Abstract
Background
Investigating the prognostic value of the Murray law-based quantitative flow ratio (μQFR) on the clinical outcome after treatment of in-stent restenosis (ISR) with a drug-coated balloon (DCB).
Methods
Patients participating in a previous randomized clinical trial for DCB-ISR were post-hoc analyzed. The primary endpoint was vessel-oriented composite endpoint (VOCE), defined as cardiac death, target vessel-related myocardial infarction, and ischemia-driven target vessel revascularization. μQFRs at baseline and after DCB angioplasty was calculated, and its prognostic value as a predictor of VOCE was explored in Cox regression.
Results
A total of 169 lesions in 169 patients were analyzed. At 1-year follow-up, 20 VOCEs occurred in 20 patients. Receiver-operating characteristic curve analysis identified a post-procedural μQFR of ≤ 0.89 as the best cut-off to predict VOCE (area under curve [AUC]: 0.74; 95% confidence interval [CI]: 0.67–0.80; p < 0.001), superior to post-procedural in-stent percent diameter stenosis, which reported an AUC of 0.61 (95% CI: 0.53–0.68; p = 0.18). Post-procedural μQFR was significantly lower in patients with VOCE compared with those without (0.88 [interquartile range: 0.79–0.94] vs. 0.96 [interquartile range: 0.91–0.98], respectively; p < 0.001). After correction for potential confounders, post-procedural μQFR ≤ 0.89 was associated with a 6-fold higher risk of VOCE than lesions with μQFR > 0.89 (hazard ratio: 5.94; 95% CI: 2.33–15.09; p < 0.001).
Conclusions
Post-procedural μQFR may become a promising predictor of clinical outcome after treatment of DES-ISR lesions by DCB angioplasty.
Keywords: quantitative flow ratio, drug-coated balloon, in-stent restenosis
Introduction
Second-generation drug eluting stents (DES) have effectively inhibited neointimal hyperplasia and hence substantially reduced the incidence of in-stent restenosis (ISR) [1, 2]. Nonetheless, recurrent ISR still occurs in late or very late phases after DES implantation in up to 7–10% of cases [3, 4], and interventional treatment of DES-ISR remains challenging. Several studies have demonstrated the efficacy of drug-coated balloons (DCB) for the treatment of ISR [5–9]. DCBs transfer an antiproliferative drug, in most cases paclitaxel, onto the vessel wall during the short time of balloon inflation, resulting in efficient inhibition of smooth muscle cell proliferation and neointimal hyperplasia [10, 11], thus circumventing the need to implant additional metallic layers in the vessel, whilst achieving a comparable net therapeutic performance to DES [9, 12, 13].
Quantitative flow ratio (QFR) is an innovative implement of computational physiology based on three-dimensional (3D) reconstruction of anatomy and hemodynamic simulation, which has shown excellent correlation and agreement with invasive wire-based fractional flow reserve (FFR) [14–16]. Previous studies have demonstrated that QFR can be used to evaluate patients with ISR [17, 18], and post-procedural QFR had the ability to predict future clinical vessel-oriented composite endpoints (VOCE) [19]. On this basis, several studies have recently explored the ability of conventional 3D-based QFR to predict the clinical outcome of ISR treated with DCB [20, 21]. Our current study appraised the prognostic value of the new-generation QFR, based on Murray bifurcation fractal law (μQFR), aided by artificial intelligence [22], to predict the incidence of vessel-oriented composite endpoint after DCB angioplasty, using data from a previous DCB-ISR trial.
Methods
Study design
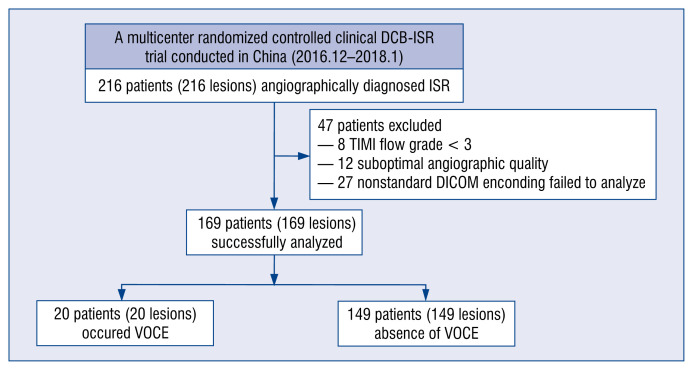
This was a post-hoc analysis of a prospective, multicenter, randomized controlled clinical trial comparing the efficacy of two different kinds of DCBs, Shenqi (Shenqi Medical, Shanghai, China) or Sequent Please (B. Braun Melsungen AG, Melsungen, Germany), for the treatment of first-occurrence DES-ISR between December 2016 and January 2018 [11]. Patients were excluded from the current analysis if the thrombolysis in myocardial infarction (TIMI) grade flow was < 3 at baseline or after DCB angioplasty, the angiography recordings were deemed of insufficient quality for μQFR analysis, or nonstandard angiographic DICOM imaging encoding failed to analyze μQFR (Fig. 1).
Figure 1.
Flow chart of this study; DCB — drug-coated balloon; ISR — in-stent restenosis; μQFR — Murray law-based quantitative flow ratio; TIMI — thrombolysis in myocardial infarction; VOCE — vessel-oriented composite endpoint.
The study complied with the principles of good clinical practice and with the Declaration of Helsinki for investigation in human beings. The study protocol was approved by the Institutional Review Board and Ethics Committee at each participating center, and all patients provided written informed consent before receiving DCB treatment.
