Abstract
Background
VEXAS (vacuoles, E1 enzyme, X-linked, auto-inflammatory, somatic) syndrome is a newly described auto-inflammatory disease. Many cases feature pulmonary infiltrates or respiratory failure. This systematic review aimed to summarize respiratory manifestations in VEXAS syndrome described to date.
Methods
Databases were searched for articles discussing VEXAS syndrome until May 2022. The research question was: What are the pulmonary manifestations in patients with VEXAS syndrome? The search was restricted to English language and those discussing clinical presentation of disease. Information on basic demographics, type and prevalence of pulmonary manifestations, co-existing disease associations and author conclusions on pulmonary involvement were extracted. The protocol was registered on the PROSPERO register of systematic reviews.
Results
Initially, 219 articles were retrieved with 36 ultimately included (all case reports or series). A total of 269 patients with VEXAS were included, 98.6% male, mean age 66.8 years at disease onset. The most frequently described pulmonary manifestation was infiltrates (43.1%; n = 116), followed by pleural effusion (7.4%; n = 20) and idiopathic interstitial pneumonia (3.3%; n = 9). Other pulmonary manifestations were: nonspecific interstitial pneumonia (n = 1), bronchiolitis obliterans (n = 3), pulmonary vasculitis (n = 6), bronchiectasis (n = 1), alveolar haemorrhage (n = 1), pulmonary embolism (n = 4), bronchial stenosis (n = 1), and alveolitis (n = 1). Several patients had one or more co-existing autoimmune/inflammatory condition. It was not reported which patients had particular pulmonary manifestations.
Conclusion
This is the first systematic review undertaken in VEXAS patients. Our results demonstrate that pulmonary involvement is common in this patient group. It is unclear if respiratory manifestations are part of the primary disease or a co-existing condition. Larger epidemiological analyses will aid further characterisation of pulmonary involvement and disease management.
Supplementary Information
The online version contains supplementary material available at 10.1007/s00296-022-05266-2.
Keywords: VEXAS, Autoinflammatory syndrome, Autoimmune disease, Respiratory, Pulmonary
Introduction
Vacuoles, E1 enzyme, X-linked, auto-inflammatory, somatic (VEXAS) syndrome is a recently described autoinflammatory condition [1]. First reported in 2020, it is due to somatic mutations in the UBA1 gene of myeloid cells and mainly affects older men. The disease is characterized by inflammatory and haematologic symptoms, arising from myeloid-driven autoinflammation and progressive bone marrow failure [2].
The systemic inflammation in patients with VEXAS syndrome can affect multiple organs, most commonly the skin, blood vessels, cartilage and lungs. This often leads to co-existing or misdiagnoses of other systemic rheumatic and autoinflammatory disorders such as Sweet syndrome, relapsing polychondritis and medium-large vessel vasculitides.
Features of patients with VEXAS syndrome continue to be under investigation and comprise a spectrum of phenomena arising from systemic inflammation. One of the most pertinent and consistent feature is pulmonary involvement, as for many autoinflammatory conditions [3–5]. Since VEXAS was first described, case reports and small cohort studies have reported various respiratory manifestations in a large proportion of this patient group including pulmonary embolism, parenchymal disease and pulmonary vasculitis [6–8]. Lung involvement is often severe, leading to significant morbidity and mortality [9].
To date, no systematic review has been undertaken of reported cases of VEXAS, or pulmonary manifestations of this disease. This systematic review aimed to summarize the respiratory manifestations of VEXAS syndrome, described in the literature.
Methods
This systematic review was conducted in accordance with the Cochrane Handbook and reported as per the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines [10, 11]. The protocol was developed by KK, SZ and MD, and registered in the PROSPERO database of systematic reviews (CRD42022355163). The research question was framed and structured using the ‘Patients, Intervention, Comparator or Control and Outcome’ (PICO) format: What are the pulmonary manifestations described in VEXAS syndrome?
The population was defined as adult patients with a clinician-confirmed diagnosis of VEXAS. The main outcome was pulmonary manifestations including (but not limited to): pulmonary infiltrates; idiopathic interstitial pneumonia; non-specific interstitial pneumonia (NSIP); bronchiolitis obliterans; pulmonary vasculitis; bronchiectasis; pleural effusion; alveolar haemorrhage; pulmonary embolism (and related MeSH terms). “Intervention” and “comparator” were not applicable to the search.
