Abstract
Significant advances have been made in recent years regarding the developmental trajectories of brain circuits and networks, revealing links between brain structure and function. Emerging evidence highlights the importance of developmental trajectories in determining early psychiatric outcomes. However, efforts to encourage crosstalk between basic developmental neuroscience and clinical practice are limited. Here, we focus on the potential advantage of considering features of neural circuit development when optimizing treatments for adolescent patient populations. Drawing on characteristics of adolescent neurodevelopment, we highlight two examples, safety cues and incentives, that leverage insights from neural circuit development and may have great promise for augmenting existing behavioral treatments for anxiety disorders during adolescence. This commentary seeks to serve as a framework to maximize the translational potential of basic research in developmental populations for strengthening psychiatric treatments. In turn, input from clinical practice including the identification of age-specific clinically relevant phenotypes will continue to guide future basic research in the same neural circuits to better reflect clinical practices. Encouraging reciprocal communication to bridge the gap between basic developmental neuroscience research and clinical implementation is an important step toward advancing both research and practice in this domain.
Keywords: Adolescence, Anxiety, Circuit neuroscience, Precision medicine, Reward, Safety, Translational
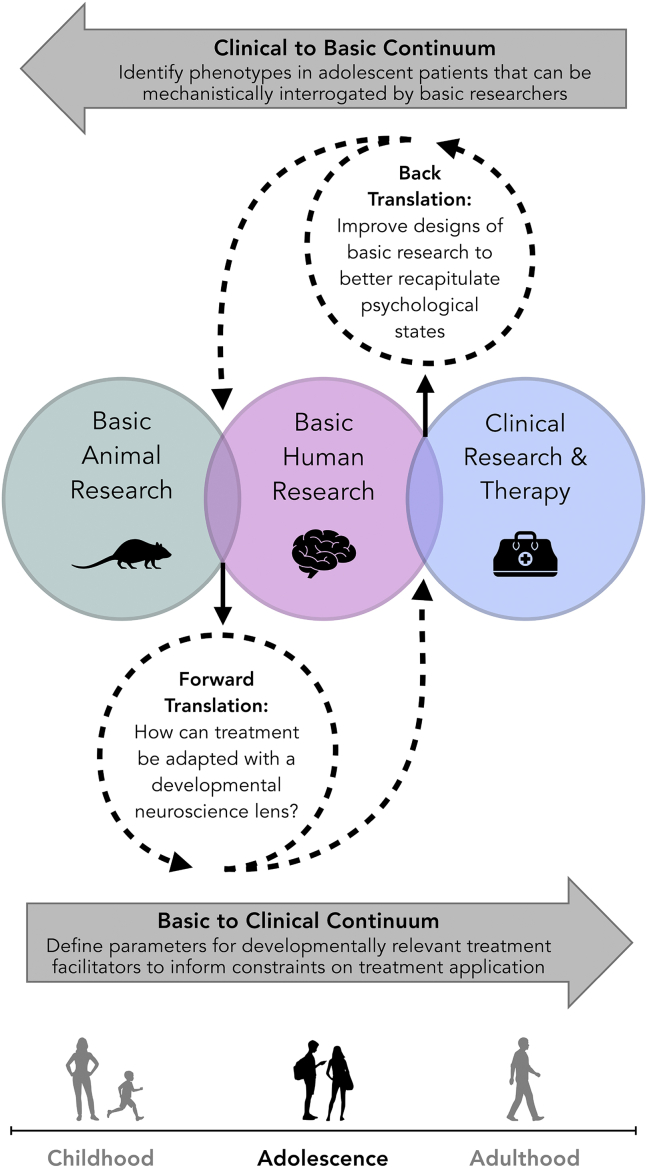
Treatment development across numerous fields of medicine often involves back-and-forth translation between research conducted in basic science laboratories and the application of clinical treatments. Termed “bench to bedside,” this collaborative arena promotes medical advances that have greatly improved patient care and outcomes. Recent efforts have highlighted the potential benefits of emphasizing the same integration of findings across basic neuroscience research and psychiatric clinical care in adults (1,2). However, much remains unknown about the relationship between brain development and behavior during adolescence, particularly with regard to both symptomatology and response to treatments in cases of psychiatric disorders. Great value may emerge from integrating basic research identifying unique features of adolescent functional neural circuitry with insights from clinical practice on effective treatment modalities (Figure 1). We posit that the incorporation of circuit-informed tools and techniques targeting clinically relevant and age-specific phenotypes identified by clinicians will provide a critical next step to increase both adherence and outcomes for the adolescent patient population.
Figure 1.
Translational utility of basic nonhuman animal research, basic human research, and clinical research and therapy.
Adolescence is a dynamic period of brain development, unique from preceding infancy and childhood as well as from later adulthood. Increases in behavioral engagement with affective cues and experiences (e.g., rewards, threats, novelty, and social interactions) and the simultaneous development of affective psychopathologies that commonly emerge during adolescence (e.g., mood, anxiety, substance use, and eating disorders) are believed to result from altered functioning in the neural circuitry underlying affective processing during adolescence. Anxiety disorders in particular pose a formidable burden, as their prevalence during development is estimated to be as high as 30% (3). Diagnoses peak at 13 years of age (4), and an even greater number of youths experience subdiagnostic symptoms (5). Furthermore, the emergence of anxiety during development has been associated with an increased severity of symptoms as well as comorbidities (including depression, substance abuse and dependence, and suicide), many of which can persist throughout adulthood. Importantly, elevated plasticity in the adolescent brain may provide a powerful opportunity for intervention to mitigate the adverse impacts of anxiety before it becomes a lifelong affliction.
Though the call has been made for back-and-forth translational approaches in adults (1,2), few similar examples exist for the treatment of adolescent anxiety. Similarities in neurobiological development and behavioral ecology during the rodent (postnatal days 29–50 in rats and mice) and human (typically defined as 12–18 years of age) adolescent periods (6,7) support translational work by providing access to both targeted examinations of the brain in animal models and the administration of cognitively complex tasks in humans. Here, we specifically focus on bridging the gap between rodent and human laboratory investigation of neural circuit development and clinical approaches to anxiety treatment during adolescence. As in other medical specialties, providing both basic research-informed treatments and conducting practice-informed research studies will be crucial to improving outcomes for adolescent anxiety.
Tailoring Treatment Based on Adolescent Neurobiology
Anxiety disorder symptoms often manifest as both the overgeneralization of fear to a range of stimuli and contexts and the underutilization of environmental indicators of safety. Thus, understanding fear circuitry development including structural and functional markers of development that are specific to adolescence and the resulting impact on fear learning may support approaches to treat adolescent anxiety [for a more comprehensive review of adolescent affective neurodevelopment, see (8, 9, 10, 11)]. Although reward circuitry is not typically considered with regard to fear learning and memory or anxiety disorders, the neural correlates supporting appetitive and aversive learning and memory involve overlapping brain regions (e.g., amygdala, ventral striatum, and prefrontal cortex; see below) and develop in parallel (10,12). Therefore, factors that impact or alter reward circuitry may also impact fear circuitry, and vice versa, highlighting the opportunity to leverage reward responding to mitigate fear responding. Below, we focus on the regulated use of safety cues and the inclusion of age-appropriate incentives as possible treatment modifications and describe how these modifications could leverage neural circuit characteristics specific to the adolescent brain.
De-emphasize Prefrontal-Based Cognitive Control
Across species, a characteristic marker of brain development during adolescence is differentially timed maturation of certain regions relative to others. The balance of maturational states, rather than the progress in one region per se, can greatly influence affective responding (13,14). Development of the prefrontal cortex is highly protracted, particularly in the context of affective regulation, continuing into the early 20s in humans (15). In contrast, the amygdala exhibits peaks in structural development in both humans (16) and rodents (17,18) during adolescence. Moreover, greater functional activation of the amygdala in response to fear-associated stimuli has been observed during adolescence relative to adults (19). Structural and functional connectivity between the amygdala and prefrontal control regions continue to strengthen across adolescence (20). Subsequently, the attainment of consistent affective control has been linked to maturation of the prefrontal cortex.
