Abstract
Policy Points.
Racism is an upstream determinant of health that influences health through many midstream and downstream factors. This Perspective traces multiple plausible causal pathways from racism to preterm birth.
Although the article focuses on the Black‐White disparity in preterm birth, a key population health indicator, it has implications for many other health outcomes.
It is erroneous to assume by default that underlying biological differences explain racial disparities in health. Appropriate science‐based policies are needed to address racial disparities in health; this will require addressing racism.
Keywords: racism and health, preterm birth, racial disparities
Preterm birth (ptb)—birth before 37 completed weeks of pregnancy—is among the most important indicators of a population's health. It is the second most common cause of infant mortality in the US population overall 1 and the leading cause among African Americans. 2 PTB has serious adverse consequences not only in infancy but across the life course; it is a major cause of childhood developmental disability 3 and a strong predictor of chronic disease in adulthood. 4 , 5 A large racial disparity in PTB has persisted for decades, with rates among African American/Black women approximately 1.5 to 1.6 times those among European American/White women. 6
Unfortunately, the causes of both PTB and the racial disparity in PTB—which are not necessarily the same—are not well established. Despite lack of evidence and/or evidence to the contrary, widespread assumptions have been made about the racial disparity in PTB; as with many health outcomes, “race” has often been assumed by default and without evidence to reflect differences in underlying biology or behaviors. 7 , 8 , 9 Evidence has been mounting over the past two decades that calls those assumptions into serious question, however, pointing to racism as a highly plausible upstream—i.e., fundamental—cause of the Black‐White disparity in PTB. 10
This paper briefly reviews the evidence about biologically plausible causes of the racial disparity in PTB, focusing on racism and associated racial differences in experiences and exposures that are rarely measured in health studies. The terms “race” and “racial” are used here to refer to social groupings according to the large geographic area (often a continent) of an individual's ancestry. Geographic ancestry often correlates with observable superficial secondary physical characteristics such as skin color, facial features, or hair texture and with differences in the way that people are treated, but has not been associated with fundamental biological differences. 11 , 12 , 13 , 14 , 15 , 16
What Is Known About the Causes of the Black‐White Disparity in PTB?
While much is unknown or uncertain, research has identified several explanations for the racial disparity in PTB that appear unlikely. Differences in the quality of prenatal care may play a role, 17 , 18 , 19 although considerable research has indicated that the PTB disparity is not explained by differences in receipt of standard prenatal care; 20 , 21 use of tobacco, alcohol, or illicit drugs; 22 , 23 , 24 , 25 , 26 or by current income or education. 27 While socioeconomic measures (such as income and/or education) around the time of pregnancy consistently predict PTB rates among White women, this has not been the case among Black women. Furthermore, the racial disparity in PTB has been widest among women of high socioeconomic status and minimal or absent among economically disadvantaged women. 28 , 29 Infections have been strongly associated with PTB, and Black women have higher rates of prenatal infection; 30 , 31 treatment of the infections, however, has not consistently improved PTB rates. 30 , 32 , 33 , 34 This raises the question whether the infections, rather than being a cause, are a marker for something else, an underlying factor—such as stress‐induced inflammation and/or immunocompromise—that causes both infections and PTB. 33 , 35
Despite lack of supporting evidence, genetic differences have often been assumed to be the principal explanation for the racial disparity in PTB. 7 , 8 , 9 This assumption has been reinforced in part because the disparity has generally persisted after adjustment for a mother's income and education around the time of pregnancy and in part because of failure to distinguish influences on PTB from influences on the racial disparity in PTB. A recent review of evidence by a panel of experts, including experts in genetics, concluded, however, that although genetic differences may contribute to PTB, they likely play a small, if any, role in the racial disparity in PTB. 10 , 13 , 36 , 37 , 38 To account for the racial disparity in PTB, a cause—for example, a genetic variant—must not only influence PTB; it must also be more prevalent and/or have a greater effect size among one racial group. No such variant has been identified by large‐scale genomic studies. 10 Gene‐environment interactions, however, may be important. 39 , 40 , 41 , 42 Potential mediators of epigenetic change include a range of social and environmental factors that are both associated with PTB risk and more prevalent among African American women, such as exposure to environmental toxins (due to environmental injustice), stress (e.g., due to experiences of racism and/or the economic hardship it produces), and diet (e.g., a recent study identified differences in gene expression associated not only with PTB but also with vitamin D insufficiency, which is more common among Black women). 43
A large body of evidence indicates that social and environmental causes play an important role in both PTB 44 and the racial disparity in PTB. 10 Among the most compelling evidence of social rather than underlying biological causes is the observation of good birth outcomes among Black immigrants to the United States from Africa—that is, the absence of a racial disparity compared with their White counterparts. 38 , 45 If genetic differences were the cause of the disparity, one would expect to see worse, not better, birth outcomes among Black women who emigrate to the United States from Africa, whose genetic endowment would not be as “diluted” by admixture with European Americans as that of African Americans. The PTB disparity, furthermore, has often been found to vary with neighborhood social conditions. 28 , 46 , 47 , 48 Many studies have observed a consistent and important role for environmental toxins such as air or ground pollution, 49 , 50 , 51 , 52 which reflect the social policies driving environmental injustice. Some studies have observed no or a minimal racial disparity in PTB among poor women and the largest racial disparity among more socioeconomically advantaged women, suggesting the role of social factors. 28 , 29 Moreover, several studies have found associations between adverse birth outcomes and experiences of racial discrimination (e.g., unfair treatment, insults, threats, vigilance); 53 , 54 , 55 , 56 stress, discussed later in the article, is thought to mediate these associations. Nutrition also may contribute. 57 , 58 Many studies have associated stress and/or lack of social support (which can modify the effects of stress) with PTB. 27 , 59 The associations between stress and PTB, however, have not been consistent, which may reflect differences in the measurement of stress or the timing of measurement, for example, only during pregnancy versus throughout a woman's life before she became pregnant. 60 , 61 A number of researchers have called attention to the need to consider exposures and conditions experienced across a woman's entire life course, and particularly in early childhood, not only during pregnancy. 62 , 63 , 64 , 65 , 66
Racism Is a Biologically Plausible Upstream Cause of the PTB Disparity
Based on the totality of available evidence, many researchers have concluded that racism in many forms is a highly plausible upstream cause of the racial disparity in PTB. 7 , 10 , 27 , 62 , 67 , 68 Racism is more than just individual acts of race‐based discrimination. Camara Jones (2018) has defined racism as follows:
Racism is a system of structuring opportunity and assigning value based on the social interpretation of how one looks (which is what we call “race”), that unfairly disadvantages some individuals and communities, unfairly advantages other individuals and communities, and saps the strength of the whole society through the waste of human resources. 69
Using the analogy of a river flowing downstream to its destination from its upstream source, an upstream cause is one that acts at the beginning of a long causal chain, setting in motion multiple midstream and/or downstream causes, which then more directly trigger the physiological mechanisms that directly produce PTB. This is illustrated by Figure 1, which, based on the literature, is a greatly simplified illustration of some of the many plausible pathways through which racism, as an upstream causal factor, may result in the racial disparity in PTB by activating midstream and downstream causes. For example, gerrymandering and voter suppression are products of racism that lead to African Americans lacking political representation. Lack of political representation puts African Americans at a disadvantage economically and environmentally because it means that their interests are not being championed and addressed, and resources are not being allocated to their communities (midstream factors). This in turn results in a range of harmful exposures downstream that directly trigger physiological mechanisms resulting in PTB.