Search strategy, databases searched and study selection
The search strategy (available in the supplementary material) was developed by two authors (KK and MD) with the help of a librarian expert in undertaking systematic reviews and clinical research (HE). The bibliographic databases Medline, Scopus, The Cochrane Library (the Cochrane Database of Systematic Reviews, the Cochrane Central Register of Controlled Trials (CENTRAL), and the Cochrane Methodology Register), and PsycINFO were searched via the Ovid platform on 13 May 2022. The search conducted had no time restriction and was limited to English-language only. All study types, excluding opinion articles, editorials and literature reviews.
Due to the relative recency of VEXAS syndrome being described in clinical practice, the search strategy was kept deliberately broad to ensure all relevant articles were captured. The ultimate focus of the review was pulmonary manifestations described in VEXAS syndrome.
Titles and abstracts were screened by AA and MD, to assess eligibility. The full articles which met the inclusion criteria were then examined in detail by AA. For validation purposes, 10% of the articles were screened at the abstract and full paper stage by a second author, KK, with input from a third reviewer (MD) where required.
Assessment of risk of bias, data extraction and synthesis
Risk of bias in each included study was assessed using the appropriate tool for each study. Risk of bias was not possible for case reports and studies. Data extraction from the included articles was undertaken by AA, with 10% of articles also reviewed and the information extracted by KK for validation. No papers or additional data or online supplemental material were required from authors.
For each selected article, in addition to basic information and patient demographics, the following were extracted: number of reported cases with lung involvement; percentage of lung involvement and their subtypes (if described); ethnicity of sample (if available); comorbidities; and summary of authors’ conclusions.
Results
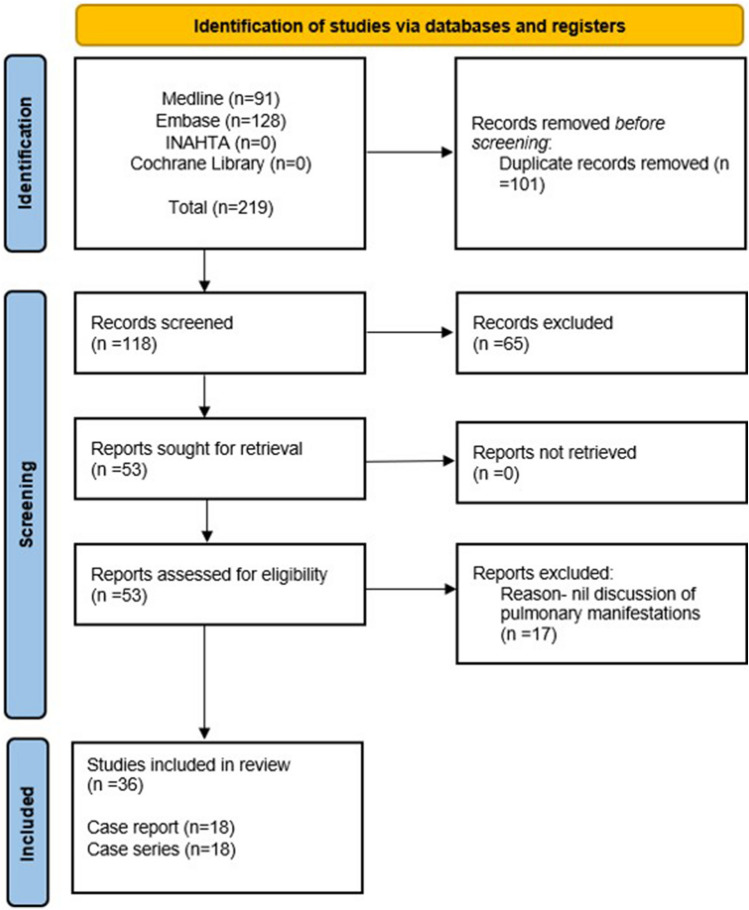
Initially, 219 articles were retrieved with 36 ultimately included (18 case reports, 18 case series; Fig. 1). A pooled total of 269 patients with VEXAS were included, 98.6% of whom were male, with an overall mean age of 66.8 years (SD 7.3) at disease onset. Cases were from: Europe (n = 181); North America (62); South America (n = 1); Asia (n = 21); Australasia (n = 4).
Fig. 1.