Continued maturation of the prefrontal cortex and associated regulatory behaviors across adolescence has emerged as a potential explanation for why treatments for anxiety that rely on prefrontal functioning may be less effective in adolescent populations (21). Certainly the prefrontal cortex makes important contributions to the regulation of emotional responding during adolescence. Yet, preceding full maturity, the refinement of connectivity between regions can contribute to the predominance of subcortical contributions to affective learning, memory, and behavior relative to lesser contributions of later maturing cognitive control regions (10,11,22). As the ability to integrate information across distributed brain regions increases across adolescence, the efficiency of affective processing also increases (23, 24, 25, 26).
This suggestion is concerning, as the most commonly used psychosocial treatment for all ages, cognitive behavioral therapy (CBT), includes core components based in extinction processes that are largely prefrontal dependent (i.e., exposure therapy). Importantly, though much work has focused on the parallels between extinction in the laboratory and exposure therapy, there are multiple cognitive components to CBT beyond the behavioral exposure components, such as cognitive restructuring, thought identification, and depersonalization. CBT has shown short-term effectiveness in reducing anxiety symptoms during late childhood and adolescence (27, 28, 29), particularly when combined with pharmacological intervention with serotonin reuptake inhibitors (5,30). However, despite evidence of symptom reduction in many patients at the time of treatment, an analysis of multiyear prospective data from adolescents treated for anxiety estimated that only ∼20% of treated youths exhibit an absence of all anxiety disorder symptoms at long-term follow-up (31). Even following a combination of behavioral and pharmacological (e.g., serotonin reuptake inhibitor) treatment, up to 50% of treated youths maintain persistent anxiety symptoms at a 6-year follow-up (32).
CBT has well-established intervention protocols for adults, and the vast majority of developmental adaptations have been geared toward being suitable for children specifically (7–12 years of age) or for a wide age range (6–18 years of age) (5,27, 28, 29). These modifications involve the addition of psychoeducation, relaxation, cognitive restructuring, and parent components, as well as both in vivo and imaginal exposures to treat symptoms of anxiety (28,32, 33, 34). Yet, because most studies examining anxiety treatment efficacy focus on children specifically or group adolescents and children together (34), there is a paucity of anxiety treatments designed for the unique developmental needs of adolescence. This may partly account for poorer treatment outcomes. Here, we suggest that modifying existing treatments to emphasize prefrontal-independent components may increase treatment efficacy during adolescence (8,35).
The use of safety cues to inhibit fear responding is one example of a promising avenue to increase the tolerability of the early stages of treatment and reduce attrition (Figure 2). Safety cues can be derived from repeatedly indicating the absence of an aversive outcome (e.g., physical or emotional distress, or catastrophe) expected following an encounter with a perceived threat. Alternatively, safety cues can be derived from positive interactions, such as those with social support figures (36). What serves as a safety cue is likely to differ between individuals, and may come in many forms (e.g., a small personal object, a photograph of a loved one, or a specific scent). Regardless of the exact modality, interacting with a safety cue should have a calming effect. Because of their explicit and specific association with safety, safety cues are capable of dampening the physiological and behavioral responses that normally arise when a threat is perceived through a process known as conditioned inhibition (37).
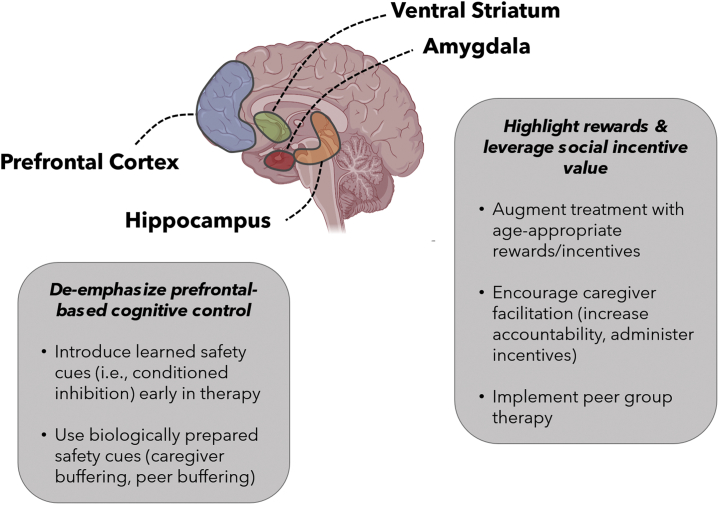
Figure 2.
Suggested treatment approaches leveraging a circuit-informed framework for anxiety disorder treatment during adolescence. The prefrontal cortex, ventral striatum, amygdala, and hippocampus are key components of affective neurocircuitry that play a strong role in fear and reward responding. Maturation of the prefrontal cortex is protracted, reducing the ability for top-down affective regulation. Therefore, bypassing the prefrontal cortex (e.g., by introducing safety cues early in therapy and engaging the hippocampus) may facilitate adolescent anxiety treatment. Simultaneously, the amygdala and ventral striatum exhibit elevated activity to rewards and social stimuli during adolescence. Therefore, identifying age-appropriate rewards and incentives and integrating caregivers or peers into treatment are promising avenues to improve treatment.
Adolescence may represent a unique opportunity to maximize the benefits of using safety cues to inhibit fear (38). Although further study of how the adolescent brain responds to safety cues is needed, in adults, safety cues act through brain regions that show elevated neural activity during adolescence [for review, see (38,39)] including the basolateral amygdala (40,41), striatum (40), and ventral hippocampus (42). Notably, safety cues have the potential to overcome the limitations of a still-developing prefrontal cortex to regulate fear. Indeed, studies in rodents indicate that the presence of a safety cue may reduce the amount of prefrontal recruitment needed to inhibit amygdala-based fear responding (40,43).
While neural mechanisms of safety learning and conditioned inhibition are an active area of research in rodents, less work has been conducted in humans. Parsing behavioral and circuit-related metrics of safety learning in nonanxious and anxious human populations will be an important area of future research (38,44). In this direction and as an important example of cross-species conservation, Meyer et al. (42) highlighted parallel ventral hippocampal–based circuits driving the fear-reducing effects of a safety cue in mice and nonanxious humans. Preclinical studies have shown that during adolescence there is enhanced plasticity and strengthening of connectivity in the hippocampus (45), which may promote the effectiveness of safety cues during this time in development. While additional translational research is necessary, these findings emphasize the potential value that can emerge from identifying adolescent-specific neural response patterns to components of treatment.
Because safety cues are stimuli distinct from a fear stimulus, they represent a departure from the safety learning that takes place during exposure therapy (or the laboratory equivalent, extinction), when the fear stimulus is encountered directly, and thus can be added as an additional feature of exposure, and other cognitive behavioral therapies. By providing a method to immediately reduce a fear response, the regulated addition of safety cues into conventional treatments may enhance opportunities to evaluate potential threats and learn about the likelihood that they will result in the anticipated distressing outcome. This is a key cognitive component of exposure therapy designed to reduce fear responding when the same threat is encountered in the future.
Establishing the parameters for introducing (and later removing) safety cues will maximize benefits of treatments incorporating safety cues as a regulated component [for additional discussion, see (38)] (Figure 1). It is critical that a safety cue does not facilitate avoidance. Avoidance could prevent an individual from facing and learning about perceived threats and interfere with the long-term ability to reduce fear (46) and has led to hesitancy among some clinicians to utilize safety cues during treatment. It is likely that considering the timing of safety cue delivery will be very important. For example, safety cues may be most effective when used subsequent to, rather than during or preceding, a fear-provoking event, respectively termed restorative and preventative safety cues (46,47).
