Abstract
Rationale and Objective
The ‘PEER-HD’ multicenter study tests the effectiveness of peer mentorship to reduce hospitalizations in patients receiving maintenance hemodialysis. In this study, we describe the feasibility, efficacy, and acceptability of the mentor training program.
Study Design
Educational program evaluation including the following aspects: (1) description of training content, (2) quantitative analysis of feasibility and acceptability of the program, and (3) quantitative pre-post analysis of efficacy of the training to impart knowledge and self-efficacy.
Setting and Participants
Data were collected using baseline clinical and sociodemographic questionnaires from mentor participants enrolled in Bronx, NY, and Nashville, TN, themselves receiving maintenance hemodialysis.
Analytical Approach
The outcome variables were the following: (1) feasibility measured by training module attendance and completion, (2) efficacy of the program to impart knowledge and self-efficacy measured by kidney knowledge and self-efficacy surveys, and (3) acceptability as measured by an 11-item survey of trainer performance and module content.
Results
The PEER-HD training program included 4 2-hour modules that covered topics including dialysis-specific knowledge and mentorship skills. Of the 16 mentor participants, 14 completed the training program. There was complete attendance to all training modules, though some patients required flexibility in scheduling and format. Performance on posttraining quizzes was consistent with high knowledge (mean scores ranged from 82.0%-90.0% correct). Mean dialysis-specific knowledge scores trended higher post training than at baseline though this difference was not statistically significant (90.0% vs 78.1%; P = 0.1). No change in mean self-efficacy scores was demonstrated from before to after training, among mentor participants (P = 0.2). Program evaluation assessments of acceptability were favorable [mean of all patient scores (0-4) within each module ranged from 3.43-3.93].
Limitations
Small sample size.
Conclusions
The PEER-HD mentor training program required accommodation to patients’ schedules but was feasible. Participants rated the program favorably, and although the comparison of performance on knowledge assessments post- and pre-program showed uptake of knowledge, this was not statistically significant.
Index Words: Peer mentor, hemodialysis, adherence, hospitalization
Graphical abstract
Plain-Language Summary.
We describe the content and implementation of a training program delivered to patients who were selected to become peer mentors and who themselves receive maintenance hemodialysis. We were interested in training peer mentors on kidney disease knowledge, communication and mentorship skills, and on how best to impart self-efficacy. We learned that despite the burden of receiving dialysis, most mentor participants successfully underwent training and did well on knowledge testing. Role playing during training sessions was a valuable component for imparting mentorship and communication skills. We also learned that similar training programs should offer flexibility and redundancy for training session schedules and should carefully evaluate the health ability of patients to participate in training.
In the United States, patients receiving maintenance dialysis spend, on average, 14 days per year in the hospital and up to 30% of them are rehospitalized within 30 days of discharge.1 Up to 30% experience depression, and quality of life and self-efficacy scores are lower in these patients than in the general population.2,3 Mortality rate is 16.3% per person-year.4 Although some patients are able to navigate their dialysis care, others struggle with adherence to the assigned treatment schedule and use health care resources suboptimally. This is especially true of racial-ethnic minority patients who have higher dialysis treatment nonadherence and hospitalization rates compared with White patients.5,6
Peer mentorship is feasible among patients with kidney failure and has shown to improve adherence to dialysis, informed decision making, and satisfaction among persons receiving mentorship.7, 8, 9 Peer mentorship programs, such as the National Kidney Foundation of Michigan10 and the ‘P2P program’ funded by Centers for Medicare and Medicaid Services,7 pair selected patients with trained individuals who receive maintenance dialysis and who demonstrate success in self-managing dialysis care.11,12 Challenges include engaging potential mentees and providing consistent and structured training to mentors to improve performance mastery.13,14 There is also a paucity of data on the efficacy of these programs to reduce morbidity and mortality.14
Implementing a peer mentorship program requires identifying, training, and supervising peer mentors. Mentor training content that has been described2,10,15 includes dialysis-specific knowledge and instructions on how to provide informational and emotional support.16 However, the success of training in imparting knowledge or self-efficacy has not been previously described, nor has there been a robust evaluation of the feasibility and acceptability of training to peer mentors. We conducted a multicenter pragmatic randomized controlled trial to test the effectiveness of a novel peer mentorship program (PEER-HD) to reduce hospitalizations in an urban racial-ethnic minority patient population. The study protocol has been described in detail elsewhere and is registered with clinicaltrials.gov (NCT-03595748).17 The objective of this study was to assess the feasibility, efficacy, and acceptability of the mentor training program, and also to describe the content of training modules and evaluate our training approach.
Methods
We presented a description of the PEER-HD mentor training program structure and content, and performed evaluation of its feasibility, efficacy, and acceptability among mentor participants. We obtained the following institutional review board approval: Einstein institutional review board no.: 2017-8531 and Vanderbilt institutional review board no.: 200895.
