Abstract
To assess the prevalence and management of atrial thrombus in patients with nonvalvular atrial fibrillation (NVAF) and identify the risk factors of the nonresolution of atrial thrombus. This single-center retrospective observational study consecutively enrolled patients with NVAF and atrial thrombus detected using transesophageal echocardiography (TEE) or cardiac computed tomography angiography (CTA) from January 2012 to December 2020. Patients with prior left atrial appendage (LAA) intervention were excluded. The primary endpoint was the presence of atrial thrombus, while the secondary endpoint was the complete resolution of atrial thrombus. The prevalence of atrial thrombus in patients with NVAF was 1.4%. Ninety patients with atrial thrombus (mean age 62.8 ± 11.9 years and 61.1% men) were finally analyzed. Atrial thrombus was in the LAA in 82 (91.1%) patients. During follow up, 60% of the patients showed complete resolution of atrial thrombus. Congestive heart failure (odds ratio [OR]: 8.94; 95% confidence interval [CI]: 1.67-47.80) and a history of ischemic stroke (OR: 8.28; 95% CI: 1.48-46.42) were independently associated with the risk of the nonresolution of atrial thrombus. The presence of atrial thrombus in patients with NVAF who received anticoagulation therapy is non-negligible. Even in anticoagulated patients, TEE or cardiac CTA might still be needed. Congestive heart failure and a history of ischemic stroke are risk factors of the nonresolution of atrial thrombus.
Keywords: atrial fibrillation, atrial thrombus, warfarin, non-vitamin K antagonist oral anticoagulants, resolution
Introduction
Atrial fibrillation (AF) affects approximately 3% of the general population, and its prevalence is increasing in the ageing population. 1 It promotes the formation of atrial thrombus, which is associated with thromboembolism and poor prognosis. 2 The CHA2DS2-VASc score (Congestive Heart Failure, Hypertension, Age ≥75 [Doubled], Diabetes Mellitus, Prior Stroke or Transient Ischemic Attack [Doubled], Vascular Disease, Age 65-74, Female) is the most recommended stratification tool for estimating the risk of thromboembolism in patients with nonvalvular atrial fibrillation (NVAF).3–5 However, the actual risk of atrial thrombus formation remains unclear.
All current guidelines by large cardiologic societies agree that patients with AF should take therapeutic oral anticoagulants (OACs) for at least 3 weeks or undergo transesophageal echocardiography (TEE) to rule out atrial thrombus before catheter ablation or cardioversion to minimize the risk of procedure-related thromboembolism.5,6 However, whether TEE could be completely avoided in patients undergoing AF ablation or cardioversion on uninterrupted anticoagulation for at least 3 weeks remains controversial.
In patients with NVAF and atrial thrombus, effective anticoagulation for at least 3 weeks and follow-up TEE are recommended to ensure thrombus resolution before cardioversion. 4 However, anticoagulation in patients with AF and atrial thrombus is challenging, since atrial thrombus may remain unresolved even after anticoagulation in some patients. The risk factors of the nonresolution of atrial thrombus have not also been fully studied.
Our study aimed to assess the prevalence and management of atrial thrombus in patients with NVAF before catheter ablation or cardioversion and identify the risk factors of the nonresolution of atrial thrombus.
Methods
Patients and Study Design
The study protocol was approved by the Ethics Committee of the First Affiliated Hospital of Nanjing Medical University, Nanjing, China (reference No. 2014-SR-113). Between January 2012 and December 2020, 6808 patients with NVAF scheduled for catheter ablation or cardioversion were admitted to our center and underwent TEE or cardiac computed tomography (CT) angiography (CTA). Patients with atrial thrombus confirmed on TEE or cardiac CTA were eligible for inclusion. Meanwhile, patients with a history of percutaneous left atrial appendage (LAA) occlusion or surgical LAA excision were excluded. The written informed consent for patients was exempted by the Ethics Committees for its retrospective nature. The demographic data, clinical data, and laboratory findings were retrieved from electronic medical records.
Stroke Risk Stratification
According to the CHA2DS2-VASc scores, the patients were stratified into 3 stroke risk categories: low (score 0 in men or 1 in women), intermediate (score 1 in men or 2 in women), and high risks (score ≥2 in men or ≥3 in women). Patients with hypertrophic cardiomyopathy were considered to be at a high risk for stroke.
Appropriate Anticoagulation Definition
The OACs used in our study included dabigatran, rivaroxaban, and warfarin. The choice of OACs was left at the discretion of the treating cardiologist. In accordance with guidelines, 4 the standard dose of dabigatran used was 150 mg twice daily (bis in die [BID]), and a lower dose of 110 mg BID was appropriate for elderly patients (age of ≥80 years), patients who concomitantly used verapamil, or patients with an increased bleeding risk (HAS-BLED score [Hypertension, Abnormal Renal/Liver Function, Stroke, Bleeding History or Predisposition, Labile INR, Elderly, Drugs/Alcohol Concomitantly] of ≥3). The standard dose of rivaroxaban was 20 mg once daily (quaque die [QD]); a reduced dose of 15 mg QD was appropriate for patients with a creatinine clearance rate of 15 to 49 mL/min calculated using the Cockcroft–Gault equation. An appropriate anticoagulation in patients treated with warfarin was defined as an on-admission prothrombin time-international normalized ratio (PT-INR) of 2.0 to 3.0.