Angiographic follow-up and endpoint definition
Patients were routinely scheduled an angiographic follow-up of 9 ± 1 months, even though some angiograms were performed earlier or later if clinically indicated. The endpoint of the study was VOCE at 1-year follow-up, defined as a composite of cardiac death, target vessel-related myocardial infarction, and ischemia-driven target vessel revascularization (TVR). A residual lesion was defined as diameter stenosis (DS) ≥ 50% in vessels ≥ 1.5 mm by visual assessment, not located within the in-segment (a ISR lesion complete treated segment + 5 mm adjacent margins) treatment by DCB angioplasty.
Quantitative coronary angiography (QCA)
Quantitative measurements of coronary angiograms were analyzed offline using QAngio XA 7.3 (Medis Medical Imaging System BV, Leiden, the Netherlands) by two well-trained observers, blind to patients’ information, at the central angiographic core laboratory, according to standard methodology [11]. Appropriate angiographic projections were selected to avoid excessive vessel foreshortening and overlap. Reference and minimal lumen diameters, percentage of DS, and lesion length were measured before and immediately after the procedure, and at follow-up. Restenosis patterns were assessed by Mehran classification [23].
μQFR computation
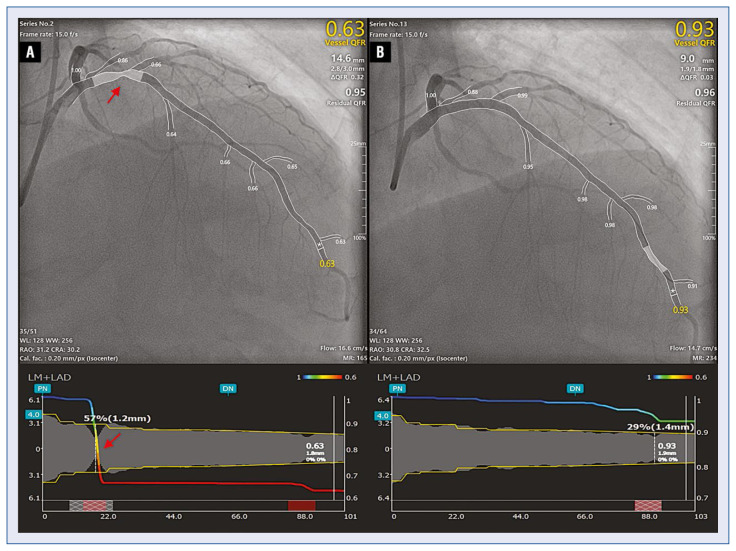
Two experienced and qualified analysts performed μQFR analysis offline, using Angioplus Galley software (Pulse Medical Imaging Technology, Shanghai, China). A single angiographic projection displaying the target vessel from the ostium to the distal segment, with the corresponding side branches and encompassing the whole target lesion, was selected as meeting the requirements of μQFR analysis. The frame with optimal definition of the target lesion was chosen as the key frame for the measurements. Lumen contour and coronary flow velocity were automatically delineated and computed, respectively, aided by artificial intelligence. In cases of inaccurate lumen delineation, minor manual editing was allowed by adding additional points markers along the lumen contour. The reference diameter was calculated along the target vessel according to the Murray fractal law, resulting in reference step-down at bifurcations. Then the physiological indexes of the main vessel and side branch were subsequently derived. In the presence of an eccentric lesion, 3D QCA analysis was performed after selecting a second projection of the target vessel, > 25° apart from the main projection. 3D μQFR was computed from the reconstruction used for 3D-QCA. A paradigmatic example is shown in Figure 2.
Figure 2.
Paradigmatic example of Murray-law based QFR measurement. Murray law-based quantitative flow ratio (μQFR) computation was solely derived from one angiographic projection view; the lumen contour and its side branches were automatically delineated; a step-down reference diameter was calculated based on the Murray bifurcation fractal law. The μQFR values at each position for both the main vessel and its side branches were readily available; A. Pre-procedural angiographic image shows an in-stent restenosis lesion (red arrow) at the proximal left anterior descending artery; the μQFR at the asterisk position was 0.63; B. After drug-coated balloon angioplasty minimal residual stenosis was detected, the the final μQFR was 0.93, as indicated by the asterisk.
Statistical analysis
Continuous variables were expressed as mean ± standard deviation or median (interquartile range) according to the data distribution determined by the Kolmogorov-Smirnov test, and they were compared using Student’s t-test or the Mann–Whitney U test, as appropriate. Categorical variables were described as counts (percentage) and compared using Pearson’s χ2 test or Fisher’s exact test, as appropriate. Receiver operator characteristic (ROC) curve analysis was performed to determine the optimal post-procedural μQFR cut-off value to predict VOCE, as determined by the Youden index. Kaplan–Meier survival analysis was performed, comparing the groups defined by the μQFR cut-off with the log-rank test. Multivariate Cox regression analysis was performed to search for independent predictors of VOCE. Proportional-hazards assumption was tested on the basis of Schoenfeld residuals. Hazard ratios (HR) and 95% confidence intervals (CI) were calculated. All statistical analyses were performed using SPSS version 22.0.0 (IBM Corporation, Armonk, New York, USA) and MedCalc version 14.12 (MedCalc Software, Ostend, Belgium). A two-sided p-value < 0.05 was considered as statistically significant.
Results
The study comprised 216 lesions in 216 patients. Forty-seven patients were excluded from the current study due to TIMI grade flow < 3 at baseline or after DCB angioplasty (8 patients), or due to insufficient quality (12 patients) or nonstandard angiographic DICOM imaging encoding (27 patients) for μQFR analysis, thus resulting in 169 patients being successfully analyzed: 20 patients with VOCE and 149 patients without VOCE (Fig. 1). One-year clinical follow-up was completed in all eligible patients with a median follow-up period of 353 days (340–371 days).
