Abstract
Background
Arboviruses are a group of viruses transmitted to vertebrate hosts by certain blood-feeding arthropods. Among urban vectors of arboviruses, mosquitoes of the genus Aedes are the most common. However, other mosquitoes may be susceptible to infection and involved in the transmission, such as Mansonia spp. Therefore, this study aimed to investigate whether Mansonia humeralis can be infected with the Mayaro virus (MAYV).
Methods
These insects were collected from 2018 to 2020 in chicken coops of rural communities in Jaci Paraná in Porto Velho, Rondônia, Brazil, while performing blood-feeding on roosters. The mosquitoes were randomly grouped in pools from which the head and thorax were macerated and checked for the presence of MAYV by quantitative reverse transcription polymerase chain reaction (RT-qPCR). The positive pools were used to infect the C6/36 cell line, and on different days post-infection, the supernatant of the infected cells was subjected to viral detection by RT-qPCR.
Results
A total of 183 pools of female mosquitoes were tested, of which 18% were positive for MAYV; some samples from insect pools inoculated into C6/36 cells showed in vitro multiplication capacity between 3 and 7 days post-infection.
Conclusions
This is the first report of Ma. humeralis mosquitoes that are naturally infected by MAYV, indicating that these vectors may be potential transmitting agents of this arbovirus.
Graphical Abstract
Supplementary Information
The online version contains supplementary material available at 10.1186/s13071-023-05707-2.
Keywords: Arboviruses, Mansonia humeralis, Mayaro fever, Viral isolation
Background
Arboviruses, or viruses transmitted by arthropods, are potentially fatal and epidemic, representing a serious public health problem with social and economic implications. Except for yellow fever and dengue, no vaccines and effective antiviral drugs or therapy are available to treat arboviral diseases. Therefore, vector control is an essential tool for preventing future outbreaks. Furthermore, screening for arboviruses in mosquito species can provide information about which vectors are participating in the local dynamics of viruses [1].
The increase in the incidence of the Mayaro virus (MAYV; genus Alphavirus, family Togaviridae) has attracted the attention of researchers and public health authorities. MAYV was discovered in 1954 when it was isolated from the blood of febrile patients with arthralgia in Trinidad and Tobago [2]. It is endemic to South America and the Caribbean. Moreover, MAYV has been detected in Argentina, Bolivia, Brazil, Ecuador, French Guiana, Haiti, Mexico, Panama, Peru, and Venezuela. Currently, four distinct genotypes are in circulation. In Brazil, epidemiological studies have shown the presence of the virus in different regions, including the north, central-west, and southeast [3, 4].
Understanding the arthropod vectors that naturally transmit MAYV is critical to elucidating the interactions between biological and environmental factors that affect the transmission of this arbovirus and identifying regions of increased outbreak probability. The primary vector of MAYV is Haemagogus janthinomys [5]. However, other mosquito species have been described as susceptible to MAYV and thus can be competent in arbovirus transmission, such as Psorophora ferox, Psorophora albipes, Sabethes spp., Culex spp., and Aedes spp. [6, 7]. Aitken et al. [8] demonstrated the vector competence of the Aedes scapularis mosquito in transmitting MAYV by inoculating the virus into female mosquitoes and inducing them to feed on the blood of infected Gallus gallus domesticus and/or Mus musculus. This mechanism was additionally used to demonstrate that MAYV can also infect Culex spp. and Anopheles spp. [6, 7, 9, 10]. Recently, MAYV was detected in eggs and male mosquitoes of the genera Aedes and Culex in Mato Grosso, Brazil, suggesting vertical transmission [11–13]. De Curcio et al. [13] also demonstrated the possible vertical transmission of MAYV by Aedes aegypti in Goiás, Brazil. In addition, the genus Mansonia has been reported to be naturally infected with several arboviruses, such as MAYV [12], Chikungunya virus (CHIKV), and Zika virus (ZIKV) [14, 15]. However, Mansonia spp. mosquitoes are not considered a vector of endemic diseases in Brazil.