Figure 1.
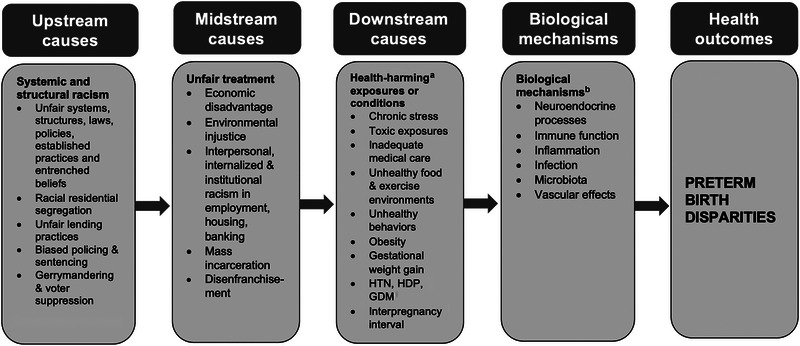
Racism Plausibly May Affect PTB As an Upstream Cause, Acting Through Midstream and Downstream Causes That More Directly Trigger the Physiologic Mechanisms Resulting in Racial Disparities in Preterm Birth
Abbreviations: GDM, gestational diabetes mellitus; HDP, hypertensive disorders of pregnancy; HTN: hypertension.
a Health‐harming (or lack of health‐promoting) exposures/conditions.
b Epigenetic effects are not displayed; they may occur through exposures at each step along the causal pathways.
Also illustrated by Figure 1, racism could contribute to the racial disparity in PTB—and other important health outcomes—through racial residential segregation. Discrimination in housing is no longer legal, but the legacy of Jim Crow laws endures in pervasive racial segregation. US census data for 2013‐2017 reveal that on a national basis, 52.6% of African Americans would have to move to achieve full integration with Whites; 70 in some cities, three out of four African Americans would need to move in order to achieve full integration. 71 Segregation tracks African Americans into neighborhoods that are less healthy in many ways, including exposure to pollution due to environmental injustice, 49 , 72 exposure to unhealthy foods, 73 , 74 and the stress of living in deteriorating areas with concentrated poverty. 75 This affects not only poor but also many middle‐class African Americans. 76 , 77 Segregation cuts African Americans off from services, homeownership, good schools, decent jobs, and economic opportunities. 78 Low levels of wealth 79 or income 80 and inferior schooling 80 , 81 result in poor health across generations.
It is important to understand that all of this can happen even without any individual now consciously intending to discriminate, because the factors involved reflect systemic and structural racism, that is, the racism built into systems and structures, including laws, policies, widely established practices, and entrenched beliefs. These include the policies, practices, and beliefs driving discriminatory treatment of African Americans by banking institutions, which are obstacles to homeownership and the ability to start or expand a business, and therefore to accumulate wealth. 75 Wealth, which is more likely to reflect economic resources across a person's entire life course, may be even more important to health than income, which measures economic resources during a limited period of time. 79
Discriminatory structures associated with racial residential segregation also include underresourced public schools. The schools are underresourced because low wealth means low property taxes, an important source of support for schools; education is key to employment opportunities. Racism also constrains the economic opportunities of African Americans through discrimination in hiring and promotions. 82 , 83
Lack of socioeconomic resources due to any discriminatory cause could affect PTB risk in many ways. Discriminatory causes could include biased lending practices, employment discrimination, inferior schools, and/or biased policing and sentencing practices; the latter stigmatize people for life, blocking their access to employment and housing opportunities. 84 , 85 , 86 Consequences of limited socioeconomic resources could include inability to purchase a nutritious diet, 73 , 74 toxic exposures (because of inability to afford housing in an environmentally safe area), 49 , 72 and the constant stress associated with facing ongoing daily challenges like child care, transportation, and feeding and sheltering one's family, without adequate resources. Most studies, however, measure socioeconomic factors very inadequately, often just one (typically current income or educational attainment) or both measures, which is not enough; the racial disparity in PTB has been observed to be reduced by about half after adjusting for more than two socioeconomic variables. 28
Stress, including stress unrelated to economic hardship, also appears likely to play an important role in the Black‐White disparity in PTB. Neuroscience has revealed that psychosocial stress (“stress”) occurs when life's demands (stressors) strain or exceed a person's adaptive resources, resulting in downstream biopsychosocial responses (the stress response) that can compromise health. 87 , 88 Considerable research links stress to PTB through stress‐induced physiological mechanisms, 89 , 90 , 91 , 92 including inflammation and immune dysregulation. 93 Stress, furthermore, influences behaviors; 94 , 95 many plausible downstream factors have potentially substantial behavioral influences. Although conclusions are not definitive in every case, studies have linked stress, directly or indirectly, to many plausible downstream factors, including diet/nutrition; 96 infection; 92 microbiota; 97 , 98 obesity; 99 prepregnancy hypertension; 100 hypertensive disorders of pregnancy (HDP); 101 prepregnancy diabetes; 102 gestational weight gain; 103 gestational diabetes; 104 and epigenetic effects. 105
Prepregnancy hypertension and HDP, such as preeclampsia, are more common among African American women and have been strongly associated with PTB. 106 , 107 , 108 , 109 The direct contribution of stress to the development of hypertension remains equivocal due to methodological inconsistencies in the research. 100 , 110 , 111 , 112 Inflammation, however, which can result from chronic stress‐related immune dysregulation, 93 has repeatedly been linked to hypertension in both pregnant and nonpregnant women. 113 , 114 , 115 Inflammation is thought to be involved in HDP; 116 , 117 preeclampsia itself has been considered “an excessive maternal inflammatory response to pregnancy.” 118 A recent study in the Netherlands found elevated hair cortisol levels (measured from three months preconception to the end of the second trimester) and anxiety scores (at hospital admission) in women with preeclampsia, a strong predictor of PTB. 119
Although African American women generally report more stress, 120 , 121 , 122 this has not consistently explained PTB disparities. 123 , 124 , 125 This may be due to studies' inadequate measurement of stress and/or an exclusive focus on stress during pregnancy rather than throughout the life course. 60 , 61 The physiologic “wear and tear” caused by chronic stress can compromise women's reproductive health long before they become pregnant. 126 Few studies, however, consider exposure of women across the entire life course, including in childhood, to chronic social stressors such as racism and/or its effects. A systematic review concluded that chronic stress is more likely than acute stress to result in prolonged elevated blood pressure, 112 supporting the hypothesis that experiences across the life course may be more important than experiences during pregnancy alone. Many researchers have concurred that there is likely an important, biologically plausible role for stress across a woman's life course (but not necessarily during pregnancy) in the PTB disparity. 62 , 63 , 64 , 65 , 66 , 127 , 128