Baseline characteristics
Baseline clinical and procedural characteristics of 169 patients and lesions finally enrolled in the study are presented in Tables 1 and 2. VOCEs occurred in 20 patients at 1-year follow-up. There were no significant differences in age, gender, body mass index, hypertension, hyperlipidemia, current smoking, medical history, clinical presentation, left ventricular ejection fraction, and number of diseased arteries between patients with and without VOCE. Diabetes mellitus and family history of coronary heart disease were significantly more prevalent among patients with VOCE than among event-free patients (Table 1).
Table 1.
Baseline clinical features of patients with available follow-up angiography suitable for Murray law-based quantitative flow ratio (μQFR) analysis after drug-coated balloon (DCB) treatment of in-stent restenosis (ISR).
| Variables | VOCE (n = 20) | Non-VOCE (n = 149) | P |
|---|---|---|---|
| Age [years] | 59.90 ± 11.57 | 62.88 ± 9.58 | 0.20 |
| Female gender | 6 (30.00%) | 35 (23.49%) | 0.58 |
| BMI [kg/m2] | 25.12 ± 3.15 | 25.60 ± 3.31 | 0.54 |
| CAD risk factors: | |||
| Diabetes mellitus | 13 (65.00%) | 56 (37.58%) | 0.03 |
| Hypertension | 15 (75.00%) | 109 (73.15%) | 0.86 |
| Hyperlipidemia | 9 (45.00%) | 49 (32.88%) | 0.28 |
| Current smoker | 6 (30.00%) | 33 (22.15%) | 0.59 |
| Medical history: | |||
| Previous MI | 9 (45.00%) | 64 (42.95%) | 0.86 |
| Previous PCI | 20 (100.00%) | 149 (100.00%) | > 0.999 |
| Family history | 6 (30.00%) | 14 (9.39%) | 0.03 |
| Clinical presentation: | 0.25 | ||
| Silent ischemia | 0 (0.00%) | 15 (10.07%) | |
| Stable angina | 1 (5.00%) | 20 (13.42%) | |
| Unstable angina | 19 (95.00%) | 114 (76.51%) | |
| LVEF [%] | 61.40 ± 7.67 | 60.56 ± 7.23 | 0.63 |
| No. of diseased arteries: | 0.86 | ||
| 1 | 6 (30.00%) | 55 (36.91%) | |
| 2 | 10 (50.00%) | 68 (45.64%) | |
| 3 | 4 (20.00%) | 26 (17.45%) | |
Values are expressed as mean ± standard deviation or number (%); BMI — body mass index; CAD — coronary artery disease; MI — myocardial infarction; LVEF — left ventricular ejection fraction; PCI — percutaneous coronary intervention; VOCE — vessel-oriented composite endpoint
Table 2.
Baseline lesion and procedural characteristics with available follow-up angiography suitable for Murray law-based quantitative flow ratio (μQFR) analysis after drug-coated balloon (DCB) treatment of in-stent restenosis (ISR).
| Variables | VOCE (n = 20) | Non-VOCE (n = 149) | P |
|---|---|---|---|
| Target vessel: | 0.81 | ||
| RCA | 6 (30.00%) | 57 (38.26%) | |
| LAD | 11 (55.00%) | 70 (46.98%) | |
| LCX | 3 (15.00%) | 22 (14.76%) | |
| Ostial lesion | 2 (10.00%) | 4 (2.68%) | 0.15 |
| Bifurcation lesion | 5 (25.00%) | 47 (31.54%) | 0.55 |
| Restenosis pattern: | 0.41 | ||
| Mehran I | 4 (20.00%) | 50 (33.56%) | |
| Mehran II | 13 (65.00%) | 73 (48.99%) | |
| Mehran III | 3 (15.00%) | 26 (17.45%) | |
| Shenqi DCB | 11 (55.00%) | 78 (52.35%) | 0.82 |
| QCA parameters: | |||
| Lesion length [mm] | 15.67 ± 5.61 | 14.99 ± 8.39 | 0.73 |
| RVD [mm] | 2.37 ± 0.40 | 2.60 ± 0.42 | 0.02 |
| MLD [mm] | 0.74 ± 0.35 | 0.95 ± 0.39 | 0.02 |
| DS [%] | 67.95 ± 12.10 | 63.20 ± 12.53 | 0.11 |
| DS after DCB [%] | 17.58 ± 15.17 | 21.11 ± 10.10 | 0.32 |
| Procedural data: | |||
| Predilation | 20 (100.00%) | 149 (100.00%) | > 0.999 |
| Cutting balloon | 5 (25.00%) | 38 (25.50%) | 0.96 |
| DCB diameter [mm] | 2.87 (2.62–3.50) | 3.00 (2.75–3.50) | 0.48 |
| DCB length [mm] | 20.00 (20.00–25.50) | 20.00 (17.00–26.00) | 0.91 |
| DCB pressure [atm] | 9 (8–9) | 9 (8–10) | 0.60 |
| DCB Inflation time [s] | 60 (60–60) | 60 (60–60) | > 0.999 |
| No. of DCB used > 1 | 1 (5.00%) | 3 (2.01%) | 0.40 |
| μQFR measurements: | |||
| Baseline μQFR | 0.50 (0.37–0.75) | 0.77 (0.63–0.85) | 0.001 |
| Post-procedural μQFR | 0.88 (0.79–0.94) | 0.96 (0.91–0.98) | < 0.001 |
| Post-procedural μQFR ≤ 0.80 | 6 (30.00%) | 6 (4.03%) | < 0.001 |
| μQFR improvement | 0.30 (0.18–0.41) | 0.18 (0.10–0.34) | 0.01 |
| Residual lesion after DCB treatment | 8 (40.00%) | 21 (14.09%) | 0.01 |