Setting, Participants, and Procedures
Mentors were identified in the following 2 study sites: (1) Albert Einstein College of Medicine, Bronx, NY, and (2) Vanderbilt University Medical Center, Nashville, TN, both academic medical centers with affiliated hemodialysis facilities located in urban communities with a high percentage of racial-ethnic minority patients and residents. The number of mentors recruited was determined by the planned enrollment of patient participants at each site for the larger trial. Mentors were individuals who were referred by dialysis facility clinical staff based on their informal assessment of medical stability and adherence to the hemodialysis procedure. The potential mentor participants were then screened by study staff according to the following inclusion criteria: experience of >1 year with dialysis, no hospitalizations within 6 months before enrollment, a serum albumin level of >3.5 mg/dL, and fluency in English or in English and Spanish. The exclusion criteria were as follows: cognitive disability, active substance use, and a life expectancy of <6 months.17 Institutional review board approval and written participant consent were obtained for all data collection and analyses. The study protocols adhered to the standards set by the Declaration of Helsinki for Human Subjects Research. Mentor participants were financially compensated for participation in training ($20 per training module), and for each completed trial study visit ($50 each visit). The modules were planned to be completed within 8 weeks after which peer mentors were subsequently assigned to patient participants identified as their mentees over the course of the primary trial.
Mentor Training Structure
The training program for the PEER-HD mentors was initially designed to be completed in person over 4 weekly 2-hour sessions. Study principal investigators and coordinators at each site conducted trainings in English. Because of the potential conflict with hemodialysis treatment schedules, the sessions were held at varying schedules and repeated as needed throughout the week to accommodate the mentors. Optional refresher sessions were planned midway through the primary trial study period. Owing to the coronavirus disease 2019 pandemic, training in groups or in person was not permitted. Trainings were therefore conducted by webinar with a group or an individual participant.
Mentor Training Content
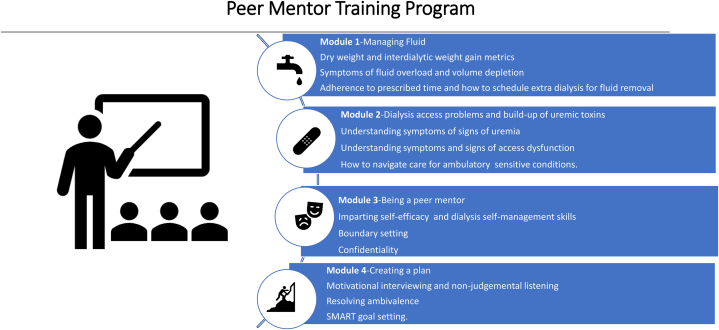
The training content was developed by the study investigators after a comprehensive literature review and consultation with peer mentorship experts. Modules were reviewed by 2 patients receiving hemodialysis, one dialysis facility social worker, and one dialysis facility nurse manager. Reviewers were compensated for their time. The content domains include the following: (1) fluid metrics; (2) dialysis access function and its association with uremic symptoms and susceptibility to infection; (3) being a peer mentor (ie, imparting knowledge and self-efficacy to assigned mentees); and (4) mastering motivational interviewing and ‘action planning,’18 including the use of the action planning SMART tool to help plan short-term goals and track achievement of those goals (Fig 1).19 The written documents of the training were not only provided in English but also translated and provided in Spanish to those mentor participants who preferred it.
Figure 1.
Description of peer mentor training program module content.
Data Collection and Key Measures
In addition to surveys collected during or immediately following the mentor training modules (1-4), surveys were collected from mentors during the primary trial at the enrollment (baseline) and 9-month (second) study visits.17 Validated surveys conducted at these 2 study visits focused on the participants’ kidney knowledge and kidney-related self-efficacy of self-care (Table 1). All data were collected by the study staff and stored within Albert Einstein College of Medicine’s REDCap database [Clinical and Translational Science Award grants UL1 TR002556 (Albert Einstein) and 5UL1TR002243-03 (Vanderbilt)].20
Table 1.
Assessments Before and After Training and the Study Domains They Inform
| Assessments | Attendance | Postmodule Training Quizzes | Module Evaluation | Summative Assessment | Kidney Knowledge (CHeKs and CHeKs Plus) |
Kidney Self-efficacy (PKDSMS) |
|---|---|---|---|---|---|---|
| Description of tools | Attendance | Developed by study team | Survey assessing trainer performance and satisfaction with module content | Developed by study team based on validated survey | CHeKs is validated; CHeKs23 Plus was developed by study team | Validated to measure kidney-related self-efficacy |
| Description of timing | % at each module, % completing all 4 modules, order of modules, and duration of completion of all 4 modules | After modules 1-3 | After each module | At completion of training (all modules) |
|
At baseline study visit and at 9 mo after enrollment |
| No. of questions | Module 1: 6 | 11 | 5 | CHeKs: 23 | 8 | |
| Module 2: 6 | CHeKs Plus: 12 | |||||
| Module 3: 5 | ||||||
| Domain informed | ||||||
| Feasibility | X | |||||
| Acceptability | X | X | ||||
| Efficacy | ||||||
| Imparting knowledge | X | X | ||||
| Imparting self-efficacy | X | |||||
Note: See Supplementary Materials for module 1-2 quizzes and the CHeKs Plus.
Abbreviations: CHeKs, Chronic Hemodialysis Knowledge Survey; CHeKs Plus, Chronic Hemodialysis Knowledge Survey Plus; PKDSMS, Perceived Kidney/Dialysis Self-Management Scale.
