Scholar]
- 84.Caldera-Crespo LA, Paidas MJ, Roy S, Schulman CI, Kenyon NS, Daunert S, et al. Experimental models of COVID-19. Front Cell Infect Microbiol. 2022;11:792584. [DOI] [PMC free article] [PubMed]
- 85.Ware LB, Matthay MA, Mebazaa A. Designing an ARDS trial for 2020 and beyond: focus on enrichment strategies. Intensive Care Med. 2020;46:2153–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Appendix A. PRISMA checklist.
Additional file 2: Appendix B. Algorithms used for all databases.
Additional file 3: Appendix C1. Definitions used for AKI and mortality follow-up time points. Appendix C2. Angiotensin-related data. Appendix C3. MINORS results for each study.
Additional file 4: Appendix D1. Funnel plot for mortality meta-analysis. Appendix D2. Funnel plot for AKI meta-analysis.
Data Availability Statement
Data can be made available upon request after publication through a collaborative process. Researchers should provide a methodically sound proposal with specific objectives in an approval proposal. Please contact the corresponding author for additional information.