| Diameter difference DCB — RVD | 0.61 (0.43–0.82) | 0.43 (0.29–0.59) | 0.004 |
| Length difference DCB — lesion | 6.36 (2.62–9.32) | 7.26 (2.65–11.88) | 0.59 |
Values are expressed as mean ± standard deviation median (25th–75th percentile) or number (%); DS — diameter stenosis; LAD — left coronary artery; LCX — left circumflex coronary artery; MLD — minimum lumen diameter; QCA — quantitative coronary angiography; RCA — right coronary artery; RVD — reference vessel diameter; VOCE — vessel-oriented composite endpoint
The distributions of target vessel, ostial lesion, bifurcation lesion, Mehran restenosis pattern, and type of DCB applied did not differ between groups. Baseline QCA parameters showed smaller reference vessel diameter (2.37 ± 0.40 vs. 2.60 ± 0.42, p = 0.02) and minimum lumen diameter (0.74 ± 0.35 vs. 0.95 ± 0.39, p = 0.02) in patients with VOCE than in event-free patients, while the mean lesion length and diameter stenosis at baseline and immediately after the procedure were similar in both groups. Procedural variables were similar between patients with or without VOCE. Patients with VOCE showed lower μQFR values than event-free patients, both at baseline (0.50 [0.37–0.75] vs. 0.77 [0.63–0.85], p = 0.001) and post-procedure (0.88 [0.79–0.94] vs. 0.96 [0.91–0.98], p < 0.001), whilst μQFR improvement, defined as difference values between post-procedure and baseline, was larger in the VOCE group (0.30 [0.18–0.41] vs. 0.18 [0.10–0.34], p = 0.01). The proportion of patients with post-procedural μQFR ≤ 0.80 was larger in the VOCE group than in the event-free group (6 [30.00%] vs. 6 [4.03%], p < 0.001) (Table 2).
Patients with residual lesion after DCB-ISR treatment were more likely to develop VOCE than patients without residual lesion (8 [40.00%] vs. 21 [14.09%], p = 0.01). Moreover, patients with greater differences between DCB diameter and reference vessel diameter (RVD) seemed more prone to develop VOCE (0.61 [0.43–0.82] vs. 0.43 [0.29–0.59], p = 0.004). The length mismatch between DCB and lesion was similar between groups (Table 2).
Definition of potential post-procedural cut-off value
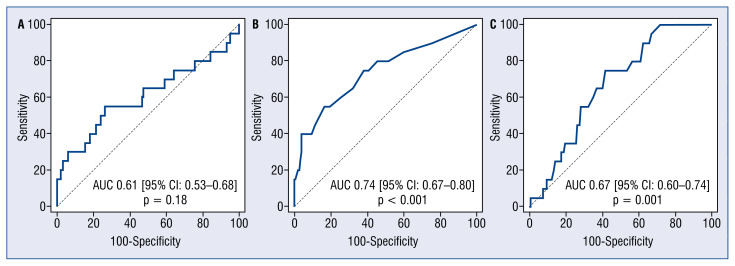
Receiver-operating characteristic curve analysis identified post-procedural μQFR ≤ 0.89 as the optimal cut-off value to predict the occurrence of VOCE, with sensitivity 55% and specificity 74% (AUC: 0.74; 95% CI: 0.67–0.80; p < 0.001). Nevertheless, there was no significant predictive value of post-procedural percent diameter stenosis (%DS) for VOCE (AUC: 0.61; 95% CI: 0.53–0.68; p = 0.18). The ROC curve for μQFR improvement also showed moderate predictive value for VOCE, with the best cut-off value > 0.20, and with sensitivity of 75% and specificity of 58% (AUC: 0.67; 95% CI: 0.60–0.74; p = 0.001) (Fig. 3).
Figure 3.
Receiver operation characteristic curves of post-procedural Murray law-based quantitative flow ratio (μQFR) measurements and diameter stenosis for the prediction of vessel-oriented composite endpoint (VOCE). Receiver operator characteristic curves for the VOCE; A. Post-procedure diameter stenosis (AUC 0.61, 95% CI 0.53–0.68; p = 0.18); B. Post-procedural μQFR (AUC 0.74, 95% CI 0.67–0.80; p < 0.001); C. Post-procedural μQFR improvement (AUC 0.67, 95% CI 0.60–0.74, p = 0.001); AUC — area under curve; CI — confidence interval.
Clinical outcomes
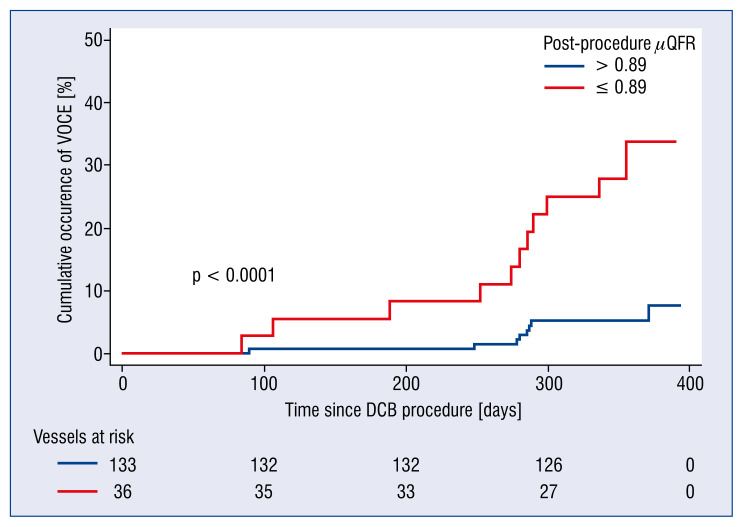
Clinical outcomes stratified by post-procedural μQFR are shown in Table 3. Patients who achieved μQFR > 0.89 after DCB treatment had significantly fewer VOCEs than those with μQFR ≤ 0.89 (6.77% vs. 30.55%, p < 0.001), mainly attributed to a higher incidence rate of TVR (6.77% vs. 27.78%, p = 0.001). Two patients presented with more than one event; both developed target vessel myocardial infarction followed by TVR. One patient with post-procedural μQFR ≤ 0.89 died of cardiac death, while the other, with post-procedural μQFR > 0.89, did not. Kaplan-Meier curves also confirmed that post-procedural μQFR ≤ 0.89 had a remarkably higher incidence rate of VOCE (Fig. 4).
