Abstract
Background
The coronavirus disease-2019 (COVID-19) pandemic has contributed to the change in the epidemiology of many infectious diseases. This study aimed to establish the pre-pandemic epidemiology of pediatric invasive bacterial infection (IBI).
Methods
A retrospective multicenter-based surveillance for pediatric IBIs has been maintained from 1996 to 2020 in Korea. IBIs caused by eight bacteria (Streptococcus pneumoniae, Haemophilus influenzae, Neisseria meningitidis, Staphylococcus aureus, Streptococcus agalactiae, Streptococcus pyogenes, Listeria monocytogenes, and Salmonella species) in immunocompetent children > 3 months of age were collected at 29 centers. The annual trend in the proportion of IBIs by each pathogen was analyzed.
Results
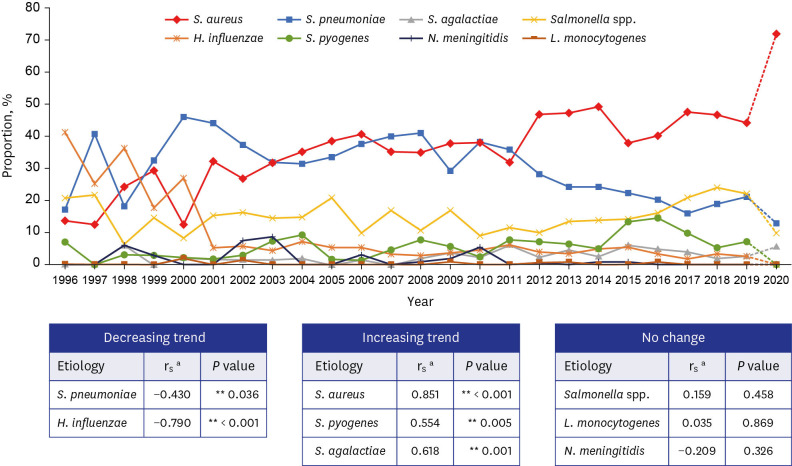
A total of 2,195 episodes were identified during the 25-year period between 1996 and 2020. S. pneumoniae (42.4%), S. aureus (22.1%), and Salmonella species (21.0%) were common in children 3 to 59 months of age. In children ≥ 5 years of age, S. aureus (58.1%), followed by Salmonella species (14.8%) and S. pneumoniae (12.2%) were common. Excluding the year 2020, there was a trend toward a decrease in the relative proportions of S. pneumoniae (rs = −0.430, P = 0.036), H. influenzae (rs = −0.922, P < 0.001), while trend toward an increase in the relative proportion of S. aureus (rs = 0.850, P < 0.001), S. agalactiae (rs = 0.615, P = 0.001), and S. pyogenes (rs = 0.554, P = 0.005).
Conclusion
In the proportion of IBIs over a 24-year period between 1996 and 2019, we observed a decreasing trend for S. pneumoniae and H. influenzae and an increasing trend for S. aureus, S. agalactiae, and S. pyogenes in children > 3 months of age. These findings can be used as the baseline data to navigate the trend in the epidemiology of pediatric IBI in the post COVID-19 era.
Keywords: Invasive Bacterial infections, Epidemiology, COVID-19, Children
Graphical Abstract
INTRODUCTION
Invasive bacterial infections (IBIs) is one of the leading causes of childhood morbidity and mortality. Epidemiology of causative bacterial organism in children varies by age and time. To monitor these changes, multicenter surveillance system for pediatric IBI has been maintained since 1996 in Korea. Two studies performed in 1996–2005 and 2006–2010 had been published, respectively.1,2 In 1996–2005, Streptococcus pneumoniae and Haemophilus influenzae were responsible for 66% of IBIs in children 3 to 59 months of age. The follow-up study in 2006–2010 revealed that S. pneumoniae and Staphylococcus aureus accounted for 75% of all IBIs in same age group.
The coronavirus disease-2019 (COVID-19) pandemic and non-pharmaceutical interventions such as containment, school closing, wearing facemasks, and handwashing have contributed to reduction in the incidence of transmissible infectious diseases among children including respiratory viral infections3,4,5,6 and IBI including S. pneumoniae, H. influenzae, and Neisseria meningitidis, which are transmitted via the respiratory route.7,8 Due to the impact of the COVID-19 pandemic on the epidemiology of diverse infectious diseases, a new epidemiological trend of IBI is expected in the post COVID-19 era.