Thrombus Assessment
TEE or cardiac CTA was selected at the discretion of the treating electrophysiologist and patient's preference should also be considered.
Transesophageal Echocardiography
We utilized the Philips EPIQ 7C ultrasound machine with the X7-1 probe. Multiple tomographic plane images were obtained to determine the presence of thrombus within the atrium. The blood flow status in the atrium was described as normal, smoke, sludge, or thrombus. Our study only focused on thrombus in the atrium, which was defined as a hypoechoic mass that could be detected in multiple views. The thrombus should be acoustically distinct from the pectinate muscles in the atrium. Images were interpreted by 2 experienced echocardiographers.
Cardiac CTA
As a high-volume tertiary center, our institution performs about 1500 cardiac CTAs per year. Therefore, abundant experience in performing cardiac CTA has been accumulated. In this study, 2-phase cardiac images were obtained using a dual-source 256-section multidetector CT system (Somatom Force; Siemens). LAA hemodynamics was assessed via delineation of filling defects using 2-phase cardiac CT. Filling defects were defined as areas of low attenuation that were not caused by motion artifacts or cardiac structures, such as the atrial trabeculae. 7 Circumscribed contrast-enhanced filling defects were identified as atrial thrombi. 8 The images were interpreted by 2 experienced radiologists.
Endpoints
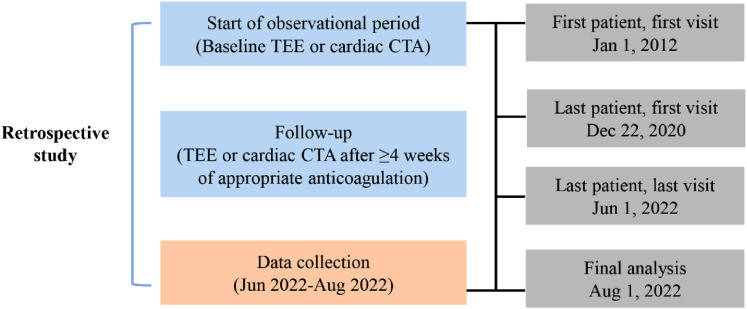
The primary endpoint was the presence of atrial thrombus, while the secondary endpoint was the complete resolution of atrial thrombus, ischemic stroke, major bleeding, and all-cause death. The patients were followed up from the date of atrial thrombus detection to the date of death, loss of follow up, or end of follow up (June 1, 2022), whichever occurred first (Figure 1).
Figure 1.
The study design.
Statistical Analysis
Continuous variables were presented as means ± standard deviations or medians (interquartile ranges [IQRs]) and compared using Student's t test or the Mann–Whitney U test, as appropriate. Categorical variables were presented as numbers (percentages) and compared using the chi-squared test or Fisher's exact test. Variables with P < .03 in the univariate analysis were entered into the multivariable logistic model to identify the risk factors of the nonresolution of atrial thrombus. Odds ratios (ORs) and the corresponding 95% confidence intervals (CIs) were estimated. Areas under the receiver operating characteristic (ROC) curves (AUC) with 95% CIs were used to assess the discriminatory capacity of the independent risk factors derived from the multivariable logistic model. In addition, we assessed the predictive value of the contemporary stroke risk stratification schemes, including the CHA2DS2-VASc score, CHADS2 score, 9 modified CHADS2 scheme by Rietbrock et al, 10 and 8th American College of Chest Physicians guidelines 11 on the nonresolution of atrial thrombus. P < .05 was considered statistically significant. All statistical analyses were performed using Statistical Package for the Social Sciences (SPSS).
Results
Prevalence of Atrial Thrombus in Patients With NVAF
During the study period, atrial thrombi were detected in 95 patients. The prevalence of atrial thrombus in patients with NVAF was 1.4% (95 of 6808). After the exclusion of 5 patients with a history of LAA intervention, 90 patients were finally included in our analysis (Figure 2). TEE was used to detect LA/LAA thrombus in 49 (54.4%), cardiac CTA in 41 (45.6%), and both TEE and cardiac CTA in 10 (11.1%) patients.
Figure 2.
Patient flow chart. Abbreviations: CTA, computed tomography angiography; LAA, left atrial appendage; TEE, transesophageal echocardiography
Characteristics of Patients With NVAF and Atrial Thrombus
The baseline characteristics of the 90 included patients are shown in Table 1. In total, 55 (61.1%) patients were men, and the mean patient age was 62.8 ± 11.9 years. Approximately 75.6% of the patients presented with nonparoxysmal AF. The mean CHA2DS2-VASc score was 3.0 ± 1.9. The most common comorbidities were hypertension (57.8%), heart failure (43.3%), stroke or transient ischemic attack (33.3%), and diabetes (25.6%). Hypertrophic cardiomyopathy was diagnosed in 13.3% of the patients. Further, 67 (74.4%), 13 (14.4%), and 10 (11.1%) patients were in the high-risk, intermediate-risk, and low-risk categories, respectively.