Table 3.
Clinical outcomes stratified by the cut-off value of post-procedural Murray law-based quantitative flow ratio (μQFR) at vessel level.
| Variables | μQFR ≤ 0.89 (n = 36) | μQFR > 0.89 (n = 133) | P |
|---|---|---|---|
| Cardiac death | 1 (2.78%) | 0 (0.00%) | 0.21 |
| Target vessel MI | 2 (5.56%) | 0 (0.00%) | 0.04 |
| Target vessel revascularization | 10 (27.78%) | 9 (6.77%) | 0.001 |
| Target lesion revascularization | 7 (19.44%) | 8 (6.01%) | 0.02 |
| VOCE | 11 (30.55%) | 9 (6.77%) | < 0.001 |
Patients with more than one event are counted only once for the composite endpoint, although each event is listed separately. P values less than 0.05 are in bold; MI — myocardial infarction; VOCE — vessel-oriented composite endpoint
Figure 4.
Kaplan-Meier curves of vessel-oriented composite endpoint (VOCE) occurrence at 1-year follow-up stratified by the best cut-off of post-procedural quantitative flow ratio (μQFR). Blue line means vessels with post-procedure μQFR above 0.89. Yellow line means vessels with values equal to or less than 0.89. The cut-off value of 0.89 was derived from receiver operator characteristic curve analysis for the best prediction of the VOCE; DCB — drug-coated balloon.
After correcting for potential confounders (diabetes mellitus, family history, ostial lesion, lesion length, residual lesion, differences in diameter DCB-RVD), post-procedural μQFR ≤ 0.89 remained associated with a 6-fold increase in the risk of VOCE (adjusted HR: 5.94; 95% CI: 2.33–15.09; p < 0.001; Table 4, Model a1). μQFR improvement > 0.20 was also associated with suboptimal clinical result at 1-year follow-up (HR: 3.75; 95% CI: 1.31–10.68, p = 0.01; Table 4, Model b1). Considered as a continuous variable, post-procedural μQFR was associated with a lower incidence of VOCE in the multivariate analysis (HR: 0.34; 95% CI: 0.23–0.51; p < 0.001), whereas μQFR improvement (each 0.10 increase) was associated with a higher incidence of VOCE (HR: 1.31; 95% CI: 1.04–1.66; p = 0.02; Table 4, Model a2 and Model b2).
Table 4.
Multivariable Cox regression analysis for predicting vessel-oriented composite endpoint (n = 20).
| HR (95% CI) | P | |
|---|---|---|
| Model a1 | ||
| Post-procedural μQFR | 5.94 (2.33–15.09) | < 0.001 |
| Diabetes mellitus | 2.64 (1.03–6.78) | 0.04 |
| Difference of DCB diameter and RVD (per 0.10-mm increase) | 1.34 (1.10–1.62) | 0.003 |
| Model a2 | ||
| Post-procedural μQFR (per 0.10-mm increase) | 0.34 (0.23–0.51) | < 0.001 |
| Diabetes mellitus | 1.61 (0.55–4.66) | 0.16 |
| Difference of DCB diameter and RVD (per 0.10-mm increase) | 1.25 (1.03–1.50) | 0.02 |
| Model b1 | ||
| μQFR improvement | 3.75 (1.31–10.68) | 0.01 |
| Diabetes mellitus | 3.15 (1.23–8.05) | 0.02 |
| Residual lesion after DCB treatment | 3.12 (1.21–8.03) | 0.02 |
| Difference of DCB diameter and RVD (per 0.10-mm increase) | 1.33 (1.11–1.59) | 0.002 |
| Model b2 | ||
| μQFR improvement (per 0.10-mm increase) | 1.31 (1.04–1.66) | 0.02 |
| Diabetes mellitus | 3.10 (1.21–7.92) | 0.02 |
| Residual lesion after DCB treatment | 3.75 (1.47–9.56) | 0.01 |
| Difference of DCB diameter and RVD (per 0.10-mm increase) | 1.32 (1.11–1.57) | 0.001 |
Independent predictors of the previous analysis were used in time-to-event analysis fitting Cox regression models with forward likelihood ratio variable selection method; p values less than 0.05 are in bold; CI — confidence interval; HR — hazard ratio; DCB — drug-coated balloon; RVD — reference vessel diameter; μQFR — Murray law-based quantitative flow ratio
Discussion
To the best of our knowledge, this post-hoc study of a previous DCB-ISR trial investigated the prognostic value of Murray law-based QFR, empowered by artificial intelligence, after DCB-ISR treatment for the first time. A low μQFR after DCB-ISR angioplasty was an independent predictor of clinical adverse events at 1-year follow-up. Post-procedural μQFR showed an optimal predictive value for the occurrence of VOCE, and a moderate predictive value was also observed for μQFR improvement.