In this study, we aimed to characterize the epidemiology of IBI in children for the 25 years from 1996 to 2020. The long-term analysis can provide us the baseline data for monitoring the post-pandemic trend in the epidemiology of pediatric IBI.
METHODS
Data collection
Twenty-nine university-affiliated hospitals participated in this study. The geographic distribution of the 29 hospitals is shown in Supplementary Fig. 1. The survey was conducted during the 25-year period from January 1996 to December 2020. In this study, we analyzed pediatric IBIs for 10 years from 2011 to 2020 and generated the dataset of 25 years by combining previous two studies of pediatric IBI in 1996–20051 and in 2006–2010.2
This study included immunocompetent children aged ≥ 3 months of age and younger than 18 years of age. The data were collected each year for the diagnosed cases from January to December in the corresponding year during the 25-year period. We extracted data from hospital discharge records and reviewed retrospectively. Initially, cases with positive culture results for the eight organisms were selected and then, investigators reviewed the medical records of the cases and determined whether each case should be included in the study based on the case definition of the study protocol. A case report form was used to record the demographic characteristics, causative organisms, site(s) of bacterial isolation, clinical manifestations.
Case definition
An IBI was defined as isolation of a bacterial organism from a usually sterile site, such as blood, cerebrospinal fluid (CSF), pleural fluid, pericardial fluid, joint fluid, bone aspirate, or a deep tissue abscess. Eight pathogens (S. pneumoniae, S. aureus, H. influenzae, N. meningitidis, Streptococcus agalactiae, Streptococcus pyogenes, Listeria monocytogenes, and Salmonella species) were analyzed as they were found to be responsible for 90–95% of IBIs among children and adolescents in Korea.9,10 Co-infection was defined as the identification of more than 2 pathogens in the same episode. If the same pathogen was found in another sample taken within 30 days of the previous positive sample or during the same admission period, the episode was considered as a single case. The organisms were obtained by culture, polymerase chain reaction or latex agglutination test. Cases with medical conditions vulnerable to IBIs, such as congenital or acquired immunodeficiency, prematurity, steroid or cancer chemotherapy, and infections associated with anatomic abnormalities were excluded. Analysis of clinical diagnosis was referred from the previous studies.1,2
Statistical analysis
Age groups were classified as follows: 3 to 23 months of age, 24 to 59 months of age, and ≥ 5 years of age. The temporal changes in the relative proportions of each causative pathogen for IBIs were analyzed using the Spearman rank correlation (rs) test for trends. All tests were 2-tailed, and P < 0.05 was considered statistically significant. All data management and statistical analysis were performed using IBM SPSS version 26.0 (SPSS Inc., Chicago, IL, USA).
Ethics statement
This study was approved by the Institutional Review Board at Seoul National University Hospital (No. 1706-125-861). The requirement of obtaining informed consent was waived due to its retrospective study design.
RESULTS
Etiology of IBI
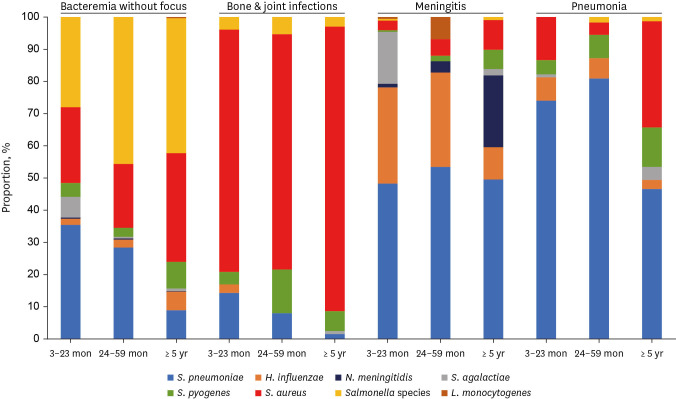
A total of 2,194 episodes of IBIs caused by 2,195 pathogens were identified in children aged 3 moths to under 18 years during the study period. There was one episode of co-infection caused by 2 pathogens in the same episode. The male to female ratio was 1.5:1. The source of the identified bacteria was available in 2,177 episodes. Same bacteria were isolated from more than two body sites in 13% (285/2,177). The source of the identified bacteria was the blood (61.7%), bone or joint fluid (19.5%), cerebrospinal fluid (11.7%), pleural fluid (3.9%), and other sterile body fluids (3.3%). The distribution of clinical diagnosis was bacteremia in 36.8%, bone and joint infection in 28.3%, meningitis in 15.2%, and pneumonia in 14.4% (Table 1). For bacteremia, S. pneumoniae accounted for 35.5% of the infants aged 3 to 23 months. On the other hand, Salmonella species was the most common causative pathogen (43.4%) in children over 24 months of age. S. pneumoniae was the most frequent cause of pneumonia and meningitis. S. aureus was the most common pathogen of bone and joint infection in all age (Fig. 1).