Recently, anthropogenic activities have led to significant ecological changes that create breeding grounds for vector proliferation and acquisition, posing a risk to human health [16, 17]. For example, Jaci Paraná, a district of Porto Velho, Rondônia, in northern Brazil, has several factors that favor the spread of vectors. This district has experienced several environmental changes, such as deforestation, rural settlements, and the establishment of extensive cattle ranching. These changes provided suitable areas for mosquito breeding, resulting in high population densities of vectors such as Mansonia spp. [18–20].
It is well known that early detection of arboviruses in mosquitoes can aid in effective decision-making about public health interventions to reduce the risk of outbreaks of these diseases. Therefore, this work describes the natural occurrence of MAYV in Ma. humeralis for the first time.
Methods
Collection of mosquitoes and processing of samples
Mosquitoes were collected from 2018 to 2020 from chicken coops while feeding on the blood of roosters (G. gallus domesticus) weighing more than 2 kg in rural communities near a hydroelectric power plant in Jaci Paraná, Porto Velho, Rondônia (RO), Brazil (latitude: −9.2654178 and longitude: −64.4008246). A manual vacuum cleaner was used to collect the mosquitoes. These mosquitoes were kept in the gut for 2 days for blood digestion and then Ma. humeralis were identified morphologically and kept at −80 °C.
A total of 1930 mosquitoes were grouped into 183 pools of female individuals and one pool of male individuals. Each pool contained 10 mosquitoes. The head and thorax of females were detached and processed, while males were processed intact. Briefly, mosquitoes were macerated in 300 µl of sterile Milli-Q water in 1.5-ml centrifuge tubes (Axygen, USA) using a pestle. The macerated mosquitoes were centrifuged at 10,000×g for 2 min, and the supernatant was then collected and stored at −80 °C until viral RNA extraction. RNA extraction was performed using the QuickExtract™ RNA Extraction Solution (Lucigen, Miami, FL, USA) kit according to the manufacturer's instructions. All reagents used for mosquito manipulation were tested in quantitative reverse transcriptase polymerase chain reaction (RT-qPCR) to exclude any internal contamination.
Molecular detection of MAYV
The presence of MAYV in the RNA samples was detected by RT-qPCR with the AriaMX system (Agilent Technologies, Santa Clara, CA, USA) using the GoTaq® Probe 1-Step RT-qPCR System Kit (Promega, Madison, WI, USA) according to the manufacturer's recommendations. The set of primers/probes amplifying the actin gene of Mansonia spp. were used as an endogenous control of RT-qPCR. We used gBlocks (Integrated DNA Technologies, Coralville, IA, USA), recognized by the primer and probe pairs designed to amplify the MAYV nonstructural polyprotein gene (GenBank: MK956954.1) or the actin gene of Mansonia spp. (GenBank: GQ981455.1) as a positive control. The viral genome copy number was calculated by plotting the quantification cycle (Cq) values reported in RT-qPCR against the standard curve [13] in which the threshold for detection of endogenous control (actin) and MAYV was determined, and Cq values above the detection threshold were considered negative. The gBlocks, probes, and primer sequences used in this study are listed in Additional file 1: Table S1 and Additional file 2: Table S2, respectively. The detection limit for the actin gene was five copies with a Cq value of 34.30 (Additional file 3: Fig. S1). For MAYV, the detection limit was 10 copies with a Cq value of 34.98 [13].
Inoculation of positive pools in cell culture
A total of four positive pools in the RT-qPCR analysis were used in the experiments in cell culture to confirm viral replication. The Aedes albopictus clone C6/36 cells (American Type Culture Collection [ATCC] CRL-1660™) were grown in TC100 medium (Vitrocell, São Paulo, Brazil) supplemented with 10% fetal bovine serum (FBS; LGC Biotechnology) at 28 °C. After 70–80% cell confluence, 100 µl of macerated supernatant from mosquito pools was filtered through a 0.22-μm sterilization membrane (Nalgene-Thermo Scientific, Waltham, MA, USA) and inoculated into C6/36 cells. Uninfected cells were used as negative controls. Cells were then incubated at 28 °C, and cell morphology was observed every 24 h for 7 days using an inverted phase-contrast microscope (Leica, DMi1). After 3 and 7 days, 2 ml of the culture supernatant was collected, viral RNA was extracted, and a new RT-qPCR was performed with these samples, as described in the “Mayaro virus recovery in the culture supernatant” section.