Coronary interventions have classically relied on the assessment of anatomic stenosis observed in angiography, even though the stenosis severity scarcely correlates with the physiological significance as a flow-limiting lesion [24, 25] and has modest predictive value for future clinical events [26]. Decision-making based on physiology has consistently proven its superiority over purely angiographic guidance in most clinical scenarios of stable coronary heart disease, thus being endorsed in international guidelines for clinical practice as the highest standard of care [27, 28]. The QFR is a novel tool to derive physiology parameters in the coronary arteries, based on 3D angiographic reconstruction and computerized hemodynamic simulation. This emerging method can efficiently identify functionally significant lesions, whilst overcoming the drawbacks of traditional wire-based invasive physiology [14, 15]. Previous studies have demonstrated that post-procedural QFR is significantly associated with clinical outcomes after percutaneous coronary intervention (PCI) [19, 29]. In the present study, we applied the most advanced and refined version of QFR, called Murray-law based QFR (μQFR), which is characterized by calculating the reference vessel diameter according to fractal geometry. This adds extra accuracy to the estimation, especially for challenging bifurcation lesions, thus achieving excellent agreement with fractional flow reserve [22].
Our study proved an inverse relationship between post-procedural μQFR and adverse clinical events after treatment of DES-ISR with DCB, an association that could not however be verified for post-procedural %DS. This finding is in line with a large corpus of evidence consistently proving the superiority of morphofunctional or computational methods over purely morphologic approaches to assess severity and prognosis [14, 16, 30]. Angiographic residual %DS has been regularly used for the assessment of procedural success in routine clinical practice [31], even though its prognostic value for future clinical events might be disputable [32]. Among morphofunctional parameters, postprocedural μQFR showed the best prognostic value for VOCE at 1-year follow-up, superior to other parameters like μQFR improvement. These findings clearly point out the paramount importance of functional residual lesion after treatment, in this case assessed by means of post-procedural μQFR, and hence indirectly suggest the need to optimize the functional (rather than the angiographic) result after PCI, in line with multiple studies.
Besides functional residual stenosis, i.e., post-procedural μQFR, and the subsequent importance of PCI optimization, an adequate DCB sizing seems also to play a relevant role for future events [33]. Obviously, an insufficient DCB balloon sizing might result in incomplete surface contact, and ultimately to inappropriate drug transfer onto the vessel wall. Nonetheless, the current study showed that the larger the oversizing of DCB diameter in regard to RVD, the higher the risk of developing VOCE at 1-year follow-up. An oversized DCB might create edge dissections of inflicting additional insult to the vessel, which might interfere with the optimal healing process, thus creating the substrate for future events. Intracoronary imaging is currently the best ancillary tool available for both accurate sizing and targeted PCI optimization, so it might be instrumental to optimize these two variables identified by our study as predictors for clinical events after treatment of DES-ISR with a DCB, namely, post-procedural μQFR and DCB diameter mismatch. Evidence about the clinical correlates of a refined interplay between physiology and intracoronary imaging to optimize PCI results is becoming increasingly strong [34].
As in many other publications, diabetes mellitus was also an independent risk factor for adverse events in the current study, probably due to a multifactorial etiology. Diabetes has been associated with more intense plaque progression and has been shown to elicit an exaggerated neointimal hyperplasia reaction [35, 36]. Like other clinical scenarios, the percutaneous treatment of ISR in diabetes patients is particularly challenging and deserves careful attention.
Limitations of the study
There are several limitations in the current study. First, this study is a post-hoc analysis of a previous DCB-ISR trial, which was not initially designed to investigate the prognostic value of μQFR on ISR. Therefore, some cases did not meet the acquisition requirements for μQFR computation. Nonetheless, this attrition of the initial sample size occurred at random and was therefore unlikely to result in selection bias. All cases were analyzed offline, which inevitably affects the precise analysis. Second, the cut-off value of post-procedural μQFR to predict vessel-oriented composite events was not exclusive, varied widely due to multiple factors, including the observed population, incidence of clinical events, lesion and procedural characteristics, etc., and should be validated in a future large randomized controlled trial. Lastly, intravascular imaging was not mandatory in the previous trial; conversely, intravascular morphology information would be beneficial to understand the underlying mechanics of low μQFR values and adverse events.
Conclusions
Post-procedural μQFR after treatment of ISR with DCB was inversely associated with the occurrence of subsequent adverse clinical events and may be considered as a promising predictor.
Acknowledgments
The authors would like to thank the dedicated efforts from the clinical research collaborators who participated in the previous DCB-ISR trial.
Footnotes
The paper was guest edited by Prof. Carlos Cortés
Conflict of interest: Shengxian Tu received a research grant from Pulse Medical Imaging Technology. All other authors have reported that they have no relationships relevant to the contents of this paper to disclose.