Flow diagram of stages of systematic literature review. INAHTA International Network of Agencies for Health Technology Assessment. Cochrane Library encompasses library of: systematic reviews; systematic review protocols; controlled clinical trials
Demographics and extracted clinical information are summarized in Table 1. 56.1% of included patients had pulmonary involvement at presentation. The most frequently described manifestation was pulmonary infiltrates (43.1%; n = 116) [1, 8, 12–34], followed by pleural effusion (7.4%; n = 20) [8, 18, 20, 22, 24, 27, 28, 32, 35] and idiopathic interstitial pneumonia (3.3%; n = 9) [14, 18, 25, 27, 28, 32, 36, 37]. Other pulmonary manifestations described were NSIP (n = 1) [14]; bronchiolitis obliterans (n = 3) [14]; pulmonary vasculitis (n = 6) [14, 24]; bronchiectasis (n = 1) [14]; alveolar haemorrhage (n = 1) [38]; pulmonary embolism (n = 4) [35, 39–41]; bronchial stenosis (n = 1) [42]; and alveolitis (n = 1) [36].
Table 1.
Article information, cohort demographics, and pulmonary manifestations, including sample size (n) for each type
| Article information | Sample size and demographics | Overall pulmonary involvement | Pulmonary manifestations | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Source | Country of study population | Type of study | Sample size (with VEXAS) | Male (%) | Mean age at onset (years) | Pulmonary involvement (n) | Pulmonary involvement (%) | Infiltrates (n) | Idiopathic interstitial pneumonia (n) | NSIP (n) | Bronchiolitis obliterans (n) | Pulmonary vasculitis (n) | Bronchiectasis (n) | Pleural effusion (n) | Alveolar haemorrhage (n) | PE (n) | Bronchial stenosis and wall thickening (n) | Alveolitis (n) |
| Van der Made 2022 [14] | Netherlands | Case series | 12 | 100 | 67 | 8 | 66.7 | 1 | 1 | 1 | 3 | 5 | 1 | 0 | 0 | 0 | 0 | 0 |
| Shaukat 2022 [20] | USA | Case report | 1 | 100 | 80 | 1 | 100 | 1 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 |
| Sharma 2022 [21] | India | Case report | 1 | 100 | 70 | 1 | 100 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Pamies 2022 [23] | Spain | Case report | 1 | 100 | 76 | 1 | 100 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Muratore 2022 [32] | Italy | Case series | 3 | 100 | 67.7 | 3 | 100 | 2 | 0 | 0 | 0 | 1 | 0 | 1 | 0 | 0 | 0 | 0 |
| Matsumoto 2022 [38] | Japan | Case report | 1 | 100 | 60 | 1 | 100 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 |
| Matsubara 2022 [25] | Japan | Case report | 1 | 100 | 68 | 1 | 100 | 1 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Martin-Nares 2022 [26] | Mexico | Case report | 1 | 100 | 77 | 1 | 100 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Loschi 2022 [39] | France | Case report | 1 | 100 | 60 | 1 | 100 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 |
| Li 2022 [27] | USA | Case series | 2 | 100 | 65 | 1 | 50 | 1 | 1 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 |
| Kao 2022 [22] | USA | Case report | 1 | 100 | 50 | 1 | 100 | 1 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 |
| Islam 2022 [28] | Australia | Case series | 3 | 100 | 67.7 | 2 | 66.7 | 2 | 2 | 0 | 0 | 0 | 0 | 2 | 0 | 0 | 0 | 0 |
| Goyal 2022 [29] | USA | Case report | 1 | 100 | 64 | 1 | 100 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Georgin-Lavialle 2022 [8] | France | Case series | 116 | 95.7 | 71 | 57 | 49.1 | 47 | 0 | 0 | 0 | 0 | 0 | 11 | 0 | 0 | 0 | 0 |
| Diarra 2022 [30] | France | Case series | 6 | 100 | 52.7 | 2 | 33.3 | 2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Ciferska 2022 [31] | Czech Republic | Case series | 3 | 100 | 72.7 | 1 | 33.3 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Beaumesnil 2022 [42] | France | Case series | 3 | 100 | 1 | 33.3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | |
| Afsahi 2022 [32] | USA | Case series | 2 | 100 | 75.5 | 1 | 50 | 1 | 1 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 |