Table 1.
Baseline Characteristics of the Study Population.
| Characteristic | Study Population (n = 90) |
|---|---|
| Age, mean ± SD, years | 62.8 ± 11.9 |
| Men, N (%) | 55 (61.1) |
| Body weight, mean ± SD, kg | 69.6 ± 14.4 |
| Type of atrial fibrillation | |
| Nonparoxysmal, N (%) | 68 (75.6) |
| Medical history | |
| Hypertension, N (%) | 52 (57.8) |
| Diabetes mellitus, N (%) | 23 (25.6) |
| Stroke/transient ischemic attack, N (%) | 30 (33.3) |
| Heart failure, N (%) | 39 (43.3) |
| Vascular disease, N (%) | 6 (6.7) |
| Hypertrophic cardiomyopathy, N (%) | 12 (13.3) |
| Dilated cardiomyopathy, N (%) | 8 (8.9) |
| Appropriate anticoagulation, N (%) | 36 (40.0) |
| CHA2DS2-VASc score, mean ± SD | 3.0 ± 1.9 |
| ≥2 in men or ≥3 in women, N (%) | 64 (71.1) |
| 1 in men or 2 in women, N (%) | 14 (15.6) |
| 0 in men or 1 in women, N (%) | 12 (13.3) |
| HAS-BLED score, mean ± SD | 2.3 ± 1.2 |
| Laboratory tests | |
| Uric acid, mean ± SD, μmol/L | 400.8 ± 116.4 |
| Creatinine clearance rate, mean ± SD, mL/min | 81.8 ± 32.2 |
| Elevated BNP/NT-proBNP, N (%) | 58 (87.9) |
| Echocardiographic data | |
| Left ventricular ejection fraction, mean ± SD, % | 59.2 ± 10.9 |
| ≥50, N (%) | 75 (84.3) |
| 41-49, N (%) | 5 (5.6) |
| ≤40, N (%) | 9 (10.1) |
| Left atrium diameter, mean ± SD, mm | 44.9 ± 6.5 |
| Moderate or severe mitral insufficiency, N (%) | 17 (19.1) |
| Moderate or severe tricuspid insufficiency, N (%) | 35 (39.3) |
| Moderate or severe aortic insufficiency, N (%) | 5 (5.6) |
Abbreviation: BNP, brain natriuretic peptide; NT-proBNP, N-terminal pro B-type natriuretic peptide; SD, standard deviation;
Location of Atrial Thrombus
Of all 90 patients, 82 (91.1%) had thrombi detected in the LAA only, 5 in the left atrial cavity only, and 3 in the right atrial cavity only (Table 2).
Table 2.
The Location of Atrial Thrombus.
| Location | N (%) |
|---|---|
| Left atrial cavity | 5 (5.6) |
| Left atrial appendage | 82 (91.1) |
| Right atrial cavity | 3 (3.3) |
| Right atrial appendage | 0 |
Anticoagulation Before and After the Detection of Atrial Thrombus
At the time of baseline TEE and cardiac CTA, 49 of 67 (73.1%) patients at a high risk for stroke were anticoagulated. Among them, 29 patients were administered with warfarin, with an on-admission PT-INR of ≥2.0 in 19 patients. Appropriate dosages and inappropriately reduced dosages of non-vitamin K antagonist oral anticoagulants (NOACs) were used in 7 and 13 patients, respectively. Among the 13 patients at an intermediate risk for stroke, 12 (92.3%) were anticoagulated; warfarin was used in 10 patients, all with an on-admission PT-INR of <2.0. Inappropriately reduced dosages of NOACs were used in 2 patients. Among the 10 patients at a low risk for stroke, 9 (90.0%) were anticoagulated before TEE or cardiac CTA; warfarin was used in 5 patients, with an on-admission PT-INR of ≥2.0 in 2 patients. Inappropriately reduced dosages of NOACs were used in 4 patients (Figure 3). The frequency of OAC use increased from 77.8% to 100% after the detection of atrial thrombus. Figure 4 shows the adjustment of OACs before and after the detection of atrial thrombus.
Figure 3.
The use of oral anticoagulants before the initial TEE or cardiac CTA examination. Abbreviations: CTA, computed tomography angiography; INRF, international normalized ratio; NOAC, non-vitamin K antagonist oral anticoagulants; TEE, transesophageal echocardiography.
Figure 4.
The adjustment of oral anticoagulants before and after the detection of atrial thrombus.