Data analysis
RT-qPCR results were analyzed using Agilent AriaMx software (version 1.8; Agilent Technologies, Santa Clara, CA, USA). Excel software was used to construct standard curves and determine the straight line of the equation, tables, and graphs. The minimum infection rate (MIR) was calculated using the formula [(number of positive pools/total specimens of the species tested) × 1000] [21] and reported with a 95% confidence interval (EpiData Analysis, 2006–2010).
Results
Mosquito screening and molecular detection of MAYV
A total of seven mosquito species of the genus Mansonia were collected and identified, including Mansonia venezuelensis, Ma. flaveola, Ma. pseudotitillans, Ma. amazonensis, Ma. titillans, Ma. wilsoni, and Ma. humeralis. Ma. humeralis was the most abundant species and was therefore selected for molecular analysis.
A total of 1930 Ma. humeralis were screened for MAYV using RT-qPCR. MAYV genetic material was detected in 34 (18%) female pools (Table 1). These data correspond to an MIR value of 19. Table 1 lists the Cq values obtained and their equivalent copy number. The Cq values ranged from 25.75 to 34.74, corresponding to a variation of 2287 to 9 viral copies/µl. Viral RNA was not identified in the male pool (data not shown).
Table 1.
Detection of Mayaro virus using RT-qPCR in pools of female Mansonia humeralis
| RT-qPCR MAYV | RT-qPCR MAYV | ||||
|---|---|---|---|---|---|
| Pool number | Cq | Number of copies/µl | Pool number | Cq | Number of copies/µl |
| 2 | 34.47 | 10 | 25 | 33.82 | 16 |
| 3 | 33.45 | 19 | 26 | 33.61 | 18 |
| 4 | 31.59 | 61 | 28 | 27.39 | 821 |
| 5 | 33.05 | 25 | 30 | 30.74 | 104 |
| 6 | 32.90 | 27 | 31b | 28.69 | 368 |
| 8 | 29.30 | 253 | 32 | 33.43 | 20 |
| 10 | 32.69 | 31 | 34 | 33.40 | 20 |
| 12 | 31.58 | 62 | 41 | 31.19 | 79 |
| 14 | 30.45 | 124 | 42 | 31.24 | 76 |
| 15 | 33.92 | 15 | 43c | 29.14 | 279 |
| 18 | 34.71 | 9 | 45 | 34.74 | 9 |
| 19 | 34.53 | 10 | 46d | 28.88 | 327 |
| 20 | 30.90 | 94 | 48 | 25.73 | 2287 |
| 21 | 32.12 | 44 | 49 | 34.41 | 11 |
| 22a | 26.27 | 1639 | 50 | 30.71 | 106 |
| 23 | 30.26 | 140 | 57 | 34.58 | 10 |
| 24 | 33.02 | 25 | 184 | 26.82 | 1167 |
a–dThese pools were used for the in vitro infection experiment
Mayaro virus recovery in the culture supernatant
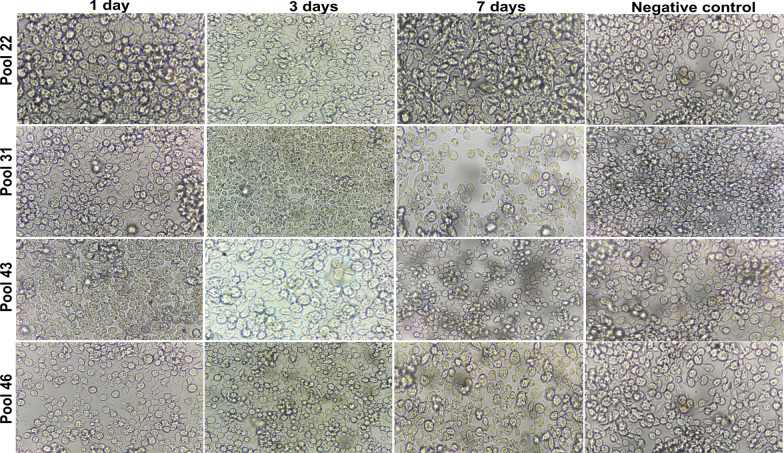
To evaluate the replication of MAYV in cell cultures, the pools with the highest viral load (22, 31, 43, and 46 samples) were inoculated into C6/36 cells. Morphological analysis showed no cytopathic effect in the cells inoculated with MAYV. However, molecular analysis showed the presence of the viral material in the cell culture. Images of the infected cells are shown in Fig. 1. All MAYV pools inoculated into C6/36 cells survived, as evidenced by the presence of the viral RNA in the culture supernatant. Replication was less evident in pools 22, 31, and 43 during infection. Otherwise, pool 46 showed remarkable viral replication capacity, as indicated by the number of viral copies/µl detected after 7 days of cultivation (Table 2). These results suggest that MAYV is present in Ma. humeralis and can survive and replicate in other cell models.