| Zakine 2021 [44] | France | Case series | 8 | 100 | 2 | 25 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| Tsuchida 2021 [43] | Japan | Case series | 14 | 85.7 | 72.1 | 7 | 50 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Thomas 2021 [33] | USA | Case report | 1 | 100 | 62 | 1 | 100 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Takahashi 2021 [34] | Japan | Case report | 1 | 100 | 55 | 1 | 100 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Staels 2021 [7] | Belgium | Case series | 2 | 100 | 72.5 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Sakuma 2021 [12] | Japan | Case report | 1 | 100 | 61 | 1 | 100 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Poulter 2021 [13] | UK | Case series | 7 | 85.7 | 72.7 | 2 | 28.5 | 2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Poulter 2021 [15] | UK | Case series | 10 | 100 | 70 | 2 | 20 | 2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Oo 2021 [40] | Singapore | Case report | 1 | 100 | 69 | 1 | 100 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 |
| Obiorah 2021 [16] | USA | Case series | 16 | 100 | 60.6 | 14 | 87 | 14 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Lotscher 2021 [17] | Switzerland | Case report | 1 | 100 | 68 | 1 | 100 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Lee 2021 [41] | Singapore | Case report | 1 | 100 | 69 | 1 | 100 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 |
| Himmelmann 2021 [18] | Switzerland | Case report | 1 | 100 | 77 | 1 | 100 | 1 | 1 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 |
| Grey 2021 [35] | Australia | Case report | 1 | 100 | 74 | 1 | 100 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 1 | 0 | 0 |
| Ferrada 2021 [19] | USA and UK | Case series | 13 | 100 | 59.8 | 10 | 77 | 10 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Delplanque 2021 [36] | France | Case series | 6 | 83 | 66.1 | 2 | 33.3 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 |
| Dehghan 2021 [37] | UK | Case report | 1 | 100 | 55 | 1 | 100 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Beck 2020 [1] | USA and UK | Case series | 25 | 100 | 62.6 | 18 | 72 | 18 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
PE pulmonary embolism
With regards to co-existing autoimmune and/or autoinflammatory diseases, pulmonary involvement was described in patients previously diagnosed with or meeting diagnostic criteria for several conditions: relapsing polychondritis (n = 33) [14, 19, 23, 31, 41–43]; ANCA-associated-vasculitis (AAV; n = 1) [24]; antibody-negative vasculitis (n = 9) [14]; IgA-vasculitis (n = 1) [23]; systemic lupus erythematosus (n = 1) [21]; Sweet syndrome (n = 2) [25, 44]; Behcet’s disease (n = 1) [38]. Pulmonary involvement in patients with these co-existing conditions, where described, are summarized in Table 1.
Discussion
Pulmonary manifestations are commonly described in patients with VEXAS syndrome, with over 50% of those included in our review having at least one type of lung pathology at initial presentation. Alongside skin lesions and haematologic manifestations, these are the most frequently described features of VEXAS and a leading cause of mortality in these patients [1, 2].
Pulmonary manifestations
The most commonly described manifestation was pulmonary infiltrates, present in 43% of cases included in our review, often co-existing with other lung pathologies such as NSIP, pulmonary vasculitis and pleural effusion [14]. A cohort study of 116 patients from the French multicentre registry, one of the largest case series in our review, also noted lung pathologies to be one of the most common clinical features of VEXAS and associated with mortality, with pulmonary infiltrates the most frequently described manifestation [8]. When assessing phenotype–genotype correlations, lung infiltrates were more common in those with the UBA1 p.Met41Thr or p.Met41Val mutations, compared with p.Met41Leu [8]. A separate study also found these two mutations to have increased association with pulmonary involvement [24].
Pulmonary embolism was identified in four cases included in our review, one co-existing with haemophagocytic lymphohistiocytosis, another with Kikuchi-Fujimoto disease (an autoinflammatory condition characterized by necrotising lymphadenitis and fever) [35, 41]. Incidence of venous thromboembolic events in VEXAS has been reported as being as high as 36% while arterial thrombotic events have an estimated incidence of 1.6% [40]. This is in part likely due to decreased ubiquitylation as a result of UBA1 gene mutation, leading to chronic inflammation and haemostatic and endothelial dysfunction [40]. As with immunosuppression, prophylaxis and treatment of thrombotic events in patients with VEXAS requires further study.