Safety cues should not replace other methods of reducing anxiety. The most efficacious approach would likely involve judiciously incorporating safety cues into existing treatments (48), providing an opportunity to augment learning about why anxiety responses manifest. Despite a large body of evidence supporting its efficacy, exposure is underutilized in the treatment of anxiety in adults (49) and youths (50,51), owing to concerns regarding patient dropout (52) and negative beliefs about patients’ ability to tolerate exposures (53). Relatedly, insufficient decreases in anxiety during the first exposure session have been linked to patient dropout (54). By providing a source of temporary relief when distress is at its highest (48,55), safety cues may mitigate patient dropout, subsequently encouraging the initiation of (56) and adherence to treatment (48).
Safety cues are often used without the explicit knowledge of the patient or the clinician. Thus, early treatment sessions could incorporate education about what a safety cue is, how to identify each individual’s cue, and how to appropriately engage with it. Just as exposures take place with increasing frequency outside of the controlled clinician’s office, then the home, and eventually generalizing to the broader world, so too could a safety cue be utilized to facilitate early engagement with treatment, followed by less frequent use. There is also the potential to use safety cues in a virtual reality setting by programming an image of the safety cue to coincide with particularly fear-provoking components of the exposure session. As basic neuroscience studies examine the modality, timing, and frequency of safety cue presentations and how they engage fear regulation circuitry during development, clinical practice will be well prepared for effective translation of safety cues into treatment.
Highlight Rewards and Leverage Social Incentive Value
Subcortical limbic regions implicated in emotional learning mature earlier in adolescence than prefrontal cortical control areas in both humans and rodents (22). For example, the amygdala and ventral striatum have been strongly implicated in affective responding during adolescence. While greater functional activation of the amygdala in response to fear-associated stimuli has been observed during adolescence relative to adulthood (19), both the amygdala and ventral striatum also exhibit increased functional activation to reward and social stimuli in adolescents (57, 58, 59), particularly when the value of the reward is high or the probability of receiving the reward is low (60,61). Neural activity driven by the amygdala, ventral striatum, and related limbic regions with limited prefrontal regulation has been proposed as a potential explanation for differing incentive salience attribution processes apparent during adolescence (8, 9, 10, 11,62,63). Both rewarding and novel outcomes drive elevations in reward circuitry activity, including dopaminergic neurons originating in the ventral tegmental area, and in the ventral striatum, a major dopaminergic contact site (64). Therefore, focusing aspects of treatment toward positive environmental stimuli may capitalize on adolescents' heightened neural responses to rewards. While already commonly used in clinical treatment, a careful consideration of which incentives to use (e.g., monetary gain, social acceptance, positive feedback), how frequently to use them, how predictable they are, and how best to administer them has the potential to both increase the salience of incentives and reduce boredom with particular incentives over time (65). Identifying these parameters will be facilitated by communication between clinicians and basic neuroscientists (Figure 1), as the former can inform what may be feasibly used in treatment, while the latter can systematically determine age-specific behavioral and neural response patterns to different incentives and how they change across multiple administrations. Using functional neuroimaging methods to characterize how reward circuitry in adolescents responds to incentives typically used in adult clinical practice is one way in which neuroscientists may help clinicians better tailor treatments for this specific population.
It may also be beneficial to direct adolescent patients to attend to positive cues, rather than to shift their focus away from threat cues (66). Initial evidence supports the use of a positive valence shift of this kind for attention bias modification training in children (6-12 years of age) (67). Similarly, in adults, introducing a novel nonthreatening stimulus following a perceived threat stimulus strengthened extinction (68). Although studies in adolescents have yet to be conducted, by engaging reward circuitry (by attending to positive cues) and reducing the requirement of mature prefrontal cognitive control networks (which are necessary to effectively ignore threat cues), this approach would likely be well suited for use during adolescence.
Alongside heightened reactivity to rewarding and novel stimuli, a notable social reorientation takes place during adolescence, resulting in a shift in focus from caregivers toward peers, coupled with an increase in the neurobiological salience of peer stimuli (59,69). In addition to this behavioral and neurobiological reorientation, social status and peer acceptance become increasingly paramount to self-evaluations (70, 71, 72). As a result, adolescents are often more motivated by the desire to obtain positive peer feedback and avoid social rejection than individuals at other ages (72). Anxious adolescents may be even more attentive to positive peer feedback (71). Methods to leverage peer incentive value may include using peers as the reward by encouraging adolescents to visit a friend subsequent to a therapy session or employing a group treatment format (73,74). The group setting has the potential to serve as a controlled environment in which adolescents can receive positive feedback from peers, providing the potential to counteract learning and memory biases of anxious adolescents in a safe, guided, and nonjudgmental environment. Peer-facilitated interventions in schools may be similarly motivating (75). Somewhat paradoxically, anxious teenagers show a striatal response indicative of reward processing when faced with confirming evidence that a peer disliked them (i.e., feedback that their prediction of negative peer feedback was correct), which may fuel social avoidance behaviors (76). Thus, the initial integration of peers in treatment may be most efficacious when the relationship between the patient and the peer is well established (i.e., predictable).
While the salience of peer interactions is enhanced during adolescence, caregivers remain important modulators of behavior (77) and the stress response (78), and commonly serve as the gatekeepers to treatment and relevant incentives. Parents are responsible in part for ensuring that their adolescent children access treatment, frequently accompanying them to visits, signing consent for their participation, and bearing primary responsibility for attrition. Parents also may be able to provide support for their adolescent child in a way that is not directly interpreted as help [i.e., invisible support (79)], which might be especially impactful for adolescents who are striving for independence. However, more work is needed to better understand how parents can contribute to successful treatment efforts.
Notably, safety cues may be inherently rewarding, and their rewarding properties have been linked to increased striatal activity (40). Thus, by leveraging elevated activity in the striatum during adolescence, safety cues may enhance existing treatments for anxiety by both facilitating fear reduction and providing an incentive to engage with treatment. The heightened salience of social relationships and the rewarding properties of social interactions have also led to the consideration of how social support figures may serve as safety cues. Research has suggested that social support figures comprise a special class of ecologically relevant prepared safety cues such that they have unique capacity to inhibit conditioned fear responses to other stimuli (36). This is reminiscent of the buffering role that social figures, particularly caregivers, provide early in life (80). In contrast to strangers, or otherwise rewarding stimuli, the presence of social support figures enhances extinction of conditioned fear and dampens fear reinstatement in nonanxious adult humans (81). Notably, the nature of the social interaction and mechanism for fear regulation is likely different during adolescence. For example, caregiver buffering activates the ventral tegmental area during infancy in rats, but there is a switch to recruitment of the prefrontal cortex as infants approach adolescence (postnatal day 28) (82). While this demonstrates recruitment of the prefrontal cortex during an initial period of independence in service of affective regulation, the ability to effectively regulate fear in the absence of the caregiver appears to benefit from continued maturation of prefrontal circuitry well into adulthood (15). For anxious adolescents, it is possible that use of parents or peers as prepared safety cues may similarly help improve fear regulation outcomes and facilitate an infrastructure for broader generalization of prefrontal-based affective control processes.
Neural Circuit Profiles Associated With Adolescent Anxiety
A deeper understanding of alterations in neural circuitry associated with adolescent anxiety is important for developing effective circuit-based treatments. For detailed review, we direct readers to (38,83). Briefly, findings from large-scale studies on neural connectivity suggest that adolescent anxiety disorders likely arise from perturbations within and between distributed brain networks as opposed to focal brain regions (84, 85, 86, 87). Anxiety is often associated with atypical functioning of the salience network of the brain, which comprises frontoamygdala circuitry that supports threat responses and attentional control, and functional connectivity between salience and executive control networks that support self-regulation and decision making (83,88). Amygdala hyperreactivity is consistently observed during threat processing among adolescents and adults with anxiety disorders (89,90). Further, the prefrontal cortex appears to be inefficiently engaged during emotional processing tasks, but the nature of frontoamygdala connectivity (hyper- or hypoconnectivity) appears to be task and age dependent such that adolescents with anxiety disorders demonstrate increased prefrontal engagement to threat and safety cues during threat appraisal, whereas blunted prefrontal recruitment has been observed among adults with anxiety disorders (91). A small, but emerging, body of research suggests that atypical functioning of reward-related circuitry is also observed in adolescents with and at risk for anxiety (92, 93, 94, 95, 96). Specifically, adolescents with anxiety exhibit neural hypersensitivity (i.e., increased striatal activity) to incentive cues signaling contingent reward relative to healthy control subjects (94). These findings are consistent with cross-species research indicating that adolescence is uniquely characterized by heightened neural sensitivity to salient affective cues, both appetitive and aversive, and they underscore the need to capitalize on intervention strategies that emphasize rewarding stimuli and circumvent the prefrontal cortex (38,86,87).
