Abstract
Rosehips (Rosa spp., Rosaceae) are wild rose bushes with more than 100 species. Its fruits vary in colour and size, depending on the species, and are recognised for their nutritional characteristics. Ten samples of Rosa canina L. and Rosa rubiginosa L. fruits were collected at different geographical points from Southern Chile. Nutrients such as crude protein and minerals and functional properties such as phenolic compounds, ascorbic acid, and also antioxidant activities were evaluated by HPLC-DAD-ESI-MS/MS. The results revealed a high content of bioactive compounds, primarily ascorbic acid (6.0 to 8.2 mg g−1 fresh weight (FW)), flavonols (427.9 ± 0.4 μg g−1 FW) and antioxidant activity. We established a relationship between the antioxidant activity using Trolox equivalent antioxidant capacity (TEAC), cupric reducing antioxidant capacity (CUPRAC) and 2,2-diphenyl radical (DPPH) methods and the concentration of uncoloured compounds, such as flavonols and catechin. This antioxidant activity was primarily associated with the samples from Gorbea, Lonquimay, Loncoche, and Villarrica localities, and all of them were of the species Rosa rubiginosa L. The results here obtained represent novel information of rosehip fruits. In this sense, the reported information about compounds and antioxidant activities in rosehip fruits allowed us to continue new lines of research in relation to the potential formulation of new functional foods and also in the treatment and/or prevention of some diseases.
Keywords: anthocyanin, antioxidant activity, phenolic compounds, rosehip
1. Introduction
The functional food characteristics of berries have been widely studied due to their high content of phenolic compounds, including flavonoids, such as anthocyanins, flavonols, flavan-3-ols, phenolic acids and hydrolysable tannins [1]. These compounds, individually or in combination, are primarily responsible for the health benefits of berries and associated with their antioxidant properties [2]. Some of these constituents are bioaccessible and bioavailable and exert anticancer, antimicrobial and anti-inflammatory activities and other activities [3]. Numerous in vitro studies indicate that plant secondary metabolites may affect various processes in mammalian cells, including gene expression, apoptosis, low-density lipoprotein oxidation, intracellular signaling, P-glycoprotein activation and the modulation of enzymatic activities associated with carcinogen activation and detoxification [4]. There is a wide variety of species from various botanical families called berry (berries in plural), which contain high levels of bioactive compounds that provide multiple health benefits [5]. Rosehip (Rosa spp.) fruits have gained attention for their important role in the health and cosmetic industries. According to the INFOR newsletter, rosehip fruit is considered a non-timber forest product (NTFP) with an important use in the cosmetic, pharmaceutical and gastronomy industries. In addition, it is positioned as the main NTFP in Chile with 30.7% of total exports, with 4420 tons of rosehip fruit, reaching USD 25.1 million, exceeding the export records in 2020. The main buyer countries include Germany, United States and Sweden. In addition, vegetable oil (7.7%) and rosehip seeds (1.3%) are also exported with amounts that reach USD 6.3 million and USD 1 million, respectively [6]. The genus Rosa has its origins in Eastern Europe [7] and includes more than 100 species. Three Rosa species are present in Chile: R. rubiginosa L., R. canina L. and R. moschata Herrm. [8]. Rosehip plants are perennial shrubs with an average height of approximately 2–3 m and strong resistance to harsh environmental conditions, such as rocky, sloping sites, poor soils and limited water access [9]. Fruits are characterised by lengths between 14.0 and 28.8 mm, diameters between 13 and 20 mm and weights of 1.2 to 2.7 g. The pulp of the fruit contributes to 42.9% to 66.5% of the fruit weight, depending on the species [10]. Various minerals, such as phosphorus, potassium, calcium, magnesium, iron, zinc, copper and manganese, have been reported in rosehip fruit [11]. Rosehip fruits have the highest content of vitamin C among fruits and vegetables (935–1230 mg 100 g−1) [10]. The fruits also contain important levels of vitamins A, B1, B2, B6, D, E and K [11,12]. The organic profile includes malic, quinic, tartaric and citric acids [13]. Fruits have a total phenolic content between 290 and 1385 mg 100 g−1 [12,13,14,15], and catechin and procyanidin B2 are the main phenolic compounds [16]. Carotenoids, such as lycopene, β-cryptoxanthin, β-carotene, rubixanthin, gazaniaxanthin and zeaxanthin, have also been identified [17,18]. High antioxidant activity has been reported for the genus [19], which is associated with the presence of flavonoids, tannins, terpenoids, xantonoids and glycerol glycoside, but significant variability exists between rose species [20,21]. Previous studies compared different species of rosehip and showed similar qualitative profiles but important differences in the content of bioactive compounds in the fruit [20,22,23]. The highest polyphenol content was reported for Rosa canina [20]. In this sense, it is important to carry out a nutritional analysis which allows knowing information such as proximal composition (moisture, ashes, protein, fat and crude fiber) and mineral content, as well as functional analysis that allows us to determine antioxidant activity, compounds phenolics and vitamins [24].
Based on these findings and the high content of functional compounds in rosehip, we hypothesized that there would be a difference in the content and concentrations of bioactive compounds in rosehip fruits, which would be influenced by the species and the geographical area of collection. The present study determined the profiles and concentrations of bioactive compounds and antioxidant activities of two species of rosehip fruits.
2. Results
2.1. Protein and Nutrient Composition
The protein content (Table 1) in the rosehip fruits ranged from 0.69 to 1.10 mg g−1, without significant differences between species or locations. The magnesium contents (0.75 to 1.45 mg g−1) also showed no significant differences between the samples and collections evaluated. Significant differences were observed for calcium, phosphorus and potassium between locations, independent of the species collected (Table 1). For calcium, the highest content was in the sample from Carahue (18.13 ± 0.25 mg g−1), and the lowest value was found in the sample from Osorno (1.95 ± 0.00 mg g−1). For phosphorus, the highest value was detected in the sample from Lonquimay (6.04 ± 0.27 mg g−1), and the lowest concentration was detected in the sample from Gorbea (0.81 ± 0.02 mg g−1). For potassium, the highest value was observed in the sample collected in Melipeuco (8.75 ± 0.12 mg g−1), and the lowest content was found in the sample collected in Loncoche (2.93 ± 0.06 mg g−1).
Table 1.
Determination of protein by the Kjeldahl method, minerals (Mg, Ca and K) by atomic absorption spectrophotometry and phosphorus by a colorimetric method in rosehip fruits. Different letters in each sub-figure indicate the presence of statistically significant differences according to the Tukey’s multiple range test (p ≤ 0.05; n = 9).