Thrombus Resolution
In total, 50 (55.6%) patients underwent follow-up TEE or cardiac CTA at a median follow-up period of 4 months (IQR: 2-7 months). The patients who did not undergo follow-up imaging were older (65.8 ± 11.4 vs 60.5 ± 11.9 years, P = .036), less likely to have hypertrophic cardiomyopathy (2.5% vs 22.0%, P = .010), and more likely to have moderate-to-severe tricuspid insufficiency (53.8% vs 28.0%, P = .017) than those who did. The other baseline characteristics did not significantly differ between these 2 groups (Supplemental Table 1). Among the patients who underwent follow-up imaging, the complete resolution rate was 60.0% (30 of 50, 95% CI: 0.53-0.67). The median resolution duration was 3 months (IQR: 2-5 months). The thrombus resolution rate among the 16 patients who underwent baseline and follow-up TEE or cardiac CTA within 4 to 12 weeks was 68.8% (11 of 16, 95% CI: 0.62-0.75). Table 3 shows that congestive heart failure, a history of ischemic stroke, and an enlarged left atrium were significantly associated with the nonresolution of atrial thrombus in the univariate analysis. The multivariable logistic regression analysis revealed that congestive heart failure (OR: 8.94; 95% CI: 1.67-47.80; P = .010) and a history of ischemic stroke (OR: 8.28; 95% CI: 1.48-46.42; P = .016) were the independent risk factors of the nonresolution of atrial thrombus. The multi-index ROC curves of heart failure and ischemic stroke and the contemporary stroke risk stratification schemes9–11 for the nonresolution of atrial thrombus are shown in Figure 5. The AUC value for the predictive probability of congestive heart failure and a history of ischemic stroke was 0.796 (95% CI: 0.673-0.919; sensitivity: 0.950; specificity: 0.533; P < .001), which was higher than that for the predictive probability of the contemporary stroke risk stratification schemes tested.
Table 3.
Univariate and Multivariable Regression Analyses for the Nonresolution of Atrial Thrombus.
| Univariate Regression | Multivariable Regression | |||||
|---|---|---|---|---|---|---|
| Variable | Odds Ratio (95% CI) | P Value | Odds Ratio (95% CI) | P Value | ||
| Age, per 10 years | 1.30 (0.79-2.12) | .304 | ||||
| Men | 0.93 (0.28-3.06) | .903 | ||||
| Body weight, per 1 kg | 0.95 (0.91-1.00) | .038 | ||||
| Nonparoxysmal atrial fibrillation | 3.28 (0.78-13.78) | .105 | ||||
| Hypertension | 0.77 (0.25-2.38) | .643 | ||||
| Diabetes mellitus | 1.77 (0.51-6.17) | .371 | ||||
| Stroke/transient ischemic attack | 4.89 (1.39-17.16) | .013 | 8.28 (1.48-46.42) | .016 | ||
| Heart failure | 5.44 (1.58-18.71) | .007 | 8.94 (1.67-47.80) | .010 | ||
| Hypertrophic cardiomyopathy | 1.33 (0.35-5.15) | .676 | ||||
| NOACs vs warfarin | 0.67 (0.21-2.10) | .488 | ||||
| Appropriate anticoagulation | 1.24 (0.38-4.00) | .721 | ||||
| Uric acid, per 1μmol/L | 1.00 (1.00-1.01) | .167 | ||||
| Creatinine clearance rate, per 1 mL/min | 0.98 (0.96-1.00) | .090 | ||||
| CHA2DS2-VASc score ≥2 in men or ≥3 in women |
3.06 (0.82-11.36) | .095 | ||||
| Left ventricular ejection fraction | 1.00 (0.95-1.06) | .870 | ||||
| Left atrium diameter, per 1 mm | 1.13 (1.02-1.25) | .024 | 1.06 (0.94-1.20) | .336 | ||
Abbreviations: CI, confidence interval; NOAC, non-vitamin K antagonist oral anticoagulants.
Figure 5.
The multi-index ROC curve of the contemporary risk stratification schemes for the nonresolution of atrial thrombus. Abbreviation: ROC, receiver operating characteristic curve.
Clinical Outcome Measures
Four patients were lost to follow up. During a median follow-up period of 64 months (IQR: 40-83 months), ischemic stroke occurred in 9 of the 86 remaining patients. One patient experienced a major gastrointestinal bleeding event during warfarin treatment. All-cause death was recorded in 10 patients (Supplemental Table 2).
Discussion
In the present retrospective study, the prevalence of atrial thrombus in patients with NVAF was 1.4%. Patients with atrial thrombi often presented with nonparoxysmal AF, heart failure, and a history of ischemic stroke and were likely to have a high CHA2DS2-VASc score and an enlarged left atrium. Extra-LAA thrombus was rare. Further, the complete thrombus resolution rate was 60.0% in patients who underwent follow-up TEE or cardiac CTA at a median follow-up period of 4 months. Congestive heart failure and a history of ischemic stroke were the independent risk factors of the nonresolution of atrial thrombus.