Sociodemographic Information
At the trial baseline study visit, which occurred before mentor training, the mentors completed questions that asked about their demographic, personal, medical, and kidney disease/hemodialysis history. They also reported on their socioeconomic status, responded to one question about food insecurity, referring to a sense of worry about not having enough food, and completed health literacy and health numeracy questionnaires. Health literacy refers to communication among providers and individuals to promote the successful application of health information to make informed health decisions.21 Health numeracy is an important component of literacy and reflects one’s ability to understand and use numbers in daily life.22 Mentor participants completed the Brief Health Literacy Scale.23 The Brief Health Literacy Scale scores range from 3-15, with higher scores corresponding to higher health literacy. Similarly, health numeracy was measured using Subject Numeracy Scale (SNS-3),24 in which total scores range from 3-18, with higher scores corresponding to higher health numeracy.
Key Measures
Feasibility
Feasibility was measured by the proportion of mentors that attended each scheduled training module and the proportion completing all 4 training modules. To consider the training program as feasible, we used a criterion of >50% attendance to all modules defined as mentors staying in the training program until its completion, and >80% attendance in any given module. Feasibility measures also included the order in which modules were attended and the duration of time taken between completed modules by the mentor participants. It was expected that all modules be completed within an 8-week period (Table 1).
Training Efficacy
We measured each mentor’s performance on training quizzes by determining the percentage of questions correctly answered, and then reported the mean of mentor scores. The first 3 modules of the training were followed by a brief, multiple-choice training quiz assessing the mentors’ knowledge of the information presented, immediately after that module was completed. These quizzes consisted mostly of items derived from the Chronic Hemodialysis Knowledge Survey (CHeKs) and CHeKs Plus and were scored based on the number of questions answered correctly and reported as a percentage. Quizzes for modules 1 and 2 had 6 items covering clinical hemodialysis concepts (Table S2). The module 3 quiz had 5 items and covered materials on how best to fill the role of a peer mentor.2,25 Mentor participants also rated themselves on their ability to carry out role-playing exercises during modules 3 and 4.25 The role-playing self-assessment consisted of 11 questions that asked about each mentor’s confidence in carrying out the tasks of a peer mentor. Each question used answer scales from 1-4, with 1 representing “Not at all confident” and 4 representing “Totally confident.” A score “Somewhat confident (3)” or higher was considered to reflect meeting training expectations. Although self-evaluation is subjective and limited in its ability to rate performance, self-rating conveys confidence in carrying out mentor tasks.
The CHeKs26 and the CHeKs Plus are surveys that measure a participant’s general knowledge about hemodialysis care. CHeKs is a valid 23-item multiple-choice survey. CHeKs Plus is an 8-item survey developed by the investigators and is aligned with the specific content of the program’s training modules. Items derived from the CHeKs Plus are included in module 1 and 2 quizzes, whereas module 2 also includes 2 items from the CHeKs (Table S1). A score of >70% was considered as achieving sufficient kidney knowledge. Self-efficacy was assessed with the validated Perceived Kidney/Dialysis Self-Management Scale,27 which is composed of 8 questions that ask about self-management skills pertaining to hemodialysis. Scores ranged from 1 (strongly disagree) to 5 (strongly agree), with higher scores indicating more confidence in self-management of one’s dialysis. These assessments were performed for mentors at their baseline study visit and 9-month study visit. Efficacy of the training program to impart knowledge and self-efficacy would be ascertained by comparing mentor scores on CHeKs and CHeKs Plus, and Perceived Kidney/Dialysis Self-Management Scale after as compared to before the training program.
Acceptability
Acceptability was assessed with an 11-item survey evaluating the performance of the trainers and satisfaction with the content of the modules.25 This survey was completed at the end of each module and was rated using a 4-point scale (1 = strongly disagree; 4 = strongly agree). An average rating of 3 (agree) or higher was considered acceptable. After all the training was completed, mentors were asked to complete a brief 4-item survey25 asking their perspectives on which topic was the most or least helpful, what tools or skills they planned to put into action, and suggestions for improvements to the training program.
Statistical Methods
STATA version 15.0 was used for all analyses. Demographics, medical history, and training module quiz results are presented using descriptive statistics. Simple proportions and summary statistics described attendance and completion of program as feasibility measures. Summary statistics of mean program-evaluation scores [standard deviation (SD)] and a description of responses on the 4-item survey are provided. To inform about the efficacy of the program to impart knowledge, simple statistics were used to ascertain the performance of the group on posttraining module quizzes and a paired t test was used to measure the change in mean scores (SD) from baseline to the 9-month trial visit, post training, on knowledge and self-efficacy surveys.
Results
A total of 24 potential mentors were approached and 16 enrolled in the study. At Albert Einstein College of Medicine, 14 mentors were approached and 10 (71%) were enrolled, with 1 not qualifying and 3 declining; at Vanderbilt, 9 mentors were approached and 3 declined, with 6 (67%) enrolled (Table S2). Participants’ median age in years was 47 (interquartile range, 37-53), and 56% were women. The majority (81%) identified as Black, African American, multiracial, or other. The latter was defined as unknown or preferred not to say (Table 2). Among those enrolled, 33% were of Hispanic descent. Only one of the mentors self-identified as both ethnically and racially White. The median health literacy score was high, with a slightly lower health numeracy score (Table 2). Despite the high educational achievement, 50% reported an annual income of less than $20,000 and 25% reported food insecurity.
Table 2.