| Location | Proteins (mg g−1) | Ca (mg g−1) | Mg (mg g−1) | P (mg g−1) | K (mg g−1) |
|---|---|---|---|---|---|
| Carahue | 0.70 ± 0.21 a | 18.13 ± 0.25 a | 0.78 ± 0.05 a | 0.90 ± 0.04 f | 4.55 ± 0.07 g |
| Gorbea | 1.01 ± 0.20 a | 4.87 ± 0.08 b | 0.79 ± 0.23 a | 0.81± 0.02 f | 6.97 ± 0.28 c |
| Icalma | 1.10 ± 0.33 a | 2.43 ± 0.01 f | 0.97 ± 0.35 a | 1.53 ± 0.15 e | 4.46 ± 0.10 g |
| Imperial | 0.77 ± 0.15 a | 2.93 ± 0.03 e | 0.81 ± 0.05 a | 2.11 ± 0.06 d | 4.92 ± 0.05 f |
| Loncoche | 0.69 ± 0.07 a | 2.99 ± 0.03 e | 0.75 ± 0.16 a | 0.82 ± 0.04 f | 2.93 ± 0.06 h |
| Lonquimay | 0.97 ± 0.20 a | 3.06 ± 0.03 e | 0.92 ± 0.12 a | 6.04 ± 0.27 a | 8.09 ± 0.08 b |
| Melipeuco | 0.99 ± 0.11 a | 3.68 ± 0.001 d | 1.45 ± 0.43 a | 4.60 ± 0.07 b | 8.75 ± 0.12 a |
| Osorno | 0.94 ± 0.27 a | 1.95 ± 0.00 g | 0.75 ± 0.04 a | 4.17 ± 0.08 c | 6.51 ± 0.11 d |
| Pitrufquén | 1.08 ± 0.19 a | 4.02 ± 0.01 c | 0.77 ± 0.53 a | 1.56 ± 0.03 e | 4.76 ± 0.17 g |
| Villarica | 1.08 ± 0.10 a | 2.46 ± 0.03 f | 0.76 ± 0.02 a | 1.97 ± 0.11 d | 6.02 ± 0.08 e |
2.2. Profiles and Concentrations of Phenolic Compounds and Ascorbic Acid in Rosehip
Phenolic compound identification was performed in rosehip fruits. Only nine phenolic compounds were tentatively identified according to their fragmentation patterns using mass spectrometry (Table 2). Of the identified compounds, one anthocyanin (cyanidin-3-glucoside), one flavan-3-ol (catechin), one hydroxycinnamic acid (galloylquinic acid) and six flavonols (primarily glycosyl derivatives of quercetin) were identified by their UV profiles and fragmentation patterns (Table 2). The identification of catechin and cyanidin-3-glucoside was confirmed via comparison to commercial standards.
Table 2.
Identification of phenolic compounds in rosehip fruits by HPLC-DAD-ESI-MS/MS.
| Peak Number | tR (min) | Abbreviation | Tentative Identification | [M]+ | [M-H]− | Product Ions | ٨max (nm) |
|---|---|---|---|---|---|---|---|
| 1 | 5.1 | CAT1 | Catechin | - | 289.1 | - | 279 |
| 2 | 10.1 | ANT1 | Cyanidin-3-glucoside | 449.1 | - | 287.1 | 516 |
| 3 | 15.3 | HCAD1 | Galloylquinic acid | - | 343.1 | 191.0; 205.0; 111.0 | 280 |
| 4 | 16.9 | FLAV1 | n.i | - | 449.1 | 269.1; 152.0 | 289 |
| 5 | 17.5 | FLAV2 | n.i | - | 433.1 | 271.1 | 278 |
| 6 | 18.5 | FLAV3 | n.i | - | 615.1 | 465.10; 301.0 | 355 |
| 7 | 19.0 | FLAV4 | Quercetin-hexoside | - | 461.1 | 300.0 | 353 |
| 8 | 19.8 | FLAV6 | Quercetin-glucuronide | - | 477.1 | 300.0 | 353 |
| 9 | 20.9 | FLAV7 | Quercetin-rhamnoside | - | 447.1 | 300.0 | 353 |
Note: n.i means no identified.
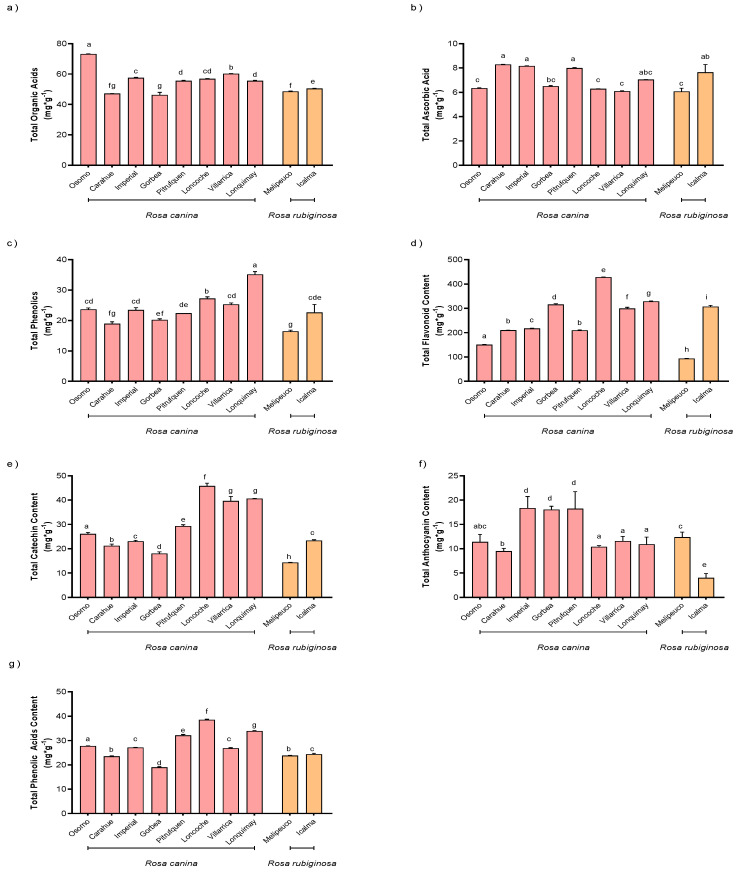
Individual phenolic compound concentrations were determined using HPLC-DAD (Table S1). The organic acid content ranged from 46.2 ± 1.66 mg g−1 to 73.2 ± 0.03 mg g−1 fresh weight (FW) (Figure 1a). The ascorbic acid concentration ranged from 6.0 to 8.2 mg g−1 FW. The lowest concentration was found in the samples collected in Melipeuco, and the highest concentration was found in the samples collected in Carahue (Figure 1b). Total phenolic content determination was consistent with the results of individual compounds. The lowest value was detected in the samples collected in Melipeuco, and the maximum value was found in the samples collected in Lonquimay (Figure 1c). The concentrations of flavonols were visibly higher and reached values of 93.6 ± 0.8 μg g−1 and 427.9 ± 0.4 μg g−1 FW in the samples collected in Melipeuco and Loncoche, respectively (Figure 1d). The contents of catechin were 14.3 ± 0.09 mg g−1 to 45.7 ± 1.15 mg g−1 FW for the samples collected in Melipeuco and Loncoche, respectively (Figure 1e). Total anthocyanin concentrations were 10.0 ± 0.0 μg g−1 and 40.1 ± 0.1 μg g−1 FW for the samples collected in Melipeuco and Pitrufquen, respectively (Figure 1f). Phenolic acid concentrations showed the same order of magnitude as anthocyanins and reached values up to 38.5 ± 0.2 mg g−1 FW in the samples collected in Loncoche (Figure 1g). A significant difference was observed between the two species collected, where R. canina had the lowest values compared to anthocyanins, flavonols, flavan-3-ols and total phenols. For the other compounds, no significant differences were observed between the two species.
Figure 1.
Contents in fruits of Rosa spp.: (a) organic acids; (b) ascorbic acid; (c) total phenolic; (d) flavonoid; (e) catechin; (f) anthocyanin; (g) phenolic acid content. Concentrations were expressed as fresh weight (FW). Different letters in each sub-figure indicate the presence of statistically significant differences according to the Tukey’s multiple range test (p ≤ 0.05; n = 9).
2.3. Colour Parameters
The present study evaluated the compositions of red, yellow, and blue colourations, colour intensity and tonality (Table 3). The highest percentage of yellow colouration was detected in the R. canina samples collected in Icalma and Melipeuco, with 78.9% ± 2.2% and 71.8% ± 7.2%, respectively. For Rosa rubiginosa, a higher percentage of red colouration was found for all of the samples, and the highest values were found in the samples collected in Pitrufquén (35.92% ± 2.62%) and Osorno (35.06% ± 1.33%). This observation is consistent with the clear difference in the appearance of these species. R. canina tended to exhibit yellow colouration, and R. rubiginosa tended to have a red colouration. The intensity of the colour of the samples ranged from 0.26 ± 0.40 to 0.97 ± 0.01, and that of the sample collected in Loncoche showed the highest value. Tonality values ranged from 0.96 ± 0.11 to 3.97 ± 0.49, and the sample collected in Imperial showed the highest tonality value.
