Abstract
Anti-SARS-CoV-2 vaccines were developed in response to the coronavirus disease 2019 (COVID-19) pandemic caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Although the BNT162b2 mRNA vaccine is effective, adverse effects have been reported. Here, we report a case of extranodal NK/T-cell lymphoma, nasal type (ENKL), of the left arm following BNT162b2 mRNA vaccination. A 73-year-old male presented with a lump in the left arm, which was the site where he received the BNT162b2 mRNA vaccine 3 months prior. He was treated with topical corticosteroids and debridement, but the tumor progressed. Additionally, fever, night sweats, and general fatigue were observed. Laboratory findings included thrombocytopenia, elevated lactate dehydrogenase, and soluble interleukin-2 receptor levels. Skin biopsy led to a diagnosis of ENKL. The patient was treated with a 50% dose of SMILE therapy and radiotherapy, resulting in regression of the tumor. It seems that latent Epstein–Barr virus (EBV)-infected NK/T cells were reactivated by vaccination and contributed to the onset of ENKL. This is the first report of ENKL after BNT162b2 mRNA vaccination. The present case highlights the possible risk of development of malignant lymphoma, including ENKL at the injection site, after BNT162b2 COVID-19 vaccination.
Keywords: Extranodal NK/T-cell lymphoma, Nasal type, Epstein–Barr virus infection, BNT162b2 mRNA COVID-19 vaccination
Introduction
Coronavirus disease 2019 (COVID-19), caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, continues to spread and remains an international public health emergency. The BNT162b2 mRNA vaccine (Pfizer/BioNtech) is effective in reducing SARS-CoV-2 infections [1]. However, local (such as redness, swelling, and tenderness) and systemic (such as fever, fatigue, and joint pain) reactions after BNT162b2 mRNA vaccination have been reported [2]. Transient lymphadenopathy has also been reported in 0.3% of BNT162b2 mRNA-vaccine recipients and is thought to cause robust vaccine-elicited immune responses [2]. However, malignant lymphoma, especially at a just injection site, rarely occurs after BNT162b2 mRNA vaccination.
Extranodal NK/T-cell lymphoma, nasal type (ENKL), is a natural killer (NK) cell malignancy mostly observed in Asian populations [3]. Approximately, 25% of patients with ENKL have disseminated disease, and the prognosis is extremely poor when treated with anthracycline-based chemotherapy [4]. The prognosis of ENKL is poor because normal NK cells express P-glycoprotein, which is associated with multidrug resistance (MDR) [5]. ENKL is associated with Epstein–Barr virus (EBV) reactivation [6]. Although EBV reactivation after COVID-19 vaccination has been reported [7], the relationship between ENKL and COVID-19 vaccination remains unclear.
Here, we report a case of ENKL at the site of the BNT162b2 mRNA COVID-19 vaccine injection. This is the first report of ENKL development following BNT162b2 vaccination.
Case presentation
A 73-year-old male received the first dose of BNT162b2 vaccination in June 2021 and second dose in July 2021. Systemic symptoms associated with vaccination were not observed. He never had COVID-19. In addition, the patient had no history of allergy including hypersensitivity to mosquito bites. His medical history included old myocardial infarction, cardiac aneurysm, hypertension, hyperuricemia, and dyslipidemia. His regular medications were warfarin, clopidogrel, nicorandil, amlodipine, enalapril, lansoprazole, carvedilol, rosuvastatin, and topiroxostat. In October 2021, the patient presented with a lump in the left arm at the site of vaccination 3 months after the second BNT162b2 vaccination. In November 2021, the patient visited nearby clinics. A 2 cm skin ulcer surrounded by subcutaneous induration and redness in his left arm was observed. Washing and topical azulene application were performed, but the subcutaneous induration remained and was accompanied by heat. In December 2021, he was treated with topical corticosteroids and debridement, but the tumor progressed. In January 2022, new ulcer appeared adjacent to the original lesion. The patient complained of fever, night sweats, general fatigue, and left arm pain. He was referred and admitted to our hospital in February 2022.