Race—or Racism? Unmeasured Social Differences
All too often, and generally unconsciously, when health researchers observe that a variable representing “race” is associated with a health outcome, they infer that the “race” variable reflects biological differences, particularly if they have adjusted for current income and/or educational attainment. The overwhelming scientific consensus today, however, is that “race” is a social, not a biological, construct. 11 , 12 , 13 , 16 “Race” is presented within quotation marks here as a reminder of its socially constructed nature. 11 , 12 , 13 , 14 , 15
All too often researchers who have concluded that “race” represents biological and/or behavioral differences have failed to consider unmeasured social differences such as those discussed earlier in the paper; this may lead to erroneous conclusions about the racial disparity in PTB and other health outcomes. As noted, structural and systemic racism have constrained African Americans’ economic opportunities—resulting in less favorable education, employment, income, wealth, and neighborhood conditions, starting in infancy. Despite this, all too often scientific papers conclude that an observed racial difference in health must reflect underlying biological differences or behavior because the researchers saw a racial difference after they “controlled for socioeconomic status (SES).” 129 It is not possible to control for SES. It is too multifaceted; it is not just a person's current income or education but both, as well as their accumulated wealth, the quality of their education and the prestige associated with it, their neighborhood socioeconomic conditions (e.g., concentrated poverty/deprivation versus concentrated privilege, environmental exposures), and their parents’ wealth, education, and income, which powerfully shaped their experiences and exposures when they were children. Socioeconomic conditions during childhood may have particularly strong effects. 130 Socioeconomic status encompasses all of these factors and more throughout a person's life, and no researcher can capture all of it. Because of racism, furthermore, at the same level of education, African Americans have far less income than Whites. 28 And at the same income, African Americans live in poorer neighborhoods and have a fraction of the accumulated wealth. 131 All of these differences can affect health in general and PTB in particular. These factors are rarely measured, yet studies often conclude a racial difference is genetic if it persists after “controlling for SES.” The “race” variable often captures unmeasured socioeconomic, psychosocial, and environmental factors.
Racism also could plausibly affect PTB not only through limiting a person's economic opportunities but also through the direct psychological effects of experiencing, anticipating, or being aware of unfair treatment of one's racial group. All of these can be stressful, regardless of one's socioeconomic resources. It is not just dramatic incidents. It is the cumulative effects of daily experiences that may be subtle or ambiguous, the effects of having to be constantly vigilant for a slight or insult—intended or not—and the cumulative effects of countless little assaults on one's self‐esteem. Or it may be the stress experienced by a woman learning that yet another unarmed Black man has been killed by the police and constantly wondering whether her husband or son is next. Chronic stress, even at a low level, is biologically plausible as a contributor to PTB through neuroendocrine and immune mechanisms. Neuroscientists have identified physiologic pathways leading from stress to health damage, including effects on inflammation and immune system functioning known to trigger labor. A large population‐based study found that the Black‐White disparity in PTB was no longer significant after adjusting for whether a woman often worried about being treated unfairly based on her race. 53 As noted earlier, several studies have observed a wider racial disparity in PTB among more socioeconomically advantaged women than among their less‐advantaged counterparts; possible explanations include greater chronic stress among higher‐income or higher‐education Black women due to paradoxically greater exposure to chronic racism at work, where they are likely to be in the minority. 53 , 132 , 133 Other hypotheses include the psychosocial price African Americans may pay for upward social mobility, including constantly feeling they must try harder than their White peers to overcome stereotypes. 132 , 133 Very few health studies have measured people's stressful experiences of racism, which are a biologically plausible and potentially very important difference.
Racism may therefore be an important contributor to the Black‐White difference in PTB and other adverse health outcomes, through many pathways, including those noted here and others. Health‐damaging (or lack of health‐promoting) pathways may operate even when no one currently intends to discriminate, because they are the effects of deeply embedded structural or systemic racism. None of these pathways is usually measured adequately, if at all, in health studies, yet this limitation is rarely acknowledged. It should be noted, however, that many of the issues discussed in this paper, while insufficiently considered in most health research, have been more adequately addressed for some time in the social science and humanities literatures. 14 , 16 , 134 , 135 , 136 , 137 , 138
While this paper focuses on preterm birth, the key arguments, particularly about plausible causal pathways from racism to health damage and unmeasured differences—are relevant to a wide range of health outcomes, particularly cardiovascular disease and diabetes, where the roles of inflammation and immune dysfunction are believed to be important, as they are in PTB. The implications of this paper therefore extend far beyond PTB. The practical implications apply to all those who read and/or conduct studies that include variables representing “race.” One must be aware that the “race” variable is always picking up the totality of unmeasured experiences that a person of that “race” may have endured, throughout that individual's life course, which could have influenced health. And even the most knowledgeable, skilled, and determined researcher could have measured at best only a tiny fraction of those experiences. When reading, designing, or analyzing results of a study of any health outcome that includes a variable representing “race,” one should always ask: are we looking at the effects of “race”—or of racism?
Addressing structural and systemic racism and their cascading effects is fundamental to improving population health and reducing health inequities, as discussed in greater detail by Brown and Hohlman,139 and also by Michener and Ford in this special issue.140 In addition, as Ray, Lantz, and Williams argue, 141 a priority agenda for promoting health equity must include reducing racial discrimination (structural as well as interpersonal) both in and far beyond the health care system.