Previous and coexisting autoimmune conditions
Our review identified several co-existing autoimmune and/or autoinflammatory conditions, with predominance of respiratory involvement, in patients diagnosed with VEXAS syndrome, most commonly relapsing polychondritis (n = 33). Patients had typical pulmonary manifestations seen in this condition, such as bronchial stenosis and bronchial wall thickening [42]. VEXAS was also diagnosed in patients with vasculitis (including Behcet’s syndrome), in some cases in conjunction with relapsing polychondritis [23]. VEXAS syndrome shares many features of vasculitis, not only pulmonary involvement, but also skin, haematological and ocular features, as well as preponderance in people of an older age. We identified one case of VEXAS in ANCA-associated vasculitis, which was characterized by pulmonary vasculitis in addition to rapidly progressive renal failure with necrotising glomerulonephritis, inflammatory manifestations and myelodysplastic features, resistant to multiple forms of immunosuppression (including cyclophosphamide and rituximab) and requiring high-dose corticosteroids to induce remission [24]. In the case of Behcet’s syndrome co-existing with VEXAS, myelodysplastic features were also present, with ground-glass changes seen on computer tomography (CT) scanning, characteristic of several cases included in our review [38]. This is not unusual in patients with Behcet’s, but it raises the need to consider VEXAS and UBA1 genetic variants especially in males with myelodysplasia.
Points to consider in treatment
Early identification of VEXAS in patients with other autoinflammatory conditions not only aids diagnosis but, importantly, can expedite management of the condition. Treatment of VEXAS is an evolving area of research and it is beyond the scope of this article to be able to comment on potential therapeutic options from the evidence presented here. Treatment strategies used in the included cases varied markedly, and included high-dose corticosteroids (most commonly reported), conventional synthetic disease-modifying antirheumatic drugs (DMARDs) including methotrexate and mycophenolate, and biologic DMARDS, including cyclophosphamide, IL-1 antagonists, IL-6 inhibitors, TNF inhibitors, rituximab and JAK inhibitors [1, 8, 24, 26, 29, 32, 35, 42]. There was one reported case of the use of ruxolitinib followed by allogeneic stem cell transplant [39]. It was often unclear in many of the cases, especially in series, as to which patients received which therapy, or were reported to have cycled through multiple agents. If reported, it was unclear whether the pulmonary manifestations were a factor in deciding which treatment to administer. Certainly, the use of methotrexate, for example, suggests that pulmonary disease was less of a concern in these particular patients. The heterogeneity of reported therapies demonstrates the many possible disease presentations and organ involvement, as well as the fact that this remains an active area of research. Similar to many autoinflammatory conditions, however, high-dose corticosteroids were the most commonly administered and effective treatment, as reported in the first article by Beck et al. [1]. Therapeutic strategies from conditions related to VEXAS may aid its management especially where there is pulmonary involvement given this is a leading cause of morbidity and mortality in these patients.
Conclusion
In conclusion, pulmonary disease is common in patients with VEXAS, most frequently described as pulmonary infiltrates. To our knowledge, this is the first systematic review conducted in patients with VEXAS syndrome, including on one of its commonest manifestations, lung involvement. It is unclear if respiratory manifestations are part of the primary disease or a separate pathology (including those predating the development of VEXAS). Based on our review it is also not clear whether those with pulmonary manifestation had any history of chronic lung conditions or not, which, if present, will likely skew respiratory outcomes and morbidity.
Our data will aid consideration of VEXAS in those with other autoimmune and autoinflammatory conditions reported in the literature, and can help to guide management strategies in this diverse patient cohort. Further epidemiological and larger cohort analyses in VEXAS patients are required to aid further characterisation of pulmonary involvement and disease management.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
The above data has been presented previously at the American College of Rheumatology Meeting 2022, and the abstract can be accessed at: https://acrabstracts.org/abstract/pulmonary-manifestations-in-vexas-vacuoles-e1-enzyme-x-linked-autoinflammatory-somatic-syndrome-a-systematic-literature-review/.
Author contributions
All co-authors take full responsibility for the integrity and accuracy of all aspects of the work.
Funding
No funding to declare.
Data availability
Data available upon request.
Declarations
Conflict of interest
Authors KK, AA, SZ and MD declare no conflicts of interest.