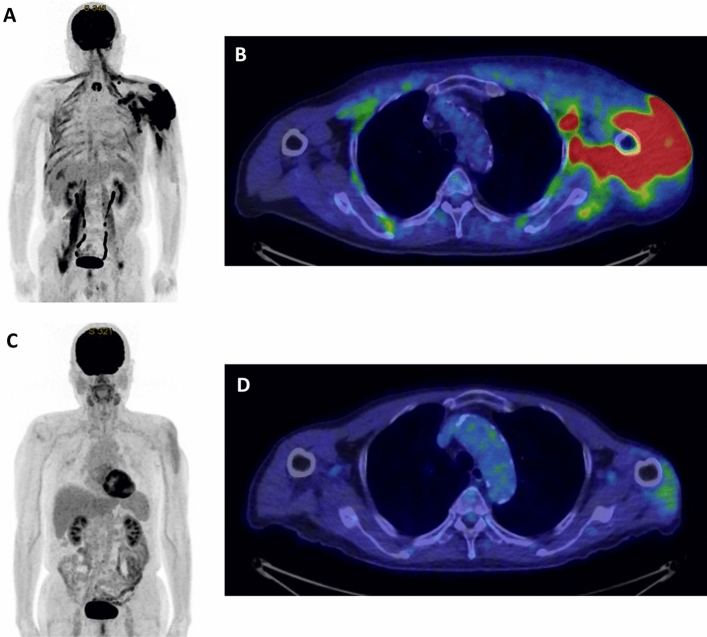
Physical examination revealed fever (body temperature, 39.1 °C) and 6 cm and 3 cm ulcerated tumors in his left arm (Fig. 1). A complete blood count revealed thrombocytopenia (platelet count, 87,000 platelets/μL). Serum biochemistry revealed elevated NT-proBNP (328 pg/mL), ferritin (2,740 ng/mL), lactate dehydrogenase (LDH; 1,371 U/L), and soluble interleukin-2 receptor (sIL-2R; 5,456 U/mL) levels. The IgG antibody titers to the viral capsid antigen (EBVCA-IgG) and nuclear antigen (EBNA) of EBV were 1:320 and 1:160, respectively. The early antigen of EBV (EBEA-IgG) was not elevated. EBV DNA was detected (7.0 × 104 copies/mL) (Table 1). Computed tomography (CT) and magnetic resonance imaging (MRI) revealed an intramuscular tumor in the left arm. Positron emission tomography (PET)–CT showed accumulation of fluorodeoxyglucose (FDG) in the left arm and muscles surrounding the humerus and left axillary lymph nodes (Fig. 2A, B). The skin biopsy was performed from the skin and subcutaneous fatty tissue of the left arm, including the ulcerated tumor and normal skin. Pathological examination showed infiltration of abnormal lymphocytes positive for cluster of differentiation (CD) 3, CD56, CD30, granzyme-B, perforin, and Epstein–Barr encoded RNA (EBER) from dermis to subcutaneous fatty tissue. Immunohistochemistry revealed that these cells were negative for CD4, CD5, CD8, CD10, CD20, CD79a, and TIA-1. The Ki-67 index was 80%. On the basis of these findings, the patient was diagnosed with ENKL. A bone marrow biopsy revealed no evidence of lymphoma involvement. We diagnosed the disease stage as Ann Arbor stage IV-B.
Fig. 1.
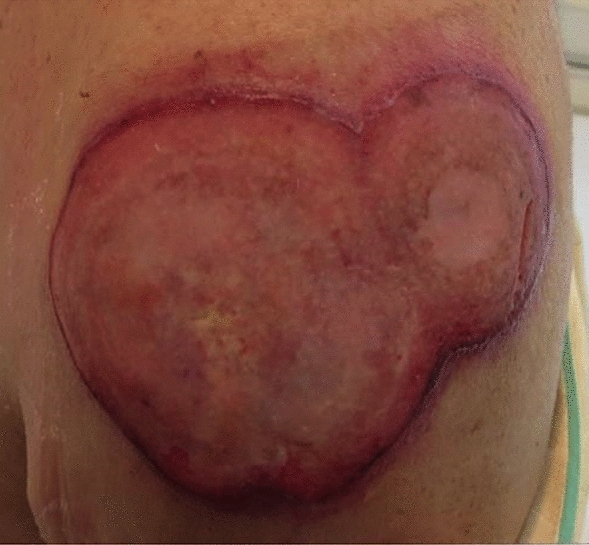
A clinical image of the ENKL lesion of the left arm
Table 1.