Owing to between-study heterogeneity, the prevalence of atrial thrombus varies considerably in the literature. In a meta-analysis, the prevalence of left atrial thrombus in patients with AF without anticoagulation was 7.4%. 2 With the widespread peri-procedural use of OACs, the prevalence of atrial thrombus declined dramatically. Lurie et al 12 reported that the prevalence of left atrial thrombus in patients with AF/AFL who received anticoagulation therapy was approximately 3%, which is higher than that reported in our study. We enrolled a younger population with fewer comorbidities and lower CHA2DS2-VASc scores, which could partly explain why the prevalence of left atrial thrombus in our study is lower. Consistent with previous reports,13–17 most patients with atrial thrombus in our study had nonparoxysmal AF, an enlarged left atrium, and a high CHA2DS2-VASc score. With the presence of these factors, the risk of atrial thrombus was more likely to be increased. The LAA is believed to be the primary site of atrial thrombus in patients with NVAF.18,19 It is essential for relieving the internal pressure in the left atrium. When the contractility of the LAA is reduced, atrial thrombus is more likely to occur. 20 In our study, atrial thrombus was located in the LAA in 91.1% of the patients, consistent with previous findings.21–23
A high CHA2DS2-VASc score is independently associated with the presence of atrial thrombus. 24 Indeed, approximately 71% of patients with atrial thrombus had a high CHA2DS2-VASc score (≥2 in men or ≥3 in women) in our study. However, notably, over one-fourth of our patients with atrial thrombus were at a low or an intermediate risk for stroke based on their CHA2DS2-VASc score, which was only sporadically reported. This finding indicated that the CHA2DS2-VASc score could not sufficiently predict the presence of atrial thrombus in patients with AF. Van Chien et al 25 demonstrated that the CHA2DS2-VASc score supplemented with the LA volume index and LA longitudinal strain parameters improved the estimation of the atrial thrombus risk. Tang et al 26 showed that hyperuricemia was significantly associated with a greater risk of left atrial thrombus in patients with AF, even when patients had a low CHA2DS2-VASc score (0 or 1). Milhem et al 27 proposed a new scoring system named atrial thrombus exclusion (ATE), which combined the components of the CHA2DS2 score (hypertension, history of stroke, and heart failure) with the D-dimer level (>270 ng/mL). In their study, an ATE score of 0 yielded a specificity of 37% and a sensitivity of 100% for ruling out atrial thrombus before catheter ablation of AF. Therefore, in cases of an inconclusive benefit–risk balance of anticoagulation therapy among patients with AF at a low or an intermediate risk for stroke based on their CHA2DS2-VASc score, the application of additional scales to estimate the risk of atrial thrombus might be especially useful. A substantial proportion of patients received suboptimal or ineffective OAC therapy in our study, which reflected the gaps between guideline recommendation and local practice. From a clinical perspective, the use of high-quality warfarin or NOACs with appropriate doses should be emphasized.
OAC treatment for at least 3 weeks before catheter ablation or cardioversion is recommended for patients with AF and stroke risk factors to minimize the risk of peri-procedural thromboembolism. 4 Some studies suggested that routine pre-ablation TEE could be avoided in patients with AF/AFL if they were on uninterrupted NOAC treatment.28,29 However, our study showed that among patients with high stroke risk with atrial thrombus, 10.4% were compliant with NOAC treatment with an appropriate dose. In addition, 25.5% of patients with atrial thrombus were at a low or an intermediate risk for stroke. Seven patients at a low risk for stroke presented with atrial thrombus at a sinus rhythm. Therefore, we speculate that peri-procedural TEE remains necessary before ablation of AF/AFL. Clinicians are generally more likely to perform imaging to exclude the presence of atrial thrombus extending beyond current AF guidelines according to the European Heart Rhythm Association survey. 30 As an invasive procedure, TEE carries some intrinsic risks, including oropharyngeal, laryngeal, esophageal, and gastric trauma. 31 Some patients may have difficulties in swallowing the probe under conscious sedation. In patients with esophageal diseases, TEE is contraindicated. In these circumstances, cardiac CTA could be considered an alternative imaging modality. Cardiac CTA has shown high sensitivity and specificity for diagnosing atrial thrombus compared with TEE. 32
Approximately 63.7% of atrial thrombus could be resolved completely with a minimum of 3 weeks of optimal anticoagulation. 33 With the prolongation of the anticoagulation duration, the resolution rate of atrial thrombus increased significantly. 34 However, in some cases, atrial thrombus might be difficult to eliminate even with intensified OAC therapy or changes in anticoagulation strategy. Lee et al 35 reported that patients with nonresolution of atrial thrombus had a higher CHA2DS2-VASc score. Al Rawahi et al 36 found that heart failure and prior stroke could predict the nonresolution of atrial thrombus. Chen et al 37 and Shah et al 38 also noted that a history of ischemic stroke and an enlarged left atrium were associated with the formation of atrial thrombus. In addition, abnormalities in various echocardiographic parameters, such as low LAA emptying velocity (<40 cm/s), high left ventricle (LV) filling pressure, or coexistence of mitral valve disease, might also be predictive of the persistence of atrial thrombus. 39 In the present study, congestive heart failure and a history of ischemic stroke were the independent risk factors of the nonresolution of atrial thrombus. The predictive value of these 2 risk factors was greater than that of the contemporary stroke risk stratification schemes tested. Therefore, anticoagulation should be intensified especially in patients with congestive heart failure and/or a history of ischemic stroke to ensure the resolution of atrial thrombus and prevent thromboembolic events.