Description of Mentor Participants
| Mentors, N=16 | |
|---|---|
| Age, y; median (IQR) | 47 (37-53) |
| Sex, n (%) | |
| Female | 9 (56.3) |
| Race, n (%) | |
| Black | 9 (56.3) |
| White | 3 (18.8) |
| Multiracial | 1 (6.25) |
| Other | 3 (18.8) |
| Ethnicity, n (%) | |
| Hispanic | 5 (33.3) |
| Non-Hispanic | 11 (66.7) |
| Married, n (%) | 4 (25) |
| Primary language, n (%) | |
| English | 14 (87.5) |
| Spanish (with some fluency in English) | 2 (12.5) |
| Highest educational achievement, n (%) | |
| Master’s degree | 2 (12.5) |
| Bachelor’s degree | 3 (18.8) |
| High school or GED | 11 (68.7) |
| Currently employed, n (%) | 2 (12.5) |
| On disability | 4 (25) |
| Unemployed | 6 (37.5) |
| Retired | 4 (25) |
| Type of insurance, n (%) | |
| Medicare | 13 (81.3) |
| Medicaid | 2 (12.5) |
| Commercial | 1 (6.3) |
| Annual income, $ (%) | |
| Annual income, n (%) | |
| 0-19,999 | 8 (50) |
| 20,000-39,999 | 2 (12.5) |
| 40,000-59,999 | 3 (18.8) |
| 60,000-99,999 | 1 (6.2) |
| >99,999 | 0 (0) |
| Do not know | 2 (12.5) |
| Report regularly worrying about not having enough money for food, n (%) | 4 (25) |
| Median Charlson score, median (IQR) | 4 (2-6.5) |
| Median health literacy score, median (IQR) | 15 (14-15) |
| Median health numeracy score, median (IQR) | 14.5 (12.5-16) |
| Etiology of ESKD (%) | |
| Diabetes | 6 (40) |
| Glomerulonephritis/lupus | 4 (26.7) |
| Hypertension | 2 (13.3) |
| Other | 3 (20) |
| Median no. of y receiving hemodialysis, median (IQR) | 7 (4.5-8) |
| Type of hemodialysis access, n (%) | |
| Arteriovenous fistula | 14 (87.5) |
| Arteriovenous graft | 2 (12.5) |
| Mean no. of medications (SD) | 10.7 (4.2) |
| No. reporting ≥1 hospitalization in the last year | 4 (25) |
| No. reporting ≥1 emergency department visit | 4 (25) |
Abbreviations: ESKD, end-stage kidney disease; GED, general educational diploma; IQR, interquartile range; SD, standard deviation.
Feasibility Regarding Attendance and Format of Training
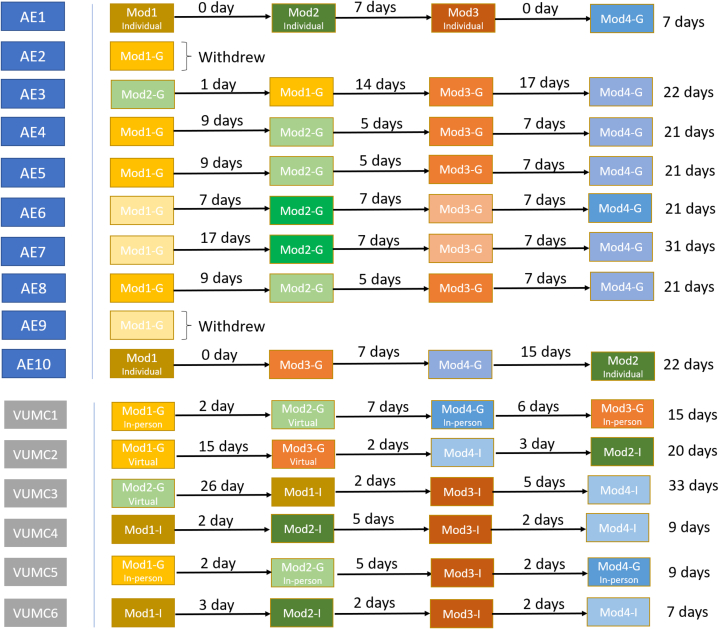
The overall attendance to the flexible and redundant schedule of training was acceptable. Two of the 16 mentors did not complete the full training. Both mentors discontinued involvement in the study after completing the first training module. Reasons for discontinuation were illness and burden on time imposed by the study activities. Five of the remaining 14 mentors did not complete modules 1-4 in order but were able to complete them out of order. The mean number of days between the modules was 6.2 and the mean number of days from the beginning of training to the end was 19.4 (Fig 2). General impressions recorded by the trainers noted a collaborative and enthusiastic atmosphere. Occasionally, one of the mentor participants would dominate the conversation, but they were successfully redirected with increased participation by others.
Figure 2.
Adherence to the training schedule at each site. Abbreviations: AE, Albert Einstein College of Medicine; G, group; I, individual; Mod, module; VUMC, Vanderbilt University Medical Center.
Efficacy of Knowledge Imparted as Measured by Module Training Quizzes
The mean number of questions answered correctly on module 1 training quiz was 90.0% (SD 10%; N=16); of the 14 mentors who completed the quiz for module 2, the mean number was 81.7% (SD 16.7%). Among the 14 mentors who completed module 3 quiz, the mean number of correct answers was 82.0% (SD 22%). The most missed question pertained to mentor skills identifying motivational interviewing and nonjudgmental listening as key to forming a relationship with the mentee. The mentor participants overall achieved threshold scores for each of the quizzes as described in the Methods section, suggesting feasibility and successful delivery of the content.