Fig. 1.
Monitoring of the effect of MAYV inoculation in C6/36 cells after 1, 3, and 7 days. The images were analyzed by phase-contrast microscopy, with ×40 magnification. The culture supernatant at these time points was collected and analyzed by RT-qPCR
Table 2.
Cq values and estimated number of viral copies/µl 3 and 7 days after infection of C6/36 with MAYV
| Pool number | Infection day | Cq | Number of copies/µl |
|---|---|---|---|
| 22 | Macerated | 26.27 | 1639 |
| 3 days | 32.48 | 35 | |
| 7 days | 32.74 | 30 | |
| 31 | Macerated | 29.69 | 198 |
| 3 days | 29.95 | 169 | |
| 7 days | 30.07 | 157 | |
| 43 | Macerated | 29.14 | 279 |
| 3 days | 32.14 | 44 | |
| 7 days | 33.94 | 15 | |
| 46 | Macerated | 28.98 | 327 |
| 3 days | 31.87 | 52 | |
| 7 days | 28.63 | 382 |
Discussion
Hundreds of arboviruses are known so far, and several species can affect domestic and wild animals [22]. However, approximately 134 have been reported to cause human diseases [23]. The main vectors of arboviruses in urban areas are Ae. aegypti and Ae. albopictus; however, other vectors are also involved in the transmission of arboviruses in urban and wild cycles. It is estimated that such arboviruses are responsible for 390 million infections annually [24]. Due to the presence of humans in forested regions and the adaptation of mosquitoes to urban areas, new vectors of arboviruses are being identified. In this sense, at least six viral RNA families and several unclassified viruses were detected in Ma. wilsoni collected in the Atlantic Forest in the northeast region of Brazil [25]. Analysis performed in the last decade indicates that novel mosquito-associated viruses primarily infect the Culex, Anopheles, Aedes, and Mansonia genera [26].
Mansonia spp. are voracious hematophagous mosquitoes whose mature stages usually breed in freshwater bodies with aquatic vegetation. Reductions in water flow lead to the proliferation of aquatic plants, increasing their populations. The dispersal of insect vectors is significant for epidemiology, as dispersal is a crucial factor in the course of disease outbreaks and the population dynamics of vector and pest arthropods [27, 28]. Jaci Paraná, Porto Velho, Brazil, is an area that has undergone several environmental changes due to deforestation, the creation of rural settlements, and the establishment of an extensive beef cattle system. These changes led to new areas with suitable vegetation for insect breeding and a high food supply for Mansonia spp. females to perform hematophagy, resulting in a high population density of Ma. humeralis in this region [25]. Nonhuman vertebrates such as chickens [29, 30] also contribute to the persistence of these viruses, as they usually serve as reservoirs for various arboviruses. In this work, an increase in the population of Mansonia spp. which fed on the blood of these animals was observed in chicken coops in Jáci Paraná. Therefore, all these processes may contribute to MAYV spreading to other hosts in nature, as is the case with other viruses such as SARS-CoV-2.