Laboratory findings at diagnosis in a patient with ENKL after BNT162b2 vaccination
| Complete blood count | Biochemistry | Serology | |||
|---|---|---|---|---|---|
| WBC | 6600/µL | TP | 6.8 g/dL | CRP | 5.16 mg/dL |
| Neu | 83% | Alb | 3.2 g/dL | IgG | 1178 mg/dL |
| Mono | 2% | AST | 124 U/L | IgA | 408 mg/dL |
| Lym | 14% | ALT | 94 U/L | IgM | 60 mg/dL |
| RBC | 578 × 104/µL | LDH | 1371 U/L | Ferritin | 2740 ng/mL |
| Hb | 17.1 g/dL | γ-GTP | 283 U/L | sIL-2R | 5456 U/mL |
| Ht | 50.40% | ALP | 73 U/L | NT-proBNP | 328 pg/mL |
| MCV | 87.2 fL | Cr | 1.15 mg/dL | HTLV-1 Ab | (−) |
| Ret | 1.0% | UA | 4.7 mg/dL | EBV DNA | 7.0 × 104 copies/mL |
| Plt | 8.7 × 104/µL | BUN | 21 mg/dL | EBVCA-IgG | 320 |
| Coagulation tests | Glc | 160 mg/dL | EBEA-IgG | < 10 | |
| Na | 133 mmol/L | EBNA | 160 | ||
| PT-INR | 1.23 | K | 4.5 mmol/L | ||
| APTT | 37.5 s | Cl | 99 mmol/L | ||
| Fib | 268 mg/dL | Ca | 8.1 mg/dL | ||
| FDP | 6.2 µg/mL | T.Bil | 1.0 mg/dL | ||
| CPK | 518 U/L | ||||
Fig. 2.
PET–CT findings. A–B PET–CT image at diagnosis. Accumulation of FDG was detected in the left arm, muscles surrounding the humerus, and the left axillary lymph nodes. C–D PET–CT images after the three courses of 50% SMILE therapy. The accumulation of FDG markedly decreased
The patient had a history of myocardial infarction and a cardiac aneurysm. Transthoracic echocardiography showed a diffuse hypokinetic ventricle with an ejection fraction (EF) of 40%. We thought it necessary to reduce the intensity of chemotherapy because the patient was older and had low cardiac function. The patient was treated with SMILE therapy reduced to 50% dose (day 1: methotrexate 1 g/m2; day 2–4: ifosfamide 750 mg/m2, etoposide 50 mg/m2, and dexamethasone 40 mg/body; and days 8, 10, 12, 14, 16, 18, and 20: L-asparaginase 3000U/m2) [8]. On day 20 of the first course of 50% SMILE therapy, the patient developed melena. The patient underwent upper gastrointestinal endoscopy, which revealed a multifocal hemorrhagic gastric ulcer. Endoscopic hemostasis was performed, and the melena disappeared. The tumor in the left arm regressed gradually. After three courses of 50% SMILE therapy, the quantity of EBV DNA in the plasma decreased (< 2.0 × 102 copies/mL). Additionally, the levels of LDH (185 U/L) and sIL-2R (631 U/mL) reduced significantly; however, anorexia, delayed clearance of methotrexate, and renal failure were also observed (decline in eGFR: 28.6 mL/min/1.73 m2). The patient discontinued the 50% SMILE therapy due to the toxicity. PET–CT was performed, and regression of the lesion in the left arm was confirmed (Fig. 2C–D). We considered the response to be a partial response (PR). The residual lesion was localized; therefore, radiotherapy was applied to the left arm (50 Gy with 25 fractions).