Table 3.
Colour parameters including colour intensity, hue, yellow, red, blue, a, b, L, C and h, by CieLAB method in fruit of rosehip from different locations. Different letters in each sub-figure indicate the presence of statistically significant differences according to the Tukey’s multiple range test (p ≤ 0.05; n = 9).
| Location | Colour Intensity |
Tonality | % Yellow | % Red | % Blue | a | B | C | L | h |
|---|---|---|---|---|---|---|---|---|---|---|
| Rosa rubiginosa L. | ||||||||||
| Carahue | 0.72 ± 0.10 b | 1.47 ± 0.20 c | 51.27 ± 5.43 cd | 34.90 ± 1.07 b | 13.83 ± 4.58 bcd | −4410.03 ± 510.28 de | 16.63 ± 4.36 ab | 3.44 ± 2.12 b | 81.78 ± 2.36 b | 62.59 ± 12.00 b |
| Gorbea | 0.57 ± 0.06 b | 1.27 ± 0.17 c | 53.49 ± 3.72 cd | 42.49 ± 3.13 a | 4.02 ± 02.03 cde | −3655.55 ± 123.05 bcd | 15.54 ± 1.31 b | 10.91 ± 2.00 a | 83.95 ± 1.60 b | 52.84 ± 5.85 b |
| Imperial | 0.76 ± 0.7 b | 1.60 ± 0.17 bc | 46.47 ± 2.12 de | 29.12 ± 1.94 c | 24.41 ± 1.04 ab | −4277.55 ± 541.50 de | 12.26 ± 3.11 b | −5.90 ± 1.98 c | 82.21 ± 1.15 b | 56.92 ± 11.08 b |
| Loncoche | 0.97 ± 0.01 a | 1.28 ± 0.08 c | 42.45 ± 1.19 de | 33.15 ± 1.25 bc | 24.40 ± 0.73 ab | −5006.74 ± 208.24 e | 10.64 ± 1.70 b | −5.44 ± 1.33 c | 76.93 ± 0.09 c | 50.92 ± 6.09 b |
| Lonquimay | 0.73 ± 0.08 b | 1.41 ± 0.09 c | 46.65 ± 0.86 de | 33.30 ± 2.55 bc | 20.06 ± 3.16 abc | −4122.90 ± 423.66 bcd | 12.23 ± 1.31 b | −0.82 ± 3.35 bc | 81.97 ± 1.95 b | 52.39 ± 3.07 b |
| Osorno | 0.58 ± 0.12 b | 1.57 ± 0. 24 bc | 54.71 ± 6.28 cd | 35.06 ± 1.33 b | 10.22 ± 5.00 cde | −3787.05 ± 105.49 bcd | 16.02 ± 2.55 b | 4.95 ± 1.28 ab | 84.77 ± 1.74 b | 59.64 ± 7.14 b |
| Pitrufquen | 0.75 ± 0.03 b | 0.96 ± 0.11 c | 34.34 ± 1.30 de | 35.92 ± 2.62 b | 29.74 ± 1.45 a | −3119.37 ± 133.29 abc | −0.57 ± 1.15 c | −4.96 ± 2.01 c | 81.69 ± 1.03 b | 15.46 ± 5.37 c |
| Villarrica | 0.61 ± 0.12 b | 2.15 ± 0.25 b | 62.28 ± 5.89 bc | 28.99 ± 0.70 c | 8.73 ± 5.23 cde | −4521.25 ± 535.90 de | 22.91 ± 1.34 a | 2.89 ± 1.88 b | 84.73 ± 2.49 b | 83.21 ± 4.54 a |
| Rosa canina L. | ||||||||||
| Melipeuco | 0.34 ± 0.03 c | 3.97 ± 0.49 a | 71.78 ± 7.28 ab | 18.18 ± 2.29 d | 10.04 ± 8.36 cde | −2893.11 ± 206.27 ab | 16.53 ± 0.80 b | −0.21 ± 3.26 bc | 91.44 ± 0.87 a | 66.33 ± 1.98 ab |
| Icalma | 0.26 ± 0.4 c | 3.66 ± 0.08 a | 78.90 ± 2.20 a | 21.57 ± 1.09 d | −0.47 ± 3.29 e | −2485.68 ± 321.96 a | 15.71 ± 1.75 b | 3.40 ± 0.74 b | 92.58 ± 0.76 a | 60.70 ± 6.09 b |
2.4. Antioxidant Activity in Rosehip Fruits
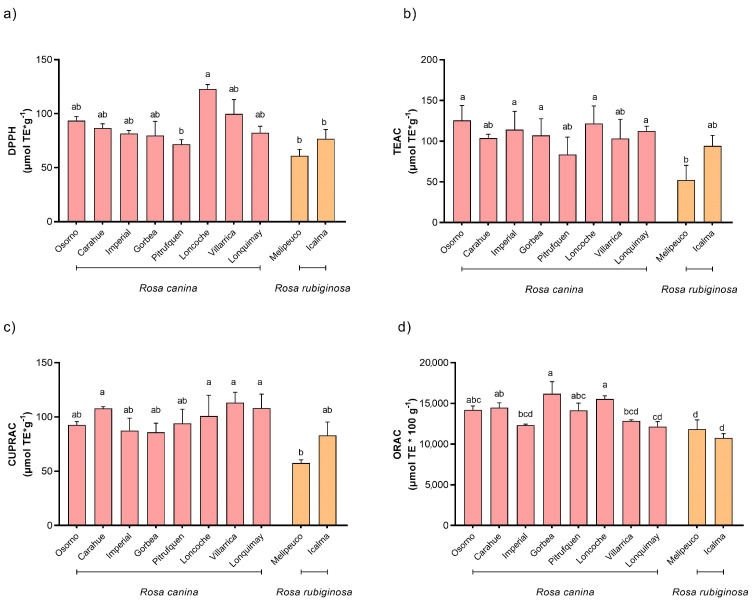
The antioxidant activity of rosehips was determined using the TEAC, DPPH, CUPRAC and ORAC methods. The DPPH method showed that the fruits collected in Loncoche had the highest levels of antioxidant activity, with concentrations of 122.8 ± 4.2 μmol g−1 (Figure 2a). The TEAC method showed that the sample with the highest value was collected in Osorno (125.5 ± 18.3 μmol g−1) (Figure 2b). The CUPRAC method found the highest antioxidant activity in the sample collected in Villarrica (113.3 ± 9.5 μmol g−1) (Figure 2c). Consistent with the quantitative composition of the fruit, the lowest values for the DPPH, TEAC and CUPRAC methods were found in the sample collected in Melipeuco. The values obtained from the ORAC method (Figure 2d) ranged from 10,776.8 ± 509.7 to 16,190.6 ± 1487.3 µmol 100 g−1, which corresponded to the samples collected in Icalma and Gorbea, respectively. The highest results in all of the methodologies used corresponded to those of R. rubiginosa samples, and the lowest values corresponded to those of R. canina samples.
Figure 2.
Antioxidant activity by spectrophotometric methods: (a) 2,2-diphenyl radical method (DPPH); (b) Trolox equivalent antioxidant capacity (TEAC); (c) cupric reducing antioxidant capacity (CUPRAC); (d) Oxygen Radical Antioxidant Capacity (ORAC). Concentrations were expressed as fresh weight (FW). Different letters in each sub-figure indicate the presence of statistically significant differences according to the Tukey’s multiple range test (p ≤ 0.05; n = 9).