Dramatic differences in the environment corresponding with shifts in school, home, and social activities can have strong implications for neurobiological and psychosocial functioning (97). As a result, despite similarities evident between adolescents and adults, the structure of the environment (e.g., focus on and proximity to peers, regulated school schedules, and parental involvement in treatment access) can lead to differential engagement of frontoamygdala and frontostriatal circuitry. Treatments that link threats and contingent rewards via safety cues have the potential to strengthen communication among neural networks. In turn, increasing crosstalk between parallel neural circuits can help set a foundation to promote the confrontation of challenging tasks and facilitate prosocial behaviors (86), both of which may be particularly effective for reducing anxiety during the adolescent period. Other considerations (biological sex, early life experiences, and access to treatment) (Table 1) are beyond the scope of this article but are likely to influence heterogeneity in symptomology and treatment outcomes for adolescent anxiety.
Table 1.
Additional Considerations
| Biological Sex | Early Life Experiences | Access to Treatment |
|---|---|---|
| Females demonstrate higher lifetime prevalence of anxiety disorders, increased intensity of symptoms, and are more likely to report having an anxiety disorder (3,4,101). These findings may be linked to the fluctuations in sex hormones associated with puberty (increasing estrogen receptors in females and testosterone in males), which impact brain development, including frontoamygdala connectivity (102,103) and thus may alter the onset and course of anxiety disorders for females and males early in adolescence. | Particularly if stressful, early life experiences may heighten risk for anxiety disorders through changes in fear learning, memory processes, and related neural circuitry (6,104, 105, 106). Subsequently, early life stress can have cascading effects on the expression of social motivations and behaviors during adolescence (107), as well as mediate the efficacy of the caregiver as a social safety cue (108,109). | Less than two thirds of children and adolescents diagnosed with anxiety currently receive treatment (101). Moreover, despite the peak in diagnoses early in adolescence, treatment for anxiety is often not sought until years later (110). Moving forward, efforts to improve treatment access in addition to treatment efficacy will be crucial to mitigate the profound impacts of anxiety disorders. |
Alongside the push for precision medicine, an increased focus on the history of each patient may also serve to improve the treatment of adolescent psychiatric disease (111,112). Numerous variables can influence heterogeneity in symptomology and treatment outcomes for adolescent anxiety. There is a strong need to consider factors that may directly influence the status of developmental processes in the adolescent brain to most effectively tailor treatment. Important considerations are listed in the table.
Unfortunately, the impact of treatment on neural circuitry in adolescents with anxiety has yet to be delineated. The little available clinical research to date has focused on identifying neurofunctional biomarkers of adolescents’ pre- to posttreatment response during CBT and serotonin reuptake inhibitor administration. Using biomarkers to predict response to treatment can be a valuable means to tailor the treatment plan to each patient. Yet, it is also crucial to determine how different treatment components impact neural circuitry during adolescence so that precision medicine can more effectively address the heterogeneous presentation of anxiety disorders and unique neurodevelopmental and social contexts of adolescence.
Conclusions
Adding to a scaffolding constructed across preceding periods of neurodevelopment, adolescence may be the last window of heightened plasticity before neural systems reach a relative state of stability, setting the stage for the infrastructure of the adult brain. The available literature paints a picture of adolescence as a period of both great vulnerability and great potential for resilience. Many have focused on how heightened plasticity during adolescence may leave the brain susceptible to environmental stressors that alter normative developmental trajectories and increase risk for psychopathology (9,98). However, in addition to susceptibility for negative outcomes, it is crucial to recognize the concomitant opportunity for enhanced intervention efficacy.
Here, we describe the use of safety cues, incentives, and their overlap in the social domain to exemplify avenues that may be particularly efficacious to bolster treatment during adolescence. Findings from circuit-based neuroscience have highlighted a strong need to emphasize features of treatment that do not rely on mature prefrontal regulation of affective neural circuitry. However, in addition to limitations in executive functions that regulate fear responding, adolescents may also experience increases in the strength or salience of fear cues. Thus, our focus on safety cues and incentives is also driven by evidence of developmental increases in functional activation of neural circuitry underlying reward and safety learning. Safety cues and incentives each may independently serve to redirect attention toward other affectively salient features of the environment and thus reduce the strength of the fear cue. This twofold means that to regulate fear responding may then pave the way for improvements to treatment administration. Our intention is not to suggest that the use of safety cues or incentives supplant existing treatments. Rather, tailoring treatment administration based on what is known of the adolescent brain and how adolescents experience the world may be a strong means to improve outcomes. For example, while CBT is commonly employed for the treatment of anxiety, certain components of CBT practice like exposure therapy are heavily dependent on prefrontal functioning. Just as laboratory measures of extinction processes have informed the implementation of exposure therapy, basic neuroscientists can focus on better characterizing the circuitry supporting the cognitive components of CBT to understand the neural bases of distorted thinking patterns that contribute to adolescent anxiety, and help clinicians address these phenotypes.
In sum, our goal is to set a foundation to bridge the gap between developmental basic neuroscience research and implementation of psychiatric care during adolescence. While much of what we have discussed has focused on how treatments may be optimized by taking a circuit-based perspective, this process is not a one-way street, but rather is an opportunity to encourage reciprocal communication between basic and clinical research with the mutually beneficial goal of increased treatment efficacy (Figure 1). For basic researchers, communicating with clinicians about the nuances of treatment in practice and identifying symptoms not currently captured by research designs will be important first steps. In turn, tailoring experiments to better operationalize and isolate behaviors resulting from current therapeutic approaches like CBT and focusing neurobiological interrogations on discrete clinically relevant and age-specific phenotypes may promote hypothesis-driven treatment optimization, replacing the lengthier trial-and-error processes carried out in the clinic. For clinicians, taking into account the underlying neural mechanisms of successful existing treatments may emphasize more efficient methods of treatment delivery, increase adherence, and improve outcomes.
This process will also require acknowledgment of the limitations in translating between research and clinical practice. Preeminently, while conservation of brain and behavioral features is apparent across rodents and humans, key differences exist in how the surrounding world is experienced and used to guide behavior. For example, although frontal regions of the rodent brain show functional homology to human and nonhuman primate prefrontal cortex during fear and reward processes, there remains ongoing debate regarding the degree to which prefrontal anatomy in rodents parallels that in humans. Nonetheless, cross-species translational circuit neuroscience work has the attractive advantage of providing access to both fine-grained appraisal of the brain in animal models and the administration of psychologically valid tasks in humans. Alongside circuit-based research endeavors, insight may also be obtained from behavioral tasks and computational models of typical and pathological behavioral states (99,100). As noted above, although extinction as a basic neuroscience parallel of exposure therapy in the clinic has gained much traction, many other features of CBT remain to be effectively modeled.
Overall, we hope that by highlighting the ways in which neural circuit development in adolescence can support ongoing clinical practice, there will be greater back-and-forth dialogue to advance both research and practice in this domain.
Acknowledgments and Disclosures
The development and writing of this article was supported by National Institutes of Mental Health Pathway to Independence Awards (Grant Nos. K99MH119320 [to HCM] and K99MH124434 [to MO]), the National Center for Advancing Translational Sciences (Grant No. TL1-TR-002386 [to DMG]), the National Science Foundation (Graduate Research Fellowship [to PAB]), a Brain and Behavior Foundation NARSAD Young Investigator Award (to MO), the National Institute of Child Health and Human Development (Grant No. R37HD083217 [to RS]), the National Institute of Mental Health (Grant No. R01MH123154 [to FSL]), and the New York Community Trust (to FSL).