Discussion
The COVID-19 pandemic, March 2020, led to the development and rapid approval of anti-SARS-CoV-2 vaccines. Although vaccination has prevented the incidence of COVID-19 and COVID-19-associated deaths, its safety profile has not been fully elucidated. Myocarditis has been reported as a severe adverse event of the BNT162b2 mRNA vaccine (Pfizer–BioNTech) [9]. Although vaccination-induced lymphadenopathy has also been reported [10], there are a few reports about the occurrence of lymphoma after BNT162b2 mRNA vaccination. Recurrence of T-cell lymphoma [11, 12] and newly diagnosed diffuse large B-cell lymphoma (DLBCL) shortly after BNT162b2 mRNA vaccination [13] has been previously reported (Table 2). Lymphoma onset in these cases occurred shortly after the injection of the vaccine (maximum of 2 weeks post-administration). Our patient took 3 months from vaccination to the onset of ENKL. This is not the typical clinical course presented in previous reports. Although the lymphoma lesions of previous reports were not observed at the vaccine injection sites, our case had a lymphoma lesion in the skin and muscle at the BNT162b2 mRNA-vaccine injection site. It is possible that our case is of different pathogenesis from previous reports, in that the disease occurred relatively late after vaccination and at the site of vaccine injection.
Table 2.
Summary of cases with malignant lymphoma after BNT162b2 vaccination
| Case | Age(years)/sex | Disease | Disease status | Latency period | BNT162b2 vaccination | Site | Lesion at injection site | Treatment | References |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 79/M | Primary cutaneous CD30-positive LPD | Recurrence | 2 days | First | Left axilla | No | NA | [11] |
| 2 | 60/M | MF | Recurrence | 4 weeks | First | Skin | No | NA | [12] |
| 3 | 73/M | MF | Recurrence | 10 days | First | Skin | No | NA | [12] |
| 4 | 67/M | DLBCL | Newly diagnosed | 14 days | Second | Left axilla | No | R-CHOP | [13] |
| 5 | 80/M | DLBCL | Newly diagnosed | 1 day | Second | Left axilla | No | R-miniCHOP + RT | [13] |
| 6 | 73/M | ENKL | Newly diagnosed | 3 months | Second | Left arm | Yes | 50% SMILE + RT | * |
M male, F female, NA not available, LPD lymphoproliferative disorder, MF mycosis fungoides, DLBCL diffuse large B-cell lymphoma, RT radiotherapy, * present case
There was no evidence of causal relationship between BNT162b2 mRNA vaccination and ENKL in this case, but we speculated that vaccination might lead to the development of ENKL in a manner similar to severe mosquito bite allergy (SMBA). SMBA is a rare EBV-positive NK-cell lymphoproliferative disorder mainly reported in children. Patients with SMBA exhibit skin lesions at bite sites (bulla, necrosis, ulcer, and necrosis), high temperature, lymphadenopathy, and hepatosplenomegaly [6, 14]. Moreover, cases of SMBA developing into ENKL, systemic chronic-active EBV infection (CAEBV), and hemophagocytic lymphohistiocytosis (HLH) have been reported [15–18]. On the other hand, the reactivation of EBV induced by BNT162b2 mRNA vaccination has been reported [7, 19, 20]. In our case, we speculated that hypersensitivity to vaccination might have triggered the reactivation of EBV and the development of ENKL. Although our case was not typical for SMBA in terms of the onset in elderly patients and long latency period from vaccination to disease onset, the skin biopsy revealed reactivation of EBV and suggested a pathology similar to SMBA. In addition, the elevated EBEA-IgG was absent in our case. In a previous study, it was reported that EBEA-IgG can be negative in the patient with hypersensitivity to mosquito bites [21]. It was also reported that the patients with NK-cell CAEBV do not always have elevated EBV-specific antibody titers in contrast to the T-cell CAEBV that have high titers of EBV-specific antibody [15]. Therefore, the low level of EBEA-IgG in our patient could suggest the reactivation of EBV in NK cells.