References
- 1.Stettler C, Wandel S, Allemann S, et al. Outcomes associated with drug-eluting and bare-metal stents: a collaborative network meta-analysis. Lancet. 2007;370(9591):937–948. doi: 10.1016/S0140-6736(07)61444-5. [DOI] [PubMed] [Google Scholar]
- 2.Stone G, Moses J, Ellis S, et al. Safety and efficacy of sirolimusand paclitaxel-eluting coronary stents. N Engl J Med. 2007;356(10):998–1008. doi: 10.1056/nejmoa067193.. [DOI] [PubMed] [Google Scholar]
- 3.Dangas GD, Claessen BE, Caixeta A, et al. In-stent restenosis in the drug-eluting stent era. J Am Coll Cardiol. 2010;56(23):1897–1907. doi: 10.1016/j.jacc.2010.07.028. [DOI] [PubMed] [Google Scholar]
- 4.Alfonso F, Byrne RA, Rivero F, et al. Current treatment of instent restenosis. J Am Coll Cardiol. 2014;63(24):2659–2673. doi: 10.1016/j.jacc.2014.02.545. [DOI] [PubMed] [Google Scholar]
- 5.Scheller B, Clever YP, Kelsch B, et al. Treatment of coronary instent restenosis with a paclitaxel-coated balloon catheter. N Engl J Med. 2006;355(20):2113–2124. doi: 10.1056/NEJMoa061254. [DOI] [PubMed] [Google Scholar]
- 6.Unverdorben M, Vallbracht C, Cremers B, et al. Paclitaxel-coated balloon catheter versus paclitaxel-coated stent for the treatment of coronary in-stent restenosis. Circulation. 2009;119(23):2986–2994. doi: 10.1161/CIRCULATIONAHA.108.839282. [DOI] [PubMed] [Google Scholar]
- 7.Adriaenssens T, Dens Jo, Ughi G, et al. Optical coherence tomography study of healing characteristics of paclitaxel-eluting balloons vs. everolimus-eluting stents for in-stent restenosis: the SEDUCE (Safety and Efficacy of a Drug elUting balloon in Coronary artery rEstenosis) randomised clinical trial. EuroIntervention. 2014;10(4):439–448. doi: 10.4244/EIJV10I4A77. [DOI] [PubMed] [Google Scholar]
- 8.Pleva L, Kukla P, Kusnierova P, et al. Comparison of the efficacy of paclitaxel-eluting balloon catheters and everolimuseluting stents in the treatment of coronary in-stent restenosis: the treatment of in-stent restenosis study. Circ Cardiovasc Interv. 2016;9(4):e003316. doi: 10.1161/CIRCINTERVENTIONS.115.003316. [DOI] [PubMed] [Google Scholar]
- 9.Giacoppo D, Matsuda Y, Fovino LN, et al. Short dual antiplatelet therapy followed by P2Y12 inhibitor monotherapy vs. prolonged dual antiplatelet therapy after percutaneous coronary intervention with second-generation drug-eluting stents: a systematic review and meta-analysis of randomized clinical trials. Eur Heart J. 2021;42(4):308–319. doi: 10.1093/eurheartj/ehaa739. [DOI] [PubMed] [Google Scholar]
- 10.Chen Y, Gao L, Qin Q, et al. Comparison of 2 different drug-coated balloons in in-stent restenosis: the RESTORE ISR China randomized trial. JACC Cardiovasc Interv. 2018;11(23):2368–2377. doi: 10.1016/j.jcin.2018.09.010. [DOI] [PubMed] [Google Scholar]
- 11.Zhu J, Liu L, Zhu Z, et al. A randomized comparison of a novel iopromide-based paclitaxel-coated balloon Shenqi versus SeQuent Please for the treatment of in-stent restenosis. Coron Artery Dis. 2021;32(6):526–533. doi: 10.1097/MCA.0000000000000994. [DOI] [PubMed] [Google Scholar]
- 12.Xu Bo, Gao R, Wang J, et al. A prospective, multicenter, randomized trial of paclitaxel-coated balloon versus paclitaxel-eluting stent for the treatment of drug-eluting stent in-stent restenosis: results from the PEPCAD China ISR trial. JACC Cardiovasc Interv. 2014;7(2):204–211. doi: 10.1016/j.jcin.2013.08.011. [DOI] [PubMed] [Google Scholar]
- 13.Giacoppo D, Alfonso F, Xu Bo, et al. Drug-Coated balloon angioplasty versus drug-eluting stent implantation in patients with coronary stent restenosis. J Am Coll Cardiol. 2020;75(21):2664–2678. doi: 10.1016/j.jacc.2020.04.006. [DOI] [PubMed] [Google Scholar]
- 14.Tu S, Barbato E, Köszegi Z, et al. Fractional flow reserve calculation from 3-dimensional quantitative coronary angiography and TIMI frame count: a fast computer model to quantify the functional significance of moderately obstructed coronary arteries. JACC Cardiovasc Interv. 2014;7(7):768–777. doi: 10.1016/j.jcin.2014.03.004. [DOI] [PubMed] [Google Scholar]
- 15.Xu Bo, Tu S, Qiao S, et al. Diagnostic accuracy of angiography-based quantitative flow ratio measurements for online assessment of coronary stenosis. J Am Coll Cardiol. 2017;70(25):3077–3087. doi: 10.1016/j.jacc.2017.10.035. [DOI] [PubMed] [Google Scholar]
- 16.Tu S, Westra J, Yang J, et al. Diagnostic accuracy of fast computational approaches to derive fractional flow reserve from diagnostic coronary angiography: the international multicenter FAVOR pilot study. JACC Cardiovasc Interv. 2016;9(19):2024–2035. doi: 10.1016/j.jcin.2016.07.013. [DOI] [PubMed] [Google Scholar]
- 17.Asano T, Katagiri Y, Collet C, et al. Functional comparison between the BuMA Supreme biodegradable polymer sirolimuseluting stent and a durable polymer zotarolimus-eluting coronary stent using quantitative flow ratio: PIONEER QFR substudy. EuroIntervention. 2018;14(5):e570–e579. doi: 10.4244/EIJD-17-00461. [DOI] [PubMed] [Google Scholar]