References
- 1. Heron M. Deaths: leading causes for 2017. Natl Vital Stat Rep. 2019;68(6):1‐77. [PubMed] [Google Scholar]
- 2. Ely D, Driscoll A. Infant mortality in the United States, 2017: data from the period linked birth/infant death file. Natl Vital Stat Rep. 2019;68(10):1‐20. https://www.cdc.gov/nchs/data/nvsr/nvsr68/nvsr68_10‐508.pdf. Accessed January 6, 2023. [PubMed] [Google Scholar]
- 3. Marlow N, Wolke D, Bracewell MA, Samara M. Neurologic and developmental disability at six years of age after extremely preterm birth. N Engl J Med. 2005;352(1):9‐19. 10.1056/NEJMoa041367. [DOI] [PubMed] [Google Scholar]
- 4. Crump C, Winkleby MA, Sundquist K, Sundquist J. Risk of diabetes among young adults born preterm in Sweden. Diabetes Care. 2011;34(5):1109‐1113. 10.2337/dc10-2108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Crump C, Howell EA, Stroustrup A, McLaughlin MA, Sundquist J, Sundquist K. Association of preterm birth with risk of ischemic heart disease in adulthood. JAMA Pediatr. 2019;173(8):736‐743. 10.1001/jamapediatrics.2019.1327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Goldenberg RL, Culhane JF, Iams JD, Romero R. Epidemiology and causes of preterm birth. Lancet. 2008;371(9606):75‐84. 10.1016/s0140-6736(08)60074-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Kramer MR, Hogue CR. What causes racial disparities in very preterm birth? A biosocial perspective. Epidemiol Rev. 2009;31(1):84‐98. 10.1093/ajerev/mxp003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Kistka ZA, Palomar L, Lee KA, et al. Racial disparity in the frequency of recurrence of preterm birth. Am J Obstet Gynecol. 2007;196(2):131.e1–131.e6. 10.1016/j.ajog.2006.06.093. [DOI] [PubMed] [Google Scholar]
- 9. Kaufman JS, Geronimus AT, James SA. Faulty interpretation of observed racial disparity in recurrent preterm birth. Am J Obstet Gynecol. 2007;197(3):327‐328. 10.1016/j.ajog.2007.04.040. [DOI] [PubMed] [Google Scholar]
- 10. Braveman P, Dominguez TP, Burke W, et al. Explaining the Black‐White disparity in preterm birth: a consensus statement from a multi‐disciplinary scientific work group convened by the March of Dimes. Front Reprod Health. 2021;3:684207. 10.3389/frph.2021.684207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Cooper RS, Kaufman JS, Ward R. Race and genomics. N Engl J Med. 2003;348(12):1166‐1170. 10.1056/NEJMsb022863. [DOI] [PubMed] [Google Scholar]
- 12. Witherspoon DJ, Wooding S, Rogers AR, et al. Genetic similarities within and between human populations. Genetics. 2007;176(1):351‐359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Yudell M, Roberts D, DeSalle R, Tishkoff S. Taking race out of human genetics. Science. 2016;351(6273):564. 10.1126/science.aac4951. [DOI] [PubMed] [Google Scholar]
- 14. Smedley A, Smedley BD. Race as biology is fiction, racism as a social problem is real: anthropological and historical perspectives on the social construction of race. Am Psychol. 2005;60(1):16‐26. 10.1037/0003-066X.60.1.16. [DOI] [PubMed] [Google Scholar]
- 15. Braveman P, Parker Dominguez T. Abandon “race.” Focus on racism. Front Public Health. 2021;9:689462. 10.3389/fpubh.2021.689462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Duster T. Race and reification in science. Science. 2005;307(5712):1050. 10.1126/science.1110303. [DOI] [PubMed] [Google Scholar]
- 17. Cox RG, Zhang L, Zotti ME, Graham J. Prenatal care utilization in Mississippi: racial disparities and implications for unfavorable birth outcomes. Matern Child Health J. 2011;15(7):931‐942. 10.1007/s10995-009-0542-6. [DOI] [PubMed] [Google Scholar]
- 18. Paul DA, Locke R, Zook K, Leef KH, Stefano JL, Colmorgen G. Racial differences in prenatal care of mothers delivering very low birth weight infants. J Perinatol. 2006;26(2):74‐78. 10.1038/sj.jp.7211428. [DOI] [PubMed] [Google Scholar]
- 19. Kogan MD, Kotelchuck M, Alexander GR, Johnson WE. Racial disparities in reported prenatal care advice from health care providers. Am J Public Health. 1994;84(1):82‐88. 10.2105/ajph.84.1.82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. DeSisto CL, Hirai AH, Collins JW, Rankin KM. Deconstructing a disparity: explaining excess preterm birth among U.S.‐born black women. Ann Epidemiol. 2018;28(4):225‐230. 10.1016/j.annepidem.2018.01.012. [DOI] [PubMed] [Google Scholar]
- 21. Lhila A, Long S. What is driving the black–white difference in low birthweight in the US? Health Econ. 2012;21(3):301‐315. 10.1002/hec.1715. [DOI] [PubMed] [Google Scholar]
- 22. Chen P, Jacobson KC. Developmental trajectories of substance use from early adolescence to young adulthood: gender and racial/ethnic differences. J Adolesc Health. 2012;50(2):154‐163. 10.1016/j.jadohealth.2011.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Truong KD, Reifsnider OS, Mayorga ME, Spitler H. Estimated number of preterm births and low birth weight children born in the United States due to maternal binge drinking. Matern Child Health J. 2013;17(4):677‐688. 10.1007/s10995-012-1048-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Evans‐Polce RJ, Vasilenko SA, Lanza ST. Changes in gender and racial/ethnic disparities in rates of cigarette use, regular heavy episodic drinking, and marijuana use: ages 14 to 32. Addict Behav. 2015;41:218‐222. 10.1016/j.addbeh.2014.10.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Keyes KM, Vo T, Wall MM, et al. Racial/ethnic differences in use of alcohol, tobacco, and marijuana: is there a cross‐over from adolescence to adulthood? Soc Sci Med. 2015;124:132‐141. 10.1016/j.socscimed.2014.11.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Cook B, Creedon T, Wang Y, et al. Examining racial/ethnic differences in patterns of benzodiazepine prescription and misuse. Drug Alcohol Depend. 2018;187:29‐34. 10.1016/j.drugalcdep.2018.02.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Institute of Medicine Committee on Understanding Premature Birth and Assuring Healthy Outcomes . Preterm Birth: Causes, Consequences, and Prevention. Washington, DC: National Academies Press; 2007. [Google Scholar]
- 28. Braveman P, Heck K, Egerter S, et al. The role of socioeconomic factors in Black–White disparities in preterm birth. Am J Public Health. 2015;105(4):694‐702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Guillory VJ, Samuels ME, Probst JC, Sharp G. Prenatal care and infant birth outcomes among Medicaid recipients. J Health Care Poor Underserved. 2003;14(2):272‐289. [DOI] [PubMed] [Google Scholar]