This case has two limitations about the relationship between BNT162b2 mRNA vaccination and ENKL. First, strictly speaking, we could not identify the exact site of vaccine administration, because we do not have the record of the site of vaccination. However, according to the patient, the site of ulcerated tumors coincided with the vaccine injection site. Second, topical steroid application and debridement might have contributed to the development of ENKL. The duration of these treatments was almost 1 month. This might be responsible for the development of ENKL. However, no steroid application and no surgical procedure were performed from the onset of a lump in the left arm in October 2021 to December 2021. In this period, the ulcer and subcutaneous induration developed without treatment. Thus, we thought that the BNT162b2 mRNA vaccination possibly contributed to the onset of ENKL. Further studies to investigate the mechanism regulating the onset of ENKL after the mRNA vaccination are needed.
This is the first report of ENKL after BNT162b2 mRNA vaccination. Although the relationship between ENKL and vaccination remains unclear, our case provides new insights into lymphomagenesis after vaccination and the pathogenesis of ENKL.
Declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
The patient gave his informed consent for this case report to be published.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Levine-Tiefenbrun M, Yelin I, Katz R, Herzel E, Golan Z, Schreiber L, et al. Initial report of decreased SARS-CoV-2 viral load after inoculation with the BNT162b2 vaccine. Nat Med. 2021;27(5):790–792. doi: 10.1038/s41591-021-01316-7. [DOI] [PubMed] [Google Scholar]
- 2.Polack FP, Thomas SJ, Kitchin N, Absalon J, Gurtman A, Lockhart S, et al. Safety and efficacy of the BNT162b2 mRNA COVID-19 vaccine. N Engl J Med. 2020;383(27):2603–2615. doi: 10.1056/NEJMoa2034577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Tse E, Kwong YL. How i treat NK/T-cell lymphomas. Blood. 2013;121(25):4997–5005. doi: 10.1182/blood-2013-01-453233. [DOI] [PubMed] [Google Scholar]
- 4.Suzuki R, Suzumiya J, Yamaguchi M, Nakamura S, Kameoka J, Kojima H, et al. Prognostic factors for mature natural killer (NK) cell neoplasms: aggressive NK cell leukemia and extranodal NK cell lymphoma, nasal type. Ann Oncol. 2009;21(5):1032–1040. doi: 10.1093/annonc/mdp418. [DOI] [PubMed] [Google Scholar]
- 5.Yamaguchi M, Suzuki R. JSH practical guidelines for hematological malignancies, 2018: II. Lymphoma-9. Extranodal NK/T-cell lymphoma, nasal type (ENKL) Int J Hematol. 2019;109(4):371–376. doi: 10.1007/s12185-019-02609-x. [DOI] [PubMed] [Google Scholar]
- 6.Swerdlow S, Campo E, Harris N, Jaffe E, Pileri S, Stein H. WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues. Lyon: IARC Press; 2016. [Google Scholar]
- 7.Herzum A, Trave I, D’Agostino F, Burlando M, Cozzani E, Parodi A. Epstein–Barr virus reactivation after COVID-19 vaccination in a young immunocompetent man: a case report. Clin Exp Vaccine Res. 2022;11(2):222. doi: 10.7774/cevr.2022.11.2.222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Yamaguchi M, Kwong YL, Kim WS, Maeda Y, Hashimoto C, Suh C, et al. Phase II study of SMILE chemotherapy for newly diagnosed stage IV, relapsed, or refractory extranodal natural killer (NK)/T-cell lymphoma, nasal type: the NK-cell tumor study group study. J Clin Oncol. 2011;29(33):4410–4416. doi: 10.1200/JCO.2011.35.6287. [DOI] [PubMed] [Google Scholar]