Our study had some limitations. First, the retrospective nature of this study might influence the outcome. Second, the study was conducted at a single center. The patients enrolled may not fully represent all patients with NVAF in every clinical setting. Moreover, the limited sample size in our study impeded the identification of more independent risk factors of the nonresolution of atrial thrombus. Third, the pre-admission PT-INR of the patients treated with warfarin could not be retrieved; thus, the time for the therapeutic range was not calculated and the quality of anticoagulation after the left atrial thrombus was detected could not be evaluated. Fourth, 44% of patients never received follow-up imaging by either cardiac CTA or TEE, which may induce nonrespondent bias. However, their baseline characteristics did not have much difference from those who received follow-up imaging. Further prospective studies with larger sample sizes are warranted.
In summary, the LAA is the dominant location of NVAF-related thrombus. The presence of atrial thrombus in patients with NVAF who received anticoagulation therapy is non-negligible. An appropriate dose of NOACs or warfarin is strongly recommended in patients with NVAF. Even in patients who received anticoagulation therapy, TEE or CTA might still be needed to exclude the presence of atrial thrombus before catheter ablation or cardioversion. Congestive heart failure and a history of ischemic stroke are risk factors of the nonresolution of atrial thrombus.
Supplemental Material
Supplemental material, sj-docx-1-cat-10.1177_10760296231168097 for Characteristics, Thrombus Resolution, and Long-Term Outcomes in Patients with Nonvalvular Atrial Fibrillation and Atrial Thrombus by Shu Yang, Yanjuan Zhang, Ning Chen, Jiaojiao Shi, Weizhu Ju, Hongwu Chen, Gang Yang, Zidun Wang, Hailei Liu, Xiaohong Jiang, Chang Cui, Minglong Chen and Mingfang Li in Clinical and Applied Thrombosis/Hemostasis
Footnotes
Authors’ Contributions: The review concept and design were developed jointly by all authors. All authors contributed to the design and data interpretation, and all provided input in each draft of the manuscript.
Data Availability: The de-identified participant data will be shared upon reasonable request.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval: Ethical approval to report this case series was obtained from the Ethics Committee of the First Affiliated Hospital of Nanjing Medical University (No. 2014-SR-113). Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Funding: The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant number 82270329).
ORCID iD: Mingfang Li https://orcid.org/0000-0001-8755-6965
Supplemental Material: Supplemental material for this article is available online.
References
- 1.Chugh SS, Havmoeller R, Narayanan K, et al. Worldwide epidemiology of atrial fibrillation: a Global Burden of Disease 2010 Study. Circulation. 2014;129(8):837‐847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Di Minno MN, Ambrosino P, Dello Russo A, Casella M, Tremoli E, Tondo C. Prevalence of left atrial thrombus in patients with non-valvular atrial fibrillation. A systematic review and meta-analysis of the literature. Thromb Haemost. 2016;115(3):663‐677. [DOI] [PubMed] [Google Scholar]
- 3.Andrade JG, Aguilar M, Atzema C, et al. The 2020 Canadian Cardiovascular Society/Canadian Heart Rhythm Society Comprehensive Guidelines for the management of atrial fibrillation. Can J Cardiol. 2020;36(12):1847‐1948. [DOI] [PubMed] [Google Scholar]
- 4.Hindricks G, Potpara T, Dagres N, et al. 2020 ESC guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS): the task force for the diagnosis and management of atrial fibrillation of the European Society of Cardiology (ESC) developed with the special contribution of the European Heart Rhythm Association (EHRA) of the ESC. Eur Heart J. 2021;42(5):373‐498. [DOI] [PubMed] [Google Scholar]
- 5.January CT, Wann LS, Calkins H, et al. 2019 AHA/ACC/HRS focused update of the 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: a report of the American College of Cardiology/American Heart Association task force on clinical practice guidelines and the Heart Rhythm Society in collaboration with the Society of Thoracic Surgeons. Circulation. 2019;140(2):e125‐e151. [DOI] [PubMed] [Google Scholar]