Efficacy of Training in Increasing Knowledge and Improving Self-efficacy
The mean scores on CHeKs at baseline and 9-month trial visit were 73.5% (SD 8.0%) and 72.7% (SD 9.9%), respectively (P = 0.6). There was a trend suggesting an increase in CHeKs-Plus scores between baseline (78.1%, SD 14.7%) and 9-month visit (90.0%, SD 14.9%), respectively (P = 0.1). Dialysis self-care self-efficacy did not differ significantly between visits [Perceived Kidney/Dialysis Self-Management Scale baseline median score was 37 (interquartile range, 33-39.5); the 9-month visit median score was 31.5 (interquartile range, 27-35); P = 0.2)].
Among the 14 mentor participants who completed the role-playing self-assessment for module 3, most strongly agreed (selected 4 from a scale of 1-4) with statements made about their skills except for one respondent who strongly disagreed with the statement about helping mentees take small steps toward goals (Table 3). Among the 14 mentors who completed the role-playing self-assessment for module 4, most also strongly agreed with the statements about their skills.
Table 3.
Rating ‘Strongly Agree’ Role-Playing Self-assessment Skills for Module 3 and Module 4
| Question, number of mentors/total (n=14) (%) | Module 3 | Module 4 |
|---|---|---|
|
13 (93) | 14 (100) |
|
12 (86) | 11 (79) |
|
11 (79) | 13 (93) |
|
13 (93) | 14 (100) |
|
9 (64) | 12 (86) |
|
11 (79) | 14 (100) |
|
13 (93) | 12 (86) |
|
13 (93) | 14 (100) |
|
13 (93) | 14 (100) |
|
12 (86) | 12 (86) |
|
13 (93) | 14 (100) |
Acceptability of Training to Mentor Participants
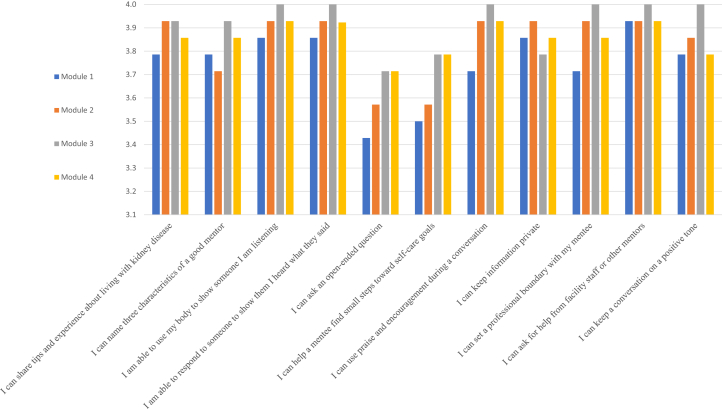
Of the mentors who completed program evaluations for all modules (n=4), 87.9% of participants strongly agreed (scored as 4) that the trainers were well prepared, 81% strongly agreed that the handouts were easy to read, and 91.4% strongly agreed that the information was useful; however, 67.2 % strongly agreed that there was enough time and opportunity to practice new skills. Furthermore, 86.2% agreed that trainers answered all questions, 86.2% agreed that the training was well organized, 87.9% agreed that they were scheduled at a convenient time, and 93.1% agreed that the training kept their interest, whereas 86.2% thought that the amount covered was appropriate (Fig 3). There was variation by module, with didactic material receiving lower ratings.
Figure 3.
Module evaluation responses by participants for modules 1-4. Scores range from 1 (strongly disagree) to 4 (strongly agree).
All mentors who completed the training (N=14) also answered a brief 4-item open-ended survey about the training program (Table 4). When asked what the most helpful tool in the training program was, most answered enhancement of communication skills, whereas the least helpful topic covered seemed to be redundancy of the dialysis-specific knowledge. Participants referred to specific topics such as dietary restrictions, transplant or communication with staff, and skills on how best to cope with dialysis when asked what was missed in the training. Most mentors answered that they would use communication skills and their own personal experience, and some mentioned they would use the educational material that was given during the training, action planning and other publicly available resources (National Kidney Foundation website), or a combination (21% of respondents), in the future.
Table 4.