3. Discussion
The mineral content detected was higher than those in previous reports for rosehips and other common fruits. The mineral contents of rosehip fruits have been reported for phosphorus (0.61 mg g−1), potassium (4.29 mg g−1), calcium (1.69 mg g−1), magnesium (0.69 mg g−1), iron (1.06 mg g−1), zinc (0.25 mg−1), copper (0.113 mg−1) and manganese (1.02 mg−1) [11]. Compared to other berries, the mineral content in rosehip is also higher. For example, raspberries have lower contents of calcium (0.25 mg g−1), phosphorus (0.29 mg g−1), potassium (0.15 mg mg−1) and magnesium (0.22 mg g−1) [25]. Blueberries contain various minerals, such as magnesium (1.5–3.3 mg 100 g−1), potassium (2.7–4.4 mg 100 g−1), phosphorus (6.0–13.3 mg 100 g−1) and calcium (0.9–2.2 mg 100 g−1) [26].
The (poly)phenolic composition of rosehip fruits has been reported. It been reported that cyanidin-3-glycoside is the only anthocyanin present in seedless extracts, and 21 other compounds, including glycosides of quercetin, taxifolin, eriodyctiol and the dihydrochalcone phoridzin, were also reported [27]. The reported phenolic compounds in crude extracts of R. canina fruits include glycosylated flavonols, flavonol aglycones, a stilbenoid, four flavan-3-ols and cyanidin-3-glycoside as anthocyanin. Fetni et al. [21] identified 46 phenolic compounds in R. canina rosehips from Slovenia, including cyanidin-3-glycoside as anthocyanin, flavan-3-ols and proanthocyanidins as the most abundant group of compounds, simple phenolic acids, flavanons, flavonols, flavanones and the dihydrochalcone phloridzin. Fetni et al. [21] recently described over 25 different compounds in R. canina fruits collected in Algeria, including O-glycosylated flavanones, O-methylated flavones, O-glycosylated flavonols and aglycones of flavanonols, but they did not report any anthocyanin.
The results for anthocyanins are consistent with the literature. Only one anthocyanin has been detected in rosehip fruits, cyanidin-3-glucoside (cy-3-glu) [20,28]. The content of cy-3-glu ranges from 9.2 to 125.7 µg g−1 expressed per dry weight (DW) [13]. The abundance and content of anthocyanins in rosehips are lower compared to those of other commonly consumed fruits [29]. The values of phenolic compounds obtained in the present research are significantly lower than in other studies, which reached the maximum of 118.56 mg g−1 [14]. Several phenolic acids and their derivatives have been identified in rosehips. The most abundant are gallic and ellagic acids [13], with concentrations between 0.18 mg g−1 and 2.44 mg g−1 DW, which are notably lower than in our study. Catechin is the main flavonoid in all Rosa spp. species. Our results are significantly higher than those reported by Elmastaş et al. [23], with values in the range of 0.225 mg g−1 to 0.472 mg g−1. Cunja et al. [22] reported values of total organic acids higher than 260 mg g−1 FW, and the higher contents of citric acid and malic acid are notable. Organic acids are fundamental to the texture and flavour of fruit [30].
Our values for the content of total phenols are comparable with those reported by other authors, where the highest and lowest levels of total phenolic compounds in rosehips were 52.94 mg g−1 and 31.08 mg g−1, respectively [17,20]. We found a high content of total phenols in rosehip fruit (67.84 mg g−1 DW), which was similar to the study of Murathan et al. [31]. It is important to consider that the Folin–Ciocalteu method is not specific for phenolic compounds, but also responds to other organic compounds, such as sugars, proteins or ascorbic acid, giving an overestimation of the phenolic content [32]. In addition, lipophilic and hydrophilic antioxidants, such as tocopherols, retinol, carotenoids, ascorbate and phenols [33], can also be identified by the Folin–Ciocalteau method.
Rosehip fruits are recognised for their high content of ascorbic acid, and our results are comparable to the literature. The ascorbic acid content in rosehip fruit ranges from 1.80 to 10.9 mg g−1 FW [19,20,34,35]. The fruit of the rosehip has a higher content of ascorbic acid compared to other fruits, such as oranges (0.44 mg g−1) [36], blueberries (0.73 mg g−1) [37,38] and strawberries (0.56 mg g−1) [39].
Colour is an important attribute of fruits that affects the consumer perception of attractiveness and quality. Phenolic compounds, carotenoids, chlorophylls and betalains normally contribute to the food colour characteristics of fruits [40]. Rosehip fruits vary in colouration, and fruits present with yellow, red and orange pigmentation, which is primarily determined by the genotype and its state of maturity [13]. The orange to red colour of rose hips is caused by their carotenoid composition, primarily β-carotene and lycopene [41]. The presence of anthocyanins in flowers, fruits and vegetables is associated with their orange, red and blue colours [42]. Fascella et al. [20] and Cunja et al. [13] observed significant differences in colour parameters between wild and cultivated rosehip fruits.
Analyses of the antioxidant capacity of a plant or fruit are essential from a nutraceutical perspective, and rosehip fruits are valuable sources of bioactive compounds, including antioxidants [20]. Some studies reported that the antioxidant activity of extracts correlated with total, rather than individual, phenolic compounds of rosehip fruit [43]. This correlation is consistent with our results, where the antioxidant activities determined by DPPH TEAC and CUPRAC were directly related to total phenols and primarily represented by flavonols and flavan-3-ols. Our results of the antioxidant activity of rosehips are consistent with the literature. Roman et al. [19] found that the antioxidant activity of rosehip extracts measured using the DPPH method yielded values of 63.35 μM Trolox 100 g−1. Another study used an ORAC assay to determine the antioxidant capacity of rosehip extracts and found that it ranged between 2727 and 4577 μmol TE g−1 DW [20]. Compared to other fruits, rosehips exhibit higher antioxidant capacity. On the other hand, a content of 30 ± 4 mmol TE 100 g −1 has been reported in ripe rosehip fruit according to the ORAC method, and a content of 9.1 ± 1.0 mmol TE 100 g−1 was measured using the TEAC method [35]. The antioxidant activity of raspberries (Rubus idaeus L.) has been reported as 29.0 μmol TE g−1 DW using the DPPH method [44]. Values from 50.8 to 99.5 μmol g−1 were determined in calafate (Berberis microphylla) using the TEAC method [45]. Values ranged from 2627 to 6747 μmol 100 g−1 DW in blueberry (Vaccinium spp), when the analysis was ORAC [46]. Oyarzún et al. [47] determined the antioxidant activity of chaura (Gaultheria sp.) fruits using the TEAC, DPPH and CUPRAC methods and obtained the maximum results of 18.72 μmol g−1, 10.22 μmol g−1 and 113.41 μmol g−1, respectively.
Fascella et al. [20] evaluated phenolic compounds and antioxidant activities in four species (R. canina, Rosa corymbifera, Rosa micrantha and Rosa sempervirens). However, a difference may be established from our results due to the species studied, because R. canina and R. sempervirens showed the highest antioxidant activities and the highest amounts of bioactive compounds. High antioxidant activity and a high content of phenolic compounds, especially flavonols, have been determined. This compound is highly important, because its biological effects are associated with its antioxidant activity related to its chemical structure [48]. Antioxidant activity is an important factor in inhibiting or delaying the oxidation of susceptible cell substrates [46]. Anti-inflammatory properties have also been reported in rosehip fruit [49]. Therefore, it is possible to use the bioactive compounds present in rosehip fruit as potential functional compounds for food processing.