- 6.Wolfes J, Ellermann C, Frommeyer G, Eckardt L. Evidence-based treatment of atrial fibrillation around the globe: comparison of the latest ESC, AHA/ACC/HRS, and CCS guidelines on the management of atrial fibrillation. Rev Cardiovasc Med. 2022;23(2):56. [DOI] [PubMed] [Google Scholar]
- 7.Du X, Wang Y. The diagnostic efficacy of cardiac CTA combined with D-dimer assay for the detection of left atrial thrombus in patients with atrial fibrillation. Am J Emerg Med. 2019;37(10):1922‐1926. [DOI] [PubMed] [Google Scholar]
- 8.Kottmaier M, Jilek C, Berglar S, et al. Exclusion of left atrial thrombus by dual-source cardiac computed tomography prior to catheter ablation for atrial fibrillation. Clin Res Cardiol. 2019;108(2):150‐156. [DOI] [PubMed] [Google Scholar]
- 9.Gage BF, Waterman AD, Shannon W, Boechler M, Rich MW, Radford MJ. Validation of clinical classification schemes for predicting stroke: results from the National Registry of Atrial Fibrillation. JAMA. 2001;285(22):2864‐2870. [DOI] [PubMed] [Google Scholar]
- 10.Rietbrock S, Heeley E, Plumb J, van Staa T. Chronic atrial fibrillation: incidence, prevalence, and prediction of stroke using the congestive heart failure, hypertension, age >75, diabetes mellitus, and prior stroke or transient ischemic attack (CHADS2) risk stratification scheme. Am Heart J. 2008;156(1):57‐64. [DOI] [PubMed] [Google Scholar]
- 11.Singer DE, Albers GW, Dalen JE, et al. Antithrombotic therapy in atrial fibrillation: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th edition). Chest. 2008;133(6 Suppl):546S‐592S. [DOI] [PubMed] [Google Scholar]
- 12.Lurie A, Wang J, Hinnegan KJ, et al. Prevalence of left atrial thrombus in anticoagulated patients with atrial fibrillation. J Am Coll Cardiol. 2021;77(23):2875‐2886. [DOI] [PubMed] [Google Scholar]
- 13.Nishikii-Tachibana M, Murakoshi N, Seo Y, et al. Prevalence and clinical determinants of left atrial appendage thrombus in patients with atrial fibrillation before pulmonary vein isolation. Am J Cardiol. 2015;116(9):1368‐1373. [DOI] [PubMed] [Google Scholar]
- 14.Yanagisawa S, Inden Y, Riku S, et al. Incidence of left atrial thrombus development and imaging approach in patients scheduled for repeat catheter ablation for atrial fibrillation. Am J Cardiol. 2021;155:52‐63. [DOI] [PubMed] [Google Scholar]
- 15.Zhan Y, Joza J, Al Rawahi M, et al. Assessment and management of the left atrial appendage thrombus in patients with nonvalvular atrial fibrillation. Can J Cardiol. 2018;34(3):252‐261. [DOI] [PubMed] [Google Scholar]
- 16.Harada M, Koshikawa M, Motoike Y, et al. Left atrial appendage thrombus prior to atrial fibrillation ablation in the era of direct oral anticoagulants. Circ J. 2018;82(11):2715‐2721. [DOI] [PubMed] [Google Scholar]
- 17.Kapłon-Cieślicka A, Budnik M, Gawałko M, et al. Atrial fibrillation type and renal dysfunction as important predictors of left atrial thrombus. Heart. 2019;105(17):1310‐1315. [DOI] [PubMed] [Google Scholar]
- 18.Zhang H, Yu M, Xia Y, Li X, Liu J, Fang P. The differences of atrial thrombus locations and variable response to anticoagulation in nonvalvular atrial fibrillation with ventricular cardiomyopathy. J Arrhythm. 2020;36(6):1016‐1022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Cresti A, García-Fernández MA, Sievert H, et al. Prevalence of extra-appendage thrombosis in non-valvular atrial fibrillation and atrial flutter in patients undergoing cardioversion: a large transoesophageal echo study. EuroIntervention. 2019;15(3):e225‐e230. [DOI] [PubMed] [Google Scholar]
- 20.Beigel R, Wunderlich NC, Ho SY, Arsanjani R, Siegel RJ. The left atrial appendage: anatomy, function, and noninvasive evaluation. JACC Cardiovasc Imaging. 2014;7(12):1251‐1265. [DOI] [PubMed] [Google Scholar]
- 21.Blackshear JL, Odell JA. Appendage obliteration to reduce stroke in cardiac surgical patients with atrial fibrillation. Ann Thorac Surg. 1996;61(2):755‐759. [DOI] [PubMed] [Google Scholar]
- 22.Di Biase L, Natale A, Romero J. Thrombogenic and arrhythmogenic roles of the left atrial appendage in atrial fibrillation. Circulation. 2018;138(18):2036‐2050. [DOI] [PubMed] [Google Scholar]