The significant increase in vector population and genetic variability allows the emergence of populations with traits that favor susceptibility to infection and make them competent to transmit different arboviruses [31]. Ma. venezuelensis and Ma. uniformis have previously been reported to be naturally infected with the arboviruses MAYV, CHIKV, and ZIKV [14, 15, 32], indicating that they are potential vectors. However, more information about their vector competence is needed. In this work, we detected the presence of MAYV viral RNA in 18% of Ma. humeralis pools collected in Jaci-Paraná district. In addition, the virus replication in cell cultures was observed in four positive pools.
The presence of MAYV in these mosquitoes indicates the occurrence of natural infection, since most of the positive pools were not engorged. Therefore, the detected virus was not acquired from vertebrate host blood. However, the isolation of MAYV from other Culicidae species, such as Aedes, Culex, Psorophora, Sabethes, and Haemagogus spp., has been described [33, 34].
MAYV is endemic to the Amazon region. An epidemiological study between 1955 and 2019 reported 1304 cases of MAYV infection in Brazil, with 1142 cases described only in the northern region [4], which includes the states of Pará, Amazonas [35], and Tocantins [36]. Ma. humeralis can travel distances greater than 2000 m from the site of adult emergence. In addition, Mello et al. [19] describe the occurrence of passive dispersion that occurs through floating aquatic macrophytes that, once containing immature species of Mansonia spp., are often dragged by river currents, favoring the distribution of these mosquitoes to different locations [18].
The increase in the population and spread of arbovirus vectors due to anthropogenic impacts could increase the transmission of emerging and re-emerging arboviruses. In this sense, the data presented in this work suggest a possible involvement of Ma. humeralis in the epidemiological cycle of MAYV in Jáci-Paraná; however, further studies are needed to clarify this mechanism.
Conclusions
This is the first report of Ma. humeralis mosquitoes naturally infected with MAYV. The detection of MAYV in the head and thorax pools and its replication in cell culture suggest the susceptibility of these mosquitoes to infection with this virus. In addition, these mosquitoes may be involved in the transmission of these arboviruses in the district of Jaci Paraná, Rondônia, Brazil. Therefore, we emphasize the need for greater attention from public health authorities and the development of effective control measures for the mosquito population in Jaci Paraná (RO) district.
Supplementary Information
Additional file 1: Table S1. gBlocks sequence used for RT-qPCR tests.
Additional file 2: Table S2. The sequence of primers to identify arboviruses and actin of insect vectors Aedes spp., Mansonia spp., and Culex spp.
Additional file 3: Fig. S1. Standard curves to identify the limit of detection of actin gene through the real-time PCR technique. The log10 dilution series, ranging from 105 to 1 copy of the amplicon of the actin gene in the block/reaction, was used to build the standard curve; the correlation coefficient (R2) values were above 0.99.
Acknowledgements
We thank the CAPES for fellowships to Sousa, F.B.; de Curcio, J.S.; Silva, L.C.; Silva, D.M.F.
Abbreviations
- MAYV
Mayaro virus
- RT-qPCR
Reverse transcription-quantitative polymerase chain reaction
Author contributions
SFB, ACE, and S-LEP designed the experiments. SFB, CJS, SLC, and SDMF performed the experiments. SFB, CJS, SLC, S-ISM, RBM, and S-LEP analyzed and/or interpreted the data. ACE, G-ZMT, RBM, and SLEPS contributed with reagents and materials. SFB, DCJS, and LEPS wrote the manuscript. All authors read and approved the final manuscript.