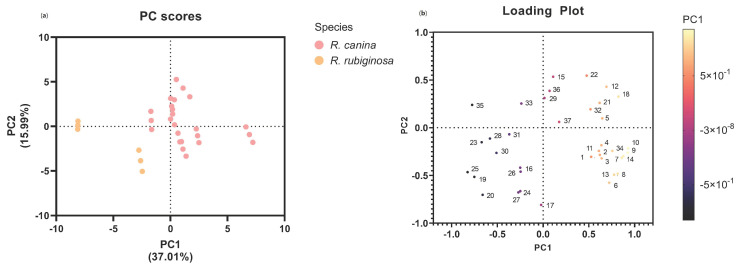
The principal component analysis (PCA) of the studied variables in the present study showed the formation of homogeneous groups of experimental variables. PC1 explained 36.2% of the total experimental variance, and PC2 explained 15.3% of the total experimental variance (Figure 3). The PCA showed a relationship between the antioxidant activity determined by DPPH, TEAC, CUPRAC and the concentrations of uncoloured compounds, such as flavonols and catechin, which were reported for other fruits [47]. This antioxidant activity was primarily associated with the samples from Gorbea, Lonquimay, Loncoche and Villarrica and all of the species R. rubiginosa. The antioxidant activity determined by ORAC was related to the presence of anthocyanins, which provided the colour intensity of primarily red.
Figure 3.
Principal component analysis (PCA) results: (a) the score plot generated by the two principal components indicating the difference between species; (b) the loading plot showing the variables that contribute to each one of the two principal components. The nomenclature of the varibles are as follows: (1) total phenolic content; (2) TEAC; (3) DPPH; (4) CUPRAC; (5) hydroxycinnamic acid 1; (6) flavonol 1; (7) flavonol 2; (8) flavonol 3; (9) flavonol 4; (10) flavonol 6; (11) flavonol 7; (12) anthocyanin 1; (13) total flavonoid content; (14) catechin; (15) total anthocyanins; (16) total protein content; (17) nitrogen content; (18) intensity of colour; (19) tone; (20) %yellow; (21)%red; (22)%blue; (23) a parameter; (24) b parameter; (25) L parameter; (26) C parameter; (27) h parameter; (28) potassium content; (29) calcium content; (30) magnesium content; (31) phosphorus content; (32) ORAC; (33) organic acid 1; (34) organic acid 2; (35) organic acid 3; (36) ascorbic acid content; (37) total organic acids.
Finally, although rosehip fruits contain higher concentrations of bioactive compounds from the phenolic compound family and also ascorbic acid that possess an important antioxidant activity, there is scarce information about the benefits for human health in relation to their consumption. In this sense, it is necessary to carry out new studies about their potential beneficial properties. On the other hand, in Chile, rosehip fruits are not cultivated in large areas, limiting their consumption to local recollection.
4. Materials and Methods
4.1. Sample Collection
Rosehip fruit samples were collected in Southern Chile between April and June 2021 in 10 different locations: Carahue (Lat. 38°43′2.38″ S, long. 73°10′6.83″ W, alt. 38,334 m a.s.l.) Nueva Imperial (Lat. 38°42′37.95″ S, long. 73°1′13.28″ W, alt. 39,964 m a.s.l.), Pitrufquén (Lat. 39°0′8.399″ S, long. 72°45′41.91″ W, alt. 90,050 m a.s.l.), Gorbea (Lat. 39°8′20.20″ S, long. 72°38′11″ W, alt. 89. 412 m a.s.l.), Loncoche (Lat. 39°19′46.36″ S, long. 72°32′14.08″ W, alt. 100,528 m a.s.l.), Villarrica (Lat. 39°16′6.35″ S, long. 72°13′49.22″ W, alt. 224,976 m a.s.l.), Osorno (Lat. 40°33′10.9″ S, long. 73°12′18.3″ W, alt. 23,524 m a.s.l.), Lonquimay (Lat. 8°50′50″ S, long. 71°40′36″ W, alt. 1160,662 m a.s.l.), Melipeuco (Lat. 38°51′8.2″ S, long. 71°45′25.2″ W, alt. 912,664 m a.s.l.) and Icalma (Lat. 38°48′00″ S, long. 71°17′00″ W, alt. 1160 m a.s.l.). Carahue and Nueva Imperial are located on the coast, whereas Lonquimay, Melipeuco and Icalma are located in mountain ranges. The species collected in Melipeuco and Icalma were identified as Rosa canina L. and Rosa rubiginosa L. for the other collection areas (Figure S1). The samples were stored at −80 °C until processing.
4.2. Protein and Mineral Determinations
For the total protein determination, 1 g of sample was placed in a digestion tube. Kjeldahl catalyst and 10 mL of 95–98% sulfuric acid were added in the tube. Then, digestion was carried out at 400 °C for 30 min. Then, 50 mL of water, 25 mL of boric acid and 2–3 drops of a mixed indicator were added, and the solution was distilled for 10 min. Finally, the distillate was titled with HCl (0.31 N), until the solution turned green or violet. The crude protein content was determined using the Kjeldahl method based on the quantification of organic nitrogen [50].
Regarding mineral determinations, for extract preparation, 1 g of sample was dried at 70 °C. Then, it was maintained in a muffle for 4–8 h at 500 °C. One millilitre of water and 10 mL of HCl 2 M were added and heated until boiling. Finally, the residues were filtered and resuspended in water for the analyses. Potassium, calcium and magnesium contents were determined after acid digestion using atomic absorption spectrophotometry (AAS; Unicam SOLAAR, mod. 969, Cambridge, UK). The phosphorus content was determined using the ammonium molybdate reaction to form molybdene blue under acidic conditions, which was quantified at 655 nm [51].
4.3. Determination of Phenolic Compounds Using HPLC
The extraction and chromatographic separation processes were performed as previously described [52]. For the preparation of the extract, 1 g of fruit was homogenised with 2 mL of an extraction solvent (90% methanol, 9% water and 1% formic acid) and sonicated for 1 min at a 40% amplitude. Subsequently, it was shaken for 10 min. Then, it was centrifuged for 10 min, and the supernatant was removed and stored in the dark. High-performance liquid chromatography-diode array detection (HPLC-DAD) analyses were performed using an HPLC system (Shimadzu, Tokyo, Japan) equipped with a quaternary LC-20AT pump, a DGU-20A5R degassing unit, a CTO-20A oven, an SIL-20a autosampler and a UV-visible diode array spectrophotometer SPD-M20A. Instrument control and data collection were performed using Lab Solutions software (version 5.96) (Shimadzu, Duisburg, Germany). The chromatographic analyses were performed according to Ruiz et al. [53]. Identity assignments were performed using an HPLC-DAD system coupled to a 6545-quadrupole time-of-flight (Q-ToF) mass spectrometer (Agilent, Waldbronn, Germany). The control software used here was a Mass Hunter workstation (version B.06.11).
Phenolic compounds were quantified using external calibration at the maximum wavelength of each compound family (520 nm for anthocyanins, 360 nm for flavonols, 320 nm for hydroxycinnamic acid and 280 nm for flavan-3-ols) using cyanidin-3-glucoside, quercetin, chlorogenic acid and catechin as standards, respectively. Total phenolic content was estimated using the Folin–Ciocalteu method (Table S2) [52].
4.4. Determination of Organic Acids
Low-molecular-weight organic acids (LMWOAs) were determined in rosehip fruit using HPLC-DAD. Briefly, 0.5 g of fruit was homogenised in 5 mL of 0.2 mol L−1 CaCl2, shaken for 10 min and centrifuged for 10 min at 4000× g. Chromatographic analyses were performed using a C18 Symmetry Waters column (250 × 4.6 mm, 5 μm) with a Novapak Waters C18 precolumn (22 × 3.9 mm, 4 μm) set at 30 °C. The chromatographic run was performed for 15 min using phosphoric acid (0.2 N, pH 2.1) as the mobile phase at 1.0 mL min−1. Quantification was performed at 210 nm via external calibration using citric acid as a standard (Table S2) [52].