Mentor Open-Ended Program-Evaluation Summary
| Questions (N=14) | Reponses | Comments by mentors |
|---|---|---|
| What would you say was the most helpful topic covered in the Peer-HD mentor training? | 64%, enhancement of communication skills; 50%, everything was helpful |
|
| What would you say was the least helpful topic covered in the Peer-HD mentor training? | Redundancy of dialysis-specific knowledge |
|
| What additional training topics should be covered in Peer-HD mentor training? | 28%, everything was covered; 21%, dietary restrictions, transplant or communication with staff; 21%, how to cope with dialysis |
|
| Which tools, materials, or resources offered do you plan to use as a mentor? | Communications skills, action planning, personal experience, and training materials |
|
| How can we improve the Peer-HD mentor training? | 57%, program well-structured and to keep the current format; more role-playing and refresher courses |
Discussion
The PEER-HD mentor training program overall was feasible and acceptable. Feasibility was demonstrated by the high proportion of those approached recruited, the high rate of attendance by mentors at scheduled training modules, and the completion of the training objectives despite disruptions introduced by the coronavirus disease 2019 pandemic. Excellent performance on postmodule quizzes demonstrated an understanding of the training content and high scores on role-playing self-assessment demonstrated perception by mentors of their ability to implement the mentor role in the trial. Favorable scoring of program-evaluation assessments for each module and favorable responses on the brief open-ended program survey were consistent with the acceptability of the training program. The performance of the mentors on health literacy, with a mean score of 15 in our study as compared with 11 and 12 in other cohorts of patients receiving in-center hemodialysis,23,28 was in part reflective of their high educational attainment, with 25% having completed a bachelor’s degree or higher (Table 2). Health numeracy was similarly higher in our mentor patients, with a mean score of 14.5 as compared with 11.0 reported for a general cohort of patients receiving maintenance hemodialysis.24 The mean score on the CHeKs survey tool, an instrument validated to evaluate patient knowledge on important kidney disease issues, was higher among our mentor patients (mean 73.5%) as compared with a larger cohort of patients receiving maintenance dialysis (65%).26 These high-baseline literacy and knowledge scores reflect oversampling of patients who may have been able to put information they received from their health care providers into action and successfully navigated life on hemodialysis. These characteristics among mentors in this study were similar to the scores reported for mentors selected for another peer mentorship study.2
Various models of peer mentorship have been used, including national programs (National Kidney Foundation Peers and the The Kidney Foundation of Canada’s Kidney Connect program16) and local programs linking individuals within a dialysis facility to each other. 7,10,14 These programs provide training on disease knowledge, and methods for informational and emotional support, but the training programs themselves have not been evaluated in detail nor were they as comprehensive in imparting knowledge and communication skills as the PEER-HD mentor training program.2,29 Moreover, our training program uniquely instructed mentors on how best to use their dialysis facility resources to manage their fluid, access, adequacy, and minor infection care (eg, upper respiratory tract infection). In this way, the training aimed to empower and equip patients as coached by their mentors to coordinate a more efficient use of resources to avoid hospitalizations.30
Racial-ethnic minorities are underrepresented among participants in kidney disease peer programs, highlighting the importance of engaging individuals of diverse backgrounds in peer support programs to better inform effective strategies.10,31 This is critical given the overrepresentation of racial-ethnic minorities among the dialysis population,32 and their well-documented greater risk of dialysis treatment nonadherence and hospitalization.3,6 It has been further hypothesized that peer programs, which use mentor/mentee pairs with similar racial-ethnic and socioeconomic backgrounds, perform better than those who do not emphasize these matching criteria.31,33 Though a notable limitation is that our program training content did not go through a formal process to address cultural sensitivity, the racial-ethnic makeup of the mentor participants was intentionally diverse to promote collaboration during the trial through pairs that shared identity characteristics. Demonstrating the likely success of training in increasing knowledge and improving mentorship skills in a purposively sampled patient population is important.16,31,33 Certainly, creating opportunities for patients as strategic partners in the success of a program promotes its acceptability and uptake, especially in patient populations that have been historically marginalized. We included patients in the development of the training, and training feedback by mentors serves as a valuable resource for future iterations of this content.31,33
The execution of a training program across 2 study sites included logistical challenges such as the restrictive schedule imposed by hemodialysis treatments and the daily health challenges faced by patients with kidney failure. We overcame some of these challenges by offering schedules that both accommodated in-center dialysis schedules and allowed ample opportunity for makeup sessions with both in-person and virtual instruction. With the onset of the coronavirus disease 2019 pandemic midway through the study period, we immediately switched all activities to virtual training modules. Although these accommodations ensured maximal participation, 2 of the 16 (12.5%) participants did not complete the training because of illness and feeling burdened by the training program. Flexibility of structure, format, and delivery is an important feature of any peer program designed for patients with kidney disease as described by peer support participants.17,31,33
As noted above, a major limitation of our descriptive and comparative analyses was the small study sample. The training was not offered in Spanish, limiting our ability to enroll Spanish only-speaking mentor participants. We did not examine the effect of training on performance during actual mentoring sessions. Participant bias may be present in some responses to the survey tools as the lead trainer at each site was a physician, which may have led to social desirability bias. The future design of peer mentorship programs should include more content on coping skills, objective assessment of role playing, and a more comprehensive screening on the health stability of candidates. Finally, the CHeKs-Plus kidney knowledge survey had not yet undergone rigorous validation analyses; however, its structure and content were modeled after other valid and established knowledge surveys increasing likelihood that its score did represent the presence or absence of related kidney knowledge. A major strength of our study was the successful engagement of a sample of racial-ethnic minority diverse patients, who serve as key members of the ecosystem to ultimately achieve kidney health equity.34
In conclusion, the PEER-HD training program was feasible, acceptable, and effective in engaging mentor participants and likely imparting dialysis-specific knowledge in patients selected to be mentors for the PEER-HD study. Programs that engage patients with kidney failure must consider challenges related to scheduling and acute health issues and should include structure evaluation to inform impact and improvements in the program itself. Participants emphasized a need for additional health communication training, which may be considered the core content in any future training program.
Article Information
Authors’ Full Names and Academic Degrees
Ladan Golestaneh, MD, MS, Rimon Golovey, MD, Mariela Navarro-Torres, MD, Christopher Roach, BS, Naomy Lantigua-Reyes, MS, Ebele M. Umeukeje, MD, MPH, Aaron Fox, MD, Michal L. Melamed, MD, MHS, and Kerri L. Cavanaugh, MD, MHS.