Ethical approval
Ethical approval not applicable.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Beck DB, Ferrada MA, Sikora KA, Ombrello AK, Collins JC, Pei W, et al. Somatic mutations in UBA1 and severe adult-onset autoinflammatory disease. N Engl J Med. 2020;383(27):2628–2638. doi: 10.1056/NEJMoa2026834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Grayson PC, Patel BA, Young NS. VEXAS syndrome. Blood. 2021;137(26):3591–3594. doi: 10.1182/blood.2021011455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Jamshidi A, Aslani S, Mahmoudi M (2018) Pulmonary manifestations of autoinflammatory disorders. Pulm Manif Prim Immunodefic Dis 193–211
- 4.Gulati M, Mani NBS, Singh P, Suri S. Pulmonary manifestations of Behçet’s disease. Thorax. 2001;56(7):572–578. doi: 10.1136/thorax.56.7.572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Dubey S, Gelder C, Pink G, Ali A, Taylor C, Shakespeare J, et al. (2021) Respiratory subtype of relapsing polychondritis frequently presents as difficult asthma: a descriptive study of respiratory involvement in relapsing polychondritis with 13 patients from a single UK centre. ERJ Open Res 7(1):00170–2020. 10.1183/23120541.00170-2020 [DOI] [PMC free article] [PubMed]
- 6.Casal Moura M, Baqir M, Tandon Y, Samec M, Reichard K, Mangaonkar A, et al. POS1377 LUNG INVOLVEMENT IN VEXAS SYNDROME. Ann Rheum Dis. 2022;81(Suppl 1):1028.1–1028. doi: 10.1136/annrheumdis-2022-eular.5232. [DOI] [Google Scholar]
- 7.Staels F, Betrains A, Woei-A-Jin FJSH, Boeckx N, Beckers M, Bervoets A, et al. Case report: VEXAS syndrome: from mild symptoms to life-threatening macrophage activation syndrome. Front Immunol. 2021;12:678927. doi: 10.3389/fimmu.2021.678927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Georgin-Lavialle S, Terrier B, Guedon AF, Heiblig M, Comont T, Lazaro E, et al. Further characterization of clinical and laboratory features in VEXAS syndrome: large-scale analysis of a multicentre case series of 116 French patients*. Br J Dermatol. 2022;186(3):564–574. doi: 10.1111/bjd.20805. [DOI] [PubMed] [Google Scholar]
- 9.(2022) A review of VEXAS syndrome in 116 French patients. Br J Dermatol 186: e115–e115. 10.1111/bjd.20989
- 10.Cochrane Handbook for Systematic Reviews of Interventions | Cochrane Training. https://training.cochrane.org/handbook/current. Accessed 31 Aug 2020
- 11.Page MJ, Moher D, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. (2021) PRISMA 2020 explanation and elaboration: updated guidance and exemplars for reporting systematic reviews. BMJ. 372. https://www.bmj.com/content/372/bmj.n160. Accessed 3 Oct 2021 [DOI] [PMC free article] [PubMed]
- 12.Sakuma M, Tanimura A, Yasui S, Ishiguro K, Kobayashi T, Ohshiro Y, et al. A Case of polychondritis-onset refractory organizing pneumonia with cytopaenia diagnosed as VEXAS syndrome: the disease course of 7 years. Rheumatology. 2021;60(10):e356–e359. doi: 10.1093/rheumatology/keab349. [DOI] [PubMed] [Google Scholar]
- 13.Poulter J, Gough A, Isaacs JD, Green M, McHugh N, Hordon L, et al. A high-throughput amplicon screen for somatic UBA1 variants in cytopenic and giant cell arteritis cohorts. J Clin Immunol. 2022;42(5):947–951. doi: 10.1007/s10875-022-01258-w. [DOI] [PubMed] [Google Scholar]
- 14.van der Made CI, Potjewijd J, Hoogstins A, Willems HPJ, Kwakernaak AJ, de Sevaux RGL, et al. Adult-onset autoinflammation caused by somatic mutations in UBA1: a Dutch case series of patients with VEXAS. J Allergy Clin Immunol. 2022;149(1):432–439.e4. doi: 10.1016/j.jaci.2021.05.014. [DOI] [PubMed] [Google Scholar]