- 9.Ammirati E, Cavalotti C, Milazzo A, Pedrotti P, Soriano F, Schroeder JW, et al. Temporal relation between second dose BNT162b2 mRNA COVID-19 vaccine and cardiac involvement in a patient with previous SARS-COV-2 infection. IJC Hear Vasc. 2021;34:100774. doi: 10.1016/j.ijcha.2021.100774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Xu G, Lu Y. COVID-19 mRNA vaccination-induced lymphadenopathy mimics lymphoma progression on FDG PET/CT. Clin Nucl Med. 2021;46(4):353–354. doi: 10.1097/RLU.0000000000003597. [DOI] [PubMed] [Google Scholar]
- 11.Brumfiel CM, Patel MH, DiCaudo DJ, Rosenthal AC, Pittelkow MR, Mangold AR. Recurrence of primary cutaneous CD30-positive lymphoproliferative disorder following COVID-19 vaccination. Leuk Lymphoma. 2021;62(10):2554–2555. doi: 10.1080/10428194.2021.1924371. [DOI] [PubMed] [Google Scholar]
- 12.Panou E, Nikolaou V, Marinos L, Kallambou S, Sidiropoulou P, Gerochristou M, et al. Recurrence of cutaneous T-cell lymphoma post viral vector COVID-19 vaccination. J Eur Acad Dermatology Venereol. 2022;36(2):e91–e93. doi: 10.1111/jdv.17736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Mizutani M, Mitsui H, Amano T, Ogawa Y, Deguchi N, Shimada S, et al. Two cases of axillary lymphadenopathy diagnosed as diffuse large B-cell lymphoma developed shortly after BNT162b2 COVID-19 vaccination. J Eur Acad Dermatology Venereol. 2022;36(8):e613–e615. doi: 10.1111/jdv.18136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tokura Y, Ishihara S, Tagawa S, Seo N, Ohshima K, Takigawa M. Hypersensitivity to mosquito bites as the primary clinical manifestation of a juvenile type of Epstein–Barr virus-associated natural killer cell leukemia/lymphoma. J Am Acad Dermatol. 2001;45(4):569–578. doi: 10.1067/mjd.2001.114751. [DOI] [PubMed] [Google Scholar]
- 15.Kimura H, Hoshino Y, Kanegane H, Tsuge I, Okamura T, Kawa K, et al. Clinical and virologic characteristics of chronic active Epstein–Barr virus infection. Blood. 2001;98(2):280–286. doi: 10.1182/blood.V98.2.280. [DOI] [PubMed] [Google Scholar]
- 16.Kimura H, Ito Y, Kawabe S, Gotoh K, Takahashi Y, Kojima S, et al. EBV-associated T/NK–cell lymphoproliferative diseases in nonimmunocompromised hosts: prospective analysis of 108 cases. Blood. 2012;119(3):673–686. doi: 10.1182/blood-2011-10-381921. [DOI] [PubMed] [Google Scholar]
- 17.Lee S, Park C-J, Cho Y-U, Jang S, Huh J, Kim H. The first Korean case of Epstein–Barr virus-positive Natural Killer/T-cell lymphoma that progressed from severe mosquito bite allergy, with coexistence of hemophagocytic lymphohistiocytosis. Ann Lab Med. 2020;40(1):80–83. doi: 10.3343/alm.2020.40.1.80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Dupuy E, Afifi L, Jonas SJ, Cheng CE, Hogeling M. A case of severe mosquito bite allergy complicated by fatal hemophagocytic lymphohistiocytosis. Pediatr Dermatol. 2022;39(3):443–446. doi: 10.1111/pde.14877. [DOI] [PubMed] [Google Scholar]
- 19.Tang LV, Hu Y. Hemophagocytic lymphohistiocytosis after COVID-19 vaccination. J Hematol Oncol. 2021;14(1):87. doi: 10.1186/s13045-021-01100-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lin TY, Yeh YH, Chen LW, Cheng CN, Chang C, Roan JN, et al. Hemophagocytic lymphohistiocytosis following BNT162b2 mRNA COVID-19 vaccination. Vaccines. 2022;10(4):573. doi: 10.3390/vaccines10040573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ishihara S, Ohshima K, Tokura Y, Yabuta R, Imaishi H, Wakiguchi H, et al. Hypersensitivity to mosquito bites conceals clonal lymphoproliferation of Epstein–Barr viral DNA-positive Natural Killer Cells. Jpn J Cancer Res. 1997;88(1):82. doi: 10.1111/j.1349-7006.1997.tb00305.x. [DOI] [PMC free article] [PubMed] [Google Scholar]