- 30. Foxman B, Wen A, Srinivasan U, et al. Mycoplasma, bacterial vaginosis–associated bacteria BVAB3, race, and risk of preterm birth in a high‐risk cohort. Am J Obstet Gynecol. 2014;210(3):226.e1‐226.e7. 10.1016/j.ajog.2013.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Paige DM, Augustyn M, Adih WK, Witter F, Chang J. Bacterial vaginosis and preterm birth: a comprehensive review of the literature. J Nurse Midwifery. 1998;43(2):83‐89. 10.1016/s0091-2182(97)00161-4. [DOI] [PubMed] [Google Scholar]
- 32. Lamont RF. Advances in the prevention of infection‐related preterm birth. Front Immunol. 2015;6:566. 10.3389/fimmu.2015.00566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Parihar AS, Katoch V, Rajguru SA, Rajpoot N, Singh P, Wakhle S. Periodontal disease: a possible risk‐factor for adverse pregnancy outcome. J Int Oral Health. 2015;7(7):137‐142. [PMC free article] [PubMed] [Google Scholar]
- 34. Sobel R, Sobel JD. Metronidazole for the treatment of vaginal infections. Expert Opin Pharmacother. 2015;16(7):1109‐1115. 10.1517/14656566.2015.1035255. [DOI] [PubMed] [Google Scholar]
- 35. Cobb CM, Kelly PJ, Williams KB, Babbar S, Angolkar M, Derman RJ. The oral microbiome and adverse pregnancy outcomes. Int J Womens Health. 2017;9:551‐559. 10.2147/IJWH.S142730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Burris HH, Lorch SA, Kirpalani H, Pursley DM, Elovitz MA, Clougherty JE. Racial disparities in preterm birth in USA: a biosensor of physical and social environmental exposures. Arch Dis Child. 2019;104(10):931‐935. 10.1136/archdischild-2018-316486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Parets SE, Knight AK, Smith AK. Insights into genetic susceptibility in the etiology of spontaneous preterm birth. Appl Clin Genet. 2015;8:283‐2890. 10.2147/tacg.S58612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. David R, Collins J Jr.. Disparities in infant mortality: what's genetics got to do with it? Am J Public Health. 2007;97(7):1191‐1197. 10.2105/AJPH.2005.068387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Parets SE, Conneely KN, Kilaru V, Menon R, Smith AK. DNA methylation provides insight into intergenerational risk for preterm birth in African Americans. Epigenetics. 2015;10(9):784‐792. 10.1080/15592294.2015.1062964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Ribeiro de Andrade Ramos B, Da Silva MG. The burden of genetic and epigenetic traits in prematurity. Reprod Sci. 2018;25(4):471‐479. 10.1177/1933719117718270. [DOI] [PubMed] [Google Scholar]
- 41. Hong X, Sherwood B, Ladd‐Acosta C, et al. Genome‐wide DNA methylation associations with spontaneous preterm birth in US blacks: findings in maternal and cord blood samples. Epigenetics. 2018;13(2):163‐172. 10.1080/15592294.2017.1287654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Mani S, Ghosh J, Lan Y, et al. Epigenetic changes in preterm birth placenta suggest a role for ADAMTS genes in spontaneous preterm birth. Hum Mol Genet. 2019;28(1):84‐95. 10.1093/hmg/ddy325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Yadama AP, Mirzakhani H, McElrath TF, Litonjua AA, Weiss ST. Transcriptome analysis of early pregnancy vitamin D status and spontaneous preterm birth. PLoS One. 2020;15(1):e0227193. 10.1371/journal.pone.0227193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Bryant AS, Worjoloh A, Caughey AB, Washington AE. Racial/ethnic disparities in obstetric outcomes and care: prevalence and determinants. Am J Obstet Gynecol. 2010;202(4):335‐343. 10.1016/j.ajog.2009.10.864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. David RJ, Collins JW Jr. Differing birth weight among infants of U.S.‐born blacks, African‐born blacks, and US‐born whites. N Engl J Med. 1997;337(17):1209‐1214. 10.1056/nejm199710233371706. [DOI] [PubMed] [Google Scholar]
- 46. Lorch SA, Enlow E. The role of social determinants in explaining racial/ethnic disparities in perinatal outcomes. Pediatr Res. 2015;79(1‐2):141‐147. 10.1038/pr.2015.199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. O'Campo P, Burke JG, Culhane J, et al. Neighborhood deprivation and preterm birth among non‐hispanic black and white women in eight geographic areas in the United States. Am J Epidemiol. 2007;167(2):155‐163. 10.1093/aje/kwm277. [DOI] [PubMed] [Google Scholar]
- 48. Kothari CL, Paul R, Dormitorio B, et al. The interplay of race, socioeconomic status and neighborhood residence upon birth outcomes in a high black infant mortality community. SSM Popul Health. 2016;2:859‐867. 10.1016/j.ssmph.2016.09.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Burris HH, Hacker MR. Birth outcome racial disparities: a result of intersecting social and environmental factors. Semin Perinatol. 2017;41(6):360‐366. 10.1053/j.semperi.2017.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Casey JA, Karasek D, Ogburn EL, et al. Retirements of coal and oil power plants in California: association with reduced preterm birth among populations nearby. Am J Epidemiol. 2018;187(8):1586‐1594. 10.1093/aje/kwy110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Wigle DT, Arbuckle TE, Turner MC, et al. Epidemiologic evidence of relationships between reproductive and child health outcomes and environmental chemical contaminants. J Toxicol Environ Health B Crit Rev. 2008;11(5‐6):373‐517. 10.1080/10937400801921320. [DOI] [PubMed] [Google Scholar]
- 52. Bekkar B, Pacheco S, Basu R, DeNicola N. Association of air pollution and heat exposure with preterm birth, low birth weight, and stillbirth in the US: a systematic review. JAMA Netw Open. 2020;3(6):e208243. 10.1001/jamanetworkopen.2020.8243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Braveman P, Heck K, Egerter S, et al. Worry about racial discrimination: a missing piece of the puzzle of Black‐White disparities in preterm birth? PLoS One. 2017;12(10):e0186151. 10.1371/journal.pone.0186151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Mustillo S, Krieger N, Gunderson EP, Sidney S, McCreath H, Kiefe CI. Self‐reported experiences of racial discrimination and Black‐White differences in preterm and low‐birthweight deliveries: the CARDIA Study. Am J Public Health. 2004;94(12):2125‐2131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Collins JW Jr., David RJ, Handler A, Wall S, Andes S. Very low birthweight in African American infants: the role of maternal exposure to interpersonal racial discrimination. Am J Public Health. 2004;94(12):2132‐2138. 10.2105/ajph.94.12.2132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Collins JW Jr., David RJ, Symons R, Handler A, Wall SN, Dwyer L. Low‐income African‐American mothers' perception of exposure to racial discrimination and infant birth weight. Epidemiology. 2000;11(3):337‐339. [DOI] [PubMed] [Google Scholar]