- 15.Poulter JA, Collins JC, Cargo C, De Tute RM, Evans P, Ospina Cardona D, et al. Novel somatic mutations in UBA1 as a cause of VEXAS syndrome. Blood. 2021;137(26):3676. doi: 10.1182/blood.2020010286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Obiorah IE, Patel BA, Groarke EM, Wang W, Trick M, Ombrello AK, et al. Benign and malignant hematologic manifestations in patients with VEXAS syndrome due to somatic mutations in UBA1. Blood Adv. 2021;5(16):3203–3215. doi: 10.1182/bloodadvances.2021004976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lötscher F, Seitz L, Simeunovic H, Sarbu AC, Porret NA, Feldmeyer L, et al. Case report: genetic double strike: VEXAS and TET2-positive myelodysplastic syndrome in a patient with long-standing refractory autoinflammatory disease. Front. 2022;12:5984. doi: 10.3389/fimmu.2021.800149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Himmelmann A, Brücker R. The VEXAS syndrome: uncontrolled inflammation and macrocytic anaemia in a 77-year-old male patient. Eur J Case Rep Intern Med. 2021;8(4):002484. doi: 10.12890/2021_002484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ferrada MA, Sikora KA, Luo Y, Wells KV, Patel B, Groarke EM, et al. Somatic mutations in UBA1 define a distinct subset of relapsing polychondritis patients with VEXAS. Arthritis Rheumatol. 2021;73(10):1886–1895. doi: 10.1002/art.41743. [DOI] [PubMed] [Google Scholar]
- 20.Shaukat F, Hart M, Burns T, Bansal P. UBA1 and DNMT3A mutations in VEXAS syndrome. A case report and literature review. Mod Rheumatol Case Reports. 2022;6(1):134–139. doi: 10.1093/mrcr/rxab021. [DOI] [PubMed] [Google Scholar]
- 21.Sharma A, Naidu G, Deo P, Beck DB. VEXAS syndrome with systemic lupus erythematosus: expanding the spectrum of associated conditions. Arthritis Rheumatol. 2022;74(2):369–371. doi: 10.1002/art.41957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kao RL, Jacobsen AA, Billington CJ, Yohe SL, Beckman AK, Vercellotti GM, et al. A case of VEXAS syndrome associated with EBV-associated hemophagocytic lymphohistiocytosis. Blood Cells, Mol Dis. 2022;93:102636. doi: 10.1016/j.bcmd.2021.102636. [DOI] [PubMed] [Google Scholar]
- 23.Pàmies A, Ferràs P, Bellaubí-Pallare N, Gimenez T, Raventós A, Colobran R. VEXAS syndrome: relapsing polychondritis and myelodysplastic syndrome with associated immunoglobulin A vasculitis. Rheumatol (United Kingdom) 2022;61(3):E69–71. doi: 10.1093/rheumatology/keab782. [DOI] [PubMed] [Google Scholar]
- 24.Muratore F, Marvisi C, Castrignanò P, Nicoli D, Farnetti E, Bonanno O, et al. VEXAS syndrome: a case series from a single-center cohort of italian patients with vasculitis. Arthritis Rheumatol. 2022;74(4):665–670. doi: 10.1002/art.41992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Matsubara A, Tsuchida N, Sakurai M, Maeda A, Uchiyama Y, Sasaki K, et al. A case of VEXAS syndrome with sweet’s disease and pulmonary involvement. J Dermatol. 2022;49(5):e177–e178. doi: 10.1111/1346-8138.16311. [DOI] [PubMed] [Google Scholar]
- 26.Martín-Nares E, Vargas-Serafín C, Delgado-de la Mora J, Montante-Montes de Oca D, Grayson PC, Larios E, et al. Orbital and periorbital inflammation in VEXAS syndrome. Scand J Rheumatol. 2022;51(4):338–341. doi: 10.1080/03009742.2022.2045791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Li P, Venkatachalam S, Cordona DO, Wilson L, Kovacsovics T, Moser KA, et al. A clinical, histopathological, and molecular study of two cases of VEXAS syndrome without a definitive myeloid neoplasm. Blood Adv. 2022;6(2):405–409. doi: 10.1182/bloodadvances.2021005243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Islam S, Cullen T, Sumpton D, Damodaran A, Heath D, Bosco A, et al. VEXAS syndrome: lessons learnt from an early Australian case series. Intern Med J. 2022;52(4):658–662. doi: 10.1111/imj.15742. [DOI] [PubMed] [Google Scholar]
- 29.Goyal A, Narayanan D, Wong W, Laga AC, Connell NT, Ritter SY, et al. Tocilizumab for treatment of cutaneous and systemic manifestations of vacuoles, E1 enzyme, X-linked, autoinflammatory, somatic (VEXAS) syndrome without myelodysplastic syndrome. JAAD Case Reports. 2022;23:15–19. doi: 10.1016/j.jdcr.2022.02.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Diarra A, Duployez N, Fournier E, Preudhomme C, Coiteux V, Magro L, et al. Successful allogeneic hematopoietic stem cell transplantation in patients with VEXAS syndrome: a 2-center experience. Blood Adv. 2022;6(3):998–1003. doi: 10.1182/bloodadvances.2021004749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ciferská H, Gregová M, Klein M, Šenolt L, Maaloufová JS, Pavelka K, et al. VEXAS syndrome: a report of three cases. Clin Exp Rheumatol. 2022;40(7):1449. doi: 10.55563/clinexprheumatol/3z07e9. [DOI] [PubMed] [Google Scholar]