4.5. Ascorbic Acid Content
The ascorbic acid content was determined using HPLC-DAD. Briefly, 5 g of fresh fruit was crushed in 25 mL of 100 mg L−1 oxalic acid and subjected to ultrasound at a 20% amplitude for 45 s, followed by centrifugation for 20 min at 4000× g. The supernatant was filtered and injected into an HPLC-DAD system. Chromatographic analysis was performed using a Zorbax Eclipse Agilent C18 column (250 × 4.6 mm, 5 µm) and a Novapak Waters C18 precolumn (22 × 3.9 mm, 4 µm) at 40 °C. Liquid chromatography was performed using an isocratic method with 2% formic acid in water (A) and 2% formic acid in acetonitrile (B) as the mobile phase at 0.7 mL min−1 at 40 °C. Quantification was performed at 254 nm via external calibration [52].
4.6. Colour Parameters
Colour determinations were performed using the CieLAB method, in a 1 mm quartz cuvette in a Genesys 10s UV–Vis spectrophotometer (Thermo Scientific, Waltham, MA, USA) [54].
4.7. Determination of Antioxidant Activity
The antioxidant activity of rosehip fruits was determined using three colourimetric methods: Trolox equivalent antioxidant capacity (TEAC), cupric reducing antioxidant capacity (CUPRAC) and the 2,2-diphenyl radical (DPPH) method [52]. The fluorimetric method was performed to estimate the oxygen radical absorbance capacity (ORAC) as reported by Ou et al. [55]. Measurements were performed in a microplate reader (SYNERGY HTX, BioTek Instruments, Winooski, VT, USA).
4.8. Statistical Analysis
For all of the studied variables, a hierarchical ANOVA was performed, where the 2 rosehip species were nested into the 10 localities considered. The significance level was established at p ≤ 0.05. The means were compared by Tukey’s multiple range test. Datasets were also subjected to principal component analysis (PCA) and correlation analyses to establish the relationships between different variables.
5. Conclusions
The present results revealed a high content of bioactive compounds, such as ascorbic acid and phenolic compounds, and related antioxidant activities in rosehip fruits. We established a relationship between phenolic compounds and their antioxidant activities, and flavonols were the most abundant compounds in the rosehip fruit. Anthocyanins were responsible for colouration. We did not establish a marked difference in the bioactive compounds between the studied species, because a difference was observed only in the content of anthocyanins and antioxidant activity using the ORAC method. No trend was observed for the other analysed parameters between the species.
The higher concentrations of bioactive compounds and the higher levels of antioxidant activity are of great interest and could contribute to the potential development of new subproducts or functional foods or in preventive or therapeutic use in some disorders and pathologies. In this sense, it is necessary to carry out new studies where the aforementioned nutraceutical characteristics of the fruits would be in use.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/molecules28083544/s1, Table S1: Concentrations of individual phenolic compounds in fruit of Rosa spp. by HPLC-DAD and their antioxidant activities by spectrometry methods. Abbreviations: HCAD1—hydroxycinnamic acid; FLAV1—flavonol 1; FLAV2—flavonol 2; FLAV3—flavonol 3; FLAV4—flavonol 4; FLAV6—flavonol 6; FLAV7—flavonol 7; ANT1—anthocyanin all with the same unit of measurement (µg g−1). Table S2: Analytical parameters for HPLC and spectrophotometric methods. Abbreviations: DL—detection limit; QL—quantification limit; LR—linear range; CV%—coefficient of variation; TEAC—Trolox equivalent antioxidant capacity; CUPRAC—cupric reducing antioxidant capacity; DPPH—2,2-diphenyl radical methods; ORAC—oxygen radical absorbance capacity. Figure S1: (A) Rosa rubiginosa; (B) Rosa canina fruits.
Author Contributions
Conceptualization, A.R; methodology, F.P., G.T., S.V. and J.N.; software, P.C. and F.J.-A.; validation, F.P. and A.R.; formal analysis, P.C and A.R; investigation, F.P.; resources, A.R.; data curation, P.C.; writing—original draft preparation, F.P.; writing—review and editing, A.R.; visualisation, A.R.; supervision, A.R.; project administration, A.R.; funding acquisition, A.R. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Conflicts of Interest
The authors declare no conflict of interest.
Sample Availability
Samples of the compounds are available from the authors.
Funding Statement
The authors would like to thank ANID/FONDECYT Grants 1230587 and 1190585 (A. Ruiz) from Agencia Nacional de Investigación y Desarrollo, Chile, for financial support and National Doctoral Scholarship ANID/CONICYT 2074/2021 (F. Peña).
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Ruiz A., Bustamante L., Vergara C., Von Baer D., Hermosín-Gutiérrez I., Obando L., Mardones C. Hydroxycinnamic Acids and Flavonols in Native Edible Berries of South Patagonia. Food Chem. 2015;167:84–90. doi: 10.1016/j.foodchem.2014.06.052. [DOI] [PubMed] [Google Scholar]
- 2.Salo H.M., Nguyen N., Alakärppä E., Klavins L., Hykkerud A.L., Karppinen K., Jaakola L., Klavins M., Häggman H. Authentication of Berries and Berry-Based Food Products. Compr. Rev. Food Sci. Food Saf. 2021;20:5197–5225. doi: 10.1111/1541-4337.12811. [DOI] [PubMed] [Google Scholar]
- 3.Vega-Galvez A., Rodríguez A., Stucken K. Antioxidant, Functional Properties and Health-Promoting Potential of Native South American Berries: A Review. J. Sci. Food Agric. 2021;101:364–378. doi: 10.1002/jsfa.10621. [DOI] [PubMed] [Google Scholar]
- 4.Działo M., Mierziak J., Korzun U., Preisner M., Szopa J., Kulma A. The Potential of Plant Phenolics in Prevention and Therapy of Skin Disorders. Int. J. Mol. Sci. 2016;17:160. doi: 10.3390/ijms17020160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Fuentes L., Figueroa C.R., Valdenegro M., Vinet R. Patagonian Berries: Healthy Potential and the Path to Becoming Functional Foods. Foods. 2019;8:289. doi: 10.3390/foods8080289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Poblete P. Instituto Forestal 2022 Productos Forestales No Madereros. 2022. [(accessed on 13 April 2023)]. Available online: https://bibliotecadigital.infor.cl/bitstream/handle/20.500.12220/31364/31364.pdf?sequence=1&isAllowed=y.