Table 1. Clinical diagnosis among 2,183 invasive infections from 1996 to 2020, by age group.
| Clinical diagnosis | No. of cases (%) by age group | Total | ||
|---|---|---|---|---|
| 3–23 mon | 24–59 mon | ≥ 5 yr | ||
| Bacteremia without focus | 277 (40.7) | 209 (43.5) | 321 (31.1) | 807 (36.8) |
| Bone and joint infection | 77 (11.3) | 74 (15.4) | 469 (45.4) | 620 (28.3) |
| Meningitis | 176 (25.8) | 58 (12.1) | 99 (9.6) | 333 (15.2) |
| Pneumonia | 115 (16.9) | 127 (26.5) | 75 (7.3) | 317 (14.4) |
| Deep organ abscess | 33 (4.8) | 6 (1.3) | 49 (4.7) | 88 (4.0) |
| Peritonitis | 2 (0.3) | 5 (1.0) | 10 (1.0) | 17 (0.8) |
| Infective endocarditis | 1 (0.1) | 1 (0.2) | 10 (1.0) | 12 (0.5) |
| Total | 681 (31.0) | 480 (21.9) | 1,033 (47.1) | 2,194a |
Values are presented as number (%).
aThere is one case of co-infection.
Fig. 1. Distribution of causative bacterial organisms in invasive infections in immunocompetent children between 1996 and 2020 according to age group and clinical diagnosis.
Distribution of causative bacterial organisms by age from 1996 to 2020
The number of cases by age group was 681 (31.0%) in 3–23 months, 480 (21.9%) in 24–59 months, and 1,033 (47.1%) in ≥ 5 years. Overall, S. aureus was the most frequently isolated organism (n = 875, 39.9%), followed by S. pneumoniae (n = 618, 28.2%), Salmonella spp. (n = 335, 15.3%), S. pyogenes (n = 135, 6.2%), H. influenzae (n = 132, 6.0%), and S. agalactiae (n = 64, 2.9%). There were a few cases of L. monocytogenes and N. meningitidis infections (Table 2). S. pneumoniae (42.4%), S. aureus (23.6%), and Salmonella species (15.7%) were common in children 3 to 59 months of age. In children ≥5 years of age, S. aureus (58.1%), followed by Salmonella species (14.8%) and S. pneumoniae (12.2%) were common (Table 2).
Table 2. Distribution of causative organisms for invasive bacterial infection from Jan 1996 to Dec 2020, by age group.
| Etiology | No. of cases (%) by age group | Total | |||
|---|---|---|---|---|---|
| 3–23 mon | 24–59 mon | 3–59 mon subtotal | ≥ 5 yr | ||
| S. aureus | 168 (24.7) | 106 (22.1) | 274 (23.6) | 601 (58.1) | 875 (39.9) |
| S. pneumoniae | 288 (42.3) | 204 (42.5) | 492 (42.4) | 126 (12.2) | 618 (28.2) |
| Salmonella spp. | 81 (11.9) | 101 (21.0) | 182 (15.7) | 153 (14.8) | 335 (15.3) |
| S. pyogenes | 25 (3.7) | 28 (5.8) | 53 (4.6) | 82 (7.9) | 135 (6.2) |
| H. influenzae | 68 (10.0) | 33 (6.9) | 101 (8.7) | 31 (3.0) | 132 (6.0) |
| S. agalactiae | 47 (6.9) | 1 (0.2) | 48 (4.1) | 16 (1.5) | 64 (2.9) |
| N. meningitidis | 3 (0.4) | 3 (0.6) | 6 (0.5) | 23 (2.2) | 29 (1.3) |
| L. monocytogenes | 1 (0.1) | 4 (0.8) | 5 (0.4) | 2 (0.2) | 7 (0.3) |
| Total | 681 (31.0) | 480 (21.9) | 1,161 (52.9) | 1,034 (47.1) | 2,195 |
Values are presented as number (%).