Funding
Academic Cooperation Program in National Defense (PROCAD)—88887.387750/2019–00/TED Nº8764786/2021.
Availability of data and materials
Not applicable.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Flávia Barreto de Sousa, Juliana Santana de Curcio, and Lívia do Carmo Silva contributed equally to the work and share first authorship
References
- 1.Viglietta M, Bellone R, Blisnick AA, Failloux AB. Vector specificity of arbovirus transmission. Front Microbiol. 2021 doi: 10.3389/fmicb.2021.773211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Anderson CR, Wattley GH, Ahin NW, Downs WG, Reese AA. Mayaro Virus: a new human disease agent. Am J Trop Med Hyg. 1957;6:1012–1016. doi: 10.4269/ajtmh.1957.6.1012. [DOI] [PubMed] [Google Scholar]
- 3.Silveira-Lacerda DP, Laschuk A, Tanuri A, Pires J, Rezza G, Eduardo C, et al. Molecular epidemiological investigation of Mayaro virus in febrile patients from Goiania city 2017–2018. Infect Gen Evol. 2021;95:104981. doi: 10.1016/j.meegid.2021.104981. [DOI] [PubMed] [Google Scholar]
- 4.Lima WG, Pereira RS, da Cruz Nizer WS, Brito JCM, Godói IP, Cardoso VN, et al. Rate of exposure to Mayaro virus (MAYV) in Brazil between 1955 and 2018: a systematic review and meta-analysis. Arch Virol. 2021;166:347–361. doi: 10.1007/s00705-020-04889-9. [DOI] [PubMed] [Google Scholar]
- 5.Hoch AL, Peterson NE, LeDuc JW, Pinheiro FP. An outbreak of Mayaro virus disease in Belterra, Brazil. III. Entomological and ecological studies. Am J Trop Med Hyg. 1981;30:689–98. doi: 10.4269/ajtmh.1981.30.689. [DOI] [PubMed] [Google Scholar]
- 6.Galindo P, Srihongse S. Transmission of arboviruses to hamsters by the bite of naturally infected Culex (Melanoconion) mosquitoes. Am J Trop Med Hyg. 1967;16:525. doi: 10.4269/ajtmh.1967.16.525. [DOI] [PubMed] [Google Scholar]
- 7.Groot H, Morales A, Vidales H. Virus isolations from forest mosquitoes in San Vicente de Chucuri Colombia. Am J Trop Med Hyg. 1961;10:397. doi: 10.4269/ajtmh.1961.10.397. [DOI] [PubMed] [Google Scholar]
- 8.Aitken TH. Virus transmission studies with Trinidadian mosquitoes. West Indian Med J. 1957;6:229. [PubMed] [Google Scholar]
- 9.Pereira Serra O, Fernandes Cardoso B, Maria Ribeiro AL, dos Santos FAL, Dezengrini SR. Mayaro virus and dengue virus 1 and 4 natural infection in culicids from Cuiabá, state of Mato Grosso Brazil. Mem Inst Oswaldo Cruz. 2016;111:20. doi: 10.1590/0074-02760150270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Galindo P, Srihongse S, de Rodaniche E, Grayson MA. An ecological survey for arboviruses in Almirante, Panama, 1959–1962. Am J Trop Med Hyg. 1966;15:385. doi: 10.4269/ajtmh.1966.15.385. [DOI] [PubMed] [Google Scholar]
- 11.Maia LMS, Bezerra MCF, Costa MCS, Souza EM, Oliveira MEB, Ribeiro ALM, et al. Natural vertical infection by dengue virus serotype 4, Zika virus and Mayaro virus in Aedes (Stegomyia) aegypti and Aedes (Stegomyia) albopictus. Med Vet Entomol. 2019;33:437–442. doi: 10.1111/mve.12369. [DOI] [PubMed] [Google Scholar]
- 12.da Silva Ferreira R, de Toni Aquino da Cruz LC, de Souza VJ, da Silva Neves NA, de Souza VC, Filho LCF, et al. Insect-specific viruses and arboviruses in adult male culicids from Midwestern Brazil. Infection, Genetics and Evolution. 2020;85:104561. [DOI] [PubMed]