- 7.Brasovan A. Ans magnesium content in brier (Rosa canina L.) fruits at the “campul lui neag” sterile coal dump (Hunedoara County, Romania) An. Univ. Din Oradea Fasc. Biol. 2011;18:5–9. [Google Scholar]
- 8.Ilyasoǧlu H. Characterization of Rosehip (Rosa canina L.) Seed and Seed Oil. Int. J. Food Prop. 2014;17:1591–1598. doi: 10.1080/10942912.2013.777075. [DOI] [Google Scholar]
- 9.Demir F., Özcan M. Chemical and Technological Properties of Rose (Rosa canina L.) Fruits Grown Wild in Turkey. J. Food Eng. 2001;47:333–336. doi: 10.1016/S0260-8774(00)00129-1. [DOI] [Google Scholar]
- 10.Soare R., Bonea D., Iancu P., Niculescu M. Biochemical and Technological Properties of Rosa canina L. Fruits from Spontaneous Flora of Oltenia, Romania. Bull. Univ. Agric. Sci. Vet. Med. Cluj-Napoca. Hortic. 2015;72:182–186. doi: 10.15835/buasvmcn-hort:10598. [DOI] [Google Scholar]
- 11.Fan C., Pacier C., Martirosyan D.M. Rose Hip (Rosa canina L): A Functional Food Perspective. Funct. Foods Health Dis. 2014;4:493–509. doi: 10.31989/ffhd.v4i12.159. [DOI] [Google Scholar]
- 12.Ercisli S. Chemical Composition of Fruits in Some Rose (Rosa spp.) Species. Food Chem. 2007;104:1379–1384. doi: 10.1016/j.foodchem.2007.01.053. [DOI] [Google Scholar]
- 13.Cunja V., Mikulic-Petkovsek M., Weber N., Jakopic J., Zupan A., Veberic R., Stampar F., Schmitzer V. Fresh from the Ornamental Garden: Hipsof Selected Rose Cultivars Rich in Phytonutrients. J. Food Sci. 2016;81:C369–C379. doi: 10.1111/1750-3841.13220. [DOI] [PubMed] [Google Scholar]
- 14.Kerasioti E., Apostolou A., Kafantaris I., Chronis K., Kokka E., Dimitriadou C., Tzanetou E.N., Priftis A., Koulocheri S.D., Haroutounian S.A., et al. Polyphenolic Composition of Rosa canina, Rosa sempervivens and Pyrocantha coccinea Extracts and Assessment of Their Antioxidant Activity in Human Endothelial Cells. Antioxidants. 2019;8:92. doi: 10.3390/antiox8040092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Koczka N., Stefanovits-Bányai É., Ombódi A. Total Polyphenol Content and Antioxidant Capacity of Rosehips of Some Rosa Species. Medicines. 2018;5:84. doi: 10.3390/medicines5030084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Fetni S., Bertella N., Ouahab A. LC–DAD/ESI–MS/MS Characterization of Phenolic Constituents in Rosa canina L. and Its Protective Effect in Cells. Biomed. Chromatogr. 2020;34:e4961. doi: 10.1002/bmc.4961. [DOI] [PubMed] [Google Scholar]
- 17.Demir N., Yildiz O., Alpaslan M., Hayaloglu A.A. Evaluation of Volatiles, Phenolic Compounds and Antioxidant Activities of Rose Hip (Rosa L.) Fruits in Turkey. LWT-Food Sci. Technol. 2014;57:126–133. doi: 10.1016/j.lwt.2013.12.038. [DOI] [Google Scholar]
- 18.Patel S. Rose Hips as Complementary and Alternative Medicine: Overview of the Present Status and Prospects. Med. J. Nutr. Metab. 2013;6:89–97. doi: 10.1007/s12349-012-0118-7. [DOI] [Google Scholar]
- 19.Roman I., Stǎnilǎ A., Stǎnilǎ S. Bioactive Compounds and Antioxidant Activity of Rosa canina L. Biotypes from Spontaneous Flora of Transylvania. Chem. Cent. J. 2013;7:73. doi: 10.1186/1752-153X-7-73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Fascella G., D’Angiolillo F., Mammano M.M., Amenta M., Romeo F.V., Rapisarda P., Ballistreri G. Bioactive Compounds and Antioxidant Activity of Four Rose Hip Species from Spontaneous Sicilian Flora. Food Chem. 2019;289:56–64. doi: 10.1016/j.foodchem.2019.02.127. [DOI] [PubMed] [Google Scholar]
- 21.Fetni S., Bertella N., Ouahab A., Martinez Zapater J.M., De Pascual-Teresa Fernandez S. Composition and Biological Activity of the Algerian Plant Rosa canina L. by HPLC-UV-MS. Arab. J. Chem. 2020;13:1105–1119. doi: 10.1016/j.arabjc.2017.09.013. [DOI] [Google Scholar]
- 22.Cunja V., Mikulic-Petkovsek M., Zupan A., Stampar F., Schmitzer V. Frost Decreases Content of Sugars, Ascorbic Acid and Some Quercetin Glycosides but Stimulates Selected Carotenes in Rosa canina Hips. J. Plant Physiol. 2015;178:55–63. doi: 10.1016/j.jplph.2015.01.014. [DOI] [PubMed] [Google Scholar]
- 23.Elmastaş M., Demir A., Genç N., Dölek Ü., Güneş M. Changes in Flavonoid and Phenolic Acid Contents in Some Rosa Species during Ripening. Food Chem. 2017;235:154–159. doi: 10.1016/j.foodchem.2017.05.004. [DOI] [PubMed] [Google Scholar]
- 24.Abedin M.J., Abdullah A.T.M., Satter M.A., Farzana T. Physical, Functional, Nutritional and Antioxidant Properties of Foxtail millet in Bangladesh. Heliyon. 2022;8:e11186. doi: 10.1016/j.heliyon.2022.e11186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Rao A.V., Snyder D.M. Raspberries and Human Health: A Review. J. Agric. Food Chem. 2010;58:3871–3883. doi: 10.1021/jf903484g. [DOI] [PubMed] [Google Scholar]
- 26.Karlsons A., Osvalde A., Čekstere G., Ponnale J. Research on the Mineral Composition of Cultivated and Wild Blueberries and Cranberries. Agron. Res. 2018;16:454–463. [Google Scholar]
- 27.Stănilă A., Diaconeasa Z., Roman I., Sima N., Măniuţiu D., Roman A., Sima R. Extraction and Characterization of Phenolic Compounds from Rose Hip (Rosa canina L.) Using Liquid Chromatography Coupled with Electrospray Ionization-Mass Spectrometry. Not. Bot. Horti Agrobot. Cluj-Napoca. 2015;43:349–354. doi: 10.15835/nbha43210028. [DOI] [Google Scholar]
- 28.Guimarães R., Barros L., Dueñas M., Carvalho A.M., Queiroz M.J.R.P., Santos-Buelga C., Ferreira I.C.F.R. Characterisation of Phenolic Compounds in Wild Fruits from Northeastern Portugal. Food Chem. 2013;141:3721–3730. doi: 10.1016/j.foodchem.2013.06.071. [DOI] [PubMed] [Google Scholar]
- 29.Wu X., Beecher G.R., Holden J.M., Haytowitz D.B., Gebhardt S.E., Prior R.L. Concentrations of Anthocyanins in Common Foods in the United States and Estimation of Normal Consumption. J. Agric. Food Chem. 2006;54:4069–4075. doi: 10.1021/jf060300l. [DOI] [PubMed] [Google Scholar]
- 30.Walker R.P., Famiani F. Organic Acids in Fruits: Metabolism, Functions and Contents. Hortic. Rev. (Am. Soc. Hortic. Sci) 2018;45:371–430. [Google Scholar]
- 31.Murathan Z.T., Zarifikhosroshahi M., Kafkas E., Sevindik E. Characterization of Bioactive Compounds in Rosehip Species from East Anatolia Region of Turkey. Ital. J. Food Sci. 2016;28:314–325. [Google Scholar]
- 32.Rebelo M.J., Rego R., Ferreira M., Oliveira M.C. Comparative Study of the Antioxidant Capacity and Polyphenol Content of Douro Wines by Chemical and Electrochemical Methods. Food Chem. 2013;141:566–573. doi: 10.1016/j.foodchem.2013.02.120. [DOI] [PubMed] [Google Scholar]