Changes in the relative proportion of bacterial organisms over time from 1996 to 2019
In this trend analysis over time, the proportion of IBI in 2020 were excluded because the distribution of causative organism in 2020 was markedly influenced by the COVID-19 pandemic and public preventive measures. Total 2,120 cases were analyzed for annual trend analysis of the relative proportion of bacterial organisms from 1996 to 2019: 484 cases from 1996–2005, 518 cases from 2006–2010 and 1,118 cases from 2011 to 2019.
The overall trends in the relative proportions of the eight causative bacterial organisms in infants and children older than 3 months of age from 1996 to 2019 are shown in Fig. 2. Statistical analysis revealed a trend toward a decrease in the relative proportions of S. pneumoniae (rs = −0.430, P = 0.036), H. influenzae (rs = −0.922, P < 0.001), while trend toward an increase in the relative proportion of S. aureus (rs = 0.850, P < 0.001), S. agalactiae (rs = 0.615, P = 0.001), and S. pyogenes (rs = 0.554, P = 0.005). To eliminate the confounding variables, the overall trends in the relative proportions of the eight organisms were analyzed with 22 hospitals which participated whole study period. The trend of relative proportion of each organisms shows similar trend and is shown in Supplementary Fig. 2.
Fig. 2. Annual trend in the proportion of eight causative organisms of invasive bacterial infections in children 3 months of age or older, 1996–2019.
aThe Spearman rank correlation (rs) test was performed using the 2-tailed test to reject the hypothesis that rs = 0 and P < 0.05 was considered statistically significant. As the proportion of invasive bacterial infections in 2020 was markedly different from the previous trend, the value of 2020 was displayed as a dotted line.
**P < 0.05.
DISCUSSION
The purpose of this study was to investigate the temporal trend change in the relative proportions of pediatric IBIs through a multicenter-based surveillance over 25 years, from 1996 to 2020 in Korea. This study includes the largest episodes of IBI in children in Korea.
The major causative organism of IBI differed according to age. S. pneumoniae, S. aureus and Salmonella species predominated in children 3 to 59 months of age. S. aureus (58.1%) was the most common pathogen in patients over 5 years of age, followed by Salmonella species (14.8%) and S. pneumoniae (12.2%). The relative proportion of S. pneumoniae and H. influenzae showed a decreasing trend from 1996 to 2019 in infants and children older than 3 months of age.
H. influenzae type b (Hib) is one of the major causes of epiglottis, pneumonia, meningitis, arthritis, pericarditis and other infections in infant and children over 3 months of age. After introduction of conjugated Hib vaccine, decline of invasive infections due to Hib infection in children was observed in worldwide.11,12 In Korea, Hib vaccine has been available since 1996 and has introduced into the National Immunization Program (NIP) in 2013.13 Following the implementation of Hib vaccination program in Korea, the vaccination coverage of Hib in 2016 was 95.0% and 96.4% in 2019.14 The decline of H. influenzae in IBI patients older than 3 months of age were observed (20.1% in the 1996–2000 period, 4.5% in the 2001–2005 period, 3.3% in the 2006–2010 period, 2.3% in the 2011–2015 period and 1.7% in the 2016–2019 period).
S. pneumoniae accounted for 45.3% of infections occurred among 3 to 59 months of age in 1996–2005 and 54.4% in 2006–2010. In the study period (2011–2020), S. pneumoniae accounted for 35.3% of IBIs, followed by S. aureus (33.3%). The most common clinical diagnosis in this age group were bacteremia without localizing signs (37.9%, 77/198), followed by pneumonia (31.8%, 63/198), and meningitis (22.2%, 44/198). In Korea, 7-valent pneumococcal conjugate vaccine (PCV7) was introduced in 2003,15 PCV10 and PCV13 were introduced in 2010 and NIP implemented PCV10 and PCV13 in 2014.16 After the introduction of PCV7 in 2003, the trend in decreased proportion of S. pneumoniae was not seen immediately. The vaccination coverage of PCV was first announced in 2017 as 96.7%, higher than 79.2% before NIP implementation.17 The reduction in proportion of S. pneumoniae was observed since 2010 through this study period (1996–2019). Unlike with Hib, S. pneumoniae has multiple serotypes that cause invasive infections, so replacement with nonvaccine serotypes among invasive pneumococcal disease after the introduction of PCVs may be responsible for the slow trend in the reduction of invasive diseases caused by S. pneumoniae. 18 While U.S., United Kingdom, and Taiwan showed a dramatic decline in IPD after introduction of PCV,19,20,21 the decreasing trend of IPD was not so evident in this study and this difference may be attributed by different study design. This study was based on retrospective, proportion-based data, while studies in U.S., United Kingdom, and Taiwan were based on prospective incidence–based surveillance.