This review is the collaborative product of thoughtful discussions between the FSL (Weill Cornell Medicine), RS (New York University), and NM (Columbia University) laboratories, including perspectives from rodent and human researchers with a core focus on affective neurodevelopment and how typical and atypical trajectories of brain development impact mental health. All authors contributed equally to conceptualization. HCM, AF, and BLC contributed to visualization. HCM, AF, AV, DMG, PAB, CH, and MO contributed to writing of the original draft. All authors contributed to writing—review and editing.
The authors report no biomedical financial interests or potential conflicts of interest.
References
- 1.Corlett P.R., Schoenbaum G. Leveraging basic science for the clinic—From bench to bedside. JAMA Psychiatry. 2021;78:331–334. doi: 10.1001/jamapsychiatry.2020.3656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ford C.L., Young L.J. Translational opportunities for circuit-based social neuroscience: Advancing 21st century psychiatry. Curr Opin Neurobiol. 2021;68:1–8. doi: 10.1016/j.conb.2020.11.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Merikangas K.R., He J., Burstein M., Swanson S.A., Avenevoli S., Cui L., et al. Lifetime prevalence of mental disorders in U.S. adolescents: Results from the National Comorbidity Survey Replication–Adolescent Supplement (NCS-A) J Am Acad Child Adolesc Psychiatry. 2010;49:980–989. doi: 10.1016/j.jaac.2010.05.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kessler R.C., Berglund P., Demler O., Jin R., Merikangas K.R., Walters E.E. Lifetime prevalence and age-of-onset distributions of DSM-IV disorders in the National Comorbidity Survey Replication. Arch Gen Psychiatry. 2005;62:593–602. doi: 10.1001/archpsyc.62.6.593. [DOI] [PubMed] [Google Scholar]
- 5.Walkup J.T., Albano A.M., Piacentini J., Birmaher B., Compton S.N., Sherrill J.T., et al. Cognitive behavioral therapy, sertraline, or a combination in childhood anxiety. N Engl J Med. 2008;359:2753–2766. doi: 10.1056/NEJMoa0804633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Callaghan B.L., Meyer H., Opendak M., Van Tieghem M., Harmon C., Li A., et al. Using a developmental ecology framework to align fear neurobiology across species. Annu Rev Clin Psychol. 2019;15:345–349. doi: 10.1146/annurev-clinpsy-050718-095727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Spear L.P. The adolescent brain and age-related behavioral manifestations. Neurosci Biobehav Rev. 2000;24:417–463. doi: 10.1016/s0149-7634(00)00014-2. [DOI] [PubMed] [Google Scholar]
- 8.Baker K.D., Den M.L., Graham B.M., Richardson R. A window of vulnerability: Impaired fear extinction in adolescence. Neurobiol Learn Mem. 2014;113:90–100. doi: 10.1016/j.nlm.2013.10.009. [DOI] [PubMed] [Google Scholar]
- 9.Gerhard D.M., Meyer H.C., Lee F.S. An adolescent sensitive period for threat responding: Impacts of stress and sex. Biol Psychiatry. 2021;89:651–658. doi: 10.1016/j.biopsych.2020.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Meyer H.C., Pattwell S.S. In: The Cognitive Neurosciences. 6th ed. Poeppel D., Mangun G.R., Gazzaniga M.S., editors. MIT Press; Cambridge, MA: 2020. Memory across development, with insights from emotional learning: A nonlinear process; pp. 243–254. [Google Scholar]
- 11.Spear L.P. Adolescent neurodevelopment. J Adolesc Health. 2013;52(2 Suppl 2):S7–S13. doi: 10.1016/j.jadohealth.2012.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lago T., Davis A., Grillon C., Ernst M. Striatum on the anxiety map: Small detours into adolescence. Brain Res. 2017;1654:177–184. doi: 10.1016/j.brainres.2016.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Casey B.J., Getz S., Galvan A. The adolescent brain. Dev Rev. 2008;28:62–77. doi: 10.1016/j.dr.2007.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Pattwell S.S., Lee F.S., Casey B.J. Fear learning and memory across adolescent development: Hormones and Behavior Special Issue: Puberty and Adolescence. Horm Behav. 2013;64:380–389. doi: 10.1016/j.yhbeh.2013.01.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cohen A.O., Breiner K., Steinberg L., Bonnie R.J., Scott E.S., Taylor-Thompson K.A., et al. When is an adolescent an adult? Assessing cognitive control in emotional and nonemotional contexts. Psychol Sci. 2016;27:549–562. doi: 10.1177/0956797615627625. [DOI] [PubMed] [Google Scholar]
- 16.Hu S., Pruessner J.C., Coupé P., Collins D.L. Volumetric analysis of medial temporal lobe structures in brain development from childhood to adolescence. NeuroImage. 2013;74:276–287. doi: 10.1016/j.neuroimage.2013.02.032. [DOI] [PubMed] [Google Scholar]
- 17.Rubinow M.J., Juraska J.M. Neuron and glia numbers in the basolateral nucleus of the amygdala from preweaning through old age in male and female rats: A stereological study. J Comp Neurol. 2009;512:717–725. doi: 10.1002/cne.21924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Saul M.L., Helmreich D.L., Callahan L.M., Fudge J.L. Differences in amygdala cell proliferation between adolescent and young adult rats. Dev Psychobiol. 2013;56:517–528. doi: 10.1002/dev.21115. [DOI] [PubMed] [Google Scholar]
- 19.Lau J.Y., Britton J.C., Nelson E.E., Angold A., Ernst M., Goldwin M., et al. Distinct neural signatures of threat learning in adolescents and adults. Proc Natl Acad Sci U S A. 2011;108:4500–4505. doi: 10.1073/pnas.1005494108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gee D.G., Humphreys K.L., Flannery J., Goff B., Telzer E.H., Shapiro M., et al. A developmental shift from positive to negative connectivity in human amygdala–prefrontal circuitry. J Neurosci. 2013;33:4584–4593. doi: 10.1523/JNEUROSCI.3446-12.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Drysdale A.T., Hartley C.A., Pattwell S.S., Ruberry E.J., Somerville L.H., Compton S.N., et al. Fear and anxiety from principle to practice: Implications for when to treat youth with anxiety disorders. Biol Psychiatry. 2014;75:e19–e20. doi: 10.1016/j.biopsych.2013.08.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Casey B.J., Jones R.M., Levita L., Libby V., Pattwell S.S., Ruberry E.J., et al. The storm and stress of adolescence: Insights from human imaging and mouse genetics. Dev Psychobiol. 2010;52:225–235. doi: 10.1002/dev.20447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Hwang K., Velanova K., Luna B. Strengthening of top-down frontal cognitive control networks underlying the development of inhibitory control: A functional magnetic resonance imaging effective connectivity study. J Neurosci. 2010;30:15535–15545. doi: 10.1523/JNEUROSCI.2825-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kundu P., Benson B.E., Rosen D., Frangou S., Leibenluft E., Luh W.-M., et al. The integration of functional brain activity from adolescence to adulthood. J Neurosci. 2018;38:3559–3570. doi: 10.1523/JNEUROSCI.1864-17.2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Durston S., Davidson M.C., Tottenham N., Galvan A., Spicer J., Fossella J.A., Casey B.J. A shift from diffuse to focal cortical activity with development. Dev Sci. 2006;9:1–8. doi: 10.1111/j.1467-7687.2005.00454.x. [DOI] [PubMed] [Google Scholar]
- 26.Gracia-Tabuenca Z., Moreno M.B., Barrios F.A., Alcauter S. Development of the brain functional connectome follows puberty-dependent nonlinear trajectories. NeuroImage. 2021;229:117769. doi: 10.1016/j.neuroimage.2021.117769. [DOI] [PubMed] [Google Scholar]