Authors’ Contributions
Research idea and study design: LG, KLC, MLM; data acquisition: LG, RG, MN-T, CR, NL-R; data analysis/interpretation: LG, KLC, AF, EMU; statistical analysis: LG, CR, RG, MN-T; supervision and mentorship: KLC, MLM, AF, EMU. Each author contributed important intellectual content during manuscript drafting or revision and accepts accountability for the overall work by ensuring that questions pertaining to the accuracy or integrity of any portion of the work are appropriately investigated and resolved.
Support
Research reported in this publication was supported by the National Institute of Diabetes and Digestive and Kidney Disease of the National Institutes of Health under award R18DK118471. The project also received support of Clinical and Translational Science Award UL1 TR002243 from the National Center for Advancing Translational Sciences. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health (NIH), nor did the NIH give any role in study design, collection, analysis, and interpretation of data.
Financial Disclosure
The authors declare that they have no relevant financial interests.
Acknowledgments
The authors would like to thank Drs Mary Beth Heisler and Jennifer St. Clair Russell for their valuable contributions to the training program. The authors would also like to thank Ms Jennie Wilson, Ms Dana Weinrib, and the 2 patients who helped review the training material content.
Peer Review
Received September 13, 2022. Evaluated by 2 external peer reviewers, with direct editorial input from the Statistical Editor, an Associate Editor, and the Editor-in-Chief. Accepted in revised form January 31, 2023.
Footnotes
Complete author and article information provided before references.
Table S1: CHeKs-Plus Survey Items (n=8) Included in Module 1 and Module 2 Posttraining Quizzes.
Table S2: Baseline Variables Stratified by Study Site.
Supplementary Material
Tables S1-S2.
References
- 1.Collins A.J., Foley R.N., Chavers B., et al. US renal data system 2013 annual data report. Am J Kidney Dis. 2014;63(suppl 1):A7. doi: 10.1053/j.ajkd.2013.11.001. [DOI] [PubMed] [Google Scholar]
- 2.St. Clair Russell J., Southerland S., Huff E.D., Thomson M., Meyer K.B., Lynch J.R. A peer-to-peer mentoring program for in-center hemodialysis: a patient-centered quality improvement program. Nephrol Nurs J. 2017;44(6):481–496. [PubMed] [Google Scholar]
- 3.Cukor D., Coplan J., Brown C., et al. Depression and anxiety in urban hemodialysis patients. Clin J Am Soc Nephrol. 2007;2(3):484–490. doi: 10.2215/cjn.00040107. [DOI] [PubMed] [Google Scholar]
- 4.United States Renal Data S. USRDS annual data report: epidemiology of kidney disease in the Unites States. Am J Kidney Dis. 2022;79(4 suppl 1):A8–A12. doi: 10.1053/j.ajkd.2022.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Al Salmi I., Larkina M., Wang M., et al. Missed hemodialysis treatments: international variation, predictors, and outcomes in the Dialysis Outcomes and Practice Patterns Study (DOPPS) Am J Kidney Dis. 2018;72(5):634–643. doi: 10.1053/j.ajkd.2018.04.019. [DOI] [PubMed] [Google Scholar]
- 6.Umeukeje E.M., Ngankam D., Beach L.B., et al. African Americans’ hemodialysis treatment adherence data assessment and presentation: a precision-based paradigm shift to support quality improvement activities. Kidney Med. 2021;4(2) doi: 10.1016/j.xkme.2021.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.St. Clair Russell J., Lupu D., Seliger S., Moss A.H. Providing supportive care to patients with kidney disease. Nephrol News Issues. 2016;30(5):28. [PubMed] [Google Scholar]
- 8.Griva K., Ng H.J., Loei J., Mooppil N., McBain H., Newman S.P. Managing treatment for end-stage renal disease—a qualitative study exploring cultural perspectives on facilitators and barriers to treatment adherence. Psychol Health. 2013;28(1):13–29. doi: 10.1080/08870446.2012.703670. [DOI] [PubMed] [Google Scholar]
- 9.Chenitz K.B., Fernando M., Shea J.A. In-center hemodialysis attendance: patient perceptions of risks, barriers, and recommendations. Hemodial Int. 2014;18(2):364–373. doi: 10.1111/hdi.12139. [DOI] [PubMed] [Google Scholar]
- 10.Perry E., Swartz J., Brown S., Smith D., Kelly G., Swartz R. Peer mentoring: a culturally sensitive approach to end-of-life planning for long-term dialysis patients. Am J Kidney Dis. 2005;46(1):111–119. doi: 10.1053/j.ajkd.2005.03.018. [DOI] [PubMed] [Google Scholar]
- 11.Bandura A. Health promotion by social cognitive means. Health Educ Behav. 2004;31(2):143–164. doi: 10.1177/1090198104263660. [DOI] [PubMed] [Google Scholar]
- 12.Bandura A. Social cognitive theory: an agentic perspective. Annu Rev Psychol. 2001;52:1–26. doi: 10.1146/annurev.psych.52.1.1. [DOI] [PubMed] [Google Scholar]
- 13.Bennett P.N., Hussein W.F., Argentina M., Damron K.C., Atwal J., Schiller B. The feasibility of remote patient peer mentoring in hemodialysis. Kidney Med. 2022;4(10) doi: 10.1016/j.xkme.2022.100529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bennett P.N., St. Clair Russell J., Atwal J., Brown L., Schiller B. Patient-to-patient peer mentor support in dialysis: improving the patient experience. Semin Dial. 2018;31(5):455–461. doi: 10.1111/sdi.12703. [DOI] [PubMed] [Google Scholar]