S. aureus remains a major cause of infection and causes significant mortality and morbidity from bacteremia, osteomyelitis, pneumonia, and endocarditis in pediatric populations.22,23,24,25 In this study, we found the most frequent cause of bacterial infection in children older than 3 months of age was S. aureus and the relative proportion of S. aureus occupied more than 60% of bone and joint infection in patients of any age. An increase in relative proportion of S. aureus infections in infant and children ≥ 3 months of age is observed from 1996 to 2019. Compared to the Methicillin-resistant Staphylococcus aureus (MRSA) rate 9.1% in 1996–2000, the MRSA rate ranged between 27–38% through 2001 to 2020 in this study. However direct comparison of MRSA rate during the study period should be interpreted with caution because the case number in early period (1996–2000) was smaller than later period. As the MRSA occupied significant portions, antibiotics such as vancomycin or clindamycin may be considered in empiric guidelines for treatment of febrile infants.23,24 This study includes pre and post COVID-19 pandemic period. From 2012 to 2017, 250 bacterial infections occurred annually on average, while in 2019 and 2020, 203 and 152 bacterial infections occurred respectively. S. aureus showed a significant increasing trend and Salmonella showed a decreasing trend in 2020. In 2018, the relative proportion of S. aureus infections was 46.7%, which rose to 71.8% in 2020. In 2018, the relative proportion of Salmonella species was 23.3%, which dropped to 9.9% in 2020. Of the 196 salmonella isolates, serogroup data were available for the 194 cases; 17 (8.8%) isolates were Salmonella typhi and 177 isolates were non-typhoidal Salmonella.
During the COVID-19 pandemic, no cases of IBI due to H. influenzae and N. meningitidis and relative proportion of infection due to S. pneumoniae also declined and these findings correspond with worldwide trend.7 Several studies revealed that containment measure and lockdown reduced IBI. The containment measures prevented invasive bacterial disease by blocking bacterial acquisition and colonization due to decreased person-to-person transmission.7,8 Future study comparing the etiology of IBI in pediatric population before and after the COVID-19 pandemic should be performed.
This study had several limitations. First, as the design of this study was focused on the relative proportion of each bacterial infection, the exact incidence of IBI caused by each organism was not evaluated. Further well-designed prospective studies which analyze annual incidence of bacterial infections are needed. Second, there were some changes in the number of participating hospitals and this may affect the trend of relative proportion of causative organisms. However, 22 hospitals continued to participate the entire study period. Nonetheless, this is the only available surveillance system for IBI in children which has been maintained for 25 years.
In conclusion, the 25-year study on the epidemiology of IBI in children revealed a decreasing trend for S. pneumoniae and H. influenzae and an increasing trend for S. aureus, S. agalactiae, and S. pyogenes in children > 3 months of age. These findings can be used as the baseline data to navigate the trend in the epidemiology of pediatric IBI in the post COVID-19 era.
Footnotes
Funding: This study was supported by the Korea Disease Control and Prevention Agency (2018E240600).
Disclosure: The authors have no conflicts of interest to disclose.
- Conceptualization: Choi EH.
- Data curation: Song SH, Lee H, Lee HJ, Song ES, Ahn JG, Park SE, Lee T, Cho HK, Lee J, Kim YJ, Jo DS, Kim JH, Kang HM, Lee JK, Kim CS, Kim DH, Kim HM, Choi JH, Eun BW, Kim NH, Cho EY, Kim YK, Oh CE, Kim KH, Ma SH, Jung HJ, Lee KS, Kim KN, Choi EH.
- Formal analysis: Song SH.
- Investigation: Song SH, Lee H, Lee HJ, Song ES, Ahn JG, Park SE, Lee T, Cho HK, Lee J, Kim YJ, Jo DS, Kim JH, Kang HM, Lee JK, Kim CS, Kim DH, Kim HM, Choi JH, Eun BW, Kim NH, Cho EY, Kim YK, Oh CE, Kim KH, Ma SH, Jung HJ, Lee KS, Kim KN, Choi EH.