- 27.Compton S.N., March J.S., Brent D., Albano A.M., Weersing R., Curry J. Cognitive-behavioral psychotherapy for anxiety and depressive disorders in children and adolescents: An evidence-based medicine review. J Am Acad Child Adolesc Psychiatry. 2004;43:930–959. doi: 10.1097/01.chi.0000127589.57468.bf. [DOI] [PubMed] [Google Scholar]
- 28.Kendall P.C. Treating anxiety disorders in children: Results of a randomized clinical trial. J Consult Clin Psychol. 1994;62:100–110. doi: 10.1037//0022-006x.62.1.100. [DOI] [PubMed] [Google Scholar]
- 29.Kendall P.C., Peterman J.S. CBT for adolescents with anxiety: Mature yet still developing. Am J Psychiatry. 2015;172:519–530. doi: 10.1176/appi.ajp.2015.14081061. [DOI] [PubMed] [Google Scholar]
- 30.Taylor J.H., Lebowitz E.R., Jakubovski E., Coughlin C.G., Silverman W.K., Bloch M.H. Monotherapy insufficient in severe anxiety? Predictors and moderators in the Child/Adolescent Anxiety Multimodal Study. J Clin Child Adolesc Psychol. 2018;47:266–281. doi: 10.1080/15374416.2017.1371028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ginsburg G.S., Becker-Haimes E.M., Keeton C., Kendall P.C., Iyengar S., Sakolsky D., et al. Results from the Child/Adolescent Anxiety Multimodal Extended Long-Term Study (CAMELS): Primary anxiety outcomes. J Am Acad Child Adolesc Psychiatry. 2018;57:471–480. doi: 10.1016/j.jaac.2018.03.017. [DOI] [PubMed] [Google Scholar]
- 32.Ginsburg G.S., Becker E.M., Keeton C.P., Sakolsky D., Piacentini J., Albano A.M., et al. Naturalistic follow-up of youths treated for pediatric anxiety disorders. JAMA Psychiatry. 2014;71:310–318. doi: 10.1001/jamapsychiatry.2013.4186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kendall P.C., Southam-Gerow M.A. Long-term follow-up of a cognitive-behavioral therapy for anxiety-disordered youth. J Consult Clin Psychol. 1996;64:724–730. doi: 10.1037//0022-006x.64.4.724. [DOI] [PubMed] [Google Scholar]
- 34.Seligman L.D., Ollendick T.H. Cognitive behavioral therapy for anxiety disorders in youth. Child Adolesc Psychiatr Clin N Am. 2011;20:217–238. doi: 10.1016/j.chc.2011.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Johnson D.C., Casey B.J. Easy to remember, difficult to forget: The development of fear regulation. Dev Cogn Neurosci. 2015;11:42–55. doi: 10.1016/j.dcn.2014.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hornstein E.A., Eisenberger N.I. A social safety net: Developing a model of social-support figures as prepared safety stimuli. Curr Dir Psychol Sci. 2018;27:25–31. doi: 10.1177/0956797616646580. [DOI] [PubMed] [Google Scholar]
- 37.Rescorla R.A. Conditioned inhibition of fear resulting from negative CS-US contingencies. J Comp Physiol Psychol. 1969;67:504–509. doi: 10.1037/h0027313. [DOI] [PubMed] [Google Scholar]
- 38.Odriozola P., Gee D.G. Learning about safety: Conditioned inhibition as a novel approach to fear reduction targeting the developing brain. Am J Psychiatry. 2021;178:136–155. doi: 10.1176/appi.ajp.2020.20020232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Foilb A.R., Christianson J.P. In: Neurobiology of Abnormal Emotion and Motivated Behaviors. Sangha S., Foti D., editors. Academic Press; San Diego: 2018. 11 - Brain mechanisms for learning and using safety signals; pp. 204–222. [Google Scholar]
- 40.Rogan M.T., Leon K.S., Perez D.L., Kandel E.R. Distinct neural signatures for safety and danger in the amygdala and striatum of the mouse. Neuron. 2005;46:309–320. doi: 10.1016/j.neuron.2005.02.017. [DOI] [PubMed] [Google Scholar]
- 41.Ng K.H., Pollock M.W., Urbanczyk P.J., Sangha S. Altering D1 receptor activity in the basolateral amygdala impairs fear suppression during a safety cue. Neurobiol Learn Mem. 2018;147:26–34. doi: 10.1016/j.nlm.2017.11.011. [DOI] [PubMed] [Google Scholar]
- 42.Meyer H.C., Odriozola P., Cohodes E.M., Mandell J.D., Li A., Yang R., et al. Ventral hippocampus interacts with prelimbic cortex during inhibition of threat response via learned safety in both mice and humans. Proc Natl Acad Sci U S A. 2019;116:26970–26979. doi: 10.1073/pnas.1910481116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ostroff L.E., Cain C.K., Bedont J., Monfils M.H., LeDoux J.E. Fear and safety learning differentially affect synapse size and dendritic translation in the lateral amygdala. Proc Natl Acad Sci U S A. 2010;107:9418–9423. doi: 10.1073/pnas.0913384107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Laing P.A.F., Harrison B.J. Safety learning and the Pavlovian conditioned inhibition of fear in humans: Current state and future directions. Neurosci Biobehav Rev. 2021;127:659–674. doi: 10.1016/j.neubiorev.2021.05.014. [DOI] [PubMed] [Google Scholar]
- 45.Pattwell S.S., Liston C., Jing D., Ninan I., Yang R.R., Witztum J., et al. Dynamic changes in neural circuitry during adolescence are associated with persistent attenuation of fear memories. Nat Commun. 2016;7 doi: 10.1038/ncomms11475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Helbig-Lang S., Petermann F. Tolerate or eliminate? A systematic review on the effects of safety behavior across anxiety disorders. Clin Psychol Sci Pract. 2010;17:218–233. [Google Scholar]
- 47.Goetz A.R., Davine T.P., Siwiec S.G., Lee H.-J. The functional value of preventive and restorative safety behaviors: A systematic review of the literature. Clin Psychol Rev. 2016;44:112–124. doi: 10.1016/j.cpr.2015.12.005. [DOI] [PubMed] [Google Scholar]
- 48.Rachman S., Radomsky A.S., Shafran R. Safety behaviour: A reconsideration. Behav Res Ther. 2008;46:163–173. doi: 10.1016/j.brat.2007.11.008. [DOI] [PubMed] [Google Scholar]
- 49.Hipol L.J., Deacon B.J. Dissemination of evidence-based practices for anxiety disorders in Wyoming: A survey of practicing psychotherapists. Behav Modif. 2013;37:170–188. doi: 10.1177/0145445512458794. [DOI] [PubMed] [Google Scholar]
- 50.Reid A.M., Bolshakova M.I., Guzick A.G., Fernandez A.G., Striley C.W., Geffken G.R., et al. Common barriers to the dissemination of exposure therapy for youth with anxiety disorders. Community Ment Health J. 2017;53:432–437. doi: 10.1007/s10597-017-0108-9. [DOI] [PubMed] [Google Scholar]
- 51.Whiteside S.P.H., Deacon B.J., Benito K., Stewart E. Factors associated with practitioners’ use of exposure therapy for childhood anxiety disorders. J Anxiety Disord. 2016;40:29–36. doi: 10.1016/j.janxdis.2016.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Kehle-Forbes S.M., Meis L.A., Spoont M.R., Polusny M.A. Treatment initiation and dropout from prolonged exposure and cognitive processing therapy in a VA outpatient clinic. Psychol Trauma. 2016;8:107–114. doi: 10.1037/tra0000065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Keleher J., Jassi A., Krebs G. Clinician-reported barriers to using exposure with response prevention in the treatment of paediatric obsessive-compulsive disorder. J Obsessive Compuls Relat Disord. 2020;24 doi: 10.1016/j.jocrd.2019.100498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Norton P.J., Hayes-Skelton S.A., Klenck S.C. What happens in session does not stay in session: Changes within exposures predict subsequent improvement and dropout. J Anxiety Disord. 2011;25:654–660. doi: 10.1016/j.janxdis.2011.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Milosevic I., Radomsky A.S. Safety behaviour does not necessarily interfere with exposure therapy. Behav Res Ther. 2008;46:1111–1118. doi: 10.1016/j.brat.2008.05.011. [DOI] [PubMed] [Google Scholar]
- 56.Milosevic I., Radomsky A.S. Incorporating the judicious use of safety behavior into exposure-based treatments for anxiety disorders: A study of treatment acceptability. J Cogn Psychother. 2013;27:155–174. doi: 10.1891/0889-8391.27.2.155. [DOI] [PubMed] [Google Scholar]