- 13.de Curcio JS, Salem-Izacc SM, Pereira Neto LM, Nunes EB, Anunciação CE, de Paula S-LE. Detection of Mayaro virus in Aedes aegypti mosquitoes circulating in Goiânia-Goiás-Brazil. Microbes Infect. 2022;24:104948. doi: 10.1016/j.micinf.2022.104948. [DOI] [PubMed] [Google Scholar]
- 14.Diallo D, Sall AA, Diagne CT, Faye O, Faye O, Ba Y, et al. Zika virus emergence in mosquitoes in Southeastern Senegal, 2011. PLoS One. 2014;9:e109442. doi: 10.1371/journal.pone.0109442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Diallo D, Sall AA, Buenemann M, Chen R, Faye O, Diagne CT, et al. Landscape ecology of sylvatic chikungunya virus and mosquito vectors in southeastern Senegal. PLoS Negl Trop Dis. 2012;6:e1649. doi: 10.1371/journal.pntd.0001649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Acosta-Ampudia Y, Monsalve DM, Rodríguez Y, Pacheco Y, Anaya JM, Ramírez-Santana C. Mayaro: an emerging viral threat? Emerg Microbes Infect. 2018;7:1–11. doi: 10.1038/s41426-018-0163-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Consoli RAGB, Oliveira RL de. Principais mosquitos de importância sanitária no Brasil. Principais mosquitos de importância sanitária no Brasil. 1994.
- 18.Brasil. Monitoramento dos casos de arboviroses urbanas causados por vírus transmitidos pelo mosquito Aedes (dengue, chikungunya e zika), semanas epidemiológicas 1 a 42, 2021. Boletim Epidemiológico Arboviroses. 2021;52.
- 19.de Mello CF, Alencar J. Dispersion pattern of Mansonia in the surroundings of the Amazon Jirau hydroelectric power plant. Sci Rep. 2021;11:24273. doi: 10.1038/s41598-021-03682-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Soares Gil LH, Mello CF, Silva JDS, Oliveira JDS, Silva SOF, Rodríguez-Planes L, et al. Evaluation of Mansonia spp. Infestation on aquatic plants in lentic and lotic environments of the Madeira river Basin in Porto Velho, Rondônia Brazil. J Am Mosq Control Assoc. 2021;37:143. doi: 10.2987/21-7007.1. [DOI] [PubMed] [Google Scholar]
- 21.Lai Z, Zhou T, Liu S, Zhou J, Xu Y, Gu J, et al. Vertical transmission of zika virus in Aedes albopictus. PLoS Negl Trop Dis. 2020 doi: 10.1371/journal.pntd.0008776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Moyes CL, Vontas J, Martins AJ, Ng LC, Koou SY, Dusfour I, et al. Contemporary status of insecticide resistance in the major Aedes vectors of arboviruses infecting humans. Sinnis P, editor. PLoS Negl Trop Dis. 2017;11:e0005625. [DOI] [PMC free article] [PubMed]
- 23.Gubler DJ. Human arbovirus infections worldwide. Ann N Y Acad Sci. 2001;951:13. doi: 10.1111/j.1749-6632.2001.tb02681.x. [DOI] [PubMed] [Google Scholar]
- 24.Wu P, Yu X, Wang P, Cheng G. Arbovirus lifecycle in mosquito: Acquisition, propagation, and transmission. Expert Rev Mol Med. 2019;21:e1. doi: 10.1017/erm.2018.6. [DOI] [PubMed] [Google Scholar]
- 25.da Silva AF, Dezordi FZ, Machado LC, de Oliveira RD, Qin S, Fan H, et al. Metatranscriptomic analysis identifies different viral-like sequences in two neotropical Mansoniini mosquito species. Virus Res. 2021;301:198455. doi: 10.1016/j.virusres.2021.198455. [DOI] [PubMed] [Google Scholar]