- 32.Afsahi V, Christensen RE, Alam M. (2022) VEXAS syndrome in dermatology. Arch Dermatol Res. https://pubmed.ncbi.nlm.nih.gov/35201420/. Accessed 26 Sep 2022 [DOI] [PubMed]
- 33.Thomas VT, Penmetcha M. Myelodysplastic syndrome associated with auto-immune inflammatory disease in VEXAS syndrome. J Hematol. 2021;10(6):274. doi: 10.14740/jh940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Takahashi N, Takeichi T, Nishida T, Sato J, Takahashi Y, Yamamura M, et al. Extensive multiple organ involvement in VEXAS syndrome. Arthritis Rheumatol (Hoboken, NJ) 2021;73(10):1896–1897. doi: 10.1002/art.41775. [DOI] [PubMed] [Google Scholar]
- 35.Grey A, Cheong PL, Lee FJ, Abadir E, Favaloro J, Yang S, et al. A case of VEXAS syndrome complicated by hemophagocytic lymphohistiocytosis. J Clin Immunol. 2021;41(7):1648–1651. doi: 10.1007/s10875-021-01070-y. [DOI] [PubMed] [Google Scholar]
- 36.Delplanque M, Aouba A, Hirsch P, Fenaux P, Graveleau J, Malard F, et al. USAID associated with myeloid neoplasm and VEXAS syndrome: two differential diagnoses of suspected adult onset still’s disease in elderly patients. J Clin Med. 2021;10(23):5586. doi: 10.3390/jcm10235586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Dehghan N, Marcon KM, Sedlic T, Beck DB, Dutz JP, Chen LYC. Vacuoles, E1 enzyme, X-linked, autoinflammatory, somatic (VEXAS) syndrome: fevers, myalgia, arthralgia, auricular chondritis, and erythema nodosum. Lancet. 2021;398(10300):621. doi: 10.1016/S0140-6736(21)01430-6. [DOI] [PubMed] [Google Scholar]
- 38.Matsumoto H, Asano T, Tsuchida N, Maeda A, Yoshida S, Yokose K, et al. Behçet’s disease with a somatic UBA1 variant: Expanding spectrum of autoinflammatory phenotypes of VEXAS syndrome. Clin Immunol. 2022;238:108996. doi: 10.1016/j.clim.2022.108996. [DOI] [PubMed] [Google Scholar]
- 39.Loschi M, Roux C, Sudaka I, Ferrero-Vacher C, Marceau-Renaut A, Duployez N, et al. Allogeneic stem cell transplantation as a curative therapeutic approach for VEXAS syndrome: a case report. Bone Marrow Transplant. 2022;57(2):315–318. doi: 10.1038/s41409-021-01544-y. [DOI] [PubMed] [Google Scholar]
- 40.Oo TM, Koay JTJ, Lee SF, Lee SMS, Lim XR, Fan BE. Thrombosis in VEXAS syndrome. J Thromb Thrombolysis. 2022;53(4):965–970. doi: 10.1007/s11239-021-02608-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Lee SMS, Fan BE, Lim JHL, Goh LL, Lee JSS, Koh LW. A case of VEXAS syndrome manifesting as Kikuchi-Fujimoto disease, relapsing polychondritis, venous thromboembolism and macrocytic anaemia. Rheumatology (Oxford) 2021;60(9):E304–E306. doi: 10.1093/rheumatology/keab200. [DOI] [PubMed] [Google Scholar]
- 42.Beaumesnil S, Boucher S, Lavigne C, Urbanski G, Lacombe V. Ear, nose, throat, and bronchial involvements in VEXAS syndrome: specifying the spectrum of clinical features. JAMA Otolaryngol Neck Surg. 2022;148(3):284–286. doi: 10.1001/jamaoto.2021.4092. [DOI] [PubMed] [Google Scholar]
- 43.Tsuchida N, Kunishita Y, Uchiyama Y, Kirino Y, Enaka M, Yamaguchi Y, et al. Pathogenic UBA1 variants associated with VEXAS syndrome in Japanese patients with relapsing polychondritis. Ann Rheum Dis. 2021;80(8):1057–1061. doi: 10.1136/annrheumdis-2021-220089. [DOI] [PubMed] [Google Scholar]
- 44.Zakine E, Schell B, Battistella M, Vignon-Pennamen MD, Chasset F, Mahévas T, et al. UBA1 variations in neutrophilic dermatosis skin lesions of patients with VEXAS syndrome. JAMA Dermatol. 2021;157(11):1349–1354. doi: 10.1001/jamadermatol.2021.3344. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data available upon request.