- 57.Ernst M., Nelson E.E., Jazbec S., McClure E.B., Monk C.S., Leibenluft E., et al. Amygdala and nucleus accumbens in responses to receipt and omission of gains in adults and adolescents. NeuroImage. 2005;25:1279–1291. doi: 10.1016/j.neuroimage.2004.12.038. [DOI] [PubMed] [Google Scholar]
- 58.Galvan A., Hare T.A., Parra C.E., Penn J., Voss H., Glover G., et al. Earlier development of the accumbens relative to orbitofrontal cortex might underlie risk-taking behavior in adolescents. J Neurosci. 2006;26:6885–6892. doi: 10.1523/JNEUROSCI.1062-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Smith A.R., Steinberg L., Strang N., Chein J. Age differences in the impact of peers on adolescents’ and adults’ neural response to reward. Dev Cogn Neurosci. 2015;11:75–82. doi: 10.1016/j.dcn.2014.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Insel C., Somerville L.H. Asymmetric neural tracking of gain and loss magnitude during adolescence. Soc Cogn Affect Neurosci. 2018;13:785–796. doi: 10.1093/scan/nsy058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Silverman M.H., Jedd K., Luciana M. Neural networks involved in adolescent reward processing: An activation likelihood estimation meta-analysis of functional neuroimaging studies. NeuroImage. 2015;122:427–439. doi: 10.1016/j.neuroimage.2015.07.083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Gee D.G., Bath K.G., Johnson C.M., Meyer H.C., Murty V.P., van den Bos W., et al. Neurocognitive development of motivated behavior: Dynamic changes across childhood and adolescence. J Neurosci. 2018;38:9433–9445. doi: 10.1523/JNEUROSCI.1674-18.2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Meyer H.C., Lee F.S. Translating developmental neuroscience to understand risk for psychiatric disorders. Am J Psychiatry. 2019;176:179–185. doi: 10.1176/appi.ajp.2019.19010091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Laviola G., Macrıì S., Morley-Fletcher S., Adriani W. Risk-taking behavior in adolescent mice: Psychobiological determinants and early epigenetic influence. Neurosci Biobehav Rev. 2003;27:19–31. doi: 10.1016/s0149-7634(03)00006-x. [DOI] [PubMed] [Google Scholar]
- 65.Kray J., Schmitt H., Lorenz C., Ferdinand N.K. The influence of different kinds of incentives on decision-making and cognitive control in adolescent development: A review of behavioral and neuroscientific studies. Front Psychol. 2018;9:768. doi: 10.3389/fpsyg.2018.00768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Shechner T., Britton J.C., Pérez-Edgar K., Bar-Haim Y., Ernst M., Fox N.A., et al. Attention biases, anxiety, and development: Toward or away from threats or rewards? Depress Anxiety. 2012;29:282–294. doi: 10.1002/da.20914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Waters A.M., Zimmer-Gembeck M.J., Craske M.G., Pine D.S., Bradley B.P., Mogg K. Look for good and never give up: A novel attention training treatment for childhood anxiety disorders. Behav Res Ther. 2015;73:111–123. doi: 10.1016/j.brat.2015.08.005. [DOI] [PubMed] [Google Scholar]
- 68.Dunsmoor J.E., Campese V.D., Ceceli A.O., LeDoux J.E., Phelps E.A. Novelty-facilitated extinction: Providing a novel outcome in place of an expected threat diminishes recovery of defensive responses. Biol Psychiatry. 2015;78:203–209. doi: 10.1016/j.biopsych.2014.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Chein J., Albert D., O’Brien L., Uckert K., Steinberg L. Peers increase adolescent risk taking by enhancing activity in the brain’s reward circuitry. Dev Sci. 2011;14:F1–F10. doi: 10.1111/j.1467-7687.2010.01035.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Nelson E.E., Jarcho J.M., Guyer A.E. Social re-orientation and brain development: An expanded and updated view. Dev Cogn Neurosci. 2016;17:118–127. doi: 10.1016/j.dcn.2015.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Rosen D., Price R.B., Ladouceur C.D., Siegle G.J., Hutchinson E., Nelson E.E., et al. Attention to peer feedback through the eyes of adolescents with a history of anxiety and healthy adolescents. Child Psychiatry Hum Dev. 2019;50:894–906. doi: 10.1007/s10578-019-00891-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Steinberg L. A social neuroscience perspective on adolescent risk-taking. Dev Rev. 2008;28:78–106. doi: 10.1016/j.dr.2007.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Silverman W.K., Kurtines W.M., Ginsburg G.S., Weems C.F., Lumpkin P.W., Carmichael D.H. Treating anxiety disorders in children with group cognitive-behaviorial therapy: A randomized clinical trial. J Consult Clin Psychol. 1999;67:995–1003. doi: 10.1037//0022-006x.67.6.995. [DOI] [PubMed] [Google Scholar]
- 74.Wolgensinger L. Cognitive behavioral group therapy for anxiety: Recent developments. Dialogues Clin Neurosci. 2015;17:347–351. doi: 10.31887/DCNS.2015.17.3/lwolgensinger. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Zhou X., Zhang Y., Furukawa T.A., Cuijpers P., Pu J., Weisz J.R., et al. Different types and acceptability of psychotherapies for acute anxiety disorders in children and adolescents. JAMA Psychiatry. 2019;76:41–50. doi: 10.1001/jamapsychiatry.2018.3070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Quarmley M.E., Nelson B.D., Clarkson T., White L.K., Jarcho J.M. I knew you weren’t going to like me! Neural response to accurately predicting rejection is associated with anxiety and depression. Front Behav Neurosci. 2019;13:219. doi: 10.3389/fnbeh.2019.00219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Guassi Moreira J.F., Telzer E.H. Mother still knows best: Maternal influence uniquely modulates adolescent reward sensitivity during risk taking. Dev Sci. 2018;21:12484. doi: 10.1111/desc.12484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Conner O.L., Siegle G.J., McFarland A.M., Silk J.S., Ladouceur C.D., Dahl R.E., et al. Mom—It helps when you’re right here! Attenuation of neural stress markers in anxious youths whose caregivers are present during fMRI. PLoS One. 2012;7 doi: 10.1371/journal.pone.0050680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Zee K.S., Bolger N. Visible and invisible social support: How, why, and when. Curr Dir Psychol Sci. 2019;28:314–320. [Google Scholar]
- 80.Gunnar M.R. Social buffering of stress in development: A career perspective. Perspect Psychol Sci. 2017;12:355–373. doi: 10.1177/1745691616680612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Hornstein E.A., Haltom K.E.B., Shirole K., Eisenberger N.I. A unique safety signal: Social-support figures enhance rather than protect from fear extinction. Clin Psychol Sci. 2017;6:407–415. [Google Scholar]
- 82.Robinson-Drummer P.A., Opendak M., Blomkvist A., Chan S., Tan S., Delmer C., et al. Infant trauma alters social buffering of threat learning: Emerging role of prefrontal cortex in preadolescence. Front Behav Neurosci. 2019;13:132. doi: 10.3389/fnbeh.2019.00132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Blackford J.U., Pine D.S. Neural substrates of childhood anxiety disorders a review of neuroimaging findings. Child Adolesc Psychiatr Clin N Am. 2012;21:501–525. doi: 10.1016/j.chc.2012.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Zugman A., Winkler A.M., Pine D.S. Recent advances in understanding neural correlates of anxiety disorders in children and adolescents. Curr Opin Psychiatry. 2021;34:617–623. doi: 10.1097/YCO.0000000000000743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Xu J., Van Dam N.T., Feng C., Luo Y., Ai H., Gu R., et al. Anxious brain networks: A coordinate-based activation likelihood estimation meta-analysis of resting-state functional connectivity studies in anxiety. Neurosci Biobehav Rev. 2019;96:21–30. doi: 10.1016/j.neubiorev.2018.11.005. [DOI] [PubMed] [Google Scholar]