- 57. Dunlop AL, Taylor RN, Tangpricha V, Fortunato S, Menon R. Maternal micronutrient status and preterm versus term birth for black and white US women. Reprod Sci. 2012;19(9):939‐948. 10.1177/1933719112438442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Rogne T, Tielemans MJ, Chong MF‐F, et al. Associations of maternal vitamin B12 concentration in pregnancy with the risks of preterm birth and low birth weight: a systematic review and meta‐analysis of individual participant data. Am J Epidemiol. 2017;185(3):212‐223. 10.1093/aje/kww212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Hetherington E, Doktorchik C, Premji SS, McDonald SW, Tough SC, Sauve RS. Preterm birth and social support during pregnancy: a systematic review and meta‐analysis. Paediatr Perinat Epidemiol. 2015;29(6):523‐535. 10.1111/ppe.12225. [DOI] [PubMed] [Google Scholar]
- 60. Curry Owens T, Jackson FM. Examining life‐course socioeconomic position, contextualized stress, and depression among well‐educated African‐American pregnant women. Womens Health Issues. 2015;25(4):382‐389. 10.1016/j.whi.2015.05.001. [DOI] [PubMed] [Google Scholar]
- 61. Jackson FM, Hogue CR, Phillips MT. The development of a race and gender‐specific stress measure for African‐American women: Jackson, Hogue, Phillips contextualized stress measure. Ethn Dis. 2005;15(4):594‐600. [PubMed] [Google Scholar]
- 62. Dominguez TP, Dunkel‐Schetter C, Glynn LM, Hobel C, Sandman CA. Racial differences in birth outcomes: the role of general, pregnancy, and racism stress. Health Psychol. 2008;27(2):194‐203. 10.1037/0278-6133.27.2.194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Geronimus AT, Hicken M, Keene D, Bound J. “Weathering” and age patterns of allostatic load scores among blacks and whites in the United States. Am J Public Health. 2006;96(5):826‐833. 10.2105/AJPH.2004.060749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Jackson FM, Phillips MT, Hogue CJ, Curry‐Owens TY. Examining the burdens of gendered racism: implications for pregnancy outcomes among college‐educated African American women. Matern Child Health J. 2001;5(2):95‐107. [DOI] [PubMed] [Google Scholar]
- 65. Geronimus AT. Black/white differences in the relationship of maternal age to birthweight: a population‐based test of the weathering hypothesis. Soc Sci Med. 1996;42(4):589‐597. 10.1016/0277-9536(95)00159-X. [DOI] [PubMed] [Google Scholar]
- 66. Lu MC, Halfon N. Racial and ethnic disparities in birth outcomes: a life‐course perspective. Matern Child Health J. 2003;7(1):13‐30. [DOI] [PubMed] [Google Scholar]
- 67. Dominguez TP. Race, racism, and racial disparities in adverse birth outcomes. Clin Obstet Gynecol. 2008;51(2):360‐370. 10.1097/GRF.0b013e31816f28de. [DOI] [PubMed] [Google Scholar]
- 68. Hogue CJR, Bremner JD. Stress model for research into preterm delivery among black women. Am J Obstet Gynecol. 2005;192(5):S47‐S55. [DOI] [PubMed] [Google Scholar]
- 69. Jones CP. Toward the science and practice of anti‐racism: launching a national campaign against racism. Ethn Dis. 2018;28(Suppl.1):231‐234. 10.18865/ed.28.S1.231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Residential segregation data for U.S. metro areas. Governing website. https://www.governing.com/gov‐data/education‐data/residential‐racial‐segregation‐metro‐areas.html. Published January 10, 2019. Accessed May 7, 2020.
- 71. Frey W. Black‐white segregation edges downward since 2000, census shows. Brookings Institution. https://www.brookings.edu/blog/the‐avenue/2018/12/17/black‐white‐segregation‐edges‐downward‐since‐2000‐census‐shows/. Published December 17, 2018. Accessed May 7, 2020.
- 72. Woodruff TJ, Parker JD, Kyle AD, Schoendorf KC. Disparities in exposure to air pollution during pregnancy. Environ Health Perspect. 2003;111(7):942‐946. 10.1289/ehp.5317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Kwate NOA. Fried chicken and fresh apples: racial segregation as a fundamental cause of fast food density in black neighborhoods. Health Place. 2008;14(1):32‐44. 10.1016/j.healthplace.2007.04.001. [DOI] [PubMed] [Google Scholar]
- 74. Morland K, Filomena S. Disparities in the availability of fruits and vegetables between racially segregated urban neighbourhoods. Public Health Nutr. 2007;10(12):1481‐1489. 10.1017/S1368980007000079. [DOI] [PubMed] [Google Scholar]
- 75. Williams DR, Lawrence JA, Davis BA. Racism and health: evidence and needed research. Annu Rev Public Health. 2019;40(1):105‐125. 10.1146/annurev-publhealth-040218-043750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Grady SC, McLafferty S. Segregation, nativity, and health: reproductive health inequalities for immigrant and native‐born black women in New York City. Urban Geogr. 2007;28(4):377‐397. 10.2747/0272-3638.28.4.377. [DOI] [Google Scholar]
- 77. Anthopolos R, Kaufman JS, Messer LC, Miranda ML. Racial residential segregation and preterm birth: built environment as a mediator. Epidemiology. 2014;25(3):397‐405. [DOI] [PubMed] [Google Scholar]
- 78. Williams DR, Collins C. Racial residential segregation: a fundamental cause of racial disparities in health. Public Health Rep. 2001;116(5):404‐416. 10.1093/phr/116.5.404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Pollack CE, Chideya S, Cubbin C, Williams B, Dekker M, Braveman P. Should health studies measure wealth?: a systematic review. Am J Prev Med. 2007;33(3):250‐264. 10.1016/j.amepre.2007.04.033. [DOI] [PubMed] [Google Scholar]
- 80. Marmot M, Friel S, Bell R, Houweling TAJ, Taylor S. Closing the gap in a generation: health equity through action on the social determinants of health. Lancet. 2008;372(9650):1661‐1669. 10.1016/S0140-6736(08)61690-6. [DOI] [PubMed] [Google Scholar]
- 81. Mirowsky J, Ross CE. Education, Social Status, and Health. Oxfordshire, UK: Routledge; 2017. [Google Scholar]