- Methodology: Song SH, Choi EH.
- Validation: Song SH, Choi EH.
- Writing - original draft: Song SH, Choi EH.
- Writing - review & editing: Lee H, Choi EH.
SUPPLEMENTARY MATERIALS
Geographic distribution of the 29 university-affiliated hospitals.
Annual trend in the proportion of eight causative organisms of invasive bacterial infections in children 3 months of age or older, 1996–2019 in 22 university-affiliated hospitals.
References
- 1.Lee JH, Cho HK, Kim KH, Kim CH, Kim DS, Kim KN, et al. Etiology of invasive bacterial infections in immunocompetent children in Korea (1996–2005): a retrospective multicenter study. J Korean Med Sci. 2011;26(2):174–183. doi: 10.3346/jkms.2011.26.2.174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Rhie K, Choi EH, Cho EY, Lee J, Kang JH, Kim DS, et al. Etiology of invasive bacterial infections in immunocompetent children in Korea (2006–2010): a retrospective multicenter study. J Korean Med Sci. 2018;33(6):e45. doi: 10.3346/jkms.2018.33.e45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hatoun J, Correa ET, Donahue SM, Vernacchio L. Social distancing for COVID-19 and diagnoses of other infectious diseases in children. Pediatrics. 2020;146(4):e2020006460. doi: 10.1542/peds.2020-006460. [DOI] [PubMed] [Google Scholar]
- 4.Huh K, Jung J, Hong J, Kim M, Ahn JG, Kim JH, et al. Impact of non-pharmaceutical interventions on the incidence of respiratory infections during the COVID-19 outbreak in Korea: a nationwide surveillance study. Clin Infect Dis. 2021;72(7):e184–e191. doi: 10.1093/cid/ciaa1682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lee H, Lee H, Song KH, Kim ES, Park JS, Jung J, et al. Impact of public health interventions on seasonal influenza activity during the COVID-19 outbreak in Korea. Clin Infect Dis. 2021;73(1):e132–e140. doi: 10.1093/cid/ciaa672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Nolen LD, Seeman S, Bruden D, Klejka J, Desnoyers C, Tiesinga J, et al. Impact of social distancing and travel restrictions on non–coronavirus disease 2019 (non–COVID-19) respiratory hospital admissions in young children in Rural Alaska. Clin Infect Dis. 2021;72(12):2196–2198. doi: 10.1093/cid/ciaa1328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Brueggemann AB, Jansen van Rensburg MJ, Shaw D, McCarthy ND, Jolley KA, Maiden MC, et al. Changes in the incidence of invasive disease due to Streptococcus pneumoniae, Haemophilus influenzae, and Neisseria meningitidis during the COVID-19 pandemic in 26 countries and territories in the Invasive Respiratory Infection Surveillance Initiative: a prospective analysis of surveillance data. Lancet Digit Health. 2021;3(6):e360–e370. doi: 10.1016/S2589-7500(21)00077-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Smith DR, Opatowski L. COVID-19 containment measures and incidence of invasive bacterial disease. Lancet Digit Health. 2021;3(6):e331–e332. doi: 10.1016/S2589-7500(21)00085-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lee JH, Song EK, Lee JA, Kim NH, Kim DH, Park KW, et al. Clinical entities and etiology of invasive bacterial infections in apparently healthy children. Korean J Pediatr. 2005;48(11):1193–1200. [Google Scholar]
- 10.Nam SG, Lee HJ. Etiology of invasive bacterial infections in apparently healthy children. Korean J Infect Dis. 1998;30(3):227–234. [Google Scholar]
- 11.Morris SK, Moss WJ, Halsey N. Haemophilus influenzae type b conjugate vaccine use and effectiveness. Lancet Infect Dis. 2008;8(7):435–443. doi: 10.1016/S1473-3099(08)70152-X. [DOI] [PubMed] [Google Scholar]
- 12.Suga S, Ishiwada N, Sasaki Y, Akeda H, Nishi J, Okada K, et al. A nationwide population-based surveillance of invasive Haemophilus influenzae diseases in children after the introduction of the Haemophilus influenzae type b vaccine in Japan. Vaccine. 2018;36(38):5678–5684. doi: 10.1016/j.vaccine.2018.08.029. [DOI] [PubMed] [Google Scholar]