- 86.Baker A.E., Galván A. Threat or thrill? the neural mechanisms underlying the development of anxiety and risk taking in adolescence. Dev Cogn Neurosci. 2020;45 doi: 10.1016/j.dcn.2020.100841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Casey B., Galván A., Somerville L.H. Beyond simple models of adolescence to an integrated circuit-based account: A commentary. Dev Cogn Neurosci. 2015;17:128–130. doi: 10.1016/j.dcn.2015.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Geng H., Li X., Chen J., Li X., Gu R. Decreased intra- and inter-salience network functional connectivity is related to trait anxiety in adolescents. Front Behav Neurosci. 2016;9:350. doi: 10.3389/fnbeh.2015.00350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Swartz J., Monk C. The role of corticolimbic circuitry in the development of anxiety disorders in children and adolescents. Curr Top Behav Neurosci. 2013;16:133–148. doi: 10.1007/7854_2013_242. [DOI] [PubMed] [Google Scholar]
- 90.Swartz J.R., Phan K.L., Angstadt M., Fitzgerald K.D., Monk C.S. Dynamic changes in amygdala activation and functional connectivity in children and adolescents with anxiety disorders. Dev Psychopathol. 2014;26:1305–1319. doi: 10.1017/S0954579414001047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Britton J.C., Grillon C., Lissek S., Norcross M.A., Szuhany K.L., Chen G., et al. Response to learned threat: An FMRI study in adolescent and adult anxiety. Am J Psychiatry. 2013;170:1195–1204. doi: 10.1176/appi.ajp.2013.12050651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Guyer A.E., Nelson E.E., Perez-Edgar K., Hardin M.G., Roberson-Nay R., Monk C.S., et al. Striatal functional alteration in adolescents characterized by early childhood behavioral inhibition. J Neurosci. 2006;26:6399–6405. doi: 10.1523/JNEUROSCI.0666-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Lahat A., Benson B.E., Pine D.S., Fox N.A., Ernst M. Neural responses to reward in childhood: Relations to early behavioral inhibition and social anxiety. Soc Cogn Affect Neurosci. 2018;13:281–289. doi: 10.1093/scan/nsw122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Benson B.E., Guyer A.E., Nelson E.E., Pine D.S., Ernst M. Role of contingency in striatal response to incentive in adolescents with anxiety. Cogn Affect Behav Neurosci. 2015;15:155–168. doi: 10.3758/s13415-014-0307-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Helfinstein S.M., Benson B., Perez-Edgar K., Bar-Haim Y., Detloff A., Pine D.S., et al. Striatal responses to negative monetary outcomes differ between temperamentally inhibited and non-inhibited adolescents. Neuropsychologia. 2011;49:479–485. doi: 10.1016/j.neuropsychologia.2010.12.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Guyer A.E., Choate V.R., Detloff A., Benson B., Nelson E.E., Perez-Edgar K., et al. Striatal functional alteration during incentive anticipation in pediatric anxiety disorders. Am J Psychiatry. 2012;169:205–212. doi: 10.1176/appi.ajp.2011.11010006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Rudolph K.D. In: Handbook of Developmental Psychopathology. Lewis M., Rudolph K.D., editors. Springer; Boston: 2014. Puberty as a developmental context of risk for psychopathology; pp. 331–354. [Google Scholar]
- 98.Andersen S.L., Teicher M.H. Stress, sensitive periods and maturational events in adolescent depression. Trends Neurosci. 2008;31:183–191. doi: 10.1016/j.tins.2008.01.004. [DOI] [PubMed] [Google Scholar]
- 99.Sharp P.B., Eldar E. Computational models of anxiety: Nascent efforts and future directions. Curr Dir Psychol Sci. 2019;28:170–176. [Google Scholar]
- 100.Ressler K.J. Translating across circuits and genetics toward progress in fear- and anxiety-related disorders. Am J Psychiatry. 2020;177:214–222. doi: 10.1176/appi.ajp.2020.20010055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Ghandour R.M., Sherman L.J., Vladutiu C.J., Ali M.M., Lynch S.E., Bitsko R.H., et al. Prevalence and treatment of depression, anxiety, and conduct problems in US children. J Pediatr. 2019;206:256–267.e3. doi: 10.1016/j.jpeds.2018.09.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Juraska J.M., Willing J. Pubertal onset as a critical transition for neural development and cognition. Brain Res. 2017;1654:87–94. doi: 10.1016/j.brainres.2016.04.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Ladouceur C.D. Neural systems supporting cognitive-affective interactions in adolescence: The role of puberty and implications for affective disorders. Front Integr Neurosci. 2012;6:65. doi: 10.3389/fnint.2012.00065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Callaghan B.L., Richardson R. The effect of adverse rearing environments on persistent memories in young rats: Removing the brakes on infant fear memories. Transl Psychiatry. 2012;2:e138. doi: 10.1038/tp.2012.65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Fareri D.S., Gabard-Durnam L., Goff B., Flannery J., Gee D.G., Lumian D.S., et al. Altered ventral striatal–medial prefrontal cortex resting-state connectivity mediates adolescent social problems after early institutional care. Dev Psychopathol. 2017;29:1865–1876. doi: 10.1017/S0954579417001456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Lange I., Goossens L., Bakker J., Michielse S., van Winkel R., Lissek S., et al. Neurobehavioural mechanisms of threat generalization moderate the link between childhood maltreatment and psychopathology in emerging adulthood. J Psychiatry Neurosci. 2019;44:185–194. doi: 10.1503/jpn.180053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Olsavsky A.K., Telzer E.H., Shapiro M., Humphreys K.L., Flannery J., Goff B., et al. Indiscriminate amygdala response to mothers and strangers after early maternal deprivation. Biol Psychiatry. 2013;74:853–860. doi: 10.1016/j.biopsych.2013.05.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Opendak M., Robinson-Drummer P., Blomkvist A., Zanca R.M., Wood K., Jacobs L., et al. Neurobiology of maternal regulation of infant fear: The role of mesolimbic dopamine and its disruption by maltreatment. Neuropsychopharmacol. 2019;44:1247–1257. doi: 10.1038/s41386-019-0340-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Gunnar M.R., Hostinar C.E. The social buffering of the hypothalamic-pituitary-adrenocortical axis in humans: Developmental and experiential determinants. Soc Neurosci. 2015;10:479–488. doi: 10.1080/17470919.2015.1070747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Wang P.S., Angermeyer M., Borges G., Bruffaerts R., Tat Chiu W., De Girolamo G., et al. Delay and failure in treatment seeking after first onset of mental disorders in the World Health Organization’s World Mental Health Survey Initiative. World Psychiatry. 2007;6:177–185. [PMC free article] [PubMed] [Google Scholar]
- 111.Manchia M., Pisanu C., Squassina A., Carpiniello B. Challenges and future prospects of precision medicine in psychiatry. Pharmacogenomics Pers Med. 2020;13:127–140. doi: 10.2147/PGPM.S198225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Tomasi J., Lisoway A.J., Zai C.C., Harripaul R., Müller D.J., Zai G.C.M., et al. Towards precision medicine in generalized anxiety disorder: Review of genetics and pharmaco(epi)genetics. J Psychiatr Res. 2019;119:33–47. doi: 10.1016/j.jpsychires.2019.09.002. [DOI] [PubMed] [Google Scholar]