- 82. Bonilla‐Silva E. Racism Without Racists: Color‐Blind Racism and the Persistence of Racial Inequality in America. 5th ed. Lanham, MD: Rowman & Littlefield; 2018. [Google Scholar]
- 83. Pager D, Shepherd H. The sociology of discrimination: racial discrimination in employment, housing, credit, and consumer markets. Annu Rev Sociol. 2008;34(1):181‐209. 10.1146/annurev.soc.33.040406.131740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. Wildeman C, Wang EA. Mass incarceration, public health, and widening inequality in the USA. Lancet. 2017;389(10077):1464‐1474. 10.1016/s0140-6736(17)30259-3. [DOI] [PubMed] [Google Scholar]
- 85. Massoglia M, Pridemore WA. Incarceration and health. Annu Rev Sociol. 2015;41:291‐310. 10.1146/annurev-soc-073014-112326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Schnittker J, John A. Enduring stigma: the long‐term effects of incarceration on health. J Health Soc Behav. 2007;48(2):115‐130. 10.1177/002214650704800202. [DOI] [PubMed] [Google Scholar]
- 87. Cohen S, Kessler RC, Gordon LU. Measuring Stress: A Guide for Health and Social Scientists. Oxford, UK: Oxford University Press; 1995:xii, 236. [Google Scholar]
- 88. Lazarus R, Folkman S. Stress,Appraisal, and Coping. New York, NY: Springer; 1984. [Google Scholar]
- 89. Dunkel Schetter C. Psychological science on pregnancy: stress processes, biopsychosocial models, and emerging research issues. Annu Rev Psychol. 2011;62:531‐558. 10.1146/annurev.psych.031809.130727. [DOI] [PubMed] [Google Scholar]
- 90. Gennaro S, Hennessy MD. Psychological and physiological stress: impact on preterm birth. J Obstet Gynecol Neonatal Nurs. 2003;32(5):668‐675. [DOI] [PubMed] [Google Scholar]
- 91. Wadhwa PD, Culhane JF, Rauh V, Barve SS. Stress and preterm birth: neuroendocrine, immune/inflammatory, and vascular mechanisms. Matern Child Health J. 2001;5(2):119‐125. 10.1023/A:1011353216619. [DOI] [PubMed] [Google Scholar]
- 92. Wadhwa PD, Culhane JF, Rauh V, et al. Stress, infection and preterm birth: a biobehavioural perspective. Paediatr Perinat Epidemiol. 2001;15(Suppl.2):17‐29. 10.1046/j.1365-3016.2001.00005.x. [DOI] [PubMed] [Google Scholar]
- 93. McEwen BS. Allostasis and the epigenetics of brain and body health over the life course: the brain on stress. JAMA Psychiatry. 2017;74(6):551‐552. 10.1001/jamapsychiatry.2017.0270. [DOI] [PubMed] [Google Scholar]
- 94. Goosby BJ, Cheadle JE, Mitchell C. Stress‐related biosocial mechanisms of discrimination and African American health inequities. Annu Rev Sociol. 2018;44(1):319‐340. 10.1146/annurev-soc-060116-053403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Lucini D, Di Fede G, Parati G, Pagani M. Impact of chronic psychosocial stress on autonomic cardiovascular regulation in otherwise healthy subjects. Hypertension. 2005;46(5):1201‐1206. 10.1161/01.HYP.0000185147.32385.4b. [DOI] [PubMed] [Google Scholar]
- 96. Lindsay KL, Buss C, Wadhwa PD, Entringer S. The interplay between maternal nutrition and stress during pregnancy: issues and considerations. Ann Nutr Metab. 2017;70(3):191‐200. 10.1159/000457136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97. Cryan JF, O'Riordan KJ, Cowan CSM, et al. The microbiota‐gut‐brain axis. Physiol Rev. 2019;99(4):1877‐2013. 10.1152/physrev.00018.2018. [DOI] [PubMed] [Google Scholar]
- 98. Hantsoo L, Jašarević E, Criniti S, et al. Childhood adversity impact on gut microbiota and inflammatory response to stress during pregnancy. Brain Behav Immun. 2019;75:240‐250. 10.1016/j.bbi.2018.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99. Tomiyama AJ. Stress and obesity. Annu Rev Psychol. 2019;70(1):703‐718. 10.1146/annurev-psych-010418-102936. [DOI] [PubMed] [Google Scholar]
- 100. Liu MY, Li N, Li WA, Khan H. Association between psychosocial stress and hypertension: a systematic review and meta‐analysis. Neurol Res. 2017;39(6):573‐580. 10.1080/01616412.2017.1317904. [DOI] [PubMed] [Google Scholar]
- 101. Zhang S, Ding Z, Liu H, et al. Association between mental stress and gestational hypertension/preeclampsia: a meta‐analysis. Obstet Gynecol Surv. 2013;68(12):825‐834. 10.1097/ogx.0000000000000001. [DOI] [PubMed] [Google Scholar]
- 102. Kumari M, Head J, Marmot M. Prospective study of social and other risk factors for incidence of type 2 diabetes in the Whitehall II study. Arch Intern Med. 2004;164(17):1873‐1880. 10.1001/archinte.164.17.1873. [DOI] [PubMed] [Google Scholar]
- 103. Kominiarek MA, Grobman W, Adam E, et al. Stress during pregnancy and gestational weight gain. J Perinatol. 2018;38(5):462‐467. 10.1038/s41372-018-0051-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104. Silveira ML, Whitcomb BW, Pekow P, et al. Perceived psychosocial stress and glucose intolerance among pregnant Hispanic women. Diabetes Metab. 2014;40(6):466‐475. 10.1016/j.diabet.2014.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105. Rogac M, Peterlin B. Epigenetic signature of chronic maternal stress load during pregnancy might be a potential biomarker for spontaneous preterm birth. Balkan J Med Genet. 2018;21(2):27‐33. 10.2478/bjmg-2018-0023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106. Gillum RF. Epidemiology of hypertension in African American women. Am Heart J. 1996;131(2):385‐395. 10.1016/s0002-8703(96)90371-3. [DOI] [PubMed] [Google Scholar]
- 107. Villar J, Carroli G, Wojdyla D, et al. Preeclampsia, gestational hypertension and intrauterine growth restriction, related or independent conditions? Am J Obstet Gynecol. 2006;194(4):921‐931. 10.1016/j.ajog.2005.10.813. [DOI] [PubMed] [Google Scholar]
- 108. Bryant AS, Seely EW, Cohen A, Lieberman E. Patterns of pregnancy‐related hypertension in Black and White women. Hypertens Pregnancy. 2005;24(3):281‐290. 10.1080/10641950500281134. [DOI] [PubMed] [Google Scholar]
- 109. Tanaka M, Jaamaa G, Kaiser M, et al. Racial disparity in hypertensive disorders of pregnancy in New York state: a 10‐year longitudinal population‐based study. Am J Public Health. 2007;97(1):163‐170. 10.2105/ajph.2005.068577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110. Ayada C, Toru Ü, Korkut Y. The relationship of stress and blood pressure effectors. Hippokratia. 2015;19(2):99‐108. [PMC free article] [PubMed] [Google Scholar]