- 23.Al-Saady NM, Obel OA, Camm AJ. Left atrial appendage: structure, function, and role in thromboembolism. Heart. 1999;82(5):547‐554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Uz O, Atalay M, Doğan M, et al. The CHA2DS2-VASc score as a predictor of left atrial thrombus in patients with non-valvular atrial fibrillation. Med Princ Pract. 2014;23(3):234‐238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Van Chien D, Thai Giang P, Son PT, Truong LV, Nguyen Son P. Novel models for the prediction of left atrial appendage thrombus in patients with chronic nonvalvular atrial fibrillation. Cardiol Res Pract. 2019;2019:1496535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Tang RB, Dong JZ, Yan XL, et al. Serum uric acid and risk of left atrial thrombus in patients with nonvalvular atrial fibrillation. Can J Cardiol. 2014;30(11):1415‐1421. [DOI] [PubMed] [Google Scholar]
- 27.Milhem A, Ingrand P, Tréguer F, et al. Exclusion of intra-atrial thrombus diagnosis using D-dimer assay before catheter ablation of atrial fibrillation. JACC Clin Electrophysiol. 2019;5(2):223‐230. [DOI] [PubMed] [Google Scholar]
- 28.Diab M, Wazni OM, Saliba WI, et al. Ablation of atrial fibrillation without left atrial appendage imaging in patients treated with direct oral anticoagulants. Circ Arrhythm Electrophysiol. 2020;13(9):e008301. [DOI] [PubMed] [Google Scholar]
- 29.Patel K, Natale A, Yang R, et al. Is transesophageal echocardiography necessary in patients undergoing ablation of atrial fibrillation on an uninterrupted direct oral anticoagulant regimen? Results from a prospective multicenter registry. Heart Rhythm. 2020;17(12):2093‐2099. [DOI] [PubMed] [Google Scholar]
- 30.Farkowski MM, Jubele K, Marín F, et al. Diagnosis and management of left atrial appendage thrombus in patients with atrial fibrillation undergoing cardioversion or percutaneous left atrial procedures: results of the European Heart Rhythm Association survey. Europace. 2020;22(1):162‐169. [DOI] [PubMed] [Google Scholar]
- 31.Hilberath JN, Oakes DA, Shernan SK, Bulwer BE, D'Ambra MN, Eltzschig HK. Safety of transesophageal echocardiography. J Am Soc Echocardiogr. 2010;23(11):1115‐1127. [DOI] [PubMed] [Google Scholar]
- 32.Vira T, Pechlivanoglou P, Connelly K, Wijeysundera HC, Roifman I. Cardiac computed tomography and magnetic resonance imaging vs. transoesophageal echocardiography for diagnosing left atrial appendage thrombi. Europace. 2019;21(1):e1‐e10. [DOI] [PubMed] [Google Scholar]
- 33.Wang NC, Sather MD, Hussain A, et al. Oral anticoagulation and left atrial thrombi resolution in nonrheumatic atrial fibrillation or flutter: a systematic review and meta-analysis. Pacing Clin Electrophysiol. 2018;41(7):767‐774. [DOI] [PubMed] [Google Scholar]
- 34.Ke HH, He Y, Lv XW, Zhang EH, Wei Z, Li JY. Efficacy and safety of rivaroxaban on the resolution of left atrial/left atrial appendage thrombus in nonvalvular atrial fibrillation patients. J Thromb Thrombolysis. 2019;48(2):270‐276. [DOI] [PubMed] [Google Scholar]
- 35.Lee WC, Fang CY, Chen YL, et al. Left atrial or left atrial appendage thrombus resolution after adjustment of oral anticoagulant treatment. J Stroke Cerebrovasc Dis. 2019;28(1):90‐96. [DOI] [PubMed] [Google Scholar]
- 36.Al Rawahi M, Samuel M, Galatas C, et al. Incidence and predictors of intracardiac thrombus on pre-electrophysiological procedure transesophageal echocardiography. CJC Open. 2019;1(5):231‐237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Chen L, Zinda A, Rossi N, et al. A new risk model of assessing left atrial appendage thrombus in patients with atrial fibrillation—using multiple clinical and transesophageal echocardiography parameters. Int J Cardiol. 2020;314:60‐63. [DOI] [PubMed] [Google Scholar]
- 38.Shah M, Mobaligh N, Niku A, Shiota T, Siegel RJ, Rader F. Predictors of left atrial appendage thrombus despite NOAC use in nonvalvular atrial fibrillation and flutter. Int J Cardiol. 2020;317:86‐90. [DOI] [PubMed] [Google Scholar]
- 39.Faggiano P, Dinatolo E, Moreo A, et al. Prevalence and rate of resolution of left atrial thrombus in patients with non-valvular atrial fibrillation: a two-center retrospective real-world study. J Clin Med. 2022;11(6):1520. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental material, sj-docx-1-cat-10.1177_10760296231168097 for Characteristics, Thrombus Resolution, and Long-Term Outcomes in Patients with Nonvalvular Atrial Fibrillation and Atrial Thrombus by Shu Yang, Yanjuan Zhang, Ning Chen, Jiaojiao Shi, Weizhu Ju, Hongwu Chen, Gang Yang, Zidun Wang, Hailei Liu, Xiaohong Jiang, Chang Cui, Minglong Chen and Mingfang Li in Clinical and Applied Thrombosis/Hemostasis