- 18.Liontou C, Mejía-Rentería H, Lauri F, et al. Quantitative flow ratio for functional evaluation of in-stent restenosis. EuroIntervention. 2021;17(5):e396–e398. doi: 10.4244/eij-d-18-00955.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Biscaglia S, Tebaldi M, Brugaletta S, et al. Prognostic value of QFR measured immediately after successful stent implantation: the international multicenter prospective HAWKEYE study. JACC Cardiovasc Interv. 2019;12(20):2079–2088. doi: 10.1016/j.jcin.2019.06.003. [DOI] [PubMed] [Google Scholar]
- 20.Cai X, Tian F, Jing J, et al. Prognostic value of quantitative flow ratio measured immediately after drug-coated balloon angioplasty for in-stent restenosis. Catheter Cardiovasc Interv. 2021;97(Suppl 2):1048–1054. doi: 10.1002/ccd.29640. [DOI] [PubMed] [Google Scholar]
- 21.Tang J, Hou H, Chu J, et al. Clinical implication of quantitative flow ratio to predict clinical events after drug-coated balloon angioplasty in patients with in-stent restenosis. Clin Cardiol. 2021;44(7):978–986. doi: 10.1002/clc.23630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Tu S, Ding D, Chang Y, et al. Diagnostic accuracy of quantitative flow ratio for assessment of coronary stenosis significance from a single angiographic view: A novel method based on bifurcation fractal law. Catheter Cardiovasc Interv. 2021;97(Suppl 2):1040–1047. doi: 10.1002/ccd.29592. [DOI] [PubMed] [Google Scholar]
- 23.Mehran R, Dangas G, Abizaid AS, et al. Angiographic patterns of in-stent restenosis: classification and implications for long-term outcome. Circulation. 1999;100(18):1872–1878. doi: 10.1161/01.cir.100.18.1872. [DOI] [PubMed] [Google Scholar]
- 24.Meijboom WB, Van Mieghem CAG, van Pelt N, et al. Comprehensive assessment of coronary artery stenoses: computed tomography coronary angiography versus conventional coronary angiography and correlation with fractional flow reserve in patients with stable angina. J Am Coll Cardiol. 2008;52(8):636–643. doi: 10.1016/j.jacc.2008.05.024. [DOI] [PubMed] [Google Scholar]
- 25.Toth G, Hamilos M, Pyxaras S, et al. Evolving concepts of angiogram: fractional flow reserve discordances in 4000 coronary stenoses. Eur Heart J. 35(40):2831–2838. doi: 10.1093/eurheartj/ehu094. [DOI] [PubMed] [Google Scholar]
- 26.Nunen Lv, Zimmermann F, Tonino P, et al. Fractional flow reserve versus angiography for guidance of PCI in patients with multivessel coronary artery disease (FAME): 5-year follow-up of a randomised controlled trial. Lancet. 2015;386(10006):1853–1860. doi: 10.1016/s0140-6736(15)00057-4. [DOI] [PubMed] [Google Scholar]
- 27.Levine GN, Bates ER, Blankenship JC, et al. 2011 ACCF/AHA/SCAI Guideline for Percutaneous Coronary Intervention: executive summary: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines and the Society for Cardiovascular Angiography and Interventions. Circulation. 2011;124(23):2574–2609. doi: 10.1161/CIR.0b013e31823a5596. [DOI] [PubMed] [Google Scholar]
- 28.NEUMANN FJ, SOUSA-UVA M, AHLSSON A, et al. 2018 ESC/EACTS Guidelines on myocardial revascularization. Eur Heart J. 2019;40(2):87–165. doi: 10.1093/eurheartj/ehy394. [DOI] [PubMed] [Google Scholar]
- 29.Ding D, Huang J, Westra J, et al. Immediate post-procedural functional assessment of percutaneous coronary intervention: current evidence and future directions. Eur Heart J. 2021;42(27):2695–2707. doi: 10.1093/eurheartj/ehab186. [DOI] [PubMed] [Google Scholar]
- 30.Gutiérrez-Chico JL, Chen Y, Yu W, et al. Diagnostic accuracy and reproducibility of optical flow ratio for functional evaluation of coronary stenosis in a prospective series. Cardiol J. 2020;27(4):350–361. doi: 10.5603/CJ.a2020.0071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Bech GJ, Pijls NH, De Bruyne B, et al. Usefulness of fractional flow reserve to predict clinical outcome after balloon angioplasty. Circulation. 1999;99(7):883–888. doi: 10.1161/01.cir.99.7.883. [DOI] [PubMed] [Google Scholar]
- 32.Lee JM, Hwang D, Choi KiH, et al. Prognostic impact of residual anatomic disease burden after functionally complete revascularization. Circ Cardiovasc Interv. 2020;13(9):e009232. doi: 10.1161/CIRCINTERVENTIONS.120.009232. [DOI] [PubMed] [Google Scholar]
- 33.Yerasi C, Case BC, Forrestal BJ, et al. Drug-coated balloon for de novo coronary artery disease: JACC state-of-the-art review. J Am Coll Cardiol. 2020;75(9):1061–1073. doi: 10.1016/j.jacc.2019.12.046. [DOI] [PubMed] [Google Scholar]
- 34.Kedhi E, Berta B, Roleder T, et al. Thin-cap fibroatheroma predicts clinical events in diabetic patients with normal fractional flow reserve: the COMBINE OCT-FFR trial. Eur Heart J. 2021 doi: 10.1093/eurheartj/ehab433. [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 35.Yang ZK, Shen Y, Dai Y, et al. Impact of coronary collateralization on long-term clinical outcomes in type 2 diabetic patients after successful recanalization of chronic total occlusion. Cardiovasc Diabetol. 2020;19(1):59. doi: 10.1186/s12933-020-01033-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Shi R, Shi Ke, Yang ZG, et al. Serial coronary computed tomography angiography-verified coronary plaque progression: comparison of stented patients with or without diabetes. Cardiovasc Diabetol. 2019;18(1):123. doi: 10.1186/s12933-019-0924-z. [DOI] [PMC free article] [PubMed] [Google Scholar]