- 111. Nagele E, Jeitler K, Horvath K, et al. Clinical effectiveness of stress‐reduction techniques in patients with hypertension: systematic review and meta‐analysis. J Hypertens. 2014;32(10):1936‐1944. 10.1097/hjh.0000000000000298. [DOI] [PubMed] [Google Scholar]
- 112. Sparrenberger F, Cichelero FT, Ascoli AM, et al. Does psychosocial stress cause hypertension? A systematic review of observational studies. J Hum Hypertens. 2009;23(1):12‐9. 10.1038/jhh.2008.74. [DOI] [PubMed] [Google Scholar]
- 113. Christian LM. Psychoneuroimmunology in pregnancy: immune pathways linking stress with maternal health, adverse birth outcomes, and fetal development. Neurosci Biobehav Rev. 2012;36(1):350‐361. 10.1016/j.neubiorev.2011.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114. De Miguel C, Rudemiller NP, Abais JM, Mattson DL. Inflammation and hypertension: new understandings and potential therapeutic targets. Curr Hypertens Rep. 2014;17(1):507. 10.1007/s11906-014-0507-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115. Agita A, Alsagaff MT. Inflammation, immunity, and hypertension. Acta Med Indones. 2017;49(2):158‐165. [PubMed] [Google Scholar]
- 116. Borzychowski AM, Sargent IL, Redman CWG. Inflammation and pre‐eclampsia. Semin Fetal Neonatal Med. 2006;11(5):309‐316. 10.1016/j.siny.2006.04.001. [DOI] [PubMed] [Google Scholar]
- 117. Ramma W, Ahmed A. Is inflammation the cause of pre‐eclampsia? Biochem Soc Trans. 2011;39(6):1619‐1627. 10.1042/bst20110672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118. Redman CW, Sacks GP, Sargent IL. Preeclampsia: an excessive maternal inflammatory response to pregnancy. Am J Obstet Gynecol. 1999;180(2 Pt 1):499‐506. 10.1016/s0002-9378(99)70239-5. [DOI] [PubMed] [Google Scholar]
- 119. van Esch J, Bolte A, Vandenbussche F, Schippers D, de Weerth C, Beijers R. Hair cortisol concentration and reported anxiety are elevated in preeclampsia. Pregnancy Hypertens. 2018;13:S105. 10.1016/j.preghy.2018.08.311. [DOI] [PubMed] [Google Scholar]
- 120. Borders AEB, Wolfe K, Qadir S, Kim KY, Holl J, Grobman W. Racial/ethnic differences in self‐reported and biologic measures of chronic stress in pregnancy. J Perinatol. 2015;35:580. 10.1038/jp.2015.18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121. Grobman WA, Parker C, Wadhwa PD, et al. Racial/ethnic disparities in measures of self‐reported psychosocial states and traits during pregnancy. Am J Perinatol. 2016;33(14):1426‐1432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122. Mukherjee S, Coxe S, Fennie K, Madhivanan P, Trepka MJ. Stressful life event experiences of pregnant women in the United States: a latent class analysis. Womens Health Issues. 2017;27(1):83‐92. 10.1016/j.whi.2016.09.007. [DOI] [PubMed] [Google Scholar]
- 123. Almeida J, Bécares L, Erbetta K, Bettegowda VR, Ahluwalia IB. Racial/ethnic inequities in low birth weight and preterm birth: the role of multiple forms of stress. Matern Child Health J. 2018;22(8):1154‐1163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124. Grobman WA, Parker CB, Willinger M, et al. Racial disparities in adverse pregnancy outcomes and psychosocial stress. Obstet Gynecol. 2018;131(2):328‐335. 10.1097/aog.0000000000002441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125. Lu MC, Chen B. Racial and ethnic disparities in preterm birth: the role of stressful life events. Am J Obstet Gynecol. 2004;191(3):691‐699. 10.1016/j.ajog.2004.04.018. [DOI] [PubMed] [Google Scholar]
- 126. Rich‐Edwards JW, Grizzard TA. Psychosocial stress and neuroendocrine mechanisms in preterm delivery. Am J Obstet Gynecol. 2005;192(5 Suppl):S30‐S35. 10.1016/j.ajog.2005.01.072. [DOI] [PubMed] [Google Scholar]
- 127. Latendresse G. The interaction between chronic stress and pregnancy: preterm birth from a biobehavioral perspective. J Midwifery Womens Health. 2009;54(1):8‐17. 10.1016/j.jmwh.2008.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128. Olson D, Severson E, Verstraeten B, Ng J, McCreary J, Metz G. Allostatic load and preterm birth. Int J Mol Sci. 2015;16(12):29856‐29874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129. Braveman PA, Cubbin C, Egerter S, et al. Socioeconomic status in health research: one size does not fit all. JAMA. 2005;294(22):2879‐2888. 10.1001/jama.294.22.2879. [DOI] [PubMed] [Google Scholar]
- 130. Shonkoff JP, Garner AS. The lifelong effects of early childhood adversity and toxic stress. Pediatrics. 2012;129(1):e232‐246. 10.1542/peds.2011-2663. [DOI] [PubMed] [Google Scholar]
- 131. Bhutta N, Chang AC, Dettling LJ. Disparities in wealth by race and ethnicity in the 2019 Survey of Consumer Finances. FEDS Notes. September 28, 2020. 10.17016/2380-7172.2797. [DOI] [Google Scholar]
- 132. Myers H, Lewis T, Dominguez T. Stress, coping, and minority health: biopsychosocial perspective on ethnic health disparities. In: Bernal G, Trimble JE, Burlew AK, Leong FTL, eds. Handbook of Racial and Ethnic Minority Psychology. Thousand Oaks, CA: SAGE; 2003:377‐400. [Google Scholar]
- 133. Cole ER, Omari SR. “Race, class and the dilemmas of upward mobility for African Americans.” Journal of Social issues 59, no. 4 (2003): 785–802. [Google Scholar]
- 134. Williams DR, Yu Y, Jackson JS, Anderson NB. Racial differences in physical and mental health: socio‐economic status, stress and discrimination. J Health Psychol. 1997;2(3):335‐351. [DOI] [PubMed] [Google Scholar]
- 135. Krieger N. Epidemiology and the web of causation: has anyone seen the spider? Soc Sci Med. 1994;39(7):887‐903. [DOI] [PubMed] [Google Scholar]
- 136. Confronting racism . Princeton University Humanities Council website. https://humanities.princeton.edu/confronting‐racism/. Accessed January 11, 2023.
- 137. Ellison R. Invisible Man. 1952. Repr., New York: Vintage; 1995:19. [Google Scholar]
- 138. Baldwin J. The Fire Next Time. 1963. Repr., New York: Vintage; 2013. [Google Scholar]
- 139. Brown TH, Hohman PA. The future of structural determinants of health: looking upstream to structural drivers. Milbank Q. 2023;101(S1):36‐60 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140. Michener J, Ford TN. Systemic racism and health: three core principles. Milbank Q. 2023;101(S1):333‐355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141. Ray R, Williams DA, Lantz PM. Upstream policy changes to improve population health and health equity: a priority agenda. Milbank Q. 2023;101(S1):20‐35. [DOI] [PMC free article] [PubMed] [Google Scholar]