- 15.Kapron K., Perry E., Bowman T., Swartz R.D. Peer resource consulting: redesigning a new future. Adv Ren Replace Ther. 1997;4(3):267–274. doi: 10.1016/s1073-4449(97)70034-7. [DOI] [PubMed] [Google Scholar]
- 16.Elliott M.J., Love S., Fox D.E., et al. ‘It’s the empathy’—defining a role for peer support among people living with chronic kidney disease: a qualitative study. BMJ Open. 2022;12(5) doi: 10.1136/bmjopen-2021-057518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Golestaneh L., Melamed M., Kim R.S., et al. Peer mentorship to improve outcomes in patients on hemodialysis (PEER-HD): a randomized controlled trial protocol. BMC Nephrol. 2022;23(1):92. doi: 10.1186/s12882-022-02701-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lenzen S.A., Daniels R., van Bokhoven M.A., van der Weijden T., Beurskens A. Disentangling self-management goal setting and action planning: a scoping review. PLoS One. 2017;12(11) doi: 10.1371/journal.pone.0188822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gutnick D., Reims K., Davis C., Gainforth H., Jay M., Cole S. Brief action planning to facilitate behavior change and support patient self-management. J Clin Outcomes Manag. 2014;21(1):17–29. [Google Scholar]
- 20.Harris P.A., Taylor R., Thielke R., Payne J., Gonzalez N., Conde J.G. Research electronic data capture (REDCap)—a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42(2):377–381. doi: 10.1016/j.jbi.2008.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Berkman N.D., Davis T.C., McCormack L. Health literacy: what is it? J Health Commun. 2010;15(suppl 2):9–19. doi: 10.1080/10810730.2010.499985. [DOI] [PubMed] [Google Scholar]
- 22.Rothman R.L., Montori V.M., Cherrington A., Pignone M.P. Perspective: the role of numeracy in health care. J Health Commun. 2008;13(6):583–595. doi: 10.1080/10810730802281791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Cavanaugh K.L., Osborn C.Y., Tentori F., Rothman R.L., Ikizler T.A., Wallston K.A. Performance of a brief survey to assess health literacy in patients receiving hemodialysis. Clin Kidney J. 2015;8(4):462–468. doi: 10.1093/ckj/sfv037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.McNaughton C.D., Cavanaugh K.L., Kripalani S., Rothman R.L., Wallston K.A. Validation of a short, 3-item version of the subjective numeracy scale. Med Decis Making. 2015;35(8):932–936. doi: 10.1177/0272989X15581800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.St. Clair Russell J.J. Virginia Commonwealth University; 2016. Development and Evaluation of a Peer-to-Peer Intervention to Increase Self-Management among Adult In-Center Hemodialysis Patients. [Google Scholar]
- 26.Cavanaugh K.L., Wingard R.L., Hakim R.M., Elasy T.A., Ikizler T.A. Patient dialysis knowledge is associated with permanent arteriovenous access use in chronic hemodialysis. Clin J Am Soc Nephrol. 2009;4(5):950–956. doi: 10.2215/CJN.04580908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wild M.G., Wallston K.A., Green J.A., et al. The Perceived Medical Condition Self-Management Scale can be applied to patients with chronic kidney disease. Kidney Int. 2017;92(4):972–978. doi: 10.1016/j.kint.2017.03.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Boyer A., Begin Y., Dupont J., et al. Health literacy level in a various nephrology population from Québec: predialysis clinic, in-centre hemodialysis and home dialysis; a transversal monocentric observational study. BMC Nephrol. 2021;22(1):259. doi: 10.1186/s12882-021-02464-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ghahramani N., Chinchilli V.M., Kraschnewski J.L., Lengerich E.J., Sciamanna C.N. Effect of peer mentoring on quality of life among ckd patients: randomized controlled trial. Kidney Dis. 2021;7(4):323–333. doi: 10.1159/000514477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Golestaneh L. Decreasing hospitalizations in patients on hemodialysis: time for a paradigm shift. Semin Dial. 2018;31(3):278–288. doi: 10.1111/sdi.12675. [DOI] [PubMed] [Google Scholar]
- 31.Taylor F., Gutteridge R., Willis C. Peer support for CKD patients and carers: overcoming barriers and facilitating access. Health Expect. 2016;19(3):617–630. doi: 10.1111/hex.12348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.System USRD. 2021 USRDS Annual Data Report: Epidemiology of Kidney Disease in the United States. https://adr.usrds.org/2021
- 33.Trasolini A., Wood E., Thomas N. A narrative review of peer support barriers and facilitators in kidney care. J Ren Care. 2021;47(1):9–16. doi: 10.1111/jorc.12350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Griffith D.M., Umeukeje E.M. Navigating to kidney health equity. J Am Soc Nephrol. 2022;33(7):1242–1244. doi: 10.1681/asn.2022040421. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Tables S1-S2.