- 13.Choi EH, Park SE, Kim YJ, Jo DS, Kim YK, Eun BW, et al. Recommended immunization schedule for children and adolescents: Committee on Infectious Diseases of the Korean Pediatric Society, 2018. Korean J Pediatr. 2019;62(7):252–256. doi: 10.3345/kjp.2019.00227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lee J, Jeong H, Kim S, Yu J, Kim G. National childhood vaccination coverage among children aged 1–3 and 6 years in Korea, 2018. Public Health Wkly Rep. 2019;12(39):1548–1558. [Google Scholar]
- 15.Cho EY, Kang HM, Lee J, Kang JH, Choi EH, Lee HJ. Changes in serotype distribution and antibiotic resistance of nasopharyngeal isolates of Streptococcus pneumoniae from children in Korea, after optional use of the 7-valent conjugate vaccine. J Korean Med Sci. 2012;27(7):716–722. doi: 10.3346/jkms.2012.27.7.716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lee JK, Yun KW, Choi EH, Kim SJ, Lee SY, Lee HJ. Changes in the serotype distribution among antibiotic resistant carriage Streptococcus pneumoniae isolates in children after the introduction of the extended-valency pneumococcal conjugate vaccine. J Korean Med Sci. 2017;32(9):1431–1439. doi: 10.3346/jkms.2017.32.9.1431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Korea Centers for Disease Control and Prevention (KCDC) National Childhood Vaccination Coverage Among Children Aged 1–3 Years in Korea, 2017. Cheongju, Korea: KCDC; 2018. [Google Scholar]
- 18.Savulescu C, Krizova P, Lepoutre A, Mereckiene J, Vestrheim DF, Ciruela P, et al. Effect of high-valency pneumococcal conjugate vaccines on invasive pneumococcal disease in children in SpIDnet countries: an observational multicentre study. Lancet Respir Med. 2017;5(8):648–656. doi: 10.1016/S2213-2600(17)30110-8. [DOI] [PubMed] [Google Scholar]
- 19.Lai CC, Lin SH, Liao CH, Sheng WH, Hsueh PR. Decline in the incidence of invasive pneumococcal disease at a medical center in Taiwan, 2000–2012. BMC Infect Dis. 2014;14(1):76. doi: 10.1186/1471-2334-14-76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tan TQ. Pediatric invasive pneumococcal disease in the United States in the era of pneumococcal conjugate vaccines. Clin Microbiol Rev. 2012;25(3):409–419. doi: 10.1128/CMR.00018-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Waight PA, Andrews NJ, Ladhani SN, Sheppard CL, Slack MP, Miller E. Effect of the 13-valent pneumococcal conjugate vaccine on invasive pneumococcal disease in England and Wales 4 years after its introduction: an observational cohort study. Lancet Infect Dis. 2015;15(5):535–543. doi: 10.1016/S1473-3099(15)70044-7. [DOI] [PubMed] [Google Scholar]
- 22.McMullan BJ, Bowen A, Blyth CC, Van Hal S, Korman TM, Buttery J, et al. Epidemiology and mortality of Staphylococcus aureus bacteremia in Australian and New Zealand children. JAMA Pediatr. 2016;170(10):979–986. doi: 10.1001/jamapediatrics.2016.1477. [DOI] [PubMed] [Google Scholar]
- 23.Ochoa TJ, Mohr J, Wanger A, Murphy JR, Heresi GP. Community-associated methicillin-resistant Staphylococcus aureus in pediatric patients. Emerg Infect Dis. 2005;11(6):966–968. doi: 10.3201/eid1106.050142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Sutter DE, Milburn E, Chukwuma U, Dzialowy N, Maranich AM, Hospenthal DR. Changing susceptibility of Staphylococcus aureus in a US pediatric population. Pediatrics. 2016;137(4):e20153099. doi: 10.1542/peds.2015-3099. [DOI] [PubMed] [Google Scholar]
- 25.Park DA, Lee SM, Peck KR, Joo EJ, Oh EG. Impact of methicillin-resistance on mortality in children and neonates with Staphylococcus aureus bacteremia: a meta-analysis. Infect Chemother. 2013;45(2):202–210. doi: 10.3947/ic.2013.45.2.202. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Geographic distribution of the 29 university-affiliated hospitals.
Annual trend in the proportion of eight causative organisms of invasive bacterial infections in children 3 months of age or older, 1996–2019 in 22 university-affiliated hospitals.