- 33.Niero G., Penasa M., Costa A., Currò S., Visentin G., Cassandro M., De Marchi M. Total Antioxidant Activity of Bovine Milk: Phenotypic Variation and Predictive Ability of Mid-Infrared Spectroscopy. Int. Dairy J. 2019;89:105–110. doi: 10.1016/j.idairyj.2018.08.014. [DOI] [Google Scholar]
- 34.Bhave A., Schulzova V., Chmelarova H., Mrnka L., Hajslova J. Assessment of Rosehips Based on the Content of Their Biologically Active Compounds. J. Food Drug Anal. 2017;25:681–690. doi: 10.1016/j.jfda.2016.12.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Al-Yafeai A., Bellstedt P., Böhm V. Bioactive Compounds and Antioxidant Capacity of Rosa rugosa Depending on Degree of Ripeness. Antioxidants. 2018;7:134. doi: 10.3390/antiox7100134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Fatin Najwa R., Azrina A. Comparison of Vitamin C Content in Citrus Fruits by Titration and High Performance Liquid Chromatography (HPLC) Methods. Int. Food Res. J. 2017;24:726–733. [Google Scholar]
- 37.De Souza V.R., Pereira P.A.P., Da Silva T.L.T., De Oliveira Lima L.C., Pio R., Queiroz F. Determination of the Bioactive Compounds, Antioxidant Activity and Chemical Composition of Brazilian Blackberry, Red Raspberry, Strawberry, Blueberry and Sweet Cherry Fruits. Food Chem. 2014;156:362–368. doi: 10.1016/j.foodchem.2014.01.125. [DOI] [PubMed] [Google Scholar]
- 38.Augusto T.R., Scheuermann Salinas E.S., Alencar S.M., D’Arce M.A.B.R., de Camargo A.C., de Souza Vieira T.M.F. Phenolic Compounds and Antioxidant Activity of Hydroalcoholic Extractsof Wild and Cultivated Murtilla (Ugni Molinae Turcz.) Food Sci. Technol. 2014;34:667–673. doi: 10.1590/1678-457X.6393. [DOI] [Google Scholar]
- 39.Nuñez Y., Pérez M., Uribe E., Vega-Gálvez A., Di Scala K. Osmotic Dehydration under High Hydrostatic Pressure: Effects on Antioxidant Activity, Total Phenolics Compounds, Vitamin C and Colour of Strawberry (Fragaria vesca) LWT-Food Sci. Technol. 2013;52:151–156. doi: 10.1016/j.lwt.2012.02.027. [DOI] [Google Scholar]
- 40.Sant’Anna V., Gurak P.D., Ferreira Marczak L.D., Tessaro I.C. Tracking Bioactive Compounds with Colour Changes in Foods—A Review. Dye. Pigment. 2013;98:601–608. doi: 10.1016/j.dyepig.2013.04.011. [DOI] [Google Scholar]
- 41.Andersson S.C., Rumpunen K., Johansson E., Olsson M.E. Carotenoid Content and Composition in Rose Hips (Rosa spp.) during Ripening, Determination of Suitable Maturity Marker and Implications for Health Promoting Food Products. Food Chem. 2011;128:689–696. doi: 10.1016/j.foodchem.2011.03.088. [DOI] [Google Scholar]
- 42.Dzhanfezova T., Barba-Espín G., Müller R., Joernsgaard B., Hegelund J.N., Madsen B., Larsen D.H., Martínez Vega M., Toldam-Andersen T.B. Anthocyanin Profile, Antioxidant Activity and Total Phenolic Content of a Strawberry (Fragaria × ananassa Duch) Genetic Resource Collection. Food Biosci. 2020;36:100620. doi: 10.1016/j.fbio.2020.100620. [DOI] [Google Scholar]
- 43.Wenzig E.M., Widowitz U., Kunert O., Chrubasik S., Bucar F., Knauder E., Bauer R. Phytochemical Composition and in Vitro Pharmacological Activity of Two Rose Hip (Rosa canina L.) Preparations. Phytomedicine. 2008;15:826–835. doi: 10.1016/j.phymed.2008.06.012. [DOI] [PubMed] [Google Scholar]
- 44.Mihailović N.R., Mihailović V.B., Ćirić A.R., Srećković N.Z., Cvijović M.R., Joksović L.G. Analysis of Wild Raspberries (Rubus idaeus L.): Optimization of the Ultrasonic-Assisted Extraction of Phenolics and a New Insight in Phenolics Bioaccessibility. Plant Foods Hum. Nutr. 2019;74:399–404. doi: 10.1007/s11130-019-00756-4. [DOI] [PubMed] [Google Scholar]
- 45.Ruiz A., Hermosín-Gutiérrez I., Mardones C., Vergara C., Herlitz E., Vega M., Dorau C., Winterhalter P., Von Baer D. Polyphenols and Antioxidant Activity of Calafate (Berberis microphylla) Fruits and Other Native Berries from Southern Chile. J. Agric. Food Chem. 2010;58:6081–6089. doi: 10.1021/jf100173x. [DOI] [PubMed] [Google Scholar]
- 46.Xu Y., Li L.Z., Cong Q., Wang W., Qi X.L., Peng Y., Song S.J. Bioactive Lignans and Flavones with in Vitro Antioxidant and Neuroprotective Properties from Rubus idaeus Rhizome. J. Funct. Foods. 2017;32:160–169. doi: 10.1016/j.jff.2017.02.022. [DOI] [Google Scholar]
- 47.Oyarzún P., Cornejo P., Gómez-Alonso S., Ruiz A. Influence of Profiles and Concentrations of Phenolic Compounds in the Coloration and Antioxidant Properties of Gaultheria poeppigii Fruits from Southern Chile. Plant Foods Hum. Nutr. 2020;75:532–539. doi: 10.1007/s11130-020-00843-x. [DOI] [PubMed] [Google Scholar]
- 48.Tai Z., Zhang F., Cai L., Shi J., Cao Q., Ding Z. Flavonol Glycosides of Pseudodrynaria Coronans and Their Antioxidant Activity. Chem. Nat. Compd. 2012;48:221–224. doi: 10.1007/s10600-012-0209-1. [DOI] [Google Scholar]
- 49.Winther K., Apel K., Thamsborg G. A Powder Made from Seeds and Shells of a Rose-Hip Subspecies (Rosa canina) Reduces Symptoms of Knee and Hip Osteoarthritis: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. Scand. J. Rheumatol. 2005;34:302–308. doi: 10.1080/03009740510018624. [DOI] [PubMed] [Google Scholar]
- 50.Sáez-Plaza P., García Asuero A., Martín J. An Annotation on the Kjeldahl Method. An. De La Real Acad. Nac. De Farm. 2019;85:14–19. [Google Scholar]
- 51.Kazaz S., Baydar H., Erbas S. Variations in Chemical Compositions of Rosa Damascena Mill, and Rosa canina L. Fruits. Czech J. Food Sci. 2009;27:178–184. doi: 10.17221/5/2009-CJFS. [DOI] [Google Scholar]
- 52.Parada J., Valenzuela T., Gómez F., Tereucán G., García S., Cornejo P., Winterhalter P., Ruiz A. Effect of Fertilization and Arbuscular Mycorrhizal Fungal Inoculation on Antioxidant Profiles and Activities in Fragaria ananassa Fruit. J. Sci. Food Agric. 2019;99:1397–1404. doi: 10.1002/jsfa.9316. [DOI] [PubMed] [Google Scholar]
- 53.Ruiz A., Sanhueza M., Gómez F., Tereucán G., Valenzuela T., García S., Cornejo P., Hermosín-Gutiérrez I. Changes in the Content of Anthocyanins, Flavonols, and Antioxidant Activity in Fragaria ananassa Var. Camarosa Fruits under Traditional and Organic Fertilization. J. Sci. Food Agric. 2019;99:2404–2410. doi: 10.1002/jsfa.9447. [DOI] [PubMed] [Google Scholar]
- 54.Prez-Magario S., Gonzalez-SanJose M.L. Prediction of Red and Rosé Wine CIELab Parameters from Simple Absorbance Measurements. J. Sci. Food Agric. 2002;82:1319–1324. doi: 10.1002/jsfa.1191. [DOI] [Google Scholar]
- 55.Ou B., Chang T., Huang D., Prior R.L. Determination of Total Antioxidant Capacity by Oxygen Radical Absorbance Capacity (ORAC) Using Fluorescein as the Fluorescence Probe: First Action 2012.23. J. AOAC Int. 2013;96:1372–1376. doi: 10.5740/jaoacint.13-175. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data presented in this study are available on request from the corresponding author.