- 26.Atoni E, Zhao L, Karungu S, Obanda V, Agwanda B, Xia H, et al. The discovery and global distribution of novel mosquito-associated viruses in the last decade (2007–2017) Rev Med Virol. 2019;29:e2079. doi: 10.1002/rmv.2079. [DOI] [PubMed] [Google Scholar]
- 27.Jeger MJ, Madden LV, van den Bosch F. The effect of transmission route on plant virus epidemic development and disease control. J Theor Biol. 2009;258:198. doi: 10.1016/j.jtbi.2009.01.012. [DOI] [PubMed] [Google Scholar]
- 28.Tallman PS, Riley-Powell AR, Schwarz L, Salmón-Mulanovich G, Southgate T, Pace C, et al. Ecosyndemics: the potential synergistic health impacts of highways and dams in the Amazon. Soc Sci Med. 2022;295:113037. doi: 10.1016/j.socscimed.2020.113037. [DOI] [PubMed] [Google Scholar]
- 29.Kading RC, Reiche ASG, Morales-Betoulle ME, Komar N. Host selection of potential West Nile virus vectors in Puerto Barrios, Guatemala, 2007. Am J Trop Med Hyg. 2013;2013:108. doi: 10.4269/ajtmh.2012.12-0223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Morales‐Betoulle ME, Komar N, Panella NA, Alvarez D, López MR, Betoulle JL, et al. West Nile virus ecology in a tropical ecosystem in Guatemala. Am J Trop Med Hyg. 2013;88:116–126. [DOI] [PMC free article] [PubMed]
- 31.Kramer LD, Ciota AT. Dissecting vectorial capacity for mosquito-borne viruses. Curr Opin Virol. 2015;15:112. doi: 10.1016/j.coviro.2015.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Aitken TH, Anderson CR. Virus transmission studies with Trinidadian mosquitoes II. Further observations. Am J Trop Med Hyg. 1959;8:41. doi: 10.4269/ajtmh.1959.8.41. [DOI] [PubMed] [Google Scholar]
- 33.Pinheiro FP, Freitas RB, Travassos Da Rosa JF, Gabbay YB, Mello WA, Leuc JW. An outbreak of Mayaro virus disease in Belterra, Brazil. I. Clinical and virological findings. Am J Trop Med Hyg. 1981;30:674. doi: 10.4269/ajtmh.1981.30.674. [DOI] [PubMed] [Google Scholar]
- 34.Long KC, Ziegler SA, Thangamani S, Hausser NL, Kochel TJ, Higgs S, et al. Experimental transmission of Mayaro virus by Aedes aegypti. Am J Trop Med Hyg. 2011;85:750–7. doi: 10.4269/ajtmh.2011.11-0359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.dos Santos Souza Marinho R, Duro RLS, Bellini Caldeira D, Galinskas J, Oliveira Mota MT, Hunter J, et al. Re-emergence of Mayaro virus and coinfection with chikungunya during an outbreak in the state of Tocantins/Brazil. BMC Res Notes. 2022;15:1. doi: 10.1186/s13104-022-06153-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Saatkamp CJ, Rodrigues LRR, Pereira AMN, Coelho JA, Marques RGB, de Souza VC, et al. Mayaro virus detection in the western region of Pará state Brazil. Rev Soc Bras Med Trop. 2021;54:e0055. doi: 10.1590/0037-8682-0055-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Table S1. gBlocks sequence used for RT-qPCR tests.
Additional file 2: Table S2. The sequence of primers to identify arboviruses and actin of insect vectors Aedes spp., Mansonia spp., and Culex spp.
Additional file 3: Fig. S1. Standard curves to identify the limit of detection of actin gene through the real-time PCR technique. The log10 dilution series, ranging from 105 to 1 copy of the amplicon of the actin gene in the block/reaction, was used to build the standard curve; the correlation coefficient (R2) values were above 0.99.
Data Availability Statement
Not applicable.